Parameter Estimation for the Exponential- Normal Convolution Model for Background Correction of Affymetrix GeneChip Data
|
|
|
- Logan Roberts
- 6 years ago
- Views:
Transcription
1 Parameter Estimation for the Exponential- Normal Convolution Model for Background Correction of Affymetrix GeneChip Data Monnie McGee, PhD Department of Statistical Science Southern Methodist University Dallas, Texas There are many methods of correcting microarray data for non-biological sources of error. Authors routinely supply software or code so that interested analysts can implement their methods. Even with a thorough reading of associated references, it is not always clear how requisite parts of the method are calculated in the software packages. However, it is important to have an understanding of such details, as this understanding is necessary for proper use of the output, or for implementing extensions to the model. In this paper, the calculation of parameter estimates used in Robust Multichip Average (RMA), a popular preprocessing algorithm for Affymetrix GeneChip brand microarrays, is elucidated. The background correction method for RMA assumes that the perfect match (PM) intensities observed result from a convolution of the true signal, assumed to be exponentially distributed, and a background noise component, assumed to have a normal distribution. A conditional expectation is calculated to estimate signal. Estimates of the mean and variance of the normal distribution and the rate parameter of the exponential distribution are needed to calculate this expectation. Simulation studies show that the current estimates are flawed; therefore, new ones are suggested. We examine the performance of preprocessing under the exponential-normal convolution model using several different methods to estimate the parameters. Friday, January 12, :00-11:00 am
2 Considerations in Adapting Clinical Trial Design for Drug Development H.M. James Hung, Ph.D. Division of Biometrics I, OB/OTS/CDER Food and Drug Administration Enhancing flexibility of clinical trial designs is one of the hot topics nowadays. Proper adaptation of clinical trial design is one of the ways for achieving this goal and has drawn much attention from clinical trialists. In past decades, the classical design has been improved to allow the flexibility for terminating the trial early if the experimental treatment is proven effective or deemed harmful or futile, based on the data accumulating during the course of the trial. Statistical validity of such an enhanced design in terms of type I error is maintained. The operational aspects of this design can still be an issue but, by and large, there have been many good models for how to deal with these aspects. As the flexibility of trial design is enhanced further, the potential risk that the resulting trial may not be interpretable increases. In this presentation we shall share our review experience, discuss the many issues arising from use of more flexible designs and hopefully stimulate further research in this area. Friday, January 19, :00-11:00 am
3 Adaptive Simon Designs for Heterogeneous Patient Populations in Phase II Cancer Trials Karen Messer, PhD Director of Biostatistics Moores UCSD Cancer Center Univeristy of California San Diego, CA In a Phase II cancer trial it may be advantageous to open enrollment to several patient populations, each with a very different null probability of response. For example in a trial of a novel therapeutic agent for relapsed Acute Myelogenous Leukemia (AML), patients in a first relapse may have a 30% probability of response under standard treatment, while patients in second relapse or higher may have only a 10% probability of response. These Phase II trials are generally uncontrolled (they often use historical controls ), and the experimental agent may be expected to induce certain Grade 3 toxicities which would not be considered dose limiting. Furthermore, historically most of these Phase II trials can be expected to prove no better than standard-of-care. Phase II trials with these characteristics are usually designed with an early stopping rule which checks for initial evidence of efficacy after a first stage enrollment target is met. If there is insufficient evidence, the trial stops for futility. We discuss the standard two-stage optimal designs in this situation, and describe their operating characteristics under heterogeneous patient enrollment. These are compared to other approaches in the literature. Simple, approximately optimal designs which account for heterogeneity are presented. We recommend a practical adaptive design strategy which we have implemented at Moores UCSD Cancer Center. Friday, February 2, :00-11:00 am
4 Statistical Analysis of Reverse-Phase Protein Arrays Kevin Coombes, PhD Department of Biostatistics & Applied Mathematics University of Texas M. D. Anderson Cancer Center Reverse-Phase Protein Arrays (RPPAs, aka protein lysate arrays, tissue lysate arrays, or lysate arrays) are recently developed tools for measuring protein expression levels in large numbers of samples. These assays are for the most part massively parallelized versions of enzyme-linked immunosorbent assays (ELISAs). In their massive parallelization, these assays are similar to cdna microarrays (for mrna) and CGH assays (for DNA). However, while those assays make thousands of measurements on a single sample ("forward-phase"), RPPAs measure one thing on hundreds of samples ("reverse-phase"). In this talk, we will attempt to place RPPAs in the broader context of other protein assays. Given this background, we will then describe issues we have encountered in modeling this data, and describe some of the tools we have developed for this purpose. Open questions will be identified. Friday, February 16, :00-11:00 am
5 Sequential Monitoring of Randomization Tests William F. Rosenberger, PhD Professor and Chairman of Statistics George Mason University Randomization provides a basis for inference, but it is rarely taken advantage of. We discuss randomization tests based on the family of linear rank tests in the context of sequential monitoring of clinical trials. Such tests are applicable for categorical, continuous, and survival time outcomes. We prove the asymptotic joint normality of sequentially monitored test statistics, which allows the computation of sequential monitoring critical values under the Lan-DeMets procedure. Since randomization tests are not based on likelihoods, the concept of information is murky. We give an alternate definition of randomization and show how to compute it for different randomization procedures. The randomization procedures we discuss are the permuted block design, stratified block design, and stratified urn design. We illustrate these results by reanalyzing a clinical trial in retinopathy. Thursday, March 1, :00-11:00 am
6 "Use of a Visual Programming Environment for Creating and Optimizing Mass Spectrometry Diagnostic Workflows" Maciek Sasinowski, Ph.D. Founder and CEO of INCOGEN, Inc. Williamsburg, VA The use of mass spectrometry for clinical applications has extraordinary potential for accurate, early, and minimally invasive diagnoses of complex diseases, such as cancer, which require sensitive diagnostic tools for prognosis and development of flexible treatment strategies. Unfortunately, current mass spectrometry data analysis options available to researchers often require improvised combinations of tools provided by instrument manufacturers, third-parties, and in-house development. The lack of unified interfaces to access existing resources presents a significant bottleneck in the research and discovery process. In this seminar, we present a modular software tool for the analysis of mass spectrometry profiling data that aims to address this bottleneck. The modules that comprise the analysis workflows can be broadly classified into three categories: signal processing tools, variable selection algorithms, and classification utilities. The software tool provides a platform that allows researchers to construct, validate, and optimize classification workflows of serum samples analyzed with time-of-flight mass spectrometry. Our work suggests that this type of flexible and interactive architecture is highly useful for 1) the development of mass spectrometry workflows and 2) biomarker discovery and validation in clinical environments. Friday, March 16, :00-11:00 am
7 " The Assessment of Multivariate Bioequivalence" Thomas Mathew, Ph.D. Department of Mathematics & Statistics, UMBC Baltimore, MD The purpose of bioequivalence testing is to compare the bioavailabilities of two drug products: a brand name drug and a generic copy, for the purpose of establishing the equivalence between the two. The primary data for bioequivalence testing consist of the concentration of the active ingredient in the blood measured at several time points, after administering the drug. The concentration is plotted against time, and three responses are obtained: the area under the plasma concentration-time curve, simply referred to as area under the curve or AUC, the maximum concentration that shows up in the blood, and the time to reach the maximum concentration. Univariate bioequivalence consists of the separate modeling and analysis of the data based on the three univariate responses. Multivariate bioequivalence consists of the joint modeling and analysis of the data on the three variables, or any two of them. It is standard practice to use mixed effects models. Very extensive literature is available on the topic of univariate bioequivalence testing; a brief review will be given in my talk, addressing the concepts of average bioequivalence, and variance bioequivalence. In the univariate case, statistical procedures have been developed to address these. The literature in the multivariate case is very limited, dealing only with average bioequivalence. In my talk, I will address various aspects of multivariate bioequivalence testing, and will describe test procedures, along with some data analysis. Friday, April 20, :00-11:00 am
8 Systems pharmacology of type 2 diabetes: a case study for pharmaceutical development Terence E. Ryan, PhD Senior Director, Systems Biology Biological Technologies Wyeth Research Collegeville, PA A key issue in drug discovery is the appropriate use of animal models to study human disease and therapeutic drug response. Animal models have, in general, been used to mimic human disease based upon relatively few points of analogy. The richness of open discovery omics platforms allows a comprehensive measurement of disease and drug response across a range of analyte classes, allowing investigators to better understand the predictive value of animal models for human disease. In a study conducted by GlaxoSmithKline, we compared disease effects and treatment response in two mouse models of type 2 diabetes and in a parallel human study. Three registered medicines for diabetes (rosglitazone, metformin, and glyburide) were studied, and detailed measurements of transcripts, lipids, metabolites, and proteins were obtained in tissues and biofluids. Integrated data analysis using various multivariate techniques allowed for the generation of predictive fingerprints which shorten the time required to demonstrate treatment efficacy in diabetes trials, as well as allowing the identification of patients most likely to respond to a particular therapy form baseline measurements. In addition, analysis uncovered a previously unsuspected mechanism for rosiglitazone activity in diabetic adipose. The use of Systems Biology approaches with large omic datasets holds great promise for deeper understandings of disease biology and pharmacology Friday, May 4, :00-11:00 am Lombardi Comprehensive Cancer Center, Room 131 (LL Lombardi)
9 Repetitive DNA and the evolution of eukaryotic gene regulation King Jordan, PhD Associate Professor School of Biology Georgia Institute of Technology Atlanta, GA At least 50% of the human genome is made up of repetitive DNA, while only 1.5% is dedicated to protein coding sequences. The functional and evolutionary significance of this repetitive DNA fraction is very much an open question. We use computational methods to explore the ways that repetitive DNA, transposable elements in particular, has influenced the structure, function and evolution of eukaryotic genomes. I will discuss recent results related to the effects of repetitive DNA on nucleosome binding affinities along vertebrate proximal promoter regions and demonstrate how this relates to the regulation of gene expression. I will also provide evidence detailing the evolution of a number of distinct classes of gene regulatory sequences from transposable elements. Taken together these results suggest a model for the evolution of eukaryotic genomes and gene regulation. This model holds that the challenges posed by transposable elements have led repeatedly to the emergence of genome defense mechanisms. These emergent regulatory mechanisms, including methylation, chromatin and RNA interference, were subsequently co-opted to serve as global regulatory systems, which in turn have provided for the regulatory complexity characteristic of the eukaryotic evolutionary lineage. Friday, May 18, :00-11:00 am Lombardi Comprehensive Cancer Center, E501 New Research Bldg.
REIMAGINING DRUG DEVELOPMENT:
 Biology Reconstructed REIMAGINING DRUG DEVELOPMENT: Accurate Disease Modeling To Drive Successful Therapies Julia Kirshner, CEO julia@zpredicta.com 1 SUCCESS RATES OF DRUG DEVELOPMENT ARE LOW, " PARTICULARLY
Biology Reconstructed REIMAGINING DRUG DEVELOPMENT: Accurate Disease Modeling To Drive Successful Therapies Julia Kirshner, CEO julia@zpredicta.com 1 SUCCESS RATES OF DRUG DEVELOPMENT ARE LOW, " PARTICULARLY
Maximizing opportunities towards achieving clinical success D R U G D I S C O V E R Y. Report Price Publication date
 F o r a c l e a r e r m a r k e t p e r s p e c t i v e Early Stage Drug Safety Strategies & Risk Management Maximizing opportunities towards achieving clinical success D R U G D I S C O V E R Y Report
F o r a c l e a r e r m a r k e t p e r s p e c t i v e Early Stage Drug Safety Strategies & Risk Management Maximizing opportunities towards achieving clinical success D R U G D I S C O V E R Y Report
DNA Microarray Technology
 CHAPTER 1 DNA Microarray Technology All living organisms are composed of cells. As a functional unit, each cell can make copies of itself, and this process depends on a proper replication of the genetic
CHAPTER 1 DNA Microarray Technology All living organisms are composed of cells. As a functional unit, each cell can make copies of itself, and this process depends on a proper replication of the genetic
Introduction to gene expression microarray data analysis
 Introduction to gene expression microarray data analysis Outline Brief introduction: Technology and data. Statistical challenges in data analysis. Preprocessing data normalization and transformation. Useful
Introduction to gene expression microarray data analysis Outline Brief introduction: Technology and data. Statistical challenges in data analysis. Preprocessing data normalization and transformation. Useful
Genomics Market Share, Size, Analysis, Growth, Trends and Forecasts to 2024 Hexa Research
 Genomics Market Share, Size, Analysis, Growth, Trends and Forecasts to 2024 Hexa Research " Increasing usage of novel genomics techniques and tools, evaluation of their benefit to patient outcome and focusing
Genomics Market Share, Size, Analysis, Growth, Trends and Forecasts to 2024 Hexa Research " Increasing usage of novel genomics techniques and tools, evaluation of their benefit to patient outcome and focusing
A Distribution Free Summarization Method for Affymetrix GeneChip Arrays
 A Distribution Free Summarization Method for Affymetrix GeneChip Arrays Zhongxue Chen 1,2, Monnie McGee 1,*, Qingzhong Liu 3, and Richard Scheuermann 2 1 Department of Statistical Science, Southern Methodist
A Distribution Free Summarization Method for Affymetrix GeneChip Arrays Zhongxue Chen 1,2, Monnie McGee 1,*, Qingzhong Liu 3, and Richard Scheuermann 2 1 Department of Statistical Science, Southern Methodist
FDA Perspectives on Biomarkers. Steven Kozlowski, M.D. Office of Biotechnology Products OPS/CDER/FDA
 FDA Perspectives on Biomarkers Steven Kozlowski, M.D. Office of Biotechnology Products OPS/CDER/FDA Importance of Biomarkers Earlier diagnosis Focusing expensive & invasive therapies on the right populations
FDA Perspectives on Biomarkers Steven Kozlowski, M.D. Office of Biotechnology Products OPS/CDER/FDA Importance of Biomarkers Earlier diagnosis Focusing expensive & invasive therapies on the right populations
E2ES to Accelerate Next-Generation Genome Analysis in Clinical Research
 www.hcltech.com E2ES to Accelerate Next-Generation Genome Analysis in Clinical Research whitepaper April 2015 TABLE OF CONTENTS Introduction 3 Challenges associated with NGS data analysis 3 HCL s NGS Solution
www.hcltech.com E2ES to Accelerate Next-Generation Genome Analysis in Clinical Research whitepaper April 2015 TABLE OF CONTENTS Introduction 3 Challenges associated with NGS data analysis 3 HCL s NGS Solution
"Stratification biomarkers in personalised medicine"
 1/12 2/12 SUMMARY REPORT "Stratification biomarkers in personalised medicine" Workshop to clarify the scope for stratification biomarkers and to identify bottlenecks in the discovery and the use of such
1/12 2/12 SUMMARY REPORT "Stratification biomarkers in personalised medicine" Workshop to clarify the scope for stratification biomarkers and to identify bottlenecks in the discovery and the use of such
POPULATION ENRICHMENT DESIGNS FOR ADAPTIVE CLINICAL TRIALS. An Aptiv Solutions White Paper
 FOR ADAPTIVE CLINICAL TRIALS An Aptiv Solutions White Paper EXECUTIVE SUMMARY The increasing pressure on governments caused by the spiraling healthcare costs is leading to a growing demand by payers for
FOR ADAPTIVE CLINICAL TRIALS An Aptiv Solutions White Paper EXECUTIVE SUMMARY The increasing pressure on governments caused by the spiraling healthcare costs is leading to a growing demand by payers for
TRANSFORMING GLOBAL GENETIC DATA INTO MEDICAL DECISIONS
 TRANSFORMING GLOBAL GENETIC DATA INTO MEDICAL DECISIONS THE DOORS ARE OPEN: FEEL FREE TO COME IN CENTOGENE UNLOCKS THE POWER OF GENETIC INSIGHTS TO IMPROVE THE QUALITY OF LIFE OF PATIENTS WITH GENETIC
TRANSFORMING GLOBAL GENETIC DATA INTO MEDICAL DECISIONS THE DOORS ARE OPEN: FEEL FREE TO COME IN CENTOGENE UNLOCKS THE POWER OF GENETIC INSIGHTS TO IMPROVE THE QUALITY OF LIFE OF PATIENTS WITH GENETIC
THE PATH TO PRECISION MEDICINE IN MULTIPLE MYELOMA. themmrf.org
 THE PATH TO PRECISION MEDICINE IN MULTIPLE MYELOMA themmrf.org ABOUT THE MULTIPLE MYELOMA RESEARCH FOUNDATION The Multiple Myeloma Research Foundation (MMRF) was established in 1998 by identical twin sisters
THE PATH TO PRECISION MEDICINE IN MULTIPLE MYELOMA themmrf.org ABOUT THE MULTIPLE MYELOMA RESEARCH FOUNDATION The Multiple Myeloma Research Foundation (MMRF) was established in 1998 by identical twin sisters
Application of Deep Learning to Drug Discovery
 Application of Deep Learning to Drug Discovery Hase T, Tsuji S, Shimokawa K, Tanaka H Tohoku Medical Megabank Organization, Tohoku University Current Situation of Drug Discovery Rapid increase of R&D expenditure
Application of Deep Learning to Drug Discovery Hase T, Tsuji S, Shimokawa K, Tanaka H Tohoku Medical Megabank Organization, Tohoku University Current Situation of Drug Discovery Rapid increase of R&D expenditure
The Five Key Elements of a Successful Metabolomics Study
 The Five Key Elements of a Successful Metabolomics Study Metabolomics: Completing the Biological Picture Metabolomics is offering new insights into systems biology, empowering biomarker discovery, and
The Five Key Elements of a Successful Metabolomics Study Metabolomics: Completing the Biological Picture Metabolomics is offering new insights into systems biology, empowering biomarker discovery, and
3.1.4 DNA Microarray Technology
 3.1.4 DNA Microarray Technology Scientists have discovered that one of the differences between healthy and cancer is which genes are turned on in each. Scientists can compare the gene expression patterns
3.1.4 DNA Microarray Technology Scientists have discovered that one of the differences between healthy and cancer is which genes are turned on in each. Scientists can compare the gene expression patterns
Predicting Microarray Signals by Physical Modeling. Josh Deutsch. University of California. Santa Cruz
 Predicting Microarray Signals by Physical Modeling Josh Deutsch University of California Santa Cruz Predicting Microarray Signals by Physical Modeling p.1/39 Collaborators Shoudan Liang NASA Ames Onuttom
Predicting Microarray Signals by Physical Modeling Josh Deutsch University of California Santa Cruz Predicting Microarray Signals by Physical Modeling p.1/39 Collaborators Shoudan Liang NASA Ames Onuttom
FACTORS CONTRIBUTING TO VARIABILITY IN DNA MICROARRAY RESULTS: THE ABRF MICROARRAY RESEARCH GROUP 2002 STUDY
 FACTORS CONTRIBUTING TO VARIABILITY IN DNA MICROARRAY RESULTS: THE ABRF MICROARRAY RESEARCH GROUP 2002 STUDY K. L. Knudtson 1, C. Griffin 2, A. I. Brooks 3, D. A. Iacobas 4, K. Johnson 5, G. Khitrov 6,
FACTORS CONTRIBUTING TO VARIABILITY IN DNA MICROARRAY RESULTS: THE ABRF MICROARRAY RESEARCH GROUP 2002 STUDY K. L. Knudtson 1, C. Griffin 2, A. I. Brooks 3, D. A. Iacobas 4, K. Johnson 5, G. Khitrov 6,
Introduction to Bioinformatics and Gene Expression Technology
 Vocabulary Introduction to Bioinformatics and Gene Expression Technology Utah State University Spring 2014 STAT 5570: Statistical Bioinformatics Notes 1.1 Gene: Genetics: Genome: Genomics: hereditary DNA
Vocabulary Introduction to Bioinformatics and Gene Expression Technology Utah State University Spring 2014 STAT 5570: Statistical Bioinformatics Notes 1.1 Gene: Genetics: Genome: Genomics: hereditary DNA
Differential Gene Expression
 Biology 4361 Developmental Biology Differential Gene Expression September 28, 2006 Chromatin Structure ~140 bp ~60 bp Transcriptional Regulation: 1. Packing prevents access CH 3 2. Acetylation ( C O )
Biology 4361 Developmental Biology Differential Gene Expression September 28, 2006 Chromatin Structure ~140 bp ~60 bp Transcriptional Regulation: 1. Packing prevents access CH 3 2. Acetylation ( C O )
IBBL: Introduction to Biobanks and their services
 IBBL: Introduction to Biobanks and their services HighTech meets Health, Oss (NL), 15 th June 2017 Monica Marchese, PhD Biomarker Validation Scientist AN ACCREDITED & CERTIFIED BIOBANK INFRASTRUCTURE FOR
IBBL: Introduction to Biobanks and their services HighTech meets Health, Oss (NL), 15 th June 2017 Monica Marchese, PhD Biomarker Validation Scientist AN ACCREDITED & CERTIFIED BIOBANK INFRASTRUCTURE FOR
Visions on the role of Statisticians in the Pharmaceutical Industry. Merete Jørgensen President EFSPI VP Biostatistics, Novo Nordisk A/S
 Visions on the role of Statisticians in the Pharmaceutical Industry Merete Jørgensen President EFSPI VP Biostatistics, Novo Nordisk A/S Overview of presentation Vision & Mission A Historical view New Statistical
Visions on the role of Statisticians in the Pharmaceutical Industry Merete Jørgensen President EFSPI VP Biostatistics, Novo Nordisk A/S Overview of presentation Vision & Mission A Historical view New Statistical
MOSAIQUES DIAGNOSTICS
 MOSAIQUES DIAGNOSTICS CLINICAL PROTEOMICS IN DRUG DEVELOPMENT INFORMATION Clinical Proteomics for early and differential diagnosis FDA Letter of Support Possibilities of a companion test in Drug Development
MOSAIQUES DIAGNOSTICS CLINICAL PROTEOMICS IN DRUG DEVELOPMENT INFORMATION Clinical Proteomics for early and differential diagnosis FDA Letter of Support Possibilities of a companion test in Drug Development
Introducing a Highly Integrated Approach to Translational Research: Biomarker Data Management, Data Integration, and Collaboration
 Introducing a Highly Integrated Approach to Translational Research: Biomarker Data Management, Data Integration, and Collaboration 1 2 Translational Informatics Overview Precision s suite of services is
Introducing a Highly Integrated Approach to Translational Research: Biomarker Data Management, Data Integration, and Collaboration 1 2 Translational Informatics Overview Precision s suite of services is
Short Course: Adaptive Clinical Trials
 Short Course: Adaptive Clinical Trials Presented at the 2 Annual Meeting of the Society for Clinical Trials Vancouver, Canada Roger J. Lewis, MD, PhD Department of Emergency Medicine Harbor-UCLA Medical
Short Course: Adaptive Clinical Trials Presented at the 2 Annual Meeting of the Society for Clinical Trials Vancouver, Canada Roger J. Lewis, MD, PhD Department of Emergency Medicine Harbor-UCLA Medical
BIOCRATES Life Sciences AG Short Company Presentation
 BIOCRATES Life Sciences AG Short Company Presentation Dr. Alexandra C. Gruber MBA MMFin Director Business Development / Marketing Austria Showcase Life Science Japan, Tokyo, December 2010 BIOCRATES Life
BIOCRATES Life Sciences AG Short Company Presentation Dr. Alexandra C. Gruber MBA MMFin Director Business Development / Marketing Austria Showcase Life Science Japan, Tokyo, December 2010 BIOCRATES Life
QIAGEN s NGS Solutions for Biomarkers NGS & Bioinformatics team QIAGEN (Suzhou) Translational Medicine Co.,Ltd
 QIAGEN s NGS Solutions for Biomarkers NGS & Bioinformatics team QIAGEN (Suzhou) Translational Medicine Co.,Ltd 1 Our current NGS & Bioinformatics Platform 2 Our NGS workflow and applications 3 QIAGEN s
QIAGEN s NGS Solutions for Biomarkers NGS & Bioinformatics team QIAGEN (Suzhou) Translational Medicine Co.,Ltd 1 Our current NGS & Bioinformatics Platform 2 Our NGS workflow and applications 3 QIAGEN s
CENTRAL NERVOUS SYSTEM (CNS) BIOMARKERS: TECHNOLOGIES AND GLOBAL MARKETS
 CENTRAL NERVOUS SYSTEM (CNS) : TECHNOLOGIES AND GLOBAL MARKETS BIO074C March 2016 Jackson Highsmith Project Analyst ISBN: 1-62296-246-X BCC Research 49 Walnut Park, Building 2 Wellesley, MA 02481 USA 866-285-7215
CENTRAL NERVOUS SYSTEM (CNS) : TECHNOLOGIES AND GLOBAL MARKETS BIO074C March 2016 Jackson Highsmith Project Analyst ISBN: 1-62296-246-X BCC Research 49 Walnut Park, Building 2 Wellesley, MA 02481 USA 866-285-7215
Official Letter from the DOH
 Issued Date 2009/04/02 Issued by DOH Ref. No 0980316268 RE The DOH issued an official letter to announce the implementation of the Guideline for BA/BE Studies on April 2, 2009 (Ref. No. 0980316265). Please
Issued Date 2009/04/02 Issued by DOH Ref. No 0980316268 RE The DOH issued an official letter to announce the implementation of the Guideline for BA/BE Studies on April 2, 2009 (Ref. No. 0980316265). Please
ICH Topic E16 Genomic Biomarkers Related to Drug Response: Context, Structure and Format of Qualification Submissions. Step 3
 European Medicines Agency June 2009 EMEA/CHMP/ICH/380636/2009 ICH Topic E16 Genomic Biomarkers Related to Drug Response: Context, Structure and Format of Qualification Submissions Step 3 NOTE FOR GUIDANCE
European Medicines Agency June 2009 EMEA/CHMP/ICH/380636/2009 ICH Topic E16 Genomic Biomarkers Related to Drug Response: Context, Structure and Format of Qualification Submissions Step 3 NOTE FOR GUIDANCE
Lecture 23: Clinical and Biomedical Applications of Proteomics; Proteomics Industry
 Lecture 23: Clinical and Biomedical Applications of Proteomics; Proteomics Industry Clinical proteomics is the application of proteomic approach to the field of medicine. Proteome of an organism changes
Lecture 23: Clinical and Biomedical Applications of Proteomics; Proteomics Industry Clinical proteomics is the application of proteomic approach to the field of medicine. Proteome of an organism changes
Towards unbiased biomarker discovery
 Towards unbiased biomarker discovery High-throughput molecular profiling technologies are routinely applied for biomarker discovery to make the drug discovery process more efficient and enable personalised
Towards unbiased biomarker discovery High-throughput molecular profiling technologies are routinely applied for biomarker discovery to make the drug discovery process more efficient and enable personalised
GENE EXPRESSION REAGENTS MARKETS (SAMPLE COPY, NOT FOR RESALE)
 TriMark Publications April 2007 Volume: TMRGER07-0401 GENE EXPRESSION REAGENTS MARKETS (SAMPLE COPY, NOT FOR RESALE) Trends, Industry Participants, Product Overviews and Market Drivers TABLE OF CONTENTS
TriMark Publications April 2007 Volume: TMRGER07-0401 GENE EXPRESSION REAGENTS MARKETS (SAMPLE COPY, NOT FOR RESALE) Trends, Industry Participants, Product Overviews and Market Drivers TABLE OF CONTENTS
Application of Deep Learning to Drug Discovery
 Application of Deep Learning to Drug Discovery Hiroshi Tanaka Tohoku Medical Megabank Orga nization, Tohoku University Current Situation of Drug Discovery Rapid increase of R&D expenditure More than 1B
Application of Deep Learning to Drug Discovery Hiroshi Tanaka Tohoku Medical Megabank Orga nization, Tohoku University Current Situation of Drug Discovery Rapid increase of R&D expenditure More than 1B
DISCOVERY AND VALIDATION OF TARGETS AND BIOMARKERS BY MASS SPECTROMETRY-BASED PROTEOMICS. September, 2011
 DISCOVERY AND VALIDATION OF TARGETS AND BIOMARKERS BY MASS SPECTROMETRY-BASED PROTEOMICS September, 2011 1 CAPRION PROTEOMICS Leading proteomics-based service provider - Biomarker and target discovery
DISCOVERY AND VALIDATION OF TARGETS AND BIOMARKERS BY MASS SPECTROMETRY-BASED PROTEOMICS September, 2011 1 CAPRION PROTEOMICS Leading proteomics-based service provider - Biomarker and target discovery
TAMU: PROTEOMICS SPECTRA 1 What Are Proteomics Spectra? DNA makes RNA makes Protein
 The Analysis of Proteomics Spectra from Serum Samples Jeffrey S. Morris Department of Biostatistics MD Anderson Cancer Center TAMU: PROTEOMICS SPECTRA 1 What Are Proteomics Spectra? DNA makes RNA makes
The Analysis of Proteomics Spectra from Serum Samples Jeffrey S. Morris Department of Biostatistics MD Anderson Cancer Center TAMU: PROTEOMICS SPECTRA 1 What Are Proteomics Spectra? DNA makes RNA makes
Whole Transcriptome Analysis of Illumina RNA- Seq Data. Ryan Peters Field Application Specialist
 Whole Transcriptome Analysis of Illumina RNA- Seq Data Ryan Peters Field Application Specialist Partek GS in your NGS Pipeline Your Start-to-Finish Solution for Analysis of Next Generation Sequencing Data
Whole Transcriptome Analysis of Illumina RNA- Seq Data Ryan Peters Field Application Specialist Partek GS in your NGS Pipeline Your Start-to-Finish Solution for Analysis of Next Generation Sequencing Data
ASIAN PATENT ATTORNEYS ASSOCIATION Recognized Group of Korea. Report to Emerging IP Rights Committee 2012, Chiang Mai
 ASIAN PATENT ATTORNEYS ASSOCIATION Recognized Group of Korea Report to Emerging IP Rights Committee 2012, Chiang Mai SPECIAL TOPIC REPORT ON Business Methods and the Laws of Nature, As Discussed by the
ASIAN PATENT ATTORNEYS ASSOCIATION Recognized Group of Korea Report to Emerging IP Rights Committee 2012, Chiang Mai SPECIAL TOPIC REPORT ON Business Methods and the Laws of Nature, As Discussed by the
Gene expression connectivity mapping and its application to Cat-App
 Gene expression connectivity mapping and its application to Cat-App Shu-Dong Zhang Northern Ireland Centre for Stratified Medicine University of Ulster Outline TITLE OF THE PRESENTATION Gene expression
Gene expression connectivity mapping and its application to Cat-App Shu-Dong Zhang Northern Ireland Centre for Stratified Medicine University of Ulster Outline TITLE OF THE PRESENTATION Gene expression
Predictive and Causal Modeling in the Health Sciences. Sisi Ma MS, MS, PhD. New York University, Center for Health Informatics and Bioinformatics
 Predictive and Causal Modeling in the Health Sciences Sisi Ma MS, MS, PhD. New York University, Center for Health Informatics and Bioinformatics 1 Exponentially Rapid Data Accumulation Protein Sequencing
Predictive and Causal Modeling in the Health Sciences Sisi Ma MS, MS, PhD. New York University, Center for Health Informatics and Bioinformatics 1 Exponentially Rapid Data Accumulation Protein Sequencing
CRO partner in Rx/CDx Co-Development
 CRO partner in Rx/CDx Co-Development DEDICATED DIALOGUE A sponsored roundtable discussion published in Pharmaceutical Executive Two Covance executives discuss a CRO s role in supporting Companion Diagnostics
CRO partner in Rx/CDx Co-Development DEDICATED DIALOGUE A sponsored roundtable discussion published in Pharmaceutical Executive Two Covance executives discuss a CRO s role in supporting Companion Diagnostics
Disclaimer This presentation expresses my personal views on this topic and must not be interpreted as the regulatory views or the policy of the FDA
 On multiplicity problems related to multiple endpoints of controlled clinical trials Mohammad F. Huque, Ph.D. Div of Biometrics IV, Office of Biostatistics OTS, CDER/FDA JSM, Vancouver, August 2010 Disclaimer
On multiplicity problems related to multiple endpoints of controlled clinical trials Mohammad F. Huque, Ph.D. Div of Biometrics IV, Office of Biostatistics OTS, CDER/FDA JSM, Vancouver, August 2010 Disclaimer
Integrated Radiology & Pathology Diagnostic Services: The Time is Now at UCLA
 Integrated Radiology & Pathology Diagnostic Services: The Time is Now at UCLA Scott W Binder MD Pritzker Professor of Clinical Pathology and Dermatology Senior Vice Chair Director, Pathology Clinical Services
Integrated Radiology & Pathology Diagnostic Services: The Time is Now at UCLA Scott W Binder MD Pritzker Professor of Clinical Pathology and Dermatology Senior Vice Chair Director, Pathology Clinical Services
Optimization of RNAi Targets on the Human Transcriptome Ahmet Arslan Kurdoglu Computational Biosciences Program Arizona State University
 Optimization of RNAi Targets on the Human Transcriptome Ahmet Arslan Kurdoglu Computational Biosciences Program Arizona State University my background Undergraduate Degree computer systems engineer (ASU
Optimization of RNAi Targets on the Human Transcriptome Ahmet Arslan Kurdoglu Computational Biosciences Program Arizona State University my background Undergraduate Degree computer systems engineer (ASU
Reflection paper on co-development of pharmacogenomic biomarkers and Assays in the context of drug development
 1 2 3 24 June 2010 EMA/CHMP/641298/2008 Committee for Medicinal Products for Human Use (CHMP) 4 5 6 7 Reflection paper on co-development of pharmacogenomic biomarkers and Assays in the context of drug
1 2 3 24 June 2010 EMA/CHMP/641298/2008 Committee for Medicinal Products for Human Use (CHMP) 4 5 6 7 Reflection paper on co-development of pharmacogenomic biomarkers and Assays in the context of drug
Proteomics And Cancer Biomarker Discovery. Dr. Zahid Khan Institute of chemical Sciences (ICS) University of Peshawar. Overview. Cancer.
 Proteomics And Cancer Biomarker Discovery Dr. Zahid Khan Institute of chemical Sciences (ICS) University of Peshawar Overview Proteomics Cancer Aims Tools Data Base search Challenges Summary 1 Overview
Proteomics And Cancer Biomarker Discovery Dr. Zahid Khan Institute of chemical Sciences (ICS) University of Peshawar Overview Proteomics Cancer Aims Tools Data Base search Challenges Summary 1 Overview
Feature Selection of Gene Expression Data for Cancer Classification: A Review
 Available online at www.sciencedirect.com ScienceDirect Procedia Computer Science 50 (2015 ) 52 57 2nd International Symposium on Big Data and Cloud Computing (ISBCC 15) Feature Selection of Gene Expression
Available online at www.sciencedirect.com ScienceDirect Procedia Computer Science 50 (2015 ) 52 57 2nd International Symposium on Big Data and Cloud Computing (ISBCC 15) Feature Selection of Gene Expression
An Overview of Bayesian Adaptive Clinical Trial Design
 An Overview of Bayesian Adaptive Clinical Trial Design Roger J. Lewis, MD, PhD Department of Emergency Medicine Harbor-UCLA Medical Center David Geffen School of Medicine at UCLA Los Angeles Biomedical
An Overview of Bayesian Adaptive Clinical Trial Design Roger J. Lewis, MD, PhD Department of Emergency Medicine Harbor-UCLA Medical Center David Geffen School of Medicine at UCLA Los Angeles Biomedical
Draft agreed by Scientific Advice Working Party 5 September Adopted by CHMP for release for consultation 19 September
 23 January 2014 EMA/CHMP/SAWP/757052/2013 Committee for Medicinal Products for Human Use (CHMP) Qualification Opinion of MCP-Mod as an efficient statistical methodology for model-based design and analysis
23 January 2014 EMA/CHMP/SAWP/757052/2013 Committee for Medicinal Products for Human Use (CHMP) Qualification Opinion of MCP-Mod as an efficient statistical methodology for model-based design and analysis
DNA Microarray Technology and Data Analysis in Cancer Research Downloaded from
 This page intentionally left blank Shaoguang Li University of Massachusetts Medical School, USA Dongguang Li Edith Cowan University, Australia World Scientific N E W J E R S E Y L O N D O N S I N G A P
This page intentionally left blank Shaoguang Li University of Massachusetts Medical School, USA Dongguang Li Edith Cowan University, Australia World Scientific N E W J E R S E Y L O N D O N S I N G A P
DNA Microarray Data Oligonucleotide Arrays
 DNA Microarray Data Oligonucleotide Arrays Sandrine Dudoit, Robert Gentleman, Rafael Irizarry, and Yee Hwa Yang Bioconductor Short Course 2003 Copyright 2002, all rights reserved Biological question Experimental
DNA Microarray Data Oligonucleotide Arrays Sandrine Dudoit, Robert Gentleman, Rafael Irizarry, and Yee Hwa Yang Bioconductor Short Course 2003 Copyright 2002, all rights reserved Biological question Experimental
Molecular Diagnostics
 Molecular Diagnostics Part II: Regulations, Markets & Companies By Prof. K. K. Jain MD, FRACS, FFPM Jain PharmaBiotech Basel, Switzerland May 2018 A Jain PharmaBiotech Report A U T H O R ' S B I O G R
Molecular Diagnostics Part II: Regulations, Markets & Companies By Prof. K. K. Jain MD, FRACS, FFPM Jain PharmaBiotech Basel, Switzerland May 2018 A Jain PharmaBiotech Report A U T H O R ' S B I O G R
Adaptive Design for Clinical Trials
 Adaptive Design for Clinical Trials Mark Chang Millennium Pharmaceuticals, Inc., Cambridge, MA 02139,USA (e-mail: Mark.Chang@Statisticians.org) Abstract. Adaptive design is a trial design that allows modifications
Adaptive Design for Clinical Trials Mark Chang Millennium Pharmaceuticals, Inc., Cambridge, MA 02139,USA (e-mail: Mark.Chang@Statisticians.org) Abstract. Adaptive design is a trial design that allows modifications
6. GENE EXPRESSION ANALYSIS MICROARRAYS
 6. GENE EXPRESSION ANALYSIS MICROARRAYS BIOINFORMATICS COURSE MTAT.03.239 16.10.2013 GENE EXPRESSION ANALYSIS MICROARRAYS Slides adapted from Konstantin Tretyakov s 2011/2012 and Priit Adlers 2010/2011
6. GENE EXPRESSION ANALYSIS MICROARRAYS BIOINFORMATICS COURSE MTAT.03.239 16.10.2013 GENE EXPRESSION ANALYSIS MICROARRAYS Slides adapted from Konstantin Tretyakov s 2011/2012 and Priit Adlers 2010/2011
Intro to Microarray Analysis. Courtesy of Professor Dan Nettleton Iowa State University (with some edits)
 Intro to Microarray Analysis Courtesy of Professor Dan Nettleton Iowa State University (with some edits) Some Basic Biology Genes are DNA sequences that code for proteins. (e.g. gene lengths perhaps 1000
Intro to Microarray Analysis Courtesy of Professor Dan Nettleton Iowa State University (with some edits) Some Basic Biology Genes are DNA sequences that code for proteins. (e.g. gene lengths perhaps 1000
Injectable modified release products
 Guideline on the pharmacokinetic and clinical evaluation of modified release dosage forms (EMA/CPMP/EWP/280/96 Corr1) Injectable modified release products Dr Sotiris Michaleas, National Expert for the
Guideline on the pharmacokinetic and clinical evaluation of modified release dosage forms (EMA/CPMP/EWP/280/96 Corr1) Injectable modified release products Dr Sotiris Michaleas, National Expert for the
International Transfers of Personal Data at sanofi-aventis R & D
 International Transfers of Personal Data at sanofi-aventis R & D Pierre-Yves Lastic, PhD Senior Director, Standards Management & Data Privacy Sanofi-aventis R&D CONFERENCE ON INTERNATIONAL TRANSFERS OF
International Transfers of Personal Data at sanofi-aventis R & D Pierre-Yves Lastic, PhD Senior Director, Standards Management & Data Privacy Sanofi-aventis R&D CONFERENCE ON INTERNATIONAL TRANSFERS OF
Lecture #1. Introduction to microarray technology
 Lecture #1 Introduction to microarray technology Outline General purpose Microarray assay concept Basic microarray experimental process cdna/two channel arrays Oligonucleotide arrays Exon arrays Comparing
Lecture #1 Introduction to microarray technology Outline General purpose Microarray assay concept Basic microarray experimental process cdna/two channel arrays Oligonucleotide arrays Exon arrays Comparing
Genomic Clinical Trials and Predictive Medicine
 Genomic Clinical Trials and Predictive Medicine Genomics is having a major impact on therapeutics development in medicine. This book contains up-to-date information on the use of genomics in the design
Genomic Clinical Trials and Predictive Medicine Genomics is having a major impact on therapeutics development in medicine. This book contains up-to-date information on the use of genomics in the design
Genomics And Pharmacogenomics In Anticancer Drug Development And Clinical Response (Cancer Drug Discovery And Development)
 Genomics And Pharmacogenomics In Anticancer Drug Development And Clinical Response (Cancer Drug Discovery And Development) Strategies for modern biomarker and drug - Oct 3, 2014 Sequencing the cancer genome
Genomics And Pharmacogenomics In Anticancer Drug Development And Clinical Response (Cancer Drug Discovery And Development) Strategies for modern biomarker and drug - Oct 3, 2014 Sequencing the cancer genome
Proteomics. Manickam Sugumaran. Department of Biology University of Massachusetts Boston, MA 02125
 Proteomics Manickam Sugumaran Department of Biology University of Massachusetts Boston, MA 02125 Genomic studies produced more than 75,000 potential gene sequence targets. (The number may be even higher
Proteomics Manickam Sugumaran Department of Biology University of Massachusetts Boston, MA 02125 Genomic studies produced more than 75,000 potential gene sequence targets. (The number may be even higher
2D gel Western blotting using antibodies against ubiquitin, SUMO and acetyl PTM
 2D gel Western blotting using antibodies against ubiquitin, SUMO and acetyl PTM Nancy Kendrick, Jon Johansen & Matt Hoelter, Kendrick Labs Inc www.kendricklabs.com Talk Outline Significance Method description
2D gel Western blotting using antibodies against ubiquitin, SUMO and acetyl PTM Nancy Kendrick, Jon Johansen & Matt Hoelter, Kendrick Labs Inc www.kendricklabs.com Talk Outline Significance Method description
Cytomics in Action: Cytokine Network Cytometry
 Cytomics in Action: Cytokine Network Cytometry Jonni S. Moore, Ph.D. Director, Clinical and Research Flow Cytometry and PathBioResource Associate Professor of Pathology & Laboratory Medicine University
Cytomics in Action: Cytokine Network Cytometry Jonni S. Moore, Ph.D. Director, Clinical and Research Flow Cytometry and PathBioResource Associate Professor of Pathology & Laboratory Medicine University
Controversy In Pharmacogenomics
 Controversy In Pharmacogenomics Alejandra M. De Jesús-Soto, Mark R. Ruprecht, Patricia Vera-González Principal Investigator: Rafael Irizarry, PhD Graduate Assistant: William Townes, MS Harvard T.H. Chan
Controversy In Pharmacogenomics Alejandra M. De Jesús-Soto, Mark R. Ruprecht, Patricia Vera-González Principal Investigator: Rafael Irizarry, PhD Graduate Assistant: William Townes, MS Harvard T.H. Chan
Humboldt Universität zu Berlin. Grundlagen der Bioinformatik SS Microarrays. Lecture
 Humboldt Universität zu Berlin Microarrays Grundlagen der Bioinformatik SS 2017 Lecture 6 09.06.2017 Agenda 1.mRNA: Genomic background 2.Overview: Microarray 3.Data-analysis: Quality control & normalization
Humboldt Universität zu Berlin Microarrays Grundlagen der Bioinformatik SS 2017 Lecture 6 09.06.2017 Agenda 1.mRNA: Genomic background 2.Overview: Microarray 3.Data-analysis: Quality control & normalization
Terminology for personalized medicine
 Terminology for personalized medicine Sarah Ali-Khan 1 PhD, Stephanie Kowal 2, Westerly Luth 2, Richard Gold 1 SJD, and Tania Bubela 2 PhD JD 1. Centre for Intellectual Property Policy (CIPP), Faculty
Terminology for personalized medicine Sarah Ali-Khan 1 PhD, Stephanie Kowal 2, Westerly Luth 2, Richard Gold 1 SJD, and Tania Bubela 2 PhD JD 1. Centre for Intellectual Property Policy (CIPP), Faculty
Outline. Analysis of Microarray Data. Most important design question. General experimental issues
 Outline Analysis of Microarray Data Lecture 1: Experimental Design and Data Normalization Introduction to microarrays Experimental design Data normalization Other data transformation Exercises George Bell,
Outline Analysis of Microarray Data Lecture 1: Experimental Design and Data Normalization Introduction to microarrays Experimental design Data normalization Other data transformation Exercises George Bell,
Proteomics and Cancer
 Proteomics and Cancer Japan Society for the Promotion of Science (JSPS) Science Dialogue Program at Niitsu Senior High School Niitsu, Niigata September 4th 2006 Vladimir Valera, M.D, PhD JSPS Postdoctoral
Proteomics and Cancer Japan Society for the Promotion of Science (JSPS) Science Dialogue Program at Niitsu Senior High School Niitsu, Niigata September 4th 2006 Vladimir Valera, M.D, PhD JSPS Postdoctoral
Analysis of Microarray Data
 Analysis of Microarray Data Lecture 1: Experimental Design and Data Normalization George Bell, Ph.D. Senior Bioinformatics Scientist Bioinformatics and Research Computing Whitehead Institute Outline Introduction
Analysis of Microarray Data Lecture 1: Experimental Design and Data Normalization George Bell, Ph.D. Senior Bioinformatics Scientist Bioinformatics and Research Computing Whitehead Institute Outline Introduction
Parameter Estimation for the Exponential-Normal Convolution Model
 Parameter Estimation for the Exponential-Normal Convolution Model Monnie McGee & Zhongxue Chen cgee@smu.edu, zhongxue@smu.edu. Department of Statistical Science Southern Methodist University ENAR Spring
Parameter Estimation for the Exponential-Normal Convolution Model Monnie McGee & Zhongxue Chen cgee@smu.edu, zhongxue@smu.edu. Department of Statistical Science Southern Methodist University ENAR Spring
Supplementary Figure 1. Utilization of publicly available antibodies in different applications.
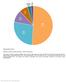 Supplementary Figure 1 Utilization of publicly available antibodies in different applications. The fraction of publicly available antibodies toward human protein targets is shown with data for the following
Supplementary Figure 1 Utilization of publicly available antibodies in different applications. The fraction of publicly available antibodies toward human protein targets is shown with data for the following
PAREXEL GENOMIC MEDICINE SERVICES. Applying genomics to enhance your drug development journey
 PAREXEL GENOMIC MEDICINE SERVICES Applying genomics to enhance your drug development journey YOUR JOURNEY. OUR MISSION. Genomic expertise to simplify the route to product approval and maximize patient
PAREXEL GENOMIC MEDICINE SERVICES Applying genomics to enhance your drug development journey YOUR JOURNEY. OUR MISSION. Genomic expertise to simplify the route to product approval and maximize patient
MICROARRAYS+SEQUENCING
 MICROARRAYS+SEQUENCING The most efficient way to advance genomics research Down to a Science. www.affymetrix.com/downtoascience Affymetrix GeneChip Expression Technology Complementing your Next-Generation
MICROARRAYS+SEQUENCING The most efficient way to advance genomics research Down to a Science. www.affymetrix.com/downtoascience Affymetrix GeneChip Expression Technology Complementing your Next-Generation
Medical Genetics Component
 2008 PATIENTS AND POPULATIONS August 7-29, 2008 MEDICAL GENETICS COMPONENT SEQUENCE DIRECTOR Thomas D. Gelehrter, M.D. Professor Emeritus of Human Genetics and Internal Medicine Department of Human Genetics
2008 PATIENTS AND POPULATIONS August 7-29, 2008 MEDICAL GENETICS COMPONENT SEQUENCE DIRECTOR Thomas D. Gelehrter, M.D. Professor Emeritus of Human Genetics and Internal Medicine Department of Human Genetics
Celerion s Symposia Series: Bridging the Gap from Phase I to Proof-of-Concept. San Francisco, CA Tue 8 th, Apr 2014
 Celerion s Symposia Series: Bridging the Gap from Phase I to Proof-of-Concept San Francisco, CA Tue 8 th, Apr 2014 Mind the Gap: Elements of a Bridging Strategy J. Fred Pritchard, Ph.D. Vice President,
Celerion s Symposia Series: Bridging the Gap from Phase I to Proof-of-Concept San Francisco, CA Tue 8 th, Apr 2014 Mind the Gap: Elements of a Bridging Strategy J. Fred Pritchard, Ph.D. Vice President,
Bioavailability and Bioequivalence Studies
 Bioavailability and Bioequivalence Studies Standard Approach Part I: Design and Conduct H. Rettig, Ph.D. LLC www.ivivc.com Note for Guidance on the Investigation of Bioavailability and Bioequivalence CPMP/EWP/QWP/1401/98
Bioavailability and Bioequivalence Studies Standard Approach Part I: Design and Conduct H. Rettig, Ph.D. LLC www.ivivc.com Note for Guidance on the Investigation of Bioavailability and Bioequivalence CPMP/EWP/QWP/1401/98
Gene Regulation Solutions. Microarrays and Next-Generation Sequencing
 Gene Regulation Solutions Microarrays and Next-Generation Sequencing Gene Regulation Solutions The Microarrays Advantage Microarrays Lead the Industry in: Comprehensive Content SurePrint G3 Human Gene
Gene Regulation Solutions Microarrays and Next-Generation Sequencing Gene Regulation Solutions The Microarrays Advantage Microarrays Lead the Industry in: Comprehensive Content SurePrint G3 Human Gene
Introduction to Bioinformatics. Fabian Hoti 6.10.
 Introduction to Bioinformatics Fabian Hoti 6.10. Analysis of Microarray Data Introduction Different types of microarrays Experiment Design Data Normalization Feature selection/extraction Clustering Introduction
Introduction to Bioinformatics Fabian Hoti 6.10. Analysis of Microarray Data Introduction Different types of microarrays Experiment Design Data Normalization Feature selection/extraction Clustering Introduction
Outline CLINICALLY RELEVANT SPECIFICATIONS. ISPE Process Validation Conference September 2017 Bethesda, MD
 CLINICALLY RELEVANT SPECIFICATIONS Patrick J Marroum Ph.D. Senior Director and ACOS Senior Research Fellow Department of Clinical Pharmacology and Pharmacometrics Abbvie Pharmaceuticals Outline CMC variables
CLINICALLY RELEVANT SPECIFICATIONS Patrick J Marroum Ph.D. Senior Director and ACOS Senior Research Fellow Department of Clinical Pharmacology and Pharmacometrics Abbvie Pharmaceuticals Outline CMC variables
Synthetic Biology. Sustainable Energy. Therapeutics Industrial Enzymes. Agriculture. Accelerating Discoveries, Expanding Possibilities. Design.
 Synthetic Biology Accelerating Discoveries, Expanding Possibilities Sustainable Energy Therapeutics Industrial Enzymes Agriculture Design Build Generate Solutions to Advance Synthetic Biology Research
Synthetic Biology Accelerating Discoveries, Expanding Possibilities Sustainable Energy Therapeutics Industrial Enzymes Agriculture Design Build Generate Solutions to Advance Synthetic Biology Research
Stem Cell Services. Driving Innovation for Stem Cell Researchers
 Driving Innovation for Stem Cell Researchers Stem Cell Services Partner with us and have access to the most advanced and comprehensive stem cell services available today. 675 W. Kendall St. Cambridge,
Driving Innovation for Stem Cell Researchers Stem Cell Services Partner with us and have access to the most advanced and comprehensive stem cell services available today. 675 W. Kendall St. Cambridge,
Personalized. Health in Canada
 Personalized Health in Canada Canadian Institutes of Health Research Personalized Medicine Signature Initiative 2010-2013 0 Dr. Morag Park CIHR Institute of Cancer Research Dr. Paul Lasko CIHR Institute
Personalized Health in Canada Canadian Institutes of Health Research Personalized Medicine Signature Initiative 2010-2013 0 Dr. Morag Park CIHR Institute of Cancer Research Dr. Paul Lasko CIHR Institute
+ + + MALARIA HIV/AIDS TUBERCULOSIS LEISHMANIASIS CHAGAS DISEASE DENGUE EBOLA ZIKA
 At the Center for Infectious Disease Research, we use systems biology approaches and cutting-edge technologies to accelerate the development of life-saving drugs, diagnostics and treatments for the world
At the Center for Infectious Disease Research, we use systems biology approaches and cutting-edge technologies to accelerate the development of life-saving drugs, diagnostics and treatments for the world
TOTAL CANCER CARE: CREATING PARTNERSHIPS TO ADDRESS PATIENT NEEDS
 TOTAL CANCER CARE: CREATING PARTNERSHIPS TO ADDRESS PATIENT NEEDS William S. Dalton, PhD, MD CEO, M2Gen & Director, Personalized Medicine Institute, Moffitt Cancer Center JULY 15, 2013 MOFFITT CANCER CENTER
TOTAL CANCER CARE: CREATING PARTNERSHIPS TO ADDRESS PATIENT NEEDS William S. Dalton, PhD, MD CEO, M2Gen & Director, Personalized Medicine Institute, Moffitt Cancer Center JULY 15, 2013 MOFFITT CANCER CENTER
A Risk-based Approach for In Vitro Companion Diagnostics Device FDA Approval Process Associated with Therapies that have Breakthrough Designation
 A Risk-based Approach for In Vitro Companion Diagnostics Device FDA Approval Process Associated with Therapies that have Breakthrough Designation A Risk-based Approach for In Vitro Companion Diagnostics
A Risk-based Approach for In Vitro Companion Diagnostics Device FDA Approval Process Associated with Therapies that have Breakthrough Designation A Risk-based Approach for In Vitro Companion Diagnostics
Venture Philanthropy Models: The Leukemia & Lymphoma Society's Therapy Acceleration Program. A FasterCures Webinar June 19, 2013
 Venture Philanthropy Models: The Leukemia & Lymphoma Society's Therapy Acceleration Program A FasterCures Webinar June 19, 2013 What is FasterCures? Founded in 2003, our mission is to save lives by saving
Venture Philanthropy Models: The Leukemia & Lymphoma Society's Therapy Acceleration Program A FasterCures Webinar June 19, 2013 What is FasterCures? Founded in 2003, our mission is to save lives by saving
Overview of Biologics Testing and Evaluation: Regulatory Requirements and Expectations. Audrey Chang, PhD, Senior Director Development Services
 Overview of Biologics Testing and Evaluation: Regulatory Requirements and Expectations Audrey Chang, PhD, Senior Director Development Services Definition of Biologics: PHS Act, section 351 Virus, therapeutic
Overview of Biologics Testing and Evaluation: Regulatory Requirements and Expectations Audrey Chang, PhD, Senior Director Development Services Definition of Biologics: PHS Act, section 351 Virus, therapeutic
Developing an Accurate and Precise Companion Diagnostic Assay for Targeted Therapies in DLBCL
 Developing an Accurate and Precise Companion Diagnostic Assay for Targeted Therapies in DLBCL James Storhoff, Ph.D. Senior Manager, Diagnostic Test Development World Cdx, Boston, Sep. 10th Molecules That
Developing an Accurate and Precise Companion Diagnostic Assay for Targeted Therapies in DLBCL James Storhoff, Ph.D. Senior Manager, Diagnostic Test Development World Cdx, Boston, Sep. 10th Molecules That
Regulatory Approaches for New Drugs: BA/BE of Topical Drug Products
 Regulatory Approaches for New Drugs: BA/BE of Topical Drug Products CAPT E. Dennis Bashaw, Pharm.D Director, Division of Clinical Pharmacology-3 Office of Clinical Pharmacology Office of Translational
Regulatory Approaches for New Drugs: BA/BE of Topical Drug Products CAPT E. Dennis Bashaw, Pharm.D Director, Division of Clinical Pharmacology-3 Office of Clinical Pharmacology Office of Translational
Industry Academic Collaboration: A Key to Successful Involvement of Patients Early in Clinical Development
 Industry Academic Collaboration: A Key to Successful Involvement of Patients Early in Clinical Development Aernout van Haarst PhD Director, European Corporate Development Feb 2016 Industry Academic Collaboration
Industry Academic Collaboration: A Key to Successful Involvement of Patients Early in Clinical Development Aernout van Haarst PhD Director, European Corporate Development Feb 2016 Industry Academic Collaboration
The University of Texas MD Anderson Cancer Center UTHealth Graduate School of Biomedical Sciences Catalog Addendum
 The University of Texas MD Anderson Cancer Center UTHealth 2016-2018 Catalog Addendum GSBS 2016-18 Catalog Addendum Table of Contents School Name Change... 1 Areas of Research Concentration Changes...
The University of Texas MD Anderson Cancer Center UTHealth 2016-2018 Catalog Addendum GSBS 2016-18 Catalog Addendum Table of Contents School Name Change... 1 Areas of Research Concentration Changes...
Dana-Farber Cancer Institute Speeds Medical Research with Advanced Data Warehouse
 Dana-Farber Cancer Institute Speeds Medical Research with Advanced Data Warehouse Dana-Farber Cancer Institute Boston, MA www.dana-farber.org Industry: Healthcare Annual Revenue: US$665.7 million Employees:
Dana-Farber Cancer Institute Speeds Medical Research with Advanced Data Warehouse Dana-Farber Cancer Institute Boston, MA www.dana-farber.org Industry: Healthcare Annual Revenue: US$665.7 million Employees:
First Annual Biomarker Symposium Quest Diagnostics Clinical Trials
 First Annual Biomarker Symposium Quest Diagnostics Clinical Trials Terry Robins, Ph.D. Director Biomarker R&D and Scientific Affairs Quest Diagnostics Clinical Trials Key Considerations: Biomarker Development
First Annual Biomarker Symposium Quest Diagnostics Clinical Trials Terry Robins, Ph.D. Director Biomarker R&D and Scientific Affairs Quest Diagnostics Clinical Trials Key Considerations: Biomarker Development
Human Genomics, Precision Medicine, and Advancing Human Health. The Human Genome. The Origin of Genomics : 1987
 Human Genomics, Precision Medicine, and Advancing Human Health Eric Green, M.D., Ph.D. Director, NHGRI The Human Genome Cells Nucleus Chromosome DNA Human Genome: 3 Billion Bases (letters) The Origin of
Human Genomics, Precision Medicine, and Advancing Human Health Eric Green, M.D., Ph.D. Director, NHGRI The Human Genome Cells Nucleus Chromosome DNA Human Genome: 3 Billion Bases (letters) The Origin of
CRYSTAL CITY V REDUX: GUIDANCE FOR INDUSTRY BIOANALYTICAL METHOD VALIDATION DRAFT GUIDANCE (2013) VI. ADDITIONAL ISSUES
 CRYSTAL CITY V REDUX: GUIDANCE FOR INDUSTRY BIOANALYTICAL METHOD VALIDATION DRAFT GUIDANCE (2013) VI. ADDITIONAL ISSUES DELAWARE VALLEY DRUG METABOLISM DISCUSSION GROUP STEVE PICCOLI, BMS RAND JENKINS,
CRYSTAL CITY V REDUX: GUIDANCE FOR INDUSTRY BIOANALYTICAL METHOD VALIDATION DRAFT GUIDANCE (2013) VI. ADDITIONAL ISSUES DELAWARE VALLEY DRUG METABOLISM DISCUSSION GROUP STEVE PICCOLI, BMS RAND JENKINS,
Comments from the FDA Working Group on SUBGROUP ANALYSES. Estelle Russek-Cohen, Ph.D. U.S. Food and Drug Administration Center for Biologics
 Comments from the FDA Working Group on SUBGROUP ANALYSES Estelle Russek-Cohen, Ph.D. U.S. Food and Drug Administration Center for Biologics 1 Outline An intro to FDA EMA and FDA on subgroups Companion
Comments from the FDA Working Group on SUBGROUP ANALYSES Estelle Russek-Cohen, Ph.D. U.S. Food and Drug Administration Center for Biologics 1 Outline An intro to FDA EMA and FDA on subgroups Companion
SNP GENOTYPING WITH iplex REAGENTS AND THE MASSARRAY SYSTEM
 SNP GENOTYPING Accurate, sensitive, flexible MassARRAY System SNP GENOTYPING WITH iplex REAGENTS AND THE MASSARRAY SYSTEM Biomarker validation Routine genetic testing Somatic mutation profiling Up to 400
SNP GENOTYPING Accurate, sensitive, flexible MassARRAY System SNP GENOTYPING WITH iplex REAGENTS AND THE MASSARRAY SYSTEM Biomarker validation Routine genetic testing Somatic mutation profiling Up to 400
Goals of pharmacogenomics
 Goals of pharmacogenomics Use drugs better and use better drugs! People inherit/exhibit differences in drug: Absorption Metabolism and degradation of the drug Transport of drug to the target molecule Excretion
Goals of pharmacogenomics Use drugs better and use better drugs! People inherit/exhibit differences in drug: Absorption Metabolism and degradation of the drug Transport of drug to the target molecule Excretion
Personalized Medicine
 Personalized Medicine Dr. Pablo Mentzinis, Director Government Relations, SAP SE Courtesy by Dominik Bertram, Marc von der Linden, Péter Adorján, SAP June 2016 1. Traditional medicine vs. personalized
Personalized Medicine Dr. Pablo Mentzinis, Director Government Relations, SAP SE Courtesy by Dominik Bertram, Marc von der Linden, Péter Adorján, SAP June 2016 1. Traditional medicine vs. personalized
Drug Development: Why Does it Cost so Much? Lewis J. Smith, MD Professor of Medicine Director, Center for Clinical Research Associate VP for Research
 Drug Development: Why Does it Cost so Much? Lewis J. Smith, MD Professor of Medicine Director, Center for Clinical Research Associate VP for Research Drug Development Process by which new chemical entities
Drug Development: Why Does it Cost so Much? Lewis J. Smith, MD Professor of Medicine Director, Center for Clinical Research Associate VP for Research Drug Development Process by which new chemical entities
Introduction to Bioinformatics
 Introduction to Bioinformatics Dr. Taysir Hassan Abdel Hamid Lecturer, Information Systems Department Faculty of Computer and Information Assiut University taysirhs@aun.edu.eg taysir_soliman@hotmail.com
Introduction to Bioinformatics Dr. Taysir Hassan Abdel Hamid Lecturer, Information Systems Department Faculty of Computer and Information Assiut University taysirhs@aun.edu.eg taysir_soliman@hotmail.com
