AccuSEQ Real-Time PCR Detection Software
|
|
|
- Domenic McCarthy
- 6 years ago
- Views:
Transcription
1 Quick Reference Card AccuSEQ Real-Time PCR Detection Software Mycoplasma SEQ Experiments Note: For safety and biohazard guidelines, refer to the Safety section in the AccuSEQ Real-Time PCR Detection Software Mycoplasma Experiments Getting Started Guide (PN ). For every chemical, read the MSDS and follow the handling instructions. Wear appropriate protective eyewear, clothing, and gloves. Overview About the AccuSEQ Software Applied Biosystems AccuSEQ Software is detection and analysis software used with Real-Time PCR (polymerase chain reaction) assays for impurity and contaminant analysis in pharmaceutical quality control and process development environments. This quick reference card provides abbreviated procedures for creating, analyzing, viewing, and interpreting Mycoplasma SEQ experiments using the AccuSEQ software. Related documents AccuSEQ Software v1.0 Help Access the Help system by pressing F1, by clicking in the toolbar of the AccuSEQ Software window, or by selecting HelpContents and Index. Documents shipped with the software: AccuSEQ Real-Time PCR Detection Software Mycoplasma Experiments Getting Started Guide AccuSEQ Real-Time PCR Detection Software Custom Experiments Quick Reference Card MicroSEQ Mycoplasma Real-Time PCR Detection Kit Protocol About Mycoplasma SEQ experiments The SEQ Experiment workflow calculates reagent volumes, determines optimum plate setup, guides you through experiment set up, uses optimized thermal cycling conditions, and performs data analysis. The Mycoplasma SEQ experiment workflow is designed to set up, run, and analyze samples prepared with the MicroSEQ Mycoplasma Real-Time PCR Detection Kit. Perform a Mycoplasma SEQ experiment to detect the presence of mycoplasma species nucleic acid sequence in a sample. Mycoplasma SEQ experiment workflow The following flowchart summarizes the steps for performing a typical Mycoplasma SEQ experiment workflow using the AccuSEQ software. Step 1: Set up the experiment (page 2) Create a new experiment. Define the experiment properties. Set up the samples and controls. Review the reaction setup. Finish the experiment setup. Step 2: Prepare the reactions (page 3) Step 3: Run the experiment (page 3) Prepare for the run. Start the run. (Optional) Monitor the run. Unload the instrument. Step 4: Review the Results Summary (page 3) Step 5: (Optional) Review the results (page 4) Review the Quality Summary. Review the amplification plot. Review the melt curve. Review the table data. Step 6: (Optional) Investigate the results (page 5) Review multiple plots. Review the analysis settings. Omit wells from the analysis. Step 7: (Optional) Export and print the data (page 5) Export data. Print a report. Print SAE reports. IMPORTANT! The Mycoplasma SEQ experiment performs a presence/absence assay and analyzes melt curve data. The SEQ presence/absence assay and the melt curve algorithm are customized for detection of specific organisms and species and may yield different results than a custom presence/ absence experiment or a custom melt curve experiment.
2 Step 1: Set up the experiment Create a new experiment 1. In the desktop, double click (AccuSEQ Software) to start the software. 2. Log in to the software. See your system administrator for username and password. Note: If you have Administrator privileges, you can enable or disable the Security, Audit, and E-Sig (SAE) settings for experiments. For more information, see the AccuSEQ Software Help. 3. In the Home screen, click Create SEQ Experiment to open the SEQ Experiment workflow. 4. In the SEQ assay selection dialog, verify that Mycoplasma Assay Software Module v1.0 is selected from the presence/absence assay group, then click Next. Define the experiment properties Experiment, reagents, run method, and target(s) are defined by the software based on the SEQ assay selected when you created the experiment. In the Experiment Properties screen: 1. Enter an experiment name that is descriptive and easy to remember. 2. (Optional) Enter a barcode to identify the barcode on the reaction plate. 3. (Optional) Enter comments to describe the experiment. 4. Verify the SEQ experiment type and assay to use, then click Next. Set up the samples and controls In the Sample Setup screen: 1. Specify the number of samples and replicates: Field Minimum Entry Samples 1 Sample replicates 1 Inhibition control replicates for each sample 1 Positive control replicates 1 Negative control replicates 1 Use at least one negative and one positive control per run, and at least one inhibition control per sample. 2. Set the Sample volume per reaction (2-10 µl). Note: Applied Biosystems recommends a sample volume per reaction of 10 µl for Mycoplasma SEQ experiments. The 7500 Fast system supports total reaction volumes from 10 to 30 µl. 3. Enter sample names. 4. (Optional) Set plot colors. 5. Click Next. Review the reaction setup The AccuSEQ software uses the sample information that you enter in the Sample Setup screen to fill the wells in the plate layout (see below) and to calculate the required reaction component volumes for each sample type, based on the Mycoplasma Real-Time PCR Detection Kit Protocol guidelines. View the plate layout The Plate Layout tab displays information about each well in the reaction plate in an illustration. Review and adjust the plate layout (click-drag or Swap wells) as needed, then click Next. IMPORTANT! If you want to modify the default plate layout, refer to the Mycoplasma Real-Time PCR Detection Kit Protocol for plate layout suggestions. Print the experiment setup Print the experiment setup for use in reaction preparation. 1. In the Print Experiment Setup screen, select the elements to include in the report: Experiment Summary Detailed Instructions Plate Layout 2. Click Preview, Save As PDF (or HTML), or Print. Finish the experiment setup Click Save & Finish in the open experiment screen, specify a name and location for the.eds file, then click OK. Specify a reason for change, if For more information, see the AccuSEQ Software Help. IMPORTANT! Do not manipulate the experiment files stored on the hard drive of the computer. If you do so, you will corrupt the experiment file and you will not be able to open it in the AccuSEQ software. Page 2
3 GR /7500 barcode on plate AccuSEQ Real-Time PCR Detection Software Mycoplasma Experiments Quick Reference Card Step 2: Prepare the reactions For a Mycoplasma SEQ experiment, you prepare PCR reactions that contain primers designed to amplify the mycoplasma targets and a reagent to detect amplification of the targets. 1. Prepare the PCR reactions as described in the printed reaction setup worksheet. 2. Pipette samples into the plate according to the printed plate layout. For details on plate sealing, see the Mycoplasma Real- Time PCR Detection Kit Protocol. Step 3: Run the experiment Prepare for the run Open the experiment 1. Select File Open from the toolbar to open the experiment you saved when you set up the experiment. 2. In the dialog box that opens, select the experiment, then click Open. (Optional) Enable the notification settings If you want the system to send notifications about the instrument run: 1. In the navigation pane, select Run Notification Settings. 2. Select Yes to enable notifications, then define the notification settings. See the AccuSEQ Software Help for more information. Start the run Load the instrument 1. Push the tray door to open it. 2. Load the plate into the plate holder in the instrument with the notched A1 position at the top-left of the tray. Ensure that the plate is properly aligned in the holder. 3. Push the tray door to close it. Apply pressure to the right side of the tray door at an angle. Start the run 1. In the navigation pane, select Run. 2. Click Start Run (green button at the top of every run screen). Specify a reason for change, if (Optional) Monitor the run 1. In the navigation pane, select Run. 2. Select a run screen from the navigation pane to monitor the progress: Amplification Plot - View the change in normalized reporter signal (ΔRn) for each cycle. Melt Curve - View the normalized reporter signal (Rn) plotted against temperature ( C). Temperature Plot - View the change in sample and block temperature ( C) throughout the run. Run Method - View the default run method. At the top of every run screen, you can view the run status. Note: During a Mycoplasma SEQ experiment run, progress bars reflect run status. During the third stage of a Mycoplasma SEQ experiment, there is a delay between melting and completion of the stage while the system analyzes the data. Unload the instrument When your 7500 Fast system displays the Run Complete message, unload the reaction plate from the instrument. 1. Push the tray door to open it. 2. Remove the reaction plate. 3. Push the tray door to close it. Step 4: Review the Results Summary Use the Call Summary, Plate Layout and Table views in the Results Summary screen to review the experiment results. 1. From the navigation pane, select Results Results Summary. 2. Review the Call Summary for results: GR2471 Sample Type Call Description GR2477 GR tray top view Push Positive Control, Inhibition Control Expected Unexpected Target detected No target detected (target should be detected) Negative Control Expected Unexpected No target detected Target detected (no target should be detected) Page 3
4 Sample Type Call As needed: To review any unexpected or inconclusive results, review the Quality Summary (see below) Export and print the data (see page 5) Sign the results Step 5: (Optional) Review the results Review the Quality Summary Description Unknown Present Target signal is present and is within the temperature range, derivative range, and C T range for the assay. Absent Target signal is not present within the temperature range, derivative range, and C T range for the assay. Inconclusive The AccuSEQ software is unable to determine a result. Use the Quality Summary to review the call assessments applied to wells of the reaction plate and view troubleshooting information. This summary includes the call assessment frequency and location for the open experiment. 1. Click Results in the navigation pane, then click Quality Summary. 2. In the Quality Summary table, look in the Frequency and Wells columns to determine which call assessments appear in the experiment. For Mycoplasma SEQ experiments, the call assessments listed below may be triggered by the experiment data. Unknown DNA in negative control No expected DNA in positive and/or inhibition control Presumptive positive Presumptive negative Unexpected result in inhibition control Sample inhibits amplification High background signal Baseline algorithm failed Thresholding algorithm failed Bad passive reference signal Fluorescence is offscale All positive controls failed All negative controls failed No inhibition control found Unable to determine the SYBR dye call No signal in well C T algorithm failed Exponential algorithm failed 3. (Optional) Click a call assessment in the table to display more information about the assessment and highlight the associated well(s) in the plate layout or table view. As needed: To further investigate any unexpected or inconclusive results, review the following: Amplification Plot (see below) Melt Curve (see page 4) Table data (see page 5) Export and print the data (see page 5) Sign the results Review the amplification plot The amplification plot displays the data collected during the cycling stage of PCR amplification. It can be viewed as: Baseline-corrected normalized reporter (ΔRn) vs. cycle Normalized reporter (Rn) vs. cycle Threshold cycle (C T ) vs. well To review the Amplification Plot: 1. Click Plot Analysis in the navigation pane, then click Amplification Plot. 2. Display all 96 wells in the amplification plot by clicking the upper left corner of the plate layout in the Plate Layout tab. 3. Use the Plot Settings tab to review the amplification plot for: C T values Irregular amplification Outliers Review the melt curve The Melt Curve plot displays the fluorescence peak data collected at each well position during the melt curve stage of an instrument run. For Mycoplasma SEQ experiments, the AccuSEQ software determines the melting temperature (T m ) and reporter signal (T m height) for each peak. To review the Melt Curve: 1. Click Plot Analysis in the navigation pane, then click Melt Curve. IMPORTANT! The T m values displayed below the melt curve correspond to the largest peak in the target range. To determine the target T m, review the table data (see below). The T m values displayed below the plot may not exactly match the values displayed in the table view because of rounding and/or interpolation. 2. Select wells in the plate layout to view in the melt curve plot. Page 4
5 Review the table data The Table View displays results data for each well in the reaction plate, including: The well number, sample name, sample type, assay, call, call assessment, and cycle 1 fluorescence (FSU) The calculated values: C T, target T m, target T m height, notarget T m, no-target T m height, and ΔRn Note: The cycle 1 fluorescence and ΔRn values are only available for display in the Table View when it is accessed from the Plot Analysis screens. To review the table data: 1. In the navigation pane, select Results or Plot Analysis, then select the Table View tab. 2. Select well(s) in the Table View to display in the corresponding plot(s). 3. Review the following values for wells with inconclusive or unexpected results: Target T m and Target T m height No-target T m and No-target T m height C T Step 6: (Optional) Investigate the results Review multiple plots 1. Click Plot Analysis in the navigation pane, then click Multiple Plots View. 2. Select Multicomponent Plot and Raw Data Plot. 3. Click a sample in the plate layout or the table view to display the corresponding plots. Review the analysis settings Note: You must have administrator or scientist privileges to access this function. The Analysis Settings dialog box displays the pre-defined threshold cycle (C T ) analysis settings for Mycoplasma SEQ experiments, optimized in the AccuSEQ software for use with the MicroSEQ Mycoplasma Real-Time PCR Detection Kit. If the pre-defined analysis settings are not suitable for your experiment, you can change the C T settings in the Analysis Settings dialog box, then reanalyze your experiment. 1. Open an analyzed Mycoplasma SEQ experiment. 2. Select Analysis Analysis Settings from the toolbar menu. 3. Define the C T settings to use for the Mycoplasma target in the table. See the AccuSEQ Software Help for more information. 4. Click Apply Analysis Settings. 5. Click Analyze to reanalyze the data with the new C T settings. Omit wells from the analysis Note: You must have administrator or scientist privileges to access this function. You may omit wells from analysis if you do not want to consider data generated by the well. 1. In the navigation pane, select Plot Analysis. 2. Using the plate layout or table view, select one or more wells to omit from analysis. 3. Right-click the well(s), then select Omit. Results for the selected well(s) are removed. 4. Click Analyze to reanalyze the data without the omitted well(s). Step 7: (Optional) Export and print the data Export data Note: You must have administrator or scientist privileges to access this function. 1. In the toolbar of an analyzed Mycoplasma SEQ experiment, click Export. 2. Complete the tasks on the Export Properties tab: a. Select the Mycoplasma SEQ data to export: Sample Setup Results Raw Data Amplification Data Multicomponent Data b. Select to export all data in one file or in separate files for each data type. c. Enter export file properties, then click Start Export. 3. Click to close the Export Data dialog box. Page 5
6 Print a report 1. In the toolbar of an analyzed Mycoplasma SEQ experiment, click Print Report. 2. Select the Mycoplasma SEQ data to include in the report: Experiment Summary Results Summary Plate Layout Amplification Plot (ΔRn vs. Cycle, Rn vs. Cycle, C T vs. Well) Melt Curve (Derivative Reporter, Normalized Reporter) Quality Summary 3. Click Preview, Save As PDF (or HTML), or Print. 4. Click to close the Print Report dialog box. Print SAE reports You can print SAE reports for the current experiment in the Experiment screen: Audit Report or E-Sig Report. For more information, see the AccuSEQ Software Help. For Research Use Only. Not for use in diagnostic procedures. NOTICE TO PURCHASER: PLEASE REFER TO THE ACCUSEQ REAL-TIME PCR DETECTION SOFTWARE MYCOPLASMA SEQ EXPERIMENTS GETTING STARTED GUIDE FOR LIMITED LABEL LICENSE OR DISCLAIMER INFORMATION. Trademarks of Applied Biosystems and its affiliated companies include: Applied Biosystems, AB (Design), AccuSEQ, and MicroSEQ. SYBR is a registered trademark of Molecular Probes, Inc. All other trademarks are the sole property of their respective owners. Copyright 2009, Applied Biosystems. All rights reserved. Part Number Rev. A 08/2009 Headquarters 850 Lincoln Centre Drive Foster City, CA USA Phone Toll Free International Sales For our office locations please call the division headquarters or refer to our Web site at
Applied Biosystems 7900HT Fast Real-Time PCR System. Performing Fast Gene Quantitation with 384-Well Plates DRAFT
 User Bulletin Applied Biosystems 7900HT Fast Real-Time PCR System October 3, 2005 SUBJECT: Performing Fast Gene Quantitation with 384-Well Plates In This User Bulletin System Overview System Requirements
User Bulletin Applied Biosystems 7900HT Fast Real-Time PCR System October 3, 2005 SUBJECT: Performing Fast Gene Quantitation with 384-Well Plates In This User Bulletin System Overview System Requirements
Perform an HRM Methylation Study
 Quick Reference Card Perform an HRM Methylation Study This quick reference card provides brief procedures for performing an HRM methylation study using MeltDoctor HRM Master Mix and High Resolution Melting
Quick Reference Card Perform an HRM Methylation Study This quick reference card provides brief procedures for performing an HRM methylation study using MeltDoctor HRM Master Mix and High Resolution Melting
AccuSEQ v2.1 Real-Time PCR Detection and Analysis Software
 DATA SHEET AccuSEQ Real-Time PCR Detection and Analysis Software AccuSEQ v2.1 Real-Time PCR Detection and Analysis Software Introduction Automated presence or absence calls for Applied Biosystems MycoSEQ
DATA SHEET AccuSEQ Real-Time PCR Detection and Analysis Software AccuSEQ v2.1 Real-Time PCR Detection and Analysis Software Introduction Automated presence or absence calls for Applied Biosystems MycoSEQ
Applied Biosystems 7500 Fast Real-Time PCR System
 User Bulletin Applied Biosystems 7500 Fast Real-Time PCR System July 2006 SUBJECT: Using Expert Mode on the Applied Biosystems 7500 Fast Real-Time PCR System with the Applied Biosystems 7500 Fast System
User Bulletin Applied Biosystems 7500 Fast Real-Time PCR System July 2006 SUBJECT: Using Expert Mode on the Applied Biosystems 7500 Fast Real-Time PCR System with the Applied Biosystems 7500 Fast System
User Bulletin. ABI PRISM 7000 Sequence Detection System DRAFT. SUBJECT: Software Upgrade from Version 1.0 to 1.1. Version 1.
 User Bulletin ABI PRISM 7000 Sequence Detection System 9/15/03 SUBJECT: Software Upgrade from Version 1.0 to 1.1 The ABI PRISM 7000 Sequence Detection System version 1.1 software is a free upgrade. Also
User Bulletin ABI PRISM 7000 Sequence Detection System 9/15/03 SUBJECT: Software Upgrade from Version 1.0 to 1.1 The ABI PRISM 7000 Sequence Detection System version 1.1 software is a free upgrade. Also
SUBJECT: System Software Patch Provides Support for Fast SYBR Green Master Mix and RNA-to-C T
 User Bulletin Applied Biosystems StepOne and StepOnePlus Real-Time PCR Systems March 2008 SUBJECT: System Software Patch Provides Support for Fast SYBR Green Master Mix and RNA-to-C T In This User Bulletin
User Bulletin Applied Biosystems StepOne and StepOnePlus Real-Time PCR Systems March 2008 SUBJECT: System Software Patch Provides Support for Fast SYBR Green Master Mix and RNA-to-C T In This User Bulletin
Data Analysis on the ABI PRISM 7700 Sequence Detection System: Setting Baselines and Thresholds. Overview. Data Analysis Tutorial
 Data Analysis on the ABI PRISM 7700 Sequence Detection System: Setting Baselines and Thresholds Overview In order for accuracy and precision to be optimal, the assay must be properly evaluated and a few
Data Analysis on the ABI PRISM 7700 Sequence Detection System: Setting Baselines and Thresholds Overview In order for accuracy and precision to be optimal, the assay must be properly evaluated and a few
Real-Time PCR ABI Prism 7500 (PSC 563, NSC 338) Real-Time PCR StepOne (PSC 563)
 Real-Time PCR ABI Prism 7500 (PSC 563, NSC 338) Real-Time PCR StepOne (PSC 563) Every time you use any of the qpcr instruments, you MUST sign the log book. Use Lab Commander to reserve a time. Each instrument
Real-Time PCR ABI Prism 7500 (PSC 563, NSC 338) Real-Time PCR StepOne (PSC 563) Every time you use any of the qpcr instruments, you MUST sign the log book. Use Lab Commander to reserve a time. Each instrument
Applied Biosystems 7500 Fast, 7500 and 7300 Real-Time PCR Systems
 PRODUCT BROCHURE Real-Time PCR Systems Applied Biosystems 7500 Fast, 7500 and 7300 Real-Time PCR Systems Real Fast. Real Versatile. Real Value. Real choices from the leader in real-time PCR. The latest
PRODUCT BROCHURE Real-Time PCR Systems Applied Biosystems 7500 Fast, 7500 and 7300 Real-Time PCR Systems Real Fast. Real Versatile. Real Value. Real choices from the leader in real-time PCR. The latest
Procedure for DNA Quantitation with Quantifiler Duo
 Procedure for DNA Quantitation with Quantifiler Duo 1.0 Purpose This procedure specifies the steps for performing quantification of DNA extracts with the Quantifiler Duo kit on the ABI 7500 instruments.
Procedure for DNA Quantitation with Quantifiler Duo 1.0 Purpose This procedure specifies the steps for performing quantification of DNA extracts with the Quantifiler Duo kit on the ABI 7500 instruments.
Applied Biosystems TaqMan Array Microfluidic Cards
 Performing Rapid Cycling Gene Quantitation on TaqMan Array Microfluidic Cards Applied Biosystems TaqMan Array Microfluidic Cards User Bulletin JULY 2010 SUBJECT: In this user bulletin Performing Rapid
Performing Rapid Cycling Gene Quantitation on TaqMan Array Microfluidic Cards Applied Biosystems TaqMan Array Microfluidic Cards User Bulletin JULY 2010 SUBJECT: In this user bulletin Performing Rapid
Georgia Bureau of Investigation - Division of Forensic Sciences DNA Procedures Manual DNA QUANTIFICATION USING QUANTIFILER HUMAN / Y HUMAN MALE KIT
 Purpose: To obtain quantitative value of human (higher primate) nuclear DNA isolated from samples. Materials: Previously extracted DNA 96 well optical reaction plate Optical adhesive cover Compression
Purpose: To obtain quantitative value of human (higher primate) nuclear DNA isolated from samples. Materials: Previously extracted DNA 96 well optical reaction plate Optical adhesive cover Compression
Protocol. Path-ID Multiplex One-Step RT-PCR Kit Protocol TaqMan probe-based multiplex one-step real-time RT-PCR detection of RNA targets
 Protocol TaqMan probe-based multiplex one-step real-time RT-PCR detection of RNA targets For Research Use Only. Not for use in diagnostic procedures. Information in this document is subject to change without
Protocol TaqMan probe-based multiplex one-step real-time RT-PCR detection of RNA targets For Research Use Only. Not for use in diagnostic procedures. Information in this document is subject to change without
Custom dye calibration for StepOne and StepOnePlus Real-Time PCR Systems
 USER BULLETIN Custom dye calibration for StepOne and StepOnePlus Real-Time PCR Systems Publication Number MAN0014609 Revision A This user bulletin is for use with the StepOne and StepOnePlus Real-Time
USER BULLETIN Custom dye calibration for StepOne and StepOnePlus Real-Time PCR Systems Publication Number MAN0014609 Revision A This user bulletin is for use with the StepOne and StepOnePlus Real-Time
ReadMe Software v2.0.1 For Applied Biosystems 7500 and 7500 Fast Real-Time PCR Systems
 850 Lincoln Centre Drive Foster City, CA 94404 U.S.A. T 650.570.6667 F 650.572.2743 www.appliedbiosystems.com ReadMe 7500 Software v2.0.1 F Applied Biosystems 7500 and 7500 Fast Real-Time PCR Systems CONTENTS
850 Lincoln Centre Drive Foster City, CA 94404 U.S.A. T 650.570.6667 F 650.572.2743 www.appliedbiosystems.com ReadMe 7500 Software v2.0.1 F Applied Biosystems 7500 and 7500 Fast Real-Time PCR Systems CONTENTS
Quantifiler Human DNA Quantification Kit Quantifiler Y Human Male DNA Quantification Kit
 Product Bulletin Human Identification Quantifiler Human DNA Quantification Kit Quantifiler Y Human Male DNA Quantification Kit The Quantifiler kits produce reliable and reproducible results, helping to
Product Bulletin Human Identification Quantifiler Human DNA Quantification Kit Quantifiler Y Human Male DNA Quantification Kit The Quantifiler kits produce reliable and reproducible results, helping to
Plexor HY System for the Applied Biosystems 7500 and 7500 FAST Real-Time PCR Systems
 TECHNICAL MANUAL Plexor HY System for the Applied Biosystems 7500 and 7500 FAST Real-Time PCR Systems Instructions for Use of Products DC1000, DC1001 and DC1500 Technical Manuals with instructions for
TECHNICAL MANUAL Plexor HY System for the Applied Biosystems 7500 and 7500 FAST Real-Time PCR Systems Instructions for Use of Products DC1000, DC1001 and DC1500 Technical Manuals with instructions for
PCR SYSTEMS. a new era in high-productivity qpcr. Applied Biosystems ViiA 7 Real-Time PCR System
 PCR SYSTEMS a new era in high-productivity qpcr Applied Biosystems ViiA 7 Real-Time PCR System a new era in high-productivity qpcr The ViiA 7 Real-Time PCR System delivers the proven reliability, sensitivity,
PCR SYSTEMS a new era in high-productivity qpcr Applied Biosystems ViiA 7 Real-Time PCR System a new era in high-productivity qpcr The ViiA 7 Real-Time PCR System delivers the proven reliability, sensitivity,
Troubleshooting Chemistry and Assay Runs
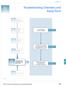 Chapter 5 Chemistry and Assay Runs Performing Calibration and Verification Runs Maintaining the 7900HT Instrument Hardware Low Precision or Irreproducibility Seepage120 Maintaining the Computer and Software
Chapter 5 Chemistry and Assay Runs Performing Calibration and Verification Runs Maintaining the 7900HT Instrument Hardware Low Precision or Irreproducibility Seepage120 Maintaining the Computer and Software
User Bulletin. Veriti 96-Well Thermal Cycler AmpFlSTR Kit Validation. Overview
 User Bulletin Veriti 96-Well Thermal Cycler AmpFlSTR Kit Validation June 2009 SUBJECT: AmpFlSTR PCR Amplification Kit validation on the Veriti 96-Well Thermal Cycler with 0.2 ml sample block format In
User Bulletin Veriti 96-Well Thermal Cycler AmpFlSTR Kit Validation June 2009 SUBJECT: AmpFlSTR PCR Amplification Kit validation on the Veriti 96-Well Thermal Cycler with 0.2 ml sample block format In
VERSANT kpcr Molecular System
 VERSANT kpcr Molecular System Application Guide Addendum Running Dynamic Assay Protocols Using the VERSANT kpcr Molecular System SMN: 10814065 Rev. C Copyrights No part of this work covered by the copyrights
VERSANT kpcr Molecular System Application Guide Addendum Running Dynamic Assay Protocols Using the VERSANT kpcr Molecular System SMN: 10814065 Rev. C Copyrights No part of this work covered by the copyrights
Relative Quantification (Mono-Color) Unknown samples (purified total RNA, mrna or cdna or genomic DNA)
 The LightCycler 480 System Short Guide Topic: Purpose: Assay Principle: Detection Format: Result: Relative Quantification (Mono-Color) Describes how to set up and perform mono-color Relative Quantification
The LightCycler 480 System Short Guide Topic: Purpose: Assay Principle: Detection Format: Result: Relative Quantification (Mono-Color) Describes how to set up and perform mono-color Relative Quantification
Quantitative Real time PCR. Only for teaching purposes - not for reproduction or sale
 Quantitative Real time PCR PCR reaction conventional versus real time PCR real time PCR principles threshold cycle C T efficiency relative quantification reference genes primers detection chemistry GLP
Quantitative Real time PCR PCR reaction conventional versus real time PCR real time PCR principles threshold cycle C T efficiency relative quantification reference genes primers detection chemistry GLP
Applied Biosystems Real-Time PCR Rapid Assay Development Guidelines
 Applied Biosystems Real-Time PCR Rapid Assay Development Guidelines Description This tutorial will discuss recommended guidelines for designing and running real-time PCR quantification and SNP Genotyping
Applied Biosystems Real-Time PCR Rapid Assay Development Guidelines Description This tutorial will discuss recommended guidelines for designing and running real-time PCR quantification and SNP Genotyping
ViiA 7 Real-Time PCR System
 ViiA 7 Real-Time PCR System Note: For safety and biohazard guidelines, refer to the Safety appendix in the Applied Biosystems ViiA 7Real- Time PCR System User Guide (PN 444661). For every chemical, read
ViiA 7 Real-Time PCR System Note: For safety and biohazard guidelines, refer to the Safety appendix in the Applied Biosystems ViiA 7Real- Time PCR System User Guide (PN 444661). For every chemical, read
Multiplex RT for TaqMan Array Human MicroRNA Panel
 f Multiplex RT for TaqMan Low Density Array Multiplex RT for TaqMan Array Human MicroRNA Panel Quick Reference Card For safety and biohazard guidelines, refer to the Safety section in the Multiplex RT
f Multiplex RT for TaqMan Low Density Array Multiplex RT for TaqMan Array Human MicroRNA Panel Quick Reference Card For safety and biohazard guidelines, refer to the Safety section in the Multiplex RT
PROTOCOL. ViralSEQ Mouse Minute Virus (MMV) Real-Time PCR Detection Kit
 PROTOCOL ViralSEQ Mouse Minute Virus (MMV) Real-Time PCR Detection Kit Publication Part Number 4445235 Rev. D Revision Date March 2011 For Research Use Only. Not intended for any animal or human therapeutic
PROTOCOL ViralSEQ Mouse Minute Virus (MMV) Real-Time PCR Detection Kit Publication Part Number 4445235 Rev. D Revision Date March 2011 For Research Use Only. Not intended for any animal or human therapeutic
Module1TheBasicsofRealTimePCR Monday, March 19, 2007
 Objectives Slide notes: Page 1 of 41 Module 1: The Basics Of Real Time PCR Slide notes: Module 1: The Basics of real time PCR Page 2 of 41 Polymerase Chain Reaction Slide notes: Here is a review of PCR,
Objectives Slide notes: Page 1 of 41 Module 1: The Basics Of Real Time PCR Slide notes: Module 1: The Basics of real time PCR Page 2 of 41 Polymerase Chain Reaction Slide notes: Here is a review of PCR,
PowerPlex Matrix Standards, 3100/3130
 TECHNICAL BULLETIN PowerPlex Matrix Standards, 3100/3130 Instruc ons for use of Product DG4650 Note: The PowerPlex Matrix Standards, 3100/3130, can be used to perform spectral calibra on on the Applied
TECHNICAL BULLETIN PowerPlex Matrix Standards, 3100/3130 Instruc ons for use of Product DG4650 Note: The PowerPlex Matrix Standards, 3100/3130, can be used to perform spectral calibra on on the Applied
Relative Quantification Applied Biosystems 7300/7500/7500 Fast Real-Time PCR System
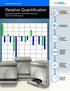 5 3 5 cdna Reverse Primer Oligo d(t) or random hexamer 3 5 cdna Getting Started Guide Relative Quantification Applied Biosystems 7300/7500/7500 Fast Real-Time PCR System Introduction and Example RQ Experiment
5 3 5 cdna Reverse Primer Oligo d(t) or random hexamer 3 5 cdna Getting Started Guide Relative Quantification Applied Biosystems 7300/7500/7500 Fast Real-Time PCR System Introduction and Example RQ Experiment
For in vitro Veterinary Diagnostics only. Real-Time PCR Detection Kit for detection of Histomonas meleagridis.
 For in vitro Veterinary Diagnostics only. Real-Time PCR Detection Kit for detection of Histomonas meleagridis www.kylt.eu DIRECTION FOR USE Art. No. 31416/31417 Kylt Histomonas meleagridis Real-Time PCR
For in vitro Veterinary Diagnostics only. Real-Time PCR Detection Kit for detection of Histomonas meleagridis www.kylt.eu DIRECTION FOR USE Art. No. 31416/31417 Kylt Histomonas meleagridis Real-Time PCR
PowerPlex 5-Dye Matrix Standards, 310
 TECHNICAL BULLETIN PowerPlex 5-Dye Matrix Standards, 310 Instructions for Use of Product DG4600 Revised 3/16 TBD023 PowerPlex 5-Dye Matrix Standards, 310 All technical literature is available at: www.promega.com/protocols/
TECHNICAL BULLETIN PowerPlex 5-Dye Matrix Standards, 310 Instructions for Use of Product DG4600 Revised 3/16 TBD023 PowerPlex 5-Dye Matrix Standards, 310 All technical literature is available at: www.promega.com/protocols/
QIAGEN Supplementary Protocol
 QIAGEN Supplementary Protocol PCR and data analysis using the cador T. equigenitalis PCR Kit on Rotor-Gene Q instruments This protocol is designed for using the cador T. equigenitalis PCR Kit for detection
QIAGEN Supplementary Protocol PCR and data analysis using the cador T. equigenitalis PCR Kit on Rotor-Gene Q instruments This protocol is designed for using the cador T. equigenitalis PCR Kit for detection
PowerPlex 5C Matrix Standards, 310
 TECHNICAL MANUAL PowerPlex 5C Matrix Standards, 310 Instruc ons for Use of Product DG5640 Printed 10/15 TMD050 PowerPlex 5C Matrix Standards, 310 All technical literature is available at: www.promega.com/protocols/
TECHNICAL MANUAL PowerPlex 5C Matrix Standards, 310 Instruc ons for Use of Product DG5640 Printed 10/15 TMD050 PowerPlex 5C Matrix Standards, 310 All technical literature is available at: www.promega.com/protocols/
Setting Up a Multiplex (two probe) Real Time 5 Nuclease Reaction on the ABI PRISM 7700 Sequence Detection System
 Setting Up a Multiplex (two probe) Real Time 5 Nuclease Reaction on the ABI PRISM 7700 Sequence Detection System Overview The goal of this tutorial is to demonstrate how to correctly label a plate set-up
Setting Up a Multiplex (two probe) Real Time 5 Nuclease Reaction on the ABI PRISM 7700 Sequence Detection System Overview The goal of this tutorial is to demonstrate how to correctly label a plate set-up
Guide to using the Bio Rad CFX96 Real Time PCR Machine
 Guide to using the Bio Rad CFX96 Real Time PCR Machine Kyle Dobbs and Peter Hansen Table of Contents Overview..3 Setup Reaction Guidelines 4 Starting up the Software 5 Setup Protocol on Software 6 Setup
Guide to using the Bio Rad CFX96 Real Time PCR Machine Kyle Dobbs and Peter Hansen Table of Contents Overview..3 Setup Reaction Guidelines 4 Starting up the Software 5 Setup Protocol on Software 6 Setup
TaqMan Pseudomonas aeruginosa Detection Kit
 USER GUIDE TaqMan Pseudomonas aeruginosa Detection Kit Catalog Number 4368604 Publication Number 4370452 Revision D For testing of Food and Environmental samples only. For testing of Food and Environmental
USER GUIDE TaqMan Pseudomonas aeruginosa Detection Kit Catalog Number 4368604 Publication Number 4370452 Revision D For testing of Food and Environmental samples only. For testing of Food and Environmental
TaqMan Cytokine Gene Expression Plate I Protocol
 TaqMan Cytokine Gene Expression Plate I Protocol For Research Use Only. Not for use in diagnostic procedures. Copyright 2003, Applied Biosystems For Research Use Only. Not for use in diagnostic procedures.
TaqMan Cytokine Gene Expression Plate I Protocol For Research Use Only. Not for use in diagnostic procedures. Copyright 2003, Applied Biosystems For Research Use Only. Not for use in diagnostic procedures.
Relative Quantification Getting Started Guide
 5 cdna Reverse Primer Oligo d(t) or random hexamer Synthesis of 1st cdna strand 3 5 cdna Applied Biosystems 7300/7500/7500 Fast Real-Time PCR System Relative Quantification Getting Started Guide Introduction
5 cdna Reverse Primer Oligo d(t) or random hexamer Synthesis of 1st cdna strand 3 5 cdna Applied Biosystems 7300/7500/7500 Fast Real-Time PCR System Relative Quantification Getting Started Guide Introduction
Touchscreen (standalone capabilities, PIN-protected user accounts, and dye calibration/rnasep functionality)
 QuantStudio 3 & 5 Real-Time PCR Systems: The Basics Touchscreen (standalone capabilities, PIN-protected user accounts, and dye calibration/rnasep functionality) USB ports Overview of the QuantStudio TM
QuantStudio 3 & 5 Real-Time PCR Systems: The Basics Touchscreen (standalone capabilities, PIN-protected user accounts, and dye calibration/rnasep functionality) USB ports Overview of the QuantStudio TM
PowerPlex 4C Matrix Standard
 TECHNICAL MANUAL PowerPlex 4C Matrix Standard Instruc ons for Use of Product DG4800 Printed 10/15 TMD048 PowerPlex 4C Matrix Standard All technical literature is available at: www.promega.com/protocols/
TECHNICAL MANUAL PowerPlex 4C Matrix Standard Instruc ons for Use of Product DG4800 Printed 10/15 TMD048 PowerPlex 4C Matrix Standard All technical literature is available at: www.promega.com/protocols/
User Manual. NGS Library qpcr Quantification Kit (Illumina compatible)
 NGS Library qpcr Quantification Kit (Illumina compatible) User Manual 384 Oyster Point Blvd, Suite 15 South San Francisco, CA 94080 Phone: 1 (888) MCLAB-88 Fax: 1 (650) 872-0253 www.mclab.com Contents
NGS Library qpcr Quantification Kit (Illumina compatible) User Manual 384 Oyster Point Blvd, Suite 15 South San Francisco, CA 94080 Phone: 1 (888) MCLAB-88 Fax: 1 (650) 872-0253 www.mclab.com Contents
Introducing a new generation of affordable choices from the leader in real-time PCR. Applied Biosystems 7500 Fast Real-Time PCR System
 7300/7500 Real-Time PCR Systems Introducing a new generation of affordable choices from the leader in real-time PCR. Applied Biosystems 7500 Real-Time PCR System Applied Biosystems 7500 Fast Real-Time
7300/7500 Real-Time PCR Systems Introducing a new generation of affordable choices from the leader in real-time PCR. Applied Biosystems 7500 Real-Time PCR System Applied Biosystems 7500 Fast Real-Time
Procedure for GeneMapper ID-X for Casework
 Procedure for GeneMapper ID-X for Casework 1.0 Purpose-This procedure specifies the steps for performing analysis on DNA samples amplified with AmpFlSTR Identifiler Plus using the GeneMapper ID-X (GMID-X)
Procedure for GeneMapper ID-X for Casework 1.0 Purpose-This procedure specifies the steps for performing analysis on DNA samples amplified with AmpFlSTR Identifiler Plus using the GeneMapper ID-X (GMID-X)
Brilliant II SYBR Green QPCR Master Mix
 Brilliant II SYBR Green QPCR Master Mix INSTRUCTION MANUAL Catalog #600828 (single kit) #600831 (10-pack kit) Revision B.01 For In Vitro Use Only 600828-12 LIMITED PRODUCT WARRANTY This warranty limits
Brilliant II SYBR Green QPCR Master Mix INSTRUCTION MANUAL Catalog #600828 (single kit) #600831 (10-pack kit) Revision B.01 For In Vitro Use Only 600828-12 LIMITED PRODUCT WARRANTY This warranty limits
PowerPlex 5-Dye Matrix Standards, 3100/3130
 TECHNICAL BULLETIN PowerPlex 5-Dye Matrix Standards, 3100/3130 Instructions for Use of Product DG4700 Note: These matrix fragments are compatible with the ABI PRISM 3100 and 3100-Avant and Applied Biosystems
TECHNICAL BULLETIN PowerPlex 5-Dye Matrix Standards, 3100/3130 Instructions for Use of Product DG4700 Note: These matrix fragments are compatible with the ABI PRISM 3100 and 3100-Avant and Applied Biosystems
Shift Swapping Quick-Guide
 Shift Swapping Quick-Guide (v 3.6.0) Shift Swapping Quick-Guide Login to Lawson Workforce Management Self Service: 1. Open Internet Explorer and type in http:// in the address bar to access the Lawson
Shift Swapping Quick-Guide (v 3.6.0) Shift Swapping Quick-Guide Login to Lawson Workforce Management Self Service: 1. Open Internet Explorer and type in http:// in the address bar to access the Lawson
BigDye Direct Cycle Sequencing Kit
 BigDye Direct Cycle Sequencing Kit QUICK REFERENCE CARD Note: For safety and biohazard guidelines, refer to the Safety section in the BigDye Direct Cycle Sequencing Kit Protocol (PN 4458040). For every
BigDye Direct Cycle Sequencing Kit QUICK REFERENCE CARD Note: For safety and biohazard guidelines, refer to the Safety section in the BigDye Direct Cycle Sequencing Kit Protocol (PN 4458040). For every
Phosphate buffered saline (PBS) for washing the cells TE buffer (nuclease-free) ph 7.5 for resuspending the SingleShot RNA control template
 Catalog # Description 172-5085 SingleShot SYBR Green Kit, 100 x 50 µl reactions For research purposes only. Introduction The SingleShot SYBR Green Kit prepares genomic DNA (gdna) free RNA directly from
Catalog # Description 172-5085 SingleShot SYBR Green Kit, 100 x 50 µl reactions For research purposes only. Introduction The SingleShot SYBR Green Kit prepares genomic DNA (gdna) free RNA directly from
TECHNICAL MANUAL. ProNex DNA QC Assay. for Use on the Bio-Rad CFX96 Touch Real-Time. PCR Detection System
 TECHNICAL MANUAL ProNex DNA QC Assay for Use on the Bio-Rad CFX96 Touch Real-Time PCR Detection System Instructions for Use of Products NG1004 and NG1005 9/17 TM513 ProNex DNA QC Assay for Use on the Bio-Rad
TECHNICAL MANUAL ProNex DNA QC Assay for Use on the Bio-Rad CFX96 Touch Real-Time PCR Detection System Instructions for Use of Products NG1004 and NG1005 9/17 TM513 ProNex DNA QC Assay for Use on the Bio-Rad
Software manual. Thermo Scientific PathoProof Norden Lab Studio Instructions for use
 Software manual Thermo Scientific PathoProof Norden Lab Studio Instructions for use PF0888A Revision Date: January 11, 2016 Contents Getting started with PathoProof Norden Lab Studio... 1 1. Overview of
Software manual Thermo Scientific PathoProof Norden Lab Studio Instructions for use PF0888A Revision Date: January 11, 2016 Contents Getting started with PathoProof Norden Lab Studio... 1 1. Overview of
MyiQ Single-Color Real-Time PCR Detection System
 MyiQ Single-Color Real-Time PCR Detection System Instruction Manual For Technical Service, Call Your Local Bio-Rad Office or, in the US, Call 1-800-4BIORAD (1-800-424-6723) ~ ~ Safety Information Grounding
MyiQ Single-Color Real-Time PCR Detection System Instruction Manual For Technical Service, Call Your Local Bio-Rad Office or, in the US, Call 1-800-4BIORAD (1-800-424-6723) ~ ~ Safety Information Grounding
Relative Quantification Getting Started Guide
 5 cdna Reverse Primer Oligo d(t) or random hexamer Synthesis of 1st cdna strand 3 5 cdna Applied Biosystems 7300/7500/7500 Fast Real-Time PCR System Relative Quantification Getting Started Guide Introduction
5 cdna Reverse Primer Oligo d(t) or random hexamer Synthesis of 1st cdna strand 3 5 cdna Applied Biosystems 7300/7500/7500 Fast Real-Time PCR System Relative Quantification Getting Started Guide Introduction
How to get started with HDPOSsmart? HDPOS smart Tutorials
 How to get started with HDPOSsmart? HDPOS smart Tutorials Now that you have installed HDPOSsmart, you would like to get started with the basics of working with the application. In this document, you get
How to get started with HDPOSsmart? HDPOS smart Tutorials Now that you have installed HDPOSsmart, you would like to get started with the basics of working with the application. In this document, you get
foodproof Magnetic Preparation Kit I
 For food testing purposes FOR IN VITRO USE ONLY foodproof Magnetic Preparation Kit I Version 1, August 2012 Application manual for the automated isolation of Gram-negative bacterial DNA from enrichment
For food testing purposes FOR IN VITRO USE ONLY foodproof Magnetic Preparation Kit I Version 1, August 2012 Application manual for the automated isolation of Gram-negative bacterial DNA from enrichment
Technical Review. Real time PCR
 Technical Review Real time PCR Normal PCR: Analyze with agarose gel Normal PCR vs Real time PCR Real-time PCR, also known as quantitative PCR (qpcr) or kinetic PCR Key feature: Used to amplify and simultaneously
Technical Review Real time PCR Normal PCR: Analyze with agarose gel Normal PCR vs Real time PCR Real-time PCR, also known as quantitative PCR (qpcr) or kinetic PCR Key feature: Used to amplify and simultaneously
About SMART Practice Aids - Field Work
 About SMART Practice Aids - Field Work SMART Practice Aids Field Work is the newest addition to PPC s SMART Audit Suite. You can now plan and execute the entire PPC audit process within the SMART Suite,
About SMART Practice Aids - Field Work SMART Practice Aids Field Work is the newest addition to PPC s SMART Audit Suite. You can now plan and execute the entire PPC audit process within the SMART Suite,
Or, from the View and Manage RFx(s) page, click the Create RFx button. Create New RFx - Use this button to create an RFQ from scratch.
 Request for Quote How to Create Create an RFQ To create any RFx (RFI, RFQ, RFP, Auction or Buyer Survey), from the View and Manage RFx(s) page, click on the Create RFx button. Follow the steps below to
Request for Quote How to Create Create an RFQ To create any RFx (RFI, RFQ, RFP, Auction or Buyer Survey), from the View and Manage RFx(s) page, click on the Create RFx button. Follow the steps below to
Data Quality Worth Sharing
 Product Bulletin Human Identification AmpFlSTR NGM and NGM SElect PCR Amplification Kits Next generation amplification chemistries including the 5 new loci from the expanded European Standard Set Enhanced
Product Bulletin Human Identification AmpFlSTR NGM and NGM SElect PCR Amplification Kits Next generation amplification chemistries including the 5 new loci from the expanded European Standard Set Enhanced
Evolution Payroll. User Guide (Basic Version) 2017 Payroll Solutions, Inc &
 Evolution Payroll User Guide (Basic Version) 2017 Payroll Solutions, Inc & www.payrollsolutions.com Contents Please note that this guide is the abbreviated version. It contains precisely enough information
Evolution Payroll User Guide (Basic Version) 2017 Payroll Solutions, Inc & www.payrollsolutions.com Contents Please note that this guide is the abbreviated version. It contains precisely enough information
The Seed Health Toolbox II. Molecular Seed Testing Methods and Processes: What are the sources of variability?
 The Seed Health Toolbox II Molecular Seed Testing Methods and Processes: What are the sources of variability? Stephan C. Brière Manager Plant Pathology Diagnostic Laboratory, Ottawa, ON Canadian Food Inspection
The Seed Health Toolbox II Molecular Seed Testing Methods and Processes: What are the sources of variability? Stephan C. Brière Manager Plant Pathology Diagnostic Laboratory, Ottawa, ON Canadian Food Inspection
Table of contents. Reports...15 Printing reports Resources...30 Accessing help...30 Technical support numbers...31
 WorldShip 2018 User Guide The WorldShip software provides an easy way to automate your shipping tasks. You can quickly process all your UPS shipments, print labels and invoices, electronically transmit
WorldShip 2018 User Guide The WorldShip software provides an easy way to automate your shipping tasks. You can quickly process all your UPS shipments, print labels and invoices, electronically transmit
Administrator Quick Reference Virtual TimeClock 15 Network Edition
 Virtual TimeClock Administrator Quick Reference Virtual TimeClock 15 Network Edition Your time clock program consists of a user status window and an administration window. The user status window is used
Virtual TimeClock Administrator Quick Reference Virtual TimeClock 15 Network Edition Your time clock program consists of a user status window and an administration window. The user status window is used
HUMAN EMBRYONIC STEM CELL (hesc) ASSESSMENT BY REAL-TIME POLYMERASE CHAIN REACTION (PCR)
 HUMAN EMBRYONIC STEM CELL (hesc) ASSESSMENT BY REAL-TIME POLYMERASE CHAIN REACTION (PCR) OBJECTIVE: is performed to analyze gene expression of human Embryonic Stem Cells (hescs) in culture. Real-Time PCR
HUMAN EMBRYONIC STEM CELL (hesc) ASSESSMENT BY REAL-TIME POLYMERASE CHAIN REACTION (PCR) OBJECTIVE: is performed to analyze gene expression of human Embryonic Stem Cells (hescs) in culture. Real-Time PCR
Applied Biosystems QuantStudio 12K Flex Real- Time PCR System: OpenArray Plate
 QUICK REFERENCE Applied Biosystems QuantStudio 12K Flex Real- Time PCR System: OpenArray Plate Pub. Part no. 4478673 Rev. A Rev. Date June 2012 In this guide WARNING! GENERAL SAFETY. Using this product
QUICK REFERENCE Applied Biosystems QuantStudio 12K Flex Real- Time PCR System: OpenArray Plate Pub. Part no. 4478673 Rev. A Rev. Date June 2012 In this guide WARNING! GENERAL SAFETY. Using this product
Completing an Internal Audit User Guide For the Reliance Assessment Database
 Completing an Internal Audit User Guide For the Reliance Assessment Database Contents Logging into Reliance... 2 Creating an Audit Plan: Planning Your Internal Audit... 4 Approving an Audit Plan... 20
Completing an Internal Audit User Guide For the Reliance Assessment Database Contents Logging into Reliance... 2 Creating an Audit Plan: Planning Your Internal Audit... 4 Approving an Audit Plan... 20
SECTION 15 CARTON REQUIREMENTS
 SECTION 15 CARTON REQUIREMENTS CARTON REQUIRMENTS OVERVIEW 1. Carton Labels are required on all shipments from all vendors 2. We suggest as a best practice extra taping on your cartons. It guards against
SECTION 15 CARTON REQUIREMENTS CARTON REQUIRMENTS OVERVIEW 1. Carton Labels are required on all shipments from all vendors 2. We suggest as a best practice extra taping on your cartons. It guards against
Brilliant SYBR Green QPCR Master Mix
 Brilliant SYBR Green QPCR Master Mix INSTRUCTION MANUAL Catalog #600548 (single kit) #929548 (10-pack kit) Revision B.01 For In Vitro Use Only 600548-12 LIMITED PRODUCT WARRANTY This warranty limits our
Brilliant SYBR Green QPCR Master Mix INSTRUCTION MANUAL Catalog #600548 (single kit) #929548 (10-pack kit) Revision B.01 For In Vitro Use Only 600548-12 LIMITED PRODUCT WARRANTY This warranty limits our
SYBR Green Realtime PCR Master Mix
 Instruction manual SYBR Green Realtime PCR Master Mix 0810 F0924K SYBR Green Realtime PCR Master Mix QPK-201T 1 ml x 1 QPK-201 1 ml x 5 Contents [1] Introduction [2] Components [3] Primer design [4] Detection
Instruction manual SYBR Green Realtime PCR Master Mix 0810 F0924K SYBR Green Realtime PCR Master Mix QPK-201T 1 ml x 1 QPK-201 1 ml x 5 Contents [1] Introduction [2] Components [3] Primer design [4] Detection
TECHNICAL MANUAL. GenePrint 24 System. Instructions for Use of Product B1870 6/16 TM465
 TECHNICAL MANUAL GenePrint 24 System Instructions for Use of Product B1870 6/16 TM465 GenePrint 24 System All technical literature is available at: www.promega.com/protocols/ Visit the web site to verify
TECHNICAL MANUAL GenePrint 24 System Instructions for Use of Product B1870 6/16 TM465 GenePrint 24 System All technical literature is available at: www.promega.com/protocols/ Visit the web site to verify
C.albicans/C.glabrata/C.krusei Real-TM Handbook
 IVD For in Vitro Diagnostic Use C.albicans/C.glabrata/C.krusei Real-TM Handbook for use with RotorGene 3000/6000 (Corbett Research), SmartCycler (Cepheid), iq icycler and iq5 (Biorad), Applied Biosystems
IVD For in Vitro Diagnostic Use C.albicans/C.glabrata/C.krusei Real-TM Handbook for use with RotorGene 3000/6000 (Corbett Research), SmartCycler (Cepheid), iq icycler and iq5 (Biorad), Applied Biosystems
Managing a FedEx Shipping Label within SoftPro 360
 Managing a FedEx Shipping Label within SoftPro 360 In order to maintain a streamlined closing process, save time and be more efficient, SoftPro 360 users now have the ability to prepare FedEx shipment
Managing a FedEx Shipping Label within SoftPro 360 In order to maintain a streamlined closing process, save time and be more efficient, SoftPro 360 users now have the ability to prepare FedEx shipment
M. tuberculosis_mpb64/is61 10 genesig Standard Kit
 TM Primerdesign Ltd M. tuberculosis_mpb64/is61 10 genesig Standard Kit 150 tests For general laboratory and research use only 1 Introduction to M.tuberculosis_MPB64/IS6110 2 Specificity MAX MIN The Primerdesign
TM Primerdesign Ltd M. tuberculosis_mpb64/is61 10 genesig Standard Kit 150 tests For general laboratory and research use only 1 Introduction to M.tuberculosis_MPB64/IS6110 2 Specificity MAX MIN The Primerdesign
RealLine Food Kit Turkey Detect
 Instruction for Use DETECTION KIT FOR DNA FROM ALL KIND OF TURKEY IN EXTREMELY PROCESSED AND CANNED FOOD AND IN OTHER PRODUCTS BY REAL-TIME-PCR This Kit is for Food Diagnostic only! CIB50700 100 tests
Instruction for Use DETECTION KIT FOR DNA FROM ALL KIND OF TURKEY IN EXTREMELY PROCESSED AND CANNED FOOD AND IN OTHER PRODUCTS BY REAL-TIME-PCR This Kit is for Food Diagnostic only! CIB50700 100 tests
Microsoft Dynamics GP. Purchase Order Processing
 Microsoft Dynamics GP Purchase Order Processing Copyright Copyright 2007 Microsoft Corporation. All rights reserved. Complying with all applicable copyright laws is the responsibility of the user. Without
Microsoft Dynamics GP Purchase Order Processing Copyright Copyright 2007 Microsoft Corporation. All rights reserved. Complying with all applicable copyright laws is the responsibility of the user. Without
Agilent 6500 Series Q-TOF LC/MS System
 Agilent 6500 Series Q-TOF LC/MS System Quick Start Guide What s New in Data Acquisition 3 Where to find information 7 Getting Started 9 Step 1. Start the Data Acquisition software 10 Step 2. Prepare the
Agilent 6500 Series Q-TOF LC/MS System Quick Start Guide What s New in Data Acquisition 3 Where to find information 7 Getting Started 9 Step 1. Start the Data Acquisition software 10 Step 2. Prepare the
FemINDICAtor qpcr Plant Gender Detection Kit on the BIO-RAD CFX96 Touch Real- Time PCR Detection System Page 1 of 11
 Page 1 of 11 Please refer to http://www.medicinalgenomics.com/product-literature/ for updated protocols and Material Safety Data Sheets (MSDS). Consult MSDS before using any new product. FEMINDICATOR is
Page 1 of 11 Please refer to http://www.medicinalgenomics.com/product-literature/ for updated protocols and Material Safety Data Sheets (MSDS). Consult MSDS before using any new product. FEMINDICATOR is
QIAGEN GeneRead QIAact Panel Cleanup Kit Handbook
 QIAGEN GeneRead QIAact Panel Cleanup Kit Handbook June 2017 For cleanup of DNA during construction of targeted, molecularly bar-coded libraries for digital sequencing with next-generation sequencing (NGS)
QIAGEN GeneRead QIAact Panel Cleanup Kit Handbook June 2017 For cleanup of DNA during construction of targeted, molecularly bar-coded libraries for digital sequencing with next-generation sequencing (NGS)
Microsoft Office Project 2010 Basic Course 01: Getting Started
 Microsoft Office Project 2010 Basic Course 01: Getting Started Slide 1 Topic A Project Management Concepts Slide 2 Project Constraints Slide 3 Phases of Project Management The initial Phase Initiating
Microsoft Office Project 2010 Basic Course 01: Getting Started Slide 1 Topic A Project Management Concepts Slide 2 Project Constraints Slide 3 Phases of Project Management The initial Phase Initiating
M. tuberculosis_mpb64/is611. genesig Advanced Kit. 150 tests. Primerdesign Ltd. For general laboratory and research use only
 TM Primerdesign Ltd M. tuberculosis_mpb64/is611 0 genesig Advanced Kit 150 tests For general laboratory and research use only 1 Introduction to M.tuberculosis_MPB64/IS6110 2 Specificity MAX MIN The Primerdesign
TM Primerdesign Ltd M. tuberculosis_mpb64/is611 0 genesig Advanced Kit 150 tests For general laboratory and research use only 1 Introduction to M.tuberculosis_MPB64/IS6110 2 Specificity MAX MIN The Primerdesign
CHAPTER 10 ACQUISITION AND PAYMENTS
 CHAPTER 10 ACQUISITION AND PAYMENTS In Chapter 9, your students set up a merchandising business and entered vendors and inventory items. In Chapter 10, they acquire inventory (make purchases) and make
CHAPTER 10 ACQUISITION AND PAYMENTS In Chapter 9, your students set up a merchandising business and entered vendors and inventory items. In Chapter 10, they acquire inventory (make purchases) and make
Relative Fluorescent Quantitation on Capillary Electrophoresis Systems:
 Application Note Relative Fluorescent Quantitation on the 3130 Series Systems Relative Fluorescent Quantitation on Capillary Electrophoresis Systems: Screening for Loss of Heterozygosity in Tumor Samples
Application Note Relative Fluorescent Quantitation on the 3130 Series Systems Relative Fluorescent Quantitation on Capillary Electrophoresis Systems: Screening for Loss of Heterozygosity in Tumor Samples
Instructions for Use. RealStar Lassa Virus RT-PCR Kit /2017 EN
 Instructions for Use RealStar Lassa Virus RT-PCR Kit 1.0 04/2017 EN RealStar Lassa Virus RT-PCR Kit 1.0 For research use only! (RUO) 641003 INS-641000-EN-S01 96 04 2017 altona Diagnostics GmbH Mörkenstr.
Instructions for Use RealStar Lassa Virus RT-PCR Kit 1.0 04/2017 EN RealStar Lassa Virus RT-PCR Kit 1.0 For research use only! (RUO) 641003 INS-641000-EN-S01 96 04 2017 altona Diagnostics GmbH Mörkenstr.
User Manual. VisiBlue qpcr mix colorant. Version 1.3 September 2012 For use in quantitative real-time PCR
 User Manual VisiBlue qpcr mix colorant Version 1.3 September 2012 For use in quantitative real-time PCR VisiBlue qpcr mix colorant Table of contents Background 4 Contents 4 Storage 4 Fluorescence data
User Manual VisiBlue qpcr mix colorant Version 1.3 September 2012 For use in quantitative real-time PCR VisiBlue qpcr mix colorant Table of contents Background 4 Contents 4 Storage 4 Fluorescence data
AmoyDx TM JAK2 V617F Mutation Detection Kit
 AmoyDx TM JAK2 V617F Mutation Detection Kit Detection of V617F mutation in the JAK2 oncogene Instructions For Use Instructions Version: B1.0 Date of Revision: July 2012 Store at -20±2 o C -1/5- Background
AmoyDx TM JAK2 V617F Mutation Detection Kit Detection of V617F mutation in the JAK2 oncogene Instructions For Use Instructions Version: B1.0 Date of Revision: July 2012 Store at -20±2 o C -1/5- Background
Neisseria gonorrhoeae Real-TM
 For in Vitro Diagnostic Use Neisseria gonorrhoeae Real-TM HANDBOOK for use with RotorGene 3000/6000/Q (Corbett Research, Qiagen), SmartCycler (Cepheid),, iq icycler and iq5 (Biorad), MX3000P and MX3005P
For in Vitro Diagnostic Use Neisseria gonorrhoeae Real-TM HANDBOOK for use with RotorGene 3000/6000/Q (Corbett Research, Qiagen), SmartCycler (Cepheid),, iq icycler and iq5 (Biorad), MX3000P and MX3005P
TaqMan PreAmp Master Mix Kit. Protocol
 TaqMan PreAmp Master Mix Kit Protocol Copyright 2007, 2010 Applied Biosystems. All rights reserved. For Research Use Only. Not for use in diagnostic procedures. Information in this document is subject
TaqMan PreAmp Master Mix Kit Protocol Copyright 2007, 2010 Applied Biosystems. All rights reserved. For Research Use Only. Not for use in diagnostic procedures. Information in this document is subject
quantitate host cell DNA with high sensitivity and throughput resdnaseq Host Cell DNA Quantitation Systems: CHO, E.
 quantitate host cell DNA with high sensitivity and throughput resdnaseq Host Cell DNA Quantitation Systems: CHO, E. coli, Vero, NS0 Precision counts Integrated real-time qpcr systems for quantitation of
quantitate host cell DNA with high sensitivity and throughput resdnaseq Host Cell DNA Quantitation Systems: CHO, E. coli, Vero, NS0 Precision counts Integrated real-time qpcr systems for quantitation of
Same TaqMan Assay quality, new format
 Product Bulletin TaqMan Array Plates TaqMan Array Plates TaqMan Gene Expression Assays delivered in ready-to-use 96-well Custom Array Plates or predefined Gene Signature Plates Flexible choose from customizable
Product Bulletin TaqMan Array Plates TaqMan Array Plates TaqMan Gene Expression Assays delivered in ready-to-use 96-well Custom Array Plates or predefined Gene Signature Plates Flexible choose from customizable
Axiom TM 2.0 Automated Target Prep Protocol
 Quick Reference Card Axiom TM 2.0 Automated Target Prep Protocol Stage 1. DNA Amplification Introduction Running the Axiom 2.0 Assay requires the following sets of steps: 1. Genomic DNA Prep, described
Quick Reference Card Axiom TM 2.0 Automated Target Prep Protocol Stage 1. DNA Amplification Introduction Running the Axiom 2.0 Assay requires the following sets of steps: 1. Genomic DNA Prep, described
DNA Genotyping from Human FFPE Samples Reliable and Reproducible
 DNA Genotyping from Human FFPE Samples Reliable and Reproducible Use this optimized protocol to obtain reliable and reproducible SNP Genotyping data from those difficult human FFPE samples. 1 FFPE DNA Isolation
DNA Genotyping from Human FFPE Samples Reliable and Reproducible Use this optimized protocol to obtain reliable and reproducible SNP Genotyping data from those difficult human FFPE samples. 1 FFPE DNA Isolation
Octet RED384 Getting Started. Sample Preparation Guidelines. Recommended concentration ranges
 Octet RED384 Getting Started Sample Preparation Guidelines Recommended concentration ranges Ligand (to be immobilized) concentration Analyte concentration 10-50 µg/ml (~µm range) 0.01-100xKD (0.1-10xKD)
Octet RED384 Getting Started Sample Preparation Guidelines Recommended concentration ranges Ligand (to be immobilized) concentration Analyte concentration 10-50 µg/ml (~µm range) 0.01-100xKD (0.1-10xKD)
KAPA PROBE FAST qpcr Master Mix (2X) Kit
 KR0397_S v1.17 Product Description s are designed for fast-cycling, real-time PCR (qpcr) using sequence-specific fluorogenic probes. These kits are compatible with all fluorogenic probe-based technologies,
KR0397_S v1.17 Product Description s are designed for fast-cycling, real-time PCR (qpcr) using sequence-specific fluorogenic probes. These kits are compatible with all fluorogenic probe-based technologies,
User Bulletin #2. ABI PRISM 7700 Sequence Detection System. Introduction. Contents. December 11, 1997
 User Bulletin #2 ABI PRISM 7700 Sequence Detection System December 11, 1997 SUBJECT: Relative Quantitation of Gene Expression Introduction Amplification of an endogenous control may be performed to standardize
User Bulletin #2 ABI PRISM 7700 Sequence Detection System December 11, 1997 SUBJECT: Relative Quantitation of Gene Expression Introduction Amplification of an endogenous control may be performed to standardize
Solar Eclipse Product Serial Numbers. Release 9.0.1
 Solar Eclipse Product Serial Numbers Release 9.0.1 Disclaimer This document is for informational purposes only and is subject to change without notice. This document and its contents, including the viewpoints,
Solar Eclipse Product Serial Numbers Release 9.0.1 Disclaimer This document is for informational purposes only and is subject to change without notice. This document and its contents, including the viewpoints,
Power SYBR Green RNA-to-C T 1-Step Kit. Protocol
 1-Step Kit Protocol For Research Use Use Only. Not intended for any animal or human therapeutic or diagnostic use. Information in this document is subject to change without notice. APPLIED BIOSYSTEMS DISCLAIMS
1-Step Kit Protocol For Research Use Use Only. Not intended for any animal or human therapeutic or diagnostic use. Information in this document is subject to change without notice. APPLIED BIOSYSTEMS DISCLAIMS
University of Massachusetts Amherst * Boston * Dartmouth * Lowell * President s Office * Worcester
 Create a Requisition through the UMASS Marketplace This job aid provides the steps required for entering a basic eprocurement requisition using the UMass Marketplace. Step 1. Log into the Finance system
Create a Requisition through the UMASS Marketplace This job aid provides the steps required for entering a basic eprocurement requisition using the UMass Marketplace. Step 1. Log into the Finance system
Purchase Order, Requisitions, Inventory Hands On. Workshop: Purchase Order, Requisitions, Inventory Hands On
 Workshop: Purchase Order, Requisitions, Inventory Hands In this follow up session to the Operations Changes in Purchase Order, Requisition, and Inventory Theory course, this hands on session will look
Workshop: Purchase Order, Requisitions, Inventory Hands In this follow up session to the Operations Changes in Purchase Order, Requisition, and Inventory Theory course, this hands on session will look
Oracle Fusion Applications
 Oracle Fusion Applications Reporting and Analytics Handbook Release 11.1.6 E41684-01 August 2013 Explains how to use Oracle Fusion Applications to perform current state analysis of business applications.
Oracle Fusion Applications Reporting and Analytics Handbook Release 11.1.6 E41684-01 August 2013 Explains how to use Oracle Fusion Applications to perform current state analysis of business applications.
AmpFlSTR NGM SElect PCR Amplification Kit. User Guide
 AmpFlSTR NGM SElect PCR Amplification Kit User Guide Information in this document is subject to change without notice. Applied Biosystems assumes no responsibility for any errors that may appear in this
AmpFlSTR NGM SElect PCR Amplification Kit User Guide Information in this document is subject to change without notice. Applied Biosystems assumes no responsibility for any errors that may appear in this
