Standard Operating Procedure
|
|
|
- Job Gaines
- 5 years ago
- Views:
Transcription
1 Title: Standard Operating Procedure Flow cytometry-based invasion phenotyping assay Authors: Michel Theron, Richard Hesketh, Sathish Subramanian & Julian Rayner Date: All procedures should be carried out inside a class II microbiological safety cabinet, to maintain aseptic conditions and protect the operator. All liquid and solid wastes must be treated in accordance with the local code of practice. 1. Materials a. Reagents Reagent name Source Catalog number 1% O 2, 3% CO 2, balance N 2 BOC CFDA SE Sigma-Aldrich Co α-chymotrypsin from bovine pancreas Sigma-Aldrich Co. C7762 Complete medium See section 1, sub-section b DDAO SE Invitrogen C34553 Dimethyl sulfoxide Sigma-Aldrich Co. D2438 Dulbecco s phosphate buffered saline Sigma-Aldrich Co. D8537 Gentamicin Invitrogen D-(+)-glucose Sigma-Aldrich Co. G7021 Glutaraldehyde Sigma-Aldrich Co. G5882 HEPES Sigma-Aldrich Co. H4034 Hoechst Invitrogen H3570 Hypoxanthine Sigma-Aldrich Co. H9636 Incomplete medium See section 1, sub-section b Neuraminidase from Vibrio cholerae Sigma-Aldrich Co. N7885 Paraformaldehyde Sigma-Aldrich Co Ribonuclease A MP Biomedicals RPMI liquid Invitrogen RPMI powder Invitrogen Sodium bicarbonate Sigma-Aldrich S5761 1
2 Reagent name Source Catalog number Sodium hydroxide VWR BDH Prolabo M SYBR Green I Invitrogen S7563 Triton X-100 Sigma-Aldrich Co. T9284 Trypsin from bovine pancreas Sigma-Aldrich Co. T1426 b. Media All media solution must be kept warm at +37 C throughout their use in the invasion assay. i. Incomplete media Reagent name Quantity RPMI powder 10.43g HEPES 7.15g Gentamicin 0.5mL D-(+)-glucose 2g Hypoxanthine 1mL of a solution at 50mg/mL in 2M NaOH MilliQ water Enough for 1000mL Adjust the ph of the solution to 7.2 and filter the solution through a 0.2µm membrane. Keep warm in a water bath at +37 C. ii. Complete media Reagent name Heat-inactivated human sera pool Incomplete media Sodium bicarbonate Quantity 50mL 450mL 16mL of a solution at 7.5% in MilliQ water Filter the solution through a 0.2µm membrane. Keep warm in a water bath at +37 C. c. Erythrocytes (RBC) and parasites i. RBC Leukocyte-depleted RBC need to be obtained in accordance with local legislation on the use of human material for research purposes. RBC need to be washed on the day the invasion assay commences, prior to their use in culture with Plasmodium falciparum parasites. 1. Label a 15mL tube with washed RBC 1. Using a disposable serological pipette, add 10mL of RPMI 1640 to the tube 2. Using a disposable serological pipette, add 4mL of leukocyte-depleted RBC to the tube 2
3 3. Centrifuge the 15mL tube at 1,800g for 5min (with brakes on) 4. Using a disposable serological pipette, aspirate and discard all of the supernatant, as well as the top layer of the cell pellet, from the 5. Using a disposable serological pipette, add enough RPMI 1640 to the tube to obtain a 50% hematocrit suspension, and pipette up and down to resuspend the cell pellet ii. Parasitized RBC (prbc) The culture of prbc to be used in the invasion assay needs to be a synchronous, ring-stage culture at the time the donor prbc are mixed with the target RBC (see section 2, sub-section c). The preparation of the donor prbc population depends on the parasitemia of the prbc culture to be phenotyped, as well as the donor:target ratio that will be used to setup the invasion assay (see section 2, sub-section c). If choosing a donor:target ratio of 1:1, a suspension of prbc at a hematocrit of 2% and a parasitemia of 1-2% needs to be prepared. If, on the other hand, a ratio of 1:2 is chosen, a suspension of prbc at a hematocrit of 2% and a parasitemia of 1.5-3% will have to be prepared. In either case, when donor prbc and target RBC are mixed in the appropriate ratio, the parasitemia will be reduced to 0.5-1%. d. Plasticware Plastic 1.5mL microfuge tubes, sterile 15mL conical tubes, sterile 50mL conical tubes, sterile 96-well, flat-bottom plate 96-well, round-bottom plate, sterile Filter-fitted pipette tips, sterile Reagent reservoirs Serological pipettes, sterile Pipette tips Brand and model (example) Eppendorf Safelock BD Falcon BD Falcon BD Falcon Corning Life Sciences Costar Gilson Diamond Filter Corning Life Sciences Costar Corning Life Sciences Costar Stripette Starlab TipOne e. Equipment Equipment Brand and model (example) Centrifuge Eppendorf 5810 Flow cytometer BD LSRII Fridge LEC S53YC Microbiological safety cabinet, class II Contained Air Solutions Bio-Class 2 Microcentrifuge VWR Galaxy MiniStar Multichannel pipette Gilson Pipetman Neo Multichannel P200N Incubator RSBiotech Galaxy R+ 3
4 Equipment Brand and model (example) Incubation culture chamber CBS Scientific M-312 Pipettes Gilson Pipetman P1000, P200, P20 and P10 Pipetting controller Brand accu-jet pro Tube rotator Miltenyi Biotec MACSmix Waterbath Grant SUB Aqua12 2. Method a. Labeling target RBC The choice of fluorescent label for target RBC is dependent on the choice of DNA dye, which itself depends on the flow cytometer that will be used to acquire the data at the end of the invasion assay. The fluorescence emission peak of CFDA SE is approximately 517nm, close to SYBR Green I at about 520nm. CFDA SE can thus only be used in combination with Hoechst The fluorescence emission peak of DDAO SE is close to 657nm. So, it can be used in combination with either Hoechst or SYBR Green I. Fluorescence label/dye combination Method sections to be completed CFDA SE & Hoechst a. i. b. c. i. c. ii. 1./c. ii. 2. c. iii. d. i. e. DDAO SE & Hoechst a. ii. b. c. i. c. ii. 1./c. ii. 2. c. iii. d. i. e. DDAO SE & SYBR Green I a. ii. b. c. i. c. ii. 1./c. ii. 2. c. iii. d. ii. e. i. CFDA SE 1. Label a 50mL tube with 2% hematocrit 2. Using a disposable serological pipette, add 12mL of RPMI 1640 to the tube 3. Using a disposable serological pipette, add 0.5mL of washed RBC to the tube to obtain a 2% hematocrit 4. Mix the cell suspension gently 5. Label a 15mL tube with stained RBC 6. Transfer 3mL of 2% hematocrit RBC suspension from the 50mL tube to the 15mL tube 7. Centrifuge the 15mL tube at 450g for 3min (moderate brakes can be applied) 8. Using a disposable serological pipette, aspirate and discard all of the supernatant from the 9. Label a 15mL tube with CFDA 10. Using a disposable serological pipette, add 3.1mL of RPMI 1640 to the CFDA tube 11. Turn off any direct light source for the reminder of the assay 12. Using a filter-fitted pipette tip, add 12.4µL of 5mM CFDA SE in DMSO to the CFDA tube to obtain a 20µM suspension 13. Mix the 20µM CFDA SE suspension thoroughly 4
5 14. Using a disposable serological pipette, add 3mL of 20µM CFDA SE to the stained RBC tube and immediately pipette the suspension up and down thoroughly to resuspend the pellet Place the tube on a rotator and switch on the rotation at maximum speed 16. Place the rotator inside an incubator at +37 C and incubate for 2h 17. Stop the rotation and centrifuge the stained RBC tube at 450g for 3min 18. Using a disposable serological pipette, aspirate and discard all of the supernatant from the 19. Using a disposable serological pipette, add 3mL of complete media to the stained RBC 20. Centrifuge the tube at 450g for 3min 21. Using a disposable serological pipette, aspirate and discard all of the supernatant from the 22. Using a disposable serological pipette, add 3mL of complete media to the stained RBC 23. Place the tube on the rotator and switch on the rotation at maximum speed 24. Place the rotator inside an incubator at +37 C and incubate for 30min 25. Stop the rotation and centrifuge the stained RBC tube at 450g for 3min 26. Using a disposable serological pipette, aspirate and discard all of the supernatant from the 27. Using a disposable serological pipette, add 3mL of incomplete media to the stained RBC 28. Centrifuge the tube at 450g for 3min 29. Repeat steps 26 to 28 once more 30. Using a disposable serological pipette, aspirate and discard all of the supernatant from the 31. Using a disposable serological pipette, add 3mL of incomplete media to the stained RBC 32. Set the stained RBC tube aside and discard all other remaining tubes At this point, it is possible to store the cells protected from light at +4 C for up to 24h, before proceeding to treat the target RBC with enzymes. ii. DDAO SE 1- Label a 50mL tube with 2% hematocrit 2- Using a disposable serological pipette, add 12mL of RPMI 1640 to the tube 3- Using a disposable serological pipette, add 0.5mL of washed RBC to the tube to obtain a 2% hematocrit 4- Mix the cell suspension gently 5- Label a 15mL tube with stained RBC 6- Transfer 3mL of 2% hematocrit RBC suspension from the 50mL tube to the 15mL tube 7- Centrifuge the 15mL tube at 450g for 3min (moderate brakes can be applied) 1 Rapidly and completely resuspending the cell pellet is important to ensure homogenous staining of the RBC population 5
6 8- Using a disposable serological pipette, aspirate and discard all of the supernatant from the 9- Label a 15mL tube with DDAO 10- Using a disposable serological pipette, add 3.1mL of RPMI 1640 to the DDAO tube 11- Turn off any direct light source for the reminder of the assay 12- Using a filter-fitted pipette tip, add 6.2µL of 5mM DDAO SE in DMSO to the DDAO tube to obtain a 10µM suspension 13- Mix the 10µM DDAO SE suspension thoroughly 14- Using a disposable serological pipette, add 3mL of 10µM DDAO SE to the stained RBC tube and immediately pipette the suspension up and down thoroughly to resuspend the pellet Place the tube on a rotator and switch on the rotation at maximum speed 16- Place the rotator inside an incubator at +37 C and incubate for 2h 17- Stop the rotation and centrifuge the stained RBC tube at 450g for 3min 18- Using a disposable serological pipette, aspirate and discard all of the supernatant from the 19- Using a disposable serological pipette, add 3mL of complete media to the stained RBC 20- Centrifuge the tube at 450g for 3min 21- Using a disposable serological pipette, aspirate and discard all of the supernatant from the 22- Using a disposable serological pipette, add 3mL of complete media to the stained RBC 23- Place the tube on the rotator and switch on the rotation at maximum speed 24- Place the rotator inside an incubator at +37 C and incubate for 30min 25- Stop the rotation and centrifuge the stained RBC tube at 450g for 3min 26- Using a disposable serological pipette, aspirate and discard all of the supernatant from the 27- Using a disposable serological pipette, add 3mL of incomplete media to the stained RBC 28- Centrifuge the tube at 450g for 3min 29- Repeat steps 26 to 28 once more 30- Using a disposable serological pipette, aspirate and discard all of the supernatant from the 31- Using a disposable serological pipette, add 3mL of incomplete media to the stained RBC 32- Set the stained RBC tube aside and discard all other remaining tubes At this point, it is possible to store the cells protected from light at +4 C for up to 24h, before proceeding to treat the target RBC with enzymes. 2 Rapidly and completely resuspending the cell pellet is important to ensure homogenous staining of the RBC population 6
7 b. Enzymatic treatments Regardless of the cell label used, stained target RBC are treated in the same manner throughout this section. 1- Label 6 microfuge tubes A to F 2- Using a filter-fitted pipette tip, transfer 400µL of cell suspension from the stained RBC tube to each of the 6 microfuge tubes 3- Using filter-fitted pipette tips, add 8µL of 1U/mL neuraminidase in RPMI 1640 to tube B and tube E 4- Place the 6 tubes on a rotator and switch on the rotation at maximum speed 5- Place the rotator inside an incubator at +37 C and incubate for 1h 6- Stop the rotation and centrifuge the microfuge tubes on a benchtop microcentrifuge for 30s 7- Using filter-fitted pipette tips, aspirate and discard the supernatant from each of the 6 tubes 8- Using a filter-fitted pipette tip, add 400µL of incomplete media to each of the 6 tubes 9- Centrifuge the tubes on a benchtop microcentrifuge for 30s 10- Using filter-fitted pipette tips, aspirate and discard the supernatant from each of the 6 tubes 11- Using filter-fitted pipette tips, add 400µL of incomplete media to each of the 6 tubes and resuspend the cell pellets by pipetting up and down 12- Using a filter-fitted pipette tip, add 2µL of 10mg/mL trypsin in RPMI 1640 to tube C 13- Using filter-fitted pipette tips, add 40µL of 10mg/mL trypsin in RPMI 1640 to tube D and tube E 14- Using a filter-fitted pipette tip, add 40µL of 10mg/mL chymotrypsin in RPMI 1640 to tube F 15- Place the 6 tubes on the rotator and switch on the rotation at maximum speed 16- Place the rotator inside an incubator at +37 C and incubate for 1h 17- Stop the rotation and centrifuge the microfuge tubes on a benchtop microcentrifuge for 30s 18- Using filter-fitted pipette tips, aspirate and discard the supernatant from each of the 6 tubes 19- Using a filter-fitted pipette tip, add 400µL of incomplete media to each of the 6 tubes 20- Centrifuge the tubes on a benchtop microcentrifuge for 30s 21- Repeat steps 18 to 20 once more 22- Using filter-fitted pipette tips, aspirate and discard the supernatant from each of the 6 tubes 23- Using a filter-fitted pipette tip, add 400µL of complete media to each of the 6 tubes resuspend the cell pellets by pipetting up and down, and discard all other remaining tubes At this point, if sub-sections a and b have been completed on the same day, it is possible to store the cells protected from light at +4 C for up to 24h, before proceeding to incubating the target cells with the donor prbc. c. Plate setup and incubation If the stained target RBC were stored at +4 C, place the tubes inside an incubator at +37 C for 5 to 10min to gently warm them up. 7
8 i. Plate layout PBS A1 B1 C1 D1 E1 F1 A2 B2 C2 D2 E2 F2 A3 B3 C3 D3 E3 F3 PBS PBS ii. Plate setup The volumes of target RBC and donor prbc to be added to the plate depend on the parasitemia of the donor prbc population. If the parasitemia is greater than 1% but less than 2%, 50µL of the donor prbc population and 50µL of the target RBC population should be added to the wells, giving a ratio of 1:1. If the parasitemia is greater than 2%, it is possible to either use a donor:target ratio of 1:1 or 1:2. A ratio of 1:2 is obtained by adding 33µL of the donor prbc population and 67µL of the target RBC population to the wells. With either ratio, the final volume will be 100µL per well. 1. 1:1 ratio 1. Label 18 wells of a 96-well, round-bottom plate, on its lid, from A to E and from 1 to 3, as indicated in sub-section c, sub-sub-section i. 2. Using a disposable serological pipette, add approximately 200µL of PBS to the 78 unlabeled wells of the plate (shaded in grey in sub-section c, sub-sub-section i) 3. Using a filter-fitted pipette tip, add 50µL of donor prbc at 2% hematocrit, 1-2% parasitemia, to each of the 18 wells labeled A1 to F Rotate the plate 180 degrees 5. Using filter-fitted pipette tips, add 50µL of target RBC at 2% hematocrit from each of the 6 microfuge tubes (see sub-section b, step 23) to the corresponding wells (e.g. 50µL from tube A to each of wells A1, A2 and A3) 4 6. Using filter-fitted pipette tips, pipette up and down the contents of each of the 18 wells, while carefully avoiding to create bubbles (a multichannel can be used to facilitate the mixing) 7. Discard all remaining tubes 3 Add the donor prbc by touching the left side wall of the well with the pipette tip 4 Add the target RBC by touching the left side wall of the well with the pipette tip. Having rotated the plate 180 degrees, this will limit the risk of carry-over between wells 8
9 2. 1:2 ratio 1. Label 18 wells of a 96-well, round-bottom plate, on its lid, from A to E and from 1 to 3, as indicated in sub-section c, sub-sub-section i. 2. Using a disposable serological pipette, add approximately 200µL of PBS to the 78 unlabeled wells of the plate (shaded in grey in sub-section c, sub-sub-section i) 3. Using a filter-fitted pipette tip, add 33µL of donor prbc at 2% hematocrit, 1.5-3% parasitemia, to each of the 18 wells labeled A1 to F Rotate the plate 180 degrees 5. Using filter-fitted pipette tips, add 67µL of target RBC at 2% hematocrit from each of the 6 microfuge tubes (see sub-section b, step 23) to the corresponding wells (e.g. 50µL from tube A to each of wells A1, A2 and A3) 6 6. Using filter-fitted pipette tips, pipette up and down the contents of each of the 18 wells, while carefully avoiding to create bubbles (a multichannel can be used to facilitate the mixing) 7. Discard all remaining tubes iii. Incubation Regardless of the donor:target ratio, the plates are treated in the same manner throughout this sub-section. 1- Place the plate inside an incubator culture chamber, pre-warmed to +37 C 2- Close the chamber and gas the chamber for 3min with a mixture of 1% O 2, 3% CO 2 and a balance of N 2 3- Place the chamber inside an incubator at +37 C and incubate for 48h d. Parasite staining If the target RBC were stained with CFDA SE, staining of the parasites will have to be done with Hoechst If the target RBC were stained with DDAO SE, either Hoechst or SYBR Green I may be used to stain the parasites, depending on the laser lines available on the flow cytometer. i. Hoechst Remove the chamber from the incubator and remove the plate from the chamber 2- Centrifuge the plate at 450g for 3min 3- Using pipette tips, draw 50µL of culture supernatant from each of the 18 wells labeled A1 to F3 (a multichannel can be used to facilitate the work throughout the whole staining procedure) 4- Using pipette tips, add 200µL of RPMI 1640 to each of the 18 wells 5- Centrifuge the plate at 450g for 3min 5 Add the donor prbc by touching the left side wall of the well with the pipette tip 6 Add the target RBC by touching the left side wall of the well with the pipette tip. Having rotated the plate 180 degrees, this will limit the risk of carry-over between wells 9
10 6- Using pipette tips, draw 200µL of supernatant from each of the 18 wells 7- Using pipette tips, add 200µL of 2µM Hoechst in RPMI 1640 to each of the 18 wells and pipette up and down to resuspend the cell pellet 8- Place the plate inside an incubator at +37 C and incubate for 1h 9- Centrifuge the plate at 450g for 3min 10- Using pipette tips, draw 200µL of supernatant from each of the 18 wells 11- Using pipette tips, add 200µL of PBS to each of the 18 wells 12- Centrifuge the plate at 450g for 3min 13- Repeat steps 10 to 12 twice more 14- Using pipette tips, draw 200µL of supernatant from each of the 18 wells 15- Using pipette tips, add 200µL of 2% paraformaldehyde/0.2% glutaraldehyde in PBS to each of the 18 wells and pipette up and down to resuspend the cell pellet 16- Place the plate inside a fridge at +4 C and incubate for 1h 17- Centrifuge the plate at 450g for 3min 18- Using pipette tips, draw 200µL of supernatant from each of the 18 wells 19- Using pipette tips, add 200µL of PBS to each of the 18 wells 20- Centrifuge the plate at 450g for 3min 21- Using pipette tips, draw 200µL of supernatant from each of the 18 wells 22- Using pipette tips, add 200µL of PBS to each of the 18 wells and pipette up and down to resuspend the cell pellet At this point, it is possible to store the plate protected from light at +4 C for later data acquisition with a flow cytometer. However, the plate has to be acquired within 7 days of the target RBC being initially stained. ii. SYBR Green I 1. Remove the chamber from the incubator and remove the plate from the chamber 2. Centrifuge the plate at 450g for 3min 3. Using pipette tips, draw 50µL of culture supernatant from each of the 18 wells labeled A1 to F3 (a multichannel can be used to facilitate the work throughout the whole staining procedure) 4. Using pipette tips, add 200µL of PBS to each of the 18 wells 5. Centrifuge the plate at 450g for 3min 6. Using pipette tips, draw 200µL of supernatant from each of the 18 wells 7. Using pipette tips, add 200µL of 2% paraformaldehyde/0.2% glutaraldehyde in PBS to each of the 18 wells and pipette up and down to resuspend the cell pellet 8. Place the plate inside a fridge at +4 C and incubate for 1h 9. Centrifuge the plate at 450g for 3min 10. Using pipette tips, draw 200µL of supernatant from each of the 18 wells 11. Using pipette tips, add 200µL of PBS to each of the 18 wells 12. Centrifuge the plate at 450g for 3min 13. Using pipette tips, add 200µL of 0.3% Triton X-100 in PBS to each of the 18 wells and pipette up and down to resuspend the cell pellet 14. Keep the plate at room temperature and incubate for 10min 15. Centrifuge the plate at 450g for 3min 10
11 16. Using pipette tips, draw 200µL of supernatant from each of the 18 wells 17. Using pipette tips, add 200µL of PBS to each of the 18 wells 18. Centrifuge the plate at 450g for 3min 19. Using pipette tips, add 200µL of 0.5mg/mL ribonuclease A in PBS to each of the 18 wells and pipette up and down to resuspend the cell pellet 20. Place the plate inside an incubator at +37 C and incubate for 1h 21. Centrifuge the plate at 450g for 3min 22. Using pipette tips, draw 200µL of supernatant from each of the 18 wells 23. Using pipette tips, add 200µL of PBS to each of the 18 wells 24. Centrifuge the plate at 450g for 3min 25. Using pipette tips, add 200µL of 1:5,000 SYBR Green I in PBS to each of the 18 wells and pipette up and down to resuspend the cell pellet 26. Place the plate inside an incubator at +37 C and incubate for 1h 27. Centrifuge the plate at 450g for 3min 28. Using pipette tips, draw 200µL of supernatant from each of the 18 wells 29. Using pipette tips, add 200µL of PBS to each of the 18 wells 30. Centrifuge the plate at 450g for 3min 31. Repeat steps 28 to 30 twice more 32. Using pipette tips, draw 200µL of supernatant from each of the 18 wells 33. Using pipette tips, add 200µL of PBS to each of the 18 wells and pipette up and down to resuspend the cell pellet At this point, it is possible to store the plate protected from light at +4 C for later data acquisition with a flow cytometer. However, the plate has to be acquired within 7 days of the target RBC being initially stained. e. Acquisition on flow cytometer Regardless of the DNA dye used to stain the parasites, the plates are treated in the same manner throughout this section. 1- Label 18 wells of a 96-well, flat-bottom plate, on its lid, from A1 to F3 2- Using pipette tips, add 250µL of PBS to each of the 18 wells (a multichannel can be used to facilitate the work) 3- Using pipette tips, draw 25µL from each of the 18 wells of the plate containing the stained cells of the invasion assay, and add this to the corresponding wells on the new plate containing only PBS 4- Pipette up and down to mix the diluted cell suspension 5- Acquire the plate containing the diluted cell suspension on a flow cytometer fitted with the appropriate laser lines for the fluorescent cell label and DNA dye chosen 7,8 7 The diluted suspension will yield a sample speed of approximately 2,000 events per second when using the lowest sample rate setting. 8 Acquire 100,000 events to have a minimum of 50,000 target cells to analyse. 11
HIV-1 Pseudovirus Neutralization Protocol using Ghost cells
 HIV-1 Pseudovirus Neutralization Protocol using Ghost cells Institute of Tropical Medicine, Belgium Neutnet code: 6A Contact person: Leo Heyndrickx; LHeyndrickx@itg.be 1. AIM: This protocol describes the
HIV-1 Pseudovirus Neutralization Protocol using Ghost cells Institute of Tropical Medicine, Belgium Neutnet code: 6A Contact person: Leo Heyndrickx; LHeyndrickx@itg.be 1. AIM: This protocol describes the
ISOLATION AND BANKING OF PBMCS FROM HUMAN WHOLE BLOOD
 ISOLATION AND BANKING OF PBMCS FROM HUMAN WHOLE BLOOD Created by: Grace Teskey Date: February 2017 Bowdish Lab, McMaster University Hamilton, ON, Canada www.bowdish.ca Purpose: To isolate peripheral blood
ISOLATION AND BANKING OF PBMCS FROM HUMAN WHOLE BLOOD Created by: Grace Teskey Date: February 2017 Bowdish Lab, McMaster University Hamilton, ON, Canada www.bowdish.ca Purpose: To isolate peripheral blood
ThiolTracker Violet (Glutathione Detection Reagent)
 ThiolTracker Violet (Glutathione Detection Reagent) Catalog nos. T10095, T10096 Table 1. Contents and storage information. Material Amount Concentration Storage* Stability ThiolTracker Violet 180 assays
ThiolTracker Violet (Glutathione Detection Reagent) Catalog nos. T10095, T10096 Table 1. Contents and storage information. Material Amount Concentration Storage* Stability ThiolTracker Violet 180 assays
Application Note. Developed for: Aerius, Odyssey Classic, Odyssey CLx, and Odyssey Sa Infrared Imaging Systems
 Application Note On-Cell Western Plate-Based Assay for Targeted Near-Infrared-Labeled Optical Imaging Agent Development: Receptor-Based Binding and Competition Assays Developed for: Aerius, Odyssey Classic,
Application Note On-Cell Western Plate-Based Assay for Targeted Near-Infrared-Labeled Optical Imaging Agent Development: Receptor-Based Binding and Competition Assays Developed for: Aerius, Odyssey Classic,
Cell surface antibody staining for flow cytometry with optional amine reactive dye. Type SOP
 1 of 5 1.0 PURPOSE 1.1 This procedure describes the process for cell surface staining with fluorescent conjugated antibody reagents and optional amine reactive dye for viability measurement 2.0 SCOPE 2.1
1 of 5 1.0 PURPOSE 1.1 This procedure describes the process for cell surface staining with fluorescent conjugated antibody reagents and optional amine reactive dye for viability measurement 2.0 SCOPE 2.1
Standard Operating Procedure. Immunology
 Standard Operating Procedure Immunology SOP Ref No SOP title Category SOP-IU008 Human PBMC isolation, freezing and thawing procedures Laboratory Version 1.1 Page 1 of 12 Isolation, freezing and thawing
Standard Operating Procedure Immunology SOP Ref No SOP title Category SOP-IU008 Human PBMC isolation, freezing and thawing procedures Laboratory Version 1.1 Page 1 of 12 Isolation, freezing and thawing
DEPARTMENT PARASITOLOGY BPRC Rijswijk. Antigen Reversal Growth Inhibition Assay with pldh read-out
 Page 1 of 6 Index Page A.1. Aim 2 A.2. Principle 2 B. Samples 2 C. Controls 2 D.1. Equipment, instruments and materials 2 D.1.1 Equipment and instruments 2 D.1.2 Materials 3 E.1. Assay preparation 4 E.1.1.
Page 1 of 6 Index Page A.1. Aim 2 A.2. Principle 2 B. Samples 2 C. Controls 2 D.1. Equipment, instruments and materials 2 D.1.1 Equipment and instruments 2 D.1.2 Materials 3 E.1. Assay preparation 4 E.1.1.
Document Title Versalyse Whole Blood Staining Method Department. Type Status CA
 1 of 6 1.0 PURPOSE This procedure describes process for preparing whole blood samples and washed blood samples with the VersaLyse method for cell surface staining and with 7AAD for viability measurement.
1 of 6 1.0 PURPOSE This procedure describes process for preparing whole blood samples and washed blood samples with the VersaLyse method for cell surface staining and with 7AAD for viability measurement.
RPCI 001 v.003 In vitro Intracellular Cytokine Staining With and Without Stimulation
 Immune Tolerance Network RPCI 001 v.003 Author: Paul Wallace and Earl Timm, Director, RPCI Laboratory of Flow Cytometry Approved by: Paul Wallace, Director, RPCI Laboratory of Flow Cytometry 1.0 Title
Immune Tolerance Network RPCI 001 v.003 Author: Paul Wallace and Earl Timm, Director, RPCI Laboratory of Flow Cytometry Approved by: Paul Wallace, Director, RPCI Laboratory of Flow Cytometry 1.0 Title
ab Propidium Iodide Flow Cytometry Kit for Cell Cycle Analysis
 ab139418 Propidium Iodide Flow Cytometry Kit for Cell Cycle Analysis Instructions for Use To determine cell cycle status in tissue culture cell lines by measuring DNA content using a flow cytometer. This
ab139418 Propidium Iodide Flow Cytometry Kit for Cell Cycle Analysis Instructions for Use To determine cell cycle status in tissue culture cell lines by measuring DNA content using a flow cytometer. This
ab Propidium Iodide Flow Cytometry Kit for Cell Cycle Analysis
 ab139418 Propidium Iodide Flow Cytometry Kit for Cell Cycle Analysis Instructions for Use To determine cell cycle status in tissue culture cell lines by measuring DNA content using a flow cytometer. This
ab139418 Propidium Iodide Flow Cytometry Kit for Cell Cycle Analysis Instructions for Use To determine cell cycle status in tissue culture cell lines by measuring DNA content using a flow cytometer. This
SOPVII-7. Panel X: NK-characterization
 Created by judith.eckl Page 1 of 8 09/06/2011 SOPVII-7 Panel X: NK-characterization Date: Author: Petra Prinz, Judith Eckl Experimenter: Date: 08/06/2011 Experiment description: Version: 1.0 Start: End:
Created by judith.eckl Page 1 of 8 09/06/2011 SOPVII-7 Panel X: NK-characterization Date: Author: Petra Prinz, Judith Eckl Experimenter: Date: 08/06/2011 Experiment description: Version: 1.0 Start: End:
Fitness Measurements of Evolved Escherichia coli Migla Miskinyte and Isabel Gordo *
 Fitness Measurements of Evolved Escherichia coli Migla Miskinyte and Isabel Gordo * Evolutionary Biology Group, Instituto Gulbenkian de Ciência, Oeiras, Portugal *For correspondence: igordo@igc.gulbenkian.pt
Fitness Measurements of Evolved Escherichia coli Migla Miskinyte and Isabel Gordo * Evolutionary Biology Group, Instituto Gulbenkian de Ciência, Oeiras, Portugal *For correspondence: igordo@igc.gulbenkian.pt
For the removal or isolation of CD45 expressing cells via a negative or positive selection principle.
 PlusCellect Human CD45 Kit Catalog Number PLS1430 For the removal or isolation of CD45 expressing cells via a negative or positive selection principle. This kit contains sufficient reagents for 25 tests
PlusCellect Human CD45 Kit Catalog Number PLS1430 For the removal or isolation of CD45 expressing cells via a negative or positive selection principle. This kit contains sufficient reagents for 25 tests
L6 Cell Growth Protocol
 L6 Cell Growth Protocol Background: Parental L6 cells were subcloned for high fusion (1, 2). Materials: 1. α-minimal Essential Medium (α-mem) Life Technologies #12571-063 2. Fetal Bovine Serum (FBS) Life
L6 Cell Growth Protocol Background: Parental L6 cells were subcloned for high fusion (1, 2). Materials: 1. α-minimal Essential Medium (α-mem) Life Technologies #12571-063 2. Fetal Bovine Serum (FBS) Life
PD-1 [Biotinylated] : PD-L1 Inhibitor Screening ELISA Assay Pair
![PD-1 [Biotinylated] : PD-L1 Inhibitor Screening ELISA Assay Pair PD-1 [Biotinylated] : PD-L1 Inhibitor Screening ELISA Assay Pair](/thumbs/82/86220857.jpg) PD-1 [Biotinylated] : PD-L1 Inhibitor Screening ELISA Assay Pair Pack Size: 96 tests / 480 tests Catalog Number:EP-101 IMPORTANT: Please carefully read this manual before performing your experiment. For
PD-1 [Biotinylated] : PD-L1 Inhibitor Screening ELISA Assay Pair Pack Size: 96 tests / 480 tests Catalog Number:EP-101 IMPORTANT: Please carefully read this manual before performing your experiment. For
Sample Preparation Demonstrated Protocol
 10x Genomics Sample Preparation Demonstrated Protocol Removal of Dead Cells from Single Cell Suspensions for Single Cell RNA Sequencing NOTICES Notices Manual Part Number CG000093 Legal Notices Rev B 2017
10x Genomics Sample Preparation Demonstrated Protocol Removal of Dead Cells from Single Cell Suspensions for Single Cell RNA Sequencing NOTICES Notices Manual Part Number CG000093 Legal Notices Rev B 2017
Protocols for Neural Progenitor Cell Expansion and Dopaminergic Neuron Differentiation
 Protocols for Neural Progenitor Cell Expansion and Dopaminergic Neuron Differentiation In vitro neurological research presents many challenges due to the difficulty in establishing high-yield neuronal
Protocols for Neural Progenitor Cell Expansion and Dopaminergic Neuron Differentiation In vitro neurological research presents many challenges due to the difficulty in establishing high-yield neuronal
T ECHNICAL MANUAL. Culture of Human Mesenchymal Stem Cells Using MesenCult -XF Medium
 T ECHNICAL MANUAL Culture of Human Mesenchymal Stem Cells Using MesenCult -XF Medium i Table of Contents 1.0 Materials... 1 1.1 MesenCult -XF Medium and Required Products... 1 1.2 Additional Required
T ECHNICAL MANUAL Culture of Human Mesenchymal Stem Cells Using MesenCult -XF Medium i Table of Contents 1.0 Materials... 1 1.1 MesenCult -XF Medium and Required Products... 1 1.2 Additional Required
VDL300.3 PRODUCTION OF RETROVIRAL VECTOR BY TRANSIENT TRANSFECTION
 1. Purpose 1.1. The purpose of this protocol is to produce retrovirus from transfected plasmid DNA. 1.2. This procedure is routinely performed in the Vector Development Laboratory (VDL) following Good
1. Purpose 1.1. The purpose of this protocol is to produce retrovirus from transfected plasmid DNA. 1.2. This procedure is routinely performed in the Vector Development Laboratory (VDL) following Good
Ren Lab ENCODE in situ HiC Protocol for Tissue
 Ren Lab ENCODE in situ HiC Protocol for Tissue Pulverization, Crosslinking of Tissue Note: Ensure the samples are kept frozen on dry ice throughout pulverization. 1. Pour liquid nitrogen into a mortar
Ren Lab ENCODE in situ HiC Protocol for Tissue Pulverization, Crosslinking of Tissue Note: Ensure the samples are kept frozen on dry ice throughout pulverization. 1. Pour liquid nitrogen into a mortar
Nanobody Library Selection by MACS
 Nanobody Library Selection by MACS Introduction We have written the protocol below with the goal of making steps of in vitro nanobody selection as clear and broadly applicable as possible, but it is important
Nanobody Library Selection by MACS Introduction We have written the protocol below with the goal of making steps of in vitro nanobody selection as clear and broadly applicable as possible, but it is important
Evolution of Escherichia coli to Macrophage Cell Line Migla Miskinyte and Isabel Gordo *
 Evolution of Escherichia coli to Macrophage Cell Line Migla Miskinyte and Isabel Gordo * Evolutionary Biology Group, Instituto Gulbenkian de Ciência, Oeiras, Portugal *For correspondence: igordo@igc.gulbenkian.pt
Evolution of Escherichia coli to Macrophage Cell Line Migla Miskinyte and Isabel Gordo * Evolutionary Biology Group, Instituto Gulbenkian de Ciência, Oeiras, Portugal *For correspondence: igordo@igc.gulbenkian.pt
Whole Spleen Flow Cytometry Assay Cathy S. Yam *, Adeline M. Hajjar *
 Whole Spleen Flow Cytometry Assay Cathy S. Yam *, Adeline M. Hajjar * Department of Comparative Medicine, University of Washington, Seattle, USA *For correspondence: csyam@u.washington.edu; hajjar@uw.edu
Whole Spleen Flow Cytometry Assay Cathy S. Yam *, Adeline M. Hajjar * Department of Comparative Medicine, University of Washington, Seattle, USA *For correspondence: csyam@u.washington.edu; hajjar@uw.edu
Sample Preparation Demonstrated Protocol
 10x Genomics Sample Preparation Demonstrated Protocol Fresh Frozen Human Peripheral Blood Mononuclear Cells for Single Cell RNA Sequencing NOTICES Notices Manual Part Number CG00039 Legal Notices Rev B
10x Genomics Sample Preparation Demonstrated Protocol Fresh Frozen Human Peripheral Blood Mononuclear Cells for Single Cell RNA Sequencing NOTICES Notices Manual Part Number CG00039 Legal Notices Rev B
Human ipsc-derived Sensory Neuron Progenitors. For the generation of ipsc-derived sensory neurons
 Human ipsc-derived Sensory Neuron Progenitors For the generation of ipsc-derived sensory neurons Product Information Catalog. No. Product Name Format Stock Conc. Storage on Arrival Thawing Instructions
Human ipsc-derived Sensory Neuron Progenitors For the generation of ipsc-derived sensory neurons Product Information Catalog. No. Product Name Format Stock Conc. Storage on Arrival Thawing Instructions
Human ipsc-derived Sensory Neuron Progenitors. For the generation of ipsc-derived sensory neurons
 Human ipsc-derived Sensory Neuron Progenitors For the generation of ipsc-derived sensory neurons Additional Reagents Product Name Supplier Product Code Mitomycin C Sigma-Aldrich M4287 Store at 4 o C for
Human ipsc-derived Sensory Neuron Progenitors For the generation of ipsc-derived sensory neurons Additional Reagents Product Name Supplier Product Code Mitomycin C Sigma-Aldrich M4287 Store at 4 o C for
SOPVII-7. Panel VII: Treg-FACS-staining
 Created by judith.eckl Page 1 of 6 09/06/2011 SOPVII-7 Panel VII: Treg-FCS-staining Date: uthor: Judith Eckl Experimenter: Date: 08/05/2011 Experiment description: Version: 1.0 Start: End: ackground information:
Created by judith.eckl Page 1 of 6 09/06/2011 SOPVII-7 Panel VII: Treg-FCS-staining Date: uthor: Judith Eckl Experimenter: Date: 08/05/2011 Experiment description: Version: 1.0 Start: End: ackground information:
HUMAN IPSC CULTURE PROTOCOLS
 HUMAN IPSC CULTURE PROTOCOLS FOR TRAINING COURSE IXCELLS BIOTECHNOLOGIES USA, LLC 10340 CAMINO SANTA FE, SUITE C, SAN DIEGO, CA 92121 TABLE OF CONTENTS CHAPTER 1 BEFORE STARTING 2-7 SECTION 1.1 COATING
HUMAN IPSC CULTURE PROTOCOLS FOR TRAINING COURSE IXCELLS BIOTECHNOLOGIES USA, LLC 10340 CAMINO SANTA FE, SUITE C, SAN DIEGO, CA 92121 TABLE OF CONTENTS CHAPTER 1 BEFORE STARTING 2-7 SECTION 1.1 COATING
Xenograft Dissociation and Mouse Cell Depletion Using Miltenyi Equipment
 1. Purpose 1.1.1. The purpose of SOP 2.7 is to provide details on how to obtain a single cell suspension from xenografts using the Miltenyi gentlemacs Octo dissociator and then mouse cell depletion on
1. Purpose 1.1.1. The purpose of SOP 2.7 is to provide details on how to obtain a single cell suspension from xenografts using the Miltenyi gentlemacs Octo dissociator and then mouse cell depletion on
VDL102.3 Production of Adenovirus in 293 Cells
 1. Purpose CENTER FOR CELL & GENE THERAPY 1.1. The purpose of this protocol is to produce adenoviral vectors from transfected plasmid DNA. 1.2. This procedure is routinely performed in the Vector Development
1. Purpose CENTER FOR CELL & GENE THERAPY 1.1. The purpose of this protocol is to produce adenoviral vectors from transfected plasmid DNA. 1.2. This procedure is routinely performed in the Vector Development
Human ipsc-derived Renal Proximal Tubular Cells. Protocol version 1.0
 Human ipsc-derived Renal Proximal Tubular Cells Protocol version 1.0 Protocol version 1.0 Table of Contents Product Information 2 Preparation of Reagents 3 Culture of Human ipsc-derived Renal Proximal
Human ipsc-derived Renal Proximal Tubular Cells Protocol version 1.0 Protocol version 1.0 Table of Contents Product Information 2 Preparation of Reagents 3 Culture of Human ipsc-derived Renal Proximal
Chemicals Ordering Information
 SOP: Cultured cells: nuclei, DNaseI treatment, crosslinking, and preserving cells for RNA Date modified: 02/02/2012 Modified by: P. Sabo, T. Canfield, S. Hansen (UW) The following protocols for cultured
SOP: Cultured cells: nuclei, DNaseI treatment, crosslinking, and preserving cells for RNA Date modified: 02/02/2012 Modified by: P. Sabo, T. Canfield, S. Hansen (UW) The following protocols for cultured
Supplementary File 3: DNA and RNA isolation
 Supplementary File 3: DNA and RNA isolation Q-CROC-02 Biopsy protocol For the purposes of this protocol, four needle core biopsies (NCBs) of lymph node tissue are isolated from each patient using a 16G
Supplementary File 3: DNA and RNA isolation Q-CROC-02 Biopsy protocol For the purposes of this protocol, four needle core biopsies (NCBs) of lymph node tissue are isolated from each patient using a 16G
High-throughput automation with the Attune NxT Autosampler: consistent results across all wells and across plates
 APPLICATION NOTE Attune NxT Flow Cytometer with Autosampler High-throughput automation with the Attune NxT Autosampler: consistent results across all wells and across plates Introduction The emerging field
APPLICATION NOTE Attune NxT Flow Cytometer with Autosampler High-throughput automation with the Attune NxT Autosampler: consistent results across all wells and across plates Introduction The emerging field
Murine in vivo CD8 + T Cell Killing Assay Myoungjoo V. Kim 1*, Weiming Ouyang 2, Will Liao 3, Michael Q. Zhang 4 and Ming O. Li 5
 Murine in vivo CD8 + T Cell Killing Assay Myoungjoo V. Kim 1*, Weiming Ouyang 2, Will Liao 3, Michael Q. Zhang 4 and Ming O. Li 5 1 Department of Immunobiology, Yale University School of Medicine, New
Murine in vivo CD8 + T Cell Killing Assay Myoungjoo V. Kim 1*, Weiming Ouyang 2, Will Liao 3, Michael Q. Zhang 4 and Ming O. Li 5 1 Department of Immunobiology, Yale University School of Medicine, New
Supplementary Note 1. Materials and Methods for the BRET procedure. Solutions and reagents
 Supplementary Note 1 Materials and Methods for the BRET procedure Solutions and reagents PEI (1 mg/ml): The polyethyleneimine (PEI) powder (Polysciences, Inc.; MW ~25 KDa; cat#: 23966) is dissolved in
Supplementary Note 1 Materials and Methods for the BRET procedure Solutions and reagents PEI (1 mg/ml): The polyethyleneimine (PEI) powder (Polysciences, Inc.; MW ~25 KDa; cat#: 23966) is dissolved in
For simultaneous purification of genomic DNA and total RNA from the same animal cells or tissues
 1. TIANamp DNA/RNA Isolation Kit For simultaneous purification of genomic DNA and total RNA from the same animal cells or tissues www.tiangen.com/en RP090603 TIANamp DNA/RNA Isolation Kit Kit Contents
1. TIANamp DNA/RNA Isolation Kit For simultaneous purification of genomic DNA and total RNA from the same animal cells or tissues www.tiangen.com/en RP090603 TIANamp DNA/RNA Isolation Kit Kit Contents
MiraCell Endothelial Cells (from ChiPSC12) Kit
 Cat. # Y50055 For Research Use MiraCell Endothelial Cells (from ChiPSC12) Kit Product Manual Table of Contents I. Description...3 II. III. IV. Components...4 Storage...4 Preparation Before Use...4 V. Protocol...5
Cat. # Y50055 For Research Use MiraCell Endothelial Cells (from ChiPSC12) Kit Product Manual Table of Contents I. Description...3 II. III. IV. Components...4 Storage...4 Preparation Before Use...4 V. Protocol...5
PlusCellect. Human SSEA-4 + Pluripotent Stem Cells Isolation Kit. Catalog Number PLS1435. For the isolation or depletion of SSEA-4 expressing cells.
 PlusCellect Human SSEA-4 + Pluripotent Stem Cells Isolation Kit Catalog Number PLS1435 For the isolation or depletion of SSEA-4 expressing cells. This kit contains sufficient reagents for 25 tests (up
PlusCellect Human SSEA-4 + Pluripotent Stem Cells Isolation Kit Catalog Number PLS1435 For the isolation or depletion of SSEA-4 expressing cells. This kit contains sufficient reagents for 25 tests (up
UNIVERSITY OF ROCHESTER MEDICAL CENTER
 Date: 04 October 2005 Authors: Sally Quataert and Jennifer Hossler Approval: Sally A. Quataert 1. Purpose: To enumerate the total number of or IgM secreting plasma B cells in human peripheral blood mononuclear
Date: 04 October 2005 Authors: Sally Quataert and Jennifer Hossler Approval: Sally A. Quataert 1. Purpose: To enumerate the total number of or IgM secreting plasma B cells in human peripheral blood mononuclear
PlusCellect. Human CD34 + Hematopoietic Stem Cells Isolation Kit. Catalog Number PLS7227
 PlusCellect Human CD34 + Hematopoietic Stem Cells Isolation Kit Catalog Number PLS7227 For the isolation or depletion of human CD34 expressing cells. This kit contains sufficient reagents for 25 tests
PlusCellect Human CD34 + Hematopoietic Stem Cells Isolation Kit Catalog Number PLS7227 For the isolation or depletion of human CD34 expressing cells. This kit contains sufficient reagents for 25 tests
Immunofluorescent staining and flow cytometric analysis of cells
 UCAN-U 0003 Version 1 November 2010 Page 1 of 7 CMCI (Center for molecular and Cellular Intervention) University Medical Center Utrecht Written By Name Function Date Signature Mark Klein Lab manager Conformation
UCAN-U 0003 Version 1 November 2010 Page 1 of 7 CMCI (Center for molecular and Cellular Intervention) University Medical Center Utrecht Written By Name Function Date Signature Mark Klein Lab manager Conformation
INTRODUCTION TABLE OF CONTENTS. Seeding Cells in Hydrogel
 INTRODUCTION AIM 3D Cell Culture Chip allows culturing of cell aggregates or dispersed cells in a 3D hydrogel which are useful in various applications such as cell migration studies. This protocol covers
INTRODUCTION AIM 3D Cell Culture Chip allows culturing of cell aggregates or dispersed cells in a 3D hydrogel which are useful in various applications such as cell migration studies. This protocol covers
ExKine Total Protein Extraction Kit
 ExKine Total Protein Extraction Kit Item NO. KTP3006 Product Name ExKine Total Protein Extraction Kit ATTENTION For laboratory research use only. Not for clinical or diagnostic use Version 201811 TABLE
ExKine Total Protein Extraction Kit Item NO. KTP3006 Product Name ExKine Total Protein Extraction Kit ATTENTION For laboratory research use only. Not for clinical or diagnostic use Version 201811 TABLE
Cytotrophoblast Isolation from Term Placenta References:
 Cytotrophoblast Isolation from Term Placenta References: CY Kuo, et al. Placental Basement Membrane Proteins are Required for Effective Cytotrophoblast Invasion in a 3D Bioprinted Placenta Model. Journal
Cytotrophoblast Isolation from Term Placenta References: CY Kuo, et al. Placental Basement Membrane Proteins are Required for Effective Cytotrophoblast Invasion in a 3D Bioprinted Placenta Model. Journal
CFSE Cell Division Assay Kit
 CFSE Cell Division Assay Kit Catalog Number KA1302 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 General Information...
CFSE Cell Division Assay Kit Catalog Number KA1302 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 General Information...
TUNEL Apoptosis Detection Kit (Green Fluorescence)
 TUNEL Apoptosis Detection Kit (Green Fluorescence) Item NO. KTA2010 Product Name TUNEL Apoptosis Detection Kit (Green Fluorescence) ATTENTION For laboratory research use only. Not for clinical or diagnostic
TUNEL Apoptosis Detection Kit (Green Fluorescence) Item NO. KTA2010 Product Name TUNEL Apoptosis Detection Kit (Green Fluorescence) ATTENTION For laboratory research use only. Not for clinical or diagnostic
03/04/2011 Mouse Tn, Tr, and B cells: DNaseI treatment and preserving cells for RNA
 SOP: Mouse naïve Tn, regulatory Tr, and B cells: DNaseI treatment and preserving cells for RNA Date modified: 03/04/2011 Modified by: E. Giste/ T. Canfield (UW) Summary Mouse naïve Tn (CD4 + /CD25 - ),
SOP: Mouse naïve Tn, regulatory Tr, and B cells: DNaseI treatment and preserving cells for RNA Date modified: 03/04/2011 Modified by: E. Giste/ T. Canfield (UW) Summary Mouse naïve Tn (CD4 + /CD25 - ),
LHCN-M2 cell culture, differentiation treatment, and cross-linking protocol.
 Cell Growth Protocol and Differentiation treatment for the LHCN-M2 Cell Line From: HudsonAlpha/Caltech ENCODE group Date: February 17, 2011 Prepared by: Chun-Hong Zhu and Woodring E. Wright (typed by Brian
Cell Growth Protocol and Differentiation treatment for the LHCN-M2 Cell Line From: HudsonAlpha/Caltech ENCODE group Date: February 17, 2011 Prepared by: Chun-Hong Zhu and Woodring E. Wright (typed by Brian
Maintenance of Novus Cerebral Cortical Neurons
 Maintenance of Novus Cerebral Cortical Neurons Catalog nos. NBP2-31284, NBP2-31285, NBP2-31287 thru NBP2-31289, NBP2-31283, NBP2-31295 thru NBP2-31298, NBP2-31344, NBP2-31350 thru NBP2-31353, NBP2-31355
Maintenance of Novus Cerebral Cortical Neurons Catalog nos. NBP2-31284, NBP2-31285, NBP2-31287 thru NBP2-31289, NBP2-31283, NBP2-31295 thru NBP2-31298, NBP2-31344, NBP2-31350 thru NBP2-31353, NBP2-31355
Kit Components Product # (50 samples) Wash Solution A Elution Buffer B
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Cells and Tissue DNA Isolation Kit Product # 53100 Product Insert
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Cells and Tissue DNA Isolation Kit Product # 53100 Product Insert
Application Protocol: CD45 CK Immunostaining for patient blood
 REPRODUCTION AND USE This document is protected by copyright and it cannot be used or shared without permission from Vortex Biosciences, Inc. Such permission is given on condition that Vortex Biosciences
REPRODUCTION AND USE This document is protected by copyright and it cannot be used or shared without permission from Vortex Biosciences, Inc. Such permission is given on condition that Vortex Biosciences
Centre for Innovative Cancer Research, Ottawa Hospital Research Institute, Ottawa, Canada;
 Ex vivo Natural Killer Cell Cytotoxicity Assay Lee-Hwa Tai 1, Christiano Tanese de Souza 1, Andrew P. Makrigiannis 2 and Rebecca Ann C. Auer 3* 1 Centre for Innovative Cancer Research, Ottawa Hospital
Ex vivo Natural Killer Cell Cytotoxicity Assay Lee-Hwa Tai 1, Christiano Tanese de Souza 1, Andrew P. Makrigiannis 2 and Rebecca Ann C. Auer 3* 1 Centre for Innovative Cancer Research, Ottawa Hospital
Standard Operating Procedure
 Standard Operating Procedure Title Subtitle NANoREG Work package/task: Owner and co-owner(s) HTS Comet Assay with and without FPG - 20 wells Comet assay with and without FPG using Trevigen 20-well slides
Standard Operating Procedure Title Subtitle NANoREG Work package/task: Owner and co-owner(s) HTS Comet Assay with and without FPG - 20 wells Comet assay with and without FPG using Trevigen 20-well slides
3T3-L1 Differentiation Protocol
 3T3-L1 Differentiation Protocol Written by Eisuke Kato on 2013/10/09 MATERIALS Dulbecco's Modified Eagles Medium (D-MEM) (High Glucose) with L-Glutamine and Phenol Red High glucose (Wako Chem 044-29765,
3T3-L1 Differentiation Protocol Written by Eisuke Kato on 2013/10/09 MATERIALS Dulbecco's Modified Eagles Medium (D-MEM) (High Glucose) with L-Glutamine and Phenol Red High glucose (Wako Chem 044-29765,
Culture of Human ipsc-derived Neural Stem Cells in a 96-Well Plate Format
 Protocol supplement version 1.0 Culture of Human ipsc-derived Neural Stem Cells in a 96-Well Plate Format Protocol Supplement This protocol supplement is to be used in addition to the Neural Stem Cell
Protocol supplement version 1.0 Culture of Human ipsc-derived Neural Stem Cells in a 96-Well Plate Format Protocol Supplement This protocol supplement is to be used in addition to the Neural Stem Cell
Propagation of H7 hesc From: UW (John Stamatoyannopoulos) ENCODE group Date: 12/17/2009 Prepared By: S. Paige/S. Hansen (UW)
 Propagation of H7 hesc From: UW (John Stamatoyannopoulos) ENCODE group Date: 12/17/2009 Prepared By: S. Paige/S. Hansen (UW) Growth and Harvest Modifications Addendum to: Propagation of H7 hesc from UW
Propagation of H7 hesc From: UW (John Stamatoyannopoulos) ENCODE group Date: 12/17/2009 Prepared By: S. Paige/S. Hansen (UW) Growth and Harvest Modifications Addendum to: Propagation of H7 hesc from UW
CFSE Cell Division Assay Kit
 CFSE Cell Division Assay Kit Item No. 10009853 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS GENERAL INFORMATION
CFSE Cell Division Assay Kit Item No. 10009853 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS GENERAL INFORMATION
VDL106.3 LARGE-SCALE AMPLIFICATION AND PURIFICATION OF ADENOVIRAL VECTOR
 1. Purpose 1.1. The purpose of this protocol is to amplify an adenoviral vector from a crude lysate (final product in SOP:VDL105.2 Preparation of Adenoviral Vector Lysate) for production of high titer
1. Purpose 1.1. The purpose of this protocol is to amplify an adenoviral vector from a crude lysate (final product in SOP:VDL105.2 Preparation of Adenoviral Vector Lysate) for production of high titer
Proteasome Activity Fluorometric Assay Kit II (Cat. # J4120)
 Proteasome Activity Fluorometric Assay Kit II (Cat. # J4120) Each supplied substrate is sufficient for use in 250 X 100 µl reactions to monitor the chymotrypsin-like (Suc-LLVY- AMC), trypsin-like (Boc-LRR-AMC)
Proteasome Activity Fluorometric Assay Kit II (Cat. # J4120) Each supplied substrate is sufficient for use in 250 X 100 µl reactions to monitor the chymotrypsin-like (Suc-LLVY- AMC), trypsin-like (Boc-LRR-AMC)
Long-lived protein degradation assay
 Long-lived protein degradation assay Shun Kageyama, Takashi Ueno, Masaaki Komatsu METHOD Pulse and chase 1. Seed cells in 24-well plates in regular medium supplemented with 10% (v/v) heat-inactivated FBS
Long-lived protein degradation assay Shun Kageyama, Takashi Ueno, Masaaki Komatsu METHOD Pulse and chase 1. Seed cells in 24-well plates in regular medium supplemented with 10% (v/v) heat-inactivated FBS
(Spin Column) For purification of DNA and RNA from formalin-fixed, paraffin-embedded. tissue sections. Instruction for Use. For Research Use Only
 AmoyDx FFPE DNA/RNA Kit (Spin Column) For purification of DNA and RNA from formalin-fixed, paraffin-embedded tissue sections Instruction for Use For Research Use Only Instruction Version: B1.0 Revision
AmoyDx FFPE DNA/RNA Kit (Spin Column) For purification of DNA and RNA from formalin-fixed, paraffin-embedded tissue sections Instruction for Use For Research Use Only Instruction Version: B1.0 Revision
ab CytoPainter Cell Plasma Membrane Staining Kit Deep Red Fluorescence
 Version 1 Last updated 27 April 2017 ab219942 CytoPainter Cell Plasma Membrane Staining Kit Deep Red Fluorescence For staining cell plasma membrane in live cells using our proprietary deep red fluorescence
Version 1 Last updated 27 April 2017 ab219942 CytoPainter Cell Plasma Membrane Staining Kit Deep Red Fluorescence For staining cell plasma membrane in live cells using our proprietary deep red fluorescence
VDL602.2 RAPID ASSAY FOR DETERMINING ADENOVIRAL VECTOR TITER
 1. Purpose 1.1. The purpose of this protocol is to determine the number of infectious adenoviral particles. 1.2. The starting material is purified adenoviral vectors. 1.3. This protocol is based upon the
1. Purpose 1.1. The purpose of this protocol is to determine the number of infectious adenoviral particles. 1.2. The starting material is purified adenoviral vectors. 1.3. This protocol is based upon the
Amaxa Cell Line Nucleofector Kit L
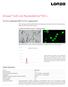 Amaxa Cell Line Nucleofector Kit L For 3T3-L1 (adipocytes) [ATCC CL-173, cryopreserved] Mouse embryonal fibroblast, differentiated into adipocytes; Fibroblast-like cells before differentiation; adipocyte-like
Amaxa Cell Line Nucleofector Kit L For 3T3-L1 (adipocytes) [ATCC CL-173, cryopreserved] Mouse embryonal fibroblast, differentiated into adipocytes; Fibroblast-like cells before differentiation; adipocyte-like
Metzger Lab Protocol Book EF May 2003
 I. Preparation for cell isolation: A. Make sure the following items are autoclaved: 1. Glassware* (1) 100 ml beaker (3) wide mouthed 1 liter bottles (1) 100 mm glass petri dish (1) 60 mm sigma coated petri
I. Preparation for cell isolation: A. Make sure the following items are autoclaved: 1. Glassware* (1) 100 ml beaker (3) wide mouthed 1 liter bottles (1) 100 mm glass petri dish (1) 60 mm sigma coated petri
Protocol for the Co-culture of ipsc-derived Microglia with ipsc-derived Cerebral Cortical Neural Culture
 Protocol for the Co-culture of ipsc-derived Microglia with ipsc-derived Cerebral Cortical Neural Culture Required reagents Product code ax0031a + b ax0041 ax0047 ax68168 (5 mg) ax0660 ax0016, ax0111 (AD
Protocol for the Co-culture of ipsc-derived Microglia with ipsc-derived Cerebral Cortical Neural Culture Required reagents Product code ax0031a + b ax0041 ax0047 ax68168 (5 mg) ax0660 ax0016, ax0111 (AD
Technical Bulletin. Caspase 10 Fluorescein (FLICA) Assay. Catalog Number CS0310 Storage Temperature 2-8 C
 Caspase 10 Fluorescein (FLICA) Assay Catalog Number CS0310 Storage Temperature 2-8 C Technical Bulletin Product Description Caspases are detected by immunoprecipitation, immunoblotting with caspase specific
Caspase 10 Fluorescein (FLICA) Assay Catalog Number CS0310 Storage Temperature 2-8 C Technical Bulletin Product Description Caspases are detected by immunoprecipitation, immunoblotting with caspase specific
Laboratory protocol for manual purification of DNA from whole sample
 Laboratory protocol for manual purification of DNA from whole sample Ethanol precipitation protocol and prepit L2P reagent for the purification of genomic DNA from the Oragene and ORAcollect families of
Laboratory protocol for manual purification of DNA from whole sample Ethanol precipitation protocol and prepit L2P reagent for the purification of genomic DNA from the Oragene and ORAcollect families of
Human Gastric Organoid Culture Protocol
 PROTOCOL Human Gastric Organoid Culture Protocol This protocol provides a procedure for subculturing normal human gastric organoids. This protocol was modified from the submerged method described in Bartfeld,
PROTOCOL Human Gastric Organoid Culture Protocol This protocol provides a procedure for subculturing normal human gastric organoids. This protocol was modified from the submerged method described in Bartfeld,
ab Comet Assay Kit (3- well slides)
 Version 1 Last updated 2 November 2018 ab238544 Comet Assay Kit (3- well slides) For the measurement of cellular DNA damage. This product is for research use only and is not intended for diagnostic use.
Version 1 Last updated 2 November 2018 ab238544 Comet Assay Kit (3- well slides) For the measurement of cellular DNA damage. This product is for research use only and is not intended for diagnostic use.
2 x 0.5ml. STORAGE CONDITIONS The kit is shipped at ambient temperature. Upon arrival, store the kit components as recommended on the reagent label.
 305PR 01 G-Biosciences, St Louis, MO. USA 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name OmniPrep For High Quality Genomic DNA Extraction INTRODUCTION
305PR 01 G-Biosciences, St Louis, MO. USA 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name OmniPrep For High Quality Genomic DNA Extraction INTRODUCTION
Plus Blood Genomic DNA Purification Kit
 Plus Blood Genomic DNA Purification Kit Cat #:DP023P/ DP023P-150 Size:50/150 reactions Store at RT For research use only 1 Description: The Plus Blood Genomic DNA Purification Kit provides a rapid, simple
Plus Blood Genomic DNA Purification Kit Cat #:DP023P/ DP023P-150 Size:50/150 reactions Store at RT For research use only 1 Description: The Plus Blood Genomic DNA Purification Kit provides a rapid, simple
TECHNICAL BULLETIN NUCLEI EZ PREP NUCLEI ISOLATION KIT. Product Number NUC-101 Store at 2-8 C
 NUCLEI EZ PREP NUCLEI ISOLATION KIT Product Number NUC-101 Store at 2-8 C TECHNICAL BULLETIN Product Description Sigma s Nuclei EZ Prep Kit is designed for the rapid isolation of nuclei from mammalian
NUCLEI EZ PREP NUCLEI ISOLATION KIT Product Number NUC-101 Store at 2-8 C TECHNICAL BULLETIN Product Description Sigma s Nuclei EZ Prep Kit is designed for the rapid isolation of nuclei from mammalian
Cell Cycle Phase Determination Kit
 Cell Cycle Phase Determination Kit Catalog Number KA1301 100 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 General
Cell Cycle Phase Determination Kit Catalog Number KA1301 100 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 General
Guidelines for WTC Derived AICS hipsc Lines Scale Up & Banking (upto 80 vials)
 Version 1.2 2017 Allen Institute for Cell Science Guidelines for WTC Derived AICS hipsc Lines Scale Up & Banking (upto 80 vials) Note: The following protocol is for banking ~60-80 vials of cells (1 million/vial)
Version 1.2 2017 Allen Institute for Cell Science Guidelines for WTC Derived AICS hipsc Lines Scale Up & Banking (upto 80 vials) Note: The following protocol is for banking ~60-80 vials of cells (1 million/vial)
Title: Micro-immunofluorescence Staining for Multichromatic FACS Analysis with Human Single Cell Suspensions
 Date: 27-December-2006 Authors: Sally A. Quataert and Matthew Cochran Approved: Title: Micro-immunofluorescence Staining for Multichromatic FACS Analysis with Human Single Cell Suspensions Purpose and
Date: 27-December-2006 Authors: Sally A. Quataert and Matthew Cochran Approved: Title: Micro-immunofluorescence Staining for Multichromatic FACS Analysis with Human Single Cell Suspensions Purpose and
with Intracellular-Compatible Universal Cell Capture Kit
 MAN-10067-01 with Intracellular-Compatible Universal Cell Capture Kit In this workflow, cells are collected and then bound to the Universal Cell Capture Beads; then the RNA and Protein samples are prepared
MAN-10067-01 with Intracellular-Compatible Universal Cell Capture Kit In this workflow, cells are collected and then bound to the Universal Cell Capture Beads; then the RNA and Protein samples are prepared
E.Z.N.A. FFPE RNA Kit. R preps R preps R preps
 E.Z.N.A. FFPE RNA Kit R6954-00 5 preps R6954-01 50 preps R6954-02 200 preps January 2015 E.Z.N.A. FFPE RNA Kit Table of Contents Introduction and Overview...2 Kit Contents/Storage and Stability...3 Preparing
E.Z.N.A. FFPE RNA Kit R6954-00 5 preps R6954-01 50 preps R6954-02 200 preps January 2015 E.Z.N.A. FFPE RNA Kit Table of Contents Introduction and Overview...2 Kit Contents/Storage and Stability...3 Preparing
Important Note: FH and media must be at room temperature (20-24 C) for accurate gradient formation
 Procedure: Lymphocyte Isolation Using a Density Gradient Materials: Method: Procedure: Lymphocyte isolation using density gradient centrifugation allows the separation of lymphocytes for crossmatch testing
Procedure: Lymphocyte Isolation Using a Density Gradient Materials: Method: Procedure: Lymphocyte isolation using density gradient centrifugation allows the separation of lymphocytes for crossmatch testing
Immunofluorescence Staining Protocol for 3 Well Chamber, removable
 Immunofluorescence Staining Protocol for 3 Well Chamber, removable This Application Note presents a simple protocol for the cultivation, fixation, and staining of cells using the 3 Well Chamber, removable.
Immunofluorescence Staining Protocol for 3 Well Chamber, removable This Application Note presents a simple protocol for the cultivation, fixation, and staining of cells using the 3 Well Chamber, removable.
USER GUIDE. HiYield TM Total RNA Extraction Kit
 USER GUIDE HiYield TM Total RNA Extraction Kit Precautions I) Handling Requirements Do not use a kit after its expiration date has passed. Some reagents contain the hazardous compounds guanidine thiocyanate
USER GUIDE HiYield TM Total RNA Extraction Kit Precautions I) Handling Requirements Do not use a kit after its expiration date has passed. Some reagents contain the hazardous compounds guanidine thiocyanate
Live and Dead Cell Assay
 ab115347 Live and Dead Cell Assay Instructions for Use Differential fluorescent labeling of live and dead cells This product is for research use only and is not intended for diagnostic use. Last Updated
ab115347 Live and Dead Cell Assay Instructions for Use Differential fluorescent labeling of live and dead cells This product is for research use only and is not intended for diagnostic use. Last Updated
Purification of cytoplasmic RNA from animal cells using the RNeasy Mini Kit
 QIAGEN Supplementary Protocol: Purification of cytoplasmic RNA from animal cells using the RNeasy Mini Kit This protocol requires the RNeasy Mini Kit. IMPORTANT: Please consult the Safety Information and
QIAGEN Supplementary Protocol: Purification of cytoplasmic RNA from animal cells using the RNeasy Mini Kit This protocol requires the RNeasy Mini Kit. IMPORTANT: Please consult the Safety Information and
PrepSEQ Sample Preparation Kits for Mycoplasma, MMV, and Vesivirus
 QUICK REFERENCE PrepSEQ Sample Preparation Kits for Mycoplasma, MMV, and Vesivirus Manual sample preparation protocols for Mycoplasma, MMV, and Vesivirus detection Catalog Numbers 445 and 4443789 Pub.
QUICK REFERENCE PrepSEQ Sample Preparation Kits for Mycoplasma, MMV, and Vesivirus Manual sample preparation protocols for Mycoplasma, MMV, and Vesivirus detection Catalog Numbers 445 and 4443789 Pub.
Phosphate buffered saline (PBS) for washing the cells TE buffer (nuclease-free) ph 7.5 for resuspending the SingleShot RNA control template
 Catalog # Description 172-5085 SingleShot SYBR Green Kit, 100 x 50 µl reactions For research purposes only. Introduction The SingleShot SYBR Green Kit prepares genomic DNA (gdna) free RNA directly from
Catalog # Description 172-5085 SingleShot SYBR Green Kit, 100 x 50 µl reactions For research purposes only. Introduction The SingleShot SYBR Green Kit prepares genomic DNA (gdna) free RNA directly from
Cardiomyocyte Differentiation Methods
 Version 1.0 2018 Allen Institute for Cell Science Cardiomyocyte Differentiation Methods The methods described below were adapted from the small molecule protocol described in Lian et al. 1, and have been
Version 1.0 2018 Allen Institute for Cell Science Cardiomyocyte Differentiation Methods The methods described below were adapted from the small molecule protocol described in Lian et al. 1, and have been
Cells and Tissue DNA Isolation Kit (Magnetic Bead System) 50 Preps Product # 59100
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Cells and Tissue DNA Isolation Kit (Magnetic Bead System) 50 Preps
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Cells and Tissue DNA Isolation Kit (Magnetic Bead System) 50 Preps
General Guidelines for Handling Feeder Free Human ipscs. Cells provided were cultured in the presence of Penicillin and Streptomycin.
 General Guidelines for Handling Feeder Free Human ipscs This document provides guidance on how to resuscitate, culture and cryopreserve human induced pluripotent stem cells (ipscs) supplied by the Human
General Guidelines for Handling Feeder Free Human ipscs This document provides guidance on how to resuscitate, culture and cryopreserve human induced pluripotent stem cells (ipscs) supplied by the Human
HIC STANDARD OPERATING PROCEDURE: Immunofluorescence Staining for Multichromatic FACS Analysis with Human Single Cell Suspensions
 Date: 15-December-2005 Authors: Sally A. Quataert and Matthew Cochran Approved: Sally A. Quataert Purpose and Scope: This procedure describes a method for multichromatic staining of human single cell suspensions
Date: 15-December-2005 Authors: Sally A. Quataert and Matthew Cochran Approved: Sally A. Quataert Purpose and Scope: This procedure describes a method for multichromatic staining of human single cell suspensions
Synchronisation of P. falciparum v1.1. Procedure. In vitro Module WorldWide Antimalarial Resistance Network (WWARN)
 Synchronisation of P. falciparum v1.1 Procedure In vitro Module WorldWide Antimalarial Resistance Network (WWARN) Suggested citation: In vitro Module, WWARN. 2010. Synchronisation of P. falciparum. WWARN
Synchronisation of P. falciparum v1.1 Procedure In vitro Module WorldWide Antimalarial Resistance Network (WWARN) Suggested citation: In vitro Module, WWARN. 2010. Synchronisation of P. falciparum. WWARN
(Spin Column) For purification of DNA and RNA from formalin-fixed, paraffin-embedded tissue sections. Instruction for Use
 AmoyDx FFPE DNA/RNA Kit (Spin Column) For purification of DNA and RNA from formalin-fixed, paraffin-embedded tissue sections Instruction for Use Instruction Version: B1.3 Revision Date: September 2015
AmoyDx FFPE DNA/RNA Kit (Spin Column) For purification of DNA and RNA from formalin-fixed, paraffin-embedded tissue sections Instruction for Use Instruction Version: B1.3 Revision Date: September 2015
IncuCyte Phagocytosis Assay
 IncuCyte Phagocytosis Assay For quantification of phagocytosis of apoptotic and non-apoptotic cell This protocol is intended for the measurement of both apoptotic (efferocytosis) and non-apoptotc phagocytosis
IncuCyte Phagocytosis Assay For quantification of phagocytosis of apoptotic and non-apoptotic cell This protocol is intended for the measurement of both apoptotic (efferocytosis) and non-apoptotc phagocytosis
Propidium Iodide Solution
 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Propidium Iodide Solution (1mg/ml in deionized water) (Cat. # 786 1272, 786 1273) think proteins!
G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Propidium Iodide Solution (1mg/ml in deionized water) (Cat. # 786 1272, 786 1273) think proteins!
XIT Genomic DNA from Cells
 464PR G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name XIT Genomic DNA from Cells For the Isolation of Genomic DNA from Cultured Cells (Cat.
464PR G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name XIT Genomic DNA from Cells For the Isolation of Genomic DNA from Cultured Cells (Cat.
3D Endothelial Pericyte Coculturing Kit Product Description Kit Components
 3D Endothelial Pericyte Coculturing Kit (3D-EPC) Cat #8728 Product Description Blood vessels are responsible for transporting blood throughout the body as part of the circulatory system. Their main components
3D Endothelial Pericyte Coculturing Kit (3D-EPC) Cat #8728 Product Description Blood vessels are responsible for transporting blood throughout the body as part of the circulatory system. Their main components
Reagents Description Storage
 MAN-10054-03 In this workflow, samples will undergo lysis using a detergent-containing buffer followed by detergent removal using spin columns. The lysate will be quantified for total protein concentration,
MAN-10054-03 In this workflow, samples will undergo lysis using a detergent-containing buffer followed by detergent removal using spin columns. The lysate will be quantified for total protein concentration,
