CompuTherm LLC Thermodynamic Databases. PanTitanium. Thermodynamic Database for Titanium-Based Alloys. Al B C. Ni Nb N. Copyright CompuTherm LLC
|
|
|
- Estella McDowell
- 6 years ago
- Views:
Transcription
1 PanTitanium Thermodynamic Database for Titanium-Based Alloys V Zr Al B C Ta Cr Sn Si Ti Cu Fe O H Ni Nb N Mo Mn Copyright CompuTherm LLC 1
2 Components A total of 19 components are included in the database as listed here: Major alloying elements: Al, Cr, Cu, Fe, Mo, Nb, Ni, Sn, Ta, Ti, V and Zr Minor alloying elements: B, C, H, Mn, N, O and Si Suggested Composition Range The suggested composition range for each element is listed in Table It should be noted that this given composition range is rather conservative. It is derived from the chemistries of the multicomponent commercial alloys that have been used to validate the current database. In the subsystems, many of these elements can be applied to a much wider composition range. In fact, some subsystems are valid in the entire composition range as given in section Table 10.1: Suggested composition range Element Composition range (wt%) Ti Al,V 0-11 Mo,Nb,Ta, Zr 0-8 Cr,Sn 0-5 Cu, Fe,Ni 0-3 B, C, H, N, O, Mn, Si Phases A total of 161 phases are included in the current database, and Table 10.2 lists those that are important for commercial titanium alloys. Information on all the other phases may be displayed through TDB viewer of Pandat and can be found at 2
3 Table 10.2: Phase Information Name Lattice Size Constituent A15_Nb3Al (3)(1) (Cr,Nb,Si,Ti)(Al,Cr,Nb,Si) Bcc (1)(3) (Al,Cr,Cu,Fe,Mo,Nb,Ni,Si,Sn,Ta,Ti,V,Zr) (B,C,H,N,O,Va) Cr3Si_A15 (3)(1)(3) (Cr,Nb,Si,Ti)(Cr,Nb,Si)(Va) DO19_Ti3Al (0.75)(0.25)(0.5) (Al,Cr,Mo,Nb,Sn,Ta,Ti,V,Zr) (Al,Cr,Mo,Nb,Si,Sn,Ta,Ti,V,Zr)(O,Va) DO22_TiAl3 (3)(1) (Al,Mo,Ti,V)(Cr,Mo,Nb,Ta,Ti,V) Diamond_A4 (1) Fcc (1)(1) Hcp (1)(0.5) L10_TiAl (1)(1) Laves_C14 (2)(1) (Al,C,Si,Sn,Ti) (Al,Cr,Cu,Fe,Mo,Nb,Ni,Si,Sn,Ta,Ti,V,Zr) (B,C,H,N,O,Va) (Al,Cr,Cu,Fe,Mo,Nb,Ni,Si,Sn,Ta,Ti,V,Zr) (B,C,H,N,O,Va) (Al,Cr,Mo,Nb,Si,Ta,Ti,V) (Al,Cr,Mo,Nb,Ta,Ti,V) (Al,Cr,Fe,Mo,Nb,Si,Ta,Ti,Zr) (Al,Cr,Fe,Mo,Nb,Ta,Ti,Zr) Laves_C15 (2)(1) (Al,Cr,Si,Ti,Zr)(Al,Cr,Si,Ti,Zr) Laves_C36 (2)(1) (Al,Cr,Ni,Zr)(Al,Cr,Ni,Zr) Liquid (1) (Al,B,C,Cr,Cu,Fe,H,Mo,N,Nb,Ni,O,Si,Sn,Ta,Ti,V,Zr) Sigma (8)(4)(18) (Al,Fe,Ni)(Cr,Mo,Nb,Ta,Ti,V) (Al,Cr,Fe,Mo,Nb,Ni,Ta,Ti,V) Sub-System Information The composition limits given in Table 10.1 are for multicomponent commercial titanium alloys in general. Complete and partial thermodynamic descriptions are developed for many binary systems as listed in Table
4 This means the current database works in a much wider composition range in many sub-systems. Table 10.3 lists all the binaries in the 18-component system. Thermodynamic descriptions are fully developed for the binaries in green color, which means that there is no composition limits if calculations are carried out for these binary systems. Only major phases are considered for the binaries in yellow color. For these binaries, phase relationships are correct in the areas near the major phases. Partial of the system is modeled for the binaries in red color as indicated by the number in front of one of the two elements. For example, Al-5B indicates that this system is modeled from pure Al to 5wt% of B. No model parameters are developed for those binaries in white color. In addition to the binaries, thermodynamic description for the key ternary Ti-Al-V system is also developed. : Full description : Full description for major phases : Partial description for certain composition range : Extrapolation Table 10.3: Current Status of Key Binary Systems C Cr Fe Mo Nb Ni Si Sn Ta Ti V Zr Al Al-C Al-Cr Al-Fe Al-Mo Al-Nb Al-Ni Al-Si Al-Sn Al-Ta Al-Ti Al-V Al-Zr C 5C-Cr C-Fe C-Mo C-Nb C-Ni C-Si C-Sn C-Ta C-Ti C-V C-Zr Cr Cr-Fe Cr-Mo Cr-Nb Cr-Ni Cr-Si Cr-Sn Cr-Ta Cr-Ti Cr-V Cr-Zr Cu 2Cu-Fe Cu-Mo Cu-Nb Cu-Ni Cu-Si Cu-Sn Cu-Ta Cu-Ti Cu-V Cu-Zr Fe Fe-Mo Fe-Nb Fe-Ni Fe-5Si Fe-Sn Fe-Ta Fe-Ti Fe-V Fe-Zr Mo Mo-Nb Mo-Ni Mo-8Si Mo-Sn Mo-Ta Mo-Ti Mo-V Mo-Zr N N-Nb N-Ni N-Si N-Sn N-Ta N-Ti N-V N-Zr Nb Nb-Ni Nb-5Si Nb-Sn Nb-Ta Nb-Ti Nb-V Nb-Zr Ni Ni-Si Ni-Sn Ni-Ta Ni-Ti Ni-V Ni-Zr Si Si-Sn 5Si-Ta Si-Ti 25Si-V 3Si-Zr Sn Sn-Ta Sn-Ti Sn-V Sn-Zr Ta Ta-Ti Ta-V Ta-Zr Ti Ti-V Ti-Zr V V-Zr 4
5 Database Validation Since this database has been designed for use with conventional - types of titanium alloys, it has been focused at the Ti-rich corner. This database has been tested by a large number of - type of titanium alloys, such as Ti64, Ti6242 and Ti6246. Table 10.4 lists the alloys and references used for validating the current database. The suggested composition ranges given in Table 10.1 are based on the compositions of these testing alloys. Users need to be careful while using the database beyond the suggested ranges. Table 10.4: Experimental Data Used for Testing the Current Titanium Database Alloy Experimental Information References transus, approach curve, partitioning [1966Cas,1979Las, Ti64 of Al and V in and. 1986Kah,1986Ro, 1991Lee,2003Fur, 2003Sem,2003Ven] Ti-144A transus, approach curve, and/or [2003Ven] partition coefficient Ti-155A transus, approach curve, and/or [2003Ven] partition coefficient Ti-6246 transus [2003Fur] Ti-6242 transus [2003Fur] IMI 834 transus [2003Fur] Ti-17 transus [2003Fur] Ti transus [2003Fur] Ti transus [2003Fur] Ti transus [2003Fur] Ti-6Al-2Nb-1Ta- transus [1984Lin] 0.8Mo Corona X approach curve [2003Boy] Ti-4.5Al-5Mo- transus [1984Yod] 1.5Cr(Corona 5) Ti transus, approach curve [1980Due] IMI 550 transus, approach curve, partitioning [2001Kha], +, and alloys listed in the handbook of Al, Mo, Sn and Si in and. transus [1994Boy] 5
6 This database can be used to calculate phase equilibria for multicomponent alloys, such as equilibrium between and. It can be used to predict phase transformation temperatures, such as -transus. The fraction of each phase as a function of temperature, partitioning of components in different phases can also be calculated. In addition to equilibrium calculations, Scheil simulations can also be carried out using this database. Some calculated examples are given below. Beta transus, the temperature at which starts to form from, is an important reference parameter in the selection of processing conditions, such as heat treatment process, for the conventional - type of titanium alloys. This temperature has been calculated for a large number of Ti64 and other titanium alloys using PanTitanium. Figure 10.1 shows a comparison between the predicted and observed beta transus temperatures for more than 150 Ti64 heats, reasonable agreement is obtained. The accuracy of the prediction depends on the reliability of the database and the accuracy of the input chemistry of the alloy. The calculated beta transus temperature is found to be very sensitive to the amount of the interstitial elements, such as C, H, N, and O. It is seen from Figure 10.1 that the predicted beta transus temperatures are in general higher than the observed ones. This can be explained by the fact that the calculated beta transus temperature corresponds to the temperature at which just starts to form, while its amount is 0%. It is very difficult to catch this exact temperature by experiments, and normally the measured temperature may corresponds to which 1~2% of phase has formed. Taking this factor into consideration, the transformation temperatures corresponding to 2% of phase are calculated for the same alloys and are compared with observed values as shown in Figure It is seen that better agreement is obtained. 6
7 Figure 10.1: Comparison between predicted and observed beta transus for more than 150 Ti64 heats. The calculated transformation temperatures correspond to 0% of phase formed and the experimental data are from [1966Cas, 2003Sem, 2003Fur]. Calculated ( o C), 2%HCP Castro 02Semiatin Furrer_1 Furrer_2 Furrer_3 Furrer_4 Furrer_ Measured ( o C) Figure 10.2: Comparison between predicted and observed beta transus for more than 150 Ti64 heats. The calculated transformation temperatures correspond to 2% of phase formed. The experimental data are from [1966Cas, 2003Sem, 2003Fur]. Figure 10.3 shows a similar comparison for other titanium alloys, including Ti662, Ti6242, Ti6246, Ti17 and so on, and reasonable agreement is 7
8 obtained. The agreement can be improved if the transformation temperatures corresponding to 2% of phase are used for comparison Calculated ( o C) Ti 6242 Ti IMI834 Ti-17 Ti Ti Measured ( o C) Figure 10.3: Comparison between predicted and observed beta transus for other titanium alloys. The calculated transformation temperatures correspond to 0% of phase formed. The relative amounts of and phases are critical in the determination of alloy properties for an - alloy. Beta approach curve, the volume fraction of beta phase as a function of temperature, is therefore important in the selection of final heat treatment temperature. Beta approach curves for two Ti64 heats are calculated as plotted in Figures 10.4 and The experimental data [2003Sem, 1966Cas] are also plotted on the diagrams for comparison; very good agreements are obtained. It should point out that the calculated phase fractions are mole fractions, while the measured values are volume fractions. However, since the molar volume of the phase is very close to that of the phase, the error induced due to the direct comparison between them is small. This can be seen in 8
9 Figure 10.4 in which both the mole factions and the volume fractions of phase are plotted Measured [03Sem] Calculated, mole Calculated, volume Fraction of Beta Phase Temperature [ o C] Figure 10.4: Beta approach curve for a Ti64 alloy with experimental data from [2003Sem] Measured [66Cas] Calculated Fraction of Beta Phase Temperature [ o C] Figure 10.5: Beta approach curve for a Ti64 alloy with experimental data from [1966Cas]. 9
10 In addition to Ti64, beta approach curves are also calculated for other titanium alloys. Figure 10.6 shows the beta approach curve for one Ti6242 alloy, and the experimental data are from Semiatin [2005Sem] Measured [05Sem] Calculated, mole Fraction of Beta Phase Temperature [ o C] Figure 10.6: Beta approach curve for a Ti-6242 alloy with experimental data from [2005Sem]. Equilibrium phase compositions are useful in understanding the partitioning of elements in different phases. These are calculated and compared with the experimental measurements for Ti64 and Ti6242 alloys. Examples are given in Figures 10.7 to Figure 10.7 shows the equilibrium compositions of Al and V in and for one Ti64 alloy. In general, the calculated equilibrium compositions agree with the experimental data very well. The calculated V concentrations in the phase are higher than the measurements at low temperatures. This is due to the fact that the grains were too small to allow an accurate analysis [1979Las]. Figures 10.8 and 10.9 show the equilibrium compositions of Al, Mo, and Ti in and for the Ti6242 alloy. 10
11 20 15 Al, Measured [03Sem] V, Measured [03Sem] Al, Calculated V, Calculated Compositions [wt%] Temperature [ o C] Figure 10.7: Equilibrium compositions of Al and V in the alpha and beta phases for a Ti64 alloy with the experimental data from [2003Sem] Compositions (wt %) Mo β Al α Al Alpha Al Beta Mo Alpha Mo Beta Pandat Al Beta Pandat Mo Beta Pandat Al Alpha Pandat Mo Alpha 5.0 Al β Mo α Temperature ( F) Figure 10.8: Equilibrium compositions of Al and Mo in the alpha and beta phases for a Ti6242 alloy with the experimental data from [2005Sem]. 11
12 90 85 Compositions [wt%] Ti Alpha Ti Beta Pandat Ti Alpha Pandat Ti Beta Temperature [ o C] Figure 10.9: Equilibrium compositions of Ti in the alpha and beta phases for a Ti6242 alloy with experimental data from [2005Sem]. Figures and show the calculated fractions for IMI550 and Corona-X alloys, respectively. Both calculations agree with experimental data very well. 12
13 1.0 Fraction of Alpha Phase Ti-IMI550 SEM [01Kha] Optical [01Kha] Calculated Temperature [ o C] Figure 10.10: Alpha Fraction curve for an IMI550 alloy with experimental data [2001Kha]. 0.8 Fraction of Alpha Phase Ti-Corona-X Exp [03Boy] Calculated Temperature [ o C] Figure 10.11: Alpha Fraction curve for a Corona-X alloy with experimental data [2001Kha]. 13
14 References [1966Cas] R. Castro and L. Seraphin, Mem. Sci. Rev. Met. 63 (1966) [1979Las] A.L. Lasalmonie and M.Loubradou, J. Mat. Sci. 14 (1979) [1980Due] T.W. Duerig, G.T. Terlinde and J.C. Williams, Met. Trans. A 11A (1980) [1984Lin] F. S. Lin, E. A. Starke, Jr., S. B. Chakrabortty and A. Gysler, Met. Trans. 15A (1984) [1984Yod] G.R. Yoder, F.H. Froes and D. Eylon, Met. Trans. 15A (1984) [1986Kah] A.I. Kahveci and G.E. Welsch, Scripta Met. 20 (1986) [1986Ro] Y. Ro, H. Onodera, and K. Ohno, Trans. Iron Steel Inst. Japan 26(4) (1986) [1991Lee] Y.T. Lee, M. Peters and G. Welsch, Met. Trans. 22A (1991) 709- [1994Boy] 714. R. Boyer, G. Welsch and E. W. Collings, Materials Properties Handbook: Titanium Alloys, ASM International, Materials Park, OH, [2001Kha] K.K. Kharia and H.J. Rack, Met. Trans. 32A (2001) [2003Boy] R. Boyer, Boeing, private communication, [2003Fur] D. Furrer, LADISH, private communication, [2003Sem] S.L. Semiatin, S.L. Knisley, P.N. Fagin, F. Zhang and D.R. Barker, Met. Trans. 34A (2003) [2003Ven] V. Venkatesh, TIMET, private communication, [2005Sem] S.L. Semiatin, AFRL, private communication,
CompuTherm LLC Thermodynamic Databases. PanMolybdenum. Thermodynamic database for multi-component Mo-rich alloys. Al B. Copyright CompuTherm LLC
 PanMolybdenum Thermodynamic database for multi-component Mo-rich alloys Zr Al B Si Ti Mo Cr Fe Re O Mn Hf Copyright CompuTherm LLC 1 Components Total of 12 components are included in the database as listed
PanMolybdenum Thermodynamic database for multi-component Mo-rich alloys Zr Al B Si Ti Mo Cr Fe Re O Mn Hf Copyright CompuTherm LLC 1 Components Total of 12 components are included in the database as listed
PanAluminum. Thermodynamic database for multi-component Aluminum-rich casting and wrought alloys. Copyright CompuTherm LLC B C Y.
 PanAluminum Thermodynamic database for multi-component Aluminum-rich casting and wrought alloys Zn Zr Ag B C Y Co V Cr Ti Cu Sr Sn Si Al Ge Fe Gd Sc Hf Sb Ni Nb Mn Mg Li Copyright CompuTherm LLC 1 Components
PanAluminum Thermodynamic database for multi-component Aluminum-rich casting and wrought alloys Zn Zr Ag B C Y Co V Cr Ti Cu Sr Sn Si Al Ge Fe Gd Sc Hf Sb Ni Nb Mn Mg Li Copyright CompuTherm LLC 1 Components
CompuTherm LLC Thermodynamic Databases. PanBMG. Thermodynamic Database for Bulk Metallic Glass BMG. Copyright CompuTherm LLC
 PanBMG Thermodynamic Database for Bulk Metallic Glass Al Zr Cu BMG Ti Ni Si Copyright CompuTherm LLC 1 Components The PanBMG thermodynamic database, including the six elements as the following, covers
PanBMG Thermodynamic Database for Bulk Metallic Glass Al Zr Cu BMG Ti Ni Si Copyright CompuTherm LLC 1 Components The PanBMG thermodynamic database, including the six elements as the following, covers
MICROSTRUCTURE CHARACTERIZATION AND IMAGING IN TITANIUM ALLOYS BY ATOMIC FORCE ACOUSTIC MICROSCOPY
 MICROSTRUCTURE CHARACTERIZATION AND IMAGING IN TITANIUM ALLOYS BY ATOMIC FORCE ACOUSTIC MICROSCOPY Sigrun HIRSEKORN, Jonas RABE, Ute RABE, FRAUNHOFER IZFP, Campus E3 1, 66123 Saarbrücken, Germany INTRODUCTION
MICROSTRUCTURE CHARACTERIZATION AND IMAGING IN TITANIUM ALLOYS BY ATOMIC FORCE ACOUSTIC MICROSCOPY Sigrun HIRSEKORN, Jonas RABE, Ute RABE, FRAUNHOFER IZFP, Campus E3 1, 66123 Saarbrücken, Germany INTRODUCTION
IMAGING OF MICROSTRUCTURAL FEATURES AND LOCAL STIFFNESS OF Α- AND Β-PHASES IN THE TITANIUM ALLOY TI- 6AL-4V BY ATOMIC FORCE ACOUSTIC MICROSCOPY
 IMAGING OF MICROSTRUCTURAL FEATURES AND LOCAL STIFFNESS OF Α- AND Β-PHASES IN THE TITANIUM ALLOY TI- 6AL-4V BY ATOMIC FORCE ACOUSTIC MICROSCOPY Jonas O. RABE, Ute RABE, Sigrun HIRSEKORN Fraunhofer Institute
IMAGING OF MICROSTRUCTURAL FEATURES AND LOCAL STIFFNESS OF Α- AND Β-PHASES IN THE TITANIUM ALLOY TI- 6AL-4V BY ATOMIC FORCE ACOUSTIC MICROSCOPY Jonas O. RABE, Ute RABE, Sigrun HIRSEKORN Fraunhofer Institute
Database. Sept , 2014, Aachen, Germany. Thermo-Calc Anwendertreffen
 Database Sept. 11-12, 2014, Aachen, Germany Thermo-Calc Anwendertreffen Thermodynamic and kinetic databases New Databases, June 2014 TCAL3 TCMG3 TCSLD2 TCSI1 TCNI7 MOBNI3 TCAL3.0 TCAL3.0 TCAL1.0 2011.05
Database Sept. 11-12, 2014, Aachen, Germany Thermo-Calc Anwendertreffen Thermodynamic and kinetic databases New Databases, June 2014 TCAL3 TCMG3 TCSLD2 TCSI1 TCNI7 MOBNI3 TCAL3.0 TCAL3.0 TCAL1.0 2011.05
ADAMIS. Alloy Database for Micro-Solders. Ag Al Sn. ADAMIS Solder. Materials Design Technology Co.,Ltd. 2-5 Odenmacho, Nihonbashi, Chuo-ku
 ADAMIS Alloy Database for Micro-Solders Zn Ag Al Sn Sb ADAMIS Solder Au Bi Pb Ni In Cu Materials Design Technology Co.,Ltd. 2-5 Odenmacho, Nihonbashi, Chuo-ku Tokyo 103-0011 JAPAN Copyright Materials Design
ADAMIS Alloy Database for Micro-Solders Zn Ag Al Sn Sb ADAMIS Solder Au Bi Pb Ni In Cu Materials Design Technology Co.,Ltd. 2-5 Odenmacho, Nihonbashi, Chuo-ku Tokyo 103-0011 JAPAN Copyright Materials Design
Titanium and titanium alloys. Josef Stráský
 Titanium and titanium alloys Josef Stráský Lecture 2: Fundamentals of Ti alloys Polymorphism Alpha phase Beta phase Pure titanium Titanium alloys alloys alloys alloys Phase transformation β α phase Hardening
Titanium and titanium alloys Josef Stráský Lecture 2: Fundamentals of Ti alloys Polymorphism Alpha phase Beta phase Pure titanium Titanium alloys alloys alloys alloys Phase transformation β α phase Hardening
HOT DEFORMATION BEHAVIOUR OF Ti-6Al-4V IN DIFFERENT STRAIN RATES
 HOT DEFORMATION BEHAVIOUR OF Ti-6Al-4V IN DIFFERENT STRAIN RATES M. Arulselvan, Dr. G. Ganesan Department of Manufacturing Engg. Department of Manufacturing Engg. Engineering Wing, DDE Faculty of Engg.
HOT DEFORMATION BEHAVIOUR OF Ti-6Al-4V IN DIFFERENT STRAIN RATES M. Arulselvan, Dr. G. Ganesan Department of Manufacturing Engg. Department of Manufacturing Engg. Engineering Wing, DDE Faculty of Engg.
Exploration of High-Entropy Alloys for Turbine Applications
 Exploration of High-Entropy Alloys for Turbine Applications Acknowledgment: "This material is based upon work supported by the Department of Energy under Award Number(s) DE-SC0013220. SBIR Program PHASE
Exploration of High-Entropy Alloys for Turbine Applications Acknowledgment: "This material is based upon work supported by the Department of Energy under Award Number(s) DE-SC0013220. SBIR Program PHASE
Calculation and application of liquidus projection
 RARE METALS Vol. 25, No. 5, Oct 2006, p. 532 Calculation and application of liquidus projection CHEN Shuanglin 1), CAO Weisheng 2), YANG Ying 1), ZHANG Fan 1), WU Kaisheng 1), DU Yong 3), and Y. Austin
RARE METALS Vol. 25, No. 5, Oct 2006, p. 532 Calculation and application of liquidus projection CHEN Shuanglin 1), CAO Weisheng 2), YANG Ying 1), ZHANG Fan 1), WU Kaisheng 1), DU Yong 3), and Y. Austin
THE REGULARITIES OF PHASE AND STRUCTURAL TRANSFORMATION IN BINARY TITANIUM ALLOYS WITH METALS OF IV VIII GROUPS OF THE PERIODIC TABLE
 DOI: 10.2478/v10077-008-0004-7 A.V. Dobromyslov Institute of Metal Physics, Ural Branch of Russian Academy of Sciences, Еkaterinburg, Russia THE REGULARITIES OF PHASE AND STRUCTURAL TRANSFORMATION IN BINARY
DOI: 10.2478/v10077-008-0004-7 A.V. Dobromyslov Institute of Metal Physics, Ural Branch of Russian Academy of Sciences, Еkaterinburg, Russia THE REGULARITIES OF PHASE AND STRUCTURAL TRANSFORMATION IN BINARY
COMPUTED PHASE EQUILIBRIA IN IRON-BASE TERNARY ALLOYS
 COMPUTED PHASE EQUILIBRIA IN IRON-BASE TERNARY ALLOYS by K.C. HARI KUMAR APPLIED MECHANICS DEPARTMENT Thesis submitted in fulfilment of the requirements of the Degree of DOCTOR OF PHILOSOPHY be vt, to
COMPUTED PHASE EQUILIBRIA IN IRON-BASE TERNARY ALLOYS by K.C. HARI KUMAR APPLIED MECHANICS DEPARTMENT Thesis submitted in fulfilment of the requirements of the Degree of DOCTOR OF PHILOSOPHY be vt, to
Recent Developments of Software and Databases
 Recent Developments of Software and Databases Aachen, Sept 1-2, 2016 Introduction 2015 Introduction 1000 Introduction 64 Introduction 46 Introduction Introduction 35 Outline - What we do About the company
Recent Developments of Software and Databases Aachen, Sept 1-2, 2016 Introduction 2015 Introduction 1000 Introduction 64 Introduction 46 Introduction Introduction 35 Outline - What we do About the company
Phase Composition of a CrMo 0.5 NbTa 0.5 TiZr High Entropy Alloy: Comparison of Experimental and Simulated Data
 Entropy 2013, 15, 3796-3809; doi:10.3390/e15093796 Article OPEN ACCESS entropy ISSN 1099-4300 www.mdpi.com/journal/entropy Phase Composition of a CrMo 0.5 NbTa 0.5 TiZr High Entropy Alloy: Comparison of
Entropy 2013, 15, 3796-3809; doi:10.3390/e15093796 Article OPEN ACCESS entropy ISSN 1099-4300 www.mdpi.com/journal/entropy Phase Composition of a CrMo 0.5 NbTa 0.5 TiZr High Entropy Alloy: Comparison of
Phase diagrams (cont.) and the Fe-C system
 Phase diagrams (cont.) and the Fe-C system Solidification: Pro-eutectic vs Eutectic Pro-eutectic solidification Ideal liquid, uniform distribution Solid Pb(Sn) () nucleates Solubility limit leads to Sn
Phase diagrams (cont.) and the Fe-C system Solidification: Pro-eutectic vs Eutectic Pro-eutectic solidification Ideal liquid, uniform distribution Solid Pb(Sn) () nucleates Solubility limit leads to Sn
Recrystallization textures in metals and alloys
 Recrystallization textures in metals and alloys Uniaxial deformation Aluminium wire F.C.C. Metals and alloys FCC wires retain deformation texture ([111]+[100]) upon recrystallisation Composition / Purity
Recrystallization textures in metals and alloys Uniaxial deformation Aluminium wire F.C.C. Metals and alloys FCC wires retain deformation texture ([111]+[100]) upon recrystallisation Composition / Purity
Chapter 2 Metallurgy and Microstructure
 Chapter 2 Metallurgy and Microstructure 2.1 The General Metallurgy of Titanium Alloys Unalloyed titanium has two allotropic forms. The low temperature form, a, exists as an hexagonal-close-packed (hcp)
Chapter 2 Metallurgy and Microstructure 2.1 The General Metallurgy of Titanium Alloys Unalloyed titanium has two allotropic forms. The low temperature form, a, exists as an hexagonal-close-packed (hcp)
the Phase Diagrams Today s Topics
 MME 291: Lecture 03 Introduction to the Phase Diagrams Prof. A.K.M.B. Rashid Department of MME BUET, Dhaka Today s Topics Concept of alloying Classification of alloys Introduction to the phase diagram
MME 291: Lecture 03 Introduction to the Phase Diagrams Prof. A.K.M.B. Rashid Department of MME BUET, Dhaka Today s Topics Concept of alloying Classification of alloys Introduction to the phase diagram
Solidification: basics and beyond Equilibrium and nonequilibrium solidification
 Solidification: basics and beyond Equilibrium and nonequilibrium solidification Prof. Dmitry Eskin BCAST NETCENG, 2015 1 Types of solidification reactions Equilibrium solidification 2 Gibbs phase rule
Solidification: basics and beyond Equilibrium and nonequilibrium solidification Prof. Dmitry Eskin BCAST NETCENG, 2015 1 Types of solidification reactions Equilibrium solidification 2 Gibbs phase rule
Titanium and titanium alloys. Josef Stráský
 Titanium and titanium alloys Josef Stráský Lecture 3: Technological aspects of Ti alloys Pure Ti metallurgy, properties and applications α+β alloys microstructures, metallurgy, heat treatment Ti-6Al-4V
Titanium and titanium alloys Josef Stráský Lecture 3: Technological aspects of Ti alloys Pure Ti metallurgy, properties and applications α+β alloys microstructures, metallurgy, heat treatment Ti-6Al-4V
Superplastic Forming Properties of TIMETAL 54M
 Superplastic Forming Properties of TIMETAL 54M October 2010 Yoji Kosaka and Phani Gudipati Henderson Technical Laboratory Titanium Metals Corporation 1 Outline Brief Introduction of TIMETAL 54M (Ti-54M)
Superplastic Forming Properties of TIMETAL 54M October 2010 Yoji Kosaka and Phani Gudipati Henderson Technical Laboratory Titanium Metals Corporation 1 Outline Brief Introduction of TIMETAL 54M (Ti-54M)
Thermo-Calc Graphical Mode Examples
 Thermo-Calc Graphical Mode Examples 1 1 Introduction These examples are for the graphical mode of Thermo-Calc; there is a separate set of examples for the console mode. The examples are stored without
Thermo-Calc Graphical Mode Examples 1 1 Introduction These examples are for the graphical mode of Thermo-Calc; there is a separate set of examples for the console mode. The examples are stored without
Modeling of Casting and Solidification Processes - MCSP 2017 Vol.14 No.5 September (b)
 https://doi.org/10.1007/s41230-017-7144-5 MCSP 2017 - Modeling of Casting and Solidification Processes Vol.14 No.5 September 2017 CHINA FOUNDRY Development of thermophysical calculator for stainless steel
https://doi.org/10.1007/s41230-017-7144-5 MCSP 2017 - Modeling of Casting and Solidification Processes Vol.14 No.5 September 2017 CHINA FOUNDRY Development of thermophysical calculator for stainless steel
Effects in Ductile Iron
 Summary of Element Effects in Ductile Iron Rick Gundlach Element Materials Technology Wixom Insert Company Logo Here DIS Annual Meeting, June 7, 2012 Muskegon, Michigan Types of Alloying Elements Substitutional
Summary of Element Effects in Ductile Iron Rick Gundlach Element Materials Technology Wixom Insert Company Logo Here DIS Annual Meeting, June 7, 2012 Muskegon, Michigan Types of Alloying Elements Substitutional
Introduction to the phase diagram Uses and limitations of phase diagrams Classification of phase diagrams Construction of phase diagrams
 Prof. A.K.M.B. Rashid Department of MME BUET, Dhaka Concept of alloying Classification of alloys Introduction to the phase diagram Uses and limitations of phase diagrams Classification of phase diagrams
Prof. A.K.M.B. Rashid Department of MME BUET, Dhaka Concept of alloying Classification of alloys Introduction to the phase diagram Uses and limitations of phase diagrams Classification of phase diagrams
www-materials.eng.cam.ac.uk/typd
 Part IB Paper 3: MATERIALS Examples Paper 3/1: TEACH YOURSELF PHASE DIAGRAMS This examples paper forms part of an interactive online resource Teach Yourself Phase Diagrams which is available at: www-materials.eng.cam.ac.uk/typd
Part IB Paper 3: MATERIALS Examples Paper 3/1: TEACH YOURSELF PHASE DIAGRAMS This examples paper forms part of an interactive online resource Teach Yourself Phase Diagrams which is available at: www-materials.eng.cam.ac.uk/typd
Isothermal Aging Behavior of Beta Titanium Manganese Alloys
 Materials Transactions, Vol. 5, No. 12 (29) pp. 2737 to 2743 Special Issue on Low Cost Reduction Processes, Roles of Low Cost Elements and Interstitial Elements, and Microstructural Control for Generalization
Materials Transactions, Vol. 5, No. 12 (29) pp. 2737 to 2743 Special Issue on Low Cost Reduction Processes, Roles of Low Cost Elements and Interstitial Elements, and Microstructural Control for Generalization
Ab-Initio Molecular Dynamics Modeling of Molten Superalloys
 Ab-Initio Molecular Dynamics Modeling of Molten Superalloys Mark Asta Department of Chemical Engineering & Materials Science ES09 Workshop University of California, Davis June 25, 2009 Acknowledgements
Ab-Initio Molecular Dynamics Modeling of Molten Superalloys Mark Asta Department of Chemical Engineering & Materials Science ES09 Workshop University of California, Davis June 25, 2009 Acknowledgements
Microstructural effects and kinetics of high temperature oxidation in Nb-Si base alloys
 Microstructural effects and kinetics of high temperature oxidation in Nb-Si base alloys E. Sarath K Menon, Triplicane A. Parthasarathy and Madan G. Mendiratta UES Inc., 4404 Dayton-Xenia Road, Dayton,
Microstructural effects and kinetics of high temperature oxidation in Nb-Si base alloys E. Sarath K Menon, Triplicane A. Parthasarathy and Madan G. Mendiratta UES Inc., 4404 Dayton-Xenia Road, Dayton,
Thermo-Calc Software. Thermo-Calc User Seminar CALCULATING THERMODYNAMIC PROPERTIES. New versions of Databases. Aachen, September 11th, 2008
 CALCULATING THERMODYNAMIC PROPERTIES Thermo-Calc User Seminar New versions of Databases Aachen, September 11th, 2008 http://www.thermocalc.com Phone: +46 8 4 99 30 E-mail: info@thermocalc.se Fax: +46 8
CALCULATING THERMODYNAMIC PROPERTIES Thermo-Calc User Seminar New versions of Databases Aachen, September 11th, 2008 http://www.thermocalc.com Phone: +46 8 4 99 30 E-mail: info@thermocalc.se Fax: +46 8
3.40 Sketch within a cubic unit cell the following planes: (a) (01 1 ) (b) (112 ) (c) (102 ) (d) (13 1) Solution
 3.40 Sketch within a cubic unit cell the following planes: (a) (01 1 ) (b) (11 ) (c) (10 ) (d) (13 1) The planes called for are plotted in the cubic unit cells shown below. 3.41 Determine the Miller indices
3.40 Sketch within a cubic unit cell the following planes: (a) (01 1 ) (b) (11 ) (c) (10 ) (d) (13 1) The planes called for are plotted in the cubic unit cells shown below. 3.41 Determine the Miller indices
Metallic Materials-Phase Diagrams
 Engineering Alloys Metals and alloys have many useful engineering properties and so have wide spread application in engineering designs. Iron and its alloys (principally steel) account for about 90 percent
Engineering Alloys Metals and alloys have many useful engineering properties and so have wide spread application in engineering designs. Iron and its alloys (principally steel) account for about 90 percent
Phase Diagrams, Solid Solutions, Phase Strengthening, Phase Transformations
 Phase Diagrams, Solid Solutions, Phase Strengthening, Phase Transformations Components and Phases Components: The elements or compounds that are mixed initially (Al and Cu). Phases: A phase is a homogenous,
Phase Diagrams, Solid Solutions, Phase Strengthening, Phase Transformations Components and Phases Components: The elements or compounds that are mixed initially (Al and Cu). Phases: A phase is a homogenous,
THE INFLUENCE OF Nb AND C ON THE SOLIDIFICATION MICROSTRUCTURES OF Fe-Ni-Cr ALLOYS
 Pergamon Scripta Materialia, Vol. 41, No. 4, pp. 449 454, 1999 Elsevier Science Ltd Copyright 1999 Acta Metallurgica Inc. Printed in the USA. All rights reserved. 1359-6462/99/$ see front matter PII S1359-6462(99)00102-5
Pergamon Scripta Materialia, Vol. 41, No. 4, pp. 449 454, 1999 Elsevier Science Ltd Copyright 1999 Acta Metallurgica Inc. Printed in the USA. All rights reserved. 1359-6462/99/$ see front matter PII S1359-6462(99)00102-5
PRE-SHOW REPORT FEATURING:
 MARCH 11 15, 2018 #TMSAnnualMeeting PHOENIX CONVENTION CENTER PHOENIX, ARIZONA, USA FEATURING: ALL-CONFERENCE PLENARY SESSION WITH TESLA AND SPACEX LEADERSHIP 2ND INTERNATIONAL CONFERENCE ON COMPUTATIONAL
MARCH 11 15, 2018 #TMSAnnualMeeting PHOENIX CONVENTION CENTER PHOENIX, ARIZONA, USA FEATURING: ALL-CONFERENCE PLENARY SESSION WITH TESLA AND SPACEX LEADERSHIP 2ND INTERNATIONAL CONFERENCE ON COMPUTATIONAL
Chapter 11: Phase Diagrams
 Chapter 11: Phase Diagrams ISSUES TO ADDRESS... When we combine two elements... what is the resulting equilibrium state? In particular, if we specify... -- the composition (e.g., wt% Cu - wt% Ni), and
Chapter 11: Phase Diagrams ISSUES TO ADDRESS... When we combine two elements... what is the resulting equilibrium state? In particular, if we specify... -- the composition (e.g., wt% Cu - wt% Ni), and
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION This file contains Supplementary information to the manuscript titled Accelerated Exploration of Multi-Principal Element Alloys with Solid Solution Phases, by O.N. Senkov, J.
SUPPLEMENTARY INFORMATION This file contains Supplementary information to the manuscript titled Accelerated Exploration of Multi-Principal Element Alloys with Solid Solution Phases, by O.N. Senkov, J.
EFFECT OF SHOT PEENING ON FATIGUE PERFORMANCE OF TWO TITANIUM ALLOYS. H. Bae, B. Flinn, M. Ramulu, G. Weber* and H. Diep*
 EFFECT OF SHOT PEENING ON FATIGUE PERFORMANCE OF TWO TITANIUM ALLOYS ABSTRACT H. Bae, B. Flinn, M. Ramulu, G. Weber* and H. Diep* University of Washington, Seattle WA, USA * The Boeing Company, Seattle
EFFECT OF SHOT PEENING ON FATIGUE PERFORMANCE OF TWO TITANIUM ALLOYS ABSTRACT H. Bae, B. Flinn, M. Ramulu, G. Weber* and H. Diep* University of Washington, Seattle WA, USA * The Boeing Company, Seattle
Chapter 9 Phase Diagrams. Dr. Feras Fraige
 Chapter 9 Phase Diagrams Dr. Feras Fraige Chapter Outline Definitions and basic concepts Phases and microstructure Binary isomorphous systems (complete solid solubility) Binary eutectic systems (limited
Chapter 9 Phase Diagrams Dr. Feras Fraige Chapter Outline Definitions and basic concepts Phases and microstructure Binary isomorphous systems (complete solid solubility) Binary eutectic systems (limited
TCFE6: TCS Steel and Fe-alloys Database
 TCFE6: TCS Steel and Fe-alloys Database Thermo-Calc Software is pleased to announce the release of TCFE6, a thermodynamic database for different kinds of steels and Fe-based alloys (stainless steels, high-speed
TCFE6: TCS Steel and Fe-alloys Database Thermo-Calc Software is pleased to announce the release of TCFE6, a thermodynamic database for different kinds of steels and Fe-based alloys (stainless steels, high-speed
Components The elements or compounds which are mixed initially. (e.g. Al & Cu)
 Why Study Phase Diagrams? The purpose of this lecture is to develop an understanding of the phase transformations which occur under conditions of slow cooling; Under such conditions equilibrium is approached,
Why Study Phase Diagrams? The purpose of this lecture is to develop an understanding of the phase transformations which occur under conditions of slow cooling; Under such conditions equilibrium is approached,
Lightweighting is a well-known
 26 COMPUTATIONAL THERMODYNAMICS AND KINETICS FOR MAGNESIUM ALLOY DEVELOPMENT Computational thermodynamics and CALPHAD modeling prove useful for selecting and developing new magnesium alloys. Alan A. Luo,
26 COMPUTATIONAL THERMODYNAMICS AND KINETICS FOR MAGNESIUM ALLOY DEVELOPMENT Computational thermodynamics and CALPHAD modeling prove useful for selecting and developing new magnesium alloys. Alan A. Luo,
Artificial Neural Networks to Predict Tensile Properties in Titanium Forgings October 7-10, 2012 Atlanta, Georgia
 Artificial Neural Networks to Predict Tensile Properties in Titanium Forgings Vikas Saraf October 7-10, 2012 Atlanta, Georgia An ICME Approach: Integrated Computational Materials Engineering Overview Predicting
Artificial Neural Networks to Predict Tensile Properties in Titanium Forgings Vikas Saraf October 7-10, 2012 Atlanta, Georgia An ICME Approach: Integrated Computational Materials Engineering Overview Predicting
The Relationship between Composition and Density in Binary Master Alloys for Titanium. Abstract
 The Relationship between Composition and Density in Binary Master Alloys for Titanium Dr. James W. Robison, Jr. and Scott M. Hawkins Reading Alloys, Inc. Abstract Each year we field several calls from
The Relationship between Composition and Density in Binary Master Alloys for Titanium Dr. James W. Robison, Jr. and Scott M. Hawkins Reading Alloys, Inc. Abstract Each year we field several calls from
MODELLING OF THE THERMO-PHYSICAL AND PHYSICAL PROPERTIES FOR SOLIDIFICATION OF AL-ALLOYS
 Light Metals 23 Edited by P. Crepeau (Warrendale, PA: TMS, 23), 999. MODELLING OF THE THERMO-PHYSICAL AND PHYSICAL PROPERTIES FOR SOLIDIFICATION OF AL-ALLOYS N.Saunders 1, X.Li 2, A.P.Miodownik 1 and J.-P.Schillé
Light Metals 23 Edited by P. Crepeau (Warrendale, PA: TMS, 23), 999. MODELLING OF THE THERMO-PHYSICAL AND PHYSICAL PROPERTIES FOR SOLIDIFICATION OF AL-ALLOYS N.Saunders 1, X.Li 2, A.P.Miodownik 1 and J.-P.Schillé
MODELLING OF THE THERMO-PHYSICAL AND PHYSICAL PROPERTIES FOR SOLIDIFICATION OF AL-ALLOYS
 Light Metals 23 Edited by P. Crepeau TMS (The Minerals, Metals & Materials Society), 23 MODELLING OF THE THERMO-PHYSICAL AND PHYSICAL PROPERTIES FOR SOLIDIFICATION OF AL-ALLOYS N.Saunders 1, X.Li 2, A.P.Miodownik
Light Metals 23 Edited by P. Crepeau TMS (The Minerals, Metals & Materials Society), 23 MODELLING OF THE THERMO-PHYSICAL AND PHYSICAL PROPERTIES FOR SOLIDIFICATION OF AL-ALLOYS N.Saunders 1, X.Li 2, A.P.Miodownik
Thermodynamic Database User s Guide
 Thermodynamic Database User s Guide Copyright 2000-2013 CompuTherm LLC Contents 1. PanAluminum... 1 1.1 Components... 2 1.2 Suggested Composition Range... 2 1.3 Phases... 2 1.4 Key Elements and Subsystems...
Thermodynamic Database User s Guide Copyright 2000-2013 CompuTherm LLC Contents 1. PanAluminum... 1 1.1 Components... 2 1.2 Suggested Composition Range... 2 1.3 Phases... 2 1.4 Key Elements and Subsystems...
Material Properties and Phase Diagrams
 PY2M20 Material Properties and Phase Diagrams Lecture 6 P. Stamenov, PhD School of Physics, TCD PY2M20-6 Microstructures in Eutectic ti Systems: I C o < 2 wt% Sn Result: - at extreme ends - polycrystal
PY2M20 Material Properties and Phase Diagrams Lecture 6 P. Stamenov, PhD School of Physics, TCD PY2M20-6 Microstructures in Eutectic ti Systems: I C o < 2 wt% Sn Result: - at extreme ends - polycrystal
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK 6 Finchwell Close Contact: Mr M Collier Handsworth Tel: +44 (0)114-2437271 Sheffield Fax: +44 (0)114-2437288 South Yorkshire E-Mail: office@metalstechnology.co.uk
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK 6 Finchwell Close Contact: Mr M Collier Handsworth Tel: +44 (0)114-2437271 Sheffield Fax: +44 (0)114-2437288 South Yorkshire E-Mail: office@metalstechnology.co.uk
Thema: Erfahrungen mit Thermocalc und Dictra auf dem Gebiet der Bondcoat phasenbestimmung und Diffusion
 Thema: Erfahrungen mit Thermocalc und Dictra auf dem Gebiet der Bondcoat phasenbestimmung und Diffusion Prof. Dr. D. R. G. Achar Institut für Werkstoffe und verfahren der Energietechnik Thermocalc Anwendertreffen
Thema: Erfahrungen mit Thermocalc und Dictra auf dem Gebiet der Bondcoat phasenbestimmung und Diffusion Prof. Dr. D. R. G. Achar Institut für Werkstoffe und verfahren der Energietechnik Thermocalc Anwendertreffen
Chapter 9: Phase Diagrams
 Chapter 9: Phase Diagrams When we combine two elements... what equilibrium state do we get? In particular, if we specify... --a composition (e.g., wt% Cu - wt% Ni), and --a temperature (T ) then... How
Chapter 9: Phase Diagrams When we combine two elements... what equilibrium state do we get? In particular, if we specify... --a composition (e.g., wt% Cu - wt% Ni), and --a temperature (T ) then... How
A master of science, engineering and entrepreneurship. Anders Engström
 A master of science, engineering and entrepreneurship Anders Engström The Ågren Symposium 2017 Outline 1. Introduction 2. Science 3. Engineering 4. Entrepreneurship Introduction My interaction with John
A master of science, engineering and entrepreneurship Anders Engström The Ågren Symposium 2017 Outline 1. Introduction 2. Science 3. Engineering 4. Entrepreneurship Introduction My interaction with John
Modeling Diffusion: Flux
 Modeling Diffusion: Flux Flux (#/area/time): J = 1 A dm dt Directional Quantity y Jy kg atoms m 2 or s m 2 s Jx Jz x z Flux can be measured for: --vacancies and interstitials --host (A) atoms --impurity
Modeling Diffusion: Flux Flux (#/area/time): J = 1 A dm dt Directional Quantity y Jy kg atoms m 2 or s m 2 s Jx Jz x z Flux can be measured for: --vacancies and interstitials --host (A) atoms --impurity
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK 6 Finchwell Close Contact: Mr M Collier Handsworth Tel: +44 (0)114-2437271 Sheffield Fax: +44 (0)114-2437288 South Yorkshire E-Mail: office@metalstechnology.co.uk
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK 6 Finchwell Close Contact: Mr M Collier Handsworth Tel: +44 (0)114-2437271 Sheffield Fax: +44 (0)114-2437288 South Yorkshire E-Mail: office@metalstechnology.co.uk
Development of high temperature Ti-Ta shape memory alloys
 , 01003 (2009) DOI:10.1051/esomat/200901003 Owned by the authors, published by EDP Sciences, 2009 Development of high temperature Ti-Ta shape memory alloys Shuichi Miyazaki 1,2,a, Hee Young Kim 1,b, and
, 01003 (2009) DOI:10.1051/esomat/200901003 Owned by the authors, published by EDP Sciences, 2009 Development of high temperature Ti-Ta shape memory alloys Shuichi Miyazaki 1,2,a, Hee Young Kim 1,b, and
Traditionally materials have been divided into three major groups: Metals, Ceramics and Polymers. In addition Composites and biomaterials.
 Tilley, Understanding solids : Traditionally materials have been divided into three major groups: Metals, Ceramics and Polymers. In addition Composites and biomaterials. Q: What characterize a material?
Tilley, Understanding solids : Traditionally materials have been divided into three major groups: Metals, Ceramics and Polymers. In addition Composites and biomaterials. Q: What characterize a material?
Phase diagrams wt% of carbon in Fe microstructure of a lead tin alloy of eutectic composition
 Phase diagrams 0.44 wt% of carbon in Fe microstructure of a lead tin alloy of eutectic composition Primer Materials For Science Teaching Spring 2018 28.6.2018 What is a phase? A phase may be defined as
Phase diagrams 0.44 wt% of carbon in Fe microstructure of a lead tin alloy of eutectic composition Primer Materials For Science Teaching Spring 2018 28.6.2018 What is a phase? A phase may be defined as
Homework #4 PROBLEM SOLUTIONS
 Homework #4 PROBLEM SOLUTIONS 4.2 Determination of the number of vacancies per cubic meter in gold at 900 C (1173 K) requires the utilization of Equations (4.1) and (4.2) as follows: N V N exp Q V kt N
Homework #4 PROBLEM SOLUTIONS 4.2 Determination of the number of vacancies per cubic meter in gold at 900 C (1173 K) requires the utilization of Equations (4.1) and (4.2) as follows: N V N exp Q V kt N
Comparing Fatigue Notch Sensitivities of Various Alloys Before and After Mechanical Surface Treating
 Comparing Fatigue Notch Sensitivities of Various Alloys Before and After Mechanical Surface Treating M. Wollmann, J. Atoura, M. Mhaede, L. Wagner 1 1 Institute of Materials Science and Engineering Clausthal
Comparing Fatigue Notch Sensitivities of Various Alloys Before and After Mechanical Surface Treating M. Wollmann, J. Atoura, M. Mhaede, L. Wagner 1 1 Institute of Materials Science and Engineering Clausthal
Modelling of materials properties and behaviour critical to casting simulation
 Materials Science and Engineering A 413 414 (2005) 465 469 Modelling of materials properties and behaviour critical to casting simulation Z. Guo a,, N. Saunders b, A.P. Miodownik b, J.-Ph. Schillé a a
Materials Science and Engineering A 413 414 (2005) 465 469 Modelling of materials properties and behaviour critical to casting simulation Z. Guo a,, N. Saunders b, A.P. Miodownik b, J.-Ph. Schillé a a
Solid Solutioning in CoCrFeNiMx (M= 4d transition metals) High-Entropy Alloys
 Solid Solutioning in CoCrFeNiMx (M= 4d transition metals) High-Entropy Alloys Sheng Guo Department of Industrial and Materials Science Chalmers University of Technology, Gothenburg, Sweden 21 September
Solid Solutioning in CoCrFeNiMx (M= 4d transition metals) High-Entropy Alloys Sheng Guo Department of Industrial and Materials Science Chalmers University of Technology, Gothenburg, Sweden 21 September
Marketplaces and Interoperability - Data Creation, Curation and Exploitation. The View of a Software Owner
 Marketplaces and Interoperability - Data Creation, Curation and Exploitation The View of a Software Owner 1. Background - Introduction to Thermo-Calc 2. Our Data - Creation - Curation - Exploitation 3.
Marketplaces and Interoperability - Data Creation, Curation and Exploitation The View of a Software Owner 1. Background - Introduction to Thermo-Calc 2. Our Data - Creation - Curation - Exploitation 3.
Thermodynamics and Microstructure: Recent Examples for Coupling of Thermodynamic and Mobility Data to the Software MICRESS
 12.09.08 Aachen Thermodynamics and Microstructure: Recent Examples for Coupling of Thermodynamic and Mobility Data to the Software MICRESS Dr. Bernd Böttger ACCESS e.v. Aachen Outline Microstructure Simulation
12.09.08 Aachen Thermodynamics and Microstructure: Recent Examples for Coupling of Thermodynamic and Mobility Data to the Software MICRESS Dr. Bernd Böttger ACCESS e.v. Aachen Outline Microstructure Simulation
(12) 1. Just one True and False question and a couple of multiple choice calculations, circle one answer for each problem, no partial credit.
 (1) 1. Just one True and False question and a couple of multiple choice calculations, circle one answer for each problem, no partial credit. The next page is left blank for your use, but no partial will
(1) 1. Just one True and False question and a couple of multiple choice calculations, circle one answer for each problem, no partial credit. The next page is left blank for your use, but no partial will
Machine learning in materials design, oil exploration, and beyond
 Machine learning in materials design, oil exploration, and beyond Gareth Conduit & Bartomeu Monserrat EP14153898.3; US 2014/177578; GB1302743.8 EP14161255.6; US 2014/223465; GB1307533.8 EP14161529.4; US
Machine learning in materials design, oil exploration, and beyond Gareth Conduit & Bartomeu Monserrat EP14153898.3; US 2014/177578; GB1302743.8 EP14161255.6; US 2014/223465; GB1307533.8 EP14161529.4; US
SUB-Programs - Calibration range Fe Base for "PMI-MASTER Pro" Spark - mode Fe 000
 SUB-Programs - Calibration range Fe Base for "PMI-MASTER Pro" Spark - mode Fe 100 Fe 200 *** Fe 250 *** Fe 300 Fe 400 Fe 500 Fe 000 Fe low alloy steel cast iron Cr hard / Ni resist stainless steel tool
SUB-Programs - Calibration range Fe Base for "PMI-MASTER Pro" Spark - mode Fe 100 Fe 200 *** Fe 250 *** Fe 300 Fe 400 Fe 500 Fe 000 Fe low alloy steel cast iron Cr hard / Ni resist stainless steel tool
solvent: component of a solution present in the greatest amount in alloy.
 Phase Equilibrium Diagrams:- Phase equilibrium diagram is a graphic relationship between temperature and weight ratios of elements and alloys contribute to the built of the diagram. Phase diagrams provide
Phase Equilibrium Diagrams:- Phase equilibrium diagram is a graphic relationship between temperature and weight ratios of elements and alloys contribute to the built of the diagram. Phase diagrams provide
Release Notes: Thermo-Calc Software Package Version 2018b
 : Thermo-Calc Software Package Version 2018b 2018 Foundation of Computational Thermodynamics: Solna, Sweden Contents Release Notes Software Development Kits (SDKs) 2 TC-Python API 2 New Features: Scheil
: Thermo-Calc Software Package Version 2018b 2018 Foundation of Computational Thermodynamics: Solna, Sweden Contents Release Notes Software Development Kits (SDKs) 2 TC-Python API 2 New Features: Scheil
CHAPTER 5 IMPERFECTIONS IN SOLIDS PROBLEM SOLUTIONS
 CHAPTER 5 IMPERFECTIONS IN SOLIDS PROBLEM SOLUTIONS Vacancies and Self-Interstitials 5.1 Calculate the fraction of atom sites that are vacant for copper at its melting temperature of 1084 C (1357 K). Assume
CHAPTER 5 IMPERFECTIONS IN SOLIDS PROBLEM SOLUTIONS Vacancies and Self-Interstitials 5.1 Calculate the fraction of atom sites that are vacant for copper at its melting temperature of 1084 C (1357 K). Assume
1. Use the Ellingham Diagram (reproduced here as Figure 0.1) to answer the following.
 315 Problems 1. Use the Ellingham Diagram (reproduced here as Figure 0.1) to answer the following. (a) Find the temperature and partial pressure of O 2 where Ni(s), Ni(l), and NiO(s) are in equilibrium.
315 Problems 1. Use the Ellingham Diagram (reproduced here as Figure 0.1) to answer the following. (a) Find the temperature and partial pressure of O 2 where Ni(s), Ni(l), and NiO(s) are in equilibrium.
Transmission Electron Microscopy Study of the Infrared Brazed High-strength Titanium Alloy
 J. Mater. Sci. Technol., 2010, 26(4), 311-316. Transmission Electron Microscopy Study of the Infrared Brazed High-strength Titanium Alloy Z.Y. Wu 1), R.K. Shiue 1) and C.S. Chang 2) 1) Department of Materials
J. Mater. Sci. Technol., 2010, 26(4), 311-316. Transmission Electron Microscopy Study of the Infrared Brazed High-strength Titanium Alloy Z.Y. Wu 1), R.K. Shiue 1) and C.S. Chang 2) 1) Department of Materials
Technologies for Process Design of Titanium Alloy Forging for Aircraft Parts
 Technologies for Process Design of Titanium Alloy Forging for Aircraft Parts Takashi CHODA *1, Dr. Hideto OYAMA *2, Shogo MURAKAMI *3 *1 Titanium Research & Development Section, Titanium Div., Iron & Steel
Technologies for Process Design of Titanium Alloy Forging for Aircraft Parts Takashi CHODA *1, Dr. Hideto OYAMA *2, Shogo MURAKAMI *3 *1 Titanium Research & Development Section, Titanium Div., Iron & Steel
PHASE EQUILIBRIUM P + F = C + 2
 PHASE EQUILIBRIUM Component: is either pure metal and/or compound of which an alloy is composed. They refer to the independent chemical species that comprise the system. Solid Solution: It consists of
PHASE EQUILIBRIUM Component: is either pure metal and/or compound of which an alloy is composed. They refer to the independent chemical species that comprise the system. Solid Solution: It consists of
Performing Scheil-Gulliver (SG) analysis in MatCalc
 Performing Scheil-Gulliver (SG) analysis in MatCalc (MatCalc 6.00.0200) P. Warczok Outline Solid phase composition after alloy solidification Back-diffusion effect Primary precipitates Modeling of solid-solid
Performing Scheil-Gulliver (SG) analysis in MatCalc (MatCalc 6.00.0200) P. Warczok Outline Solid phase composition after alloy solidification Back-diffusion effect Primary precipitates Modeling of solid-solid
Mohammad Anwar Karim Id :
 Department of Mechanical and Industrial Engineering ME 8109 Casting and Solidification of Materials EFFECTS OF RAPID SOLIDIFICATION ON MICROSTRUCTURE AND PROPERTIES OF AL, MG & TI ALLOYS Winter 2012 Presented
Department of Mechanical and Industrial Engineering ME 8109 Casting and Solidification of Materials EFFECTS OF RAPID SOLIDIFICATION ON MICROSTRUCTURE AND PROPERTIES OF AL, MG & TI ALLOYS Winter 2012 Presented
Characterization of Phases in an As-cast Copper-Manganese- Aluminum Alloy
 J. Mater. Sci. Technol., Vol.22 No.6, 2006 779 Characterization of Phases in an As-cast Copper-Manganese- Aluminum Alloy J.Iqbal, F.Hasan and F.Ahmad Department of Metallurgical and Materials Engineering,
J. Mater. Sci. Technol., Vol.22 No.6, 2006 779 Characterization of Phases in an As-cast Copper-Manganese- Aluminum Alloy J.Iqbal, F.Hasan and F.Ahmad Department of Metallurgical and Materials Engineering,
Phase Diagrams. Phases
 Phase Diagrams Reading: Callister Ch. 10 What is a phase? What is the equilibrium i state t when different elements are mixed? What phase diagrams tell us. How phases evolve with temperature and composition
Phase Diagrams Reading: Callister Ch. 10 What is a phase? What is the equilibrium i state t when different elements are mixed? What phase diagrams tell us. How phases evolve with temperature and composition
Microstructure and creep resistance of Ti-rich Mo + Mo5Si3 + Mo5SiB2 alloys
 Engineering Conferences International ECI Digital Archives Beyond Nickel-Based Superalloys II Proceedings 7-18-2016 Microstructure and creep resistance of Ti-rich Mo + Mo5Si3 + Mo5SiB2 alloys Daniel Schliephake
Engineering Conferences International ECI Digital Archives Beyond Nickel-Based Superalloys II Proceedings 7-18-2016 Microstructure and creep resistance of Ti-rich Mo + Mo5Si3 + Mo5SiB2 alloys Daniel Schliephake
Use of Modeling in Production of Titanium Alloys. Titanium 2006 San Diego Dr Stephen Fox Dr. Vasisht Venkatesh TIMET
 Use of Modeling in Production of Titanium Alloys Titanium 2006 San Diego Dr Stephen Fox Dr. Vasisht Venkatesh TIMET Why use computer modeling? Vacuum Arc Remelting Rolling, forging etc Thermodynamic modeling
Use of Modeling in Production of Titanium Alloys Titanium 2006 San Diego Dr Stephen Fox Dr. Vasisht Venkatesh TIMET Why use computer modeling? Vacuum Arc Remelting Rolling, forging etc Thermodynamic modeling
Database name: TCS Si-based alloys Database Database acronym: TCSI
 TCSI1 - TCS Ultrapure Silicon Database Database name: TCS Si-based alloys Database Database acronym: TCSI Database owner: Thermo-Calc Software AB Database version: 1.2 TCSI is a thermodynamic database
TCSI1 - TCS Ultrapure Silicon Database Database name: TCS Si-based alloys Database Database acronym: TCSI Database owner: Thermo-Calc Software AB Database version: 1.2 TCSI is a thermodynamic database
INSTITUTE OF AERONAUTICAL ENGINEERING (Autonomous) Dundigal, Hyderabad
 INSTITUTE OF AERONAUTICAL ENGINEERING (Autonomous) Dundigal, Hyderabad -500 043 MECHANICAL ENGINEERING TUTORIAL QUESTION BANK Course Name METALLURGY AND MATERIAL SCIENCE Course Code AME005 Class III Semester
INSTITUTE OF AERONAUTICAL ENGINEERING (Autonomous) Dundigal, Hyderabad -500 043 MECHANICAL ENGINEERING TUTORIAL QUESTION BANK Course Name METALLURGY AND MATERIAL SCIENCE Course Code AME005 Class III Semester
Chapter 10: Phase Transformations
 Chapter 10: Phase Transformations ISSUES TO ADDRESS... Transforming one phase into another takes time. Fe C FCC (Austenite) Eutectoid transformation Fe 3 C (cementite) + (ferrite) (BCC) How does the rate
Chapter 10: Phase Transformations ISSUES TO ADDRESS... Transforming one phase into another takes time. Fe C FCC (Austenite) Eutectoid transformation Fe 3 C (cementite) + (ferrite) (BCC) How does the rate
The peritectic transformation, where δ (ferrite) and L (liquid)
 Cellular automaton simulation of peritectic solidification of a C-Mn steel *Su Bin, Han Zhiqiang, and Liu Baicheng (Key Laboratory for Advanced Materials Processing Technology (Ministry of Education),
Cellular automaton simulation of peritectic solidification of a C-Mn steel *Su Bin, Han Zhiqiang, and Liu Baicheng (Key Laboratory for Advanced Materials Processing Technology (Ministry of Education),
Heat transfer coefficient and latent heat of martensite in a medium-carbon steel
 International Communications in Heat and Mass Transfer, volume 39, issue 10, December 2012, Pages 1519 1521. Heat transfer coefficient and latent heat of martensite in a medium-carbon steel Hala Salman
International Communications in Heat and Mass Transfer, volume 39, issue 10, December 2012, Pages 1519 1521. Heat transfer coefficient and latent heat of martensite in a medium-carbon steel Hala Salman
atoms = 1.66 x g/amu
 CHAPTER 2 Q1- How many grams are there in a one amu of a material? A1- In order to determine the number of grams in one amu of material, appropriate manipulation of the amu/atom, g/mol, and atom/mol relationships
CHAPTER 2 Q1- How many grams are there in a one amu of a material? A1- In order to determine the number of grams in one amu of material, appropriate manipulation of the amu/atom, g/mol, and atom/mol relationships
New functionalities of TC-DICTRA
 New functionalities of TC-DICTRA Thermo-Calc Anwendertreffen und Schulung Aachen 11-13 th of June 2003 Dr. Anders Engström Stockholm Technology Park SE-113 47 Stockholm, Sweden Outline Introduction Thermo-Calc
New functionalities of TC-DICTRA Thermo-Calc Anwendertreffen und Schulung Aachen 11-13 th of June 2003 Dr. Anders Engström Stockholm Technology Park SE-113 47 Stockholm, Sweden Outline Introduction Thermo-Calc
Chapter 10: Phase Transformations
 Chapter 10: Phase Transformations ISSUES TO ADDRESS... Transforming one phase into another takes time. Fe (Austenite) Eutectoid transformation Fe 3 C (cementite) + C FCC (ferrite) (BCC) How does the rate
Chapter 10: Phase Transformations ISSUES TO ADDRESS... Transforming one phase into another takes time. Fe (Austenite) Eutectoid transformation Fe 3 C (cementite) + C FCC (ferrite) (BCC) How does the rate
ATI FLAT ROLLED PRODUCTS Brackenridge, PA for technical competence in the field of
 A2LA has accredited Brackenridge, PA for technical competence in the field of Mechanical Testing This laboratory is accredited in accordance with the recognized International Standard ISO/IEC 17025:2005
A2LA has accredited Brackenridge, PA for technical competence in the field of Mechanical Testing This laboratory is accredited in accordance with the recognized International Standard ISO/IEC 17025:2005
Materials Engineering. Phase transformation Phase diagrams
 Materials Engineering Phase transformation Phase diagrams Phase Transformation Why is it important for us? o Temperature, chemical composition and pressure can change the properties of materials o Understanding
Materials Engineering Phase transformation Phase diagrams Phase Transformation Why is it important for us? o Temperature, chemical composition and pressure can change the properties of materials o Understanding
Influence of Vanadium and Tungsten on the Bainite start temperature
 Influence of Vanadium and Tungsten on the Bainite start temperature Author: Andreas Malmberg Mentors: Mats Hillert and Lars Höglund 2013-05-08 Abstract This paper tries to display the influence of the
Influence of Vanadium and Tungsten on the Bainite start temperature Author: Andreas Malmberg Mentors: Mats Hillert and Lars Höglund 2013-05-08 Abstract This paper tries to display the influence of the
Partition coeff. at constant temp. and press. if ml < 0 k < 1. İf ml > 0 k >1
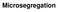 Microsegregation Partition coeff. k=cs/cl at constant temp. and press. if ml < 0 k < 1 İf ml > 0 k >1 Models for Microsegregation 1 no mixing in liquid no diffusion in solid no coarsening 2. partial
Microsegregation Partition coeff. k=cs/cl at constant temp. and press. if ml < 0 k < 1 İf ml > 0 k >1 Models for Microsegregation 1 no mixing in liquid no diffusion in solid no coarsening 2. partial
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Hareness Circle Industrial Estate Aberdeen Scotland AB12 3LY Contact: Mrs D McAdam Tel: +44 (0)1224-890020 Fax: +44 (0)1224-890220 E-Mail:
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Hareness Circle Industrial Estate Aberdeen Scotland AB12 3LY Contact: Mrs D McAdam Tel: +44 (0)1224-890020 Fax: +44 (0)1224-890220 E-Mail:
Lecture 7: Solid State Reactions Phase Diagrams and Mixing
 Lecture 7: Solid State Reactions Phase Diagrams and Mixing Prof Ken Durose, Univ of Liverpool Text book for this lecture: Callister Materials Science and Engineering Learning objectives 1.Solid state reactions
Lecture 7: Solid State Reactions Phase Diagrams and Mixing Prof Ken Durose, Univ of Liverpool Text book for this lecture: Callister Materials Science and Engineering Learning objectives 1.Solid state reactions
Dynamic Young s Modulus and Mechanical Properties of Ti Hf Alloys
 Materials Transactions, Vol. 45, No. 5 (2004) pp. 1549 to 1554 Special Issue on Recent Research and Developments in Titanium and Its Alloys #2004 The Japan Institute of Metals Dynamic Young s Modulus and
Materials Transactions, Vol. 45, No. 5 (2004) pp. 1549 to 1554 Special Issue on Recent Research and Developments in Titanium and Its Alloys #2004 The Japan Institute of Metals Dynamic Young s Modulus and
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK 19 Wildmere Road Banbury Oxfordshire OX16 3JU Contact: Mr J Guntrip Tel: +44 (0)1295 261211 Fax: +44 (0)1295 263096 E-Mail: qualityuk@wmtr.com
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK 19 Wildmere Road Banbury Oxfordshire OX16 3JU Contact: Mr J Guntrip Tel: +44 (0)1295 261211 Fax: +44 (0)1295 263096 E-Mail: qualityuk@wmtr.com
Globularization Behavior of ELI Grade Ti-6Al-4V Alloy during Non-Isothermal Multi-Step Forging
 Materials Transactions, Vol. 49, No. 1 (8) pp. 215 to 223 #8 The Japan Institute of Metals EXPRESS REGULAR ARTICLE Globularization Behavior of ELI Grade Ti-6Al-4V Alloy during Non-Isothermal Multi-Step
Materials Transactions, Vol. 49, No. 1 (8) pp. 215 to 223 #8 The Japan Institute of Metals EXPRESS REGULAR ARTICLE Globularization Behavior of ELI Grade Ti-6Al-4V Alloy during Non-Isothermal Multi-Step
Physical Metallurgy Friday, January 28, 2011; 8:30 12:00 h
 Physical Metallurgy Friday, January 28, 2011; 8:30 12:00 h Always motivate your answers All sub-questions have equal weight in the assessment Question 1 Precipitation-hardening aluminium alloys are, after
Physical Metallurgy Friday, January 28, 2011; 8:30 12:00 h Always motivate your answers All sub-questions have equal weight in the assessment Question 1 Precipitation-hardening aluminium alloys are, after
Graphical Mode Examples Guide Thermo-Calc Version 2015b
 Graphical Mode Examples Guide Thermo-Calc Version 2015b 1995-2015 Foundation of Computational Thermodynamics Stockholm, Sweden About the Graphical Mode Examples These examples are for Thermo-Calc in graphical
Graphical Mode Examples Guide Thermo-Calc Version 2015b 1995-2015 Foundation of Computational Thermodynamics Stockholm, Sweden About the Graphical Mode Examples These examples are for Thermo-Calc in graphical
Simulation of Shrinkage Defect Formation in Exhaust Manifold
 Simulation of Shrinkage Defect Formation in Exhaust Manifold Castings Guo Zhanli*, Yim Young-Hoon**, Kijima Shuya***, Yoon Joong-Mook**, Schillé Jean-Philippe* * Sente Software Ltd., Surrey Technology
Simulation of Shrinkage Defect Formation in Exhaust Manifold Castings Guo Zhanli*, Yim Young-Hoon**, Kijima Shuya***, Yoon Joong-Mook**, Schillé Jean-Philippe* * Sente Software Ltd., Surrey Technology
