Lab Question from Board papers Class-09[2018] 1. How will you separate a mixture of iron filings, iodine and common salt?
|
|
|
- Frederica Manning
- 6 years ago
- Views:
Transcription
1 Lab Question from Board papers Class-09[2018] SET-1 Short Answer Type Questions (2 marks) 1. How will you separate a mixture of iron filings, iodine and common salt? Ans. (i) Remove iron filings with the help of magnet. (ii) Iodine can be removed by sublimation whereas common salt will be left behind. 2. Write three observations when iron filings and Sulphur are mixed together in a china dish. Ans. (i) A heterogeneous mixture results. (ii) The constituent particles present can be seen. (ii,) The Constituents can be separated by magnet, physical method. 3. Give four properties of FeS. Ans. (i) It is greyish-black in colour. (ii) It does not dissolve in carbon disulphide. (iii) It liberates H,S when treated with dil H2SO4. (iv) It is not attracted by magnet. 4. What is the formula of iron sulphide? Can we separate FeS into iron and sulphur by physical method? Ans. FeS is the formula of iron sulphide. We cannot separate elements of a compound by physical method. 5. What precautions should we observe while handling carbon disulphide and why? Ans. (i) It should be kept away from flame because it is highly inflammable (catches fire). (ii) It should be kept in airtight bottle because it is highly volatile (vaporizable). SET-2 Short Answer Type Questions (2 marks) 1. On burning magnesium ribbon in air, a white ash is obtained. Name the product and type of change. Ans. 2Mg + 2Mg0(s) Magnesium oxide is formed and it is a chemical change. 2. What are observations when zinc granules are added to dil. sulphuric acid in a test tube? Ans. (i) Colourless, odourless gas with bubbles comes out. (ii) Gradual decrease in size of zinc granules. (iii) Heat energy is evolved in the reaction. P a g e 1
2 3. What is the formula of hydrated copper sulphate? What is its colour? What will be the formula and colour of compound obtained on heating. Ans. CuSO4.5H2O is blue coloured hydrated copper sulphate. CuSO4, is anhydrous copper sulphate which is dirty white. 4.What happens when iron nails are added to copper sulphate solution? What do you conclude about reactivity of iron and copper? Ans. Reddish brown precipitate of copper is formed and solution becomes pale green. It shows iron is more reactive than copper. 5. What happens when barium chloride solution is mined with sodium sulphate solution? What type of reaction it is? Ans. White ppt. of BaSO 4 will be formed. It is precipitation reaction. SET-3 Short Answer Type Questions (2 marks.) 1. What are precautions to be observed while conducting experiment to find relationship between weight of block and applied force to just balance it? Ans. (i) Putty should be frictionless. (ii) Spring balance should not have zero error. If it has, it should be taken into consideration. (iii) The hanging pan should be vertical. 2. What type of thread should be used to move the block kept horizontally on the surface and why? Ans. It should be less elastic, unable tube stretched. It is because force applied is exerted only on the block and not on the thread. 3. Define coefficient of limiting fiction. Ans. It is ratio of force of friction to the normal reaction. 4. State two factors which determine the magnitude of limiting friction. Ans. (i)the nature of surface. (ii) The normal reaction force between two surfaces. SET- 4 Short Answer Type Questions (2 marks.) 1. How will you prepare colloidal solution of starch? [CBSE (CCE) 2014] Ans. (i) Make a thin paste of starch. (ii) Add thin paste of starch to boiling water with constant stirring. P a g e 2
3 2. What is correct procedure for preparing colloidal solution of egg albumin? Ans. (i) Break the egg shell. (ii) Take only the white portion. (iii) Add it to water. (iv) Keep on stirring continuously. 3. Give three characteristics of colloidal solution by which it can be identified. Ans. (i) Components cannot be separated by ordinary filtration. (ii) It is translucent. (iii)it shows Tyndall effect. 4. Give two features of true solution by which it can be identified. Ans. (i) It is homogeneous and transparent. (ii) It passes through filter paper. 5. How will you identify suspension? Ans. (i) It is opaque. (ii) The components can be separated by filtration. (iii) Particles settle down due to gravity after some time. (iv) Particles can be seen by naked eye. SET-5 Short Answer Type Questions (2 marks) 1. Four students were asked to add water to glucose powder, milk, sand and soil separately in four beakers. Classify the mixtures as a true solution, colloid and suspension: [CBSE (CCE) 2014] Ans. Glucose will form true solution. Milk will form colloidal solution. Sand and soil will form suspension. 2. Rama took a fine chalk powder, egg albumin starch powder and alum powder in four test tubes A, B, C and D. After adding water to all of test tubes, identify the test tube as true solution, suspension, and colloidal. [CBSE (CCE Ans. 'A' will have suspension, 'B', 'C' will have colloidal solution and 'D' will have true solution. 3. When light passes through a solution of alum in water one cannot see the path of light through it, why? Give two examples of solution where similar results will be observed. [CBSE (CCE) 2015] Ans. It is because particles of true solution are very small and cannot scatter light. Sugar solution and salt solution are examples which will show similar results. P a g e 3
4 4. Identify two clear and transparent solutions from the following mixtures: [CBSE (CCE) 2015] (i) Milk and water (ii) sugar and water (iii) chalk powder and water (iv) Starch powder and water (v) glucose and water Ans. (ii) and (n) are clear and transparent solutions because these are true solutions. 5. You are asked to prepare aqueous solutions of sodium chloride and starch powder. How would you compare them on the basis of transparency and stability? [CBSE (CCE) 2015] Ans. Aqueous solution of sodium chloride will be transparent and more stable. Colloidal solution of starch is translucent and less stable than sodium chloride solution. SET- 6 Short Answer Type Questions (2 marks.) 1. (a) Mention the phylum of the following animals; (i) Earthworm (ii) Cockroach (b) Funaria differs from Dryopteris, How? (give one point) Ans: (a) (i) Annelida (ii) Arthropoda (b) Dryopteris belong to pteridophyta while funaria belongs to bryophyta. 2. Write any two precautions that you will take while preparing a stained temporary mount of cheek cells. Ans: Precautions Ensure toothpick used to scrape the cheek is clean, so it does not cause infection to the cheek. Extra glycerine stain should be removed using blotting paper. 3. Classify the following as chemical or physical changes: (i) Reaction of barium chloride solution with sodium sulphate solution (p) (ii) Passing of electric current, through water and the water breaking down into hydrogen and oxygen gases.(c) (iii) Dissolving common salt in water. (p) (iv) Heating sulphur powder with iron. (p) P a g e 4
5 4. (i) If we want to determine the volume of a solid by immersing it in water, then whatkind of solid should we take? (ii) If a body floats on the surface of a liquid, then how much is the weight of the liquid displaced as compared to weight of body? Ans: (i) The solid should be heavier than water and insoluble in it. (ii) Equal to the weight of the body. 5. In an experiment to determine the loss weight of an object when immersed in a liquid, on what factors the force acting on an object depends Ans. The weight of a body acting downwards, up thrust on the body due to liquid acting vertically upward. 6. Identify the given organism in the picture given below and write its one specific feature and one adaptive feature Ans: Picture is - Cockroach. Specific feature - Jointed legs, compound eye. Adaptive feature - Omnivorous, body divided into head, thorax and abdomen. SET-7 Short Answer Type Questions (2 marks) 1. Write two important precautions to be taken in an experiment to determine the density of a solid by using a spring balance and a measuring cylinder. Ans. (i)while weighing body should be fully immersed in water / liquid the solid should not touch the walls of the beaker or cylinder. (ii) Lower meniscus of the water level in the measuring cylinder should be read. 2. In an experiment on measurement of loss in weight of an iron ball immersed in tap water and salty water separately, when is the maximum loss in weight of iron ball observed? Give reason. Ans. In tap water because tap water is less dense than salty water and exerts less up thrust hence maximum loss in weight in the solid. 3.(a) Animals of which phylum have jointed appendages? (b) Why cockroaches are generally found in kitchens or godown? P a g e 5
6 Ans. (a) Arthropoda (b) Because they are nocturnal insects and are found in places where moisture and warmth are available. 4. A piece of copper of mass 106g is dipped in a measuring cylinder containing water at 70ml mark. The water rises to 82ml mark. Find (a) volume of copper piece (b) density of copper. (c) Relative density of copper Ans: (a) Volume of copper piece = V2 V1 = = 13 ml =13cc (b) Density of copper = m/v = 106/13 = 8.15 g/cc (c) Relative density of copper = Density of copper/(density of water) = 8.15/1 = A student uses a spring balance of least count 10g wt and range 500 g wt. he records the weight of a small iron cube in air, in tap water and in salty water. If his three readings taken in this order are W1, W2 and W3. Relate the three readings W1, W2 and W3 using Archimedes principle. Ans: W 1 > W 2 > W 3 It is because greater the density of the fluid greater up thrust will be reduced by the fluid into the object SET-8 Short Answer Type Questions (2 marks) 1. An object of volume 200 cm3 is floating on a fluid with half of its portion inside the fluid as below. Find the volume and weight of the fluid displaced by the object Ans: If a object is floating half inside (i) Volume of water displaced = volume of object inside water = 100cm 3 [half portion of object inside the fluid] (iii) Weight of water displaced = Vdg = 100cm 3 x 1 g/cm m/s 2 3 gm/cm 3 = 980N 2. A cube of copper and spherical ball of iron having the same volume, immersed is salty water. Find the buoyant force acting on these objects with reasons. Ans. A cube of copper and spherical ball of iron experience same amount of buoyant force in both cases because buoyant force canot depend on shape of object. As both cubical and spherical have the same volume, same amount of water (may be salty) will get displaced and hence same buoyant force in both cases P a g e 6
7 3. The volume of a solid cube of copper of mass 900 g is 100 cm3. This cube is immersed in water. Find the mass of water displaced by it? If this cube is immersed in salt solution then how will the mass of liquid displaced by the cube change? Give reason for your answer. Ans: (i) Volume of water displaced = volume of object inside water = 100cm 3 (ii) Masss of water = density of water x Volume of water = 100cm 3 x 1g/cm3 = 100g No change occur as volume of water depends on volume of object not on the density of fluid 4. Write two main precautions to be taken to read the water level in the graduated cylinder. Ans: When reading a scale on the side of an instrument filled up with a Colourless liquid in this case water, we have to take the reading of the lower meniscus. However in case of colored solutions or mercury the reading of upper meniscus is taken. The reading has to be taken at the same level as that of the eye to prevent any parallax error. 5. In which of the two, glycerine or kerosene, the loss in weight of a solid when fully immersed in them will be more and why? Ans. The weight loss of given solid will be more in glycerine than in kerosene.this is because density of glycerine (1.26g/cm³) is more than density of Kerosene (0.8 g/cm³) SET-9 Short Answer Type Questions (2 marks) 1. In an experiment on measurement of loss in weight of an iron ball immersed in tap water and salty water separately, when is the maximum loss in weight of iron ball observed? Give reason. Ans. As the density of the salty water is higher than the tap water. Therefore the wt. of the iron ball is less in salty water. 2(i) If we want to determine the volume of a solid by immersing it in water, then what kind of solid should we take? (ii) If a body floats on the surface of a liquid, then how much is the weight of the liquid displaced as compared to weight of body? Ans. (i) solid must be heavier and insoluble in water =>heavier because as it should get completely immersed in water => insoluble as it will not dissolve in water which will tend to give the wrong weight (ii) the weight of the liquid displaced is equal to weight of body P a g e 7
8 3. In an experiment to establish the relation between the loss in weight of a solid when fully immersed in tap and salty water separately a student inferred the correct result as follows: (Complete the result by filling appropriate words) (a) The loss in weight of the solid immersed in the liquid is to the weight of the liquid displaced by that solid which proves the Principle. (b) The loss in weight of the solid in strongly salty water is as compared to the tap water for the same solid as up thrust in salty water is than in tap water. Ans.(i) Equal,Archimedes principle (ii) more, greater 4. If the weight of displaced water by an object weighing 50 N is 10 N, then find the buoyant force of water on the object. What will be the weight of object in water? Ans: Buoyant force act on object = weight loss = 50N -10N = 40N 5. Mention the type of thermometer that should be used to determine the melting point of ice in laboratory? What should be the position of bulb in thermometer? Ans. Thermometer used for measuring the melting point of ice is laboratory thermometer. The two precautions includes : (i) Thermometer knob should not touch the bottom. (ii) Ice should be perfectly crushed. SET-10 Short Answer Type Questions (2 marks) 1. After a solid starts melting, we observe that the temperature remains constant until the whole of the solid has melted. Where the energy does goes? Ans. After solid start melting, the temperature remain constant until all the solid get melted because the energy is used to overcome the latent heat of fusion and in breaking the strong intermolecular force of attraction. 2. Sheema was asked to prepare four separate mixtures in four beakers A, B, C, D by mixing sugar, fine sand, thin paste of starch and chalk powder respectively in water and then categories each as stable or unstable. What will be the correct categorization? Ans. The mixture in beaker A and C will be stable. The mixture in beaker B and D will be unstable. P a g e 8
INDIAN SCHOOL MUSCAT SENIOR SECTION DEPARTMENT OF CHEMISTRY CLASS X- PRACTICAL WORKSHEET
 INDIAN SCHOOL MUSCAT SENIOR SECTION DEPARTMENT OF CHEMISTRY CLASS X- PRACTICAL WORKSHEET Different types of chemical reactions Experiment No: 1(a) Combination reaction Objectives: To study the Combination
INDIAN SCHOOL MUSCAT SENIOR SECTION DEPARTMENT OF CHEMISTRY CLASS X- PRACTICAL WORKSHEET Different types of chemical reactions Experiment No: 1(a) Combination reaction Objectives: To study the Combination
Properties A Metal B Non- metal Electronic configuration?? Nature of oxides?? Oxidizing or reducing action?? Conduction of heat and electricity??
 CLASS: X NCERT (CBSE) SCIENCE: Chemistry Page: 1 Question 1: Compare the properties of a typical metal and a non-metal on the basis of the following. Fill in Column A, B. Properties A Metal B Non- metal
CLASS: X NCERT (CBSE) SCIENCE: Chemistry Page: 1 Question 1: Compare the properties of a typical metal and a non-metal on the basis of the following. Fill in Column A, B. Properties A Metal B Non- metal
Covered with a thin layer of oxide at ordinary temperatures.
 1 More about Metals Physical properties of metals In general metals have luster, are malleable and ductile, good conductors of heat and electricity and have high boiling and melting points and nonmetals
1 More about Metals Physical properties of metals In general metals have luster, are malleable and ductile, good conductors of heat and electricity and have high boiling and melting points and nonmetals
CBSE QUESTION PAPER CLASS-X SCIENCE
 CBSE QUESTION PAPER CLASS-X SCIENCE SECTION - A Q.1. Why decomposition reactions are called the opposite of combination reactions? 1mark Q.2. What happens to the resistance of a conductor when its area
CBSE QUESTION PAPER CLASS-X SCIENCE SECTION - A Q.1. Why decomposition reactions are called the opposite of combination reactions? 1mark Q.2. What happens to the resistance of a conductor when its area
look down at cross on paper paper cross on paper
 1 The equation for the reaction between sodium thiosulfate and hydrochloric acid is given below. Na 2 S 2 O 3 (aq) + 2HCl (aq) 2NaCl (aq) + S(s) + SO 2 (g) + H 2 O(l) The speed of this reaction was investigated
1 The equation for the reaction between sodium thiosulfate and hydrochloric acid is given below. Na 2 S 2 O 3 (aq) + 2HCl (aq) 2NaCl (aq) + S(s) + SO 2 (g) + H 2 O(l) The speed of this reaction was investigated
Iron filings (Fe) 56g IRON + SULPHUR IRON SULPHIDE
 W.S.51. Chemical reactions. All of the different materials around us have been formed by chemical reactions from about one hundred simple elements. The diagram below shows a chemical reaction between the
W.S.51. Chemical reactions. All of the different materials around us have been formed by chemical reactions from about one hundred simple elements. The diagram below shows a chemical reaction between the
EXPERIMENT 3: Identification of a Substance by Physical Properties
 EXPERIMENT 3: Identification of a Substance by Physical Properties Materials: Hot plate Digital balance Capillary tubes (3) Thermometer Beakers (250 ml) Watch glass Graduated Cylinder (10 ml) Mel-Temp
EXPERIMENT 3: Identification of a Substance by Physical Properties Materials: Hot plate Digital balance Capillary tubes (3) Thermometer Beakers (250 ml) Watch glass Graduated Cylinder (10 ml) Mel-Temp
CHEM 1215 LAB NOTES EXPT #2: PHYSICAL AND CHEMICAL CHANGES 1
 CHEM 1215 LAB NOTES EXPT #2: PHYSICAL AND CHEMICAL CHANGES 1 TECHNIQUES: chemical and physical changes, reactions, observations READING: PHYSICAL AND CHEMICAL CHANGES e.g. Tro chapter 1 SAFETY: Safety
CHEM 1215 LAB NOTES EXPT #2: PHYSICAL AND CHEMICAL CHANGES 1 TECHNIQUES: chemical and physical changes, reactions, observations READING: PHYSICAL AND CHEMICAL CHANGES e.g. Tro chapter 1 SAFETY: Safety
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS Cambridge Checkpoint SCIENCE
 Centre Number Candidate Number Name www.xtremepapers.com UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS Cambridge Checkpoint SCIENCE 1113/02 Paper 2 November 2005 Candidates answer on the Question
Centre Number Candidate Number Name www.xtremepapers.com UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS Cambridge Checkpoint SCIENCE 1113/02 Paper 2 November 2005 Candidates answer on the Question
85 Q.51 Which of the following carbonates would give the metal when heated with carbon? (1) MgCO 3 (2) PbCO 3 (3) K 2 CO 3 (4) CuCO 3
 Metal and metal reactivity / Section 2 / Sect2pp.doc / S. W. Tse / P.1 85 Q.51 Which of the following carbonates would give the metal when heated with carbon? (1) MgCO 3 (2) PbCO 3 (3) K 2 CO 3 (4) CuCO
Metal and metal reactivity / Section 2 / Sect2pp.doc / S. W. Tse / P.1 85 Q.51 Which of the following carbonates would give the metal when heated with carbon? (1) MgCO 3 (2) PbCO 3 (3) K 2 CO 3 (4) CuCO
METALS AND THEIR COMPOUNDS
 METALS AND THEIR COMPOUNDS Metals are elements whose atoms ionize by electron loss, while non-metals are elements whose atoms ionize by electron gain. Metals are in groups 1, 2 and 3 of the periodic table.
METALS AND THEIR COMPOUNDS Metals are elements whose atoms ionize by electron loss, while non-metals are elements whose atoms ionize by electron gain. Metals are in groups 1, 2 and 3 of the periodic table.
To study the chemical reaction of an iron nail with aqueous copper sulphate solution; and to study the burning of magnesium ribbon in air.
 Materials 1 AIM To study the chemical reaction of an iron nail with aqueous copper sulphate solution; and to study the burning of magnesium ribbon in air. (a) Chemical reaction of iron nail with copper
Materials 1 AIM To study the chemical reaction of an iron nail with aqueous copper sulphate solution; and to study the burning of magnesium ribbon in air. (a) Chemical reaction of iron nail with copper
Oxidation and Reduction
 Oxidation and Reduction An oxidation reaction is one in which oxygen is added to a substance. Example: Methane is oxidised when it burns in air. Oxygen is added to the carbon in methane, forming carbon
Oxidation and Reduction An oxidation reaction is one in which oxygen is added to a substance. Example: Methane is oxidised when it burns in air. Oxygen is added to the carbon in methane, forming carbon
Skills in Science. Lab equipment. (Always draw 2D) Drawings below are NOT to scale. Beaker - A general purpose container with a pouring lip.
 Skills in Science Safety: Do NOT enter or leave the lab without permission from a teacher. Keep the gaps between tables clear of stools and bags. Never run in the lab. Do not throw things around in the
Skills in Science Safety: Do NOT enter or leave the lab without permission from a teacher. Keep the gaps between tables clear of stools and bags. Never run in the lab. Do not throw things around in the
Partner: Cathy 22 March Separation and Qualitative Determination of Cations and Anions
 Partner: Cathy 22 March 2012 Separation and Qualitative Determination of Cations and Anions Purpose: The purpose of this lab is to identify the cations and anions components in the unknown solution. This
Partner: Cathy 22 March 2012 Separation and Qualitative Determination of Cations and Anions Purpose: The purpose of this lab is to identify the cations and anions components in the unknown solution. This
For more sample papers visit :
 For more sample papers visit : www.4ono.com CBSE Sample Paper-02 SCIENCE (Theory)Class X Time allowed: 3 hours Maximum Marks: 90 General Instructions: a) All questions are compulsory. b) The question paper
For more sample papers visit : www.4ono.com CBSE Sample Paper-02 SCIENCE (Theory)Class X Time allowed: 3 hours Maximum Marks: 90 General Instructions: a) All questions are compulsory. b) The question paper
To identify and classify various types of chemical reactions.
 Cycle of Copper Reactions Minneapolis Community and Technical College v.11.17 Objectives: To observe and document copper s chemical changes in five different reactions and verify that copper is conserved
Cycle of Copper Reactions Minneapolis Community and Technical College v.11.17 Objectives: To observe and document copper s chemical changes in five different reactions and verify that copper is conserved
The table below shows some of the properties of the elements cobalt and nickel.
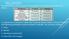 BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet
 Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet Name: Sec: Date: Q1.Choose the best answer. 1. Which of the following is an element? a) steam b) sugar c)dry ice d) sulphur 2. Which
Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet Name: Sec: Date: Q1.Choose the best answer. 1. Which of the following is an element? a) steam b) sugar c)dry ice d) sulphur 2. Which
CONSERVATION OF MATTER AND CHEMICAL PROPERTIES
 1 CONSERVATION OF MATTER AND CHEMICAL PROPERTIES I. OBJECTIVES AND BACKGROUND The object of this experiment is to demonstrate the conservation of matter- or more particularly, the conservation of "atoms"
1 CONSERVATION OF MATTER AND CHEMICAL PROPERTIES I. OBJECTIVES AND BACKGROUND The object of this experiment is to demonstrate the conservation of matter- or more particularly, the conservation of "atoms"
Teknik Bioseparasi. Dina Wahyu. Genap/ Maret 2014
 4. Teknik Bioseparasi Dina Wahyu Genap/ Maret 2014 Outline Chemical Reaction Engineering 1 2 3 4 5 6 7 Pendahuluan mempelajari ruang lingkup teknik bioseparasi dan teknik cel disruption Teknik Pemisahan
4. Teknik Bioseparasi Dina Wahyu Genap/ Maret 2014 Outline Chemical Reaction Engineering 1 2 3 4 5 6 7 Pendahuluan mempelajari ruang lingkup teknik bioseparasi dan teknik cel disruption Teknik Pemisahan
Preparation of copper(ii) sulfate from copper(ii) nitrate
 Student s Name: Date: Background Preparation of copper(ii) sulfate from copper(ii) nitrate The purpose of this laboratory activity is to prepare copper(ii) sulfate from copper(ii) nitrate. This is done
Student s Name: Date: Background Preparation of copper(ii) sulfate from copper(ii) nitrate The purpose of this laboratory activity is to prepare copper(ii) sulfate from copper(ii) nitrate. This is done
Salinity in Seawater
 Salinity in Seawater Objective To familiarize students with the different methods used for measuring salinity of water. Introduction: Salinity exerts profound impacts on the marine environment. It controls
Salinity in Seawater Objective To familiarize students with the different methods used for measuring salinity of water. Introduction: Salinity exerts profound impacts on the marine environment. It controls
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
DETERMINATION of the EMPIRICAL FORMULA
 DETERMINATION of the EMPIRICAL FORMULA One of the fundamental statements of the atomic theory is that elements combine in simple whole number ratios. This observation gives support to the theory of atoms,
DETERMINATION of the EMPIRICAL FORMULA One of the fundamental statements of the atomic theory is that elements combine in simple whole number ratios. This observation gives support to the theory of atoms,
GRAVIMETRIC DETERMINATION OF SULFATE IN AN UNKNOWN SOLUTION
 GRAVIMETRIC DETERMINATION OF SULFATE IN AN UNKNOWN SOLUTION AIM The main objective of this experiment is to determine the concentration of sulfate ion in an unknown solution by using gravimetry. INTRODUCTION
GRAVIMETRIC DETERMINATION OF SULFATE IN AN UNKNOWN SOLUTION AIM The main objective of this experiment is to determine the concentration of sulfate ion in an unknown solution by using gravimetry. INTRODUCTION
A TEACHER-DIRECTED, GUIDED DISCOVERY ACTIVITY FOR ADVANCED PHYSICAL SCIENCE STUDENTS
 A TEACHER-DIRECTED, GUIDED DISCOVERY ACTIVITY FOR ADVANCED PHYSICAL SCIENCE STUDENTS 1 2 FEATURES OF THIS ACTIVITY Problem solving Cooperative learning Requires student reasoning Requires student initiative
A TEACHER-DIRECTED, GUIDED DISCOVERY ACTIVITY FOR ADVANCED PHYSICAL SCIENCE STUDENTS 1 2 FEATURES OF THIS ACTIVITY Problem solving Cooperative learning Requires student reasoning Requires student initiative
Labs Covered. Le Chatelier. Calculating Ka. Kitchen Chemistry Project. Calculating Kc. Redox lab
 Labs Covered Le Chatelier Calculating Kc Calculating Ka Redox lab Kitchen Chemistry Project LeChatelier Sample Questions Fe +3 (aq) + SCN -1 (aq) FeSCN +2 (aq) Light orange colourless red a) To create
Labs Covered Le Chatelier Calculating Kc Calculating Ka Redox lab Kitchen Chemistry Project LeChatelier Sample Questions Fe +3 (aq) + SCN -1 (aq) FeSCN +2 (aq) Light orange colourless red a) To create
MAHESH TUTORIALS I.C.S.E.
 ICSE X MAES TUTORIALS I.C.S.E. Marks : 80 Exam No. : MT/ICSE/I SPA 033SUBJECT : CEMISTRY Time : 2 hrs. Topics : Sulphuric acid, Ammonia, Analytical Chemistry, Organic Chemistry Cl, Nitric acid, Metallurgy
ICSE X MAES TUTORIALS I.C.S.E. Marks : 80 Exam No. : MT/ICSE/I SPA 033SUBJECT : CEMISTRY Time : 2 hrs. Topics : Sulphuric acid, Ammonia, Analytical Chemistry, Organic Chemistry Cl, Nitric acid, Metallurgy
6 METALS & NON METALS
 Grade-8 Chemistry Chapter- 6 METALS & NON METALS 1. What happens in each of the following cases? a) Magnesium is burnt in oxygen. Ans: When magnesium burns in oxygen it produces magnesium oxide with dazzling
Grade-8 Chemistry Chapter- 6 METALS & NON METALS 1. What happens in each of the following cases? a) Magnesium is burnt in oxygen. Ans: When magnesium burns in oxygen it produces magnesium oxide with dazzling
OC30 Conduct a qualitative experiment to detect the presence of dissolved solids in water samples, and test water for hardness (soap test)
 Chemistry: 6. Water Please remember to photocopy 4 pages onto one sheet by going A3 A4 and using back to back on the photocopier Syllabus OC14 Use cobalt chloride paper or anhydrous copper sulfate to test
Chemistry: 6. Water Please remember to photocopy 4 pages onto one sheet by going A3 A4 and using back to back on the photocopier Syllabus OC14 Use cobalt chloride paper or anhydrous copper sulfate to test
The Crystal Forest Favorite Holiday Demonstrations
 The Crystal Forest Favorite Holiday Demonstrations SCIENTIFIC Introduction Put a new twist on crystal growing. In this class participation demonstration, students cut out and assemble miniature trees and
The Crystal Forest Favorite Holiday Demonstrations SCIENTIFIC Introduction Put a new twist on crystal growing. In this class participation demonstration, students cut out and assemble miniature trees and
Periodic Trends and the Properties of Elements
 Page 1 - The Alkaline Earth Metals Introduction The periodic table is the most recognized symbol of chemistry across the world. It is a valuable tool that allows scientists not only to classify the elements
Page 1 - The Alkaline Earth Metals Introduction The periodic table is the most recognized symbol of chemistry across the world. It is a valuable tool that allows scientists not only to classify the elements
Estimation of total, permanent and temporary hardness of water (EDTA method)
 Estimation of total, permanent and temporary hardness of water (EDTA method) Expt. No.: Date: Aim: To estimate the amount of total, permanent and temporary hardness in the collected sample of water. A
Estimation of total, permanent and temporary hardness of water (EDTA method) Expt. No.: Date: Aim: To estimate the amount of total, permanent and temporary hardness in the collected sample of water. A
» Talc is a native, hydrous magnesium silicate, sometimes containing a small proportion of aluminum silicate.
 Change to read: Talc» Talc is a native, hydrous magnesium silicate, sometimes containing a small proportion of aluminum silicate. Packaging and storage Preserve in well closed containers. Identification
Change to read: Talc» Talc is a native, hydrous magnesium silicate, sometimes containing a small proportion of aluminum silicate. Packaging and storage Preserve in well closed containers. Identification
Equation Writing and Predicting Products Chemistry I Acc
 Introduction: Equation Writing and Predicting Products Chemistry I Acc If you examine your bicycle after it has been left out in the rain a number of times you will find that it has begun to rust. Rust
Introduction: Equation Writing and Predicting Products Chemistry I Acc If you examine your bicycle after it has been left out in the rain a number of times you will find that it has begun to rust. Rust
IDENTIFYING UNKNOWN SUBSTANCES
 IDENTIFYING UNKNOWN SUBSTANCES LAB 15 EXPERIMENT STUDENT BOOK Chapter 1, page 25 TOOLBOX Page 4 and 36 Goal Identify unknown substances with the help of different tests. 1. What is the independent variable
IDENTIFYING UNKNOWN SUBSTANCES LAB 15 EXPERIMENT STUDENT BOOK Chapter 1, page 25 TOOLBOX Page 4 and 36 Goal Identify unknown substances with the help of different tests. 1. What is the independent variable
5-4 Chemical changes Trilogy
 5-4 Chemical changes Trilogy.0 A student investigated the reaction of sodium carbonate with dilute hydrochloric acid. The student used the apparatus shown in Figure. Figure Sodium carbonate This is the
5-4 Chemical changes Trilogy.0 A student investigated the reaction of sodium carbonate with dilute hydrochloric acid. The student used the apparatus shown in Figure. Figure Sodium carbonate This is the
GENARAL INTRODUCTION TO METALLURGY :Std: XI-CHEMISTRY
 GENARAL INTRODUCTION TO METALLURGY :Std: XI-CHEMISTRY 1. What is matrix? The ore is generally associated with rock impurities like clay, sand etc. called gangue or matrix 2. What is mineral? The natural
GENARAL INTRODUCTION TO METALLURGY :Std: XI-CHEMISTRY 1. What is matrix? The ore is generally associated with rock impurities like clay, sand etc. called gangue or matrix 2. What is mineral? The natural
SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example,
 SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example, density, thermal or electrical conductivity, solubility,
SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example, density, thermal or electrical conductivity, solubility,
1. Elemental mercury is a shiny, silver-colored, dense liquid that flows easily. Are these characteristics of mercury physical or chemical properties?
 Chemistry Dr. Saulmon 2014-15 School Year Unit 2: Basics of Chemistry Problem Set 3 Wednesday, September 3, 2014 1. Elemental mercury is a shiny, silver-colored, dense liquid that flows easily. Are these
Chemistry Dr. Saulmon 2014-15 School Year Unit 2: Basics of Chemistry Problem Set 3 Wednesday, September 3, 2014 1. Elemental mercury is a shiny, silver-colored, dense liquid that flows easily. Are these
Boiling point in C. Colour in aqueous solution. Fluorine 188 colourless. Chlorine 35 pale green. Bromine X orange.
 Q1.This question is about halogens and their compounds. The table below shows the boiling points and properties of some of the elements in Group 7 of the periodic table. Element Boiling point in C Colour
Q1.This question is about halogens and their compounds. The table below shows the boiling points and properties of some of the elements in Group 7 of the periodic table. Element Boiling point in C Colour
Compounds & Reactions Week 1. Writing Formulas & Balancing Equations. Write the chemical formula for each molecular (covalent) compound.
 Compounds & Reactions Week 1 Name Writing Formulas & Balancing Equations Write the chemical formula for each ionic compound. 1. Lithium fluoride 2. Copper (II) chloride 3. Manganese (II) oxide 4. Potassium
Compounds & Reactions Week 1 Name Writing Formulas & Balancing Equations Write the chemical formula for each ionic compound. 1. Lithium fluoride 2. Copper (II) chloride 3. Manganese (II) oxide 4. Potassium
O 3, SIO 2. O, CAO, Al 2 AND MG IN ANT-HILL SOIL SAMPLES WITHIN ABRAKA TOWN IN NIGERIA
 Int. J. Agric.Sc & Vet.Med. 2014 Ekakitie A O and Osakwe A A, 2014 Research Paper ISSN 2320-3730 www.ijasvm.com Vol. 2, No. 3, August 2014 2014 www.ijasvm.com. All Rights Reserved DETERMINATION OF FE 2,
Int. J. Agric.Sc & Vet.Med. 2014 Ekakitie A O and Osakwe A A, 2014 Research Paper ISSN 2320-3730 www.ijasvm.com Vol. 2, No. 3, August 2014 2014 www.ijasvm.com. All Rights Reserved DETERMINATION OF FE 2,
Mark Scheme (Results) November 2009
 Scheme (Results) November 2009 IGCSE IGCSE Chemistry (4335) Paper F Edexcel Limited. Registered in England and Wales No. 4496750 Registered Office: One90 High Holborn, London WCV 7BH 4335_F Nov 2009 i
Scheme (Results) November 2009 IGCSE IGCSE Chemistry (4335) Paper F Edexcel Limited. Registered in England and Wales No. 4496750 Registered Office: One90 High Holborn, London WCV 7BH 4335_F Nov 2009 i
CHAPTER 6. Natural Mineral Mineral Content Elements In The Minerals Bauxite Aluminium oxide Aluminium, oxygen. Cassiterite Tin oxide Tin, oxygen
 CHAPTER 6 6.1 Minerals Found In The Earth s Crust mineral : is a naturally occurring solid element or compound with a definite crystalline structure and chemical composition. natural elements : gold, silver
CHAPTER 6 6.1 Minerals Found In The Earth s Crust mineral : is a naturally occurring solid element or compound with a definite crystalline structure and chemical composition. natural elements : gold, silver
Analysis of Calcium Carbonate Tablets
 Experiment 9 Analysis of Calcium Carbonate Tablets Prepared by Ross S. Nord, Eastern Michigan University PURPOSE To perform a gravimetric exercise to determine weight percent of active ingredient in a
Experiment 9 Analysis of Calcium Carbonate Tablets Prepared by Ross S. Nord, Eastern Michigan University PURPOSE To perform a gravimetric exercise to determine weight percent of active ingredient in a
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 3 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 3 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
OPERATORS PERSPECTIVE : OPTIMISATION OF A NEW PACKAGE WATER TREATMENT PLANT. Melina Entwistle. North East Water Authority
 OPERATORS PERSPECTIVE : OPTIMISATION OF A NEW PACKAGE WATER TREATMENT PLANT Paper Presented by : Melina Entwistle Author: Melina Entwistle, Water and Wastewater Treatment Operator, North East Water Authority
OPERATORS PERSPECTIVE : OPTIMISATION OF A NEW PACKAGE WATER TREATMENT PLANT Paper Presented by : Melina Entwistle Author: Melina Entwistle, Water and Wastewater Treatment Operator, North East Water Authority
Periodic Properties: Analysis of Group I, II and Transition Metals
 Periodic Properties: Analysis of Group I, II and Transition Metals Procedure All solutions should be mixed thoroughly after any additions. Clearly record all observations (indicating what you did), even
Periodic Properties: Analysis of Group I, II and Transition Metals Procedure All solutions should be mixed thoroughly after any additions. Clearly record all observations (indicating what you did), even
Corrosion Control and Cathodic Protection Data Sheet
 Data Sheet CORROSION CONTROL Corrosion control is the application of engineering principles and procedures to minimise corrosion to an acceptable level by the most economical method. It is rarely practical
Data Sheet CORROSION CONTROL Corrosion control is the application of engineering principles and procedures to minimise corrosion to an acceptable level by the most economical method. It is rarely practical
Mon. Tues. Wed. Thurs. Fri. AM or PM B
 Name: (cf. Honesty Declaration Statement on page 20) Laboratory Day (circle) Lab Room Locker Lab. Session (circle) Lab. Section Mon. Tues. Wed. Thurs. Fri. AM or PM B Date experiment is performed MARK:
Name: (cf. Honesty Declaration Statement on page 20) Laboratory Day (circle) Lab Room Locker Lab. Session (circle) Lab. Section Mon. Tues. Wed. Thurs. Fri. AM or PM B Date experiment is performed MARK:
Part A (Level 1) A Matching (4 marks, 1 mark each) B Multiple-choice questions (20 marks, 2 marks each) Name: ( ) Time and Marks Class: Date:
 Unit 5 S1 Science Test The wonderful solvent - water Name: ( ) Time and Marks Class: Date: Part A: 35 min / 100 marks Parts A & B: 45 min / 120 marks Note: 1 Attempt ALL questions. 2 Write your answers
Unit 5 S1 Science Test The wonderful solvent - water Name: ( ) Time and Marks Class: Date: Part A: 35 min / 100 marks Parts A & B: 45 min / 120 marks Note: 1 Attempt ALL questions. 2 Write your answers
TITANIUM DIOXIDE. SYNONYMS Titania; CI Pigment white 6; CI (1975) No ; INS No. 171 DEFINITION DESCRIPTION FUNCTIONAL USES CHARACTERISTICS
 TITANIUM DIOXIDE Prepared at the 71 st JECFA (2009) and published in FAO JECFA Monographs 7 (2009), superseding specifications prepared at the 67 th JECFA (2006) and published in FAO JECFA Monographs 3
TITANIUM DIOXIDE Prepared at the 71 st JECFA (2009) and published in FAO JECFA Monographs 7 (2009), superseding specifications prepared at the 67 th JECFA (2006) and published in FAO JECFA Monographs 3
Grade 7: Mixtures and Solutions Lesson: Potash Solution Mining Dissolving Potash
 Grade 7: Mixtures and Solutions Lesson: Potash Solution Mining Dissolving Potash Overview Students will discover through guided inquiry and hands on activities that one of Saskatchewan s valuable mineral
Grade 7: Mixtures and Solutions Lesson: Potash Solution Mining Dissolving Potash Overview Students will discover through guided inquiry and hands on activities that one of Saskatchewan s valuable mineral
Significance of Water Observation Lab
 Significance of Water Observation Lab It has been said that the chemistry of life is water chemistry. Because of its chemical properties, water is the medium in which most of life s chemical reactions
Significance of Water Observation Lab It has been said that the chemistry of life is water chemistry. Because of its chemical properties, water is the medium in which most of life s chemical reactions
Basic Survival Chemistry
 Basic Survival Chemistry Francis Glode hclo3.tk Notation: The numbers in parentheses indicate the reaction temperature in Celsius. RT indicates that the reaction proceeds at room temperature.
Basic Survival Chemistry Francis Glode hclo3.tk Notation: The numbers in parentheses indicate the reaction temperature in Celsius. RT indicates that the reaction proceeds at room temperature.
NCEA Level 1 Chemistry (90933) 2012 page 1 of 5. Q Evidence Achievement Achievement with Merit Achievement with Excellence NØ N1 N2 A3 A4 M5 M6 E7 E8
 Assessment Schedule 2012 NCEA Level 1 Chemistry (90933) 2012 page 1 of 5 Chemistry: Demonstrate understanding of aspects of selected elements (90933) Evidence Statement Q Evidence with Merit with Excellence
Assessment Schedule 2012 NCEA Level 1 Chemistry (90933) 2012 page 1 of 5 Chemistry: Demonstrate understanding of aspects of selected elements (90933) Evidence Statement Q Evidence with Merit with Excellence
Separating Mixtures WORKSHEETS
 Attention Teachers KEEP IT SIMPLE SCIENCE Separating Year 7 Chemical Sciences WORKSHEETS KISS topic number Topic 03.7C KISS Worksheets are designed to consolidate students knowledge & understanding and/or
Attention Teachers KEEP IT SIMPLE SCIENCE Separating Year 7 Chemical Sciences WORKSHEETS KISS topic number Topic 03.7C KISS Worksheets are designed to consolidate students knowledge & understanding and/or
Metals and Non-metals
 CHAPTER 3 Metals and Non-metals In Class IX you have learnt about various elements. You have seen that elements can be classified as metals or non-metals on the basis of their properties. Think of some
CHAPTER 3 Metals and Non-metals In Class IX you have learnt about various elements. You have seen that elements can be classified as metals or non-metals on the basis of their properties. Think of some
Standard Test Procedures Manual
 STP 205-10 Standard Test Procedures Manual Section: 1. SCOPE 1.1. Description of Test This method describes the quantitative determination of the distribution of particle sizes in soils. The distribution
STP 205-10 Standard Test Procedures Manual Section: 1. SCOPE 1.1. Description of Test This method describes the quantitative determination of the distribution of particle sizes in soils. The distribution
Metals And Their Properties- Physical and Chemical
 Metals And Their Properties- Physical and Chemical All the things around us are made of 100 or so elements. These elements were classified by Lavoisier in to metals and non-metals by studying their properties.
Metals And Their Properties- Physical and Chemical All the things around us are made of 100 or so elements. These elements were classified by Lavoisier in to metals and non-metals by studying their properties.
PVA polymer slime. PVA polymer slime
 PVA polymer slime PVA polymer slime A solution of polyvinyl alcohol (PVA) can be made into a slime by adding borax solution, which creates crosslinks between polymer chains. In this activity, some interesting
PVA polymer slime PVA polymer slime A solution of polyvinyl alcohol (PVA) can be made into a slime by adding borax solution, which creates crosslinks between polymer chains. In this activity, some interesting
[ Cl ] - [[Mg 2+ ] ] Experiment 7: Oxidation-Reduction Reactions. transfer e -
![[ Cl ] - [[Mg 2+ ] ] Experiment 7: Oxidation-Reduction Reactions. transfer e - [ Cl ] - [[Mg 2+ ] ] Experiment 7: Oxidation-Reduction Reactions. transfer e -](/thumbs/74/70175814.jpg) Experiment 7: OxidationReduction Reactions PURPOSE Become familiar with the concepts of oxidation and reduction and how these reactions occur. Carry out several such reactions and learn to recognize when
Experiment 7: OxidationReduction Reactions PURPOSE Become familiar with the concepts of oxidation and reduction and how these reactions occur. Carry out several such reactions and learn to recognize when
Chapter 8. Gravimetric Analysis
 Chapter 8 Gravimetric Analysis Gravimetric analysis is the use of weighing to determine the amount of a component in your sample. Gravimetric analysis, or gravimetry is normally performed either as a :
Chapter 8 Gravimetric Analysis Gravimetric analysis is the use of weighing to determine the amount of a component in your sample. Gravimetric analysis, or gravimetry is normally performed either as a :
What Is The Chemical Formula For Copper 2 Sulfate
 What Is The Chemical Formula For Copper 2 Sulfate Free Download WHAT IS THE CHEMICAL FORMULA FOR COPPER 2 SULFATE WHAT IS THE FORMULA FOR COPPER(II) SULFATE? REFERENCE Sun, 17 Dec 2017 13:50:00 GMT the
What Is The Chemical Formula For Copper 2 Sulfate Free Download WHAT IS THE CHEMICAL FORMULA FOR COPPER 2 SULFATE WHAT IS THE FORMULA FOR COPPER(II) SULFATE? REFERENCE Sun, 17 Dec 2017 13:50:00 GMT the
SCIENCE REVISION CARDS For KS3 and Common Entrance
 SCIENCE REVISION CARDS For KS3 and Common Entrance I recommend printing the following pages onto card and cutting them into 4 equal pieces. They will then fit into a file box for revision purposes. Many
SCIENCE REVISION CARDS For KS3 and Common Entrance I recommend printing the following pages onto card and cutting them into 4 equal pieces. They will then fit into a file box for revision purposes. Many
EXTRA CREDIT - EXPERIMENT G ELECTROCHEMISTRY ACTIVITY OF METALS
 EXTRA CREDIT - EXPERIMENT G ELECTROCHEMISTRY ACTIVITY OF METALS INTRODUCTION The objective of this experiment is to develop an abbreviated activity series of metals using: 1. Displacement reactions 2.
EXTRA CREDIT - EXPERIMENT G ELECTROCHEMISTRY ACTIVITY OF METALS INTRODUCTION The objective of this experiment is to develop an abbreviated activity series of metals using: 1. Displacement reactions 2.
REPORT NUMBER REPORT DATE SEND TO ISSUE DATE Apr 18, Apr 18, 2017 RECEIVED DATE Apr 05, 2017
 PAGE 1/7 Sample ID: EWA PELLETS Lab Number: 2653281 Date Sampled: 2017-04-04 0850 Carbon nitrogen ratio C/N 6 : 1 0.1 Calculation Auto-2017/04/12 Auto-2017/04/18 Carbon (total) 38.53 % 0.050 ASTM D 5373
PAGE 1/7 Sample ID: EWA PELLETS Lab Number: 2653281 Date Sampled: 2017-04-04 0850 Carbon nitrogen ratio C/N 6 : 1 0.1 Calculation Auto-2017/04/12 Auto-2017/04/18 Carbon (total) 38.53 % 0.050 ASTM D 5373
Nickel Electroplating
 Nickel Electroplating In a galvanic or voltaic electrochemical cell, the spontaneous reaction occurs and electrons flow from the anode (oxidation) to the cathode (reduction). In an electrolytic cell, a
Nickel Electroplating In a galvanic or voltaic electrochemical cell, the spontaneous reaction occurs and electrons flow from the anode (oxidation) to the cathode (reduction). In an electrolytic cell, a
How Salty Is Our Water?
 Title: How Salty Is Our Water? (Water Chemistry) Grade(s): 6-7 Introduction: Corals require a relatively constant environment. However, due to the proximity to the surface of the ocean, the salinity of
Title: How Salty Is Our Water? (Water Chemistry) Grade(s): 6-7 Introduction: Corals require a relatively constant environment. However, due to the proximity to the surface of the ocean, the salinity of
2. Crystallization. A. Background
 2. Crystallization A. Background Crystallization is one of several available techniques available to purify organic compounds. Unlike other techniques, however, crystallization is specific to the purification
2. Crystallization A. Background Crystallization is one of several available techniques available to purify organic compounds. Unlike other techniques, however, crystallization is specific to the purification
The empirical formula of a compound
 The empirical formula of a compound Reference: Chapter 1, Section 1.2, pages 21 24 Please note Aim A full risk assessment should be carried out prior to commencing this experiment. Personal safety equipment
The empirical formula of a compound Reference: Chapter 1, Section 1.2, pages 21 24 Please note Aim A full risk assessment should be carried out prior to commencing this experiment. Personal safety equipment
Experiment 3 * Thermochromism in the Ionic Conductor, Cu 2 HgI 4
 Experiment 3 * Thermochromism in the Ionic Conductor, Cu 2 HgI 4 *This lab taken from Teaching General Chemistry; A Materials Science Companion, Eds. Ellis, A.B.; Geselbracht, M.J.; Johnson, B.J.; Lisensky,
Experiment 3 * Thermochromism in the Ionic Conductor, Cu 2 HgI 4 *This lab taken from Teaching General Chemistry; A Materials Science Companion, Eds. Ellis, A.B.; Geselbracht, M.J.; Johnson, B.J.; Lisensky,
PHASE CHANGES. Time Temperature Observations. Name(s)
 3 5 PHASE CHANGES PHASE CHANGES Name(s) The activities presented here focus on the energy changes that occur in substances undergoing a phase change. The first activity will take the most time to complete.
3 5 PHASE CHANGES PHASE CHANGES Name(s) The activities presented here focus on the energy changes that occur in substances undergoing a phase change. The first activity will take the most time to complete.
to the presentation Teaching Thermodynamics: Chemical Potential from the Beginning Regina Rüffler, Georg Job
 to the presentation Teaching Thermodynamics: Chemical Potential from the Beginning Regina Rüffler, Georg Job Thermo International 2006 Boulder, August 3, 2006 FOUNDATION Further informations on the homepage:
to the presentation Teaching Thermodynamics: Chemical Potential from the Beginning Regina Rüffler, Georg Job Thermo International 2006 Boulder, August 3, 2006 FOUNDATION Further informations on the homepage:
Activity 2.1. Activity 2.2. Looking at animal cells. Looking at plant cells
 Activity 2.1 Looking at animal cells Skills C1, C2 a source of animal cells, for example some macerated liver or scrapings from the lining of the trachea from a set of sheep or other lungs (obtainable
Activity 2.1 Looking at animal cells Skills C1, C2 a source of animal cells, for example some macerated liver or scrapings from the lining of the trachea from a set of sheep or other lungs (obtainable
Method 6.1 Boiling house products: Brix, pol and purity
 Section 6: Boiling house products p 1/6 Method 6.1 Boiling house products: Brix, pol and purity 1. Rationale This method is applicable to A-, B- and C- (or final) molasses, A-, B- and C-nutsch, A-, B-
Section 6: Boiling house products p 1/6 Method 6.1 Boiling house products: Brix, pol and purity 1. Rationale This method is applicable to A-, B- and C- (or final) molasses, A-, B- and C-nutsch, A-, B-
Experiment. Molar Mass of an Unknown Sulfate Salt by Gravimetric Techniques 1
 Experiment. Molar Mass of an Unknown Sulfate Salt by Gravimetric Techniques 1 This lab is to reacquaint you with some basic laboratory techniques and serves as a warm-up to the experiments in this course.
Experiment. Molar Mass of an Unknown Sulfate Salt by Gravimetric Techniques 1 This lab is to reacquaint you with some basic laboratory techniques and serves as a warm-up to the experiments in this course.
Lab 1 Basics of Energy Production I
 1 Lab 1 Basics of Energy Production I Relevant SOLs: PS.1a, PS.1b, PS.1d, PS.1k, PS.1m, PS.2e, PS.2f, PS.5a, PS.5c, PS.6a, PS.6b, PS.6c, PS.7a, PS.7b, PS.9c, 3.1h, 3.1j, 3.11a, 3.11b, 4.3d, 5.3a, 5.4c,
1 Lab 1 Basics of Energy Production I Relevant SOLs: PS.1a, PS.1b, PS.1d, PS.1k, PS.1m, PS.2e, PS.2f, PS.5a, PS.5c, PS.6a, PS.6b, PS.6c, PS.7a, PS.7b, PS.9c, 3.1h, 3.1j, 3.11a, 3.11b, 4.3d, 5.3a, 5.4c,
Make Five. A game about the chemical formulas of some common minerals
 Make Five A game about the chemical formulas of some common minerals By defi nition, a mineral has a defi nite chemical composition. It s sort of like a recipe. This game is for students who want to (or
Make Five A game about the chemical formulas of some common minerals By defi nition, a mineral has a defi nite chemical composition. It s sort of like a recipe. This game is for students who want to (or
yk4lm (1:26) vd_90 (4:37)
 Properties of Water Video Notes http://www.youtube.com/watch?v=2vfld5 yk4lm (1:26) http://www.youtube.com/watch?v=ivu4nl vd_90 (4:37) https://www.youtube.com/watch?v=0ens nj4zfz8#t=4.4600086 (4:58) new
Properties of Water Video Notes http://www.youtube.com/watch?v=2vfld5 yk4lm (1:26) http://www.youtube.com/watch?v=ivu4nl vd_90 (4:37) https://www.youtube.com/watch?v=0ens nj4zfz8#t=4.4600086 (4:58) new
Significance of Water Observation Lab
 Significance of Water Observation Lab It has been said that the chemistry of life is water chemistry. Because of its chemical properties, water is the medium in which most of life s chemical reactions
Significance of Water Observation Lab It has been said that the chemistry of life is water chemistry. Because of its chemical properties, water is the medium in which most of life s chemical reactions
YEAR 7 SCIENCE EXAMINATION. Semester MULTIPLE CHOICE QUESTION BOOK 1 MATERIAL REQUIRED / RECOMMENDED FOR THIS PAPER:
 YEAR 7 SCIENCE EXAMINATION Semester 2 2016 MULTIPLE CHOICE QUESTION BOOK 1 STUDENT NAME: TEACHER: DATE: Time allowed for this exam: (Book 1 and Book 2 Combined) Reading time before commencing work: Working
YEAR 7 SCIENCE EXAMINATION Semester 2 2016 MULTIPLE CHOICE QUESTION BOOK 1 STUDENT NAME: TEACHER: DATE: Time allowed for this exam: (Book 1 and Book 2 Combined) Reading time before commencing work: Working
The oxides of nitrogen and the details of the oxygen states of nitrogen and the N:O ratio can be presented in a tabular form as:
 Oxides of Nitrogen Introduction to oxides of nitrogen Nirogen has a position in second period of group V in the modern periodic table. It has molecular formula N2. It has atomic number 7 and atomic weight
Oxides of Nitrogen Introduction to oxides of nitrogen Nirogen has a position in second period of group V in the modern periodic table. It has molecular formula N2. It has atomic number 7 and atomic weight
METHOD #: Approved for NPDES (Issued 1971) Oxygen, Dissolved (Modified Winkler, Full-Bottle Technique) ANALYTE: CAS # O Oxygen
 METHOD #: 360.2 Approved for NPDES (Issued 1971) TITLE: Oxygen, Dissolved (Modified Winkler, Full-Bottle Technique) ANALYTE: CAS # O Oxygen 7782-44-7 INSTRUMENTATION: Titration, Probe STORET No. 00300
METHOD #: 360.2 Approved for NPDES (Issued 1971) TITLE: Oxygen, Dissolved (Modified Winkler, Full-Bottle Technique) ANALYTE: CAS # O Oxygen 7782-44-7 INSTRUMENTATION: Titration, Probe STORET No. 00300
Preparation of Cyclohexene From Cyclohexanol
 EXPERIMENT 9 Alkene Synthesis From Alcohol Preparation of Cyclohexene From Cyclohexanol Purpose: a) Preparation of an alkene by dehydration (elimination of water) of an alcohol in the presence of an acid
EXPERIMENT 9 Alkene Synthesis From Alcohol Preparation of Cyclohexene From Cyclohexanol Purpose: a) Preparation of an alkene by dehydration (elimination of water) of an alcohol in the presence of an acid
Name Lab Section Date. Sediment Lab
 Name Lab Section Date. Investigating Stokes Law Sediment Lab ds = density of solid, g/cm dw = density of water, g/cm g = gravity, 980 cm/second 2 D = particle diameter in centimeters μ = molecular viscosity,
Name Lab Section Date. Investigating Stokes Law Sediment Lab ds = density of solid, g/cm dw = density of water, g/cm g = gravity, 980 cm/second 2 D = particle diameter in centimeters μ = molecular viscosity,
Experiment E: Martensitic Transformations
 Experiment E: Martensitic Transformations Introduction: The purpose of this experiment is to introduce students to a family of phase transformations which occur by shear rather than diffusion. In metals,
Experiment E: Martensitic Transformations Introduction: The purpose of this experiment is to introduce students to a family of phase transformations which occur by shear rather than diffusion. In metals,
EDICT ± OF GOVERNMENT
 EDICT ± OF GOVERNMENT Inordertopromotepubliceducationandpublicsafety,equal justiceforal,abeterinformedcitizenry,theruleoflaw,world tradeandworldpeace,thislegaldocumentisherebymade availableonanoncommercialbasis,asitistherightofal
EDICT ± OF GOVERNMENT Inordertopromotepubliceducationandpublicsafety,equal justiceforal,abeterinformedcitizenry,theruleoflaw,world tradeandworldpeace,thislegaldocumentisherebymade availableonanoncommercialbasis,asitistherightofal
Float standard programme
 Float standard programme A/M/K Dimension (mm):ø40 x 27 POM 18 1 20 0.9 22.5 0.8 26 0.7 T/C/I Dimension (mm): Ø30 x 44 27.5 1 30.5 0.9 34.5 0.8 39.5 0.7 R/S/U Dimension (mm):ø30 x 44 NBR 19.5 1 22 0.9 24.5
Float standard programme A/M/K Dimension (mm):ø40 x 27 POM 18 1 20 0.9 22.5 0.8 26 0.7 T/C/I Dimension (mm): Ø30 x 44 27.5 1 30.5 0.9 34.5 0.8 39.5 0.7 R/S/U Dimension (mm):ø30 x 44 NBR 19.5 1 22 0.9 24.5
Recrystallization with a Single Solvent
 Experiment: Recrystallization Part II: Purification of Solids In Part I of the recrystallization experiment, you learned about the factors which make a good recrystallization solvent, and you learned how
Experiment: Recrystallization Part II: Purification of Solids In Part I of the recrystallization experiment, you learned about the factors which make a good recrystallization solvent, and you learned how
*20GSD5201* Double Award Science: Chemistry. Unit C2 Higher Tier TUESDAY 9 JUNE 2015, AFTERNOON [GSD52] *GSD52* *G5802* TIME 1 hour 15 minutes.
![*20GSD5201* Double Award Science: Chemistry. Unit C2 Higher Tier TUESDAY 9 JUNE 2015, AFTERNOON [GSD52] *GSD52* *G5802* TIME 1 hour 15 minutes. *20GSD5201* Double Award Science: Chemistry. Unit C2 Higher Tier TUESDAY 9 JUNE 2015, AFTERNOON [GSD52] *GSD52* *G5802* TIME 1 hour 15 minutes.](/thumbs/76/73786999.jpg) Centre Number Candidate Number General Certificate of Secondary Education 2015 Double Award Science: Chemistry Unit C2 Higher Tier [GSD52] *GSD52* *G5802* *GSD52* TUESDAY 9 JUNE 2015, AFTERNOON TIME 1
Centre Number Candidate Number General Certificate of Secondary Education 2015 Double Award Science: Chemistry Unit C2 Higher Tier [GSD52] *GSD52* *G5802* *GSD52* TUESDAY 9 JUNE 2015, AFTERNOON TIME 1
The Sense of Smell - Practical work
 CONTENTS Extraction of Natural Perfume Ingredients Activity 1 Limonene from citrus fruits Part A: Expression Part B: Steam distillation Part C: Solvent extraction Part D: Determination of yield Part E:
CONTENTS Extraction of Natural Perfume Ingredients Activity 1 Limonene from citrus fruits Part A: Expression Part B: Steam distillation Part C: Solvent extraction Part D: Determination of yield Part E:
COMPOSITION OF SEAWATER
 COMPOSITION OF SEAWATER Ocean water is a combination of freshwater and a variety of dissolved substances. Salinity is a measure of the amount of dissolved salts in seawater, measured in parts per thousand
COMPOSITION OF SEAWATER Ocean water is a combination of freshwater and a variety of dissolved substances. Salinity is a measure of the amount of dissolved salts in seawater, measured in parts per thousand
Method 5.1 Syrup and remelt: Brix, pol and purity
 Section 5: Syrup and remelt p 1/5 Method 5.1 Syrup and remelt: Brix, pol and purity 1. Rationale This method is applicable to factory syrups and remelt and may be used to obtain data for factory control
Section 5: Syrup and remelt p 1/5 Method 5.1 Syrup and remelt: Brix, pol and purity 1. Rationale This method is applicable to factory syrups and remelt and may be used to obtain data for factory control
Electricity and Chemistry
 Electricity and Chemistry Electrochemistry: It is a branch of chemistry that deals with the reactions involving the conversion of chemical energy into electrical energy and vice-versa. Electrochemical
Electricity and Chemistry Electrochemistry: It is a branch of chemistry that deals with the reactions involving the conversion of chemical energy into electrical energy and vice-versa. Electrochemical
TWEED RIVER HIGH SCHOOL 2006 PRELIMINARY CHEMISTRY. Unit 2 Metals
 TWEED RIVER HIGH SCHOOL 2006 PRELIMINARY CHEMISTRY Unit 2 Metals Part 2 Metals differ in their reactivity with other chemicals and this influences their uses. Describe observable changes when metals react
TWEED RIVER HIGH SCHOOL 2006 PRELIMINARY CHEMISTRY Unit 2 Metals Part 2 Metals differ in their reactivity with other chemicals and this influences their uses. Describe observable changes when metals react
Raw Water Activities
 Name: Raw Water Activities Directions: Record your observations during each activity in the boxes below. Draw a picture of your test if you d like. Answer the discussion questions below each notes box.
Name: Raw Water Activities Directions: Record your observations during each activity in the boxes below. Draw a picture of your test if you d like. Answer the discussion questions below each notes box.
ADVANCED AP PLACEMENT CHEMISTRY. Activity Series. Introduction. Objective. Chemicals and Equipment
 ADVANCED AP PLACEMENT CHEMISTRY Introduction Activity Series An activity series of metals is a table of metals arranged in the order of their decreasing chemical activity or the ease at which the metal
ADVANCED AP PLACEMENT CHEMISTRY Introduction Activity Series An activity series of metals is a table of metals arranged in the order of their decreasing chemical activity or the ease at which the metal
