Laboratory Service Report
|
|
|
- Ruth Dorsey
- 5 years ago
- Views:
Transcription
1 Laboratory Service Report SA SA Client SA /14/ :00 C702-DLP ROCHESTER 0/17/ :39 eningoencephalitis Comp Panel, S REPORTED 0/16/ :26 Lymphocytic Choriomeningitis Ab LC IgG H 1:16 Y03 LC Ig H 1:40 Y03 COENT: Due to a low/negative IgG and a positive Ig, the presence easles (Rubeola) Ab Panel, IFA easles (Rubeola) IgG, IFA H 1: Y03 easles (Rubeola) Ig, IFA H 1:40 Y03 COENT: Due to a low/negative IgG and a positive Ig, the presence umps Antibody Panel, IFA umps Ab IgG, IFA H 1:16 Y03 umps Ab Ig, IFA H 1:40 Y03 COENT: Due to a low/negative IgG and a positive Ig, the presence E. Equine Enceph Virus Ab Panel, IFA E. Equine Enceph. Virus IgG H 1:64 Y03 ***ing Site Legend on Last Page of Report*** 0/14/ :00 Page 1 of ** Reprinted ** >> Continued on Next Page >>
2 Laboratory Service Report SA SA Client SA /14/ :00 C702-DLP ROCHESTER 0/17/ :39 E. Equine Enceph. Virus Ig H 1:12 Y03 RECENT/CURRENT INFECTION Calif Enceph Virus Ab Pnl, IFA CA Enceph. Virus IgG H 1:16 Y03 CA Enceph. Virus Ig H 1:32 Y03 COENT: Due to a low/negative IgG and a positive Ig, the presence St. Louis Enceph Virus Ab, IFA St. Louis Enceph. Virus IgG H 1:256 Y03 St. Louis Enceph. Virus Ig H >=1:256 Y03 RECENT/CURRENT INFECTION W. Equine Enceph Ab Panel, IFA W. Equine Enceph. Virus IgG H 1:16 Y03 W. Equine Enceph. Virus Ig H 1:32 Y03 COENT: Due to a low/negative IgG and a positive Ig, the presence IFA REFERENCE RANGES: IgG (ALL VIRUSES EXCEPT EASLES) <1:16 IgG (EASLES VIRUS) <1: Ig (LC, EASLES, UPS) <1:20 ***ing Site Legend on Last Page of Report*** 0/14/ :00 Page 2 of ** Reprinted ** >> Continued on Next Page >>
3 Laboratory Service Report SA SA Client SA /14/ :00 C702-DLP ROCHESTER 0/17/ :39 Ig (ENCEPHALITIS VIRUSES) <1:16 Cytomegalovirus (CV) Ab Pnl, ELISA Cytomegalovirus IgG, ELISA H 1.10 Y03 Cytomegalovirus Ig, ELISA H 1.10 Y03 REFERENCE RANGE: IgG <0.0 Ig <0.90 INTERPRETIVE CRITERIA: IgG: <0.0 Antibody not detected Equivocal > or = 1.00 Antibody detected Ig: <0.90 Antibody not detected Equivocal > or = 1.10 Antibody detected Antibody to lymphocytic choriomeningitis virus is often detectable within a few days of clinical symptoms. The presence of mumps IgG antibody in the absence of mumps Ig antibody indicates prior exposure and immunity to mumps virus. easles Ig antibody is typically detectable for only 30 days after rash onset. Detection of CV IgG antibody indicates prior exposure. CV Ig antibody may persist for up to 2 years following primary infection. Human infections caused by arboviruses are seasonal, from mid-summer to late summer. Typical geographic distributions are: Eastern equine encephalitis virus from New England to Texas, California encephalitis virus in the north central states, St. Louis encephalitis virus throughout the southern, southwestern, and west central states, and Western encephalitis virus throughout the western states. For all viruses mentioned, the presence of Ig antibody or a four fold increase in IgG titer between acute and convalescent sera indicates recent or current infection. NOTE: Positive results for arbovirus antibody ***ing Site Legend on Last Page of Report*** 0/14/ :00 Page 3 of ** Reprinted ** >> Continued on Next Page >>
4 Laboratory Service Report SA SA Client SA /14/ :00 C702-DLP ROCHESTER 0/17/ :39 are CDC reportable. Please contact your local public health agency. The LC, measles, and mumps antibody tests were developed and their performance characteristics have been determined by Focus Diagnostics. They have not been cleared or approved by the U.S. Food and Drug Administration. The FDA has determined that such clearance or approval is not necessary. ance characteristics refer to the analytical performance of the tests. Adenovirus Antibody Adenovirus Ab H 1: Y03 Influenza Virus A and B Ab Influenza A Ab H 1: Y03 Influenza B Ab H 1:16 Y03 Varicella-Zoster Virus Antibody Varicella-Zoster Virus Ab H 1: Y03 Coxsackie A Antibodies, Serum Coxsackie A2 Ab H 1: Y03 Coxsackie A4 Ab H 1:16 Y03 Coxsackie A7 Ab H 1:32 Y03 Coxsackie A9 Ab H >=1:64 Y03 Coxsackie A10 Ab H 1: Y03 Coxsackie A16 Ab H 1:16 Y03 Coxsackie B(1-6) Antibodies, Serum Coxsackie B1 Ab H >=1:64 Y03 Coxsackie B2 Ab H 1:32 Y03 Coxsackie B3 Ab H 1:16 Y03 Coxsackie B4 Ab H 1: Y03 ***ing Site Legend on Last Page of Report*** 0/14/ :00 Page 4 of ** Reprinted ** >> Continued on Next Page >>
5 Laboratory Service Report SA SA Client SA /14/ :00 C702-DLP ROCHESTER 0/17/ :39 Coxsackie B5 Ab H 1: Y03 Coxsackie B6 Ab H 1:16 Y03 Echovirus Antibodies Echovirus 4 Ab H 1: Y03 Echovirus 7 Ab H 1:16 Y03 Echovirus 9 Ab H 1:32 Y03 Echovirus 11 Ab H >=1:64 Y03 Echovirus 30 Ab H 1: Y03 CF REFERENCE RANGES: <1: CF INTERPRETIVE CRITERIA: <1: Antibody Not Detected 1: - 1:16 (Coxsackie A,B, Echovirus) Equivocal 1: - 1:32 (Adenovirus, Influenza A,B) Equivocal 1: - 1:12 (Varicella Zoster) Equivocal > or = 1:32 (Coxsackie A,B, Echovirus) Antibody Detected > or = 1:64 (Adenovirus, Influenza A,B) Antibody Detected > or = 1:256 (Varicella Zoster) Antibody Detected Single titers in the appropriate "antibody detected" range are suggestive of recent infection. Due to the shortlived nature of complement fixing antibodies, equivocal titers may also be indicative of recent infection. A four fold or greater change in titer between acute and convalescent sera is considered confirmatory evidence of infection. Among the enteroviruses (Coxsackie A, B, and Echovirus), there is considerable crossreactivity; however, the highest titer is usually associated with the infecting serotype. ***ing Site Legend on Last Page of Report*** 0/14/ :00 Page 5 of ** Reprinted ** >> Continued on Next Page >>
6 Laboratory Service Report SA SA Client SA /14/ :00 C702-DLP ROCHESTER 0/17/ :39 The complement fixation tests were developed and their performance characteristics have been determined by Focus Diagnostics. ance characteristics refer to the analytical performance of these tests. HSV 1/2 Ig and Type-Specific IgG HSV 1 IgG Index 5.25 Y03 HSV 1 IgG HSV-1 IgG ANTIBODY DETECTED HSV 2 IgG Index 4.12 Y03 HSV 2 IgG HSV-2 IgG ANTIBODY DETECTED HSV 1/2 Ig Index 1.51 Y03 HSV 1/2 Ig Confirmatory IFA POSITIVE Y03 HSV 1/2 Ig HSV 1/2 Ig ANTIBODY DETECTED REFERENCE RANGE: <0.90 IgG INDEX ANTIBODY STATUS <0.90 Antibody not detected Equivocal >1.10 Antibody detected Ig INDEX ANTIBODY STATUS <0.90 Antibody not detected Equivocal > or = 1.10 Antibody detected The HerpeSelect test system utilizes recombinant type-specific HSV-1 and HSV-2 antigens to detect only type-specific IgG antibodies. Results from these serologic assays must be correlated with ***ing Site Legend on Last Page of Report*** 0/14/ :00 Page 6 of ** Reprinted ** >> Continued on Next Page >>
7 Laboratory Service Report SA SA Client SA /14/ :00 C702-DLP ROCHESTER 0/17/ :39 clinical history and other data to evaluate the patient's HSV status. As with all serologic tests, false positives may occur. Repeat testing or utilization of a different assay may be indicated in some settings (e.g., patients with a low likelihood of HSV infection). The HSV Ig ELISA detects type-common as well as type-specific Ig antibodies; thus, the typespecificity of any HSV Ig antibodies detected cannot be reliably determined. All samples giving an equivocal or positive Ig ELISA result are confirmed by an IFA procedure. As with the HSV Ig ELISA, however, Ig reactivity in the IFA is not type-specific. West Nile Virus Ab Panel, ELISA West Nile Virus IgG H 1.31 Y03 West Nile Virus Ig H 1.31 Y03 RECENT/CURRENT INFECTION DUE TO EXPOSURE TO FLAVIVIRUSES WHICH AY INCLUDE WEST NILE VIRUS REFERENCE RANGE: IgG <1.30 Ig <0.90 INTERPRETIVE CRITERIA IgG: <1.30 Antibody not detected Equivocal >=1.50 Antibody detected Ig: <0.90 Antibody not detected Equivocal >1.10 Antibody detected West Nile Virus (WNV) Ig is usually detectable by the time symptoms appear, but IgG may not be detectable until day 4 or day 5 of illness. Although WNV Ig persists for more than a year in some patients with WNV encephalitis, detection of WNV Ig remains a reliable indicator of recent ***ing Site Legend on Last Page of Report*** 0/14/ :00 Page 7 of ** Reprinted ** >> Continued on Next Page >>
8 Laboratory Service Report SA SA Client SA /14/ :00 C702-DLP ROCHESTER 0/17/ :39 infection for most patients. Antibodies induced by other flavivirus infections (e.g., Dengue, St. Louis Encephalitis) may show crossreactivity with WNV; thus, antibody detection using this panel is not diagnostically conclusive for WNV infection. diagnosis should be based on clinical assessment and confirmatory assays, such as the plaque reduction neutralization test. * ing Site: Focus Diagnostics, Inc. Y Corporate Avenue Cypress, CA Lab Director: 0/14/ :00 Page of ** Reprinted ** ** End of Report **
Test Definition: FMEPC Meningoencephalitis (Encephalitis) Panel, CSF
 Reporting Title: Meningoencephalitis Panel CSF Performing Location: Focus Diagnostics, Specimen Requirements: Submit 5 ml spinal fluid (CSF). Refrigerate specimen after collection, and ship at refrigerate
Reporting Title: Meningoencephalitis Panel CSF Performing Location: Focus Diagnostics, Specimen Requirements: Submit 5 ml spinal fluid (CSF). Refrigerate specimen after collection, and ship at refrigerate
Test Definition: FMEPL Meningoencephalitis (Encephalitis) Panel, CSF
 Reporting Title: Meningoencephalitis Panel CSF Performing Location: Focus Diagnostics, Specimen Requirements: Submit 5 ml spinal fluid (CSF). Refrigerate specimen after collection, and ship at refrigerate
Reporting Title: Meningoencephalitis Panel CSF Performing Location: Focus Diagnostics, Specimen Requirements: Submit 5 ml spinal fluid (CSF). Refrigerate specimen after collection, and ship at refrigerate
Test Definition: FENAP Encephalitis Antibody Panel (CSF)
 Reporting Title: Performing Location: Focus Diagnostics, Specimen Requirements: Submit 5 ml of spinal fluid (CSF). Refrigerate specimen after collecting and ship at refrigerate temperature in a sterile,
Reporting Title: Performing Location: Focus Diagnostics, Specimen Requirements: Submit 5 ml of spinal fluid (CSF). Refrigerate specimen after collecting and ship at refrigerate temperature in a sterile,
Diagnostic Microbiology
 Diagnostic Microbiology 320 MIC Lecture: 4 Identification of Microbes 3/8/2014 1 Agglutination and Precipitation Reactions Agglutination testing: Antibody cross links whole-cell antigens, forming complexes
Diagnostic Microbiology 320 MIC Lecture: 4 Identification of Microbes 3/8/2014 1 Agglutination and Precipitation Reactions Agglutination testing: Antibody cross links whole-cell antigens, forming complexes
Type Specificity of Complement-Requiring and Immunoglobulin M Neutralizing Antibody in Initial Herpes Simplex Virus Infections of Humans
 INFECTION AND IMMUNITY, OCt. 1975, p. 728-732 Copyright C) 1975 American Society for Microbiology Vol. 12, No. 14 Printed in U.S.A. Type Specificity of Complement-Requiring and Immunoglobulin M Neutralizing
INFECTION AND IMMUNITY, OCt. 1975, p. 728-732 Copyright C) 1975 American Society for Microbiology Vol. 12, No. 14 Printed in U.S.A. Type Specificity of Complement-Requiring and Immunoglobulin M Neutralizing
Inaccuracy of Certain Commercial Enzyme Immunoassays in Diagnosing Genital Infections With Herpes Simplex Virus Types 1 or 2
 Microbiology and Infectious Disease / INACCURACY OF CERTAIN HSV ELISA TESTS Inaccuracy of Certain Commercial Enzyme Immunoassays in Diagnosing Genital Infections With Herpes Simplex Virus Types 1 or 2
Microbiology and Infectious Disease / INACCURACY OF CERTAIN HSV ELISA TESTS Inaccuracy of Certain Commercial Enzyme Immunoassays in Diagnosing Genital Infections With Herpes Simplex Virus Types 1 or 2
Individual-Based Correlates of Protection
 Module 8 Evaluating Immunological Correlates of Protection Session 3 Evaluating Correlates of Protection Using Individual, Population, and Titer-specific Approaches Ivan S.F. Chan, Ph.D. Merck Research
Module 8 Evaluating Immunological Correlates of Protection Session 3 Evaluating Correlates of Protection Using Individual, Population, and Titer-specific Approaches Ivan S.F. Chan, Ph.D. Merck Research
The following is an overview of diagnostic techniques for Ebola infection in humans.
 IBM Deep Dive Topic 1/14/2015 Alex // operonlabs.com Diagnostics and Ebola: The following is an overview of diagnostic techniques for Ebola infection in humans. Current Status: The current status of Ebola
IBM Deep Dive Topic 1/14/2015 Alex // operonlabs.com Diagnostics and Ebola: The following is an overview of diagnostic techniques for Ebola infection in humans. Current Status: The current status of Ebola
Issued by: LABORATORY MANAGER Original Date: March 14, 2001 Approved by: Laboratory Director Revision Date: October 10, 2003 CEREBRAL SPINAL FLUID
 Policy # MI/VIR/03/v02 Page 1 of 5 Section: Subject Title: Cerebral Spinal Fluid (CSF) Issued by: LABORATORY MANAGER Original Date: March 14, 2001 Approved by: Laboratory Director Revision Date: October
Policy # MI/VIR/03/v02 Page 1 of 5 Section: Subject Title: Cerebral Spinal Fluid (CSF) Issued by: LABORATORY MANAGER Original Date: March 14, 2001 Approved by: Laboratory Director Revision Date: October
Performance of commercial enzyme-linked immunosorbent assays (ELISA) for diagnosis of HSV-1 and HSV-2 infection in a clinical setting
 Performance of commercial enzyme-linked immunosorbent assays (ELISA) for diagnosis of HSV-1 and HSV-2 infection in a clinical setting Elfriede Ampong Agyemang A thesis submitted in partial fulfillment
Performance of commercial enzyme-linked immunosorbent assays (ELISA) for diagnosis of HSV-1 and HSV-2 infection in a clinical setting Elfriede Ampong Agyemang A thesis submitted in partial fulfillment
National Institute of Virology(NIV), Pune
 National Institute of Virology(NIV), Pune Japanese Encephalitis Vaccine Product/Process: Chimeric peptide vaccine candidate against Japanese Encephalitis virus. Application/Uses: Japanese encephalitis
National Institute of Virology(NIV), Pune Japanese Encephalitis Vaccine Product/Process: Chimeric peptide vaccine candidate against Japanese Encephalitis virus. Application/Uses: Japanese encephalitis
YELLOW FEVER DIAGNOSIS IN OUTBREAK CONTEXTS: CHALLENGES AND LIMITATIONS
 YELLOW FEVER DIAGNOSIS IN OUTBREAK CONTEXTS: CHALLENGES AND LIMITATIONS 10/19/2017 Dr. Cristina Domingo, Robert Koch Institute, Berlin, Germany YELLOW FEVER DIAGNOSIS IN OUTBREAK CONTEXTS: CHALLENGES AND
YELLOW FEVER DIAGNOSIS IN OUTBREAK CONTEXTS: CHALLENGES AND LIMITATIONS 10/19/2017 Dr. Cristina Domingo, Robert Koch Institute, Berlin, Germany YELLOW FEVER DIAGNOSIS IN OUTBREAK CONTEXTS: CHALLENGES AND
Guidance for Industry and FDA Staff. Class II Special Controls Guidance Document: Herpes Simplex Virus Types 1 and 2 Serological Assays
 Reprinted from FDA s website by Guidance for Industry and FDA Staff Class II Special Controls Guidance Document: Herpes Simplex Virus Types 1 and 2 Serological Assays Document issued on: April 3, 2007
Reprinted from FDA s website by Guidance for Industry and FDA Staff Class II Special Controls Guidance Document: Herpes Simplex Virus Types 1 and 2 Serological Assays Document issued on: April 3, 2007
1.0 Abstract. Palivizumab P Study Results Final
 1.0 Abstract Title: Prospective, Multi-Center, Observational Program to Assess RSV Hospitalization Rate in Population of Children at High-risk of Serious RSV Illness Who Received Palivizumab Immunoprophylaxis
1.0 Abstract Title: Prospective, Multi-Center, Observational Program to Assess RSV Hospitalization Rate in Population of Children at High-risk of Serious RSV Illness Who Received Palivizumab Immunoprophylaxis
Neutralizing Antibody Responses to Varicella-Zoster Virus
 INFECTION AND IMMUNrrY, Sept. 1975, p. B6-613 Copyright 1975 American Society for Microbiology Vol. 12, No. 3 Printed in U.S.A. Neutralizing Antibody Responses to Varicella-Zoster Virus NATHALIE J. SCHMIDT
INFECTION AND IMMUNrrY, Sept. 1975, p. B6-613 Copyright 1975 American Society for Microbiology Vol. 12, No. 3 Printed in U.S.A. Neutralizing Antibody Responses to Varicella-Zoster Virus NATHALIE J. SCHMIDT
Use of Sindbis/Eastern Equine Encephalitis Chimeric Viruses in Plaque Reduction Neutralization Tests for Arboviral Disease Diagnostics
 CLINICAL AND VACCINE IMMUNOLOGY, Sept. 2011, p. 1486 1491 Vol. 18, No. 9 1556-6811/11/$12.00 doi:10.1128/cvi.05129-11 Copyright 2011, American Society for Microbiology. All Rights Reserved. Use of Sindbis/Eastern
CLINICAL AND VACCINE IMMUNOLOGY, Sept. 2011, p. 1486 1491 Vol. 18, No. 9 1556-6811/11/$12.00 doi:10.1128/cvi.05129-11 Copyright 2011, American Society for Microbiology. All Rights Reserved. Use of Sindbis/Eastern
Diagnostic Virology. Diagnostic Virology
 Diagnostic Virology Diagnostic Virology The purpose is to diagnose viral diseases in the patient This can be achieved by: Directly detecting the virus or viral products Indirectly detecting an immunological
Diagnostic Virology Diagnostic Virology The purpose is to diagnose viral diseases in the patient This can be achieved by: Directly detecting the virus or viral products Indirectly detecting an immunological
CE Marked, Health Canada Licensed, and FDA Cleared / Exempt Products
 Molecular Products MOL1001 / MOL1101 3M Integrated Cycler (110 V / 220 V) CE Canada FDA MOL2150 Simplexa HSV 1 & 2 Direct CE Canada FDA MOL2160 Simplexa HSV 1 & 2 Positive Control Pack CE Canada FDA MOL2200
Molecular Products MOL1001 / MOL1101 3M Integrated Cycler (110 V / 220 V) CE Canada FDA MOL2150 Simplexa HSV 1 & 2 Direct CE Canada FDA MOL2160 Simplexa HSV 1 & 2 Positive Control Pack CE Canada FDA MOL2200
ELISA. MODULE 4 Objective 4.2 Lesson B
 MODULE 4 Objective 4.2 Lesson B ELISA Course Advanced Biotechnology Unit Biotechnology Basics Essential Question How is biotechnolog y being used to advance medical diagnostics? TEKS 130.364 1A-K, 2H,
MODULE 4 Objective 4.2 Lesson B ELISA Course Advanced Biotechnology Unit Biotechnology Basics Essential Question How is biotechnolog y being used to advance medical diagnostics? TEKS 130.364 1A-K, 2H,
Detection of Anti-Arboviral Immunoglobulin G by Using a Monoclonal Antibody-Based Capture Enzyme-Linked Immunosorbent Assay
 JOURNAL OF CLINICAL MICROBIOLOGY, May 2000, p. 1827 1831 Vol. 38, No. 5 0095-1137/00/$04.00 0 Detection of Anti-Arboviral Immunoglobulin G by Using a Monoclonal Antibody-Based Capture Enzyme-Linked Immunosorbent
JOURNAL OF CLINICAL MICROBIOLOGY, May 2000, p. 1827 1831 Vol. 38, No. 5 0095-1137/00/$04.00 0 Detection of Anti-Arboviral Immunoglobulin G by Using a Monoclonal Antibody-Based Capture Enzyme-Linked Immunosorbent
ToRCH IgG/IgM Assay Development
 ToRCH IgG/IgM Assay Development www.meridianbioscience.com/lifescience ToRCH Overview ToRCH is an acronym for a group of infections that can cause significant birth defects and even fetal death. Meridian
ToRCH IgG/IgM Assay Development www.meridianbioscience.com/lifescience ToRCH Overview ToRCH is an acronym for a group of infections that can cause significant birth defects and even fetal death. Meridian
Detection of Serotype-Specific Antibodies to the Four Dengue Viruses Using an Immune Complex Binding (ICB) ELISA
 Detection of Serotype-Specific Antibodies to the Four Dengue Viruses Using an Immune Complex Binding (ICB) ELISA Petra Emmerich 1, Angela Mika 2, Herbert Schmitz 1 * 1 Department of Virology, Bernhard
Detection of Serotype-Specific Antibodies to the Four Dengue Viruses Using an Immune Complex Binding (ICB) ELISA Petra Emmerich 1, Angela Mika 2, Herbert Schmitz 1 * 1 Department of Virology, Bernhard
PPTA Regulatory Workshop June 13, 2016
 PPTA Regulatory Workshop June 13, 2016 DOROTY SCOTT Dr. Dorothy Scott is the Branch Chief for the Laboratory of Plasma Derivatives, in the Office of Blood Research and Review, CBER. Her group is responsible
PPTA Regulatory Workshop June 13, 2016 DOROTY SCOTT Dr. Dorothy Scott is the Branch Chief for the Laboratory of Plasma Derivatives, in the Office of Blood Research and Review, CBER. Her group is responsible
Immunoassay Diagnosis of Adenovirus Infections in Children
 JOURNAL OF CLINICAL MICROBIOLOGY, Nov. 1983, p. 1190-1195 0095-1137/83/11 1190-06$02.00/0 Copyright 1983, American Society for Microbiology Vol. 18, No. 5 Immunoassay Diagnosis of Adenovirus Infections
JOURNAL OF CLINICAL MICROBIOLOGY, Nov. 1983, p. 1190-1195 0095-1137/83/11 1190-06$02.00/0 Copyright 1983, American Society for Microbiology Vol. 18, No. 5 Immunoassay Diagnosis of Adenovirus Infections
HerpeSelect 1 and 2 Immunoblot IgG (K000238; Focus Diagnostics, Inc.)
 I. GENERAL INFORMATION Submitter: Theranos, Inc. 1701 Page Mill Road Palo Alto, CA 94304 Phone: 650-838-9292 Fax: 650-838-9165 Contact Person: Brad Arington Associate Director, Regulatory Phone: 650-856-7304
I. GENERAL INFORMATION Submitter: Theranos, Inc. 1701 Page Mill Road Palo Alto, CA 94304 Phone: 650-838-9292 Fax: 650-838-9165 Contact Person: Brad Arington Associate Director, Regulatory Phone: 650-856-7304
Virus-Specific Immunoglobulin G Subclasses in Herpes Simplex and Varicella-Zoster Virus Infections
 JOURNAL OF CLINICAL MICROBIOLOGY, JUIY 1984, p. 94-98 Vol. 20, No. 1 0095-1137/84/070094-05$00/0 Copyright 1984, American Society for Microbiology Virus-Specific Immunoglobulin G Subclasses in Herpes Simplex
JOURNAL OF CLINICAL MICROBIOLOGY, JUIY 1984, p. 94-98 Vol. 20, No. 1 0095-1137/84/070094-05$00/0 Copyright 1984, American Society for Microbiology Virus-Specific Immunoglobulin G Subclasses in Herpes Simplex
TaqPath Zika Virus Kit (ZIKV) Instructions for Use
 Page 1 of 19 TaqPath Zika Virus Kit (ZIKV) Instructions for Use Thermo Fisher Scientific For in vitro Diagnostic Use For Prescription Use Only For Use Under an Emergency Use Authorization (EUA) Only Page
Page 1 of 19 TaqPath Zika Virus Kit (ZIKV) Instructions for Use Thermo Fisher Scientific For in vitro Diagnostic Use For Prescription Use Only For Use Under an Emergency Use Authorization (EUA) Only Page
The Sensitivity Comparison of Immunodiagnostic Assays for Diagnosing Dengue Fever
 ORIGINAL ARTICLE Korean J Clin Lab Sci. 2016;48(4):275-279 https://doi.org/10.15324/kjcls.2016.48.4.275 pissn 1738-3544 eissn 2288-1662 Korean J Clin Lab Sci. Vol. 48, No. 4, December 2016 275 The Sensitivity
ORIGINAL ARTICLE Korean J Clin Lab Sci. 2016;48(4):275-279 https://doi.org/10.15324/kjcls.2016.48.4.275 pissn 1738-3544 eissn 2288-1662 Korean J Clin Lab Sci. Vol. 48, No. 4, December 2016 275 The Sensitivity
IgG Antibodies TO Rubella Virus ELISA Kit Protocol
 IgG Antibodies TO Rubella Virus ELISA Kit Protocol (Cat. No.:EK-310-81) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com INTENDED
IgG Antibodies TO Rubella Virus ELISA Kit Protocol (Cat. No.:EK-310-81) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com INTENDED
trial. Key trial data points:
 February 23, 2015 ADMA Biologics Announces Positive Data on Primary and Secondary Endpoints from its Pivotal Phase III Clinical Trial for RI-002 at the AAAAI Medical Conference RAMSEY, N.J., Feb. 23, 2015
February 23, 2015 ADMA Biologics Announces Positive Data on Primary and Secondary Endpoints from its Pivotal Phase III Clinical Trial for RI-002 at the AAAAI Medical Conference RAMSEY, N.J., Feb. 23, 2015
Immunogenicity of Therapeutic Proteins. Steven J Swanson, Ph.D. Executive Director, Clinical Immunology
 Immunogenicity of Therapeutic Proteins Steven J Swanson, Ph.D. Executive Director, Clinical Immunology swanson@amgen.com Causes of Immunogenicity Sequence differences between therapeutic protein and endogenous
Immunogenicity of Therapeutic Proteins Steven J Swanson, Ph.D. Executive Director, Clinical Immunology swanson@amgen.com Causes of Immunogenicity Sequence differences between therapeutic protein and endogenous
Emergency Use Assessment and Listing of IVDs
 P r e q u a l i f i c a t i o n T e a m - D i a g n o s t i c s Instructions for Submission Requirements: In vitro diagnostics (IVDs) Detecting Zika Virus Nucleic Acid or Antigen Emergency Use Assessment
P r e q u a l i f i c a t i o n T e a m - D i a g n o s t i c s Instructions for Submission Requirements: In vitro diagnostics (IVDs) Detecting Zika Virus Nucleic Acid or Antigen Emergency Use Assessment
Office of. Integrated Surveillance and Informatics Services (ISIS) Standard Operating Procedures
 Office of Integrated Surveillance and Informatics Services (ISIS) Standard Operating Procedures GUIDE TO EVENT CLASSIFICATION AND LABORATORY INTERPRETATION Q:\RESOURCES\ISIS SURVEILLANCE\Case Classification
Office of Integrated Surveillance and Informatics Services (ISIS) Standard Operating Procedures GUIDE TO EVENT CLASSIFICATION AND LABORATORY INTERPRETATION Q:\RESOURCES\ISIS SURVEILLANCE\Case Classification
Received 19 September 2008/Returned for modification 4 November 2008/Accepted 13 November 2008
 CLINICAL AND VACCINE IMMUNOLOGY, Jan. 2009, p. 55 60 Vol. 16, No. 1 1556-6811/09/$08.00 0 doi:10.1128/cvi.00351-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Comparison of
CLINICAL AND VACCINE IMMUNOLOGY, Jan. 2009, p. 55 60 Vol. 16, No. 1 1556-6811/09/$08.00 0 doi:10.1128/cvi.00351-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Comparison of
Step-by-Step Description of ELISA
 Step-by-Step Description of ELISA The protocols in this kit rely on indirect antibody capture ELISA. The steps in this assay are: Step 1: Antigen is added to the wells of the microplate strip and incubated
Step-by-Step Description of ELISA The protocols in this kit rely on indirect antibody capture ELISA. The steps in this assay are: Step 1: Antigen is added to the wells of the microplate strip and incubated
Design Verification T IgG ELISA C Determination of Diagnostic Sensitivity and Diagnostic Specificity... 2 Crossreactivity... 3
 Design Verification TOXO IgG ELISA CONTENTS 1 Determination of Diagnostic Sensitivity and Diagnostic Specificity... 2 1.1 External Study:... 2 1.2 Internal Evaluation... 3 2 Crossreactivity... 3 3 Potentially
Design Verification TOXO IgG ELISA CONTENTS 1 Determination of Diagnostic Sensitivity and Diagnostic Specificity... 2 1.1 External Study:... 2 1.2 Internal Evaluation... 3 2 Crossreactivity... 3 3 Potentially
Herpes Simplex 1 IgM (HSV1 IgM)
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Observations about complement were carried out by Nuthall Pfeiffer and Bordet in the 1800's.
 COMPLEMENT SYSTEM Observations about complement were carried out by Nuthall Pfeiffer and Bordet in the 1800's. Researchers compared cholera vibrio with immune fresh serum in the test tube and; Cholera
COMPLEMENT SYSTEM Observations about complement were carried out by Nuthall Pfeiffer and Bordet in the 1800's. Researchers compared cholera vibrio with immune fresh serum in the test tube and; Cholera
Nipah-available assays and reference materials Need for real time diagnostics in proximity to Outbreak-Mobile lab
 Nipah-available assays and reference materials Need for real time diagnostics in proximity to Outbreak-Mobile lab Christina F. Spiropoulou Viral Special Pathogens Branch Centers for Disease Control and
Nipah-available assays and reference materials Need for real time diagnostics in proximity to Outbreak-Mobile lab Christina F. Spiropoulou Viral Special Pathogens Branch Centers for Disease Control and
Human IgG Rubella ELISA Kit
 Human IgG Rubella ELISA Kit Catalog No: IRAPKT2020 Lot No: SAMPLE INTENDED USE The Rubella IgG ELISA is intended for use in evaluating a patient s serologic status to the rubella virus infection. It is
Human IgG Rubella ELISA Kit Catalog No: IRAPKT2020 Lot No: SAMPLE INTENDED USE The Rubella IgG ELISA is intended for use in evaluating a patient s serologic status to the rubella virus infection. It is
Pre-existing anti-viral vector antibodies in gene therapy
 Pre-existing anti-viral vector antibodies in gene therapy Impact on assays, study conduct and data interpretation Mark N Milton, Executive Director DMPK-Bx, Novartis AAPS NBC, May 2016 Gene Therapy Treatment
Pre-existing anti-viral vector antibodies in gene therapy Impact on assays, study conduct and data interpretation Mark N Milton, Executive Director DMPK-Bx, Novartis AAPS NBC, May 2016 Gene Therapy Treatment
Infectious Diseases Serology Explained
 This image cannot currently be displayed. Infectious Diseases Serology Explained David Dickeson Centre for Infectious Diseases & Microbiology Laboratory Services Why use serology? To determine immune status
This image cannot currently be displayed. Infectious Diseases Serology Explained David Dickeson Centre for Infectious Diseases & Microbiology Laboratory Services Why use serology? To determine immune status
SERION ELISA classic Antibody Quantification
 YOUR GLOBAL PARTNER IN DIAGNOSTICS SERION ELISA classic Antibody Quantification Optical Density OD 3,0 2,0 Sample OD 1,0 0,1 1 10 100 1000 Sample Activity U/ml Antibody Activity serion multianalyt TM Diagnosis
YOUR GLOBAL PARTNER IN DIAGNOSTICS SERION ELISA classic Antibody Quantification Optical Density OD 3,0 2,0 Sample OD 1,0 0,1 1 10 100 1000 Sample Activity U/ml Antibody Activity serion multianalyt TM Diagnosis
Chapter 17: Immunization & Immune Testing. 1. Immunization 2. Diagnostic Immunology
 Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
1. Immunization. What is Immunization? 12/9/2016. Chapter 17: Immunization & Immune Testing. 1. Immunization 2. Diagnostic Immunology
 Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
dengue virus by multiplex RT-PCR and real-time SYBR green RT-PCR
 Original Article Singapore Med 2007, 48 (7) : 662 Rapid detection and serotyping of dengue virus by multiplex RT-PCR and real-time SYBR green RT-PCR Yong Y K, Thayan R, Chong H T, Tan C T, Sekaran S D
Original Article Singapore Med 2007, 48 (7) : 662 Rapid detection and serotyping of dengue virus by multiplex RT-PCR and real-time SYBR green RT-PCR Yong Y K, Thayan R, Chong H T, Tan C T, Sekaran S D
21 st Century Herpes Solutions. William Halford, PhD RVx, Chief Science Officer
 21 st Century Herpes Solutions William Halford, PhD RVx, Chief Science Officer Scope of Herpes Problem: by the numbers A major public health concern HSV-1 and/or HSV-2 HSV-2 infections ~100 million recurrent
21 st Century Herpes Solutions William Halford, PhD RVx, Chief Science Officer Scope of Herpes Problem: by the numbers A major public health concern HSV-1 and/or HSV-2 HSV-2 infections ~100 million recurrent
CHAPTER 24. Immunology
 CHAPTER 24 Diagnostic i Microbiology and Immunology Growth-Dependent Diagnostic Methods Isolation of Pathogens from Clinical Specimens Proper sampling and culture of a suspected pathogen is the most reliable
CHAPTER 24 Diagnostic i Microbiology and Immunology Growth-Dependent Diagnostic Methods Isolation of Pathogens from Clinical Specimens Proper sampling and culture of a suspected pathogen is the most reliable
Dream or reality?!"# $$%&' ( ) **+**
 Dream or reality? 1!"# $$%&' &# ( ) **+**,' 2 1 Assays based on specific recognition of nucleic acids, e.g. RNA or DNA Detection = qualitative tests e.g. Chlamydia trachomatis, Neisseria gonorrhoeae etc.
Dream or reality? 1!"# $$%&' &# ( ) **+**,' 2 1 Assays based on specific recognition of nucleic acids, e.g. RNA or DNA Detection = qualitative tests e.g. Chlamydia trachomatis, Neisseria gonorrhoeae etc.
Received 10 May 2007/Returned for modification 2 August 2007/Accepted 2 November 2007
 CLINICAL AND VACCINE IMMUNOLOGY, Jan. 2008, p. 159 163 Vol. 15, No. 1 1556-6811/08/$08.00 0 doi:10.1128/cvi.00218-07 Copyright 2008, American Society for Microbiology. All Rights Reserved. Rapid, Sensitive,
CLINICAL AND VACCINE IMMUNOLOGY, Jan. 2008, p. 159 163 Vol. 15, No. 1 1556-6811/08/$08.00 0 doi:10.1128/cvi.00218-07 Copyright 2008, American Society for Microbiology. All Rights Reserved. Rapid, Sensitive,
Received 10 May 2007/Returned for modification 2 August 2007/Accepted 2 November 2007
 CLINICAL AND VACCINE IMMUNOLOGY, Jan. 2008, p. 159 163 Vol. 15, No. 1 1556-6811/08/$08.00 0 doi:10.1128/cvi.00218-07 Copyright 2008, American Society for Microbiology. All Rights Reserved. Rapid, Sensitive,
CLINICAL AND VACCINE IMMUNOLOGY, Jan. 2008, p. 159 163 Vol. 15, No. 1 1556-6811/08/$08.00 0 doi:10.1128/cvi.00218-07 Copyright 2008, American Society for Microbiology. All Rights Reserved. Rapid, Sensitive,
Potency Bioassays: Cell-based assay
 Potency Bioassays: Cell-based assay Nongyao Somdach Institution of Biological Product Department of Medical Sciences Ministry of Public Health 1 Outline Potency-What?-Why? Vaccine-What?- Why? Potency assay
Potency Bioassays: Cell-based assay Nongyao Somdach Institution of Biological Product Department of Medical Sciences Ministry of Public Health 1 Outline Potency-What?-Why? Vaccine-What?- Why? Potency assay
CDRH: Advancing Regulatory Science. Murray Malin, MD Center for Devices and Radiological Health March 29, 2011
 CDRH: Advancing Regulatory Science Murray Malin, MD Center for Devices and Radiological Health March 29, 2011 Medical Countermeasures Diagnostic and Detection Devices Personal Protective Equipment (PPE)
CDRH: Advancing Regulatory Science Murray Malin, MD Center for Devices and Radiological Health March 29, 2011 Medical Countermeasures Diagnostic and Detection Devices Personal Protective Equipment (PPE)
Measurement of uncertainty for Elisa Tests. University of Hasselt, Center for Statistics, Hasselt, Belgium
 Appendix 58 Measurement of uncertainty for Elisa Tests Toussaint, J.F. 1*, Assam, P. 2, Caij, B. 1, Dekeyser, F. 1, Imberechts, H. 1, Knapen, K. 1, Goris N. 1, Molenberghs, G. 2, Mintiens, K. 1, De Clercq,
Appendix 58 Measurement of uncertainty for Elisa Tests Toussaint, J.F. 1*, Assam, P. 2, Caij, B. 1, Dekeyser, F. 1, Imberechts, H. 1, Knapen, K. 1, Goris N. 1, Molenberghs, G. 2, Mintiens, K. 1, De Clercq,
Qualitative determination of Anti-Treponema pallidum antibodies For in vitro use only Store at 2-8 C
 Qualitative determination of Anti-Treponema pallidum antibodies For in vitro use only Store at 2-8 C INTRODUCTION Syphilis is a venereal disease caused by infection with T. pallidum. The microorganism
Qualitative determination of Anti-Treponema pallidum antibodies For in vitro use only Store at 2-8 C INTRODUCTION Syphilis is a venereal disease caused by infection with T. pallidum. The microorganism
Learning from Past Emergency Use Authorizations
 Learning from Past Emergency Use Authorizations Elliot P. Cowan, Ph.D. Principal, Partners in Diagnostics, LLC PDC Workshop on New and Innovative Approaches to Laboratory Diagnosis of Zika, Dengue and
Learning from Past Emergency Use Authorizations Elliot P. Cowan, Ph.D. Principal, Partners in Diagnostics, LLC PDC Workshop on New and Innovative Approaches to Laboratory Diagnosis of Zika, Dengue and
Best practice for tissue and tests for screening for PD/SAV? Dr. Hilde Sindre Fish health research group Norwegian Veterinary Institute
 Best practice for tissue and tests for screening for PD/SAV? Dr. Hilde Sindre Fish health research group Norwegian Veterinary Institute Diagnostic criteria SAV/PD (from the OIE manual) A suspected case
Best practice for tissue and tests for screening for PD/SAV? Dr. Hilde Sindre Fish health research group Norwegian Veterinary Institute Diagnostic criteria SAV/PD (from the OIE manual) A suspected case
LECTURE: 26 SIMPLE SEROLOGICAL LABORATORY TECHNIQUES LEARNING OBJECTIVES:
 LECTURE: 26 Title SIMPLE SEROLOGICAL LABORATORY TECHNIQUES LEARNING OBJECTIVES: The student should be able to: Define the term "simple serological techniques". Describe the benefit of the use of serological
LECTURE: 26 Title SIMPLE SEROLOGICAL LABORATORY TECHNIQUES LEARNING OBJECTIVES: The student should be able to: Define the term "simple serological techniques". Describe the benefit of the use of serological
Vaccines and other immunological antimicrobial therapy 2
 Vaccines and other immunological antimicrobial therapy 2 The Development of New Vaccines Golden age of immunology: 1870-1910 Introduction of antibiotics: decrease vaccine development Safety issue and fear
Vaccines and other immunological antimicrobial therapy 2 The Development of New Vaccines Golden age of immunology: 1870-1910 Introduction of antibiotics: decrease vaccine development Safety issue and fear
CHAPTER 3 DEVELOPMENT OF DENV GROUP SPECIFIC REAL TIME RT-PCR
 CHAPTER 3 DEVELOPMENT OF DENV GROUP SPECIFIC REAL TIME RT-PCR 28 Dengue is diagnosed by either detecting virus or antibody to the virus in blood. Isolation of virus in cell culture or in infant mouse brain
CHAPTER 3 DEVELOPMENT OF DENV GROUP SPECIFIC REAL TIME RT-PCR 28 Dengue is diagnosed by either detecting virus or antibody to the virus in blood. Isolation of virus in cell culture or in infant mouse brain
Technical Reports LEISHMANIA RECOMBINANT ANTIGENS KMP11 AND K39
 Technical Reports LEISHMANIA RECOMBINANT ANTIGENS KMP11 AND K39 The aim of the research presented in this technical report is to show the evaluation of the Leishmania recombinant antigens KMP-11 and K39
Technical Reports LEISHMANIA RECOMBINANT ANTIGENS KMP11 AND K39 The aim of the research presented in this technical report is to show the evaluation of the Leishmania recombinant antigens KMP-11 and K39
Enzyme-Linked Immunosorbent Assays Using Novel Japanese Encephalitis Virus Antigen Improve the Accuracy of Clinical Diagnosis of Flavivirus Infections
 CLINICAL AND VACCINE IMMUNOLOGY, May 2008, p. 825 835 Vol. 15, No. 5 1556-6811/08/$08.00 0 doi:10.1128/cvi.00004-08 Copyright 2008, American Society for Microbiology. All Rights Reserved. Enzyme-Linked
CLINICAL AND VACCINE IMMUNOLOGY, May 2008, p. 825 835 Vol. 15, No. 5 1556-6811/08/$08.00 0 doi:10.1128/cvi.00004-08 Copyright 2008, American Society for Microbiology. All Rights Reserved. Enzyme-Linked
The Molecular Virology Support Core: Adenoviral Vectors and Beyond
 The : Adenoviral Vectors and Beyond Christoph A. Kahl, Ph.D. Viral Vector Workshop, May 5th, 2011 Outline 1) Overview of the MVSC 2) Adenoviral Vectors 3) Expertise and Services Overview What does the
The : Adenoviral Vectors and Beyond Christoph A. Kahl, Ph.D. Viral Vector Workshop, May 5th, 2011 Outline 1) Overview of the MVSC 2) Adenoviral Vectors 3) Expertise and Services Overview What does the
Enzyme-Linked Immunosorbent Assays Using Novel Japanese Encephalitis Virus Antigen Improve the Accuracy of Clinical Diagnosis of Flavivirus Infections
 CLINICAL AND VACCINE IMMUNOLOGY, May 2008, p. 825 835 Vol. 15, No. 5 1556-6811/08/$08.00 0 doi:10.1128/cvi.00004-08 Copyright 2008, American Society for Microbiology. All Rights Reserved. Enzyme-Linked
CLINICAL AND VACCINE IMMUNOLOGY, May 2008, p. 825 835 Vol. 15, No. 5 1556-6811/08/$08.00 0 doi:10.1128/cvi.00004-08 Copyright 2008, American Society for Microbiology. All Rights Reserved. Enzyme-Linked
Herpes Simplex 1 IgM (HSV1 IgM)
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Critical Quality Attributes for Live Viral Vaccines. Mark S. Galinski Vaccine Analytical Sciences, MedImmune, Mountain View, CA USA
 Critical Quality Attributes for Live Viral Vaccines Mark S. Galinski Vaccine Analytical Sciences, MedImmune, Mountain View, CA USA Terminology Quality Release Standards The specifications and procedures
Critical Quality Attributes for Live Viral Vaccines Mark S. Galinski Vaccine Analytical Sciences, MedImmune, Mountain View, CA USA Terminology Quality Release Standards The specifications and procedures
Herpes Simplex Virus Type 1 (HSV-1) IgM ELISA Kit Protocol
 Herpes Simplex Virus Type 1 (HSV-1) IgM ELISA Kit Protocol (Cat. No.:EK-310-93) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com
Herpes Simplex Virus Type 1 (HSV-1) IgM ELISA Kit Protocol (Cat. No.:EK-310-93) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com
Lyme and other Tickborne diseases 2018 What do we know? What do we need to know?
 Lyme and other Tickborne diseases 2018 What do we know? What do we need to know? Holly Ahern, MS, BS, MT(ASCP) Assoc. Prof. of Microbiology, SUNY Adirondack; VP Lyme Action Network ahernh@sunyacc.edu From
Lyme and other Tickborne diseases 2018 What do we know? What do we need to know? Holly Ahern, MS, BS, MT(ASCP) Assoc. Prof. of Microbiology, SUNY Adirondack; VP Lyme Action Network ahernh@sunyacc.edu From
Determinations of Antibody to Herpes Simplex Virus in Sera
 JOURNAL OF CLINICAL MICROBIOLOGY, May 1982, p. 815-823 Vol. 15, No. 5 95-1137/82/5815-9$2./ Evaluation and Reporting of Enzyme Immunoassay Determinations of Antibody to Herpes Simplex Virus in Sera and
JOURNAL OF CLINICAL MICROBIOLOGY, May 1982, p. 815-823 Vol. 15, No. 5 95-1137/82/5815-9$2./ Evaluation and Reporting of Enzyme Immunoassay Determinations of Antibody to Herpes Simplex Virus in Sera and
FDA Draft Guidance on Immunogenicity Testing
 FDA Draft Guidance on Immunogenicity Testing Susan Kirshner, Ph.D. Associate Chief, Laboratory of Immunology Division of Therapeutic Proteins OBP/CDER/FDA EBF 2010 Guidance for Industry Assay Development
FDA Draft Guidance on Immunogenicity Testing Susan Kirshner, Ph.D. Associate Chief, Laboratory of Immunology Division of Therapeutic Proteins OBP/CDER/FDA EBF 2010 Guidance for Industry Assay Development
Immunogenicity. How to deal with? Nathalie Macé Sanofi, Biomarkers & Biological analyses Unit
 Immunogenicity How to deal with? Nathalie Macé Sanofi, Biomarkers & Biological analyses Unit Club Phase I, 22 March 2016 1 Outline Introduction to immunogenicity Analytical challenges for immunogenicity
Immunogenicity How to deal with? Nathalie Macé Sanofi, Biomarkers & Biological analyses Unit Club Phase I, 22 March 2016 1 Outline Introduction to immunogenicity Analytical challenges for immunogenicity
ANTIBODIES A LABORATORY SECOND EDITION
 page 1 / 5 page 2 / 5 antibodies a laboratory second pdf Thyroid autoantibodies are antibodies that develop when a person's immune system mistakenly targets components of the thyroid gland or thyroid proteins,
page 1 / 5 page 2 / 5 antibodies a laboratory second pdf Thyroid autoantibodies are antibodies that develop when a person's immune system mistakenly targets components of the thyroid gland or thyroid proteins,
Detection, Pathogenesis, and Therapy of Respiratory Syncytial
 CLINICAL MICROBIOLOGY REVIEWS, Jan. 1988, p. 27-39 Vol. 1, No. 1 0893-8512/88/010027-13$02.00/0 Copyright C 1988, American Society for Microbiology Detection, Pathogenesis, and Therapy of Respiratory Syncytial
CLINICAL MICROBIOLOGY REVIEWS, Jan. 1988, p. 27-39 Vol. 1, No. 1 0893-8512/88/010027-13$02.00/0 Copyright C 1988, American Society for Microbiology Detection, Pathogenesis, and Therapy of Respiratory Syncytial
Selection and use of Ebola in vitro diagnostic (IVD) assays
 EMERGENCY GUIDANCE Selection and use of Ebola in vitro diagnostic (IVD) assays June 2015 World Health Organization 2015. All rights reserved. The designations employed and the presentation of the material
EMERGENCY GUIDANCE Selection and use of Ebola in vitro diagnostic (IVD) assays June 2015 World Health Organization 2015. All rights reserved. The designations employed and the presentation of the material
Assays for Immunogenicity: Are We There Yet?
 Assays for Immunogenicity: Are We There Yet? Mark Wener, MD Department of Laboratory Medicine & Rheumatology Division Department of Medicine University of Washington Seattle, WA 98195 wener@uw.edu Goals:
Assays for Immunogenicity: Are We There Yet? Mark Wener, MD Department of Laboratory Medicine & Rheumatology Division Department of Medicine University of Washington Seattle, WA 98195 wener@uw.edu Goals:
May Immunohistochemistry. Immunology. Microbiology. Referral Testing. ROS1 screening
 May 2017 Immunohistochemistry ROS1 screening Immunology Immunofluorescence (IFA) assay platform change ANCA (Anti-Neutrophil Cytoplasmic Ab) Panel for Vasculitis Anti-Mitochondrial Antibody Anti-Native
May 2017 Immunohistochemistry ROS1 screening Immunology Immunofluorescence (IFA) assay platform change ANCA (Anti-Neutrophil Cytoplasmic Ab) Panel for Vasculitis Anti-Mitochondrial Antibody Anti-Native
Procedures for Identifying Pathogens and Diagnosing Infection
 Chapter 17 Procedures for Identifying Pathogens and Diagnosing Infection 1 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 17.1 An Overview of Clinical Microbiology
Chapter 17 Procedures for Identifying Pathogens and Diagnosing Infection 1 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 17.1 An Overview of Clinical Microbiology
RIDASCREEN Masern / Measles Virus IgG, IgM
 RIDASCREEN Masern / Measles Virus IgG, IgM Article no: K5421 (IgG) K5431 (IgM) R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 61 51 81 02-0 / Fax: +49 61 51 81 02-20 1.
RIDASCREEN Masern / Measles Virus IgG, IgM Article no: K5421 (IgG) K5431 (IgM) R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 61 51 81 02-0 / Fax: +49 61 51 81 02-20 1.
Lab. 7: Serological Tests ELISA. 320 MIC Microbial Diagnosis 320 MBIO PRACTICAL. Amal Alghamdi 2018
 Lab. 7: 320 MIC Microbial Diagnosis Serological Tests ELISA. 320 MBIO PRACTICAL Amal Alghamdi 2018 1 Infection and Immunity Serology is the study of immune bodies in human blood. These are products of
Lab. 7: 320 MIC Microbial Diagnosis Serological Tests ELISA. 320 MBIO PRACTICAL Amal Alghamdi 2018 1 Infection and Immunity Serology is the study of immune bodies in human blood. These are products of
Received 31 March 2011/Returned for modification 11 May 2011/Accepted 13 June 2011
 CLINICAL AND VACCINE IMMUNOLOGY, Aug. 2011, p. 1336 1342 Vol. 18, No. 8 1556-6811/11/$12.00 doi:10.1128/cvi.05061-11 Copyright 2011, American Society for Microbiology. All Rights Reserved. Varicella-Zoster
CLINICAL AND VACCINE IMMUNOLOGY, Aug. 2011, p. 1336 1342 Vol. 18, No. 8 1556-6811/11/$12.00 doi:10.1128/cvi.05061-11 Copyright 2011, American Society for Microbiology. All Rights Reserved. Varicella-Zoster
Antibody-dependent Enhancement of Tick-borne Encephalitis Virus Infectivity
 J. gen. Virol. (1985), 66, 1831-1837. Printed in Great Britain 1831 Key words: antibody-mediated enhancement/tbev/flavivirus Antibody-dependent Enhancement of Tick-borne Encephalitis Virus Infectivity
J. gen. Virol. (1985), 66, 1831-1837. Printed in Great Britain 1831 Key words: antibody-mediated enhancement/tbev/flavivirus Antibody-dependent Enhancement of Tick-borne Encephalitis Virus Infectivity
Dengue Vaccine Development. Status, challenges, and WHO priorities. 24/05/2004 WHO Initiative for Vaccine Research
 Dengue Vaccine Development Status, challenges, and WHO priorities Key issues Vaccination appears as the only sustainable strategy for disease prevention, no vaccine available until today; Circulation of
Dengue Vaccine Development Status, challenges, and WHO priorities Key issues Vaccination appears as the only sustainable strategy for disease prevention, no vaccine available until today; Circulation of
PSEUDOMONAS-CF-IgG ELISA KIT
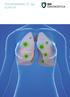 PSEUDOMONAS-CF-IgG ELISA KIT PSEUDOMONAS-CF-IgG ELISA Kit is for the quantitative measurement of the antibody level to P. aeruginosa in human serum samples. Application Chronic P. aeruginosa infection
PSEUDOMONAS-CF-IgG ELISA KIT PSEUDOMONAS-CF-IgG ELISA Kit is for the quantitative measurement of the antibody level to P. aeruginosa in human serum samples. Application Chronic P. aeruginosa infection
SPECIMEN COLLECTION AND TRANSPORT TABLE
 SPECIMEN COLLECTION AND TRANSPORT TABLE City of Milwaukee Health Department Public Health Laboratories 841 N. Broadway, Milwaukee, WI 53202-3653 Extremes in temperature (cold and hot) during transport
SPECIMEN COLLECTION AND TRANSPORT TABLE City of Milwaukee Health Department Public Health Laboratories 841 N. Broadway, Milwaukee, WI 53202-3653 Extremes in temperature (cold and hot) during transport
MUMPS IgM. ReQuest. Reagents Reagents are sufficient for 96 determinations. Allow reagents to reach room temperature before use.
 REF 01-200M 96 Test Set Intended use: Immunoenzymatic capture method for the qualitative determination of IgM-class antibodies to mumps virus in human serum. For in vitro diagnostic use only. Summary and
REF 01-200M 96 Test Set Intended use: Immunoenzymatic capture method for the qualitative determination of IgM-class antibodies to mumps virus in human serum. For in vitro diagnostic use only. Summary and
Evaluation of Chimeric Japanese Encephalitis and Dengue Viruses for Use in Diagnostic Plaque Reduction Neutralization Tests
 CLINICAL AND VACCINE IMMUNOLOGY, July 2009, p. 1052 1059 Vol. 16, No. 7 1556-6811/09/$08.00 0 doi:10.1128/cvi.00095-09 Copyright 2009, American Society for Microbiology. All Rights Reserved. Evaluation
CLINICAL AND VACCINE IMMUNOLOGY, July 2009, p. 1052 1059 Vol. 16, No. 7 1556-6811/09/$08.00 0 doi:10.1128/cvi.00095-09 Copyright 2009, American Society for Microbiology. All Rights Reserved. Evaluation
IgG Antibodies To Toxoplasma. Gondii ELISA Kit
 IgG Antibodies To Toxoplasma Catalog No: IRAPKT1405 Gondii ELISA Kit Lot No: SAMPLE INTENDED USE The Toxoplasma IgG ELISA is intended for use in evaluating a patient s serologic status to Toxoplasma gondii
IgG Antibodies To Toxoplasma Catalog No: IRAPKT1405 Gondii ELISA Kit Lot No: SAMPLE INTENDED USE The Toxoplasma IgG ELISA is intended for use in evaluating a patient s serologic status to Toxoplasma gondii
Ag-Ab reactions Tests for Ag-Ab reactions. EISA SALEHI PhD. Immunology Dept. TUMS
 Ag-Ab reactions Tests for Ag-Ab reactions EISA SALEHI PhD. Immunology Dept. TUMS Importance of Ag-Ab Reactions Understand the mechanisms of defense Abs as tools in: Treatment Diagnosis As biomarkers As
Ag-Ab reactions Tests for Ag-Ab reactions EISA SALEHI PhD. Immunology Dept. TUMS Importance of Ag-Ab Reactions Understand the mechanisms of defense Abs as tools in: Treatment Diagnosis As biomarkers As
Isolation of Flaviviruses from Blood Specimens from Palm Oil and Rubber Plantation Workers in Central Malaysia
 Isolation of Flaviviruses from Blood Specimens from Palm Oil and Rubber Plantation Workers in Central Malaysia Principal Investigators: Donald S. Burket MAJ, MC Ananda Nisalak, M.D. Douglas M. Watt, Ph.D.
Isolation of Flaviviruses from Blood Specimens from Palm Oil and Rubber Plantation Workers in Central Malaysia Principal Investigators: Donald S. Burket MAJ, MC Ananda Nisalak, M.D. Douglas M. Watt, Ph.D.
Antibodies by Enzyme-Linked Immunosorbent Assay
 JOURNAL OF CLINICAL MICROBIOLOGY, Aug. 1983, p. 287-291 0095-1137/83/080287-05$02.00/0 Vol. 18, No. 2 Determination of Herpes Simplex Virus Type-Specific Antibodies by Enzyme-Linked Immunosorbent Assay
JOURNAL OF CLINICAL MICROBIOLOGY, Aug. 1983, p. 287-291 0095-1137/83/080287-05$02.00/0 Vol. 18, No. 2 Determination of Herpes Simplex Virus Type-Specific Antibodies by Enzyme-Linked Immunosorbent Assay
VIROLOGY SERVICE OFFER
 Eidgenössisches Departement für Verteidigung, Bevölkerungsschutz und Sport VBS VIROLOGY SERVICE OFFER CLINICAL SAMPLES The preferred method for verifying the particular is printed in bold. Pathogen Examination
Eidgenössisches Departement für Verteidigung, Bevölkerungsschutz und Sport VBS VIROLOGY SERVICE OFFER CLINICAL SAMPLES The preferred method for verifying the particular is printed in bold. Pathogen Examination
Attribution: University of Michigan Medical School, Department of Microbiology and Immunology
 Attribution: University of Michigan Medical School, Department of Microbiology and Immunology License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution
Attribution: University of Michigan Medical School, Department of Microbiology and Immunology License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution
INFECTION RATES OF RESPIRATORY SYNCYTIAL VIRUS IN PEDIATRIC PATIENTS ATTENDING PHRA MONGKUTKLAO HOSPITAL, BANGKOK
 INFECTION RATES OF RESPIRATORY SYNCYTIAL VIRUS IN PEDIATRIC PATIENTS ATTENDING PHRA MONGKUTKLAO HOSPITAL, BANGKOK SURANG TANTIVANICH, 0RALAK CHITYOTHIN and SAVANAT THARAVANIJ Department of Microbiology
INFECTION RATES OF RESPIRATORY SYNCYTIAL VIRUS IN PEDIATRIC PATIENTS ATTENDING PHRA MONGKUTKLAO HOSPITAL, BANGKOK SURANG TANTIVANICH, 0RALAK CHITYOTHIN and SAVANAT THARAVANIJ Department of Microbiology
Parvovirus B19 IgG, IgM ELISA KIT
 Parvovirus B19 IgG, IgM ELISA KIT Cat. No.:DEIA05717 Pkg.Size:96T Intended use The Parvovirus B19 Tests are enzyme immunoassays (EIA) for the quantitative determination of IgG and semi-quantitative determination
Parvovirus B19 IgG, IgM ELISA KIT Cat. No.:DEIA05717 Pkg.Size:96T Intended use The Parvovirus B19 Tests are enzyme immunoassays (EIA) for the quantitative determination of IgG and semi-quantitative determination
About the LIAISON MDX
 The quality of treatment starts with diagnosis USA Molecular Product MENU Simplexa Molecular Kits LIAISON MDX Molecular Reagents Primer Pairs US. Molecular Menu About the LIAISON MDX The LIAISON MDX is
The quality of treatment starts with diagnosis USA Molecular Product MENU Simplexa Molecular Kits LIAISON MDX Molecular Reagents Primer Pairs US. Molecular Menu About the LIAISON MDX The LIAISON MDX is
Unit title: Infectious Diseases
 Unit title: Infectious Diseases Unit code: M/601/0231 QCF level: 5 Credit value: 15 Aim This unit provides an understanding of infection and disease and uses the dynamic relationship between micro-organism
Unit title: Infectious Diseases Unit code: M/601/0231 QCF level: 5 Credit value: 15 Aim This unit provides an understanding of infection and disease and uses the dynamic relationship between micro-organism
