Government-Owned Inventions; Availability for Licensing. SUMMARY: The inventions listed below are owned by an agency of the U.S.
|
|
|
- Ferdinand James
- 6 years ago
- Views:
Transcription
1 This document is scheduled to be published in the Federal Register on 06/10/2015 and available online at and on FDsys.gov [Billing Code P] DEPARTMENT OF HEALTH AND HUMAN SERVICES National Institutes of Health Government-Owned Inventions; Availability for Licensing AGENCY: National Institutes of Health, HHS ACTION: Notice SUMMARY: The inventions listed below are owned by an agency of the U.S. Government and are available for licensing in the U.S. in accordance with 35 U.S.C. 209 and 37 CFR Part 404 to achieve expeditious commercialization of results of federallyfunded research and development. Foreign patent applications are filed on selected inventions to extend market coverage for companies and may also be available for licensing. FOR FURTHER INFORMATION: Licensing information and copies of the U.S. patent applications listed below may be obtained by writing to the indicated licensing contact at the Office of Technology Transfer, National Institutes of Health, 6011 Executive Boulevard, Suite 325, Rockville, Maryland ; telephone:
2 2 7057; fax: A signed Confidential Disclosure Agreement will be required to receive copies of the patent applications. SUPPLEMENTARY INFORMATION: Technology descriptions follow. Boron Amino Acid Mimetics for PET Imaging of Cancer Description of Technology: Available for licensing and commercial development as imaging agents for positron emission tomography of cancer are boramino acid compounds. The inventors showed that mimetics created by substituting the carboxylate group (-COO - ) of an amino acid with trifluoroborate (-BF - 3 ) are metabolically stable and allow for the use of fluorene-18 ( 18 F) as the radiolabel. Using boroamino acid for 18 F-labeling allows for integrating the 18 F radiolabel into the core molecular backbone rather than the side-chains thus increasing the agent's target specificity. There is a direct relationship between amino acid uptake and cancer cell replication, where the uptake is extensively upregulated in most cancer cells. This uptake increases as cancer progresses, leading to greater uptake in high-grade tumors and metastases. Amino acids act as signaling molecules for proliferation and may also reprogram metabolic networks in the buildup of biomass. This invention provides for an unmet need for traceable amino acid mimics, including those based on naturallyoccurring amino acids, which may be non-invasively detected by imaging technology, including for clinical diagnosis and anti-cancer drug evaluation. Potential Commercial Applications: Cancer imaging
3 3 Anti-cancer drug development Competitive Advantages: Fluorene-18 labeling Metabolic stability Development Stage: Early-stage In vitro data available In vivo data available (animal) Inventors: Xiaoyuan Chen and Zhibo Liu (NIBIB) Publications: 1. Liu Z, et al. Preclinical evaluation of a high-affinity 18F-trifluoroborate octreotate derivative for somatostatin receptor imaging. J Nucl Med Sep;55(9): [PMID ] 2. Liu Z, et al. (18)F-trifluoroborate derivatives of [des-arg(10)]kallidin for imaging bradykinin b1 receptor expression with positron emission tomography. Mol Pharm Mar 2;12(3): [PMID ] Intellectual Property: HHS Reference No. E /0 - US Provisional Patent Application 62/155,085 filed April 30, 2015 Licensing Contact: Michael Shmilovich, Esq., CLP; or ; shmilovm@mail.nih.gov Collaborative Research Opportunity: The National Institute of Biomedical Imaging and Bioengineering is seeking statements of capability or interest from parties
4 4 interested in collaborative research to further develop, evaluate or commercialize Boramino Acid Mimetics for Use in Cancer Imaging. For collaboration opportunities, please contact Cecilia Pazman at Resolution Enhancement for Light Sheet Microscopy Systems Description of Technology: The invention pertains to a technique for enhancing the resolution of images in light sheet microscopy by adding additional enhanced depthof-focus optical arrangements and high numerical aperture objective lenses. The technique employs an arrangement of three objective lenses and a processor for combining captured images. The image composition utilizes the greater resolving power of the third high numerical aperture objective lens by imaging the light sheet and enhanced depth-of-focus arrangement resulting in improved overall resolution of the light sheet system. The depth of field arrangement could be a simple oscillation of the third objective, a "layer cake," or cubic phase mask component. Any loss in lateral resolution that results from the depth of field arrangement may be compensated for by deconvolution. In some embodiments, other optics, such as an axicon or annular aperture, can provide extended depth of field. Potential Commercial Applications: High speed imaging Fast single cell and cellular dynamics imaging Superresolution and single molecule imaging 3D single particle tracking 3D superresolution imaging in thick samples
5 5 Competitive Advantages: Resolution enhancement in light microscopy Development Stage: In vitro data available Inventors: Hari Shroff (NIBIB), Yicong Wu (NIBIB), Sara Abrahamsson Intellectual Property: HHS Reference No. E /0 - US Application No. 62/054,484 filed September 24, 2014 Related Technology: HHS Reference No. E /0 Licensing Contact: Michael Shmilovich, Esq., CLP; or ; shmilovm@mail.nih.gov Collaborative Research Opportunity: The National Institute of Biomedical Imaging and Bioengineering is seeking statements of capability or interest from parties interested in collaborative research to further develop, evaluate or commercialize Resolution Enhancement Technique for Light Sheet Microscopy Systems. For collaboration opportunities, please contact Cecilia Pazman at or pazmance@nhlbi.nih.gov. Device for Selective Partitioning of Frozen Cellular Products Description of Technology: Cryopreservation using liquid nitrogen frozen polyvinyl bags allows for storing cellular materials for extended periods while maintaining their activity and viability. Such bags are commonly used in the clinic to store blood products including blood cells, plasma, hematopoietic stem cells, umbilical cord blood for future uses including transplantation. These materials, typically obtained in limited quantities, may be of great therapeutic value, as is the case of stem cells or cord blood derived cells which can be used to potentially treat a number of diseases. Currently,
6 6 even if only a small portion of the cryopreserved sample is needed the whole bag must be thawed, wasting much of the sample or rendering the remaining sample susceptible to contamination since it cannot be effectively refrozen or sterilized. The present device meets an unmet need for retrieving a portion of a frozen sample stored in polyvinyl cryopreserved bags, resealing the remainder of the sample and preserving the cryopreserved state and integrity of the rest of the cellular product without compromising viability and sterility. Potential Commercial Applications: Cryopreservation Cellular Products Hematopoietic stem cells Umbilical cord blood ipscs Transplantation Chronic spinal cord injury Neurological disorders Cancer immunotherapy Cell banking Cell replacement therapy Competitive Advantages: Partitioning cryopreserved cell products Maintenance of sterility of partitioned product Maintenance of viability of partitioned product
7 7 Resealing of cryopreservation bag Multiple use of patient derived cellular products Development Stage: Prototype Inventors: Richard Childs, Sumithira Vasu, Herb Cullis, PJ Broussard, Kevin Clark, Eric Harting (all rights assigned to the US Government) Intellectual Property: HHS Reference No. E /0 - US Provisional App. 61/175,131 Int'l App. PCT/US2010/ Canadian App. 2,760,363 EP App IL App US Patent 8,790,597 US Patent App. 14/305,578 Licensing Contact: Michael Shmilovich, Esq., CLP; or ; shmilovm@mail.nih.gov Collaborative Research Opportunity: The National Heart, Lung, and Blood Institute is seeking statements of capability or interest from parties interested in collaborative research to further develop, evaluate, or commercialize Device for Partitioning Cryopreserved Cellular Products. For collaboration opportunities, please contact Cecilia Pazman, Ph.D. at or pazmance@nhlbi.nih.gov. Dated: June 4, Richard U. Rodriguez,
8 Acting Director, Office of Technology Transfer National Institutes of Health [FR Doc Filed: 6/9/ :45 am; Publication Date: 6/10/2015] 8
Stem Cell Services. Driving Innovation for Stem Cell Researchers
 Driving Innovation for Stem Cell Researchers Stem Cell Services Partner with us and have access to the most advanced and comprehensive stem cell services available today. 675 W. Kendall St. Cambridge,
Driving Innovation for Stem Cell Researchers Stem Cell Services Partner with us and have access to the most advanced and comprehensive stem cell services available today. 675 W. Kendall St. Cambridge,
Availability of Masked and De-identified Non-Summary Safety and Efficacy Data; Request for
 This document is scheduled to be published in the Federal Register on 06/04/2013 and available online at http://federalregister.gov/a/2013-13083, and on FDsys.gov 4160-01-P DEPARTMENT OF HEALTH AND HUMAN
This document is scheduled to be published in the Federal Register on 06/04/2013 and available online at http://federalregister.gov/a/2013-13083, and on FDsys.gov 4160-01-P DEPARTMENT OF HEALTH AND HUMAN
The science behind Betalutin : why is it unique? Roy H. Larsen PhD Sciencons AS, Oslo, Norway
 The science behind Betalutin : why is it unique? Roy H. Larsen PhD Sciencons AS, Oslo, Norway Speaker credentials Roy H. Larsen, PhD >25 years of experience in research on targeted radionuclide therapy
The science behind Betalutin : why is it unique? Roy H. Larsen PhD Sciencons AS, Oslo, Norway Speaker credentials Roy H. Larsen, PhD >25 years of experience in research on targeted radionuclide therapy
The New News in Stem Cell Research Andrés Bratt-Leal, PhD 12/1/2017
 The New News in Stem Cell Research Andrés Bratt-Leal, PhD 12/1/2017 Cell Therapy and Parkinson s Disease Very specific neural degeneration >50% of DA neurons are gone by diagnosis 1 million in the USA,
The New News in Stem Cell Research Andrés Bratt-Leal, PhD 12/1/2017 Cell Therapy and Parkinson s Disease Very specific neural degeneration >50% of DA neurons are gone by diagnosis 1 million in the USA,
Measurement of Hematopoietic Stem Cell Potency Prior to Transplantation
 WHITE PAPER Measurement of Hematopoietic Stem Cell Potency Prior to Transplantation February, 2009 This White Paper is a forward-looking statement. It represents the present state of the art and future
WHITE PAPER Measurement of Hematopoietic Stem Cell Potency Prior to Transplantation February, 2009 This White Paper is a forward-looking statement. It represents the present state of the art and future
A gateway to academic excellence for Biotech and Pharma
 A gateway to academic excellence for Biotech and Pharma 6 8 12 Connecting meaningful endpoints: Ksilink s comprehensive translational effort Ksilink s patient centered, translational technology Engaging
A gateway to academic excellence for Biotech and Pharma 6 8 12 Connecting meaningful endpoints: Ksilink s comprehensive translational effort Ksilink s patient centered, translational technology Engaging
Fujifilm s Initiatives in Regenerative Medicine
 Fujifilm s Initiatives in Regenerative Medicine Shigetaka Komori Chairman and CEO FUJIFILM Holdings Corporation March 30, 2015 Priority Business: Healthcare Expand business with focus on six core business
Fujifilm s Initiatives in Regenerative Medicine Shigetaka Komori Chairman and CEO FUJIFILM Holdings Corporation March 30, 2015 Priority Business: Healthcare Expand business with focus on six core business
GUIDANCE ON THE EVALUATION OF NON ACCREDITED QUALIFICATIONS
 GUIDANCE ON THE EVALUATION OF NON ACCREDITED QUALIFICATIONS 1. Introduction 1.1 This document provides guidance notes for the assessment of academic qualifications that have not been formally accredited
GUIDANCE ON THE EVALUATION OF NON ACCREDITED QUALIFICATIONS 1. Introduction 1.1 This document provides guidance notes for the assessment of academic qualifications that have not been formally accredited
REIMAGINING DRUG DEVELOPMENT:
 Biology Reconstructed REIMAGINING DRUG DEVELOPMENT: Accurate Disease Modeling To Drive Successful Therapies Julia Kirshner, CEO julia@zpredicta.com 1 SUCCESS RATES OF DRUG DEVELOPMENT ARE LOW, " PARTICULARLY
Biology Reconstructed REIMAGINING DRUG DEVELOPMENT: Accurate Disease Modeling To Drive Successful Therapies Julia Kirshner, CEO julia@zpredicta.com 1 SUCCESS RATES OF DRUG DEVELOPMENT ARE LOW, " PARTICULARLY
Cytomics in Action: Cytokine Network Cytometry
 Cytomics in Action: Cytokine Network Cytometry Jonni S. Moore, Ph.D. Director, Clinical and Research Flow Cytometry and PathBioResource Associate Professor of Pathology & Laboratory Medicine University
Cytomics in Action: Cytokine Network Cytometry Jonni S. Moore, Ph.D. Director, Clinical and Research Flow Cytometry and PathBioResource Associate Professor of Pathology & Laboratory Medicine University
European Regenerative Medicine Firms & Their Strategic Approaches. Michael Morrison University of York
 European Regenerative Medicine Firms & Their Strategic Approaches Michael Morrison University of York OVERVIEW Creating the European RM Universe of firms Characterizing the European RM Universe Strategic
European Regenerative Medicine Firms & Their Strategic Approaches Michael Morrison University of York OVERVIEW Creating the European RM Universe of firms Characterizing the European RM Universe Strategic
Molecular Imaging: Definition, Overview and Goals
 This tutorial will define what is currently considered molecular imaging. It will provide history and an overview, discuss the goals and the advantages of molecular imaging. It will clarify what is and
This tutorial will define what is currently considered molecular imaging. It will provide history and an overview, discuss the goals and the advantages of molecular imaging. It will clarify what is and
HCT/P Regulation vs 361 Products
 HCT/P Regulation - 351 vs 361 Products Presented by: Paul Gadiock February 15, 2017 Arent Fox LLP Washington, DC New York, NY Los Angeles, CA San Francisco, CA 1 Presentation Overview Introduction Public
HCT/P Regulation - 351 vs 361 Products Presented by: Paul Gadiock February 15, 2017 Arent Fox LLP Washington, DC New York, NY Los Angeles, CA San Francisco, CA 1 Presentation Overview Introduction Public
PROTOCOL FOR A RESEARCH SAMPLE REPOSITORY FOR ALLOGENEIC HEMATOPOIETIC STEM CELL TRANSPLANTATION, OTHER CELLULAR THERAPIES AND MARROW TOXIC INJURIES
 Center for International Blood and Marrow Transplant Research PROTOCOL FOR A RESEARCH SAMPLE REPOSITORY FOR ALLOGENEIC HEMATOPOIETIC STEM CELL TRANSPLANTATION, OTHER CELLULAR THERAPIES AND MARROW TOXIC
Center for International Blood and Marrow Transplant Research PROTOCOL FOR A RESEARCH SAMPLE REPOSITORY FOR ALLOGENEIC HEMATOPOIETIC STEM CELL TRANSPLANTATION, OTHER CELLULAR THERAPIES AND MARROW TOXIC
Managing Risk and Uncertainty Through the Drug Life cycle. Recent FDA Initiatives. Theresa Mullin, PhD
 Managing Risk and Uncertainty Through the Drug Life cycle Recent FDA Initiatives Theresa Mullin, PhD Director Office of Strategic Programs US FDA Center for Drug Evaluation and Research October 14, 2014
Managing Risk and Uncertainty Through the Drug Life cycle Recent FDA Initiatives Theresa Mullin, PhD Director Office of Strategic Programs US FDA Center for Drug Evaluation and Research October 14, 2014
Des cellules-souches dans le poumon : pourquoi faire?
 Des cellules-souches dans le poumon : pourquoi faire? Karl-Heinz Krause Dept. of Pathology and Immunology, Medical Faculty Dept. of Genetic and Laboratory Medicine, University Hospitals Geneva, Switzerland
Des cellules-souches dans le poumon : pourquoi faire? Karl-Heinz Krause Dept. of Pathology and Immunology, Medical Faculty Dept. of Genetic and Laboratory Medicine, University Hospitals Geneva, Switzerland
Office for Human Subject Protection. University of Rochester
 POLICY 1. Purpose Outline the responsibilities and regulatory requirements when conducting human subject research that involves the use of drugs, agents, biological products, or nutritional products (e.g.,
POLICY 1. Purpose Outline the responsibilities and regulatory requirements when conducting human subject research that involves the use of drugs, agents, biological products, or nutritional products (e.g.,
Innovating out of Crisis
 Innovating out of Crisis Shigetaka Komori Chairman and CEO FUJIFILM Holdings Corporation 1 st June, 2015 Business Fields of Fujifilm Group Digital Camera Photo book Optical devise FY2015/3 Digital minilab
Innovating out of Crisis Shigetaka Komori Chairman and CEO FUJIFILM Holdings Corporation 1 st June, 2015 Business Fields of Fujifilm Group Digital Camera Photo book Optical devise FY2015/3 Digital minilab
Blue Marble University Biology Course Handbook
 Blue Marble University Biology Course Handbook A Summary of Course Descriptions Courses and Content Subject to Change Without Notice BlueMarbleUniversity.com Info@bluemarbleuniversity.com Stem Cell Biology
Blue Marble University Biology Course Handbook A Summary of Course Descriptions Courses and Content Subject to Change Without Notice BlueMarbleUniversity.com Info@bluemarbleuniversity.com Stem Cell Biology
Nanotechnology: A Brief History and Its Convergence with Medicine. Weston Daniel, PhD Director of Program Management
 Nanotechnology: A Brief History and Its Convergence with Medicine Weston Daniel, PhD Director of Program Management Outline Introduction The Nanoscale Applications Realization of a Vision There s Plenty
Nanotechnology: A Brief History and Its Convergence with Medicine Weston Daniel, PhD Director of Program Management Outline Introduction The Nanoscale Applications Realization of a Vision There s Plenty
Medical Devices; Immunology and Microbiology Devices; Classification of the Nucleic Acid-
 This document is scheduled to be published in the Federal Register on 10/16/2017 and available online at https://federalregister.gov/d/2017-22287, and on FDsys.gov 4164-01-P DEPARTMENT OF HEALTH AND HUMAN
This document is scheduled to be published in the Federal Register on 10/16/2017 and available online at https://federalregister.gov/d/2017-22287, and on FDsys.gov 4164-01-P DEPARTMENT OF HEALTH AND HUMAN
US FDA: CMC Issues for INDs
 ISBTC Global Regulatory Summit October 29, 2008 US FDA: CMC Issues for INDs Keith Wonnacott, Ph.D. keith.wonnacott@fda.hhs.gov US Food and Drug Administration Center for Biologics Evaluation and Research
ISBTC Global Regulatory Summit October 29, 2008 US FDA: CMC Issues for INDs Keith Wonnacott, Ph.D. keith.wonnacott@fda.hhs.gov US Food and Drug Administration Center for Biologics Evaluation and Research
NUCLEAR REGULATORY COMMISSION [NRC ] Yttrium-90 Microsphere Brachytherapy Sources and Devices TheraSphere.
![NUCLEAR REGULATORY COMMISSION [NRC ] Yttrium-90 Microsphere Brachytherapy Sources and Devices TheraSphere. NUCLEAR REGULATORY COMMISSION [NRC ] Yttrium-90 Microsphere Brachytherapy Sources and Devices TheraSphere.](/thumbs/75/72620659.jpg) This document is scheduled to be published in the Federal Register on 11/07/2017 and available online at https://federalregister.gov/d/2017-24129, and on FDsys.gov [7590-01-P] NUCLEAR REGULATORY COMMISSION
This document is scheduled to be published in the Federal Register on 11/07/2017 and available online at https://federalregister.gov/d/2017-24129, and on FDsys.gov [7590-01-P] NUCLEAR REGULATORY COMMISSION
Copyright. Jeremiah J. Kelly (2015). All rights reserved. Further dissemination without express written consent strictly prohibited.
 Statutory Framework for Biologics Drugs Investigational Use Application IND Pre-Market Approval Applications 505(b)(1) NDA 505(b)(2) NDA 505(j) ANDA Over-the-Counter (OTC) Non- Rx Drugs Monograph Biologics
Statutory Framework for Biologics Drugs Investigational Use Application IND Pre-Market Approval Applications 505(b)(1) NDA 505(b)(2) NDA 505(j) ANDA Over-the-Counter (OTC) Non- Rx Drugs Monograph Biologics
Cell Therapy Services Your Product. Our Passion.
 Pharma&Biotech Cell Therapy Services Your Product. Our Passion. Complete Portfolio of Services for cgmp Manufacturing of Cell-based Products Pharma&Biotech Cell Therapy Services for cgmp Manufacturing
Pharma&Biotech Cell Therapy Services Your Product. Our Passion. Complete Portfolio of Services for cgmp Manufacturing of Cell-based Products Pharma&Biotech Cell Therapy Services for cgmp Manufacturing
Partnering with the NIH: Technology Transfer. Why partner?
 Partnering with the NIH: Technology Transfer Jennifer Wong, M.S. Technology Development Coordinator National Institute of Mental Health Why partner? Enhance company s portfolio De-risks developing a technology
Partnering with the NIH: Technology Transfer Jennifer Wong, M.S. Technology Development Coordinator National Institute of Mental Health Why partner? Enhance company s portfolio De-risks developing a technology
Research and Innovation in Drug Discovery and Diagnostics
 INTERNATIONAL CONFERENCE Intellectual Property and Health Innovation Challenges for the future April 28, 2014, Athens, Greece Research and Innovation in Drug Discovery and Diagnostics Alexander Pintzas,
INTERNATIONAL CONFERENCE Intellectual Property and Health Innovation Challenges for the future April 28, 2014, Athens, Greece Research and Innovation in Drug Discovery and Diagnostics Alexander Pintzas,
NIH Public Access Author Manuscript Biochem Pharmacol (Los Angel). Author manuscript; available in PMC 2014 October 24.
 NIH Public Access Author Manuscript Published in final edited form as: Biochem Pharmacol (Los Angel). ; 1:. doi:10.4172/2167-0501.1000e128. Application of Positron Emission Tomography in Drug Development
NIH Public Access Author Manuscript Published in final edited form as: Biochem Pharmacol (Los Angel). ; 1:. doi:10.4172/2167-0501.1000e128. Application of Positron Emission Tomography in Drug Development
HIGH SCHOOL STUDENT SCIENCE WEEK. St. Paul s Hospital Vancouver, BC
 HIGH SCHOOL STUDENT SCIENCE WEEK St. Paul s Hospital Vancouver, BC Sponsors 2 AGENDA Location: UBC James Hogg Research Centre (JHRC), St. Paul s Hospital, Room 166 Burrard Building, 1081 Burrard Street,
HIGH SCHOOL STUDENT SCIENCE WEEK St. Paul s Hospital Vancouver, BC Sponsors 2 AGENDA Location: UBC James Hogg Research Centre (JHRC), St. Paul s Hospital, Room 166 Burrard Building, 1081 Burrard Street,
Patient Handbook on Stem Cell Therapies
 Patient Handbook on Stem Cell Therapies WWW.ISSCR.ORG WWW.CLOSERLOOKATSTEMCELLS.ORG Patient Handbook on Stem Cell Therapies Introduction We have all heard about the extraordinary promise that stem cell
Patient Handbook on Stem Cell Therapies WWW.ISSCR.ORG WWW.CLOSERLOOKATSTEMCELLS.ORG Patient Handbook on Stem Cell Therapies Introduction We have all heard about the extraordinary promise that stem cell
Stem Cell Principle -
 Effective Date: 31.10.2017 Doc ID: 20290214 Version: 1.0 Status: Approved Planned Effective Date: 31-Oct-2017 00:00 CET (Server Date) Stem Cell Principle - Rationale Research on human stem cells and their
Effective Date: 31.10.2017 Doc ID: 20290214 Version: 1.0 Status: Approved Planned Effective Date: 31-Oct-2017 00:00 CET (Server Date) Stem Cell Principle - Rationale Research on human stem cells and their
Antisense Therapeutics Ltd ASX:ANP January 2017
 Antisense Therapeutics Ltd ASX:ANP January 2017 Forward Looking Statements This presentation contains forward-looking statements regarding the Company s business and the therapeutic and commercial potential
Antisense Therapeutics Ltd ASX:ANP January 2017 Forward Looking Statements This presentation contains forward-looking statements regarding the Company s business and the therapeutic and commercial potential
leading the way in research & development
 leading the way in research & development people. passion. possibilities. ABBVIE 2 immunology AbbVie Immunology has a demonstrated record of success in identifying and developing both small molecule and
leading the way in research & development people. passion. possibilities. ABBVIE 2 immunology AbbVie Immunology has a demonstrated record of success in identifying and developing both small molecule and
Moving Forward: Advantages and Challenges of Closed-Loop Control for Cell-Based Therapies
 Moving Forward: Advantages and Challenges of Closed-Loop Control for Cell-Based Therapies Guy-Bart Stan Control Engineering Synthetic Biology Group Centre for Synthetic Biology and Innovation Department
Moving Forward: Advantages and Challenges of Closed-Loop Control for Cell-Based Therapies Guy-Bart Stan Control Engineering Synthetic Biology Group Centre for Synthetic Biology and Innovation Department
Holland Innovation Network Team Boston: Walter de Wit Julia Knoeff
 Holland Innovation Network Team Boston: Walter de Wit Julia Knoeff Boston and Cambridge in Massachusetts are innovation-focused hubs with world-leading universities such as Harvard, Boston University and
Holland Innovation Network Team Boston: Walter de Wit Julia Knoeff Boston and Cambridge in Massachusetts are innovation-focused hubs with world-leading universities such as Harvard, Boston University and
TRENDS AND PRACTICE TIPS IN THERAPEUTIC ANTIBODY PATENTING
 TRENDS AND PRACTICE TIPS IN THERAPEUTIC ANTIBODY PATENTING 16 BY PEI WU AND JOHN P. IWANICKI Antibody technologies have evolved sideby-side with the advancement of molecular cloning, DNA sequencing, phage
TRENDS AND PRACTICE TIPS IN THERAPEUTIC ANTIBODY PATENTING 16 BY PEI WU AND JOHN P. IWANICKI Antibody technologies have evolved sideby-side with the advancement of molecular cloning, DNA sequencing, phage
Corporate Medical Policy Genetic Testing for Fanconi Anemia
 Corporate Medical Policy Genetic Testing for Fanconi Anemia File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_testing_for_fanconi_anemia 03/2015 3/2017 3/2018 12/2017 Description
Corporate Medical Policy Genetic Testing for Fanconi Anemia File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_testing_for_fanconi_anemia 03/2015 3/2017 3/2018 12/2017 Description
SORRENTO THERAPEUTICS, INC. (Exact name of registrant as specified in its charter)
 UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, DC 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 Date of Report (Date of earliest event
UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, DC 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 Date of Report (Date of earliest event
Industry-Academic Corporation Kyungpook National University
 Industry-Academic Corporation Title (Name of Technology) Composition for Glucose Sensing Comprising of Nanofibrous Membrane and Method for Manufacturing Non-enzymatic Glucose BlOsensor Using the Same 1
Industry-Academic Corporation Title (Name of Technology) Composition for Glucose Sensing Comprising of Nanofibrous Membrane and Method for Manufacturing Non-enzymatic Glucose BlOsensor Using the Same 1
Applied Protein Services
 Applied Protein Services Applied Protein Services A Window into the Future Development risk and attrition rates remain two of the greatest challenges to a successful biopharmaceutical pipeline. To help
Applied Protein Services Applied Protein Services A Window into the Future Development risk and attrition rates remain two of the greatest challenges to a successful biopharmaceutical pipeline. To help
Myeloma Bone Disease Current and Future Treatment. A Publication of The Bone and Cancer Foundation
 Myeloma Bone Disease Current and Future Treatment A Publication of The Bone and Cancer Foundation What Is Multiple Myeloma? Multiple Myeloma is a cancer that results from a malignant change in a particular
Myeloma Bone Disease Current and Future Treatment A Publication of The Bone and Cancer Foundation What Is Multiple Myeloma? Multiple Myeloma is a cancer that results from a malignant change in a particular
Singapore nanotechnology combats fatal brain infections
 P ublic release date: 28-Jun-2009 Contact: Cathy Yarbrough sciencematter@yahoo.com 858-243-1814 Agency for Science, Technology and Research (A*STAR), Singapore Singapore nanotechnology combats fatal brain
P ublic release date: 28-Jun-2009 Contact: Cathy Yarbrough sciencematter@yahoo.com 858-243-1814 Agency for Science, Technology and Research (A*STAR), Singapore Singapore nanotechnology combats fatal brain
Agios Pharmaceuticals, Inc. (Exact Name of Registrant as Specified in Charter)
 UNITED STATES SECURITIES AND EXCHANGE COMMISSION WASHINGTON, DC 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 Date of report (Date of earliest event
UNITED STATES SECURITIES AND EXCHANGE COMMISSION WASHINGTON, DC 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 Date of report (Date of earliest event
Functional Assessment and Clinical Outcome
 Tissue Engineering Functional Assessment and Clinical Outcome STEVEN A. GOLDSTEIN Orthopaedic Research Laboratories, Department of Orthopaedic Surgery, University of Michigan, Ann Arbor, Michigan 48109,
Tissue Engineering Functional Assessment and Clinical Outcome STEVEN A. GOLDSTEIN Orthopaedic Research Laboratories, Department of Orthopaedic Surgery, University of Michigan, Ann Arbor, Michigan 48109,
Regeneration of spinal cord injury (SCI) : What we know so far. By: Kendra Michaud
 Regeneration of spinal cord injury (SCI) : What we know so far By: Kendra Michaud INTRODUCTION BACKGROUND/IMPACT Neerumalla, Dr. Ravali. Spinal Cord Injury - Causes, Symptoms, Diagnosis, Treatment & Prevention.
Regeneration of spinal cord injury (SCI) : What we know so far By: Kendra Michaud INTRODUCTION BACKGROUND/IMPACT Neerumalla, Dr. Ravali. Spinal Cord Injury - Causes, Symptoms, Diagnosis, Treatment & Prevention.
Stem Cells and Regenerative Medicine
 Q 0 Stem Cells and Regenerative Medicine Technologies and capabilities to add value to your company Differentiation HA-PPS for cartilage Parallel culture optimisation Stem Cell Therapies Fetal EPCs and
Q 0 Stem Cells and Regenerative Medicine Technologies and capabilities to add value to your company Differentiation HA-PPS for cartilage Parallel culture optimisation Stem Cell Therapies Fetal EPCs and
SORRENTO THERAPEUTICS, INC. (Exact Name of Registrant as Specified in its Charter)
 UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM 8-K CURRENT REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 Date of Report (Date of earliest event
UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM 8-K CURRENT REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 Date of Report (Date of earliest event
Technology Development Funding Program Round 3
 Technology Development Funding Program Round 3 CWRU January 26, 2015 1 Program Overview NIH Center for Accelerated Innovations (NCAI) at Cleveland Clinic Mission to improve translation of basic science
Technology Development Funding Program Round 3 CWRU January 26, 2015 1 Program Overview NIH Center for Accelerated Innovations (NCAI) at Cleveland Clinic Mission to improve translation of basic science
Important Updates in USP <797> and USP <823> Eric S. Kastango, MBA, BS Pharm, FASHP Clinical IQ, LLC & CriticalPoint, LLC Madison, New Jersey
 Important Updates in USP and USP Eric S. Kastango, MBA, BS Pharm, FASHP Clinical IQ, LLC & CriticalPoint, LLC Madison, New Jersey Disclosures Principal-Clinical IQ, LLC Healthcare consulting
Important Updates in USP and USP Eric S. Kastango, MBA, BS Pharm, FASHP Clinical IQ, LLC & CriticalPoint, LLC Madison, New Jersey Disclosures Principal-Clinical IQ, LLC Healthcare consulting
LUPAS Luminescent Polymers for in vivo Imaging of Amyloid Signatures
 LUPAS Luminescent Polymers for in vivo Imaging of Amyloid Signatures A research project for innovative diagnostics for neurodegenerative disorders Funded by the European Union under the 7 th Framework
LUPAS Luminescent Polymers for in vivo Imaging of Amyloid Signatures A research project for innovative diagnostics for neurodegenerative disorders Funded by the European Union under the 7 th Framework
Production Assistance for Cellular Therapies PACT
 Production Assistance for Cellular Therapies PACT Traci Heath Mondoro, PhD NHLBI Bethesda, Maryland Renewed PACT Program Renewed January 15, 2010 Scope and size expanded Cell Processing Facilities (CPFs)
Production Assistance for Cellular Therapies PACT Traci Heath Mondoro, PhD NHLBI Bethesda, Maryland Renewed PACT Program Renewed January 15, 2010 Scope and size expanded Cell Processing Facilities (CPFs)
NCI Research Networks Model for Collaboration with Bioelectronics Round Table
 NCI Research Networks Model for Collaboration with Bioelectronics Round Table Larry Clarke, Cancer Imaging Program DCDT, NCI Detail: NIBIB Guest Scientist: NIST 1 Bioelectronics Round Table Research Triangle
NCI Research Networks Model for Collaboration with Bioelectronics Round Table Larry Clarke, Cancer Imaging Program DCDT, NCI Detail: NIBIB Guest Scientist: NIST 1 Bioelectronics Round Table Research Triangle
Global Regulatory Perspective Workshop
 Global Regulatory Perspective Workshop ISCT meeting Singapore 25 May 2016 Dr Nicolas FERRY ANSM, France Avertissement Lien d intérêt : personnel salarié de l ANSM (opérateur de l Etat). La présente intervention
Global Regulatory Perspective Workshop ISCT meeting Singapore 25 May 2016 Dr Nicolas FERRY ANSM, France Avertissement Lien d intérêt : personnel salarié de l ANSM (opérateur de l Etat). La présente intervention
Immunotherapy in myeloma
 Immunotherapy in myeloma This Horizons Infosheet contains information on immunotherapy, a type of treatment being investigated in myeloma. The Horizons Infosheet series provides information relating to
Immunotherapy in myeloma This Horizons Infosheet contains information on immunotherapy, a type of treatment being investigated in myeloma. The Horizons Infosheet series provides information relating to
InVivo Therapeutics. Developing Innovative Products for Spinal Cord Injury
 1 Developing Innovative Products for Spinal Cord Injury 2 Forward-Looking Statements Before we begin, we would like to remind everyone that during our presentation, we will be making forward-looking statements
1 Developing Innovative Products for Spinal Cord Injury 2 Forward-Looking Statements Before we begin, we would like to remind everyone that during our presentation, we will be making forward-looking statements
Molecule-specific diagnosis of metabolic diseases
 Powered by Website address: https://www.gesundheitsindustriebw.de/en/article/news/molecule-specific-diagnosis-ofmetabolic-diseases/ Molecule-specific diagnosis of metabolic diseases Lysosomal storage diseases
Powered by Website address: https://www.gesundheitsindustriebw.de/en/article/news/molecule-specific-diagnosis-ofmetabolic-diseases/ Molecule-specific diagnosis of metabolic diseases Lysosomal storage diseases
Texas Vendor Drug Program Fee-For-Service Medicaid Synagis Authorization Request
 Form 1033 September 2017-E Texas Vendor Drug Program Fee-For-Service Medicaid Synagis Authorization Request About Human Respiratory Syncytial Virus (RSV) causes respiratory tract infections and serious
Form 1033 September 2017-E Texas Vendor Drug Program Fee-For-Service Medicaid Synagis Authorization Request About Human Respiratory Syncytial Virus (RSV) causes respiratory tract infections and serious
Flow CAST : Testing Potency and Efficacy of Inhibitors of PI3K δ, PI3Kγ, BTK and SYK Activity
 Flow CAST : Testing Potency and Efficacy of Inhibitors of PI3K δ, PI3Kγ, BTK and SYK Activity Michele Romano, PhD Product Manager Flow CAST is for Research Use Only. Not for use in diagnostic procedures.
Flow CAST : Testing Potency and Efficacy of Inhibitors of PI3K δ, PI3Kγ, BTK and SYK Activity Michele Romano, PhD Product Manager Flow CAST is for Research Use Only. Not for use in diagnostic procedures.
A French Biotechnology company dedicated to the development and production of human endocrine cell lines. Innovation In Healthcare April 2012
 A French Biotechnology company dedicated to the development and production of human endocrine cell lines Innovation In Healthcare 16-17 April 2012 1 French Biotechnology company Founded in S2 2004 in Paris
A French Biotechnology company dedicated to the development and production of human endocrine cell lines Innovation In Healthcare 16-17 April 2012 1 French Biotechnology company Founded in S2 2004 in Paris
Reduce costs of the early drug development process Increase probality of developing drugs that enter late clinical phases
 Aktivitetsplan 5 - Application for Strategic collaboration on joint development and implementation of service offerings between Bioneer A/S and Fraunhofer Institute for Biomedical Engineering (Særskilt
Aktivitetsplan 5 - Application for Strategic collaboration on joint development and implementation of service offerings between Bioneer A/S and Fraunhofer Institute for Biomedical Engineering (Særskilt
Global Leader in Viral Vector Technologies
 Global Leader in Viral Vector Technologies Gene Therapy Vaccines - Discovery 1 SIRION BIOTECH ANY GENE TO ANY CELL Expert for viral vectors Competitive proprietary viral vector platforms High-end enabling
Global Leader in Viral Vector Technologies Gene Therapy Vaccines - Discovery 1 SIRION BIOTECH ANY GENE TO ANY CELL Expert for viral vectors Competitive proprietary viral vector platforms High-end enabling
MEDICAL PHYSICS (MED PHYS)
 Medical Physics (MED PHYS) 1 MEDICAL PHYSICS (MED PHYS) MED PHYS/PHYSICS 265 INTRODUCTION TO MEDICAL PHYSICS Primarily for premeds and other students in the medical and biological sciences. Applications
Medical Physics (MED PHYS) 1 MEDICAL PHYSICS (MED PHYS) MED PHYS/PHYSICS 265 INTRODUCTION TO MEDICAL PHYSICS Primarily for premeds and other students in the medical and biological sciences. Applications
Primiano Pio Di Mauro, Biomedical Engineer, PhD in Bioengineering
 Primiano Pio Di Mauro, Biomedical Engineer, PhD in Bioengineering pri.dimauro@gmail.com Mobile: 0034 689326511 Carrer de la Diputació 478 1º2ª, 08013, Barcelona, Spain https://www.linkedin.com/in/pridimauro/
Primiano Pio Di Mauro, Biomedical Engineer, PhD in Bioengineering pri.dimauro@gmail.com Mobile: 0034 689326511 Carrer de la Diputació 478 1º2ª, 08013, Barcelona, Spain https://www.linkedin.com/in/pridimauro/
Lessons learned: NCI s FLT F-18 IND and F-18 NaF NDA. Paula M. Jacobs, Ph.D. Deputy Associate Director, DCTD. NCI Cancer Imaging Program
 Lessons learned: NCI s FLT F-18 IND and F-18 NaF NDA Paula M. Jacobs, Ph.D. Deputy Associate Director, DCTD. NCI Cancer Imaging Program April 13, 2010 Disclaimers Opinions are mine alone NCI, NIH, and
Lessons learned: NCI s FLT F-18 IND and F-18 NaF NDA Paula M. Jacobs, Ph.D. Deputy Associate Director, DCTD. NCI Cancer Imaging Program April 13, 2010 Disclaimers Opinions are mine alone NCI, NIH, and
[Billing code: S] FEDERAL TRADE COMMISSION. Agency Information Collection Activities; Submission for OMB Review; Comment Request
![[Billing code: S] FEDERAL TRADE COMMISSION. Agency Information Collection Activities; Submission for OMB Review; Comment Request [Billing code: S] FEDERAL TRADE COMMISSION. Agency Information Collection Activities; Submission for OMB Review; Comment Request](/thumbs/76/73663408.jpg) This document is scheduled to be published in the Federal Register on 09/09/2016 and available online at http://federalregister.gov/a/2016-21675, and on FDsys.gov [Billing code: 6750-01-S] FEDERAL TRADE
This document is scheduled to be published in the Federal Register on 09/09/2016 and available online at http://federalregister.gov/a/2016-21675, and on FDsys.gov [Billing code: 6750-01-S] FEDERAL TRADE
The 3Rs: are Human Stem Cells and Organs on Chip alternatives?
 The 3Rs: are Human Stem Cells and Organs on Chip alternatives? Towards precision medicine in future healthcare Christine Mummery Leiden University Medical Centre University Twente hdmt Three kinds of human
The 3Rs: are Human Stem Cells and Organs on Chip alternatives? Towards precision medicine in future healthcare Christine Mummery Leiden University Medical Centre University Twente hdmt Three kinds of human
Ready or Not: The New Medical Device Regulations Are Here!
 Ready or Not: The New Medical Device Regulations Are Here! Felicia R Cochran, PhD, CMPP TM feliciacochran@earthlink.net FR Cochran 1 General Disclaimers This presentation represents the knowledge, professional
Ready or Not: The New Medical Device Regulations Are Here! Felicia R Cochran, PhD, CMPP TM feliciacochran@earthlink.net FR Cochran 1 General Disclaimers This presentation represents the knowledge, professional
Roche, Roche Molecular Diagnostics and more
 , Molecular and more [Monte Wetzel, PhD] Patients have questions. We provide answers. Group Clear focus on Healthcare Innovation with two strong pillars Pharma Pharma Genentech Chugai Molecular Professional
, Molecular and more [Monte Wetzel, PhD] Patients have questions. We provide answers. Group Clear focus on Healthcare Innovation with two strong pillars Pharma Pharma Genentech Chugai Molecular Professional
Clinical Research at MSU
 Financial Administration Development Program: Clinical Research at MSU Kristen Burt, Office of Regulatory Affairs (ORA) Sharon Schooley, Clinical & Translational Sciences Institute (CTSI) February 24,
Financial Administration Development Program: Clinical Research at MSU Kristen Burt, Office of Regulatory Affairs (ORA) Sharon Schooley, Clinical & Translational Sciences Institute (CTSI) February 24,
FDA Oversight of Gene Therapy
 FDA Oversight of Gene Therapy Celia M. Witten, Ph.D., M.D. Deputy Director, FDA CBER Rare Disease and Orphan Products Breakthrough Summit NORD October 17, 2016 Hyatt Regency Crystal City Crystal City,
FDA Oversight of Gene Therapy Celia M. Witten, Ph.D., M.D. Deputy Director, FDA CBER Rare Disease and Orphan Products Breakthrough Summit NORD October 17, 2016 Hyatt Regency Crystal City Crystal City,
Voyager Therapeutics - A Spinout from UMass Gene Therapy and RNAi Technologies
 University of Massachusetts Medical School escholarship@umms UMass Center for Clinical and Translational Science Research Retreat 2016 UMass Center for Clinical and Translational Science Research Retreat
University of Massachusetts Medical School escholarship@umms UMass Center for Clinical and Translational Science Research Retreat 2016 UMass Center for Clinical and Translational Science Research Retreat
21 CFR Ch. I ( Edition) Personnel Design controls Identification Traceability.
 Pt. 820 device together with an explanation of the basis for the estimate; (iv) Information describing the applicant s clinical experience with the device since the HDE was initially approved. This information
Pt. 820 device together with an explanation of the basis for the estimate; (iv) Information describing the applicant s clinical experience with the device since the HDE was initially approved. This information
MDR ID: Definition: Applicable:
 MDR Classification: (Reference Medical Device Regulation EU 2017/745, Annex VIII) Product: Product Name 1. DURATION OF USE MDR ID: Definition: Applicable: - Invasive Device: Continue Go to 2. Invasive
MDR Classification: (Reference Medical Device Regulation EU 2017/745, Annex VIII) Product: Product Name 1. DURATION OF USE MDR ID: Definition: Applicable: - Invasive Device: Continue Go to 2. Invasive
Exam MOL3007 Functional Genomics
 Faculty of Medicine Department of Cancer Research and Molecular Medicine Exam MOL3007 Functional Genomics Tuesday May 29 th 9.00-13.00 ECTS credits: 7.5 Number of pages (included front-page): 5 Supporting
Faculty of Medicine Department of Cancer Research and Molecular Medicine Exam MOL3007 Functional Genomics Tuesday May 29 th 9.00-13.00 ECTS credits: 7.5 Number of pages (included front-page): 5 Supporting
Insights into the Rare Disease Drug Approval Landscape: Trends and Current Development
 Insights into the Rare Disease Drug Approval Landscape: Trends and Current Development Mike Lanthier Operations Research Analyst FDA Office of the Commissioner NORD Rare Diseases & Orphan Products Breakthrough
Insights into the Rare Disease Drug Approval Landscape: Trends and Current Development Mike Lanthier Operations Research Analyst FDA Office of the Commissioner NORD Rare Diseases & Orphan Products Breakthrough
ADDIS ABABA UNIVERSITY CENTER OF BIOMEDICAL ENGINEERING
 ADDIS ABABA UNIVERSITY CENTER OF BIOMEDICAL ENGINEERING November 2013 History of Biomedical Engineering Definition of Biomedical Engineering Achievements of Biomedical Engineering Streams in Biomedical
ADDIS ABABA UNIVERSITY CENTER OF BIOMEDICAL ENGINEERING November 2013 History of Biomedical Engineering Definition of Biomedical Engineering Achievements of Biomedical Engineering Streams in Biomedical
International Transfers of Personal Data at sanofi-aventis R & D
 International Transfers of Personal Data at sanofi-aventis R & D Pierre-Yves Lastic, PhD Senior Director, Standards Management & Data Privacy Sanofi-aventis R&D CONFERENCE ON INTERNATIONAL TRANSFERS OF
International Transfers of Personal Data at sanofi-aventis R & D Pierre-Yves Lastic, PhD Senior Director, Standards Management & Data Privacy Sanofi-aventis R&D CONFERENCE ON INTERNATIONAL TRANSFERS OF
Antibody-based HLA (Human Leucocyte Antigen ) tissue-typing technologies
 Human AB Serum Product Description Human AB Serum is a vital cell culture reagent for some human cell types providing growth factors, vitamins, nutrients as well as trace elements and transport factors,
Human AB Serum Product Description Human AB Serum is a vital cell culture reagent for some human cell types providing growth factors, vitamins, nutrients as well as trace elements and transport factors,
Lecture 23: Clinical and Biomedical Applications of Proteomics; Proteomics Industry
 Lecture 23: Clinical and Biomedical Applications of Proteomics; Proteomics Industry Clinical proteomics is the application of proteomic approach to the field of medicine. Proteome of an organism changes
Lecture 23: Clinical and Biomedical Applications of Proteomics; Proteomics Industry Clinical proteomics is the application of proteomic approach to the field of medicine. Proteome of an organism changes
Is FMT A Drug? Lance Shea, M.S., J.D. Washington Square, Suite Connecticut Ave., NW Washington, DC, D
 Is FMT A Drug? Lance Shea, M.S., J.D. Washington Square, Suite 1100 1050 Connecticut Ave., NW Washington, DC, 20036 D 202-861-1648 LShea@bakerlaw.com FMT Scenarios Intra-Office Bank Product The Issue The
Is FMT A Drug? Lance Shea, M.S., J.D. Washington Square, Suite 1100 1050 Connecticut Ave., NW Washington, DC, 20036 D 202-861-1648 LShea@bakerlaw.com FMT Scenarios Intra-Office Bank Product The Issue The
Challenges associated with supply to larger patients populations: Elements of the CTL019 experience
 Cell and Gene Therapy Challenges associated with supply to larger patients populations: Elements of the CTL019 experience Daniel Stark Head External Supply Cell- and Gene Therapies Development and Manufacturing
Cell and Gene Therapy Challenges associated with supply to larger patients populations: Elements of the CTL019 experience Daniel Stark Head External Supply Cell- and Gene Therapies Development and Manufacturing
Medical Devices; Immunology and Microbiology Devices; Classification of the Automated
 This document is scheduled to be published in the Federal Register on 11/14/2017 and available online at https://federalregister.gov/d/2017-24585, and on FDsys.gov 4164-01-P DEPARTMENT OF HEALTH AND HUMAN
This document is scheduled to be published in the Federal Register on 11/14/2017 and available online at https://federalregister.gov/d/2017-24585, and on FDsys.gov 4164-01-P DEPARTMENT OF HEALTH AND HUMAN
Simple, intuitive and accessible MRI solution for preclinical research. M-Series Compact MRI Systems
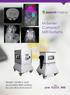 Simple, intuitive and accessible MRI solution for preclinical research M-Series Compact MRI Systems Application Oriented Imaging Anatomy and Morphology In vivo soft tissue imaging for morphological characterization.
Simple, intuitive and accessible MRI solution for preclinical research M-Series Compact MRI Systems Application Oriented Imaging Anatomy and Morphology In vivo soft tissue imaging for morphological characterization.
a) JOURNAL OF BIOLOGICAL CHEMISTRY b) PNAS c) NATURE
 a) JOURNAL OF BIOLOGICAL CHEMISTRY b) c) d) ........................ JOURNAL OF BIOLOGICAL CHEMISTRY MOLECULAR PHARMACOLOGY TRENDS IN PHARMACOLOGICAL S AMERICAN JOURNAL OF PHYSIOLOGY-HEART AND CIRCULATORY
a) JOURNAL OF BIOLOGICAL CHEMISTRY b) c) d) ........................ JOURNAL OF BIOLOGICAL CHEMISTRY MOLECULAR PHARMACOLOGY TRENDS IN PHARMACOLOGICAL S AMERICAN JOURNAL OF PHYSIOLOGY-HEART AND CIRCULATORY
nanoprecipitation mpeg-pla 2 nd Emulsion mpeg-pla in DCM
 THERAPEUTIC NANOTECHNOLOGY LAB MODULE Location: BioNano Lab, 3119 Micro and Nanotechnology Laboratory (MNTL) Instructor: Jianjun Cheng, Assistant Professor of Materials Science and Engineering Lab Assistants:
THERAPEUTIC NANOTECHNOLOGY LAB MODULE Location: BioNano Lab, 3119 Micro and Nanotechnology Laboratory (MNTL) Instructor: Jianjun Cheng, Assistant Professor of Materials Science and Engineering Lab Assistants:
LTX 109. BIO International Philadelphia 2015
 LTX 109 BIO International Philadelphia 2015 Lytix Biopharma company background Established in 2003 to develop and commercialize medical applications of lytic peptides originating from the University of
LTX 109 BIO International Philadelphia 2015 Lytix Biopharma company background Established in 2003 to develop and commercialize medical applications of lytic peptides originating from the University of
Pharmacy Compounding of Human Drug Products Under Section 503A of the Federal Food, Drug, and Cosmetic Act
 Pharmacy Compounding of Human Drug Products Under Section 503A of the Federal Food, Drug, and Cosmetic Act Guidance U.S. Department of Health and Human Services Food and Drug Administration Center for
Pharmacy Compounding of Human Drug Products Under Section 503A of the Federal Food, Drug, and Cosmetic Act Guidance U.S. Department of Health and Human Services Food and Drug Administration Center for
Authorizations of Emergency Use of In Vitro Diagnostic Devices for Detection of Zika Virus;
 This document is scheduled to be published in the Federal Register on 06/17/2016 and available online at http://federalregister.gov/a/2016-14380, and on FDsys.gov 4164-01-P DEPARTMENT OF HEALTH AND HUMAN
This document is scheduled to be published in the Federal Register on 06/17/2016 and available online at http://federalregister.gov/a/2016-14380, and on FDsys.gov 4164-01-P DEPARTMENT OF HEALTH AND HUMAN
Insight to Gene Techno Science Co.,Ltd
 Unlimited drug discovery from the beginning Ticker symbol: 4584 Insight to Gene Techno Science Co.,Ltd November 2017 2 Corporate Overview Chief Executive Founded March 2001 Masaharu Tani, President Listed
Unlimited drug discovery from the beginning Ticker symbol: 4584 Insight to Gene Techno Science Co.,Ltd November 2017 2 Corporate Overview Chief Executive Founded March 2001 Masaharu Tani, President Listed
Stem Cells. Part 1: What is a Stem Cell? STO Stem cells are unspecialized. What does this mean?
 STO-120 Stem Cells Part 1: What is a Stem Cell? Stem cells differ from other kinds of cells in the body. When a stem cell divides by mitosis, each new cell has the potential to either remain a stem cell
STO-120 Stem Cells Part 1: What is a Stem Cell? Stem cells differ from other kinds of cells in the body. When a stem cell divides by mitosis, each new cell has the potential to either remain a stem cell
