Clinical Data Standards Governance. Tammy Burns, Expert Clinical Data Manager 11 th January 2013
|
|
|
- Abigail Flynn
- 6 years ago
- Views:
Transcription
1 Clinical Data Standards Governance Tammy Burns, Expert Clinical Data Manager 11 th January 2013
2 Standards Used and Formal Govenance Standard Types General and Therapeutic Area Data Standards Collection Modules EMD Serono/Merck Serono SDTM Standards EMD Serono/Merck Serono ADaM Standards Controlled Terminology Value Level Metadata GBS Output Standards Standard algorithms The official CDISC Standards Standard validated programs and tools Standard mappings 2 Clinical Data Standards Governance
3 Objective of the WI To describe the management of the general and therapeutic area data standards, Study Data Tabulation Model (SDTM) and Analysis Data Model (ADaM) in relation to the creation, use, maintenance and documentation of the standards, to ensure data consistency and to facilitate regulatory submissions 3 Clinical Data Standards Governance
4 Standards Governance Requires empowerment of governance teams to reinforce the standards within the organisation and a strong support from upper management It requires the governance teams to be advocates for the use of standards increasing the acceptance across the organisation Allocating resources for standards management is a must to achieve the benefits 4 Clinical Data Standards Governance
5 Governance Teams Standards Governance Teams General Data Standards (DS) Team Therapeutic Area Data Standards (TA DS) Team for each of the main Therapeutic Areas Membership DS Team Lead Clinical Data Standards Librarian(s) Controlled Terminology Librarian TA DS Team Lead Controlled Terminology Librarian Merck Serono (MS) SDTM Team Merck Serono (MS) ADaM Team SDTM Team Lead (appointed by the Head of Global Clinical Data Science or designee) SDTM Librarian(s) Controlled Terminology Librarian MS ADaM Team Representative appointed by the MS ADaM Team Lead MS ADaM Team Lead (appointed by the Head of Global Biostatistics or designee) Statistical Standards Librarian (ADaM) Statistical Programmer(s) Biostatistician(s) 5 Presentation title in footer 00 Month 0000
6 Standards Governance Teams (ctd) Organisation Level Function EMD Serono/Merck Serono Clinical Governance Head of GDM and GBS Standard Type General Data Standards Team TA Data Standards Team SDTM Team ADaM Team Project PDM PDM Specialist / PDM Specialist /Project Stats Prog / Biostat Trial TDM CRO TDM CRO TDM CRO Trial Stats Prog Trial Biostat CRO 6 Clinical Data Standards Governance
7 When is the governance process used? Previous governance only required an exemption when not complying with the existing standard. Study specific forms did not require approval/governance process. The new governance requires if the current clinical data standards do not cover all the requirements for setting up any of the trial specifications (e.g. ecrf, SDTM, ADaM) or anything which does not comply with an existing standard a New, Change or Exemption Request Form is completed and approved prior to implementation. 7 Clinical Data Standards Governance
8 Completion of Metadata on the Clinical Data Standards New, Change or Exemption Request Form When the governance was updated to incorporate the governance of SDTM, CT and ADaM Standards it was felt that the SDTM metadata should be provided as early as when the CRFs were being designed We then agreed to take a flexible approach to the completion of the New, Change or Exemption form so that the process can be adapted to what is required and what is feasible. Definition of all metadata is now not required at the CRF creation phase. However, where possible, the minimum information should be collected (i.e. CT and VLM) and an SDTM Expert should review the request to evaluate the impact on the SDTM 8 Clinical Data Standards Governance
9 When, How Much and Where Do We Govern How do we balance strict timelines for go-live of a CRF with time to manage and mandate the use of standards? Do we govern the SDTM at the same time as the General Clinical Data Collection Standards? Do we govern all forms and modules including study specific? 9 Clinical Data Standards Governance
10 Standards and Governance Team Objectives Development of new standards Assessing the impact of new releases of CDISC standards and related guidance from Health Authorities on the company specific standards Freezing standards which are no longer in use Providing general rules and conventions for company specific use of general data standards, control terminology, CDISC SDTM and ADaM, as applicable Develop and provide training 10 Clinical Data Standards Governance
11 Standards and Governance Team Objectives Providing expert guidance on the application of the standards Compliance to existing standards Management of exemptions Track all exemptions and review them for potential changes to the standards Track and review all requests for updates to the standards and make the changes or provide rationale as to why a request is not made 11 Clinical Data Standards Governance
October, Integration and Implementation of CDISC Standards
 Integration and Implementation of CDISC Standards October, 2008 Barbara Lentz, Associate Director, Electronic Data Management Standards, Processes & Training Pat Majcher, Statistical Reporting Services
Integration and Implementation of CDISC Standards October, 2008 Barbara Lentz, Associate Director, Electronic Data Management Standards, Processes & Training Pat Majcher, Statistical Reporting Services
CDISC Data Standards Validation
 CDISC Data Standards Validation How can it be done? Peter Van Reusel Business Unit Director Business & Decision Life Sciences Tel +32 2 774 11 00 Fax +32 2 774 11 99 Mobile +32 476 54 59 17 peter.vanreusel@businessdecision.com
CDISC Data Standards Validation How can it be done? Peter Van Reusel Business Unit Director Business & Decision Life Sciences Tel +32 2 774 11 00 Fax +32 2 774 11 99 Mobile +32 476 54 59 17 peter.vanreusel@businessdecision.com
Roadmap for Managing Multiple CRO Vendors
 PharmaSUG 2017 - Paper PO27 Roadmap for Managing Multiple CRO Vendors Veena Nataraj, Shire, Lexington, MA Karin LaPann, Shire, Lexington, MA ABSTRACT When working with CROs, the relationship established
PharmaSUG 2017 - Paper PO27 Roadmap for Managing Multiple CRO Vendors Veena Nataraj, Shire, Lexington, MA Karin LaPann, Shire, Lexington, MA ABSTRACT When working with CROs, the relationship established
End-to-End Management of Clinical Trials Data
 End-to-End Management of Clinical Trials Data A Revolutionary Step Toward Supporting Clinical Trials Analysis Over the Next Decades of Clinical Research WHITE PAPER SAS White Paper Table of Contents Introduction....
End-to-End Management of Clinical Trials Data A Revolutionary Step Toward Supporting Clinical Trials Analysis Over the Next Decades of Clinical Research WHITE PAPER SAS White Paper Table of Contents Introduction....
CDASH Clinical Data Acquisition Standards Harmonization
 CDISC Italian User Group Meeting 16 November 2007 CDASH Clinical Data Acquisition Standards Harmonization 1 Outline Background Organization Goals Timelinesand Process Review of progress Results Future
CDISC Italian User Group Meeting 16 November 2007 CDASH Clinical Data Acquisition Standards Harmonization 1 Outline Background Organization Goals Timelinesand Process Review of progress Results Future
Aepodia is a team of experts with more than 15 years
 Aepodia is a team of experts with more than 15 years experience in early clinical development and translational science with small startup and large global pharmaceutical companies. Aepodia s team has
Aepodia is a team of experts with more than 15 years experience in early clinical development and translational science with small startup and large global pharmaceutical companies. Aepodia s team has
PharmaSUG Paper PO12
 PharmaSUG 2016 - Paper PO12 CDISC Standards End-to-End: Transitional Hurdles Alyssa Wittle, Chiltern International, King of Prussia, PA Christine McNichol, Chiltern International, King of Prussia, PA Antonio
PharmaSUG 2016 - Paper PO12 CDISC Standards End-to-End: Transitional Hurdles Alyssa Wittle, Chiltern International, King of Prussia, PA Christine McNichol, Chiltern International, King of Prussia, PA Antonio
CDASH 2.0. Berlin Éanna Kiely CDISC Engineer
 CDASH 2.0 Berlin Éanna Kiely CDISC Engineer 2016-09-13 Agenda CDASH 1.1 & CDASHIG 2.0 CDASH Concepts TAUG Examples Current Regulatory Perspective on CDASH Questions 2 CDASH: CDISC End to End Clinical Data
CDASH 2.0 Berlin Éanna Kiely CDISC Engineer 2016-09-13 Agenda CDASH 1.1 & CDASHIG 2.0 CDASH Concepts TAUG Examples Current Regulatory Perspective on CDASH Questions 2 CDASH: CDISC End to End Clinical Data
Chapter 1. Pharmaceutical Industry Overview
 Chapter 1 Pharmaceutical Industry Overview 1.1 Introduction 2 1.2 Regulations 2 1.2.1 Health Insurance Portability and Accountability Act 2 1.2.2 The Code of Federal Regulations 3 1.2.3 Guidance for Industry
Chapter 1 Pharmaceutical Industry Overview 1.1 Introduction 2 1.2 Regulations 2 1.2.1 Health Insurance Portability and Accountability Act 2 1.2.2 The Code of Federal Regulations 3 1.2.3 Guidance for Industry
PharmaSUG 2017 Paper DS14
 PharmaSUG 2017 Paper DS14 Considerations in Submitting Standardized Electronic Data Under the Animal Rule: The Use of SDTMIG and SENDIG Domains, and the Need for New Domains and Concepts Fred Wood, Accenture
PharmaSUG 2017 Paper DS14 Considerations in Submitting Standardized Electronic Data Under the Animal Rule: The Use of SDTMIG and SENDIG Domains, and the Need for New Domains and Concepts Fred Wood, Accenture
Clinical Data Acquisition Standards Harmonization (CDASH)
 Clinical Data Acquisition Standards Harmonization () Prepared by: CDISC Team Section Table of Contents Page 1.0 Orientation... 1 1.1 Purpose... 1 1.2 Organization of this Document... 1 1.2.1 General Notes...2
Clinical Data Acquisition Standards Harmonization () Prepared by: CDISC Team Section Table of Contents Page 1.0 Orientation... 1 1.1 Purpose... 1 1.2 Organization of this Document... 1 1.2.1 General Notes...2
The use of electronic Health Records in Clinical Research - The value of CDISC Standards
 The use of electronic Health Records in Clinical Research - The value of CDISC Standards FH-Prof. Dr. Jozef Aerts University of Applied Sciences FH Joanneum Graz, Austria Who is Jozef Aerts? CDISC volunteer
The use of electronic Health Records in Clinical Research - The value of CDISC Standards FH-Prof. Dr. Jozef Aerts University of Applied Sciences FH Joanneum Graz, Austria Who is Jozef Aerts? CDISC volunteer
CDISC UK Network face to face Reading
 CDISC UK Network face to face Reading Paul Houston, CDISC Head of European Operations CDISC Update and European Activity 2 CDISC Values and Principals Core value Foster CDISC community is altruistic
CDISC UK Network face to face Reading Paul Houston, CDISC Head of European Operations CDISC Update and European Activity 2 CDISC Values and Principals Core value Foster CDISC community is altruistic
Data Integrity in Clinical Trials the Sponsor Perspective
 Data Integrity in Clinical Trials the Sponsor Perspective Maximilian Stroebe, GSK Vaccines Three differences to Commercial Manufacturing Long process chains Source Data, Clinical Sites and Outsourcing
Data Integrity in Clinical Trials the Sponsor Perspective Maximilian Stroebe, GSK Vaccines Three differences to Commercial Manufacturing Long process chains Source Data, Clinical Sites and Outsourcing
Statistical Review and Data Standards: It s Gettin Better All The Time
 Statistical Review and Data Standards: It s Gettin Better All The Time Steve Wilson, Dr.P.H., CAPT USPHS Office of Biostatistics FDA/OMPT/CDER 2017 PharmaSUG Baltimore, MD May 17, 2017 Disclaimer This
Statistical Review and Data Standards: It s Gettin Better All The Time Steve Wilson, Dr.P.H., CAPT USPHS Office of Biostatistics FDA/OMPT/CDER 2017 PharmaSUG Baltimore, MD May 17, 2017 Disclaimer This
Electronic Health Records and Clinical Data Interchange Standards
 Electronic Health Records and Clinical Data Interchange Standards Stephen E. Wilson, Dr.P.H., CAPT USPHS Director Division of Biostatistics III Center for Drug Evaluation and Research US Food and Drug
Electronic Health Records and Clinical Data Interchange Standards Stephen E. Wilson, Dr.P.H., CAPT USPHS Director Division of Biostatistics III Center for Drug Evaluation and Research US Food and Drug
EUROPEAN COMMISSION ENTERPRISE DIRECTORATE-GENERAL QUESTIONS. and ANSWERS
 EUROPEAN COMMISSION ENTERPRISE DIRECTORATE-GENERAL Consumer goods Pharmaceuticals Brussels, F2/SM D (2008) QUESTIONS and ANSWERS The rules governing medicinal products in the European Union VOLUME 2 NOTICE
EUROPEAN COMMISSION ENTERPRISE DIRECTORATE-GENERAL Consumer goods Pharmaceuticals Brussels, F2/SM D (2008) QUESTIONS and ANSWERS The rules governing medicinal products in the European Union VOLUME 2 NOTICE
Department At-A-Glance
 Integrated Site Services Functional Role Integrated Site Services (ISS) includes global and country Start-Up and Regulatory units (RSU) and Integrated Patients & Site Services (IPSS). Our department s
Integrated Site Services Functional Role Integrated Site Services (ISS) includes global and country Start-Up and Regulatory units (RSU) and Integrated Patients & Site Services (IPSS). Our department s
Applying SDTM and ADaM to the Construction of Datamarts in Support of Cross-indication Regulatory Requests
 Applying SDTM and ADaM to the Construction of Datamarts in Support of Cross-indication Regulatory Requests Data-what? This presentation will discuss some of the complexities involved when integrating study
Applying SDTM and ADaM to the Construction of Datamarts in Support of Cross-indication Regulatory Requests Data-what? This presentation will discuss some of the complexities involved when integrating study
Relevant Standards: Standards for Registered Training Organisations (RTOs) 2015: Transition of training products
 Australian Computer Society Policies and Procedures: Manage transition from superseded training Relevant Standards: Standards for Registered Training Organisations (RTOs) 2015: 1.26-1.27 Transition of
Australian Computer Society Policies and Procedures: Manage transition from superseded training Relevant Standards: Standards for Registered Training Organisations (RTOs) 2015: 1.26-1.27 Transition of
Guidelines for Developing Randomization Procedures R03
 Guidelines for Developing Randomization Procedures R03 Table of Contents 1. Purpose 2. Objective 3. Process 4. Description of Outputs 5. Field Procedures for a Double Blind Trial Involving the Research
Guidelines for Developing Randomization Procedures R03 Table of Contents 1. Purpose 2. Objective 3. Process 4. Description of Outputs 5. Field Procedures for a Double Blind Trial Involving the Research
LOINC in Regulated Clinical Research a Lab LOINC Steeringg Committee Meeting 08 June 2017
 LOINC in Regulated Clinical Research a Lab LOINC Steeringg Committee Meeting 08 June 2017 Lauren Becnel, Ph.D. VP, Biomedical Informatics & Alliances, CDISC Asst Prof, Duncan Comprehensive Cancer Center,
LOINC in Regulated Clinical Research a Lab LOINC Steeringg Committee Meeting 08 June 2017 Lauren Becnel, Ph.D. VP, Biomedical Informatics & Alliances, CDISC Asst Prof, Duncan Comprehensive Cancer Center,
DATA SHARING AGREEMENT
 DATA SHARING AGREEMENT Effective Date: This Data Sharing Agreement ( Agreement ) is effective as of the Effective Date between EMD Serono, One Technology Place, Rockland, MA 02370 and Name ( Institution
DATA SHARING AGREEMENT Effective Date: This Data Sharing Agreement ( Agreement ) is effective as of the Effective Date between EMD Serono, One Technology Place, Rockland, MA 02370 and Name ( Institution
INVESTIGATIONAL DEVICE EXEMPTION APPLICATION. IDE Title (if title being used)
 INVESTIGATIONAL DEVICE EXEMPTION APPLICATION IDE Title (if title being used) Name of Sponsor Investigator, MD X Professor, Department Icahn School of Medicine at Mount Sinai Date of Submission Form version
INVESTIGATIONAL DEVICE EXEMPTION APPLICATION IDE Title (if title being used) Name of Sponsor Investigator, MD X Professor, Department Icahn School of Medicine at Mount Sinai Date of Submission Form version
Optimizing Your Study Data Submissions to FDA Updates from CDER and CBER
 This single PDF file contains the slides for all three presentations in the webinar: Optimizing Your Study Data Submissions to FDA Updates from CDER and CBER Please page down to find the slides for all
This single PDF file contains the slides for all three presentations in the webinar: Optimizing Your Study Data Submissions to FDA Updates from CDER and CBER Please page down to find the slides for all
PhilaSUG Spring 2007 Meeting Wednesday, June 13 th
 Spring 2007 Meeting Announcement PhilaSUG Spring 2007 Meeting Wednesday, June 13 th The Philadelphia Area SAS Users Group Spring Meeting will be on Wednesday, June 13, 2007 at 1:00 PM, and will be hosted
Spring 2007 Meeting Announcement PhilaSUG Spring 2007 Meeting Wednesday, June 13 th The Philadelphia Area SAS Users Group Spring Meeting will be on Wednesday, June 13, 2007 at 1:00 PM, and will be hosted
Swiss Multiple Sclerosis Society Rules of Procedure for the Scientific Advisory Board
 Swiss Multiple Sclerosis Society Rules of Procedure for the Scientific Advisory Board Based on Art. 8 of the Statutes, the following Rules of Procedure for the Scientific Advisory Board (SAB) are adopted:
Swiss Multiple Sclerosis Society Rules of Procedure for the Scientific Advisory Board Based on Art. 8 of the Statutes, the following Rules of Procedure for the Scientific Advisory Board (SAB) are adopted:
Public release of clinical information in drug submissions and medical device applications
 Public release of clinical information in drug submissions and medical device applications Health Products and Food Branch March 10, 2017 Health Canada is the federal department responsible for helping
Public release of clinical information in drug submissions and medical device applications Health Products and Food Branch March 10, 2017 Health Canada is the federal department responsible for helping
An Industry Perspective: Reducing the Complexity and Impact of Regulatory Changes in Latin America
 An Industry Perspective: Reducing the Complexity and Impact of Regulatory Changes in Latin America SPEAKERS: Janett Mugaburu-Richards, M.S. Pfizer Inc. Kavita Ramalingam Iyer, Ph.D Merck & Co., Inc. Contents
An Industry Perspective: Reducing the Complexity and Impact of Regulatory Changes in Latin America SPEAKERS: Janett Mugaburu-Richards, M.S. Pfizer Inc. Kavita Ramalingam Iyer, Ph.D Merck & Co., Inc. Contents
Standard for Exchange of Nonclinical Data Implementation Guide: Nonclinical Studies
 Standard for Exchange of Nonclinical Data Implementation Guide: Nonclinical Studies Prepared by the CDISC Standard for Exchange of Nonclinical Data Team Notes to Readers This is the implementation guide
Standard for Exchange of Nonclinical Data Implementation Guide: Nonclinical Studies Prepared by the CDISC Standard for Exchange of Nonclinical Data Team Notes to Readers This is the implementation guide
CHALLENGES OF GLOBAL CLINICAL TRIAL SUPPLY SOURCING SOME SPECIFICS FOR EMERGING MARKETS
 Clinical Trial Supply 28 th 29 th October 2014 Princeton, NJ - USA CHALLENGES OF GLOBAL CLINICAL TRIAL SUPPLY SOURCING SOME SPECIFICS FOR EMERGING MARKETS 1 Agenda The challenges associated with various
Clinical Trial Supply 28 th 29 th October 2014 Princeton, NJ - USA CHALLENGES OF GLOBAL CLINICAL TRIAL SUPPLY SOURCING SOME SPECIFICS FOR EMERGING MARKETS 1 Agenda The challenges associated with various
Data Quality and Integrity: From Clinical Monitoring to Marketing Approval
 Data Quality and Integrity: From Clinical Monitoring to Marketing Approval Nancy Detich, Ph.D., C.C.R.P. Senior Scientist, Clinical Strategy 18 November 2010 1 Objectives Identify the importance of accuracy,
Data Quality and Integrity: From Clinical Monitoring to Marketing Approval Nancy Detich, Ph.D., C.C.R.P. Senior Scientist, Clinical Strategy 18 November 2010 1 Objectives Identify the importance of accuracy,
White Paper INTRODUCING A COMPLEXITY SCORING SYSTEM IN CONTRACT NEGOTIATION
 White Paper INTRODUCING A COMPLEXITY SCORING SYSTEM IN CONTRACT NEGOTIATION Table of Contents 1. Introduction... 3 2. The Key Drivers For Study Startup Performance... 3 3. Contracts Negotiation Parallel
White Paper INTRODUCING A COMPLEXITY SCORING SYSTEM IN CONTRACT NEGOTIATION Table of Contents 1. Introduction... 3 2. The Key Drivers For Study Startup Performance... 3 3. Contracts Negotiation Parallel
OCTC 2012 CRO Selection
 OCTC 2012 CRO Selection Colin Macaulay Viron Therapeutics Inc. 15 Nov 2012 Viron Therapeutics Inc. Virtual Biotech Company (6) Phase 2a (48 pt) clinical trial in acute coronary syndrome (ACS) completed
OCTC 2012 CRO Selection Colin Macaulay Viron Therapeutics Inc. 15 Nov 2012 Viron Therapeutics Inc. Virtual Biotech Company (6) Phase 2a (48 pt) clinical trial in acute coronary syndrome (ACS) completed
Safety Measures in the new Pharmacovigilance System
 Safety Measures in the new Pharmacovigilance System Dr. Harald Tietz Director Global Patient Safety & Regulatory Affairs, Germany Lilly Deutschland GmbH Documentation and reporting requirements: Centralisation
Safety Measures in the new Pharmacovigilance System Dr. Harald Tietz Director Global Patient Safety & Regulatory Affairs, Germany Lilly Deutschland GmbH Documentation and reporting requirements: Centralisation
EAUTIFUL BDATA AGGREGATION EXTRAVAGANZA. Implementing a Clinical Data Repository and Analytics Platform in 90 Days
 DATA IS SIMPLY EAUTIFUL BDATA AGGREGATION EXTRAVAGANZA This artful imagery is an abstract representation of data coming from a myriad of sources to create a single, impactful image. Created by data artist,
DATA IS SIMPLY EAUTIFUL BDATA AGGREGATION EXTRAVAGANZA This artful imagery is an abstract representation of data coming from a myriad of sources to create a single, impactful image. Created by data artist,
Introduction to Clinical Research
 Introduction to Clinical Research What is Clinical Research? Clinical research is medical research that involves people like you. People volunteer to participate in carefully conducted investigations that
Introduction to Clinical Research What is Clinical Research? Clinical research is medical research that involves people like you. People volunteer to participate in carefully conducted investigations that
HITSP Construct HL7 Standard
 EHR Lab Results Reporting HL7 /IS01 Clinical Document Architecture Release 2 (CDA R2) /C37 - Lab Report Document U.S. Realm - Interoperability Specification: Lab Result to EHR (ORU^R01) (HL7.1) September,
EHR Lab Results Reporting HL7 /IS01 Clinical Document Architecture Release 2 (CDA R2) /C37 - Lab Report Document U.S. Realm - Interoperability Specification: Lab Result to EHR (ORU^R01) (HL7.1) September,
Life Sciences Regulatory Publishing Software Suite
 Easily manage submissions across multiple regions Solution Suite Overview Life sciences companies today are challenged by regulatory submissions processes that vary by country and region. The DXC Technology
Easily manage submissions across multiple regions Solution Suite Overview Life sciences companies today are challenged by regulatory submissions processes that vary by country and region. The DXC Technology
CFAST Therapeutic Area Program Steering Committee 2013 Meeting Summaries (January-August)
 CFAST Therapeutic Area Program Steering Committee 2013 Meeting Summaries (January-August) Participant full names and titles: Wayne: Wayne Kubick, Chief Technology Officer, CDISC (Chair) Becky: Rebecca
CFAST Therapeutic Area Program Steering Committee 2013 Meeting Summaries (January-August) Participant full names and titles: Wayne: Wayne Kubick, Chief Technology Officer, CDISC (Chair) Becky: Rebecca
EU Portal and Database Update
 EU Portal and Database Update TOPRA 4 October 2017 Presented by Noémie Manent on 4 October 2017 Committees and Inspections Department An agency of the European Union Table of content EMA Portal and Database
EU Portal and Database Update TOPRA 4 October 2017 Presented by Noémie Manent on 4 October 2017 Committees and Inspections Department An agency of the European Union Table of content EMA Portal and Database
The SDTMIG: What s New in 3.2
 Accenture Life Sciences Rethink Reshape Restructure for better health outcomes. The SDTMIG: What s New in 3.2 Fred Wood Data Standards Consulting Lead Accenture Life Sciences RTP CDISC User Network March
Accenture Life Sciences Rethink Reshape Restructure for better health outcomes. The SDTMIG: What s New in 3.2 Fred Wood Data Standards Consulting Lead Accenture Life Sciences RTP CDISC User Network March
Building (Earthquake-prone buildings) Amendment Bill Interim Report
 < Local Councils play an active role in keeping our communities safe.> Building (Earthquake-prone buildings) Amendment Bill Interim Report Local Government New Zealand s submission to Local Government
< Local Councils play an active role in keeping our communities safe.> Building (Earthquake-prone buildings) Amendment Bill Interim Report Local Government New Zealand s submission to Local Government
Welcome to the American College of Toxicology s Webinar Series. We will begin at 11AM EDT
 Welcome to the American College of Toxicology s Webinar Series We will begin at 11AM EDT Preparing for Nonclinical edata Regulatory Submissions to the US FDA SEND and Beyond September 5, 2013, 11:00 AM
Welcome to the American College of Toxicology s Webinar Series We will begin at 11AM EDT Preparing for Nonclinical edata Regulatory Submissions to the US FDA SEND and Beyond September 5, 2013, 11:00 AM
Job Evaluation Policy
 Job Evaluation Policy (Revised 1 June 2013) 1. Aims The University uses a single job evaluation system for all categories of job, in order to: - Establish the relative size of all jobs. - Enable all jobs
Job Evaluation Policy (Revised 1 June 2013) 1. Aims The University uses a single job evaluation system for all categories of job, in order to: - Establish the relative size of all jobs. - Enable all jobs
PK-PD Concepts and Programming Bangalore, India. 6th December India SDE
 PK-PD Concepts and Programming Bangalore, India 6th December 2014 India SDE 2014 1 Welcome Dear Single Day Event Attendee, We, on behalf of the organising committee would like to welcome you all to this
PK-PD Concepts and Programming Bangalore, India 6th December 2014 India SDE 2014 1 Welcome Dear Single Day Event Attendee, We, on behalf of the organising committee would like to welcome you all to this
Specific pathologist responses for Standard for Exchange of Nonclinical Data (SEND)
 J Toxicol Pathol 2017; 30: 201 207 Review Specific pathologist responses for Standard for Exchange of Nonclinical Data (SEND) Atsushi Watanabe 1, 2*, Osamu Kusuoka 1, Norihiro Sato 1, Osamu Nakazono 1,
J Toxicol Pathol 2017; 30: 201 207 Review Specific pathologist responses for Standard for Exchange of Nonclinical Data (SEND) Atsushi Watanabe 1, 2*, Osamu Kusuoka 1, Norihiro Sato 1, Osamu Nakazono 1,
Procurement procedure decision trees
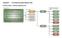 Procurement procedure decision trees Procurement procedure 1 Infrastructure: A staged is typically appropriate when: methods for a staged delivery model Scale is small to medium. Complexity, uncertainty
Procurement procedure decision trees Procurement procedure 1 Infrastructure: A staged is typically appropriate when: methods for a staged delivery model Scale is small to medium. Complexity, uncertainty
CMC Strategy Forum Prague Enda Moran EBE Satellite Session, 06 May 2013
 CMC Strategy Forum Prague 2013 Enda Moran EBE Satellite Session, 06 May 2013 European Biopharmaceutical Enterprises EBE s VISION: fostering innovation, and promote favorable business and regulatory conditions
CMC Strategy Forum Prague 2013 Enda Moran EBE Satellite Session, 06 May 2013 European Biopharmaceutical Enterprises EBE s VISION: fostering innovation, and promote favorable business and regulatory conditions
PharmaSUG PO14
 PharmaSUG 2014 - PO14 Route to SDTM Implementation in In-Vitro Diagnostic Industry: Simple or Twisted Carey Smoak, Roche Molecular Systems, Inc., Pleasanton, CA Sofia Shamas, MaxisIT Inc., NJ Chowdagam
PharmaSUG 2014 - PO14 Route to SDTM Implementation in In-Vitro Diagnostic Industry: Simple or Twisted Carey Smoak, Roche Molecular Systems, Inc., Pleasanton, CA Sofia Shamas, MaxisIT Inc., NJ Chowdagam
Bury Local Care Organisation Provider Alliance
 Job Description Post: Project Manager Band: 6 Location/Base: Responsible to: Main Contacts: Bury Town Centre Senior Programme Manager Bury Local Care Organisation Provider Alliance Job Summary The Project
Job Description Post: Project Manager Band: 6 Location/Base: Responsible to: Main Contacts: Bury Town Centre Senior Programme Manager Bury Local Care Organisation Provider Alliance Job Summary The Project
The European Medicines Agency Inspections ANNEX IV TO PROCEDURE FOR CONDUCTING GCP INSPECTIONS REQUESTED BY THE EMEA:
 The European Medicines Agency Inspections London, 20 September 2007 EMEA/INS/GCP/197221/2005 Procedure no.: INS/GCP/3/IV ANNEX IV TO PROCEDURE FOR CONDUCTING GCP INSPECTIONS REQUESTED BY THE EMEA: SPONSOR
The European Medicines Agency Inspections London, 20 September 2007 EMEA/INS/GCP/197221/2005 Procedure no.: INS/GCP/3/IV ANNEX IV TO PROCEDURE FOR CONDUCTING GCP INSPECTIONS REQUESTED BY THE EMEA: SPONSOR
MSCI 689: Career Development for the Biomedical PhD Scientist Introduction to Medical Writing and Drug Development
 MSCI 689: Career Development for the Biomedical PhD Scientist Introduction to Medical Writing and Drug Development Danielle K Lewis, PhD April 28, 2017 One-Stop Shop Regulatory Submissions Regulatory Operations
MSCI 689: Career Development for the Biomedical PhD Scientist Introduction to Medical Writing and Drug Development Danielle K Lewis, PhD April 28, 2017 One-Stop Shop Regulatory Submissions Regulatory Operations
Analyzing which software needs to be utilized to ensure a timely and more automated process
 Clinical Data Integration & Management March 20 th Program Day One 08:15 Registration and refreshments 08:50 Chair s opening remarks Analyzing which software needs to be utilized to ensure a timely and
Clinical Data Integration & Management March 20 th Program Day One 08:15 Registration and refreshments 08:50 Chair s opening remarks Analyzing which software needs to be utilized to ensure a timely and
POLICY AND PROCEDURE MANUAL
 POLICY AND PROCEDURE MANUAL PBRC: POLICY NO. 378.00 ORIGIN DATE: 5/25/2016 IMPACTS: CLINICAL RESEARCH LAST REVISED: 1/1/2017 SUBJECT: SOURCE: PURPOSE PENNINGTON BIOMEDICAL CLINICALTRIALS.GOV POLICY INSTITUTIONAL
POLICY AND PROCEDURE MANUAL PBRC: POLICY NO. 378.00 ORIGIN DATE: 5/25/2016 IMPACTS: CLINICAL RESEARCH LAST REVISED: 1/1/2017 SUBJECT: SOURCE: PURPOSE PENNINGTON BIOMEDICAL CLINICALTRIALS.GOV POLICY INSTITUTIONAL
Investigating Clinical Trial Costs Comparative Analysis of Trial Cost Components in Key Geographies Table of Contents
 Investigating Clinical Trial Costs Comparative Analysis of Trial Cost Components in Key Geographies Table of Contents Executive summary 14 Introduction and background to clinical trials 14 Financial considerations
Investigating Clinical Trial Costs Comparative Analysis of Trial Cost Components in Key Geographies Table of Contents Executive summary 14 Introduction and background to clinical trials 14 Financial considerations
COMPUTER SYSTEMS VALIDATION
 COMPUTER SYSTEMS VALIDATION DOCUMENT NO.: POL007 v1.0 AUTHOR: Lorn Mackenzie ISSUE DATE: 04 AUG 2017 1 INTRODUCTION 1.1 The Academic & Clinical Central Office for Research & Development (ACCORD) is a joint
COMPUTER SYSTEMS VALIDATION DOCUMENT NO.: POL007 v1.0 AUTHOR: Lorn Mackenzie ISSUE DATE: 04 AUG 2017 1 INTRODUCTION 1.1 The Academic & Clinical Central Office for Research & Development (ACCORD) is a joint
Validation of Participants
 Validation of Participants Clean Sky 2014 Negotiation Kick-off Info Day Pamela Gorini Henk Van Belle REA - Unit C3 Participant Validation and Support 19th June 2014 Summary Part 1 How to obtain a PIC Part
Validation of Participants Clean Sky 2014 Negotiation Kick-off Info Day Pamela Gorini Henk Van Belle REA - Unit C3 Participant Validation and Support 19th June 2014 Summary Part 1 How to obtain a PIC Part
Quality Assurance of Investigational Medicinal Products. Sue McKenzie PPQA (A)
 Quality Assurance of Investigational Medicinal Products Sue McKenzie PPQA (A) Agenda for Today IMP Regulations - CTD and Annex 13 Role of QP for IMP Challenges of IMP Manufacture for the QP IMP Regulations
Quality Assurance of Investigational Medicinal Products Sue McKenzie PPQA (A) Agenda for Today IMP Regulations - CTD and Annex 13 Role of QP for IMP Challenges of IMP Manufacture for the QP IMP Regulations
ICH Topic E16 Genomic Biomarkers Related to Drug Response: Context, Structure and Format of Qualification Submissions. Step 3
 European Medicines Agency June 2009 EMEA/CHMP/ICH/380636/2009 ICH Topic E16 Genomic Biomarkers Related to Drug Response: Context, Structure and Format of Qualification Submissions Step 3 NOTE FOR GUIDANCE
European Medicines Agency June 2009 EMEA/CHMP/ICH/380636/2009 ICH Topic E16 Genomic Biomarkers Related to Drug Response: Context, Structure and Format of Qualification Submissions Step 3 NOTE FOR GUIDANCE
arena that impact on clinical development
 RESEARCH DEVELOPMENT Changes in the regulatory arena that impact on clinical development 23 May 2011 Agenda Voluntary Harmonisation Procedure PIP and Paediatric requirements Changes to EudraCT and European
RESEARCH DEVELOPMENT Changes in the regulatory arena that impact on clinical development 23 May 2011 Agenda Voluntary Harmonisation Procedure PIP and Paediatric requirements Changes to EudraCT and European
8/2/2017. Key attributes of scientific excellence: rigor, innovation, and relevance. Medical Physics 3.0. Key Attributes of Scientific Excellence
 Medical Physics 3.0 in Design Key attributes of scientific excellence: rigor, innovation, and relevance Maryellen Giger, Ph.D. A. N. Pritzker Professor of Radiology / Medical Physics The University of
Medical Physics 3.0 in Design Key attributes of scientific excellence: rigor, innovation, and relevance Maryellen Giger, Ph.D. A. N. Pritzker Professor of Radiology / Medical Physics The University of
Quality Assurance QA STANDARD OPERATING PROCEDURE FOR FDA or Pharmaceutical Sponsored Audits
 Quality Assurance QA 601.01 STANDARD OPERATING PROCEDURE FOR FDA or Pharmaceutical Sponsored Audits Approval: Nancy Paris, MS, FACHE President and CEO 24 May 2017 (Signature and Date) Approval: Frederick
Quality Assurance QA 601.01 STANDARD OPERATING PROCEDURE FOR FDA or Pharmaceutical Sponsored Audits Approval: Nancy Paris, MS, FACHE President and CEO 24 May 2017 (Signature and Date) Approval: Frederick
Author Signature: Date: 10 October 2017 The author is signing to confirm the technical content of this document
 SOP Title: Investigator s Brochure Content, Design, Amendments, Filing & Distribution Author: Clinical Research and Development Office (CRDO) Author Signature: Date: 10 October 2017 The author is signing
SOP Title: Investigator s Brochure Content, Design, Amendments, Filing & Distribution Author: Clinical Research and Development Office (CRDO) Author Signature: Date: 10 October 2017 The author is signing
University of Virginia Cancer Center
 University of Virginia Cancer Center Data and Safety Monitoring Plan for Clinical Research March 18, 2016 Approved: National Cancer Institute April 14 th, 2016 Table of Contents 1. Introduction... 3 2.
University of Virginia Cancer Center Data and Safety Monitoring Plan for Clinical Research March 18, 2016 Approved: National Cancer Institute April 14 th, 2016 Table of Contents 1. Introduction... 3 2.
Impact of the transposition of the European Clinical Trials Directive. CEMO, Paris 17 November 2004
 Impact of the transposition of the European Clinical Trials Directive CEMO, Paris 17 November 2004 Dr Martine Dehlinger-Kremer VP Regulatory Affairs International Agenda Overview of key areas of Directive
Impact of the transposition of the European Clinical Trials Directive CEMO, Paris 17 November 2004 Dr Martine Dehlinger-Kremer VP Regulatory Affairs International Agenda Overview of key areas of Directive
OVERVIEW OF DRUG REGISTRATION REQUIREMENTS FOR PHARMACEUTICALS IN EMERGING MARKET
 Badjatya et al Journal of Drug Delivery & Therapeutics; 2013, 3(2), 227-232 227 Available online at http://jddtonline.info REVIEW ARTICLE OVERVIEW OF DRUG REGISTRATION REQUIREMENTS FOR PHARMACEUTICALS
Badjatya et al Journal of Drug Delivery & Therapeutics; 2013, 3(2), 227-232 227 Available online at http://jddtonline.info REVIEW ARTICLE OVERVIEW OF DRUG REGISTRATION REQUIREMENTS FOR PHARMACEUTICALS
STANDING COMMITTEE ON FINANCE AND ECONOMIC AFFAIRS. Consideration Of Bill 148 (Fair Workplaces, Better Jobs Act, 2017)
 STANDING COMMITTEE ON FINANCE AND ECONOMIC AFFAIRS Consideration Of Bill 148 (Fair Workplaces, Better Jobs Act, 2017) Submissions of the Association of Canadian Search, Employment & Staffing Services (ACSESS)
STANDING COMMITTEE ON FINANCE AND ECONOMIC AFFAIRS Consideration Of Bill 148 (Fair Workplaces, Better Jobs Act, 2017) Submissions of the Association of Canadian Search, Employment & Staffing Services (ACSESS)
The EFGCP Report on The Procedure for the Ethical Review of Protocols for Clinical Research Projects in Europe (Update: April 2011) Bulgaria
 The Procedure for the Ethical Review of Protocols for Clinical Research Projects in Europe (Update: April 2011) Bulgaria Question 1: What laws or regulations apply to an application for conducting a clinical
The Procedure for the Ethical Review of Protocols for Clinical Research Projects in Europe (Update: April 2011) Bulgaria Question 1: What laws or regulations apply to an application for conducting a clinical
Risk-Based Monitoring Update Volume IV
 Risk-Based Monitoring Update Volume IV 1. Introduction TransCelerate s Risk-Based Monitoring (RBM) initiative continues to produce tangible outputs during the first half of 2015. The purpose of RBM Update
Risk-Based Monitoring Update Volume IV 1. Introduction TransCelerate s Risk-Based Monitoring (RBM) initiative continues to produce tangible outputs during the first half of 2015. The purpose of RBM Update
Good Clinical Practice (GCP) & Clinical Trial Registries
 Good Clinical Practice (GCP) & Clinical Trial Registries The Fifth Annual Pharmaceutical Regulatory and Compliance Congress and Best Practice Forum November 14-17, 2004 Kate Maloney, RN, MS, CPHQ Manager,
Good Clinical Practice (GCP) & Clinical Trial Registries The Fifth Annual Pharmaceutical Regulatory and Compliance Congress and Best Practice Forum November 14-17, 2004 Kate Maloney, RN, MS, CPHQ Manager,
A D D E N D U M # Clarification: General QUESTIONS RECEIVED REGARDING TECHNICAL INTERFACE WITH Versaterm CAD AND COUNTY RESPONSES
 A D D E N D U M # 7 October 17, 2017 Address all questions to: Kathi Braeme-Burr, Sr. Procurement Analyst Multnomah County Purchasing 501 SE Hawthorne Blvd. Portland, OR 97214 503-988-7550 E-mail: Kathi.braeme-burr@multco.us
A D D E N D U M # 7 October 17, 2017 Address all questions to: Kathi Braeme-Burr, Sr. Procurement Analyst Multnomah County Purchasing 501 SE Hawthorne Blvd. Portland, OR 97214 503-988-7550 E-mail: Kathi.braeme-burr@multco.us
Designing a Disease-Specific Master Protocol
 Designing a Disease-Specific Master Protocol Lisa M. LaVange, PhD Director, Office of Biostatistics OTS/CDER/FDA Pediatric Master Protocols Workshop September 23, 2016 FDA, White Oak Campus Acknowledgments
Designing a Disease-Specific Master Protocol Lisa M. LaVange, PhD Director, Office of Biostatistics OTS/CDER/FDA Pediatric Master Protocols Workshop September 23, 2016 FDA, White Oak Campus Acknowledgments
2016 CRO LEADERSHIP AWARDS. Page Life Science Leader s readership of pharmaceutical and biopharmaceutical executives have told us about
 List Of Winners Page 28-34 Company Profiles Page 35-42 Life Science Leader s readership of pharmaceutical and biopharmaceutical executives have told us about their struggles in efficiently vetting potential
List Of Winners Page 28-34 Company Profiles Page 35-42 Life Science Leader s readership of pharmaceutical and biopharmaceutical executives have told us about their struggles in efficiently vetting potential
10:00 Examining the Challenges with Sourcing the Right CRO and Vendors for Global Trials
 Outsourcing in Clinical Trials East Asia 6 th December 2017 Program Day One 08:30 Registration and Refreshments 08:50 Chair s Opening Speech 09:00 The Key Elements to Success: Learning about the Essential
Outsourcing in Clinical Trials East Asia 6 th December 2017 Program Day One 08:30 Registration and Refreshments 08:50 Chair s Opening Speech 09:00 The Key Elements to Success: Learning about the Essential
@TandFOpen. How are subscription publishers making the transition to open access ( OA)?
 @TandFOpen How are subscription publishers making the transition to open access ( OA)? V Gardner, Open Access Publisher, Taylor & Francis / Routledge UKSG Nov 2013 screen 2 Overview The third age Transition
@TandFOpen How are subscription publishers making the transition to open access ( OA)? V Gardner, Open Access Publisher, Taylor & Francis / Routledge UKSG Nov 2013 screen 2 Overview The third age Transition
The University of Texas MD Anderson Cancer Center UTHealth Graduate School of Biomedical Sciences Catalog Addendum
 The University of Texas MD Anderson Cancer Center UTHealth 2016-2018 Catalog Addendum GSBS 2016-18 Catalog Addendum Table of Contents School Name Change... 1 Areas of Research Concentration Changes...
The University of Texas MD Anderson Cancer Center UTHealth 2016-2018 Catalog Addendum GSBS 2016-18 Catalog Addendum Table of Contents School Name Change... 1 Areas of Research Concentration Changes...
5 th Training Workshop for Micro-, Small- and Medium- Sized Enterprises (SMEs)
 5 th Training Workshop for Micro-, Small- and Medium- Sized Enterprises (SMEs) GCP Requirements and Compliance Dr. Katalina Mettke BfArM, Germany Agenda Specialities of Early Development Trials Examples
5 th Training Workshop for Micro-, Small- and Medium- Sized Enterprises (SMEs) GCP Requirements and Compliance Dr. Katalina Mettke BfArM, Germany Agenda Specialities of Early Development Trials Examples
International Transfers of Personal Data at sanofi-aventis R & D
 International Transfers of Personal Data at sanofi-aventis R & D Pierre-Yves Lastic, PhD Senior Director, Standards Management & Data Privacy Sanofi-aventis R&D CONFERENCE ON INTERNATIONAL TRANSFERS OF
International Transfers of Personal Data at sanofi-aventis R & D Pierre-Yves Lastic, PhD Senior Director, Standards Management & Data Privacy Sanofi-aventis R&D CONFERENCE ON INTERNATIONAL TRANSFERS OF
Sponsorship of Clinical Research Studies
 Sponsorship of Clinical Research Studies Category: Summary: Equality Impact Assessment undertaken: Policy The UK Policy Framework for Health and Social Care 2017 (UKPF) and The Medicines for Human Use
Sponsorship of Clinical Research Studies Category: Summary: Equality Impact Assessment undertaken: Policy The UK Policy Framework for Health and Social Care 2017 (UKPF) and The Medicines for Human Use
CDISC Standards in the Regulatory Submission Process
 CDISC 2010 CDISC Standards in the Regulatory Submission Process What s New and What s Ahead 26 January 2012 Moderator Frank Newby, CDISC COO CDISC 2012 2011 Webinar Agenda SDTM Amendment 1 Wayne Kubick,
CDISC 2010 CDISC Standards in the Regulatory Submission Process What s New and What s Ahead 26 January 2012 Moderator Frank Newby, CDISC COO CDISC 2012 2011 Webinar Agenda SDTM Amendment 1 Wayne Kubick,
ICH Q3D Guideline Impact on the Users: Perspective of a Finished Product Manufacturer John Glennon
 ICH Q3D Guideline Impact on the Users: Perspective of a Finished Product Manufacturer John Glennon 9 November 2016 Disclaimer The views and opinions expressed in this presentation are those of the author
ICH Q3D Guideline Impact on the Users: Perspective of a Finished Product Manufacturer John Glennon 9 November 2016 Disclaimer The views and opinions expressed in this presentation are those of the author
Introduction to CMC Regulatory Affairs
 Introduction to CMC Regulatory Affairs Bharathi Mamidipudi Regulatory Affairs Consultant II Syner-G Pharma Consulting, LLC Northeastern University, Boston November 10, 2016 My Background Experience ~4
Introduction to CMC Regulatory Affairs Bharathi Mamidipudi Regulatory Affairs Consultant II Syner-G Pharma Consulting, LLC Northeastern University, Boston November 10, 2016 My Background Experience ~4
Report on recognising organisations as awarding bodies
 Report on recognising organisations as awarding bodies The recognition process February 2008 QCA/08/3569 Contents The recognition process... 3 Background... 3 Current recognition process... 3 Reducing
Report on recognising organisations as awarding bodies The recognition process February 2008 QCA/08/3569 Contents The recognition process... 3 Background... 3 Current recognition process... 3 Reducing
What is ISO 30300? Who, when, where, why and how to implement
 What is ISO 30300? Who, when, where, why and how to implement Barcelona, October 28th 2011 Carlota Bustelo Judith Ellis Index 1. What is ISO 30300: MSR? a) Background of MSR initiative b) What is a MSR?
What is ISO 30300? Who, when, where, why and how to implement Barcelona, October 28th 2011 Carlota Bustelo Judith Ellis Index 1. What is ISO 30300: MSR? a) Background of MSR initiative b) What is a MSR?
Case Studies in Capacity Acquisition:
 Case Studies in Capacity Acquisition: institutional strategies for sourcing Research Data Management (RDM) services Constance Malpas, OCLC Research Research Data and the Evolving Scholarly Record Growing
Case Studies in Capacity Acquisition: institutional strategies for sourcing Research Data Management (RDM) services Constance Malpas, OCLC Research Research Data and the Evolving Scholarly Record Growing
Non-clinical documentation Overview of Requirements
 3 rd EMEA-. Non-clinical Aspects Non-clinical documentation Overview of Requirements EMEA Pre-Authorisation Human Unit 3 rd EMEA-. Non-clinical Aspects Outline Overview of Legal and Regulatory requirements
3 rd EMEA-. Non-clinical Aspects Non-clinical documentation Overview of Requirements EMEA Pre-Authorisation Human Unit 3 rd EMEA-. Non-clinical Aspects Outline Overview of Legal and Regulatory requirements
Asia-Pacific Conference on Transformative Agenda
 Asia-Pacific Conference on Transformative Agenda Group B Statistics Denmark- set-up 24-26 August 2016, Bangkok Vision We envisage that in 2020 Statistics Denmark is transformed into a generator of data
Asia-Pacific Conference on Transformative Agenda Group B Statistics Denmark- set-up 24-26 August 2016, Bangkok Vision We envisage that in 2020 Statistics Denmark is transformed into a generator of data
Global Contract Research Organization (CRO) Market: Size, Trends & Forecasts ( ) November 2017
 Global Contract Research Organization (CRO) Market: Size, Trends & Forecasts (2017-2021) November 2017 Global Contract Research Organization (CRO) Market : Coverage Executive Summary and Scope Introduction/Market
Global Contract Research Organization (CRO) Market: Size, Trends & Forecasts (2017-2021) November 2017 Global Contract Research Organization (CRO) Market : Coverage Executive Summary and Scope Introduction/Market
REGULATORY REQUIREMENTS FOR CLINICAL TRIAL APPROVAL MEDICAL DEVICES DR D DIALE MRS P NKAMBULE. 02 December 2015
 REGULATORY REQUIREMENTS FOR CLINICAL TRIAL APPROVAL MEDICAL DEVICES DR D DIALE MRS P NKAMBULE 02 December 2015 LAYOUT Background to clinical trials Stakeholders in clinical trials Legislative requirements
REGULATORY REQUIREMENTS FOR CLINICAL TRIAL APPROVAL MEDICAL DEVICES DR D DIALE MRS P NKAMBULE 02 December 2015 LAYOUT Background to clinical trials Stakeholders in clinical trials Legislative requirements
COMMUNITY SERVICE WORKER 1 COMMUNITY SERVICE WORKER 2
 B001 B002 Established 01-30-91 COMMUNITY SERVICE WORKER 1 COMMUNITY SERVICE WORKER 2 DEFINITION To provide a variety of services to the community and to assist departments in accomplishing their missions;
B001 B002 Established 01-30-91 COMMUNITY SERVICE WORKER 1 COMMUNITY SERVICE WORKER 2 DEFINITION To provide a variety of services to the community and to assist departments in accomplishing their missions;
-Regulation (EC) No.1394/2007 -Regulation (EC) No. 668/2009
 Introduction to Advanced Therapy Medicinal Products Regulation -Regulation (EC) No.1394/2007 -Regulation (EC) No. 668/2009 -Directive 2009/120/EC Dr. Maura O Donovan F.R.C.O.G. MA MD M.R.C.P.I. CAT member
Introduction to Advanced Therapy Medicinal Products Regulation -Regulation (EC) No.1394/2007 -Regulation (EC) No. 668/2009 -Directive 2009/120/EC Dr. Maura O Donovan F.R.C.O.G. MA MD M.R.C.P.I. CAT member
POSITION DESCRIPTION SENIOR ENGINEER
 POSITION DESCRIPTION SENIOR ENGINEER Position Title: Classification: Department: Position Type: Organisational Context: Senior Engineer SGW Enterprise Agreement - SEO Infrastructure and Planning Team Full
POSITION DESCRIPTION SENIOR ENGINEER Position Title: Classification: Department: Position Type: Organisational Context: Senior Engineer SGW Enterprise Agreement - SEO Infrastructure and Planning Team Full
EU scientific regulatory support mechanisms and initiatives for innovation in drug development: the EMA perspective
 EU scientific regulatory support mechanisms and initiatives for innovation in drug development: the EMA perspective FAMHP Workshop, Brussels, 2 nd May 2016 Presented by Zahra Hanaizi Product Development
EU scientific regulatory support mechanisms and initiatives for innovation in drug development: the EMA perspective FAMHP Workshop, Brussels, 2 nd May 2016 Presented by Zahra Hanaizi Product Development
Health research and innovation services
 Health research and innovation services Health research and innovation services ALIDANS PROVIDES SERVICES AND PRODUCTS FOR RESEARCH AND INNOVATION (R&I) IN THE HEALTH FIELD, THANKS TO A WORLDWIDE NETWORK
Health research and innovation services Health research and innovation services ALIDANS PROVIDES SERVICES AND PRODUCTS FOR RESEARCH AND INNOVATION (R&I) IN THE HEALTH FIELD, THANKS TO A WORLDWIDE NETWORK
Business and Management. Case Study for Paper 1. Reach Out
 Business and Management Case Study for Paper 1 2011 Reach Out 1 Reach Out Task Book. Reach out is your Case study for paper 1. To do well you need to be so familiar with the Case Study that you do not
Business and Management Case Study for Paper 1 2011 Reach Out 1 Reach Out Task Book. Reach out is your Case study for paper 1. To do well you need to be so familiar with the Case Study that you do not
Comparison of admission rates to neonatal units between PO screening and non-po screening Units
 London - Westminster Research Ethics Committee 4 Minshull Street Manchester M1 3DZ Telephone: 0207 104 8012 Please note: This is the favourable opinion of the REC only and does not allow you to start your
London - Westminster Research Ethics Committee 4 Minshull Street Manchester M1 3DZ Telephone: 0207 104 8012 Please note: This is the favourable opinion of the REC only and does not allow you to start your
Expanded Access. to Investigational Drugs & Biologics. for Treatment Use
 SJMHS Research Compliance Office Guidance Document Expanded Access to Investigational Drugs & Biologics for Treatment Use May 2015 1 Expanded Access to Investigational Drugs & Biologics for Treatment Use
SJMHS Research Compliance Office Guidance Document Expanded Access to Investigational Drugs & Biologics for Treatment Use May 2015 1 Expanded Access to Investigational Drugs & Biologics for Treatment Use
Pre-registration Pharmacist National Recruitment Scheme via Oriel Employer Guidance Handbook 2018
 Pre-registration Pharmacist National Recruitment Scheme via Oriel Employer Guidance Handbook 2018 Contents 1. Introduction... 3 1.1 Benefits of recruiting nationally... 3 Diagram 1 Pre-registration Pharmacist
Pre-registration Pharmacist National Recruitment Scheme via Oriel Employer Guidance Handbook 2018 Contents 1. Introduction... 3 1.1 Benefits of recruiting nationally... 3 Diagram 1 Pre-registration Pharmacist
