Supplementary Appendix
|
|
|
- Trevor Jefferson
- 5 years ago
- Views:
Transcription
1 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Zhu J, Zhou L, XingWu F. Tracking neural stem cells in patients with brain trauma. N Engl J Med 2006;355:
2 1. Isolation procedure of NSCs Supplementary Information About 5-20g exposed brain tissues, which were usually contused and mixed with broken hair and blood among the cranial fracture bones, but sometimes just in the hair, were collected directly into cold DMEM-F12-N2 Medium (Gibco) containing 20% FBS, and rinsed five times. Because debris of brain tissue no longer retained its normal architecture, it was difficult to distinguish sub-cortical structure from cortical structure. But in some cases, we obtained large pieces of brain tissue, in which we could distinguish cortex from sub-cortical structure. After resection and removal of pia and arachnoid tissues, specimens were minced into slurries of µl with sterile scalpel blades, and incubated in prewarmed papain/dnase I solution (final concentrations: 0.114U/ml papain; 0.1 U/ml DNase; Sigma) for 30 minutes at 37 C. The resulting crude tissue homogenate was plated into uncoated flask with complete medium. We used complete medium for NSC culture: a mixture of 44 ml of Neurobasal (NB) medium (Gibco), 0.5 ml of glutamine, 0.5 ml of B27 (Gibco), 5 ml of fetal calf serum, 0.04 ml of gentamicin (Cellgro) and supplemented with epidermal growth factor (EGF) (20ng/ml; Sigma) and fibroblast growth factor-2 (FGF2) (20ng/ml; Sigma). After plating, 50% of the medium was replaced, 2 times weekly. Non-adherent cells and debris from the removed supernatant were pelleted by centrifugation and re-introduced into the cultures together with the fresh medium 1. After 7 days in culture, plates were agitated by sharp rapping with a marking pen and 100% of the culture medium and non-adherent material was removed; the removed medium was centrifuged to pellet cell debris and non-adherent cells and to recover complete medium as supernatant. After one week, the procedure was repeated, except that the non-adherent fraction was discarded. In this way, an additional population of cells was recovered from the non-adherent fraction. All the cells were eventually combined to form a single population of cultured cells. We succeeded in isolating neural progenitor cells (NPCs)/NSCs in the 16 cases out of 22 adult patients from the injured brain tissues and propagate them in vitro. Among 16 cases, NPCs/NSCs were isolated from frontal lobes in 6 cases, from parietal lobes in 7 cases, from temporal lobe in 2 cases, from occipital lobe in 1 case. This suggests that debris of brain tissue in open head injury, at least in some cases, can serve as a source of NPCs/NSCs. We failed to culture NPCs from 6 cases, which we attribute to (i) contamination of brain tissue (ii) exposure outside of the brain for a long time (the average exposure time was 8.1 hours in failed cases and 3.3 hours in successful cases), and (iii) inadequate quantity of brain tissue. We found that it is critical not to let the brain tissues dry, and to keep them in the collecting medium containing 20% FBS under low temperature (4-8 C) for transfer to culture medium. 2. Biochemical and physical characteristics of the NSCs Clonal assay: Clonogenic studies were done to confirm that the progenitor cells possessed self-renewal properties. Single spheres were dissociated into single cells after about 30 days of culture and plated in a 96-well plate with 0.5 ml/well of DMEM-F12-N2 with bfgf/pdgf-aa.
3 Seven days later, the cells were switched to serum free medium with bfgf alone (30 ng/ml). Over the next 7 days, we observed the formation of neurospheres spherical masses of cells that expand from single parental progenitors, and a cloning efficiency of %. The neurospheres were typically >150 m in diameter and included 68±18.2 cells/sphere. For secondary cloning, the neurospheres were passaged at the 50- to 100- cell stage, by dissociation to single cells, 2.5% of the secondly seeded cells gave rise to a new culture of neural stem cells, which could reach 3 5 x 10 5 cells after 40 days. This clonogenic potential was observed in samples derived from10 of the 16 individuals studied. Furthermore, each neurosphere can give rise to a large clonal expansion in the numbers of sphere (average number of secondary spheres from each primary sphere = 81.2±5.6). However, over subsequent passages up to passage 12, the numbers of new spheres that formed from dissociation of each single sphere dropped. The proliferation rate of the cells derived from frontal lobes was higher than that of other region. We found that NSCs isolated from sub-cortex proliferated significantly more strongly than those isolated from cortical matter. (P<0.01). Differentiation and Immunostaining: To assay the differentiation potential of the adult human NSCs, individual clonally derived spheres were plated as whole spheres or dissociated spheres in 24-well plates on a substrate of poly-l-ornithine plus laminin (BD Biosciences) in DMEM-F12-N2 medium with 15% fetal calf serum. Media were changed every 3 4 days, and cells were allowed to migrate and differentiate over the course of 2 weeks, at which time the plates were fixed in fresh 4% paraformaldehyde. Fixed cells were incubated for 15 min in a blocking solution containing 0.2% Triton X-100, and 5% normal goat serum before incubation with the primary antibody. Immunohistochemical analysis of the cells was tested by using antibodies directed to specific markers. Nestin and Pax6 was used for undifferentiated cells; beta-tubuliniii, microtubule-associated protein 2 (MAP2) and neurofilament-m were used for neuron; glial fibrillary acidic protein (GFAP) was used for glia; GalC and the O4 sulfatide antigen were used for oligodendrocytes. Under these culture conditions, the spheres consisted of 8.9±2.3% III-tubulin+ neurons, 61.3±5.7% GFAP+ astrocytes and 5.8±1.7% O4+ oligodendrocytes. Strikingly, the percentage of neurons derived from different regions differed significantly. Cultures derived from temporal and occipital lobes gave rise to 8.2±1.2% and 4.3±0.7% neurons, respectively (n=3), whereas 5.0±1.1% of the cells derived from parietal lobes were III-tubulin+ (n=7). A higher proportion of neurons (11.9±1.9%; n=6) was generated from tissue excised from frontal lobes.
4 Fig. 1. A. NSCs isolated from sub-cortex proliferated more strongly than those isolated from cortical matter (P<0.01). B. The percentage of neurons derived from different regions differed. Cultures derived from temporal and occipital lobes had 8.2±1.2% and 4.3±0.7% neurons, respectively (n=3), whereas, 5.0±1.1% of the cells derived from parietal lobes were III-tubulin+ (n=7). A higher proportion of neurons (11.9±1.9%; n=6) was generated in frontal lobes. asterisk: P < 0.01 Electrophysiology Characteristics: We performed electrophysiological experiments to investigate if the adult NSCs give rise to functional neurons in vitro and in vivo. It will be crucial to determine the functional significance of these newly generated cells in the brain. Whole-cell recordings were obtained with an Axonclamp200B amplifier (Axon Instruments). Cells were visualized on an inverted Nikon microscope with mercury arc lamp attachment. A holding potential of -70 mv and voltage steps of 10 mv with 100 ms durations were applied to the recorded cells through the patch electrodes. Resting membrane potentials was ±8.12mV. Action potentials were recorded (Fig.3A). APa was 39.23±5.06m; APd was10.14±1.25ms. A total of 47 NSCs-derived cells were recorded, in 4 cultures derived from 4 patients. Of these, 21 showed voltage-activated sodium ion currents (INa) of >100 na, and 9 had INa > 600 (Fig.3B), compatible with the fast sodium currents of neuronal depolarization. Accordingly, whereas two of seven cells with INa > 800 generated stimulus-evoked action potentials, none did so with INa < 800. In addition, none of morphologically non-neuronal cells showed substantial current-induced sodium currents. For in vivo study, we use a virus vector expressing green fluorescent protein (GFP) to labels NSCs, and that can be visualized in live brain slice. Adult human NSCs labeled with GFP gene were grafted into brain of nude mice. Eight weeks after transplantation, brains of the mice were removed into a chilled solution containing: 110 mm choline Cl -, 2.5 mm KCl, 1.3 mm NaH 2 PO 4, 25 mm NaHCO 3, 0.5 mm CaCl 2, 7 mm MgCl 2, 20 mm dextrose, 1.3 mm Na + ascorbate, 0.6 mm Na + pyruvate, 5.5 mm kynurenic acid. Brain slices were cut into sections of µm, transferred to a chamber containing artificial cerebrospinal fluid (124 mm NaCl, 5 mm KCl, 26 mm NaHCO 3, 0.1 mm CaCl 2, 1.3 mm MgCl 2, 10 mm D-glucose), bubbled with 95% O 2 /5% CO 2 (ph 7.4, 320 mosm), and stored at 30 o C. Using fluorescent optics, slices were scanned for GFP+ cells. Microelectrodes (6±8MQ) were pulled from borosilicate glass and filled with 140 mm potassium gluconate, 11 mm EGTA 10 mm HEPES, 35 mm KOH and 1 mm CaCl 2 (ph 8.2; 300 mosm). Criteria to include cells in the analysis were: (1) visual confirmation of GFP in the pipette tip; (2) resting potential less than -50mV; and (3) leak current less than
5 300 pa. The action potential or continuous K+ currents were recorded by patch clamp in the GFP-positive neurons derived from adult NSCs following transplantation in vivo (Fig.3C). Fig. 2 (A) Action potentials of the neurons derived from NSCs was recorded by whole cell patch clamp. (B) Inward Na+ current in neurons derived from NSCs was recorded in vitro. (C) Continuous K+ currents recorded by patch clamp in a GFP positive neuron derived from NSC in vivo. 3. Gene transfer experiment to distinguish MRI signal generated by neural stem cell migration from MRI signal potentially generated by macrophages It is important to differentiate between true migration of SPIO-labeled NSC to the site of injury and the possibility that the host's macrophages engulfed the foreign cells, acquired the tracking label, and then tracked to the injury site. A retroviral marker gene is expressed only in live cells, and not dead cells or macrophages that may engulf the foreign cells that express the marker gene. We thus used a retroviral construct containing the gene that encodes the green fluorescent protein (GFP) to distinguish between SPIO-labelled NSCs, and the possibility that magnetic signal is generated by macrophages that have engulfed the labeled NSCs. We transduced NSCs with retroviral green fluorescent protein (GFP), and selected the GFP-positive NSCs for SPIO labeling. Then the GFP and SPIO double-labeled NSCs were contra-laterally implanted into a rat brain modelling traumatic brain injury. An identical staining pattern of GFP and Prussian blue (the latter indicating SPIO) indicates that the hypointensity signals in MRI are derived from the SPIO-labeled NSCs, rather than macrophages. The experiment was conducted as follows. First, we established traumatic brain injury in rats. One week later, SPIO and GFP double-labeled NSCs were contra-laterally implanted into the brain. Weekly MR imaging showed that the hypointensity signal migrated from the implantation site to the injured region during the 3 weeks of follow-up (Fig. 3). Histology confirmed that a large number of Prussian blue positive cells had migrated toward the lesion at 21 days after implantation, corresponding to the planes of the MRI investigation (Fig. 4). In parallel, the GFP labeled cells in coronal brain sections were consistent with Prussian blue positive cells (Fig. 5). We therefore conclude that the hypointensity signals did not come from engulfment of the implanted cells by macrophages, but rather, from SPIO-labeled NSCs.
6 Figure 3. Serial in vivo tracking of transhemispheric stem cell migration. One day after transplantation of NSCs, a hypointensity signal can be detected at injection tract (A). Over the following 21 days post-grafting in the same animal, transplanted cells migrated along the corpus callosum to reach the site of damage (B:7 days after transplantation; C: 14 days after transplantation; D: 21 days after transplantation). This indicates that migration of stem cells to the contralateral hemisphere mainly relies on transcallosal migration. Figure 4. Histological corroboration of in vivo observations. At 21 days, Prussian blue staining showed SPIO labeled NSCs migrated from injection tract (B), crossed corpus callosum (C), and infiltrated into the lesion (D), corresponding to the planes of the MRI investigation (A). Figure 5. Fluorescent photographs showed that GFP labeled cells migrated from injection site (A), crossed corpus callosum (B), and infiltrated into the lesion (C), which was consistent with Prussian blue positive cells (B),(C) and (D) in the figure 2. References 1. Zhu J, Zhou L, Zhang L, Zhu W, Pang L, Wang Y. Isolation and cloning of human neural stem cells along with intracerebral grafting and transgene expression in mice. Chinese J. Nervous & Mental Diseases. 2001,
Table of Contents. 2.1 NeuroCult NCFC Assay Kit (Rat) Components Additional Required Reagents Required Equipment...
 i Table of Contents 1.0 Overview of the NeuroCult NCFC Assay 2.0 Materials 2.1 NeuroCult NCFC Assay Kit (Rat) Components... 4 2.2 Additional Required Reagents... 4 2.3 Required Equipment... 4 3.0 Preparation
i Table of Contents 1.0 Overview of the NeuroCult NCFC Assay 2.0 Materials 2.1 NeuroCult NCFC Assay Kit (Rat) Components... 4 2.2 Additional Required Reagents... 4 2.3 Required Equipment... 4 3.0 Preparation
Rat Cortical Stem Cells
 Rat Cortical Stem Cells Catalog Number NSC001 For cortical stem cell expansion and differentiation into neurons, astrocytes, and oligodendrocytes. This package insert must be read in its entirety before
Rat Cortical Stem Cells Catalog Number NSC001 For cortical stem cell expansion and differentiation into neurons, astrocytes, and oligodendrocytes. This package insert must be read in its entirety before
All quality control test results are reported on a lot specific Certificate of Analysis which is available at or upon request.
 PRIME-XV Neural Basal Medium PRIME-XV Neural Basal Medium is a chemically-defined basal medium optimized for the culture and maintenance of neuronal cells when supplemented with PRIME-XV IS21 Supplement
PRIME-XV Neural Basal Medium PRIME-XV Neural Basal Medium is a chemically-defined basal medium optimized for the culture and maintenance of neuronal cells when supplemented with PRIME-XV IS21 Supplement
NEURONAL CELL CULTURE MATRIX FOR BETTER MAINTENANCE AND SURVIVAL OF NEURONAL CELL CULTURES IN TISSUE CULTURE.
 NEURONAL CELL CULTURE MATRIX FOR BETTER MAINTENANCE AND SURVIVAL OF NEURONAL CELL CULTURES IN TISSUE CULTURE. D. R. Aguirre, N. DiMassa, Chrystal Johnson, H. Eran, R. Perez, C.V.R. Sharma, M.V.R. Sharma,
NEURONAL CELL CULTURE MATRIX FOR BETTER MAINTENANCE AND SURVIVAL OF NEURONAL CELL CULTURES IN TISSUE CULTURE. D. R. Aguirre, N. DiMassa, Chrystal Johnson, H. Eran, R. Perez, C.V.R. Sharma, M.V.R. Sharma,
All quality control test results are reported on a lot specific Certificate of Analysis which is available at or upon request.
 PRIME-XV NPC Expansion XSFM PRIME-XV NPC Expansion XSFM is a xeno-and serum-free medium optimized for the cultivation and expansion of neural progenitor cells (NPC) that are able to maintain their ability
PRIME-XV NPC Expansion XSFM PRIME-XV NPC Expansion XSFM is a xeno-and serum-free medium optimized for the cultivation and expansion of neural progenitor cells (NPC) that are able to maintain their ability
Mouse/Rat Neural Stem Cell Functional Identification Kit
 Mouse/Rat Neural Stem Cell Functional Identification Kit Catalog Number SC013 Reagents for the identification of mouse/rat Neural Stem Cells (NSCs) by in vitro functional differentiation. This package
Mouse/Rat Neural Stem Cell Functional Identification Kit Catalog Number SC013 Reagents for the identification of mouse/rat Neural Stem Cells (NSCs) by in vitro functional differentiation. This package
SUPPLEMENTARY NOTE 3. Supplememtary Note 3, Wehr et al., Monitoring Regulated Protein-Protein Interactions Using Split-TEV 1
 SUPPLEMENTARY NOTE 3 Fluorescent Proteolysis-only TEV-Reporters We generated TEV reporter that allow visualizing TEV activity at the membrane and in the cytosol of living cells not relying on the addition
SUPPLEMENTARY NOTE 3 Fluorescent Proteolysis-only TEV-Reporters We generated TEV reporter that allow visualizing TEV activity at the membrane and in the cytosol of living cells not relying on the addition
Xeno-Free Culture and Differentiation of Neural Stem Cells into Neurons
 Xeno-Free Culture and Differentiation of Neural Stem Cells into Neurons Publication Part Number MAN0007497 Revision 1.0 Introduction As the field of neuroscience moves closer to the reality of neural stem
Xeno-Free Culture and Differentiation of Neural Stem Cells into Neurons Publication Part Number MAN0007497 Revision 1.0 Introduction As the field of neuroscience moves closer to the reality of neural stem
Propagation of H7 hesc From: UW (John Stamatoyannopoulos) ENCODE group Date: 12/17/2009 Prepared By: S. Paige/S. Hansen (UW)
 Propagation of H7 hesc From: UW (John Stamatoyannopoulos) ENCODE group Date: 12/17/2009 Prepared By: S. Paige/S. Hansen (UW) Growth and Harvest Modifications Addendum to: Propagation of H7 hesc from UW
Propagation of H7 hesc From: UW (John Stamatoyannopoulos) ENCODE group Date: 12/17/2009 Prepared By: S. Paige/S. Hansen (UW) Growth and Harvest Modifications Addendum to: Propagation of H7 hesc from UW
Components Neural induction basal medium (Cat. # ) 100 ml x 5 Neural induction medium supplement 50x (Cat. # ) 2 ml x 5
 HPSC Neural Induction Medium (PSCNIM) Catalog #5931 Introduction Human embryonic stem cells (hesc) and induced pluripotent stem cells (hipsc) possess the capability of differentiating into all derivatives
HPSC Neural Induction Medium (PSCNIM) Catalog #5931 Introduction Human embryonic stem cells (hesc) and induced pluripotent stem cells (hipsc) possess the capability of differentiating into all derivatives
In vivo recording, forepaw denervation, and isolation of slices: Methods for mapping the forepaw/lower jaw border in anesthetized adult rat primary
 Supplementary Methods In vivo recording, forepaw denervation, and isolation of slices: Methods for mapping the forepaw/lower jaw border in anesthetized adult rat primary somatosensory cortex (S1), forepaw
Supplementary Methods In vivo recording, forepaw denervation, and isolation of slices: Methods for mapping the forepaw/lower jaw border in anesthetized adult rat primary somatosensory cortex (S1), forepaw
RUTGERS. Executive Director NJ Commission on Spinal Cord Research PO Box 360 Trenton, NJ
 RUTGERS W. M. KECK CENTER FOR COLLABORATIVE NEUROSCIENCE 604 Allison Road, D251, Piscataway, NJ 08854-8082 USA Dept. of Cell Biology and Neuroscience (732) 445-6sn, (732) 445-2061, Fax: (732) 445-2063
RUTGERS W. M. KECK CENTER FOR COLLABORATIVE NEUROSCIENCE 604 Allison Road, D251, Piscataway, NJ 08854-8082 USA Dept. of Cell Biology and Neuroscience (732) 445-6sn, (732) 445-2061, Fax: (732) 445-2063
Section of Morphological and Behavioral Neuroscience Protocol for low density primary neuron cultures
 Institute of Pharmacology and Toxicology Section of Morphological and Behavioral Neuroscience Protocol for low density primary neuron cultures B. Lardi-Studler, C. Sidler, J.-M. Fritschy Revised March
Institute of Pharmacology and Toxicology Section of Morphological and Behavioral Neuroscience Protocol for low density primary neuron cultures B. Lardi-Studler, C. Sidler, J.-M. Fritschy Revised March
In Vitro Proliferation and Differentiation of Human Neural Stem and Progenitor Cells Using NeuroCult or NeuroCult -XF
 In Vitro Proliferation and Differentiation of Human Neural Stem and Progenitor Cells Using NeuroCult or NeuroCult -XF i Table of Contents 1.0 Materials and Reagents... 1 1.1 Storage of Basal Media and
In Vitro Proliferation and Differentiation of Human Neural Stem and Progenitor Cells Using NeuroCult or NeuroCult -XF i Table of Contents 1.0 Materials and Reagents... 1 1.1 Storage of Basal Media and
CHO K V 4.3 Cell Line
 B SYS GmbH CHO K V 4.3 Cell Line Specification Sheet B SYS GmbH B SYS GmbH CHO K V 4.3 Cells Page 2 TABLE OF CONTENTS 1. PRODUCT SHIPMENT...3 1.1. Product Format...3 1.2. Mycoplasma Certificate...3 1.3.
B SYS GmbH CHO K V 4.3 Cell Line Specification Sheet B SYS GmbH B SYS GmbH CHO K V 4.3 Cells Page 2 TABLE OF CONTENTS 1. PRODUCT SHIPMENT...3 1.1. Product Format...3 1.2. Mycoplasma Certificate...3 1.3.
The Ultimate Low Cell Binding Surface
 The Ultimate Low Cell Binding Surface Thermo Scientific Nunc HydroCell Surface SINGLE CELL CULTURES Grow cells that are sensitive to activation and differentiation signals arising from cell adhesion CELL
The Ultimate Low Cell Binding Surface Thermo Scientific Nunc HydroCell Surface SINGLE CELL CULTURES Grow cells that are sensitive to activation and differentiation signals arising from cell adhesion CELL
Neural Stem Cells (ipsc from Blood Cells; Male)
 Applied StemCell, Inc. (866) 497-4180 www.appliedstemcell.com Datasheet Neural Stem Cells (ipsc from Blood Cells; Male) Product Information Catalog Number ASE-9234 (Male) Description Applied StemCell's
Applied StemCell, Inc. (866) 497-4180 www.appliedstemcell.com Datasheet Neural Stem Cells (ipsc from Blood Cells; Male) Product Information Catalog Number ASE-9234 (Male) Description Applied StemCell's
CHO-Tet herg Cell Line
 F B SYS GmbH CHO-Tet herg Cell Line Specification Sheet B SYS GmbH B SYS GmbH CHO-Tet herg Cells Page 2 TABLE OF CONTENTS 1 BACKGROUND...3 1.1 Drug-induced QT Prolongation...3 1.2 Regulatory Issues...3
F B SYS GmbH CHO-Tet herg Cell Line Specification Sheet B SYS GmbH B SYS GmbH CHO-Tet herg Cells Page 2 TABLE OF CONTENTS 1 BACKGROUND...3 1.1 Drug-induced QT Prolongation...3 1.2 Regulatory Issues...3
Corning PureCoat rlaminin-521 (Human) for Expansion and Differentiation of Human Neural Stem Cells
 PureCoat rlaminin-521 (Human) for Expansion and Differentiation of Human Neural Stem Cells Application Note Audrey Bergeron 1, Hilary Sherman 1, Pilar Pardo 1, Hannah Gitschier 1, Himabindu Nandivada 2,
PureCoat rlaminin-521 (Human) for Expansion and Differentiation of Human Neural Stem Cells Application Note Audrey Bergeron 1, Hilary Sherman 1, Pilar Pardo 1, Hannah Gitschier 1, Himabindu Nandivada 2,
CHO Na V 1.4 Cell Line
 B SYS GmbH CHO Na V 1.4 Cell Line Specification Sheet B SYS GmbH B SYS GmbH CHO Na V 1.4 Cells Page 2 TABLE OF CONTENTS 1. B'SYS Na V 1.4 CELL LINE...2 1.1 THE VOLTAGE GATED SODIUM CHANNELS NA V 1.4...2
B SYS GmbH CHO Na V 1.4 Cell Line Specification Sheet B SYS GmbH B SYS GmbH CHO Na V 1.4 Cells Page 2 TABLE OF CONTENTS 1. B'SYS Na V 1.4 CELL LINE...2 1.1 THE VOLTAGE GATED SODIUM CHANNELS NA V 1.4...2
Cell Surface-Anchored Fluorescent Aptamer Sensor Enables. Imaging of Chemical Transmitter Dynamics
 Supporting Information for Cell Surface-Anchored Fluorescent Aptamer Sensor Enables Imaging of Chemical Transmitter Dynamics Takeshi Tokunaga, Shigeyuki Namiki, Katsuhiro Yamada, Takahiro Imaishi, Hiroshi
Supporting Information for Cell Surface-Anchored Fluorescent Aptamer Sensor Enables Imaging of Chemical Transmitter Dynamics Takeshi Tokunaga, Shigeyuki Namiki, Katsuhiro Yamada, Takahiro Imaishi, Hiroshi
Alternatives to animal use 2. Ex vivo and in vitro experimental techniques. Állatkísérletek helyettesítése 2: Sejt és szövettenyésztés.
 Reference number: 035/14 UNIVERSITY OF SZEGED, Department of Biochemistry, Faculty of Medicine Animal experiments theory and practice C level Alternatives to animal use 2. Ex vivo and in vitro experimental
Reference number: 035/14 UNIVERSITY OF SZEGED, Department of Biochemistry, Faculty of Medicine Animal experiments theory and practice C level Alternatives to animal use 2. Ex vivo and in vitro experimental
Supplementary Figure 1: Derivation and characterization of RN ips cell lines. (a) RN ips cells maintain expression of pluripotency markers OCT4 and
 Supplementary Figure 1: Derivation and characterization of RN ips cell lines. (a) RN ips cells maintain expression of pluripotency markers OCT4 and SSEA4 after 10 passages in mtesr 1 medium. (b) Schematic
Supplementary Figure 1: Derivation and characterization of RN ips cell lines. (a) RN ips cells maintain expression of pluripotency markers OCT4 and SSEA4 after 10 passages in mtesr 1 medium. (b) Schematic
Dissolve it in water and filter it through 0.22µm. (It should dissolve pretty fast in water, just mix it well. Sometimes I let it sit for 30 min)
 CLEANING COVERSLIPS FOR NEURON PLATING (from J. Wesseling) PLUS SUGGESTIONS FOR SUBTRATES Note: Use coverslips from Fisher and do everything using glass material. The cleaning procedure can be substituted
CLEANING COVERSLIPS FOR NEURON PLATING (from J. Wesseling) PLUS SUGGESTIONS FOR SUBTRATES Note: Use coverslips from Fisher and do everything using glass material. The cleaning procedure can be substituted
Hippocampal Neuron Dissociation Transfection and Culture in Microfluidics Chambers Yang Geng *
 Hippocampal Neuron Dissociation Transfection and Culture in Microfluidics Chambers Yang Geng * Department of Pediatrics and Bioengineering, Stanford University School of Medicine, Stanford, USA *For correspondence:
Hippocampal Neuron Dissociation Transfection and Culture in Microfluidics Chambers Yang Geng * Department of Pediatrics and Bioengineering, Stanford University School of Medicine, Stanford, USA *For correspondence:
Human ESC-derived Neural Progenitor Expansion Starter Panel. Reagents for the expansion of human neural progenitors.
 Human ESC-derived Neural Progenitor Expansion Starter Panel Catalog Number SC016 Reagents for the expansion of human neural progenitors. This package insert must be read in its entirety before using this
Human ESC-derived Neural Progenitor Expansion Starter Panel Catalog Number SC016 Reagents for the expansion of human neural progenitors. This package insert must be read in its entirety before using this
Accumax Cell Dissociation Solution
 http://www.accutase.com/accumax.html 2015/01/30 Accumax Cell Dissociation Solution Accumax is a ready to use non-mammalian, non-bacterial replacement for all applications of trypsin and collagenase in
http://www.accutase.com/accumax.html 2015/01/30 Accumax Cell Dissociation Solution Accumax is a ready to use non-mammalian, non-bacterial replacement for all applications of trypsin and collagenase in
SOP 2 Solid Tumor Processing for Cell Culture and Xenografts
 Texas Cancer Cell Repository Cell Culture and Xenograft Repository http://cancer.ttuhsc.edu www.txccr.org www.cogcell.org SOP 2 Solid Tumor Processing for Cell Culture and Xenografts Receiving the Sample:
Texas Cancer Cell Repository Cell Culture and Xenograft Repository http://cancer.ttuhsc.edu www.txccr.org www.cogcell.org SOP 2 Solid Tumor Processing for Cell Culture and Xenografts Receiving the Sample:
Protocol Reprogramming Human Fibriblasts using the Dox Inducible Reprogramming Polycistronic Lentivirus Set: Human 4F2A LoxP
 STEMGENT Page 1 OVERVIEW The following protocol describes the reprogramming of one well of BJ Human Fibroblasts (BJ cells) into induced pluripotent stem (ips) cells in a 6-well format. Transduction efficiency
STEMGENT Page 1 OVERVIEW The following protocol describes the reprogramming of one well of BJ Human Fibroblasts (BJ cells) into induced pluripotent stem (ips) cells in a 6-well format. Transduction efficiency
Xeno-Free Systems for hesc & hipsc. Facilitating the shift from Stem Cell Research to Clinical Applications
 Xeno-Free Systems for hesc & hipsc Facilitating the shift from Stem Cell Research to Clinical Applications NutriStem Defined, xeno-free (XF), serum-free media (SFM) specially formulated for growth and
Xeno-Free Systems for hesc & hipsc Facilitating the shift from Stem Cell Research to Clinical Applications NutriStem Defined, xeno-free (XF), serum-free media (SFM) specially formulated for growth and
Cortical Neural Induction Kit. Protocol version 1.0
 Cortical Neural Induction Kit Protocol version 1.0 Protocol version 1.0 Table of Contents Product Information 2 Preparation of Reagents 3 Protocol Overview 4 Seeding ipscs 4 Cortical Neural Induction
Cortical Neural Induction Kit Protocol version 1.0 Protocol version 1.0 Table of Contents Product Information 2 Preparation of Reagents 3 Protocol Overview 4 Seeding ipscs 4 Cortical Neural Induction
Therapeutic Cell Replacement. Steven McLoon Department of Neuroscience University of Minnesota
 Therapeutic Cell Replacement Steven McLoon Department of Neuroscience University of Minnesota 1 Neuronal Death Neurons are lost due to four main causes: Trauma Toxin Hypoxia (typically loss of air or blood
Therapeutic Cell Replacement Steven McLoon Department of Neuroscience University of Minnesota 1 Neuronal Death Neurons are lost due to four main causes: Trauma Toxin Hypoxia (typically loss of air or blood
IncuCyte S3 Neuronal Activity Assay
 IncuCyte S3 Neuronal Activity Assay For the kinetic quantification of neuronal activity and functional connectivity This protocol provides an overview of the methodology which uses the lentiviral based
IncuCyte S3 Neuronal Activity Assay For the kinetic quantification of neuronal activity and functional connectivity This protocol provides an overview of the methodology which uses the lentiviral based
IncuCyte S3 Neuronal Activity Assay
 IncuCyte S3 Neuronal Activity Assay For the kinetic quantification of neuronal activity and functional connectivity This protocol provides an overview of the methodology which uses the lentiviral based
IncuCyte S3 Neuronal Activity Assay For the kinetic quantification of neuronal activity and functional connectivity This protocol provides an overview of the methodology which uses the lentiviral based
CHO herg-duo Cell Line
 B SYS GmbH CHO herg-duo Cell Line Specification Sheet B SYS GmbH B SYS GmbH CHO herg DUO Cells Page 2 TABLE OF CONTENTS 1. BACKGROUND...3 1.1. DRUG-INDUCED QT PROLONGATION...3 1.2. REGULATORY ISSUES...3
B SYS GmbH CHO herg-duo Cell Line Specification Sheet B SYS GmbH B SYS GmbH CHO herg DUO Cells Page 2 TABLE OF CONTENTS 1. BACKGROUND...3 1.1. DRUG-INDUCED QT PROLONGATION...3 1.2. REGULATORY ISSUES...3
Corning BioCoat Matrigel Matrix 6-well Plates for Embryonic Stem (ES) Cell Culture. Catalog Number Guidelines for Use
 Corning BioCoat Matrigel Matrix 6-well Plates for Embryonic Stem (ES) Cell Culture Catalog Number 354671 Guidelines for Use Discovery Labware, Inc., Two Oak Park, Bedford, MA 01730, Tel: 1.978.442.2200
Corning BioCoat Matrigel Matrix 6-well Plates for Embryonic Stem (ES) Cell Culture Catalog Number 354671 Guidelines for Use Discovery Labware, Inc., Two Oak Park, Bedford, MA 01730, Tel: 1.978.442.2200
Culture of Human ipsc-derived Neural Stem Cells in a 96-Well Plate Format
 Protocol supplement version 1.0 Culture of Human ipsc-derived Neural Stem Cells in a 96-Well Plate Format Protocol Supplement This protocol supplement is to be used in addition to the Neural Stem Cell
Protocol supplement version 1.0 Culture of Human ipsc-derived Neural Stem Cells in a 96-Well Plate Format Protocol Supplement This protocol supplement is to be used in addition to the Neural Stem Cell
MimEX TM GI Human Descending Colon Stem Cells
 MimEX TM GI Human Descending Colon Stem Cells Adult Gastrointestinal Stem Cells for Use With MimEX GI Reagents Catalog Number: MIM006 Size: 1 vial, ~ 100,000 cells PRODUCT DESCRIPTION The MimEX GI Tissue
MimEX TM GI Human Descending Colon Stem Cells Adult Gastrointestinal Stem Cells for Use With MimEX GI Reagents Catalog Number: MIM006 Size: 1 vial, ~ 100,000 cells PRODUCT DESCRIPTION The MimEX GI Tissue
Skull optical clearing window for in vivo imaging of the mouse cortex at synaptic resolution
 SUPPLEMENTARY INFORMATION for Skull optical clearing window for in vivo imaging of the mouse cortex at synaptic resolution Yanjie Zhao 1,2, Tingting Yu 1,2, Chao Zhang 1,2, Zhao Li 1,2, Qingming Luo 1,2,
SUPPLEMENTARY INFORMATION for Skull optical clearing window for in vivo imaging of the mouse cortex at synaptic resolution Yanjie Zhao 1,2, Tingting Yu 1,2, Chao Zhang 1,2, Zhao Li 1,2, Qingming Luo 1,2,
Lab Module 7: Cell Adhesion
 Lab Module 7: Cell Adhesion Tissues are made of cells and materials secreted by cells that occupy the spaces between the individual cells. This material outside of cells is called the Extracellular Matrix
Lab Module 7: Cell Adhesion Tissues are made of cells and materials secreted by cells that occupy the spaces between the individual cells. This material outside of cells is called the Extracellular Matrix
Protocol Using the Reprogramming Ecotropic Retrovirus Set: Mouse OSKM to Reprogram MEFs into ips Cells
 STEMGENT Page 1 OVERVIEW The following protocol describes the reprogramming of one well of mouse embryonic fibroblast (MEF) cells into induced pluripotent stem (ips) cells in a 6-well format. Transduction
STEMGENT Page 1 OVERVIEW The following protocol describes the reprogramming of one well of mouse embryonic fibroblast (MEF) cells into induced pluripotent stem (ips) cells in a 6-well format. Transduction
B-27 Plus Neuronal Culture System
 USER GUIDE B-27 Plus Neuronal Culture System Catalog Number A3653401 Pub. No. MAN0017319 Rev. 1.0 WARNING! Read the Safety Data Sheets (SDSs) and follow the handling instructions. Wear appropriate protective
USER GUIDE B-27 Plus Neuronal Culture System Catalog Number A3653401 Pub. No. MAN0017319 Rev. 1.0 WARNING! Read the Safety Data Sheets (SDSs) and follow the handling instructions. Wear appropriate protective
Supplementary Figure 1.
 Supplementary Figure 1. VEH NI Supplementary Figure 1. Cannula tip placement in mice infused with vehicle (VEH) or anisomycin (NI). Cannula tip placement in mice infused with VEH (open circles) or NI (solid
Supplementary Figure 1. VEH NI Supplementary Figure 1. Cannula tip placement in mice infused with vehicle (VEH) or anisomycin (NI). Cannula tip placement in mice infused with VEH (open circles) or NI (solid
Protocol Using a Dox-Inducible Polycistronic m4f2a Lentivirus to Reprogram MEFs into ips Cells
 STEMGENT Page 1 OVERVIEW The following protocol describes the transduction and reprogramming of one well of Oct4-GFP mouse embryonic fibroblasts (MEF) using the Dox Inducible Reprogramming Polycistronic
STEMGENT Page 1 OVERVIEW The following protocol describes the transduction and reprogramming of one well of Oct4-GFP mouse embryonic fibroblasts (MEF) using the Dox Inducible Reprogramming Polycistronic
Development of Novel Advanced Cell Culture Surfaces that Provide Better Cell Growth and Attachment for Cell-Based Assays
 Development of Novel Advanced Cell Culture Surfaces that Provide Better Cell Growth and Attachment for Cell-Based Assays Elizabeth J. Abraham, Ph.D. BD Biosciences Outline Overview of surfaces and culture
Development of Novel Advanced Cell Culture Surfaces that Provide Better Cell Growth and Attachment for Cell-Based Assays Elizabeth J. Abraham, Ph.D. BD Biosciences Outline Overview of surfaces and culture
Amaxa Basic Neuron SCN Nucleofector Kit
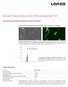 Amaxa Basic Neuron SCN Nucleofector Kit For Primary Mouse Hippocampal Neurons (Small-Cell-Number) Embryonic mouse hippocampal neurons transfected using the Nucleofector Technology A B Approximately 100.000
Amaxa Basic Neuron SCN Nucleofector Kit For Primary Mouse Hippocampal Neurons (Small-Cell-Number) Embryonic mouse hippocampal neurons transfected using the Nucleofector Technology A B Approximately 100.000
Neural Stem Cell Characterization Kit
 Neural Stem Cell Characterization Kit Catalog No. SCR019 FOR RESEARCH USE ONLY Not for use in diagnostic procedures USA & Canada Phone: +1(800) 437-7500 Fax: +1 (951) 676-9209 Europe +44 (0) 23 8026 2233
Neural Stem Cell Characterization Kit Catalog No. SCR019 FOR RESEARCH USE ONLY Not for use in diagnostic procedures USA & Canada Phone: +1(800) 437-7500 Fax: +1 (951) 676-9209 Europe +44 (0) 23 8026 2233
The Effects of Isotretinoin on C2C12 Stem Cells. Colm Parrish Pi,sburgh Central Catholic HS Grade 11
 The Effects of Isotretinoin on C2C12 Stem Cells Colm Parrish Pi,sburgh Central Catholic HS Grade 11 Tissue Engineering The development and manipulation of artificial implants, laboratory-grown tissues,
The Effects of Isotretinoin on C2C12 Stem Cells Colm Parrish Pi,sburgh Central Catholic HS Grade 11 Tissue Engineering The development and manipulation of artificial implants, laboratory-grown tissues,
Engraftment of human induced pluripotent stem cell-derived hepatocytes in. immunocompetent mice via 3D co-aggregation and encapsulation
 Engraftment of human induced pluripotent stem cell-derived hepatocytes in immunocompetent mice via 3D co-aggregation and encapsulation Wei Song 1, Yen-Chun Lu 1, Angela S. Frankel 2, Duo An 1, Robert E.
Engraftment of human induced pluripotent stem cell-derived hepatocytes in immunocompetent mice via 3D co-aggregation and encapsulation Wei Song 1, Yen-Chun Lu 1, Angela S. Frankel 2, Duo An 1, Robert E.
Supporting Protocols
 Supporting Protocols This protocol may be used prior to immunostaining cells, organoids, or patient-derived xenografts cultured in TissueSpec ECM Hydrogels. Introduction Cells and organoids may form complex
Supporting Protocols This protocol may be used prior to immunostaining cells, organoids, or patient-derived xenografts cultured in TissueSpec ECM Hydrogels. Introduction Cells and organoids may form complex
To isolate single GNS 144 cell clones, cells were plated at a density of 1cell/well
 Supplemental Information: Supplemental Methods: Cell culture To isolate single GNS 144 cell clones, cells were plated at a density of 1cell/well in 96 well Primaria plates in GNS media and incubated at
Supplemental Information: Supplemental Methods: Cell culture To isolate single GNS 144 cell clones, cells were plated at a density of 1cell/well in 96 well Primaria plates in GNS media and incubated at
Neural Precursor Cell-Based Screening & Bioassay Kit
 Neural Precursor Cell-Based Screening & Bioassay Kit Catalog Number SC014 Reagents for screening toxins or other bioactive agents for their effects on neural precursor cell proliferation and neuronal differentiation.
Neural Precursor Cell-Based Screening & Bioassay Kit Catalog Number SC014 Reagents for screening toxins or other bioactive agents for their effects on neural precursor cell proliferation and neuronal differentiation.
A20 Ubiquitin Ligase-Mediated Polyubiquitination of RIP1 Inhibits Caspase-8 Cleavage and TRAIL-Induced Apoptosis in Glioblastoma
 A20 Ubiquitin Ligase-Mediated Polyubiquitination of RIP1 Inhibits Caspase-8 Cleavage and TRAIL-Induced Apoptosis in Glioblastoma Anita C. Bellail, Jeffrey J. Olson, Xiaolu Yang, Zhijian J. Chen, and Chunhai
A20 Ubiquitin Ligase-Mediated Polyubiquitination of RIP1 Inhibits Caspase-8 Cleavage and TRAIL-Induced Apoptosis in Glioblastoma Anita C. Bellail, Jeffrey J. Olson, Xiaolu Yang, Zhijian J. Chen, and Chunhai
T ECHNICAL MANUAL. Culture of Human Mesenchymal Stem Cells Using MesenCult -XF Medium
 T ECHNICAL MANUAL Culture of Human Mesenchymal Stem Cells Using MesenCult -XF Medium i Table of Contents 1.0 Materials... 1 1.1 MesenCult -XF Medium and Required Products... 1 1.2 Additional Required
T ECHNICAL MANUAL Culture of Human Mesenchymal Stem Cells Using MesenCult -XF Medium i Table of Contents 1.0 Materials... 1 1.1 MesenCult -XF Medium and Required Products... 1 1.2 Additional Required
PRIMARY NEURONAL CULTURE
 PRIMARY NEURONAL CULTURE Products for Your Research Scientists Helping Scientists WWW.STEMCELL.COM TABLE OF CONTENTS 3 Primary Neuronal Culture NeuroCult SM1 and BrainPhys Neuronal Medium 4 Increased Neuronal
PRIMARY NEURONAL CULTURE Products for Your Research Scientists Helping Scientists WWW.STEMCELL.COM TABLE OF CONTENTS 3 Primary Neuronal Culture NeuroCult SM1 and BrainPhys Neuronal Medium 4 Increased Neuronal
Rat Fetal Neural Stem Cells
 Rat Fetal Neural Stem Cells Catalog nos. N7744-100, N7744-200 Rev. date: 7 April 2009 Manual part no. A11229 MAN0001642 User Manual ii Table of Contents Contents and Storage... iv Additional Products...
Rat Fetal Neural Stem Cells Catalog nos. N7744-100, N7744-200 Rev. date: 7 April 2009 Manual part no. A11229 MAN0001642 User Manual ii Table of Contents Contents and Storage... iv Additional Products...
VDL602.2 RAPID ASSAY FOR DETERMINING ADENOVIRAL VECTOR TITER
 1. Purpose 1.1. The purpose of this protocol is to determine the number of infectious adenoviral particles. 1.2. The starting material is purified adenoviral vectors. 1.3. This protocol is based upon the
1. Purpose 1.1. The purpose of this protocol is to determine the number of infectious adenoviral particles. 1.2. The starting material is purified adenoviral vectors. 1.3. This protocol is based upon the
Metzger Lab Protocol Book EF May 2003
 I. Preparation for cell isolation: A. Make sure the following items are autoclaved: 1. Glassware* (1) 100 ml beaker (3) wide mouthed 1 liter bottles (1) 100 mm glass petri dish (1) 60 mm sigma coated petri
I. Preparation for cell isolation: A. Make sure the following items are autoclaved: 1. Glassware* (1) 100 ml beaker (3) wide mouthed 1 liter bottles (1) 100 mm glass petri dish (1) 60 mm sigma coated petri
InVERT moulding for scalable control of tissue microarchitecture
 InVERT moulding for scalable control of tissue microarchitecture Stevens KR, Ungrin MD, Schwartz RE, Ng SY, Carvalho B, Christine KS, Chaturvedi R, Li CY, Zandstra PW, Chen CS, Bhatia SN Supplementary
InVERT moulding for scalable control of tissue microarchitecture Stevens KR, Ungrin MD, Schwartz RE, Ng SY, Carvalho B, Christine KS, Chaturvedi R, Li CY, Zandstra PW, Chen CS, Bhatia SN Supplementary
MicroRNAs Modulate Hematopoietic Lineage Differentiation
 Chen et al., page 1 MicroRNAs Modulate Hematopoietic Lineage Differentiation Chang-Zheng Chen, Ling Li, Harvey F. Lodish, David. Bartel Supplemental Online Material Methods Cell isolation Murine bone marrow
Chen et al., page 1 MicroRNAs Modulate Hematopoietic Lineage Differentiation Chang-Zheng Chen, Ling Li, Harvey F. Lodish, David. Bartel Supplemental Online Material Methods Cell isolation Murine bone marrow
Which hydrogel preparation for immunostaining protocol should I use?
 Protocol: Preparation of TissueSpec hydrogels for immunostaining This protocol may be used prior to immunostaining cells, organoids, or patient-derived xenografts cultured in TissueSpec matrix hydrogels.
Protocol: Preparation of TissueSpec hydrogels for immunostaining This protocol may be used prior to immunostaining cells, organoids, or patient-derived xenografts cultured in TissueSpec matrix hydrogels.
Protocol Reprogramming MEFs using the Dox Inducible Reprogramming Lentivirus Set: Mouse OKSM
 STEMGENT Page 1 OVERVIEW The following protocol describes the reprogramming of one well of mouse embryonic fibroblasts (MEFs) into induced pluripotent stem (ips) cells in a 6-well format. Transduction
STEMGENT Page 1 OVERVIEW The following protocol describes the reprogramming of one well of mouse embryonic fibroblasts (MEFs) into induced pluripotent stem (ips) cells in a 6-well format. Transduction
STANDARD OPERATING PROCEDURE
 STANDARD OPERATING PROCEDURE Culture of the Rat Macrophage Cell Line, NR8383 SOP number: WP1/001 Protocol prepared by: Ewelina Hoffman Developed under NC3R project: NC/C013203/1 INTRODUCTION Procedure
STANDARD OPERATING PROCEDURE Culture of the Rat Macrophage Cell Line, NR8383 SOP number: WP1/001 Protocol prepared by: Ewelina Hoffman Developed under NC3R project: NC/C013203/1 INTRODUCTION Procedure
Week 1: Protein isolation and quantification
 Week 1: Protein isolation and quantification Objective The objective of this lab exercise is to obtain protein samples from fruit fly larvae, BCS and FBS, all of which are then quantitated in the preparation
Week 1: Protein isolation and quantification Objective The objective of this lab exercise is to obtain protein samples from fruit fly larvae, BCS and FBS, all of which are then quantitated in the preparation
DOPA Maturation Media
 Applied StemCell, Inc. (866) 97-180 www.appliedstemcell.com Datasheet Media Product Information Catalog Number Description ASE-933DM Media is serum free media produced using Applied StemCell s proprietary
Applied StemCell, Inc. (866) 97-180 www.appliedstemcell.com Datasheet Media Product Information Catalog Number Description ASE-933DM Media is serum free media produced using Applied StemCell s proprietary
Cell Culture in Cardiovascular Research
 Cell Culture in Cardiovascular Research Tamás Csont, Csaba Csonka, Anikó Görbe Department of Biochemistry, Faculty of Medicine, University of Szeged Cardiovascular Research Group Nov 25, 2011 Introduction
Cell Culture in Cardiovascular Research Tamás Csont, Csaba Csonka, Anikó Görbe Department of Biochemistry, Faculty of Medicine, University of Szeged Cardiovascular Research Group Nov 25, 2011 Introduction
hpsc Maintenance Media
 hpsc Maintenance Media Brigitte Arduini, version 2, 2013-Jun-12 Initially, it was found that pluripotency of human pluripotent stem cells (hpscs) can be maintained when plated in co-culture with mouse
hpsc Maintenance Media Brigitte Arduini, version 2, 2013-Jun-12 Initially, it was found that pluripotency of human pluripotent stem cells (hpscs) can be maintained when plated in co-culture with mouse
Mesenchymal Progenitor Cells (MPCs) Involvement In Ectopic Fat Formation In Muscular Dystrophy
 Mesenchymal Progenitor Cells (MPCs) Involvement In Ectopic Fat Formation In Muscular Dystrophy Jihee Sohn, MS 1, Ying Tang 2, Bing Wang, MD,PhD 3, Aiping Lu 4, Johnny Huard, Ph.D 5. 1 university of pittsburgh,
Mesenchymal Progenitor Cells (MPCs) Involvement In Ectopic Fat Formation In Muscular Dystrophy Jihee Sohn, MS 1, Ying Tang 2, Bing Wang, MD,PhD 3, Aiping Lu 4, Johnny Huard, Ph.D 5. 1 university of pittsburgh,
Lineage-specific Reporter Knock-in ipscs: MAP2-Nanoluc-Halotag ipsc (Heterozygous)
 Lineage-specific Reporter Knock-in ipscs: MAP2-Nanoluc-Halotag ipsc (Heterozygous) Applied StemCell, Inc. (866) 497-4180 www.appliedstemcell.com Datasheet Product Information Catalog Number Description
Lineage-specific Reporter Knock-in ipscs: MAP2-Nanoluc-Halotag ipsc (Heterozygous) Applied StemCell, Inc. (866) 497-4180 www.appliedstemcell.com Datasheet Product Information Catalog Number Description
Lineage-specific Reporter Knock-in ipscs: AAVS-DCX-GFP ipsc (Heterozygous)
 Lineage-specific Reporter Knock-in ipscs: AAVS-DCX-GFP ipsc (Heterozygous) Applied StemCell, Inc. (866) 497-4180 www.appliedstemcell.com Datasheet Product Information Catalog Number Description Amount
Lineage-specific Reporter Knock-in ipscs: AAVS-DCX-GFP ipsc (Heterozygous) Applied StemCell, Inc. (866) 497-4180 www.appliedstemcell.com Datasheet Product Information Catalog Number Description Amount
In vitro Human Umbilical Vein Endothelial Cells (HUVEC) Tube-formation Assay. Josephine MY Ko and Maria Li Lung *
 In vitro Human Umbilical Vein Endothelial Cells (HUVEC) Tube-formation Assay Josephine MY Ko and Maria Li Lung * Clinical Oncology Department, The Univerisity of Hong Kong, Hong Kong, Hong Kong SAR *For
In vitro Human Umbilical Vein Endothelial Cells (HUVEC) Tube-formation Assay Josephine MY Ko and Maria Li Lung * Clinical Oncology Department, The Univerisity of Hong Kong, Hong Kong, Hong Kong SAR *For
ipsc Reprogramming from Human Peripheral Blood Using Sendai Virus Mediated Gene Transfer
 ipsc Reprogramming from Human Peripheral Blood Using Sendai Virus Mediated Gene Transfer Wenli Yang 1, Jason A. Mills 2, Spencer Sullivan 3, Ying Liu 1, Deborah L. French 2 and Paul Gadue 2, 1 Institute
ipsc Reprogramming from Human Peripheral Blood Using Sendai Virus Mediated Gene Transfer Wenli Yang 1, Jason A. Mills 2, Spencer Sullivan 3, Ying Liu 1, Deborah L. French 2 and Paul Gadue 2, 1 Institute
STEMdiff Astrocyte Differentiation Kit and STEMdiff Astrocyte Maturation Kit
 Kit and Kit Catalog #08540 Catalog #08550 1 Kit 1 Kit Product Description Kit (Catalog #08540) is used to rapidly and efficiently generate astrocyte precursors from neural progenitor cells (NPCs) derived
Kit and Kit Catalog #08540 Catalog #08550 1 Kit 1 Kit Product Description Kit (Catalog #08540) is used to rapidly and efficiently generate astrocyte precursors from neural progenitor cells (NPCs) derived
Safe Harbor Knock-in ipscs: AAVS1-CAG-GFP Chr19 ipsc (Heterozygous)
 Safe Harbor Knock-in ipscs: AAVS1-CAG-GFP Chr19 ipsc (Heterozygous) Applied StemCell, Inc. (866) 497-4180 www.appliedstemcell.com Datasheet Product Information Catalog Number Description Amount Parental
Safe Harbor Knock-in ipscs: AAVS1-CAG-GFP Chr19 ipsc (Heterozygous) Applied StemCell, Inc. (866) 497-4180 www.appliedstemcell.com Datasheet Product Information Catalog Number Description Amount Parental
Peri.4U. Human ipsc-derived peripheral neurons
 Peri.4U Human ipsc-derived peripheral neurons Manual Version 1.0 / July 2018 Contents 1. General Information 2 2. Safety Information 2 3. Material 3 3.1 Cells and media provided by Ncardia 3 3.2 Storage
Peri.4U Human ipsc-derived peripheral neurons Manual Version 1.0 / July 2018 Contents 1. General Information 2 2. Safety Information 2 3. Material 3 3.1 Cells and media provided by Ncardia 3 3.2 Storage
Protocols for Neural Progenitor Cell Expansion and Dopaminergic Neuron Differentiation
 Protocols for Neural Progenitor Cell Expansion and Dopaminergic Neuron Differentiation In vitro neurological research presents many challenges due to the difficulty in establishing high-yield neuronal
Protocols for Neural Progenitor Cell Expansion and Dopaminergic Neuron Differentiation In vitro neurological research presents many challenges due to the difficulty in establishing high-yield neuronal
Neurosphere Nestin SOX 2 Merge Nestin SOX 2 Merge
 Neurosphere Nestin SOX 2 Merge Nestin SOX 2 Merge A B Fig. S1. Generation and characterization of anscs in vitro. anscs were isolated and expanded from the SVZ region of adult naïve C57BL/6 mice and cultured
Neurosphere Nestin SOX 2 Merge Nestin SOX 2 Merge A B Fig. S1. Generation and characterization of anscs in vitro. anscs were isolated and expanded from the SVZ region of adult naïve C57BL/6 mice and cultured
Therapeutic Cell Replacement. Steven McLoon Department of Neuroscience University of Minnesota
 Therapeutic Cell Replacement Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Coffee Hour Wednesday (Dec 13) Thursday (Dec 14) Friday (Dec 15) 9:00-10:00am Surdyk s Café in
Therapeutic Cell Replacement Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Coffee Hour Wednesday (Dec 13) Thursday (Dec 14) Friday (Dec 15) 9:00-10:00am Surdyk s Café in
Generation and Culture of Neural Progenitor Cells using the STEMdiff Neural System
 Generation and Culture of Neural Progenitor Cells using the STEMdiff Neural System i Table of Contents 1.0 Introduction... 1 2.0 Materials, Reagents and Equipment... 2 2.1 Materials Required for Neural
Generation and Culture of Neural Progenitor Cells using the STEMdiff Neural System i Table of Contents 1.0 Introduction... 1 2.0 Materials, Reagents and Equipment... 2 2.1 Materials Required for Neural
3T3-L1 Differentiation Protocol
 3T3-L1 Differentiation Protocol Written by Eisuke Kato on 2013/10/09 MATERIALS Dulbecco's Modified Eagles Medium (D-MEM) (High Glucose) with L-Glutamine and Phenol Red High glucose (Wako Chem 044-29765,
3T3-L1 Differentiation Protocol Written by Eisuke Kato on 2013/10/09 MATERIALS Dulbecco's Modified Eagles Medium (D-MEM) (High Glucose) with L-Glutamine and Phenol Red High glucose (Wako Chem 044-29765,
StemXVivo. Mouse Oligodendrocyte Differentiation Kit. Catalog Number SC004
 StemXVivo Mouse Oligodendrocyte Differentiation Kit Catalog Number SC004 For the differentiation of mouse pluripotent stem cells to oligodendrocytes. This package insert must be read in its entirety before
StemXVivo Mouse Oligodendrocyte Differentiation Kit Catalog Number SC004 For the differentiation of mouse pluripotent stem cells to oligodendrocytes. This package insert must be read in its entirety before
Supplementary Methods. Li J.-Y. et al. Lewy bodies in grafted neurons in Parkinson s patients suggest host to. graft disease propagation
 1 Supplementary Methods Li J.-Y. et al. Lewy bodies in grafted neurons in Parkinson s patients suggest host to graft disease propagation Neural transplantation and clinical assessment Detailed information
1 Supplementary Methods Li J.-Y. et al. Lewy bodies in grafted neurons in Parkinson s patients suggest host to graft disease propagation Neural transplantation and clinical assessment Detailed information
Cor.4U on QPatch Cells provided by AxioGenesis
 Application Report: Cor.4U on QPatch This report shows how cardiomyocytes from Axiogenesis can be used on the QPatch. Action potentials can be induced in a controlled and reproducible manner using a mix
Application Report: Cor.4U on QPatch This report shows how cardiomyocytes from Axiogenesis can be used on the QPatch. Action potentials can be induced in a controlled and reproducible manner using a mix
Cor.At. Cardiomyocytes derived from Mouse Embryonic Stem Cells. Protocol. Cor.At Cardiomyocytes Transfection
 Use our Discoveries to Help Advance Yours Cor.At Cardiomyocytes derived from Mouse Embryonic Stem Cells Protocol Cor.At Cardiomyocytes Transfection Using the Nucleofector Technology (Amaxa/Lonza Cologne)
Use our Discoveries to Help Advance Yours Cor.At Cardiomyocytes derived from Mouse Embryonic Stem Cells Protocol Cor.At Cardiomyocytes Transfection Using the Nucleofector Technology (Amaxa/Lonza Cologne)
Kit Components (Included) Cat # # of vials Product Name Quantity Storage Human Renal Cortical Epithelial Cells (HRCEpiC)
 3D Renal Tubule Formation Kit 3D-RTF Cat # 3D-4110 Product Description The human kidney is frequently exposed to drugs and toxic compounds, which can result in nephrotoxicity. A drug s uptake and clearance
3D Renal Tubule Formation Kit 3D-RTF Cat # 3D-4110 Product Description The human kidney is frequently exposed to drugs and toxic compounds, which can result in nephrotoxicity. A drug s uptake and clearance
Neural induction - Dual SMAD inhibition
 Neural induction - Dual SMAD inhibition Mark J. Tomishima, SKI Stem Cell Research Facility http://stemcells.mskcc.org, tomishim@mskcc.org This protocol is based on Chambers et al. 2009. [Plating pluripotent
Neural induction - Dual SMAD inhibition Mark J. Tomishima, SKI Stem Cell Research Facility http://stemcells.mskcc.org, tomishim@mskcc.org This protocol is based on Chambers et al. 2009. [Plating pluripotent
Protocol for the Co-culture of ipsc-derived Microglia with ipsc-derived Cerebral Cortical Neural Culture
 Protocol for the Co-culture of ipsc-derived Microglia with ipsc-derived Cerebral Cortical Neural Culture Required reagents Product code ax0031a + b ax0041 ax0047 ax68168 (5 mg) ax0660 ax0016, ax0111 (AD
Protocol for the Co-culture of ipsc-derived Microglia with ipsc-derived Cerebral Cortical Neural Culture Required reagents Product code ax0031a + b ax0041 ax0047 ax68168 (5 mg) ax0660 ax0016, ax0111 (AD
Genome Edited ipscs APOE -/- Knockout
 Applied StemCell, Inc. (866) 497-4180 www.appliedstemcell.com Datasheet Genome Edited ipscs APOE -/- Knockout Product Information Catalog Number Description Amount Parental Cell Line Gene Knockout Generated
Applied StemCell, Inc. (866) 497-4180 www.appliedstemcell.com Datasheet Genome Edited ipscs APOE -/- Knockout Product Information Catalog Number Description Amount Parental Cell Line Gene Knockout Generated
Defense Technical Information Center Compilation Part Notice
 UNCLASSIFIED Defense Technical Information Center Compilation Part Notice ADP019695 TITLE: Fabricating Neural and Cardiomyogenic Stem Cell Structures by a Novel Rapid Prototyping--the Inkjet Printing Method
UNCLASSIFIED Defense Technical Information Center Compilation Part Notice ADP019695 TITLE: Fabricating Neural and Cardiomyogenic Stem Cell Structures by a Novel Rapid Prototyping--the Inkjet Printing Method
Sabrina Jedlicka. Interactions of Neurons and Materials
 Sabrina Jedlicka Interactions of Neurons and Materials Neurological Disorders Over 600 known neurological disorders ú Diseases of the CNS and PNS Epilepsy Alzheimer s Parkinson s Etc. ú Diseases that attack
Sabrina Jedlicka Interactions of Neurons and Materials Neurological Disorders Over 600 known neurological disorders ú Diseases of the CNS and PNS Epilepsy Alzheimer s Parkinson s Etc. ú Diseases that attack
Amaxa Cell Line Nucleofector Kit L
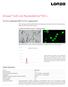 Amaxa Cell Line Nucleofector Kit L For 3T3-L1 (adipocytes) [ATCC CL-173, cryopreserved] Mouse embryonal fibroblast, differentiated into adipocytes; Fibroblast-like cells before differentiation; adipocyte-like
Amaxa Cell Line Nucleofector Kit L For 3T3-L1 (adipocytes) [ATCC CL-173, cryopreserved] Mouse embryonal fibroblast, differentiated into adipocytes; Fibroblast-like cells before differentiation; adipocyte-like
EZ-iPSC Generation kit - EPISOMAL. ASK-3013 (Episomal vector based ipsc reprogramming kit)
 Applied StemCell, Inc. (866) 497-4180 www.appliedstemcell.com Datasheet EZ-iPSC Generation kit - EPISOMAL Product Information Catalog Number Description ASK-3013 (Episomal vector based ipsc reprogramming
Applied StemCell, Inc. (866) 497-4180 www.appliedstemcell.com Datasheet EZ-iPSC Generation kit - EPISOMAL Product Information Catalog Number Description ASK-3013 (Episomal vector based ipsc reprogramming
Supporting Information
 Supporting Information Wang et al. 10.1073/pnas.1008382107 SI Methods Tissue Fixation. Adult rats, mice, and Oprd1 exon 1-deleted mice (Jackson Lab) were anesthetized and perfused with 20 ml of warm saline,
Supporting Information Wang et al. 10.1073/pnas.1008382107 SI Methods Tissue Fixation. Adult rats, mice, and Oprd1 exon 1-deleted mice (Jackson Lab) were anesthetized and perfused with 20 ml of warm saline,
ReproRNA -OKSGM is a non-integrating, self-replicating RNA-based reprogramming vector for generating induced pluripotent stem (ips)
 Kit for generating ips cells using ReproRNA -OKSGM, a non-integrating, self-replicating RNA reprogramming vector Product Description ReproRNA -OKSGM is a non-integrating, self-replicating RNA-based reprogramming
Kit for generating ips cells using ReproRNA -OKSGM, a non-integrating, self-replicating RNA reprogramming vector Product Description ReproRNA -OKSGM is a non-integrating, self-replicating RNA-based reprogramming
ETS-Embryo Medium. In Vitro Culture Media. Version 1.0
 ETS-Embryo Medium In Vitro Culture Media Version 1.0 3D Culture Media Product Catalogue No Storage ETS-Embryo Medium, 25 ml (5 x 5 ml) M13-25 -20 C ETS-Embryo Medium, 100 ml M13-100 -20 C Additional Reagents
ETS-Embryo Medium In Vitro Culture Media Version 1.0 3D Culture Media Product Catalogue No Storage ETS-Embryo Medium, 25 ml (5 x 5 ml) M13-25 -20 C ETS-Embryo Medium, 100 ml M13-100 -20 C Additional Reagents
HUMAN IPSC CULTURE PROTOCOLS
 HUMAN IPSC CULTURE PROTOCOLS FOR TRAINING COURSE IXCELLS BIOTECHNOLOGIES USA, LLC 10340 CAMINO SANTA FE, SUITE C, SAN DIEGO, CA 92121 TABLE OF CONTENTS CHAPTER 1 BEFORE STARTING 2-7 SECTION 1.1 COATING
HUMAN IPSC CULTURE PROTOCOLS FOR TRAINING COURSE IXCELLS BIOTECHNOLOGIES USA, LLC 10340 CAMINO SANTA FE, SUITE C, SAN DIEGO, CA 92121 TABLE OF CONTENTS CHAPTER 1 BEFORE STARTING 2-7 SECTION 1.1 COATING
Fitness Measurements of Evolved Escherichia coli Migla Miskinyte and Isabel Gordo *
 Fitness Measurements of Evolved Escherichia coli Migla Miskinyte and Isabel Gordo * Evolutionary Biology Group, Instituto Gulbenkian de Ciência, Oeiras, Portugal *For correspondence: igordo@igc.gulbenkian.pt
Fitness Measurements of Evolved Escherichia coli Migla Miskinyte and Isabel Gordo * Evolutionary Biology Group, Instituto Gulbenkian de Ciência, Oeiras, Portugal *For correspondence: igordo@igc.gulbenkian.pt
VitroGel ready-to-use tunable hydrogel for 3D cell culture and beyond
 VitroGel ready-to-use tunable hydrogel for 3D cell culture and beyond Cat No: TWG001 VItroGel 3D (10 ml high concentration) Cat No: TWG002 VitroGel 3D-RGD (10 ml high concentration) Handbook Rev. June
VitroGel ready-to-use tunable hydrogel for 3D cell culture and beyond Cat No: TWG001 VItroGel 3D (10 ml high concentration) Cat No: TWG002 VitroGel 3D-RGD (10 ml high concentration) Handbook Rev. June
3D Mammary Colony-Forming Cell Assay Giusy Tornillo 1* and Sara Cabodi 2
 3D Mammary Colony-Forming Cell Assay Giusy Tornillo 1* and Sara Cabodi 2 1 Cardiff School of Biosciences, European Cancer Stem Cell Research Institute, Cardiff University, Cardiff, UK; 2 Department of
3D Mammary Colony-Forming Cell Assay Giusy Tornillo 1* and Sara Cabodi 2 1 Cardiff School of Biosciences, European Cancer Stem Cell Research Institute, Cardiff University, Cardiff, UK; 2 Department of
Supplementary Figure 1 A green: cytokeratin 8
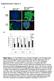 Supplementary Figure 1 A green: cytokeratin 8 green: α-sma red: α-sma blue: DAPI blue: DAPI Panc-1 Panc-1 Panc-1+hPSC Panc-1+hPSC monoculture coculture B Suppl. Figure 1: A, Immunofluorescence staining
Supplementary Figure 1 A green: cytokeratin 8 green: α-sma red: α-sma blue: DAPI blue: DAPI Panc-1 Panc-1 Panc-1+hPSC Panc-1+hPSC monoculture coculture B Suppl. Figure 1: A, Immunofluorescence staining
