Measurement and Density - Experiment 1
|
|
|
- Dominic Short
- 6 years ago
- Views:
Transcription
1 Measurement and Density - Experiment 1 Purpose: The purpose of this experiment is to acquaint you with some common metric units of length, volume, and mass. You will also measure the density of an unknown metal and use this to try to identify the metal. Procedure: Record all of your observations in the blanks provided using the metric system. Include the units on each measurement and show all of your calculations. For lengths, use centimeters (cm); for volume, cubic centimeters (cm 3 ); and for mass, grams (g). Note that 1 cm 3 = 1 milliliter (ml) and 1000 ml = 1 Liter (L). Experiment #1 1. Measure and record the length, width, and thickness of a book. (You can following the example but you will get different values). (Example) (Your book) a. Length (L) : cm b. Width (W) : cm c. Thickness (T) : 2.00 cm The above three are examples of direct measurements. Indirect measurements follow from mathematical operations (addition, subtraction, multiplication, division) on direct measurements. 2. Using, length, width, and height, we indirectly measure the volume. For the Example, a calculator will help you determine that its volume must be near units. The volume is reported as 982 cm 3. a. What is the volume of your book (show your work)? b. The volume was found by a (direct / indirect ) measurement. (Choose one) 3. Now measure the mass of the book. a. What is the mass of the book? b. How many sheets of paper are in the book? 4. Indirect measurements. a. What is the thickness of each sheet? HINT: thickness of book / # sheets b. What is the mass of each sheet? c. Would you expect to get the same answer if you weighed one sheet by itself, why? 1
2 Experiment #2 - There is a sample of unknown liquid on your desk. In this next experiment you will identify an unknown liquid. This liquid was found in a bottle labeled organic unknown - experiment #4". A search of our stockroom found an old organic lab prep notebook describing experiment #4". It said there were four possible organic unknowns: butanol, water, carbon tetrachloride, and chloroform. We will do some measurements on the unknown liquid to help us determine which one of these four compounds is in the bottle. 1. What is the number on your unknown bottle? 2. Determine the mass (g) of your unknown liquid. a. What is the mass of the empty 50 ml erlenmeyer flask which is on your lab bench? (± 0.01 g) Fill the 50 ml erlenmeyer flask about 3/4 full with your unknown liquid. b. What is the mass in grams (g) of the flask and unknown liquid? (± 0.01 g) c. What is the mass in grams (g) of the unknown liquid alone? (± 0.01 g) HINT: ((mass of liquid and flask) (mass of flask)) 3. Determining the volume of your unknown liquid. Use a small plastic funnel to carefully pour the unknown liquid into a 100 ml graduated cylinder. The volume is measured at the bottom of the curved upper surface. a. What is the volume of the unknown liquid? (± 0.1 ml) Pour the unknown liquid back into the unknown bottle. b. Calculate the weight in grams (g) of 1 milliliter (1 ml) of your unknown liquid? HINT: mass of liquid / volume of liquid (± 0.1 g/ml) What we have calculated in the previous step is the density of the unknown liquid. Density is defined as weight per unit volume. With liquids, density is usually reported as grams per milliliter (the number of grams in one milliliter). With solids, density is usually reported as grams per cubic centimeter (the number of grams in one cubic centimeter). mass m Density = = d = volume v The density is a characteristic property of any substance and can be used in the identification of that substance. In order to determine the identity of our unknown liquid, we must look up the known value for each of the densities of our four possible liquids. 4. Find the known value of densities in the Handbook of Chemistry and Physics. chloroform butanol (Methane, trichloro-) (1-butanol) carbon tetrachloride (Methane, tetrachloro-) water 2 Based upon these four known values and the value you calculated for your unknown liquid, what is the identity of the unknown liquid?
3 Experiment #3 - There is a sample of unknown metal on your desk. The city of Monmouth is cleaning up an old abandoned metallurgy factory. They find several crates containing unidentified cans filled with metal. In order to dispose of these metals properly, they must first be identified. Luckily they also found several shipping receipts so they know that the metal is Aluminum, Nickel, Lead, Tin (white), Cadmium, Bismuth, or Zinc. We will indirectly determine the density of these metals so the workers can properly dispose of them. The volume of an irregular solid is not easily determined by measuring its dimensions, but can be determined by the displacement of water. 1. What is the number of your unknown metal? 2. Determine the mass of your unknown metal. a. What is the mass of your unknown metal and container? (± 0.01 g) b. What is the mass of the container? (± 0.01 g) c. What is the mass of the unknown metal (m)? (± 0.01 g) Pour exactly 50.0 milliliters of water into your 100 ml graduated cylinder (note: 1 milliliter equals 1 cubic centimeter). Hold your graduated cylinder at an angle of 45 degrees. SLOWLY AND CAREFULLY add the metal to the water in the cylinder. Use ALL of your metal. Gently tap the graduated cylinder with your hand to displace any air bubbles which are attached to the pieces of metal. 3. Determining the density of your unknown metal. a. What is the volume of water that you put in your cylinder? (± 0.1 ml) b. What is the volume of the metal and the water combined? (± 0.1 ml) c. What is the measured volume of the metal alone (V)? (± 0.1 ml) d. What is the density (m / V) of your unknown metal? (± 0.01 g/ml) 4. Look up the known value of these densities in the Handbook of Chemistry and Physics. The metals listed on the shipping receipts are listed below. Aluminum Nickel Zinc Lead Cadmium Bismuth Tin (white) 5. Use your calculated density and the "literature" values to identify your unknown metal so the workers can properly dispose of it. a. Which metal is your unknown? TA checking unknown liquid and metal: Unknown Liquid Unknown Metal When you are finished, leave the metal pieces on a paper towel on top of your desk. 3
4 Post-Lab Calculations To be done before leaving lab for the day!!! The following calculations are all based on a cultural chemistry lab book. The length (L) of the book in centimeters is 21.6 cm (± 0.1 cm). The length (L) of the book in inches is 8.5 in (± 0.1 in). 1. How many centimeters are present in one inch? (± 0.01 cm) 2. How many centimeters (cm) are present in 7.7 inches? (± 0.1 cm) 3. How many meters (m) are present in 7.7 inches? (± m) 4. How many millimeters (mm) are present in 7.7 inches? (± 1 mm) 5. How many inches (in) are present in 27.9 centimeters? (± 0.1 in) 6. How many inches (in) are in 1.74 meters? (± 0.1 in) 7. How many inches (in) are in 28.4 millimeters? (± 0.01 in) 8. How many millimeters (mm) are in 1.39 meters (m)? (± 10 mm) 9. How many centimeters (cm) are in 1.83 meters (m)? (± 1 cm) 10. How many meters (m) are in 43 millimeters (mm)? (± m) 11. How many meters (m) are in 27 centimeters (cm)? (± 0.01 m) 4
5 Continuing with our calculations on the cultural chemistry book: The length (L) of the book in centimeters is 21.6 cm (± 0.1 cm). The width (W) of the book in centimeters is 15.5 cm (± 0.1 cm). The thickness (T) of the book in centimeters is 1.3 cm (± 0.1 cm). The mass (m) of the book in grams is g (± 0.1 g). 12. Calculate the volume of the book (L*W*T) cm 3 (± 1cm 3 ) 13. If 1 cubic centimeter is equal to 1 ml, what is the volume of the book in ml? (± 1 ml) 14. Calculate the volume of the book in liters (L) : (± L) 15. What is the weight of the book in kilograms? (± kg) 16. What is the weight of the book in milligrams (mg)? (± 100 mg) 17. What is the density (in grams per cm 3 ) of the paper used to print the book? (± 0.01 g/cm 3 ). 5
Experiment 1: The Densities of Liquids and Solids (from Masterson & Hurley)
 Experiment 1: The Densities of Liquids and Solids (from Masterson & Hurley) One of the fundamental properties of any sample of matter is its density, which is its mass per unit of volume. The density of
Experiment 1: The Densities of Liquids and Solids (from Masterson & Hurley) One of the fundamental properties of any sample of matter is its density, which is its mass per unit of volume. The density of
SIGNIFICANT FIGURES WORKSHEET
 SIGNIFICANT FIGURES WORKSHEET PART 1 - Determine the number of significant figures in the following numbers. 1.) 0.02 2.) 0.020 3.) 501 4.) 501.0 5.) 5,000 6.) 5,000. 7.) 6,051.00 8.) 0.0005 9.) 0.1020
SIGNIFICANT FIGURES WORKSHEET PART 1 - Determine the number of significant figures in the following numbers. 1.) 0.02 2.) 0.020 3.) 501 4.) 501.0 5.) 5,000 6.) 5,000. 7.) 6,051.00 8.) 0.0005 9.) 0.1020
DETERMINATION OF DENSITY
 DETERMINATION OF DENSITY Dr. Barbara B. Bunn. Adapted from Experiments in General Chemistry ; Wiley 1996J.G. Wardeska, T.T-S. Huang, R.W. Kopp; used by permission. Tested by Ms. Janice Orr s Chemistry
DETERMINATION OF DENSITY Dr. Barbara B. Bunn. Adapted from Experiments in General Chemistry ; Wiley 1996J.G. Wardeska, T.T-S. Huang, R.W. Kopp; used by permission. Tested by Ms. Janice Orr s Chemistry
CHAPTER 1. Conversions. General Plan for Converting Measurements. State the relationship between the unit given and the unit sought as an equality.
 CHAPTER 1 Conversions One of the aims of chemistry is to describe changes to tell what changed, how it changed, and what it changed into. Another aim of chemistry is to look at matter and its changes and
CHAPTER 1 Conversions One of the aims of chemistry is to describe changes to tell what changed, how it changed, and what it changed into. Another aim of chemistry is to look at matter and its changes and
Density What is density? How do you measure density?
 Density What is density? How do you measure density? What is density? Density describes how much mass is in a given volume of a material. Mass: the amount of matter in an object. Volume: the amount of
Density What is density? How do you measure density? What is density? Density describes how much mass is in a given volume of a material. Mass: the amount of matter in an object. Volume: the amount of
Introduction to Density
 Introduction to Density INTRODUCTION Which is heavier, a pound of aluminum or a pound of lead? The answer, of course, is neither, but many people confuse the words "heavy" and "dense". "Heavy" refers to
Introduction to Density INTRODUCTION Which is heavier, a pound of aluminum or a pound of lead? The answer, of course, is neither, but many people confuse the words "heavy" and "dense". "Heavy" refers to
Name Honors Chemistry / /
 Name Honors Chemistry / / SOL Questions Chapter 1 Each of the following questions below appeared on an SOL Chemistry Exam. For each of the following bubble in the correct answer on your scantron. 1. The
Name Honors Chemistry / / SOL Questions Chapter 1 Each of the following questions below appeared on an SOL Chemistry Exam. For each of the following bubble in the correct answer on your scantron. 1. The
Salinity in Seawater
 Salinity in Seawater Objective To familiarize students with the different methods used for measuring salinity of water. Introduction: Salinity exerts profound impacts on the marine environment. It controls
Salinity in Seawater Objective To familiarize students with the different methods used for measuring salinity of water. Introduction: Salinity exerts profound impacts on the marine environment. It controls
Density Answers to the End of module questions
 IDS 101 Density Answers to the End of module questions 1. You are experimenting with the mysterious substance X. You carefully measure the mass of a uniform 2 ml sample of pure substance X. You slice it
IDS 101 Density Answers to the End of module questions 1. You are experimenting with the mysterious substance X. You carefully measure the mass of a uniform 2 ml sample of pure substance X. You slice it
v = 5.00cm x 3.00cm x 2.00cm 2. What is the volume of a marble that has a mass of 3.0 g and a density of 2.7 g/ml? Formula (density) Substitution
 Name: STUDY GUIDE: MEASUREMENT, DENSITY, MASS, VOLUME, AND DIMENSIONAL ANALYSIS (2016) 1. A solid block is 5.00 cm high, 3.00 cm wide, and 2.00 cm long. It has a mass of 129 g. What is the density? Formula
Name: STUDY GUIDE: MEASUREMENT, DENSITY, MASS, VOLUME, AND DIMENSIONAL ANALYSIS (2016) 1. A solid block is 5.00 cm high, 3.00 cm wide, and 2.00 cm long. It has a mass of 129 g. What is the density? Formula
2.3 Density and Density Calculations
 2.3 Density and Density Calculations When people say that lead is heavier than wood, they do not mean that a pea sized piece of lead weighs more than a truckload of pine logs. What they mean is that a
2.3 Density and Density Calculations When people say that lead is heavier than wood, they do not mean that a pea sized piece of lead weighs more than a truckload of pine logs. What they mean is that a
2. Crystallization. A. Background
 2. Crystallization A. Background Crystallization is one of several available techniques available to purify organic compounds. Unlike other techniques, however, crystallization is specific to the purification
2. Crystallization A. Background Crystallization is one of several available techniques available to purify organic compounds. Unlike other techniques, however, crystallization is specific to the purification
Name Lab Section Date. Sediment Lab
 Name Lab Section Date. Investigating Stokes Law Sediment Lab ds = density of solid, g/cm dw = density of water, g/cm g = gravity, 980 cm/second 2 D = particle diameter in centimeters μ = molecular viscosity,
Name Lab Section Date. Investigating Stokes Law Sediment Lab ds = density of solid, g/cm dw = density of water, g/cm g = gravity, 980 cm/second 2 D = particle diameter in centimeters μ = molecular viscosity,
Experiment 2: Preparation of the Artificial Sweetener Dulcin
 Experiment 2: Preparation of the Artificial Sweetener Dulcin Organic compounds known as sugars are carbohydrates that occur widely in nature. For example, sucrose (aka table sugar) is found in sugar can,
Experiment 2: Preparation of the Artificial Sweetener Dulcin Organic compounds known as sugars are carbohydrates that occur widely in nature. For example, sucrose (aka table sugar) is found in sugar can,
This slide show has been prepared under fair use exemption of the U.S. Copyright Law and are restricted from further use.
 This slide show has been prepared under fair use exemption of the U.S. Copyright Law and are restricted from further use. Density & Dimensional Analysis Density - the volume which is occupied by a specific
This slide show has been prepared under fair use exemption of the U.S. Copyright Law and are restricted from further use. Density & Dimensional Analysis Density - the volume which is occupied by a specific
EXPERIMENT 3: Identification of a Substance by Physical Properties
 EXPERIMENT 3: Identification of a Substance by Physical Properties Materials: Hot plate Digital balance Capillary tubes (3) Thermometer Beakers (250 ml) Watch glass Graduated Cylinder (10 ml) Mel-Temp
EXPERIMENT 3: Identification of a Substance by Physical Properties Materials: Hot plate Digital balance Capillary tubes (3) Thermometer Beakers (250 ml) Watch glass Graduated Cylinder (10 ml) Mel-Temp
H N 2. Decolorizing carbon O. O Acetanilide
 Experiment 1: Recrystallization of Acetanilide Reading Assignment Mohrig 2 4 (Glassware, Reagents, & Heating) & 14 15 (Melting Point & Recrystallization) The purification of organic compounds is a tedious,
Experiment 1: Recrystallization of Acetanilide Reading Assignment Mohrig 2 4 (Glassware, Reagents, & Heating) & 14 15 (Melting Point & Recrystallization) The purification of organic compounds is a tedious,
TREATMENT PLANT OPERATOR TRAINEE Examination Study Guide
 TREATMENT PLANT OPERATOR TRAINEE Examination Study Guide Mobile County Personnel Board 1809 Government St., Mobile AL 36606 May, 2018 A calculator and this page will be made available to examinees when
TREATMENT PLANT OPERATOR TRAINEE Examination Study Guide Mobile County Personnel Board 1809 Government St., Mobile AL 36606 May, 2018 A calculator and this page will be made available to examinees when
EXPERIMENT. The Reaction of Magnesium with Hydrochloric Acid; The Molar Volume of Hydrogen
 EXPERIMENT The Reaction of Magnesium with Hydrochloric Acid; The Molar Volume of Hydrogen PURPOSE In this experiment you will determine the volume of the hydrogen gas which is produced when a sample of
EXPERIMENT The Reaction of Magnesium with Hydrochloric Acid; The Molar Volume of Hydrogen PURPOSE In this experiment you will determine the volume of the hydrogen gas which is produced when a sample of
There are 2 different types of numbers Exact -Measured
 Significant Figures When measuring or using our calculators we must determine the correct answer; our calculators are mindless drones and don t know the correct answer. There are 2 different types of numbers
Significant Figures When measuring or using our calculators we must determine the correct answer; our calculators are mindless drones and don t know the correct answer. There are 2 different types of numbers
Analysis of Calcium Carbonate Tablets
 Experiment 9 Analysis of Calcium Carbonate Tablets Prepared by Ross S. Nord, Eastern Michigan University PURPOSE To perform a gravimetric exercise to determine weight percent of active ingredient in a
Experiment 9 Analysis of Calcium Carbonate Tablets Prepared by Ross S. Nord, Eastern Michigan University PURPOSE To perform a gravimetric exercise to determine weight percent of active ingredient in a
Copper Odyssey. Chemical Reactions of Copper
 Name Lab Partner(s) Copper Odyssey Chemical Reactions of Copper Date Period Elemental copper metal will be converted into copper (II) ion and then brought through a series of compound conversions until
Name Lab Partner(s) Copper Odyssey Chemical Reactions of Copper Date Period Elemental copper metal will be converted into copper (II) ion and then brought through a series of compound conversions until
Experiment 2: The Chromatography of Organic Compounds
 Experiment 2: The Chromatography of Organic Compounds INTRODUCTION When performing an organic reaction, it is very common to observe the formation of other compounds in addition to your desired product;
Experiment 2: The Chromatography of Organic Compounds INTRODUCTION When performing an organic reaction, it is very common to observe the formation of other compounds in addition to your desired product;
ADDITIONAL PRACTICE WITH WRITING CONVERSION FACTORS
 ADDITIONAL PRACTICE WITH WRITING CONVERSION FACTORS Note: This worksheet is for additional practice. It is not due for a grade. Note: Unless specifically told otherwise, assume that percentage-by-mass
ADDITIONAL PRACTICE WITH WRITING CONVERSION FACTORS Note: This worksheet is for additional practice. It is not due for a grade. Note: Unless specifically told otherwise, assume that percentage-by-mass
TYPES OF CHEMICAL REACTIONS PART I INTRODUCTION
 EXPERIMENT 10 (2 Weeks) Chemistry 100 Laboratory TYPES OF CHEMICAL REACTIONS PART I INTRODUCTION It is useful to classify reactions into different types, because products of reactions can be predicted.
EXPERIMENT 10 (2 Weeks) Chemistry 100 Laboratory TYPES OF CHEMICAL REACTIONS PART I INTRODUCTION It is useful to classify reactions into different types, because products of reactions can be predicted.
Experiment 3: The Chromatography of Organic Compounds
 Experiment 3: The Chromatography of Organic Compounds INTRODUCTION Very often, in an organic synthesis, a reaction will proceed to produce multiple products or perhaps will only partially form the desired
Experiment 3: The Chromatography of Organic Compounds INTRODUCTION Very often, in an organic synthesis, a reaction will proceed to produce multiple products or perhaps will only partially form the desired
1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter.
 1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter. What is the mass of this object? A) 0.5 g B) 2 g C) 18 g D) 36 g Base your answers to questions 2 through
1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter. What is the mass of this object? A) 0.5 g B) 2 g C) 18 g D) 36 g Base your answers to questions 2 through
IDENTIFYING UNKNOWN SUBSTANCES
 IDENTIFYING UNKNOWN SUBSTANCES LAB 15 EXPERIMENT STUDENT BOOK Chapter 1, page 25 TOOLBOX Page 4 and 36 Goal Identify unknown substances with the help of different tests. 1. What is the independent variable
IDENTIFYING UNKNOWN SUBSTANCES LAB 15 EXPERIMENT STUDENT BOOK Chapter 1, page 25 TOOLBOX Page 4 and 36 Goal Identify unknown substances with the help of different tests. 1. What is the independent variable
GEL Hydrogeology (Groundwater) LAB 2: POROSITY & HYDRAULIC CONDUCTIVITY - Porosity Segment - Grade: /25
 GEL 4250 - Hydrogeology (Groundwater) LAB 2: POROSITY & HYDRAULIC CONDUCTIVITY - Porosity Segment - Name: Section: Grade: /25 COMPLETE & TURN IN ONLY PAGES THAT HAVE A FIELD FOR YOUR NAME. ALL OTHER PAGES
GEL 4250 - Hydrogeology (Groundwater) LAB 2: POROSITY & HYDRAULIC CONDUCTIVITY - Porosity Segment - Name: Section: Grade: /25 COMPLETE & TURN IN ONLY PAGES THAT HAVE A FIELD FOR YOUR NAME. ALL OTHER PAGES
The table below shows some of the properties of the elements cobalt and nickel.
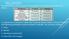 BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
Calculating Ballast By Troy Averill
 Calculating Ballast By Troy Averill Lesson Overview: Students use the dimensions of the cargo hold of a ship s hull to calculate the volume of material that can be contained. Students will then use the
Calculating Ballast By Troy Averill Lesson Overview: Students use the dimensions of the cargo hold of a ship s hull to calculate the volume of material that can be contained. Students will then use the
DETERMINATION of the EMPIRICAL FORMULA
 DETERMINATION of the EMPIRICAL FORMULA One of the fundamental statements of the atomic theory is that elements combine in simple whole number ratios. This observation gives support to the theory of atoms,
DETERMINATION of the EMPIRICAL FORMULA One of the fundamental statements of the atomic theory is that elements combine in simple whole number ratios. This observation gives support to the theory of atoms,
Determine whether the metal is magnesium, iron, or zinc based on the value of the calculated molar mass.
 Gases Part A: A student working at METAL Company found an unlabelled bottle of a metal in the lab. The metal could be magnesium, iron, or zinc. Each of these metals react with dilute hydrochloric acid
Gases Part A: A student working at METAL Company found an unlabelled bottle of a metal in the lab. The metal could be magnesium, iron, or zinc. Each of these metals react with dilute hydrochloric acid
INTRODUCTION TO ELECTROCHEMISTRY: CURRENT, VOLTAGE, & BATTERIES. Introduction. Electrochemistry Revised 4/28/14
 INTRODUCTION TO ELECTROCHEMISTRY: CURRENT, VOLTAGE, & BATTERIES Introduction Electrochemical Cells In this part of the experiment, four half cells are created by immersing metal strips of zinc, copper,
INTRODUCTION TO ELECTROCHEMISTRY: CURRENT, VOLTAGE, & BATTERIES Introduction Electrochemical Cells In this part of the experiment, four half cells are created by immersing metal strips of zinc, copper,
Soil Particle Density Protocol
 Soil Particle Density Protocol Purpose To measure the soil particle density of each horizon in a soil profile Overview Students weigh a sample of dry, sieved soil from a horizon, mix it with distilled
Soil Particle Density Protocol Purpose To measure the soil particle density of each horizon in a soil profile Overview Students weigh a sample of dry, sieved soil from a horizon, mix it with distilled
Forensics with TI-Nspire Technology
 Forensics with TI-Nspire Technology 2013 Texas Instruments Incorporated 1 education.ti.com Science Objectives Identify characteristics of different soils to demonstrate that a suspect has been at a scene.
Forensics with TI-Nspire Technology 2013 Texas Instruments Incorporated 1 education.ti.com Science Objectives Identify characteristics of different soils to demonstrate that a suspect has been at a scene.
Experiment 30A ENERGY CONTENT OF FUELS
 Experiment 30A ENERGY CONTENT OF FUELS FV 12/10/2012 MATERIALS: 12-oz. aluminum beverage can with top cut out and holes on side, thermometer, 100 ml graduated cylinder, 800 ml beaker, long-stem lighter,
Experiment 30A ENERGY CONTENT OF FUELS FV 12/10/2012 MATERIALS: 12-oz. aluminum beverage can with top cut out and holes on side, thermometer, 100 ml graduated cylinder, 800 ml beaker, long-stem lighter,
A TEACHER-DIRECTED, GUIDED DISCOVERY ACTIVITY FOR ADVANCED PHYSICAL SCIENCE STUDENTS
 A TEACHER-DIRECTED, GUIDED DISCOVERY ACTIVITY FOR ADVANCED PHYSICAL SCIENCE STUDENTS 1 2 FEATURES OF THIS ACTIVITY Problem solving Cooperative learning Requires student reasoning Requires student initiative
A TEACHER-DIRECTED, GUIDED DISCOVERY ACTIVITY FOR ADVANCED PHYSICAL SCIENCE STUDENTS 1 2 FEATURES OF THIS ACTIVITY Problem solving Cooperative learning Requires student reasoning Requires student initiative
D =? D = M = 10 g = 5 g/cm 3 M = 10 g V 2 cm 3 V = 2 cm 3 5 g/cm 3
 Using the formula for density, answer the following calculation problems. M D = (g/ml or g/cm 3 ) Density = Mass D = M M = (g) Volume V D V V = (ml or cm 3 ) For all of your problems, please complete them
Using the formula for density, answer the following calculation problems. M D = (g/ml or g/cm 3 ) Density = Mass D = M M = (g) Volume V D V V = (ml or cm 3 ) For all of your problems, please complete them
An Oxidation-Reduction Titration: The Reaction of Fe 2+ and Ce 4+
 An Oxidation-Reduction Titration: The Reaction of Fe 2+ and Ce 4+ LAB ADV COMP 8 From Advanced Chemistry with Vernier, Vernier Software & Technology, 2004 INTRODUCTION A titration, as you recall, is a
An Oxidation-Reduction Titration: The Reaction of Fe 2+ and Ce 4+ LAB ADV COMP 8 From Advanced Chemistry with Vernier, Vernier Software & Technology, 2004 INTRODUCTION A titration, as you recall, is a
Mon. Tues. Wed. Thurs. Fri. AM or PM B
 Name: (cf. Honesty Declaration Statement on page 20) Laboratory Day (circle) Lab Room Locker Lab. Session (circle) Lab. Section Mon. Tues. Wed. Thurs. Fri. AM or PM B Date experiment is performed MARK:
Name: (cf. Honesty Declaration Statement on page 20) Laboratory Day (circle) Lab Room Locker Lab. Session (circle) Lab. Section Mon. Tues. Wed. Thurs. Fri. AM or PM B Date experiment is performed MARK:
EXPERIMENT 6. Determination of the Ideal Gas Law Constant - R. Magnesium metal reacts with hydrochloric acid according to the following reaction,
 EXPERIMENT 6 Determination of the Ideal Gas Law Constant - R Magnesium metal reacts with hydrochloric acid according to the following reaction, Mg + 2 HCl MgCl 2 + H 2 (g) In this experiment you will use
EXPERIMENT 6 Determination of the Ideal Gas Law Constant - R Magnesium metal reacts with hydrochloric acid according to the following reaction, Mg + 2 HCl MgCl 2 + H 2 (g) In this experiment you will use
CONSERVATION OF MATTER AND CHEMICAL PROPERTIES
 1 CONSERVATION OF MATTER AND CHEMICAL PROPERTIES I. OBJECTIVES AND BACKGROUND The object of this experiment is to demonstrate the conservation of matter- or more particularly, the conservation of "atoms"
1 CONSERVATION OF MATTER AND CHEMICAL PROPERTIES I. OBJECTIVES AND BACKGROUND The object of this experiment is to demonstrate the conservation of matter- or more particularly, the conservation of "atoms"
Melting Point 1. Figure 2. A close-up of the "business end" of the Mel-Temp apparatus. Figure 1. The mel-temp device.
 Melting Point 1 The temperature at which a solid melts is known as the melting point (MP) of that substance. The melting point is a physical property of a solid and can be used to help identify a substance.
Melting Point 1 The temperature at which a solid melts is known as the melting point (MP) of that substance. The melting point is a physical property of a solid and can be used to help identify a substance.
GRAVIMETRIC DETERMINATION OF SULFATE IN AN UNKNOWN SOLUTION
 GRAVIMETRIC DETERMINATION OF SULFATE IN AN UNKNOWN SOLUTION AIM The main objective of this experiment is to determine the concentration of sulfate ion in an unknown solution by using gravimetry. INTRODUCTION
GRAVIMETRIC DETERMINATION OF SULFATE IN AN UNKNOWN SOLUTION AIM The main objective of this experiment is to determine the concentration of sulfate ion in an unknown solution by using gravimetry. INTRODUCTION
Lab #7H Photosynthesis and Respiration
 Lab #7H Photosynthesis and Respiration Plants make sugar, storing the energy of the sun into chemical energy, by the process of photosynthesis. When they require energy, they can tap the stored energy
Lab #7H Photosynthesis and Respiration Plants make sugar, storing the energy of the sun into chemical energy, by the process of photosynthesis. When they require energy, they can tap the stored energy
COC Biotechnology Program
 COC Biotechnology Program DNA FINGERPRINTING: VERSION C In the time it takes you to complete this lab, your DNA could be extracted, amplified, analyzed and compared. Everything from a criminal past to
COC Biotechnology Program DNA FINGERPRINTING: VERSION C In the time it takes you to complete this lab, your DNA could be extracted, amplified, analyzed and compared. Everything from a criminal past to
Chem 2115 Experiment #9. Consumer Chemistry: Determining the Iron Content in Supplements
 Chem 2115 Experiment #9 Consumer Chemistry: Determining the Iron Content in Supplements OBJECTIVE: The goal of this experiment is to use the quantitative technique of spectrophotometry to determine the
Chem 2115 Experiment #9 Consumer Chemistry: Determining the Iron Content in Supplements OBJECTIVE: The goal of this experiment is to use the quantitative technique of spectrophotometry to determine the
Recrystallization with a Single Solvent
 Experiment: Recrystallization Part II: Purification of Solids In Part I of the recrystallization experiment, you learned about the factors which make a good recrystallization solvent, and you learned how
Experiment: Recrystallization Part II: Purification of Solids In Part I of the recrystallization experiment, you learned about the factors which make a good recrystallization solvent, and you learned how
LABORATORY 3 SOIL ANALYSIS
 VEGETATION DESCRIPTION AND ANALYSIS 2017 LABORATORY 3 SOIL ANALYSIS OBJECTIVE This lab will obtain four key soil parameters from the samples collected from Shawnee Gowan s Grizzly Glacier project relevés.
VEGETATION DESCRIPTION AND ANALYSIS 2017 LABORATORY 3 SOIL ANALYSIS OBJECTIVE This lab will obtain four key soil parameters from the samples collected from Shawnee Gowan s Grizzly Glacier project relevés.
EXPERIMENT 5. Molecular Absorption Spectroscopy: Determination of Iron with 1,10-Phenanthroline
 EXPERIMENT 5 Molecular Absorption Spectroscopy: Determination of Iron with 1,10-Phenanthroline UNKNOWN Submit a clean, labeled 100-mL volumetric flask to the instructor so that your unknown iron solution
EXPERIMENT 5 Molecular Absorption Spectroscopy: Determination of Iron with 1,10-Phenanthroline UNKNOWN Submit a clean, labeled 100-mL volumetric flask to the instructor so that your unknown iron solution
To identify and classify various types of chemical reactions.
 Cycle of Copper Reactions Minneapolis Community and Technical College v.11.17 Objectives: To observe and document copper s chemical changes in five different reactions and verify that copper is conserved
Cycle of Copper Reactions Minneapolis Community and Technical College v.11.17 Objectives: To observe and document copper s chemical changes in five different reactions and verify that copper is conserved
The total amount in grams of solid material dissolved in 1 kg of seawater.
 MS20 Laboratory Objectives To understand the definition of salinity To learn the sources and sinks for salt in the ocean To measure the salinity of a water sample by mass evaporation To measure the salinity
MS20 Laboratory Objectives To understand the definition of salinity To learn the sources and sinks for salt in the ocean To measure the salinity of a water sample by mass evaporation To measure the salinity
ALTERNATIVE TRANSPORTATION FUELS LAB
 ALTERNATIVE TRANSPORTATION FUELS LAB Purpose: To examine the energy content of various alternative fuels which could be used for powering our vehicles in the future. As gasoline becomes increasingly expensive,
ALTERNATIVE TRANSPORTATION FUELS LAB Purpose: To examine the energy content of various alternative fuels which could be used for powering our vehicles in the future. As gasoline becomes increasingly expensive,
EXTRA CREDIT - EXPERIMENT G ELECTROCHEMISTRY ACTIVITY OF METALS
 EXTRA CREDIT - EXPERIMENT G ELECTROCHEMISTRY ACTIVITY OF METALS INTRODUCTION The objective of this experiment is to develop an abbreviated activity series of metals using: 1. Displacement reactions 2.
EXTRA CREDIT - EXPERIMENT G ELECTROCHEMISTRY ACTIVITY OF METALS INTRODUCTION The objective of this experiment is to develop an abbreviated activity series of metals using: 1. Displacement reactions 2.
Total Dissolved Solids
 Total Dissolved Solids LabQuest 12 INTRODUCTION Solids are found in streams in two forms, suspended and dissolved. Suspended solids include silt, stirred-up bottom sediment, decaying plant matter, or sewage-treatment
Total Dissolved Solids LabQuest 12 INTRODUCTION Solids are found in streams in two forms, suspended and dissolved. Suspended solids include silt, stirred-up bottom sediment, decaying plant matter, or sewage-treatment
Stream Ecology Lab Module 4/5 Determining Discharge and Sediment Yield in a Small Stream
 Stream Ecology Lab Module 4/5 Determining Discharge and Sediment Yield in a Small Stream Introduction: In this lab you will determine the total suspended solids yield per year for two streams a local stream
Stream Ecology Lab Module 4/5 Determining Discharge and Sediment Yield in a Small Stream Introduction: In this lab you will determine the total suspended solids yield per year for two streams a local stream
REINFORCED CONCRETE CULVERT STORM DRAIN AND SEWER PIPE FIELD SECTION 1026
 (Page 1 of 8) REINFORCED CONCRETE CULVERT STORM DRAIN AND SEWER PIPE FIELD SECTION 1026 1026.1 SCOPE. To establish procedures for the inspection of reinforced concrete pipe and the approval of modified
(Page 1 of 8) REINFORCED CONCRETE CULVERT STORM DRAIN AND SEWER PIPE FIELD SECTION 1026 1026.1 SCOPE. To establish procedures for the inspection of reinforced concrete pipe and the approval of modified
Deciding how to handle waste produced from manufacturing
 27 Reclaiming the Metal l a b o r at o ry Deciding how to handle waste produced from manufacturing is challenging. It can be diluted or incinerated, as you saw in previous activities. In some cases, it
27 Reclaiming the Metal l a b o r at o ry Deciding how to handle waste produced from manufacturing is challenging. It can be diluted or incinerated, as you saw in previous activities. In some cases, it
Copper Smelting by an Ancient Method
 Copper Smelting by an Ancient Method EXPERIMENT ## Prepared by Paul C. Smithson, Berea College, based on Yee et al., 004 Using beads of a copper-containing mineral, students will produce beads of nearly
Copper Smelting by an Ancient Method EXPERIMENT ## Prepared by Paul C. Smithson, Berea College, based on Yee et al., 004 Using beads of a copper-containing mineral, students will produce beads of nearly
Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3
 Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3 Directions to Students: Do not open this booklet until you are told to do so. Please respond to the following items by marking the best answer
Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3 Directions to Students: Do not open this booklet until you are told to do so. Please respond to the following items by marking the best answer
POLLUTANTS: HOW MUCH TOTAL OR HOW MUCH PER UNIT OF WATER?
 POLLUTANTS: HOW MUCH TOTAL OR HOW MUCH PER UNIT OF WATER? 9-12 OBJECTIVES The student will do the following: 1. Define the concept of concentration as different from an expression of total amount. 2. Explain
POLLUTANTS: HOW MUCH TOTAL OR HOW MUCH PER UNIT OF WATER? 9-12 OBJECTIVES The student will do the following: 1. Define the concept of concentration as different from an expression of total amount. 2. Explain
Bio EOC Topics for Living Things, Metric Measurement, Microscope and The Scientific Method
 Bio EOC Topics for Living Things, Metric Measurement, Microscope and The Scientific Method Laboratory Equipment o Identification of laboratory equipment; for example beaker, triple beam balance, microscope,
Bio EOC Topics for Living Things, Metric Measurement, Microscope and The Scientific Method Laboratory Equipment o Identification of laboratory equipment; for example beaker, triple beam balance, microscope,
The Release of Base Metals During Acidic Leaching of Fly Ash
 The Release of Base Metals During Acidic Leaching of Fly Ash George Kazonich and Ann G. Kim U.S. Department of Energy Federal Energy Technology Center P.O. Box 19 Pittsburgh, PA 153 ABSTRACT Since 199,
The Release of Base Metals During Acidic Leaching of Fly Ash George Kazonich and Ann G. Kim U.S. Department of Energy Federal Energy Technology Center P.O. Box 19 Pittsburgh, PA 153 ABSTRACT Since 199,
Significance of Water Observation Lab
 Significance of Water Observation Lab It has been said that the chemistry of life is water chemistry. Because of its chemical properties, water is the medium in which most of life s chemical reactions
Significance of Water Observation Lab It has been said that the chemistry of life is water chemistry. Because of its chemical properties, water is the medium in which most of life s chemical reactions
Field Name Data Type Notes. Text Required Text Required. Text Required Text Required. Required Number Required. Number Required.
 CSV Invoice Format Column Position 1 2 3 4 5 6 7 8 9 10 Campus* Field Name Data Type Notes Supplier * Invoice * Order * Invoice Date* Total Invoice Amount* Total Sales Tax Amount* Discount Amount Discount
CSV Invoice Format Column Position 1 2 3 4 5 6 7 8 9 10 Campus* Field Name Data Type Notes Supplier * Invoice * Order * Invoice Date* Total Invoice Amount* Total Sales Tax Amount* Discount Amount Discount
Gravimetric Analysis: Determination of % Sulfur in Fertilizer
 Gravimetric Analysis: Determination % Sulfur in Fertilizer This is another "real world" sample experiment in this case we will analyze a fertilizer sample for the sulfate content and express the result
Gravimetric Analysis: Determination % Sulfur in Fertilizer This is another "real world" sample experiment in this case we will analyze a fertilizer sample for the sulfate content and express the result
Name # Date Period! FINDING&VOLUME&AND&DENSITY& !!! !!!!!!!
 Name # Date Period FINDING&VOLUME&AND&DENSITY& TherectangularshapedboxinFigure1isfilledwithdrysand.Findthe volumeanddensityofthesand. 1.Firstfindthevolumeofthebox.Thisisalsothevolumeofthesand. Usethediagramtofindthedimensionsofthebox.
Name # Date Period FINDING&VOLUME&AND&DENSITY& TherectangularshapedboxinFigure1isfilledwithdrysand.Findthe volumeanddensityofthesand. 1.Firstfindthevolumeofthebox.Thisisalsothevolumeofthesand. Usethediagramtofindthedimensionsofthebox.
Measuring Manganese Concentration Using Spectrophotometry
 Measuring Manganese Concentration Using Spectrophotometry Objectives To use spectroscopy to determine the amount of Manganese is an unknown sample. Scenario Your have just joined a "Green Team" at the
Measuring Manganese Concentration Using Spectrophotometry Objectives To use spectroscopy to determine the amount of Manganese is an unknown sample. Scenario Your have just joined a "Green Team" at the
The entire document shall be read and understood before proceeding with a test. ISTA 3B Page 1 of 35
 Packaged-Products for Less-Than-Truckload (LTL) Shipment ISTA 3 Series General Simulation Performance Test PROCEDURE VERSION DATE Last TECHNICAL Change: MARCH 2017 Last EDITORIAL Change: MARCH 2017 For
Packaged-Products for Less-Than-Truckload (LTL) Shipment ISTA 3 Series General Simulation Performance Test PROCEDURE VERSION DATE Last TECHNICAL Change: MARCH 2017 Last EDITORIAL Change: MARCH 2017 For
An Insulated Cola Bottle
 An Insulated Cola Bottle Computer 12 Insulation slows the flow of heat. Glass, plastics such as Styrofoam, wool, fiber glass, aluminum foil, air, and a vacuum are some of the many materials used for heat
An Insulated Cola Bottle Computer 12 Insulation slows the flow of heat. Glass, plastics such as Styrofoam, wool, fiber glass, aluminum foil, air, and a vacuum are some of the many materials used for heat
esources / ^ Vocabulary Process Skill
 / ^ Vocabulary natural resource, C38 renewable resource, C40 nonrenewable resource, C41 pollution, C42 esources Why do miners dig deep into Earth? They are looking for useful materials, such as metals,
/ ^ Vocabulary natural resource, C38 renewable resource, C40 nonrenewable resource, C41 pollution, C42 esources Why do miners dig deep into Earth? They are looking for useful materials, such as metals,
Rev Experiment 10
 Experiment 10 SPECTROPHOTOMETRIC DETERMINATION OF IRON IN DRINKING WATER 2 lab periods Reading: 1) Chapter 17, pg 393-403, Quantitative Chemical Analysis, 8 h Edition, Daniel C. Harris (7 th Edition: Chapter
Experiment 10 SPECTROPHOTOMETRIC DETERMINATION OF IRON IN DRINKING WATER 2 lab periods Reading: 1) Chapter 17, pg 393-403, Quantitative Chemical Analysis, 8 h Edition, Daniel C. Harris (7 th Edition: Chapter
MATHEMATICS Y4 Measuring 4602 Units to estimate or measure length, mass or capacity. Equipment MathsGoGoGo
 MATHEMATICS Y4 Measuring 4602 Units to estimate or measure length, mass or capacity. Equipment Paper, pencil, ruler Various measuring instruments, balance scales Maths Go Go Go 4602 Units to estimate and
MATHEMATICS Y4 Measuring 4602 Units to estimate or measure length, mass or capacity. Equipment Paper, pencil, ruler Various measuring instruments, balance scales Maths Go Go Go 4602 Units to estimate and
Eutrophication: Too Much of a Good Thing?
 Name Class Date Skills Practice Lab DATASHEET FOR IN-TEXT LAB Eutrophication: Too Much of a Good Thing? Plants depend on nutrients such as phosphates and nitrates to survive. However, when people release
Name Class Date Skills Practice Lab DATASHEET FOR IN-TEXT LAB Eutrophication: Too Much of a Good Thing? Plants depend on nutrients such as phosphates and nitrates to survive. However, when people release
2. 47 mm grid marked, white sterile 0.45 micron membranes (Millipore or equivalent) 4. Vacuum pump capable of inches of vacuum
 Microbiological Methods IX-B- 1 PRESUMPTIVE MEMBRANE FILTER METHOD AND CONFIRMATION OF PSEUDOMONAS AERUGINOSA PRINCIPLE SCOPE Presumptive Pseudomonas bacteria are quantitated by a membrane filter technique,
Microbiological Methods IX-B- 1 PRESUMPTIVE MEMBRANE FILTER METHOD AND CONFIRMATION OF PSEUDOMONAS AERUGINOSA PRINCIPLE SCOPE Presumptive Pseudomonas bacteria are quantitated by a membrane filter technique,
By Authority Of THE UNITED STATES OF AMERICA Legally Binding Document
 By Authority Of THE UNITED STATES OF AMERICA Legally Binding Document By the Authority Vested By Part 5 of the United States Code 552(a) and Part 1 of the Code of Regulations 51 the attached document has
By Authority Of THE UNITED STATES OF AMERICA Legally Binding Document By the Authority Vested By Part 5 of the United States Code 552(a) and Part 1 of the Code of Regulations 51 the attached document has
OPERATIONAL MEMO GEN-14 Revision 3. November, Volatile Organic Compound (VOC) Sediment Sampling Method 5035 Field Sampling Procedure
 MICHIGAN DEPARTMENT OF ENVIRONMENTAL QUALITY INTEROFFICE COMMUNICATION OPERATIONAL MEMO GEN-14 TO: FROM: SUBJECT: All Waste Management Division Staff Jim Sygo, Chief, Waste Management Division Volatile
MICHIGAN DEPARTMENT OF ENVIRONMENTAL QUALITY INTEROFFICE COMMUNICATION OPERATIONAL MEMO GEN-14 TO: FROM: SUBJECT: All Waste Management Division Staff Jim Sygo, Chief, Waste Management Division Volatile
Preparation of Cyclohexene From Cyclohexanol
 EXPERIMENT 9 Alkene Synthesis From Alcohol Preparation of Cyclohexene From Cyclohexanol Purpose: a) Preparation of an alkene by dehydration (elimination of water) of an alcohol in the presence of an acid
EXPERIMENT 9 Alkene Synthesis From Alcohol Preparation of Cyclohexene From Cyclohexanol Purpose: a) Preparation of an alkene by dehydration (elimination of water) of an alcohol in the presence of an acid
As you saw in the last activity, a computer is made of many parts,
 23 Producing Circuit Boards R EA D I N G As you saw in the last activity, a computer is made of many parts, each manufactured from one or more materials. One essential part of a computer, and of many other
23 Producing Circuit Boards R EA D I N G As you saw in the last activity, a computer is made of many parts, each manufactured from one or more materials. One essential part of a computer, and of many other
Grade Six: Energy Lesson 6.1: Forms of Energy
 Grade Six: Energy Lesson 6.1: Forms of Energy Lesson Concept Energy can be in different forms. Link Lesson 6.1 Forms of Energy is an introduction to the many forms of energy. All forms of energy can do
Grade Six: Energy Lesson 6.1: Forms of Energy Lesson Concept Energy can be in different forms. Link Lesson 6.1 Forms of Energy is an introduction to the many forms of energy. All forms of energy can do
Engineering with Renewable Energy: Solar Water Pumping
 Engineering with Renewable Energy: Solar Water Pumping AUTHOR: Jamie Repasky DESCRIPTION: Students will learn that energy from a renewable resource can be converted to electrical energy to do work by engineering
Engineering with Renewable Energy: Solar Water Pumping AUTHOR: Jamie Repasky DESCRIPTION: Students will learn that energy from a renewable resource can be converted to electrical energy to do work by engineering
Experiment 13: Determination of Molecular Weight by Freezing Point Depression
 1 Experiment 13: Determination of Molecular Weight by Freezing Point Depression Objective: In this experiment, you will determine the molecular weight of a compound by measuring the freezing point of a
1 Experiment 13: Determination of Molecular Weight by Freezing Point Depression Objective: In this experiment, you will determine the molecular weight of a compound by measuring the freezing point of a
Methods: Record the date and time in the data book.
 Chemical Station Materials & Methods Materials: 3 Tall Plastic Vials with Caps 1 Small Glass Vial with Cap 1 Nitrate Tablet #1 1 ph Tablet 1 Nitrate Tablet #2 1 Phosphate Tablet 2 Dissolved Oxygen (DO)
Chemical Station Materials & Methods Materials: 3 Tall Plastic Vials with Caps 1 Small Glass Vial with Cap 1 Nitrate Tablet #1 1 ph Tablet 1 Nitrate Tablet #2 1 Phosphate Tablet 2 Dissolved Oxygen (DO)
Chem 355 Jasperse DISTILLATION
 Chem 355 Jasperse DISTILLATION 1 Background Distillation is a widely used technique for purifying liquids. The basic distillation process involves heating a liquid such that liquid molecules vaporize.
Chem 355 Jasperse DISTILLATION 1 Background Distillation is a widely used technique for purifying liquids. The basic distillation process involves heating a liquid such that liquid molecules vaporize.
Absolutely. Positively. Precise.
 1 Absolutely. Positively. Precise. Troemner s Philosophy Calibration & Traceability Philosophy Troemner is the world s largest independent mass calibration company. Our philosophy is to provide the highest
1 Absolutely. Positively. Precise. Troemner s Philosophy Calibration & Traceability Philosophy Troemner is the world s largest independent mass calibration company. Our philosophy is to provide the highest
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
Calcium and Magnesium; Chlorophosphonazo Rapid Liquid Method Method to 1000 µg/l Ca and Mg as CaCO 3 (ULR) Pour-Thru Cell
 Hardness, Total DOC316.53.01045 Calcium and Magnesium; Chlorophosphonazo Rapid Liquid Method Method 8374 4 to 1000 µg/l Ca and Mg as CaCO 3 (ULR) Pour-Thru Cell Scope and application: For boiler and ultrapure
Hardness, Total DOC316.53.01045 Calcium and Magnesium; Chlorophosphonazo Rapid Liquid Method Method 8374 4 to 1000 µg/l Ca and Mg as CaCO 3 (ULR) Pour-Thru Cell Scope and application: For boiler and ultrapure
The Planet s Lungs: Planting Giant Sequoias to Combat Carbon Dioxide
 The Planet s Lungs: Planting Giant Sequoias to Combat Carbon Dioxide By Matthew Auman Carbon dioxide concentration in the atmosphere is increasing. This is not controversial. Global warming has been cited
The Planet s Lungs: Planting Giant Sequoias to Combat Carbon Dioxide By Matthew Auman Carbon dioxide concentration in the atmosphere is increasing. This is not controversial. Global warming has been cited
Materials. Materials. NOTE Delta Education Customer Service can be reached at
 w Weather and Water Measuring Matter Materials Materials Contents Kit Inventory List... 42 Materials Supplied by the Teacher... 45 Preparing the Kit for Your Classroom... 47 Care, Reuse, and Recycling...
w Weather and Water Measuring Matter Materials Materials Contents Kit Inventory List... 42 Materials Supplied by the Teacher... 45 Preparing the Kit for Your Classroom... 47 Care, Reuse, and Recycling...
Module 2, Add on Lesson Turbidity Sensor. Student. 90 minutes
 Module 2, Add on Lesson Turbidity Sensor Student 90 minutes Purpose Construct a sensor to measure the turbidity of water Graph data and reason about curves and linear relationships Calibrate the turbidity
Module 2, Add on Lesson Turbidity Sensor Student 90 minutes Purpose Construct a sensor to measure the turbidity of water Graph data and reason about curves and linear relationships Calibrate the turbidity
Evaluation copy. Total Dissolved Solids. Computer INTRODUCTION
 Total Dissolved Solids Computer 12 INTRODUCTION Solids are found in streams in two forms, suspended and dissolved. Suspended solids include silt, stirred-up bottom sediment, decaying plant matter, or sewage-treatment
Total Dissolved Solids Computer 12 INTRODUCTION Solids are found in streams in two forms, suspended and dissolved. Suspended solids include silt, stirred-up bottom sediment, decaying plant matter, or sewage-treatment
SOP-C-130 Determination of Total Solids and Percent Solids
 i. Identification of the method a. SM 2540 G & B (approved 2011) ii. Applicable matrix or matrices a. Nonpotable water (Not TNI accredited) b. Soils and other solids (not TNI accredited) iii. Limits of
i. Identification of the method a. SM 2540 G & B (approved 2011) ii. Applicable matrix or matrices a. Nonpotable water (Not TNI accredited) b. Soils and other solids (not TNI accredited) iii. Limits of
Methods for determination of mercury in LP gas
 (JLPGA-S-07) Methods for determination of mercury in LP gas 1. Scope This standard specifies the methods for determination of gaseous mercury in vaporized liquid liquefied petroleum (LP) gas. Remarks 1
(JLPGA-S-07) Methods for determination of mercury in LP gas 1. Scope This standard specifies the methods for determination of gaseous mercury in vaporized liquid liquefied petroleum (LP) gas. Remarks 1
, to form the products magnesium oxide, MgO(s), and magnesium nitride, Mg 3
 Active Metals in the Periodic Table Periodic Trends and the Properties of the Elements SCIENTIFIC Introduction Like good experiments, good demonstrations often lead to other demonstrations. What follows
Active Metals in the Periodic Table Periodic Trends and the Properties of the Elements SCIENTIFIC Introduction Like good experiments, good demonstrations often lead to other demonstrations. What follows
LAB 6: Agarose Gel Electrophoresis of Restriction Digested Plasmid DNA
 LAB 6: Agarose Gel Electrophoresis of Restriction Digested Plasmid DNA I. Objectives The purpose of today s lab is to learn how to set up and run an agarose gel, separate DNA fragments on the gel, and
LAB 6: Agarose Gel Electrophoresis of Restriction Digested Plasmid DNA I. Objectives The purpose of today s lab is to learn how to set up and run an agarose gel, separate DNA fragments on the gel, and
PROJECT NO.: X
 February 23, 2011 REPORT NO.: 1012045 PROJECT NO.: 206-1 X Please find enclosed the analytical report, including the Sample Summary, Sample Narrative and Chain of Custody for your sample set received November
February 23, 2011 REPORT NO.: 1012045 PROJECT NO.: 206-1 X Please find enclosed the analytical report, including the Sample Summary, Sample Narrative and Chain of Custody for your sample set received November
Commonwealth of Pennsylvania PA Test Method No. 507 Department of Transportation October Pages LABORATORY TESTING SECTION. Method of Test for
 Commonwealth of Pennsylvania PA Test Method No. 507 Department of Transportation 6 Pages 1. SCOPE LABORATORY TESTING SECTION Method of Test for BULK SPECIFIC GRAVITY (SATURATED SURFACE-DRY BASIS), ABSORPTION,
Commonwealth of Pennsylvania PA Test Method No. 507 Department of Transportation 6 Pages 1. SCOPE LABORATORY TESTING SECTION Method of Test for BULK SPECIFIC GRAVITY (SATURATED SURFACE-DRY BASIS), ABSORPTION,
NATIONAL STANDARD OF THE PEOPLE S REPUBLIC OF CHINA (Zhenghua Renmin Gongheguo Guojia Biaozhun)
 1/9 GB NATIONAL STANDARD OF THE PEOPLE S REPUBLIC OF CHINA (Zhenghua Renmin Gongheguo Guojia Biaozhun) GB 5009.15-2014 FOOD SAFETY NATIONAL STANDARD DETERMINATION OF CADMIUM IN FOODS Issued 28 January
1/9 GB NATIONAL STANDARD OF THE PEOPLE S REPUBLIC OF CHINA (Zhenghua Renmin Gongheguo Guojia Biaozhun) GB 5009.15-2014 FOOD SAFETY NATIONAL STANDARD DETERMINATION OF CADMIUM IN FOODS Issued 28 January
Chapter 1.6. Polished Single-Crystal Silicon, Prime Wafers (all numbers nominal) Wafer Specification Table. Diameter 100 mm 4-inch 150 mm 6-inch
 Chapter 1.6 I - Substrate Specifications Polished Single-Crystal Silicon, Prime Wafers (all numbers nominal) Wafer Specification Table Diameter 100 mm 4-inch 150 mm 6-inch Thickness 525 µm 20.5 mils 675
Chapter 1.6 I - Substrate Specifications Polished Single-Crystal Silicon, Prime Wafers (all numbers nominal) Wafer Specification Table Diameter 100 mm 4-inch 150 mm 6-inch Thickness 525 µm 20.5 mils 675
