Gases and the atmosphere
|
|
|
- Charity Johns
- 5 years ago
- Views:
Transcription
1 Gases and the atmosphere Chemical reactions in the atmosphere Ozone cycle Ozone depletion Greenhouse effect Global Warming Pollution in the troposphere 1
2 Gases and the atmosphere Review Composition of the atmosphere Use mole fraction and ppm Pressure profile of atmosphere Low pressure means few molecular collisions Chemical reactions occur less frequently Temperature profile of atmosphere High temperature means avg. KE of molecules is high! 2
3 Composition of the atmosphere mole fraction: x i = moles of i total moles N 2 and O 2 represent > 99% of atm 3
4 Composition of the atmosphere For other components Eg. Neon use parts per million (ppm). ppm = x i 10 6 x Ne = Ne concentration = ppm If you know the barometric pressure, you can determine the partial pressure. P BAR = atm= P TOT P Ne = x Ne P TOT = ( atm) = atm 4
5 Pressure decreases exponentially with altitude. WHY? Pressure at any altitude depends on weight of gas above it. What is pressure? 5
6 Pressure and altitude Altitude P collision frequency (km) (atm) (sec -1 ) Low pressure means fewer collisions When collision frequency is low, chemical reactions occur less frequently 6
7 Regions of Atmosphere Based on Temperature Profile Thermosphere: High energy radiation is absorbed Ions formed. Mesosphere: Density of gases is small. Stratosphere: Warming caused by ozone cycle Ultraviolet light absorbed Troposphere: Life: where we live! weather planes Present life forms could not survive (above ground) if all solar radiation reaches earth. 7
8 Distribution of Radiation 8
9 Penetration of Solar Radiation 9
10 Reactions in the atmosphere Photochemical Reactions: reactions that involve the absorption or emission of a photon. Electronic excitation of molecules NO 2 (g) + hν NO 2* (g) Photodissociation bond broken by absorption of a photon (light) O 2 + hν O + O E = hν Photon needs sufficient energy to break bond (Bond energy for O 2 = 495kJ/mol) Photoionization λν = c λ < 242 nm removal of an outer (valence electron) from a molecule by absorption of a photon N 2 + hν N e - 10
11 Thermosphere (Ionosphere) 11
12 Warming of the Stratosphere Ozone concentration peaks in stratosphere ~25km, concentration 10ppm Presence of ozone causes temperature increase in stratosphere. 12
13 Ozone O 3 Structure 2 resonance forms: bent, bond angle = 117 bond length = 1.28Å (O 2 is 1.21Å) (in agreement with VSEPR predictions) Properties: Light blue gas Pungent odor (smell near electrical discharges) ΔH f = 142kJ/mol (reactive less stable than O 2 ) 13
14 formation of O 3 Why is ozone so important? Ozone cycle O 2 + hν 2O λ < 242nm O + O 2 O 3 uv blocking by O 3 O 3 + hν O 2 + O λ < 320nm Solar radiation (ultraviolet) is converted to heat The (small) amount of O 3 in stratosphere reflects the delicate balance between creation and destruction. 14
15 Problem: Ozone depletion What causes it? Chlorofluorocarbons (CFCs) Examples: CFCl 3 CF 2 Cl 2 freon-11 freon-12 Properties relatively inert easily liquefied Non-combustible volatile evaporate and diffuse upward easily Uses aerosol propellants coolants for refrig. solvents for cleaning polymer manufacturing 15
16 CFC destruction by ozone CF 2 Cl 2 + hν CF 2 Cl + Cl (λ<240nm) Cl + O 3 ClO + O 2 ClO + O Cl + O 2 NET: O 3 + O 2O 2 Cl atom from CFC catalyzes O 3 destruction (speeds up reaction, but is not used up). 1 Cl atom destroys ~ 2 million O 3 molecules Global concentration of O 3 has been on the decline since
17 Greenhouse Effect Energy comes to earth from the sun. Some is reflected by atmosphere Some is absorbed Most of uv and visible reaches the earth s surface: causes warming. Earth radiates heat back out. mainly infrared energy 17
18 The earth s energy balance Distribution of Radiation Solar radiation warms the earth Earth radiates infrared light back 18
19 H 2 O and CO 2 thermally regulate the atmosphere. Greenhouse effect Absorption of infrared radiation: Activates stretching and bending modes keeps heat from being released back into space Without the greenhouse effect, Earth would be ~33 colder (water ice) 19
20 What is Global Warming? Over the past century atmospheric CO 2 has increased ~25% Mean global temperature has increased ~1 Increased quantities of CO 2 may be causing global warming. 20
21 How serious is the problem? Need to know about Carbon cycle: interconversion of C in the environment 21
22 The Carbon Cycle Sources of atmospheric CO 2 Natural volcanic eruption sources decaying organic materials respiration Man-made fuel combustion sources deforestation cement manufacturing Sinks for CO 2 Formation of rock Dissolves in oceans Photosynthesis 22
23 Summary ~200 billion metric tons (bmt) of C in the form of CO 2 is removed each year from the atmosphere. ~110 bmt removed by photosynthesis ~ 90 bmt removed by absorption in ocean (seashells, coral, limestone ) ~203 bmt of C are released into the atmosphere respiration decomposition deforestation ~3.5% of CO 2 released fossil fuel use 23
24 Summary Projections CO 2 levels are expected to double in 25 years change in global T (?) 1-5m rise in ocean levels (but then more CO 2 dissolves!) shift in climate and crop production See National Geographic s Sept Edition 24
Gases and the atmosphere
 Gases and the atmosphere Chemical reactions in the atmosphere Ozone cycle Ozone depletion Greenhouse effect Global Warming Pollution in the troposphere 1 Gases and the atmosphere Review Composition of
Gases and the atmosphere Chemical reactions in the atmosphere Ozone cycle Ozone depletion Greenhouse effect Global Warming Pollution in the troposphere 1 Gases and the atmosphere Review Composition of
Lecture Presentation. Chapter 18. Chemistry of the Environment. James F. Kirby Quinnipiac University Hamden, CT Pearson Education, Inc.
 Lecture Presentation Chapter 18 James F. Kirby Quinnipiac University Hamden, CT Atmosphere The atmosphere consists of the troposphere, stratosphere (combined 99.9 mass %), mesosphere, and thermosphere.
Lecture Presentation Chapter 18 James F. Kirby Quinnipiac University Hamden, CT Atmosphere The atmosphere consists of the troposphere, stratosphere (combined 99.9 mass %), mesosphere, and thermosphere.
Chapter 11: Atmosphere
 To get you thinking This is our atmosphere. All life on Earth exists within this tiny protective blanket. Why is the atmosphere important to us? What do you think it does for us? Chapter 11: Atmosphere
To get you thinking This is our atmosphere. All life on Earth exists within this tiny protective blanket. Why is the atmosphere important to us? What do you think it does for us? Chapter 11: Atmosphere
Chapter 18 Chemistry of the Environment
 Chapter 18 Chemistry of the Environment Earth s Atmosphere Outer Regions of the Atmosphere Ozone in the Upper Atmosphere Chemistry of the Troposphere You will need to read the Course Pack to compliment
Chapter 18 Chemistry of the Environment Earth s Atmosphere Outer Regions of the Atmosphere Ozone in the Upper Atmosphere Chemistry of the Troposphere You will need to read the Course Pack to compliment
THE BIG ISLAND of Hawaii
 THE BIG ISLAND of Hawaii WHAT S AHEAD 18.1 EARTH S ATMOSPHERE 18.2 HUMAN ACTIVITYES AND EARTH S ATMOSPHERE 18.3 EARTH S WATER 18.4 HUMAN ACTIVITYES AND EARTH S WATER 18.5 GREEN CHEMISTRY CHAPTER 18.1 EARTH
THE BIG ISLAND of Hawaii WHAT S AHEAD 18.1 EARTH S ATMOSPHERE 18.2 HUMAN ACTIVITYES AND EARTH S ATMOSPHERE 18.3 EARTH S WATER 18.4 HUMAN ACTIVITYES AND EARTH S WATER 18.5 GREEN CHEMISTRY CHAPTER 18.1 EARTH
The September Equinox is today: Sep 23rd! It s considered the traditional end of Summer and the beginning of Fall
 More coming up in Topic #11 (class notes p 61) The September Equinox is today: Sep 23rd! It s considered the traditional end of Summer and the beginning of Fall The Sun s rays have greatest intensity right
More coming up in Topic #11 (class notes p 61) The September Equinox is today: Sep 23rd! It s considered the traditional end of Summer and the beginning of Fall The Sun s rays have greatest intensity right
1 Characteristics of the Atmosphere
 CHAPTER 22 1 Characteristics of the Atmosphere SECTION The Atmosphere KEY IDEAS As you read this section, keep these questions in mind: What are the layers of Earth s atmosphere? How has Earth s atmosphere
CHAPTER 22 1 Characteristics of the Atmosphere SECTION The Atmosphere KEY IDEAS As you read this section, keep these questions in mind: What are the layers of Earth s atmosphere? How has Earth s atmosphere
Chemistry in the Environment
 Chemistry in the Environment Section 261 Earth s Atmosphere In your textbook, read about the terms used to describe the physical and chemical properties of Earth s atmosphere Complete each statement 1
Chemistry in the Environment Section 261 Earth s Atmosphere In your textbook, read about the terms used to describe the physical and chemical properties of Earth s atmosphere Complete each statement 1
ASTRONOMY 161. Introduction to Solar System Astronomy. Class 16
 ASTRONOMY 161 Introduction to Solar System Astronomy Class 16 Earth s Atmosphere Monday, February 19 Earth s Atmosphere: Key Concepts (1) The Earth s atmosphere consists mainly of nitrogen (N 2 ) and
ASTRONOMY 161 Introduction to Solar System Astronomy Class 16 Earth s Atmosphere Monday, February 19 Earth s Atmosphere: Key Concepts (1) The Earth s atmosphere consists mainly of nitrogen (N 2 ) and
Vocabulary for Section 1.1: Pressure. Atmosphere. Troposphere. Ionosphere O zone Layer. Ultraviolet radiation. Chlorofluorocarbon (CFC) Stratosphere
 Name: Date: Module I: Section 1.1 Vocabulary and Study Guide for: Atmosphere Objectives: You will learn to identify the gases in Earth s atmosphere. You will learn to describe the structure of Earth s
Name: Date: Module I: Section 1.1 Vocabulary and Study Guide for: Atmosphere Objectives: You will learn to identify the gases in Earth s atmosphere. You will learn to describe the structure of Earth s
Topic # 7 Part II ATMOSPHERIC STRUCTURE & CHEMICAL COMPOSITION
 Topic # 7 Part II ATMOSPHERIC STRUCTURE & CHEMICAL COMPOSITION All about the GASES IN THE ATMOSPHERE, esp. GREENHOUSE GASES! Class Notes pp 37-41 REVIEW: ATMOSPHERIC STRUCTURE The changes in temperature
Topic # 7 Part II ATMOSPHERIC STRUCTURE & CHEMICAL COMPOSITION All about the GASES IN THE ATMOSPHERE, esp. GREENHOUSE GASES! Class Notes pp 37-41 REVIEW: ATMOSPHERIC STRUCTURE The changes in temperature
Energy and the Earth. Key words: Incoming Solar Radiation, Electromagnetic wave, Greenhouse effect, conduction, convection, radiation.
 S c i e n c e Energy and the Earth Key words: Incoming Solar Radiation, Electromagnetic wave, Greenhouse effect, conduction, convection, radiation. Energy transfer Heat is energy in transit from warmer
S c i e n c e Energy and the Earth Key words: Incoming Solar Radiation, Electromagnetic wave, Greenhouse effect, conduction, convection, radiation. Energy transfer Heat is energy in transit from warmer
Earth s Atmosphere. Composition & Structure
 Earth s Atmosphere Composition & Structure Essential Questions 1. What is the Earth s atmosphere made of? 2. What are the different layers of the atmosphere? 3. What causes atmospheric pressure? Historical
Earth s Atmosphere Composition & Structure Essential Questions 1. What is the Earth s atmosphere made of? 2. What are the different layers of the atmosphere? 3. What causes atmospheric pressure? Historical
How our atmosphere evolved
 Earth s Atmosphere How our atmosphere evolved I. The early atmosphere did not support life. It contained deadly gases such as Methane and Ammonia. There was very little Oxygen. II. Over time, gases were
Earth s Atmosphere How our atmosphere evolved I. The early atmosphere did not support life. It contained deadly gases such as Methane and Ammonia. There was very little Oxygen. II. Over time, gases were
Chapter 19 Global Change. Wednesday, April 18, 18
 Chapter 19 Global Change Module 62 Global Climate Change and the Greenhouse Effect After reading this module you should be able to distinguish among global change, global climate change, and global warming.
Chapter 19 Global Change Module 62 Global Climate Change and the Greenhouse Effect After reading this module you should be able to distinguish among global change, global climate change, and global warming.
3/4/2014. Air Pollution. Chapter 15 Air Pollution and Stratospheric Ozone Depletion. Major Air Pollutants. Primary Pollutants
 Air Pollution Air pollution- the introduction of chemicals, particulate matter, or microorganisms into the atmosphere at concentrations high enough to harm plants, animals, and materials such as buildings,
Air Pollution Air pollution- the introduction of chemicals, particulate matter, or microorganisms into the atmosphere at concentrations high enough to harm plants, animals, and materials such as buildings,
Chapter 18 Environmental Chemistry
 Chapter 18 Environmental Chemistry I) Earth s Atmosphere A) 4 regions based on temperature 1) Troposphere Temp. dec. w. inc. altitude - min. of ~ 215 K at 10 km (at tropopause) 2) Stratosphere Temp. inc.
Chapter 18 Environmental Chemistry I) Earth s Atmosphere A) 4 regions based on temperature 1) Troposphere Temp. dec. w. inc. altitude - min. of ~ 215 K at 10 km (at tropopause) 2) Stratosphere Temp. inc.
Climate Change Vocabulary Global Challenges for the 21 st Century Tony Del Vecchio, M.Ed. Atmosphere
 Atmosphere The mixture of gases surrounding the Earth. The Earth's atmosphere consists of about 79.1% nitrogen (by volume), 20.9% oxygen, 0.036% carbon dioxide and trace amounts of other gases. The atmosphere
Atmosphere The mixture of gases surrounding the Earth. The Earth's atmosphere consists of about 79.1% nitrogen (by volume), 20.9% oxygen, 0.036% carbon dioxide and trace amounts of other gases. The atmosphere
GLOBAL WARMING COMPUTER LAB
 GLOBAL WARMING COMPUTER LAB A COMPUTER SIMULATION PROGRAM ON TEMPERATURE CHANGE AND SEA LEVEL RISING After performing this computer simulation lab you will be able to: 1) understand the greenhouse effect
GLOBAL WARMING COMPUTER LAB A COMPUTER SIMULATION PROGRAM ON TEMPERATURE CHANGE AND SEA LEVEL RISING After performing this computer simulation lab you will be able to: 1) understand the greenhouse effect
TODAY: TOPIC #6 WRAP UP!! Atmospheric Structure & Composition
 TODAY: TOPIC #6 WRAP UP!! Atmospheric Structure & Composition There s one more thing to correct in our the depiction of incoming Solar....... the atmosphere is NOT totally TRANSPARENT to INCOMING Solar
TODAY: TOPIC #6 WRAP UP!! Atmospheric Structure & Composition There s one more thing to correct in our the depiction of incoming Solar....... the atmosphere is NOT totally TRANSPARENT to INCOMING Solar
Chapter 15 Air Pollution and Stratospheric Ozone Depletion
 Chapter 15 Air Pollution and Stratospheric Ozone Depletion Air Pollution Air pollution- the introduction of chemicals, particulate matter, or microorganisms into the atmosphere at concentrations high enough
Chapter 15 Air Pollution and Stratospheric Ozone Depletion Air Pollution Air pollution- the introduction of chemicals, particulate matter, or microorganisms into the atmosphere at concentrations high enough
Atmospheric Chemistry (Option 1B)
 Atmospheric Chemistry (Option 1B) Introduction Option 1B consists of the following subsections: 1B.1 Oxygen 1B.2 Nitrogen 1B.3 Carbon Dioxide 1B.4 Atmospheric Pollution 1B.5 The Ozone Layer Some chemistry
Atmospheric Chemistry (Option 1B) Introduction Option 1B consists of the following subsections: 1B.1 Oxygen 1B.2 Nitrogen 1B.3 Carbon Dioxide 1B.4 Atmospheric Pollution 1B.5 The Ozone Layer Some chemistry
Topic # 7 ATMOSPHERIC STRUCTURE & CHEMICAL COMPOSITION
 Topic # 7 ATMOSPHERIC STRUCTURE & CHEMICAL COMPOSITION All about the GASES IN THE ATMOSPHERE, esp. GREENHOUSE GASES! Class Notes pp 37-41 OBJECTIVES: To understand: -- the VERTICALSTRUCTURE of the atmosphere
Topic # 7 ATMOSPHERIC STRUCTURE & CHEMICAL COMPOSITION All about the GASES IN THE ATMOSPHERE, esp. GREENHOUSE GASES! Class Notes pp 37-41 OBJECTIVES: To understand: -- the VERTICALSTRUCTURE of the atmosphere
Acid deposition accumulation of potential acid-forming particles on a surface acids can result from natural causes
 1 Air Quality Issues: Part 2 - Acid Deposition, Greenhouse Gases EVPP 111 Lecture Dr. Largen 2 Air Quality Issues Air Pollution Indoor Air Pollution Acid Deposition Greenhouse Gases & Global Warming 3
1 Air Quality Issues: Part 2 - Acid Deposition, Greenhouse Gases EVPP 111 Lecture Dr. Largen 2 Air Quality Issues Air Pollution Indoor Air Pollution Acid Deposition Greenhouse Gases & Global Warming 3
Thursday Sep 25th SIT WITH YOUR GROUP TODAY! Topic # 6 Atmospheric Structure & Chemical Composition
 Thursday Sep 25th SIT WITH YOUR GROUP TODAY! Topic # 6 Atmospheric Structure & Chemical Composition Self Test 4 & RQ-4 on The Laws of Thermodynamics are now posted. The readings that will prepare you for
Thursday Sep 25th SIT WITH YOUR GROUP TODAY! Topic # 6 Atmospheric Structure & Chemical Composition Self Test 4 & RQ-4 on The Laws of Thermodynamics are now posted. The readings that will prepare you for
Earth's Atmosphere. Atmospheric Layers. Atmospheric Layers
 Earth's Atmosphere Today we will talk about the part of Earth that is most important to our survival - the atmosphere Earth's atmosphere is unique in the Solar System and has changed greatly over time
Earth's Atmosphere Today we will talk about the part of Earth that is most important to our survival - the atmosphere Earth's atmosphere is unique in the Solar System and has changed greatly over time
Chapter 5. The Earth s Atmosphere
 Chapter 5 The Earth s Atmosphere Layers of the Earth Earth largest of the inner planets Gravity strong enough to hold gases. Lots of spheres Equator divided the Earth into two hemispheres Lithosphere-
Chapter 5 The Earth s Atmosphere Layers of the Earth Earth largest of the inner planets Gravity strong enough to hold gases. Lots of spheres Equator divided the Earth into two hemispheres Lithosphere-
Carbon Cycle LT 1-6 Review. LT 1 I can describe the three primary processes of the biological (short) carbon cycle.
 Carbon Cycle LT 1-6 Review Name Date LT 1 I can describe the three primary processes of the biological (short) carbon cycle. 1. What is the Law of Conservation of Matter and how does it apply to the carbon
Carbon Cycle LT 1-6 Review Name Date LT 1 I can describe the three primary processes of the biological (short) carbon cycle. 1. What is the Law of Conservation of Matter and how does it apply to the carbon
Greenhouse Effect. How we stay warm
 Greenhouse Effect How we stay warm The Sun s energy reaches Earth through Radiation (heat traveling through Space) How much solar radiation reaches Earth? The Earth s surface only absorbs 51% of incoming
Greenhouse Effect How we stay warm The Sun s energy reaches Earth through Radiation (heat traveling through Space) How much solar radiation reaches Earth? The Earth s surface only absorbs 51% of incoming
4/12. There is so much pollution in the air now that if it weren t for our lungs there d be no place to put it all. Robert Orben
 4/12 There is so much pollution in the air now that if it weren t for our lungs there d be no place to put it all. Robert Orben Chapter 15 Air Pollution and Stratospheric Ozone Depletion Air Pollution
4/12 There is so much pollution in the air now that if it weren t for our lungs there d be no place to put it all. Robert Orben Chapter 15 Air Pollution and Stratospheric Ozone Depletion Air Pollution
1. The diagram below shows a greenhouse.
 1. The diagram below shows a greenhouse. 5. A gradual increase in atmospheric carbon dioxide would warm Earth s because carbon dioxide is a A) poor reflector of ultraviolet radiation B) good reflector
1. The diagram below shows a greenhouse. 5. A gradual increase in atmospheric carbon dioxide would warm Earth s because carbon dioxide is a A) poor reflector of ultraviolet radiation B) good reflector
Atmospheric Chemistry
 Atmospheric Chemistry Research Dr. Husam T. Majeed Department of Atmospheric Sciences 2nd Year Class First Semester 2018-2019 Course Syllabus 1. Introduction & Review Atmospheric Chemistry Box Models and
Atmospheric Chemistry Research Dr. Husam T. Majeed Department of Atmospheric Sciences 2nd Year Class First Semester 2018-2019 Course Syllabus 1. Introduction & Review Atmospheric Chemistry Box Models and
Environmental Science Std.-9 Chp.7 Atmosphere and Climate
 Environmental Science Std.-9 Chp.7 Atmosphere and Climate 2018-19 Q.1. Name the layers of the atmosphere. Troposphere Stratosphere Mesosphere Thermosphere Exosphere GREENHOUSE EARTH: Q.2. (a) What is the
Environmental Science Std.-9 Chp.7 Atmosphere and Climate 2018-19 Q.1. Name the layers of the atmosphere. Troposphere Stratosphere Mesosphere Thermosphere Exosphere GREENHOUSE EARTH: Q.2. (a) What is the
Atmosphere. Earth s Atmosphere
 chapter 15 3 Atmosphere section 1 Earth s Atmosphere Before You Read Imagine you are on a spaceship looking down at Earth. Would the view be perfectly clear? What do you think you might see surrounding
chapter 15 3 Atmosphere section 1 Earth s Atmosphere Before You Read Imagine you are on a spaceship looking down at Earth. Would the view be perfectly clear? What do you think you might see surrounding
ATMOSPHERE GASES, LAYERS, TEMPERATURES
 ATMOSPHERE GASES, LAYERS, TEMPERATURES 2/27/2007 1 EARTH S S ATMOSPHERE FROM SPACE 2/27/2007 Vicki Drake, SMC 2 COMPONENTS OF THE GLOBAL CLIMATE SYSTEM 2/27/2007 Vicki Drake, SMC 3 WHAT IS THE ATMOSPHERE?
ATMOSPHERE GASES, LAYERS, TEMPERATURES 2/27/2007 1 EARTH S S ATMOSPHERE FROM SPACE 2/27/2007 Vicki Drake, SMC 2 COMPONENTS OF THE GLOBAL CLIMATE SYSTEM 2/27/2007 Vicki Drake, SMC 3 WHAT IS THE ATMOSPHERE?
The Earth s Atmosphere-I. GEOL 1350: Introduction To Meteorology
 The Earth s Atmosphere-I GEOL 1350: Introduction To Meteorology 1 Overview What is the composition of Atmosphere? How did the atmosphere arrive at its current state? 2 Earth s Atmosphere Earth s atmosphere
The Earth s Atmosphere-I GEOL 1350: Introduction To Meteorology 1 Overview What is the composition of Atmosphere? How did the atmosphere arrive at its current state? 2 Earth s Atmosphere Earth s atmosphere
Lecture 11: Global Warming. Human Acticities. Natural Climate Changes. Global Warming: Natural or Man-Made CO 2 CH 4
 Lecture 11: Global Warming Human Acticities CO 2 CH 4 The initial appearance of human species: last 100,000 to 200,000 years Development of the first civilization: the last 10,000 years What is the sensitivity
Lecture 11: Global Warming Human Acticities CO 2 CH 4 The initial appearance of human species: last 100,000 to 200,000 years Development of the first civilization: the last 10,000 years What is the sensitivity
Biogeochemical Cycles
 Biogeochemical Cycles Biogeochemical Cycles refers to the cycling of materials between living things and the environment. Text Pages 50 51, 62 69 1 The Oxygen Cycle the movement of oxygen between the atmosphere
Biogeochemical Cycles Biogeochemical Cycles refers to the cycling of materials between living things and the environment. Text Pages 50 51, 62 69 1 The Oxygen Cycle the movement of oxygen between the atmosphere
Casterlin Environmental Systems pg. 1
 s of the Earth's Atmosphere The atmosphere is divided into five layers. It is thickest near the surface and thins out with height until it eventually merges with space. 1. The troposphere is the first
s of the Earth's Atmosphere The atmosphere is divided into five layers. It is thickest near the surface and thins out with height until it eventually merges with space. 1. The troposphere is the first
BIOGEOCHEMICAL CYCLES INTRODUCTION THE CYCLING PROCESS TWO CYCLES: CARBON CYCLE NITROGEN CYCLE HUMAN IMPACTS GLOBAL WARMING AQUATIC EUTROPHICATION
 BIOGEOCHEMICAL CYCLES INTRODUCTION THE CYCLING PROCESS TWO CYCLES: CARBON CYCLE NITROGEN CYCLE HUMAN IMPACTS GLOBAL WARMING AQUATIC EUTROPHICATION BIOGEOCHEMICAL CYCLES: The RECYCLING of MATERIALS through
BIOGEOCHEMICAL CYCLES INTRODUCTION THE CYCLING PROCESS TWO CYCLES: CARBON CYCLE NITROGEN CYCLE HUMAN IMPACTS GLOBAL WARMING AQUATIC EUTROPHICATION BIOGEOCHEMICAL CYCLES: The RECYCLING of MATERIALS through
4.4 CLIMATE CHANGE. Concentrations of gases in the atmosphere affect climates experiences at the Earth s surface
 4.4 CLIMATE CHANGE Concentrations of gases in the atmosphere affect climates experiences at the Earth s surface Greenhouse Gases (GHG) Carbon dioxide and water vapour are the most significant greenhouse
4.4 CLIMATE CHANGE Concentrations of gases in the atmosphere affect climates experiences at the Earth s surface Greenhouse Gases (GHG) Carbon dioxide and water vapour are the most significant greenhouse
Assessing Rates of Climate Change
 Assessing Rates of Climate Change What is the difference between climate and weather Climate = average weather over many years (ex: 30 year averages) Climate forcings (things that affect global temperatures)
Assessing Rates of Climate Change What is the difference between climate and weather Climate = average weather over many years (ex: 30 year averages) Climate forcings (things that affect global temperatures)
The Effects of Volcano-Induced Ozone Depletion on Short-Lived Climate Forcing in the Arctic
 C53C-0852 The Effects of Volcano-Induced Ozone Depletion on Short-Lived Climate Forcing in the Arctic Peter L. Ward US Geological Survey Retired Teton Tectonics Jackson, WY 307-733-3664 cell 307-413-4055
C53C-0852 The Effects of Volcano-Induced Ozone Depletion on Short-Lived Climate Forcing in the Arctic Peter L. Ward US Geological Survey Retired Teton Tectonics Jackson, WY 307-733-3664 cell 307-413-4055
Earth and Space Science (Earth's Atmosphere) Grade 7 Science Grade 7 Science Start Date: December 02, 2013 End Date : December 20, 2013
 Unit Overview Atmospheric properties Content Elaborations The atmosphere has different properties at different elevations and contains a mixture of gases that cycle through the lithosphere, biosphere,
Unit Overview Atmospheric properties Content Elaborations The atmosphere has different properties at different elevations and contains a mixture of gases that cycle through the lithosphere, biosphere,
Some resources (more websites later)
 Some resources (more websites later) Intergovernmental Panel Climate Change 2001: The Scientific Basis at http://www.ipcc.ch/pub/reports.htm John Houghton Global Warming - the complete briefing Cambridge
Some resources (more websites later) Intergovernmental Panel Climate Change 2001: The Scientific Basis at http://www.ipcc.ch/pub/reports.htm John Houghton Global Warming - the complete briefing Cambridge
G-5 ACTIVITY ON VOLCANISM & CLIMATE THE ANSWERS!
 G-5 ACTIVITY ON VOLCANISM & CLIMATE THE ANSWERS! #1. List 4 reasons why Tambora in 1815 resulted in the largest GLOBAL cooling: #1 Low latitude eruption both hemispheres #2 Large amount of eruptive material
G-5 ACTIVITY ON VOLCANISM & CLIMATE THE ANSWERS! #1. List 4 reasons why Tambora in 1815 resulted in the largest GLOBAL cooling: #1 Low latitude eruption both hemispheres #2 Large amount of eruptive material
Environmental Science Tuesday,&September&8,&2015&
 Environmental Science Tuesday,&September&8,&2015& Do-Now: Dynamic Earth Vocab. 1. Begin(your(Ch.3(Dynamic(Earth(vocabulary( Recall:(you(may(use(the(vocabulary(builder(WS((in( the(cabinet)(or(you(may(use(flashcards((
Environmental Science Tuesday,&September&8,&2015& Do-Now: Dynamic Earth Vocab. 1. Begin(your(Ch.3(Dynamic(Earth(vocabulary( Recall:(you(may(use(the(vocabulary(builder(WS((in( the(cabinet)(or(you(may(use(flashcards((
Planetary Energy Balance
 Planetary Energy Balance Overview of Planetary Energy Balance Energy coming into the Earth s atmosphere from the sun is always in balance with the energy leaving Earth s atmosphere going back out into
Planetary Energy Balance Overview of Planetary Energy Balance Energy coming into the Earth s atmosphere from the sun is always in balance with the energy leaving Earth s atmosphere going back out into
The Earth s Global Energy Balance
 The Earth s Global Energy Balance Electromagnetic Radiation Insolation over the Globe World Latitude Zones Composition of the Atmosphere Sensible Heat and Latent Heat Transfer The Global Energy System
The Earth s Global Energy Balance Electromagnetic Radiation Insolation over the Globe World Latitude Zones Composition of the Atmosphere Sensible Heat and Latent Heat Transfer The Global Energy System
Chapter 17: Atmospheric Science and Air Pollution I. Central Case: The 1952 Killer Smog of London
 Chapter 17: Atmospheric Science and Air Pollution I. Central Case: The 1952 Killer Smog of London A. Thick smog first settled over the city on December 5, 1952, when many residents stoked: B. A wind finally
Chapter 17: Atmospheric Science and Air Pollution I. Central Case: The 1952 Killer Smog of London A. Thick smog first settled over the city on December 5, 1952, when many residents stoked: B. A wind finally
Earth Science Lesson Plan Quarter 2, Week 1, Day 1
 Earth Science Lesson Plan Quarter 2, Week 1, Day 1 1 Outcomes for Today Standard Focus: Earth Sciences 4.c Students know the different atmospheric gases that absorb the Earth s thermal radiation and the
Earth Science Lesson Plan Quarter 2, Week 1, Day 1 1 Outcomes for Today Standard Focus: Earth Sciences 4.c Students know the different atmospheric gases that absorb the Earth s thermal radiation and the
L 18 Thermodynamics [3] Thermodynamics- review. electromagnetic spectrum. radiation. What produces thermal radiation?
![L 18 Thermodynamics [3] Thermodynamics- review. electromagnetic spectrum. radiation. What produces thermal radiation? L 18 Thermodynamics [3] Thermodynamics- review. electromagnetic spectrum. radiation. What produces thermal radiation?](/thumbs/87/96937790.jpg) L 18 Thermodynamics [3] Heat transfer processes convection conduction Physics of the atmosphere the ozone layer Greenhouse effect climate change Thermodynamics- review Thermodynamics is the science dealing
L 18 Thermodynamics [3] Heat transfer processes convection conduction Physics of the atmosphere the ozone layer Greenhouse effect climate change Thermodynamics- review Thermodynamics is the science dealing
Triple Beam Balance: add the three together: 700g + 20g + 2.9g = 722.9g Metric base unit for mass is gram.
 6 th Grade 2 nd Nine Week CSA Study Guide 2015-16 SOL 6.1b: Make precise and consistent measurements and estimations. Graduated Cylinder: read from the lowest part of the water line curve. Reading: 56ml
6 th Grade 2 nd Nine Week CSA Study Guide 2015-16 SOL 6.1b: Make precise and consistent measurements and estimations. Graduated Cylinder: read from the lowest part of the water line curve. Reading: 56ml
2 Atmospheric Heating
 CHAPTER 15 2 Atmospheric Heating SECTION The Atmosphere BEFORE YOU READ After you read this section, you should be able to answer these questions: How does energy travel from the sun to Earth? What are
CHAPTER 15 2 Atmospheric Heating SECTION The Atmosphere BEFORE YOU READ After you read this section, you should be able to answer these questions: How does energy travel from the sun to Earth? What are
Questions 1 4 refer to the diagram of the earth s atmosphere shown above. 3. The section of the atmosphere responsible for our daily weather.
 Unit 6 Practice Test Questions 1 4 refer to the diagram of the earth s atmosphere shown above 1. The ozone layer is found at this location. 2. The atmosphere is at its warmest point at this location. 3.
Unit 6 Practice Test Questions 1 4 refer to the diagram of the earth s atmosphere shown above 1. The ozone layer is found at this location. 2. The atmosphere is at its warmest point at this location. 3.
Name: Class: Date: 6. Most air pollution is produced by a. thermal inversions. c. ozone layer depletion. b. fuel burning. d. volcanic eruptions.
 Name: Class: Date: Air Test Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of the following is often used to remove poisonous gases from industrial
Name: Class: Date: Air Test Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of the following is often used to remove poisonous gases from industrial
How is the atmosphere different from outer space? a mixture of gases that surrounds the Earth
 Chapter 15 Atmosphere Section 1 Objectives Describe the composition of Earth's atmosphere. Explain why air pressure changes with altitude. Explain how air temperature changes with atmospheric composition.
Chapter 15 Atmosphere Section 1 Objectives Describe the composition of Earth's atmosphere. Explain why air pressure changes with altitude. Explain how air temperature changes with atmospheric composition.
Global Warming Science Solar Radiation
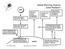 SUN Ozone and Oxygen absorb 190-290 nm. Latent heat from the surface (evaporation/ condensation) Global Warming Science Solar Radiation Turbulent heat from the surface (convection) Some infrared radiation
SUN Ozone and Oxygen absorb 190-290 nm. Latent heat from the surface (evaporation/ condensation) Global Warming Science Solar Radiation Turbulent heat from the surface (convection) Some infrared radiation
Lecture 2: Greenhouse Gases - Basic Background on Atmosphere - GHG Emission and Concentration Rise - California Regulation (AB32)
 Lecture 2: Greenhouse Gases - Basic Background on Atmosphere - GHG Emission and Concentration Rise - California Regulation (AB32) METR 113/ENVS 113 Spring Semester 2011 February 15, 2011 Suggested Reading
Lecture 2: Greenhouse Gases - Basic Background on Atmosphere - GHG Emission and Concentration Rise - California Regulation (AB32) METR 113/ENVS 113 Spring Semester 2011 February 15, 2011 Suggested Reading
Global Climate Change. The sky is falling! The sky is falling!
 Global Climate Change The sky is falling! The sky is falling! 1 Global Climate Change Radiative Equilibrium, Solar and Earth Radiation Atmospheric Greenhouse Effect Greenhouse Gases Global Climate Change
Global Climate Change The sky is falling! The sky is falling! 1 Global Climate Change Radiative Equilibrium, Solar and Earth Radiation Atmospheric Greenhouse Effect Greenhouse Gases Global Climate Change
9th Period Environmental Science Chapter 15: The Atmosphere
 Section 15.1: Earth s Atmosphere 9th Period Environmental Science Chapter 15: The Atmosphere Properties of the Atmosphere: nitrogen, oxygen, water vapor and 1% other gases. air pressure is higher at the
Section 15.1: Earth s Atmosphere 9th Period Environmental Science Chapter 15: The Atmosphere Properties of the Atmosphere: nitrogen, oxygen, water vapor and 1% other gases. air pressure is higher at the
Chapter 15. Atmosphere Notes
 Chapter 15 Atmosphere Notes The Air Around You Weather: The condition of the Earth s atmosphere at a particular time and place The Air Around You Atmosphere: the envelope of gases that surround the planet
Chapter 15 Atmosphere Notes The Air Around You Weather: The condition of the Earth s atmosphere at a particular time and place The Air Around You Atmosphere: the envelope of gases that surround the planet
Introduction to Photolysis AOSC 433/633 & CHEM 433/633 Ross Salawitch. Importance of Radicals
 Introduction to Photolysis AOSC 433/633 & CHEM 433/633 Ross Salawitch Class Web Site: http://www.atmos.umd.edu/~rjs/class/spr2013 Lecture 10 5 March 2013 1 Importance of Radicals With a few exceptions,
Introduction to Photolysis AOSC 433/633 & CHEM 433/633 Ross Salawitch Class Web Site: http://www.atmos.umd.edu/~rjs/class/spr2013 Lecture 10 5 March 2013 1 Importance of Radicals With a few exceptions,
Stratospheric Ozone. Science Concepts. Stratospheric Ozone. Ozone Chemistry Creation Process Destruction Processes Equilibrium.
 Stratospheric Ozone 12-1 Stratospheric Ozone Ozone Chlorofluorocarbons (CFCs) CFCs Science Concepts Ozone Chemistry Creation Process Destruction Processes Equilibrium CFC Chemistry Antarctic Ozone Hole
Stratospheric Ozone 12-1 Stratospheric Ozone Ozone Chlorofluorocarbons (CFCs) CFCs Science Concepts Ozone Chemistry Creation Process Destruction Processes Equilibrium CFC Chemistry Antarctic Ozone Hole
TROPICS: insolation high year round, high sun angle and ~ constant duration
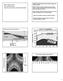 GE 101, February 6, 14 Finish insolation variation Global environmental issues associated with insolation TRPICS: insolation high year round, high sun angle and ~ constant duration MID-LATITUDES: insolation
GE 101, February 6, 14 Finish insolation variation Global environmental issues associated with insolation TRPICS: insolation high year round, high sun angle and ~ constant duration MID-LATITUDES: insolation
Earth s Atmosphere Lecture 14 3/6/2014
 Earth s Atmosphere Lecture 14 3/6/2014 MRS 1 Due Tuesday Second exam will be postponed until after spring break The sun drives the climate of Earth http://www.spaceweather.com/images2002/18mar02/cme_c3_big.gif
Earth s Atmosphere Lecture 14 3/6/2014 MRS 1 Due Tuesday Second exam will be postponed until after spring break The sun drives the climate of Earth http://www.spaceweather.com/images2002/18mar02/cme_c3_big.gif
Chapter 13 The Earths Atmosphere
 Chapter 3 The Earths Atmosphere Name: Class: Date: Time: 79 minutes Marks: 79 marks Comments: Page of 28 The bar chart shows some of the gases in the atmospheres of Earth today and Mars today. (b) Complete
Chapter 3 The Earths Atmosphere Name: Class: Date: Time: 79 minutes Marks: 79 marks Comments: Page of 28 The bar chart shows some of the gases in the atmospheres of Earth today and Mars today. (b) Complete
Name SID Number Final VERSION A
 Name SID Number Final VERSION A 1. The main source of energy, that is heating planet earth is (1) renewable energy (2) natural gas (3) the sun (4) oil (5) coal 2. Which of the following mainly causes acid
Name SID Number Final VERSION A 1. The main source of energy, that is heating planet earth is (1) renewable energy (2) natural gas (3) the sun (4) oil (5) coal 2. Which of the following mainly causes acid
Chemistry 471/671. Global Climate Change
 Chemistry 471/671 Global Climate Change The Greenhouse Effect and Global Warming These are NOT the same thing! To begin with, let s make the distinction 2 Earth Average temperature 15 C Average pressure:
Chemistry 471/671 Global Climate Change The Greenhouse Effect and Global Warming These are NOT the same thing! To begin with, let s make the distinction 2 Earth Average temperature 15 C Average pressure:
Answers to the Questions Posed by Judge Alsup
 Answers to the Questions Posed by Judge Alsup Don Wuebbles Department of Atmospheric Sciences University of Illinois SF March 21, 2018 Date Name of Meeting 1 Addressing Some Questions for the Tutorial
Answers to the Questions Posed by Judge Alsup Don Wuebbles Department of Atmospheric Sciences University of Illinois SF March 21, 2018 Date Name of Meeting 1 Addressing Some Questions for the Tutorial
READING QUESTIONS: Global Climate Change GEOL /WI. 2. Fill in the blanks in the following sentences from the textbook (p.
 READING QUESTIONS: Global Climate Change GEOL 131 18/WI NAME DUE: Tuesday, April 24 57pts Climate and Geology (p. 382) 1. List the five spheres included in the climate system. (5 pts) Detecting Climate
READING QUESTIONS: Global Climate Change GEOL 131 18/WI NAME DUE: Tuesday, April 24 57pts Climate and Geology (p. 382) 1. List the five spheres included in the climate system. (5 pts) Detecting Climate
6-4 Charting a Course for the Future Slide 2 of 30
 6-4 Charting a Course for the Future 2 of 30 Ozone Depletion What are two types of global change of concern to biologists? the ozone layer high in the atmosphere the global climate system 3 of 30 Ozone
6-4 Charting a Course for the Future 2 of 30 Ozone Depletion What are two types of global change of concern to biologists? the ozone layer high in the atmosphere the global climate system 3 of 30 Ozone
Climate Change and Ozone Depletion
 Climate Change and Ozone Depletion Troposphere 78% N 2, 21% O 2, 0.09% Ar,, 0.035% CO 2 Global warming occurs here Stratosphere global sunscreen (ozone layer) Keeps 95% of the sun s s harmful UV radiation
Climate Change and Ozone Depletion Troposphere 78% N 2, 21% O 2, 0.09% Ar,, 0.035% CO 2 Global warming occurs here Stratosphere global sunscreen (ozone layer) Keeps 95% of the sun s s harmful UV radiation
Chapter 4: The Global Energy System
 Discovering Physical Geography Third Edition by Alan Arbogast Chapter 4: The Global Energy System The Electromagnetic Spectrum and Solar Energy Solar Energy as Radiation Electromagnetic energy transmitted
Discovering Physical Geography Third Edition by Alan Arbogast Chapter 4: The Global Energy System The Electromagnetic Spectrum and Solar Energy Solar Energy as Radiation Electromagnetic energy transmitted
Case 3:17-cv WHA Document Filed 03/23/18 Page 1 of 14 EXHIBIT 4
 Case 3:17-cv-06011-WHA Document 183-4 Filed 03/23/18 Page 1 of 14 EXHIBIT 4 Case 3:17-cv-06011-WHA Document 183-4 Filed 03/23/18 Page 2 of 14 Answers to the Questions Posed by Judge Alsup Don Wuebbles
Case 3:17-cv-06011-WHA Document 183-4 Filed 03/23/18 Page 1 of 14 EXHIBIT 4 Case 3:17-cv-06011-WHA Document 183-4 Filed 03/23/18 Page 2 of 14 Answers to the Questions Posed by Judge Alsup Don Wuebbles
Funding provided by NOAA Sectoral Applications Research Project ALTERING CLIMATE. Basic Climatology Oklahoma Climatological Survey
 Funding provided by NOAA Sectoral Applications Research Project ALTERING CLIMATE Basic Climatology Oklahoma Climatological Survey ATMOSPHERIC POLLUTANTS Acid Rain Acid Rain is the precipitation that carries
Funding provided by NOAA Sectoral Applications Research Project ALTERING CLIMATE Basic Climatology Oklahoma Climatological Survey ATMOSPHERIC POLLUTANTS Acid Rain Acid Rain is the precipitation that carries
WELCOME TO PERIOD 12: CONSEQUENCES OF USING CHEMICAL ENERGY Homework #11 is due today.
 WELCOME TO PERIOD 12: CONSEQUENCES OF USING CHEMICAL ENERGY Homework #11 is due today. Midterm 1 grade curve: A 29-33: 17% (of 166 students) B 25-28: 34% C 20-24: 32% D 15-19: 14% E 0-14: 3% Average score
WELCOME TO PERIOD 12: CONSEQUENCES OF USING CHEMICAL ENERGY Homework #11 is due today. Midterm 1 grade curve: A 29-33: 17% (of 166 students) B 25-28: 34% C 20-24: 32% D 15-19: 14% E 0-14: 3% Average score
Earth s Atmosphere Lecture 14 2/28/2013
 Earth s Atmosphere Lecture 14 2/28/2013 MRS 1 Due Tuesday The Earth s atmosphere has changed substantially over our planet s history First gases surrounding Earth were originally hydrogen and helium (during
Earth s Atmosphere Lecture 14 2/28/2013 MRS 1 Due Tuesday The Earth s atmosphere has changed substantially over our planet s history First gases surrounding Earth were originally hydrogen and helium (during
Convection. L 18 Thermodynamics [3] Conduction. heat conduction. radiation
![Convection. L 18 Thermodynamics [3] Conduction. heat conduction. radiation Convection. L 18 Thermodynamics [3] Conduction. heat conduction. radiation](/thumbs/72/66535645.jpg) L 18 Thermodynamics [3] Heat transfer processes convection conduction Thermodynamics of the atmosphere Greenhouse effect and climate change Effect of the ozone layer Convection heat is transferred from
L 18 Thermodynamics [3] Heat transfer processes convection conduction Thermodynamics of the atmosphere Greenhouse effect and climate change Effect of the ozone layer Convection heat is transferred from
ChE 505 Chapter 1N revised 01/17/13
 ENVIRONMENTAL REACTION ENGINEERING 1.1 Introduction Chemical reactions play a key role in generation of pollutants (e.g. combustion of fossil fuels) as well as in pollution abatement (e.g. automobile exhaust
ENVIRONMENTAL REACTION ENGINEERING 1.1 Introduction Chemical reactions play a key role in generation of pollutants (e.g. combustion of fossil fuels) as well as in pollution abatement (e.g. automobile exhaust
Name SID Number Final VERSION B
 Name SID Number Final VERSION B 1. Which of the following is true: (1) The world population is about 15 billion people (2) Half of the energy used on earth today comes from fossil fuel (3) Reducing all
Name SID Number Final VERSION B 1. Which of the following is true: (1) The world population is about 15 billion people (2) Half of the energy used on earth today comes from fossil fuel (3) Reducing all
Name SID Number Final VERSION B
 Name SID Number Final VERSION B 1. Greenhouses. (1) absorb infrared and absorb ultraviolet radiation (2) does not absorb infrared and does not absorb ultraviolet radiation (3) does not absorb infrared
Name SID Number Final VERSION B 1. Greenhouses. (1) absorb infrared and absorb ultraviolet radiation (2) does not absorb infrared and does not absorb ultraviolet radiation (3) does not absorb infrared
Page 2. Q1.Greenhouse gases affect the temperature of the Earth. Which gas is a greenhouse gas? Tick one box. Argon. Methane. Nitrogen.
 Q1.Greenhouse gases affect the temperature of the Earth. (a) Which gas is a greenhouse gas? Tick one box. Argon Methane Nitrogen Oxygen (b) An increase in global temperature will cause climate change.
Q1.Greenhouse gases affect the temperature of the Earth. (a) Which gas is a greenhouse gas? Tick one box. Argon Methane Nitrogen Oxygen (b) An increase in global temperature will cause climate change.
Climate Change. Greenhouse Effect & Global Warming
 Climate Change What is climate change Greenhouse Effect & Global Warming Global Warming = World wide increase in average Temp. Cause = greenhouse effect 1 Some would argue the Global Warming trend is natural
Climate Change What is climate change Greenhouse Effect & Global Warming Global Warming = World wide increase in average Temp. Cause = greenhouse effect 1 Some would argue the Global Warming trend is natural
Earth Systems and Interactions
 CHAPTER The Earth System Earth Systems and Interactions What do you think? Read the three statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree
CHAPTER The Earth System Earth Systems and Interactions What do you think? Read the three statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree
Introduction. Introduction. Introduction. Outline Last IPCC report : 2001 Last IPCC report :
 Introduction Greenhouse Gases & Climate Change Laurent Bopp LSCE, Paris When did the story start? ¾1827 Fourier hypothesizes greenhouse effect ¾1860 Tyndal identifies CO2 and water vapor as heat trapping
Introduction Greenhouse Gases & Climate Change Laurent Bopp LSCE, Paris When did the story start? ¾1827 Fourier hypothesizes greenhouse effect ¾1860 Tyndal identifies CO2 and water vapor as heat trapping
Green Chemistry Five ways in which the Chemical industry can become Greener Changing to renewable sources Use of alternatives to hazardous chemicals
 Green Chemistry Green Chemistry refers to the processes in the chemical industry that are being reinvented to make them more sustainable. The term sustain means to keep going. If we use resources faster
Green Chemistry Green Chemistry refers to the processes in the chemical industry that are being reinvented to make them more sustainable. The term sustain means to keep going. If we use resources faster
Introduction. August 27, 2014
 Introduction August 27, 2014 Atmospheric sciences focuses on understanding the atmosphere of the earth and other planets. The motivations for studying atmospheric sciences are largely: weather forecasting,
Introduction August 27, 2014 Atmospheric sciences focuses on understanding the atmosphere of the earth and other planets. The motivations for studying atmospheric sciences are largely: weather forecasting,
ATM S 211 Final Examination June 4, 2007
 ATM S 211 Final Examination June 4, 2007 Name This examination consists of a total of 100 points. In each of the first two sections, you have a choice of which questions to answer. Please note that you
ATM S 211 Final Examination June 4, 2007 Name This examination consists of a total of 100 points. In each of the first two sections, you have a choice of which questions to answer. Please note that you
Atmosphere and Climate Change Section 1. Chapter 13 Atmosphere and Climate Change Section 1: Climate and Climate Change DAY ONE
 Chapter 13 Atmosphere and Climate Change Section 1: Climate and Climate Change DAY ONE Climate Climate is the average weather conditions in an area over a long period of time. Climate is determined by
Chapter 13 Atmosphere and Climate Change Section 1: Climate and Climate Change DAY ONE Climate Climate is the average weather conditions in an area over a long period of time. Climate is determined by
LABEL AND EXPLAIN THE PROCESSES AT EACH NUMBER IN THE DIAGRAM ABOVE
 HYDROLOGIC CYCLE 3 4 5 2 5 1B 6B 1A 6A 7 6C LABEL AND EXPLAIN THE PROCESSES AT EACH NUMBER IN THE DIAGRAM ABOVE 1A. Evaporation of water from oceans 1B. Evaporation of water from land sources (water and
HYDROLOGIC CYCLE 3 4 5 2 5 1B 6B 1A 6A 7 6C LABEL AND EXPLAIN THE PROCESSES AT EACH NUMBER IN THE DIAGRAM ABOVE 1A. Evaporation of water from oceans 1B. Evaporation of water from land sources (water and
Environmental Science Diagnostic Practice Exam
 Name: Period: Environmental Science Diagnostic Practice Exam * This test will be used to determine your current levels of understanding on Environmental Science topics. You will be graded based on effort
Name: Period: Environmental Science Diagnostic Practice Exam * This test will be used to determine your current levels of understanding on Environmental Science topics. You will be graded based on effort
CALIFORNIA EDUCATION AND THE ENVIRONMENT INITIATIVE
 Water Vapor: A GHG Lesson 3 page 1 of 2 Water Vapor: A GHG Water vapor in our atmosphere is an important greenhouse gas (GHG). On a cloudy day we can see evidence of the amount of water vapor in our atmosphere.
Water Vapor: A GHG Lesson 3 page 1 of 2 Water Vapor: A GHG Water vapor in our atmosphere is an important greenhouse gas (GHG). On a cloudy day we can see evidence of the amount of water vapor in our atmosphere.
6-4 Charting a Course for the Future Slide 1 of 30
 6-4 Charting a Course for the Future 1 of 30 6-4 Charting a Course for the Future Researchers are gathering data to monitor and evaluate the effects of human activities on important systems in the biosphere.
6-4 Charting a Course for the Future 1 of 30 6-4 Charting a Course for the Future Researchers are gathering data to monitor and evaluate the effects of human activities on important systems in the biosphere.
Chapter 19: Global Change
 1 Summary Of the Case Study Polar Bear population in the Antarctic going down because temperatures are going up and melting the caps. Polar bears are losing their habitat, they also can t get their food
1 Summary Of the Case Study Polar Bear population in the Antarctic going down because temperatures are going up and melting the caps. Polar bears are losing their habitat, they also can t get their food
7.014 Lecture 20: Biogeochemical Cycles April 1, 2007
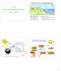 Global Nutrient Cycling - Biogeochemical Cycles 7.14 Lecture 2: Biogeochemical Cycles April 1, 27 Uptake Bioelements in Solution Weathering Precipitation Terrestrial Biomass Decomposition Volatile Elements
Global Nutrient Cycling - Biogeochemical Cycles 7.14 Lecture 2: Biogeochemical Cycles April 1, 27 Uptake Bioelements in Solution Weathering Precipitation Terrestrial Biomass Decomposition Volatile Elements
2.2 - Nutrient Cycles. Carbon Cycle
 2.2 - Nutrient Cycles Carbon Cycle Nutrients What are nutrients? Chemicals (C,O, N, P, H...) needed for life There is a constant amount of these nutrients on Earth and they are stored in different places.
2.2 - Nutrient Cycles Carbon Cycle Nutrients What are nutrients? Chemicals (C,O, N, P, H...) needed for life There is a constant amount of these nutrients on Earth and they are stored in different places.
The Carbon Cycle. the atmosphere the landmass of Earth (including the interior) all of Earth s water all living organisms
 The Carbon Cycle Carbon is an essential part of life on Earth. About half the dry weight of most living organisms is carbon. It plays an important role in the structure, biochemistry, and nutrition of
The Carbon Cycle Carbon is an essential part of life on Earth. About half the dry weight of most living organisms is carbon. It plays an important role in the structure, biochemistry, and nutrition of
