NOMENCLATURE (ie naming compounds)
|
|
|
- Silvester McLaughlin
- 6 years ago
- Views:
Transcription
1 NOMENCLATURE (ie naming compounds) An ionic compounds (salt) may be any one of thousands of chemical compounds How do chemists describe which salt they are discussing? There is a system that we will follow when writing the names or the formulas for ionic compounds.
2 NOMENCLATURE (ie naming compounds) Let s look at monatomic ions first. Monatomic of course implies ONE atom in this case with a charge. For example: lithium ion = Li + sodium ion = Na + barium ion = Ba 2+
3 NOMENCLATURE (ie naming compounds) Let s look at monatomic ions first. Monatomic of course implies ONE atom in this case with a charge. For example: lithium ion = Li + sodium ion = Na + barium ion = Ba 2+ All cations!
4 NOMENCLATURE (ie naming compounds) Let s look at monatomic ions first. Monatomic of course implies ONE atom in this case with a charge. But for nonmetals: fluorine as an ion becomes = fluoride ion = F - chlorine becomes chloride ion = Cl - oxygen becomes oxide ion = O 2-
5 NOMENCLATURE (ie naming compounds) Let s look at monatomic ions first. Monatomic of course implies ONE atom in this case with a charge. But for nonmetals: fluorine as an ion becomes = fluoride ion = F - chlorine becomes chloride ion = Cl - oxygen becomes oxide ion = O 2- Anions!
6 NOMENCLATURE (ie naming compounds) Let s look at monatomic ions first. Monatomic of course implies ONE atom in this case with a charge. But for nonmetals: fluorine as an ion becomes = fluoride ion = F - chlorine becomes chloride ion = Cl - oxygen becomes oxide ion = O 2- So for nonmetals you drop the ending and replace it with ide. Anions!
7 To write a name for an ionic compound - it is as easy as just writing the names of the ions present. example: NaF is made of the sodium and the fluoride ions.
8 To write a name for an ionic compound - it is as easy as just writing the names of the ions present. example: NaF is made of the sodium and the fluoride ions. becomes known as sodium fluoride
9 To write a name for an ionic compound - it is as easy as just writing the names of the ions present. example: NaF is made of the sodium and the fluoride ions. becomes known as sodium fluoride AlCl3 is made of the aluminum and the chloride ions.
10 To write a name for an ionic compound - it is as easy as just writing the names of the ions present. example: NaF is made of the sodium and the fluoride ions. becomes known as sodium fluoride AlCl3 is made of the aluminum and the chloride ions. becomes known as aluminum chloride
11 Try these two yourself: K2S BaI2
12 Try these two yourself: K2S BaI2 that s potassium sulfide. and that s right this one is barium iodide.
13 But what about these two: CuCl CuCl2
14 But what about these two: CuCl CuCl2 At first glance they both should be copper chloride. They have different formulas so they have to have different names. Right?
15 But what about these two: CuCl CuCl2 Remember from earlier discussions that transition elements can form multiple charges. This means that they can also form multiple compounds with the same anion.
16 But what about these two: CuCl CuCl2 At one time the one on the left was known as cuprous chloride and the one of the right was known as cupric chloride.
17 But what about these two: CuCl CuCl2 But now the one on the left is called copper(i) chloride while the one on the right is called copper (II) chloride.
18 But what about these two: CuCl CuCl2 But now the one on the left is called copper(i) chloride while the one on the right is called copper (II) chloride. copper (I) ion chloride ion copper (II) ion chloride ion Hence the different names.
19 As mentioned you name what you see: FeO Fe2O3
20 As mentioned you name what you see: FeO is iron (II) oxide Fe2O3 is iron (III) oxide
21 As mentioned you name what you see: FeO is made up of Fe 2+ and O 2- Fe2O3 is made up of Fe 3+ and O 2- is iron (II) oxide is iron (III) oxide
22 As mentioned you name what you see: FeO is made up of Fe 2+ and O 2- is iron (II) oxide Fe2O3 is made up of Fe 3+ and O 2- is iron (III) oxide How do we know iron (II) vs. iron (III)? The charge on an oxide ion is ALWAYS 2-. So the example on the left iron has to be 2+ to cancel it out.
23 As mentioned you name what you see: FeO is made up of Fe 2+ and O 2- is iron (II) oxide Fe2O3 is made up of Fe 3+ and O 2- is iron (III) oxide How do we know iron (II) vs. iron (III)? The charge on an oxide ion is ALWAYS 2-. But in the example on the right there are three oxide ions making 6-. So there had to be two iron 3+ to make that one neutral.
24 Try these two yourself: CoCl3 MnO
25 Try these two yourself: CoCl3 Well chloride is ALWAYS 1- and there are three of them so the cobalt had to be 3+. So cobalt (III) chloride. MnO And oxide is ALWAYS 2- and there is only one of them so the manganese had to be 2+. So manganese (II) oxide.
26 So now the charges go: Roman Numerals RN for Sn & Pb
27 Nomenclature YOUR TURN: Write the names for the following ionic compounds: 1. K3N 2. CaBr2 3. Al2S3 4. Na3P 5. RaF2 6. CrO 7. Cr2O3 8. SnCl4 9. BeI2 10. PbO
Nomenclature. A systematic method of writing chemical formulas and naming compounds
 Nomenclature A systematic method of writing chemical formulas and naming compounds Chemical symbols Symbols are used to represent elements Either one capital letter, or a capital letter with a lower case
Nomenclature A systematic method of writing chemical formulas and naming compounds Chemical symbols Symbols are used to represent elements Either one capital letter, or a capital letter with a lower case
» Please take out your periodic table.
 » Please take out your periodic table. Ionic Compounds Before we begin What are ions? How are they made? (Hint: Remember what happens to VALENCE electrons.) (Put a line down the middle of the white board.)
» Please take out your periodic table. Ionic Compounds Before we begin What are ions? How are they made? (Hint: Remember what happens to VALENCE electrons.) (Put a line down the middle of the white board.)
1. Name the first element by its name. 2. The second element has the ending ide. 3. The number of atoms of each element is indicated with Greek
 Binary compounds containing two nonmetals 1. Name the first element by its name. 2. The second element has the ending ide. 3. The number of atoms of each element is indicated with Greek prefixes. Greek
Binary compounds containing two nonmetals 1. Name the first element by its name. 2. The second element has the ending ide. 3. The number of atoms of each element is indicated with Greek prefixes. Greek
Worksheet 5 - Naming Ionic Compounds Part 1 (Binary compounds with Group 1, 2 or 13 metals) Naming
 Worksheet 5 - Naming Ionic Compounds Part 1 (Binary compounds with Group 1, 2 or 13 metals) Binary compounds: have only two kinds of elements, if there are three or more it is not a binary compound. Naming
Worksheet 5 - Naming Ionic Compounds Part 1 (Binary compounds with Group 1, 2 or 13 metals) Binary compounds: have only two kinds of elements, if there are three or more it is not a binary compound. Naming
Chemical Formulas. In a chemical formula, the element with the positive charge is always written first.
 Bonding Part 2 Chemical Formulas Chemical formulas a group of chemical symbols and numbers that represent the number of atoms of each element that make up a compound. ( H O) 2 In a chemical formula, the
Bonding Part 2 Chemical Formulas Chemical formulas a group of chemical symbols and numbers that represent the number of atoms of each element that make up a compound. ( H O) 2 In a chemical formula, the
Summer Assignment Coversheet
 Summer Assignment Coversheet Course: A.P. Chemistry Teachers Names: Mary Engels Assignment Title: Summer Assignment A Review Assignment Summary/Purpose: To review the Rules for Solubility, Oxidation Numbers,
Summer Assignment Coversheet Course: A.P. Chemistry Teachers Names: Mary Engels Assignment Title: Summer Assignment A Review Assignment Summary/Purpose: To review the Rules for Solubility, Oxidation Numbers,
Chapter 5. Cycle 4 Chemistry I Lesson 4. WARMUP: Manganese oxide scenario
 Cycle 4 Chemistry I Lesson 4 WARMUP: Manganese oxide scenario You own a factory that produces alkaline batteries. One of the ingredients in your batteries is manganese oxide, MnO 2, a black powder. However,
Cycle 4 Chemistry I Lesson 4 WARMUP: Manganese oxide scenario You own a factory that produces alkaline batteries. One of the ingredients in your batteries is manganese oxide, MnO 2, a black powder. However,
Name: Mods: Date Agenda Homework
 Name: Mods: UNIT 6 FORMULA WRITING, FORMULA WEIGHTS, and PERCENT COMPOSITION Date Agenda Homework Wed Dec 3 Thurs Dec 4 Fri Dec 5 Mon Dec 8 Tues Dec 9 Wed Dec 10 (1/2 Day) Thurs Dec 11 Fri Dec 12 Mon Dec
Name: Mods: UNIT 6 FORMULA WRITING, FORMULA WEIGHTS, and PERCENT COMPOSITION Date Agenda Homework Wed Dec 3 Thurs Dec 4 Fri Dec 5 Mon Dec 8 Tues Dec 9 Wed Dec 10 (1/2 Day) Thurs Dec 11 Fri Dec 12 Mon Dec
Formula & Equation Writing
 Formula & Equation Writing Book column Be Na Mg column Ca lithium Ionic Equations Ionic Formulae Balanced Equations Formula Equations Word Equations carbonate Transition Metals C 3 valency valency Using
Formula & Equation Writing Book column Be Na Mg column Ca lithium Ionic Equations Ionic Formulae Balanced Equations Formula Equations Word Equations carbonate Transition Metals C 3 valency valency Using
21. sodium nitrite 31. potassium carbonate. 23. aluminum hydroxide 33. nickel (II) carbonate. 24. ammonium hydroxide 34.
 N.E. Packet 1 Nomenclature WS 1 (Ionic Compounds) Name: Date: Per: Write the name for each of the following compounds. 1. CaCO 3 11. BaSO 4 2. FeO 12. Zn(NO 3 ) 2 3. K 2 SO 3 13. CuSO 4 4. AgCl 14. AlCl
N.E. Packet 1 Nomenclature WS 1 (Ionic Compounds) Name: Date: Per: Write the name for each of the following compounds. 1. CaCO 3 11. BaSO 4 2. FeO 12. Zn(NO 3 ) 2 3. K 2 SO 3 13. CuSO 4 4. AgCl 14. AlCl
Ionic Naming Quiz #1. Ionic Naming Quiz #1. March 26, Day 05 Ionic Quiz, SDR Note, Exit Card.notebook
 Ionic Naming Quiz #1 Name the following ionic compounds: 1) NaBr sodium bromide 2) Sc(OH) 3 scandium hydroxide 3) V 2 (SO 4 ) 3 vanadium (III) sulfate 4) NH 4 F ammonium fluoride 5) CaCO 3 calcium carbonate
Ionic Naming Quiz #1 Name the following ionic compounds: 1) NaBr sodium bromide 2) Sc(OH) 3 scandium hydroxide 3) V 2 (SO 4 ) 3 vanadium (III) sulfate 4) NH 4 F ammonium fluoride 5) CaCO 3 calcium carbonate
Ionic Naming Quiz #1. Ionic Naming Quiz #2 Write the formulas for the following ionic compounds:
 Ionic Naming Quiz #1 Name the following ionic compounds: 1) NaBr sodium bromide 2) Sc(OH) 3 scandium hydroxide 3) V 2 (SO 4 ) 3 vanadium (III) sulfate 4) NH 4 F ammonium fluoride 5) CaCO 3 calcium carbonate
Ionic Naming Quiz #1 Name the following ionic compounds: 1) NaBr sodium bromide 2) Sc(OH) 3 scandium hydroxide 3) V 2 (SO 4 ) 3 vanadium (III) sulfate 4) NH 4 F ammonium fluoride 5) CaCO 3 calcium carbonate
Formula & Equation Writing
 Formula & Equation Writing Book 2 H H Al Al H Al(H) 3 H Ionic Equations Ionic Formulae Balanced Equations Formula Equations Word Equations Transition Metals Using Brackets Awkward Customers More than 2
Formula & Equation Writing Book 2 H H Al Al H Al(H) 3 H Ionic Equations Ionic Formulae Balanced Equations Formula Equations Word Equations Transition Metals Using Brackets Awkward Customers More than 2
Balance the following equations: 1. Al + N 2 AlN. 2. Fe + O 2 Fe 3 O CaCO 3 CaO + CO 2 4. NH 4 NO 3 N 2 O + H 2 O. 5. KI + Cl 2 KCl + I 2
 Balance the following equations: 1. Al + N 2 AlN 2. Fe + O 2 Fe 3 O 4 3. CaCO 3 CaO + CO 2 4. NH 4 NO 3 N 2 O + H 2 O 5. KI + Cl 2 KCl + I 2 6. Pb(NO 3 ) 2 + HCl PbCl 2 + HNO 3 7. BaO 2 BaO + O 2 8. Al
Balance the following equations: 1. Al + N 2 AlN 2. Fe + O 2 Fe 3 O 4 3. CaCO 3 CaO + CO 2 4. NH 4 NO 3 N 2 O + H 2 O 5. KI + Cl 2 KCl + I 2 6. Pb(NO 3 ) 2 + HCl PbCl 2 + HNO 3 7. BaO 2 BaO + O 2 8. Al
Slide 1 / 76. Slide 2 / 76. Slide 3 / In the periodic table, the elements are arranged in.
 1 In the periodic table, the elements are arranged in. Slide 1 / 76 alphabetical order order of increasing atomic number order of increasing metallic properties order of increasing neutron content reverse
1 In the periodic table, the elements are arranged in. Slide 1 / 76 alphabetical order order of increasing atomic number order of increasing metallic properties order of increasing neutron content reverse
Slide 1 / 76. Slide 2 / 76. Slide 4 / 76. Slide 3 / 76. Slide 6 / 76. Slide 5 / In the periodic table, the elements are arranged in.
 Slide 1 / 76 1 In the periodic table, the elements are arranged in Slide 2 / 76 2 lements exhibit similar physical and chemical properties. alphabetical order order of increasing atomic number order of
Slide 1 / 76 1 In the periodic table, the elements are arranged in Slide 2 / 76 2 lements exhibit similar physical and chemical properties. alphabetical order order of increasing atomic number order of
INTERPRETING CHEMICAL FORMULAS AND SYMBOLS
 INTERPRETING CHEMICAL FORMULAS AND SYMBOLS THE FORMULA OR SYMBOL REPRESENTS: CHEMICAL FORMULA OR SYMBOL NUMBER OF ATOMS TOTAL CHARGE NUMBER OF PROTONS NUMBER OF ELECTRONS Atom Molecule Monatomic Cation
INTERPRETING CHEMICAL FORMULAS AND SYMBOLS THE FORMULA OR SYMBOL REPRESENTS: CHEMICAL FORMULA OR SYMBOL NUMBER OF ATOMS TOTAL CHARGE NUMBER OF PROTONS NUMBER OF ELECTRONS Atom Molecule Monatomic Cation
SNC 2D0-01 Chemistry Review Chapters 5 and 6
 SNC 2D0-01 Chemistry Review Chapters 5 and 6 Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. A chemical change is distinguished from a physical
SNC 2D0-01 Chemistry Review Chapters 5 and 6 Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. A chemical change is distinguished from a physical
Name: Date: Period: Page: Predicting Products. Predict the products, and balance each reaction. Use the reaction type as a hint.
 Name: Date: Period: Page: Predicting Products Predict the products, and balance each reaction. Use the reaction type as a hint. For reactions with a transition metal, the most likely charge on the metal
Name: Date: Period: Page: Predicting Products Predict the products, and balance each reaction. Use the reaction type as a hint. For reactions with a transition metal, the most likely charge on the metal
Inorganic Naming Worksheet
 Inorganic Naming Worksheet A) Write the formulas for the following names: 1. ammonium sulfite 2. sodium nitrite 3. cupric bromate 4. aluminum sulfate 5. potassium nitrate 6. ferrous bicarbonate 7. lead
Inorganic Naming Worksheet A) Write the formulas for the following names: 1. ammonium sulfite 2. sodium nitrite 3. cupric bromate 4. aluminum sulfate 5. potassium nitrate 6. ferrous bicarbonate 7. lead
Chemistry. Ionic Compounds. Inside:
 hemistry Judy van der Velden. reative ommons licence. onic Bonding Practice Grade 10 cience onic ompounds onic compounds are chemical compounds in which ions are held together in a lattice structure by
hemistry Judy van der Velden. reative ommons licence. onic Bonding Practice Grade 10 cience onic ompounds onic compounds are chemical compounds in which ions are held together in a lattice structure by
Nomenclature. I. Simple Ions (Ions that contain only one atom) II. Complex Ions. I A. Negative Ions Negative ions have names ending in -IDE
 Nomenclature I. Simple Ions (Ions that contain only one atom) I A. Negative Ions Negative ions have names ending in -IDE O = oxide ion, Cl = chloride ion, P = phosphide ion I B. Positive Ions 1. If there
Nomenclature I. Simple Ions (Ions that contain only one atom) I A. Negative Ions Negative ions have names ending in -IDE O = oxide ion, Cl = chloride ion, P = phosphide ion I B. Positive Ions 1. If there
Which of these is the formula for disulfur heptoxide? A. S 2 O 7 B. S 7 O 2 C. SO 2 D. N 2 O
 Which of these is the formula for disulfur heptoxide? A. S 2 O 7 B. S 7 O 2 C. SO 2 D. N 2 O Which of these is the correct chemical formula for a molecule of oxygen? A. O B. O -2 C. O +2 D. O 2 Which of
Which of these is the formula for disulfur heptoxide? A. S 2 O 7 B. S 7 O 2 C. SO 2 D. N 2 O Which of these is the correct chemical formula for a molecule of oxygen? A. O B. O -2 C. O +2 D. O 2 Which of
S1 Building Blocks Summary Notes
 S1 Building Blocks Summary Notes Atoms & Molecules 1 We are developing our understanding of atoms and molecules. Atoms are the simplest building blocks of every substance in the universe. There are just
S1 Building Blocks Summary Notes Atoms & Molecules 1 We are developing our understanding of atoms and molecules. Atoms are the simplest building blocks of every substance in the universe. There are just
Compounds & Reactions Week 1. Writing Formulas & Balancing Equations. Write the chemical formula for each molecular (covalent) compound.
 Compounds & Reactions Week 1 Name Writing Formulas & Balancing Equations Write the chemical formula for each ionic compound. 1. Lithium fluoride 2. Copper (II) chloride 3. Manganese (II) oxide 4. Potassium
Compounds & Reactions Week 1 Name Writing Formulas & Balancing Equations Write the chemical formula for each ionic compound. 1. Lithium fluoride 2. Copper (II) chloride 3. Manganese (II) oxide 4. Potassium
Predicting Reaction Products
 Predicting Reaction Products Once you classify a chemical reaction, write the products and balance the reaction. Review Diatomic Molecules! Elements that exist only as compounds in the natural world: Iodine
Predicting Reaction Products Once you classify a chemical reaction, write the products and balance the reaction. Review Diatomic Molecules! Elements that exist only as compounds in the natural world: Iodine
Detroit Water and Sewerage Department Water Quality Division Laboratory Analysis of Water Samples Collected at Lake Huron Plant 6/9/2015
 Lake Huron Plant Turbidity NTU 0.35 0.05 0.3/95% (1) Total Solids mg/l 136 162 500 10 Total Dissolved Solids mg/l 145 136 500 10 Aluminum Al mg/l 0.510 0.142 0.05-0.2 0.005 Iron Fe mg/l 0.082 0.118 0.3
Lake Huron Plant Turbidity NTU 0.35 0.05 0.3/95% (1) Total Solids mg/l 136 162 500 10 Total Dissolved Solids mg/l 145 136 500 10 Aluminum Al mg/l 0.510 0.142 0.05-0.2 0.005 Iron Fe mg/l 0.082 0.118 0.3
3. [7 points] How many significant figures should there be in the answer to the following problem?
![3. [7 points] How many significant figures should there be in the answer to the following problem? 3. [7 points] How many significant figures should there be in the answer to the following problem?](/thumbs/83/87492512.jpg) 1 of 6 10/20/2009 3:52 AM 1. [7 points] What is the correct name for ZnF 2? (a) zinc fluorine (b) zinc (II) fluoride (c) zinc fluoride (d) fluoride zinc 2. [7 points] What is the correct name for P 2 O
1 of 6 10/20/2009 3:52 AM 1. [7 points] What is the correct name for ZnF 2? (a) zinc fluorine (b) zinc (II) fluoride (c) zinc fluoride (d) fluoride zinc 2. [7 points] What is the correct name for P 2 O
Unit 5 ~ Learning Guide Name:
 Unit 5 ~ Learning Guide Name: Instructions: Using a pencil, complete the following notes as you work through the related lessons. Show ALL work as is explained in the lessons. You are required to have
Unit 5 ~ Learning Guide Name: Instructions: Using a pencil, complete the following notes as you work through the related lessons. Show ALL work as is explained in the lessons. You are required to have
JSUNIL TUTORIAL, SAMASTIPUR
 Chapter 1 Chemical Reactions and Equations Q 1. Why should a magnesium ribbon be cleaned before burning in air? Ans. Before burning in air, the magnesium ribbon is cleaned by rubbing with a sandpaper.
Chapter 1 Chemical Reactions and Equations Q 1. Why should a magnesium ribbon be cleaned before burning in air? Ans. Before burning in air, the magnesium ribbon is cleaned by rubbing with a sandpaper.
Lake Huron Water Treatment Plant Mineral Report
 Lake Huron Water Treatment Plant Mineral Report Turbidity NTU 0.40 0.05 0.3/95% (1) Total Solids mg/l 160 155 500 10 Total Dissolved Solids mg/l 101 0 500 10 Aluminum Al mg/l < 0.050 0.060 0.05-0.2 0.005
Lake Huron Water Treatment Plant Mineral Report Turbidity NTU 0.40 0.05 0.3/95% (1) Total Solids mg/l 160 155 500 10 Total Dissolved Solids mg/l 101 0 500 10 Aluminum Al mg/l < 0.050 0.060 0.05-0.2 0.005
Lake Huron Water Treatment Plant Mineral Report
 Lake Huron Water Treatment Plant Mineral Report Turbidity NTU 0.30 0.06 0.3/95% (1) Total Solids mg/l 94 91 500 10 Total Dissolved Solids mg/l 81 51 500 10 Aluminum Al mg/l < 0.050 < 0.050 0.05-0.2 0.005
Lake Huron Water Treatment Plant Mineral Report Turbidity NTU 0.30 0.06 0.3/95% (1) Total Solids mg/l 94 91 500 10 Total Dissolved Solids mg/l 81 51 500 10 Aluminum Al mg/l < 0.050 < 0.050 0.05-0.2 0.005
A. REGULAR BINARY COMPOUNDS
 A. REGULAR BINARY COMPOUNDS Worksheet A1 anions...±±± F -1 O -2 N -3 C -4 cations...999 name: fluoride name: oxide name: nitride name: carbide Li +1 Li 1 F 1 or LiF Li 2 O 1 or Li 2 O Li 3 N Li 4 C name:
A. REGULAR BINARY COMPOUNDS Worksheet A1 anions...±±± F -1 O -2 N -3 C -4 cations...999 name: fluoride name: oxide name: nitride name: carbide Li +1 Li 1 F 1 or LiF Li 2 O 1 or Li 2 O Li 3 N Li 4 C name:
1. Which of the given statements about the reaction below are incorrect?
 1. Which of the given statements about the reaction below are incorrect? 2PbO(s) + C(s) 2Pb(s) + CO 2 (g) a. Lead is getting reduced b. Carbon dioxide is getting oxidised c. Carbon is getting oxidised
1. Which of the given statements about the reaction below are incorrect? 2PbO(s) + C(s) 2Pb(s) + CO 2 (g) a. Lead is getting reduced b. Carbon dioxide is getting oxidised c. Carbon is getting oxidised
Detroit Water and Sewerage Department Water Quality Division Laboratory Analysis of Water Samples Collected at Lake Huron Plant 10/14/2014
 Lake Huron Plant Turbidity NTU 0.43 0.04 0.3/95% (1) Total Solids mg/l 103 116 500 10 Total Dissolved Solids mg/l 112 142 500 10 Aluminum Al mg/l < 0.050 < 0.050 0.05-0.2 0.005 Iron Fe mg/l 0.053 < 0.050
Lake Huron Plant Turbidity NTU 0.43 0.04 0.3/95% (1) Total Solids mg/l 103 116 500 10 Total Dissolved Solids mg/l 112 142 500 10 Aluminum Al mg/l < 0.050 < 0.050 0.05-0.2 0.005 Iron Fe mg/l 0.053 < 0.050
Today! Demonstrations of Redox Chemistry! Electrochemistry! electrons moving about! equilibrium with a control knob! The disappearing Aluminum Rod!
 Today! Electrochemistry! electrons moving about! equilibrium with a control knob! Redox chemistry! oxidation and reduction! Demonstrations of Redox Chemistry! The disappearing Aluminum Rod! Alkali Metals
Today! Electrochemistry! electrons moving about! equilibrium with a control knob! Redox chemistry! oxidation and reduction! Demonstrations of Redox Chemistry! The disappearing Aluminum Rod! Alkali Metals
Double Award Science: Chemistry
 New Specification Centre Number Candidate Number General Certificate of Secondary Education 2014 2015 Double Award Science: Chemistry Unit C1 Higher Tier ML [GSD22] WEDNESDAY 25 FEBRUARY 2015, MORNING
New Specification Centre Number Candidate Number General Certificate of Secondary Education 2014 2015 Double Award Science: Chemistry Unit C1 Higher Tier ML [GSD22] WEDNESDAY 25 FEBRUARY 2015, MORNING
Sn 2+ (aq) + 2 Ag + (aq) Sn 4+ (aq) + 2 Ag(s),
 1. Which change in oxidation number represents oxidation? A) Sn 2+ (aq) Sn 4+ (aq) B) Sn 2+ (aq) Sn(s) C) Sn 4+ (aq) Sn 2+ (aq) D) Sn 4+ (aq) Sn(s) E) Sn(s) Sn 2 (aq) 2. In the reaction Sn 2+ (aq) + 2
1. Which change in oxidation number represents oxidation? A) Sn 2+ (aq) Sn 4+ (aq) B) Sn 2+ (aq) Sn(s) C) Sn 4+ (aq) Sn 2+ (aq) D) Sn 4+ (aq) Sn(s) E) Sn(s) Sn 2 (aq) 2. In the reaction Sn 2+ (aq) + 2
Ionic Bonding Drill. Compound Formula. tin (II) nitride. silver oxide. lithium sulfide. magnesium sulfide. copper (I) nitride.
 tin (II) nitride silver oxide lithium sulfide magnesium sulfide copper (I) nitride boron iodide potassium fluoride potassium bromide sodium phosphide iron (II) chloride copper (I) bromide lead (II) sulfide
tin (II) nitride silver oxide lithium sulfide magnesium sulfide copper (I) nitride boron iodide potassium fluoride potassium bromide sodium phosphide iron (II) chloride copper (I) bromide lead (II) sulfide
GCSE 4472/02 CHEMISTRY 2 HIGHER TIER ADDITIONAL SCIENCE/CHEMISTRY. P.M. MONDAY, 20 May hour For Examiner s use only Question Maximum Mark 1.
 Surname Centre Number Candidate Number Other Names 0 GCSE 4472/02 ADDITIONAL SCIENCE/CHEMISTRY CHEMISTRY 2 HIGHER TIER P.M. MONDAY, 20 May 2013 1 hour For s use Question Maximum Mark 1. 8 Mark Awarded
Surname Centre Number Candidate Number Other Names 0 GCSE 4472/02 ADDITIONAL SCIENCE/CHEMISTRY CHEMISTRY 2 HIGHER TIER P.M. MONDAY, 20 May 2013 1 hour For s use Question Maximum Mark 1. 8 Mark Awarded
Oxidation-Reduction Reactions
 Oxidation-Reduction Reactions REACTIONS 20.1 The Meaning of Oxidation and Reduction Essential Understanding Oxidation and reduction are opposite chemical processes during which electrons are shifted between
Oxidation-Reduction Reactions REACTIONS 20.1 The Meaning of Oxidation and Reduction Essential Understanding Oxidation and reduction are opposite chemical processes during which electrons are shifted between
Naming and Classifying the Elements. nobelium antimony. oxygen arsenic. phosphorus beryllium. platinum bismuth. plutonium boron.
 6.2 Naming and Classifying the Elements To learn more about the origins of the names of the elements, visit www.science.nelson.com GO The names of the elements have varied origins. Some elements are named
6.2 Naming and Classifying the Elements To learn more about the origins of the names of the elements, visit www.science.nelson.com GO The names of the elements have varied origins. Some elements are named
Solubility Equilibrium
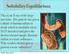 Solubility Equilibrium This is an X-ray of the large intestine. The patient was given a drink of barium sulfate to drink which is insoluble (does NOT dissolve) and gives the doctor a better image. Barium
Solubility Equilibrium This is an X-ray of the large intestine. The patient was given a drink of barium sulfate to drink which is insoluble (does NOT dissolve) and gives the doctor a better image. Barium
NAME PER "ALICE" REFERENCE NOTEBOOK
 NAME PER ACTIVE LEARNING IN CHEMISTRY EDUCATION "ALICE" REFERENCE NOTEBOOK This document may be used (when directed) during tests and quizzes, provided that you do not alter it in any way. Do not add any
NAME PER ACTIVE LEARNING IN CHEMISTRY EDUCATION "ALICE" REFERENCE NOTEBOOK This document may be used (when directed) during tests and quizzes, provided that you do not alter it in any way. Do not add any
Solve: 1. a 4 a 5 = 2. ( )/10 4 = 3. a(2a) 2 = 4. (6-1 ) 3 = a /10 4 a(4a 2 ) a 3
 Solve: 1. a 4 a 5 = 2. (10 2 10 3 )/10 4 = 3. a(2a) 2 = 4. (6-1 ) 3 = a 9 10 5 /10 4 a(4a 2 ) 6 2 10 1 4a 3 5. 12y 2 / 4y 3 = 6. (a 3 ) 0 = 7. x 2 y 4 z 3 x 1 y 2 z 5 = 3y -1 1 x 1 y 2 z -2 Solve for x:
Solve: 1. a 4 a 5 = 2. (10 2 10 3 )/10 4 = 3. a(2a) 2 = 4. (6-1 ) 3 = a 9 10 5 /10 4 a(4a 2 ) 6 2 10 1 4a 3 5. 12y 2 / 4y 3 = 6. (a 3 ) 0 = 7. x 2 y 4 z 3 x 1 y 2 z 5 = 3y -1 1 x 1 y 2 z -2 Solve for x:
AP Chemistry A. Allan Chapter 18 - The Representative Elements: Groups 1A through 4A
 AP Chemistry A. Allan Chapter 18 - The Representative Elements: Groups 1A through 4A 18.1 A Survey of the Representative Elements A. Basic Trends 1. Metals tend to lose electrons and form cations 2. Nonmetals
AP Chemistry A. Allan Chapter 18 - The Representative Elements: Groups 1A through 4A 18.1 A Survey of the Representative Elements A. Basic Trends 1. Metals tend to lose electrons and form cations 2. Nonmetals
Appendix U. Mapping of Water Quality Concentration Contours across the Province
 ALDERON IRON ORE CORP. ENVIRONMENTAL IMPACT STATEMENT KAMI IRON ORE MINE AND RAIL INFRASTRUCTURE, LABRADOR Appendix U Mapping of Water Quality Concentration Contours across the Province Figure 17I.1 Alkalinity
ALDERON IRON ORE CORP. ENVIRONMENTAL IMPACT STATEMENT KAMI IRON ORE MINE AND RAIL INFRASTRUCTURE, LABRADOR Appendix U Mapping of Water Quality Concentration Contours across the Province Figure 17I.1 Alkalinity
. The X-ion in this salt has 28 electrons. The metal X is. A) Ni B) Zn C) Fe D) V E) Pd 30) The charge on the manganese in the salt MnF
 8) Magnesium reacts with a certain element to form a compound with the general formula MgX. What would the most likely formula be for the compound formed between potassium and element X? A) KX B) KX C)
8) Magnesium reacts with a certain element to form a compound with the general formula MgX. What would the most likely formula be for the compound formed between potassium and element X? A) KX B) KX C)
Inorganic Chemistry Lesson 16 Classification of elements. Part 2. Periodic Law
 Inorganic Chemistry Lesson 16 Classification of elements. Part 2. Periodic Law March 2, 2014 1 Atomic mass and the properties of elements. If we arrange halogens according to their nonmetallicity (which
Inorganic Chemistry Lesson 16 Classification of elements. Part 2. Periodic Law March 2, 2014 1 Atomic mass and the properties of elements. If we arrange halogens according to their nonmetallicity (which
ADDITIONAL SCIENCE/CHEMISTRY. A.M. TUESDAY, 14 January hour
 Surname Centre Number Candidate Number Other Names 0 GCSE 4472/02 ADDITIONAL SCIENCE/CEMISTRY CEMISTRY 2 IGER TIER A.M. TUESDAY, 14 January 2014 1 hour For s use Question Maximum Mark Mark Awarded 1. 8
Surname Centre Number Candidate Number Other Names 0 GCSE 4472/02 ADDITIONAL SCIENCE/CEMISTRY CEMISTRY 2 IGER TIER A.M. TUESDAY, 14 January 2014 1 hour For s use Question Maximum Mark Mark Awarded 1. 8
ADDITIONAL SCIENCE/CHEMISTRY. A.M. TUESDAY, 14 January hour
 Surname Centre Number Candidate Number Other Names 0 GCSE 4472/02 ADDITIONAL SCIENCE/CEMISTRY CEMISTRY 2 IGER TIER A.M. TUESDAY, 14 January 2014 1 hour For s use Question Maximum Mark Mark Awarded 1. 8
Surname Centre Number Candidate Number Other Names 0 GCSE 4472/02 ADDITIONAL SCIENCE/CEMISTRY CEMISTRY 2 IGER TIER A.M. TUESDAY, 14 January 2014 1 hour For s use Question Maximum Mark Mark Awarded 1. 8
Enthalpy " H o " Entropy "S o " Gibbs energy
 THERMODYNAMIC VALUES AT STANDARD STATE (298K) Data Retrieved From: Kots, Treichal, Weaver Chemistry & Chemical Reactivity (Sixth Edition) COPYRIGHT 2006 Species Name Enthalpy " H o " Entropy "S o " Gibbs
THERMODYNAMIC VALUES AT STANDARD STATE (298K) Data Retrieved From: Kots, Treichal, Weaver Chemistry & Chemical Reactivity (Sixth Edition) COPYRIGHT 2006 Species Name Enthalpy " H o " Entropy "S o " Gibbs
Duncan. UNIT 8 - Chemical Equations BALANCING EQUATIONS PRACTICE WORKSHEET 14.) C2H6 + O2 CO2 + H2O. 2.) Na + I2 NaI 3.) N2 + O2 N2O 4.
 BALANCING EQUATIONS PRACTICE WORKSHEET 1.) CH4 + O2 CO2 + H2O 2.) Na + I2 NaI 3.) N2 + O2 N2O 4.) N2 + H2 NH3 5.) KI + Cl2 KCl + I2 6.) HCl + Ca(OH)2 CaCl2 + H2O 7.) KClO3 KCl + O2 8.) K3PO4 + HCl KCl
BALANCING EQUATIONS PRACTICE WORKSHEET 1.) CH4 + O2 CO2 + H2O 2.) Na + I2 NaI 3.) N2 + O2 N2O 4.) N2 + H2 NH3 5.) KI + Cl2 KCl + I2 6.) HCl + Ca(OH)2 CaCl2 + H2O 7.) KClO3 KCl + O2 8.) K3PO4 + HCl KCl
Thermal decomposition. Metal carbonates
 Decomposition reactions Copy correctly Up to 3% of a workbook Copying or scanning from ESA workbooks is subject to the New Zealand Copyright Act which limits copying to 3% of this workbook. Many compounds
Decomposition reactions Copy correctly Up to 3% of a workbook Copying or scanning from ESA workbooks is subject to the New Zealand Copyright Act which limits copying to 3% of this workbook. Many compounds
CERTIFICATE OF ANALYSIS
 BAY 3, 4500 5TH STREET NE CALGARY, AB CANADA T2L 7C3 MAXXAM JOB #: A814851 Received: 2008/04/02, 13:15 Matrix: Water # s Received: 1 This report supersedes all previous reports with the same Maxxam job
BAY 3, 4500 5TH STREET NE CALGARY, AB CANADA T2L 7C3 MAXXAM JOB #: A814851 Received: 2008/04/02, 13:15 Matrix: Water # s Received: 1 This report supersedes all previous reports with the same Maxxam job
Chapter 17 Solubility and Complex Ion Equilibria
 Chem 1046 General Chemistry by Ebbing and Gammon, 8th Edition Prof George W.J. Kenney, Jr Last Update: 27Nov2008 Chapter 17 Solubility and Complex Ion Equilibria These Notes are to SUPPLIMENT the Text,
Chem 1046 General Chemistry by Ebbing and Gammon, 8th Edition Prof George W.J. Kenney, Jr Last Update: 27Nov2008 Chapter 17 Solubility and Complex Ion Equilibria These Notes are to SUPPLIMENT the Text,
Appendix C1: Batch Kinetics Tests
 Appendix C1: Batch Kinetics Tests This appendix contains the entire data set for the batch kinetics tests for the potential biofilter components. These tests were performed to provide estimates of optimal
Appendix C1: Batch Kinetics Tests This appendix contains the entire data set for the batch kinetics tests for the potential biofilter components. These tests were performed to provide estimates of optimal
ADDITIONAL SCIENCE/CHEMISTRY
 Surname Centre Number Candidate Number Other Names 0 GCSE 4472/02 S16-4472-02 ADDITIONAL SCIENCE/CHEMISTRY CHEMISTRY 2 HIGHER TIER A.M. THURSDAY, 19 May 2016 1 hour For s use Question Maximum Mark Mark
Surname Centre Number Candidate Number Other Names 0 GCSE 4472/02 S16-4472-02 ADDITIONAL SCIENCE/CHEMISTRY CHEMISTRY 2 HIGHER TIER A.M. THURSDAY, 19 May 2016 1 hour For s use Question Maximum Mark Mark
AP Chemistry
 AP Chemistry 2018-2019 Introduction Advanced Placement Chemistry is a college level course; THIS IS freshman level college chemistry. You will need to be dedicated and work very hard if you are to be successful.
AP Chemistry 2018-2019 Introduction Advanced Placement Chemistry is a college level course; THIS IS freshman level college chemistry. You will need to be dedicated and work very hard if you are to be successful.
+ 10Cl 2Mn H 2 O + 5Cl 2. Deduce the half-equation for the oxidation of chloride ions to chlorine.
 Q1.Chlorine is an important industrial chemical. (a) Chlorine is formed when KMnO 4 reacts with hydrochloric acid. The ionic equation for this redox reaction is 16H + + 2MnO 4 + 10Cl 2Mn 2+ + 8H 2 O +
Q1.Chlorine is an important industrial chemical. (a) Chlorine is formed when KMnO 4 reacts with hydrochloric acid. The ionic equation for this redox reaction is 16H + + 2MnO 4 + 10Cl 2Mn 2+ + 8H 2 O +
P.M. FRIDAY, 12 June hour For Examiner s use only
 Surname Centre Number Candidate Number Other Names 0 GCSE 4462/02 S15-4462-02 SCIENCE A/CHEMISTRY CHEMISTRY 1 HIGHER TIER P.M. FRIDAY, 12 June 2015 1 hour For s use Question Maximum Mark Mark Awarded 1.
Surname Centre Number Candidate Number Other Names 0 GCSE 4462/02 S15-4462-02 SCIENCE A/CHEMISTRY CHEMISTRY 1 HIGHER TIER P.M. FRIDAY, 12 June 2015 1 hour For s use Question Maximum Mark Mark Awarded 1.
The ion with Vanadium in its oxidation state of 5 exists as a solid compound in the form of a VO 3
 Chemistry of some Transition elements: V, Mn, Co, Cu, Cr and Ag Vanadium Vanadium has four main oxidation states. Each one has a different colour. VO 2 VO 2 V 3 V 2 O.N. 5 yellow O.N. 4 blue O.N. 3 green
Chemistry of some Transition elements: V, Mn, Co, Cu, Cr and Ag Vanadium Vanadium has four main oxidation states. Each one has a different colour. VO 2 VO 2 V 3 V 2 O.N. 5 yellow O.N. 4 blue O.N. 3 green
QUESTIONNAIRE FOR GENERAL WTP REQUESTED DATA FOR THE WATER TREATMENT PLANT (WTP) - Application : groundwater (well pit); urban wastewater;
 REQUESTED DATA FOR THE WATER TREATMENT PLANT (WTP) - Application : ENVIRONMENTAL DATA - Temperature min. : C - Temperature max. : C - Altitude (m.a.s.l) : m - Plant location (geographic area, seismic zone,
REQUESTED DATA FOR THE WATER TREATMENT PLANT (WTP) - Application : ENVIRONMENTAL DATA - Temperature min. : C - Temperature max. : C - Altitude (m.a.s.l) : m - Plant location (geographic area, seismic zone,
The Reactivity Series
 The Reactivity Series 1 of 33 Boardworks Ltd 2016 The Reactivity Series 2 of 33 Boardworks Ltd 2016 What is the reactivity series 3 of 33 Boardworks Ltd 2016 It is possible to arrange a group of elements
The Reactivity Series 1 of 33 Boardworks Ltd 2016 The Reactivity Series 2 of 33 Boardworks Ltd 2016 What is the reactivity series 3 of 33 Boardworks Ltd 2016 It is possible to arrange a group of elements
SOLUBILITY STUDY GUIDE- Multiple Choice Section
 SOLUBILITY STUDY GUIDE- Multiple Choice Section Multiple Choice Section: This study guide is a compilation of questions from provincial exams since 2000. I urge you to become intimately familiar with question
SOLUBILITY STUDY GUIDE- Multiple Choice Section Multiple Choice Section: This study guide is a compilation of questions from provincial exams since 2000. I urge you to become intimately familiar with question
A.M. MONDAY, 18 January minutes
 Candidate Name Centre Number Candidate Number 0 GCSE 240/01 ADDITIONAL SCIENCE FOUNDATION TIER CHEMISTRY 2 A.M. MONDAY, 18 January 2010 45 minutes ADDITIONAL MATERIALS In addition to this paper you may
Candidate Name Centre Number Candidate Number 0 GCSE 240/01 ADDITIONAL SCIENCE FOUNDATION TIER CHEMISTRY 2 A.M. MONDAY, 18 January 2010 45 minutes ADDITIONAL MATERIALS In addition to this paper you may
Chemistry. TOPIC : Redox Reactions. products are PH3 and NaH2PO2. This reaction is an examplee of. (b) Reduction (d) Neutralization
 TOPIC : Redox Reactions Date : Marks : 1 mks Time : ½ hr 1. When P reacts with caustic soda, the products are PH and NaHPO. This reaction is an examplee of (a) Oxidation (c) Oxidation and reduction (redox)
TOPIC : Redox Reactions Date : Marks : 1 mks Time : ½ hr 1. When P reacts with caustic soda, the products are PH and NaHPO. This reaction is an examplee of (a) Oxidation (c) Oxidation and reduction (redox)
CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK. Due Date Assignment On-Time (100) Late (70)
 Name Period CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK Due Date Assignment On-Time (100) Late (70) 1.1 1.2 1.3 Warm Ups Extra Credit Notes, Homework, Exam Reviews and Their KEYS located
Name Period CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK Due Date Assignment On-Time (100) Late (70) 1.1 1.2 1.3 Warm Ups Extra Credit Notes, Homework, Exam Reviews and Their KEYS located
GCSE 4462/01 CHEMISTRY 1 FOUNDATION TIER SCIENCE A/CHEMISTRY. A.M. Tuesday, 13 January hour JAN W PMT.
 Surname Centre Number Candidate Number Other Names 0 GCSE 4462/01 W15-4462-01 SCIENCE A/CHEMISTRY CHEMISTRY 1 FOUNDATION TIER A.M. Tuesday, 13 January 2015 1 hour For s use Question Maximum Mark Mark Awarded
Surname Centre Number Candidate Number Other Names 0 GCSE 4462/01 W15-4462-01 SCIENCE A/CHEMISTRY CHEMISTRY 1 FOUNDATION TIER A.M. Tuesday, 13 January 2015 1 hour For s use Question Maximum Mark Mark Awarded
Boiling point in C. Colour in aqueous solution. Fluorine 188 colourless. Chlorine 35 pale green. Bromine X orange.
 Q1.This question is about halogens and their compounds. The table below shows the boiling points and properties of some of the elements in Group 7 of the periodic table. Element Boiling point in C Colour
Q1.This question is about halogens and their compounds. The table below shows the boiling points and properties of some of the elements in Group 7 of the periodic table. Element Boiling point in C Colour
Combination Reactions 2H 2 + O 2 2H 2 O. 2Na + I 2 2NaI. Ca + Cl 2 CaCl 2. C + O 2 CO 2 or 2C + O 2 2CO 3H 2 + N 2 2NH 3
 Combination Reactions Predict the product and write balanced reactions for each of the following. 1. Hydrogen burned in oxygen. 2H 2 + O 2 2H 2 O 2. Sodium plus iodine. 2Na + I 2 2NaI 3. Calcium burned
Combination Reactions Predict the product and write balanced reactions for each of the following. 1. Hydrogen burned in oxygen. 2H 2 + O 2 2H 2 O 2. Sodium plus iodine. 2Na + I 2 2NaI 3. Calcium burned
Compiled by Rahul Arora What do you mean by corrosion? How can you prevent it?
 Rahul Arora 12. What do you mean by corrosion? How can you prevent it? 13. MnO2 + 4HCl MnCl2 + 2H2O + Cl2 In the above equation, name the compound which is oxidized and which is reduced? 14. Match the
Rahul Arora 12. What do you mean by corrosion? How can you prevent it? 13. MnO2 + 4HCl MnCl2 + 2H2O + Cl2 In the above equation, name the compound which is oxidized and which is reduced? 14. Match the
EPA Primary. (mg/l as CaCO3) (mg/l as CaCO3)
 NORTH TEXAS MUNICIPAL WATER DISTRICT - Wylie Water Analysis Jan-2018 Mineral Analysis Raw Treated Standards Residue on Evaporation 412 456 500 1000 Silica (SiO2) 3.63 3.41 Iron (Fe) 0.378 0.259 0.3 0.3
NORTH TEXAS MUNICIPAL WATER DISTRICT - Wylie Water Analysis Jan-2018 Mineral Analysis Raw Treated Standards Residue on Evaporation 412 456 500 1000 Silica (SiO2) 3.63 3.41 Iron (Fe) 0.378 0.259 0.3 0.3
A.M. MONDAY, 18 January minutes
 Candidate Name Centre Number Candidate Number 0 GCSE 240/02 ADDITIONAL SCIENCE IGER TIER CEMISTRY 2 A.M. MONDAY, 18 January 2010 45 minutes ADDITIONAL MATERIALS In addition to this paper you may require
Candidate Name Centre Number Candidate Number 0 GCSE 240/02 ADDITIONAL SCIENCE IGER TIER CEMISTRY 2 A.M. MONDAY, 18 January 2010 45 minutes ADDITIONAL MATERIALS In addition to this paper you may require
Our country, our future S2 CHEMISTRY DURATION: 2 HOUR
 Our country, our future S2 CHEMISTRY Exam 1 DURATION: 2 HOUR INSTRUCTIONS: This paper consists of two sections A and B, Attempt all questions in section A and B For section A, circle the most correct alternative
Our country, our future S2 CHEMISTRY Exam 1 DURATION: 2 HOUR INSTRUCTIONS: This paper consists of two sections A and B, Attempt all questions in section A and B For section A, circle the most correct alternative
[ Cl ] - [[Mg 2+ ] ] Experiment 7: Oxidation-Reduction Reactions. transfer e -
![[ Cl ] - [[Mg 2+ ] ] Experiment 7: Oxidation-Reduction Reactions. transfer e - [ Cl ] - [[Mg 2+ ] ] Experiment 7: Oxidation-Reduction Reactions. transfer e -](/thumbs/74/70175814.jpg) Experiment 7: OxidationReduction Reactions PURPOSE Become familiar with the concepts of oxidation and reduction and how these reactions occur. Carry out several such reactions and learn to recognize when
Experiment 7: OxidationReduction Reactions PURPOSE Become familiar with the concepts of oxidation and reduction and how these reactions occur. Carry out several such reactions and learn to recognize when
3. Give the molecular formula for the following ionic compounds (note both fixed and variable charged metals)
 Dr. Taylor Naming ompounds Practice Fall 2007 1. Give the name for the following ionic compounds containing fixed charge metals: A. Li 2 B. K 2 r 4. sl D. Rb 3 2 E. MgS F. Mg 3 N 2 G. abr 2 4 2. a 3 P
Dr. Taylor Naming ompounds Practice Fall 2007 1. Give the name for the following ionic compounds containing fixed charge metals: A. Li 2 B. K 2 r 4. sl D. Rb 3 2 E. MgS F. Mg 3 N 2 G. abr 2 4 2. a 3 P
ICSE-Science 2 (Chemistry) 2004
 ICSE-Science 2 (Chemistry) 2004 Answers to this Paper must be written on the paper provided separately. You will not be allowed to write during the first 15 minutes. This time is to be spent in reading
ICSE-Science 2 (Chemistry) 2004 Answers to this Paper must be written on the paper provided separately. You will not be allowed to write during the first 15 minutes. This time is to be spent in reading
Q1. From the following list of substances, choose the substances which meet the description given in parts (i) to (v) below :
 Questions:- Q1. From the following list of substances, choose the substances which meet the description given in parts (i) to (v) below : Ammonium chloride, ammonium nitrate, chlorine, dilute hydrochloric
Questions:- Q1. From the following list of substances, choose the substances which meet the description given in parts (i) to (v) below : Ammonium chloride, ammonium nitrate, chlorine, dilute hydrochloric
CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK. Due Date Assignment On-Time (100) Late (70)
 Name KEY Period CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK Due Date Assignment On-Time (100) Late (70) 1.1 1.2 1.3 Warm Ups Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic
Name KEY Period CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK Due Date Assignment On-Time (100) Late (70) 1.1 1.2 1.3 Warm Ups Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic
3.3 Minerals. Describe the characteristics that define minerals.
 3.3 Minerals Describe the characteristics that define minerals. Are you a mineral? There used to be a TV commercial that said "you are what you eat." If that s true - and to some extent it is - then you
3.3 Minerals Describe the characteristics that define minerals. Are you a mineral? There used to be a TV commercial that said "you are what you eat." If that s true - and to some extent it is - then you
Unit I PART I. MATH TOOLS FOR CHEMISTRY I. The Metric System The metric system is the scientific system of units of measurement
 CHEMISTRY 100 LECTURE Unit I PART I. MATH TOOLS FOR CHEMISTRY I. The Metric System The metric system is the scientific system of units of measurement Length Volume Mass METRIC BASIC UNITS LENGTH MASS VOLUME
CHEMISTRY 100 LECTURE Unit I PART I. MATH TOOLS FOR CHEMISTRY I. The Metric System The metric system is the scientific system of units of measurement Length Volume Mass METRIC BASIC UNITS LENGTH MASS VOLUME
*16GSD2201* Double Award Science: Chemistry. Unit C1 Higher Tier THURSDAY 19 MAY 2016, MORNING [GSD22] *GSD22* TIME 1 hour.
![*16GSD2201* Double Award Science: Chemistry. Unit C1 Higher Tier THURSDAY 19 MAY 2016, MORNING [GSD22] *GSD22* TIME 1 hour. *16GSD2201* Double Award Science: Chemistry. Unit C1 Higher Tier THURSDAY 19 MAY 2016, MORNING [GSD22] *GSD22* TIME 1 hour.](/thumbs/85/92047421.jpg) Centre Number Candidate Number General Certificate of Secondary Education 2015 2016 Double Award Science: Chemistry Unit C1 Higher Tier [GSD22] *GSD22* *G5802* *GSD22* THURSDAY 19 MAY 2016, MORNING TIME
Centre Number Candidate Number General Certificate of Secondary Education 2015 2016 Double Award Science: Chemistry Unit C1 Higher Tier [GSD22] *GSD22* *G5802* *GSD22* THURSDAY 19 MAY 2016, MORNING TIME
The table gives some information about a family of molecules in crude oil. Show information from the table in the most appropriate way on the grid.
 ## The table gives some information about a family of molecules in crude oil. NUMBER OF CARBON ATOMS IN MOLECULE MASS OF MOLECULE (atomic units) 1 16 2 30 4 58 Show information from the table in the most
## The table gives some information about a family of molecules in crude oil. NUMBER OF CARBON ATOMS IN MOLECULE MASS OF MOLECULE (atomic units) 1 16 2 30 4 58 Show information from the table in the most
P.M. THURSDAY, 15 January minutes
 Candidate Name Centre Number Candidate Number 0 GCSE 236/01 SCIENCE FOUNDATION TIER CHEMISTRY 1 P.M. THURSDAY, 15 January 2009 45 minutes For Examiner s use Total Mark ADDITIONAL MATERIALS In addition
Candidate Name Centre Number Candidate Number 0 GCSE 236/01 SCIENCE FOUNDATION TIER CHEMISTRY 1 P.M. THURSDAY, 15 January 2009 45 minutes For Examiner s use Total Mark ADDITIONAL MATERIALS In addition
Inorganic Reactions and Methods Volume 2
 Inorganic Reactions and Methods Volume 2 The Formation of the Bond to Hydrogen (Part 2) Editor J.J. Zuckerman Subject Index Editor A.P. Hagen Contents of Volume 2 How to Use this Book Preface to the Series
Inorganic Reactions and Methods Volume 2 The Formation of the Bond to Hydrogen (Part 2) Editor J.J. Zuckerman Subject Index Editor A.P. Hagen Contents of Volume 2 How to Use this Book Preface to the Series
Electrochemistry Written Response
 Electrochemistry Written Response January 1999 7. Balance the following redox reaction in acidic solution: RuO 4 + P Ru(OH) 2 2+ + H 3 PO 3 (acid) (3 marks) 8. A technician tests the concentration of methanol,
Electrochemistry Written Response January 1999 7. Balance the following redox reaction in acidic solution: RuO 4 + P Ru(OH) 2 2+ + H 3 PO 3 (acid) (3 marks) 8. A technician tests the concentration of methanol,
TWEED RIVER HIGH SCHOOL 2006 PRELIMINARY CHEMISTRY. Unit 2 Metals
 TWEED RIVER HIGH SCHOOL 2006 PRELIMINARY CHEMISTRY Unit 2 Metals Part 2 Metals differ in their reactivity with other chemicals and this influences their uses. Describe observable changes when metals react
TWEED RIVER HIGH SCHOOL 2006 PRELIMINARY CHEMISTRY Unit 2 Metals Part 2 Metals differ in their reactivity with other chemicals and this influences their uses. Describe observable changes when metals react
ADDITIONAL SCIENCE/CHEMISTRY
 Surname Centre Number Candidate Number Other Names 0 GCSE 4472/02 S15-4472-02 ADDITIONAL SCIENCE/CHEMISTRY CHEMISTRY 2 HIGHER TIER A.M. THURSDAY, 14 May 2015 1 hour For s use Question Maximum Mark Mark
Surname Centre Number Candidate Number Other Names 0 GCSE 4472/02 S15-4472-02 ADDITIONAL SCIENCE/CHEMISTRY CHEMISTRY 2 HIGHER TIER A.M. THURSDAY, 14 May 2015 1 hour For s use Question Maximum Mark Mark
Solutions Unit Exam Name Date Period
 Name Date Period Ms. Roman Page 1 Regents Chemistry 1. Which mixture can be separated by using the equipment shown below? 6. Which ion, when combined with chloride ions, Cl, forms an insoluble substance
Name Date Period Ms. Roman Page 1 Regents Chemistry 1. Which mixture can be separated by using the equipment shown below? 6. Which ion, when combined with chloride ions, Cl, forms an insoluble substance
Please write the balanced net ionic reaction for each one. Then answer the accompanying question.
 AP Chemistry Net Ionic Rx Practice Test A CLASS SET PLEASE RETURN!! Please write the balanced net ionic reaction for each one. Then answer the accompanying question. 1) A piece of potassium is dropped
AP Chemistry Net Ionic Rx Practice Test A CLASS SET PLEASE RETURN!! Please write the balanced net ionic reaction for each one. Then answer the accompanying question. 1) A piece of potassium is dropped
I. PHYSICAL PROPERTIES PROPERTY METALS NON-METALS
 Elements can be classified as metals and non-metals on the basis of their properties. Example of some metals are : Iron (Fe), Aluminium (Al), Silver (Ag), Copper (Cu) Examples of some non-metals are :
Elements can be classified as metals and non-metals on the basis of their properties. Example of some metals are : Iron (Fe), Aluminium (Al), Silver (Ag), Copper (Cu) Examples of some non-metals are :
GCSE 0236/02 SCIENCE HIGHER TIER CHEMISTRY 1
 Surname Other Names Centre Number 0 Candidate Number GCSE 0236/02 SCIENCE IGER TIER CEMISTRY 1 A.M. TUESDAY, 12 June 2012 45 minutes For s use Question Maximum Mark 1. 7 Mark Awarded 2. 8 ADDITIONAL MATERIALS
Surname Other Names Centre Number 0 Candidate Number GCSE 0236/02 SCIENCE IGER TIER CEMISTRY 1 A.M. TUESDAY, 12 June 2012 45 minutes For s use Question Maximum Mark 1. 7 Mark Awarded 2. 8 ADDITIONAL MATERIALS
Chapter: The d and f Block Elements
 Chapter: The d and f Block Elements Introduction to d block elements Question 1 In Tc ( Z = 43) and Tb( Z = 65) which one is inner transition metal and which one is transition metal and why? The outer
Chapter: The d and f Block Elements Introduction to d block elements Question 1 In Tc ( Z = 43) and Tb( Z = 65) which one is inner transition metal and which one is transition metal and why? The outer
METALLIC CRYSTALS. tend to be densely packed. have several reasons for dense packing: have the simplest crystal structures.
 METALLIC CRYSTALS tend to be densely packed. have several reasons for dense packing: -Typically, only one element is present, so all atomic radii are the same. -Metallic bonding is not directional. -Nearest
METALLIC CRYSTALS tend to be densely packed. have several reasons for dense packing: -Typically, only one element is present, so all atomic radii are the same. -Metallic bonding is not directional. -Nearest
GCSE 4462/01 CHEMISTRY 1 FOUNDATION TIER SCIENCE A/CHEMISTRY. P.M. FRIDAY, 12 June hour JUN S
 Surname Centre Number Candidate Number Other Names 0 GCSE 4462/01 S15-4462-01 SCIENCE A/CHEMISTRY CHEMISTRY 1 FOUNDATION TIER P.M. FRIDAY, 12 June 2015 1 hour For s use Question Maximum Mark Mark Awarded
Surname Centre Number Candidate Number Other Names 0 GCSE 4462/01 S15-4462-01 SCIENCE A/CHEMISTRY CHEMISTRY 1 FOUNDATION TIER P.M. FRIDAY, 12 June 2015 1 hour For s use Question Maximum Mark Mark Awarded
Chemical Elements and the Periodic Table
 Chemical Elements and the Periodic Table Periodic Table Odd elements quiz Some of the more obscure elements are ever so odd. Which of these are too strange to be true? Mark words you find difficult/important!
Chemical Elements and the Periodic Table Periodic Table Odd elements quiz Some of the more obscure elements are ever so odd. Which of these are too strange to be true? Mark words you find difficult/important!
General Certificate of Secondary Education ADDITIONAL SCIENCE. FOUNDATION TIER (Grades G-C) P.M. FRIDAY, 18 January 2008 (45 minutes)
 Candidate Name Centre Number Candidate Number General Certificate of Secondary Education 24/1 ADDITIONAL SCIENCE FOUNDATION TIER (Grades G-C) CHEMISTRY 2 P.M. FRIDAY, 18 January 28 (45 minutes) For Examiner
Candidate Name Centre Number Candidate Number General Certificate of Secondary Education 24/1 ADDITIONAL SCIENCE FOUNDATION TIER (Grades G-C) CHEMISTRY 2 P.M. FRIDAY, 18 January 28 (45 minutes) For Examiner
Extracting Metals by Electrolysis
 Extracting Metals by Electrolysis 1 of 17 Boardworks Ltd 2016 Extracting Metals by Electrolysis 2 of 17 Boardworks Ltd 2016 Methods of extracting metals 3 of 17 Boardworks Ltd 2016 Most metals are found
Extracting Metals by Electrolysis 1 of 17 Boardworks Ltd 2016 Extracting Metals by Electrolysis 2 of 17 Boardworks Ltd 2016 Methods of extracting metals 3 of 17 Boardworks Ltd 2016 Most metals are found
