Workshop on Medical Device Cleanliness: How Clean is Clean Enough? Sponsored by ASTM Committee F04 on Medical and Surgical Materials and Devices
|
|
|
- Meryl Harvey
- 5 years ago
- Views:
Transcription
1 Breaking the Myth Case Medical Page 1 Our Position Caustic cleaners are unnecessary for instrument processing and present significant hazards to health and safety Properly formulated ph neutral instrument chemistries with excellent surfactants and synergistic enzymes can offer a safe and proven alternative The need to provide validated cleaning protocols for reusable medical devices has become a focal point in medical device reprocessing. Page 2 page 1
2 Sustainability Meeting the needs of our current generation without impacting the needs of future generations to meet their own needs Social responsibility to protect the public from exposure to harm All manufacturers are obligated to design instrument chemistries to control measures which may lead to possible harm. Page 3 Precautionary Principle Ethical responsibility to maintaining the integrity of natural systems Willingness to take action in advance of definitive scientific proof when a delay could prove costly to society and nature as well as selfish to future generations Page 4 page 2
3 Breaking the Chain Cleaning, decontamination and subsequent sterilization are essential steps in breaking the chain of infection. Page 5 Cleaning The removal, usually with detergent and water, of adherent visible soil such as blood, protein substances and other debris from the surfaces, crevices, serrations, joints and lumens of instruments, devices and equipment by a manual or mechanical process that prepares the items for safe handling and further decontamination. Page 6 page 3
4 Ineffective Cleaning Can affect the ability of medical devices To function properly Decreases the useful life of the device Increases costs for repair and replacement Can interfere with the effectiveness of subsequent sterilization or disinfection Increases the risk of nosocomial infection Page 7 Green Chemistry Chemical products and processes designed to reduce or eliminate hazardous substances Pollution prevention focus of EPA s Green Chemistry program Page 8 page 4
5 Caustic Cleaners Alkaline detergents followed by acid neutralizers Present hazards to waste water stream Cause pitting and corrosion of surgical devices May result in safety issues for patients and staff Page 9 Warning The use of elevated temperatures in a cleaning process will cause denaturation and precipitation of soil components (blood) and make them more difficult to remove. Page 10 page 5
6 Ideal Cleaning Agents Non-corrosive Free-rinsing Non-abrasive Low-foaming Biodegradable Environmentally Friendly Nontoxic in specified use dilution Provide for rapid soil dispersion or suspension May be used in all water types Page 11 Cleaning Reusable Devices The critical first step in the decontamination process Multi-step process including manual and automated cleaning Followed by a thorough rinse with high purity water Manual cleaning dependent on individual s performance. Mechanical reproducible. Page 12 page 6
7 Validation The FDA places primary responsibility for developing and validating methods for effective processing of the medical device on the manufacturer of the device. Device manufacturers must provide procedures that must be easily replicated and verified by users Page 13 The Validation Enzymatic detergents are often recommended for cleaning reusable devices. Yet, there was little data to confirm the efficacy of enzymatic detergents for effective soil removal from patient-used medical devices Validation: Simulated-use evaluation of enzymatic cleaning Evaluated static soak, manual cleaning, automated cleaning, and the recommended process Inoculated devices Organic soil reduction Bioburden reduction Page 14 page 7
8 Validation of Process Pre-cleaning using a ph neutral multienzymatic foam Brief rinse with tap water Ultra sonic cleaning with ph neutral detergent Manual cleaning with ph neutral multienzymatic cleaner Final rinse with tap water Page 15 Results Hemostats soiled with ATS and microorganisms processed by the manufacturer s products and method resulted in a 6 Log 10 reduction bioburden and > 99.9% reduction in organic material post processing. Conclusion The protocol of cleaning that is recommended by the manufacturer provides efficient cleaning of medical devices by providing soil and bioburden reduction. Page 16 page 8
9 The ph Effect To determine if an increase in ph (alkalinity level) will improve or change the outcome of the cleaning protocol Two parallel studies were conducted of the entire cleaning protocol One using neutral ph detergent One using the same ingredients but with a ph of 9 (alkaline) Page 17 ph Effect Results There was no significant difference in outcome between the ph neutral and the alkaline detergents In fact, there was a slight improvement in efficacy with the ph neutral detergent in replicate studies. Page 18 page 9
10 Why Use Enzymatic Cleaners? Each enzyme is a catalyst working on a particular substrate much as a lock and key. If one enzyme is used only one type of organic soil will be broken down. Protease breaks down protein Lipase breaks down lipids (fats). Amylase breaks down starch and carbohydrates. Multiple enzymes are needed to attack organic soils, so that they may be washed away. Hot water is not recommended nor needed. Page 19 Why are Detergents Needed? Detergents are required for a thorough cleaning and removal of organic and inorganic soils, detergent residue, including enzymes. They may be used in hot water wash cycles for thermal disinfection. Detergents with chelating and sequestering agents are required for removal of salts, minerals and other hard water ions. In automated systems, when filtered, deionized or RO water systems are used lower concentrations of detergents may be used. Page 20 page 10
11 What Instrument Manufacturers Say Chemicals used in cleaning and decontamination processes should be able to remove the type of soil found on the item while, at the same time, preserving the integrity of the item. Such cleaning agents should have the following properties: Ability to remove organic and inorganic soil. Ability to prevent deposits from hard water ions. Low foaming. Free rinsing. Neutral ph (range 7-8) Very acid or alkaline solutions can damage the inert layer that keeps stainless steel instruments from corroding. Anodized aluminum containers require ph neutral products for decontamination and to maintain their corrosion resistance and useful life. Page 21 Why Green Products? Environmentally friendly products are safe for the patient, staff, and the community. Color, dye, fragrance mask the degradation of the product (fading, sediment, mold!) and the odor of denatured enzymes. Most water systems in each of our cities limit the level of alkalinity in the water supply to a ph of 8.5, yet the alkaline detergents used may reach a ph of 14, the highest level possible and are dumped in our water supply, destroy medical devices, and if used on patients without rinsing, will cause injury or death. Page 22 page 11
12 Summary Medical devices are critical for patient care. No instrument company recommends caustic detergents, because of the potential damage to the device. Breaking the myth means that there are instrument chemistries that are ph neutral, validated in independent studies, that are environmentally friendly, safe and effective, and demonstrate efficacy and sustainability. Page 23 page 12
Water for Instrument Processing
 Water for Instrument Processing by Marcia Frieze, Case Medical Water, the universal solvent Water can dissolve more substances than any other liquid. It is essentially nonionic or neutral. While alkaline
Water for Instrument Processing by Marcia Frieze, Case Medical Water, the universal solvent Water can dissolve more substances than any other liquid. It is essentially nonionic or neutral. While alkaline
HemoCheck TM Sample Policy. SUBJECT: Detection of blood residue on various surfaces
 HemoCheck TM Sample Policy SUBJECT: Detection of blood residue on various surfaces DEPARTMENT: Central Service APPROVED BY: EFFECTIVE: REVISED: 05/2018 PURPOSE: To test for detection of blood residue on
HemoCheck TM Sample Policy SUBJECT: Detection of blood residue on various surfaces DEPARTMENT: Central Service APPROVED BY: EFFECTIVE: REVISED: 05/2018 PURPOSE: To test for detection of blood residue on
HemoCheck TM Sample Policy. SUBJECT: Detection of blood residue on various surfaces
 HemoCheck TM Sample Policy SUBJECT: Detection of blood residue on various surfaces DEPARTMENT: Central Service APPROVED BY: EFFECTIVE: REVISED: 07/2016 PURPOSE: To test for detection of blood residue on
HemoCheck TM Sample Policy SUBJECT: Detection of blood residue on various surfaces DEPARTMENT: Central Service APPROVED BY: EFFECTIVE: REVISED: 07/2016 PURPOSE: To test for detection of blood residue on
Technical Data Monograph. The Instrument Protection Properties of Prolystica 2X Concentrate Neutral Detergent Versus Other Neutral Detergent Products
 Technical Data Monograph The Instrument Protection Properties of Prolystica 2X Concentrate Neutral Detergent Versus Other Neutral Detergent Products Table of Contents 1. Background.....................................................1
Technical Data Monograph The Instrument Protection Properties of Prolystica 2X Concentrate Neutral Detergent Versus Other Neutral Detergent Products Table of Contents 1. Background.....................................................1
Guide for Cleaning, Sterilization and Storage of Introducers for Caldera Medical Implants
 Guide for Cleaning, Sterilization and Storage of Introducers for Caldera Medical Implants M Manufactured by: Caldera Medical, Inc. 5171 Clareton Drive Agoura Hills, CA 91301 U.S. Toll Free: 866-4-CALDERA
Guide for Cleaning, Sterilization and Storage of Introducers for Caldera Medical Implants M Manufactured by: Caldera Medical, Inc. 5171 Clareton Drive Agoura Hills, CA 91301 U.S. Toll Free: 866-4-CALDERA
Guide for Cleaning, Sterilization and Storage of Reusable Introducers
 Guide for Cleaning, Sterilization and Storage of Reusable Introducers M Manufactured by: Caldera Medical, Inc. 5171 Clareton Drive Agoura Hills, CA 91301 U.S. Toll Free: 866-4-CALDERA Telephone: 818-879-6555
Guide for Cleaning, Sterilization and Storage of Reusable Introducers M Manufactured by: Caldera Medical, Inc. 5171 Clareton Drive Agoura Hills, CA 91301 U.S. Toll Free: 866-4-CALDERA Telephone: 818-879-6555
3.03 Understand support services
 3.03 Understand support services Clean and Decontaminate the Healthcare Environment Manage Hazardous Materials and Wastes Manage and Store Materials 3.03 Understand support services 2 Clean and Decontaminate
3.03 Understand support services Clean and Decontaminate the Healthcare Environment Manage Hazardous Materials and Wastes Manage and Store Materials 3.03 Understand support services 2 Clean and Decontaminate
How is Sterilization Validation Performed by the Medical Device Manufacturer?
 How is Sterilization Validation Performed by the Medical Device Manufacturer? Rod Parker, Ph.D. Sr. Principal Scientist Stryker Instruments Division May 9, 2016 DISCLOSURE I am an employee of Stryker Corporation.
How is Sterilization Validation Performed by the Medical Device Manufacturer? Rod Parker, Ph.D. Sr. Principal Scientist Stryker Instruments Division May 9, 2016 DISCLOSURE I am an employee of Stryker Corporation.
The Power of Prolystica Cleaning Chemistries SO MUCH MORE THAN. One Integrated Approach to Healthcare.
 Sterile Endoscopy Processing Solutions Department The Power of Prolystica Cleaning Chemistries SO MUCH MORE THAN One Integrated Approach to Healthcare. World class cleaning is an expectation of Prolystica.
Sterile Endoscopy Processing Solutions Department The Power of Prolystica Cleaning Chemistries SO MUCH MORE THAN One Integrated Approach to Healthcare. World class cleaning is an expectation of Prolystica.
Biofilms 101 Remediation Strategies in Meat Processing
 Biofilms 101 Remediation Strategies in Meat Processing Jeremy M. Adler Director of Technology and Innovation American Meat Science Association April 28, 2016 Webinar Biofilms How does Pseudomonas know
Biofilms 101 Remediation Strategies in Meat Processing Jeremy M. Adler Director of Technology and Innovation American Meat Science Association April 28, 2016 Webinar Biofilms How does Pseudomonas know
Best Practices for Reprocessing Endoscopes
 Best Practices for Reprocessing Endoscopes Reprocessing of Endoscopes Reprocessing Room Standards The resources referenced include: Canadian Standards Association Provincial Infectious Disease Advisory
Best Practices for Reprocessing Endoscopes Reprocessing of Endoscopes Reprocessing Room Standards The resources referenced include: Canadian Standards Association Provincial Infectious Disease Advisory
The Basics of Alkaline In-Process Cleaning. for Metal Substrates. John Sparks Oakite Products, Inc. Berkeley Heights, New Jersey
 The Basics of Alkaline In-Process Cleaning for Metal Substrates John Sparks Oakite Products, Inc. Berkeley Heights, New Jersey This paper pertains mainly to alkaline cleaners, but includes solvents and
The Basics of Alkaline In-Process Cleaning for Metal Substrates John Sparks Oakite Products, Inc. Berkeley Heights, New Jersey This paper pertains mainly to alkaline cleaners, but includes solvents and
Enzyme Wizard Multi Enzyme cleaning formulations contain ingredients that are naturally occurring. They consist of four main ingredients natural
 Safe for people, pets and the planet. Enzyme Wizard cleaning products are 100% biodegradable - no caustic chemicals soap free - ph neutral - totally eco-friendly. Enzyme Wizard Multi Enzyme cleaning formulations
Safe for people, pets and the planet. Enzyme Wizard cleaning products are 100% biodegradable - no caustic chemicals soap free - ph neutral - totally eco-friendly. Enzyme Wizard Multi Enzyme cleaning formulations
*Slides updated to include newer guidelines in February 2016 by Shelby Lassiter, BSN, RN, CPHQ, CIC
 *Slides updated to include newer guidelines in February 2016 by Shelby Lassiter, BSN, RN, CPHQ, CIC 1 2 3 4 * 5 6 7 If the instrument is not clean, the organic soil can harbor embedded microorganisms.
*Slides updated to include newer guidelines in February 2016 by Shelby Lassiter, BSN, RN, CPHQ, CIC 1 2 3 4 * 5 6 7 If the instrument is not clean, the organic soil can harbor embedded microorganisms.
Professional instrument reprocessing in CSSD
 Professional instrument reprocessing in CSSD The reprocessing cycle in CSSD Fig. 1 The objective of the surgical instrument reprocessing cycle Only proper, professional manual as well as automatic in central
Professional instrument reprocessing in CSSD The reprocessing cycle in CSSD Fig. 1 The objective of the surgical instrument reprocessing cycle Only proper, professional manual as well as automatic in central
DISCOVER INNOVATION DRIVING INCREASED EFFICIENCY
 ULTRASONIC CLEANING SYSTEMS AND SONIC IRRIGATORS Central Sterile Services DISCOVER INNOVATION DRIVING INCREASED EFFICIENCY One Integrated Approach to Healthcare. Advancements in surgical instruments have
ULTRASONIC CLEANING SYSTEMS AND SONIC IRRIGATORS Central Sterile Services DISCOVER INNOVATION DRIVING INCREASED EFFICIENCY One Integrated Approach to Healthcare. Advancements in surgical instruments have
Monitoring Flexible Endoscope Channels to Assure Cleaning
 Monitoring Flexible Endoscope Channels to Assure Cleaning Dr. Michelle J. Alfa, Ph.D., FCCM Medical Director, Clinical Microbiology, Diagnostic Services of Manitoba, Winnipeg, Canada Disclaimers: Dr. M.
Monitoring Flexible Endoscope Channels to Assure Cleaning Dr. Michelle J. Alfa, Ph.D., FCCM Medical Director, Clinical Microbiology, Diagnostic Services of Manitoba, Winnipeg, Canada Disclaimers: Dr. M.
One such problem is blood residual inside the channels of a robotic arm.
 SUBJECT: Robotic Arm Testing for residue blood DEPARTMENT: Central Service APPROVED BY: EFFECTIVE: REVISED: 07/2016 PURPOSE: To test for detection of blood residue inside and outside of a robotic arm and
SUBJECT: Robotic Arm Testing for residue blood DEPARTMENT: Central Service APPROVED BY: EFFECTIVE: REVISED: 07/2016 PURPOSE: To test for detection of blood residue inside and outside of a robotic arm and
Lotus. Reprocessing instructions for reusable instrumentation
 Lotus Reprocessing instructions for reusable instrumentation Reprocessing instructions These instructions relate to the Lotus Transducers & Waveguides only. Following use remove the Single Use Handpiece
Lotus Reprocessing instructions for reusable instrumentation Reprocessing instructions These instructions relate to the Lotus Transducers & Waveguides only. Following use remove the Single Use Handpiece
Lotus. Reprocessing instructions for reusable instrumentation. French/Swiss protocol
 Lotus Reprocessing instructions for reusable instrumentation French/Swiss protocol Reprocessing instructions These instructions relate to the Lotus Transducers & Waveguides only. Following use remove the
Lotus Reprocessing instructions for reusable instrumentation French/Swiss protocol Reprocessing instructions These instructions relate to the Lotus Transducers & Waveguides only. Following use remove the
Manner of Assessment Code (check all that apply) & Surveyor Notes. Yes N/A. Yes. Yes N/A N/A. Yes N/A. N/A Yes N/A
 Section 4. L Reprocessing of Semi-Critical Equipment Elements to be assessed Manner of Assessment Code (check all that apply) & Surveyor Notes Manner of Assessment Code (check all that apply) & Surveyor
Section 4. L Reprocessing of Semi-Critical Equipment Elements to be assessed Manner of Assessment Code (check all that apply) & Surveyor Notes Manner of Assessment Code (check all that apply) & Surveyor
SUBJECT: Detection of Blood residue inside the biopsy channel of a scope
 SUBJECT: Detection of Blood residue inside the biopsy channel of a scope DEPARTMENT: Central Service/ Endoscopy APPROVED BY: EFFECTIVE: REVISED: 2/2012 PURPOSE: To test for detection of blood residue inside
SUBJECT: Detection of Blood residue inside the biopsy channel of a scope DEPARTMENT: Central Service/ Endoscopy APPROVED BY: EFFECTIVE: REVISED: 2/2012 PURPOSE: To test for detection of blood residue inside
3.03 Understand support services
 3.03 Understand support services Clean and Decontaminate the Healthcare Environment Manage Hazardous Materials and Wastes Manage and Store Materials 3.03 Understand support services 2 Clean and Decontaminate
3.03 Understand support services Clean and Decontaminate the Healthcare Environment Manage Hazardous Materials and Wastes Manage and Store Materials 3.03 Understand support services 2 Clean and Decontaminate
Appendix 2. Revised 510(k) Summary
 Appendix 2 Revised 510(k) Summary April 1, 2010 Appendix 2: Page 1 of 11 510(k) Summary For SYSTEM 1E Liquid Chemical Sterilant Processing System STERIS Corporation 5960 Heisley Road Mentor, OH 44060 Phone:
Appendix 2 Revised 510(k) Summary April 1, 2010 Appendix 2: Page 1 of 11 510(k) Summary For SYSTEM 1E Liquid Chemical Sterilant Processing System STERIS Corporation 5960 Heisley Road Mentor, OH 44060 Phone:
Validation of cleaning and sterilization of reusable medical devices
 Synergy makes sence Validation of cleaning and sterilization of reusable medical devices MG-FSI72-119 Last revision: June 2012 Aurélien BIGNON 5, Chemin du Catupolan - 69120 Vaulx en Velin - France - Tel.
Synergy makes sence Validation of cleaning and sterilization of reusable medical devices MG-FSI72-119 Last revision: June 2012 Aurélien BIGNON 5, Chemin du Catupolan - 69120 Vaulx en Velin - France - Tel.
Sterilization, disinfection and reprocessing
 Sterilization, disinfection and reprocessing David R. Woodard, MSc, CIC, FSHEA Definitions Cleaning the removal of visible soil from the surfaces, joints and lumen of instruments or devices. This is done
Sterilization, disinfection and reprocessing David R. Woodard, MSc, CIC, FSHEA Definitions Cleaning the removal of visible soil from the surfaces, joints and lumen of instruments or devices. This is done
Products made in Switzerland
 Products made in Switzerland HYGIENIC AND MEDICALLY CLEAR DIFFERENT PRODUCTS BUT THE SAME QUALITY LABEL HYGIENIC AND MEDICALLY CLEAR 2 MDD TECHNOLOGIES FOR: 1. Cleaning 2. Disinfection 3. High Level Disinfection
Products made in Switzerland HYGIENIC AND MEDICALLY CLEAR DIFFERENT PRODUCTS BUT THE SAME QUALITY LABEL HYGIENIC AND MEDICALLY CLEAR 2 MDD TECHNOLOGIES FOR: 1. Cleaning 2. Disinfection 3. High Level Disinfection
Principles of Preservation
 From ISP/Sutton Laboratories Principles of Preservation What is preservation? When we speak as formulators about preservation, we are referring to the protection of our products from contamination by bacteria,
From ISP/Sutton Laboratories Principles of Preservation What is preservation? When we speak as formulators about preservation, we are referring to the protection of our products from contamination by bacteria,
The Role of Detergents and Disinfectants in Instrument Cleaning and Reprocessing
 CE ONLINE The Role of Detergents and Disinfectants in Instrument Cleaning and Reprocessing An Online Continuing Education Activity Sponsored By Grant funds provided by Welcome to The Role of Detergents
CE ONLINE The Role of Detergents and Disinfectants in Instrument Cleaning and Reprocessing An Online Continuing Education Activity Sponsored By Grant funds provided by Welcome to The Role of Detergents
MEMBRANE PRODUCTS PLEASE CONTACT YOUR LOCAL NORLEX CHEMICALS PARTNER FOR MORE INFORMATION ON THESE TYPES OF PRODUCTS.
 MEMBRANE PRODUCTS PLEASE CONTACT YOUR LOCAL NORLEX CHEMICALS PARTNER FOR MORE INFORMATION ON THESE TYPES OF PRODUCTS. NORLEX CHEMICALS A/S BISTRUPVEJ 172 3460 BIRKERØD DENMARK TLF: +45 45 94 09 94 info@norlex.com
MEMBRANE PRODUCTS PLEASE CONTACT YOUR LOCAL NORLEX CHEMICALS PARTNER FOR MORE INFORMATION ON THESE TYPES OF PRODUCTS. NORLEX CHEMICALS A/S BISTRUPVEJ 172 3460 BIRKERØD DENMARK TLF: +45 45 94 09 94 info@norlex.com
Technical Data Monograph. Stainless Steel Instrument Maintenance Using Prolystica Restore Descaler & Neutralizing Detergent
 Technical Data Monograph Stainless Steel Instrument Maintenance Using Prolystica Restore Descaler & Neutralizing Detergent Table of Contents 1. Background...3 2. Experimental Studies...4 3. Passivation...5
Technical Data Monograph Stainless Steel Instrument Maintenance Using Prolystica Restore Descaler & Neutralizing Detergent Table of Contents 1. Background...3 2. Experimental Studies...4 3. Passivation...5
6/12/2016. Objectives. Manufacturers Instructions for Use: Impact on Patient Safety. Background
 Manufacturers Instructions for Use: Impact on Patient Safety N A N C Y C H O B I N, R N, A A S, A C S P, C S P M, C F E R P R E S I D E N T, S T E R I L E P R O C E S S I N G U N I V E R S I T Y, L L C
Manufacturers Instructions for Use: Impact on Patient Safety N A N C Y C H O B I N, R N, A A S, A C S P, C S P M, C F E R P R E S I D E N T, S T E R I L E P R O C E S S I N G U N I V E R S I T Y, L L C
Antiseptic and Disinfectant Product Group
 Antiseptic and Disinfectant Product Group one drop of is enough for more hygiene ENDOSCOPE AND MEDICAL DEVICE DISINFECTANTS Points to be paid attention to while disinfection of instruments Disinfection
Antiseptic and Disinfectant Product Group one drop of is enough for more hygiene ENDOSCOPE AND MEDICAL DEVICE DISINFECTANTS Points to be paid attention to while disinfection of instruments Disinfection
Amalgatome SD Sterilization Tray User Guide
 Amalgatome SD Sterilization Tray User Guide EXSURCO MEDICAL 10804 GREEN ROAD WAKEMAN, OH 44889 PN: X101489 REV. B This page intentionally left blank. AMALGATOME SD STERILIZATION TRAY USER GUIDE Instructions
Amalgatome SD Sterilization Tray User Guide EXSURCO MEDICAL 10804 GREEN ROAD WAKEMAN, OH 44889 PN: X101489 REV. B This page intentionally left blank. AMALGATOME SD STERILIZATION TRAY USER GUIDE Instructions
Getinge da Vinci Solution
 Getinge da Vinci Solution A comprehensive cleaning solution for da Vinci EndoWrist instruments This document is intended to provide information to an international audience outside of the US. Validated,
Getinge da Vinci Solution A comprehensive cleaning solution for da Vinci EndoWrist instruments This document is intended to provide information to an international audience outside of the US. Validated,
What Does Validation Involve?
 Practical instructions for the Validation of cleaning and disinfection methods according to the recommendation of the German Society for Hospital Hygiene (DGKH) and according to the pren ISO 15883. Laws,
Practical instructions for the Validation of cleaning and disinfection methods according to the recommendation of the German Society for Hospital Hygiene (DGKH) and according to the pren ISO 15883. Laws,
Sterilization of Schweickhardt Instruments
 Sterilization of Schweickhardt Instruments INTRODUCTION l Most dental instruments are made of corrosion-resistant highgrade steels. The requirements for grades of steel are defined in national and international
Sterilization of Schweickhardt Instruments INTRODUCTION l Most dental instruments are made of corrosion-resistant highgrade steels. The requirements for grades of steel are defined in national and international
CAUTION: FEDERAL LAW (USA) RESTRICTS THESE DEVICES TO SALE BY OR ON THE ORDER OF A PHYSICIAN.
 Manufacturer Silony Medical GmbH Leinfelder Strasse 60 70771 Leinfelden-Echterdingen Germany US Representative: Silony Medical Corp. 8200 NW 27TH STR, STE#104, DORAL, FL 33122 USA Phone: +1 305 916 0016
Manufacturer Silony Medical GmbH Leinfelder Strasse 60 70771 Leinfelden-Echterdingen Germany US Representative: Silony Medical Corp. 8200 NW 27TH STR, STE#104, DORAL, FL 33122 USA Phone: +1 305 916 0016
Standards of Infection Prevention in Reprocessing Flexible Gastrointestinal Endoscopes Webinar FAQ
 Standards of Infection Prevention in Reprocessing Flexible Gastrointestinal Endoscopes Webinar FAQ Disclaimer: The Society of Gastroenterology Nurses and Associates, Inc. assume no responsibility for the
Standards of Infection Prevention in Reprocessing Flexible Gastrointestinal Endoscopes Webinar FAQ Disclaimer: The Society of Gastroenterology Nurses and Associates, Inc. assume no responsibility for the
HISTORY OF CHLORINE DIOXIDE
 HISTORY OF CHLORINE DIOXIDE 1814 Sir Humphrey Davy, a British chemist, discovers chlorine dioxide (CD), a result of potassium chlorate reacting with sulfuric acid. Although he fully understood the chemistry,
HISTORY OF CHLORINE DIOXIDE 1814 Sir Humphrey Davy, a British chemist, discovers chlorine dioxide (CD), a result of potassium chlorate reacting with sulfuric acid. Although he fully understood the chemistry,
CDWA3 / CDWA4 CDWAH. Page 4. BASKET ZONE: water pressure, spray system performance, detergent chemistry and concentration, temperature.
 Rev. 2 March 2017 Control of cleaning and washing processes of medical instruments and materials is highly important since its outcome affects the success of subsequent disinfection and/or sterilization
Rev. 2 March 2017 Control of cleaning and washing processes of medical instruments and materials is highly important since its outcome affects the success of subsequent disinfection and/or sterilization
Why wait until catastrophe strikes? Prevent the Chain Reaction of Equipment Wear, Maximize Heat Transfer, Protect Equipment Life
 Why wait until catastrophe strikes? Prevent the Chain Reaction of Equipment Wear, Maximize Heat Transfer, Protect Equipment Life IAS has pioneered and utilizes a new generation high-velocity, chemical
Why wait until catastrophe strikes? Prevent the Chain Reaction of Equipment Wear, Maximize Heat Transfer, Protect Equipment Life IAS has pioneered and utilizes a new generation high-velocity, chemical
Cleaning and Sanitizing Products BRU-R-EZ CELL-R-MASTR DIACTOLATE. Features:
 BRU-R-EZ Clean your mash tun, brew kettle, fermenters, heat exchanger, lines, kegs, etc. more quickly and effectively than the routine caustic products with BRU-R-EZ. Ultra rinsability leaves a perfect
BRU-R-EZ Clean your mash tun, brew kettle, fermenters, heat exchanger, lines, kegs, etc. more quickly and effectively than the routine caustic products with BRU-R-EZ. Ultra rinsability leaves a perfect
Applied Sanitation in Home. Rick Theiner LOGIC, Inc.
 Applied Sanitation in Home Fermentation Hobbies Rick Theiner LOGIC, Inc. What is LOGIC, Inc.? Longtime manufacturer of the two best cleansing and sanitizing products for homebrewers Founded and owned by
Applied Sanitation in Home Fermentation Hobbies Rick Theiner LOGIC, Inc. What is LOGIC, Inc.? Longtime manufacturer of the two best cleansing and sanitizing products for homebrewers Founded and owned by
Guide to Optimal Steam Generation
 Central Sterile Services HOW TO: Guide to Optimal Steam Generation Introduction Successful steam sterilization is much more than just achieving verification of sterility with biological indicators. Most
Central Sterile Services HOW TO: Guide to Optimal Steam Generation Introduction Successful steam sterilization is much more than just achieving verification of sterility with biological indicators. Most
Technical Data Monograph
 Technical Data Monograph Compatibility of Prolystica 2X Concentrate Presoak and Cleaner and Other Products with Soft Metal Substrates Table of Contents 1. Purpose.......................................................1
Technical Data Monograph Compatibility of Prolystica 2X Concentrate Presoak and Cleaner and Other Products with Soft Metal Substrates Table of Contents 1. Purpose.......................................................1
THE EXPERTS IN INDUSTRIAL CLEANING
 THE EXPERTS IN INDUSTRIAL CLEANING CASTROL OFFERS YOU A COMPREHENSIVE RANGE OF WORLD-CLASS INDUSTRIAL CLEANERS AND PRODUCT SUPPORT SERVICES THAT WILL HELP YOU TO: > Achieve more reliable cleanliness of
THE EXPERTS IN INDUSTRIAL CLEANING CASTROL OFFERS YOU A COMPREHENSIVE RANGE OF WORLD-CLASS INDUSTRIAL CLEANERS AND PRODUCT SUPPORT SERVICES THAT WILL HELP YOU TO: > Achieve more reliable cleanliness of
Recommendation
 1 2 3 4 5 6 7 1 Instruments reception Reception of a new instrument MUST be performed by a person aware of proper procedures. After control of order conformity, this material can be unpacked and it will
1 2 3 4 5 6 7 1 Instruments reception Reception of a new instrument MUST be performed by a person aware of proper procedures. After control of order conformity, this material can be unpacked and it will
CLEANING IN THE AGE OF MAGNESIUM AND CALCIUM CHLORIDE
 CLEANING IN THE AGE OF MAGNESIUM AND CALCIUM CHLORIDE Tony Vertin Ver-tech Labs, Rockford, MN www.ver-techlabs.com www.vtlvehiclecare.com The Corrosive Effects of Magnesium and Calcium Chloride Protective
CLEANING IN THE AGE OF MAGNESIUM AND CALCIUM CHLORIDE Tony Vertin Ver-tech Labs, Rockford, MN www.ver-techlabs.com www.vtlvehiclecare.com The Corrosive Effects of Magnesium and Calcium Chloride Protective
SUBJECT: Detection of Blood (hemoglobin) residue inside the biopsy channel of an endoendoscope
 SUBJECT: Detection of Blood (hemoglobin) residue inside the biopsy channel of an endoendoscope DEPARTMENT: Central Service/ Endoscopy APPROVED BY: EFFECTIVE: REVISED: 6/2016 PURPOSE: To test for detection
SUBJECT: Detection of Blood (hemoglobin) residue inside the biopsy channel of an endoendoscope DEPARTMENT: Central Service/ Endoscopy APPROVED BY: EFFECTIVE: REVISED: 6/2016 PURPOSE: To test for detection
stain Benefits Premium Professional Clean
 stain Provides superior spot removal from carpet and fabric with the additional benefit of odour control. Eliminates stains and odours contributing to a freshness that welcomes guests and instills confidence
stain Provides superior spot removal from carpet and fabric with the additional benefit of odour control. Eliminates stains and odours contributing to a freshness that welcomes guests and instills confidence
A Case Study from EP3: Pollution Prevention Assessment from a Cattle Hide Tannery. HBI Pub. 10/31/94
 A Case Study from EP3: Pollution Prevention Assessment from a Cattle Hide Tannery HBI-94-003-01 Pub. 10/31/94 What is EP3? The amount of pollutants and waste generated by industrial facilities has become
A Case Study from EP3: Pollution Prevention Assessment from a Cattle Hide Tannery HBI-94-003-01 Pub. 10/31/94 What is EP3? The amount of pollutants and waste generated by industrial facilities has become
Human Alanine Aminotransferase (ALT) ELISA Kit. For Reference Only
 Human Alanine Aminotransferase (ALT) ELISA Kit Catalog No.: abx572210 Size: 96T Range: 0.312 ng/ml - 20 ng/ml Sensitivity: < 0.132 ng/ml Storage: Store standard, detection reagent A, detection reagent
Human Alanine Aminotransferase (ALT) ELISA Kit Catalog No.: abx572210 Size: 96T Range: 0.312 ng/ml - 20 ng/ml Sensitivity: < 0.132 ng/ml Storage: Store standard, detection reagent A, detection reagent
Endoscope Decontamination. A Tube in a Hole??
 Endoscope Decontamination A Tube in a Hole?? John Prendergast AE(D) MIHEEM Senior Decontamination Engineer (Wales) May 2016 Note This presentation is my personal interpretation and not NHS Wales policy.
Endoscope Decontamination A Tube in a Hole?? John Prendergast AE(D) MIHEEM Senior Decontamination Engineer (Wales) May 2016 Note This presentation is my personal interpretation and not NHS Wales policy.
Administrative Policies and Procedures. Cross Reference: Date Issued: 2/14 Date Reviewed: 9/14 Date: Revised: 12/14 Attachment: None Page of 1 of 6
 Administrative Policies and Procedures Originating Venue: Infection Control Policy No.: IC 2306 Title: Cystoscope Reprocessing Policy & Procedure Cross Reference: Date Issued: 2/14 Date Reviewed: 9/14
Administrative Policies and Procedures Originating Venue: Infection Control Policy No.: IC 2306 Title: Cystoscope Reprocessing Policy & Procedure Cross Reference: Date Issued: 2/14 Date Reviewed: 9/14
Flexible Endoscopes: The A,B,C s of Monitoring Manual Cleaning Efficacy!
 Flexible Endoscopes: The A,B,C s of Monitoring Manual Cleaning Efficacy! Dr. Michelle J. Alfa, Ph.D., FCCM Medical Director, Clinical Microbiology, Diagnostic Services of Manitoba, Winnipeg, Canada Disclaimers:
Flexible Endoscopes: The A,B,C s of Monitoring Manual Cleaning Efficacy! Dr. Michelle J. Alfa, Ph.D., FCCM Medical Director, Clinical Microbiology, Diagnostic Services of Manitoba, Winnipeg, Canada Disclaimers:
Guidelines for Selection and Use of Disinfectants
 Guidelines for Selection and Use of Disinfectants Ref: (a) APIC Guidelines for Infection Control Practice, American Journal of Infection Control; April 1990, Vol 18, 99-113. To assist health care professionals
Guidelines for Selection and Use of Disinfectants Ref: (a) APIC Guidelines for Infection Control Practice, American Journal of Infection Control; April 1990, Vol 18, 99-113. To assist health care professionals
protect public health protect the most sensitive among us protect the environment prevent the spread of antibiotic resistant bacteria
 TM PCS Process Cleaning Solutions is a company devoted to cleaning in such a way as to: protect public health protect the most sensitive among us protect the environment prevent the spread of antibiotic
TM PCS Process Cleaning Solutions is a company devoted to cleaning in such a way as to: protect public health protect the most sensitive among us protect the environment prevent the spread of antibiotic
RBS detergents for laboratories
 RBS detergents for laboratories High performance solutions For reliable and reproducible results Competence & experience RBS products are mainly intended for water-based cleaning processes of glassware
RBS detergents for laboratories High performance solutions For reliable and reproducible results Competence & experience RBS products are mainly intended for water-based cleaning processes of glassware
Loaner Program Guidance
 Loaner Program Guidance This document is valid only on the date printed. If unsure of the print date, please re-print to ensure use of the latest revision of this document (available at www.depuysynthes.com).
Loaner Program Guidance This document is valid only on the date printed. If unsure of the print date, please re-print to ensure use of the latest revision of this document (available at www.depuysynthes.com).
INSTRUMENT CLEANING & STERILISATION FOAMING BATH FORMULATION. Surgistain. Rapid Multi-Enzyme Cleaner. Kimguard Sterile Wrap. Instrument brushes $32.
 1Ltr Rapid Multi-Enzyme Cleaner Ÿ Proprietary combination enzyme Ÿ concentrate Ÿ Unparalleled combination of speed Ÿ and efficacy Ÿ Fully effective against proteins, Ÿ carbohydrates, lipids and Ÿ mucopolysaccharides
1Ltr Rapid Multi-Enzyme Cleaner Ÿ Proprietary combination enzyme Ÿ concentrate Ÿ Unparalleled combination of speed Ÿ and efficacy Ÿ Fully effective against proteins, Ÿ carbohydrates, lipids and Ÿ mucopolysaccharides
Daraclean Selection. Finding the right formulation MAGNAFLUX SOURCE
 Daraclean Selection Finding the right formulation MAGNAFLUX SOURCE - 2005 Daraclean Cleaners There are fourteen different Daraclean aqueous cleaner formulations How do you choose the right one for a specific
Daraclean Selection Finding the right formulation MAGNAFLUX SOURCE - 2005 Daraclean Cleaners There are fourteen different Daraclean aqueous cleaner formulations How do you choose the right one for a specific
CLEANING AND HANDLING OF MICROPORT INSTRUMENTS The following languages are included in this packet:
 EN CLEANING AND HANDLING OF MICROPORT INSTRUMENTS 150802-1 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português
EN CLEANING AND HANDLING OF MICROPORT INSTRUMENTS 150802-1 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português
Validation Strategy for an Automated Endoscope Reprocessor
 Validation Strategy for an Automated Endoscope Reprocessor November 16, 2010 Bradley Catalone AER Validation Strategy System Approach page 1 AER Validation Strategy AER Scope-specific connectors Requires
Validation Strategy for an Automated Endoscope Reprocessor November 16, 2010 Bradley Catalone AER Validation Strategy System Approach page 1 AER Validation Strategy AER Scope-specific connectors Requires
Cleaning and Sanitizing
 Cleaning and Sanitizing Presented by Dalia M. El-Sheikh Mokhtar Harb Zainab Abd-Elkareem ١ CLEANING: is the complete removal food soil using appropriate detergent chemicals under recommended conditions.
Cleaning and Sanitizing Presented by Dalia M. El-Sheikh Mokhtar Harb Zainab Abd-Elkareem ١ CLEANING: is the complete removal food soil using appropriate detergent chemicals under recommended conditions.
ProGarda. Natural chlorine, chlorate, QAC and sythetic free process washing aids for the food industry
 ProGarda Natural chlorine, chlorate, QAC and sythetic free process washing aids for the food industry COMPLIES WITH MRL REGULATIONS CHLORINE, CHLORATES AND QAC s THE PROBLEM Chlorates used in plant protection
ProGarda Natural chlorine, chlorate, QAC and sythetic free process washing aids for the food industry COMPLIES WITH MRL REGULATIONS CHLORINE, CHLORATES AND QAC s THE PROBLEM Chlorates used in plant protection
Bio-Burden Reduction in Biological Laboratories
 Bio-Burden Reduction in Biological Laboratories In biological laboratories samples worked with can harbor microorganisms that could be pathogens. Work is also done with microorganisms that are known pathogens.
Bio-Burden Reduction in Biological Laboratories In biological laboratories samples worked with can harbor microorganisms that could be pathogens. Work is also done with microorganisms that are known pathogens.
Designing an effective cleaning procedure for medical devices through laboratory studies
 Designing an effective cleaning procedure for medical devices through laboratory studies Elizabeth Rivera Technical Service Specialist STERIS Corporation elizabeth_rivera@steris.com October 2012 Agenda
Designing an effective cleaning procedure for medical devices through laboratory studies Elizabeth Rivera Technical Service Specialist STERIS Corporation elizabeth_rivera@steris.com October 2012 Agenda
catalyst substrate active site
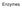 Enzymes ENZYMES Enzyme is protein in nature, it acts as an organic catalyst which speeds up many chemical reactions in organisms. Enzyme works on a substance called substrate and change it into product.
Enzymes ENZYMES Enzyme is protein in nature, it acts as an organic catalyst which speeds up many chemical reactions in organisms. Enzyme works on a substance called substrate and change it into product.
STERILIZATION VALIDATION IN THIS SECTION REPROCESSING VALIDATIONS FOR REUSABLE MEDICAL DEVICES. Radiation Sterilization Validation
 STERILIZATION VALIDATION All sterilization processes require validation of the efficacy and reproducibility of the process. Depending on the type of sterilization, this may be accomplished by partial,
STERILIZATION VALIDATION All sterilization processes require validation of the efficacy and reproducibility of the process. Depending on the type of sterilization, this may be accomplished by partial,
Module 7B DNA Collection, Extraction, and Analysis. Forensic Science Teacher Professional Development
 Module 7B DNA Collection, Extraction, and Analysis Forensic Science Teacher Professional Development Unit 3 Sample Collection, DNA Extraction, and DNA Quantitation Part 1 Sample Collection DNA can be found
Module 7B DNA Collection, Extraction, and Analysis Forensic Science Teacher Professional Development Unit 3 Sample Collection, DNA Extraction, and DNA Quantitation Part 1 Sample Collection DNA can be found
Stripol NI Ultra Product Code: Revised Date: 12/11/2012. Stripol NI Ultra
 Stripol NI Ultra DESCRIPTION Stripol NI Ultra is a non-regulated, one component liquid designed to rapidly strip nickel and nickel alloys from all substrates typically encountered in the metal finishing
Stripol NI Ultra DESCRIPTION Stripol NI Ultra is a non-regulated, one component liquid designed to rapidly strip nickel and nickel alloys from all substrates typically encountered in the metal finishing
Principles of Chlorine Dioxide Gas as a Decontamination Method
 Principles of Chlorine Dioxide Gas as a Decontamination Method In order for any decontamination method to be effective, the following points must be satisfied The decontamination method must: Be able to
Principles of Chlorine Dioxide Gas as a Decontamination Method In order for any decontamination method to be effective, the following points must be satisfied The decontamination method must: Be able to
A Better Way to a Healthier Environment ENHANCING BRAND CONTROLLING COSTS & PROTECTING HUMAN HEALTH
 A Better Way to a Healthier Environment SUSTAINABLE & ECO-FRIENDLY SANITATION FOR ENHANCING BRAND CONTROLLING COSTS & PROTECTING HUMAN HEALTH Environmental Partners / NA Green, Clean And Hygienic Biological
A Better Way to a Healthier Environment SUSTAINABLE & ECO-FRIENDLY SANITATION FOR ENHANCING BRAND CONTROLLING COSTS & PROTECTING HUMAN HEALTH Environmental Partners / NA Green, Clean And Hygienic Biological
AFFINITY HIS-TAG PURIFICATION
 DESCRIPTION Nickel Affinity Cartridges 5ml are used for purification of histidine-tagged proteins in native or denaturing conditions. This cartridge can be used with an automated chromatography system,
DESCRIPTION Nickel Affinity Cartridges 5ml are used for purification of histidine-tagged proteins in native or denaturing conditions. This cartridge can be used with an automated chromatography system,
511 ULTRAKOOL SS DESCRIPTION:
 511 ULTRAKOOL SS DESCRIPTION: UltraKool SS is a premium multi-purpose semi-synthetic, very low foaming, biostable, nonstaining metalworking fluid that is recommended for a multitude of machining and grinding
511 ULTRAKOOL SS DESCRIPTION: UltraKool SS is a premium multi-purpose semi-synthetic, very low foaming, biostable, nonstaining metalworking fluid that is recommended for a multitude of machining and grinding
SEASTROM Manufacturing Co., Inc Seastrom Street Twin Falls, Idaho Fax (208)
 MATERIAL CHARACTERISTICS - METAL Aluminum 2024 Aircraft Grade 1/3 the weight of steel Heat treat to increase hardness Good electrical conductivity (30% of copper) Good heat conductivity (70 btu/hr/ft²/
MATERIAL CHARACTERISTICS - METAL Aluminum 2024 Aircraft Grade 1/3 the weight of steel Heat treat to increase hardness Good electrical conductivity (30% of copper) Good heat conductivity (70 btu/hr/ft²/
TEXTILE INDUSTRY AND ENVIRONMENT. W.J.K.Dushyanthi Ranpatige Research Officer Industrial Technology Institute
 TEXTILE INDUSTRY AND ENVIRONMENT W.J.K.Dushyanthi Ranpatige Research Officer Industrial Technology Institute The textile industry One of the major contributors to many Asian economies and one of the main
TEXTILE INDUSTRY AND ENVIRONMENT W.J.K.Dushyanthi Ranpatige Research Officer Industrial Technology Institute The textile industry One of the major contributors to many Asian economies and one of the main
The stainless steels used in the manufacture of surgical instruments can be divided into two main classes.
 Introduction Instruments represent a significant material asset within the overall investment of a hospital. The care and maintenance these instruments receive is critical to their performance during surgery
Introduction Instruments represent a significant material asset within the overall investment of a hospital. The care and maintenance these instruments receive is critical to their performance during surgery
LIQUID PENETRANTS USER S MANUAL in accordance to specifications: EN 571, ASTME 1417, AMS 2644, MIL, ASME, DIN, UNI, BS, AFNOR, etc..
 LIQUID PENETRANTS USER S MANUAL in accordance to specifications: EN 571, ASTME 1417, AMS 2644, MIL, ASME, DIN, UNI, BS, AFNOR, etc.. The liquid penetrant examination method is an effective means for detecting
LIQUID PENETRANTS USER S MANUAL in accordance to specifications: EN 571, ASTME 1417, AMS 2644, MIL, ASME, DIN, UNI, BS, AFNOR, etc.. The liquid penetrant examination method is an effective means for detecting
Water The Universal Solvent
 1 Water The Universal Solvent Ever changing and Universally Different QLD SRACA September 2012 Water the universal solvent Water is the most fundamental requirement for your cleaning, thermal disinfection
1 Water The Universal Solvent Ever changing and Universally Different QLD SRACA September 2012 Water the universal solvent Water is the most fundamental requirement for your cleaning, thermal disinfection
Ideal Sterilization Method. Sterilization. Sterilization Practices in Healthcare Facilities
 Sterilization Practices in Healthcare Facilities Virtual Tour of Central Processing William A. Rutala, Ph.D., M.P.H. Director, Hospital Epidemiology, Occupational Health and Safety Program, UNC Health
Sterilization Practices in Healthcare Facilities Virtual Tour of Central Processing William A. Rutala, Ph.D., M.P.H. Director, Hospital Epidemiology, Occupational Health and Safety Program, UNC Health
Endoscope Washer Disinfector Cleaning Process Challenge Devices Research & Validation
 Endoscope Washer Disinfector Cleaning Process Challenge Devices Research & Validation Richard Bancroft, B.Sc (Hons), FRSB Science & Technical Director STERIS Corporation Registered Authorising Engineer
Endoscope Washer Disinfector Cleaning Process Challenge Devices Research & Validation Richard Bancroft, B.Sc (Hons), FRSB Science & Technical Director STERIS Corporation Registered Authorising Engineer
Low Temperature Processing. An Update on Guidelines, Standards and Requirements
 Low Temperature Processing An Update on Guidelines, Standards and Requirements Continuing Education Contact Hours Participants must complete the entire presentation/seminar to achieve successful completion
Low Temperature Processing An Update on Guidelines, Standards and Requirements Continuing Education Contact Hours Participants must complete the entire presentation/seminar to achieve successful completion
Loaner Programme Guidance
 Loaner Programme Guidance This document is valid only on the date of printing. In case of doubt about the date of printing, please re-print to ensure that the latest revised version of this document is
Loaner Programme Guidance This document is valid only on the date of printing. In case of doubt about the date of printing, please re-print to ensure that the latest revised version of this document is
Loaner Programme Guidance
 Loaner Programme Guidance This document is valid only on the date of printing. In case of doubt about the date of printing, please re-print to ensure that the latest revised version of this document is
Loaner Programme Guidance This document is valid only on the date of printing. In case of doubt about the date of printing, please re-print to ensure that the latest revised version of this document is
Monitoring your decontamination and reprocessing cycle
 Monitoring your decontamination and reprocessing cycle 1er Journee National sur la Sterilisation La Societe Marocaine de Sterilisation Maroc 2011 Ahmed Ghatwary/SteriGMS/Pharcomedic Index 1-Cleaning step,
Monitoring your decontamination and reprocessing cycle 1er Journee National sur la Sterilisation La Societe Marocaine de Sterilisation Maroc 2011 Ahmed Ghatwary/SteriGMS/Pharcomedic Index 1-Cleaning step,
ENDOSCOPE REPROCESSING AND INFECTION CONTROL FOR ENDOSCOPY
 ENDOSCOPE REPROCESSING AND INFECTION CONTROL FOR ENDOSCOPY Assist. Prof. Pochamana Phisalprapa, M.D., M.Sc. Department of Medicine, Faculty of Medicine Siriraj Hospital, Mahidol University GI ENDOSCOPY
ENDOSCOPE REPROCESSING AND INFECTION CONTROL FOR ENDOSCOPY Assist. Prof. Pochamana Phisalprapa, M.D., M.Sc. Department of Medicine, Faculty of Medicine Siriraj Hospital, Mahidol University GI ENDOSCOPY
: Focus MP 11 Multi-Purpose Cleaner
 SECTION 1: Identification 1.1. Identification Product form Product name Revision date: 04/15/2015 : Mixture Product code : 155-2101 : 1.2. Relevant identified uses of the substance or mixture and uses
SECTION 1: Identification 1.1. Identification Product form Product name Revision date: 04/15/2015 : Mixture Product code : 155-2101 : 1.2. Relevant identified uses of the substance or mixture and uses
ABCs of Validation. Ruth L. Petran. Corporate Scientist, Food Safety 03 April 2012
 1 ABCs of Validation Ruth L. Petran Corporate Scientist, Food Safety 03 April 2012 2 Topics Validation starts with product and process design Traditional validation Important to focus on in-plant situations
1 ABCs of Validation Ruth L. Petran Corporate Scientist, Food Safety 03 April 2012 2 Topics Validation starts with product and process design Traditional validation Important to focus on in-plant situations
March 28, 2019 Maintaining a Clean Brewery Presenter: Richard Rench Q&A Session
 March 28, 2019 Maintaining a Clean Brewery Presenter: Richard Rench Q&A Session 1. Does increasing caustic solution temp above 60-70ºC no longer increase effectiveness significantly, or does it just no
March 28, 2019 Maintaining a Clean Brewery Presenter: Richard Rench Q&A Session 1. Does increasing caustic solution temp above 60-70ºC no longer increase effectiveness significantly, or does it just no
0 50 Very soft Soft Medium Hard
 THE RIGHT WATER CHEMISTRY: UNDERSTANDING THE AQUEOUS INFLUENCE UPON METALWORKING FLUID PERFORMANCE Bob Trivett, Senior Chemist PICO Chemical Corporation Chicago Heights, Illinois December 10, 2003 INTRODUCTION
THE RIGHT WATER CHEMISTRY: UNDERSTANDING THE AQUEOUS INFLUENCE UPON METALWORKING FLUID PERFORMANCE Bob Trivett, Senior Chemist PICO Chemical Corporation Chicago Heights, Illinois December 10, 2003 INTRODUCTION
SANITIZING OF MILK CASE WASHERS AND TRANSPORT CONVEYORS: AN OVERVIEW OF CURRENT PRACTICES AND ADVANTAGES OF CHLORINE DIOXIDE USE
 SANITIZING OF MILK CASE WASHERS AND TRANSPORT CONVEYORS: AN OVERVIEW OF CURRENT PRACTICES AND ADVANTAGES OF CHLORINE DIOXIDE USE A PURELINE WHITE PAPER SEPTEMBER, 2010 1 EXECUTIVE SUMMARY; THE ISSUES There
SANITIZING OF MILK CASE WASHERS AND TRANSPORT CONVEYORS: AN OVERVIEW OF CURRENT PRACTICES AND ADVANTAGES OF CHLORINE DIOXIDE USE A PURELINE WHITE PAPER SEPTEMBER, 2010 1 EXECUTIVE SUMMARY; THE ISSUES There
PowerMax Soil DNA Isolation Kit
 PowerMax Soil DNA Isolation Kit Catalog No. Quantity 12988-10 10 Preps Instruction Manual Inhibitor Removal Technology (IRT) is a registered trademark of MO BIO Laboratories, Inc. and is covered by the
PowerMax Soil DNA Isolation Kit Catalog No. Quantity 12988-10 10 Preps Instruction Manual Inhibitor Removal Technology (IRT) is a registered trademark of MO BIO Laboratories, Inc. and is covered by the
Decontamination of Reusable Medical Devices: A Risk Management Perspective. By Paul Holland
 Decontamination of Reusable Medical Devices: A Risk Management Perspective. By Paul Holland Contents: Why we should manage risks and relevant definitions. Framework documentation. Perception to healthcare
Decontamination of Reusable Medical Devices: A Risk Management Perspective. By Paul Holland Contents: Why we should manage risks and relevant definitions. Framework documentation. Perception to healthcare
We combine enzymes and chemistry to enable the next level of low temperature washing with Lavergy Pro 104 L
 Home Care We combine enzymes and chemistry to enable the next level of low temperature washing with Lavergy Pro 104 L Add the Power of Lavergy High Performance at Low Washing Temperatures Little changes
Home Care We combine enzymes and chemistry to enable the next level of low temperature washing with Lavergy Pro 104 L Add the Power of Lavergy High Performance at Low Washing Temperatures Little changes
Introduction. Kit components. 50 Preps GF-PC-050. Product Catalog No. 200 Preps GF-PC Preps GF-PC Preps SAMPLE
 Introduction The GF-1 PCR Clean Up Kit is a system designed for rapid clean up of DNA bands ranging from 100bp to 20kb. The GF-1 PCR Clean Up Kit contains special buffers to provide the correct salt concentration
Introduction The GF-1 PCR Clean Up Kit is a system designed for rapid clean up of DNA bands ranging from 100bp to 20kb. The GF-1 PCR Clean Up Kit contains special buffers to provide the correct salt concentration
BREW. Protect Your Brand! Craft Brewery Sanitation Program. Complete Line of Cleaners/Sanitizers SSOP Support GHS Training Local Deliveries/Support
 BREW Craft Brewery Sanitation Program Protect Your Brand! Complete Line of Cleaners/Sanitizers SSOP Support GHS Training Local Deliveries/Support Providing Quality Maintenance and Chemical Specialty Solutions
BREW Craft Brewery Sanitation Program Protect Your Brand! Complete Line of Cleaners/Sanitizers SSOP Support GHS Training Local Deliveries/Support Providing Quality Maintenance and Chemical Specialty Solutions
3034 Owen Drive Antioch, TN USA Fax: Maybachstraße 10. Revised 01/17
 CLN VersaPak Modular Instrument Tray Symmetry Surgical Inc. 3034 Owen Drive Antioch, TN 37013 USA 1-800-251-3000 Fax: 1-615-964-5566 www.symmetrysurgical.com EC REP Symmetry Surgical GmbH 78532 Tuttlingen,
CLN VersaPak Modular Instrument Tray Symmetry Surgical Inc. 3034 Owen Drive Antioch, TN 37013 USA 1-800-251-3000 Fax: 1-615-964-5566 www.symmetrysurgical.com EC REP Symmetry Surgical GmbH 78532 Tuttlingen,
Responses to Questions for SYSTEM 1E Process Monitoring and Validation Webinar
 Responses to Questions for SYSTEM 1E Process Monitoring and Validation Webinar MONITORING THE SYSTEM 1E PROCESSOR Q: I want to confirm that there is no need for a Biological Indicator (BI) and that use
Responses to Questions for SYSTEM 1E Process Monitoring and Validation Webinar MONITORING THE SYSTEM 1E PROCESSOR Q: I want to confirm that there is no need for a Biological Indicator (BI) and that use
