QM-1 Supplement QUALITY MANAGEMENT SYSTEMS MANUAL SUPPLEMENT. Revision 7. Quality Director / Management Representative n. B.
|
|
|
- Bennett Jenkins
- 6 years ago
- Views:
Transcription
1 QUALITY MANAGEMENT SYSTEMS MANUAL SUPPLEMENT Revision 7 APPROVED BY: TITLE DATE B. Olevson Quality Director / Management Representative n
2 DOCUMENT REVISION HISTORY Paragraph Revisions: or Rev: Date: Section: All Initial Release 0 12/14/09 Table 1, Section 7, Product Realization. Removed IP 7.1.2, Production Job Planning requirements 1 7/10/10 covered in IP and IP Replaced IP with CQP /13/10 Table 1 Replaced IP with CQP /18/10 Table 1 Replaced CQP with IP Replaced CQP with IP /7/13 Table 1 Replaced IP with IP /18/ Deleted reference to Quality Manager 6 10/3/15 All Complete rewrite to match the new numbering of clauses of the Quality Manual and revisions of ISO9001:2015, AS9100D & ISO 13485: /7/17 Page: 2 of 11 This document contains proprietary information and is not to be distributed to or copied for a third party without the express written permission of Veridiam, Inc. This document is to be
3 1. PURPOSE 1.1 The purpose of this document is to provide supplemental information to QM-1 Quality Management System Manual. 1.2 This Supplement depicts by section, the second tier procedures that address how the Quality Manual requirements are met and the correlation between different standards applicable to Veridiam. 2 SCOPE 2.2 This supplemental procedure is applicable for El Cajon 2.3 This document requires only the approval of the Quality Management Representative. 2.4 In the event that a second tier procedure changes, this document is revised to reflect those changes. 2.5 Newly developed procedures may also be added. 2.6 Training of applicable personnel is required. 3 REFERENCES 3.1 Table 1 lists, by section, QM-1 Quality Management Systems Manual, a correlation between the different standards, procedures and documents applicable to each section Page: 3 of 11 This document contains proprietary information and is not to be distributed to or copied for a third party without the express written permission of Veridiam, Inc. This document is to be
4 TABLE Context of the Organization 4.1 Understanding the Organization and Its Context 4.2 Understanding the needs and expectations of interested parties 4.3 Determining the scope of quality management system 4.4 Quality Management System and Its processes General Requirements Quality Manual 4.1 General Requirements General Quality Manual 5.0 Leadership 5.1 Management commitment 5.1 Management Commitment IP Context of the Organization QM-1 Quality Management Systems Manual QM-1 (p 5.1) Customer focus 5.2 Customer focus IP Customer-Related process Administration 5.2 Quality Policy and communication 5.3 Organizational Roles, Responsibilities and Authorities Responsibility and Authority Management Representative 5.3 Quality Policy QM-1 (p 5.2) 5.5 Responsibility, Authority and communication QM-1 (5.3) QM-1 (p 5.3) 6.0 Planning 6.1 Actions to address Risks and Opportunities QMS Planning Preventive Action QM-1 (p 6.1) QS1227, COTO log and Risk assessment PROD0092, Contract Review PURCH0004, Receiving Inspection/Purchase Req (RIPR) M0001 Appointment of Management Representative
5 6.2 Quality Objectives and Planning to Achieve them 6.3 Quality Management System Planning of Changes Quality Objectives OP Monitoring and Measurement of KPI s QMS Planning QM-1 (p 6.2, 6.3) 7.0 Support QS1230 KPI & PEAR Form Provision of Resources 6.1 Provision of resources QM-1 (p 7.1.1) Assignment of Personnel 6.2 Human Resources QM-1 (p 7.1.2) IP Personnel Competence Infrastructure 6.3 Infrastructure IP Maintenance Procedures QS1030, Corrective - Breakdown - Process Improvement Maintenance Review Work Environment 6.4 Work Environment QM-1 (p 7.1.4) Monitoring and Measuring IP Control & Calibration of of Resources Measurement and Test Equipment 7.6 Control of monitoring and measuring equipment 7.2 Competence 6.2 Human Resources IP Training and Qualification Awareness 6.2 Human Resources Records 7.3 IP Personnel Competence QS0084, Gage Evaluation Form QS1190, Calibration Extension Request Form QS1195, Calibration Certificate Review Checklist QS1207, Calibration Certificate Review Heat Treat Furnace WA-297, Guide for SAT and TUS report review TRAINING0001 Training Record HR0020, Training waiver form QS0385, Training waiver form Page: 5 of 11
6 7.4 Communication Internal Communication 7.5 Documented Information Documentation requirements General General Creating and updating Medical device file Control of documented Control of information Documents Control of records QM-1 (p 7.4) IP Document Control IP Quality Record Control IP Software QA Controls OP Creating a DHR for ISI 8.0 Operation 8.1 Operation Planning and Control 7.1 Planning of product realization IP Context of the Organization IP Product Development Operation Risk 7.1 Planning of product management realization Configuration Management IP Product Development IP Configuration Management QS1027, QMS Training Matrix QS Production Department Orientation Checklist QS1202, New Hire Orientation Checklist QS0055, Document Release Authorization (DRA) form QS1109, PPI Review and Comment Form QS1106, Document Approval Matrix QS1210, Document Review Record QS1227, COTO log and Risk assessment QS1177 Engineering Change Request (ECR) Form QS1178 Engineering Change Order (ECO) Form QS1179 Engineering Change Notification (ECN) Form QS1180 Configuration Definition Form QS1181 Standard Cost Definition Form Page: 6 of 11
7 8.1.3 Product safety IP Purchase Order Prevention of Counterfeit Administration Parts QS1182 Effectivity Assessment and Disposition Form PU0001 Standard Purchase Order Quality Clauses PU0002 ASL Approved Supplier List QS1003 Purchase Requisition (PR) 8.2 Requirements for product and services 7.2 Customer related processes Customer communication Communication Determination of Determine requirements of requirements related to products and services product Review requirements of Review of products and services requirements related to Changes to requirements of products and services 8.4 Control of externally provided process, products and services product Review of requirements related to product General Purchasing process IP Customer-Related process Administration 7.4 Purchasing IP Purchase Order Administration Customer satisfaction survey PROD0092, Contract Review PU0001 Standard Purchase Order Quality Clauses PU0002 ASL Approved Supplier List QS1003 Purchase Requisition (PR) QS1117 Quality Clause Flow Down Guidelines QS1234 Traveling Requisitions Page: 7 of 11
8 8.4.2 Type and extent of control Purchasing process Verification of purchased products Information for external providers 8.5 Production and Service Provision Purchasing information Verification of purchased products 7.5 Production and service provision IP Verification of Purchased Product IP Supplier Selection & Evaluation IP Traveler Requirements (Green Card) QS1003 Purchase Requisition (PR) NCM0035 Non Conformance Report QS0018 Approved Supplier List (ASL) Action Form QS0060 Supplier Corrective Action Report (SCAR) QS0207 Supplier Evaluation PU0002 Approved Supplier List (ASL) PROD TRAVELER CHANGE NOTICE Control of production and service provision Identification and Traceability Control of production and service provision Validation of processes for production and service provision Identification Traceability IP Process Validation IP Identification & Traceability of Products PROD0051 Accept Status Green Tag PROD0052 Hold Status Yellow TAG PROD0053 Reject Status Red Tag PROD0054 Conditional Release Blue Tag Page: 8 of 11
9 8.5.3 Property belonging to customers or external providers Customer property Preservation Preservation of product Control of changes Control of design and development changes 8.6 Release of Product and services 8.7 Control of Nonconforming Outputs Verification of purchased products Monitoring and measurement of product 8.3 Control of nonconforming product IP Handling Customer Property IP Preservation of Product IP Configuration Management IP Verification of Purchased Product IP Monitoring and Measurement of Product IP Product Certification IP Control of Nonconforming Product Customer/Supplier Specific QS1177 Engineering Change Request (ECR) Form QS1178 Engineering Change Order (ECO) Form QS1179 Engineering Change Notification (ECN) Form QS1180 Configuration Definition Form QS1181 Standard Cost Definition Form QS1182 Effectivity Assessment and Disposition Form Customer Specific (MQCP, Fixed Process, etc) Customer specific NCM0035 Non Conformance Report 9.0 Performance Evaluation General 8.1 General Monitoring and measurement of processes IP Analysis of Data OP Monitoring and Measurement of KPI s QS1230 KPI & PEAR Form Page: 9 of 11
10 9.1.2 Customer Satisfaction Feedback IP Customer-Related process Administration Customer satisfaction survey PROD0092, Contract Review Analysis and evaluation 8.4 Analysis of data IP Analysis of Data 9.2 Internal Audit Internal Audit IP Internal Audit Program QS0259 Audit Plan and Summary Report QS0208 On-Site Audit Checklist QS1196 QMS Checklist QS1211 Internal Audit Agenda QS1226 Auditor Skill Matrix 9.3 Management Review 5.6 Management review IP Management Review QS1228, Management Review General General Management Review Review input Inputs Management Review Outputs Review output 10.0 Improvement 10.1 General General IP Context of the Organization IP Preventive Action Presentation QS1229, Management Review Meeting minutes QS1227, COTO log and Risk assessment 10.2 Nonconformity and Corrective Action 8.3 Control of nonconforming products Corrective Action IP Control of Nonconforming Product IP Corrective Action NCM0035 Non Conformance Report QS0060, Supplier Corrective Action Report (SCAR) Problem Improvement Request (PIR) Form (Rolls Royce SABRE requirement 8D Page: 10 of 11
11 root cause analysis tool) QS1192, 8D Report QS1193, CAR Status Tracker 10.3 Continual Improvement Preventive Action QM-1 (p 10.3) IP Preventive Action Page: 11 of 11
Correlation matrices between ISO 9001:2008 and ISO 9001:2015
 Correlation matrices between ISO 9001:2008 and ISO 9001:2015 ISO 9001:2015 ISO 9001:2008 1 Scope 1 Scope 1.1 General 4 Context of the organization 4 Quality management system 4.1 Understanding the organization
Correlation matrices between ISO 9001:2008 and ISO 9001:2015 ISO 9001:2015 ISO 9001:2008 1 Scope 1 Scope 1.1 General 4 Context of the organization 4 Quality management system 4.1 Understanding the organization
9110 Correlation matrices
 9110 Correlation matrices 9110:2016 to 9110:2012 9110:2012 to 9110:2016 This document provides correlation matrices from 9110:2016 to 9110:2012 and 9110:2012 to 9110:2016. This document can be used to
9110 Correlation matrices 9110:2016 to 9110:2012 9110:2012 to 9110:2016 This document provides correlation matrices from 9110:2016 to 9110:2012 and 9110:2012 to 9110:2016. This document can be used to
ISO 13485: :2015 CLIENT TRANSITION CHECKLIST
 - 9001:2015 CLIENT TRANSITION CHECKLIST Audit Conclusions: All requirements have been addressed. The organization is recommended for ISO 13485:2016 certification. Recommendation for registration is dependent
- 9001:2015 CLIENT TRANSITION CHECKLIST Audit Conclusions: All requirements have been addressed. The organization is recommended for ISO 13485:2016 certification. Recommendation for registration is dependent
Clause Map IATF 16949:2016 to ISO/TS 16949:2009
 Table of Contents Table of Contents Foreword Foreword + Foreword Automotive QMS Standard + History + Goal + 0.5 Goal of this Technical Specification + Remarks for Certification + Remarks for Certification
Table of Contents Table of Contents Foreword Foreword + Foreword Automotive QMS Standard + History + Goal + 0.5 Goal of this Technical Specification + Remarks for Certification + Remarks for Certification
Correlation Matrix & Change Summary
 The correlation matrix compares the new requirements of ISO 9001:2015 to the requirements of ISO 9001:2008, and provides a summary of the changes. Correlation Matrix & Change Summary Introduction Correlation
The correlation matrix compares the new requirements of ISO 9001:2015 to the requirements of ISO 9001:2008, and provides a summary of the changes. Correlation Matrix & Change Summary Introduction Correlation
4. Quality management system 4. Context of the organization. 5 Management responsibility 5 Leadership
 AS 9100:2009 (Rev.C) AS 9100:2016 (Rev.D) 4. Quality management system 4. Context of the organization 4.1 General requirements 4.4 Quality management system and its processes products and 4.2 Documentation
AS 9100:2009 (Rev.C) AS 9100:2016 (Rev.D) 4. Quality management system 4. Context of the organization 4.1 General requirements 4.4 Quality management system and its processes products and 4.2 Documentation
Sections of the Standard. Evidence / Comments. (Y) / Nonconforming (NC)
 Date: Sections of the Standard 4 Context of the Organization 4.1 Understanding the organization and its context 4.2 Understanding the needs and expectations of interested parties 4.3 Determining the scope
Date: Sections of the Standard 4 Context of the Organization 4.1 Understanding the organization and its context 4.2 Understanding the needs and expectations of interested parties 4.3 Determining the scope
Supplier Quality System Survey
 Supplier Division Address Date: Quality Contact: Reports to: Title: Title: Phone Fax e-mail Plant size (ft 2 ): Union: Contract expires: Total Employment: Direct Labor: Shifts: QA employees: This report
Supplier Division Address Date: Quality Contact: Reports to: Title: Title: Phone Fax e-mail Plant size (ft 2 ): Union: Contract expires: Total Employment: Direct Labor: Shifts: QA employees: This report
AESOP 15604; ISSUE 2; STATUS PENDING APPROVAL; AUTHORITY CARL BLAZIK This document is the property of NSF ISR. Page 1 of 9
 This document is the property of NSF ISR. Page 1 of 9 ISO 9001 Quality Management Systems registration provides a set of uniform requirements for a quality management system. A number of quality management
This document is the property of NSF ISR. Page 1 of 9 ISO 9001 Quality Management Systems registration provides a set of uniform requirements for a quality management system. A number of quality management
ISO 9001 PMA and 14 CFR 21 QUALITY POLICIES HANDBOOK
 ISO 9001 PMA and 14 CFR 21 QUALITY POLICIES HANDBOOK Origination Date: XXXX Document Identifier: Date: Document Revision: QMS-00 QMS Quality Policies Handbook Latest Revision Date Orig Abstract: This quality
ISO 9001 PMA and 14 CFR 21 QUALITY POLICIES HANDBOOK Origination Date: XXXX Document Identifier: Date: Document Revision: QMS-00 QMS Quality Policies Handbook Latest Revision Date Orig Abstract: This quality
AS9100 PMA and 14 CFR 21 QUALITY HANDBOOK
 AS9100 PMA and 14 CFR 21 QUALITY HANDBOOK Origination Date: XXXX Document Identifier: Date: Document Status: Document Link: Latest Revision Date Draft, Redline, Released, Obsolete Location on Server (if
AS9100 PMA and 14 CFR 21 QUALITY HANDBOOK Origination Date: XXXX Document Identifier: Date: Document Status: Document Link: Latest Revision Date Draft, Redline, Released, Obsolete Location on Server (if
INTEGRATING ISO 9000 METHODOLOGIES WITH PROJECT QUALITY MANAGEMENT
 INTEGRATING ISO 9000 METHODOLOGIES WITH PROJECT QUALITY MANAGEMENT M a r ch 2015 OBJECTIVE ISO and Project Quality Management Process Are they different or the same? ISO 9000 QMS FAMILY ISO 9000:2005 Vocabulary
INTEGRATING ISO 9000 METHODOLOGIES WITH PROJECT QUALITY MANAGEMENT M a r ch 2015 OBJECTIVE ISO and Project Quality Management Process Are they different or the same? ISO 9000 QMS FAMILY ISO 9000:2005 Vocabulary
Business Management System: Requirements 00-BMS-002
 Business Management System: Requirements 00-BMS-002 Preface This document defines the applicability of International s requirements to the Business Management System (BMS) processes implemented at GM Nameplate
Business Management System: Requirements 00-BMS-002 Preface This document defines the applicability of International s requirements to the Business Management System (BMS) processes implemented at GM Nameplate
Vendor Qualification Survey
 1200 West 96 th St Minneapolis, MN 55431 Ph: 952-888-7900 Fax: 952-888-2719 Vendor Qualification Survey Vendor Information Company Name: Date: Address: City: Phone Number: email address: Product or Service
1200 West 96 th St Minneapolis, MN 55431 Ph: 952-888-7900 Fax: 952-888-2719 Vendor Qualification Survey Vendor Information Company Name: Date: Address: City: Phone Number: email address: Product or Service
ISO/TC 176/SC 2 Document N1224, July 2014
 ISO/TC 176/SC 2 Document N1224, July 2014 Correlation matrices between ISO 9001:2008 and ISO/DIS 9001 This document gives correlation matrices from ISO 9001:2008 to the current Draft International Standard
ISO/TC 176/SC 2 Document N1224, July 2014 Correlation matrices between ISO 9001:2008 and ISO/DIS 9001 This document gives correlation matrices from ISO 9001:2008 to the current Draft International Standard
Comparison ISO/TS (1999) to QS 9000, 3 rd edition (1998)
 1 SCOPE QS 9000: new: Introduction, applicability In addition to the applicability for supplier sites for production and services and their subcontractors for: - parts or materials, or - services like
1 SCOPE QS 9000: new: Introduction, applicability In addition to the applicability for supplier sites for production and services and their subcontractors for: - parts or materials, or - services like
ISO 9001 QUALITY MANUAL
 ISO 9001 QUALITY MANUAL Origination Date: 10/01/14 Document Identifier: AIF quality control manual Date: 10/01/14 Project: Customer review Document Status: Released Document Link: www.aeroindfast.com Abstract:
ISO 9001 QUALITY MANUAL Origination Date: 10/01/14 Document Identifier: AIF quality control manual Date: 10/01/14 Project: Customer review Document Status: Released Document Link: www.aeroindfast.com Abstract:
AERO GEAR, INC. SUPPLIER QUALITY MANUAL
 AERO GEAR, INC. SUPPLIER QUALITY MANUAL REVISION HISTORY REVISION EFFECTIVE DATE SUMMARY OF CHANGES A 30-JAN-1998 Original Issue B 17-OCT-2002 Added note re: subcontractors may not subcontract to another
AERO GEAR, INC. SUPPLIER QUALITY MANUAL REVISION HISTORY REVISION EFFECTIVE DATE SUMMARY OF CHANGES A 30-JAN-1998 Original Issue B 17-OCT-2002 Added note re: subcontractors may not subcontract to another
Quality Management System Manual
 This Page 1 of 47 of the Quality Management System Manual If issued as a controlled copy, the serial number of this copy is Quality Management System Manual Certified to AS9100 Revision C Printed copies
This Page 1 of 47 of the Quality Management System Manual If issued as a controlled copy, the serial number of this copy is Quality Management System Manual Certified to AS9100 Revision C Printed copies
QUALITY MANAGEMENT SYSTEM POLICIES AND PROCEDURES
 QUALITY MANAGEMENT SYSTEM POLICIES AND PROCEDURES Origination Date: XXXX Document Identifier: Date: Document Revision: QMS-00 QMS Policies and Procedures Latest Revision Date Abstract: This handbook documents
QUALITY MANAGEMENT SYSTEM POLICIES AND PROCEDURES Origination Date: XXXX Document Identifier: Date: Document Revision: QMS-00 QMS Policies and Procedures Latest Revision Date Abstract: This handbook documents
QM-1 QUALITY MANAGEMENT SYSTEMS MANUAL. Revision 10
 QM-1 QUALITY MANAGEMENT SYSTEMS MANUAL Revision 10 Page: 1 of 54 TABLE OF CONTENTS 1. DOCUMENT REVISION HISTORY... 5 2. INTRODUCTION... 6 2.1 Purpose... 6 2.2 Scope... 7 Figure 1: Sequence and Interaction
QM-1 QUALITY MANAGEMENT SYSTEMS MANUAL Revision 10 Page: 1 of 54 TABLE OF CONTENTS 1. DOCUMENT REVISION HISTORY... 5 2. INTRODUCTION... 6 2.1 Purpose... 6 2.2 Scope... 7 Figure 1: Sequence and Interaction
ISO 9001:2015 QUALITY MANAGEMENT SYSTEM POLICIES AND PROCEDURES
 ISO 9001:2015 QUALITY MANAGEMENT SYSTEM POLICIES AND PROCEDURES Origination Date: XXXX Document Identifier: Date: Document Revision: QMS-00 Policies and Procedures Latest Revision Date Abstract: This handbook
ISO 9001:2015 QUALITY MANAGEMENT SYSTEM POLICIES AND PROCEDURES Origination Date: XXXX Document Identifier: Date: Document Revision: QMS-00 Policies and Procedures Latest Revision Date Abstract: This handbook
QUALITY MANUAL QM0492 AS9100 SUPPLEMENT II
 QUALITY MANUAL QM0492 AS9100 SUPPLEMENT II REV C Foreword This document defines the quality management system requirements to be applied in facilities registered to the aerospace standard AS9100. This
QUALITY MANUAL QM0492 AS9100 SUPPLEMENT II REV C Foreword This document defines the quality management system requirements to be applied in facilities registered to the aerospace standard AS9100. This
QMS Team: MR and all HODs (Internal Auditors) MR March 10. Quality policy Define quality policy The Steering committee Objectives and targets
 QMS Roles, Responsibility and Authority Process Clause Activities Records Required Responsibility Authority Deadline Clause 4: Process Development 4.1 Develop processes and sequence, operation controls
QMS Roles, Responsibility and Authority Process Clause Activities Records Required Responsibility Authority Deadline Clause 4: Process Development 4.1 Develop processes and sequence, operation controls
QUALITY POLICIES HANDBOOK
 QUALITY POLICIES HANDBOOK Origination Date: (mo/yr) Document Identifier: Date: Document Status: Document Link: Latest Revision Date Draft, Redline, Released, Obsolete Location on Server (if used) Abstract:
QUALITY POLICIES HANDBOOK Origination Date: (mo/yr) Document Identifier: Date: Document Status: Document Link: Latest Revision Date Draft, Redline, Released, Obsolete Location on Server (if used) Abstract:
QUALITY MANUAL. Origination Date: XXXX. Latest Revision Date. Revision Orig
 QUALITY MANUAL Origination Date: XXXX Document Identifier: Date: Document Status: Latest Revision Date Revision Orig Abstract: This document describes the tailored quality management system for the build
QUALITY MANUAL Origination Date: XXXX Document Identifier: Date: Document Status: Latest Revision Date Revision Orig Abstract: This document describes the tailored quality management system for the build
QUALITY MANUAL ECO# REVISION DATE MGR QA A 2/25/2008 R.Clement J.Haislip B 6/17/2008 T.Finneran J.Haislip
 UHV SPUTTERING INC Page 1 of 18 ECO REVISION HISTORY ECO# REVISION DATE MGR QA 1001 A 2/25/2008 R.Clement J.Haislip 1017 B 6/17/2008 T.Finneran J.Haislip 1071 C 1/13/2011 R.Clement J.Haislip 1078 D 5/15/2013
UHV SPUTTERING INC Page 1 of 18 ECO REVISION HISTORY ECO# REVISION DATE MGR QA 1001 A 2/25/2008 R.Clement J.Haislip 1017 B 6/17/2008 T.Finneran J.Haislip 1071 C 1/13/2011 R.Clement J.Haislip 1078 D 5/15/2013
QUALITY MANUAL QM0492 AS9100 SUPPLEMENT II
 QUALITY MANUAL QM0492 AS9100 SUPPLEMENT II REV E Foreword This document defines the quality management system requirements to be applied in facilities registered to the aerospace standard AS9100. This
QUALITY MANUAL QM0492 AS9100 SUPPLEMENT II REV E Foreword This document defines the quality management system requirements to be applied in facilities registered to the aerospace standard AS9100. This
Correlation Matrix EN 9100:2016 EN 9100:2009
 Correlation Matrix EN 9100:2016 EN 9100:2009 EN 9100:2016 EN 9100:2009 4 Context of the organization 4 Quality management system 4.2 Understanding the organization and its context Understanding the needs
Correlation Matrix EN 9100:2016 EN 9100:2009 EN 9100:2016 EN 9100:2009 4 Context of the organization 4 Quality management system 4.2 Understanding the organization and its context Understanding the needs
IAQG 9101:2014 (Rev. E)
 IAQG 9101:2014 (Rev. E) Changes Overview Conformity plus Performance Equals Effectiveness Prepared by the IAQG 9101 team 2014-04-08 Presentation Objectives Part One: Provide Overview of 9101:2014 (Rev.
IAQG 9101:2014 (Rev. E) Changes Overview Conformity plus Performance Equals Effectiveness Prepared by the IAQG 9101 team 2014-04-08 Presentation Objectives Part One: Provide Overview of 9101:2014 (Rev.
Quality Systems Manual
 Quality Systems Manual ISO9001:2008 Prepared By: Date: 06/28/2013 President Approved by: Date: 06/28/2013 Quality Assurance Manager Introduction Cal-Tron Corp has developed and implemented a Quality Management
Quality Systems Manual ISO9001:2008 Prepared By: Date: 06/28/2013 President Approved by: Date: 06/28/2013 Quality Assurance Manager Introduction Cal-Tron Corp has developed and implemented a Quality Management
0. 0 TABLE OF CONTENTS
 QUALITY MANUAL Conforming to ISO 9001:2008 0. 0 TABLE OF CONTENTS Section Description ISO 9001 Clause Page 0 TABLE OF CONTENTS n/a 2 1 PIMA VALVE, INC. DESCRIPTION n/a 3 2 QUALITY MANUAL DESCRIPTION 4.2.2
QUALITY MANUAL Conforming to ISO 9001:2008 0. 0 TABLE OF CONTENTS Section Description ISO 9001 Clause Page 0 TABLE OF CONTENTS n/a 2 1 PIMA VALVE, INC. DESCRIPTION n/a 3 2 QUALITY MANUAL DESCRIPTION 4.2.2
ISO 9001:2008 Quality Management System QMS Manual
 2501 Kutztown Road Reading, PA, 19605 Tel. 610-929-3330 Fax 610-921-6861 ISO 9001:2008 Quality Management System QMS Manual The information contained in this document is Fidelity Technologies Corporation
2501 Kutztown Road Reading, PA, 19605 Tel. 610-929-3330 Fax 610-921-6861 ISO 9001:2008 Quality Management System QMS Manual The information contained in this document is Fidelity Technologies Corporation
Quality Manual QM -07 Approval: D. Wheeler. AARD Spring & Stamping Quality Manual. Quality Manual. Page 1 of 24
 Quality Manual Page 1 of 24 ISO 9001:2015 Standard to Quality Manual Section Matrix ISO 9001:2015 Quality Manual Section 1 Scope 1 Scope 1.1 General 4 Context of the organization 4 Quality management system
Quality Manual Page 1 of 24 ISO 9001:2015 Standard to Quality Manual Section Matrix ISO 9001:2015 Quality Manual Section 1 Scope 1 Scope 1.1 General 4 Context of the organization 4 Quality management system
4. RELATED DOCUMENTS AND FORMS 4.1. AS ISO Cambridge Valley Machining, Inc. QMS procedures, including, but not limited to:
 Approved By: Purchasing Manager Candice Lane (signature on file) Date: 5/26/17 Approved By: Process Owner James D. Moore (signature on file) Date: 5/26/17 1. PURPOSE 1.1. To define Cambridge Valley Machining,
Approved By: Purchasing Manager Candice Lane (signature on file) Date: 5/26/17 Approved By: Process Owner James D. Moore (signature on file) Date: 5/26/17 1. PURPOSE 1.1. To define Cambridge Valley Machining,
CAPITAL AVIONICS, INC. Quality Manual
 CAPITAL AVIONICS, INC. Issued 31 July 2018 Conforms to ISO 9001:2015 2018 ; all rights reserved. This document may contain proprietary information and may only be released to third parties with approval
CAPITAL AVIONICS, INC. Issued 31 July 2018 Conforms to ISO 9001:2015 2018 ; all rights reserved. This document may contain proprietary information and may only be released to third parties with approval
QUALITY MANAGEMENT SYSTEM POLICIES AND PROCEDURES
 Your Company Name QUALITY MANAGEMENT SYSTEM POLICIES AND PROCEDURES Origination Date: XXXX Document Identifier: Date: Document Revision: QMS-00 QMS Policies and Procedures Latest Revision Date Abstract:
Your Company Name QUALITY MANAGEMENT SYSTEM POLICIES AND PROCEDURES Origination Date: XXXX Document Identifier: Date: Document Revision: QMS-00 QMS Policies and Procedures Latest Revision Date Abstract:
Quality System Manual - Section 00
 Quality System Manual - Section 00 INDEX AND REVISION STATUS Issued by: Quality Assurance Eff. Date: 00/00/00 Rev.: A Pg. 1 of 4 Organization of this manual is the same as the sectional organization of
Quality System Manual - Section 00 INDEX AND REVISION STATUS Issued by: Quality Assurance Eff. Date: 00/00/00 Rev.: A Pg. 1 of 4 Organization of this manual is the same as the sectional organization of
Reliance Aerospace Solutions
 Reliance Aerospace Solutions Quality Manual The information contained in this document is the property of Reliance Aerospace Solutions, a division of Reliance Steel & Aluminum Company This manual is a
Reliance Aerospace Solutions Quality Manual The information contained in this document is the property of Reliance Aerospace Solutions, a division of Reliance Steel & Aluminum Company This manual is a
Fitting ISO 9001:2000 into a 20 Element Quality System
 Fitting ISO 9001:2000 into a 20 Element Quality System ASQ Washington DC Section 0509 Rockville Maryland Presented by Norman P. Moreau, P.E., CSQE, CQA President Theseus Professional Services 410-857-0023
Fitting ISO 9001:2000 into a 20 Element Quality System ASQ Washington DC Section 0509 Rockville Maryland Presented by Norman P. Moreau, P.E., CSQE, CQA President Theseus Professional Services 410-857-0023
ADVANCED INTERNAL AUDIT WORKSHOP
 ADVANCED INTERNAL AUDIT WORKSHOP By Paul J. Kunder RABQSA QMS Lead Auditor/Aerospace Industry Experienced Auditor RABQSA Approved Aerospace Auditor Transition Training (AATT) Trainer Vice Chair U.S. Z1A
ADVANCED INTERNAL AUDIT WORKSHOP By Paul J. Kunder RABQSA QMS Lead Auditor/Aerospace Industry Experienced Auditor RABQSA Approved Aerospace Auditor Transition Training (AATT) Trainer Vice Chair U.S. Z1A
List of Documented Information (Document & Records)
 As per IATF 16949:2016 Clause Type Clause Reference 4.3 Determining the scope of the quality management system 5.1.1.1 Corporate responsibility The only permitted exclusion for this Automotive QMS Standard
As per IATF 16949:2016 Clause Type Clause Reference 4.3 Determining the scope of the quality management system 5.1.1.1 Corporate responsibility The only permitted exclusion for this Automotive QMS Standard
Quality Systems Manual Rev. NC Issued July 9 / 2018
 NMT Specialized Machining Inc 290 Shoemaker Street Kitchener, Ontario Canada N2E 3E1 Quality Systems Manual Rev. NC Issued July 9 / 2018 Conforms to AS9100 Rev D and ISO 9001:2015 Table of Contents Introduction
NMT Specialized Machining Inc 290 Shoemaker Street Kitchener, Ontario Canada N2E 3E1 Quality Systems Manual Rev. NC Issued July 9 / 2018 Conforms to AS9100 Rev D and ISO 9001:2015 Table of Contents Introduction
NMT Specialized Machining Inc & NMT General Machining Inc AS 9100 Rev C Quality Systems Manual
 NMT Specialized Machining Inc & NMT General Machining Inc AS 9100 Rev C Quality Systems Manual 290 Shoemaker Street Kitchener, Ontario Canada N2E 3E1 Table of Contents Introduction 5 Documentation Scheme..
NMT Specialized Machining Inc & NMT General Machining Inc AS 9100 Rev C Quality Systems Manual 290 Shoemaker Street Kitchener, Ontario Canada N2E 3E1 Table of Contents Introduction 5 Documentation Scheme..
UNIVERSAL STAINLESS NORTH JACKSON FACILITY. Quality Systems Manual
 UNIVERSAL STAINLESS NORTH JACKSON FACILITY Revision 8 Issued 12-5-17 Conforms to AS9100 Rev. D and ISO 9001:2015 This document may contain proprietary information and may only be released to third parties
UNIVERSAL STAINLESS NORTH JACKSON FACILITY Revision 8 Issued 12-5-17 Conforms to AS9100 Rev. D and ISO 9001:2015 This document may contain proprietary information and may only be released to third parties
Lobo Ventures Ltd. Precision Mold & Tool Government Division Warfield San Antonio, TX, 78216
 Effective date: 03/27/2018 Page 1 of 31 Lobo Ventures Ltd. Precision Mold & Tool Government Division 12027 Warfield San Antonio, TX, 78216 QUALITY MANUAL AS9100 Rev. D Effective date: 03/27/2018 Page
Effective date: 03/27/2018 Page 1 of 31 Lobo Ventures Ltd. Precision Mold & Tool Government Division 12027 Warfield San Antonio, TX, 78216 QUALITY MANUAL AS9100 Rev. D Effective date: 03/27/2018 Page
QUALITY MANAGEMENT SYSTEM MANUAL
 High Temp Metals, Inc. 12500 Foothill Blvd. Sylmar, CA 91342-6038 Phone: 800-500-2141 818-362-5357 FAX: 818-362-9884 Email: sales@hightempmetals.com QUALITY MANAGEMENT SYSTEM MANUAL This document is uncontrolled
High Temp Metals, Inc. 12500 Foothill Blvd. Sylmar, CA 91342-6038 Phone: 800-500-2141 818-362-5357 FAX: 818-362-9884 Email: sales@hightempmetals.com QUALITY MANAGEMENT SYSTEM MANUAL This document is uncontrolled
QUALITY MANUAL QM0492 ISO SUPPLEMENT III
 QUALITY MANUAL QM0492 ISO 13485 SUPPLEMENT III REV. ORIG Foreword This document defines the quality management system requirements to be applied when processing product for clients within the medical sector
QUALITY MANUAL QM0492 ISO 13485 SUPPLEMENT III REV. ORIG Foreword This document defines the quality management system requirements to be applied when processing product for clients within the medical sector
UNCONTROLLED DOCUMENT
 Rheem Manufacturing Company Fort Smith, Arkansas Original Release: 4/06/09 Revision Date: 10/25/16 Quality Management System D. Presley 05 1 of 1 Manual Contents A. Johnson 10/25/16 0 Section Title Revision
Rheem Manufacturing Company Fort Smith, Arkansas Original Release: 4/06/09 Revision Date: 10/25/16 Quality Management System D. Presley 05 1 of 1 Manual Contents A. Johnson 10/25/16 0 Section Title Revision
UNIVERSITY OF NAIROBI
 Q# UNIVERSITY OF NAIROBI 4 CONTEXTS OF THE ORGANIZATION Yes No Remarks 4.1 UNDERSTANDING THE ORGANIZATION AND ITS CONTEXT 1 Have we determined the external and internal issues that are relevant to our
Q# UNIVERSITY OF NAIROBI 4 CONTEXTS OF THE ORGANIZATION Yes No Remarks 4.1 UNDERSTANDING THE ORGANIZATION AND ITS CONTEXT 1 Have we determined the external and internal issues that are relevant to our
NATO STANDARD AQAP-2110 NATO QUALITY ASSURANCE REQUIREMENTS FOR DESIGN, DEVELOPMENT AND PRODUCTION
 NATO STANDARD AQAP-2110 NATO QUALITY ASSURANCE REQUIREMENTS FOR DESIGN, DEVELOPMENT AND PRODUCTION Edition D Version 1 JUNE 2016 NORTH ATLANTIC TREATY ORGANIZATION ALLIED QUALITY ASSURANCE PUBLICATION
NATO STANDARD AQAP-2110 NATO QUALITY ASSURANCE REQUIREMENTS FOR DESIGN, DEVELOPMENT AND PRODUCTION Edition D Version 1 JUNE 2016 NORTH ATLANTIC TREATY ORGANIZATION ALLIED QUALITY ASSURANCE PUBLICATION
ISO 9001: 2000 (December 13, 2000) QUALITY MANAGEMENT SYSTEM DOCUMENTATION OVERVIEW MATRIX
 In completing your Documented Quality Management System Review, it is important that the following matrix be completed and returned to us as soon as possible. This will save time during the review and
In completing your Documented Quality Management System Review, it is important that the following matrix be completed and returned to us as soon as possible. This will save time during the review and
SUPPLIER QUALITY SYSTEM SURVEY
 SUPPLIER QUALITY SYSTEM SURVEY DATE (M/d/yy): 55 Dragon Court Woburn, MA 01801 Tel: (781) 933-7300 Fax: (781) 935-4529 GENERAL INFORMATION Supplier Name: Physical Address: City: State: Zip: Sr. Company
SUPPLIER QUALITY SYSTEM SURVEY DATE (M/d/yy): 55 Dragon Court Woburn, MA 01801 Tel: (781) 933-7300 Fax: (781) 935-4529 GENERAL INFORMATION Supplier Name: Physical Address: City: State: Zip: Sr. Company
Issued by: Chris Abrey Eff. Date: 10/1/13 Rev: A Cover QUALITY MANUAL
 Quality System Manual Accurate Perforating & Metal Fabricating Company Issued by: Chris Abrey Eff. Date: 10/1/13 Rev: A Cover QUALITY MANUAL Approved Date: 10/1/13 By: Chris Abrey Quality System Manual
Quality System Manual Accurate Perforating & Metal Fabricating Company Issued by: Chris Abrey Eff. Date: 10/1/13 Rev: A Cover QUALITY MANUAL Approved Date: 10/1/13 By: Chris Abrey Quality System Manual
PCA ELECTRONICS, INC. Quality Manual
 PCA ELECTRONICS, INC. Conforms to ISO 9001:2015 (c) 2018 PCA Electronics, Inc.; all rights reserved. This document may contain proprietary information and may only be released to third parties with approval
PCA ELECTRONICS, INC. Conforms to ISO 9001:2015 (c) 2018 PCA Electronics, Inc.; all rights reserved. This document may contain proprietary information and may only be released to third parties with approval
PRECISE INDUSTRIES INC. Quality Manual
 PRECISE INDUSTRIES INC Revision N Issued July 5, 2017 Conforms to AS9100 Rev. D and ISO 9001:2015 Copyright Year2017 [PRECISE INDUSTRIES INC]; all rights reserved. This document may contain proprietary
PRECISE INDUSTRIES INC Revision N Issued July 5, 2017 Conforms to AS9100 Rev. D and ISO 9001:2015 Copyright Year2017 [PRECISE INDUSTRIES INC]; all rights reserved. This document may contain proprietary
IMPORTANCE OF AUDITING ISO 9001: 2015
 IMPORTANCE OF AUDITING ISO 9001: 2015 PMI, PMP, PMBOK and the PMI Registered Education Provider logo are registered marks of the Project Management Institute, Inc. At the end of the class, please fill
IMPORTANCE OF AUDITING ISO 9001: 2015 PMI, PMP, PMBOK and the PMI Registered Education Provider logo are registered marks of the Project Management Institute, Inc. At the end of the class, please fill
BAITY SCREW MACHINE PRODUCTS QUALITY MANUAL
 BAITY SCREW MACHINE PRODUCTS QUALITY MANUAL Page 1 of 33 TABLE OF CONTENTS SECTION TITLE PAGE 0 Company Introduction... 4 0 Organizational Chart.. 5 1 Scope.. 6 2 Related Documents... 6 3 Terminology...
BAITY SCREW MACHINE PRODUCTS QUALITY MANUAL Page 1 of 33 TABLE OF CONTENTS SECTION TITLE PAGE 0 Company Introduction... 4 0 Organizational Chart.. 5 1 Scope.. 6 2 Related Documents... 6 3 Terminology...
AS 9100 Rev C Quality Systems Manual AS-050C-QM
 AS 9100 Rev C Quality Systems Manual AS-050C-QM Innovative Control Systems, Inc. 10801 N. 24 th Ave. Suite 101-103 Phoenix, AZ 85029 U.S.A. www.icsaero.com +01-602-861-6984 VOICE +01-602-588-9440 FAX Table
AS 9100 Rev C Quality Systems Manual AS-050C-QM Innovative Control Systems, Inc. 10801 N. 24 th Ave. Suite 101-103 Phoenix, AZ 85029 U.S.A. www.icsaero.com +01-602-861-6984 VOICE +01-602-588-9440 FAX Table
NATO STANDARD AQAP-2310 NATO QUALITY ASSURANCE REQUIREMENTS FOR AVIATION, SPACE AND DEFENCE SUPPLIERS
 NATO STANDARD AQAP-2310 NATO QUALITY ASSURANCE REQUIREMENTS FOR AVIATION, SPACE AND DEFENCE SUPPLIERS Edition B Version 1 DECEMBER 2017 NORTH ATLANTIC TREATY ORGANIZATION ALLIED QUALITY ASSURANCE PUBLICATION
NATO STANDARD AQAP-2310 NATO QUALITY ASSURANCE REQUIREMENTS FOR AVIATION, SPACE AND DEFENCE SUPPLIERS Edition B Version 1 DECEMBER 2017 NORTH ATLANTIC TREATY ORGANIZATION ALLIED QUALITY ASSURANCE PUBLICATION
Quality Systems Compliance LLC is your Compliance Partner
 Mark Durivage, Managing Principal Consultant and Member Quality Systems Compliance LLC 3118 Chanson Valley Road Lambertville, MI 48144 mark.durivage@qscompliance.com www.qscompliance.com 419-265-2862 This
Mark Durivage, Managing Principal Consultant and Member Quality Systems Compliance LLC 3118 Chanson Valley Road Lambertville, MI 48144 mark.durivage@qscompliance.com www.qscompliance.com 419-265-2862 This
Reference Paragraphs. Appendix B AQS Auditor Handbook
 Introduction Auditing is a basic tool to assess effective implementation of and conformity to Quality Management System (QMS) requirements. In addition to the determination of conformity, this handbook
Introduction Auditing is a basic tool to assess effective implementation of and conformity to Quality Management System (QMS) requirements. In addition to the determination of conformity, this handbook
ISO 9001:2015 Gap Analysis Check Sheet
 CONTEXT OF THE ORGANIZATION 4.1 Understanding the organization and its context Organization shall determine external and internal issues that are relevant to its purpose and strategic direction and that
CONTEXT OF THE ORGANIZATION 4.1 Understanding the organization and its context Organization shall determine external and internal issues that are relevant to its purpose and strategic direction and that
Quality Manual Template ISO 9001:2015 Quality Management System
 Quality Manual Template Table of Contents 1 INTRODUCTION... 5 2 QUALITY MANAGEMENT PRINCIPLES... 6 3 REFERENCES & DEFINITIONS... 6 4 CONTEXT OF THE ORGANIZATION... 8 4.1 ORGANIZATIONAL CONTEXT... 8 4.2
Quality Manual Template Table of Contents 1 INTRODUCTION... 5 2 QUALITY MANAGEMENT PRINCIPLES... 6 3 REFERENCES & DEFINITIONS... 6 4 CONTEXT OF THE ORGANIZATION... 8 4.1 ORGANIZATIONAL CONTEXT... 8 4.2
AVNET Logistics & EM Americas. Quality Manual
 AVNET Logistics & EM Americas Quality Manual Avnet is committed to ensuring customer satisfaction while meeting all customer and applicable legal, statutory and regulatory requirements. This is accomplished
AVNET Logistics & EM Americas Quality Manual Avnet is committed to ensuring customer satisfaction while meeting all customer and applicable legal, statutory and regulatory requirements. This is accomplished
14620 Henry Road Houston, Texas PH: FX: WEB: QUALITY MANUAL
 14620 Henry Road Houston, Texas 77060 PH: 281-447-3980 FX: 281-447-3988 WEB: www.texasinternational.com QUALITY MANUAL ISO 9001:2008 API Spec Q1, 9th Edition API Spec 8C 5 Th Edition MANUAL NUMBER: Electronic
14620 Henry Road Houston, Texas 77060 PH: 281-447-3980 FX: 281-447-3988 WEB: www.texasinternational.com QUALITY MANUAL ISO 9001:2008 API Spec Q1, 9th Edition API Spec 8C 5 Th Edition MANUAL NUMBER: Electronic
Supplier Quality Requirements Document (SQRD)
 Supplier Quality Requirements Document (SQRD) D127135 Rev. AE Supplier Quality Requirements Document Page 2 of 8 1.0 SCOPE This document applies to all worldwide production suppliers of Coherent, Inc.
Supplier Quality Requirements Document (SQRD) D127135 Rev. AE Supplier Quality Requirements Document Page 2 of 8 1.0 SCOPE This document applies to all worldwide production suppliers of Coherent, Inc.
Supplier Quality Survey. 1. Type of Business: g) Commodities supplied? Supplier Changes/comments: 2. Headcount breakdown by group: Purchasing
 Supplier: Phone: Prime Contact/Title: Sales Contact/Title: Address: Fax: e-mail address e-mail address Quality Contact/Title: e-mail address 1. Type of Business: a) Number of years in business? b) Company
Supplier: Phone: Prime Contact/Title: Sales Contact/Title: Address: Fax: e-mail address e-mail address Quality Contact/Title: e-mail address 1. Type of Business: a) Number of years in business? b) Company
I. Development, Submittal, and Acceptance of a Quality Management System
 I. Development, Submittal, and Acceptance of a Quality Management System 1. Scope Under the Contract, quality assurance and quality control management are the responsibility of the Contractor. The Contractor
I. Development, Submittal, and Acceptance of a Quality Management System 1. Scope Under the Contract, quality assurance and quality control management are the responsibility of the Contractor. The Contractor
Quality System Manual - Section 00
 Quality System Manual - Section 00 INDEX AND REVISION STATUS Issued by: Quality Assurance Eff. Date: 06/10/2014 Rev.: A Pg. 1 of 4 QUALITY SYSTEM MANUAL SECTION 0 - INDEX AND REVISION STATUS SECTION 1
Quality System Manual - Section 00 INDEX AND REVISION STATUS Issued by: Quality Assurance Eff. Date: 06/10/2014 Rev.: A Pg. 1 of 4 QUALITY SYSTEM MANUAL SECTION 0 - INDEX AND REVISION STATUS SECTION 1
Objective Evidence Record (OER)
 This material is derived from SAE AS9101D, which is copyrighted intellectual property of SAE international. SAE is not responsible for outcomes resulting from use of this material. APPENDIX A OBJECTIVE
This material is derived from SAE AS9101D, which is copyrighted intellectual property of SAE international. SAE is not responsible for outcomes resulting from use of this material. APPENDIX A OBJECTIVE
Desk Audit of. Based on Federal Transit Administration (FTA) Quality Assurance and Quality Control Guidelines FTA-IT
 Desk Audit of Based on Federal Transit Administration (FTA) Quality Assurance and Quality Control Guidelines FTA-IT-90-5001-02.1 Reviewed by: Element Requirements Applicable 1. Is a quality policy defined
Desk Audit of Based on Federal Transit Administration (FTA) Quality Assurance and Quality Control Guidelines FTA-IT-90-5001-02.1 Reviewed by: Element Requirements Applicable 1. Is a quality policy defined
Continual Improvement
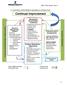 15. CONTINUAL IMPROVEMENT SEQUENCE & INTERACTION Continual Customer and Stakeholders Requirements Resource / Support Finance Document Control Information System Human Resources Competence Training Performance
15. CONTINUAL IMPROVEMENT SEQUENCE & INTERACTION Continual Customer and Stakeholders Requirements Resource / Support Finance Document Control Information System Human Resources Competence Training Performance
POLYCRAFT INCORPORATED QUALITY MANUAL Quality Manual QM -10 Approval: D. Wheeler.
 Polycraft, Inc. QM - 10 Title: Polycraft, Inc. Quality Manual Copy Approval: D. Wheeler This Quality Manual is the sole property of Polycraft, Inc. and intended for exclusive use by the organization. This
Polycraft, Inc. QM - 10 Title: Polycraft, Inc. Quality Manual Copy Approval: D. Wheeler This Quality Manual is the sole property of Polycraft, Inc. and intended for exclusive use by the organization. This
Summary of ISO 9001:2015 New and Changed Requirements
 This is a summary of the new and changed ISO 9001:2015 requirements compared to ISO 9001:2008. 4. Context of the Organization 4.1 Changes Understanding the Organization and its Context New requirement
This is a summary of the new and changed ISO 9001:2015 requirements compared to ISO 9001:2008. 4. Context of the Organization 4.1 Changes Understanding the Organization and its Context New requirement
INSERT COMPANY NAME/LOGO HERE
 This gap analysis checklist is prepared for use in evaluating a Quality Management System (QMS) against the requirements of the new Aerospace standard. The AS 9100 Rev D standard includes the requirements
This gap analysis checklist is prepared for use in evaluating a Quality Management System (QMS) against the requirements of the new Aerospace standard. The AS 9100 Rev D standard includes the requirements
Quality Systems Manual
 Quality Systems Manual This manual was developed to support ISO 9001 elements 1445 Oakland Drive, San Jose, CA 95112-2702 - Tel: (408) 294-9897 - Fax: (408) 297-1540 http://www.gorillacircuits.com Reference
Quality Systems Manual This manual was developed to support ISO 9001 elements 1445 Oakland Drive, San Jose, CA 95112-2702 - Tel: (408) 294-9897 - Fax: (408) 297-1540 http://www.gorillacircuits.com Reference
Specification for Quality Programs for the Petroleum, Petrochemical and Natural Gas Industry
 Addendum 1 June 2010 Effective Date: December 1, 2010 Specification for Quality Programs for the Petroleum, Petrochemical and Natural Gas Industry ANSI/API SPECIFICATION Q1 EIGHTH EDITION, DECEMBER 2007
Addendum 1 June 2010 Effective Date: December 1, 2010 Specification for Quality Programs for the Petroleum, Petrochemical and Natural Gas Industry ANSI/API SPECIFICATION Q1 EIGHTH EDITION, DECEMBER 2007
25 D.L. Martin Drive Mercersburg, PA (717)
 QUALITY MANUAL D. L. MARTIN CO. 25 D.L. Martin Drive Mercersburg, PA 17236 (717) 328-2141 Revision 14 August 2012 Michael A. White Manager, QA & Engineering D.L. Martin Co. Quality Manual UNCONTROLLED
QUALITY MANUAL D. L. MARTIN CO. 25 D.L. Martin Drive Mercersburg, PA 17236 (717) 328-2141 Revision 14 August 2012 Michael A. White Manager, QA & Engineering D.L. Martin Co. Quality Manual UNCONTROLLED
Conducting Quality Audit based on ISO 9001:2015
 Conducting Quality Audit based on ISO 9001:2015 FREE Professional Development Seminar Series At the end of the class, please fill the training feedback and referral forms! Return the form at the reception!
Conducting Quality Audit based on ISO 9001:2015 FREE Professional Development Seminar Series At the end of the class, please fill the training feedback and referral forms! Return the form at the reception!
QUALITY MANUAL. Number: M-001 Revision: C Page 1 of 18 THIS DOCUMENT IS CONSIDERED UNCONTROLLED UNLESS ISSUED IDENTIFIED AS CONTROLLED
 Page 1 of 18 THIS DOCUMENT IS CONSIDERED UNCONTROLLED UNLESS ISSUED IDENTIFIED AS CONTROLLED Page 2 of 18 REVISION HISTORY DATE CHANGE DESCRIPTION 10/11/06 Original release 10/21/09 Revised to ISO9001:2008
Page 1 of 18 THIS DOCUMENT IS CONSIDERED UNCONTROLLED UNLESS ISSUED IDENTIFIED AS CONTROLLED Page 2 of 18 REVISION HISTORY DATE CHANGE DESCRIPTION 10/11/06 Original release 10/21/09 Revised to ISO9001:2008
ISO 9001:2015. Quality Management System. Manual
 ISO 9001:2015 Quality Management System Manual Introduction Company has made the Strategic Business Decision to develop and implement an effective Quality Management Systems (QMS) across all areas of the
ISO 9001:2015 Quality Management System Manual Introduction Company has made the Strategic Business Decision to develop and implement an effective Quality Management Systems (QMS) across all areas of the
ISO/DIS 9001: 2014 comparison with ISO 9001:2008. ISO 9001:2015 Updates. (Based on Draft International Standard, DIS) ISO/DIS 9001 ISO 9001:2008
 ISO 9001:2015 Updates (Based ondraft International Standard, DIS) August 2014 Page 1 ISO 9001:2015 Updates (Based on Draft International Standard, DIS) ISO/DIS 9001: 2014 comparison with ISO 9001:2008
ISO 9001:2015 Updates (Based ondraft International Standard, DIS) August 2014 Page 1 ISO 9001:2015 Updates (Based on Draft International Standard, DIS) ISO/DIS 9001: 2014 comparison with ISO 9001:2008
ISO 9001: 2015 Quality Management System Certification. Awareness Training
 ISO 9001: 2015 Quality Management System Certification Awareness Training ISO 9001: 2015 STRUCTURE The new standard is modeled around the ISO Directive Annex SL, a high level structure (HSL) based on the
ISO 9001: 2015 Quality Management System Certification Awareness Training ISO 9001: 2015 STRUCTURE The new standard is modeled around the ISO Directive Annex SL, a high level structure (HSL) based on the
20 September 2017 Document No. QM-ISO revision T ASTRONAUTICS CORPORATION OF AMERICA S. AS 9100 and FAA QUALITY MANUAL. Proprietary Notice
 20 September 2017 Document No. QM-ISO revision T ASTRONAUTICS CORPORATION OF AMERICA S AS 9100 and FAA QUALITY MANUAL Proprietary Notice Information disclosed herein is the property of Astronautics Corporation
20 September 2017 Document No. QM-ISO revision T ASTRONAUTICS CORPORATION OF AMERICA S AS 9100 and FAA QUALITY MANUAL Proprietary Notice Information disclosed herein is the property of Astronautics Corporation
9100 Team July, IAQG is a trademark the International Aerospace Quality Group. Copyright 2014 IAQG. All rights reserved.
 9100 Series 2016 Revision Overview 9100 Team July, 2014 1 9100 Revision The Plan 9100 Series Revision High Level Plan The 9100 is based on ISO 9001 and is thus affected by the ISO TC176 revision activity
9100 Series 2016 Revision Overview 9100 Team July, 2014 1 9100 Revision The Plan 9100 Series Revision High Level Plan The 9100 is based on ISO 9001 and is thus affected by the ISO TC176 revision activity
Quality Manual ISO 9001:2000
 Quality Manual ISO 9001:2000 Page 2 of 23 TABLE OF CONTENTS COVER PAGE...1 TABLE OF CONTENTS...2 SIGNATURES...3 QUALITY POLICY...4 INTRODUCTION...5 CORPORATE PROFILE...9 4.O QUALITY MANAGEMENT SYSTEM...10
Quality Manual ISO 9001:2000 Page 2 of 23 TABLE OF CONTENTS COVER PAGE...1 TABLE OF CONTENTS...2 SIGNATURES...3 QUALITY POLICY...4 INTRODUCTION...5 CORPORATE PROFILE...9 4.O QUALITY MANAGEMENT SYSTEM...10
APS Cleaning Quality Management System Scope of Certification The provision of commercial and industrial cleaning services throughout Queensland.
 Quality Management System Scope of Certification The provision of commercial and industrial cleaning services throughout Queensland. Table of Contents Contents 1. Introduction... 3 1.1. Process Approach...
Quality Management System Scope of Certification The provision of commercial and industrial cleaning services throughout Queensland. Table of Contents Contents 1. Introduction... 3 1.1. Process Approach...
Document Number: QM001 Page 1 of 19. Rev Date: 10/16/2009 Rev Num: 1. Quality Manual. Quality Manual. Controlled Copy
 QM001 Page 1 of 19 Quality Manual QM001 Page 2 of 19 Table of Contents Page Company Profile 4 Approval 4 Revision History 4 Distribution List 4 1.0 Scope 5 Section 2: Normative Reference 6 2.0 Quality
QM001 Page 1 of 19 Quality Manual QM001 Page 2 of 19 Table of Contents Page Company Profile 4 Approval 4 Revision History 4 Distribution List 4 1.0 Scope 5 Section 2: Normative Reference 6 2.0 Quality
P. 1. Identify the Differences between ISO9001:2000 與 ISO9001:2008 ISO9001:2008 ISO9001:2000 版本的異同. 5 January 2009 ISO 9000 SERIES
 Identify the Differences between ISO9001:2000 and ISO 9001:2008 審視 ISO9001:2000 與 ISO9001:2008 版本的異同 ISO 9000 SERIES ISO 19011 ISO9000 5 January 2009 ISO9001 ISO9004 2 ISO 9000 SERIES ISO 9001 ISO 9000
Identify the Differences between ISO9001:2000 and ISO 9001:2008 審視 ISO9001:2000 與 ISO9001:2008 版本的異同 ISO 9000 SERIES ISO 19011 ISO9000 5 January 2009 ISO9001 ISO9004 2 ISO 9000 SERIES ISO 9001 ISO 9000
AS9101 Revision E Understanding the Changes
 AS9101 Revision E Understanding the Changes Introduction Aviation, space and defense (ASD) organizations are highly regulated to ensure their products are safe and reliable, conform to statutory and regulatory
AS9101 Revision E Understanding the Changes Introduction Aviation, space and defense (ASD) organizations are highly regulated to ensure their products are safe and reliable, conform to statutory and regulatory
QUALITY MANUAL ISO 9001 QUALITY MANAGEMENT SYSTEM
 QUALITY MANUAL ISO 9001 QUALITY MANAGEMENT SYSTEM APPROVED BY: JOSE ALBERTO APPROVED ON: 08/10/2017 Rev.: 1 M001 QUALITY MANAGEMENT SYSTEM PAGE: 1 of 20 TABLE OF CONTENTS SECTION TITLE PAGE Table of Contents
QUALITY MANUAL ISO 9001 QUALITY MANAGEMENT SYSTEM APPROVED BY: JOSE ALBERTO APPROVED ON: 08/10/2017 Rev.: 1 M001 QUALITY MANAGEMENT SYSTEM PAGE: 1 of 20 TABLE OF CONTENTS SECTION TITLE PAGE Table of Contents
SUPPLIER QUALITY SYSTEM SURVEY
 SUPPLIER NAME DATE WEB ADDRESS ADDRESS CITY STATE ZIP PUBLICLY OR PRIVATELY HELD? TEL FAX CONTACT INFORMATION POSITION CONTACT NAME PHONE FAX E-MAIL PRESIDENT OPERATIONS MGR. QUALITY MGR. ENGINEERING MGR.
SUPPLIER NAME DATE WEB ADDRESS ADDRESS CITY STATE ZIP PUBLICLY OR PRIVATELY HELD? TEL FAX CONTACT INFORMATION POSITION CONTACT NAME PHONE FAX E-MAIL PRESIDENT OPERATIONS MGR. QUALITY MGR. ENGINEERING MGR.
Overview of Draft API Standard 20M
 Overview of Draft API Standard 20M Qualification of Suppliers of Machining Services for Use in the Petroleum and Natural Gas Industries API STANDARD 20M Slide 1 of 9 Overview Standard 20M provides methods
Overview of Draft API Standard 20M Qualification of Suppliers of Machining Services for Use in the Petroleum and Natural Gas Industries API STANDARD 20M Slide 1 of 9 Overview Standard 20M provides methods
ISO 9001:2015: Change or Not to Change or How to Transition from ISO9001:2008 and ISO9001:2015. Joan Richter ASQ Baltimore (Section 502) June 2016
 ISO 9001:2015: Change or Not to Change or How to Transition from ISO9001:2008 and ISO9001:2015 Joan Richter ASQ Baltimore (Section 502) June 2016 HISTORY OF ISO 9001 Standard (28 Years of ISO) 1987 Procedures
ISO 9001:2015: Change or Not to Change or How to Transition from ISO9001:2008 and ISO9001:2015 Joan Richter ASQ Baltimore (Section 502) June 2016 HISTORY OF ISO 9001 Standard (28 Years of ISO) 1987 Procedures
Railroad Friction Products Corporation
 Railroad Friction Products Corporation Our Quality Philosophy: "At Railroad Friction Products Corporation we will consistently provide products that meet and exceed customer and regulatory requirements
Railroad Friction Products Corporation Our Quality Philosophy: "At Railroad Friction Products Corporation we will consistently provide products that meet and exceed customer and regulatory requirements
Quality Manual ISO 9001:2008 ISO 9001:2015
 Quality Manual ISO 9001:2008 ISO 9001:2015 SAE CIRCUITS, INC. 4820 63 rd Street Suite 100 Boulder, CO 80301 USA www.saecircuits.com Table of Contents 1. Company Information 3 2. QMS Scope and Exclusions
Quality Manual ISO 9001:2008 ISO 9001:2015 SAE CIRCUITS, INC. 4820 63 rd Street Suite 100 Boulder, CO 80301 USA www.saecircuits.com Table of Contents 1. Company Information 3 2. QMS Scope and Exclusions
Document Type: Main Process: Revision Level: Page: POLICY QUALITY ASSURANCE 3 1 of 6 Process Owner Title:
 POLICY QUALITY ASSURANCE 3 1 of 6 Process Owner A. SCOPE This document contains requirements for conducting business with Tri Star Metals, LLC and its customers. It is applicable to suppliers that provide
POLICY QUALITY ASSURANCE 3 1 of 6 Process Owner A. SCOPE This document contains requirements for conducting business with Tri Star Metals, LLC and its customers. It is applicable to suppliers that provide
QUALITY MANUAL Revision H
 QUALITY MANUAL Revision H JADE PRECISION MEDICAL COMPONENTS, LLC 3063 B Philmont Avenue Huntingdon Valley, PA 19006 Page: 1 of 15 Table of Contents 1. Purpose & Scope... 2 2. Applicable Standards... 2
QUALITY MANUAL Revision H JADE PRECISION MEDICAL COMPONENTS, LLC 3063 B Philmont Avenue Huntingdon Valley, PA 19006 Page: 1 of 15 Table of Contents 1. Purpose & Scope... 2 2. Applicable Standards... 2
