MoGUL: Detecting Common Insertions and Deletions in a Population
|
|
|
- Warren Kelley
- 6 years ago
- Views:
Transcription
1 MoGUL: Detecting Common Insertions and Deletions in a Population Seunghak Lee 1,2, Eric Xing 2, and Michael Brudno 1,3, 1 Department of Computer Science, University of Toronto, Canada 2 School of Computer Science, Carnegie Mellon University, USA 3 Banting and Best Dept. of Medical Research, University of Toronto, Canada Abstract. While the discovery of structural variants in the human population is ongoing, most methods for this task assume that the genome is sequenced to high coverage (e.g. 40x), and use the combined power of the many sequenced reads and mate pairs to identify the variants. In contrast, the 1000 Genomes Project hopes to sequence hundreds of human genotypes, but at low coverage (4-6x), and most of the current methods are unable to discover insertion/deletion and structural variants from this data. In order to identify indels from multiple low-coverage individuals we have developed the MoGUL (Mixture of Genotypes Variant Locator) framework, which identifies potential locations with indels by examining mate pairs generated from all sequenced individuals simultaneously, uses a Bayesian network with appropriate priors to explicitly model each individual as homozygous or heterozygous for each locus, and computes the expected Minor Allele Frequency (MAF) for all predicted variants. We have used MoGUL to identify variants in 1000 Genomes data, as well as in simulated genotypes, and show good accuracy at predicting indels, especially for MAF > 0.06 and indel size > 20 base pairs. 1 Introduction Next generation sequencing technologies have dramatically decreased the cost of sequencing human genomes. These technologies are enabling the 1000 Genomes Project - an ambitious undertaking to reconstruct hundreds of genotypes and understand the polymorphisms present in the human population. The resequencing of humans for the 1000 Genomes Project uses a combination of approaches, including deep sequencing of several individuals and whole-exome resequencing via DNA-capture. Simultaneously, the largest fraction of individuals will be sequenced via a low-coverage whole-genome shotgun approach, where each individual will be sequenced to 4-6x coverage. At this point in time it is not clear if this low coverage will be sufficient to identify a large fraction of the human variation, especially structural genomic polymorphisms. To whom correspondence should be addressed, brudno@cs.toronto.edu
2 2 While methods for the discovery of SNPs from read mapping have been available for some time [1], and the past two years have seen several tools developed specifically for discerning SNPs from NGS data ([2,3,4]), the development of algorithms for the identification of larger, structural variants (SVs), including insertions and deletions (indels), is still a very active research area. While the identification of very small indels can be accomplished by directly analyzing the read mappings, with 36bp reads it is difficult to identify indels > 10 bases. The identification of larger indels and other rearrangements is typically accomplished via the mate pair, or pair-end mapping technique (see [5] for a review). In this approach, two reads are sequenced from the two ends of a DNA fragment (the insert). Because the size of the DNA fragment is (approximately) known, structural variants can be identified by comparing the expected insert size to the distance between the mapped reads in the reference genome: if these are significantly different (the mate pair is termed discordant), it is likely that an SV has occurred between the two mappings. The past few years have seen the development of several novel methodologies and tools for SV discovery based on the analysis of discordant mate pairs, including a formal framework for identification of structural variants [6], tools that allow for flexible clustering of mate pairs to identify SVs [7], maximum parsimony and maximum likelihood approaches for SV detection [8], as well as tools that combine pair-end mapping with careful analysis of unpaired reads to assemble SV breakpoints [9]. Previously we proposed MoDIL [10], a method for SV identification based on the analysis of all matepairs (concordant and discordant) that span a particular genomic location. MoDIL (Mixture of Distributions Indel Locator) fits two (possibly shifted) distributions of insert sizes (corresponding to the two haploid genotypes in a diploid) to the observed mapped distances at each location in the genome. By analyzing these distributions it is possible to discover much smaller indels than with other mate pair-based approaches. MoDIL, however, cannot be directly applied to low coverage individuals, including the bulk of the 1000 genomes data, as it requires at least 20 inserts covering a genomic locus to identify indels (it is difficult to accurately fit two distributions with fewer data points). In the 1000 Genomes data, each locus is expected to be covered, on average, by 4 mate pairs in each individual. While the total coverage from all individuals is much higher, and most polymorphisms are di-allelic (i.e. there are only two alleles at a given locus in the human population), MoDIL expects the fractions of matepairs sampled from each haplotype to be approximately equal. In contrast, in the 1000 Genomes data the fractions are determined by the allele frequencies and will vary across the loci. In this work we build a Bayesian approach for the discovery of indel polymorphisms from mixtures of large numbers of genotypes, such as 1000 genomes data. Our approach, MoGUL (Mixture of Genotypes Variant Locator), builds a Bayesian network that uses priors to explicitly model each individual as homozygous or heterozygous, and computes the expected Minor Allele Frequency (MAF) at each location along the chromosome. We use MoGUL to identify variants in the 1000 Genomes data and simulated genotypes, and demonstrate that
3 3 it allows for the identification of indels > 30 bases for MAF > 0.04, while indels as small as 20 bases can be identified for MAF > Density NA12716 NA11920 NA07346 NA19225 NA Mapped distance mean of insert size Fig. 1. Distribution of insert sizes from different individuals, shifted so that they are all centered at zero. Note the discrepancies among the individual distribution, necessitating modeling them as separate random variables. Here mean of insert sizes are set to be zero. 2 Methods The main difficulty in identifying indels from paired-end data is differentiating mate pairs coming from a locus with an indel from those with an anomalous insert size. The insert size from each individual l follows a distribution, p(y l ) (see Fig. 1), and individual mate pairs generated from the tail of the distribution are impossible to discern from mate pairs overlapping an indel. Previous methods, such as MoDIL [10] and BreakDancer [9], use support from other mate pairs, generated by the high matepair coverage to separate these cases. While each individual in our dataset will have only a few mate pairs sampled at every genomic location, our algorithm combines the mate pairs generated from many individuals to achieve sufficient coverage. MoGUL models mate pairs as generated from either one or two unknown distributions, corresponding to the two possible alleles at this location among the human genotypes. Our algorithm does not consider tri-allelic variants, which are rare. Our algorithm starts by mapping all of the matepairs onto the reference genome. We use the MrFAST tool [11], which identifies mappings for every mate pair that has at most 2 mismatches in each read and has the mapped distance (the distance between the forward and reverse reads of the pair) closest to the expected insert size. If this mapped distance is within 3 standard deviations,
4 4 only the best mapping is identified. If no such mapping is found, all possible mappings for the two reads are returned, and our algorithm considers all of them. For every genomic location we identify those matepairs that would be affected if that location was the site of an indel. These matepairs will have the two reads mapping on opposite sides of the genomic location, and we refer to this set of matepairs as a cluster (see next section). Ifthegenomiclocationisthesiteofanindelthatispolymorphicinthehuman population, matepairs in the corresponding cluster may be generated from two distributions, corresponding to the two alleles (with and without the indel). Using a Bayesian network we infer the size of the indel, as well as the individuals with indels for each cluster. Because our model may identify the same indel calls from multiple clusters, a final post-processing step is used to combine these calls and to compute the log likelihood ratio between our model and the null model. For simplicity, in the following sections we will call a matepair discordant if there is significant disparity between insert size and mapped distance, and concordant otherwise. Note that these terms are only used for convenience we do not a priori assign matepairs to these categories. 2.1 Clustering Matepairs We first generate clusters with mappings of matepairs for each genomic locus, and determine whether or not the locus contains a common indel. In this step we find a set of mate pairs C from L number of individuals, all of which overlap with a particular genomic location. Figure 2 illustrates our clustering scheme. For each matepair we look at one base after the left read and all matepairs overlapping the location form a cluster C. We explain how we detect indels from these clusters by example. Suppose the matepairs in Figure 2 are from the same matepair library with the first two matepairs discordant and the rest concordant. In such a case, as shown in Figure 2, the first two matepairs agree on a certain indel size (they have similar mapped distance), and the indel can be detected from the second to the fourth cluster containing the two discordant matepairs (we merge indel calls in a post-processing step). If we use all the clusters generated by this scheme, the number of clusters will be close to the number of matepairs, and the algorithm will be too slow. Instead, we filter out clusters if it is very likely that there is no indel iat the corresponding location. For each individual l, we compute the likelihood that the mate pairs were generated from a cluster with no indel (p-value). If there is at least one individual with significant p-value (< 0.001) or two individuals with less significant p-value (< 0.05), the locus is deemed significant. We define the p-value as the probability of having at least predicted size of indel (> γ) given no indels. Let {D l1,...,d ln } represent independent and identically distributed random variables corresponding to the mapped distances of mate pairs generated from the l-th individual with insert size distribution p(y l ), mean µ Yl and standard deviation σ Yl. Their mean follows the Gaussian distribution with mean equal to the mean of the insert size µ Yl and standard
5 5 deviationofσ Yl / naccordingtothecentrallimittheorem.wedefinethep-value for the individual l with the size of indel γ as follows: p-value = P(X;0,σ Yl / n) = γ 0 P(X;γ,σ Yl / n) Here, X = D µ Yl is the expected size of the indel, and P(X) follows the Gaussian distribution. The second equality can be proven via symmetry of Gaussian. In computing the p-value we correct for the possibility that the cluster contains a heterozygous indel by using a shifted sample mean: γ = 2γ. Fig. 2. This figure shows how to generate clusters with mapped matepairs in the reference genome. Matepairs are colored by red or blue representing different individuals. For each matepair X i, we generate a cluster consisting of all mate pairs overlapping a genomic location of one base after the left read of the matepair X i (the locations of the arrows). 2.2 Detecting Common Indels Using a Bayesian network The clusters from Sec. 2.1 include mate pairs generated from many individuals, all of which have unique distributions of insert sizes (see Figure 1). We define the variable X lm as the expected size of indel from the m-th matepair of individual l: X lm = D lm µ Yl where D lm is mapped distance of the m-th matepair of the individual l and µ Yl is mean of the insert size distribution p(y l ). We will use random variabe X li instead of D li because it shifts the distributions for all individuals so that they are all centered at zero. Given a cluster of matepairs as an input, we developed a Bayesian network (Figure 3) to infer the size of the indel polymorphism (if one exists), and haplotypes of individuals that contain the indel. The Bayesian networkgeneratesmatepairs{x lm },whileinternalstatescorrespondtothepresence/absence of indel and its heterozygosity. All random variables are defined for an input cluster, rather than the whole individual genome. We model the individual l with random variable Z l : { 0 if individual l has no indel Z l = 1 if individual l has an indel.
6 6 α Z l L π M l β γ X lm Q lm θ l Fig. 3. Bayesian network for detecting common indels at a particular locus in the genome. Here L represents the number of individuals and M l is the number of matepairs from individual l. The random variable Z l determines whether individual l has an indel or not. If individual l has an indel (Z l = 1), Q lm generates a matepair X lm and θ l controls the heterozygosity of Z l. Matepair, X lm, is generated from distribution of insert sizes with zero mean or with shifted mean of γ if X lm has an indel. If individual l has no indel (Z l = 0), matepairs {X lm } M l m=1 are generated from p(y l ) with zero mean. Priors π and θ l are controlled by α and β parameters. WeusetherandomvariableQ lm tomodelthetwocopiesofchromosomes(alleles) in individual l. Note that subscript l refers to individual l and m denotes m-th matepair generated from this individual: 0 if Z l = 1 and chromosome contains no indel Q lm = 1 if Z l = 1 and chromosome contains an indel 2 if Z l = 0. As shown in Figure 3 we can generate X lm given Z l, Q lm and size of indel γ. For example, if Q lm = 1, X lm is generated from p(y l ) with an indel size of γ. If {Z l = 0 Q lm = 0} then X lm is generated from p(y l ) with no indel. For simplicity we omit the p(y l )s in Fig. 3. To avoid overfitting problems we applied Bayesian priors π and θ l to Z l and Q lm, respectively. Wesmooththedistributionofp(Y l ),anddefineanewprobabilitydistribution of insert sizes, q(x l ), for individual l as follows: { k q(x l ) = iσ Yl y µ Yl <k i+1σ Yl p Yl (y) if k i σ Yl X l < k i+1 σ Yl p Yl (y) if k j+1 σ Y l X l < k j σ Y l k j+1 σy l y µy l < k j σy l Herewesump Yl (y)sovertheintervals[k i σ Yl,k i+1 σ Yl )fordeletionsand[ k j+1 σ Y l, k j σ Y l ) for insertions. In our experiments, we used 10 values of k i and k j s (i,j {1,2,...,10}, k 1 = k 1 = 0). Probability distributions of the random variables in
7 7 Figure 3 are defined as follows: p(z l = z π) = π z (1 π) 1 z wherez = 0ifindividuallhasnoindelandP(Z l = 0) = π andp(z l = 1) = 1 π. p(q lm = q Z l = 1,θ l ) = θ q l (1 θ l) 1 q where q = 0 if the chromosome contains no indel, and 1 otherwise. If Z 1 = 0, we do not generate matepair X lm from Q lm and set q = 2. We generate X lm from the following distribution: { q(xlm ) if {Z p(x lm Z l,q lm,γ) = l = 0 q = 0} q(x lm γ) if q = 1. The priors π and θ l follow the beta distribution, which is the conjugate prior of binomial distributions. To infer the states of our model, we find maximum a posteriori (MAP) solution because it is fast and deterministic. We initialize our model using heuristics (e.g. Q lm = 1 if X lm > σ l ) and random configurations, and run the model multiple times to avoid local maxima. Given current states of the model the update rules are given as follows (updated states are denoted by (*)): π = u+α 1 1 L+α 1 +α 2 2 whereuisthenumberofindividualswithnoindel.inpracticeweuseα = {30,1} because most variants have a small MAF [12]. θ l = v +β 1 1 M l +β 1 +β 2 2 where v is the number of matepairs with Q lm = 0 in individual l, and we set β = {5,5}, favoring heterozygous indels, as these are more likely under a neutral evolutionary model. We update the random variables γ, Z l and Q lm as follows: Z l = argmax Z l {0,1} γ = argmax γ P(Z l π) L M l l=1m=1 M l m=1 P(X lm Z l,q lm,γ) P(X lm Z l,γ,q lm )P(Q lm Z l,θ l ) Q lm = argmax P(Q lm Z l,θ l )P(X lm Z l,q lm,γ). Q lm {0,1,2} This algorithm is iterated, with each hidden random variable updated until the posterior probability of the model cannot be improved by the value of the threshold (e.g. τ = 10 4 ).
8 8 2.3 Merging and Assigning Confidence to Indel Calls In the post-processing step we merge duplicated indel calls. As shown in Sec. 2.1, a single indel may be found in multiple clusters. We merge indel calls if they meet three criteria: (1) the predicted indel regions overlap, (2) the expected size of the indel is similar (< σ mix ), (3) the sets of individuals for whom the indel is predicted overlap. Here σ mix is the standard deviation of insert sizes from all individuals. To assign confidence values for every cluster we compute the log likelihood ratio R between our model and null model as follows: R = L M l l=1 m=1 logp(x lm Z l,q lm,γ) L M l l=1 m=1 logp(x lm Z l,q lm,0). We discard indel calls if the log likelihood ratio is not significantly larger than a pre-specified threshold (by default, 30). 3 Results In the sections below, we use two different approaches to validate our algorithms. First, we use simulated data to evaluate how well MoGUL performs at different variant frequencies, and then use MoGUL to perform variant discovery on one chromosome of the current 1000 genomes dataset, that includes 124 individuals sequenced at approximately 4x. 3.1 Simulation results We first validate our model through simulation results. In our simulation, we sampled matepairs from 120 individuals, with the matepair library size of each individual l following the experimental distribution p(y l ). We generated indels of base pairs and implanted them in the individual genomes, varying the minor allele frequency (MAF) from 0.02 to 0.5. Fig. 4 shows the heatmap for the performance of MoGUL. MoGUL works well for MAF greater than 0.06, for indels > 20 base pairs. To investigate the precision and recall rate of MoGUL we generated 10,000 clusters with 50 individuals (100 haplotypes) clusters contained implanted indels of base pairs, while 100 clusters contained implanted indels of base pairs. For each individual we sampled matepairs with approximately 2-3x read coverage. We detected indels for these individuals using MoGUL. The recall and precision rates of our algorithm are shown in Table genome project pilot dataset In order to validate MoGUL on real data, we downloaded low coverage individuals generated by the pilot project for the 1000 genomes project from the NCBI
9 9 Minor allele frequency (MAF) Size of deletion Fig. 4. Heatmap representing the performance of MoGUL. The color of each cell indicates average error rate of 20 MoGUL simulations for a given combination of deletion size (X axis) and Minor Allele Frequencies (Y axis). If the size of indel predictions by MoGUL is more than 10bp away from the true size of deletion we consider it incorrect. trace archive, aligned these to the NCBI reference genome with MrFAST [11], and predicted indels for all of these on chromosome 20. The results are summarized in Table 1. Overall, MoGUL predicted 3,545 events in any individual on chromosome 20. This is approximately 630 events per individual. We compare these indels to previously discovered variants both across the population, and for one specific individual, NA To our knowledge, the only previous study that has characterize small to medium size indels in the human populations is by Mills et al [13]. They used low coverage Sanger-style reads from 36 individuals to identify indels via the split-read mapping approach. Thus they are able to identify the exact size of the indel, while the MoGUL method infers it indirectly from the discordant mappings. Overall, the overlap between MoGUL and the indels of Mills et al was statistically significant. While exact sensitivity and specificity of the two methods is difficult to analyze, as different (and fewer) individuals were used for the Mills et al study, the size correlation of overlapping indels was very strong, and the overall error of MoGUL size estimates was small (see Figure 6). In order to enable the direct comparison of indel discovery from single high coverage individual versus multiple low coverage individuals, we included in our dataset a down-sampled version of the NA18507 Yoruban genome which we previously analyzed using the MoDIL method. Remarkably, MoGUL was able to identify 83% of the indels > 50bp (20/24) that were previously detected by MoDIL, while identifying several additional variants that were missed by MoDIL,
10 10 Table 1. Comparison between indel calls in chromosome 20 located by our approach with the datasets generated by Mills et al. [13] (all MoGUL indels), and MoDIL [10] (only indels in NA18507, the same individual as was studied by Lee et al., was considered). For the simulation experiments we consider the indel call is correct if the difference between the true indel size and the predicted one is less than 10bp and the log likelihood ratio is greater than 10. Population NA18507 Simulation Length Type MoGUL Mills et al. Overlap MoGUL MoDIL Overlap Recall Precision 100bp INS DEL bp INS DEL bp INS DEL possibly due to low coverage in the NA18507 individual specifically. Of the events 20-50bp, 40% (43/108) were recovered by MoGUL. In Figure 5 we plot the minor allele frequency of the variants discovered by our method. The distribution agrees with the expected curve until MAF 0.07, but then drops rapidly demonstrating MoGUL s inability to identify indels at low minor allele frequencies. 4 Discussion The identification of various polymorphisms in the human population is an important step towards understanding the landscape of human genotypes. In this paper we present MoGUL: the Mixture of Genotypes Variant Locator, a tool to identify common insertion/deletion polymorphisms from many individuals sequenced at low coverage. We validate our approach via simulated data at various allele frequencies, as well as with data from the 1000 Genomes project. MoGUL can identify indels >20 base pairs with at least 0.06 MAF, using the current low coverage data; it is expected that the coverage will double to 6-8x per individual for the final 1000 Genomes project data release, and we are hopeful that MoGUL s performance will further improve on this larger dataset. Another application of MoGUL is resequencing of biopsy tissues, where the diseased (tumourous) tissue is biopsied (and sequenced) together with the healthy surrounding tissue, leading to a mixture of several genotypes at each location. Simultaneously, MoGUL is only capable of recapturing a small fraction of the rare variants that predominate in the human population. While capturing common genotypes is important, it is thought that rare alleles, ones with MAF < 0.01, are much more likely to be evolutionarily harmful and disease related [12]. Designing methods that can find these variants from paired-end data, possibly incorporating direct information on read matches, as in the Pindel tool [14], is an important avenue for further research.
11 Number of indels Minor allele frequency (MAF) Fig. 5. Distribution of minor allele frequencies for indels in the 1000 genomes dataset. a 500 b 70 MoGUL indel sizes (bp) Mills et al. indel sizes (bp) Number of indels Mills et al. minus MoGUL (bp) Fig. 6. (A) A scatter plot showing the lengths of overlapping indels between the Mills et al. dataset and MoGUL predictions. Overall the lengths are highly correlated. The cluster of indels of length 300 corresponds to Alu element activity. (B) The absolute error in the estimation of indel length. The predicted lengths of the indels are very close (typically within 10 bases) of the true indel size. Overall the distribution of the error follows a Gaussian, as expected from the model (see [10] for details). The outliers may indicate either false positives for either dataset or tri-allelic variants.
12 12 5 Acknowledgments We would like to thank Lisa Brooks of the NIH for permission to use 1000 genomes data, and to Can Alkan and Fereydoun Hormozdiari for providing us with the MrFAST mapping tool. References 1. Marth, G. T. et al. A general approach to single-nucleotide polymorphism discovery. Nature Genetics 23, (1999). 2. Li, H., Ruan, J. & Durbin, R. Mapping short dna sequencing reads and calling variants using mapping quality scores. Genome Research 18, (2008). 3. Li, R. et al. Snp detection for massively parallel whole-genome resequencing. Genome Research 19, (2009). 4. Hoberman, R. et al. A probabilistic approach for SNP discovery in high-throughput human resequencing data. Genome Research 19, (2009). 5. Medvedev, P., Stanciu, M. & Brudno, M. Computational methods for discovering structural variation with high throughput sequencing. Nature Methods 6, S13 S20 (2009). 6. Lee, S., Cheran, E. & Brudno, M. A robust framework for detecting structural variations in a genome. Bioinformatics 24, i59 i67 (2008). 7. Korbel, J. et al. Paired-end mapping reveals extensive structural variation in the human genome. Science 318, (2007). 8. Hormozdiari, F., Alkan, C., Eichler, E. E. & Sahinalp, S. C. Combinatorial algorithms for structural variation detection in high-throughput sequenced genomes. Genome Research 19, (2009). 9. Chen, K. et al. Breakdancer: an algorithm for high-resolution mapping of genomic structural variation. Nature Methods 6, (2009). 10. Lee, S., Hormozdiari, F., Alkan, C. & Brudno, M. MoDIL: Detecting INDEL Variation with Mixtures of Distributions. Nature Methods 6, (2009). 11. Alkan, C. et al. Personalized copy number and segmental duplication maps using next-generation sequencing. Nature Genetics 41, (2009). 12. Kryukov, G., Shpunt, A., Stamatoyannopoulos, J.& Sunyaev, S. Power of deep, allexon resequencing for discovery of human trait genes. Proceedings of the National Academy of Sciences 106, (2009). 13. Mills, R. et al. An initial map of insertion and deletion (INDEL) variation in the human genome. Genome Research 16, (2006). 14. Ye, K., Schulz, M. H., Long, Q., Apweiler, R. & Ning, Z. Pindel: a pattern growth approach to detect break points of large deletions and medium sized insertions from paired-end short reads. Bioinformatics, (2009).
SNP calling and VCF format
 SNP calling and VCF format Laurent Falquet, Oct 12 SNP? What is this? A type of genetic variation, among others: Family of Single Nucleotide Aberrations Single Nucleotide Polymorphisms (SNPs) Single Nucleotide
SNP calling and VCF format Laurent Falquet, Oct 12 SNP? What is this? A type of genetic variation, among others: Family of Single Nucleotide Aberrations Single Nucleotide Polymorphisms (SNPs) Single Nucleotide
CS273B: Deep Learning in Genomics and Biomedicine. Recitation 1 30/9/2016
 CS273B: Deep Learning in Genomics and Biomedicine. Recitation 1 30/9/2016 Topics Genetic variation Population structure Linkage disequilibrium Natural disease variants Genome Wide Association Studies Gene
CS273B: Deep Learning in Genomics and Biomedicine. Recitation 1 30/9/2016 Topics Genetic variation Population structure Linkage disequilibrium Natural disease variants Genome Wide Association Studies Gene
Characterization of Allele-Specific Copy Number in Tumor Genomes
 Characterization of Allele-Specific Copy Number in Tumor Genomes Hao Chen 2 Haipeng Xing 1 Nancy R. Zhang 2 1 Department of Statistics Stonybrook University of New York 2 Department of Statistics Stanford
Characterization of Allele-Specific Copy Number in Tumor Genomes Hao Chen 2 Haipeng Xing 1 Nancy R. Zhang 2 1 Department of Statistics Stonybrook University of New York 2 Department of Statistics Stanford
Variation detection based on second generation sequencing data. Xin LIU Department of Science and Technology, BGI
 Variation detection based on second generation sequencing data Xin LIU Department of Science and Technology, BGI liuxin@genomics.org.cn 2013.11.21 Outline Summary of sequencing techniques Data quality
Variation detection based on second generation sequencing data Xin LIU Department of Science and Technology, BGI liuxin@genomics.org.cn 2013.11.21 Outline Summary of sequencing techniques Data quality
Runs of Homozygosity Analysis Tutorial
 Runs of Homozygosity Analysis Tutorial Release 8.7.0 Golden Helix, Inc. March 22, 2017 Contents 1. Overview of the Project 2 2. Identify Runs of Homozygosity 6 Illustrative Example...............................................
Runs of Homozygosity Analysis Tutorial Release 8.7.0 Golden Helix, Inc. March 22, 2017 Contents 1. Overview of the Project 2 2. Identify Runs of Homozygosity 6 Illustrative Example...............................................
Mutations during meiosis and germ line division lead to genetic variation between individuals
 Mutations during meiosis and germ line division lead to genetic variation between individuals Types of mutations: point mutations indels (insertion/deletion) copy number variation structural rearrangements
Mutations during meiosis and germ line division lead to genetic variation between individuals Types of mutations: point mutations indels (insertion/deletion) copy number variation structural rearrangements
Phasing of 2-SNP Genotypes based on Non-Random Mating Model
 Phasing of 2-SNP Genotypes based on Non-Random Mating Model Dumitru Brinza and Alexander Zelikovsky Department of Computer Science, Georgia State University, Atlanta, GA 30303 {dima,alexz}@cs.gsu.edu Abstract.
Phasing of 2-SNP Genotypes based on Non-Random Mating Model Dumitru Brinza and Alexander Zelikovsky Department of Computer Science, Georgia State University, Atlanta, GA 30303 {dima,alexz}@cs.gsu.edu Abstract.
Personal Genomics Platform White Paper Last Updated November 15, Executive Summary
 Executive Summary Helix is a personal genomics platform company with a simple but powerful mission: to empower every person to improve their life through DNA. Our platform includes saliva sample collection,
Executive Summary Helix is a personal genomics platform company with a simple but powerful mission: to empower every person to improve their life through DNA. Our platform includes saliva sample collection,
Measurement of Molecular Genetic Variation. Forces Creating Genetic Variation. Mutation: Nucleotide Substitutions
 Measurement of Molecular Genetic Variation Genetic Variation Is The Necessary Prerequisite For All Evolution And For Studying All The Major Problem Areas In Molecular Evolution. How We Score And Measure
Measurement of Molecular Genetic Variation Genetic Variation Is The Necessary Prerequisite For All Evolution And For Studying All The Major Problem Areas In Molecular Evolution. How We Score And Measure
Axiom mydesign Custom Array design guide for human genotyping applications
 TECHNICAL NOTE Axiom mydesign Custom Genotyping Arrays Axiom mydesign Custom Array design guide for human genotyping applications Overview In the past, custom genotyping arrays were expensive, required
TECHNICAL NOTE Axiom mydesign Custom Genotyping Arrays Axiom mydesign Custom Array design guide for human genotyping applications Overview In the past, custom genotyping arrays were expensive, required
LUMPY: A probabilistic framework for structural variant discovery
 LUMPY: A probabilistic framework for structural variant discovery Ryan M Layer 1, Aaron R Quinlan* 1,2,3,4 and Ira M Hall* 2,4 1 Department of Computer Science 2 Department of Biochemistry and Molecular
LUMPY: A probabilistic framework for structural variant discovery Ryan M Layer 1, Aaron R Quinlan* 1,2,3,4 and Ira M Hall* 2,4 1 Department of Computer Science 2 Department of Biochemistry and Molecular
Variant Detection in Next Generation Sequencing Data. John Osborne Sept 14, 2012
 + Variant Detection in Next Generation Sequencing Data John Osborne Sept 14, 2012 + Overview My Bias Talk slanted towards analyzing whole genomes using Illumina paired end reads with open source tools
+ Variant Detection in Next Generation Sequencing Data John Osborne Sept 14, 2012 + Overview My Bias Talk slanted towards analyzing whole genomes using Illumina paired end reads with open source tools
Computational methods for the analysis of rare variants
 Computational methods for the analysis of rare variants Shamil Sunyaev Harvard-M.I.T. Health Sciences & Technology Division Combine all non-synonymous variants in a single test Theory: 1) Most new missense
Computational methods for the analysis of rare variants Shamil Sunyaev Harvard-M.I.T. Health Sciences & Technology Division Combine all non-synonymous variants in a single test Theory: 1) Most new missense
Nature Biotechnology: doi: /nbt Supplementary Figure 1. Number and length distributions of the inferred fosmids.
 Supplementary Figure 1 Number and length distributions of the inferred fosmids. Fosmid were inferred by mapping each pool s sequence reads to hg19. We retained only those reads that mapped to within a
Supplementary Figure 1 Number and length distributions of the inferred fosmids. Fosmid were inferred by mapping each pool s sequence reads to hg19. We retained only those reads that mapped to within a
Variant calling in NGS experiments
 Variant calling in NGS experiments Jorge Jiménez jjimeneza@cipf.es BIER CIBERER Genomics Department Centro de Investigacion Principe Felipe (CIPF) (Valencia, Spain) 1 Index 1. NGS workflow 2. Variant calling
Variant calling in NGS experiments Jorge Jiménez jjimeneza@cipf.es BIER CIBERER Genomics Department Centro de Investigacion Principe Felipe (CIPF) (Valencia, Spain) 1 Index 1. NGS workflow 2. Variant calling
Genome-wide association studies (GWAS) Part 1
 Genome-wide association studies (GWAS) Part 1 Matti Pirinen FIMM, University of Helsinki 03.12.2013, Kumpula Campus FIMM - Institiute for Molecular Medicine Finland www.fimm.fi Published Genome-Wide Associations
Genome-wide association studies (GWAS) Part 1 Matti Pirinen FIMM, University of Helsinki 03.12.2013, Kumpula Campus FIMM - Institiute for Molecular Medicine Finland www.fimm.fi Published Genome-Wide Associations
Chromosome-scale scaffolding of de novo genome assemblies based on chromatin interactions. Supplementary Material
 Chromosome-scale scaffolding of de novo genome assemblies based on chromatin interactions Joshua N. Burton 1, Andrew Adey 1, Rupali P. Patwardhan 1, Ruolan Qiu 1, Jacob O. Kitzman 1, Jay Shendure 1 1 Department
Chromosome-scale scaffolding of de novo genome assemblies based on chromatin interactions Joshua N. Burton 1, Andrew Adey 1, Rupali P. Patwardhan 1, Ruolan Qiu 1, Jacob O. Kitzman 1, Jay Shendure 1 1 Department
Genomic models in bayz
 Genomic models in bayz Luc Janss, Dec 2010 In the new bayz version the genotype data is now restricted to be 2-allelic markers (SNPs), while the modeling option have been made more general. This implements
Genomic models in bayz Luc Janss, Dec 2010 In the new bayz version the genotype data is now restricted to be 2-allelic markers (SNPs), while the modeling option have been made more general. This implements
BST227 Introduction to Statistical Genetics. Lecture 8: Variant calling from high-throughput sequencing data
 BST227 Introduction to Statistical Genetics Lecture 8: Variant calling from high-throughput sequencing data 1 PC recap typical genome Differs from the reference genome at 4-5 million sites ~85% SNPs ~15%
BST227 Introduction to Statistical Genetics Lecture 8: Variant calling from high-throughput sequencing data 1 PC recap typical genome Differs from the reference genome at 4-5 million sites ~85% SNPs ~15%
Systematic comparison of CRISPR/Cas9 and RNAi screens for essential genes
 CORRECTION NOTICE Nat. Biotechnol. doi:10.1038/nbt. 3567 Systematic comparison of CRISPR/Cas9 and RNAi screens for essential genes David W Morgens, Richard M Deans, Amy Li & Michael C Bassik In the version
CORRECTION NOTICE Nat. Biotechnol. doi:10.1038/nbt. 3567 Systematic comparison of CRISPR/Cas9 and RNAi screens for essential genes David W Morgens, Richard M Deans, Amy Li & Michael C Bassik In the version
POPULATION GENETICS Winter 2005 Lecture 18 Quantitative genetics and QTL mapping
 POPULATION GENETICS Winter 2005 Lecture 18 Quantitative genetics and QTL mapping - from Darwin's time onward, it has been widely recognized that natural populations harbor a considerably degree of genetic
POPULATION GENETICS Winter 2005 Lecture 18 Quantitative genetics and QTL mapping - from Darwin's time onward, it has been widely recognized that natural populations harbor a considerably degree of genetic
De Novo Assembly of High-throughput Short Read Sequences
 De Novo Assembly of High-throughput Short Read Sequences Chuming Chen Center for Bioinformatics and Computational Biology (CBCB) University of Delaware NECC Third Skate Genome Annotation Workshop May 23,
De Novo Assembly of High-throughput Short Read Sequences Chuming Chen Center for Bioinformatics and Computational Biology (CBCB) University of Delaware NECC Third Skate Genome Annotation Workshop May 23,
Mate-pair library data improves genome assembly
 De Novo Sequencing on the Ion Torrent PGM APPLICATION NOTE Mate-pair library data improves genome assembly Highly accurate PGM data allows for de Novo Sequencing and Assembly For a draft assembly, generate
De Novo Sequencing on the Ion Torrent PGM APPLICATION NOTE Mate-pair library data improves genome assembly Highly accurate PGM data allows for de Novo Sequencing and Assembly For a draft assembly, generate
Sequence assembly. Jose Blanca COMAV institute bioinf.comav.upv.es
 Sequence assembly Jose Blanca COMAV institute bioinf.comav.upv.es Sequencing project Unknown sequence { experimental evidence result read 1 read 4 read 2 read 5 read 3 read 6 read 7 Computational requirements
Sequence assembly Jose Blanca COMAV institute bioinf.comav.upv.es Sequencing project Unknown sequence { experimental evidence result read 1 read 4 read 2 read 5 read 3 read 6 read 7 Computational requirements
Midterm 1 Results. Midterm 1 Akey/ Fields Median Number of Students. Exam Score
 Midterm 1 Results 10 Midterm 1 Akey/ Fields Median - 69 8 Number of Students 6 4 2 0 21 26 31 36 41 46 51 56 61 66 71 76 81 86 91 96 101 Exam Score Quick review of where we left off Parental type: the
Midterm 1 Results 10 Midterm 1 Akey/ Fields Median - 69 8 Number of Students 6 4 2 0 21 26 31 36 41 46 51 56 61 66 71 76 81 86 91 96 101 Exam Score Quick review of where we left off Parental type: the
Amapofhumangenomevariationfrom population-scale sequencing
 doi:.38/nature9534 Amapofhumangenomevariationfrom population-scale sequencing The Genomes Project Consortium* The Genomes Project aims to provide a deep characterization of human genome sequence variation
doi:.38/nature9534 Amapofhumangenomevariationfrom population-scale sequencing The Genomes Project Consortium* The Genomes Project aims to provide a deep characterization of human genome sequence variation
Read Mapping and Variant Calling. Johannes Starlinger
 Read Mapping and Variant Calling Johannes Starlinger Application Scenario: Personalized Cancer Therapy Different mutations require different therapy Collins, Meredith A., and Marina Pasca di Magliano.
Read Mapping and Variant Calling Johannes Starlinger Application Scenario: Personalized Cancer Therapy Different mutations require different therapy Collins, Meredith A., and Marina Pasca di Magliano.
Machine learning applications in genomics: practical issues & challenges. Yuzhen Ye School of Informatics and Computing, Indiana University
 Machine learning applications in genomics: practical issues & challenges Yuzhen Ye School of Informatics and Computing, Indiana University Reference Machine learning applications in genetics and genomics
Machine learning applications in genomics: practical issues & challenges Yuzhen Ye School of Informatics and Computing, Indiana University Reference Machine learning applications in genetics and genomics
Supplementary Note: Detecting population structure in rare variant data
 Supplementary Note: Detecting population structure in rare variant data Inferring ancestry from genetic data is a common problem in both population and medical genetic studies, and many methods exist to
Supplementary Note: Detecting population structure in rare variant data Inferring ancestry from genetic data is a common problem in both population and medical genetic studies, and many methods exist to
Genetic load. For the organism as a whole (its genome, and the species), what is the fitness cost of deleterious mutations?
 Genetic load For the organism as a whole (its genome, and the species), what is the fitness cost of deleterious mutations? Anth/Biol 5221, 25 October 2017 We saw that the expected frequency of deleterious
Genetic load For the organism as a whole (its genome, and the species), what is the fitness cost of deleterious mutations? Anth/Biol 5221, 25 October 2017 We saw that the expected frequency of deleterious
Reliable classification of two-class cancer data using evolutionary algorithms
 BioSystems 72 (23) 111 129 Reliable classification of two-class cancer data using evolutionary algorithms Kalyanmoy Deb, A. Raji Reddy Kanpur Genetic Algorithms Laboratory (KanGAL), Indian Institute of
BioSystems 72 (23) 111 129 Reliable classification of two-class cancer data using evolutionary algorithms Kalyanmoy Deb, A. Raji Reddy Kanpur Genetic Algorithms Laboratory (KanGAL), Indian Institute of
AP BIOLOGY Population Genetics and Evolution Lab
 AP BIOLOGY Population Genetics and Evolution Lab In 1908 G.H. Hardy and W. Weinberg independently suggested a scheme whereby evolution could be viewed as changes in the frequency of alleles in a population
AP BIOLOGY Population Genetics and Evolution Lab In 1908 G.H. Hardy and W. Weinberg independently suggested a scheme whereby evolution could be viewed as changes in the frequency of alleles in a population
H3A - Genome-Wide Association testing SOP
 H3A - Genome-Wide Association testing SOP Introduction File format Strand errors Sample quality control Marker quality control Batch effects Population stratification Association testing Replication Meta
H3A - Genome-Wide Association testing SOP Introduction File format Strand errors Sample quality control Marker quality control Batch effects Population stratification Association testing Replication Meta
Introduction to Bioinformatics
 Introduction to Bioinformatics Richard Corbett Canada s Michael Smith Genome Sciences Centre Vancouver, British Columbia June 28, 2017 Our mandate is to advance knowledge about cancer and other diseases
Introduction to Bioinformatics Richard Corbett Canada s Michael Smith Genome Sciences Centre Vancouver, British Columbia June 28, 2017 Our mandate is to advance knowledge about cancer and other diseases
Gap hunting to characterize clustered probe signals in Illumina methylation array data
 DOI 10.1186/s13072-016-0107-z Epigenetics & Chromatin RESEARCH Gap hunting to characterize clustered probe signals in Illumina methylation array data Shan V. Andrews 1,2, Christine Ladd Acosta 1,2,3, Andrew
DOI 10.1186/s13072-016-0107-z Epigenetics & Chromatin RESEARCH Gap hunting to characterize clustered probe signals in Illumina methylation array data Shan V. Andrews 1,2, Christine Ladd Acosta 1,2,3, Andrew
SVMerge Output File Format Specification Sheet
 SVMerge Output File Format Specification Sheet Document Number: 30165 Document Revision: C For Research Use Only. Not for use in diagnostic procedures. Copyright 2017 Bionano Genomics, Inc. All Rights
SVMerge Output File Format Specification Sheet Document Number: 30165 Document Revision: C For Research Use Only. Not for use in diagnostic procedures. Copyright 2017 Bionano Genomics, Inc. All Rights
Computational Workflows for Genome-Wide Association Study: I
 Computational Workflows for Genome-Wide Association Study: I Department of Computer Science Brown University, Providence sorin@cs.brown.edu October 16, 2014 Outline 1 Outline 2 3 Monogenic Mendelian Diseases
Computational Workflows for Genome-Wide Association Study: I Department of Computer Science Brown University, Providence sorin@cs.brown.edu October 16, 2014 Outline 1 Outline 2 3 Monogenic Mendelian Diseases
By the end of this lecture you should be able to explain: Some of the principles underlying the statistical analysis of QTLs
 (3) QTL and GWAS methods By the end of this lecture you should be able to explain: Some of the principles underlying the statistical analysis of QTLs Under what conditions particular methods are suitable
(3) QTL and GWAS methods By the end of this lecture you should be able to explain: Some of the principles underlying the statistical analysis of QTLs Under what conditions particular methods are suitable
Figure S1. Unrearranged locus. Rearranged locus. Concordant read pairs. Region1. Region2. Cluster of discordant read pairs, bundle
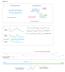 Figure S1 a Unrearranged locus Rearranged locus Concordant read pairs Region1 Concordant read pairs Cluster of discordant read pairs, bundle Region2 Concordant read pairs b Physical coverage 5 4 3 2 1
Figure S1 a Unrearranged locus Rearranged locus Concordant read pairs Region1 Concordant read pairs Cluster of discordant read pairs, bundle Region2 Concordant read pairs b Physical coverage 5 4 3 2 1
From Variants to Pathways: Agilent GeneSpring GX s Variant Analysis Workflow
 From Variants to Pathways: Agilent GeneSpring GX s Variant Analysis Workflow Technical Overview Import VCF Introduction Next-generation sequencing (NGS) studies have created unanticipated challenges with
From Variants to Pathways: Agilent GeneSpring GX s Variant Analysis Workflow Technical Overview Import VCF Introduction Next-generation sequencing (NGS) studies have created unanticipated challenges with
It s not a fundamental force like mutation, selection, and drift.
 What is Genetic Draft? It s not a fundamental force like mutation, selection, and drift. It s an effect of mutation at a selected locus, that reduces variation at nearby (linked) loci, thereby reducing
What is Genetic Draft? It s not a fundamental force like mutation, selection, and drift. It s an effect of mutation at a selected locus, that reduces variation at nearby (linked) loci, thereby reducing
Structural variation. Marta Puig Institut de Biotecnologia i Biomedicina Universitat Autònoma de Barcelona
 Structural variation Marta Puig Institut de Biotecnologia i Biomedicina Universitat Autònoma de Barcelona Genetic variation How much genetic variation is there between individuals? What type of variants
Structural variation Marta Puig Institut de Biotecnologia i Biomedicina Universitat Autònoma de Barcelona Genetic variation How much genetic variation is there between individuals? What type of variants
Genome-Wide Association Studies (GWAS): Computational Them
 Genome-Wide Association Studies (GWAS): Computational Themes and Caveats October 14, 2014 Many issues in Genomewide Association Studies We show that even for the simplest analysis, there is little consensus
Genome-Wide Association Studies (GWAS): Computational Themes and Caveats October 14, 2014 Many issues in Genomewide Association Studies We show that even for the simplest analysis, there is little consensus
Bioinformatics small variants Data Analysis. Guidelines. genomescan.nl
 Next Generation Sequencing Bioinformatics small variants Data Analysis Guidelines genomescan.nl GenomeScan s Guidelines for Small Variant Analysis on NGS Data Using our own proprietary data analysis pipelines
Next Generation Sequencing Bioinformatics small variants Data Analysis Guidelines genomescan.nl GenomeScan s Guidelines for Small Variant Analysis on NGS Data Using our own proprietary data analysis pipelines
Why can GBS be complicated? Tools for filtering, error correction and imputation.
 Why can GBS be complicated? Tools for filtering, error correction and imputation. Edward Buckler USDA-ARS Cornell University http://www.maizegenetics.net Many Organisms Are Diverse Humans are at the lower
Why can GBS be complicated? Tools for filtering, error correction and imputation. Edward Buckler USDA-ARS Cornell University http://www.maizegenetics.net Many Organisms Are Diverse Humans are at the lower
Improving the Accuracy of Base Calls and Error Predictions for GS 20 DNA Sequence Data
 Improving the Accuracy of Base Calls and Error Predictions for GS 20 DNA Sequence Data Justin S. Hogg Department of Computational Biology University of Pittsburgh Pittsburgh, PA 15213 jsh32@pitt.edu Abstract
Improving the Accuracy of Base Calls and Error Predictions for GS 20 DNA Sequence Data Justin S. Hogg Department of Computational Biology University of Pittsburgh Pittsburgh, PA 15213 jsh32@pitt.edu Abstract
General aspects of genome-wide association studies
 General aspects of genome-wide association studies Abstract number 20201 Session 04 Correctly reporting statistical genetics results in the genomic era Pekka Uimari University of Helsinki Dept. of Agricultural
General aspects of genome-wide association studies Abstract number 20201 Session 04 Correctly reporting statistical genetics results in the genomic era Pekka Uimari University of Helsinki Dept. of Agricultural
Sequencing technologies. Jose Blanca COMAV institute bioinf.comav.upv.es
 Sequencing technologies Jose Blanca COMAV institute bioinf.comav.upv.es Outline Sequencing technologies: Sanger 2nd generation sequencing: 3er generation sequencing: 454 Illumina SOLiD Ion Torrent PacBio
Sequencing technologies Jose Blanca COMAV institute bioinf.comav.upv.es Outline Sequencing technologies: Sanger 2nd generation sequencing: 3er generation sequencing: 454 Illumina SOLiD Ion Torrent PacBio
Complementary Technologies for Precision Genetic Analysis
 Complementary NGS, CGH and Workflow Featured Publication Zhu, J. et al. Duplication of C7orf58, WNT16 and FAM3C in an obese female with a t(7;22)(q32.1;q11.2) chromosomal translocation and clinical features
Complementary NGS, CGH and Workflow Featured Publication Zhu, J. et al. Duplication of C7orf58, WNT16 and FAM3C in an obese female with a t(7;22)(q32.1;q11.2) chromosomal translocation and clinical features
Tangram: a comprehensive toolbox for mobile element insertion detection
 Wu et al. BMC Genomics 2014, 15:795 METHODOLOGY ARTICLE Open Access Tangram: a comprehensive toolbox for mobile element insertion detection Jiantao Wu 1,Wan-PingLee 1, Alistair Ward 1, Jerilyn A Walker
Wu et al. BMC Genomics 2014, 15:795 METHODOLOGY ARTICLE Open Access Tangram: a comprehensive toolbox for mobile element insertion detection Jiantao Wu 1,Wan-PingLee 1, Alistair Ward 1, Jerilyn A Walker
LAB ACTIVITY ONE POPULATION GENETICS AND EVOLUTION 2017
 OVERVIEW In this lab you will: 1. learn about the Hardy-Weinberg law of genetic equilibrium, and 2. study the relationship between evolution and changes in allele frequency by using your class to represent
OVERVIEW In this lab you will: 1. learn about the Hardy-Weinberg law of genetic equilibrium, and 2. study the relationship between evolution and changes in allele frequency by using your class to represent
Illumina (Solexa) Throughput: 4 Tbp in one run (5 days) Cheapest sequencing technology. Mismatch errors dominate. Cost: ~$1000 per human genme
 Illumina (Solexa) Current market leader Based on sequencing by synthesis Current read length 100-150bp Paired-end easy, longer matepairs harder Error ~0.1% Mismatch errors dominate Throughput: 4 Tbp in
Illumina (Solexa) Current market leader Based on sequencing by synthesis Current read length 100-150bp Paired-end easy, longer matepairs harder Error ~0.1% Mismatch errors dominate Throughput: 4 Tbp in
Oral Cleft Targeted Sequencing Project
 Oral Cleft Targeted Sequencing Project Oral Cleft Group January, 2013 Contents I Quality Control 3 1 Summary of Multi-Family vcf File, Jan. 11, 2013 3 2 Analysis Group Quality Control (Proposed Protocol)
Oral Cleft Targeted Sequencing Project Oral Cleft Group January, 2013 Contents I Quality Control 3 1 Summary of Multi-Family vcf File, Jan. 11, 2013 3 2 Analysis Group Quality Control (Proposed Protocol)
Lecture 8: Sequencing and SNP. Sept 15, 2006
 Lecture 8: Sequencing and SNP Sept 15, 2006 Announcements Random questioning during literature discussion sessions starts next week for real! Schedule changes Moved QTL lecture up Removed landscape genetics
Lecture 8: Sequencing and SNP Sept 15, 2006 Announcements Random questioning during literature discussion sessions starts next week for real! Schedule changes Moved QTL lecture up Removed landscape genetics
Measuring transcriptomes with RNA-Seq. BMI/CS 776 Spring 2016 Anthony Gitter
 Measuring transcriptomes with RNA-Seq BMI/CS 776 www.biostat.wisc.edu/bmi776/ Spring 2016 Anthony Gitter gitter@biostat.wisc.edu Overview RNA-Seq technology The RNA-Seq quantification problem Generative
Measuring transcriptomes with RNA-Seq BMI/CS 776 www.biostat.wisc.edu/bmi776/ Spring 2016 Anthony Gitter gitter@biostat.wisc.edu Overview RNA-Seq technology The RNA-Seq quantification problem Generative
Random Allelic Variation
 Random Allelic Variation AKA Genetic Drift Genetic Drift a non-adaptive mechanism of evolution (therefore, a theory of evolution) that sometimes operates simultaneously with others, such as natural selection
Random Allelic Variation AKA Genetic Drift Genetic Drift a non-adaptive mechanism of evolution (therefore, a theory of evolution) that sometimes operates simultaneously with others, such as natural selection
User Bulletin. Veriti 96-Well Thermal Cycler AmpFlSTR Kit Validation. Overview
 User Bulletin Veriti 96-Well Thermal Cycler AmpFlSTR Kit Validation June 2009 SUBJECT: AmpFlSTR PCR Amplification Kit validation on the Veriti 96-Well Thermal Cycler with 0.2 ml sample block format In
User Bulletin Veriti 96-Well Thermal Cycler AmpFlSTR Kit Validation June 2009 SUBJECT: AmpFlSTR PCR Amplification Kit validation on the Veriti 96-Well Thermal Cycler with 0.2 ml sample block format In
7-1. Read this exercise before you come to the laboratory. Review the lecture notes from October 15 (Hardy-Weinberg Equilibrium)
 7-1 Biology 1001 Lab 7: POPULATION GENETICS PREPARTION Read this exercise before you come to the laboratory. Review the lecture notes from October 15 (Hardy-Weinberg Equilibrium) OBECTIVES At the end of
7-1 Biology 1001 Lab 7: POPULATION GENETICS PREPARTION Read this exercise before you come to the laboratory. Review the lecture notes from October 15 (Hardy-Weinberg Equilibrium) OBECTIVES At the end of
Whole genome sequencing in drug discovery research: a one fits all solution?
 Whole genome sequencing in drug discovery research: a one fits all solution? Marc Sultan, September 24th, 2015 Biomarker Development, Translational Medicine, Novartis On behalf of the BMD WGS pilot team:
Whole genome sequencing in drug discovery research: a one fits all solution? Marc Sultan, September 24th, 2015 Biomarker Development, Translational Medicine, Novartis On behalf of the BMD WGS pilot team:
Data Mining and Applications in Genomics
 Data Mining and Applications in Genomics Lecture Notes in Electrical Engineering Volume 25 For other titles published in this series, go to www.springer.com/series/7818 Sio-Iong Ao Data Mining and Applications
Data Mining and Applications in Genomics Lecture Notes in Electrical Engineering Volume 25 For other titles published in this series, go to www.springer.com/series/7818 Sio-Iong Ao Data Mining and Applications
Using the Trio Workflow in Partek Genomics Suite v6.6
 Using the Trio Workflow in Partek Genomics Suite v6.6 This user guide will illustrate the use of the Trio/Duo workflow in Partek Genomics Suite (PGS) and discuss the basic functions available within the
Using the Trio Workflow in Partek Genomics Suite v6.6 This user guide will illustrate the use of the Trio/Duo workflow in Partek Genomics Suite (PGS) and discuss the basic functions available within the
Molecular Markers CRITFC Genetics Workshop December 9, 2014
 Molecular Markers CRITFC Genetics Workshop December 9, 2014 Molecular Markers Tools that allow us to collect information about an individual, a population, or a species Application in fisheries mating
Molecular Markers CRITFC Genetics Workshop December 9, 2014 Molecular Markers Tools that allow us to collect information about an individual, a population, or a species Application in fisheries mating
The effect of RAD allele dropout on the estimation of genetic variation within and between populations
 Molecular Ecology (2013) 22, 3165 3178 doi: 10.1111/mec.12089 The effect of RAD allele dropout on the estimation of genetic variation within and between populations MATHIEU GAUTIER,* KARIM GHARBI, TIMOTHEE
Molecular Ecology (2013) 22, 3165 3178 doi: 10.1111/mec.12089 The effect of RAD allele dropout on the estimation of genetic variation within and between populations MATHIEU GAUTIER,* KARIM GHARBI, TIMOTHEE
A GENOTYPE CALLING ALGORITHM FOR AFFYMETRIX SNP ARRAYS
 Bioinformatics Advance Access published November 2, 2005 The Author (2005). Published by Oxford University Press. All rights reserved. For Permissions, please email: journals.permissions@oxfordjournals.org
Bioinformatics Advance Access published November 2, 2005 The Author (2005). Published by Oxford University Press. All rights reserved. For Permissions, please email: journals.permissions@oxfordjournals.org
An introductory overview of the current state of statistical genetics
 An introductory overview of the current state of statistical genetics p. 1/9 An introductory overview of the current state of statistical genetics Cavan Reilly Division of Biostatistics, University of
An introductory overview of the current state of statistical genetics p. 1/9 An introductory overview of the current state of statistical genetics Cavan Reilly Division of Biostatistics, University of
APPLICATION INFORMATION
 A-1932A APPLICATION INFORMATION Genetic Analysis: CEQ Series AUTOMATED BINNING PROCESS FOR THE GENERATION OF LOCUS TAGS Heather Gull, Dana Campbell, and Mark Dobbs Beckman Coulter, Inc. Short tandem repeats
A-1932A APPLICATION INFORMATION Genetic Analysis: CEQ Series AUTOMATED BINNING PROCESS FOR THE GENERATION OF LOCUS TAGS Heather Gull, Dana Campbell, and Mark Dobbs Beckman Coulter, Inc. Short tandem repeats
less sensitive than RNA-seq but more robust analysis pipelines expensive but quantitiatve standard but typically not high throughput
 Chapter 11: Gene Expression The availability of an annotated genome sequence enables massively parallel analysis of gene expression. The expression of all genes in an organism can be measured in one experiment.
Chapter 11: Gene Expression The availability of an annotated genome sequence enables massively parallel analysis of gene expression. The expression of all genes in an organism can be measured in one experiment.
Bayesian Variable Selection and Data Integration for Biological Regulatory Networks
 Bayesian Variable Selection and Data Integration for Biological Regulatory Networks Shane T. Jensen Department of Statistics The Wharton School, University of Pennsylvania stjensen@wharton.upenn.edu Gary
Bayesian Variable Selection and Data Integration for Biological Regulatory Networks Shane T. Jensen Department of Statistics The Wharton School, University of Pennsylvania stjensen@wharton.upenn.edu Gary
Cornell Probability Summer School 2006
 Colon Cancer Cornell Probability Summer School 2006 Simon Tavaré Lecture 5 Stem Cells Potten and Loeffler (1990): A small population of relatively undifferentiated, proliferative cells that maintain their
Colon Cancer Cornell Probability Summer School 2006 Simon Tavaré Lecture 5 Stem Cells Potten and Loeffler (1990): A small population of relatively undifferentiated, proliferative cells that maintain their
MUTATION RATES. Genetica per Scienze Naturali a.a prof S. Presciuttini
 MUTATION RATES Questo documento è pubblicato sotto licenza Creative Commons Attribuzione Non commerciale Condividi allo stesso modo http://creativecommons.org/licenses/by-nc-sa/2.5/deed.it 1. Mutation
MUTATION RATES Questo documento è pubblicato sotto licenza Creative Commons Attribuzione Non commerciale Condividi allo stesso modo http://creativecommons.org/licenses/by-nc-sa/2.5/deed.it 1. Mutation
Accelerate High Throughput Analysis for Genome Sequencing with GPU
 Accelerate High Throughput Analysis for Genome Sequencing with GPU ATIP - A*CRC Workshop on Accelerator Technologies in High Performance Computing May 7-10, 2012 Singapore BingQiang WANG, Head of Scalable
Accelerate High Throughput Analysis for Genome Sequencing with GPU ATIP - A*CRC Workshop on Accelerator Technologies in High Performance Computing May 7-10, 2012 Singapore BingQiang WANG, Head of Scalable
Linking Genetic Variation to Important Phenotypes
 Linking Genetic Variation to Important Phenotypes BMI/CS 776 www.biostat.wisc.edu/bmi776/ Spring 2018 Anthony Gitter gitter@biostat.wisc.edu These slides, excluding third-party material, are licensed under
Linking Genetic Variation to Important Phenotypes BMI/CS 776 www.biostat.wisc.edu/bmi776/ Spring 2018 Anthony Gitter gitter@biostat.wisc.edu These slides, excluding third-party material, are licensed under
LS50B Problem Set #7
 LS50B Problem Set #7 Due Friday, March 25, 2016 at 5 PM Problem 1: Genetics warm up Answer the following questions about core concepts that will appear in more detail on the rest of the Pset. 1. For a
LS50B Problem Set #7 Due Friday, March 25, 2016 at 5 PM Problem 1: Genetics warm up Answer the following questions about core concepts that will appear in more detail on the rest of the Pset. 1. For a
Mapping strategies for sequence reads
 Mapping strategies for sequence reads Ernest Turro University of Cambridge 21 Oct 2013 Quantification A basic aim in genomics is working out the contents of a biological sample. 1. What distinct elements
Mapping strategies for sequence reads Ernest Turro University of Cambridge 21 Oct 2013 Quantification A basic aim in genomics is working out the contents of a biological sample. 1. What distinct elements
Sequencing technologies. Jose Blanca COMAV institute bioinf.comav.upv.es
 Sequencing technologies Jose Blanca COMAV institute bioinf.comav.upv.es Outline Sequencing technologies: Sanger 2nd generation sequencing: 3er generation sequencing: 454 Illumina SOLiD Ion Torrent PacBio
Sequencing technologies Jose Blanca COMAV institute bioinf.comav.upv.es Outline Sequencing technologies: Sanger 2nd generation sequencing: 3er generation sequencing: 454 Illumina SOLiD Ion Torrent PacBio
PERSPECTIVES. A gene-centric approach to genome-wide association studies
 PERSPECTIVES O P I N I O N A gene-centric approach to genome-wide association studies Eric Jorgenson and John S. Witte Abstract Genic variants are more likely to alter gene function and affect disease
PERSPECTIVES O P I N I O N A gene-centric approach to genome-wide association studies Eric Jorgenson and John S. Witte Abstract Genic variants are more likely to alter gene function and affect disease
KNN-MDR: a learning approach for improving interactions mapping performances in genome wide association studies
 Abo Alchamlat and Farnir BMC Bioinformatics (2017) 18:184 DOI 10.1186/s12859-017-1599-7 METHODOLOGY ARTICLE Open Access KNN-MDR: a learning approach for improving interactions mapping performances in genome
Abo Alchamlat and Farnir BMC Bioinformatics (2017) 18:184 DOI 10.1186/s12859-017-1599-7 METHODOLOGY ARTICLE Open Access KNN-MDR: a learning approach for improving interactions mapping performances in genome
A Bayesian Graphical Model for Genome-wide Association Studies (GWAS)
 A Bayesian Graphical Model for Genome-wide Association Studies (GWAS) Laurent Briollais 1,2, Adrian Dobra 3, Jinnan Liu 1, Hilmi Ozcelik 1 and Hélène Massam 4 Prosserman Centre for Health Research, Samuel
A Bayesian Graphical Model for Genome-wide Association Studies (GWAS) Laurent Briollais 1,2, Adrian Dobra 3, Jinnan Liu 1, Hilmi Ozcelik 1 and Hélène Massam 4 Prosserman Centre for Health Research, Samuel
BTRY 7210: Topics in Quantitative Genomics and Genetics
 BTRY 7210: Topics in Quantitative Genomics and Genetics Jason Mezey Biological Statistics and Computational Biology (BSCB) Department of Genetic Medicine jgm45@cornell.edu January 29, 2015 Why you re here
BTRY 7210: Topics in Quantitative Genomics and Genetics Jason Mezey Biological Statistics and Computational Biology (BSCB) Department of Genetic Medicine jgm45@cornell.edu January 29, 2015 Why you re here
Detecting selection on nucleotide polymorphisms
 Detecting selection on nucleotide polymorphisms Introduction At this point, we ve refined the neutral theory quite a bit. Our understanding of how molecules evolve now recognizes that some substitutions
Detecting selection on nucleotide polymorphisms Introduction At this point, we ve refined the neutral theory quite a bit. Our understanding of how molecules evolve now recognizes that some substitutions
CS 5984: Application of Basic Clustering Algorithms to Find Expression Modules in Cancer
 CS 5984: Application of Basic Clustering Algorithms to Find Expression Modules in Cancer T. M. Murali January 31, 2006 Innovative Application of Hierarchical Clustering A module map showing conditional
CS 5984: Application of Basic Clustering Algorithms to Find Expression Modules in Cancer T. M. Murali January 31, 2006 Innovative Application of Hierarchical Clustering A module map showing conditional
An introduction to genetics and molecular biology
 An introduction to genetics and molecular biology Cavan Reilly September 5, 2017 Table of contents Introduction to biology Some molecular biology Gene expression Mendelian genetics Some more molecular
An introduction to genetics and molecular biology Cavan Reilly September 5, 2017 Table of contents Introduction to biology Some molecular biology Gene expression Mendelian genetics Some more molecular
Alignment. J Fass UCD Genome Center Bioinformatics Core Wednesday December 17, 2014
 Alignment J Fass UCD Genome Center Bioinformatics Core Wednesday December 17, 2014 From reads to molecules Why align? Individual A Individual B ATGATAGCATCGTCGGGTGTCTGCTCAATAATAGTGCCGTATCATGCTGGTGTTATAATCGCCGCATGACATGATCAATGG
Alignment J Fass UCD Genome Center Bioinformatics Core Wednesday December 17, 2014 From reads to molecules Why align? Individual A Individual B ATGATAGCATCGTCGGGTGTCTGCTCAATAATAGTGCCGTATCATGCTGGTGTTATAATCGCCGCATGACATGATCAATGG
Chapter 14: Genes in Action
 Chapter 14: Genes in Action Section 1: Mutation and Genetic Change Mutation: Nondisjuction: a failure of homologous chromosomes to separate during meiosis I or the failure of sister chromatids to separate
Chapter 14: Genes in Action Section 1: Mutation and Genetic Change Mutation: Nondisjuction: a failure of homologous chromosomes to separate during meiosis I or the failure of sister chromatids to separate
Exploring the Genetic Basis of Congenital Heart Defects
 Exploring the Genetic Basis of Congenital Heart Defects Sanjay Siddhanti Jordan Hannel Vineeth Gangaram szsiddh@stanford.edu jfhannel@stanford.edu vineethg@stanford.edu 1 Introduction The Human Genome
Exploring the Genetic Basis of Congenital Heart Defects Sanjay Siddhanti Jordan Hannel Vineeth Gangaram szsiddh@stanford.edu jfhannel@stanford.edu vineethg@stanford.edu 1 Introduction The Human Genome
Harnessing the power of RADseq for ecological and evolutionary genomics
 STUDY DESIGNS Harnessing the power of RADseq for ecological and evolutionary genomics Kimberly R. Andrews 1, Jeffrey M. Good 2, Michael R. Miller 3, Gordon Luikart 4 and Paul A. Hohenlohe 5 Abstract High-throughput
STUDY DESIGNS Harnessing the power of RADseq for ecological and evolutionary genomics Kimberly R. Andrews 1, Jeffrey M. Good 2, Michael R. Miller 3, Gordon Luikart 4 and Paul A. Hohenlohe 5 Abstract High-throughput
Fine-scale mapping of meiotic recombination in Asians
 Fine-scale mapping of meiotic recombination in Asians Supplementary notes X chromosome hidden Markov models Concordance analysis Alternative simulations Algorithm comparison Mongolian exome sequencing
Fine-scale mapping of meiotic recombination in Asians Supplementary notes X chromosome hidden Markov models Concordance analysis Alternative simulations Algorithm comparison Mongolian exome sequencing
Quality Measures for CytoChip Microarrays
 Quality Measures for CytoChip Microarrays How to evaluate CytoChip Oligo data quality in BlueFuse Multi software. Data quality is one of the most important aspects of any microarray experiment. This technical
Quality Measures for CytoChip Microarrays How to evaluate CytoChip Oligo data quality in BlueFuse Multi software. Data quality is one of the most important aspects of any microarray experiment. This technical
RNA-Sequencing analysis
 RNA-Sequencing analysis Markus Kreuz 25. 04. 2012 Institut für Medizinische Informatik, Statistik und Epidemiologie Content: Biological background Overview transcriptomics RNA-Seq RNA-Seq technology Challenges
RNA-Sequencing analysis Markus Kreuz 25. 04. 2012 Institut für Medizinische Informatik, Statistik und Epidemiologie Content: Biological background Overview transcriptomics RNA-Seq RNA-Seq technology Challenges
Linkage Disequilibrium. Adele Crane & Angela Taravella
 Linkage Disequilibrium Adele Crane & Angela Taravella Overview Introduction to linkage disequilibrium (LD) Measuring LD Genetic & demographic factors shaping LD Model predictions and expected LD decay
Linkage Disequilibrium Adele Crane & Angela Taravella Overview Introduction to linkage disequilibrium (LD) Measuring LD Genetic & demographic factors shaping LD Model predictions and expected LD decay
Measuring transcriptomes with RNA-Seq
 Measuring transcriptomes with RNA-Seq BMI/CS 776 www.biostat.wisc.edu/bmi776/ Spring 2017 Anthony Gitter gitter@biostat.wisc.edu These slides, excluding third-party material, are licensed under CC BY-NC
Measuring transcriptomes with RNA-Seq BMI/CS 776 www.biostat.wisc.edu/bmi776/ Spring 2017 Anthony Gitter gitter@biostat.wisc.edu These slides, excluding third-party material, are licensed under CC BY-NC
Addresses 1 Department of Computer Science and Engineering, 2 School of Life Sciences, The Chinese University of Hong
 Title page Title OMSV enables accurate and comprehensive identification of large structural variations from nanochannel-based single-molecule optical maps Authors Le Li 1, (lil@cse.cuhk.edu.hk), Alden
Title page Title OMSV enables accurate and comprehensive identification of large structural variations from nanochannel-based single-molecule optical maps Authors Le Li 1, (lil@cse.cuhk.edu.hk), Alden
Sequence Assembly and Alignment. Jim Noonan Department of Genetics
 Sequence Assembly and Alignment Jim Noonan Department of Genetics james.noonan@yale.edu www.yale.edu/noonanlab The assembly problem >>10 9 sequencing reads 36 bp - 1 kb 3 Gb Outline Basic concepts in genome
Sequence Assembly and Alignment Jim Noonan Department of Genetics james.noonan@yale.edu www.yale.edu/noonanlab The assembly problem >>10 9 sequencing reads 36 bp - 1 kb 3 Gb Outline Basic concepts in genome
JS 190- Population Genetics- Assessing the Strength of the Evidence Pre class activities
 JS 190- Population Genetics- Assessing the Strength of the Evidence I. Pre class activities a. Quiz then Review Assignments and Schedule II. Learning Objectives a. Overview of Validation Developmental
JS 190- Population Genetics- Assessing the Strength of the Evidence I. Pre class activities a. Quiz then Review Assignments and Schedule II. Learning Objectives a. Overview of Validation Developmental
Alignment methods. Martijn Vermaat Department of Human Genetics Center for Human and Clinical Genetics
 Alignment methods Martijn Vermaat Department of Human Genetics Center for Human and Clinical Genetics Alignment methods Sequence alignment Assembly vs alignment Alignment methods Common issues Platform
Alignment methods Martijn Vermaat Department of Human Genetics Center for Human and Clinical Genetics Alignment methods Sequence alignment Assembly vs alignment Alignment methods Common issues Platform
Evaluation of Genome wide SNP Haplotype Blocks for Human Identification Applications
 Ranajit Chakraborty, Ph.D. Evaluation of Genome wide SNP Haplotype Blocks for Human Identification Applications Overview Some brief remarks about SNPs Haploblock structure of SNPs in the human genome Criteria
Ranajit Chakraborty, Ph.D. Evaluation of Genome wide SNP Haplotype Blocks for Human Identification Applications Overview Some brief remarks about SNPs Haploblock structure of SNPs in the human genome Criteria
Linkage & Genetic Mapping in Eukaryotes. Ch. 6
 Linkage & Genetic Mapping in Eukaryotes Ch. 6 1 LINKAGE AND CROSSING OVER! In eukaryotic species, each linear chromosome contains a long piece of DNA A typical chromosome contains many hundred or even
Linkage & Genetic Mapping in Eukaryotes Ch. 6 1 LINKAGE AND CROSSING OVER! In eukaryotic species, each linear chromosome contains a long piece of DNA A typical chromosome contains many hundred or even
Introduction to Next Generation Sequencing (NGS) Andrew Parrish Exeter, 2 nd November 2017
 Introduction to Next Generation Sequencing (NGS) Andrew Parrish Exeter, 2 nd November 2017 Topics to cover today What is Next Generation Sequencing (NGS)? Why do we need NGS? Common approaches to NGS NGS
Introduction to Next Generation Sequencing (NGS) Andrew Parrish Exeter, 2 nd November 2017 Topics to cover today What is Next Generation Sequencing (NGS)? Why do we need NGS? Common approaches to NGS NGS
A shotgun introduction to sequence assembly (with Velvet) MCB Brem, Eisen and Pachter
 A shotgun introduction to sequence assembly (with Velvet) MCB 247 - Brem, Eisen and Pachter Hot off the press January 27, 2009 06:00 AM Eastern Time llumina Launches Suite of Next-Generation Sequencing
A shotgun introduction to sequence assembly (with Velvet) MCB 247 - Brem, Eisen and Pachter Hot off the press January 27, 2009 06:00 AM Eastern Time llumina Launches Suite of Next-Generation Sequencing
