A quantitative protocol for intensity-based live cell FRET imaging.
|
|
|
- Tyrone Mason
- 6 years ago
- Views:
Transcription
1 A quantitative protocol for intensity-based live cell FRET imaging. Kaminski CF, Rees EJ, Schierle GS. Methods Mol Biol. 2014; 1076: Department of Chemical Engineering and Biotechnology, Pembroke Street University of Cambridge. Key Words Fluorescence, FRET, Förster Resonance Energy Transfer, live cell imaging, microscopy, sensitised emission Summary Förster resonance energy transfer (FRET) has become one of the most ubiquitous and powerful methods to quantify protein interactions in molecular biology. FRET refers to the sensitization of an acceptor molecule through transfer of energy from a nearby donor and it can occur if the emission band of the donor exhibits spectral overlap with the absorption band of the acceptor molecule. Numerous methods exist to quantify FRET levels from interacting protein labels including fluorescence lifetime, acceptor photobleaching, and polarization resolved imaging (1--3). For live cell imaging, however, sensitized emission FRET (sefret) is the most powerful and robust method of FRET signal quantification (4). It is fast, can be applied using straight forward microscopy equipment and offers information not only on strength of interaction, but, uniquely, also on the relative changes between interacting and noninteracting moieties in the reaction, referred to as FRET stoichiometry. A rigorous and quantitative application of sefret is far from trivial, however, and requires appropriate calibration experiments and constructs, control over hardware settings and appropriate image processing steps. This protocol presents a rigorous method to perform quantitative sefret measurements in live cells, providing the maximum possible information content from the measurement. The theoretical development and validation of the method is described in detail in (5) where it is also demonstrated in the kinetic ( time lapse ) analysis of protein interactions governing mitosis. The present protocol gives a detailed recipe for application of sefret. It is written specifically for use with CFP (cyan fluorescence protein) as donor fluorophore and YFP (yellow fluorescent protein) as acceptor fluorophore, a popular choice for many experiments. The protocol is however valid for any other FRET fluorophore pair, and we indicate how to adapt the protocol in such situations. We also provide a software programme that automates the calibration tasks outlined in this protocol and which is available for free to download (6). 1. Introduction to sefret methodology The principal features of sefret measurements are shown in figure 1. Two fluorescence filter sets are required: one which is optimized to detect fluorescence from the donor ( donor emission channel, here CFP) and similarly one optimized for emission from the acceptor ( acceptor emission channel, here YFP). [Figure 1 near here] Excitation of the donor molecule, D, (solid blue arrow), leads to fluorescence emission into the donor emission channel (light blue). In the absence of acceptors and FRET, this is the only signal emitted. If FRET occurs (red arrow, labeled FRET) the acceptor molecule, A, becomes sensitized, and the sefret signal emitted from A is detected in the acceptor channel. This is indicated as pathway 3 (solid red arrow). The efficiency of energy transfer from D to A is denoted by E and the so called Förster radius is defined to be the distance at which E=50%. However, in practice the sefret signal is contaminated by signal cross talk. There are two contributions to cross talk in sefret based on the CFP/YFP - and most other 1
2 fluorophore - pairings. Cross excitation refers to the direct excitation of the acceptor at the donor excitation wavelength (path 1, dashed blue arrow). For example, YFP has a finite probability of being excited at the CFP excitation wavelength. Contamination also arises from donor bleed through (path 2, red dashed red arrow), e.g. CFP fluorescence will leak into the YFP fluorescence band. Signal cross talk is indistinguishable from the true sefret signals, and, if unaccounted for, can generate false positives. To correct for these effects we calculate a corrected sefret efficiency, which we refer to as cfret (5), to yield: cfret = I DA! AER " I AA! DER " I DD Here the first term on the right hand side denotes the uncorrected signal, the second term accounts for the acceptor cross excitation and the last term for the donor bleed through. Table 1 denotes the nomenclature in detail. [Table 1 near here] The AER and DER are dye specific parameters that are ideally constant for a given sample and can be determined from control samples containing, respectively, donor (CFP) only and acceptor only (YFP). A measureable value for cfret signifies that FRET takes place between donor and acceptor, however, its value also depends on the stoichiometries of free donors and acceptors and interacting FRET pairs. To obtain information on the relative number of free and interacting fluorophores we further normalize cfret either by the acceptor or the donor concentration. This leads to the finally sought quantities: and The nomenclatures for the terms in these equations are given in table 2. dfret is the true FRET efficiency E normalized by which is the fraction of the donor molecules that undergo FRET. Similarly afret is the acceptor normalized FRET efficiency. The two may be significantly different as Figure 2a and 2b illustrate. For the situation depicted in Fig 2a) we have afret>>dfret signifying that the number of donor molecules participating in FRET interactions is much smaller than the number of acceptor molecules. Similarly in b) the opposite is true. The evolution of afret and dfret and their interdependence thus offer important information in the stoichiometry of biological interactions, information that is usually hidden in other modalities of performing FRET (e.g. lifetime measurements). Examples of how this can be used to follow kinase activity in living cells are given in (5). In what follows we give a recipe on how to quantify afret and dfret in biological samples. Example MATLAB scripts are provided together with example data as a guide to performing the image processing required for sefret. [Figure 2 near here] [Table 2 near here] 2. Materials 2
3 Biological sample preparation In addition to the biological FRET sample of interest 3 control samples are required to determine,,, DER, and AER (See Note 1). 1. Donor only control sample (used as negative control and for determination of DER): Transfect cells (the protocol is verified for mammalian cells) using the CFP-tagged protein of interest (see Note 2). 2. Acceptor only control sample (used as negative control and for determination of AER): Transfect cells with YFP-tagged protein of interest in analogy to step Positive control (required for determination of, ): Transfect cells with CFP-YFP linker construct exhibiting a known amount of FRET. The protocol as follows is valid for a CFP-YFP tandem construct obtained by excising the EYFP gene (Enhanced Yellow Fluorescent Protein) from the peyfp-n1 vector, and ligating it into the Multiple Cloning Site (MCS) of the pecfp-c1 vector (Enhanced Cyan Fluorescent Protein; both originally obtained from Clontech, Palo Alto, CA, USA) such that the two fluorophores in the construct are separated by a sequence of 18 amino acids (GLRSRAQASNSAVEGSAM). The plasmid vector maps are available from and respectively (see Note 3). Propagate the plasmid in E.coli (recommended host strains are DH5-alpha or HB101) and use kanamycin (30 µg/ml) to select positive strains. Isolate DNA and transfect cells according to standard protocols. If required, stable transformants can be selected by G418 (neomycin). See also Note Samples for FRET analysis: Transfect cells with pecfp-fusion- and peyfpfusion proteins. Aim to obtain similar expression levels for both proteins. Microscope set-up The protocol described here permits system independent determination of FRET efficiency and system specific attributes (filter transmission, detector gains etc.) are taken into account through control experiments. However, for optimal sensitivity such filter sets and excitation wavelengths should be used as to maximize FRET signals whilst minimizing donor bleed through and acceptor direct excitation. For CFP / YFP based FRET the following settings are representative examples for either widefield or confocal imaging set-ups. Wide field microscope settings: Sensitive CCD cameras with good dynamic range (12 bit or greater) should be chosen as detectors, and a linear response ensured. Note: camera / detector settings must remain constant for control and sample measurements. If acceptor and donor channels are measured sequentially, ensure that no sample movement occurs between images (see Note 5). [Table 3 near here] Confocal microscope settings. Choose PMT voltage settings to ensure a linear signal response. Set laser excitation powers to obtain similar signal levels for cfp and yfp. Check for photobleaching and adjust settings to minimize this effect. Minimise sample movement artefacts (e.g. use line averaging rather than frame averaging if available and minimize time delays between consecutive measurements of donor and acceptor channels). 3
4 [Table 4 near here] 3. Methods 3.1 Measurement of DER Use the donor only sample. Keep magnification and gain settings constant throughout and subtract any d.c. background signal from all obtained images before further processing (see Note 6). Set up the microscope in sequential acquisition mode (if available) and obtain the following set of images, from which the DER can be calculated: 1. Set excitation light source to donor excitation setting and the emission channel to the donor emission bandpass to obtain signal I DD (e.g nm excitation / nm emission filter for widefield system). 2. Set excitation light source to donor excitation setting and the emission channel to acceptor emission bandpass to obtain signal I DA (e.g nm 3. Calculate the DER for every image pixel according to: 4. Repeat this calibration measurement on at least five different areas of the donor-only sample. 3.2 Measurement of AER Use the acceptor only sample. Keep magnification and gain settings same as above and constant throughout and subtract d.c. background signal from all obtained images before further processing (see Note 6). Set up the microscope in sequential acquisition mode (if available) and obtain the following set of images, from which the AER can be calculated. : 1. Set excitation light source to donor excitation setting and the emission channel to acceptor emission bandpass to obtain signal I DA (e.g nm excitation / nm emission filter for widefield system). 2. Set excitation light source to acceptor excitation setting and the emission channel to acceptor emission bandpass to obtain signal I AA (e.g nm 3. Calculate the AER for every image pixel according to: 4 4. Repeat this calibration measurement on at least five different areas of the acceptor-only sample. 3.3 System calibration with tandem FRET construct Use the sample containing the cfp-yfp linker construct. Keep magnifaction and gain settings same as above and constant throughout and subtract any d.c. background signal from all obtained images before further processing (see Note 6). Set up the microscope in sequential acquisition mode (if available) and obtain the following set of images, from which the cross talk corrected FRET signal, cfret, can be calculated. : 1. Set excitation light source to donor excitation setting and the emission channel to the donor emission bandpass to obtain signal I DD. (e.g nm excitation / nm emission filter for widefield system).
5 2. Set excitation light source to donor excitation setting and the emission channel to acceptor emission bandpass to obtain signal I DA (e.g nm 3. Set excitation light source to acceptor excitation setting and the emission channel to acceptor emission bandpass to obtain signal I AA (e.g nm 4. Calculate the cfret for every image pixel according to: cfret = I DA! AER " I AA! DER " I DD 5. Select regions (pixels) in your image where FRET occurs and determine through calculation of (See Note 7, 8): 6. Select regions (pixels) in your image where FRET occurs and determine through calculation of (See Note 7): 7. Repeat measurements on five areas of linker-only sample. 3.4 Measurement of dfret and afret Use the biological sample with cfp and yfp fusion proteins of interest. Keep magnifaction and gain settings same as above and constant throughout and subtract any d.c. background signal from all obtained images before further processing (see Note 6). Set up the microscope in sequential acquisition mode (if available) and obtain the following set of images, from which dfret and afret can be determined: 1. Set excitation light source to donor excitation setting and the emission channel to the donor emission bandpass to obtain signal I DD. (e.g nm excitation / nm emission filter for widefield system). 2. Set excitation light source to donor excitation setting and the emission channel to acceptor emission bandpass to obtain signal I DA (e.g nm 3. Set excitation light source to acceptor excitation setting and the emission channel to acceptor emission bandpass to obtain signal I AA (e.g nm 4. Calculate the cfret for every image pixel according to: cfret = I DA! AER " I AA! DER " I DD 5. Calculate dfret for every image pixel according to: See Notes 9 and Calculate afret for every image pixel according to: See Notes 9 and 10. 5
6 4. Notes: 1) The protocol here is specifically written for use with cfp and yfp fusion proteins and for the linker construct presented in paragraph 3 of the Materials section. It has furthermore been validated for use in live HeLa cells. The protocol is however valid for other FRET fluorophore pairs, cell types, and linker constructs. In this case appropriate filter sets and excitation lines have to be chosen, and, crucially, the FRET efficiency of the positive FRET tandem control construct needs to be determined independently, as described in Note 7. 2) To ensure strong FRET signal levels it is advisable to devise constructs labeled near the binding sites of the proteins. E.g. if the interaction is likely to occur in the C- terminal region of the protein of interest, it is advisable to construct a C-terminal fusion protein, e.g. via use of the pecfp-n1 vector (see Vice versa if the interaction is more likely to happen in the N-terminal region, use the pecfp-c1 plasmid, instead. Follow similar procedure for the yfp-fusion proteins. 3) The plasmid for the positive control construct is available from the authors. In live HeLa cells this construct was determined to yield a FRET efficiency of E=38 percent. Other linker constructs may also be used but require an independent determination of FRET efficiency (see (5) for details). 4) Optionally, a further negative control sample containing empty pecfp and peyfp plasmids can be used. These are not strictly necessary but provide a useful means to validate the calibration procedure described in the Methods section 3.1, 3.2, and 3.3 and in Note 9. 5) Use other filter sets / laser settings as appropriate if FRET pairs other than cfp/yfp are used in the experiments. 6) For background subtraction identify dark regions in measured images. The average brightness of these regions allows for a d.c. background subtraction from the images. Incorrect background subtraction would yield erroneous FRET results. Perform background subtraction for all measurements in this protocol. The MATLAB script provided in (6) as an example of sefret image processing already includes a background correction which is suitable for the sample data. 7) For the 18 aa cfp-yfp linker construct expressed in live HeLa cells (see Materials section) substitute E=0.38 in the equations for and. If fixed cells are used or a different calibration construct is used (e.g. different FRET fluorophores or different linker system) then E needs to be determined independently for this construct and the appropriate value for E substituted. E can be determined from a measurement of the donor fluorescence lifetime as: where is the donor fluorescence lifetime in the presence of FRET and is the donor lifetime in the absence of FRET (i.e. the natural lifetime of the donor). For details refer to (5). 8) Measure in at least five different image areas to determine and, to check for consistency of and enable averaging. 9) A control experiment to check that the calibration has been successful and the correct data for AER and DER have been obtained is to measure a negative control sample, which contains non interacting pecfp and peyfp plasmids (or corresponding plasmids if other fluorophores are used in the experiment). For this sample bot afret and dfret should be 0 if correct values for DER, AER and α and β were used. 10) The consistency of obtained datasets can be verified through a repeat determination of the AER from a corresponding measurement of the acceptor only sample at the end of the experiment. If this value differs from that determined at the start of the experiment this is indicative of changes in the experimental conditions (e.g. laser power fluctuations, microscope settings, etc.) which may invalidate results. 6
7 5. References: 1. Lakowicz J.R. (2006) Principles of Fluorescence Spectroscopy. ISBN Jares-Erijman E.A., Jovin T.M. (2006) Imaging molecular interactions in living cells by FRET microscopy. Current opinion in chemical biology 10(5), van Munster EB, Gadella TWJ (2005) Microscopy Techniques. doi: /b Chakrabortee S, Meersman F, Kaminski Schierle GS, et al. (2010) Catalytic and chaperone-like functions in an intrinsically disordered protein associated with desiccation tolerance. Proceedings of the National Academy of Sciences of the United States of America 107(37), Elder A. D, Domin A., Kaminski Schierle GS, et al. (2009) A quantitative protocol for dynamic measurements of protein interactions by Förster resonance energy transfer-sensitized fluorescence emission. Journal of The Royal Society Interface 6, S59 S Figure Captions Figure 1. Principle of sefret Figure 2. Illustration to exemplify the difference between donor-normalised and acceptor-normalised FRET values. In a) there is an abundance of non interacting donors, whereas all acceptors are interacting. Here dfret is less than afret. In b) there is an overabundance of acceptor and the opposite holds true. A simultaneous determination of afret and dfret thus establishes both whether energy transfer is taking place and also whether there is an overabundance of non interacting donors / acceptors. Table Captions Table 1. Quantities required to correct for signal cross talk in sefret Table 2. Nomenclature of sefret calibration constants Table 3. Widefield microscope filter setup Table 4. Confocal microscope filter setup Tables 7
8 I DA AER DER I AA I DD Uncorrected FRET signal: Signal in Acceptor channel upon donor excitation Acceptor excitation ratio: corrects for direct excitation of acceptor (path 1 in figure 1) Donor emission ratio: Determines bleed through of donor fluorescence into the acceptor channel (path 2 in figure 1) Signal obtained in acceptor channel, upon excitation at acceptor excitation wavelength Signal obtained in donor channel, upon excitation at the donor excitation wavelength Table 1 Table 2 Donor normalized FRET efficiency (i.e. fraction of interacting donor χ D times FRET efficiency E) Acceptor normalized FRET efficiency (i.e. fraction of interacting acceptor χ A times FRET efficiency E). Relates to relative quantum yields and signal detection efficiencies between donor and acceptors. Relates to lineshapes and excitation efficiencies of donor and acceptor molecules Excitation wavelength Donor excitation filter Acceptor excitation filter Donor emission filter Acceptor emission filter Table nm, nm nm nm Donor excitation wavelength Acceptor excitation wavelength Donor emission filter Acceptor emission filter Table nm 514 nm nm nm 8
9 Figure 1 Figure 2 9
Welcome! openmicberkeley.wordpress.com. Open Berkeley
 Welcome! openmicberkeley.wordpress.com Agenda Jen Lee: Introduction to FRET Marla Feller: Using FRET sensors to look at time resolved measurements Becky Lamason: Using FRET to determine if a bacterial
Welcome! openmicberkeley.wordpress.com Agenda Jen Lee: Introduction to FRET Marla Feller: Using FRET sensors to look at time resolved measurements Becky Lamason: Using FRET to determine if a bacterial
TECHNICAL BULLETIN. In Vitro Bacterial Split Fluorescent Protein Fold n Glow Solubility Assay Kits
 In Vitro Bacterial Split Fluorescent Protein Fold n Glow Solubility Assay Kits Catalog Numbers APPA001 In Vitro Bacterial Split GFP "Fold 'n' Glow" Solubility Assay Kit (Green) APPA008 In Vitro Bacterial
In Vitro Bacterial Split Fluorescent Protein Fold n Glow Solubility Assay Kits Catalog Numbers APPA001 In Vitro Bacterial Split GFP "Fold 'n' Glow" Solubility Assay Kit (Green) APPA008 In Vitro Bacterial
Challenges to measuring intracellular Ca 2+ Calmodulin: nature s Ca 2+ sensor
 Calcium Signals in Biological Systems Lecture 3 (2/9/0) Measuring intracellular Ca 2+ signals II: Genetically encoded Ca 2+ sensors Henry M. Colecraft, Ph.D. Challenges to measuring intracellular Ca 2+
Calcium Signals in Biological Systems Lecture 3 (2/9/0) Measuring intracellular Ca 2+ signals II: Genetically encoded Ca 2+ sensors Henry M. Colecraft, Ph.D. Challenges to measuring intracellular Ca 2+
Monitoring and Optimizing the Lipopolysaccharides-plasmid DNA interaction by FLIM-FRET
 Transactions on Science and Technology Vol. 4, No. 3-3, 342-347, 2017 Monitoring and Optimizing the Lipopolysaccharides-plasmid DNA interaction by FLIM-FRET Nur Syahadatain Abdul Razak 1#, Clarence M.
Transactions on Science and Technology Vol. 4, No. 3-3, 342-347, 2017 Monitoring and Optimizing the Lipopolysaccharides-plasmid DNA interaction by FLIM-FRET Nur Syahadatain Abdul Razak 1#, Clarence M.
Simultaneous multi-color, multiphoton fluorophore excitation using dual-color fiber lasers
 Multiphoton Microscopy / Fiber Laser Simultaneous multi-color, multiphoton fluorophore excitation using dual-color fiber lasers Matthias Handloser, Tim Paasch-Colberg, Bernhard Wolfring TOPTICA Photonics
Multiphoton Microscopy / Fiber Laser Simultaneous multi-color, multiphoton fluorophore excitation using dual-color fiber lasers Matthias Handloser, Tim Paasch-Colberg, Bernhard Wolfring TOPTICA Photonics
Partha Roy
 Fluorescence microscopy http://micro.magnet.fsu.edu/primer/index.html Partha Roy 1 Lecture Outline Definition of fluorescence Common fluorescent reagents Construction ti of a fluorescence microscope Optical
Fluorescence microscopy http://micro.magnet.fsu.edu/primer/index.html Partha Roy 1 Lecture Outline Definition of fluorescence Common fluorescent reagents Construction ti of a fluorescence microscope Optical
Introduction to Fluorescence Jablonski Diagram
 ntroduction to Fluorescence Jablonski Diagram Excited Singlet Manifold S1 internal conversion S2 k -isc k isc Excited riplet Manifold 1 S0 k nr k k' f nr fluorescence k p phosphorescence Singlet round
ntroduction to Fluorescence Jablonski Diagram Excited Singlet Manifold S1 internal conversion S2 k -isc k isc Excited riplet Manifold 1 S0 k nr k k' f nr fluorescence k p phosphorescence Singlet round
Time-resolved Measurements Using the Agilent Cary Eclipse Fluorescence Spectrophotometer A Versatile Instrument for Accurate Measurements
 Time-resolved Measurements Using the Agilent Cary Eclipse Fluorescence Spectrophotometer A Versatile Instrument for Accurate Measurements Technical Overview Authors Dr. Fabian Zieschang, Katherine MacNamara,
Time-resolved Measurements Using the Agilent Cary Eclipse Fluorescence Spectrophotometer A Versatile Instrument for Accurate Measurements Technical Overview Authors Dr. Fabian Zieschang, Katherine MacNamara,
FLUORESCENT PEPTIDES. Outstanding Performance and Wide Application Range
 FLUORESCENT PEPTIDES Peptides and amino acids labeled with and Tide Quencher TM We offer peptides and amino acids tagged with fluorescent dyes. They meet highest demands in fluorescence intensity and photo-stability,
FLUORESCENT PEPTIDES Peptides and amino acids labeled with and Tide Quencher TM We offer peptides and amino acids tagged with fluorescent dyes. They meet highest demands in fluorescence intensity and photo-stability,
Spectral Separation of Multifluorescence Labels with the LSM 510 META
 Microscopy from Carl Zeiss Spectral Separation of Multifluorescence Labels with the LSM 510 META Indians living in the South American rain forest can distinguish between almost 200 hues of green in their
Microscopy from Carl Zeiss Spectral Separation of Multifluorescence Labels with the LSM 510 META Indians living in the South American rain forest can distinguish between almost 200 hues of green in their
Introduction to N-STORM
 Introduction to N-STORM Dan Metcalf Advanced Imaging Manager Outline Introduction Principles of STORM Applications N-STORM overview Biological Scale Mitochondrion Microtubule Amino Acid 1Å Kinesin 1nm
Introduction to N-STORM Dan Metcalf Advanced Imaging Manager Outline Introduction Principles of STORM Applications N-STORM overview Biological Scale Mitochondrion Microtubule Amino Acid 1Å Kinesin 1nm
Quantum Dot applications in Fluorescence Imaging for Calibration and Molecular Imaging
 Quantum Dot applications in Fluorescence Imaging for Calibration and Molecular Imaging Introduction In this application note, we will discuss the application of quantum dots in fluorescence imaging, both
Quantum Dot applications in Fluorescence Imaging for Calibration and Molecular Imaging Introduction In this application note, we will discuss the application of quantum dots in fluorescence imaging, both
Masayoshi Honda, Jeehae Park, Robert A. Pugh, Taekjip Ha, and Maria Spies
 Molecular Cell, Volume 35 Supplemental Data Single-Molecule Analysis Reveals Differential Effect of ssdna-binding Proteins on DNA Translocation by XPD Helicase Masayoshi Honda, Jeehae Park, Robert A. Pugh,
Molecular Cell, Volume 35 Supplemental Data Single-Molecule Analysis Reveals Differential Effect of ssdna-binding Proteins on DNA Translocation by XPD Helicase Masayoshi Honda, Jeehae Park, Robert A. Pugh,
Confocal Microscopes. Evolution of Imaging
 Confocal Microscopes and Evolution of Imaging Judi Reilly Hans Richter Massachusetts Institute of Technology Environment, Health & Safety Office Radiation Protection What is Confocal? Pinhole diaphragm
Confocal Microscopes and Evolution of Imaging Judi Reilly Hans Richter Massachusetts Institute of Technology Environment, Health & Safety Office Radiation Protection What is Confocal? Pinhole diaphragm
Quantitative comparison of different fluorescent protein couples for fast FRET-FLIM acquisition
 Biophysical Journal, Volume 97 Supporting Material Quantitative comparison of different fluorescent protein couples for fast FRET-FLIM acquisition Sergi Padilla, Nicolas Audugé, Hervé Lalucque, Jean Claude
Biophysical Journal, Volume 97 Supporting Material Quantitative comparison of different fluorescent protein couples for fast FRET-FLIM acquisition Sergi Padilla, Nicolas Audugé, Hervé Lalucque, Jean Claude
Fluorescence Imaging with One Nanometer Accuracy Lab
 I. Introduction. Fluorescence Imaging with One Nanometer Accuracy Lab Traditional light microscope is limited by the diffraction limit of light, typically around 250 nm. However, many biological processes
I. Introduction. Fluorescence Imaging with One Nanometer Accuracy Lab Traditional light microscope is limited by the diffraction limit of light, typically around 250 nm. However, many biological processes
ADVANCED PRACTICAL COURSE IN BIOPHYSICS: FRET
 : FRET 1 INTRODUCTION Fluorescence spectroscopy and fluorescence microscopy are essential tools in biology. Biological molecules can be labeled with fluorescent molecules and thus, their localization and
: FRET 1 INTRODUCTION Fluorescence spectroscopy and fluorescence microscopy are essential tools in biology. Biological molecules can be labeled with fluorescent molecules and thus, their localization and
CONFOCAL APPLICATION LETTER. June 2009 No. 35. FRET with FLIM Quantitative In-vivo Biochemistry
 CONFOCAL APPLICATION LETTER June 2009 No. 35 resolution FRET with FLIM Quantitative In-vivo Biochemistry Fig. 1 Emission spectrum of donor (here ECFP, blue line) must overlap with excitation spectrum of
CONFOCAL APPLICATION LETTER June 2009 No. 35 resolution FRET with FLIM Quantitative In-vivo Biochemistry Fig. 1 Emission spectrum of donor (here ECFP, blue line) must overlap with excitation spectrum of
Practical light microscopy: an introduction
 Practical light microscopy: an introduction Dr. Mark Leake, Oxford University www.physics.ox.ac.uk/users/leake Aim of today s talk: Explanation of the very (very) basics of how a light microscope works
Practical light microscopy: an introduction Dr. Mark Leake, Oxford University www.physics.ox.ac.uk/users/leake Aim of today s talk: Explanation of the very (very) basics of how a light microscope works
Comparison of LANCE Ultra TR-FRET to PerkinElmer s Classical LANCE TR-FRET Platform for Kinase Applications
 Comparison of LANCE Ultra TR-FRET to PerkinElmer s Classical LANCE TR-FRET Platform for Kinase Applications LANCE ULTRA TR-FRET TECHNOLOGY A P P L I C A T I O N N O T E Introduction Protein kinases play
Comparison of LANCE Ultra TR-FRET to PerkinElmer s Classical LANCE TR-FRET Platform for Kinase Applications LANCE ULTRA TR-FRET TECHNOLOGY A P P L I C A T I O N N O T E Introduction Protein kinases play
Flow Cytometry - The Essentials
 Flow Cytometry - The Essentials Pocket Guide to Flow Cytometry: 1. Know your Cytometer 2. Understanding Fluorescence and Fluorophores 3. Gating Process 4. Controls 5. Optimization 6. Panel Building 7.
Flow Cytometry - The Essentials Pocket Guide to Flow Cytometry: 1. Know your Cytometer 2. Understanding Fluorescence and Fluorophores 3. Gating Process 4. Controls 5. Optimization 6. Panel Building 7.
Green Fluorescent Protein. Avinash Bayya Varun Maturi Nikhileswar Reddy Mukkamala Aravindh Subhramani
 Green Fluorescent Protein Avinash Bayya Varun Maturi Nikhileswar Reddy Mukkamala Aravindh Subhramani Introduction The Green fluorescent protein (GFP) was first isolated from the Jellyfish Aequorea victoria,
Green Fluorescent Protein Avinash Bayya Varun Maturi Nikhileswar Reddy Mukkamala Aravindh Subhramani Introduction The Green fluorescent protein (GFP) was first isolated from the Jellyfish Aequorea victoria,
Chapter 4 Fluorescence Resonance Energy Transfer (FRET) by Minor Groove-Associated Cyanine-Polyamide Conjugates
 Chapter 4 Fluorescence Resonance Energy Transfer (FRET) by Minor Groove-Associated Cyanine-Polyamide Conjugates The work described in this chapter was accomplished in collaboration with V. Rucker (Dervan
Chapter 4 Fluorescence Resonance Energy Transfer (FRET) by Minor Groove-Associated Cyanine-Polyamide Conjugates The work described in this chapter was accomplished in collaboration with V. Rucker (Dervan
Application of Quantum Mechanics to Biology
 Application of Quantum Mechanics to Biology How can we apply quantum mechanics to biology? Polymers of nucleotides and amino acids - millions of atoms bounded into a large molecule Visual System Must turn
Application of Quantum Mechanics to Biology How can we apply quantum mechanics to biology? Polymers of nucleotides and amino acids - millions of atoms bounded into a large molecule Visual System Must turn
Innovations To Meet Your Needs
 Innovations To Meet Your Needs Cooled CCD Camera 1340 x 1037 pixel resolution for greatest image quality 12-bit precision provides 3 orders of linear dynamic range Windows and Power Macintosh Software
Innovations To Meet Your Needs Cooled CCD Camera 1340 x 1037 pixel resolution for greatest image quality 12-bit precision provides 3 orders of linear dynamic range Windows and Power Macintosh Software
Cationic Vector Intercalation into the Lipid Membrane Enables Intact Polyplex DNA Escape from Endosomes for Gene Delivery
 Cationic Vector Intercalation into the Lipid Membrane Enables Intact Polyplex DNA Escape from Endosomes for Gene Delivery Sriram Vaidyanathan, 1 Junjie Chen, 2 Bradford G. Orr, 3 Mark M. Banaszak Holl
Cationic Vector Intercalation into the Lipid Membrane Enables Intact Polyplex DNA Escape from Endosomes for Gene Delivery Sriram Vaidyanathan, 1 Junjie Chen, 2 Bradford G. Orr, 3 Mark M. Banaszak Holl
HiPer Real-Time PCR Teaching Kit
 HiPer Real-Time PCR Teaching Kit Product Code: HTBM032 Number of experiments that can be performed: 10 Duration of Experiment Protocol: 1.5 hours Storage Instructions: The kit is stable for 12 months from
HiPer Real-Time PCR Teaching Kit Product Code: HTBM032 Number of experiments that can be performed: 10 Duration of Experiment Protocol: 1.5 hours Storage Instructions: The kit is stable for 12 months from
Technical Review. Real time PCR
 Technical Review Real time PCR Normal PCR: Analyze with agarose gel Normal PCR vs Real time PCR Real-time PCR, also known as quantitative PCR (qpcr) or kinetic PCR Key feature: Used to amplify and simultaneously
Technical Review Real time PCR Normal PCR: Analyze with agarose gel Normal PCR vs Real time PCR Real-time PCR, also known as quantitative PCR (qpcr) or kinetic PCR Key feature: Used to amplify and simultaneously
Comparing the Agilent 2100 Bioanalyzer Performance to Traditional DNA Analysis Techniques
 Comparing the Agilent 2100 Bioanalyzer Performance to Traditional DNA Analysis Techniques Application Note Author Deborah Vitale Agilent Technologies, Inc. Palo Alto, CA, USA Abstract This Application
Comparing the Agilent 2100 Bioanalyzer Performance to Traditional DNA Analysis Techniques Application Note Author Deborah Vitale Agilent Technologies, Inc. Palo Alto, CA, USA Abstract This Application
DeltaPro. DeltaFlex FORENSICS PARTICLE CHARACTERIZATION ELEMENTAL ANALYSIS FLUORESCENCE GRATINGS & OEM SPECTROMETERS OPTICAL COMPONENTS RAMAN
 DeltaFlex DeltaPro ELEMENTAL ANALYSIS FLUORESCENCE GRATINGS & OEM SPECTROMETERS OPTICAL COMPONENTS FORENSICS PARTICLE CHARACTERIZATION RAMAN SPECTROSCOPIC ELLIPSOMETRY SPR IMAGING Why measure fluorescence
DeltaFlex DeltaPro ELEMENTAL ANALYSIS FLUORESCENCE GRATINGS & OEM SPECTROMETERS OPTICAL COMPONENTS FORENSICS PARTICLE CHARACTERIZATION RAMAN SPECTROSCOPIC ELLIPSOMETRY SPR IMAGING Why measure fluorescence
Transport of Potato Lipoxygenase into the Vacuole Larsen, Mia Kruse Guldstrand; Welinder, Karen Gjesing; Jørgensen, Malene
 Aalborg Universitet Transport of Potato Lipoxygenase into the Vacuole Larsen, Mia Kruse Guldstrand; Welinder, Karen Gjesing; Jørgensen, Malene Publication date: 2009 Document Version Publisher's PDF, also
Aalborg Universitet Transport of Potato Lipoxygenase into the Vacuole Larsen, Mia Kruse Guldstrand; Welinder, Karen Gjesing; Jørgensen, Malene Publication date: 2009 Document Version Publisher's PDF, also
Supplemental Information. A Versatile Tool for Live-Cell Imaging. and Super-Resolution Nanoscopy Studies. of HIV-1 Env Distribution and Mobility
 Cell Chemical Biology, Volume 24 Supplemental Information A Versatile Tool for Live-Cell Imaging and Super-Resolution Nanoscopy Studies of HIV-1 Env Distribution and Mobility Volkan Sakin, Janina Hanne,
Cell Chemical Biology, Volume 24 Supplemental Information A Versatile Tool for Live-Cell Imaging and Super-Resolution Nanoscopy Studies of HIV-1 Env Distribution and Mobility Volkan Sakin, Janina Hanne,
Resolution of Microscopes Visible light is nm Dry lens(0.5na), green(530nm light)=0.65µm=650nm for oil lens (1.4NA) UV light (300nm) = 0.13µm f
 Microscopes and Microscopy MCB 380 Good information sources: Alberts-Molecular Biology of the Cell http://micro.magnet.fsu.edu/primer/ http://www.microscopyu.com/ Approaches to Problems in Cell Biology
Microscopes and Microscopy MCB 380 Good information sources: Alberts-Molecular Biology of the Cell http://micro.magnet.fsu.edu/primer/ http://www.microscopyu.com/ Approaches to Problems in Cell Biology
Lecture Four. Molecular Approaches I: Nucleic Acids
 Lecture Four. Molecular Approaches I: Nucleic Acids I. Recombinant DNA and Gene Cloning Recombinant DNA is DNA that has been created artificially. DNA from two or more sources is incorporated into a single
Lecture Four. Molecular Approaches I: Nucleic Acids I. Recombinant DNA and Gene Cloning Recombinant DNA is DNA that has been created artificially. DNA from two or more sources is incorporated into a single
FLUORESCENCE MICROSCOPY TECHNIQUES PRACTICAL MANUAL FOR
 FLUORESCENCE PRACTICAL MANUAL FOR MICROSCOPY TECHNIQUES Sohail Ahmed Sudhaharan Thankiah Radek Machán Martin Hof Andrew H. A. Clayton Graham Wright Jean-Baptiste Sibarita Thomas Korte Andreas Herrmann
FLUORESCENCE PRACTICAL MANUAL FOR MICROSCOPY TECHNIQUES Sohail Ahmed Sudhaharan Thankiah Radek Machán Martin Hof Andrew H. A. Clayton Graham Wright Jean-Baptiste Sibarita Thomas Korte Andreas Herrmann
QImaging Camera Application Notes Multicolor Immunofluorescence Imaging
 QImaging Camera Application Notes Multicolor Immunofluorescence Imaging In order to image localization of intracellular proteins with high specificity, it is frequently necessary to multiplex antibody
QImaging Camera Application Notes Multicolor Immunofluorescence Imaging In order to image localization of intracellular proteins with high specificity, it is frequently necessary to multiplex antibody
Azure Biosystems Western Blotting Workflow
 Azure Biosystems Western Blotting Workflow PROBE PLAN SEPARATE ANALYZE VISUALIZE PLAN Plan your experiment and choose your detection method Chemiluminescent Western Blotting The most common method for
Azure Biosystems Western Blotting Workflow PROBE PLAN SEPARATE ANALYZE VISUALIZE PLAN Plan your experiment and choose your detection method Chemiluminescent Western Blotting The most common method for
Performance of the Micro Photon Devices PDM 50CT SPAD detector with PicoQuant TCSPC systems
 Technical Note Performance of the Micro Photon Devices PDM 5CT SPAD detector with PicoQuant TCSPC systems Rolf Krahl, Andreas Bülter, Felix Koberling, PicoQuant GmbH These measurements were performed to
Technical Note Performance of the Micro Photon Devices PDM 5CT SPAD detector with PicoQuant TCSPC systems Rolf Krahl, Andreas Bülter, Felix Koberling, PicoQuant GmbH These measurements were performed to
Supplementary information to accompany: A novel role for the DNA repair gene Rad51 in Netrin-1 signalling
 Supplementary information to accompany: A novel role for the DNA repair gene Rad51 in Netrin-1 signalling Glendining KA 1, Markie D 2, Gardner RJM 4, Franz EA 3, Robertson SP 4, Jasoni CL 1 Supplementary
Supplementary information to accompany: A novel role for the DNA repair gene Rad51 in Netrin-1 signalling Glendining KA 1, Markie D 2, Gardner RJM 4, Franz EA 3, Robertson SP 4, Jasoni CL 1 Supplementary
Determining fluorescence Limit of Detection with Nanoparticle Tracking Analysis (NTA)
 Determining fluorescence Limit of Detection with Nanoparticle Tracking Analysis (NTA) FLUORESCENCE DETECTION PARTICLE SIZE PARTICLE CONCENTRATION Introduction The ability to detect nanoparticle fluorescence
Determining fluorescence Limit of Detection with Nanoparticle Tracking Analysis (NTA) FLUORESCENCE DETECTION PARTICLE SIZE PARTICLE CONCENTRATION Introduction The ability to detect nanoparticle fluorescence
Genome Sequence Assembly
 Genome Sequence Assembly Learning Goals: Introduce the field of bioinformatics Familiarize the student with performing sequence alignments Understand the assembly process in genome sequencing Introduction:
Genome Sequence Assembly Learning Goals: Introduce the field of bioinformatics Familiarize the student with performing sequence alignments Understand the assembly process in genome sequencing Introduction:
Detection of Homotypic Protein Interactions with Green Fluorescent Protein Proximity Imaging (GFP-PRIM)
 11 Detection of Homotypic Protein Interactions with Green Fluorescent Protein Proximity Imaging (GFP-PRIM) Dino A. De Angelis Cellular Biochemistry and Biophysics Program, Memorial Sloan-Kettering Cancer
11 Detection of Homotypic Protein Interactions with Green Fluorescent Protein Proximity Imaging (GFP-PRIM) Dino A. De Angelis Cellular Biochemistry and Biophysics Program, Memorial Sloan-Kettering Cancer
Exploring Similarities of Conserved Domains/Motifs
 Exploring Similarities of Conserved Domains/Motifs Sotiria Palioura Abstract Traditionally, proteins are represented as amino acid sequences. There are, though, other (potentially more exciting) representations;
Exploring Similarities of Conserved Domains/Motifs Sotiria Palioura Abstract Traditionally, proteins are represented as amino acid sequences. There are, though, other (potentially more exciting) representations;
Photon Upconversion Sensitized Nanoprobes for
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supporting Information Photon Upconversion Sensitized Nanoprobes for Sensing and Imaging of ph
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supporting Information Photon Upconversion Sensitized Nanoprobes for Sensing and Imaging of ph
Applicability of Hyperspectral Fluorescence Imaging to Mineral Sorting
 Institute of Industrial Information Technology Applicability of Hyperspectral Fluorescence Imaging to Mineral Sorting Optical Characterization of Materials, March 19, 2015 Sebastian Bauer, M.Sc. (Head:
Institute of Industrial Information Technology Applicability of Hyperspectral Fluorescence Imaging to Mineral Sorting Optical Characterization of Materials, March 19, 2015 Sebastian Bauer, M.Sc. (Head:
CytoPainter Golgi Staining Kit Green Fluorescence
 ab139483 CytoPainter Golgi Staining Kit Green Fluorescence Instructions for Use Designed for the detection of Golgi bodies by microscopy This product is for research use only and is not intended for diagnostic
ab139483 CytoPainter Golgi Staining Kit Green Fluorescence Instructions for Use Designed for the detection of Golgi bodies by microscopy This product is for research use only and is not intended for diagnostic
Bottom-up systems biology using optical proteomics
 Bottom-up systems biology using optical proteomics Systems biology option, Biochemistry Dr. Mark Leake, Oxford University www.physics.ox.ac.uk/users/leake Some previous single-molecule techniques Leake
Bottom-up systems biology using optical proteomics Systems biology option, Biochemistry Dr. Mark Leake, Oxford University www.physics.ox.ac.uk/users/leake Some previous single-molecule techniques Leake
Transmission Mode Photocathodes Covering the Spectral Range
 Transmission Mode Photocathodes Covering the Spectral Range 6/19/2002 New Developments in Photodetection 3 rd Beaune Conference June 17-21, 2002 Arlynn Smith, Keith Passmore, Roger Sillmon, Rudy Benz ITT
Transmission Mode Photocathodes Covering the Spectral Range 6/19/2002 New Developments in Photodetection 3 rd Beaune Conference June 17-21, 2002 Arlynn Smith, Keith Passmore, Roger Sillmon, Rudy Benz ITT
UV Fluorescence Polarization as a Means to Investigate Protein Conformational and Mass Change
 A p p l i c a t i o n N o t e UV Fluorescence Polarization as a Means to Investigate Protein Conformational and Mass Change Using Intrinsic Tryptophan Fluorescence in Conjunction with UV-capable Polarizers
A p p l i c a t i o n N o t e UV Fluorescence Polarization as a Means to Investigate Protein Conformational and Mass Change Using Intrinsic Tryptophan Fluorescence in Conjunction with UV-capable Polarizers
Before You Begin. Calibration Protocols
 Before You Begin Read through this entire protocol sheet carefully before you start your experiment and prepare any materials you may need. Calibration Protocols 1. OD 600 Reference point You will use
Before You Begin Read through this entire protocol sheet carefully before you start your experiment and prepare any materials you may need. Calibration Protocols 1. OD 600 Reference point You will use
How to perform-control immunostaining experiment - microscopist subjective point of view. Pawel Pasierbek
 How to perform-control immunostaining experiment - microscopist subjective point of view. Pawel Pasierbek Immunolabeling and fluorescent detection became such a standard procedure in the biomedical research
How to perform-control immunostaining experiment - microscopist subjective point of view. Pawel Pasierbek Immunolabeling and fluorescent detection became such a standard procedure in the biomedical research
nanodsf 2bind: Your service provider for biophysical characterization of proteins Precisely revealing protein folding and stability
 nanodsf Precisely revealing protein folding and stability 2bind: Your service provider for biophysical characterization of proteins This booklet was written and designed by 2bind 08 2015 Any reproduction
nanodsf Precisely revealing protein folding and stability 2bind: Your service provider for biophysical characterization of proteins This booklet was written and designed by 2bind 08 2015 Any reproduction
Roche Molecular Biochemicals Technical Note No. LC 10/2000
 Roche Molecular Biochemicals Technical Note No. LC 10/2000 LightCycler Overview of LightCycler Quantification Methods 1. General Introduction Introduction Content Definitions This Technical Note will introduce
Roche Molecular Biochemicals Technical Note No. LC 10/2000 LightCycler Overview of LightCycler Quantification Methods 1. General Introduction Introduction Content Definitions This Technical Note will introduce
Bioinstrumentation Light Sources Lasers or LEDs?
 Bioinstrumentation Light Sources Lasers or LEDs? A comprehensive analysis of all the factors involved in designing and building life sciences instrumentation reveals that lasers provide superior performance
Bioinstrumentation Light Sources Lasers or LEDs? A comprehensive analysis of all the factors involved in designing and building life sciences instrumentation reveals that lasers provide superior performance
Fluorescence Nanoscopy
 Fluorescence Nanoscopy Keith A. Lidke University of New Mexico panda3.phys.unm.edu/~klidke/index.html Optical Microscopy http://en.wikipedia.org/wiki/k%c3%b6hler_illumination 30 µm Fluorescent Probes Michalet
Fluorescence Nanoscopy Keith A. Lidke University of New Mexico panda3.phys.unm.edu/~klidke/index.html Optical Microscopy http://en.wikipedia.org/wiki/k%c3%b6hler_illumination 30 µm Fluorescent Probes Michalet
GEL IMAGING. The detection and quantitation of specific subsets of posttranslationally
 High Sensitivity Detection and Quantitation of Phosphoproteins in 1D and 2D Gels using the ProXPRESS 2D Proteomic Imager and ProFINDER 2D Image Analysis Software GEL IMAGING A P P L I C A T I O N N O T
High Sensitivity Detection and Quantitation of Phosphoproteins in 1D and 2D Gels using the ProXPRESS 2D Proteomic Imager and ProFINDER 2D Image Analysis Software GEL IMAGING A P P L I C A T I O N N O T
RAMAN STRUCTURAL STUDY OF OLEFIN BLENDS
 RAMAN STRUCTURAL STUDY OF OLEFIN BLENDS Authors: Yu.V. Zavgorodnev, E.A. Sagitova, G.Yu. Nikolaeva, K.A. Prokhorov, P.P. Pashinin, L.A. Novokshonova, T.M. Ushakova, E.E. Starchak, E.M. Antipov DOI: 10.12684/alt.1.79
RAMAN STRUCTURAL STUDY OF OLEFIN BLENDS Authors: Yu.V. Zavgorodnev, E.A. Sagitova, G.Yu. Nikolaeva, K.A. Prokhorov, P.P. Pashinin, L.A. Novokshonova, T.M. Ushakova, E.E. Starchak, E.M. Antipov DOI: 10.12684/alt.1.79
CBI Toolbox Tour 2015
 CBI Toolbox Tour 2015 Thermophoresis (NanoTemper) NT.115 & NT.LabelFree Images: NanoTemper Circular Dichroism Jasco J-1500 Spectrometer Six Position Turreted Peltier Temperature Control System Automated
CBI Toolbox Tour 2015 Thermophoresis (NanoTemper) NT.115 & NT.LabelFree Images: NanoTemper Circular Dichroism Jasco J-1500 Spectrometer Six Position Turreted Peltier Temperature Control System Automated
Lecture 13: Analysis of 2D gels
 Lecture 13: Analysis of 2D gels A complete proteomic analysis aims at collecting quantitative information about all protein in a sample. A normal 2 Dimensional gel electrophoresis results are analyzed
Lecture 13: Analysis of 2D gels A complete proteomic analysis aims at collecting quantitative information about all protein in a sample. A normal 2 Dimensional gel electrophoresis results are analyzed
Multiplex PCR reaction
 Multiplexed Detection of Pathogens using Fluorescence Resonance Energy Transfer in a Spatial Detection Format Multiplex PCR reaction Bruce Applegate Collaborators: Sergei Savikhin Pina Fratamico Wilfredo
Multiplexed Detection of Pathogens using Fluorescence Resonance Energy Transfer in a Spatial Detection Format Multiplex PCR reaction Bruce Applegate Collaborators: Sergei Savikhin Pina Fratamico Wilfredo
Using ULS24 CMOS Bio-imager as a Readout Sensor for Chemiluminescence Immunoassay and DNA Hybridization Assay
 Using ULS24 CMOS Bio-imager as a Readout Sensor for Chemiluminescence Immunoassay and DNA Hybridization Assay Updated: Nov 11, 2016 Introduction Immunoassay is a widely used method for detecting the presence
Using ULS24 CMOS Bio-imager as a Readout Sensor for Chemiluminescence Immunoassay and DNA Hybridization Assay Updated: Nov 11, 2016 Introduction Immunoassay is a widely used method for detecting the presence
Methods of Culturing Microorganisms. Chapter 3. Five Basic Techniques of Culturing Bacteria. Topics
 Chapter 3 Topics Methods of Culturing Microorganisms Microscope (History, Types, Definitions) Staining (Gram s) Methods of Culturing Microorganisms Five basic techniques of culturing Media Microbial growth
Chapter 3 Topics Methods of Culturing Microorganisms Microscope (History, Types, Definitions) Staining (Gram s) Methods of Culturing Microorganisms Five basic techniques of culturing Media Microbial growth
CENTER FOR BRAIN EXPERIMENT
 CENTER FOR BRAIN EXPERIMENT Section of Brain Structure Associate Professor: ARII, Tatsuo, PhD 1967 Graduated from Tohoku University, Faculty of Science. Completed the doctoral course in Engineering, Nagoya
CENTER FOR BRAIN EXPERIMENT Section of Brain Structure Associate Professor: ARII, Tatsuo, PhD 1967 Graduated from Tohoku University, Faculty of Science. Completed the doctoral course in Engineering, Nagoya
Lecture 25 (11/15/17)
 Lecture 25 (11/15/17) Reading: Ch9; 328-332 Ch25; 990-995, 1005-1012 Problems: Ch9 (study-guide: applying); 1,2 Ch9 (study-guide: facts); 7,8 Ch25 (text); 1-3,5-7,9,10,13-15 Ch25 (study-guide: applying);
Lecture 25 (11/15/17) Reading: Ch9; 328-332 Ch25; 990-995, 1005-1012 Problems: Ch9 (study-guide: applying); 1,2 Ch9 (study-guide: facts); 7,8 Ch25 (text); 1-3,5-7,9,10,13-15 Ch25 (study-guide: applying);
Double-pulse fluorescence lifetime measurements
 Journal of Microscopy, Vol. 186, Pt 3, June 1997, pp. 212 220. Received 15 August 1996; accepted 14 January 1997 Double-pulse fluorescence lifetime measurements A. H. BUIST,* M. MÜLLER,* E. J. GIJSBERS,*
Journal of Microscopy, Vol. 186, Pt 3, June 1997, pp. 212 220. Received 15 August 1996; accepted 14 January 1997 Double-pulse fluorescence lifetime measurements A. H. BUIST,* M. MÜLLER,* E. J. GIJSBERS,*
Rapid Learning Center Presents. Teach Yourself AP Biology in 24 Hours
 Rapid Learning Center Chemistry :: Biology :: Physics :: Math Rapid Learning Center Presents Teach Yourself AP Biology in 24 Hours 1/35 *AP is a registered trademark of the College Board, which does not
Rapid Learning Center Chemistry :: Biology :: Physics :: Math Rapid Learning Center Presents Teach Yourself AP Biology in 24 Hours 1/35 *AP is a registered trademark of the College Board, which does not
Targeted modification of gene function exploiting homology directed repair of TALENmediated double strand breaks in barley
 Targeted modification of gene function exploiting homology directed repair of TALENmediated double strand breaks in barley Nagaveni Budhagatapalli a, Twan Rutten b, Maia Gurushidze a, Jochen Kumlehn a,
Targeted modification of gene function exploiting homology directed repair of TALENmediated double strand breaks in barley Nagaveni Budhagatapalli a, Twan Rutten b, Maia Gurushidze a, Jochen Kumlehn a,
Single-Molecule FISH. Single-Molecule Fluorescence Resonance Energy Transfer. Synonyms. Definition. Photophysical Basis of smfret.
 Transfer 2329 ingle-molecule FIH ingle-molecule RNA FIH ingle-molecule Fluorescence Resonance Energy Transfer Alexander E. Johnson-Buck 1, Mario R. Blanco 2 and Nils G. Walter 1 1 Department of Chemistry,
Transfer 2329 ingle-molecule FIH ingle-molecule RNA FIH ingle-molecule Fluorescence Resonance Energy Transfer Alexander E. Johnson-Buck 1, Mario R. Blanco 2 and Nils G. Walter 1 1 Department of Chemistry,
Certificate of Analysis
 Certificate of Analysis pet6xhn Expression Vector Set Contents Product Information... 1 pet6xhn-n, pet6xhn-c, and pet6xhn-gfpuv Vector Information... 2 Location of Features... 4 Additional Information...
Certificate of Analysis pet6xhn Expression Vector Set Contents Product Information... 1 pet6xhn-n, pet6xhn-c, and pet6xhn-gfpuv Vector Information... 2 Location of Features... 4 Additional Information...
Monitoring Protein:Protein Interactions in Living Cells Using NanoBRET Technology. Danette L. Daniels, Ph.D. Group Leader, Functional Proteomics
 Monitoring Protein:Protein Interactions in Living Cells Using NanoBRET Technology Danette L. Daniels, Ph.D. Group Leader, Functional Proteomics Proteomics and studying dynamic interactions AP-MS Proteomics
Monitoring Protein:Protein Interactions in Living Cells Using NanoBRET Technology Danette L. Daniels, Ph.D. Group Leader, Functional Proteomics Proteomics and studying dynamic interactions AP-MS Proteomics
Sanger Sequencing: Troubleshooting Guide
 If you need help analysing your Sanger sequencing output, this guide can help. CONTENTS 1 Introduction... 2 2 Sequence Data Evaluation... 2 3 Troubleshooting... 4 3.1 Reviewing the Sequence... 4 3.1.1
If you need help analysing your Sanger sequencing output, this guide can help. CONTENTS 1 Introduction... 2 2 Sequence Data Evaluation... 2 3 Troubleshooting... 4 3.1 Reviewing the Sequence... 4 3.1.1
PERKINELMER, INC. AlphaPlex Assay Development Guide
 PERKINELMER, INC. AlphaPlex Assay Development Guide For Laboratory Use Only Research Chemicals for Research Purposes Only Notes and Precautions In this guide, AlphaPlex Acceptor Beads refer to AlphaPlex
PERKINELMER, INC. AlphaPlex Assay Development Guide For Laboratory Use Only Research Chemicals for Research Purposes Only Notes and Precautions In this guide, AlphaPlex Acceptor Beads refer to AlphaPlex
Fluorescence Quenching of Human Serum Albumin by Caffeine
 CHEM 411L Instrumental Analysis Laboratory Revision 2.1 Fluorescence Quenching of Human Serum Albumin by Caffeine In this laboratory exercise we will examine the fluorescence of Human Serum Albumin (HSA)
CHEM 411L Instrumental Analysis Laboratory Revision 2.1 Fluorescence Quenching of Human Serum Albumin by Caffeine In this laboratory exercise we will examine the fluorescence of Human Serum Albumin (HSA)
Supporting Information for Matching nanoantenna field confinement to FRET distances enhances Förster energy transfer rates
 Supporting Information for Matching nanoantenna field confinement to FRET distances enhances Förster energy transfer rates Petru Ghenuche, Mathieu Mivelle, Juan de Torres, Satish Babu Moparthi, Hervé Rigneault,
Supporting Information for Matching nanoantenna field confinement to FRET distances enhances Förster energy transfer rates Petru Ghenuche, Mathieu Mivelle, Juan de Torres, Satish Babu Moparthi, Hervé Rigneault,
Exchange Kinetics and Activity analysis of GTP-Binding Proteins with Fluorescent GTP-Analogs
 Exchange Kinetics and Activity analysis of GTP-Binding Proteins with Fluorescent GTP-Analogs Chandandeep Kaur, Ahmed A. Heikal Department of Biomedical Engineering, University of California, Irvine CA92617;
Exchange Kinetics and Activity analysis of GTP-Binding Proteins with Fluorescent GTP-Analogs Chandandeep Kaur, Ahmed A. Heikal Department of Biomedical Engineering, University of California, Irvine CA92617;
Direct visualization, sizing and concentration measurement of fluorescently labeled nanoparticles using NTA
 Direct visualization, sizing and concentration measurement of fluorescently labeled nanoparticles using NTA NANOSIGHT RANGE Visualize and Measure Nanoparticle Size and Concentration PARTICLE SIZE PARTICLE
Direct visualization, sizing and concentration measurement of fluorescently labeled nanoparticles using NTA NANOSIGHT RANGE Visualize and Measure Nanoparticle Size and Concentration PARTICLE SIZE PARTICLE
An Introduction to Fluorescence Resonance Energy Transfer (FRET) Technology and its Application in Bioscience
 W h i t e P a p e r An Introduction to Fluorescence Resonance Energy Transfer (FRET) Technology and its Application in Bioscience Paul Held, Laboratory Manager, Applications Department, BioTek Instruments,
W h i t e P a p e r An Introduction to Fluorescence Resonance Energy Transfer (FRET) Technology and its Application in Bioscience Paul Held, Laboratory Manager, Applications Department, BioTek Instruments,
SUMOstar Gene Fusion Technology
 Gene Fusion Technology NEW METHODS FOR ENHANCING FUNCTIONAL PROTEIN EXPRESSION AND PURIFICATION IN INSECT CELLS White Paper June 2007 LifeSensors Inc. 271 Great Valley Parkway Malvern, PA 19355 www.lifesensors.com
Gene Fusion Technology NEW METHODS FOR ENHANCING FUNCTIONAL PROTEIN EXPRESSION AND PURIFICATION IN INSECT CELLS White Paper June 2007 LifeSensors Inc. 271 Great Valley Parkway Malvern, PA 19355 www.lifesensors.com
Protocols for cloning SEC-based repair templates using SapTrap assembly
 Protocols for cloning SEC-based repair templates using SapTrap assembly Written by Dan Dickinson (ddickins@live.unc.edu) and last updated July 2016. Overview SapTrap (Schwartz and Jorgensen, 2016) is a
Protocols for cloning SEC-based repair templates using SapTrap assembly Written by Dan Dickinson (ddickins@live.unc.edu) and last updated July 2016. Overview SapTrap (Schwartz and Jorgensen, 2016) is a
FRET imaging of multiple focal planes to analyze the organization and conformation of Transferrin-Receptor in polarized cells
 FRET imaging of multiple focal planes to analyze the organization and conformation of Transferrin-Receptor in polarized cells Horst Wallrabe a, Ammasi Periasamy b and Margarida Barroso c a Keck Center
FRET imaging of multiple focal planes to analyze the organization and conformation of Transferrin-Receptor in polarized cells Horst Wallrabe a, Ammasi Periasamy b and Margarida Barroso c a Keck Center
Chapter 1 Nanophotonics and Single Molecules
 Chapter 1 Nanophotonics and Single Molecules W.E. Moerner(*ü ), P. James Schuck, David P. Fromm, Anika Kinkhabwala, Samuel J. Lord, Stefanie Y. Nishimura, Katherine A. Willets, Arvind Sundaramurthy, Gordon
Chapter 1 Nanophotonics and Single Molecules W.E. Moerner(*ü ), P. James Schuck, David P. Fromm, Anika Kinkhabwala, Samuel J. Lord, Stefanie Y. Nishimura, Katherine A. Willets, Arvind Sundaramurthy, Gordon
PERFORMANCE MADE EASY REAL-TIME PCR
 PERFORMANCE MADE EASY REAL-TIME PCR The MyGo Pro real-time PCR instrument provides unmatched performance in a convenient format. Novel Full Spectrum Optics deliver 120 optical channels of fluorescence
PERFORMANCE MADE EASY REAL-TIME PCR The MyGo Pro real-time PCR instrument provides unmatched performance in a convenient format. Novel Full Spectrum Optics deliver 120 optical channels of fluorescence
STRUCTURAL BIOLOGY. α/β structures Closed barrels Open twisted sheets Horseshoe folds
 STRUCTURAL BIOLOGY α/β structures Closed barrels Open twisted sheets Horseshoe folds The α/β domains Most frequent domain structures are α/β domains: A central parallel or mixed β sheet Surrounded by α
STRUCTURAL BIOLOGY α/β structures Closed barrels Open twisted sheets Horseshoe folds The α/β domains Most frequent domain structures are α/β domains: A central parallel or mixed β sheet Surrounded by α
Gene Expression Technology
 Gene Expression Technology Bing Zhang Department of Biomedical Informatics Vanderbilt University bing.zhang@vanderbilt.edu Gene expression Gene expression is the process by which information from a gene
Gene Expression Technology Bing Zhang Department of Biomedical Informatics Vanderbilt University bing.zhang@vanderbilt.edu Gene expression Gene expression is the process by which information from a gene
Lecture 22: Single-molecule Dynamics: rotation, diffusion, energy transfer, and powerful application in biological systems
 Lecture 22: Single-molecule Dynamics: rotation, diffusion, energy transfer, and powerful application in biological systems Why and how to study molecule dynamics: rotation and diffusion. 2 research cases.
Lecture 22: Single-molecule Dynamics: rotation, diffusion, energy transfer, and powerful application in biological systems Why and how to study molecule dynamics: rotation and diffusion. 2 research cases.
Reading for lecture 11
 Reading for lecture 11 1. Optical Tweezers, Myosin 2. Atomic Force Microscopy (AFM) 3. Single-Molecule Fluorescence Microscopy 4. Patch-Clamp 5. Genetic Techniques Key references are included in italics
Reading for lecture 11 1. Optical Tweezers, Myosin 2. Atomic Force Microscopy (AFM) 3. Single-Molecule Fluorescence Microscopy 4. Patch-Clamp 5. Genetic Techniques Key references are included in italics
Chapter 10: Classification of Microorganisms
 Chapter 10: Classification of Microorganisms 1. The Taxonomic Hierarchy 2. Methods of Identification 1. The Taxonomic Hierarchy Phylogenetic Tree of the 3 Domains Taxonomic Hierarchy 8 successive taxa
Chapter 10: Classification of Microorganisms 1. The Taxonomic Hierarchy 2. Methods of Identification 1. The Taxonomic Hierarchy Phylogenetic Tree of the 3 Domains Taxonomic Hierarchy 8 successive taxa
Imagerie et spectroscopie de fluorescence par excitation non radiative
 Imagerie et spectroscopie de fluorescence par excitation non radiative comment s affranchir de la limite de diffraction Rodolphe Jaffiol, Cyrille Vézy, Marcelina Cardoso Dos Santos LNIO, UTT, Troyes NanoBioPhotonics
Imagerie et spectroscopie de fluorescence par excitation non radiative comment s affranchir de la limite de diffraction Rodolphe Jaffiol, Cyrille Vézy, Marcelina Cardoso Dos Santos LNIO, UTT, Troyes NanoBioPhotonics
A Survey of Laser Types. Gas Lasers
 Mihail Pivtoraiko Andrei Rozhkov Applied Optics Winter 2003 A Survey of Laser Types Laser technology is available to us since 1960 s, and since then has been quite well developed. Currently, there is a
Mihail Pivtoraiko Andrei Rozhkov Applied Optics Winter 2003 A Survey of Laser Types Laser technology is available to us since 1960 s, and since then has been quite well developed. Currently, there is a
A Comparison of AlphaLISA and TR-FRET Homogeneous Immunoassays in Serum-Containing Samples
 application Note A Comparison of and Homogeneous Immunoassays in Serum-Containing Samples Authors Anuradha Prasad, PhD, Catherine Lautenschlager, PhD, Stephen Hurt, PhD, David Titus, PhD and Stéphane Parent,
application Note A Comparison of and Homogeneous Immunoassays in Serum-Containing Samples Authors Anuradha Prasad, PhD, Catherine Lautenschlager, PhD, Stephen Hurt, PhD, David Titus, PhD and Stéphane Parent,
Imaging Nucleic Acid Gels on the Odyssey Fc Imager
 Imaging Nucleic Acid Gels on the Odyssey Fc Imager Developed for: Odyssey Fc Imaging System Published September 2011. The most recent version of this protocol is posted at: https://www.licor.com/bio/support/
Imaging Nucleic Acid Gels on the Odyssey Fc Imager Developed for: Odyssey Fc Imaging System Published September 2011. The most recent version of this protocol is posted at: https://www.licor.com/bio/support/
1. A brief overview of sequencing biochemistry
 Supplementary reading materials on Genome sequencing (optional) The materials are from Mark Blaxter s lecture notes on Sequencing strategies and Primary Analysis 1. A brief overview of sequencing biochemistry
Supplementary reading materials on Genome sequencing (optional) The materials are from Mark Blaxter s lecture notes on Sequencing strategies and Primary Analysis 1. A brief overview of sequencing biochemistry
Improving the Accuracy of Base Calls and Error Predictions for GS 20 DNA Sequence Data
 Improving the Accuracy of Base Calls and Error Predictions for GS 20 DNA Sequence Data Justin S. Hogg Department of Computational Biology University of Pittsburgh Pittsburgh, PA 15213 jsh32@pitt.edu Abstract
Improving the Accuracy of Base Calls and Error Predictions for GS 20 DNA Sequence Data Justin S. Hogg Department of Computational Biology University of Pittsburgh Pittsburgh, PA 15213 jsh32@pitt.edu Abstract
Click chemistry for advanced drug discovery applications of human protein kinase CK2
 Click chemistry for advanced drug discovery applications of human protein kinase CK2 Christian Nienberg 1, *, Anika Retterath 1, Kira Sophie Becher 2, Henning D. Mootz 2 and Joachim Jose 1 1 Institut für
Click chemistry for advanced drug discovery applications of human protein kinase CK2 Christian Nienberg 1, *, Anika Retterath 1, Kira Sophie Becher 2, Henning D. Mootz 2 and Joachim Jose 1 1 Institut für
Final exam. Please write your name on the exam and keep an ID card ready.
 Biophysics of Macromolecules Prof. R. Jungmann and Prof. J. Lipfert SS 2017 Final exam Final exam First name: Last name: Student number ( Matrikelnummer ): Please write your name on the exam and keep an
Biophysics of Macromolecules Prof. R. Jungmann and Prof. J. Lipfert SS 2017 Final exam Final exam First name: Last name: Student number ( Matrikelnummer ): Please write your name on the exam and keep an
Photoluminescence Spectroscopy on Chemically Synthesized Nanoparticles
 Photoluminescence Spectroscopy on Chemically Synthesized Nanoparticles Torben Menke 1 Institut für Angewandte Physik und Zentrum für Mikrostrukturforschung Universität Hamburg Diploma Talk, 18.12.27 1
Photoluminescence Spectroscopy on Chemically Synthesized Nanoparticles Torben Menke 1 Institut für Angewandte Physik und Zentrum für Mikrostrukturforschung Universität Hamburg Diploma Talk, 18.12.27 1
The Human Protein PRR14 Tethers Heterochromatin to the Nuclear Lamina During Interphase and Mitotic Exit
 Cell Reports, Volume 5 Supplemental Information The Human Protein PRR14 Tethers Heterochromatin to the Nuclear Lamina During Interphase and Mitotic Exit Andrey Poleshko, Katelyn M. Mansfield, Caroline
Cell Reports, Volume 5 Supplemental Information The Human Protein PRR14 Tethers Heterochromatin to the Nuclear Lamina During Interphase and Mitotic Exit Andrey Poleshko, Katelyn M. Mansfield, Caroline
Nature Methods: doi: /nmeth Supplementary Figure 1. Retention of RNA with LabelX.
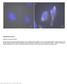 Supplementary Figure 1 Retention of RNA with LabelX. (a) Epi-fluorescence image of single molecule FISH (smfish) against GAPDH on HeLa cells expanded without LabelX treatment. (b) Epi-fluorescence image
Supplementary Figure 1 Retention of RNA with LabelX. (a) Epi-fluorescence image of single molecule FISH (smfish) against GAPDH on HeLa cells expanded without LabelX treatment. (b) Epi-fluorescence image
Synthetic Biology for
 Synthetic Biology for Plasmids and DNA Digestion Plasmids Plasmids are small DNA molecules that are separate from chromosomal DNA They are most commonly found as double stranded, circular DNA Typical plasmids
Synthetic Biology for Plasmids and DNA Digestion Plasmids Plasmids are small DNA molecules that are separate from chromosomal DNA They are most commonly found as double stranded, circular DNA Typical plasmids
See what s really in your sample
 See what s really in your sample Dye-free quantification of the biomolecules that you desire and the contaminants that you fear From the nucleic acid experts! Sample to Insight With QIAxpert Tell DNA from
See what s really in your sample Dye-free quantification of the biomolecules that you desire and the contaminants that you fear From the nucleic acid experts! Sample to Insight With QIAxpert Tell DNA from
