HEALTH AND HUMAN SERVICES: HHS PROFILES
|
|
|
- Curtis Long
- 6 years ago
- Views:
Transcription
1 SMARTER STATE CASE STUDIES HEALTH AND HUMAN SERVICES: HHS PROFILES Using Expert Networking to Improve Safety and Efficiency of Medical Device Review TALENT BANK February 10, 2016 We would like to thank Jessica Hernandez and Nichole Rosamilia, Office of Science and Engineering Laboratories, Center for Devices and Radiological Health, U.S. Food and Drug Administration, for their assistance. This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License
2 BASIC INFORMATION Owner Center for Devices and Radiological Health, Food and Drug Administration, Department of Health and Human Services Project Contacts Jessica N. Hernandez, MA, MS, Team Leader, Innovation and Technology Solutions (ITS), Center for Devices and Radiological Health, U.S. Food and Drug Administration Nichole Rosamilia, Technical Information Specialist, Center for Devices and Radiological Health, U.S. Food and Drug Administration Project Summary An expert network for the regulatory review of medical devices Sector Health Audience Employees of the Department of Health and Human Services Problem that it is trying to solve It is difficult to quickly convene a qualified group of reviewers for the diversity of new medical devices coming to market. Platform Customized version of Harvard Catalyst Profiles Design basics Employees create profiles drawing from a number of data sources describing their skills and experiences. Representatives of the Office for Device Evaluation (ODE) use those expertise profiles to identify the most qualified individuals to participate in the regulatory review of a given medical device. KEY TAKEAWAYS What s new? Previously there was no rapid, systematic way for the ODE to identify capable reviewers within HHS resulting on a reliance on the rolodex approach involving the usual suspects. Incentives for participation The incentives for an individual to participate in HHS Profiles are threefold: 1) recognition of high-level departmental buy-in and interest in the tool; 2) the opportunity to play a meaningful part in improving public health; and 3) leveraging an outlet for articulating and sharing professional expertise. Challenges The central challenge for the initial HHS Profiles pilot involves the translation of a given medical device to be reviewed into a collection of relevant skills or knowledge areas which can be used as search parameters on the platform. Anticipated impact/metrics HHS Profiles will seek to increase the speed and effectiveness of the regulatory review of medical devices. Metrics of success will range from the diversity of disciplinary backgrounds represented in reviews to the average time required for the entire review process. Why is this project interesting? HHS Profiles is a clear example of the potential impacts on public life of better matching the supply of expertise within government (i.e., medical experts within HHS) to the demand for expertise in government (i.e., the need for qualified regulatory reviewers of an innovative new medical device).and searching for expertise, that can originate a wide variety of use cases. It is capturing expertise from institutional data such as human resources data on previous projects and positions.
3 THE CONTEXT The FDA s Center for Devices and Radiological Health (CDRH) manages the process of premarket approval and post-market review of all medical devices. CDRH ensures that patients and providers have timely and continued access to safe, effective, and high-quality medical devices and safe radiation-emitting products, and facilitates medical device innovation by advancing regulatory science, providing industry with predictable, consistent, transparent, and efficient regulatory pathways, and ensuring consumer confidence in devices marketed in the U.S. 1 Every year, the CDRH evaluates thousands of devices of varying degrees of patient risk and complexity many of which employ novel technology. To ensure the safety and efficacy of these products, the FDA is faced with the challenge of finding the right expertise to help it quickly and effectively assess new products. The pathway to regulatory review and compliance for low-risk items like tongue depressors is straightforward. But life-sustaining or high-risk devices such as pacemakers and breast implants require judicious and timely Premarket Approval (PMA) by those with the right know-how. If these medical devices reach patients prior to being properly tested, there may well be very real human costs. 2 Unlike post-market reviews, the PMA process is undertaken exclusively by internal staff. Previously, the FDA had wrestled with the idea of allowing those outside the agency more broadly to review medical devices, but this foundered out of a fear of conflict of interest and undue influence. Hence the agency has to rely upon its own internal staff augmented by some outside experts, who are hired as temporary employees to man these review panels. As is common in many governmental practices, such as patent examination or regulatory drafting, the FDA is faced with the task of evaluating large quantities of complex information with resort only to a too-small pool of people lacking the requisite diversity and laboring under the strain of too much work. 3 Traditionally, reviews are undertaken by a set of usual suspects identified by staff from the FDA Office of Device Evaluation (ODE) Beth Simone Noveck, Bridging the Knowledge Gap: In Search of Expertise, Democracy 34, Fall 2014, Beth Simone Noveck, Smart Citizens: Smarter State: The Technologies of Expertise and the Future of Governing. Cambrigdge: Harvard University Press, 2015.
4 THE CHALLENGE In its regulatory review efforts, the FDA confronts several challenges related to 4 : D Agility Finding and convening a qualified regulatory review panel can take as long as nine months. The FDA has also seen a rapid increase in new medical devices being developed and submitted for review: In 2010 and 2011, an average of 49.5 high-risk medical device recalls were initiated, compared to an average of 24 over the three preceding years; D Expertise The current pool from which experts are identified is limited and may not include people experienced with the specific potentially novel technologies featured in innovative medical devices; D Diversity It is hard to identify people from diverse disciplines with relevant prior knowledge at each of the many stages of device review; and D Complexity The regulatory review process lags behind the market in the ability to keep pace with advancements in technology. In addition, some devices require compliance with multiple sets of agency rules. These challenges have real effects on the medical device industry and, potentially, public health. Long review times are bad for firms due to the potential for significant financial stress after large up-front investments, as well as due to the opportunity costs sacrificed during the review timeline. 5 Ineffective reviews are bad for the public because devices with the potential to kill or injure may be approved for widespread use. On the other hand, unnecessarily long review times for truly safe products are bad for the public, since under these circumstances lifesaving products may take longer to reach patients. As of 2012, it takes an average of 266 days for a device to pass through the PMA process. 6 The average PMA review time for a medical device increases markedly for unique devices those with no predicate devices. Unique, innovative new devices are reviewed over the course of, on average, 18.1 months 7.2 months longer than the approval time for the second such device. 7 Particularly regarding new and unique devices, extended review times are somewhat expected given the danger to public health that would arise from unsafe devices reaching market and, indeed, longer approval timeframes currently correlate to fewer subsequent reports of adverse 4 Beth Simone Noveck, If We Only Knew What We Know: Open Regulatory Review at the FDA, Yale Law & Policy Review 32, no. 2, 2014, 5 Ariel Dora Stern, Innovation under Regulatory Uncertainty: Evidence from Medical Technology, July 15, 2014, 6 FDA, Improvements in Device Review: Results of CDRH s Plan of Action for Premarket Review of Devices, November 2012, 7 Ariel Dora Stern, Innovation under Regulatory Uncertainty: Evidence from Medical Technology, July 15, 2014,
5 events. 8 HHS Profiles is rooted in the belief that better targeting using the expertise within the FDA and all of HHS for reviews could lead to faster, industry-benefitting reviews that do not sacrifice thoroughness or safety. EXPERIMENTATION WITH HHS PROFILES As part of a broader effort across HHS, the FDA s Office of Science and Engineering Laboratories (OSEL) at CDRH is launching an expert networking pilot project in response to these regulatory challenges. HHS Profiles is the agency s attempt to identify, quickly and intelligently, reviewers with specific areas of expertise for the pre-market review of medical devices. The FDA is partnering with the GovLab and its MacArthur Foundation Research Network on Opening Governance to develop and implement pilot projects to assess the effectiveness of using Profiles in this way. Profiles imports and analyzes white pages (detailed contact) information, publications, and other data sources to create and maintain a complete, searchable library of web-based electronic CVs for experts within HHS. Profiles uses the Harvard Profiles Research Networking Software 9 platform developed by Harvard Medical School with support from the National Institutes of Health. The platform s User Group includes over 300 members from institutions around the world, such as the medical and biomedical faculties at Harvard, Penn State, Boston University, and UCSF. 10 The HHS Profiles pilot program which will be launched in 2016 with funding from the General Services Administration s The Great Pitch investment contest 11 will evaluate several potential impacts of the introduction of this piece of expert networking software during the medical device review process. Within the target population of FDA and HHS personnel that Profiles is uploading into its database, it is possible to compare targeting participation with Profiles against status quo methods to understand the impact of general matching, and of matching based on academic degrees and publications. Does targeting on the basis of academic credentials have the downside of reaching those who are too busy to help and whose professional norms discourage such volunteer activity? Good academics may, for example, turn out to be bad participants because they are trained to contribute in ways that are unhelpful to regulators (i.e., academics used to writing long articles are often not all that useful to busy government professionals). 12 The experimental rollout of the software will facilitate causal analysis of the software platform s 8 Diana M. Zuckerman, Paul Brown, Steven E. Nissen, Medical Device Recalls and the FDA Approval Process, February 2014, files_pilot/ Beth Simone Noveck, Smart Citizens: Smarter State: The Technologies of Expertise and the Future of Governing. Cambrigdge: Harvard University Press, 2015.
6 impact, including its effect on review panel composition, time needed for a panel to be convened, time required for a panel to review a new medical device, and subsequent safety outcomes for all products reviewed e.g. product recalls and adverse event reports. The results of this pilot have the potential to inform and improve the process of regulatory review and will be relevant to the design of more effective regulatory review. Moreover, adding empirical research into the rollout of HHS Profiles is a chance to experiment not simply between policies, but with how we make policy. The agile empirical manner in which the experimentation will occur is meant to help develop replicable methods for studying governance innovations in the wild, and accelerating the pace of research in government as a result. ABOUT THE GOVLAB The GovLab s mission is to improve people s lives by changing how we govern. Our goal is to strengthen the ability of institutions including but not limited to governments and people to work more openly, collaboratively, effectively and legitimately to make better decisions and solve public problems. For more information, please visit: ABOUT SMARTER STATE New tools what GovLab calls technologies of expertise are making it possible to match the supply of citizen expertise to the demand for it in government. Smarter State is a GovLab initiative to design and test how public decision-making could improve if institutions knew how to use the technologies of expertise to tap the wisdom of citizens and civil servants.
Enhancing Staff Training for New Medical Device Reviewers
 Enhancing Staff Training for New Medical Device Reviewers Carole C. Carey, BSEE, MEng. carole.carey@fda.hhs.gov Center for Devices and Radiological Health U.S. Food and Drug Administration TOPICS Addressing
Enhancing Staff Training for New Medical Device Reviewers Carole C. Carey, BSEE, MEng. carole.carey@fda.hhs.gov Center for Devices and Radiological Health U.S. Food and Drug Administration TOPICS Addressing
General Comments. May 30, Division of Dockets Management (HFA-305) Food and Drug Administration 5630 Fishers Lane, Room 1061 Rockville, MD 20852
 701 Pennsylvania Avenue, Ste. 800 Washington, DC 20004 2654 Tel: 202 783 8700 Fax: 202 783 8750 www.advamed.org Division of Dockets Management (HFA-305) Food and Drug Administration 5630 Fishers Lane,
701 Pennsylvania Avenue, Ste. 800 Washington, DC 20004 2654 Tel: 202 783 8700 Fax: 202 783 8750 www.advamed.org Division of Dockets Management (HFA-305) Food and Drug Administration 5630 Fishers Lane,
MODEL-BASED DRUG DEVELOPMENT: A FDA CRITICAL PATH OPPORTUNITY
 MODEL-BASED DRUG DEVELOPMENT: A FDA CRITICAL PATH OPPORTUNITY Donald R. Stanski MD Scientific Advisor, Office of the Commissioner, FDA Professor of Anesthesia Stanford University, CA THE OPINIONS EXPRESSED
MODEL-BASED DRUG DEVELOPMENT: A FDA CRITICAL PATH OPPORTUNITY Donald R. Stanski MD Scientific Advisor, Office of the Commissioner, FDA Professor of Anesthesia Stanford University, CA THE OPINIONS EXPRESSED
Bipartisan Policy Center, Top Medical Innovation Priorities
 Bipartisan Policy Center, Top Medical Priorities FDA: Advancing Medical is a Bipartisan Policy Center initiative led by former Senator Bill Frist, MD, former Congressman Bart Gordon, and BPC Health Initiative
Bipartisan Policy Center, Top Medical Priorities FDA: Advancing Medical is a Bipartisan Policy Center initiative led by former Senator Bill Frist, MD, former Congressman Bart Gordon, and BPC Health Initiative
Building the Elegant Credit Union
 Building the Elegant Credit Union Several years ago we wrote The Elegant Credit Union based on our extensive experience working with credit unions, banks, insurance companies, and other financial-services
Building the Elegant Credit Union Several years ago we wrote The Elegant Credit Union based on our extensive experience working with credit unions, banks, insurance companies, and other financial-services
Chapter 7E: Nurturing Human Capital/Focus on Staff
 Chapter 7E: Nurturing Human Capital/Focus on Staff Starting Points The following points made by Jim Collins (author of the bestseller Good to Great; Why Some Companies Make the Leap And others Don t) are
Chapter 7E: Nurturing Human Capital/Focus on Staff Starting Points The following points made by Jim Collins (author of the bestseller Good to Great; Why Some Companies Make the Leap And others Don t) are
THE CULTURE CANVAS A Working Guide and Checklist to Support the Development of a High-Performing Culture
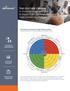 denison TM THE CULTURE CANVAS A Working Guide and Checklist to Support the Development of a High-Performing Culture The Denison Model of High Performance A Systems Approach to Understanding and Managing
denison TM THE CULTURE CANVAS A Working Guide and Checklist to Support the Development of a High-Performing Culture The Denison Model of High Performance A Systems Approach to Understanding and Managing
Making Models Publicly Available: Successes and Challenges
 Making Models Publicly Available: Successes and Challenges Tina Morrison PhD Office of Device Evaluation Center for Devices and Radiological Health U.S. Food and Drug Administration Modeling & Simulation
Making Models Publicly Available: Successes and Challenges Tina Morrison PhD Office of Device Evaluation Center for Devices and Radiological Health U.S. Food and Drug Administration Modeling & Simulation
Evidence-Based HR in Action
 AN EXCERPT FROM: Evidence-Based HR in Action R-1427-08-CS Case Studies Trusted Insights for Business Worldwide For the complete report, visit: www.conference-board.org/ebhr The Conference Board Mission
AN EXCERPT FROM: Evidence-Based HR in Action R-1427-08-CS Case Studies Trusted Insights for Business Worldwide For the complete report, visit: www.conference-board.org/ebhr The Conference Board Mission
U.S. FDA CENTER FOR DEVICES AND RADIOLOGICAL HEALTH UPDATE
 U.S. FDA CENTER FOR DEVICES AND RADIOLOGICAL HEALTH UPDATE Jeff Shuren Director Center for Devices and Radiological Health CDRH STRATEGIC PRIORITIES UPDATE The Center for Devices and Radiological Health
U.S. FDA CENTER FOR DEVICES AND RADIOLOGICAL HEALTH UPDATE Jeff Shuren Director Center for Devices and Radiological Health CDRH STRATEGIC PRIORITIES UPDATE The Center for Devices and Radiological Health
Collaboration for Research Integrity and Transparency
 Collaboration for Research Integrity and Transparency A PROGRAM OF YALE LAW SCHOOL, YALE SCHOOL OF MEDICINE AND YALE SCHOOL OF PUBLIC HEALTH To: From: Collaboration for Research Integrity and Transparency
Collaboration for Research Integrity and Transparency A PROGRAM OF YALE LAW SCHOOL, YALE SCHOOL OF MEDICINE AND YALE SCHOOL OF PUBLIC HEALTH To: From: Collaboration for Research Integrity and Transparency
Democratized Data Driving Discovery RENCI Strategic Plan 2017
 Democratized Data Driving Discovery RENCI Strategic Plan 2017 Stanley C. Ahalt, Ph.D. Director, Renaissance Computing Institute (RENCI) Professor, Department of Computer Science, UNC-Chapel Hill Associate
Democratized Data Driving Discovery RENCI Strategic Plan 2017 Stanley C. Ahalt, Ph.D. Director, Renaissance Computing Institute (RENCI) Professor, Department of Computer Science, UNC-Chapel Hill Associate
Health Data Research UK
 Health Data Research UK Applicant pack Job description and person specification Post: Location: Salary: Digital Innovation Hub Project Manager London 50,000-70,000 dependent on experience Duration: Initial
Health Data Research UK Applicant pack Job description and person specification Post: Location: Salary: Digital Innovation Hub Project Manager London 50,000-70,000 dependent on experience Duration: Initial
Data Transparency and Quality Metrics
 Data Transparency and Quality Metrics Ann Ferriter Division of Analysis and Program Operations Office of Compliance Center for Devices and Radiological Health Why are we interested in transparency? What
Data Transparency and Quality Metrics Ann Ferriter Division of Analysis and Program Operations Office of Compliance Center for Devices and Radiological Health Why are we interested in transparency? What
Murray Sheldon, MD Associate Director for Technology and Innovation Center for Devices and Radiological Health (CDRH) Office of the Center Director
 Murray Sheldon, MD Associate Director for Technology and Innovation Center for Devices and Radiological Health (CDRH) Office of the Center Director The National Academies Innovation Policy Forum September
Murray Sheldon, MD Associate Director for Technology and Innovation Center for Devices and Radiological Health (CDRH) Office of the Center Director The National Academies Innovation Policy Forum September
Compliance Central: CDRH 2017 Trends and 2018 Priorities
 Compliance Central: CDRH 2017 Trends and 2018 Priorities CAPT Sean M. Boyd, MPH, USPHS Deputy Director for Regulatory Affairs CDRH Office of Compliance Assessing Benefit-Risk Benefit Risk Other Factors
Compliance Central: CDRH 2017 Trends and 2018 Priorities CAPT Sean M. Boyd, MPH, USPHS Deputy Director for Regulatory Affairs CDRH Office of Compliance Assessing Benefit-Risk Benefit Risk Other Factors
Tina Morrison, Ph.D. Chair, Committee on Modeling and Simulation Office of the Chief Scientist U.S. Food and Drug Administration
 Tina Morrison, Ph.D. Chair, Committee on Modeling and Simulation Office of the Chief Scientist U.S. Food and Drug Administration Tina.Morrison@fda.hhs.gov Joseph Pellettiere Overview FDA s Strategic Plan
Tina Morrison, Ph.D. Chair, Committee on Modeling and Simulation Office of the Chief Scientist U.S. Food and Drug Administration Tina.Morrison@fda.hhs.gov Joseph Pellettiere Overview FDA s Strategic Plan
Regulatory Affairs in Life Science
 www.topra.org/careers Regulatory Affairs in Life Science Samantha Alsbury, Head of Professional Development, TOPRA ENABLING AND PROMOTING EXCELLENCE IN THE HEALTHCARE REGULATORY PROFESSION What is healthcare
www.topra.org/careers Regulatory Affairs in Life Science Samantha Alsbury, Head of Professional Development, TOPRA ENABLING AND PROMOTING EXCELLENCE IN THE HEALTHCARE REGULATORY PROFESSION What is healthcare
VOLUME RECRUITING. Guide to Using Video for Volume Recruiting
 VOLUME RECRUITING Guide to Using Video for Volume Recruiting 1 INTRODUCTION At a time where there is uncertainty facing the global economy, companies are thinking twice about where to allocate its funds
VOLUME RECRUITING Guide to Using Video for Volume Recruiting 1 INTRODUCTION At a time where there is uncertainty facing the global economy, companies are thinking twice about where to allocate its funds
CDRH Device Approval
 CDRH Device Approval Mary S. Pastel, ScD Deputy Director for Radiological Health Office of In Vitro Diagnostics and Radiological Health (OIR) Center for Devices and Radiological Health (CDRH) US Food and
CDRH Device Approval Mary S. Pastel, ScD Deputy Director for Radiological Health Office of In Vitro Diagnostics and Radiological Health (OIR) Center for Devices and Radiological Health (CDRH) US Food and
Core Values and Concepts
 Core Values and Concepts These beliefs and behaviors are embedded in highperforming organizations. They are the foundation for integrating key performance and operational requirements within a results-oriented
Core Values and Concepts These beliefs and behaviors are embedded in highperforming organizations. They are the foundation for integrating key performance and operational requirements within a results-oriented
TALENT ACQUISITION AND MANAGEMENT STRATEGIES for Hourly Workers
 TALENT ACQUISITION AND MANAGEMENT STRATEGIES for Hourly Workers An icims Guide to Attracting and Retaining the Best Hourly Talent 2016 icims, Inc. All rights reserved. TABLE OF Contents 3-4 5 6-13 14-19
TALENT ACQUISITION AND MANAGEMENT STRATEGIES for Hourly Workers An icims Guide to Attracting and Retaining the Best Hourly Talent 2016 icims, Inc. All rights reserved. TABLE OF Contents 3-4 5 6-13 14-19
Introduction to the FDA. Historical Context. Changes over Time FDA - BE-200 9/23/2004. Copyright C. S. Tritt, Ph.D. 1
 Introduction to the FDA C. S. Tritt, Ph.D. October 21, 2003 Historical Context In the early 1900 s people were being ripped off, made sick and even killed by bad food, patent medicines and cosmetics with
Introduction to the FDA C. S. Tritt, Ph.D. October 21, 2003 Historical Context In the early 1900 s people were being ripped off, made sick and even killed by bad food, patent medicines and cosmetics with
Evolving Regulatory Pathways for Medical Devices FDLI Annual Conference Access materials at fdli.org/annual2018
 Evolving Regulatory Pathways for Medical Devices CDRH Policy Perspective/Key Initiatives CDRH 2018-2020 Strategic Priorities Simplicity & Collaborative Communities Implementing statutory changes, Cures
Evolving Regulatory Pathways for Medical Devices CDRH Policy Perspective/Key Initiatives CDRH 2018-2020 Strategic Priorities Simplicity & Collaborative Communities Implementing statutory changes, Cures
External Defibrillator Improvement Initiative
 External Defibrillator Improvement Initiative November 2010 Center for Devices and Radiological Health U.S. Food and Drug Administration External Defibrillator Improvement Initiative Table of Contents
External Defibrillator Improvement Initiative November 2010 Center for Devices and Radiological Health U.S. Food and Drug Administration External Defibrillator Improvement Initiative Table of Contents
Canada s Archives: A vision and areas of focus for
 Canada s Archives: A vision and areas of focus for 2015-2025 Community consultation In January 2014, the archival community convened the Canadian Archives Summit: Towards a New Blueprint for Canada s Recorded
Canada s Archives: A vision and areas of focus for 2015-2025 Community consultation In January 2014, the archival community convened the Canadian Archives Summit: Towards a New Blueprint for Canada s Recorded
Assessing the Status of PAP-DIB Implementation
 Assessing the Status of PAP-DIB Implementation Hari Bucur-Marcu Four years have already passed since the heads of state and government convened in the Euro-Atlantic Partnership Council (EAPC) at the NATO
Assessing the Status of PAP-DIB Implementation Hari Bucur-Marcu Four years have already passed since the heads of state and government convened in the Euro-Atlantic Partnership Council (EAPC) at the NATO
The Johns Hopkins Bloomberg School of Public Health Managing Long-Term Care Services for Aging Populations NOTES:
 This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike License. Your use of this material constitutes acceptance of that license and the conditions of use of materials on this
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike License. Your use of this material constitutes acceptance of that license and the conditions of use of materials on this
E-PROCUREMENT CHANGE MANAGEMENT
 E-PROCUREMENT CHANGE MANAGEMENT PART ONE STRATEGY Digitizing business processes is inevitable; the challenge for procurement leaders will be changing perceptions internally to win the support needed to
E-PROCUREMENT CHANGE MANAGEMENT PART ONE STRATEGY Digitizing business processes is inevitable; the challenge for procurement leaders will be changing perceptions internally to win the support needed to
SAP Fieldglass Datasheet WHY VMS? LEVERAGING A VENDOR MANAGEMENT SYSTEM FOR YOUR EXTERNAL WORKFORCE
 SAP Fieldglass Datasheet WHY VMS? LEVERAGING A VENDOR MANAGEMENT SYSTEM FOR YOUR EXTERNAL WORKFORCE LEVERAGE VENDOR MANAGEMENT SYSTEM (VMS) TO PROCURE AND MANAGE YOUR FLEXIBLE WORKFORCE Finding and managing
SAP Fieldglass Datasheet WHY VMS? LEVERAGING A VENDOR MANAGEMENT SYSTEM FOR YOUR EXTERNAL WORKFORCE LEVERAGE VENDOR MANAGEMENT SYSTEM (VMS) TO PROCURE AND MANAGE YOUR FLEXIBLE WORKFORCE Finding and managing
March 2, Dear Chairman Upton, Ranking Member Pallone, and Representative DeGette:
 March 2, 2015 The Honorable Fred Upton Chairman Energy and Commerce Committee House of Representatives Washington, DC 20515 The Honorable Frank Pallone Ranking Member Energy and Commerce Committee House
March 2, 2015 The Honorable Fred Upton Chairman Energy and Commerce Committee House of Representatives Washington, DC 20515 The Honorable Frank Pallone Ranking Member Energy and Commerce Committee House
JOB LEVEL GUIDE Career Track: TECHNICAL AND ADMINISTRATIVE SUPPORT
 JOB LEVEL GUIDE Career Track: TECHNICAL AND ADMINISTRATIVE SUPPORT This career path typically includes staff whose primary duties are administrative. Support staff are responsible for providing support
JOB LEVEL GUIDE Career Track: TECHNICAL AND ADMINISTRATIVE SUPPORT This career path typically includes staff whose primary duties are administrative. Support staff are responsible for providing support
Conflict Resolution and Public Participation Center of Expertise (CPCX) FY Strategic Plan DRAFT DATE: 2 October 2014
 Conflict Resolution and Public Participation Center of Expertise (CPCX) FY 2015-2020 Strategic Plan DRAFT DATE: 2 October 2014 The U.S. Army Corps of Engineers (Corps) collaborates with a purpose: to deliver
Conflict Resolution and Public Participation Center of Expertise (CPCX) FY 2015-2020 Strategic Plan DRAFT DATE: 2 October 2014 The U.S. Army Corps of Engineers (Corps) collaborates with a purpose: to deliver
Improving Morale and Increasing Psychological Safety
 Improving Morale and Increasing Psychological Safety Steven Lovett, Ph.D. Chief, Psychology VA Palo Alto Lisa K. Kearney, Ph.D., ABPP Associate Director for Education VA Center for Integrated Healthcare
Improving Morale and Increasing Psychological Safety Steven Lovett, Ph.D. Chief, Psychology VA Palo Alto Lisa K. Kearney, Ph.D., ABPP Associate Director for Education VA Center for Integrated Healthcare
QbD BASED SCALE UP SERVICES THE DPT LABS APPROACH
 DPT Thought Leadership Issue 14: 4 th in a Series QbD BASED SCALE UP SERVICES THE DPT LABS APPROACH INTRODUCTION This paper describes the approach and benefits of using DPT Labs QbD Scale Up service and
DPT Thought Leadership Issue 14: 4 th in a Series QbD BASED SCALE UP SERVICES THE DPT LABS APPROACH INTRODUCTION This paper describes the approach and benefits of using DPT Labs QbD Scale Up service and
FDA s Center for Devices and Radiological Health: Strategic Priorities for 2017 and Beyond
 FDA s Center for Devices and Radiological Health: Strategic Priorities for 2017 and Beyond Jeff Shuren, MD, JD Center for Devices and Radiological Health U.S. Food and Drug Administration May 4, 2017 1
FDA s Center for Devices and Radiological Health: Strategic Priorities for 2017 and Beyond Jeff Shuren, MD, JD Center for Devices and Radiological Health U.S. Food and Drug Administration May 4, 2017 1
DXC Eclipse White Paper. How to mitigate the risks and leverage the power of the cloud
 How to mitigate the risks and leverage the power of the cloud Table of contents The time to move to the cloud is now 2 Cost benefits of a cloud-based approach 2 Beware of the race to services 3 How to
How to mitigate the risks and leverage the power of the cloud Table of contents The time to move to the cloud is now 2 Cost benefits of a cloud-based approach 2 Beware of the race to services 3 How to
Mr. Kyle Peters Vice President Operations Policy and Country Services World Bank 1818 H Street Washington, DC Dear Mr.
 Mr. Kyle Peters Vice President Operations Policy and Country Services World Bank 1818 H Street Washington, DC 20433 Dear Mr. Peters, It is our pleasure to share the following submission with you in response
Mr. Kyle Peters Vice President Operations Policy and Country Services World Bank 1818 H Street Washington, DC 20433 Dear Mr. Peters, It is our pleasure to share the following submission with you in response
STRATEGIC PLAN
 STRATEGIC PLAN 2018 2020 TABLE OF CONTENTS Welcome Context Founding Mandate Vision Mission Values Building the LMIC Together Our Approach Strategic Goals Measuring Success Monitoring Progress Ensuring
STRATEGIC PLAN 2018 2020 TABLE OF CONTENTS Welcome Context Founding Mandate Vision Mission Values Building the LMIC Together Our Approach Strategic Goals Measuring Success Monitoring Progress Ensuring
Accelerating the pace and impact of digital transformation
 RESEARCH REPORT Accelerating the pace and impact of digital transformation How the manufacturing organizations view the digital agenda A Harvard Business Review Analytics Services study in association
RESEARCH REPORT Accelerating the pace and impact of digital transformation How the manufacturing organizations view the digital agenda A Harvard Business Review Analytics Services study in association
Innovation and Technology in Breast Cancer 2019 A research initiative from Breast Cancer Foundation NZ.
 Request for Proposals Innovation and Technology in Breast Cancer 2019 A research initiative from Breast Cancer Foundation NZ. Overview Breast Cancer Foundation NZ (BCFNZ) is committed to funding research
Request for Proposals Innovation and Technology in Breast Cancer 2019 A research initiative from Breast Cancer Foundation NZ. Overview Breast Cancer Foundation NZ (BCFNZ) is committed to funding research
Ten Steps to Evaluate and Select A Mid-Market Budgeting, Forecasting, and Reporting Solution
 Ten Steps to Evaluate and Select A Mid-Market Budgeting, Forecasting, and Reporting Solution August 2007 BPM Partners, Inc. Six Landmark Square Stamford, CT 06901 203-359-5677 www.bpmpartners.com Introduction
Ten Steps to Evaluate and Select A Mid-Market Budgeting, Forecasting, and Reporting Solution August 2007 BPM Partners, Inc. Six Landmark Square Stamford, CT 06901 203-359-5677 www.bpmpartners.com Introduction
In today s evolving world of work, business is hungry for adaptive culture and speed-to-innovation. Because of this, HR is going Agile.
 In today s evolving world of work, business is hungry for adaptive culture and speed-to-innovation. Because of this, HR is going Agile. Agile methodology, popularized by software development, helps teams
In today s evolving world of work, business is hungry for adaptive culture and speed-to-innovation. Because of this, HR is going Agile. Agile methodology, popularized by software development, helps teams
Retraining and reskilling workers in the age of automation
 Pablo Illanes, Susan Lund, Mona Mourshed, Scott Rutherford, and Magnus Tyreman Retraining and reskilling workers in the age of automation January 2018 Executives increasingly see investing in retraining
Pablo Illanes, Susan Lund, Mona Mourshed, Scott Rutherford, and Magnus Tyreman Retraining and reskilling workers in the age of automation January 2018 Executives increasingly see investing in retraining
TOWARD A SUSTAINABLE BIOMEDICAL RESEARCH ENTERPRISE
 TOWARD A SUSTAINABLE BIOMEDICAL RESEARCH ENTERPRISE The American biomedical research enterprise is the world leader in health-related discovery and innovation. This system has trained students and faculty
TOWARD A SUSTAINABLE BIOMEDICAL RESEARCH ENTERPRISE The American biomedical research enterprise is the world leader in health-related discovery and innovation. This system has trained students and faculty
Clean Energy Fund Proposal
 Clean Energy Fund Proposal Association of Energy Engineers, New York Chapter March 17, 2015 2 Agenda Policy Background Market Development Portfolio Covering residential, commercial, and industrial sectors
Clean Energy Fund Proposal Association of Energy Engineers, New York Chapter March 17, 2015 2 Agenda Policy Background Market Development Portfolio Covering residential, commercial, and industrial sectors
WALKING THE AI TALK HEALTHCARE:
 HEALTHCARE: WALKING THE AI TALK ACCENTURE SURVEY SHOWS HEALTHCARE ORGANIZATIONS BELIEVE IN ARTIFICIAL INTELLIGENCE, AND THEY ARE LEADING THE WAY IN ADOPTING MACHINE LEARNING-ENABLED PROCESSES. Artificial
HEALTHCARE: WALKING THE AI TALK ACCENTURE SURVEY SHOWS HEALTHCARE ORGANIZATIONS BELIEVE IN ARTIFICIAL INTELLIGENCE, AND THEY ARE LEADING THE WAY IN ADOPTING MACHINE LEARNING-ENABLED PROCESSES. Artificial
PESIT Bangalore South Campus Hosur road, 1km before Electronic City, Bengaluru -100 Department of Information Sciences and Engineering
 Management & Entrepreneurship for IT industry INTERNAL ASSESSMENT TEST 1 Solution Document 1. A group of people wants to start a new engineering college. The group s long term vision is to make this college
Management & Entrepreneurship for IT industry INTERNAL ASSESSMENT TEST 1 Solution Document 1. A group of people wants to start a new engineering college. The group s long term vision is to make this college
Visionary Leadership. Systems Perspective. Student-Centered Excellence
 Core Values and Concepts These beliefs and behaviors are embedded in high-performing organizations. They are the foundation for integrating key performance and operational requirements within a results-oriented
Core Values and Concepts These beliefs and behaviors are embedded in high-performing organizations. They are the foundation for integrating key performance and operational requirements within a results-oriented
THE $1 TRILLION OPPORTUNITY IN DIGITAL SUPPORT FUNCTIONS
 THE $1 TRILLION OPPORTUNITY IN DIGITAL SUPPORT FUNCTIONS By Fabrice Roghé, Sukand Ramachandran, Yann Letourneux, and Nicholas Clark Most executives understand the power of digital technology to improve
THE $1 TRILLION OPPORTUNITY IN DIGITAL SUPPORT FUNCTIONS By Fabrice Roghé, Sukand Ramachandran, Yann Letourneux, and Nicholas Clark Most executives understand the power of digital technology to improve
Wales Millennium Centre Behavioral Competencies Framework 1
 Wales Millennium Centre Behavioural Competencies Framework Be Reflective Ensuring we understand who our customers are Taking time to listen to customers Proactively engaging with customers to find out
Wales Millennium Centre Behavioural Competencies Framework Be Reflective Ensuring we understand who our customers are Taking time to listen to customers Proactively engaging with customers to find out
McGill ITS Strategic Plan
 McGill Strategic Plan Context: Our 5 Year Roadmap Approved in April 205 Focuses on improvements to the following areas Teaching and Learning Research Student systems Campus Administration Must be cyclically
McGill Strategic Plan Context: Our 5 Year Roadmap Approved in April 205 Focuses on improvements to the following areas Teaching and Learning Research Student systems Campus Administration Must be cyclically
II. General Policies
 II. General Policies Enterprises should take fully into account established policies in the countries in which they operate, and consider the views of other stakeholders. In this regard: A. Enterprises
II. General Policies Enterprises should take fully into account established policies in the countries in which they operate, and consider the views of other stakeholders. In this regard: A. Enterprises
CAREERS IN SCIENCE WHAT ARE MY OPTIONS?
 CAREERS IN SCIENCE WHAT ARE MY OPTIONS? Lou Ann S. Brown, Ph.D. Assistant Dean School of Medicine Director Office of Postdoctoral Education Professor of Pediatrics and Biochemistry Children s Research
CAREERS IN SCIENCE WHAT ARE MY OPTIONS? Lou Ann S. Brown, Ph.D. Assistant Dean School of Medicine Director Office of Postdoctoral Education Professor of Pediatrics and Biochemistry Children s Research
Voluntary Pilot Meeting Preview: How will CDRH apply assessments in the voluntary program?
 Voluntary Pilot Meeting Preview: How will CDRH apply assessments in the voluntary program? Cisco Vicenty Case for Quality Program Manager Center for Devices and Radiological Health U.S. Food and Drug Administration,
Voluntary Pilot Meeting Preview: How will CDRH apply assessments in the voluntary program? Cisco Vicenty Case for Quality Program Manager Center for Devices and Radiological Health U.S. Food and Drug Administration,
Introduction 1. Retail execution: What works today 3. Additional enablers to retail execution that we see on the horizon
 Introduction 1 Retail execution: What works today 3 Additional enablers to retail execution that we see on the horizon 9 Retail execution: What is not working 12 Retail execution: What is required for
Introduction 1 Retail execution: What works today 3 Additional enablers to retail execution that we see on the horizon 9 Retail execution: What is not working 12 Retail execution: What is required for
Version: 4/27/16 APPENDIX C RBA GUIDE
 Version: 4/27/16 APPENDIX C RBA GUIDE Common Metrics Results-Based Accountability Guide CTSA Program Version I. Introduction 1 What is Results-Based Accountability? Results-Based Accountability ( RBA )
Version: 4/27/16 APPENDIX C RBA GUIDE Common Metrics Results-Based Accountability Guide CTSA Program Version I. Introduction 1 What is Results-Based Accountability? Results-Based Accountability ( RBA )
Tips for Eliminating Unexpected Risk from Incentive Compensation Plans
 5 Tips for Eliminating Unexpected Risk from Incentive Compensation Plans Overview To understand how your incentive compensation plan will work before it s rolled out to the sales force it is important
5 Tips for Eliminating Unexpected Risk from Incentive Compensation Plans Overview To understand how your incentive compensation plan will work before it s rolled out to the sales force it is important
Accelerating the pace and impact of digital transformation
 RESEARCH REPORT Accelerating the pace and impact of digital transformation How the consumer goods and retail organizations view the digital agenda A Harvard Business Review Analytic Services study in association
RESEARCH REPORT Accelerating the pace and impact of digital transformation How the consumer goods and retail organizations view the digital agenda A Harvard Business Review Analytic Services study in association
WHITE PAPER. Change Management: Driving the Long-Term Success of Your Workforce Management Solution
 WHITE PAPER Change Management: Driving the Long-Term Success of Your Workforce Management Solution How Do You Measure the Success of a Technology Project? When your organization embarks on a technology
WHITE PAPER Change Management: Driving the Long-Term Success of Your Workforce Management Solution How Do You Measure the Success of a Technology Project? When your organization embarks on a technology
Real World Evidence Generation in the 21 st Century: National Evaluation System for health Technology (NEST)
 Real World Evidence Generation in the 21 st Century: National Evaluation System for health Technology (NEST) Bill Murray, President & CEO IDEAL Conference Presentation May 5, 2017 1 www.mdic.org Vision
Real World Evidence Generation in the 21 st Century: National Evaluation System for health Technology (NEST) Bill Murray, President & CEO IDEAL Conference Presentation May 5, 2017 1 www.mdic.org Vision
Overcoming Statistical Challenges to the Reuse of Data within the Mini-Sentinel Distributed Database
 September 5, 2012 September 5, 2012 Meeting Summary Overcoming Statistical Challenges to the Reuse of Data within the Mini-Sentinel Distributed Database With passage of the Food and Drug Administration
September 5, 2012 September 5, 2012 Meeting Summary Overcoming Statistical Challenges to the Reuse of Data within the Mini-Sentinel Distributed Database With passage of the Food and Drug Administration
Adopting Site Quality Management to Optimize Risk-Based Monitoring
 WHITE PAPER Adopting Site Quality Management to Optimize Risk-Based Monitoring In today s pressure-packed environment, the quest for improved data quality at a lower cost is of paramount importance to
WHITE PAPER Adopting Site Quality Management to Optimize Risk-Based Monitoring In today s pressure-packed environment, the quest for improved data quality at a lower cost is of paramount importance to
Terms of Reference Framework for World Bank Financed Information and Communication Technologies (ICT) Expert Services
 Terms of Reference Framework for World Bank Financed Information and Communication Technologies (ICT) Expert Services The World Bank (ICT Sector Unit and the World Bank Institute) intends to establish
Terms of Reference Framework for World Bank Financed Information and Communication Technologies (ICT) Expert Services The World Bank (ICT Sector Unit and the World Bank Institute) intends to establish
How to Future-Proof Your Indirect Tax Team WHITE PAPER
 How to Future-Proof Your Indirect Tax Team WHITE PAPER Survive Smarter: Why Tax Digitalization Means Act Now or Get Left Behind 2 Technology is increasing the rate at which pressures impact your indirect
How to Future-Proof Your Indirect Tax Team WHITE PAPER Survive Smarter: Why Tax Digitalization Means Act Now or Get Left Behind 2 Technology is increasing the rate at which pressures impact your indirect
Human Resources and Organisational Management
 58 chapter 4 Human Resources and Organisational Organisational Development The institutional review process conducted in the year under review, led to significant restructuring and the organisational redesign.
58 chapter 4 Human Resources and Organisational Organisational Development The institutional review process conducted in the year under review, led to significant restructuring and the organisational redesign.
How it works: Questions from the OCAT 2.0
 Social Sector Practice How it works: Questions from the OCAT 2.0 OCAT 2.0 is an updated and improved version of our original OCAT survey. It asks nonprofit staff to rate their organization s operational
Social Sector Practice How it works: Questions from the OCAT 2.0 OCAT 2.0 is an updated and improved version of our original OCAT survey. It asks nonprofit staff to rate their organization s operational
Multifamily Energy Efficiency Initiative Developing Next Practice Energy Efficiency Options
 Multifamily Energy Efficiency Initiative Developing Next Practice Energy Efficiency Options Initiative Work Plan October 2012 Introduction The carbon emissions associated with electricity generation and
Multifamily Energy Efficiency Initiative Developing Next Practice Energy Efficiency Options Initiative Work Plan October 2012 Introduction The carbon emissions associated with electricity generation and
T H E B O T T O M L I N E
 P R O G R A M : C U S T O M E R R E L A T I O N S H I P M A N A G E M E N T R O I C A S E S T U D Y D O C U M E N T Q 93 J U N E 2 0 1 6 ACUMEN SOLUTIONS GOVERNMENT AGENCY A N A L Y S T S Rebecca Wettemann,
P R O G R A M : C U S T O M E R R E L A T I O N S H I P M A N A G E M E N T R O I C A S E S T U D Y D O C U M E N T Q 93 J U N E 2 0 1 6 ACUMEN SOLUTIONS GOVERNMENT AGENCY A N A L Y S T S Rebecca Wettemann,
Scott Gottlieb, MD Commissioner of Food and Drugs Food and Drug Administration New Hampshire Avenue Silver Spring, MD 20993
 July 31, 2018 Scott Gottlieb, MD Commissioner of Food and Drugs Food and Drug Administration 10903 New Hampshire Avenue Silver Spring, MD 20993 RE: Medical Device Safety Action Plan: Protecting Patients,
July 31, 2018 Scott Gottlieb, MD Commissioner of Food and Drugs Food and Drug Administration 10903 New Hampshire Avenue Silver Spring, MD 20993 RE: Medical Device Safety Action Plan: Protecting Patients,
MENTORING G UIDE MENTEES. for BY TRIPLE CREEK ASSOCIATES, INC Mentoring Guide for Mentees
 MENTORING G UIDE for MENTEES BY TRIPLE CREEK ASSOCIATES, INC. www.3creek.com 800-268-4422 Mentoring Guide for Mentees 2002 1 Table of Contents What Is Mentoring?... 3 Who Is Involved?... 3 Why Should People
MENTORING G UIDE for MENTEES BY TRIPLE CREEK ASSOCIATES, INC. www.3creek.com 800-268-4422 Mentoring Guide for Mentees 2002 1 Table of Contents What Is Mentoring?... 3 Who Is Involved?... 3 Why Should People
Transformation Enablement
 Offering Overview Transformation Enablement Ensure your organization s digital transformation initiatives achieve radically different levels of performance and quickly capture value with Cognizant Digital
Offering Overview Transformation Enablement Ensure your organization s digital transformation initiatives achieve radically different levels of performance and quickly capture value with Cognizant Digital
FINANCE & BUSINESS AT PENN STATE...
 Table of Contents FINANCE & BUSINESS AT PENN STATE... 3 Mission & Vision... 3 Organizational Profile... 3 F&B Situational Analysis... 3 F&B Alignment with University Priorities... 4 STRATEGIC PRIORITIES...
Table of Contents FINANCE & BUSINESS AT PENN STATE... 3 Mission & Vision... 3 Organizational Profile... 3 F&B Situational Analysis... 3 F&B Alignment with University Priorities... 4 STRATEGIC PRIORITIES...
Innovation and Technology in Breast Cancer 2018 A research initiative from Breast Cancer Foundation NZ.
 Request for Proposals Innovation and Technology in Breast Cancer 2018 A research initiative from Breast Cancer Foundation NZ. Overview Breast Cancer Foundation NZ (BCFNZ) is committed to funding research
Request for Proposals Innovation and Technology in Breast Cancer 2018 A research initiative from Breast Cancer Foundation NZ. Overview Breast Cancer Foundation NZ (BCFNZ) is committed to funding research
What do we need to realize in silico medicine in the future?
 October 11, 2016 EU Parliament, Brussels What do we need to realize in silico medicine in the future? Modeling and Simulation at U.S. FDA Tina Morrison, Ph.D. Chair, Modeling and Simulation Working Group
October 11, 2016 EU Parliament, Brussels What do we need to realize in silico medicine in the future? Modeling and Simulation at U.S. FDA Tina Morrison, Ph.D. Chair, Modeling and Simulation Working Group
Joy E. Spicer, President & CEO. March 28, 2011
 An Elegrity, Inc. White Paper 160 Pine Street, Suite 720 San Francisco, CA 94111 415-821-0900 http://www.elegrity.com http://blog.elegrity.com WHY LAW FIRMS CAN T SURVIVE WITHOUT BUSINESS PROCESS MANAGEMENT
An Elegrity, Inc. White Paper 160 Pine Street, Suite 720 San Francisco, CA 94111 415-821-0900 http://www.elegrity.com http://blog.elegrity.com WHY LAW FIRMS CAN T SURVIVE WITHOUT BUSINESS PROCESS MANAGEMENT
STRONG INNOVATORS ARE LEAN INNOVATORS
 STRONG INNOVATORS ARE LEAN INNOVATORS By Michael Ringel, Andrew Taylor, and Hadi Zablit This article is an excerpt from The Most Innovative Companies 2015: Four Factors that Differentiate Leaders (BCG
STRONG INNOVATORS ARE LEAN INNOVATORS By Michael Ringel, Andrew Taylor, and Hadi Zablit This article is an excerpt from The Most Innovative Companies 2015: Four Factors that Differentiate Leaders (BCG
HarbisonWalker International. Core Competencies
 HarbisonWalker International Core Competencies HWI Core Competency Model 1 TABLE OF CONTENTS Core Competencies Accountability/Drive 3 Innovation 4 Collaboration 5 Customer Focus 6 Adapting to Change 7
HarbisonWalker International Core Competencies HWI Core Competency Model 1 TABLE OF CONTENTS Core Competencies Accountability/Drive 3 Innovation 4 Collaboration 5 Customer Focus 6 Adapting to Change 7
Since its inception in 2007, the National Fund
 EXECUTIVE SUMMARY Systems Change in the National Fund for Workforce Solutions By Lisa Soricone November 2015 Since its inception in 2007, the National Fund for Workforce Solutions has been committed to
EXECUTIVE SUMMARY Systems Change in the National Fund for Workforce Solutions By Lisa Soricone November 2015 Since its inception in 2007, the National Fund for Workforce Solutions has been committed to
Innovating at Internet Speed: How to balance speed and efficiency in the digital age
 Innovating at Internet Speed: How to balance speed and efficiency in the digital age Alan W. Brown Professor of Entrepreneurship and Innovation Surrey Business School University of Surrey, UK alan.w.brown@surrey.ac.uk
Innovating at Internet Speed: How to balance speed and efficiency in the digital age Alan W. Brown Professor of Entrepreneurship and Innovation Surrey Business School University of Surrey, UK alan.w.brown@surrey.ac.uk
Developing the future of manufacturing and supply chain
 Developing the future of manufacturing and supply chain Industry 4.0 Industry 4.0 is the new source of substantial productivity gains Industry 4.0 spans an exciting array of digital technologies that are
Developing the future of manufacturing and supply chain Industry 4.0 Industry 4.0 is the new source of substantial productivity gains Industry 4.0 spans an exciting array of digital technologies that are
Medical Device Regulatory Science: A View from 20 Years at FDA s Device Center
 Medical Device Regulatory Science: A View from 20 Years at FDA s Device Center Greg Campbell, Ph.D. President, GCStat Consulting former Director, Division of Biostatistics, CDRH, FDA GCStat@verizon.net
Medical Device Regulatory Science: A View from 20 Years at FDA s Device Center Greg Campbell, Ph.D. President, GCStat Consulting former Director, Division of Biostatistics, CDRH, FDA GCStat@verizon.net
MEDICAL DEVICE CLINICAL INVESTIGATIONS AND ISO 14155
 MEDICAL DEVICE CLINICAL INVESTIGATIONS AND ISO 14155 EXECUTIVE SUMMARY Medical device regulations around the world generally require manufacturers of most types of medical devices to supply data as part
MEDICAL DEVICE CLINICAL INVESTIGATIONS AND ISO 14155 EXECUTIVE SUMMARY Medical device regulations around the world generally require manufacturers of most types of medical devices to supply data as part
JOB ONE: REIMAGINE TODAY S STATE GOVERNMENT WORKFORCE
 JOB ONE: REIMAGINE TODAY S STATE GOVERNMENT WORKFORCE Research Findings and Recommendations MARCH 2019 Competing for talent has long been challenging for state governments. Today s leaders recognize the
JOB ONE: REIMAGINE TODAY S STATE GOVERNMENT WORKFORCE Research Findings and Recommendations MARCH 2019 Competing for talent has long been challenging for state governments. Today s leaders recognize the
2. Is there a list of commonly used resources (textbooks, software, videos, case studies, etc.) for program directors for each program level?
 EDUCATION RELATED FREQUENTLY ASKED QUESTIONS 1. How often are the curriculum maps revised? The maps are reviewed annually by the curricula workgroup of the Council for Excellence in Education (CEE). Minor
EDUCATION RELATED FREQUENTLY ASKED QUESTIONS 1. How often are the curriculum maps revised? The maps are reviewed annually by the curricula workgroup of the Council for Excellence in Education (CEE). Minor
EXECUTIVE SUMMARY. Public engagement is a condition for effective governance
 EXECUTIVE SUMMARY Public engagement is a condition for effective governance Governments alone cannot deal with complex global and domestic challenges, such as climate change or soaring obesity levels.
EXECUTIVE SUMMARY Public engagement is a condition for effective governance Governments alone cannot deal with complex global and domestic challenges, such as climate change or soaring obesity levels.
A Guide to Competencies and Behavior Based Interviewing
 A Guide to Competencies and Behavior Based Interviewing 9.14.2015 HR Toolkit http://www.unitedwayofcolliercounty.org/maphr 2015 Competence is the ability of an individual to do a job properly. Job competencies
A Guide to Competencies and Behavior Based Interviewing 9.14.2015 HR Toolkit http://www.unitedwayofcolliercounty.org/maphr 2015 Competence is the ability of an individual to do a job properly. Job competencies
REQUIRED DOCUMENT FROM HIRING UNIT
 Terms of reference GENERAL INFORMATION Title: UNPDF Finalization Consultant Project Name : Strengthening Coordination Reports to: UN Resident Coordinator, UNPDF Steering Group, RC Office Duty Station:
Terms of reference GENERAL INFORMATION Title: UNPDF Finalization Consultant Project Name : Strengthening Coordination Reports to: UN Resident Coordinator, UNPDF Steering Group, RC Office Duty Station:
Dictionary of Functional Competencies for Career Banding
 Dictionary of Functional Competencies for Career Banding NC Office of State Personnel Revised April 18, 2007 Advocacy: Skills and abilities in developing and presenting the client s, patient s, and/or
Dictionary of Functional Competencies for Career Banding NC Office of State Personnel Revised April 18, 2007 Advocacy: Skills and abilities in developing and presenting the client s, patient s, and/or
HEALTHCARE RECRUITING LEXICON
 HEALTHCARE RECRUITING LEXICON Hiring the right candidates is critical for any healthcare organization looking to stay ahead of the challenges facing the healthcare industry. However, the terminology and
HEALTHCARE RECRUITING LEXICON Hiring the right candidates is critical for any healthcare organization looking to stay ahead of the challenges facing the healthcare industry. However, the terminology and
Artificial Intelligence in Life Sciences: The Formula for Pharma Success Across the Drug Lifecycle
 Volume XX, Issue 60 Artificial Intelligence in Life Sciences: The Formula for Pharma Success Across the Drug Lifecycle Artificial intelligence (AI) has the potential to transform the pharmaceutical industry.
Volume XX, Issue 60 Artificial Intelligence in Life Sciences: The Formula for Pharma Success Across the Drug Lifecycle Artificial intelligence (AI) has the potential to transform the pharmaceutical industry.
Business Plan. Public Service Secretariat
 2006-2008 Business Plan Public Service Secretariat Message from the Minister I am pleased to present the business plan for the Public Service Secretariat. The plan identifies Government s strategic direction
2006-2008 Business Plan Public Service Secretariat Message from the Minister I am pleased to present the business plan for the Public Service Secretariat. The plan identifies Government s strategic direction
a Recruitment Software Provider that fits
 5 Steps to Help You Win Over Key Stakeholders and Invest in a Talent Acquisition Software Suite That You Will Love. a Research ebook by: HIRING INSIGHTS WHY INVEST IN TALENT ACQUISITION? B2C technology
5 Steps to Help You Win Over Key Stakeholders and Invest in a Talent Acquisition Software Suite That You Will Love. a Research ebook by: HIRING INSIGHTS WHY INVEST IN TALENT ACQUISITION? B2C technology
PORTLAND PUBLIC SCHOOLS HUMAN RESOURCE SERVICES AND DELIVERY
 PORTLAND PUBLIC SCHOOLS HUMAN RESOURCE SERVICES AND DELIVERY January 2013 Overview Portland Public School District (the District or PPS) contracted with AKT to create a new vision and values for their
PORTLAND PUBLIC SCHOOLS HUMAN RESOURCE SERVICES AND DELIVERY January 2013 Overview Portland Public School District (the District or PPS) contracted with AKT to create a new vision and values for their
Community Empowerment (Scotland) Act Part 2 Community Planning Guidance
 Community Empowerment (Scotland) Act 2015 Part 2 Community Planning Guidance December 2016 Contents page number Part 1 Strategic overview of community planning 3 Foreword 4 Purpose of community planning
Community Empowerment (Scotland) Act 2015 Part 2 Community Planning Guidance December 2016 Contents page number Part 1 Strategic overview of community planning 3 Foreword 4 Purpose of community planning
Competencies. Working in Partnership. Creativity and Innovation Organisational and People Development
 Competencies Leadership Competency Framework - Summary Technical Expertise Demonstrates an avid interest in continuously enhancing current skills and learning new ones; applies advanced functional or technical
Competencies Leadership Competency Framework - Summary Technical Expertise Demonstrates an avid interest in continuously enhancing current skills and learning new ones; applies advanced functional or technical
SOLUTION BRIEF MAINFRAME SERVICES FROM CA TECHNOLOGIES
 SOLUTION BRIEF MAINFRAME SERVICES FROM CA TECHNOLOGIES Can You Make Your Mainframe Platform the Strongest Player in a Team Computing Environment, Supporting Mobile, Big Data, Massive Transaction Processing
SOLUTION BRIEF MAINFRAME SERVICES FROM CA TECHNOLOGIES Can You Make Your Mainframe Platform the Strongest Player in a Team Computing Environment, Supporting Mobile, Big Data, Massive Transaction Processing
Accenture Federal Services. US Department of Homeland Security: At a Tipping Point Agile Solutions or Aging Infrastructure?
 Accenture Federal Services US Department of Homeland Security: At a Tipping Point Agile Solutions or Aging Infrastructure? Despite our best efforts to stall the process of aging, growing older is a constant
Accenture Federal Services US Department of Homeland Security: At a Tipping Point Agile Solutions or Aging Infrastructure? Despite our best efforts to stall the process of aging, growing older is a constant
CFO LEADERSHIP BEYOND FINANCE PROGRAM
 CFO LEADERSHIP BEYOND FINANCE PROGRAM FEI Canada has partnered with Smith School of Business to develop a best in class uniquely positioned program for its members. Queen s Executive Education is ranked
CFO LEADERSHIP BEYOND FINANCE PROGRAM FEI Canada has partnered with Smith School of Business to develop a best in class uniquely positioned program for its members. Queen s Executive Education is ranked
How Often Should Companies Survey Employees?
 How Often Should Companies Survey Employees? Emerging Trends and Practical Considerations. 2017 Perceptyx, Inc. 28765 Single Oak Drive, Suite 250, Temecula California 92590 951.676.4414 www.perceptyx.com
How Often Should Companies Survey Employees? Emerging Trends and Practical Considerations. 2017 Perceptyx, Inc. 28765 Single Oak Drive, Suite 250, Temecula California 92590 951.676.4414 www.perceptyx.com
