Press Release
|
|
|
- Jody Sherman
- 6 years ago
- Views:
Transcription
1 Press Release Shire Wins Patent Trial against Watson Concerning LIALDA LIALDA Patent Upheld and Extends through June 8, 2020 [Lexington, MA] [March 29, 2016] Shire plc (LSE: SHP, NASDAQ: SHPG) and its subsidiary, Shire Development LLC, announced today that the United States District Court for Southern Florida has upheld its patent for LIALDA (mesalamine) delayed release tablets for the induction of remission in adults with active, mild to moderate ulcerative colitis and for the maintenance of remission of ulcerative colitis. The decision follows litigation by Shire against Watson Pharmaceuticals Inc., Watson Laboratories, Inc.-Florida, Watson Pharma, Inc. and Watson Laboratories, Inc. (collectively Watson, now Actavis ) in connection with their Abbreviated New Drug Application ( ANDA ) for a generic version of LIALDA. Judge Donald M. Middlebrooks issued a ruling holding that Actavis proposed ANDA formulation infringes US Patent No. 6,773,720 ( 720 patent ), the Orange Book listed patent for LIALDA, which extends through June 8, Accordingly, Judge Middlebrooks entered an injunction prohibiting the FDA from approving the ANDA formulation until the expiration of the 720 patent and an injunction against Actavis prohibiting them from making, using, selling or importing their proposed ANDA product until after the expiration of the 720 patent. This is the second patent infringement trial in which the proposed ANDA product from Actavis (formerly Watson) has been found to infringe the 720 patent. Shire is very pleased that the court has once again ruled in our favour, reaffirming the validity of the patent protecting LIALDA, said James Harrington, Senior Vice President, Global Head of Intellectual Property, Shire. This ruling supports the innovation and value we continue to bring to the patients who benefit from this important medicine that allows them to lead better lives. Shire s LIALDA remains the only once-daily mesalamine product indicated for both the induction of remission of mild to moderate ulcerative colitis and the maintenance of remission of ulcerative colitis. Indication Lialda is a prescription medication approved for the induction of remission in patients with active, mild to moderate ulcerative colitis (UC) and for the maintenance of remission of UC. Important Safety Information Do not take Lialda (mesalamine) if you are allergic to salicylates, such as aspirin, or medications that contain aspirin; aminosalicylates; mesalamine; or any other ingredients in Lialda. Tell your doctor if you: have or have had kidney problems. Kidney problems have been reported with medications that contain mesalamine, such as Lialda. Your doctor may check to see how your kidneys are working before starting Lialda and periodically while taking Lialda. have symptoms including cramping, stomach ache, bloody diarrhea, fever, headache, and rash. Medications that contain mesalamine, such as Lialda, have been associated Registered in Jersey, No , 22 Grenville Street, St Helier, Jersey JE4 8PX
2 with a condition that may be hard to tell apart from a UC flare. Call your doctor right away if you have any of these symptoms. He or she may tell you to stop taking Lialda. are allergic to sulfasalazine, as you may also be allergic to Lialda or medications that contain mesalamine. have or have had heart-related allergic reactions, such as inflammation of the heart muscle (myocarditis) or the lining of the heart (pericarditis). These reactions have been seen in patients taking Lialda or medications that contain mesalamine. Your chance of having these types of reactions may increase when taking Lialda. have or have had liver problems. Problems with liver function have been reported in patients who have or have had liver problems and were taking medications that contain mesalamine, such as Lialda. have a stomach blockage. It may take longer for Lialda to start working. The most common side effects reported in clinical studies of Lialda were: ulcerative colitis headache passing gas abnormal liver function test results stomach ache In clinical studies of Lialda, inflammation of the pancreas also occurred. If this happens to you, your doctor may tell you to stop taking Lialda. Other side effects may occur. Before starting Lialda, tell your doctor about all medications you are taking, including: non-steroidal anti-inflammatory drugs (NSAIDs), such as ibuprofen and naproxen. Taking these medications with Lialda may increase your chance of kidney problems. azathioprine and 6-mercaptopurine. Taking these medications with Lialda may increase your chance of blood disorders. Please see Full Prescribing Information for Lialda (mesalamine) You are encouraged to report negative side effects of prescription drugs to the FDA. Visit or call FDA For further information please contact: Investor Relations Sarah Elton-Farr seltonfarr@shire.com Robert Coates rcoates@shire.com Media Michele Galen mgalen@shire.com Gwen Fisher gfisher@shire.com Brooke Clarke brclarke@shire.com
3 NOTES TO EDITORS Shire enables people with life-altering conditions to lead better lives. Our strategy is to focus on developing and marketing innovative specialty medicines to meet significant unmet patient needs. We focus on providing treatments in Rare Diseases, Neuroscience, Gastrointestinal and Internal Medicine and we are developing treatments for symptomatic conditions treated by specialist physicians in other targeted therapeutic areas, such as Ophthalmics. 3
4 Forward-Looking Statements Statements included herein that are not historical facts, including without limitation statements concerning our announced business combination with Baxalta and the timing and financial and strategic benefits thereof, our 20x20 ambition that targets $20 billion in combined product sales by 2020, as well as other targets for future financial results, capital structure, performance and sustainability of the combined company, the combined company s future strategy, plans, objectives, expectations and intentions, the anticipated timing of clinical trials and approvals for, and the commercial potential of, inline or pipeline products are forward-looking statements. Such forward-looking statements involve a number of risks and uncertainties and are subject to change at any time. In the event such risks or uncertainties materialize, Shire s results could be materially adversely affected. The risks and uncertainties include, but are not limited to, the following: the proposed combination with Baxalta may not be completed due to a failure to satisfy certain closing conditions, including any shareholder or regulatory approvals or the receipt of applicable tax opinions; disruption from the proposed transaction with Baxalta may make it more difficult to conduct business as usual or maintain relationships with patients, physicians, employees or suppliers; the combined company may not achieve some or all of the anticipated benefits of Baxalta s spin-off from Baxter International, Inc. ( Baxter ) and the proposed transaction may have an adverse impact on Baxalta s existing arrangements with Baxter, including those related to transition, manufacturing and supply services and tax matters; the failure to achieve the strategic objectives with respect to the proposed combination with Baxalta may adversely affect the combined company s financial condition and results of operations; products and product candidates may not achieve commercial success; product sales from ADDERALL XR and INTUNIV are subject to generic competition; the failure to obtain and maintain reimbursement, or an adequate level of reimbursement, by third-party payers in a timely manner for the combined company s products may affect future revenues, financial condition and results of operations, particularly if there is pressure on pricing of products to treat rare diseases; supply chain or manufacturing disruptions may result in declines in revenue for affected products and commercial traction from competitors; regulatory actions associated with product approvals or changes to manufacturing sites, ingredients or manufacturing processes could lead to significant delays, an increase in operating costs, lost product sales, an interruption of research activities or the delay of new product launches; the successful development of products in various stages of research and development is highly uncertain and requires significant expenditures and time, and there is no guarantee that these products will receive regulatory approval; the actions of certain customers could affect the combined company s ability to sell or market products profitably, and fluctuations in buying or distribution patterns by such customers can adversely affect the combined company s revenues, financial condition or results of operations; investigations or enforcement action by regulatory authorities or law enforcement agencies relating to the combined company s activities in the highly regulated markets in which it operates may result in significant legal costs and the payment of substantial compensation or fines; adverse outcomes in legal matters and other disputes, including the combined company s ability to enforce and defend patents and other intellectual property rights required for its business, could have a material adverse effect on the combined company s revenues, financial condition or results of operations; Shire is undergoing a corporate reorganization and was the subject of an unsuccessful acquisition proposal and the consequent uncertainty could adversely affect the combined company s ability to attract and/or retain the highly skilled personnel needed to meet its strategic objectives; failure to achieve the strategic objectives with respect to Shire s acquisition of NPS Pharmaceuticals Inc. or Dyax may adversely affect the combined company s financial condition and results of operations; the combined company will be dependent on information technology and its systems and infrastructure face certain risks, including from service disruptions, the loss of sensitive or confidential information, cyber-attacks and other security breaches or data leakages that could have a material adverse effect on the combined company s revenues, financial condition or results of operations; the combined company may be unable to retain and hire key personnel and/or maintain its relationships with customers, suppliers and other business partners; difficulties in integrating Dyax or Baxalta into Shire may lead to the combined company not being able to realize the expected operating efficiencies, cost savings, revenue enhancements, synergies or other benefits at the time anticipated or at all; and other risks and uncertainties detailed from time to time in Shire s, Dyax s or Baxalta s filings with the Securities and Exchange Commission ( SEC ), including those risks outlined in Baxalta s current 4
5 Registration Statement on Form S-1, as amended, and in ITEM 1A: Risk Factors in Shire s Annual Report on Form 10-K for the year ended December 31, All forward-looking statements attributable to us or any person acting on our behalf are expressly qualified in their entirety by this cautionary statement. Readers are cautioned not to place undue reliance on these forward-looking statements that speak only as of the date hereof. Except to the extent otherwise required by applicable law, we do not undertake any obligation to republish revised forward-looking statements to reflect events or circumstances after the date hereof or to reflect the occurrence of unanticipated events. 5
Press Release
 Press Release www.shire.com This press release is intended for a global audience. Shire Receives Extension of Market Authorization in Europe for Revestive (Teduglutide) for the Treatment of Paediatric
Press Release www.shire.com This press release is intended for a global audience. Shire Receives Extension of Market Authorization in Europe for Revestive (Teduglutide) for the Treatment of Paediatric
Agios Pharmaceuticals, Inc. (Exact Name of Registrant as Specified in Charter)
 UNITED STATES SECURITIES AND EXCHANGE COMMISSION WASHINGTON, DC 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 Date of report (Date of earliest event
UNITED STATES SECURITIES AND EXCHANGE COMMISSION WASHINGTON, DC 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 Date of report (Date of earliest event
Momenta Pharmaceuticals Reports First Quarter 2017 Financial Results
 May 2, 2017 Momenta Pharmaceuticals Reports First Quarter 2017 Financial Results --Company reports Glatopa 20 mg product revenues of $23 million, a 58% increase over the same period in 2016-- --Ended the
May 2, 2017 Momenta Pharmaceuticals Reports First Quarter 2017 Financial Results --Company reports Glatopa 20 mg product revenues of $23 million, a 58% increase over the same period in 2016-- --Ended the
Supplemental Financial Schedules May 19, 2015
 Supplemental Financial Schedules May 19, 2015 The presentations and related schedules contain financial measures that are not calculated in accordance with generally accepted accounting principles (GAAP).
Supplemental Financial Schedules May 19, 2015 The presentations and related schedules contain financial measures that are not calculated in accordance with generally accepted accounting principles (GAAP).
SMARTSTOP SELF STORAGE, INC. / EXTRA SPACE STORAGE INC. PROPOSED MERGER EMPLOYEE Q&A
 SMARTSTOP SELF STORAGE, INC. / EXTRA SPACE STORAGE INC. PROPOSED MERGER EMPLOYEE Q&A 1. Why is SmartStop merging with Extra Space? SmartStop Self Storage, Inc. ( SmartStop ) and Extra Space Storage Inc.
SMARTSTOP SELF STORAGE, INC. / EXTRA SPACE STORAGE INC. PROPOSED MERGER EMPLOYEE Q&A 1. Why is SmartStop merging with Extra Space? SmartStop Self Storage, Inc. ( SmartStop ) and Extra Space Storage Inc.
Intec. Pharma Unfolding drug delivery solutions. Investor Presentation. Nasdaq: NTEC. June 2017
 Intec Pharma Unfolding drug delivery solutions Investor Presentation Nasdaq: NTEC June 2017 Forward Looking Statements This presentation by Intec Pharma Ltd. (referred to as we or our ) contains forward-looking
Intec Pharma Unfolding drug delivery solutions Investor Presentation Nasdaq: NTEC June 2017 Forward Looking Statements This presentation by Intec Pharma Ltd. (referred to as we or our ) contains forward-looking
REFERENCE CODE GDHC218CFR PUBLICAT ION DATE FEBRUARY 2014 ULCERATIVE COLITIS - US DRUG FORECAST AND MARKET ANALYSIS TO 2022
 REFERENCE CODE GDHC218CFR PUBLICAT ION DATE FEBRUARY 2014 ULCERATIVE COLITIS - US DRUG FORECAST AND MARKET ANALYSIS TO 2022 Executive Summary Sales for Ulcerative Colitis in the United States In 2012,
REFERENCE CODE GDHC218CFR PUBLICAT ION DATE FEBRUARY 2014 ULCERATIVE COLITIS - US DRUG FORECAST AND MARKET ANALYSIS TO 2022 Executive Summary Sales for Ulcerative Colitis in the United States In 2012,
Second Quarter 2017 Earnings Teleconference. August 1, 2017
 Second Quarter 2017 Earnings Teleconference August 1, 2017 Introduction Chuck Triano Senior Vice President, Investor Relations Forward-Looking Statements and Non-GAAP Financial Information 3 Our discussions
Second Quarter 2017 Earnings Teleconference August 1, 2017 Introduction Chuck Triano Senior Vice President, Investor Relations Forward-Looking Statements and Non-GAAP Financial Information 3 Our discussions
Treatment: Nutrition and Medication
 INFLAMMATORY BOWEL DISEASE (IBD): CROHN S DISEASE Treatment: Nutrition and Medication While there is no cure for Crohn s disease, there are a number of options to help treat it. The goals of treatment
INFLAMMATORY BOWEL DISEASE (IBD): CROHN S DISEASE Treatment: Nutrition and Medication While there is no cure for Crohn s disease, there are a number of options to help treat it. The goals of treatment
Swissmedic Accepts Santhera's Filing of SNT-MC17 in Friedreich's Ataxia
 Published: 07:00 04.10.2007 GMT+2 /HUGIN /Source: Santhera Pharmaceuticals Holding AG /SWX: SANN /ISIN: CH0027148649 Swissmedic Accepts Santhera's Filing of SNT-MC17 in Friedreich's Ataxia Liestal and
Published: 07:00 04.10.2007 GMT+2 /HUGIN /Source: Santhera Pharmaceuticals Holding AG /SWX: SANN /ISIN: CH0027148649 Swissmedic Accepts Santhera's Filing of SNT-MC17 in Friedreich's Ataxia Liestal and
People Characteristics: A predictor of cyber security incidents?
 People Characteristics: A predictor of cyber security incidents? Esther Bongenaar HR Analytics Manager Vasilis Giagkoulas HR Analytics Analyst Copyright of Shell International Shell HR Analytics 6 October
People Characteristics: A predictor of cyber security incidents? Esther Bongenaar HR Analytics Manager Vasilis Giagkoulas HR Analytics Analyst Copyright of Shell International Shell HR Analytics 6 October
LEVERAGING OUR BIOLOGICS EXPERTISE IN BIOSIMILARS SCOTT FORAKER VICE PRESIDENT AND GENERAL MANAGER, BIOSIMILARS
 LEVERAGING OUR BIOLOGICS EXPERTISE IN BIOSIMILARS SCOTT FORAKER VICE PRESIDENT AND GENERAL MANAGER, BIOSIMILARS SAFE HARBOR STATEMENT This presentation contains forward-looking statements that are based
LEVERAGING OUR BIOLOGICS EXPERTISE IN BIOSIMILARS SCOTT FORAKER VICE PRESIDENT AND GENERAL MANAGER, BIOSIMILARS SAFE HARBOR STATEMENT This presentation contains forward-looking statements that are based
Shire announces third quarter earnings and reiterates full year guidance
 Press Release Shire announces third quarter earnings and reiterates full year guidance Strong commercial execution and pipeline progression October 23, 2015 Shire (LSE: SHP, NASDAQ: SHPG) announces unaudited
Press Release Shire announces third quarter earnings and reiterates full year guidance Strong commercial execution and pipeline progression October 23, 2015 Shire (LSE: SHP, NASDAQ: SHPG) announces unaudited
SORRENTO THERAPEUTICS, INC. (Exact Name of Registrant as Specified in its Charter)
 UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM 8-K CURRENT REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 Date of Report (Date of earliest event
UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM 8-K CURRENT REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 Date of Report (Date of earliest event
WESTFALIA AUTOMOTIVE ACQUISITION AUGUST 25, 2016
 WESTFALIA AUTOMOTIVE ACQUISITION AUGUST 25, 2016 1 SAFE HARBOR STATEMENT FORWARD-LOOKING STATEMENTS This presentation may contain "forward-looking statements" as defined in the Private Securities Litigation
WESTFALIA AUTOMOTIVE ACQUISITION AUGUST 25, 2016 1 SAFE HARBOR STATEMENT FORWARD-LOOKING STATEMENTS This presentation may contain "forward-looking statements" as defined in the Private Securities Litigation
Second Quarter 2016 Financial Results. August 4, 2016
 Second Quarter 2016 Financial Results August 4, 2016 Cautionary Note Regarding Forward-Looking Statements This presentation and various remarks we make during this presentation contain forward-looking
Second Quarter 2016 Financial Results August 4, 2016 Cautionary Note Regarding Forward-Looking Statements This presentation and various remarks we make during this presentation contain forward-looking
Form 8-K. Sally Beauty Holdings, Inc. - SBH. Filed: December 19, 2006 (period: December 19, 2006)
 THE FOLLOWING INFORMATION, DOCUMENT OR REPORT IS BEING MADE AVAILABLE BY SALLY HOLDINGS LLC TO THE HOLDERS OF ITS 9.25% SENIOR NOTES DUE 2014 AND ITS 10.5% SENIOR SUBORDINATED NOTES DUE 2016 AS WELL AS
THE FOLLOWING INFORMATION, DOCUMENT OR REPORT IS BEING MADE AVAILABLE BY SALLY HOLDINGS LLC TO THE HOLDERS OF ITS 9.25% SENIOR NOTES DUE 2014 AND ITS 10.5% SENIOR SUBORDINATED NOTES DUE 2016 AS WELL AS
UNITED STATES SECURITIES AN D EXCHANGE COMMISSION Washington, D.C FORM SD
 UNITED STATES SECURITIES AN D EXCHANGE COMMISSION Washington, D.C. 20549 FORM SD SPECIALIZED DISCLOSURE REPORT VALEANT PHARMACEUTICALS INTERNATIONAL, INC. (Exact name of the registrant as specified in
UNITED STATES SECURITIES AN D EXCHANGE COMMISSION Washington, D.C. 20549 FORM SD SPECIALIZED DISCLOSURE REPORT VALEANT PHARMACEUTICALS INTERNATIONAL, INC. (Exact name of the registrant as specified in
Why did we have to find out about this from the media instead of our supervisors?
 Week 1 Q&A Dozens of employees have submitted questions through the integration@pgnmail.com mailbox and the Integration SharePoint site. Each week, the company will provide answers to some of the most
Week 1 Q&A Dozens of employees have submitted questions through the integration@pgnmail.com mailbox and the Integration SharePoint site. Each week, the company will provide answers to some of the most
uniqure Completes Strategic Review to Refocus its Pipeline, Reduce Operating Costs and Deliver Long-Term Shareholder Value
 uniqure Completes Strategic Review to Refocus its Pipeline, Reduce Operating Costs and Deliver Long-Term Shareholder Value ~ Company Structure Simplified; Manufacturing to be Consolidated into Lexington,
uniqure Completes Strategic Review to Refocus its Pipeline, Reduce Operating Costs and Deliver Long-Term Shareholder Value ~ Company Structure Simplified; Manufacturing to be Consolidated into Lexington,
HIKMA PHARMACEUTICALS PLC. Merrill Lynch Middle East & North Africa (MENA) Conference 13 December r 2005
 HIKMA PHARMACEUTICALS PLC Merrill Lynch Middle East & North Africa (MENA) Conference 13 December r 2005 Important notice This document and its contents are confidential and may not be redistributed or
HIKMA PHARMACEUTICALS PLC Merrill Lynch Middle East & North Africa (MENA) Conference 13 December r 2005 Important notice This document and its contents are confidential and may not be redistributed or
Santen Acquisition of InnFocus, Developer of MicroShunt Glaucoma Implant Device
 Santen Acquisition of InnFocus, Developer of MicroShunt Glaucoma Implant Device Akira Kurokawa President & CEO August 2, 2016 Copyright 2016 Santen Pharmaceutical Co., Ltd. All rights reserved. 1 External
Santen Acquisition of InnFocus, Developer of MicroShunt Glaucoma Implant Device Akira Kurokawa President & CEO August 2, 2016 Copyright 2016 Santen Pharmaceutical Co., Ltd. All rights reserved. 1 External
SILVERCORP REPORTS SILVER PRODUCTION OF 5.0 MILLION OUNCES, CASH FLOWS FROM OPERATIONS OF $31.9 MILLION, OR $0
 NEWS RELEASE Trading Symbol: TSX: SVM SILVERCORP REPORTS SILVER PRODUCTION OF 5.0 MILLION OUNCES, CASH FLOWS FROM OPERATIONS OF $31.9 MILLION, OR $0.19 PER SHARE, FOR FISCAL YEAR ENDED MARCH 31, 2016 VANCOUVER,
NEWS RELEASE Trading Symbol: TSX: SVM SILVERCORP REPORTS SILVER PRODUCTION OF 5.0 MILLION OUNCES, CASH FLOWS FROM OPERATIONS OF $31.9 MILLION, OR $0.19 PER SHARE, FOR FISCAL YEAR ENDED MARCH 31, 2016 VANCOUVER,
STRATEGIC WORKFORCE PLANNING AT ROYAL DUTCH SHELL (RDS)
 STRATEGIC WORKFORCE PLANNING AT ROYAL DUTCH SHELL (RDS) EAPM Congress Manchester November 2013 Dilek Kizilcik, Taher Ebeid Global Talent Advisor November 2013 3 DEFINITIONS AND CAUTIONARY NOTE The companies
STRATEGIC WORKFORCE PLANNING AT ROYAL DUTCH SHELL (RDS) EAPM Congress Manchester November 2013 Dilek Kizilcik, Taher Ebeid Global Talent Advisor November 2013 3 DEFINITIONS AND CAUTIONARY NOTE The companies
Safe Harbour Statement
 Management Presentation May, 2013 Safe Harbour Statement This presentation contains certain forward-looking statements, which relate to future events or the Company s future performance, that include terms
Management Presentation May, 2013 Safe Harbour Statement This presentation contains certain forward-looking statements, which relate to future events or the Company s future performance, that include terms
AUTODESK, INC. CORPORATE GOVERNANCE GUIDELINES. Adopted December 15, Most Recently Amended December 15, 2016
 AUTODESK, INC. CORPORATE GOVERNANCE GUIDELINES Adopted December 15, 1995 Most Recently Amended December 15, 2016 These guidelines and principles have been adopted by the Board of Directors (the Board )
AUTODESK, INC. CORPORATE GOVERNANCE GUIDELINES Adopted December 15, 1995 Most Recently Amended December 15, 2016 These guidelines and principles have been adopted by the Board of Directors (the Board )
Pfizer To Acquire Hospira
 For immediate release: February 5, 2015 Media Contact: Joan Campion (212) 733-2798 Investor Contact: Ryan Crowe (212) 733-8160 Pfizer To Acquire Hospira - Transaction will significantly enhance Pfizer
For immediate release: February 5, 2015 Media Contact: Joan Campion (212) 733-2798 Investor Contact: Ryan Crowe (212) 733-8160 Pfizer To Acquire Hospira - Transaction will significantly enhance Pfizer
Creating the global leader in Rare Diseases
 Creating the global leader in Rare Diseases Fourth quarter and full year results to December 31, 2015 4 Flemming Ornskov, M.D. CEO Jeff Poulton CFO Phil Vickers, Ph.D. Head of R&D February 11, 2016 SAFE
Creating the global leader in Rare Diseases Fourth quarter and full year results to December 31, 2015 4 Flemming Ornskov, M.D. CEO Jeff Poulton CFO Phil Vickers, Ph.D. Head of R&D February 11, 2016 SAFE
Enoxaparin (Lovenox )
 What is enoxaparin (Lovenox )? Enoxaparin is also called Lovenox Enoxaparin is an anticoagulant, or blood thinner, that works to prevent blood clots from forming in your body or treat blood clots after
What is enoxaparin (Lovenox )? Enoxaparin is also called Lovenox Enoxaparin is an anticoagulant, or blood thinner, that works to prevent blood clots from forming in your body or treat blood clots after
R1 to Acquire Intermedix and Selected as RCM Services Provider for Ascension Physician Business
 R1 to Acquire Intermedix and Selected as RCM Services Provider for Ascension Physician Business Investor Presentation February 26, 2018 Forward-Looking Statements and Non-GAAP Financial Measures This presentation
R1 to Acquire Intermedix and Selected as RCM Services Provider for Ascension Physician Business Investor Presentation February 26, 2018 Forward-Looking Statements and Non-GAAP Financial Measures This presentation
Investor Update. Jefferies Global Healthcare Conference June Because people depend on us
 Investor Update Jefferies Global Healthcare Conference June 2014 Because people depend on us Forward-looking statement This presentation and information communicated verbally to you may contain certain
Investor Update Jefferies Global Healthcare Conference June 2014 Because people depend on us Forward-looking statement This presentation and information communicated verbally to you may contain certain
January (San Francisco, CA) January 8, 2018
 January 2017 J.P. Morgan 36 th Annual Management Healthcare Presentation Conference (San Francisco, CA) January 8, 2018 DISCLAIMER Certain information contained in this presentation relates to or is based
January 2017 J.P. Morgan 36 th Annual Management Healthcare Presentation Conference (San Francisco, CA) January 8, 2018 DISCLAIMER Certain information contained in this presentation relates to or is based
Disclaimer. 2
 www.valirx.com 1 Disclaimer This Document comprises an institutional presentation (the Presentation ) which has been prepared by and is the sole responsibility of ValiRx plc (the Company ). The content
www.valirx.com 1 Disclaimer This Document comprises an institutional presentation (the Presentation ) which has been prepared by and is the sole responsibility of ValiRx plc (the Company ). The content
R&D Strategy and Pipeline Transformation. Philip Vickers, Ph.D. Global Head of Research & Development
 R&D Strategy and Pipeline Transformation Philip Vickers, Ph.D. Global Head of Research & Development Through growth and transformation, patients at the very heart of our thinking HEMOPHILIA SLE Pancreatic
R&D Strategy and Pipeline Transformation Philip Vickers, Ph.D. Global Head of Research & Development Through growth and transformation, patients at the very heart of our thinking HEMOPHILIA SLE Pancreatic
Corporate Presentation November 2017 All amounts in USD unless otherwise stated
 Corporate Presentation November 2017 All amounts in USD unless otherwise stated Disclaimer Certain information and statements in this presentation, including all statements that are not historical facts,
Corporate Presentation November 2017 All amounts in USD unless otherwise stated Disclaimer Certain information and statements in this presentation, including all statements that are not historical facts,
Sales Force Productivity
 Mary Kay Kaufmann Vice President, NBT Team Leader I&IS Division Mark Stoll Vice President, Field Operations I&IS Division Investor Day 2006 8/29/2006 9:43 AM metis\roadshow Presentation\07 Roadshow Secondary
Mary Kay Kaufmann Vice President, NBT Team Leader I&IS Division Mark Stoll Vice President, Field Operations I&IS Division Investor Day 2006 8/29/2006 9:43 AM metis\roadshow Presentation\07 Roadshow Secondary
Maximizing Market Access: THE 5 MOST CRITICAL QUESTIONS TO ASK WHEN LAUNCHING A SPECIALTY DRUGS
 Maximizing Market Access: THE 5 MOST CRITICAL QUESTIONS TO ASK WHEN LAUNCHING A SPECIALTY DRUGS by Jan Nielsen, division president, Access & Patient Support Are You Asking the Right Questions? We ve all
Maximizing Market Access: THE 5 MOST CRITICAL QUESTIONS TO ASK WHEN LAUNCHING A SPECIALTY DRUGS by Jan Nielsen, division president, Access & Patient Support Are You Asking the Right Questions? We ve all
A GUIDE TO THIS REFLECTIONS B RESEARCH STUDY IF YOU RE FIGHTING BREAST CANCER, YOU RE NOT ALONE
 A GUIDE TO THIS REFLECTIONS B327-02 RESEARCH STUDY IF YOU RE FIGHTING BREAST CANCER, YOU RE NOT ALONE Do you have breast cancer that has spread to outside the breast? Has your tumor tested positive for
A GUIDE TO THIS REFLECTIONS B327-02 RESEARCH STUDY IF YOU RE FIGHTING BREAST CANCER, YOU RE NOT ALONE Do you have breast cancer that has spread to outside the breast? Has your tumor tested positive for
STATEMENT SANDRA KWEDER, M.D DEPUTY DIRECTOR, OFFICE OF NEW DRUGS CENTER FOR DRUG EVALUATION AND RESEARCH U.S. FOOD AND DRUG ADMINISTRATION BEFORE THE
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Rockville MD 20857 STATEMENT OF SANDRA KWEDER, M.D DEPUTY DIRECTOR, OFFICE OF NEW DRUGS CENTER FOR DRUG EVALUATION
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Rockville MD 20857 STATEMENT OF SANDRA KWEDER, M.D DEPUTY DIRECTOR, OFFICE OF NEW DRUGS CENTER FOR DRUG EVALUATION
FUTURE OF ENERGY SURVEY - NIGERIA. March 2015, Nigeria
 FUTURE OF ENERGY SURVEY - NIGERIA March 015, Nigeria Copyright Company by name Shell International appears here BV DEFINITIONS AND CAUTIONARY NOTE The companies in which Royal Dutch Shell plc directly
FUTURE OF ENERGY SURVEY - NIGERIA March 015, Nigeria Copyright Company by name Shell International appears here BV DEFINITIONS AND CAUTIONARY NOTE The companies in which Royal Dutch Shell plc directly
CRO partner in Rx/CDx Co-Development
 CRO partner in Rx/CDx Co-Development DEDICATED DIALOGUE A sponsored roundtable discussion published in Pharmaceutical Executive Two Covance executives discuss a CRO s role in supporting Companion Diagnostics
CRO partner in Rx/CDx Co-Development DEDICATED DIALOGUE A sponsored roundtable discussion published in Pharmaceutical Executive Two Covance executives discuss a CRO s role in supporting Companion Diagnostics
LifeCycle Pharma A/S IMPROVING TREATMENTS IMPROVING LIVES. March 2011
 LifeCycle Pharma A/S IMPROVING TREATMENTS IMPROVING LIVES March 2011 FORWARD LOOKING STATEMENTS This presentation contains forward looking statements. All statements other than statements of historical
LifeCycle Pharma A/S IMPROVING TREATMENTS IMPROVING LIVES March 2011 FORWARD LOOKING STATEMENTS This presentation contains forward looking statements. All statements other than statements of historical
Investor Presenta,on. Dawson Reimer President & COO. Albert D. Friesen, PhD. Chair & CEO
 Investor Presenta,on Albert D. Friesen, PhD Chair & CEO Dawson Reimer President & COO April 2015 Disclaimer This presentation is for informational purposes only and should not be considered as an offer
Investor Presenta,on Albert D. Friesen, PhD Chair & CEO Dawson Reimer President & COO April 2015 Disclaimer This presentation is for informational purposes only and should not be considered as an offer
An Update on the Technical and Commercial Prospects for CCS
 An Update on the Technical and Commercial Prospects for CCS Angus Gillespie Vice President CO 2 Shell Global Solutions NL Carbon Capture and Storage An update on the technical and commercial prospects
An Update on the Technical and Commercial Prospects for CCS Angus Gillespie Vice President CO 2 Shell Global Solutions NL Carbon Capture and Storage An update on the technical and commercial prospects
Bank of America Merrill Lynch Healthcare Conference. September 13, 2013
 Bank of America Merrill Lynch Healthcare Conference September 13, 2013 Safe Harbor Statement This presentation contains forward-looking statements that are based on management s current expectations and
Bank of America Merrill Lynch Healthcare Conference September 13, 2013 Safe Harbor Statement This presentation contains forward-looking statements that are based on management s current expectations and
Acquisition of Amplify Snack Brands, Inc. December 18, 2017
 Acquisition of Amplify Snack Brands, Inc. December 18, 2017 1 FORWARD LOOKING STATEMENTS This presentation contains forward-looking statements. Many of these forward-looking statements can be identified
Acquisition of Amplify Snack Brands, Inc. December 18, 2017 1 FORWARD LOOKING STATEMENTS This presentation contains forward-looking statements. Many of these forward-looking statements can be identified
Outline of the Business Alliance
 Notification with Respect to the Basic Agreement Between FUJIFILM Holdings Corporation, Taisho Pharmaceutical Co., Ltd. and Toyama Chemical Co., Ltd. Regarding a Strategic Capital and Business Alliance
Notification with Respect to the Basic Agreement Between FUJIFILM Holdings Corporation, Taisho Pharmaceutical Co., Ltd. and Toyama Chemical Co., Ltd. Regarding a Strategic Capital and Business Alliance
Annual Meeting of Shareholders. May 28, 2014
 Annual Meeting of Shareholders May 28, 2014 1 CAUTIONARY STATEMENT PURSUANT TO SAFE HARBOR PROVISIONS OF THE PRIVATE SECURITIES LITIGATION REFORM ACT OF 1995 This document, which includes any exhibits
Annual Meeting of Shareholders May 28, 2014 1 CAUTIONARY STATEMENT PURSUANT TO SAFE HARBOR PROVISIONS OF THE PRIVATE SECURITIES LITIGATION REFORM ACT OF 1995 This document, which includes any exhibits
SORRENTO THERAPEUTICS, INC. (Exact name of registrant as specified in its charter)
 UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, DC 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 Date of Report (Date of earliest event
UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, DC 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 Date of Report (Date of earliest event
A critical review of the current landscape Presentations
 CLINICAL INNOVATION: Fair & Effective Incentives for New Uses of Established Drugs A critical review of the current landscape Presentations Presenters: Christoph de Coster Taylor Wessing LLP Elaine Herrmann
CLINICAL INNOVATION: Fair & Effective Incentives for New Uses of Established Drugs A critical review of the current landscape Presentations Presenters: Christoph de Coster Taylor Wessing LLP Elaine Herrmann
Arthur P. Bedrosian, Chief Executive Officer Brian Kearns, Chief Financial Officer. Investor Presentation February 2009
 Arthur P. Bedrosian, Chief Executive Officer Brian Kearns, Chief Financial Officer Investor Presentation February 2009 Safe Harbor Statement Except for historical facts, the statements in this presentation,
Arthur P. Bedrosian, Chief Executive Officer Brian Kearns, Chief Financial Officer Investor Presentation February 2009 Safe Harbor Statement Except for historical facts, the statements in this presentation,
ANDA ANDA APPROVAL
 DEPARTMENT OF HEALTH & HUMAN SERVICES ANDA 206726 Food and Drug Administration Silver Spring, MD 20993 ANDA APPROVAL Mylan Pharmaceuticals, Inc. 781 Chestnut Ridge Road P.O. Box 4310 Morgantown, WV 26504
DEPARTMENT OF HEALTH & HUMAN SERVICES ANDA 206726 Food and Drug Administration Silver Spring, MD 20993 ANDA APPROVAL Mylan Pharmaceuticals, Inc. 781 Chestnut Ridge Road P.O. Box 4310 Morgantown, WV 26504
Scenarios & Energy Transitions
 Scenarios & Energy Transitions Jeremy Bentham Vice President Global Business Environment Head of Shell Scenarios November 2017 1 Warning: uncertainties ahead This presentation contains data from Shell
Scenarios & Energy Transitions Jeremy Bentham Vice President Global Business Environment Head of Shell Scenarios November 2017 1 Warning: uncertainties ahead This presentation contains data from Shell
Quarterly Report for the Period Ended 31 March 2018
 Quarterly Report for the Period Ended 31 March 2018 Highlights Cash receipts of $617,440 received during the March quarter. Main royalty contributors were the SK-II Power Booster (throughout Asia) and
Quarterly Report for the Period Ended 31 March 2018 Highlights Cash receipts of $617,440 received during the March quarter. Main royalty contributors were the SK-II Power Booster (throughout Asia) and
Cloud Computing: HCM SaaS
 Cloud Computing: HCM SaaS Chris Leone Senior Vice President Applications Development 92 Safe Harbor Statement "Safe Harbor" Statement: Statements in this presentation relating to Oracle's future plans,
Cloud Computing: HCM SaaS Chris Leone Senior Vice President Applications Development 92 Safe Harbor Statement "Safe Harbor" Statement: Statements in this presentation relating to Oracle's future plans,
Investor Presentation. June 2015
 Investor Presentation June 2015 Important Information for Investors and Shareholders This communication does not constitute an offer to buy or sell or the solicitation of an offer to buy or sell any securities
Investor Presentation June 2015 Important Information for Investors and Shareholders This communication does not constitute an offer to buy or sell or the solicitation of an offer to buy or sell any securities
investor meeting SANTA CLARA
 investor meeting 2 0 1 5 SANTA CLARA investor meeting 2 0 1 5 SANTA CLARA Mark Henninger Vice President, Finance Director, Investor Relations Risk Factors Today s presentations contain forward-looking
investor meeting 2 0 1 5 SANTA CLARA investor meeting 2 0 1 5 SANTA CLARA Mark Henninger Vice President, Finance Director, Investor Relations Risk Factors Today s presentations contain forward-looking
U.S. General Services Administration. What s New Federal Agencies. Steve Babine Vice President Citi
 U.S. General Services Administration What s New Federal Agencies Steve Babine Vice President Citi July 2012 This material is intended for use by the GSA only 2012 GSA SmartPay Conference Fine Tune Your
U.S. General Services Administration What s New Federal Agencies Steve Babine Vice President Citi July 2012 This material is intended for use by the GSA only 2012 GSA SmartPay Conference Fine Tune Your
DUCHENNE MUSCULAR DYSTROPHY CLINICAL DEVELOPMENT PROGRAM
 DUCHENNE MUSCULAR DYSTROPHY CLINICAL DEVELOPMENT PROGRAM Dana R. Martin, PharmD Sarepta Therapeutics PPMD ANNUAL CONNECT CONFERENCE ORLANDO, FLORIDA JUNE 27, 2016 FORWARD LOOKING STATEMENTS This presentation,
DUCHENNE MUSCULAR DYSTROPHY CLINICAL DEVELOPMENT PROGRAM Dana R. Martin, PharmD Sarepta Therapeutics PPMD ANNUAL CONNECT CONFERENCE ORLANDO, FLORIDA JUNE 27, 2016 FORWARD LOOKING STATEMENTS This presentation,
Medicis to Acquire LipoSonix
 Medicis to Acquire LipoSonix Medicis Expands Aesthetics Pipeline to Body Contouring SCOTTSDALE, Ariz. and SEATTLE, June 16, 2008 (PRIME NEWSWIRE) -- Medicis (NYSE:MRX) and LipoSonix, Inc. today jointly
Medicis to Acquire LipoSonix Medicis Expands Aesthetics Pipeline to Body Contouring SCOTTSDALE, Ariz. and SEATTLE, June 16, 2008 (PRIME NEWSWIRE) -- Medicis (NYSE:MRX) and LipoSonix, Inc. today jointly
FINANCIAL PERSPECTIVES
 FINANCIAL PERSPECTIVES M³ - Meet Merck Management Marcus Kuhnert, CFO Darmstadt December 10, 2015 Disclaimer Cautionary Note Regarding Forward-Looking Statements This communication may include forward-looking
FINANCIAL PERSPECTIVES M³ - Meet Merck Management Marcus Kuhnert, CFO Darmstadt December 10, 2015 Disclaimer Cautionary Note Regarding Forward-Looking Statements This communication may include forward-looking
Financial and other information related to the intended combination of Intrum Justitia and Lindorff published by Intrum Justitia
 Stockholm 12 June 08:10 CET (page 1 of 5) PRESS RELEASE Intrum Justitia AB (publ) Corporate identity no.:556607-7581 Financial and other information related to the intended combination of Intrum Justitia
Stockholm 12 June 08:10 CET (page 1 of 5) PRESS RELEASE Intrum Justitia AB (publ) Corporate identity no.:556607-7581 Financial and other information related to the intended combination of Intrum Justitia
VISIO MOBILE DIAGNOSTICS ROLLS OUT COMPUMED'S EHEALTH TECHNOLOGY TO MARKETS ACROSS OHIO
 VISIO MOBILE DIAGNOSTICS ROLLS OUT COMPUMED'S EHEALTH TECHNOLOGY TO MARKETS ACROSS OHIO LOS ANGELES, May 27, 2010 -- CompuMed, Inc. (http://www.compumed.net), a telemedicine and medical informatics company,
VISIO MOBILE DIAGNOSTICS ROLLS OUT COMPUMED'S EHEALTH TECHNOLOGY TO MARKETS ACROSS OHIO LOS ANGELES, May 27, 2010 -- CompuMed, Inc. (http://www.compumed.net), a telemedicine and medical informatics company,
MEDICATION GUIDE RIBAVIRIN TABLETS Rx Only Read this Medication Guide carefully before you start taking ribavirin tablets and read the Medication
 MEDICATION GUIDE RIBAVIRIN TABLETS Rx Only Read this Medication Guide carefully before you start taking ribavirin tablets and read the Medication Guide each time you get more ribavirin tablets. There may
MEDICATION GUIDE RIBAVIRIN TABLETS Rx Only Read this Medication Guide carefully before you start taking ribavirin tablets and read the Medication Guide each time you get more ribavirin tablets. There may
Spark Therapeutics, Inc. (Exact Name of Registrant as Specified in its Charter)
 UNITED STATES SECURITIES AND EXCHANGE COMMISSION WASHINGTON, D.C. 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 Date of Report (Date of earliest event
UNITED STATES SECURITIES AND EXCHANGE COMMISSION WASHINGTON, D.C. 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 Date of Report (Date of earliest event
Prescription Drug Pricing. Page 1
 Prescription Drug Pricing Page 1 Prescription Drugs As a Percent of Total National Health Expenditures, 1991-2014 12% 10% 8% 6% 4% 2% 0% 1991 1992 1993 1994 1995 1996 1997 1998 1999 2000 2001 2002 2003
Prescription Drug Pricing Page 1 Prescription Drugs As a Percent of Total National Health Expenditures, 1991-2014 12% 10% 8% 6% 4% 2% 0% 1991 1992 1993 1994 1995 1996 1997 1998 1999 2000 2001 2002 2003
Oracle Buys ClearTrial Adds Leading Cloud-based Clinical Trial Operations and Analytics Applications to the Oracle Health Sciences Suite
 Oracle Buys ClearTrial Adds Leading Cloud-based Clinical Trial Operations and Analytics Applications to the Oracle Health Sciences Suite April 12, 2012 Oracle is currently reviewing
Oracle Buys ClearTrial Adds Leading Cloud-based Clinical Trial Operations and Analytics Applications to the Oracle Health Sciences Suite April 12, 2012 Oracle is currently reviewing
A full-service CRO with integrated early-stage capabilities
 CHARLES RIVER TO ACQUIRE ARGENTA and BIOFOCUS A full-service CRO with integrated early-stage capabilities James C. Foster Chairman, President & CEO Thomas F. Ackerman Executive Vice President & CFO March
CHARLES RIVER TO ACQUIRE ARGENTA and BIOFOCUS A full-service CRO with integrated early-stage capabilities James C. Foster Chairman, President & CEO Thomas F. Ackerman Executive Vice President & CFO March
Investor Presentation. November 26, 2013
 Investor Presentation November 26, 2013 Forward-Looking Statements This presentation may contain forward-looking statements as defined under U.S. federal securities laws. These statements reflect management's
Investor Presentation November 26, 2013 Forward-Looking Statements This presentation may contain forward-looking statements as defined under U.S. federal securities laws. These statements reflect management's
TRUSTED IDENTITIES TRUSTED DEVICES TRUSTED TRANSACTIONS. Investor Presentation - December 2017
 TRUSTED IDENTITIES TRUSTED DEVICES TRUSTED TRANSACTIONS Investor Presentation - December 2017 Safe Harbor Statement This presentation contains forward-looking statements within the meaning of Section 21E
TRUSTED IDENTITIES TRUSTED DEVICES TRUSTED TRANSACTIONS Investor Presentation - December 2017 Safe Harbor Statement This presentation contains forward-looking statements within the meaning of Section 21E
Impact of Patient Engagement on Project Valuation
 Impact of Patient Engagement on Project Valuation Bennett Levitan, MD-PhD Epidemiology, Janssen Research and Development CTTI Patient Groups & Clinical Trials Expert Meeting January 22, 2015 2 Disclosures
Impact of Patient Engagement on Project Valuation Bennett Levitan, MD-PhD Epidemiology, Janssen Research and Development CTTI Patient Groups & Clinical Trials Expert Meeting January 22, 2015 2 Disclosures
Delivering On Our Strategy
 Delivering On Our Strategy Q2 2017 Results August 3, 2017 Flemming Ornskov, MD, MPH CEO Jeff Poulton CFO Safe Harbor Statement Under The Private Securities Litigation Reform Act Of 1995 Statements included
Delivering On Our Strategy Q2 2017 Results August 3, 2017 Flemming Ornskov, MD, MPH CEO Jeff Poulton CFO Safe Harbor Statement Under The Private Securities Litigation Reform Act Of 1995 Statements included
Portfolio management and product development in Actavis. 8 May 2007 Sigurdur O. Olafsson Deputy CEO
 Portfolio management and product development in Actavis 8 May 2007 Sigurdur O. Olafsson Deputy CEO Disclaimer Any statement contained in this presentation that refers to Actavis estimated or anticipated
Portfolio management and product development in Actavis 8 May 2007 Sigurdur O. Olafsson Deputy CEO Disclaimer Any statement contained in this presentation that refers to Actavis estimated or anticipated
Threat-Centric Security for the Digital World. David Goeckeler SVP/GM, Cisco Security Business Group October 2015
 Threat-Centric Security for the Digital World David Goeckeler SVP/GM, Cisco Security Business Group October 2015 FORWARD-LOOKING STATEMENTS This presentation contains projections and other forward-looking
Threat-Centric Security for the Digital World David Goeckeler SVP/GM, Cisco Security Business Group October 2015 FORWARD-LOOKING STATEMENTS This presentation contains projections and other forward-looking
Drugs, Dollars and Dynamics: The Ups and Downs of Generic Pharmaceutical Pricing. Presented by: Pharmacy Healthcare Solutions, Inc.
 Drugs, Dollars and Dynamics: The Ups and Downs of Generic Pharmaceutical Pricing Presented by: Pharmacy Healthcare Solutions, Inc. Learning Objectives 1. Understand factors influencing generic pharmaceutical
Drugs, Dollars and Dynamics: The Ups and Downs of Generic Pharmaceutical Pricing Presented by: Pharmacy Healthcare Solutions, Inc. Learning Objectives 1. Understand factors influencing generic pharmaceutical
SILVERCORP REPORTS Q3 RESULTS: NET INCOME OF $12.7 MILLION, $0.08 PER SHARE AND PROVIDES FISCAL 2019 PRODUCTION AND CASH COSTS GUIDANCE
 NEWS RELEASE Trading Symbol: TSX: SVM NYSE AMERICAN: SVM SILVERCORP REPORTS Q3 RESULTS: NET INCOME OF $12.7 MILLION, $0.08 PER SHARE AND PROVIDES FISCAL 2019 PRODUCTION AND CASH COSTS GUIDANCE VANCOUVER,
NEWS RELEASE Trading Symbol: TSX: SVM NYSE AMERICAN: SVM SILVERCORP REPORTS Q3 RESULTS: NET INCOME OF $12.7 MILLION, $0.08 PER SHARE AND PROVIDES FISCAL 2019 PRODUCTION AND CASH COSTS GUIDANCE VANCOUVER,
ASIT BIOTECH ANNOUNCES ITS INTENTION TO LAUNCH AN INITIAL PUBLIC OFFERING AND TO LIST ITS SHARES ON EURONEXT BRUSSELS AND EURONEXT PARIS
 This announcement does not constitute, or form part of, an offer or invitation to sell or issue, or any solicitation of an offer to purchase or subscribe for securities. This announcement is not a prospectus
This announcement does not constitute, or form part of, an offer or invitation to sell or issue, or any solicitation of an offer to purchase or subscribe for securities. This announcement is not a prospectus
The Mochida Pharmaceutical Group s Medium Term Management Plan
 The Mochida Pharmaceutical Group s 17-19 Medium Term Management Plan This material is an English translation of the press release issued on April 3, 2017 in Japanese, and the Japanese release is given
The Mochida Pharmaceutical Group s 17-19 Medium Term Management Plan This material is an English translation of the press release issued on April 3, 2017 in Japanese, and the Japanese release is given
Capital Market Day June 12, 2012
 Capital Market Day 2012 June 12, 2012 Product Segments IV Drugs John Ducker Member of the Management Board President Region North America Capital Market Day Fresenius Kabi, June 12, 2012 Agenda 1 2 3 4
Capital Market Day 2012 June 12, 2012 Product Segments IV Drugs John Ducker Member of the Management Board President Region North America Capital Market Day Fresenius Kabi, June 12, 2012 Agenda 1 2 3 4
July 13, Dear Secretary Price:
 July 13, 2017 The Honorable Thomas E. Price, M.D. Secretary U.S. Department of Health and Human Services 200 Independence Avenue, SW Washington, DC 20201 Dear Secretary Price: At our meeting on May 16,
July 13, 2017 The Honorable Thomas E. Price, M.D. Secretary U.S. Department of Health and Human Services 200 Independence Avenue, SW Washington, DC 20201 Dear Secretary Price: At our meeting on May 16,
1 st Quarter 2007 Earnings. April 19, 2007
 1 st Quarter 2007 Earnings April 19, 2007 Forward-Looking Statement The statements in this presentation that are not historical facts are forward-looking statements based on current expectations of future
1 st Quarter 2007 Earnings April 19, 2007 Forward-Looking Statement The statements in this presentation that are not historical facts are forward-looking statements based on current expectations of future
Biopharmaceuticals Investor & Analyst Day
 Adequate picture Biopharmaceuticals Investor & Analyst Day Strategic perspective on Merck KGaA, Darmstadt, Germany Karl-Ludwig Kley CEO Darmstadt, Germany September 18, 2014 Disclaimer Remarks All comparative
Adequate picture Biopharmaceuticals Investor & Analyst Day Strategic perspective on Merck KGaA, Darmstadt, Germany Karl-Ludwig Kley CEO Darmstadt, Germany September 18, 2014 Disclaimer Remarks All comparative
Antisense Therapeutics Ltd ASX:ANP January 2017
 Antisense Therapeutics Ltd ASX:ANP January 2017 Forward Looking Statements This presentation contains forward-looking statements regarding the Company s business and the therapeutic and commercial potential
Antisense Therapeutics Ltd ASX:ANP January 2017 Forward Looking Statements This presentation contains forward-looking statements regarding the Company s business and the therapeutic and commercial potential
The Competition Council launched for public consultation the report on sector inquiry on pharma market
 The Competition Council launched for public consultation the report on sector inquiry on pharma market One of the conclusions of the sector inquiry on pharma sector carried out by the Competition Council
The Competition Council launched for public consultation the report on sector inquiry on pharma market One of the conclusions of the sector inquiry on pharma sector carried out by the Competition Council
Pharmaceutical Promotion and Social Media
 Pharmaceutical Promotion and Social Media Marianne Slivkova, J.D., MPH Senior Corporate Counsel Bristol Myers Squibb Opinions expressed are solely those of the author and may not represent the views of
Pharmaceutical Promotion and Social Media Marianne Slivkova, J.D., MPH Senior Corporate Counsel Bristol Myers Squibb Opinions expressed are solely those of the author and may not represent the views of
BIOLOGIC MEDICATIONS IN THE TREATMENT OF IBD. crohnsandcolitis.ca
 BIOLOGIC MEDICATIONS IN THE TREATMENT OF IBD crohnsandcolitis.ca BIOLOGIC MEDICATIONS IN THE TREATMENT OF IBD INFLAMMATION AND IMMUNITY IN CROHN S AND COLITIS Inflammation is the body s normal and natural
BIOLOGIC MEDICATIONS IN THE TREATMENT OF IBD crohnsandcolitis.ca BIOLOGIC MEDICATIONS IN THE TREATMENT OF IBD INFLAMMATION AND IMMUNITY IN CROHN S AND COLITIS Inflammation is the body s normal and natural
Allergan plc Board of Directors Corporate Governance Guidelines
 Allergan plc Board of Directors Corporate Governance Guidelines I. Roles and Responsibilities of the Board of Directors The Board of Directors (the Board ), elected by the shareholders, is the ultimate
Allergan plc Board of Directors Corporate Governance Guidelines I. Roles and Responsibilities of the Board of Directors The Board of Directors (the Board ), elected by the shareholders, is the ultimate
Interim report January September 2017
 Interim report January September 2017 Conference call, November 13 th, 2017 at 3:00 p.m. (CET) Dial-in number: SE: +46 8 566 425 08, US: +1 646 502 51 18 Peter Wolpert, CEO & Founder Anna Ljung, CFO Disclaimer
Interim report January September 2017 Conference call, November 13 th, 2017 at 3:00 p.m. (CET) Dial-in number: SE: +46 8 566 425 08, US: +1 646 502 51 18 Peter Wolpert, CEO & Founder Anna Ljung, CFO Disclaimer
uniqure Announces First Clinical Data From Second Dose Cohort of AMT-060 in Ongoing Phase I/II trial in Patients with Severe Hemophilia B
 uniqure Announces First Clinical Data From Second Dose Cohort of AMT-060 in Ongoing Phase I/II trial in Patients with Severe Hemophilia B -- Second-dose Cohort Demonstrates Dose Response with All Patients
uniqure Announces First Clinical Data From Second Dose Cohort of AMT-060 in Ongoing Phase I/II trial in Patients with Severe Hemophilia B -- Second-dose Cohort Demonstrates Dose Response with All Patients
National MS Society Information Sourcebook
 National MS Society Information Sourcebook www.nationalmssociety.org/sourcebook Interferons The interferons are a group of natural proteins that are produced by human cells in response to viral infection
National MS Society Information Sourcebook www.nationalmssociety.org/sourcebook Interferons The interferons are a group of natural proteins that are produced by human cells in response to viral infection
APITOPE ANNOUNCES ITS INTENTION TO LAUNCH AN INITIAL PUBLIC OFFERING AND LISTING ON EURONEXT BRUSSELS
 APITOPE ANNOUNCES ITS INTENTION TO LAUNCH AN INITIAL PUBLIC OFFERING AND LISTING ON EURONEXT BRUSSELS HASSELT, Belgium, and CHEPSTOW, UK, 31 October 2017 - Apitope (the Company or Apitope ), a clinical
APITOPE ANNOUNCES ITS INTENTION TO LAUNCH AN INITIAL PUBLIC OFFERING AND LISTING ON EURONEXT BRUSSELS HASSELT, Belgium, and CHEPSTOW, UK, 31 October 2017 - Apitope (the Company or Apitope ), a clinical
Bob Picciano Senior Vice President IBM ANALYTICS International Business Machines Corporation
 Bob Picciano Senior Vice President IBM ANALYTICS 2015 International Business Machines Corporation 1 Forward-Looking Statement Certain comments made during this event and in the presentation materials may
Bob Picciano Senior Vice President IBM ANALYTICS 2015 International Business Machines Corporation 1 Forward-Looking Statement Certain comments made during this event and in the presentation materials may
Hydrogen KIVI NIRIA CONGRESS. Eindhoven, 6 th November Dr. Jurgen Louis, Technology Manager Shell Hydrogen. Copyright of Royal Dutch Shell plc 1
 Hydrogen KIVI NIRIA CONGRESS Eindhoven, 6 th November 2013 Dr. Jurgen Louis, Technology Manager Shell Hydrogen 1 DEFINITIONS & CAUTIONARY NOTE The companies in which Royal Dutch Shell plc directly and
Hydrogen KIVI NIRIA CONGRESS Eindhoven, 6 th November 2013 Dr. Jurgen Louis, Technology Manager Shell Hydrogen 1 DEFINITIONS & CAUTIONARY NOTE The companies in which Royal Dutch Shell plc directly and
YASHAJIT SAHA & ABHISHEK SHARMA, SUBJECT MATTER EXPERTS, RESEARCH & ANALYTICS ADVANCED ANALYTICS: A REMEDY FOR COMMERCIAL SUCCESS IN PHARMA.
 YASHAJIT SAHA & ABHISHEK SHARMA, SUBJECT MATTER EXPERTS, RESEARCH & ANALYTICS ADVANCED ANALYTICS: A REMEDY FOR COMMERCIAL SUCCESS IN PHARMA wns wns ADVANCED ANALYTICS: A REMEDY FOR COMMERCIAL SUCCESS IN
YASHAJIT SAHA & ABHISHEK SHARMA, SUBJECT MATTER EXPERTS, RESEARCH & ANALYTICS ADVANCED ANALYTICS: A REMEDY FOR COMMERCIAL SUCCESS IN PHARMA wns wns ADVANCED ANALYTICS: A REMEDY FOR COMMERCIAL SUCCESS IN
Inflammatory Bowel Disease
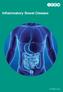 Inflammatory Bowel Disease ICONplc.com Inflammatory bowel disease (IBD) is a chronic, relapsing, inflammatory disorder of the gastrointestinal tract and includes Ulcerative Colitis (UC) and Crohn s Disease
Inflammatory Bowel Disease ICONplc.com Inflammatory bowel disease (IBD) is a chronic, relapsing, inflammatory disorder of the gastrointestinal tract and includes Ulcerative Colitis (UC) and Crohn s Disease
COMPANY OVERVIEW JOHN MORIKIS CHAIRMAN, PRESIDENT & CHIEF EXECUTIVE OFFICER FINANCIAL COMMUNITY PRESENTATION OCTOBER 3, 2017
 COMPANY OVERVIEW JOHN MORIKIS CHAIRMAN, PRESIDENT & CHIEF EXECUTIVE OFFICER Forward Looking Statement The presentations today will contain certain forward looking statements," as defined under U.S. federal
COMPANY OVERVIEW JOHN MORIKIS CHAIRMAN, PRESIDENT & CHIEF EXECUTIVE OFFICER Forward Looking Statement The presentations today will contain certain forward looking statements," as defined under U.S. federal
NASDAQ: ATRS. Investor Presentation January 2017
 NASDAQ: ATRS Investor Presentation January 2017 Safe Harbor Statement This presentation contains forward-looking statements within the meaning of the safe harbor provisions of the Private Securities Litigation
NASDAQ: ATRS Investor Presentation January 2017 Safe Harbor Statement This presentation contains forward-looking statements within the meaning of the safe harbor provisions of the Private Securities Litigation
Emerging Trends in Regulatory Outsourcing WHITE PAPER.
 WHITE PAPER www.makrocare.com/consulting You are not alone in the quest for bringing your products to market and improve the quality of the life for millions! MakroCare brings you the insight on how a
WHITE PAPER www.makrocare.com/consulting You are not alone in the quest for bringing your products to market and improve the quality of the life for millions! MakroCare brings you the insight on how a
New Cardinal Health (Post-Spin)
 New Cardinal Health (Post-Spin) George Barrett Vice Chairman and Chief Executive Officer Healthcare Supply Chain Services and Chief Executive Officer Cardinal Health, Inc. (post-spin) January 14, 2009
New Cardinal Health (Post-Spin) George Barrett Vice Chairman and Chief Executive Officer Healthcare Supply Chain Services and Chief Executive Officer Cardinal Health, Inc. (post-spin) January 14, 2009
