Corresponding Physical or Chemical Property bread rising through the use of baking soda carbonates decompose to form CO 2
|
|
|
- Eustace Matthews
- 6 years ago
- Views:
Transcription
1 Substance = a chemical (element or mixture) Substance mixture chemical process = chemical reaction (new substances result) physical process = same substances present through out Physical or chemical processes? Process Corresponding Physical or Chemical Property bread rising through the use of baking soda carbonates decompose to form CO 2 boiling water apples rotting biomolecules such as carbohydrates can be digested (often oxidized) by microorganisms burning coal carbon burns in oxygen freezing liquid freezing point distilling a liquid boiling/condensation point evaporating a liquid vapor pressure exploding dynamite undergoes a disproportionation reaction decomposes titration of an acid with a base acid-base neutralization filtration of a solid from a liquid particle size (molecules versus small macroscopic particles) condensation of water formation of polyethylene from ethylene baking of a potato nitroglycerin exploding high pressure gas cylinder exploding iron rusting crushing of stone tarnishing of silver melting lead Popcorn popping flint and steel producing sparks smoke scattering light water condensing on metal metal getting hot addition across double bond thermal decomposition of biomolecules denaturation of proteins vaporization of water undergoes a disproportionation reaction decomposes tendency of gas to expand reactivity of iron with oxygen hardness, lack of malleability reactivity of silver with oxygen melting point
2 Physical vs. chemical property? Property Physical or chemical? Intensive or Extensive? color density reaction with oxygen tendency of metal to corrode solubility of sugar in water density soluble (dissolves) in water metal can tarnish or corrode mass melting point Intensive property Does not depend on size of sample (property of material making up sample) Same of all samples Can be used to identify the sample (qualitative analysis = what is it?) Extensive property Depends on size of sample property of specific sample being studied Varies with sample size Can be used to determine how much is present (Qualitative Analysis how much is there? or How concentrated is it?)
3 Sample Substance vs. Mixture Homogenous vs. Heterogenous gasohol (gasoline with alcohol added) gasoline solid butter snickers bar grape juice apple juice apple cider orange juice tomato juice uncooked hamburger cooked hamburger freshly brewed coffee vinegar slice of white bread whipped cream wedding ring sweat Nile River Water Concho River Water human blood air compressed air in a scuba tank table salt sand copper nitrogen gas argon gas carbon dioxide gas mineral water brass (a Zn/Cu alloy) sucrose (table sugar) HCl O 2 O 3 beer I 2 Ce ClF S 8 filtered water chicken noodle soup element vs. compound
4 trail mix snack fruit salad cool aid drink (already mixed) cool aid mix (in packet) sand and water alcohol dissolved in water damp air clouds Properties of a sample of 24 carat gold (classify them) a g b. doe not tarnish c. density = 19 g/ml d. insoluble in water e. has a yellowish color f. is shiny and reflects light (has luster) g. is malleable (can be deformed without breaking) Properties of a sample of rose gold (18 carat) (classify them) an alloy a g b. doe not tarnish c. density = 12.7 g/ml d. insoluble in water e. has a pink color f. is shiny and reflects light (has luster) g. is malleable (can be deformed without breaking) h. 75% gold, 21% copper and 4% silver Properties of sodium (Na) a. density is greater than kerosene and less than water b. lower melting point than most metals c. excellent conductor of heat and electricity d. soft (can be cut with a knife) e. freshly cut sodium is shiny, but rapidly tarnishes in air f. it reacts vigorously with water to form H 2 and NaOH Properties of H 2 a. At room temperature, less dense than any other gas
5 b. reacts vigorously with water c. Liquefied below -253 o C d. Gas does not conduct electricity Properties of formaldehyde (CH 2 O) a. characteristic acrid smell b. soluble in water c. burns in air d. gas at room temperature e. colorless Properties of silver a. tarnishes over time b. tarnished silver can be polished to a shiny metallic shine (luster) c. reacts with chlorine to produce a white solid d. It sinks in water Give three properties that you can use to distinguish between... a. salt, copper, sand b. sugar, water, oxygen
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
1. Elemental mercury is a shiny, silver-colored, dense liquid that flows easily. Are these characteristics of mercury physical or chemical properties?
 Chemistry Dr. Saulmon 2014-15 School Year Unit 2: Basics of Chemistry Problem Set 3 Wednesday, September 3, 2014 1. Elemental mercury is a shiny, silver-colored, dense liquid that flows easily. Are these
Chemistry Dr. Saulmon 2014-15 School Year Unit 2: Basics of Chemistry Problem Set 3 Wednesday, September 3, 2014 1. Elemental mercury is a shiny, silver-colored, dense liquid that flows easily. Are these
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 3 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 3 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
Iron filings (Fe) 56g IRON + SULPHUR IRON SULPHIDE
 W.S.51. Chemical reactions. All of the different materials around us have been formed by chemical reactions from about one hundred simple elements. The diagram below shows a chemical reaction between the
W.S.51. Chemical reactions. All of the different materials around us have been formed by chemical reactions from about one hundred simple elements. The diagram below shows a chemical reaction between the
Metal and Non Metals
 Metal and Non Metals Malleable Ductile Sonorous Conductor Insulator KEYWORDS Rusting Brass Calcium Chloride Galvanising Reactivity Metals on the Periodic table Metals Non Metals Metals and their properties
Metal and Non Metals Malleable Ductile Sonorous Conductor Insulator KEYWORDS Rusting Brass Calcium Chloride Galvanising Reactivity Metals on the Periodic table Metals Non Metals Metals and their properties
Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet
 Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet Name: Sec: Date: Q1.Choose the best answer. 1. Which of the following is an element? a) steam b) sugar c)dry ice d) sulphur 2. Which
Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet Name: Sec: Date: Q1.Choose the best answer. 1. Which of the following is an element? a) steam b) sugar c)dry ice d) sulphur 2. Which
Draw a ring around the correct word in the box to complete the sentence.
 Q. Iron is the main structural metal used in the world. (a) The diagram represents the particles in iron, Fe. Draw a ring around the correct word in the box to complete the sentence. Iron is described
Q. Iron is the main structural metal used in the world. (a) The diagram represents the particles in iron, Fe. Draw a ring around the correct word in the box to complete the sentence. Iron is described
SCIENCE REVISION CARDS For KS3 and Common Entrance
 SCIENCE REVISION CARDS For KS3 and Common Entrance I recommend printing the following pages onto card and cutting them into 4 equal pieces. They will then fit into a file box for revision purposes. Many
SCIENCE REVISION CARDS For KS3 and Common Entrance I recommend printing the following pages onto card and cutting them into 4 equal pieces. They will then fit into a file box for revision purposes. Many
METALS AND THEIR COMPOUNDS
 METALS AND THEIR COMPOUNDS Metals are elements whose atoms ionize by electron loss, while non-metals are elements whose atoms ionize by electron gain. Metals are in groups 1, 2 and 3 of the periodic table.
METALS AND THEIR COMPOUNDS Metals are elements whose atoms ionize by electron loss, while non-metals are elements whose atoms ionize by electron gain. Metals are in groups 1, 2 and 3 of the periodic table.
(A) Copper. (B) Zinc. (C) Iron. (D) Wood. (A) Zinc. (B) Wood. (C) Rubber. (D) Plastic.
 Downloaded from MATERIALS : METALS AND NON-METALS 1.Give one example of each: metals and non-metals. 2.Name the metal, which is the best conductor of heat and electricity. 3.Name the property by which
Downloaded from MATERIALS : METALS AND NON-METALS 1.Give one example of each: metals and non-metals. 2.Name the metal, which is the best conductor of heat and electricity. 3.Name the property by which
Properties A Metal B Non- metal Electronic configuration?? Nature of oxides?? Oxidizing or reducing action?? Conduction of heat and electricity??
 CLASS: X NCERT (CBSE) SCIENCE: Chemistry Page: 1 Question 1: Compare the properties of a typical metal and a non-metal on the basis of the following. Fill in Column A, B. Properties A Metal B Non- metal
CLASS: X NCERT (CBSE) SCIENCE: Chemistry Page: 1 Question 1: Compare the properties of a typical metal and a non-metal on the basis of the following. Fill in Column A, B. Properties A Metal B Non- metal
SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example,
 SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example, density, thermal or electrical conductivity, solubility,
SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example, density, thermal or electrical conductivity, solubility,
Covered with a thin layer of oxide at ordinary temperatures.
 1 More about Metals Physical properties of metals In general metals have luster, are malleable and ductile, good conductors of heat and electricity and have high boiling and melting points and nonmetals
1 More about Metals Physical properties of metals In general metals have luster, are malleable and ductile, good conductors of heat and electricity and have high boiling and melting points and nonmetals
Metals And Their Properties- Physical and Chemical
 Metals And Their Properties- Physical and Chemical All the things around us are made of 100 or so elements. These elements were classified by Lavoisier in to metals and non-metals by studying their properties.
Metals And Their Properties- Physical and Chemical All the things around us are made of 100 or so elements. These elements were classified by Lavoisier in to metals and non-metals by studying their properties.
85 Q.51 Which of the following carbonates would give the metal when heated with carbon? (1) MgCO 3 (2) PbCO 3 (3) K 2 CO 3 (4) CuCO 3
 Metal and metal reactivity / Section 2 / Sect2pp.doc / S. W. Tse / P.1 85 Q.51 Which of the following carbonates would give the metal when heated with carbon? (1) MgCO 3 (2) PbCO 3 (3) K 2 CO 3 (4) CuCO
Metal and metal reactivity / Section 2 / Sect2pp.doc / S. W. Tse / P.1 85 Q.51 Which of the following carbonates would give the metal when heated with carbon? (1) MgCO 3 (2) PbCO 3 (3) K 2 CO 3 (4) CuCO
(3) The compound boron nitride (BN) has a high melting point (2967 ºC), high density, and is very hard. What is the best classification of this solid?
 Solids and Liquids Name: Period: (1) Identify the type of solid formed by each compound. (a) Ag (b) CO 2 (c) SiO 2 (d) wax (e) MgCl 2 (f) Fe (g) graphite (h) SO 2 (i) CaCO 3 (j) I 2 (k) rubber (l) SiC
Solids and Liquids Name: Period: (1) Identify the type of solid formed by each compound. (a) Ag (b) CO 2 (c) SiO 2 (d) wax (e) MgCl 2 (f) Fe (g) graphite (h) SO 2 (i) CaCO 3 (j) I 2 (k) rubber (l) SiC
2.3 Chemical Changes corrosion Kinds of Corrosion
 2.3 Chemical Changes Have you ever wondered why metal car bodies rust but plastic bumpers do not? As you know, different substances have different physical properties, such as colour and hardness, and
2.3 Chemical Changes Have you ever wondered why metal car bodies rust but plastic bumpers do not? As you know, different substances have different physical properties, such as colour and hardness, and
OC30 Conduct a qualitative experiment to detect the presence of dissolved solids in water samples, and test water for hardness (soap test)
 Chemistry: 6. Water Please remember to photocopy 4 pages onto one sheet by going A3 A4 and using back to back on the photocopier Syllabus OC14 Use cobalt chloride paper or anhydrous copper sulfate to test
Chemistry: 6. Water Please remember to photocopy 4 pages onto one sheet by going A3 A4 and using back to back on the photocopier Syllabus OC14 Use cobalt chloride paper or anhydrous copper sulfate to test
CHAPTER 6. Natural Mineral Mineral Content Elements In The Minerals Bauxite Aluminium oxide Aluminium, oxygen. Cassiterite Tin oxide Tin, oxygen
 CHAPTER 6 6.1 Minerals Found In The Earth s Crust mineral : is a naturally occurring solid element or compound with a definite crystalline structure and chemical composition. natural elements : gold, silver
CHAPTER 6 6.1 Minerals Found In The Earth s Crust mineral : is a naturally occurring solid element or compound with a definite crystalline structure and chemical composition. natural elements : gold, silver
CHAPTER 9. The Uses of Sulphuric Acid
 CHAPTER 9 MANUFACTURED SUBSTANCES IN INDUSTRY SULPHURIC ACID The Uses of Sulphuric Acid 175 POLLUTION of Sulphur DIOXIDE Formation of Acid Rain Burning of Sulphur Burning of Sulphur Dioxide 176 Effect
CHAPTER 9 MANUFACTURED SUBSTANCES IN INDUSTRY SULPHURIC ACID The Uses of Sulphuric Acid 175 POLLUTION of Sulphur DIOXIDE Formation of Acid Rain Burning of Sulphur Burning of Sulphur Dioxide 176 Effect
An Organized Table Worksheet Due Thursday Name: Date: Period:
 An Organized Table Worksheet Due Thursday Name: Date: Period: The Periodic Table of Elements In 1871, the first periodic table was developed by Dmitrii Mendeleev. Mendeleev is known as the father of the
An Organized Table Worksheet Due Thursday Name: Date: Period: The Periodic Table of Elements In 1871, the first periodic table was developed by Dmitrii Mendeleev. Mendeleev is known as the father of the
Skills in Science. Lab equipment. (Always draw 2D) Drawings below are NOT to scale. Beaker - A general purpose container with a pouring lip.
 Skills in Science Safety: Do NOT enter or leave the lab without permission from a teacher. Keep the gaps between tables clear of stools and bags. Never run in the lab. Do not throw things around in the
Skills in Science Safety: Do NOT enter or leave the lab without permission from a teacher. Keep the gaps between tables clear of stools and bags. Never run in the lab. Do not throw things around in the
EXPERIMENT 3: Identification of a Substance by Physical Properties
 EXPERIMENT 3: Identification of a Substance by Physical Properties Materials: Hot plate Digital balance Capillary tubes (3) Thermometer Beakers (250 ml) Watch glass Graduated Cylinder (10 ml) Mel-Temp
EXPERIMENT 3: Identification of a Substance by Physical Properties Materials: Hot plate Digital balance Capillary tubes (3) Thermometer Beakers (250 ml) Watch glass Graduated Cylinder (10 ml) Mel-Temp
2.2 Physical Properties. Physical Properties. Notes due EOC
 Physical Properties Notes due EOC There are pitchers of ice water and lemonade on a picnic table. How do you know which liquid is in each pitcher? It s easy! Lemonade is yellow and has a tart taste that
Physical Properties Notes due EOC There are pitchers of ice water and lemonade on a picnic table. How do you know which liquid is in each pitcher? It s easy! Lemonade is yellow and has a tart taste that
INDIAN SCHOOL MUSCAT SENIOR SECTION DEPARTMENT OF CHEMISTRY CLASS X- PRACTICAL WORKSHEET
 INDIAN SCHOOL MUSCAT SENIOR SECTION DEPARTMENT OF CHEMISTRY CLASS X- PRACTICAL WORKSHEET Different types of chemical reactions Experiment No: 1(a) Combination reaction Objectives: To study the Combination
INDIAN SCHOOL MUSCAT SENIOR SECTION DEPARTMENT OF CHEMISTRY CLASS X- PRACTICAL WORKSHEET Different types of chemical reactions Experiment No: 1(a) Combination reaction Objectives: To study the Combination
Electricity and Chemistry
 Electricity and Chemistry Electrochemistry: It is a branch of chemistry that deals with the reactions involving the conversion of chemical energy into electrical energy and vice-versa. Electrochemical
Electricity and Chemistry Electrochemistry: It is a branch of chemistry that deals with the reactions involving the conversion of chemical energy into electrical energy and vice-versa. Electrochemical
Extracting and using metals. ores. native. Only the most unreactive metals such as gold and platinum are found as native metals.
 Extracting and using metals Only the most unreactive metals such as gold and platinum are found as native metals. ores All the other metals we use are extracted from their ores by chemical processes. native
Extracting and using metals Only the most unreactive metals such as gold and platinum are found as native metals. ores All the other metals we use are extracted from their ores by chemical processes. native
Oxidation and Reduction
 Oxidation and Reduction An oxidation reaction is one in which oxygen is added to a substance. Example: Methane is oxidised when it burns in air. Oxygen is added to the carbon in methane, forming carbon
Oxidation and Reduction An oxidation reaction is one in which oxygen is added to a substance. Example: Methane is oxidised when it burns in air. Oxygen is added to the carbon in methane, forming carbon
The table below shows some of the properties of the elements cobalt and nickel.
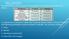 BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
NCEA Level 1 Chemistry (90933) 2012 page 1 of 5. Q Evidence Achievement Achievement with Merit Achievement with Excellence NØ N1 N2 A3 A4 M5 M6 E7 E8
 Assessment Schedule 2012 NCEA Level 1 Chemistry (90933) 2012 page 1 of 5 Chemistry: Demonstrate understanding of aspects of selected elements (90933) Evidence Statement Q Evidence with Merit with Excellence
Assessment Schedule 2012 NCEA Level 1 Chemistry (90933) 2012 page 1 of 5 Chemistry: Demonstrate understanding of aspects of selected elements (90933) Evidence Statement Q Evidence with Merit with Excellence
Copper Odyssey. Chemical Reactions of Copper
 Name Lab Partner(s) Copper Odyssey Chemical Reactions of Copper Date Period Elemental copper metal will be converted into copper (II) ion and then brought through a series of compound conversions until
Name Lab Partner(s) Copper Odyssey Chemical Reactions of Copper Date Period Elemental copper metal will be converted into copper (II) ion and then brought through a series of compound conversions until
GCSE BITESIZE Examinations
 GCSE BITESIZE Examinations General Certificate of Secondary Education AQA SCIENCE A CHY1A Unit Chemistry C1a (Products from Rocks) AQA Chemistry Unit Chemistry C1a (Products from Rocks) FOUNDATION TIER
GCSE BITESIZE Examinations General Certificate of Secondary Education AQA SCIENCE A CHY1A Unit Chemistry C1a (Products from Rocks) AQA Chemistry Unit Chemistry C1a (Products from Rocks) FOUNDATION TIER
Chapter 11 The Periodic Table
 Chapter 11 The Periodic Table Lesson 1 Using the Periodic Table Textbook pages 391 397 Lesson 2 Metals Textbook pages 401 402 Lesson 3 Nonmetals and Metalloids Textbook pages 409 410 & 413-414 1 Chapter
Chapter 11 The Periodic Table Lesson 1 Using the Periodic Table Textbook pages 391 397 Lesson 2 Metals Textbook pages 401 402 Lesson 3 Nonmetals and Metalloids Textbook pages 409 410 & 413-414 1 Chapter
Lesson note 5: Separation of Mixtures. Mixtures. Mixtures. Separation Methods: Many different types of mixtures Examples:
 Lesson note 5: Separation of Mixtures Mixtures Many different types of mixtures Examples: - air (gas-gas mixture), - sea water(liquid-solid mixture), - crude oil (liquid-liquid mixture) The mixtures can
Lesson note 5: Separation of Mixtures Mixtures Many different types of mixtures Examples: - air (gas-gas mixture), - sea water(liquid-solid mixture), - crude oil (liquid-liquid mixture) The mixtures can
Teknik Bioseparasi. Dina Wahyu. Genap/ Maret 2014
 4. Teknik Bioseparasi Dina Wahyu Genap/ Maret 2014 Outline Chemical Reaction Engineering 1 2 3 4 5 6 7 Pendahuluan mempelajari ruang lingkup teknik bioseparasi dan teknik cel disruption Teknik Pemisahan
4. Teknik Bioseparasi Dina Wahyu Genap/ Maret 2014 Outline Chemical Reaction Engineering 1 2 3 4 5 6 7 Pendahuluan mempelajari ruang lingkup teknik bioseparasi dan teknik cel disruption Teknik Pemisahan
The diagram shows some of the substances used and produced at a cement works. . Write the correct number in the box to complete each sentence.
 Q. A limestone quarry is in an area of natural beauty and near several villages. The company operating the quarry wants planning permission to build a new cement works in the quarry. The diagram shows
Q. A limestone quarry is in an area of natural beauty and near several villages. The company operating the quarry wants planning permission to build a new cement works in the quarry. The diagram shows
(a) To find out which is the more reactive metal, zinc or tin, the following experiment could be carried out. piece of zinc shiny surface
 1 The reactivity series lists metals in order of reactivity. (a) To find out which is the more reactive metal, zinc or tin, the following experiment could be carried out. piece of zinc shiny surface tin(ii)
1 The reactivity series lists metals in order of reactivity. (a) To find out which is the more reactive metal, zinc or tin, the following experiment could be carried out. piece of zinc shiny surface tin(ii)
Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3
 Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3 Directions to Students: Do not open this booklet until you are told to do so. Please respond to the following items by marking the best answer
Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3 Directions to Students: Do not open this booklet until you are told to do so. Please respond to the following items by marking the best answer
3 The Formation, Mining, and Use of Minerals
 CHAPTER 3 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
CHAPTER 3 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
THE PERIODIC TABLE. Chapter 15
 THE PERIODIC TABLE Chapter 15 MENDELEEV 1869 The first version of the periodic table was published by Dmitri Mendeleev, a Russian chemist He arranged the elements in order of increasing atomic mass This
THE PERIODIC TABLE Chapter 15 MENDELEEV 1869 The first version of the periodic table was published by Dmitri Mendeleev, a Russian chemist He arranged the elements in order of increasing atomic mass This
concentration of acid in mol / dm 3 temperature / C ti / min
 1 (a A small piece of marble, calcium carbonate, was added to 5 cm 3 of hydrochloric acid at 25 C. The time taken for the reaction to stop was measured. CaCO 3 (s) + 2HCl(aq) CaCl 2 (aq) + CO 2 (g) + H
1 (a A small piece of marble, calcium carbonate, was added to 5 cm 3 of hydrochloric acid at 25 C. The time taken for the reaction to stop was measured. CaCO 3 (s) + 2HCl(aq) CaCl 2 (aq) + CO 2 (g) + H
TWEED RIVER HIGH SCHOOL 2006 PRELIMINARY CHEMISTRY. Unit 2 Metals
 TWEED RIVER HIGH SCHOOL 2006 PRELIMINARY CHEMISTRY Unit 2 Metals Part 2 Metals differ in their reactivity with other chemicals and this influences their uses. Describe observable changes when metals react
TWEED RIVER HIGH SCHOOL 2006 PRELIMINARY CHEMISTRY Unit 2 Metals Part 2 Metals differ in their reactivity with other chemicals and this influences their uses. Describe observable changes when metals react
CHEM 1215 LAB NOTES EXPT #2: PHYSICAL AND CHEMICAL CHANGES 1
 CHEM 1215 LAB NOTES EXPT #2: PHYSICAL AND CHEMICAL CHANGES 1 TECHNIQUES: chemical and physical changes, reactions, observations READING: PHYSICAL AND CHEMICAL CHANGES e.g. Tro chapter 1 SAFETY: Safety
CHEM 1215 LAB NOTES EXPT #2: PHYSICAL AND CHEMICAL CHANGES 1 TECHNIQUES: chemical and physical changes, reactions, observations READING: PHYSICAL AND CHEMICAL CHANGES e.g. Tro chapter 1 SAFETY: Safety
Equation Writing and Predicting Products Chemistry I Acc
 Introduction: Equation Writing and Predicting Products Chemistry I Acc If you examine your bicycle after it has been left out in the rain a number of times you will find that it has begun to rust. Rust
Introduction: Equation Writing and Predicting Products Chemistry I Acc If you examine your bicycle after it has been left out in the rain a number of times you will find that it has begun to rust. Rust
METALS AND NON-METALS
 METALS AND NON-METALS VERY SHORT ANSWER TYPE QUESTIONS [1 MARK] 1. A green layer is gradually formed on a copper plate left exposed to air for a week in a bathroom. What could this green substance be?
METALS AND NON-METALS VERY SHORT ANSWER TYPE QUESTIONS [1 MARK] 1. A green layer is gradually formed on a copper plate left exposed to air for a week in a bathroom. What could this green substance be?
1. Scaling. H.O.: H-5/21, KRISHNA NAGAR, DELHI Tel.: , Fax:
 Boiler Water Problems and Its Causes Water is the essential medium for steam generation. Conditioning it properly can increase the efficiency of boiler and as well as extend the boiler s life. Treating
Boiler Water Problems and Its Causes Water is the essential medium for steam generation. Conditioning it properly can increase the efficiency of boiler and as well as extend the boiler s life. Treating
Labs Covered. Le Chatelier. Calculating Ka. Kitchen Chemistry Project. Calculating Kc. Redox lab
 Labs Covered Le Chatelier Calculating Kc Calculating Ka Redox lab Kitchen Chemistry Project LeChatelier Sample Questions Fe +3 (aq) + SCN -1 (aq) FeSCN +2 (aq) Light orange colourless red a) To create
Labs Covered Le Chatelier Calculating Kc Calculating Ka Redox lab Kitchen Chemistry Project LeChatelier Sample Questions Fe +3 (aq) + SCN -1 (aq) FeSCN +2 (aq) Light orange colourless red a) To create
Food Processing Agricultural Commodities for Residue Analysis
 Food Processing Agricultural Commodities for Residue Analysis presented by Joshua Bevan Mission Statement...to provide a facility and/or resources to assist food companies with food related issues that
Food Processing Agricultural Commodities for Residue Analysis presented by Joshua Bevan Mission Statement...to provide a facility and/or resources to assist food companies with food related issues that
GRAVIMETRIC DETERMINATION OF SULFATE IN AN UNKNOWN SOLUTION
 GRAVIMETRIC DETERMINATION OF SULFATE IN AN UNKNOWN SOLUTION AIM The main objective of this experiment is to determine the concentration of sulfate ion in an unknown solution by using gravimetry. INTRODUCTION
GRAVIMETRIC DETERMINATION OF SULFATE IN AN UNKNOWN SOLUTION AIM The main objective of this experiment is to determine the concentration of sulfate ion in an unknown solution by using gravimetry. INTRODUCTION
The oxides of nitrogen and the details of the oxygen states of nitrogen and the N:O ratio can be presented in a tabular form as:
 Oxides of Nitrogen Introduction to oxides of nitrogen Nirogen has a position in second period of group V in the modern periodic table. It has molecular formula N2. It has atomic number 7 and atomic weight
Oxides of Nitrogen Introduction to oxides of nitrogen Nirogen has a position in second period of group V in the modern periodic table. It has molecular formula N2. It has atomic number 7 and atomic weight
Physical Behavior of Metals
 Activity 4 Physical Behavior of Metals GOALS In this activity you will: Discover what an alloy is. Make a brass-coated penny. Determine how the properties of a metal are affected by making it into an alloy.
Activity 4 Physical Behavior of Metals GOALS In this activity you will: Discover what an alloy is. Make a brass-coated penny. Determine how the properties of a metal are affected by making it into an alloy.
GCE Environmental Technology. Energy from Biomass. For first teaching from September 2013 For first award in Summer 2014
 GCE Environmental Technology Energy from Biomass For first teaching from September 2013 For first award in Summer 2014 Energy from Biomass Specification Content should be able to: Students should be able
GCE Environmental Technology Energy from Biomass For first teaching from September 2013 For first award in Summer 2014 Energy from Biomass Specification Content should be able to: Students should be able
 Stoichiometric Calculations 1. The weight of calcium carbonate required to produce carbon-dioxide that is sufficient for conversion of one 0.1 mole sodium carbonate to sodium bicarbonate is 1) 1gm 2) 10gm
Stoichiometric Calculations 1. The weight of calcium carbonate required to produce carbon-dioxide that is sufficient for conversion of one 0.1 mole sodium carbonate to sodium bicarbonate is 1) 1gm 2) 10gm
Water Quality in East Chicago. December 12, 2016
 Water Quality in East Chicago December 12, 2016 What is lead? Lead is a toxic colorless, odorless, and tasteless metal It is found throughout the environment In the past lead was often used in: Paint Gasoline
Water Quality in East Chicago December 12, 2016 What is lead? Lead is a toxic colorless, odorless, and tasteless metal It is found throughout the environment In the past lead was often used in: Paint Gasoline
Alloys and Solid Solutions
 Alloys and Solid Solutions Chemistry 123 Spring 2008 Dr. Woodward Solutions Solid Solution 14 Carat Gold Liquid Solution Vodka Gaseous Solution Air Solution = A homogeneous mixture 1 Alloys An alloy is
Alloys and Solid Solutions Chemistry 123 Spring 2008 Dr. Woodward Solutions Solid Solution 14 Carat Gold Liquid Solution Vodka Gaseous Solution Air Solution = A homogeneous mixture 1 Alloys An alloy is
GENARAL INTRODUCTION TO METALLURGY :Std: XI-CHEMISTRY
 GENARAL INTRODUCTION TO METALLURGY :Std: XI-CHEMISTRY 1. What is matrix? The ore is generally associated with rock impurities like clay, sand etc. called gangue or matrix 2. What is mineral? The natural
GENARAL INTRODUCTION TO METALLURGY :Std: XI-CHEMISTRY 1. What is matrix? The ore is generally associated with rock impurities like clay, sand etc. called gangue or matrix 2. What is mineral? The natural
Partner: Cathy 22 March Separation and Qualitative Determination of Cations and Anions
 Partner: Cathy 22 March 2012 Separation and Qualitative Determination of Cations and Anions Purpose: The purpose of this lab is to identify the cations and anions components in the unknown solution. This
Partner: Cathy 22 March 2012 Separation and Qualitative Determination of Cations and Anions Purpose: The purpose of this lab is to identify the cations and anions components in the unknown solution. This
Oxygen. Oxygen is one of the fundamental resources required by life forms on Earth. Aquatic ecosystems have a wide assortment of life forms.
 Oxygen Oxygen is one of the fundamental resources required by life forms on Earth. Aquatic ecosystems have a wide assortment of life forms. Oxygen is also required for some natural chemical decays. What
Oxygen Oxygen is one of the fundamental resources required by life forms on Earth. Aquatic ecosystems have a wide assortment of life forms. Oxygen is also required for some natural chemical decays. What
Safety Precautions. Hydrochloric acid, nitric acid and gold salts are corrosive.
 Stefan s Dental Gold Experiment By Stefan aka Garage Chemist Loosely translated from German to English by Steven Sackett 12/08/2007 The original German version is located here: Versuchschemie.de Safety
Stefan s Dental Gold Experiment By Stefan aka Garage Chemist Loosely translated from German to English by Steven Sackett 12/08/2007 The original German version is located here: Versuchschemie.de Safety
Element, Mixture, Compound Lab
 Element, Mixture, Compound Lab Background: Material engineers and mechanical engineers are focused on understanding different materials so that they can create new materials with desired properties. For
Element, Mixture, Compound Lab Background: Material engineers and mechanical engineers are focused on understanding different materials so that they can create new materials with desired properties. For
Mark Scheme (Results) Summer Pearson Edexcel GCSE in Chemistry (5CH1F) Paper 01
 Mark Scheme (Results) Summer 2014 Pearson Edexcel GCSE in Chemistry (5CH1F) Paper 01 Edexcel and BTEC Qualifications Edexcel and BTEC qualifications come from Pearson, the world s leading learning company.
Mark Scheme (Results) Summer 2014 Pearson Edexcel GCSE in Chemistry (5CH1F) Paper 01 Edexcel and BTEC Qualifications Edexcel and BTEC qualifications come from Pearson, the world s leading learning company.
CALIFORNIA EDUCATION AND THE ENVIRONMENT INITIATIVE
 Water Vapor: A GHG Lesson 3 page 1 of 2 Water Vapor: A GHG Water vapor in our atmosphere is an important greenhouse gas (GHG). On a cloudy day we can see evidence of the amount of water vapor in our atmosphere.
Water Vapor: A GHG Lesson 3 page 1 of 2 Water Vapor: A GHG Water vapor in our atmosphere is an important greenhouse gas (GHG). On a cloudy day we can see evidence of the amount of water vapor in our atmosphere.
CBSE QUESTION PAPER CLASS-X SCIENCE
 CBSE QUESTION PAPER CLASS-X SCIENCE SECTION - A Q.1. Why decomposition reactions are called the opposite of combination reactions? 1mark Q.2. What happens to the resistance of a conductor when its area
CBSE QUESTION PAPER CLASS-X SCIENCE SECTION - A Q.1. Why decomposition reactions are called the opposite of combination reactions? 1mark Q.2. What happens to the resistance of a conductor when its area
AQA GCSE Design and Technology 8552
 AQA GCSE Design and Technology 8552 Metals and alloys Unit 3 Materials and their working properties 3 Objectives Know the primary sources of materials for producing metals and alloys Be able to recognise
AQA GCSE Design and Technology 8552 Metals and alloys Unit 3 Materials and their working properties 3 Objectives Know the primary sources of materials for producing metals and alloys Be able to recognise
REPORT NUMBER REPORT DATE SEND TO ISSUE DATE Apr 18, Apr 18, 2017 RECEIVED DATE Apr 05, 2017
 PAGE 1/7 Sample ID: EWA PELLETS Lab Number: 2653281 Date Sampled: 2017-04-04 0850 Carbon nitrogen ratio C/N 6 : 1 0.1 Calculation Auto-2017/04/12 Auto-2017/04/18 Carbon (total) 38.53 % 0.050 ASTM D 5373
PAGE 1/7 Sample ID: EWA PELLETS Lab Number: 2653281 Date Sampled: 2017-04-04 0850 Carbon nitrogen ratio C/N 6 : 1 0.1 Calculation Auto-2017/04/12 Auto-2017/04/18 Carbon (total) 38.53 % 0.050 ASTM D 5373
SUBJECT: Integrated Science TEACHER: Mr. S. Campbell DATE: GRADE: 7 DURATION: 2 wks GENERAL TOPIC: The Sun As The Main Source Of Energy / Fuels and
 SUBJECT: Integrated Science TEACHER: Mr. S. Campbell DATE: GRADE: 7 DURATION: 2 wks GENERAL TOPIC: The Sun As The Main Source Of Energy / Fuels and their uses in the home. The Sun As The Main Source Of
SUBJECT: Integrated Science TEACHER: Mr. S. Campbell DATE: GRADE: 7 DURATION: 2 wks GENERAL TOPIC: The Sun As The Main Source Of Energy / Fuels and their uses in the home. The Sun As The Main Source Of
Assistant Lecturer. Sahar Mohammed Shakir Assistant Lecturer. Abdul Hafeedh Hameed Assistant Lecturer. Ali Basim
 Assistant Lecturer Sahar Mohammed Shakir Assistant Lecturer Abdul Hafeedh Hameed Assistant Lecturer Ali Basim Solid organic cpd.s when isolated from organic reaction are impure; they are contaminated with
Assistant Lecturer Sahar Mohammed Shakir Assistant Lecturer Abdul Hafeedh Hameed Assistant Lecturer Ali Basim Solid organic cpd.s when isolated from organic reaction are impure; they are contaminated with
Chemistry CH1FP. (Jun15CH1FP01) General Certificate of Secondary Education Foundation Tier June Unit Chemistry C1. Unit Chemistry C1 TOTAL
 Centre Number Surname Candidate Number For Examiner s Use Other Names Candidate Signature Examiner s Initials Question Mark Science A Unit Chemistry C1 Chemistry Unit Chemistry C1 Tuesday 9 June 2015 General
Centre Number Surname Candidate Number For Examiner s Use Other Names Candidate Signature Examiner s Initials Question Mark Science A Unit Chemistry C1 Chemistry Unit Chemistry C1 Tuesday 9 June 2015 General
One of the main ores of zinc is zinc blende, ZnS. There are two stages in the extraction of zinc from this ore.
 1 Two of the main uses of zinc are for galvanising and for making alloys. One of the main ores of zinc is zinc blende, ZnS. There are two stages in the extraction of zinc from this ore. (a) Stage 1 Zinc
1 Two of the main uses of zinc are for galvanising and for making alloys. One of the main ores of zinc is zinc blende, ZnS. There are two stages in the extraction of zinc from this ore. (a) Stage 1 Zinc
Lab #3: Law of Definite Proportions
 Name Lab #3: Law of Definite Proportions Sept. 21, 2016 Purpose To find the percent composition and therefore definite ratio of the elements in magnesium oxide. Background When magnesium and oxygen are
Name Lab #3: Law of Definite Proportions Sept. 21, 2016 Purpose To find the percent composition and therefore definite ratio of the elements in magnesium oxide. Background When magnesium and oxygen are
Estimation of total, permanent and temporary hardness of water (EDTA method)
 Estimation of total, permanent and temporary hardness of water (EDTA method) Expt. No.: Date: Aim: To estimate the amount of total, permanent and temporary hardness in the collected sample of water. A
Estimation of total, permanent and temporary hardness of water (EDTA method) Expt. No.: Date: Aim: To estimate the amount of total, permanent and temporary hardness in the collected sample of water. A
5-4 Chemical changes Trilogy
 5-4 Chemical changes Trilogy.0 A student investigated the reaction of sodium carbonate with dilute hydrochloric acid. The student used the apparatus shown in Figure. Figure Sodium carbonate This is the
5-4 Chemical changes Trilogy.0 A student investigated the reaction of sodium carbonate with dilute hydrochloric acid. The student used the apparatus shown in Figure. Figure Sodium carbonate This is the
, to form the products magnesium oxide, MgO(s), and magnesium nitride, Mg 3
 Active Metals in the Periodic Table Periodic Trends and the Properties of the Elements SCIENTIFIC Introduction Like good experiments, good demonstrations often lead to other demonstrations. What follows
Active Metals in the Periodic Table Periodic Trends and the Properties of the Elements SCIENTIFIC Introduction Like good experiments, good demonstrations often lead to other demonstrations. What follows
MAINTAINING DRIP IRRIGATION SYSTEMS
 MAINTAINING DRIP IRRIGATION SYSTEMS Bill Lamont Penn State Horticulture and Extension wlamont@psu.edu This article is a re-print from an article previously published in The Vegetable & Small Fruit Gazette,
MAINTAINING DRIP IRRIGATION SYSTEMS Bill Lamont Penn State Horticulture and Extension wlamont@psu.edu This article is a re-print from an article previously published in The Vegetable & Small Fruit Gazette,
*20GSD5201* Double Award Science: Chemistry. Unit C2 Higher Tier TUESDAY 9 JUNE 2015, AFTERNOON [GSD52] *GSD52* *G5802* TIME 1 hour 15 minutes.
![*20GSD5201* Double Award Science: Chemistry. Unit C2 Higher Tier TUESDAY 9 JUNE 2015, AFTERNOON [GSD52] *GSD52* *G5802* TIME 1 hour 15 minutes. *20GSD5201* Double Award Science: Chemistry. Unit C2 Higher Tier TUESDAY 9 JUNE 2015, AFTERNOON [GSD52] *GSD52* *G5802* TIME 1 hour 15 minutes.](/thumbs/76/73786999.jpg) Centre Number Candidate Number General Certificate of Secondary Education 2015 Double Award Science: Chemistry Unit C2 Higher Tier [GSD52] *GSD52* *G5802* *GSD52* TUESDAY 9 JUNE 2015, AFTERNOON TIME 1
Centre Number Candidate Number General Certificate of Secondary Education 2015 Double Award Science: Chemistry Unit C2 Higher Tier [GSD52] *GSD52* *G5802* *GSD52* TUESDAY 9 JUNE 2015, AFTERNOON TIME 1
Removing Heavy Metals from Wastewater
 Removing Heavy Metals from Wastewater Engineering Research Center Report David M. Ayres Allen P. Davis Paul M. Gietka August 1994 1 Removing Heavy Metals From Wastewater Introduction This manual provides
Removing Heavy Metals from Wastewater Engineering Research Center Report David M. Ayres Allen P. Davis Paul M. Gietka August 1994 1 Removing Heavy Metals From Wastewater Introduction This manual provides
Name Product identifier % GHS-US classification
 SECTION 1: Identification 1.1. Identification Product form Substance name Water Date of issue: 11/15/2013 Revision date: 01/25/2017 Supersedes: 01/25/2017 Version: 1.2 : Substance : Water CAS No : 7732-18-5
SECTION 1: Identification 1.1. Identification Product form Substance name Water Date of issue: 11/15/2013 Revision date: 01/25/2017 Supersedes: 01/25/2017 Version: 1.2 : Substance : Water CAS No : 7732-18-5
5072 CHEMISTRY (NEW PAPERS WITH SPA) TOPIC 9: METALS 5067 CHEMISTRY (NEW PAPERS WITH PRACTICAL EXAM) TOPIC 9: METALS
 5072 CHEMISTRY (NEW PAPERS WITH SPA) TOPIC 9: METALS 5067 CHEMISTRY (NEW PAPERS WITH PRACTICAL EXAM) TOPIC 9: METALS SUB-TOPIC 9.3 TO 5 EXTRACTION OF METALS; RECYLING OF METALS; IRON LEARNING OUTCOMES
5072 CHEMISTRY (NEW PAPERS WITH SPA) TOPIC 9: METALS 5067 CHEMISTRY (NEW PAPERS WITH PRACTICAL EXAM) TOPIC 9: METALS SUB-TOPIC 9.3 TO 5 EXTRACTION OF METALS; RECYLING OF METALS; IRON LEARNING OUTCOMES
Water cycles through ecosystems.
 Water cycles through ecosystems. Water is stored on Earth s surface in lakes, rivers, and oceans. Water is found underground, filling the spaces between soil particles and cracks in rocks. Large amounts
Water cycles through ecosystems. Water is stored on Earth s surface in lakes, rivers, and oceans. Water is found underground, filling the spaces between soil particles and cracks in rocks. Large amounts
7.3 Bonding in Metals > Chapter 7 Ionic and Metallic Bonding. 7.3 Bonding in Metals. 7.1 Ions 7.2 Ionic Bonds and Ionic Compounds
 Chapter 7 Ionic and Metallic Bonding 7.1 Ions 7.2 Ionic Bonds and Ionic Compounds 7.3 Bonding in Metals 1 Copyright Pearson Education, Inc., or its affiliates. All Rights Reserved. CHEMISTRY & YOU What
Chapter 7 Ionic and Metallic Bonding 7.1 Ions 7.2 Ionic Bonds and Ionic Compounds 7.3 Bonding in Metals 1 Copyright Pearson Education, Inc., or its affiliates. All Rights Reserved. CHEMISTRY & YOU What
Hydrogen Sulphide and Mercaptan Removal
 Natural Gas The significance of natural gas as an energy source is increasing rapidly. Yet its discovery and exploitation are far more recent than those of oil as gas projects have begun to develop since
Natural Gas The significance of natural gas as an energy source is increasing rapidly. Yet its discovery and exploitation are far more recent than those of oil as gas projects have begun to develop since
REVISION NO : 6 January-2015
 1. PRODUCT AND COMPANY IDENTIFICATION Product Name Magnalium Powder (Alloy of Magnesium and Aluminium) Chemical Symbol Mg/Al Mg Al CAS No 7439-95-4 7429-90-5 EINECS No 231-104-6 231-072-3 Supplier Name
1. PRODUCT AND COMPANY IDENTIFICATION Product Name Magnalium Powder (Alloy of Magnesium and Aluminium) Chemical Symbol Mg/Al Mg Al CAS No 7439-95-4 7429-90-5 EINECS No 231-104-6 231-072-3 Supplier Name
MAHESH TUTORIALS I.C.S.E.
 ICSE X MAES TUTORIALS I.C.S.E. Marks : 80 Exam No. : MT/ICSE/I SPA 033SUBJECT : CEMISTRY Time : 2 hrs. Topics : Sulphuric acid, Ammonia, Analytical Chemistry, Organic Chemistry Cl, Nitric acid, Metallurgy
ICSE X MAES TUTORIALS I.C.S.E. Marks : 80 Exam No. : MT/ICSE/I SPA 033SUBJECT : CEMISTRY Time : 2 hrs. Topics : Sulphuric acid, Ammonia, Analytical Chemistry, Organic Chemistry Cl, Nitric acid, Metallurgy
TYPES OF CHEMICAL REACTIONS PART I INTRODUCTION
 EXPERIMENT 10 (2 Weeks) Chemistry 100 Laboratory TYPES OF CHEMICAL REACTIONS PART I INTRODUCTION It is useful to classify reactions into different types, because products of reactions can be predicted.
EXPERIMENT 10 (2 Weeks) Chemistry 100 Laboratory TYPES OF CHEMICAL REACTIONS PART I INTRODUCTION It is useful to classify reactions into different types, because products of reactions can be predicted.
Material Safety Data Sheet
 MSDS nº 020 Review: 02 Date: 30/01/2015 Page: 1/6 1. PRODUCT AND COMPANY IDENTIFICATION 1.1 Identification of the product Stanlux Pell Gold 1.2 Main recommended uses Masterbatch 1.3 Company s identification
MSDS nº 020 Review: 02 Date: 30/01/2015 Page: 1/6 1. PRODUCT AND COMPANY IDENTIFICATION 1.1 Identification of the product Stanlux Pell Gold 1.2 Main recommended uses Masterbatch 1.3 Company s identification
Chapter 9 Forms of Energy
 Chapter 9 Forms of Energy 9.1 What is energy? Energy is the ability to make things move. It is all around us but sometimes we don t recognize it. In order to properly describe energy, we classify it according
Chapter 9 Forms of Energy 9.1 What is energy? Energy is the ability to make things move. It is all around us but sometimes we don t recognize it. In order to properly describe energy, we classify it according
SCIENCE Course of Study
 6 th GRADE SPECIAL EDUCATION SCIENCE Course of Study Findlay City Schools 2005 6 TH GRADE BENCHMARK: (A) Use skills of scientific inquiry processes (e.g., hypothesis, record keeping, description and explanation).
6 th GRADE SPECIAL EDUCATION SCIENCE Course of Study Findlay City Schools 2005 6 TH GRADE BENCHMARK: (A) Use skills of scientific inquiry processes (e.g., hypothesis, record keeping, description and explanation).
PurAwater Softener. Hard White Film, Scale Buildup, Haze on Fixtures, Lower Water Pressure
 Hard White Film, Scale Buildup, Haze on Fixtures, Lower Water Pressure PurAwater Softener The hardness of water is a measure of the amount of minerals, primarily calcium and magnesium, it contains. Water
Hard White Film, Scale Buildup, Haze on Fixtures, Lower Water Pressure PurAwater Softener The hardness of water is a measure of the amount of minerals, primarily calcium and magnesium, it contains. Water
2015 O LEVEL CHEMISTRY 5073/02
 OF SCIENCE 2015 O LEVEL CHEMISTRY 5073/02 SECTION A 1. The table shows some common oxidation states for some elements in their compounds. element common oxidation states metal / non-metal A 2 non-metal
OF SCIENCE 2015 O LEVEL CHEMISTRY 5073/02 SECTION A 1. The table shows some common oxidation states for some elements in their compounds. element common oxidation states metal / non-metal A 2 non-metal
Abrasive Flo Dry Finishing Media
 Abrasive Flo Dry Finishing Media North Kingstown, RI Phone: 401-667-7902 info@belairfinishing.com www.belairfinishing.com D E S C R I P T I O N O F C O M P O S I T I O N S ORGANIC MEDIA D E S C R I P T
Abrasive Flo Dry Finishing Media North Kingstown, RI Phone: 401-667-7902 info@belairfinishing.com www.belairfinishing.com D E S C R I P T I O N O F C O M P O S I T I O N S ORGANIC MEDIA D E S C R I P T
: Magnesium Sulfate, 2.25% w/v
 Date of issue: 12/23/2013 Revision date: 11/08/2017 Supersedes: 11/08/2017 Version: 1.1 SECTION 1: Identification 1.1. Identification Product form Product name Product code : Mixtures : Magnesium Sulfate,
Date of issue: 12/23/2013 Revision date: 11/08/2017 Supersedes: 11/08/2017 Version: 1.1 SECTION 1: Identification 1.1. Identification Product form Product name Product code : Mixtures : Magnesium Sulfate,
Chapter 20 CHEMISTRY. Metallurgy and the Chemistry of Metals. Dr. Ibrahim Suleiman
 CHEMISTRY Chapter 20 Metallurgy and the Chemistry of Metals Dr. Ibrahim Suleiman GENERAL PROPERTIES AND STRUCTURE OF METALS opaque good conductors of heat and electricity high malleability and ductility
CHEMISTRY Chapter 20 Metallurgy and the Chemistry of Metals Dr. Ibrahim Suleiman GENERAL PROPERTIES AND STRUCTURE OF METALS opaque good conductors of heat and electricity high malleability and ductility
SAFETY DATA SHEET WORKING COPY Finished product
 Page 1 of 5 SAFETY DATA SHEET WORKING COPY Finished product Date Prepared : 03/26/2015 SDS No : F1 1. PRODUCT AND COMPANY IDENTIFICATION GENERAL USE: Injection Molding PRODUCT DESCRIPTION: Glass Filled
Page 1 of 5 SAFETY DATA SHEET WORKING COPY Finished product Date Prepared : 03/26/2015 SDS No : F1 1. PRODUCT AND COMPANY IDENTIFICATION GENERAL USE: Injection Molding PRODUCT DESCRIPTION: Glass Filled
Food. Overview. Optimizing Production and Freezing Processes for the Food Industry. Food
 Overview Optimizing Production and Freezing Processes for the Industry With over 100 years experience in the food and beverage industry, we have the know-how to help you optimize your production and freezing
Overview Optimizing Production and Freezing Processes for the Industry With over 100 years experience in the food and beverage industry, we have the know-how to help you optimize your production and freezing
Material Safety Data Sheet. : Oleo Pine Resin
 Material Safety Data Sheet Oleo Pine Resin Section 1 - Chemical Product and Company Identification MSDS Name : Oleo Pine Resin Synonyms : Pine Resin Turpentine Company Identification : Tradeasia International
Material Safety Data Sheet Oleo Pine Resin Section 1 - Chemical Product and Company Identification MSDS Name : Oleo Pine Resin Synonyms : Pine Resin Turpentine Company Identification : Tradeasia International
Boiling point in C. Colour in aqueous solution. Fluorine 188 colourless. Chlorine 35 pale green. Bromine X orange.
 Q1.This question is about halogens and their compounds. The table below shows the boiling points and properties of some of the elements in Group 7 of the periodic table. Element Boiling point in C Colour
Q1.This question is about halogens and their compounds. The table below shows the boiling points and properties of some of the elements in Group 7 of the periodic table. Element Boiling point in C Colour
Properties of Minerals
 What is a mineral? Properties of Minerals Mineral 5 characteristics: naturally occurring (not made by people) inorganic (cannot come from living things) solid (definite shape and volume) crystal structure
What is a mineral? Properties of Minerals Mineral 5 characteristics: naturally occurring (not made by people) inorganic (cannot come from living things) solid (definite shape and volume) crystal structure
Water for Instrument Processing
 Water for Instrument Processing by Marcia Frieze, Case Medical Water, the universal solvent Water can dissolve more substances than any other liquid. It is essentially nonionic or neutral. While alkaline
Water for Instrument Processing by Marcia Frieze, Case Medical Water, the universal solvent Water can dissolve more substances than any other liquid. It is essentially nonionic or neutral. While alkaline
NIEMI (ENERGY) CARBON AND THE ENERGY WE USE
 NIEMI (ENERGY) CARBON AND THE ENERGY WE USE Photograph of the La Cygne Power Plant (Kansas City Power and Light) Photo by Tina M. Niemi June 14, 2007 VERSION #2 1 Lesson #1 Combustion Exploration What
NIEMI (ENERGY) CARBON AND THE ENERGY WE USE Photograph of the La Cygne Power Plant (Kansas City Power and Light) Photo by Tina M. Niemi June 14, 2007 VERSION #2 1 Lesson #1 Combustion Exploration What
