PROGRAM FOR THE MANAGEMENT AND DISPOSAL OF RADIOACTIVE WASTE
|
|
|
- Olivia Roberts
- 6 years ago
- Views:
Transcription
1 RADIATION SAFETY DEPARTMENT West Virginia University Health Sciences Center WVU Hospitals Jefferson Memorial Hospital G-139 Health Sciences Center PO Box 9006 Morgantown, WV Phone: Fax: PROGRAM FOR THE MANAGEMENT AND DISPOSAL OF RADIOACTIVE WASTE 1. GENERAL CONSIDERATIONS (For the purpose of this policy the Radiation Safety Department will be here and throughout this document referred to as RSD and Authorized Radiation User is referred to as ARU/PI.) West Virginia University developed these procedures to ensure the safe and proper handling, labeling and packaging of radioactive waste and to continually be in compliance with all federal, state and local regulatory requirements and the Radiation Safety Manual (RSM). Appropriate waste disposal records MUST be maintained at all times. Radioactive waste MUST be stored in appropriate containers. Radioactive waste MUST be appropriately labeled. Integrity of the waste containers MUST be assured. Radioactive waste MUST be secured against unauthorized access or removal. The disposition of all forms of radioactive waste is the responsibility of the Radiation Safety Department office. All radioactive waste is picked up by the Radiation Safety Department and processed for final disposition. Radioactive waste must never be disposed of in regular trash. Properly labeled radioactive waste container should be readily available in labs utilizing radioactive materials. The Radiation Safety Department provides both liquid and solid waste containers to use in the work area for radioactive waste disposal. Radiation users should complete a RSD Radioactive Waste Removal Request form to request containers or waste removal from their lab. It is important that this Program be strictly adhered to by all radiation users. Not only for regulatory compliance and safety, but to effectively minimize the volume of waste generated to reduce the financial burden emanating from waste disposal. Changes in regulatory guidelines and escalating decommissioning costs have forced West Virginia University administration to adopt a new criterion that would amend the financial assurance requirements in the areas of decontamination, decommissioning and radioactive material waste disposal under the institutions radioactive materials broad scope license (WVU Exp ) and WVRHP materials license. Current authorized radiation users (ARU/PI), and new applicants requesting non-human use of radiation and radionuclides materials authorization, are required to provide a written commitment to WVU that ascertains funding is available for all elements of radioactive material final disposition under their Radiation Safety Committee application approval. The ARU/PI s department head must also ratify this commitment whereby indicating any costs not covered by the ARU/PI s research grant will be the financial responsibility of the stated department. This is a straight forward way to ensure adequate funding is available for timely radioactive waste removal and decommissioning. Page 1 of 7
2 In addition, all radioactive waste disposal costs are to be considered and added as a direct charge on grants and contracts when financial support is sought for the purchase of radioactive materials. The fee schedule for waste processing and disposal will be reviewed annually by RSD. Fees are derived using a number of variables that includes a fee schedule provided by the contracted vendor for radioactive waste removal. Each ARU/PI is financially responsible for the procurement and disposition of all radioactive materials they obtain under their Non-Human Use of Radiation & Radionuclide Committee authorization. All waste processing, supplies and disposal costs will be direct billed to the ARU/PI quarterly and payment must be submitted to the Radiation Safety Department within 30 days of invoice receipt. II. IMPORTANT RULES TO REMEMBER 1. Do not allow waste containers to overflow. If you will be conducting an experiment on a particular date that will generate an abundance of waste, notify RSD in advance so that arrangements can be made for immediate removal by completing a RSD Radioactive Waste Removal Request form (see appendices). 2. Radioactive waste should not be held for decay purposes in any laboratory. 3. All radioactive waste must be properly labeled with activity, isotope, compound, ph level; date, ARU/PI and clearly visible label bearing the appropriate radiation symbol (see appendices). 4. The exterior of all containers must be free of removable contamination. 5. RSD will not remove waste from a laboratory that has not been properly processed and prepared for pickup. 6. RSD does not permit laboratories to dispose of radioactive materials through sanitary sewer. 7. Fax a copy of the Radioactive Material Inventory and Waste Disposal Record form (see appendices) and RSD Radioactive Waste Removal Request to RSD for a waste pickup. 8. The Radioactive Material Inventory and Waste Disposal Record form should always include a written record of each time radioactive solution is removed from the vial and used or transferred to a designated radioactive waste container. 9. Maintain an accurate account of all waste placed in waste containers. The completed Radioactive Waste Composition Record (see appendices) should always be attached to the outside of the waste container in clear view. 10. All contaminated materials must be discarded as radioactive waste if it cannot be adequately decontaminated. 11. Prior to removal or disposal of empty contamination free containers to unrestricted areas or regular trash, deface the radioactive material label or clearly indicate on the container that it no longer contains radioactive materials. 12. Radioactive waste should be properly secured at all times. 13. Waste barrels should not contain any detectable free standing liquids. 14. Radioactive waste may not contain pyrophoric materials or materials that react violently with water. 15. The physical form of all waste must be specified, e.g., compressed paper, glass, etc. 16. Biological (excluding animal carcasses), pathogenic or infectious material or equipment (e.g., syringes, test tubes, capillary tubes) used to handle such material shall be treated so that the material, if not radioactive, can be disposed of as infectious waste. 17. The radionuclide and activity must be specified in microcuries or millicuries. The radionuclide and activity must coincide with the barrel sheet. Page 2 of 7
3 III. SUGGESTIONS ON HOW TO MINIMIZE WASTE 1. Use radioisotopes with a half-life of less than 90 days whenever possible. 2. Order only the quantity of radionuclides required for the experiment. 3. Do not discard non-contaminated trash into radioactive waste containers. 4. Survey potentially contaminated items (e.g. disposable gloves, trays, absorbent papers) in the work areas. If there is no contamination, they can be disposed of in regular trash after completely removing any radiation warning symbols and/or markings. Prior to discarding, double check to ensure that all radiation labels are properly defaced. IV. WASTE DISPOSAL For disposal purposes, radioactive wastes should be segregated into 5 categories: A. Dry Solid Radioactive Waste B. Solid Biological Radioactive Waste C. Liquid Radioactive Waste D. Liquid Scintillation Vials E. Radioactive/Hazardous Mixed Waste (After radioactive waste is categorized you should segregate waste according to the half-life of the radionuclide and then by isotope.) Note: Short half-life waste is not to be mixed with long half-life waste. When short half-life waste is mixed with long half-life waste the entire container of waste must be processed as long half-life and shipped accordingly. (Short half-life waste is radionuclides with a half-life of 60 days or less including Sulfur-35.) DISPOSAL OF DRY SOLID RADIOACTIVE WASTE (NON-BIOLOGICAL) 1. Obtain a 5 gallon pail containing a yellow plastic bag liner from RSD. (Note: 55 gallon drums are available to users who anticipate generating large amounts of waste in short periods of time.) 2. Fold the top of yellow bag down over the side of the pail. 3. Needles and other sharp objects should be placed in a puncture proof container prior to being placed in the yellow plastic bag or pail. 4. Wrap broken glassware in paper towels to prevent the plastic bag from being punctured and to avoid a potential safety hazard. 5. Place only radioactive materials in the waste pail, carboy, drum, etc. Non-radioactive materials will increase the volume and thus increase waste disposal costs. 6. Waste containers surveyed for contamination with a reading greater than 2 mr/hr, at a distance of one meter from the container, should be sealed accordingly with tape whether the bag is full or not. Always seal bag according to Bagging & Sealing instructions (Section VII). 7. When the waste container and/or yellow plastic bag are approximately 90% full, close the bag, 8. RSD will arrange to pickup the waste within 2 working days. Page 3 of 7
4 DISPOSAL OF SECONDARY CONTAINERS/LEAD FROM STOCK VIAL SHIELDING 1. The plastic outer secondary containers utilized by manufacturers to hold the stock vial during shipment must be processed separately. If the pig contains no lead, the pig and stock vial can be disposed in solid waste. 2. The lead liner used for added shielding (lead pig) cannot be placed in either radioactive or regular waste for disposal. 3. The container and lead pig will be processed for disposal separate from other solid waste. 4. Lead is an ingestion hazard and disposable gloves are recommended during handling. 5. Survey the container and lead to confirm there is no contamination. 6. When contamination is found, decontaminate accordingly. 7. Place empty, contamination free containers and lead in separate container. No radioactive waste label is required. 8. When the waste container and/or yellow plastic bag are approximately 90% full, close the bag, 9. RSD will arrange to pickup the waste within 2 working days. DISPOSAL OF SHARPS 1. Includes all hypodermic needles, syringes, glass pasteur pipettes, scalpel blades, razor blades, broken glass, blood vials, etc. 2. Sharps should be placed in containers that are rigid, leak proof, puncture resistant and distinctly labeled radioactive and biohazardous. 3. Ensure that container is sealed to prevent loss of contents. 4. When the waste container and/or yellow plastic bag are approximately 90% full, close the bag, 5. RSD will arrange to pickup the waste within 2 working days. DISPOSAL OF SOLID BIOLOGICAL RADIOACTIVE WASTE 1. Radioactive biological waste is defined as radioactive contaminated: animal carcasses feces radioactive plants animal bedding tissue samples 2. Materials must be properly prepared to prevent the plastic bag from being torn and/or punctured. This may entail padding parts of the biological waste with gauze pads or other materials. 3. Radiation Safety will provide plastic bags upon receipt of a RSD Radioactive Waste Removal Request form. 4. When the waste container and/or yellow plastic bag are approximately 90% full, close the bag, 5. RSD will arrange to pickup the waste within 2 working days. Page 4 of 7
5 DISPOSAL OF LIQUID WASTE 1. Liquid waste intended for disposal must be segregated according to the chemical composition of the liquid: Aqueous liquids with no organic solvents Liquid scintillation fluids Radioactive/hazardous mixed liquid waste 2. Radiation Safety provides sturdy Nalgene carboys upon receipt of a RSD Radioactive Waste Removal Request form. 3. Fill the carboy to the fill line mark, indicated by a black line, on the container. 4. A carboy that is overfull is a hazardous during transport and will not be picked up by Radiation Safety. 5. Each carboy must be labeled "Caution, Radioactive Material." 6. The use of glass waste containers is prohibited. If you have any old materials stored in glass containers, place it in a secondary break-resistant container in a location where it will not be dropped or knocked over. 7. Prior to pick up, complete both sides of the yellow Radioactivity Precautions materials tag attached to the carboy. All information must be provided before RSD can pickup the carboy. Radionuclide Activity Date Chemical Form ph Solvent Department Authorized Radiation User/Investigator Volume 8. A carboy is provided for one time use only and will not be returned to the laboratory (Smaller carboys can be made available to the lab upon request). 9. When the content of the carboy reaches the fill line, seal the top and fax a completed RSD Radioactive Waste Removal Request form to RSS. 10. RSD will arrange to pickup the waste within 2 working days. DISPOSAL OF WASH WATER 1. Wash water is not required to be disposed of as liquid radioactive waste. This is the only instance that disposal can occur via the sewage system and the following guidelines must be followed: Designate a sink in the laboratory as a Radioactive Waste Sink and request RSD to post it accordingly. Do not use an unmarked sink for washing contaminated glassware and disposing of wash water. Rinse the sink with plenty of water after washing contaminated items. This will reduce the contamination in the sink and also further dilute the small amount of radionuclide released into the sewage system. DISPOSAL OF LIQUID SCINTILLATION VIALS 1. Liquid scintillation vials are available in glass or plastic, have a capacity of 20 ml or less for use in scintillation counting. 2. Plastic scintillation vials are recommended by RSD to reduce waste disposal costs. 3. Glass and plastic vials can be placed in the same waste container. 4. Liquid scintillation vials are required to be packed separately from other items such as gloves, etc. 5. Do not put any other waste except LS vials into a designated LS vial waste container. Page 5 of 7
6 6. In most instances, "cocktails" are utilized to produce scintillations that are counted. 7. Liquid scintillation fluid containing non-hazardous solvents should be utilized at all times. 8. Only under special written authorization from the RSO, can cocktails that contain solvents such as xylene or tolulene be used and will be classified as hazardous material waste during disposal processing. 9. Liquid scintillation fluid containing hazardous and radioactive materials will require special processing and transportation that will result in supplemental disposal costs. 10. Pails containing hazardous scintillation fluid should be opened in a hood and, if feasible, stored in a hood. 11. Place used vials into LS vial pail and record current activity. 12. When the waste container and/or yellow plastic bag are approximately 90% full, close the bag, 13. RSD will arrange to pickup the waste within 2 working days. DISPOSAL OF RADIOACTIVE/HAZARDOUS MIXED WASTE 1. Radioactive waste that contains additional hazards, (e.g., biohazard or chemical hazard) is considered mixed waste. 2. Storage and disposal must also comply with all applicable Federal, state, and local regulatory requirements. 3. The charges for mixed waste are determined on a case-by case basis dependent upon the chemical and radioisotope. 4. The disposal of mixed waste is very costly. Specialized processing and transportation is required. 5. When the waste container and/or yellow plastic bag are approximately 90% full, close the bag, 6. RSD will arrange to pickup the waste within 2 working days. DISPOSAL OF SEALED SOURCES AND RADIATION PRODUCING DEVICES 1. The ARU/PI must provide RSD written notification of the decision to dispose of a sealed source or radiation producing device. 2. RSD will make arrangements for a site visit, contamination surveys, decommissioning, and disposal. 3. RSD will investigate disposal options and costs and forward those results back to the ARU/PI. 4. The decommissioning and disposal project would then be scheduled. VII. BAGGING & SEALING INSTRUCTIONS 1. Place all radioactive materials and biological waste in YELLOW plastic bags 2. Properly pack materials to ensure contents will not puncture or tear the bag. 3. Additional bags may be required around the original bag to prevent leakage or puncture and then seal accordingly. 4. Non-radioactive biological waste should not be placed in yellow plastic bags. 5. Seal the bag by twisting the top of the bag tightly. 6. Wrap tape tightly two or three times around the twisted section. 7. Fold twisted section in half and wrap the tape two or three more turns. 8. Always re-bag contents, if a tear should occur, prior to transferring materials to the waste storage container. Page 6 of 7
7 VIII. ANIMAL WASTE INVENTORY CONTROL 1. Waste inventory control and identification will be recorded using a pre-numbered yellow 4- ply, Radionuclide Identification Form provided by RSD to the user (see appendices). 2. Complete one form for each animal or small group of animals when they are ready for disposal and pickup. 3. Using a permanent marker, transfer the pre-printed number on top of the form to the outside surface of the corresponding bag. 4. Attach one copy of the pre-numbered yellow form to the outside of the bag. 5. Always store biological waste in a freezer properly posted for biological and radioactive waste. 6. Freezing is required. 7. To request a waste pickup, complete and fax the RSD Radioactive Waste Removal Request 8. RSD will arrange to pickup the waste within 2 working days. 9. At pickup, provide RSD with the 1st and 3rd copy of the Radionuclide Identification Form for the bag labeled with the identical number to be removed and the 4th copy should be retained by the lab. IX. SUPPLIES & MATERIALS REQUIRED 1. RSD provides the ARU/PI with the required waste maintenance forms, pails, carboys, drums yellow plastic bags for a nominal fee. 2. Waste and other pertinent departmental forms can be downloaded from the RSD web site 3. All waste and related paperwork should be readily available when RSD arrives to process and remove waste. 4. It is just good practice to have all waste ready for pickup prior to faxing your request to RSD. 5. Additional radiation tags, stickers and containers are available upon request. X. WASTE STORAGE IN LABS 1. Radioactive waste should be stored away from other personnel located in the lab and counting instruments. 2. Do not store liquid waste containers on the floor unless a secondary container is used in the event of a leak or spill. 3. Do not stack waste containers. 4. Adequately secure waste at all times from unauthorized removal or access. 5. Depending on your lab setup, locked cabinets may be required to adequately secure waste materials in disposal containers. 6. Good housekeeping practices should be maintained at all times. 7. The waste storage area should be wipe tested and area surveyed monthly to eliminate the possibility of contamination. Appendices: Radioactive Waste Disposal Request Waste Composition Record (30-55 gal) Waste Composition Record (5 gal) Yellow Radioactivity Precautions String Tag Record Usage Form Prenumbered Animal Biological Waste Form Page 7 of 7
2/07. PROCEDURE for DISPOSAL of RADIOACTIVE MATERIALS
 2/07 CORNELL UNIVERSITY PROCEDURE for DISPOSAL of RADIOACTIVE MATERIALS This procedure has been developed to ensure the safety of those individuals who handle radioactive waste and to comply with University,
2/07 CORNELL UNIVERSITY PROCEDURE for DISPOSAL of RADIOACTIVE MATERIALS This procedure has been developed to ensure the safety of those individuals who handle radioactive waste and to comply with University,
By: Georgia Institute of Technology
 Date: / / Page 1 of 7 1.0 PURPOSE 2.0 SCOPE To describe the handling, storage, and disposal of radioactive waste and the documentation requirements of these activities. This procedure applies to all radioactive
Date: / / Page 1 of 7 1.0 PURPOSE 2.0 SCOPE To describe the handling, storage, and disposal of radioactive waste and the documentation requirements of these activities. This procedure applies to all radioactive
WVU IACUC SOP: Radioactive Materials Used in Animal Studies
 Institutional Animal Care and Use Committee Effective: 11/1/10 Ver.1 SOP#: 11-010 WVU IACUC WVU IACUC SOP: Radioactive Materials Used in Animal Studies 1. General requirements must be met before using
Institutional Animal Care and Use Committee Effective: 11/1/10 Ver.1 SOP#: 11-010 WVU IACUC WVU IACUC SOP: Radioactive Materials Used in Animal Studies 1. General requirements must be met before using
Laboratory Close-Out Guidelines
 Office of Laboratory Safety 2300 I Street, NW Ross Hall, Suite B- 05 Washington, DC 20037 t. 202-994- 8258 I labsafety@gwu.edu Guidelines The Principal Investigator (PI) is responsible for leaving laboratories
Office of Laboratory Safety 2300 I Street, NW Ross Hall, Suite B- 05 Washington, DC 20037 t. 202-994- 8258 I labsafety@gwu.edu Guidelines The Principal Investigator (PI) is responsible for leaving laboratories
LABORATORY WASTE DISPOSAL GUIDE
 LABORATORY WASTE DISPOSAL GUIDE Purpose This guide will help lab members properly identify and manage hazardous waste in their laboratory. Identifying the types of waste generated is crucial to handling
LABORATORY WASTE DISPOSAL GUIDE Purpose This guide will help lab members properly identify and manage hazardous waste in their laboratory. Identifying the types of waste generated is crucial to handling
WHO NEEDS TO KNOW THIS PROGRAM All NYU employees that generate, handle or transport RMW should be familiar with this written program.
 Title: NYU Regulated Medical Waste Safety Written Program Effective Date: October 2002 Revision Date: April 14, 2017 Issuing Authority: Responsible Officer: VP, Facilities and Construction Management Director
Title: NYU Regulated Medical Waste Safety Written Program Effective Date: October 2002 Revision Date: April 14, 2017 Issuing Authority: Responsible Officer: VP, Facilities and Construction Management Director
Radioactive Material User Annual Refresher Training. Created by SU Environmental Health & Safety Services
 Created by SU Environmental Health & Safety Services Syracuse University Office of Human Resources 2015 Welcome to Syracuse University s On-Line Radioactive Material User Refresher Training. This Refresher
Created by SU Environmental Health & Safety Services Syracuse University Office of Human Resources 2015 Welcome to Syracuse University s On-Line Radioactive Material User Refresher Training. This Refresher
OREGON HEALTH & SCIENCE UNIVERSITY RADIATION SAFETY OPERATING PROCEDURE 1801 RADIOACTIVE WASTE HANDLING & STORAGE
 OREGON HEALTH & SCIENCE UNIVERSITY RADIATION SAFETY OPERATING PROCEDURE 1801 RADIOACTIVE WASTE HANDLING & STORAGE I. PURPOSE: To describe the manner in which radioactive waste shall be stored and/or disposed
OREGON HEALTH & SCIENCE UNIVERSITY RADIATION SAFETY OPERATING PROCEDURE 1801 RADIOACTIVE WASTE HANDLING & STORAGE I. PURPOSE: To describe the manner in which radioactive waste shall be stored and/or disposed
RADIOISOTOPE ACQUISITION AND DISPOSITION
 RADIOISOTOPE ACQUISITION AND DISPOSITION PURPOSE This procedure specifies measures for the control of radioactive materials from initial acquisition to final disposition. It describes the prerequisites
RADIOISOTOPE ACQUISITION AND DISPOSITION PURPOSE This procedure specifies measures for the control of radioactive materials from initial acquisition to final disposition. It describes the prerequisites
UNIVERSITY OF RICHMOND REGULATED MEDICAL WASTE MANAGEMENT GUIDELINES
 UNIVERSITY OF RICHMOND REGULATED MEDICAL WASTE MANAGEMENT GUIDELINES October 2016 Table of Contents Section Page I. Introduction.... 1 II. Characteristics of Regulated Medical Waste 1-2 III. Exclusions...2-3
UNIVERSITY OF RICHMOND REGULATED MEDICAL WASTE MANAGEMENT GUIDELINES October 2016 Table of Contents Section Page I. Introduction.... 1 II. Characteristics of Regulated Medical Waste 1-2 III. Exclusions...2-3
Radiation Safety Program Hunter College of the City University of New York
 Radiation Safety Program Hunter College of the City University of New York 1. Guidelines for All Users of Radioactive Materials This document presents the guidelines for all persons using radioactive materials.
Radiation Safety Program Hunter College of the City University of New York 1. Guidelines for All Users of Radioactive Materials This document presents the guidelines for all persons using radioactive materials.
Radioactive Waste Radiation Safety Orientation Open Source Booklet 7 (June 1, 2018)
 Radioactive Waste Radiation Safety Orientation Open Source Booklet 7 (June 1, 2018) For more information, refer to the Radiation Safety Manual, 2017 RSP-3 and the Quick Step for waste in the radiation
Radioactive Waste Radiation Safety Orientation Open Source Booklet 7 (June 1, 2018) For more information, refer to the Radiation Safety Manual, 2017 RSP-3 and the Quick Step for waste in the radiation
Radiation Safety Manual
 University of California, Merced Environment, Health and Safety Radiation Safety Manual Approved by: Signature: Title: Date: TABLE OF CONTENTS 1. ORGANIZATION AND RESPONSIBILITIES 1.1 POLICY 1.2 RADIATION
University of California, Merced Environment, Health and Safety Radiation Safety Manual Approved by: Signature: Title: Date: TABLE OF CONTENTS 1. ORGANIZATION AND RESPONSIBILITIES 1.1 POLICY 1.2 RADIATION
Medical, Pathological and Low-level Radioactive Waste Management Plan for Incinerator Revised 3-08
 Background Medical, Pathological and Low-level Radioactive Waste Management Plan for Incinerator Revised 3-08 Many WSU laboratories and departments generate medical waste (contaminated solids), pathological
Background Medical, Pathological and Low-level Radioactive Waste Management Plan for Incinerator Revised 3-08 Many WSU laboratories and departments generate medical waste (contaminated solids), pathological
Infectious Waste Disposal Procedure 25 PA Code 284 Ursinus College Collegeville, PA 19426
 The purpose of the Infectious Waste Disposal procedure is to establish procedures for the safe handling and proper disposal of infectious waste generated at in accordance with regulation 25 PA Code Chapter
The purpose of the Infectious Waste Disposal procedure is to establish procedures for the safe handling and proper disposal of infectious waste generated at in accordance with regulation 25 PA Code Chapter
University of Richmond Radiation Safety Manual
 University of Richmond Radiation Safety Manual Office of Safety Services and Risk Management Revised: October 2017 i TABLE OF CONTENTS A. Purpose....1 B. Responsibility 1. Radiation Safety Committee.......1
University of Richmond Radiation Safety Manual Office of Safety Services and Risk Management Revised: October 2017 i TABLE OF CONTENTS A. Purpose....1 B. Responsibility 1. Radiation Safety Committee.......1
BIOMEDICAL WASTE MANAGEMENT POLICY
 BIOMEDICAL WASTE MANAGEMENT POLICY Managing biomedical waste in a safe and environmentally responsible manner is a key focus of the Environmental Compliance and Sustainability office. 1.0 PURPOSE In an
BIOMEDICAL WASTE MANAGEMENT POLICY Managing biomedical waste in a safe and environmentally responsible manner is a key focus of the Environmental Compliance and Sustainability office. 1.0 PURPOSE In an
Dartmouth College. Institutional Biosafety Committee. Biohazardous Waste Disposal Guide IBC Approved: 10/3/18
 Biohazardous Waste Disposal IBC Approved: 10/3/18 I. DEFINITION OF BIOHAZARDOUS WASTE: Biohazardous waste is any waste generated from working in biological or biomedical laboratories that may contain infectious
Biohazardous Waste Disposal IBC Approved: 10/3/18 I. DEFINITION OF BIOHAZARDOUS WASTE: Biohazardous waste is any waste generated from working in biological or biomedical laboratories that may contain infectious
Dartmouth College. Institutional Biosafety Committee. Biohazardous Waste Disposal Guide IBC Approved: 4/5/17
 Institutional Biosafety Committee Biohazardous Waste Disposal IBC Approved: 4/5/17 I. DEFINITION OF BIOHAZARDOUS WASTE: Biohazardous waste is any waste generated from working in biological or biomedical
Institutional Biosafety Committee Biohazardous Waste Disposal IBC Approved: 4/5/17 I. DEFINITION OF BIOHAZARDOUS WASTE: Biohazardous waste is any waste generated from working in biological or biomedical
8.0 Radioactive Waste
 8.0 Radioactive Waste 8.1 Solid Radioactive Waste Management Solid radioactive waste is disposed of by land burial at licensed, low-level radioactive waste facilities or by holding short-lived wastes for
8.0 Radioactive Waste 8.1 Solid Radioactive Waste Management Solid radioactive waste is disposed of by land burial at licensed, low-level radioactive waste facilities or by holding short-lived wastes for
Performance-Based Inspection Field Guide
 Non-radioactive material worker Performance-Based Inspection Field Guide 1. Have you completed the on-line Radiation Safety Course? If yes, get the individual s name.# to verify attendance If no, instruct
Non-radioactive material worker Performance-Based Inspection Field Guide 1. Have you completed the on-line Radiation Safety Course? If yes, get the individual s name.# to verify attendance If no, instruct
You are required to complete this course with a passing score of 85% or higher if you:
 OHS Biosafety BIO301L Medical Waste Management for Labs Training You are required to complete this course with a passing score of 85% or higher if you: Offer medical waste for transport from a UAB campus
OHS Biosafety BIO301L Medical Waste Management for Labs Training You are required to complete this course with a passing score of 85% or higher if you: Offer medical waste for transport from a UAB campus
The University of Texas Health Science Center at San Antonio Environmental Health & Safety Department Radiation Safety Division
 Page 1 of 6 The University of Texas Health Science Center at San Antonio Environmental Health & Safety Department Radiation Safety Division RADIOACTIVE MATERIAL AUTHORIZATION INITIAL APPLICATION AMENDENT
Page 1 of 6 The University of Texas Health Science Center at San Antonio Environmental Health & Safety Department Radiation Safety Division RADIOACTIVE MATERIAL AUTHORIZATION INITIAL APPLICATION AMENDENT
Policy & Procedure Manual
 Environmental Health & Safety Policy & Procedure Manual Title: 1. Purpose: To establish policies, work practices, and systematic procedures for the handling, packaging, collection, treatment, and disposal
Environmental Health & Safety Policy & Procedure Manual Title: 1. Purpose: To establish policies, work practices, and systematic procedures for the handling, packaging, collection, treatment, and disposal
DEPARTMENT OF BIOLOGICAL SCIENCES MEDICAL/BIOHAZARDOUS WASTE DISPOSAL AUTOCLAVE TRAINING
 DEPARTMENT OF BIOLOGICAL SCIENCES MEDICAL/BIOHAZARDOUS WASTE DISPOSAL AUTOCLAVE TRAINING INTRODUCTION In accordance with the California Medical Waste Management Act, Health and Safety Code Stanford University
DEPARTMENT OF BIOLOGICAL SCIENCES MEDICAL/BIOHAZARDOUS WASTE DISPOSAL AUTOCLAVE TRAINING INTRODUCTION In accordance with the California Medical Waste Management Act, Health and Safety Code Stanford University
THE UNIVERSITY OF TULSA RADIATION SAFETY PROGRAM. Table of Contents
 v6.13.2013 THE UNIVERSITY OF TULSA RADIATION SAFETY PROGRAM Table of Contents I. PURPOSE OF THE RADIATION SAFETY PROGRAM II. III. IV. DEFINITIONS SCOPE OF THE PROGRAM DELEGATION OF AUTHORITY AND RESPONSIBILITY
v6.13.2013 THE UNIVERSITY OF TULSA RADIATION SAFETY PROGRAM Table of Contents I. PURPOSE OF THE RADIATION SAFETY PROGRAM II. III. IV. DEFINITIONS SCOPE OF THE PROGRAM DELEGATION OF AUTHORITY AND RESPONSIBILITY
Laboratory Relocation Guidelines. Overview 2. Laboratory Close Out Contacts..3. Initial Laboratory Close Out Checklist...3
 Laboratory Relocation Guidelines Due to the variety and amount of hazardous materials (biological, chemical, and radioactive) routinely present in biomedical laboratories, laboratory relocations have the
Laboratory Relocation Guidelines Due to the variety and amount of hazardous materials (biological, chemical, and radioactive) routinely present in biomedical laboratories, laboratory relocations have the
For ethidium bromide waste that is generated at Harvard, each waste stream is to be managed or accumulated as described below.
 LABORATORY CHEMICAL WASTE FAQS ETHIDIUM BROMIDE 1. Q - How should I manage and dispose of ethidium bromide waste? Answer - Ethidium bromide is a mutagen that requires special storage, handling, and disposal
LABORATORY CHEMICAL WASTE FAQS ETHIDIUM BROMIDE 1. Q - How should I manage and dispose of ethidium bromide waste? Answer - Ethidium bromide is a mutagen that requires special storage, handling, and disposal
Consignment of Waste to the Disposal of Hazardous Waste Service
 Radioactive Waste LR 31 Who should read this document This document should be read by the Head of Department, Departmental Radiation Protection Supervisors, users of radioactive materials and their supervisors.
Radioactive Waste LR 31 Who should read this document This document should be read by the Head of Department, Departmental Radiation Protection Supervisors, users of radioactive materials and their supervisors.
WASTE MANAGEMENT GUIDE. Creighton University Environmental Health and Safety
 WASTE MANAGEMENT GUIDE Creighton University Environmental Health and Safety www.creighton.edu/ehs 402 546 6400 Overview With over 150 teaching and research labs, as well as other areas such as fine arts
WASTE MANAGEMENT GUIDE Creighton University Environmental Health and Safety www.creighton.edu/ehs 402 546 6400 Overview With over 150 teaching and research labs, as well as other areas such as fine arts
Biological and Regulated Medical Waste Plan
 Biological and Regulated Medical Waste Plan TUFTS ENVIRONMENTAL HEALTH AND SAFETY OCTOBER 2016 Table of Contents I. Purpose... 3 II. Responsibilities... 3 III. Sink and sewer discharge of specific medical
Biological and Regulated Medical Waste Plan TUFTS ENVIRONMENTAL HEALTH AND SAFETY OCTOBER 2016 Table of Contents I. Purpose... 3 II. Responsibilities... 3 III. Sink and sewer discharge of specific medical
LABORATORY WASTE MANAGEMENT PROGRAMMES
 LABORATORY WASTE MANAGEMENT PROGRAMMES Sylvia Wanjiru Kamau INTERNATIONAL LIVESTOCK RESEARCH INSTITUTE LABORATORY MANAGEMENT AND EQUIPMENT OPERATIONS WORKSHOP. 17 TH -21 ST JUNE 2012 TRAINING OBJECTIVES
LABORATORY WASTE MANAGEMENT PROGRAMMES Sylvia Wanjiru Kamau INTERNATIONAL LIVESTOCK RESEARCH INSTITUTE LABORATORY MANAGEMENT AND EQUIPMENT OPERATIONS WORKSHOP. 17 TH -21 ST JUNE 2012 TRAINING OBJECTIVES
University Health Services Health and Safety
 ADVISORY NO. 10.2: MANAGEMENT OF BIOLOGICAL AND INFECTIOUS WASTES The following recommendations are based on the Ohio Revised Code (ORC), Ohio Administrative Code (OAC) and EPA guidelines regarding biological
ADVISORY NO. 10.2: MANAGEMENT OF BIOLOGICAL AND INFECTIOUS WASTES The following recommendations are based on the Ohio Revised Code (ORC), Ohio Administrative Code (OAC) and EPA guidelines regarding biological
Biomedical Waste Management Guide
 Biomedical Waste Management Guide Yale University Office of Environmental Health & Safety 135 College Street, Suite 100, New Haven, CT 06510 Updated April 2017 Telephone: 203-785-3550 / Fax: 203-785-7588
Biomedical Waste Management Guide Yale University Office of Environmental Health & Safety 135 College Street, Suite 100, New Haven, CT 06510 Updated April 2017 Telephone: 203-785-3550 / Fax: 203-785-7588
INFECTIOUS/BIOLOGICAL WASTE MANAGEMENT PROTOCOL
 INFECTIOUS/BIOLOGICAL WASTE MANAGEMENT PROTOCOL UNIVERSITY RISK MANAGEMENT Occupational Safety and Health Programs 19 Hagood Avenue, Suite 908 Charleston, SC 29425 843-792-3604 Revised: November 2016 TABLE
INFECTIOUS/BIOLOGICAL WASTE MANAGEMENT PROTOCOL UNIVERSITY RISK MANAGEMENT Occupational Safety and Health Programs 19 Hagood Avenue, Suite 908 Charleston, SC 29425 843-792-3604 Revised: November 2016 TABLE
Regulated Medical Waste Management and Proper Waste Segregation. State-Specific Information Washington
 Regulated Medical Waste Management and Proper Waste Segregation State-Specific Information Washington Biomedical Waste Regulation In the State of Washington biomedical waste, where regulated, is regulated
Regulated Medical Waste Management and Proper Waste Segregation State-Specific Information Washington Biomedical Waste Regulation In the State of Washington biomedical waste, where regulated, is regulated
Laboratory Safety Reminders
 Laboratory Safety Reminders July 2012 Mount Holyoke College Environmental Health and Safety Safe Use of Radioactive Materials in MHC Research Labs Research Protocols A Radioactive Materials (RAM) use Protocol
Laboratory Safety Reminders July 2012 Mount Holyoke College Environmental Health and Safety Safe Use of Radioactive Materials in MHC Research Labs Research Protocols A Radioactive Materials (RAM) use Protocol
Date June 2, 2006 Page 1 of 5. Approved By
 Date June 2, 2006 Page 1 of 5 Reviewed By Lisa Arceneaux Approved By 1. PURPOSE The purpose of this procedure is to provide a viable method of waste management of radioactive waste when the half-life of
Date June 2, 2006 Page 1 of 5 Reviewed By Lisa Arceneaux Approved By 1. PURPOSE The purpose of this procedure is to provide a viable method of waste management of radioactive waste when the half-life of
Swette Center for Environmental Biotechnology Radiation Safety Policy
 Reviewed: BR date RKB date CIT date Swette Center for Environmental Biotechnology Radiation Safety Policy Scope, Application and Purpose: The purpose of the Center for Environmental Biotechnology Radiation
Reviewed: BR date RKB date CIT date Swette Center for Environmental Biotechnology Radiation Safety Policy Scope, Application and Purpose: The purpose of the Center for Environmental Biotechnology Radiation
Delaware State University
 Delaware State University University Area Responsible: Risk and Safety Management Policy Number and Name: 7-22: Laboratories Closeout Approval Date: 7/28/11 Reviewed: 7/26/2013 Revisions: 8/13/2013 Related
Delaware State University University Area Responsible: Risk and Safety Management Policy Number and Name: 7-22: Laboratories Closeout Approval Date: 7/28/11 Reviewed: 7/26/2013 Revisions: 8/13/2013 Related
THE UNIVERSITY OF NEWCASTLE- DISCIPLINE OF MEDICAL BIOCHEMISTRY
 THE UNIVERSITY OF NEWCASTLE- DISCIPLINE OF MEDICAL BIOCHEMISTRY STANDARD OPERATING PROCEDURE PROCEDURE NO: GDP 020 MOD: 1st Issue Page: 1 of 5 Procedure Type: General Discipline Procedure Title: Handling
THE UNIVERSITY OF NEWCASTLE- DISCIPLINE OF MEDICAL BIOCHEMISTRY STANDARD OPERATING PROCEDURE PROCEDURE NO: GDP 020 MOD: 1st Issue Page: 1 of 5 Procedure Type: General Discipline Procedure Title: Handling
Western Michigan University
 Western Michigan University Radiation Safety Source Inventory and Control Program Purpose A. To ensure all Western Michigan University licensed material and radiation producing machines are controlled
Western Michigan University Radiation Safety Source Inventory and Control Program Purpose A. To ensure all Western Michigan University licensed material and radiation producing machines are controlled
Appendix J. University of Alabama at Birmingham Medical Waste Management Plan
 University of Alabama at Birmingham Medical Waste Management Plan I. Definitions per the Alabama Department of Environmental Management Land Division 13-Solid Waste Program, Chapter 335-13-1, Medical Waste
University of Alabama at Birmingham Medical Waste Management Plan I. Definitions per the Alabama Department of Environmental Management Land Division 13-Solid Waste Program, Chapter 335-13-1, Medical Waste
RADIOACTIVE MATERIALS SAFETY REFRESHER EXAM
 RADIOACTIVE MATERIALS SAFETY REFRESHER EXAM DIRECTIONS: Please print this quiz and fill in the information requested. Mark the correct answer to each question. When completed either scan or email the quiz
RADIOACTIVE MATERIALS SAFETY REFRESHER EXAM DIRECTIONS: Please print this quiz and fill in the information requested. Mark the correct answer to each question. When completed either scan or email the quiz
University of Maryland Baltimore. Radiation Safety Procedure
 University of Maryland Baltimore Procedure Number: 2.1 Radiation Safety Procedure Title: Ordering, Receiving, Opening, And Transferring Packages Containing Revision Number: 0 Technical Review and Approval:
University of Maryland Baltimore Procedure Number: 2.1 Radiation Safety Procedure Title: Ordering, Receiving, Opening, And Transferring Packages Containing Revision Number: 0 Technical Review and Approval:
LABORATORY CLOSURE AND DECOMMISSIONING POLICY
 LABORATORY CLOSURE AND DECOMMISSIONING POLICY Yale University is committed to the health and safety of its students, faculty, staff and visitors as well as the surrounding community and environment in
LABORATORY CLOSURE AND DECOMMISSIONING POLICY Yale University is committed to the health and safety of its students, faculty, staff and visitors as well as the surrounding community and environment in
Hazardous Materials Management Policies and Procedures
 Hazardous Materials Management Policies and Procedures Table of Contents I. Purpose... 2 II. Scope... 2 III. Policy... 2 III.A. Methods for Managing Hazardous Materials... 2 III.A.1. Ordering... 2 III.A.2.
Hazardous Materials Management Policies and Procedures Table of Contents I. Purpose... 2 II. Scope... 2 III. Policy... 2 III.A. Methods for Managing Hazardous Materials... 2 III.A.1. Ordering... 2 III.A.2.
Biosafety Checklist. 07-BiosafetyChecklist-LTC-SOP-v2.0-17Feb of 6
 Title: Biosafety Checklist Origination Date: 31 Jan 1997 Total Pages: 5 Effective Date: 17 Feb 2012 SOP Number LTC-SOP-07 v2.0 Supersedes SOP Written By: ACTG/IMPAACT Lab Tech Committee Dated: 01 Jun 2004
Title: Biosafety Checklist Origination Date: 31 Jan 1997 Total Pages: 5 Effective Date: 17 Feb 2012 SOP Number LTC-SOP-07 v2.0 Supersedes SOP Written By: ACTG/IMPAACT Lab Tech Committee Dated: 01 Jun 2004
Health Sciences Campus Biomedical Waste Management Standard Operating Procedure (SOP)
 Health Sciences Campus Biomedical Waste Management Standard Operating Procedure (SOP) NOTE: This SOP for biological waste management does not supersede the requirements for radioactive and/or hazardous
Health Sciences Campus Biomedical Waste Management Standard Operating Procedure (SOP) NOTE: This SOP for biological waste management does not supersede the requirements for radioactive and/or hazardous
RADIATION SAFETY MANUAL
 RADIATION SAFETY MANUAL Environmental Health, Safety & Risk Management September 1, 2015 Table of Contents A. Introduction... 3 B. Responsibilities... 4 1. Radiation Safety Officer... 4 2. Department of
RADIATION SAFETY MANUAL Environmental Health, Safety & Risk Management September 1, 2015 Table of Contents A. Introduction... 3 B. Responsibilities... 4 1. Radiation Safety Officer... 4 2. Department of
CLEMSON UNIVERSITY LAB CLOSE-OUT POLICY/PROCEDURES FOR HAZARDOUS SUBSTANCES, EQUIPMENT, SUPPLIES, ETC.
 CLEMSON UNIVERSITY LAB CLOSE-OUT POLICY/PROCEDURES FOR HAZARDOUS SUBSTANCES, EQUIPMENT, SUPPLIES, ETC. The purpose of this policy is to ensure that all hazardous materials, lab equipment, etc. is properly
CLEMSON UNIVERSITY LAB CLOSE-OUT POLICY/PROCEDURES FOR HAZARDOUS SUBSTANCES, EQUIPMENT, SUPPLIES, ETC. The purpose of this policy is to ensure that all hazardous materials, lab equipment, etc. is properly
Radiation Safety Operating Procedure. Shipping and Transporting
 1.0 Statement All shipments of open source nuclear substances to and from Memorial University shall be handled as detailed in this procedure to ensure compliance with appropriate regulations and in a manner
1.0 Statement All shipments of open source nuclear substances to and from Memorial University shall be handled as detailed in this procedure to ensure compliance with appropriate regulations and in a manner
Guidelines for Laboratory Closure
 Guidelines for Laboratory Closure These guidelines are to facilitate the process of laboratory closure when an investigator is vacating, relocating, or renovating a lab space. These guidelines only cover
Guidelines for Laboratory Closure These guidelines are to facilitate the process of laboratory closure when an investigator is vacating, relocating, or renovating a lab space. These guidelines only cover
Standard Operating Procedure. Handling of Radioactive Material. PC-SOP-IM-005-v02
 Standard Operating Procedure Handling of Radioactive Material Revision History Version Reason for Revision Date 01 New SOP August/21/2013 02 Review and update February/12/2015 Summary The content of this
Standard Operating Procedure Handling of Radioactive Material Revision History Version Reason for Revision Date 01 New SOP August/21/2013 02 Review and update February/12/2015 Summary The content of this
DISPOSAL OF HAZARDOUS WASTE, CHEMICAL WASTE AND UNUSED CHEMICALS
 NO. 3071 DISPOSAL OF HAZARDOUS WASTE, CHEMICAL WASTE AND UNUSED CHEMICALS 1.0 PURPOSE. Oklahoma City Community College (OCCC) recognizes that employees have a right and need to know the properties and
NO. 3071 DISPOSAL OF HAZARDOUS WASTE, CHEMICAL WASTE AND UNUSED CHEMICALS 1.0 PURPOSE. Oklahoma City Community College (OCCC) recognizes that employees have a right and need to know the properties and
PI s Name Date Bldg./Rm#
 PI s Name Date Bldg./Rm# Animal Biosafety Level 3 (ABSL-3) Yes No 1. Is access to the animal facility limited or restricted only to those persons authorized for program or support purposes? Yes No 2. Does
PI s Name Date Bldg./Rm# Animal Biosafety Level 3 (ABSL-3) Yes No 1. Is access to the animal facility limited or restricted only to those persons authorized for program or support purposes? Yes No 2. Does
In-Country shipment : How to safely ship human blood samples from suspected Ebola cases within a country by road, rail and sea
 In-Country shipment : How to safely ship human blood samples from suspected Ebola cases within a country by road, rail and sea Step 1: Before handling the sample, prepare all shipping equipment (1) Manage
In-Country shipment : How to safely ship human blood samples from suspected Ebola cases within a country by road, rail and sea Step 1: Before handling the sample, prepare all shipping equipment (1) Manage
Chapter 4 Procurement and Transfer of Radioactive Materials
 Chapter 4 Procurement and Transfer of Radioactive Materials Radioactive material may be procured only by authorized radionuclide projects. Project directors must work with their support personnel and the
Chapter 4 Procurement and Transfer of Radioactive Materials Radioactive material may be procured only by authorized radionuclide projects. Project directors must work with their support personnel and the
Laboratory Decontamination and Decommissioning Policy and Procedures. Office of Environmental Health and Safety
 Laboratory Decontamination and Decommissioning Policy and Procedures Office of Environmental Health and Safety July 2018 1. Purpose and Scope 1.1 It is the policy of Cleveland State University that laboratory
Laboratory Decontamination and Decommissioning Policy and Procedures Office of Environmental Health and Safety July 2018 1. Purpose and Scope 1.1 It is the policy of Cleveland State University that laboratory
Nuclear Substance(Class 7) Receiving: RefresherTraining
 Nuclear Substance(Class 7) Receiving: RefresherTraining Refresher Training Instructions The following Class 7 Receiving Refresher training is intended for nuclear substance workers who have previously
Nuclear Substance(Class 7) Receiving: RefresherTraining Refresher Training Instructions The following Class 7 Receiving Refresher training is intended for nuclear substance workers who have previously
How to safely ship human blood samples from suspected Ebola cases within a country by road, rail and sea
 INTERIM GUIDELINE How to safely ship human blood samples from suspected Ebola cases within a country by road, rail and sea 2014 Step 1: Before handling the sample, prepare all shipping equipment Step 1a:
INTERIM GUIDELINE How to safely ship human blood samples from suspected Ebola cases within a country by road, rail and sea 2014 Step 1: Before handling the sample, prepare all shipping equipment Step 1a:
Biomedical Waste Management Plan
 Biomedical Waste Management Plan USF Biomedical Waste Management Plan for: Facility Address: Facility Phone Number: Facility Contact: Division of Environmental Health and Safety 4202 E. Fowler Ave, OPM
Biomedical Waste Management Plan USF Biomedical Waste Management Plan for: Facility Address: Facility Phone Number: Facility Contact: Division of Environmental Health and Safety 4202 E. Fowler Ave, OPM
This is Chapter 6, part-two of the Radiation Safety Short Course: Radioisotope Laboratory Techniques. In this section we will review what Radiation
 This is Chapter 6, part-two of the Radiation Safety Short Course: Radioisotope Laboratory Techniques. In this section we will review what Radiation Safety personnel evaluate when completing the quarterly
This is Chapter 6, part-two of the Radiation Safety Short Course: Radioisotope Laboratory Techniques. In this section we will review what Radiation Safety personnel evaluate when completing the quarterly
How to safely ship human blood samples from Lassa cases within a country by road, rail and sea
 How to safely ship human blood samples from Lassa cases within a country by road, rail and sea Interim Guidance February 2018 Step 1: Before handling the sample, prepare all shipping equipment Step 1a:
How to safely ship human blood samples from Lassa cases within a country by road, rail and sea Interim Guidance February 2018 Step 1: Before handling the sample, prepare all shipping equipment Step 1a:
GUIDELINES FOR SHIPPING NONPATHOGENIC BIOLOGICAL CULTURES AND NON-INFECTIOUS BIOLOGICAL MATERIALS
 GUIDELINES FOR SHIPPING NONPATHOGENIC BIOLOGICAL CULTURES AND NON-INFECTIOUS BIOLOGICAL MATERIALS Table of Contents General Information Regulations Definitions Recommended packing procedure Universal precautions
GUIDELINES FOR SHIPPING NONPATHOGENIC BIOLOGICAL CULTURES AND NON-INFECTIOUS BIOLOGICAL MATERIALS Table of Contents General Information Regulations Definitions Recommended packing procedure Universal precautions
Environmental Health and Safety Equipment Release Form
 Environmental Health and Equipment Release Form Date: Location of Origin: Principal Investigator/Supervisor: Type of Equipment: Destination/Service Department: Service to Be Performed: Contaminated: Not
Environmental Health and Equipment Release Form Date: Location of Origin: Principal Investigator/Supervisor: Type of Equipment: Destination/Service Department: Service to Be Performed: Contaminated: Not
RADIOACTIVE WASTE MANAGEMENT PLAN
 RADIOACTIVE WASTE MANAGEMENT PLAN Copies of this plan will be made available to all members of the campus community with the potential to generate radioactive waste. Contents I. Purpose II. Regulatory
RADIOACTIVE WASTE MANAGEMENT PLAN Copies of this plan will be made available to all members of the campus community with the potential to generate radioactive waste. Contents I. Purpose II. Regulatory
Radioactive Material Safety Manual
 Radioactive Material Safety Manual Radiation Safety Office URI Bay Campus 16 Reactor Road Narragansett, RI 02882 Phone: 401-874-2600 Fax: 401-874-9452 www.uri.edu/radiation 02/2015 Updated 07/2017 Table
Radioactive Material Safety Manual Radiation Safety Office URI Bay Campus 16 Reactor Road Narragansett, RI 02882 Phone: 401-874-2600 Fax: 401-874-9452 www.uri.edu/radiation 02/2015 Updated 07/2017 Table
MODEL EXPOSURE CONTROL PLAN
 MODEL EXPOSURE CONTROL PLAN The Model Exposure Control Plan is intended to serve as an employer guide to the OSHA Bloodborne Pathogens standard. A central component of the requirements of the standard
MODEL EXPOSURE CONTROL PLAN The Model Exposure Control Plan is intended to serve as an employer guide to the OSHA Bloodborne Pathogens standard. A central component of the requirements of the standard
nc.us/swhome/
 Appendix 2 Waste Management Rules in North Carolina http://wastenot.enr.state.nc.us/swhome/12rul.htm#http://wastenot.enr.state. nc.us/swhome/ Home About DWM Contact Us Site Map Search Laws, Rules & Regulations
Appendix 2 Waste Management Rules in North Carolina http://wastenot.enr.state.nc.us/swhome/12rul.htm#http://wastenot.enr.state. nc.us/swhome/ Home About DWM Contact Us Site Map Search Laws, Rules & Regulations
Standard Operating Procedure Title: Good laboratory practices (GLP) for microbiology and chemistry laboratories
 Standard Operating Procedure Title: Good laboratory practices (GLP) for microbiology and chemistry laboratories Department Micro Laboratory Document no MICLAB 155 Title Good laboratory practices (GLP)
Standard Operating Procedure Title: Good laboratory practices (GLP) for microbiology and chemistry laboratories Department Micro Laboratory Document no MICLAB 155 Title Good laboratory practices (GLP)
Radiation Safety Policy & Procedures
 Radiation Safety Policy & Procedures Emergency Numbers RADIATION SAFETY OFFICER, RAMIN VAKILI... FIRE, POLICE, AND AMBULANCE...- CAMPUSE SECURITY... TABLE OF CONTENTS. PREAMBLE.... SCOPE.... POLICY....
Radiation Safety Policy & Procedures Emergency Numbers RADIATION SAFETY OFFICER, RAMIN VAKILI... FIRE, POLICE, AND AMBULANCE...- CAMPUSE SECURITY... TABLE OF CONTENTS. PREAMBLE.... SCOPE.... POLICY....
Laboratory Waste Disposal
 Laboratory Waste Disposal 1. Purpose This guideline details the procedures to follow in disposing of hazardous waste that is generated in the laboratory in order to minimise risks associated with the disposal
Laboratory Waste Disposal 1. Purpose This guideline details the procedures to follow in disposing of hazardous waste that is generated in the laboratory in order to minimise risks associated with the disposal
EHS Assist Radioactive Material User Instructions. Link to EHS Assist from the EHS homepage
 EHS Assist s Web Address: http://www.ehs.osu.edu Link to EHS Assist from the EHS homepage Log-In using assigned USER ID and PASSWORD provided by Environmental Health and Safety Main Menu Waste Pickup Used
EHS Assist s Web Address: http://www.ehs.osu.edu Link to EHS Assist from the EHS homepage Log-In using assigned USER ID and PASSWORD provided by Environmental Health and Safety Main Menu Waste Pickup Used
Radiation Safety Manual
 Table of Contents I. Purpose... 3 II. Scope... 3 III. Procedures... 3 III.A. Responsibilities... 3 III.A.1. Radiation Safety Committee... 3 III.A.2. Radiation Safety Officer... 4 III.A.3. Licensed Principal
Table of Contents I. Purpose... 3 II. Scope... 3 III. Procedures... 3 III.A. Responsibilities... 3 III.A.1. Radiation Safety Committee... 3 III.A.2. Radiation Safety Officer... 4 III.A.3. Licensed Principal
Employee Environmental, Health & Safety Manual. INFECTIOUS WASTE MANAGEMENT PROGRAM Latest Revision: 05/27/08
 Note: Print document as needed for MATC Departmental purposes; it is user responsibility to ensure use of latest revision by CONTENT checking online at: http://matcmadison.edu/ad/facilities/ehs/ under
Note: Print document as needed for MATC Departmental purposes; it is user responsibility to ensure use of latest revision by CONTENT checking online at: http://matcmadison.edu/ad/facilities/ehs/ under
RADIATION SAFETY INSPECTION CHECKLIST Section A : Contact Information (Principle Investigator) Last Name: First Name: Extension:
 RADIATION SAFETY INSPECTION CHECKLIST Section A : Contact Information (Principle Investigator) Last Name: First Name: Extension: Department: Building: Room: Section B: Inspection Date of Inspection: Time
RADIATION SAFETY INSPECTION CHECKLIST Section A : Contact Information (Principle Investigator) Last Name: First Name: Extension: Department: Building: Room: Section B: Inspection Date of Inspection: Time
BALL STATE UNIVERSITY RADIATION PROTECTION PROCEDURES* USER S GUIDE
 BALL STATE UNIVERSITY RADIATION PROTECTION PROCEDURES* USER S GUIDE At Ball State University, there are two categories of material users. Authorized Users and Supervised Users. Authorized Users must have
BALL STATE UNIVERSITY RADIATION PROTECTION PROCEDURES* USER S GUIDE At Ball State University, there are two categories of material users. Authorized Users and Supervised Users. Authorized Users must have
THE RICHARD STOCKTON COLLEGE OF NEW JERSEY. Natural Sciences and Mathematics Department. Radiation Safety Program
 Revised February 2004 THE RICHARD STOCKTON COLLEGE OF NEW JERSEY Natural Sciences and Mathematics Department Radiation Safety Program 1. Introduction The United States Nuclear Regulatory Commission (NRC)
Revised February 2004 THE RICHARD STOCKTON COLLEGE OF NEW JERSEY Natural Sciences and Mathematics Department Radiation Safety Program 1. Introduction The United States Nuclear Regulatory Commission (NRC)
NOTE: Do NOT overfill sharps containers; there should be NO more than 50ml of residual liquid in the mailback kit when returned
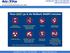 These DO go in the Mailback SHARPS Containers Sharps Waste Such As: Needles & syringes Razor blades Orthodon?c wires Scalpel blades & lancets Glass pipebes, slides & tubes Broken, contaminated glass Staples
These DO go in the Mailback SHARPS Containers Sharps Waste Such As: Needles & syringes Razor blades Orthodon?c wires Scalpel blades & lancets Glass pipebes, slides & tubes Broken, contaminated glass Staples
Michigan Small Animal Imaging Resource In Vivo Cellular and Molecular Imaging Center. Department of Radiology. University of Michigan
 Date: August 29, 2013 Version: 8 Page: 1 of 6 A. PURPOSE: Many animal use protocols entail the administration or exposure of the research animals to potentially hazardous chemical agents or pathogens.
Date: August 29, 2013 Version: 8 Page: 1 of 6 A. PURPOSE: Many animal use protocols entail the administration or exposure of the research animals to potentially hazardous chemical agents or pathogens.
Environment, Health and Safety Guidelines For Moving Downtown
 Environment, Health & Safety Services Jacobs School of Medicine and Biomedical Sciences Environment, Health and Safety Guidelines For Moving Downtown 220 Winspear Ave., Buffalo, NY 14215-1034 Office: (716)
Environment, Health & Safety Services Jacobs School of Medicine and Biomedical Sciences Environment, Health and Safety Guidelines For Moving Downtown 220 Winspear Ave., Buffalo, NY 14215-1034 Office: (716)
Animal Facility Biosafety Level 3 Checklist (date: April 16, 1998)
 Date: Location: Responsible: Project Title: Inspector: _ Animal Facility Biosafety Level 3 Checklist (date: April 16, 1998) These questions are based on the Biosafety Level 3 section of Biosafety in Microbiological
Date: Location: Responsible: Project Title: Inspector: _ Animal Facility Biosafety Level 3 Checklist (date: April 16, 1998) These questions are based on the Biosafety Level 3 section of Biosafety in Microbiological
California Polytechnic State University San Luis Obispo. Radiation Safety Manual
 California Polytechnic State University San Luis Obispo Radiation Safety Manual November 2016 1. ADMINISTRATIVE PROCEDURES AND RESPONSIBILITIES The purpose of the Radiation Safety Program is to assure
California Polytechnic State University San Luis Obispo Radiation Safety Manual November 2016 1. ADMINISTRATIVE PROCEDURES AND RESPONSIBILITIES The purpose of the Radiation Safety Program is to assure
Waste Disposal Policies and Procedures
 Table of Contents I. Purpose... 3 II. Scope... 3 III. Policy... 3 III.A. Ordering... 3 III.B. Ordinary Trash... 3 III.C. Sharps... 4 III.C.1. Sharps, Non-Infectious, Non-Contaminated... 4 III.C.2. Sharps,
Table of Contents I. Purpose... 3 II. Scope... 3 III. Policy... 3 III.A. Ordering... 3 III.B. Ordinary Trash... 3 III.C. Sharps... 4 III.C.1. Sharps, Non-Infectious, Non-Contaminated... 4 III.C.2. Sharps,
Radiation Safety Services. Prospective Radiation User. Request to Use Radioactive Material (RM) Here at UMASS
 UNIVERSITY OF MASSACHUSETTS/AMHERST ENVIRONMENTAL HEALTH & SAFETY 117 Draper Hall, 40 Campus Center Way AMHERST, MA 01003-9244 413.545.2682 413.545.2600 fax FROM: TO: SUBJECT: Radiation Safety Services
UNIVERSITY OF MASSACHUSETTS/AMHERST ENVIRONMENTAL HEALTH & SAFETY 117 Draper Hall, 40 Campus Center Way AMHERST, MA 01003-9244 413.545.2682 413.545.2600 fax FROM: TO: SUBJECT: Radiation Safety Services
Philip E. Hamrick, Ph.D. National Institute of Environmental Health Sciences Research Triangle Park, NC
 MANAGEMENT OF LOW-LEVEL RADIOACTIVE WASTES Philip E. Hamrick, Ph.D. National Institute of Environmental Health Sciences Research Triangle Park, NC The procedures for the management of low-level radioactive
MANAGEMENT OF LOW-LEVEL RADIOACTIVE WASTES Philip E. Hamrick, Ph.D. National Institute of Environmental Health Sciences Research Triangle Park, NC The procedures for the management of low-level radioactive
TJEMS Drug Box Program Best Practices Reviewed: Updated: November 2015
 TJEMS Drug Box Program Best Practices Reviewed: Updated: November 2015 The TJEMS Drug Box Program Best Practices relate to the use of the TJEMS Drug Box. These best practices sever to provide guidance
TJEMS Drug Box Program Best Practices Reviewed: Updated: November 2015 The TJEMS Drug Box Program Best Practices relate to the use of the TJEMS Drug Box. These best practices sever to provide guidance
Guidelines for the Use of Cytotoxic or Chemotherapeutic Drugs
 Guidelines for the Use of Cytotoxic or Chemotherapeutic Drugs Caltech Environment, Health and Safety 1200 E. California Blvd. M/C 25-6 Pasadena, CA 91125 626.395.6727 safety@caltech.edu www.safety.caltech.edu
Guidelines for the Use of Cytotoxic or Chemotherapeutic Drugs Caltech Environment, Health and Safety 1200 E. California Blvd. M/C 25-6 Pasadena, CA 91125 626.395.6727 safety@caltech.edu www.safety.caltech.edu
Chapter 7 Transportation of Radioactive Material
 Chapter 7 Transportation of Radioactive Material Radioactive material that is possessed by UIC must be transported in a safe and legal manner. This chapter explains the requirements for transportation
Chapter 7 Transportation of Radioactive Material Radioactive material that is possessed by UIC must be transported in a safe and legal manner. This chapter explains the requirements for transportation
MIXED WASTE MANAGEMENT PROGRAM. UNIVERSITY RISK MANAGEMENT Radiation Safety 19 Hagood Avenue, Suite 301 Charleston SC
 MIXED WASTE MANAGEMENT PROGRAM UNIVERSITY RISK MANAGEMENT Radiation Safety 19 Hagood Avenue, Suite 301 Charleston SC 29425 843-792-4255 Revised: January 2015 TABLE OF CONTENTS INTRODUCTION... 3 DEFINITION...
MIXED WASTE MANAGEMENT PROGRAM UNIVERSITY RISK MANAGEMENT Radiation Safety 19 Hagood Avenue, Suite 301 Charleston SC 29425 843-792-4255 Revised: January 2015 TABLE OF CONTENTS INTRODUCTION... 3 DEFINITION...
Animal cell and tissue culture. Lab 1
 Animal cell and tissue culture Lab 1 Tissue culture Laboratory Safety Outline Lab Safety Biohazards Biosafety Levels Biosafety Cabinets Decontamination Biological Waste Introduction A cell culture laboratory
Animal cell and tissue culture Lab 1 Tissue culture Laboratory Safety Outline Lab Safety Biohazards Biosafety Levels Biosafety Cabinets Decontamination Biological Waste Introduction A cell culture laboratory
MODEL EXPOSURE CONTROL PLAN
 MODEL EXPOSURE CONTROL PLAN The Model Exposure Control Plan is intended to serve employers as an example exposure control plan which is required by the Bloodborne Pathogens Standard. A central component
MODEL EXPOSURE CONTROL PLAN The Model Exposure Control Plan is intended to serve employers as an example exposure control plan which is required by the Bloodborne Pathogens Standard. A central component
Title: Engineering Services - Pneumatic Tube System Operational - Guidelines
 Document Owner: Timothy Markijohn Content Expert: Bobby Schnetzler Last Approved Date: 05/30/2015 Printed copies are for reference only. Please refer to the electronic copy for the latest version. I. Purpose
Document Owner: Timothy Markijohn Content Expert: Bobby Schnetzler Last Approved Date: 05/30/2015 Printed copies are for reference only. Please refer to the electronic copy for the latest version. I. Purpose
NJIT Biological Waste Management for Laboratories. NJIT Environmental Health and Safety Department Spring, 2017
 NJIT Biological Waste Management for Laboratories NJIT Environmental Health and Safety Department Spring, 2017 Biological Waste In broad terms, biological waste may be defined as: - liquid or solid waste
NJIT Biological Waste Management for Laboratories NJIT Environmental Health and Safety Department Spring, 2017 Biological Waste In broad terms, biological waste may be defined as: - liquid or solid waste
ENVIRONMENTAL MANAGEMENT GUIDE FOR SMALL LABORATORIES
 United States Office of the EPA 233-B-00-001 Environmental Protection Administrator May 2000 Agency (2131) ENVIRONMENTAL MANAGEMENT GUIDE FOR SMALL LABORATORIES 3.5 Biologically Active Substances and Wastes
United States Office of the EPA 233-B-00-001 Environmental Protection Administrator May 2000 Agency (2131) ENVIRONMENTAL MANAGEMENT GUIDE FOR SMALL LABORATORIES 3.5 Biologically Active Substances and Wastes
Appendix B: Decommissioning Checklist for Lab Move
 Laboratory Decommissioning/Commissioning Document - LS-003 12 Appendix B: Decommissioning Checklist for Lab Move This form is for lab moves only from one UTK location to another UTK location General Information
Laboratory Decommissioning/Commissioning Document - LS-003 12 Appendix B: Decommissioning Checklist for Lab Move This form is for lab moves only from one UTK location to another UTK location General Information
Biological Safety Program. For. Otterbein University
 Biological Safety Program For Otterbein University Developed in conformance with the Ohio EPA Infectious Waste Regulation, OAC 3745-27 Table of Contents PURPOSE... 2 GENERAL STATEMENT... 2 POLICY... 3
Biological Safety Program For Otterbein University Developed in conformance with the Ohio EPA Infectious Waste Regulation, OAC 3745-27 Table of Contents PURPOSE... 2 GENERAL STATEMENT... 2 POLICY... 3
WARTBURG COLLEGE ENVIRONMENTAL AND OCCUPATIONAL SAFETY PROGRAM HAZARDOUS WASTE PLAN
 3 February, 2001 WARTBURG COLLEGE ENVIRONMENTAL AND OCCUPATIONAL SAFETY PROGRAM TABLE OF CONTENTS HAZARDOUS WASTE PLAN 1. General 2. Responsibilities 3. Program Development 4. References 5. Management
3 February, 2001 WARTBURG COLLEGE ENVIRONMENTAL AND OCCUPATIONAL SAFETY PROGRAM TABLE OF CONTENTS HAZARDOUS WASTE PLAN 1. General 2. Responsibilities 3. Program Development 4. References 5. Management
RADIATION PROCEDURES MANUAL Procedure Cover Sheet
 RADIATION PROCEDURES MANUAL Procedure Cover Sheet Procedure Title: Receipt of Package Containing Radioactive Material Procedure Number: TSO-10-18-REV 1 Effective Date: April 23, 2011 Date: April 23, 2011
RADIATION PROCEDURES MANUAL Procedure Cover Sheet Procedure Title: Receipt of Package Containing Radioactive Material Procedure Number: TSO-10-18-REV 1 Effective Date: April 23, 2011 Date: April 23, 2011
