Safety, Efficacy & Microbiological Consideration
|
|
|
- Beatrix McKenzie
- 6 years ago
- Views:
Transcription
1 Safety, Efficacy & Microbiological Consideration Endoscope Cleaning and Disinfection System (With Optional Cleaning Cycle that Eliminates Manual Cleaning) 1
2 Medivators is a registered trademark of Minntech Corporation. Medivators ADVANTAGE is a trademark of Minntech Corporation. Olympus is a registered trademark of Olympus Corporation. Pentax is a registered trademark of Pentax Precision Instrument Corporation. Fujinon is a registered trademark of Fujinon Corporation ADVANTAGE PLUS , Minntech Corporation All rights reserved. This publication is protected by copyright. Copying, disclosure to others, or the use of this publication is prohibited without the express written consent of Minntech Corporation. Minntech reserves the right to make changes in the specifications shown herein without notice or obligation. Contact your Medivators representative or Medivators customer service for more information. 2
3 INTENDED USE The Medivators ADVANTAGE Plus Endoscope Reprocessing System is cleared by the Food and Drug Administration (FDA) to test, clean, disinfect, and rinse endoscopes, such as fiber-optic and video endoscopes, between patient uses. The ADVANTAGE Plus system is indicated to provide high level disinfection of heat sensitive semi-critical endoscopes and related accessories. Manual cleaning of endoscopes is not required prior to placement in the ADVANTAGE Plus system. The scopes must be precleaned immediately after use. The ADVANTAGE Plus Endoscope Reprocessing System uses Rapicide PA High Level Disinfectant to provide high level disinfection of endoscopes when used according to the direction for use. The system uses Intercept Detergent in the cleaning cycle at a concentration of 0.5%. PRODUCT DESCRIPTION The Advantage Plus automated endoscope reprocessor tests, cleans, and high-level disinfects endoscopes and related accessories between uses. This advanced system performs automated cleaning of endoscopes, a feature which eliminates the need for manual cleaning. Built-in continuous leak testing and channel blockage monitoring provide additional levels of safety to ensure consistent results. Results are then recorded in the sophisticated data management storage system. The Advantage Plus has two large, easy loading independent basins, which perform asynchronous reprocessing and accommodate most endoscope models. Endoscopes are connected to the ADVANTAGE Hook-up Connector Block*, which contains individual connectors for each endoscope channel. These validated hook-ups test for channel connectivity blockage and outer sheath leaks and provide perfusion of internal endoscope channels. Upon successful completion of the connectivity, blockage and leak tests, the system proceeds to rinse the instrument and start the cleaning and disinfection cycle. The Advantage Plus utilizes Intercept Detergent and the high level disinfectant Rapicide PA, a single-use peracetic acid. Both Intercept and Rapicide PA have been validated for use in the Advantage Plus system. Endoscopes will not require manual cleaning if the wash cycle utilizing Intercept Detergent is performed. For reprocessing cycles that will not include the wash cycle, manual cleaning of the endoscope is required. The ADVANTAGE Plus also has the ability to perform a 70% isopropyl alcohol flush with subsequent air purge to assist in drying of endoscope channels. *It is important to use the correct connections (hookups) for connecting the scope to the machine. A list of endoscope and corresponding hookups can be found on the online interactive hookup guide: 3
4 PRODUCT FEATURES Asynchronous operation of two large independent basins A dedicated personal computer (PC) for cycle recording, powerful quality assurance reporting, easy backups, networking availability, and remote diagnostics Contains second hard drive for easy data backups Hook-up blocks dedicated to a family of endoscopes to ensure correct connectivity and flow rates to meet manufacturers specifications Individual channel identification and blockage monitoring Continuous endoscope leak testing throughout entire reprocessing cycle Remote diagnostics for operator assistance and troubleshooting Uses single-shot, environmentally friendly Rapicide PA High Level Disinfectant FDA-cleared optional cleaning claim cycle that eliminates manual cleaning Easy-to-fill detergent and alcohol reservoirs 4
5 REPROCESSING CYCLE The Advantage Plus has an average reprocessing cycle time of 35 minutes, which includes leak testing, washing, disinfecting, rinsing and a final alcohol and air purge. The Advantage Plus utilizes an advanced software program that continuously monitors all system sensors. This highly advanced program was designed specifically for the Advantage Plus System and monitors each phase of the reprocessing cycle. Each overall cycle on the Advantage Plus consists of a combination of fourteen possible phases: Phase Phase Usage Details Number 1 Leak Test 2 Flush Rinse approximately 5 liters of water 3 Wash 3.5 liters water plus 10.8 ml Intercept detergent 4 Flush Rinse approximately 5 liters of water 5 Post Rinse 10 liters of water 6 Wash 3.5 liters water plus detergent 7 Flush rinse approximately 5 liters of water 8 Post Rinse 10 liters of water 9 Disinfect 10 liters of Rapicide PA 10 Flush Rinse 5 liters of water 11 Post Rinse 5 liters of water 12 Post Rinse 5 liters of water 13 Final Rinse 5 liters of water 14 Alcohol Purge/Vent ml per cycle depending on the scope channel configuration The minimum requirements for a cycle are 1, 2, 3, 4, 9, 10, 13, 14 which can be configured with any different combination of washes and rinses. 5
6 LEAK TESTING The Advantage Plus automated endoscope reprocessor performs leak testing during the first phase of the reprocessing cycle by pressurizing the endoscope. The pressure level is maintained throughout each following cycle until reprocessing is complete. If a gross leak is detected, the Advantage Plus will automatically stop the reprocessing cycle so no fluid enters the endoscope preventing further damage. If a small leak is detected in the endoscope but its sheath remains pressurized, no fluid invasion can occur so the Advantage Plus will finish the disinfection cycle. Therefore, the leak testing capability of the Advantage Plus System minimizes associated endoscope repair costs while providing the endoscope manufacturer with a high-level disinfected endoscope. CHANNEL MONITORING & BLOCKAGE DETECTION The Advantage Plus uses specially designed Hook-up Connector Blocks, which are specific for each endoscope make and model. Individual connections are made via the Connector Block tubing to the endoscope channels: air, water, suction, biopsy, etc. Through these connections, the advanced software system performs continuous monitoring of each channel connection and will alert the operator if a connection is not secure or if there is blockage. These quality assurance procedures provide consistency not offered with manual reprocessing. AUTOMATED CLEANING The FDA has cleared the Medivators Advantage Plus Endoscope Reprocessing System with a new cleaning cycle claim that eliminates the need for manual cleaning of endoscopes. Automated cleaning of endoscopes must be performed using Medivators Intercept Detergent in the high performance cleaning cycle. This automated cleaning cycle has been shown to be equivalent to manual cleaning, achieving results that meet the AAMI TIR-30 standard for residual soil. The endoscope cleaning phase for the ADVANTAGE Plus consists of four phases: 1. Phase 2 Flush phase. This phase flushes the endoscope channels with fresh water. The outside of the endoscope is also sprayed with fresh water using the spray arm. This is the initial flush of the endoscope. This phase also flushes the water up to temperature. This phase is also part of cycles programmed without the cleaning claim wash phase. 2. Phase 3 Detergent Wash phase. This phase dispenses detergent into the basin to a detergent concentration of 0.5%. The basin is filled with 3.5 liters of fresh water. The endoscope channels are rigorously flushed with the detergent solution. The exterior soaks in the 3.5 liter solution and is sprayed with the solution using the spray arm and the hydrodynamic spray tower. 3. Phase 4 Second Flush phase. This phase is used to remove the bulk of the detergent solution from the basin and the endoscope. This phase flushes the endoscope channels with fresh water. The outside of the endoscope is also sprayed with fresh water using the spray arm and the hydrodynamic spray tower. The drain is open and the water level is not maintained in the basin. 4. Phase 5 Post rinse. This rinse fills the basin with 10 liters of water. The endoscope channels are flushed while the exterior soaks in 10 liters of water and the exterior is sprayed with using the spray arm and the hydrodynamic spray tower. Air is then blown through the channels to remove residual rinse water prior to the introduction of disinfectant. This ensures surface contact with disinfectant and minimizes dilution. 6
7 Cleaning Validation Validation of the automated cleaning by the Advantage Plus was done both with Artificial Test Soils (ATS) and with clinically soiled endoscopes. The endoscopes chosen were representative of the most complex scopes in three different classes: bronchoscopes, gastroscopes, and colonoscopes. The scopes in the simulated use testing were soiled in the channel lumens and on the surface with ATS, to the worst case limits reported (Alfa, 1999). The soils used were the Alfa test soil (Alfa, 2002) for the gastroscopes and colonoscopes, and a respiratory test soil (based on published reports of respiratory soil composition) for the bronchoscopes. The test markers followed were the total protein, total carbohydrate, and total hemoglobin. The total protein detection was done using the BCA assay (Smith et al., 1985), a standard method widely used in biochemistry and medicinal chemistry. The total carbohydrate detection was done using the anthrone/acid method (Morris, 1948), which produces a very stable color change and does not require prior hydrolysis of the polysaccharides. The hemoglobin quantitation was performed using cation exchange on a PolyCAT HPLC column, after cyano-derivatizing the hemoglobin. All of the methods were validated with the soils, including matrix validation. The quantitation for each analyte was reported in total (per scope) and also normalized to the total surface area that was soiled. The target criteria were taken from AAMI TIR-30. The goal of the cleaning phase was to achieve more than 90% reduction of protein, more than 90% reduction of carbohydrate, and a residue of <6.4 µg protein/cm 2, <1.8 µg carbohydrate/cm 2, and <2.2 µg hemoglobin/cm 2. All criteria were required to be met in all cases. A minimum of 5 repetitions were completed for each scope, in addition to 5 positive control (soiled but not cleaned, then recovered) and 5 negative control (not soiled, recovered) repetitions. The simulated studies were performed on scopes from three different manufacturers; in each case, the scopes used were representative of the most complicated and difficult to clean instruments in their class. The scopes were soiled in all lumens and on the surface, and dried for 1 hour. The total amount of soiling was determined both by dry weight difference and by the use of positive controls (where the soil was immediately recovered without being cleaned). Negative controls were performed in each case by performing soil recoveries on the scopes prior to the soiling/cleaning tests (i.e., on unsoiled scopes). This demonstrated that the scopes used did not have residual soil from previous use. Soiled/cleaned scopes were processed on the cleaning cycle of the Advantage Plus, using 0.3% Intercept cleaner, which is worst-case (the normal cycle uses 0.6% Intercept); this worst-case condition insures that variation in dilution is accounted for. After cleaning, the residual soil was recovered by brushing and flushing the channels and scrubbing the surface; all cleaning brushes and scrubs were recovered as well, using sonication and extraction. The recovery method for all cases (positive controls, negative controls, and soiled/cleaned scopes) was validated by demonstrating that >90% of the soil was recovered, and that successive soil recoveries of the same scope did not produce significant residuals of the analytes. The recovered soil was analyzed for the markers, and the data were totaled for each scope, and also normalized to the total surface area inoculated. The data, shown in tables 1,2, and 3, show that the target residual criteria were met in all cases. 7
8 Table 1. Residual soil recovered after cleaning, Olympus scopes Residuals Endoscope Channel protein carbohydrate hemoglobin Olympus Bronchoscope B/S BF-P30 Out Whole Olympus Gastroscope B/S GIF 2T-160 B Air water Elevator Out Whole Olympus Colonoscope B/S XCF-H160AYL Air water Elevator Out Whole
9 Table 1. Residual soil after cleaning, Pentax scopes. Residuals Endoscope Channel protein carbohydrate hemoglobin Pentax B/S Bronchoscope Valve VB 1530 Out Whole Pentax B/S Gastroscope Air EG 3830K water Elevator Out Whole Pentax B/S Colonoscope Air EC 3830FK water Elevator Out Whole
10 Table 2. Residual soil after cleaning, Fuji scopes. Residuals Endoscope Channel protein carbohydrate hemoglobin Fuji Bronchoscope B/S EB-470S Valve Out Whole Fuji Gastroscope B/S ED-530XT Air water Out Whole Fuji Colonoscope B/S EC-450LS5 Air water Elevator Out Whole The cleaning, indicated by the markers followed, achieved >99.9% removal of organic soil, and the average of residual soil was well below the target levels identified in AAMI TIR-30. This demonstrates that the Advantage Plus system was effective in removing the organic load from flexible endoscopes. 10
11 Clinically Soiled Scopes In addition to the simulated soiling/recovery, clinically soiled endoscopes were also cleaned, in order to evaluate the cleaning efficacy on actual patient soil, and also to demonstrate that the automated cycle achieves that same level of soil removal/residual as manual cleaning. Scopes were obtained from 2 different endoscopy clinics, representing multiple manufacturers, models, and classes (bronchoscopes, gastroscopes, and colonoscopes). The manual cleaning was performed by the clinical staff, in accordance with their normal cleaning methods. The scopes for automated cleaning were picked up from the clinics immediately after use and brought to the laboratory for processing on the Advantage Plus cleaning cycle (cleaning cycle only). Both the manually cleaned scopes and the scopes cleaned on the automated cycle of the Advantage Plus were harvested for residual soil by the same method that was validated and used in the simulated soiling. The same markers for residual soil (protein, carbohydrate, and hemoglobin) were followed. Three colonoscopes of each model and type were performed by each method (automated and manual). The results are shown in table 4. Both of the cleaning processes met the criteria set in AAMI TIR-30, for all scopes tested. In all cases the automated cycle of the Advantage Plus performed comparably to manual cleaning. Table 3. Cleaning of clinically soiled scopes, by automated and by manual cleaning. Scope Channels Protein residual, Carbohydrate residual, Hemoglobin residual, Automated Clean Manual Clean Automated Clean Manual Clean Automated Clean Bron. Biopsy BF-P40 Out Manual Clean Whole Gastro B/S GIF-H180 Air Water Out Whole Colon B/S PCF- H180AL Air Water Elevator Out Whole All of the cleaning processes met the criteria set in AAMI TIR-30. These data show that the automated cleaning cycle on the Advantage plus performs as effectively as manual cleaning in removing the organic load of even the most complicated endoscopes. 11
12 HIGH LEVEL DISINFECTION High level disinfection is achieved with Rapicide PA High Level Disinfectant, a single-use environmentally friendly peracetic acid. Rapicide PA enters the basin and is perfused through internal endoscope channels via hookups. Rapicide PA Solutions A and B are combined to produce a single use-solution for High-Level Disinfection (HLD) of endoscopes with peracetic acid (PAA) as the biocidal active agent. Rapicide PA also contains corrosion inhibitors and water conditioning agents. The Advantage Plus System is constructed with high-grade components compatible with peracetic acid. The characteristics of the single use-solution are: Active Ingredient: Peracetic Acid PAA Concentration: ppm Minimum Recommended Concentration (MRC): 850 ppm Reuse Period: Single use High-Level Disinfection Claim: 5 30ºC (Use conditions may vary by country. Please contact your Minntech distributor for information) Dilution Required: 1:1 to 48 water Activation Required: No Corrosion Inhibitors: Yes Rapicide PA has been shown to be effective in killing vegetative bacteria, M. tuberculosis, fungi and viruses, MRSA, VRE C. difficile and other spores of Bacillus and Clostridium species (36). Test strips are used to determine whether an effective concentration of active ingredient is present following reprocessing. TESTS Antimicrobial Activity Results Sporicidal Clostridium sporogenes spores Total Kill Sporicidal Bacillus subtilis spores Total Kill Tuberculocidal Mycobacterium bovis (BCG) Total Kill Virucidal Polio virus type 2 Complete Inactivation Virucidal Herpes Simplex virus type 1 Complete Inactivation Virucidal Human Immunodeficiency Virus type 1 Fungicidal Trichophyton mentagrophytes Total Kill Bactericidal Pseudomonas aeruginosa Total Kill Bactericidal Staphylococcus aureus Total Kill Bactericidal Salmonella enterica Total Kill Complete Inactivation Simulated Use Mycobacterium terrae >6 log Reduction In-Use GI Tract Normal Flora Total Kill 12
13 AUTOMATED ALCOHOL FLUSH & AIR PURGE After the high-level disinfection cycle is completed, the endoscope is given a final rinse and purged with alcohol and air to assist in drying all channels. This automated alcohol flush and air purge is an important step to sustaining a high-level disinfected endoscope. Table 3. Total Residues Extracted from Colonoscopes after ADVANTAGE Plus Processing Disinfectant Safe residual level (mg) Range of total extracted residuals from 3 endoscopes (mg) Required Endoscope Rinses Rapicide PA 250 mg 0 2 Electrical Safety Testing All electrical safety aspects of the ADVANTAGE Plus have been tested by an outside testing service (Intertek Testing Services) for compliance with UL Standard for Safety Requirements for Electrical Equipment. It has also been tested for emissions that may affect other devices, and for immunity to the effects that other devices may have on the reprocessor. These test data meet the requirements of IEC Testing on file with shows the machine meets all required standards and will be safe under its labeled conditions. Reprocessor Self-Disinfection The ADVANTAGE Plus needs to be sanitized at regular intervals to ensure that water pathways and filters do not become contaminated. Testing was performed on the ADVANTAGE Plus using the self-disinfection cycle with Rapicide PA. Concentrations of both disinfectants relative to the MRC in the water filter were determined in three replicate runs during the selfdisinfection hold period. Results in Table 4 demonstrate that Rapicide PA disinfectant concentrations are within the normal use concentration and well above the MRC during the disinfection hold period. Table 4. Disinfectant Concentrations During ADVANTAGE Plus Self-Disinfection Cycles Disinfectant MRC Range of baseline disinfectant Range during selfdisinfection Rapicide PA 850 ppm ppm ppm 13
14 Material Compatibility Material compatibility testing has been performed on all components of the ADVANTAGE Plus that will be contacted by the labeled disinfectants. Metals, ceramics, plastics, composites and elastomers were exposed to Rapicide PA at full strength at recommended use temperatures. Components were exposed for 325 hours to meet the equivalent of 1.5 years of field use. All materials had parameter changes less than the 5% criterion and are acceptable for use with disinfectants under labeled use conditions. Software Verification Laboratory studies were conducted to verify that the ADVANTAGE Plus will perform its specified functions. Each system and sensor has been studied, and the complete integrated system has been shown to perform in an effective manner. Software system parameter verification testing included: Control of machine draining Control of the pre-rinse and final rinse requirements Execution of the proper self-disinfection cycle Software detection of error conditions verification testing included: Compromised endoscope sheath (leak detection) Disconnected or improperly connected channels Endoscope channel blockage Reservoir temperatures above or below labeled parameters Open/ajar basin lids Low water or disinfectant level in basin Deviation from maximum/minimum cycle step times Disinfectant overflow sensor Disinfectant low level sensor Electrical power supply at incorrect cycle phase Disinfection contact times inadequate Unsatisfactory completion of self-disinfection phase 14
15 References AAMI TIR 30 (2003). A compendium of processes, materials, test methods, and acceptance criteria for cleaning reusable medical devices. Alfa, MJ, Degagne, P, Olson, N (1999), Worst-case soiling levels for patient-used flexible endoscopes before and after cleaning. Am J. Infection Control 27:5, Alfa, MJ (2002). Artificial Test Soil. US Patent 6,447,990. Morris, DL (1948). Quantitative determination of carbohydrates with Dreywood s anthrone reagent. Science 107:254-5 Smith, P.K. et al. (1985). Measurement of protein using bicinchonic acid. Anal. Biochem. 150:
16 Distributed in Canada by: CARSEN Medical Inc Amber Street Markham, ON L3R 3B
HIGH-LEVEL DISINFECTANT RAPICIDE. Safety, Efficacy, and Microbiological Considerations
 HIGH-LEVEL DISINFECTANT RAPICIDE PA Safety, Efficacy, and Microbiological Considerations 1 50096-959 Rev D 2011, MEDIVATORS, A Minntech Corporation Business Group All rights reserved. This publication
HIGH-LEVEL DISINFECTANT RAPICIDE PA Safety, Efficacy, and Microbiological Considerations 1 50096-959 Rev D 2011, MEDIVATORS, A Minntech Corporation Business Group All rights reserved. This publication
High-Level Disinfectant and Sterilant. Rapicide SAFETY, EFFICACY AND MICROBIOLOGICAL CONSIDERATIONS
 High-Level Disinfectant and Sterilant Rapicide SAFETY, EFFICACY AND MICROBIOLOGICAL CONSIDERATIONS Contents Product Description... 3 Intended Use... 3 Safety... 4 Storage, Use and Disposal... 4 Efficacy
High-Level Disinfectant and Sterilant Rapicide SAFETY, EFFICACY AND MICROBIOLOGICAL CONSIDERATIONS Contents Product Description... 3 Intended Use... 3 Safety... 4 Storage, Use and Disposal... 4 Efficacy
FREQUENTLY ASKED QUESTIONS FOR THE ADVANTAGE
 FREQUENTLY ASKED QUESTIONS FOR THE ADVANTAGE What is an Automated Endoscope Reprocessor? An Automated Endoscopes Reprocessor (AER) performs all the cycle steps (fresh water preflush, rinsing, disinfection,
FREQUENTLY ASKED QUESTIONS FOR THE ADVANTAGE What is an Automated Endoscope Reprocessor? An Automated Endoscopes Reprocessor (AER) performs all the cycle steps (fresh water preflush, rinsing, disinfection,
Rapicide High-Level Disinfectant and Sterilant. Safety, Efficacy, and Microbiological Considerations
 Rapicide High-Level Disinfectant and Sterilant Safety, Efficacy, and Microbiological Considerations CONTENTS Product Description.......... 1 Safety...................... 3 Efficacy Summary............
Rapicide High-Level Disinfectant and Sterilant Safety, Efficacy, and Microbiological Considerations CONTENTS Product Description.......... 1 Safety...................... 3 Efficacy Summary............
Validation Strategy for an Automated Endoscope Reprocessor
 Validation Strategy for an Automated Endoscope Reprocessor November 16, 2010 Bradley Catalone AER Validation Strategy System Approach page 1 AER Validation Strategy AER Scope-specific connectors Requires
Validation Strategy for an Automated Endoscope Reprocessor November 16, 2010 Bradley Catalone AER Validation Strategy System Approach page 1 AER Validation Strategy AER Scope-specific connectors Requires
Flexible Endoscopes: The A,B,C s of Monitoring Manual Cleaning Efficacy!
 Flexible Endoscopes: The A,B,C s of Monitoring Manual Cleaning Efficacy! Dr. Michelle J. Alfa, Ph.D., FCCM Medical Director, Clinical Microbiology, Diagnostic Services of Manitoba, Winnipeg, Canada Disclaimers:
Flexible Endoscopes: The A,B,C s of Monitoring Manual Cleaning Efficacy! Dr. Michelle J. Alfa, Ph.D., FCCM Medical Director, Clinical Microbiology, Diagnostic Services of Manitoba, Winnipeg, Canada Disclaimers:
AUTOMATED ENDOSCOPE REPROCESSOR. OER-Pro Easy. Fast. Reliable.
 AUTOMATED ENDOSCOPE REPROCESSOR OER-Pro Easy. Fast. Reliable. Olympus Endoscope Reprocessor The only reprocessor designed by an endoscope manufacturer. The OER-Pro provides high-level disinfection of Olympus
AUTOMATED ENDOSCOPE REPROCESSOR OER-Pro Easy. Fast. Reliable. Olympus Endoscope Reprocessor The only reprocessor designed by an endoscope manufacturer. The OER-Pro provides high-level disinfection of Olympus
Infection Prevention in Endoscopy, Keeping Patients and Practioners Safe
 Infection Prevention in Endoscopy, Keeping Patients and Practioners Safe August 11, 2015 Cheri Ackert-Burr, RN, MSN, CNOR Clinical Education Manager, Medivators Disclosures 1.Successful completion: Participants
Infection Prevention in Endoscopy, Keeping Patients and Practioners Safe August 11, 2015 Cheri Ackert-Burr, RN, MSN, CNOR Clinical Education Manager, Medivators Disclosures 1.Successful completion: Participants
Products made in Switzerland
 Products made in Switzerland HYGIENIC AND MEDICALLY CLEAR DIFFERENT PRODUCTS BUT THE SAME QUALITY LABEL HYGIENIC AND MEDICALLY CLEAR 2 MDD TECHNOLOGIES FOR: 1. Cleaning 2. Disinfection 3. High Level Disinfection
Products made in Switzerland HYGIENIC AND MEDICALLY CLEAR DIFFERENT PRODUCTS BUT THE SAME QUALITY LABEL HYGIENIC AND MEDICALLY CLEAR 2 MDD TECHNOLOGIES FOR: 1. Cleaning 2. Disinfection 3. High Level Disinfection
ADVANTAGE PLUS Pass-Thru Endoscope Reprocessing System
 ADVANTAGE PLUS Pass-Thru Endoscope Reprocessing System Advanced pass-through design Fast and efficient turnover Hands-free operation Built on proven ADVANTAGE PLUS technology FAST AND FLEXIBLE THE FUTURE
ADVANTAGE PLUS Pass-Thru Endoscope Reprocessing System Advanced pass-through design Fast and efficient turnover Hands-free operation Built on proven ADVANTAGE PLUS technology FAST AND FLEXIBLE THE FUTURE
Standards of Infection Prevention in Reprocessing Flexible Gastrointestinal Endoscopes Webinar FAQ
 Standards of Infection Prevention in Reprocessing Flexible Gastrointestinal Endoscopes Webinar FAQ Disclaimer: The Society of Gastroenterology Nurses and Associates, Inc. assume no responsibility for the
Standards of Infection Prevention in Reprocessing Flexible Gastrointestinal Endoscopes Webinar FAQ Disclaimer: The Society of Gastroenterology Nurses and Associates, Inc. assume no responsibility for the
Best Practices for Reprocessing Endoscopes
 Best Practices for Reprocessing Endoscopes Reprocessing of Endoscopes Reprocessing Room Standards The resources referenced include: Canadian Standards Association Provincial Infectious Disease Advisory
Best Practices for Reprocessing Endoscopes Reprocessing of Endoscopes Reprocessing Room Standards The resources referenced include: Canadian Standards Association Provincial Infectious Disease Advisory
Is contamination of bronchoscopes really a problem?
 Is contamination of bronchoscopes really a problem? 1 Is contamination of bronchoscopes really a problem? High Level Disinfection does not methodically clean bronchoscopes What we know 210 More than 210
Is contamination of bronchoscopes really a problem? 1 Is contamination of bronchoscopes really a problem? High Level Disinfection does not methodically clean bronchoscopes What we know 210 More than 210
Christina Bradley Endoscope Decontamination A Webber Training Teleclass. Christina Bradley
 ENDOSCOPE DECONTAMINATION Christina Bradley Hospital Infection Research Laboratory City Hospital NHS Trust Dudley Road, Birmingham B18 7QH Hosted by Paul Webber paul@webbertraining.com www.webbertraining.com
ENDOSCOPE DECONTAMINATION Christina Bradley Hospital Infection Research Laboratory City Hospital NHS Trust Dudley Road, Birmingham B18 7QH Hosted by Paul Webber paul@webbertraining.com www.webbertraining.com
8,000 facilities count on it
 Proven Trusted Safe 8,000 facilities count on it 45 countries use it 10 years of experience prove it Proven performance at every level CIDEX OPA Solution is trusted for its performance and professional
Proven Trusted Safe 8,000 facilities count on it 45 countries use it 10 years of experience prove it Proven performance at every level CIDEX OPA Solution is trusted for its performance and professional
Recommended Procedure for Sampling Gastrointestinal Flexible Endoscopes
 SAMPLING GUIDE* Recommended Procedure for Sampling Gastrointestinal Flexible Endoscopes This document provides the recommended procedure for obtaining acceptable samples from a flexible endoscope without
SAMPLING GUIDE* Recommended Procedure for Sampling Gastrointestinal Flexible Endoscopes This document provides the recommended procedure for obtaining acceptable samples from a flexible endoscope without
The New Choice in Disinfection Technology
 The New Choice in Disinfection Technology www.contecinc.com Introducing The New Choice in Disinfection Technology Accel TB Is A Powerful Disinfectant Against: VIRUCIDAL: 1 Min. at 20 C Proven effectiveness
The New Choice in Disinfection Technology www.contecinc.com Introducing The New Choice in Disinfection Technology Accel TB Is A Powerful Disinfectant Against: VIRUCIDAL: 1 Min. at 20 C Proven effectiveness
STERILIZATION VALIDATION IN THIS SECTION REPROCESSING VALIDATIONS FOR REUSABLE MEDICAL DEVICES. Radiation Sterilization Validation
 STERILIZATION VALIDATION All sterilization processes require validation of the efficacy and reproducibility of the process. Depending on the type of sterilization, this may be accomplished by partial,
STERILIZATION VALIDATION All sterilization processes require validation of the efficacy and reproducibility of the process. Depending on the type of sterilization, this may be accomplished by partial,
Responses to Questions for SYSTEM 1E Process Monitoring and Validation Webinar
 Responses to Questions for SYSTEM 1E Process Monitoring and Validation Webinar MONITORING THE SYSTEM 1E PROCESSOR Q: I want to confirm that there is no need for a Biological Indicator (BI) and that use
Responses to Questions for SYSTEM 1E Process Monitoring and Validation Webinar MONITORING THE SYSTEM 1E PROCESSOR Q: I want to confirm that there is no need for a Biological Indicator (BI) and that use
Best Practices for High Level Disinfection Part I
 Best Practices for High Level Disinfection Part I Nancy Chobin, RN, AAS, ACSP, CSPM, CFER 2017 Sterile Processing University, LLC. **This in-service has been Approved by the CBSPD for 1.5 CEUs. The Bible
Best Practices for High Level Disinfection Part I Nancy Chobin, RN, AAS, ACSP, CSPM, CFER 2017 Sterile Processing University, LLC. **This in-service has been Approved by the CBSPD for 1.5 CEUs. The Bible
Guidelines for the use of disinfectants in hospital settings. Eleni Tomprou Infection Control Nurse, MSc General Hospital of Athens Polykliniki
 Guidelines for the use of disinfectants in hospital settings Eleni Tomprou Infection Control Nurse, MSc General Hospital of Athens Polykliniki Introduction USA 46.5 million surgical procedures Even more
Guidelines for the use of disinfectants in hospital settings Eleni Tomprou Infection Control Nurse, MSc General Hospital of Athens Polykliniki Introduction USA 46.5 million surgical procedures Even more
EVOTECH endoscope cleaner and reprocessor (ECR) simulated-use and clinical-use evaluation of cleaning efficacy
 RESEARCH ARTICLE Open Access Research article EVOTECH endoscope cleaner and reprocessor (ECR) simulated-use and clinical-use evaluation of cleaning efficacy Michelle J Alfa* 1,2,3, Pat DeGagne 2, Nancy
RESEARCH ARTICLE Open Access Research article EVOTECH endoscope cleaner and reprocessor (ECR) simulated-use and clinical-use evaluation of cleaning efficacy Michelle J Alfa* 1,2,3, Pat DeGagne 2, Nancy
ENDOSCOPIC REPROCESSING ETD DOUBLE Advancing the Efficiency of Reprocessing.
 11157 ENDOSCOPIC REPROCESSING ETD DOUBLE Advancing the Efficiency of Reprocessing. ETD DOUBLE: INTRODUCING THE FIRST OLYMPUS PASS- THROUGH WASHER-DISINFECTOR With the ETD Double, Olympus Has Advanced Its
11157 ENDOSCOPIC REPROCESSING ETD DOUBLE Advancing the Efficiency of Reprocessing. ETD DOUBLE: INTRODUCING THE FIRST OLYMPUS PASS- THROUGH WASHER-DISINFECTOR With the ETD Double, Olympus Has Advanced Its
St Boniface Research Centre Winnipeg, Manitoba Canada
 Medical Device Reprocessing: Can we Ban the Biofilm?! St Boniface Research Centre Winnipeg, Manitoba Canada Nancy Olson & Michelle Alfa Dr. Michelle J. Alfa, Ph.D., FCCM Professor, University of Manitoba,
Medical Device Reprocessing: Can we Ban the Biofilm?! St Boniface Research Centre Winnipeg, Manitoba Canada Nancy Olson & Michelle Alfa Dr. Michelle J. Alfa, Ph.D., FCCM Professor, University of Manitoba,
SERIE 4. rapid, reliable, efficient
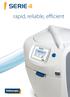 SERIE 4 rapid, reliable, efficient SERIE 4 Intuitive an machine 12/07/2014 15:23:00 1 35 C The power at your fingertips A NEW GENERATION OF INTERFACE The Serie 4 PA brings the benefit of a new intuitive
SERIE 4 rapid, reliable, efficient SERIE 4 Intuitive an machine 12/07/2014 15:23:00 1 35 C The power at your fingertips A NEW GENERATION OF INTERFACE The Serie 4 PA brings the benefit of a new intuitive
Validation of cleaning and sterilization of reusable medical devices
 Synergy makes sence Validation of cleaning and sterilization of reusable medical devices MG-FSI72-119 Last revision: June 2012 Aurélien BIGNON 5, Chemin du Catupolan - 69120 Vaulx en Velin - France - Tel.
Synergy makes sence Validation of cleaning and sterilization of reusable medical devices MG-FSI72-119 Last revision: June 2012 Aurélien BIGNON 5, Chemin du Catupolan - 69120 Vaulx en Velin - France - Tel.
C. difficile as a Cleaning Problem New Deep Cleaning Process Beats Disinfection
 C. difficile as a Cleaning Problem New Deep Cleaning Process Beats Disinfection PCS is proud to introduce our new MicroClean Deep Cleaning Process, which promises to change the industry s view on cleaning
C. difficile as a Cleaning Problem New Deep Cleaning Process Beats Disinfection PCS is proud to introduce our new MicroClean Deep Cleaning Process, which promises to change the industry s view on cleaning
Comparison of ANSI/AAMI ST-91 Standards, SGNA Standards and AORN Guidelines on Processing Endoscopes March 9, 2017
 New guidelines for processing endoscopes, published May 2015 Precleaning AORN 2017 Endoscopy Guideline N/A Review N/A 1. Precleaning; 2. Leak testing; 3. Manual cleaning; 4. Rinse after cleaning; 5. Visual
New guidelines for processing endoscopes, published May 2015 Precleaning AORN 2017 Endoscopy Guideline N/A Review N/A 1. Precleaning; 2. Leak testing; 3. Manual cleaning; 4. Rinse after cleaning; 5. Visual
VHP Sterilization and Prion Inactivation
 WFHSS Workshop Opatija Croatia March 27 th 2009 VHP Sterilization and Prion Inactivation Dr. Georgia Alevizopoulou Clinical Specialist Eastern Europe, Middle East & Africa VHP Sterilization and Prion Inactivation
WFHSS Workshop Opatija Croatia March 27 th 2009 VHP Sterilization and Prion Inactivation Dr. Georgia Alevizopoulou Clinical Specialist Eastern Europe, Middle East & Africa VHP Sterilization and Prion Inactivation
Disinfectant Qualification A Multifaceted Study
 Disinfectant Qualification A Multifaceted Study In this article, Ziva Abraham, Microrite, Inc., gives us a brief overview of the disinfectant qualification study process. An effective cleaning and disinfection
Disinfectant Qualification A Multifaceted Study In this article, Ziva Abraham, Microrite, Inc., gives us a brief overview of the disinfectant qualification study process. An effective cleaning and disinfection
CMS, FDA and AAMI Requirements for Bicarbonate Systems Made Simple. Compiled January 2012 by AmeriWater Incorporated
 CMS, FDA and AAMI Requirements for Bicarbonate Systems Made Simple Compiled January 2012 by AmeriWater Incorporated Background on the Standards FDA, 21 CFR Sec. 876.5820(a) states that hemodialysis systems
CMS, FDA and AAMI Requirements for Bicarbonate Systems Made Simple Compiled January 2012 by AmeriWater Incorporated Background on the Standards FDA, 21 CFR Sec. 876.5820(a) states that hemodialysis systems
DISINFECTION QUALIFI CATION TESTING CONSIDERATIONS FOR THE ASEPTIC AND CLEANROOM MANUFACTURING ENVIRONMENT
 DISINFECTION QUALIFI CATION TESTING CONSIDERATIONS FOR THE ASEPTIC AND CLEANROOM MANUFACTURING ENVIRONMENT by Dave Rottjakob, M.T. (ASCP) December, 2013 INTRODUCTION Obtaining the highest confidence that
DISINFECTION QUALIFI CATION TESTING CONSIDERATIONS FOR THE ASEPTIC AND CLEANROOM MANUFACTURING ENVIRONMENT by Dave Rottjakob, M.T. (ASCP) December, 2013 INTRODUCTION Obtaining the highest confidence that
If All Else Fails Read the Directions. Disclosure. Manufacturer s Instructions. Presenter: Sue Lafferty CHICA Canada Conference, May 2010
 If All Else Fails Read the Directions The Importance of Medical Device Manufacturer s Instructions Presenter: Sue Lafferty CHICA Canada Conference, May 2010 Disclosure Member of 3M Canada Speakers Bureau:
If All Else Fails Read the Directions The Importance of Medical Device Manufacturer s Instructions Presenter: Sue Lafferty CHICA Canada Conference, May 2010 Disclosure Member of 3M Canada Speakers Bureau:
National Endoscopy Programme. Decontamination Standards for Flexible Endoscopes
 National Endoscopy Programme Decontamination Standards for Flexible Endoscopes Updated March 2008 Introduction Providing an effective endoscope decontamination service within a safe environment is an essential
National Endoscopy Programme Decontamination Standards for Flexible Endoscopes Updated March 2008 Introduction Providing an effective endoscope decontamination service within a safe environment is an essential
Oxivir. Tough on Pathogens, Not on People Fast*, Effective, Responsible
 Oxivir Tough on Pathogens, Not on People Fast*, Effective, Responsible * Fast vs. leading competition based on Federal master label comparison as of 11/1/13. Creating a Safer Environment for Patients Healthcare-Associated
Oxivir Tough on Pathogens, Not on People Fast*, Effective, Responsible * Fast vs. leading competition based on Federal master label comparison as of 11/1/13. Creating a Safer Environment for Patients Healthcare-Associated
Competency Guide: Care and Handling of Rigid Endoscopes
 Competency Guide: Care and Handling of Rigid Endoscopes This document details the basic steps to correctly process and maintain a rigid endoscope. This document can be used to perform a competency review
Competency Guide: Care and Handling of Rigid Endoscopes This document details the basic steps to correctly process and maintain a rigid endoscope. This document can be used to perform a competency review
Clostridium difficile Update Dr. Michelle Alfa, Winnipeg Sponsored by ARJO
 Clostridium difficile: Update Dr. Michelle Alfa Ph.D, FCCM Diagnostic Services of Manitoba St. Boniface General Hospital Site Hosted by Paul Webber paul@webbertraining.com Sponsored by www.arjo.com www.webbertraining.com
Clostridium difficile: Update Dr. Michelle Alfa Ph.D, FCCM Diagnostic Services of Manitoba St. Boniface General Hospital Site Hosted by Paul Webber paul@webbertraining.com Sponsored by www.arjo.com www.webbertraining.com
Efficacy Report Summarization for SoClean 2
 Efficacy Report Summarization for SoClean 2 October 2017 SoClean Inc 36 Town Forest Road Oxford, Massachusetts 01540 USA Tel. 508-363-0418 Email info@soclean.com SoClean 2 is USA FDA Registered 3009534409
Efficacy Report Summarization for SoClean 2 October 2017 SoClean Inc 36 Town Forest Road Oxford, Massachusetts 01540 USA Tel. 508-363-0418 Email info@soclean.com SoClean 2 is USA FDA Registered 3009534409
Chemical Sterilants and High-Level Disinfectants: Managing Practical, Safe and Effective Use
 Chemical Sterilants and High-Level Disinfectants: Managing Practical, Safe and Effective Use by Donald P. Satchell, Ph.D. Objectives After completion of this self-study activity, the learner will be able
Chemical Sterilants and High-Level Disinfectants: Managing Practical, Safe and Effective Use by Donald P. Satchell, Ph.D. Objectives After completion of this self-study activity, the learner will be able
Transposal Ultra Evacuation Unit Instructions for Use
 Transposal Ultra Evacuation Unit Instructions for Use Model No.: UL-EV100 Ultra Evac Instructions for Use Manual Effective Date: 07.23.2014 Ultra Evac Instructions for Use Manual Effective Date: 07.23.2014
Transposal Ultra Evacuation Unit Instructions for Use Model No.: UL-EV100 Ultra Evac Instructions for Use Manual Effective Date: 07.23.2014 Ultra Evac Instructions for Use Manual Effective Date: 07.23.2014
Best Practices for Reprocessing Robotic Instruments
 Best Practices for Reprocessing Robotic Instruments Nupur Jain Manager, Validation Engineering Services Intuitive Surgical, US Agenda 1. Background on robotic instruments 2. Reprocessing procedure for
Best Practices for Reprocessing Robotic Instruments Nupur Jain Manager, Validation Engineering Services Intuitive Surgical, US Agenda 1. Background on robotic instruments 2. Reprocessing procedure for
Guidelines for Selection and Use of Disinfectants
 Guidelines for Selection and Use of Disinfectants Ref: (a) APIC Guidelines for Infection Control Practice, American Journal of Infection Control; April 1990, Vol 18, 99-113. To assist health care professionals
Guidelines for Selection and Use of Disinfectants Ref: (a) APIC Guidelines for Infection Control Practice, American Journal of Infection Control; April 1990, Vol 18, 99-113. To assist health care professionals
#003. Liquid Chemical Sterilization Story. The. A Continuing Education Study Guide presented by
 #003 S t u d y G u i d e The Liquid Chemical Sterilization Story A Continuing Education Study Guide presented by Notes The Liquid Chemical Sterilization Story Study Guide #003 Nurses: To receive 1.2
#003 S t u d y G u i d e The Liquid Chemical Sterilization Story A Continuing Education Study Guide presented by Notes The Liquid Chemical Sterilization Story Study Guide #003 Nurses: To receive 1.2
ENDORA Endoscope Tracking System
 ENDORA Endoscope Tracking System Procedure Manual Clean Reprocess Dry & Store Procedure Storage Manual Cleaning Workflow Software Designed to Prevent Endoscope Reprocessing Lapses Reprocessing THE COMPLETE
ENDORA Endoscope Tracking System Procedure Manual Clean Reprocess Dry & Store Procedure Storage Manual Cleaning Workflow Software Designed to Prevent Endoscope Reprocessing Lapses Reprocessing THE COMPLETE
REUSABLE MEDICAL DEVICE VALIDATION
 All sterilization processes require validation of the efficacy and reproducibility of the process. Depending on the type of sterilization, this may be accomplished by partial, sub-lethal, or repetitive
All sterilization processes require validation of the efficacy and reproducibility of the process. Depending on the type of sterilization, this may be accomplished by partial, sub-lethal, or repetitive
Parameters exerting Influence on Cleaning of flexible Endoscops
 Parameters exerting Influence on Cleaning of flexible Endoscops Heike Martiny Technische Hygiene Campus Benjamin Franklin Charité Universitätsmedizin Berlin ÖGSV Jahrestagung und wfhss-kongress 03. - 05.
Parameters exerting Influence on Cleaning of flexible Endoscops Heike Martiny Technische Hygiene Campus Benjamin Franklin Charité Universitätsmedizin Berlin ÖGSV Jahrestagung und wfhss-kongress 03. - 05.
West Virginia Department of Health and Human Resources Office of Environmental Health Services Infectious Medical Waste Program
 IW-20 Rev. 8/04 West Virginia Department of Health and Human Resources Office of Environmental Health Services Infectious Medical Waste Program Application for Alternative Treatment Technology Evaluation
IW-20 Rev. 8/04 West Virginia Department of Health and Human Resources Office of Environmental Health Services Infectious Medical Waste Program Application for Alternative Treatment Technology Evaluation
Safe to Handle? Comparing Manually and Machine-Washed Medical Devices
 Safe to Handle? Comparing Manually and Machine-Washed Medical Devices Kaumudi Kulkarni, Dzelma Kaczorowski, Alex Bonkowski, Stephen Kovach, and Ralph Basile About the Authors Kaumudi Kulkarni, MS, is manager
Safe to Handle? Comparing Manually and Machine-Washed Medical Devices Kaumudi Kulkarni, Dzelma Kaczorowski, Alex Bonkowski, Stephen Kovach, and Ralph Basile About the Authors Kaumudi Kulkarni, MS, is manager
مادة االدوية املرحلة الثالثة أ.م.د. حسام الدين سامل
 مادة االدوية املرحلة الثالثة أ.م.د. حسام الدين سامل 2017-2016 ANTISEPTICS AND DISINFECTANTS Dr. Husam Aldeen Salim General information They have specific use and their selectivity is very low. Disinfectants
مادة االدوية املرحلة الثالثة أ.م.د. حسام الدين سامل 2017-2016 ANTISEPTICS AND DISINFECTANTS Dr. Husam Aldeen Salim General information They have specific use and their selectivity is very low. Disinfectants
Draft Guidance for Industry and FDA Staff Processing/Reprocessing Medical Devices in Health Care Settings: Validation Methods and Labeling
 Draft Guidance for Industry and FDA Staff Processing/Reprocessing Medical Devices in Health Care Settings: Validation Methods and Labeling DRAFT GUIDANCE This guidance document is being distributed for
Draft Guidance for Industry and FDA Staff Processing/Reprocessing Medical Devices in Health Care Settings: Validation Methods and Labeling DRAFT GUIDANCE This guidance document is being distributed for
Principles of Chlorine Dioxide Gas as a Decontamination Method
 Principles of Chlorine Dioxide Gas as a Decontamination Method In order for any decontamination method to be effective, the following points must be satisfied The decontamination method must: Be able to
Principles of Chlorine Dioxide Gas as a Decontamination Method In order for any decontamination method to be effective, the following points must be satisfied The decontamination method must: Be able to
The Complete Solution for Clean Room Aerosol-Based Disinfection MINNCARE DRY FOG SYSTEM
 The Complete Solution for Clean Room Aerosol-Based Disinfection Current Methods of Room Disinfection Surface Wiping Manual Spraying (with Spray Bottles) Heating Process (Vaporization) Cold Process - Wet
The Complete Solution for Clean Room Aerosol-Based Disinfection Current Methods of Room Disinfection Surface Wiping Manual Spraying (with Spray Bottles) Heating Process (Vaporization) Cold Process - Wet
QUALITY ASSURANCE IN AN MDRD
 QUALITY ASSURANCE IN AN MDRD MAINTENANCE AND PREVENTATIVE MAINTENANCE MDRD is a machine dependent department washers, pasteurizers, cart washer, sterilizer, ultrasonic, heat sealers, RO water systems All
QUALITY ASSURANCE IN AN MDRD MAINTENANCE AND PREVENTATIVE MAINTENANCE MDRD is a machine dependent department washers, pasteurizers, cart washer, sterilizer, ultrasonic, heat sealers, RO water systems All
CONTROL OF MICROBIAL GROWTH - DISINFECTANTS AND ANTISEPTICS
 CONTROL OF MICROBIAL GROWTH - DISINFECTANTS AND ANTISEPTICS Specific control measures can be used to kill or inhibit the growth of microorganisms. A procedure which leads to the death of cells is broadly
CONTROL OF MICROBIAL GROWTH - DISINFECTANTS AND ANTISEPTICS Specific control measures can be used to kill or inhibit the growth of microorganisms. A procedure which leads to the death of cells is broadly
CONTROL OF MICROBIAL GROWTH - DISINFECTANTS AND ANTISEPTICS
 CONTROL OF MICROBIAL GROWTH - DISINFECTANTS AND ANTISEPTICS Specific control measures can be used to kill or inhibit the growth of microorganisms. A procedure which leads to the death of cells is broadly
CONTROL OF MICROBIAL GROWTH - DISINFECTANTS AND ANTISEPTICS Specific control measures can be used to kill or inhibit the growth of microorganisms. A procedure which leads to the death of cells is broadly
TOXICITY, EFFICACY, STABILITY AND CORROSION TEST RESULTS
 TOXICITY, EFFICACY, STABILITY AND CORROSION TEST RESULTS MICROCYN TECHNOLOGY TOXICITY STUDIES SKIN IRRITATION This study was conducted to assess the potential of Microcyn to produce dermal irritation and
TOXICITY, EFFICACY, STABILITY AND CORROSION TEST RESULTS MICROCYN TECHNOLOGY TOXICITY STUDIES SKIN IRRITATION This study was conducted to assess the potential of Microcyn to produce dermal irritation and
Hopefully you will learn.. Objectives. Contact Transmission. Pathway of Disease Transmission. Contact Precautions Direct Indirect
 Kathy Zegarski BS, CIC Kettering Medical Center Infection Prevention and Control March 6, 2010 1 2 Objectives Discuss issues and standards associated with infection prevention in the endoscopy setting
Kathy Zegarski BS, CIC Kettering Medical Center Infection Prevention and Control March 6, 2010 1 2 Objectives Discuss issues and standards associated with infection prevention in the endoscopy setting
Case 6. Fight Against Nosocomial Infections
 Case 6 Fight Against Nosocomial Infections Decontamination and microbiological surveillance of endoscopes Prof. dr. Isabel Leroux-Roels Laboratory of Medical Microbiology & Infection Control Team UZ Gent
Case 6 Fight Against Nosocomial Infections Decontamination and microbiological surveillance of endoscopes Prof. dr. Isabel Leroux-Roels Laboratory of Medical Microbiology & Infection Control Team UZ Gent
Tests to Support Sterility Claim. Imtiaz Ahmed
 Tests to Support Sterility Claim Imtiaz Ahmed Sterile Product As per TGO 77, a sterile product must comply with the requirements of the following tests: Sterility Test Bacterial Endotoxins Test Appendix
Tests to Support Sterility Claim Imtiaz Ahmed Sterile Product As per TGO 77, a sterile product must comply with the requirements of the following tests: Sterility Test Bacterial Endotoxins Test Appendix
Getinge da Vinci Solution
 Getinge da Vinci Solution A comprehensive cleaning solution for da Vinci EndoWrist instruments This document is intended to provide information to an international audience outside of the US. Validated,
Getinge da Vinci Solution A comprehensive cleaning solution for da Vinci EndoWrist instruments This document is intended to provide information to an international audience outside of the US. Validated,
Guide for Cleaning, Sterilization and Storage of Introducers for Caldera Medical Implants
 Guide for Cleaning, Sterilization and Storage of Introducers for Caldera Medical Implants M Manufactured by: Caldera Medical, Inc. 5171 Clareton Drive Agoura Hills, CA 91301 U.S. Toll Free: 866-4-CALDERA
Guide for Cleaning, Sterilization and Storage of Introducers for Caldera Medical Implants M Manufactured by: Caldera Medical, Inc. 5171 Clareton Drive Agoura Hills, CA 91301 U.S. Toll Free: 866-4-CALDERA
Disinfection of Endoscopes & Accessories. Luis S. Marsano, MD Professor of Medicine University of Louisville Kentucky
 Disinfection of Endoscopes & Accessories Luis S. Marsano, MD Professor of Medicine University of Louisville Kentucky Sources for Transmission of Infections Contaminated Endoscope Bottle of water for Endoscope
Disinfection of Endoscopes & Accessories Luis S. Marsano, MD Professor of Medicine University of Louisville Kentucky Sources for Transmission of Infections Contaminated Endoscope Bottle of water for Endoscope
Summary of Proposed Legislative Provisions
 Summary of Proposed Legislative Provisions The evidence obtained by the Committee on Oversight and Government Reform during its investigation of duodenoscope-related patient infections identified significant
Summary of Proposed Legislative Provisions The evidence obtained by the Committee on Oversight and Government Reform during its investigation of duodenoscope-related patient infections identified significant
Automated methods in the Microbiology Lab-Issues and Troubleshooting! Presented By: Dr B. Oboudi
 In the name of GOD Automated methods in the Microbiology Lab-Issues and Troubleshooting! Presented By: Dr B. Oboudi Overview Background/history Plate streakers and Gram stainers Blood cultures Automated
In the name of GOD Automated methods in the Microbiology Lab-Issues and Troubleshooting! Presented By: Dr B. Oboudi Overview Background/history Plate streakers and Gram stainers Blood cultures Automated
Microbiological Cleaning Method Validation
 Microbiological Cleaning Method Validation The purpose of cleaning procedures should never be to reduce bioburden to an acceptable level! Fergus O Connell QA Manager Eurofins ams Laboratories www.eurofins.com
Microbiological Cleaning Method Validation The purpose of cleaning procedures should never be to reduce bioburden to an acceptable level! Fergus O Connell QA Manager Eurofins ams Laboratories www.eurofins.com
In use test of a Washer-disinfector reprocessing of flexible endoscopes
 of a Washer-disinfector reprocessing of flexible endoscopes BHT INNOVA E4 Clean Endoscope Copenhagen Hospital Corporation Denmark of INNOVA E4 Washer Disinfector from BHT, Germany. An aldehyde free thermo-chemical
of a Washer-disinfector reprocessing of flexible endoscopes BHT INNOVA E4 Clean Endoscope Copenhagen Hospital Corporation Denmark of INNOVA E4 Washer Disinfector from BHT, Germany. An aldehyde free thermo-chemical
GPS III Platelet Separation System. Leadership through Technology
 GPS III Platelet Separation System Leadership through Technology Quality and Simplicity Redefined with You in Mind Quality GPS III Platelet Separation System Biomet Biologics continues to push the envelope
GPS III Platelet Separation System Leadership through Technology Quality and Simplicity Redefined with You in Mind Quality GPS III Platelet Separation System Biomet Biologics continues to push the envelope
Evaluation of Dry Fogging System for Microbial Inactivation
 Evaluation of Dry Fogging System for Microbial Inactivation Carol Stansfield Senior Biosafety Officer Safety & Environmental Services Canadian Science Centre for Human and Animal Health, Winnipeg, Manitoba
Evaluation of Dry Fogging System for Microbial Inactivation Carol Stansfield Senior Biosafety Officer Safety & Environmental Services Canadian Science Centre for Human and Animal Health, Winnipeg, Manitoba
AFS 8D / 16D Degassed Water Purification Systems
 AFS 8D / 16D Degassed Water Purification Systems Cost-effective, low-maintenance solutions for clinical analyzers with degassed pure water needs up to 320 L daily Cost-effective low-maintenance solutions
AFS 8D / 16D Degassed Water Purification Systems Cost-effective, low-maintenance solutions for clinical analyzers with degassed pure water needs up to 320 L daily Cost-effective low-maintenance solutions
Report prepared on behalf of Medivators Inc. by Nova Biologicals, Inc., Title of Report: Laboratory Evaluation of Endoscope Waterbottles
 Report prepared on behalf of Medivators Inc. by Nova Biologicals, Inc., 2005 Title of Report: Laboratory Evaluation of Endoscope Waterbottles Author: Paul J. Pearce, Bachelor of Science (BS), Master of
Report prepared on behalf of Medivators Inc. by Nova Biologicals, Inc., 2005 Title of Report: Laboratory Evaluation of Endoscope Waterbottles Author: Paul J. Pearce, Bachelor of Science (BS), Master of
Prevention of Transmission of the Superbug Carbapenem-Resistant Enterobacteriaceae (CRE) during Gastrointestinal Endoscopy
 Prevention of Transmission of the Superbug Carbapenem-Resistant Enterobacteriaceae (CRE) during Gastrointestinal Endoscopy Sponsored by: Presentation by: Lawrence F. Muscarella, Ph.D. President LFM Healthcare
Prevention of Transmission of the Superbug Carbapenem-Resistant Enterobacteriaceae (CRE) during Gastrointestinal Endoscopy Sponsored by: Presentation by: Lawrence F. Muscarella, Ph.D. President LFM Healthcare
Avoid recurrent microbial contamination using trending of historical data. El Azab Walid Technical Service Manager STERIS
 Avoid recurrent microbial contamination using trending of historical data El Azab Walid Technical Service Manager STERIS Agenda Factors influencing the cleaning and disinfection performance Recurring microbial
Avoid recurrent microbial contamination using trending of historical data El Azab Walid Technical Service Manager STERIS Agenda Factors influencing the cleaning and disinfection performance Recurring microbial
á61ñ MICROBIOLOGICAL EXAMINATION OF NONSTERILE PRODUCTS: MICROBIAL ENUMERATION TESTS
 USP 40 Microbiological Tests / á61ñ Microbiological Examination 1 á61ñ MICROBIOLOGICAL EXAMINATION OF NONSTERILE PRODUCTS: MICROBIAL ENUMERATION TESTS INTRODUCTION The tests described hereafter will allow
USP 40 Microbiological Tests / á61ñ Microbiological Examination 1 á61ñ MICROBIOLOGICAL EXAMINATION OF NONSTERILE PRODUCTS: MICROBIAL ENUMERATION TESTS INTRODUCTION The tests described hereafter will allow
Lumen claims of the STERRAD 100NX Sterilizer: Testing performance limits of this technology
 Lumen claims of the STERRAD 100NX Sterilizer: Testing performance limits of this technology Magda Diab-Elschahawi, Alexander Blacky, Walter Koller Clinical Institute of Hospital Hygiene Medical University
Lumen claims of the STERRAD 100NX Sterilizer: Testing performance limits of this technology Magda Diab-Elschahawi, Alexander Blacky, Walter Koller Clinical Institute of Hospital Hygiene Medical University
A GLIMPSE AT THE TRUE COST OF REPROCESSING ENDOSCOPES:
 by Cori L. Ofstead, MSPH; Mariah R. Quick, MPH; John E. Eiland, MS, RN; Steven J. Adams, RN, CRCST A GLIMPSE AT THE TRUE COST OF REPROCESSING ENDOSCOPES: RESULTS OF A PILOT PROJECT INTRODUCTION AND METHODS
by Cori L. Ofstead, MSPH; Mariah R. Quick, MPH; John E. Eiland, MS, RN; Steven J. Adams, RN, CRCST A GLIMPSE AT THE TRUE COST OF REPROCESSING ENDOSCOPES: RESULTS OF A PILOT PROJECT INTRODUCTION AND METHODS
Choosing an Effective Sanitizer
 Choosing an Effective Sanitizer, CEO - Shepard Bros., Inc. NWFSS 2017 Outline Antimicrobial Definitions Cleaning Comes First Biofilms Overview of Sanitizers Common Products New Technologies Organic Processing
Choosing an Effective Sanitizer, CEO - Shepard Bros., Inc. NWFSS 2017 Outline Antimicrobial Definitions Cleaning Comes First Biofilms Overview of Sanitizers Common Products New Technologies Organic Processing
The University of Ferrara Study At the St. Anne Hospital
 The University of Ferrara Study At the St. Anne Hospital The Research Centre For Pollution Control In High Sterile Rooms Conducted A Year-Long Study (2010 2011) On The TESTING OF BIO-STABILIZATION TECHNIQUES
The University of Ferrara Study At the St. Anne Hospital The Research Centre For Pollution Control In High Sterile Rooms Conducted A Year-Long Study (2010 2011) On The TESTING OF BIO-STABILIZATION TECHNIQUES
Endoscope Reprocessor Specification Installation Instructions
 Endoscope Reprocessor Specification Installation Instructions Endoscope Reprocessing System Introduction The following document outlines the space requirements, water supply specifications, drain specifications,
Endoscope Reprocessor Specification Installation Instructions Endoscope Reprocessing System Introduction The following document outlines the space requirements, water supply specifications, drain specifications,
3/26/2015. This quote sums up the activity going on right now with flexible scopes
 This quote sums up the activity going on right now with flexible scopes Flexible Endoscope Cleaning If you want to truly understand something try to change it Kurt Lewis Objectives To understand and make
This quote sums up the activity going on right now with flexible scopes Flexible Endoscope Cleaning If you want to truly understand something try to change it Kurt Lewis Objectives To understand and make
Cleaning Procedures. These guidelines are intended for Bulk Pharmaceutical Chemicals but almost every word applies to Drug Products
 CLEANING DEVELOPMENT Cleaning Procedures as the FDA See It These guidelines are intended for Bulk Pharmaceutical Chemicals but almost every word applies to Drug Products BPC CLEANING (& Drug Products)
CLEANING DEVELOPMENT Cleaning Procedures as the FDA See It These guidelines are intended for Bulk Pharmaceutical Chemicals but almost every word applies to Drug Products BPC CLEANING (& Drug Products)
Active Oxygen Tablets
 Introduction We are pleased to introduce a new and unique technology within the field of surface disinfection: A powerful and efficient surface disinfectant in user-friendly effervescent tablet form. This
Introduction We are pleased to introduce a new and unique technology within the field of surface disinfection: A powerful and efficient surface disinfectant in user-friendly effervescent tablet form. This
determine optimum instrument settings for their own instruments and establish their own daily values.
 PC7 (770/488) SETUP KIT 6607121 PN 4299504-C FLOW CYTOMETER ALIGNMENT VERIFICATION FLUOROSPHERES FLOW CYTOMETER DETECTOR STANDARDIZATION FLUOROSPHERES INTENDED USE For Research Use Only. Not for use in
PC7 (770/488) SETUP KIT 6607121 PN 4299504-C FLOW CYTOMETER ALIGNMENT VERIFICATION FLUOROSPHERES FLOW CYTOMETER DETECTOR STANDARDIZATION FLUOROSPHERES INTENDED USE For Research Use Only. Not for use in
Designing an effective cleaning procedure for medical devices through laboratory studies
 Designing an effective cleaning procedure for medical devices through laboratory studies Elizabeth Rivera Technical Service Specialist STERIS Corporation elizabeth_rivera@steris.com October 2012 Agenda
Designing an effective cleaning procedure for medical devices through laboratory studies Elizabeth Rivera Technical Service Specialist STERIS Corporation elizabeth_rivera@steris.com October 2012 Agenda
sterility assurance Prove the power and your processes
 sterility assurance Prove the power and efficacy of your processes sterility assurance programs: an introduction YOUR CHALLENGES Contamination events cost a lot of time and money. Sterility problems can
sterility assurance Prove the power and efficacy of your processes sterility assurance programs: an introduction YOUR CHALLENGES Contamination events cost a lot of time and money. Sterility problems can
500 Multipurpose Sanitising Wipes
 Product Data 500 Multipurpose Sanitising Wipes Product FAct Sheet PRODUCT FACT SHEET 500 MULTIPURPOSE SANITISING WIPES ISSUED 11-11-2013 TECHNICAL SPECIFICATION PRODUCT BRAND DESCRIPTION MULTIPURPOSE SANITISING
Product Data 500 Multipurpose Sanitising Wipes Product FAct Sheet PRODUCT FACT SHEET 500 MULTIPURPOSE SANITISING WIPES ISSUED 11-11-2013 TECHNICAL SPECIFICATION PRODUCT BRAND DESCRIPTION MULTIPURPOSE SANITISING
USER S MANUAL. Flexible Nasopharyngo-Laryngoscope
 USER S MANUAL COGENTIX MEDICAL Flexible Nasopharyngo-Laryngoscope ENT-4000/4500 NOTE: Federal (USA) law restricts this device to sale by, or on the order of, a physician or other appropriately licensed
USER S MANUAL COGENTIX MEDICAL Flexible Nasopharyngo-Laryngoscope ENT-4000/4500 NOTE: Federal (USA) law restricts this device to sale by, or on the order of, a physician or other appropriately licensed
Mar Cor Purification. Minncare Dry Fog Technology. An Integral Part of Modern Critical Area Disinfection Procedures MAR COR
 Minncare Dry Fog Technology An Integral Part of Modern Critical Area Disinfection Procedures MAR COR The Safest & Most Effective Clean Room Bio-Decon Clean Room Lock crabs Isolator Biohazard Cabinet Animal
Minncare Dry Fog Technology An Integral Part of Modern Critical Area Disinfection Procedures MAR COR The Safest & Most Effective Clean Room Bio-Decon Clean Room Lock crabs Isolator Biohazard Cabinet Animal
Best Practices for High Level Disinfection - Revisited. Objectives. The Bible for Chemicals ST-58 FDA. What is Available? 9/6/2015
 Best Practices for High Level Disinfection - Revisited Nancy Chobin, RN, AAS, ACSP, CSPM President, Sterile Processing University, LLC Copyright 2015 Objectives To define disinfection To review general
Best Practices for High Level Disinfection - Revisited Nancy Chobin, RN, AAS, ACSP, CSPM President, Sterile Processing University, LLC Copyright 2015 Objectives To define disinfection To review general
EASY-Spray Columns. Guidance for column set up and installation Tips to maximize column lifetime
 EASY-Spray Columns Guidance for column set up and installation Tips to maximize column lifetime EASY-Spray Column Tips and Tricks This document provides guidance for Thermo Scientific EASY-Spray column
EASY-Spray Columns Guidance for column set up and installation Tips to maximize column lifetime EASY-Spray Column Tips and Tricks This document provides guidance for Thermo Scientific EASY-Spray column
Ortho-phthalaldehyde: a possible alternative to glutaraldehyde for high level disinfection
 Journal of Applied Microbiology 1999, 86, 1039 1046 Ortho-phthalaldehyde: a possible alternative to glutaraldehyde for high level disinfection S.E. Walsh, J-Y. Maillard and A.D. Russell Welsh School of
Journal of Applied Microbiology 1999, 86, 1039 1046 Ortho-phthalaldehyde: a possible alternative to glutaraldehyde for high level disinfection S.E. Walsh, J-Y. Maillard and A.D. Russell Welsh School of
Method Suitability Report Membrane Filtration Sterility Test with QTMicro Apparatus
 Method Suitability Report Membrane Filtration Sterility Test with QTMicro Apparatus Bevacizumab (Avastin) 1.25 mg/0.05ml This report provides details on the specifics of a Membrane Filtration Sterility
Method Suitability Report Membrane Filtration Sterility Test with QTMicro Apparatus Bevacizumab (Avastin) 1.25 mg/0.05ml This report provides details on the specifics of a Membrane Filtration Sterility
Foundations in Microbiology Seventh Edition. Talaro Chapter 11 Physical and Chemical Agents for Microbial Control
 Foundations in Microbiology Seventh Edition Talaro Chapter 11 Physical and Chemical Agents for Microbial Control 11.1 Controlling Microorganisms Physical, chemical, and mechanical methods to destroy or
Foundations in Microbiology Seventh Edition Talaro Chapter 11 Physical and Chemical Agents for Microbial Control 11.1 Controlling Microorganisms Physical, chemical, and mechanical methods to destroy or
Breaking new ground in
 Breaking new ground in Sterilisation Disinfecting Water purification 2009. The written and pictorial material in this document is the property of Envirolyte Industries International Ltd and its partners
Breaking new ground in Sterilisation Disinfecting Water purification 2009. The written and pictorial material in this document is the property of Envirolyte Industries International Ltd and its partners
Water for Instrument Processing
 Water for Instrument Processing by Marcia Frieze, Case Medical Water, the universal solvent Water can dissolve more substances than any other liquid. It is essentially nonionic or neutral. While alkaline
Water for Instrument Processing by Marcia Frieze, Case Medical Water, the universal solvent Water can dissolve more substances than any other liquid. It is essentially nonionic or neutral. While alkaline
AAG PRINCIPLE SPECIMEN. REF (150 tests) ANNUAL REVIEW Reviewed by: Date. Date INTENDED USE
 IMMAGE Immunochemistry Systems Chemistry Information Sheet Copyright 2010 Beckman Coulter, Inc. Alpha 1 -Acid Glycoprotein REF 447780 (150 tests) For In Vitro Diagnostic Use ANNUAL REVIEW Reviewed by:
IMMAGE Immunochemistry Systems Chemistry Information Sheet Copyright 2010 Beckman Coulter, Inc. Alpha 1 -Acid Glycoprotein REF 447780 (150 tests) For In Vitro Diagnostic Use ANNUAL REVIEW Reviewed by:
This document is a preview generated by EVS
 INTERNATIONAL STANDARD ISO 18416 Second edition 2015-12-01 Corrected version 2016-12-15 Cosmetics Microbiology Detection of Candida albicans Cosmétiques Microbiologie Détection de Candida albicans Reference
INTERNATIONAL STANDARD ISO 18416 Second edition 2015-12-01 Corrected version 2016-12-15 Cosmetics Microbiology Detection of Candida albicans Cosmétiques Microbiologie Détection de Candida albicans Reference
