Aspiring to Measure Quality Culture
|
|
|
- Peregrine Shelton
- 5 years ago
- Views:
Transcription
1 Aspiring to Measure Quality Culture Denyse Baker PDA Director of Science and Regulatory Affairs Pharmaceutical Quality Congress The Unintended Consequences of Quality June 14-16, Bethesda, MD 1
2 Parenteral Drug Association Founded in 1946 Mission: To advance pharmaceutical and biopharmaceutical manufacturing science and regulation so members can better serve patients. Science Based: Science is the foundation of our organization. We utilize a scientific approach to meet challenges and continuously improve. It is not subjective or emotional, but rather a logical, open, rational and transparent process. 2
3 Why Should I Focus on Quality Culture? A company with a highly developed culture of quality spends, on average, $350 million less annually fixing mistakes than a company with a poorly developed one. Regulators are increasingly focused on quality culture as they recognize its impact on the production of quality products. We have the privilege of serving patients. Harvard Business Review April 2014
4 PDA s 2014 Quality Metric Conference Can you objectively measure Quality Culture? Quality Culture is a set of behaviors, beliefs, values, attitudes and governance (subjective) Mature Quality Attributes goes beyond Traditional Quality Systems to create a framework for a strong Quality Culture (objective) Hypothesis Mature Quality Attributes have a strong relationship to positive Quality Culture behavior Identify a set of Mature Quality Attributes that are a surrogate for Quality Culture Behaviors
5 Relationship established with Survey Results Behaviors Behaviors Mature Quality Attributes Enhanced Quality Systems (Q8, 9, 10, 11) Risk management, MR, QbD, etc. Other Systems Quality goals, rewards & recognition, personnel development, etc. Mature Quality Attribute Higher Quality Maturity is accompanied with Higher Quality Behavior.
6 St. Gallen confirms PDA s Survey Outcome PDA Survey Analysis 2014 St. Gallen Analysis 2017 R 2 = 0.34 R 2 = pharmaceutical sites of different size and focus within St. Gallen database confirm PDA 96% of variability of Quality Behavior can be explained by the Quality Maturity Attributes Thomas Friedli, PDA 2017 Metrics Conference
7 Quality Culture Model and Tool Objective June 2015 Develop a quality culture maturity model as an assessment tool to be used during audits Simple Objective and verifiable Pilot Launch May 2016 On-Site Self-Assessment Tool and Training Internal Sites, Suppliers, CMOs All Employee Survey Pilot Data, Benchmarking, Feedback
8 On-site assessment tool 5 Categories Leadership Commitment Communication & Collaboration Employee Ownership Continuous Improvement Technical Excellence 8
9 On-site assessment tool 12 Attributes Leadership Commitment Communication & Collaboration Employee Ownership 1. Leadership Commitment to Quality 3. Quality Communications 2. Enabling Capable Resources 4. Collaboration with Auditors 5. Understanding Quality Goals 6. Safety Culture Continuous Improvement Technical Excellence 7. CAPA robustness 11.Utilization of new proven technologies 8. Management Review and Metrics 12. Maturity of Systems 9. Clear Quality Objectives 10.Internal Stakeholder Feedback 9
10 On-site assessment tool 24 Sub Attributes Leadership Commitment 1. Leadership Commitment to Quality Accountability and Quality Planning 2. Enabling Capable Resources Feedback and Coaching Training & staff development Rewards and Recognition Continuous Improvement Communication & Collaboration 3. Quality Communications Quality Communications 4. Collaboration with Auditors Collaboration Operations Readiness & Knowledge Behaviors Technical Excellence Employee Ownership 5. Understanding Quality Goals Impact on Product Quality Patient Impact 6. Safety Culture EHS Program Targets 7. CAPA robustness Root Cause Human Error 8. Management Review and Metrics Management Reviews Metrics 9. Clear Quality Objectives Continuous Improvement 10. Internal Stakeholder Feedback Internal Stakeholder Feedback Quality Culture Survey 11. Utilization of new proven technologies Manufacturing Technologies New Technology 12. Maturity of Systems QMS Processes Maturity Model Responsibilities 10
11 Sample of the PDA Quality Culture Tool 5. Understanding Quality Goals Impact on Product Quality Patient Impact 11
12 Analysis 2 weeks Assessment 1 3 days Preparation 2 3 weeks Quality Click to edit Culture text Assessment Process Pre-assessment questionnaire Planning (interviews or panels) Pre-scoring Kickoff by site leadership Perform assessment & document results Daily wrap with site leadership Send data to PDA & provide feedback Continuous improvement discussion with site PDA data analysis 12
13 Pilot Program Participants A cross Section of the Pharma Industry: ~40 sites from 20 firms, 64 assessors Business Function Product Class Site Locations Other
14 Feedback from participants Quality culture assessment without audit mindset encouraged openness The tool provided clear framework and scoring method Enable firms to follow a structured way to assess quality culture Tool is valuable to align expectations between CMO and firms Tool provides roadmap for improvement 14
15 Lessons Learned Through The Pilot Site leadership to set expectation of the assessment Differentiate the quality culture assessment vs. GMP audit Advance planning is key! Assessor s skill sets the tone Survey Translation for sites that don t speak English Best to have more than one assessor at the site 15
16 Where is PDA in the Quality Culture Program? Define Develop assessment tool Control Improve Measure Analyze On-site assessment & survey tools PILOT ANALYSIS UNDERWAY 16
17 Future work Wrap up pilot and present findings at September PDA/FDA Joint Conference Launch tool Q PDA database with assessment data for benchmarking Expand assessment tool for suppliers and corporate functions Workshop/Users Group for sharing practical experiences in driving improvement using the tool. 17
18 PDA Quality Culture Team Cylia Chen (Amgen) Team Lead Chuck Bornhoeft (Upsher-Smith) Rick Burdick (Consultant) Anne Eickhoff (GSK Vaccines) Machelle Eppler (Patheon) Matija Gabrovsek (Novartis) Sandra Lueken (MedImmune/AZ) Steve Mendivil (Amgen) Pritesh Patel (Novartis) Brianna Peterson (Boehringer Ingelheim) Sue Schniepp (Regulatory Compliance Associates) Jan Paul Zonnenberg (PricewaterhouseCoopers) Tara Gooen (FDA) Gerald Heddell (MHRA) PDA Staff: Denyse Baker, Rich Levy, David Talmage
PDA Quality Culture Program Overview
 PDA Quality Culture Program Overview Are you ready to assess your site s quality culture? Cylia Chen, PDA Quality Culture team PDA SoCal Chapter Nov. 2016 1 What is FDA (and industry) Striving for? A maximally
PDA Quality Culture Program Overview Are you ready to assess your site s quality culture? Cylia Chen, PDA Quality Culture team PDA SoCal Chapter Nov. 2016 1 What is FDA (and industry) Striving for? A maximally
Tuesday, February 21, :15 a.m. 5:30 p.m. Registration Open. 8:15 a.m. 8:30 a.m. Continental Breakfast
 2017 PDA Pharmaceutical Quality Metrics and Quality Culture Conference February 21 22, 2017 Bethesda North Marriott Hotel & Conference Center Bethesda, MD As of January 30, 2017 Tuesday, February 21, 2017
2017 PDA Pharmaceutical Quality Metrics and Quality Culture Conference February 21 22, 2017 Bethesda North Marriott Hotel & Conference Center Bethesda, MD As of January 30, 2017 Tuesday, February 21, 2017
PDA: A Global Association
 Quality Culture Metrics? PDA s Seeking Answers PDA: A Global Association Steven Mendivil PDA Quality Metric Task Force Leader 1 FDA is Interested in Quality Metrics FDASIA (7/12) - Section 706 Opportunity
Quality Culture Metrics? PDA s Seeking Answers PDA: A Global Association Steven Mendivil PDA Quality Metric Task Force Leader 1 FDA is Interested in Quality Metrics FDASIA (7/12) - Section 706 Opportunity
Building Quality Culture and Capabilities
 Building Quality Culture and Capabilities IPA Advanced GMP Workshop November 2018 McKinsey & Company 1 The typical questions we face on quality culture : 1 2 How much does it matter? Can you measure it?
Building Quality Culture and Capabilities IPA Advanced GMP Workshop November 2018 McKinsey & Company 1 The typical questions we face on quality culture : 1 2 How much does it matter? Can you measure it?
Digital Transformation Blueprint. The Dawn of the Digital Industrial
 Digital Transformation Blueprint The Dawn of the Digital Industrial The age of the Industrial Internet of Things is upon us Let GE Digital help get you get started. Build your Digital Transformation Blueprint
Digital Transformation Blueprint The Dawn of the Digital Industrial The age of the Industrial Internet of Things is upon us Let GE Digital help get you get started. Build your Digital Transformation Blueprint
Strategies to Revise Quality Policy and Quality Objectives. Eileen Cortes February 22, 2017
 Strategies to Revise Quality Policy and Quality Objectives Eileen Cortes February 22, 2017 Agenda Elements of Compliance Quality Policy Outline Quality Strategy and Objectives Case Study Open Discussion
Strategies to Revise Quality Policy and Quality Objectives Eileen Cortes February 22, 2017 Agenda Elements of Compliance Quality Policy Outline Quality Strategy and Objectives Case Study Open Discussion
FDA s Draft Guidance Request for Quality Metrics: What All Drug and Biologics Manufacturers Need to Know June 2017
 FDA s Draft Guidance Request for Quality Metrics: What All Drug and Biologics Manufacturers Need to Know June 2017 Discussion Topics Quality Concepts The Journey The Guidance Details Industry Responses
FDA s Draft Guidance Request for Quality Metrics: What All Drug and Biologics Manufacturers Need to Know June 2017 Discussion Topics Quality Concepts The Journey The Guidance Details Industry Responses
AGENDA. 12:00 p.m. 1:00 p.m. Registration 1:00 p.m. 4:00 p.m. Pre-Conference Workshop: Data Integrity Problems on the Rise
 AGENDA Pre-Conference Workshop, June 14 12:00 p.m. 1:00 p.m. Registration 1:00 p.m. 4:00 p.m. Pre-Conference Workshop: Data Integrity Problems on the Rise Data integrity issues have become a common and
AGENDA Pre-Conference Workshop, June 14 12:00 p.m. 1:00 p.m. Registration 1:00 p.m. 4:00 p.m. Pre-Conference Workshop: Data Integrity Problems on the Rise Data integrity issues have become a common and
Click to edit Master title style
 Measuring Performance, Driving Excellence Click to edit Master title style ISPE Nordic, Stockholm WTC November 26 th 2015 Dr. Nuala Calnan, Research Fellow, Dublin Institute of Technology 11/26/2015 1
Measuring Performance, Driving Excellence Click to edit Master title style ISPE Nordic, Stockholm WTC November 26 th 2015 Dr. Nuala Calnan, Research Fellow, Dublin Institute of Technology 11/26/2015 1
Effective Quality Oversight of Pharmaceutical Contract Manufacturing Organizations (COM) COURSE DESCRIPTION
 Effective Quality Oversight of Pharmaceutical Contract Manufacturing Organizations (COM) COURSE DESCRIPTION The globalization of the pharmaceutical supply chain has led increased use of Contract Manufacturing
Effective Quality Oversight of Pharmaceutical Contract Manufacturing Organizations (COM) COURSE DESCRIPTION The globalization of the pharmaceutical supply chain has led increased use of Contract Manufacturing
Quality Metrics Journey to Excellence. Steve Greer Corp QA External Engagement Leader Procter & Gamble
 Quality Metrics Journey to Excellence Steve Greer Corp QA External Engagement Leader Procter & Gamble Acknowledgements Tara Gooen Bizjak FDA, CDER/OPQ/OPPQ Ashley Boam FDA, CDER/OPQ/OPPQ Alex Viehman FDA,
Quality Metrics Journey to Excellence Steve Greer Corp QA External Engagement Leader Procter & Gamble Acknowledgements Tara Gooen Bizjak FDA, CDER/OPQ/OPPQ Ashley Boam FDA, CDER/OPQ/OPPQ Alex Viehman FDA,
Effective Quality Oversight of Pharmaceutical Contract Manufacturing Organizations (CMOs)
 Effective Quality Oversight of Pharmaceutical Contract Manufacturing Organizations (CMOs) *** LIMITED TIME OFFER: FREE $100 AMAZON GIFT CARD! *** REGISTER TODAY! The globalization of the pharmaceutical
Effective Quality Oversight of Pharmaceutical Contract Manufacturing Organizations (CMOs) *** LIMITED TIME OFFER: FREE $100 AMAZON GIFT CARD! *** REGISTER TODAY! The globalization of the pharmaceutical
CMC Activities for Commercialization: PAI, QMS, and BLA/NDA. NEPDA Dinner Meeting Cambridge, MA 16 May 2018
 CMC Activities for Commercialization: PAI, QMS, and BLA/NDA Advancing from Clinical to Commercial The Commercialization Roadmap NEPDA Dinner Meeting Cambridge, MA 16 May 2018 Standards Certification Education
CMC Activities for Commercialization: PAI, QMS, and BLA/NDA Advancing from Clinical to Commercial The Commercialization Roadmap NEPDA Dinner Meeting Cambridge, MA 16 May 2018 Standards Certification Education
SUPPLIER PERFORMANCE PROGRAM
 SUPPLIER PERFORMANCE PROGRAM Without question, supplier relationships have a significant impact on AbbVie's corporate success. AbbVie Purchasing and Supplier Management has established a formal metrics
SUPPLIER PERFORMANCE PROGRAM Without question, supplier relationships have a significant impact on AbbVie's corporate success. AbbVie Purchasing and Supplier Management has established a formal metrics
Can Quality Solve the Equation?
 Pricing Authority Expectations Supply Chain Complexity Counterfeiting Can Quality Solve the Equation? Quality Drugs to Patients & An Engaged Workforce Jennifer Magnani & Anders Vinther Article in PDA Journal
Pricing Authority Expectations Supply Chain Complexity Counterfeiting Can Quality Solve the Equation? Quality Drugs to Patients & An Engaged Workforce Jennifer Magnani & Anders Vinther Article in PDA Journal
Largest Independent Pharmaceutical Benchmarking Survey
 Largest Independent Pharmaceutical Benchmarking Survey Operational Excellence in the Pharmaceutical Industry II Operational Excellence in the Pharmaceutical Industry Largest Independent Pharmaceutical
Largest Independent Pharmaceutical Benchmarking Survey Operational Excellence in the Pharmaceutical Industry II Operational Excellence in the Pharmaceutical Industry Largest Independent Pharmaceutical
Establishing and Sustaining a Quality Culture Jacqueline Elbonne, Ph.D., Senior Vice President Global Quality, Merck. August 2016
 Establishing and Sustaining a Quality Culture Jacqueline Elbonne, Ph.D., Senior Vice President Global Quality, Merck August 2016 Merck s Mission Our Mission To discover, develop and provide innovative
Establishing and Sustaining a Quality Culture Jacqueline Elbonne, Ph.D., Senior Vice President Global Quality, Merck August 2016 Merck s Mission Our Mission To discover, develop and provide innovative
Quality Metrics and Data Driven Decisions. Disclaimer
 Quality Metrics and Data Driven Decisions PDA Pharmaceutical Quality Metrics and Quality Culture Conference February 21, 2017 CDR Tara Gooen Bizjak Senior Science Policy Advisor Div. of Regulations, Guidance,
Quality Metrics and Data Driven Decisions PDA Pharmaceutical Quality Metrics and Quality Culture Conference February 21, 2017 CDR Tara Gooen Bizjak Senior Science Policy Advisor Div. of Regulations, Guidance,
Quality Metrics: A Regulatory Perspective
 Quality Metrics: A Regulatory Perspective By Tara Gooen Bizjak, MBSci CDER Office of Policy for Pharmaceutical Quality Presented By Dr. Ademola Daramola US FDA Office of International Programs Indian Pharmaceutical
Quality Metrics: A Regulatory Perspective By Tara Gooen Bizjak, MBSci CDER Office of Policy for Pharmaceutical Quality Presented By Dr. Ademola Daramola US FDA Office of International Programs Indian Pharmaceutical
Parenteral Drug Association 2020 Strategic Plan
 Parenteral Drug Association 2020 Strategic Plan November 2015 Connecting People, Science and Regulation Table of Contents Introduction...3 Strategic Planning Committee...6 Mission, Vision, Values and Motto...7
Parenteral Drug Association 2020 Strategic Plan November 2015 Connecting People, Science and Regulation Table of Contents Introduction...3 Strategic Planning Committee...6 Mission, Vision, Values and Motto...7
A Potential Innovative CMC Solution: Responding To Public Health Needs With An Accelerated Clinical Pathway A Vaccine Example
 A Potential Innovative CMC Solution: Responding To Public Health Needs With An Accelerated Clinical Pathway A Vaccine Example January 2018 Natalie A. Christian Integrated Development and Supply Team Lead
A Potential Innovative CMC Solution: Responding To Public Health Needs With An Accelerated Clinical Pathway A Vaccine Example January 2018 Natalie A. Christian Integrated Development and Supply Team Lead
How a project approach will build change management capability across your organization
 capability across your How a project approach will build change management capability across your Prosci Webinar How a project approach will build change management Enterprise Change Management Organizational
capability across your How a project approach will build change management capability across your Prosci Webinar How a project approach will build change management Enterprise Change Management Organizational
Pharma EXPO 6 9 November 2016 Chicago, IL
 DESIGN TO VALUE: An End-to-end Lifecycle Methodology, Enabling Customer-centric, High-quality Products, Made Efficiently and Reliably Susan Neadle Senior Director Design to Value & Product Quality Management
DESIGN TO VALUE: An End-to-end Lifecycle Methodology, Enabling Customer-centric, High-quality Products, Made Efficiently and Reliably Susan Neadle Senior Director Design to Value & Product Quality Management
EMA perspective on Quality Metrics
 EMA perspective on Quality Metrics Indian Pharmaceutical Alliance Annual Congress, Mumbai India Presented by Brendan Cuddy on 24 February 2017 Head of Manufacturing and Quality Compliance, European Medicines
EMA perspective on Quality Metrics Indian Pharmaceutical Alliance Annual Congress, Mumbai India Presented by Brendan Cuddy on 24 February 2017 Head of Manufacturing and Quality Compliance, European Medicines
Can quality metrics drive quality culture?
 Can quality metrics drive quality culture? Jeney Isbel, Site Quality Director PGS Melbourne Tuesday 25 th July 2017, 10:00 10:30am Slide 1 PharmOut 2017 About Pfizer Who We Are and What We Do One of the
Can quality metrics drive quality culture? Jeney Isbel, Site Quality Director PGS Melbourne Tuesday 25 th July 2017, 10:00 10:30am Slide 1 PharmOut 2017 About Pfizer Who We Are and What We Do One of the
Comments from Cross-Industry Quality Metrics Collaboration Group regarding Docket FDA D-2537: Request for Quality Metrics.
 November 25, 2015 Division of Dockets Management (HFA-305) Food and Drug Administration Department of Health and Human Services 5630 Fishers Lane Room 1061 Rockville, MD 20852 Comments from Cross-Industry
November 25, 2015 Division of Dockets Management (HFA-305) Food and Drug Administration Department of Health and Human Services 5630 Fishers Lane Room 1061 Rockville, MD 20852 Comments from Cross-Industry
Tim Moore Global Head, Pharmaceutical Technical Operations Biologics, Genentech a member of the Roche Group
 Ensuring the Supply of Quality Biopharmaceuticals: Integrated Manufacturing Network & Continual Improvement of Manufacturing Process Tim Moore Global Head, Pharmaceutical Technical Operations Biologics,
Ensuring the Supply of Quality Biopharmaceuticals: Integrated Manufacturing Network & Continual Improvement of Manufacturing Process Tim Moore Global Head, Pharmaceutical Technical Operations Biologics,
TRAINING, AUDITING, INTERIM MANAGEMENT AND CONSULTANCY
 GMP SERVICES TRAINING, AUDITING, INTERIM MANAGEMENT AND CONSULTANCY PCS was founded in 1990 with one simple mission: help companies struggling with GMP in achieving regulatory compliance. We have maintained
GMP SERVICES TRAINING, AUDITING, INTERIM MANAGEMENT AND CONSULTANCY PCS was founded in 1990 with one simple mission: help companies struggling with GMP in achieving regulatory compliance. We have maintained
Building Quality Culture and Capabilities Jaidev S Rajpal Partner, McKinsey & Company IPA CONFERENCE FEBRUARY 2019
 Building Quality Culture and Capabilities Jaidev S Rajpal Partner, IPA CONFERENCE FEBRUARY 2019 CULTURE MATTERS 1 Quality culture matters: Increasingly companies believe that culture matters; this is also
Building Quality Culture and Capabilities Jaidev S Rajpal Partner, IPA CONFERENCE FEBRUARY 2019 CULTURE MATTERS 1 Quality culture matters: Increasingly companies believe that culture matters; this is also
Driving effective quality governance using Quality Metrics
 Driving effective quality governance using Quality Metrics IPA Advanced GMP Workshop November 208 McKinsey & Company Contents Perspectives on best practices in Quality Metrics Group exercise 2 Regulatory
Driving effective quality governance using Quality Metrics IPA Advanced GMP Workshop November 208 McKinsey & Company Contents Perspectives on best practices in Quality Metrics Group exercise 2 Regulatory
Pharmaceutical Quality for 21 st Century Initiative
 Quality Metrics Alicia Mozzachio, RPh, MPH Senior Advisor for International Activities Office of Policy for Pharmaceutical Quality (OPPQ) Center for Drug Evaluation and Research U.S. Food and Drug Administration
Quality Metrics Alicia Mozzachio, RPh, MPH Senior Advisor for International Activities Office of Policy for Pharmaceutical Quality (OPPQ) Center for Drug Evaluation and Research U.S. Food and Drug Administration
LIFE SCIENCE BENCHMARKING STUDY. Trends and Best Practices Based on Our Work with the US FDA and Over 250 Companies
 LIFE SCIENCE 01 BENCHMARKING STUDY Trends and Best Practices Based on Our Work with the US FDA and Over 0 Companies SUMMARY In this report, UL EduNeering has identified the 01/01 compliance and business
LIFE SCIENCE 01 BENCHMARKING STUDY Trends and Best Practices Based on Our Work with the US FDA and Over 0 Companies SUMMARY In this report, UL EduNeering has identified the 01/01 compliance and business
Global Trends in Audits: Demonstrating Training Effectiveness
 Global Trends in Audits: Demonstrating Training Effectiveness UL and the UL logo are trademarks of UL LLC 2013. Agenda Goals for this Discussion Our Panel Industry Audit Trends Our Benchmarking Responses
Global Trends in Audits: Demonstrating Training Effectiveness UL and the UL logo are trademarks of UL LLC 2013. Agenda Goals for this Discussion Our Panel Industry Audit Trends Our Benchmarking Responses
Modernizing Quality Management
 Modernizing Quality Management Priya Durvasula Head of IT, Global Quality Raghunandan Satyanarayan Associate Director September 17, 2018 1 Speaker Information Priya Durvasula, PhD Head of IT, Global Quality
Modernizing Quality Management Priya Durvasula Head of IT, Global Quality Raghunandan Satyanarayan Associate Director September 17, 2018 1 Speaker Information Priya Durvasula, PhD Head of IT, Global Quality
White paper Audits in automated production
 White paper Audits in automated production data management for automation Introduction: Audits in automated production In the field of production management, audits have become an important tool for analysing
White paper Audits in automated production data management for automation Introduction: Audits in automated production In the field of production management, audits have become an important tool for analysing
Culture & the QP. Frank Hallinan Annual QP Forum 16 April 2015.
 Culture & the QP. Frank Hallinan. 2015 Annual QP Forum 16 April 2015. The Big Picture: NOT about Handel, Heaney or, Hurling! What is responsibility of QP? Certify individual batches of a medicine. Assure
Culture & the QP. Frank Hallinan. 2015 Annual QP Forum 16 April 2015. The Big Picture: NOT about Handel, Heaney or, Hurling! What is responsibility of QP? Certify individual batches of a medicine. Assure
Strategic Plan f y f y
 Strategic Plan fy 2017 - fy 2019 Introduction Fiscal year 2017 marks the 10th year for the University of Texas System (UT System) Supply Chain Alliance (Alliance). As we celebrate the accomplishment of
Strategic Plan fy 2017 - fy 2019 Introduction Fiscal year 2017 marks the 10th year for the University of Texas System (UT System) Supply Chain Alliance (Alliance). As we celebrate the accomplishment of
Developing a Robust Quality System to Assure Data Integrity
 Developing a Robust Quality System to Assure Data Integrity Maryann Gribbin Chief Compliance Officer, Faith & Royale Consultants Co-Chair PDA Data Integrity Task Force Data Integrity Facts or information
Developing a Robust Quality System to Assure Data Integrity Maryann Gribbin Chief Compliance Officer, Faith & Royale Consultants Co-Chair PDA Data Integrity Task Force Data Integrity Facts or information
Dear Colleagues, Looking forward to seeing you in Singapore, Andiyanto Sutandar, Technical Director, HGP Asia Pte. Ltd.
 2018 PDA Annual Singapore Conference New Developments in Bio/Pharmaceutical Manufacturing and Regulation October 30-31, 2018 Westin Singapore Singapore, Singapore Dear Colleagues, The 2018 PDA Annual Singapore
2018 PDA Annual Singapore Conference New Developments in Bio/Pharmaceutical Manufacturing and Regulation October 30-31, 2018 Westin Singapore Singapore, Singapore Dear Colleagues, The 2018 PDA Annual Singapore
Advanced Performance Management for Government
 Advanced Performance Management for Government Dates Offered: June 25-27, 2018 December 10-12, 2018 GOVERNMENT PERFORMANCE LEADER CAREER PATH: Obtain the skills and a pathway to taking a leadership role
Advanced Performance Management for Government Dates Offered: June 25-27, 2018 December 10-12, 2018 GOVERNMENT PERFORMANCE LEADER CAREER PATH: Obtain the skills and a pathway to taking a leadership role
Zydus Hospira Oncology Pvt. Ltd.
 Company, Position & Person Profile Zydus Hospira Oncology Pvt. Ltd. 6/7/15 About Zydus Hospira Oncology Pvt. Ltd. Zydus Hospira Oncology Private Limited (ZHOPL) is a joint venture company in Ahmedabad,
Company, Position & Person Profile Zydus Hospira Oncology Pvt. Ltd. 6/7/15 About Zydus Hospira Oncology Pvt. Ltd. Zydus Hospira Oncology Private Limited (ZHOPL) is a joint venture company in Ahmedabad,
GxP Auditing, Remediation, and Staff Augmentation
 GxP Auditing, Remediation, and Staff Augmentation TABLE OF CONTENTS 3 Introduction 4 GxP Auditing 4 GMP Auditing 5 GCP Auditing 6 GLP Auditing 7 Pharmacovigilance Auditing 7 Vendor/Supplier Auditing 8
GxP Auditing, Remediation, and Staff Augmentation TABLE OF CONTENTS 3 Introduction 4 GxP Auditing 4 GMP Auditing 5 GCP Auditing 6 GLP Auditing 7 Pharmacovigilance Auditing 7 Vendor/Supplier Auditing 8
IPA Quality Forum Reflections on the Journey so far
 IPA Quality Forum Reflections on the Journey so far India Pharmaceutical Forum 2018 23rd Feb 2018 The IPA Quality forum was formed with a vision of helping the Indian pharma industry achieve excellence
IPA Quality Forum Reflections on the Journey so far India Pharmaceutical Forum 2018 23rd Feb 2018 The IPA Quality forum was formed with a vision of helping the Indian pharma industry achieve excellence
Investors in People Introducing the sixth generation Standard. Gordon Stopani Accredited Practitioner
 Investors in People Introducing the sixth generation Standard Gordon Stopani Accredited Practitioner What is Investors in People (IIP)? People Management Standard Tool for supporting the achievement of
Investors in People Introducing the sixth generation Standard Gordon Stopani Accredited Practitioner What is Investors in People (IIP)? People Management Standard Tool for supporting the achievement of
Changes in the EXCiPACT 2017 Edition Requirements from the 2012 Edition
 Changes in the EXCiPACT 2017 Edition Requirements from the 2012 Edition Blue text indicates changes to both GMP and GDP requirements Orange text indicates specific changes to the GDP requirements Items
Changes in the EXCiPACT 2017 Edition Requirements from the 2012 Edition Blue text indicates changes to both GMP and GDP requirements Orange text indicates specific changes to the GDP requirements Items
Connecting People, Science and Regulation
 Connecting People, Science and Regulation Bethesda Towers 4350 East West Highway, Ste 600 Bethesda, MD 20814 USA Tel: +1 (301) 656-5900 Fax: +1 (301) 986-0296 www.pda.org PDA Europe ggmbh Am Borsigturm
Connecting People, Science and Regulation Bethesda Towers 4350 East West Highway, Ste 600 Bethesda, MD 20814 USA Tel: +1 (301) 656-5900 Fax: +1 (301) 986-0296 www.pda.org PDA Europe ggmbh Am Borsigturm
Company, Position & Person Profile. Cadila Healthcare Limited
 Company, Position & Person Profile Cadila Healthcare Limited About Zydus Cadila Zydus Cadila is a fully integrated, global healthcare provider, with strengths all along the pharmaceutical value chain.
Company, Position & Person Profile Cadila Healthcare Limited About Zydus Cadila Zydus Cadila is a fully integrated, global healthcare provider, with strengths all along the pharmaceutical value chain.
Voluntary Pilot Meeting Preview: How will CDRH apply assessments in the voluntary program?
 Voluntary Pilot Meeting Preview: How will CDRH apply assessments in the voluntary program? Cisco Vicenty Case for Quality Program Manager Center for Devices and Radiological Health U.S. Food and Drug Administration,
Voluntary Pilot Meeting Preview: How will CDRH apply assessments in the voluntary program? Cisco Vicenty Case for Quality Program Manager Center for Devices and Radiological Health U.S. Food and Drug Administration,
External Quality Assessment Are You Ready? Institute of Internal Auditors
 External Quality Assessment Are You Ready? Institute of Internal Auditors Objectives Describe frameworks used to assess the quality of an IA activity Discuss benefits, challenges, and success factors related
External Quality Assessment Are You Ready? Institute of Internal Auditors Objectives Describe frameworks used to assess the quality of an IA activity Discuss benefits, challenges, and success factors related
# MAKE CAPA COOL Workstream Report Out. 02/25/19 Project Chair: Kathryn Merrill (Medtronic)
 # MAKE CAPA COOL Workstream Report Out 02/25/19 Project Chair: Kathryn Merrill (Medtronic) #makecapacool Project CASE FOR QUALITY CAPA WORKSTREAM Industry-wide struggle resulting from the current CAPA
# MAKE CAPA COOL Workstream Report Out 02/25/19 Project Chair: Kathryn Merrill (Medtronic) #makecapacool Project CASE FOR QUALITY CAPA WORKSTREAM Industry-wide struggle resulting from the current CAPA
University of Limerick Quality Management System Framework for Support Units
 University of Limerick Quality Management System Framework for Support Units Revision 1 Approved by Governing Authority Strategic Planning & Quality Assurance Committee (GASPQA) 26 January 2016 Contents
University of Limerick Quality Management System Framework for Support Units Revision 1 Approved by Governing Authority Strategic Planning & Quality Assurance Committee (GASPQA) 26 January 2016 Contents
An Introduction to Double Dragon Consulting
 An Introduction to Double Dragon Consulting www.doubledragonconsulting.com About DDC Our Experience: - All dosage forms, aseptic processes, API manufacture, OTC - Remediation of 483 observations, Warning
An Introduction to Double Dragon Consulting www.doubledragonconsulting.com About DDC Our Experience: - All dosage forms, aseptic processes, API manufacture, OTC - Remediation of 483 observations, Warning
Risk Culture: The Heart and Soul of Enterprise Risk Management
 Risk Culture: The Heart and Soul of Enterprise Risk Management Philadelphia AFP Conference May 4, 2017 Edmund Green, Managing Director Risk Consulting KPMG, LLP Agenda Introductions What is Culture The
Risk Culture: The Heart and Soul of Enterprise Risk Management Philadelphia AFP Conference May 4, 2017 Edmund Green, Managing Director Risk Consulting KPMG, LLP Agenda Introductions What is Culture The
Pharmaceutical Good Manufacturing Practice. Contents are subject to change. For the latest updates visit
 Pharmaceutical Good Manufacturing Practice Page 1 of 6 Why Attend A robust quality system that meets the requirements of pharmaceutical regulators is an essential element of every good pharmaceutical manufacturing
Pharmaceutical Good Manufacturing Practice Page 1 of 6 Why Attend A robust quality system that meets the requirements of pharmaceutical regulators is an essential element of every good pharmaceutical manufacturing
CSR / Sustainability Governance and Management Assessment By Coro Strandberg President, Strandberg Consulting
 Introduction CSR / Sustainability Governance and Management Assessment By Coro Strandberg President, Strandberg Consulting www.corostrandberg.com November 2015 Companies which adopt CSR or sustainability
Introduction CSR / Sustainability Governance and Management Assessment By Coro Strandberg President, Strandberg Consulting www.corostrandberg.com November 2015 Companies which adopt CSR or sustainability
ISO FDA QSR. ISO and FDA QSR: A Step by Step Guide to Complying with Medical Device QMS Requirements
 ISO 13485 FDA QSR ISO 13485 and FDA QSR: A Step by Step Guide to Complying with Medical Device QMS Requirements Jon Speer Founder & VP of QA/RA greenlight.guru Table of Contents 1 Introduction 4 QMS Philosophy
ISO 13485 FDA QSR ISO 13485 and FDA QSR: A Step by Step Guide to Complying with Medical Device QMS Requirements Jon Speer Founder & VP of QA/RA greenlight.guru Table of Contents 1 Introduction 4 QMS Philosophy
Automating the Defense: Really Taking Advantage of Automated Sharing
 Automating the Defense: Really Taking Advantage of Automated Sharing Michael Vermilye The Johns Hopkins University Applied Physics Laboratory 2017 by The Johns Hopkins Applied Physics Laboratory. Material
Automating the Defense: Really Taking Advantage of Automated Sharing Michael Vermilye The Johns Hopkins University Applied Physics Laboratory 2017 by The Johns Hopkins Applied Physics Laboratory. Material
McKinsey ACQUA Assessment of Capabilities in Quality. Making the organization flow to Quality Excellence
 McKinsey ACQUA Assessment of Capabilities in Quality Making the organization flow to Quality Excellence What is McKinsey Assessment of Quality Capabilities (ACQUA) and which impact can it bring to your
McKinsey ACQUA Assessment of Capabilities in Quality Making the organization flow to Quality Excellence What is McKinsey Assessment of Quality Capabilities (ACQUA) and which impact can it bring to your
CDER /OPQ Office of Surveillance Quality Metrics in Surveillance
 CDER /OPQ Office of Surveillance Quality Metrics in Surveillance Russell Wesdyk Acting Director Office of Surveillance Office of Surveillance Goals Builds from the shared vision A maximally efficient,
CDER /OPQ Office of Surveillance Quality Metrics in Surveillance Russell Wesdyk Acting Director Office of Surveillance Office of Surveillance Goals Builds from the shared vision A maximally efficient,
Quality Culture A Regulatory Perspective Regulatory Compliance Associates Inc. All Rights Reserved
 Quality Culture A Regulatory Perspective 8/15/2016 Quality Culture- The What Quality culture starts with leadership that understands that human behavior and motivations are critical to meeting ongoing
Quality Culture A Regulatory Perspective 8/15/2016 Quality Culture- The What Quality culture starts with leadership that understands that human behavior and motivations are critical to meeting ongoing
BIOTECH, MED DEVICE & PHARMA
 WHITEPAPER Key Account Management Strategies for BIOTECH, MED DEVICE & PHARMA INTRODUCTION Changes in the healthcare landscape have led to changes in customers. In an environment where healthcare providers
WHITEPAPER Key Account Management Strategies for BIOTECH, MED DEVICE & PHARMA INTRODUCTION Changes in the healthcare landscape have led to changes in customers. In an environment where healthcare providers
Division of Docket Management (HFA-305) Food and Drug Administration 5630 Fishers Lane, Room 1061 Rockville, MD 20852
 Connecting People, Science and Regulation Bethesda Towers 4350 East West Highway Suite 600 Bethesda, MD 20814 USA Tel: +1 (301) 656-5900 Fax: +1 (301) 986-0296 www.pda.org PDA Europe ggmbh Am Borsigturm
Connecting People, Science and Regulation Bethesda Towers 4350 East West Highway Suite 600 Bethesda, MD 20814 USA Tel: +1 (301) 656-5900 Fax: +1 (301) 986-0296 www.pda.org PDA Europe ggmbh Am Borsigturm
Modernizing Pharmaceutical Quality Systems; Studying Quality Metrics and Quality Culture;
 This document is scheduled to be published in the Federal Register on 06/29/2018 and available online at https://federalregister.gov/d/2018-14005, and on FDsys.gov 4164-01-P DEPARTMENT OF HEALTH AND HUMAN
This document is scheduled to be published in the Federal Register on 06/29/2018 and available online at https://federalregister.gov/d/2018-14005, and on FDsys.gov 4164-01-P DEPARTMENT OF HEALTH AND HUMAN
PDA Task Force for Single Use Systems
 PDA Task Force for Single Use Systems Robert Repetto- Chairman Pfizer Morten Munk- Co-Chair CMC Biologics April, 2011 PDA Annual Meeting, San Antonio, Texas 1 2 What is a Single Use System?? 3 Technical
PDA Task Force for Single Use Systems Robert Repetto- Chairman Pfizer Morten Munk- Co-Chair CMC Biologics April, 2011 PDA Annual Meeting, San Antonio, Texas 1 2 What is a Single Use System?? 3 Technical
The emdr Requirement. Best Practices for Enhancing Collaboration with the FDA
 The emdr Requirement Best Practices for Enhancing Collaboration with the FDA Executive Summary The Food and Drug Administration (FDA) is tasked with the job of ensuring that all products produced within
The emdr Requirement Best Practices for Enhancing Collaboration with the FDA Executive Summary The Food and Drug Administration (FDA) is tasked with the job of ensuring that all products produced within
Zydus Hospira Oncology Pvt. Ltd.
 Company, Position & Person Profile Zydus Hospira Oncology Pvt. Ltd. 6/7/15 About Zydus Hospira Oncology Pvt. Ltd. Zydus Hospira Oncology Private Limited (ZHOPL) is a joint venture company in Ahmedabad,
Company, Position & Person Profile Zydus Hospira Oncology Pvt. Ltd. 6/7/15 About Zydus Hospira Oncology Pvt. Ltd. Zydus Hospira Oncology Private Limited (ZHOPL) is a joint venture company in Ahmedabad,
PDA Bioburden and Biofilm Workshop
 The Parenteral Drug Association presents the... PDA Bioburden and Biofilm Workshop Controlling Microbial Contamination to Assure Product Quality, Patient Safety Save $100 when you register for the workshop
The Parenteral Drug Association presents the... PDA Bioburden and Biofilm Workshop Controlling Microbial Contamination to Assure Product Quality, Patient Safety Save $100 when you register for the workshop
The Boardroom DEVELOPING SALES LEADERS.
 Developing Sales Leaders The Boardroom are a best practice sales and sales leadership Assessment and Development Programme, providing high value client-centric solutions to companies from all sectors across
Developing Sales Leaders The Boardroom are a best practice sales and sales leadership Assessment and Development Programme, providing high value client-centric solutions to companies from all sectors across
PHASE APPROPRIATE GMP FOR BIOLOGICAL PROCESSES
 PHASE APPROPRIATE GMP FOR BIOLOGICAL PROCESSES PRE-CLINICAL TO COMMERCIAL PRODUCTION Trevor Deeks Editor Phase Appropriate GMP for Biological Processes Pre-clinical to commercial production Trevor Deeks
PHASE APPROPRIATE GMP FOR BIOLOGICAL PROCESSES PRE-CLINICAL TO COMMERCIAL PRODUCTION Trevor Deeks Editor Phase Appropriate GMP for Biological Processes Pre-clinical to commercial production Trevor Deeks
GxP Auditing, Remediation, and Staff Augmentation
 GxP Auditing, Remediation, and Staff Augmentation TABLE OF CONTENTS 3 Introduction 4 GxP Auditing 4 GMP Auditing 5 GCP Auditing 5 GLP Auditing 6 Pharmacovigilance Auditing 6 Vendor/Supplier Auditing 7
GxP Auditing, Remediation, and Staff Augmentation TABLE OF CONTENTS 3 Introduction 4 GxP Auditing 4 GMP Auditing 5 GCP Auditing 5 GLP Auditing 6 Pharmacovigilance Auditing 6 Vendor/Supplier Auditing 7
MDIC Case for Quality 2020 Strategic Plan Work streams. June 27, 2018
 MDIC Case for Quality 2020 Strategic Plan Work streams June 27, 2018 Background & Timeline February, 2018 MDIC receives proposals for assistance with the Case for Quality 2020 Strategic Plan Development
MDIC Case for Quality 2020 Strategic Plan Work streams June 27, 2018 Background & Timeline February, 2018 MDIC receives proposals for assistance with the Case for Quality 2020 Strategic Plan Development
Committed to Excellence Assessment
 Committed to Excellence Assessment Applicant Guide The objective of this guide is to help you prepare for Committed to Excellence Assessment. Following the advice in this guide will help maximise the benefits
Committed to Excellence Assessment Applicant Guide The objective of this guide is to help you prepare for Committed to Excellence Assessment. Following the advice in this guide will help maximise the benefits
Leading Practice: Approaches to Organizational Change Management
 Leading Practice: Approaches to Organizational Change Management Abstract This document provides recommended approaches to organizational change management (OCM) when implementing CA Project and Portfolio
Leading Practice: Approaches to Organizational Change Management Abstract This document provides recommended approaches to organizational change management (OCM) when implementing CA Project and Portfolio
Applying a Knowledge-Based Approach to Pharmaceutical Supply Chain Temperature Control Management
 May 1-3, 2012 Javits Center New York, NY Applying a Knowledge-Based Approach to Pharmaceutical Supply Chain Control Management Arminda O. Montero Program Manager, Distribution QA Global Pharmaceutical
May 1-3, 2012 Javits Center New York, NY Applying a Knowledge-Based Approach to Pharmaceutical Supply Chain Control Management Arminda O. Montero Program Manager, Distribution QA Global Pharmaceutical
THE CULTURE CANVAS A Working Guide and Checklist to Support the Development of a High-Performing Culture
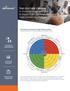 denison TM THE CULTURE CANVAS A Working Guide and Checklist to Support the Development of a High-Performing Culture The Denison Model of High Performance A Systems Approach to Understanding and Managing
denison TM THE CULTURE CANVAS A Working Guide and Checklist to Support the Development of a High-Performing Culture The Denison Model of High Performance A Systems Approach to Understanding and Managing
Best practices in Quality Metrics. Vivek Arora Partner, McKinsey & Company IPA CONFERENCE FEBRUARY 2019
 Best practices in Quality Metrics Vivek Arora Partner, McKinsey & Company IPA CONFERENCE FEBRUARY 209 Management reviews & quality metrics have existed for a while Management review should provide assurance
Best practices in Quality Metrics Vivek Arora Partner, McKinsey & Company IPA CONFERENCE FEBRUARY 209 Management reviews & quality metrics have existed for a while Management review should provide assurance
Committed to Excellence Assessment
 Committed to Excellence Assessment Applicant Guide 2015 www.qualityscotland.co.uk Quality Scotland, West End House, 28 Drumsheugh Gardens, Edinburgh EH3 7RN Tel: 0131 225 5765 The objective of this guide
Committed to Excellence Assessment Applicant Guide 2015 www.qualityscotland.co.uk Quality Scotland, West End House, 28 Drumsheugh Gardens, Edinburgh EH3 7RN Tel: 0131 225 5765 The objective of this guide
Defining Quality for Your Organization
 Defining Quality for Your Organization Align your mission and vision with the organization s mission & vision Agents, back-office staff and managers strive to meet a range of quality standards. There are
Defining Quality for Your Organization Align your mission and vision with the organization s mission & vision Agents, back-office staff and managers strive to meet a range of quality standards. There are
Executive Summary. The State of Employee Engagement pg. 2
 the STATE of EMPLOYEE ENGAGEMENt 2018 v Executive Summary 2018 has been named The Year of Employee Experience by Forbes; highlighting an increasing need for employers to offer engaging experiences to retain
the STATE of EMPLOYEE ENGAGEMENt 2018 v Executive Summary 2018 has been named The Year of Employee Experience by Forbes; highlighting an increasing need for employers to offer engaging experiences to retain
E-PROCUREMENT CHANGE MANAGEMENT
 E-PROCUREMENT CHANGE MANAGEMENT PART ONE STRATEGY Digitizing business processes is inevitable; the challenge for procurement leaders will be changing perceptions internally to win the support needed to
E-PROCUREMENT CHANGE MANAGEMENT PART ONE STRATEGY Digitizing business processes is inevitable; the challenge for procurement leaders will be changing perceptions internally to win the support needed to
Anthony M. Tocco CCEP, CIA, CFE Chief Compliance Officer DTE Energy
 Anthony M. Tocco CCEP, CIA, CFE Chief Compliance Officer DTE Energy Define a Performance Assessment Governance Planning the Assessment Selecting the Assessor Common Assessment Practices Rating Scales Communication
Anthony M. Tocco CCEP, CIA, CFE Chief Compliance Officer DTE Energy Define a Performance Assessment Governance Planning the Assessment Selecting the Assessor Common Assessment Practices Rating Scales Communication
The Impact of Quality Culture on Quality Risk Management. FDA Perspective on Quality Culture; how it Impacts Risk Management
 The Impact of Quality Culture on Quality Risk Management FDA Perspective on Quality Culture; how it Impacts Risk Management Teresa Gorecki Practice Lead Compliance Architects Agenda The WHAT Definitions
The Impact of Quality Culture on Quality Risk Management FDA Perspective on Quality Culture; how it Impacts Risk Management Teresa Gorecki Practice Lead Compliance Architects Agenda The WHAT Definitions
Executional Excellence
 Executional Excellence Managing Compliance Productivity, Risk and Reputation Dalton Smart June 7, 2005 Overview Managing Risk with Process Excellence Executional Excellence Examples Key Challenges and
Executional Excellence Managing Compliance Productivity, Risk and Reputation Dalton Smart June 7, 2005 Overview Managing Risk with Process Excellence Executional Excellence Examples Key Challenges and
GxP Auditing, Remediation, and Quality System Resourcing
 GxP Auditing, Remediation, and Quality System Resourcing TABLE OF CONTENTS 3 Introduction 4 GxP Auditing 4 GMP Auditing 5 GCP Auditing 6 GLP Auditing 7 Pharmacovigilance Auditing 7 Vendor/Supplier Auditing
GxP Auditing, Remediation, and Quality System Resourcing TABLE OF CONTENTS 3 Introduction 4 GxP Auditing 4 GMP Auditing 5 GCP Auditing 6 GLP Auditing 7 Pharmacovigilance Auditing 7 Vendor/Supplier Auditing
Q10 PHARMACEUTICAL QUALITY SYSTEM
 Q10 PHARMACEUTICAL QUALITY SYSTEM This draft guidance, when finalized, will represent the Food and Drug Administration's (FDA's) current thinking on this topic. It does not create or confer any rights
Q10 PHARMACEUTICAL QUALITY SYSTEM This draft guidance, when finalized, will represent the Food and Drug Administration's (FDA's) current thinking on this topic. It does not create or confer any rights
IAQG Strategic Plan and Pace of Progress 2017
 IAQG Mission The mission of the International Aerospace Quality Group (IAQG) is to implement quality initiatives for improvements throughout the aerospace product and services value stream. For that purpose,
IAQG Mission The mission of the International Aerospace Quality Group (IAQG) is to implement quality initiatives for improvements throughout the aerospace product and services value stream. For that purpose,
Cost-effective Purification of Vaccines, Data Integrity Systems and CTDs. DRAFT AGENDA Rio de Janeiro, 18 to 20 June 2018
 Cost-effective Purification of Vaccines, Data Integrity Systems and CTDs DRAFT AGENDA Rio de Janeiro, 18 to 20 June 2018 DAY1, Monday 18 June - Welcome & Introduction Plenary session for all participants
Cost-effective Purification of Vaccines, Data Integrity Systems and CTDs DRAFT AGENDA Rio de Janeiro, 18 to 20 June 2018 DAY1, Monday 18 June - Welcome & Introduction Plenary session for all participants
EEI Leading Indicator Study: Preliminary Results
 EEI Leading Indicator Study: Preliminary Results Edison Electric Institute Occupational Safety & Health Committee Conference Indianapolis, Indiana April 23, 2013 Larry Pena & Blakely Smith Southern California
EEI Leading Indicator Study: Preliminary Results Edison Electric Institute Occupational Safety & Health Committee Conference Indianapolis, Indiana April 23, 2013 Larry Pena & Blakely Smith Southern California
A conversation about the framework for performance excellence PERFORM LIKE A ROCK STAR!
 A conversation about the framework for performance excellence PERFORM LIKE A ROCK STAR! Like Olympic athletes, pursing performance excellence in our work implies that we optimize every factor that goes
A conversation about the framework for performance excellence PERFORM LIKE A ROCK STAR! Like Olympic athletes, pursing performance excellence in our work implies that we optimize every factor that goes
RMF is scheduled to operate for three years, from January December 2017.
 Responsible Microfinance Facility Funded by the Agence Française de Développement and Managed by the Social Performance Task Force, in collaboration with the Smart Campaign Frequently Asked Questions 1.
Responsible Microfinance Facility Funded by the Agence Française de Développement and Managed by the Social Performance Task Force, in collaboration with the Smart Campaign Frequently Asked Questions 1.
7 th APRAST SMS/SSP Workshop Outcome 31 August 2015 Bangkok, Thailand
 7 th APRAST SMS/SSP Workshop Outcome 31 August 2015 Bangkok, Thailand SSP/ SMS Implementation Challenges Obstacles Barriers - Needs - Regulatory & Industry perspective Outcome: Lack of Ministerial understanding
7 th APRAST SMS/SSP Workshop Outcome 31 August 2015 Bangkok, Thailand SSP/ SMS Implementation Challenges Obstacles Barriers - Needs - Regulatory & Industry perspective Outcome: Lack of Ministerial understanding
ISPE Annual Meeting 29 October 1 November 2017 San Diego, CA. Need systems approach to pharmaceutical manufacturing
 Aligning Quality Excellence with ICH(Quality Systems Management) Prabir K Basu Need systems approach to pharmaceutical manufacturing Pharmaceutical manufacturing needs to be approached as a system that
Aligning Quality Excellence with ICH(Quality Systems Management) Prabir K Basu Need systems approach to pharmaceutical manufacturing Pharmaceutical manufacturing needs to be approached as a system that
2013/2014 UL EduNeering ComplianceWire Client Benchmarking Study: Quality Metrics Being Used to Drive Continuous Improvement
 2013/2014 UL EduNeering ComplianceWire Client Benchmarking Study: Quality Metrics Being Used to Drive Continuous Improvement UL and the UL logo are trademarks of UL LLC 2013 Agenda About our benchmarking
2013/2014 UL EduNeering ComplianceWire Client Benchmarking Study: Quality Metrics Being Used to Drive Continuous Improvement UL and the UL logo are trademarks of UL LLC 2013 Agenda About our benchmarking
Ensuring Quality & Regulatory Compliance when Collaborating with a Service Provider
 DPT Thought Leadership Issue 2 Ensuring Quality & Regulatory Compliance when Collaborating with a Service Provider I N T R O D U C T I O N In recent years, the pharmaceutical industry has given greater
DPT Thought Leadership Issue 2 Ensuring Quality & Regulatory Compliance when Collaborating with a Service Provider I N T R O D U C T I O N In recent years, the pharmaceutical industry has given greater
An Industry Perspective
 Controls An Industry Perspective Presented by: Jay Gerondale Amgen Inc. Overview Why are We Here? What are Controls? Control Elements Product Development Steps Conclusion Bridging Making a Combination
Controls An Industry Perspective Presented by: Jay Gerondale Amgen Inc. Overview Why are We Here? What are Controls? Control Elements Product Development Steps Conclusion Bridging Making a Combination
Challenges to Building Quality End to End
 Challenges to Building Quality End to End FDA/PQRI Conference- OCTOBER 2015 Michael Thien, Sc. D. SVP, Global Science, Technology & Commercialization Merck Manufacturing Division Two key dimensions of
Challenges to Building Quality End to End FDA/PQRI Conference- OCTOBER 2015 Michael Thien, Sc. D. SVP, Global Science, Technology & Commercialization Merck Manufacturing Division Two key dimensions of
The Governance Institute s Fax Poll Results December Board Quality Committees
 The Governance Institute s Fax Poll Results December 2002 Board Quality Committees We are what we repeatedly do. Excellence, then, is not an act, but a habit. Aristotle As healthcare watchdogs move into
The Governance Institute s Fax Poll Results December 2002 Board Quality Committees We are what we repeatedly do. Excellence, then, is not an act, but a habit. Aristotle As healthcare watchdogs move into
Quality Assurance in nonregulated. pharmaceutical industry. Thomas Steckler
 Quality Assurance in nonregulated research of the pharmaceutical industry Thomas Steckler The views expressed in this presentation are solely those of the individual authors, and do not necessarily reflect
Quality Assurance in nonregulated research of the pharmaceutical industry Thomas Steckler The views expressed in this presentation are solely those of the individual authors, and do not necessarily reflect
Regulatory Affairs & Quality Advisory Board. A Parenteral Drug Association (PDA) Advisory Board
 Regulatory Affairs & Quality Advisory Board A Parenteral Drug Association (PDA) Advisory Board Purpose of Presentation RAQAB has a goal of improving communication of RAQAB activities to the membership
Regulatory Affairs & Quality Advisory Board A Parenteral Drug Association (PDA) Advisory Board Purpose of Presentation RAQAB has a goal of improving communication of RAQAB activities to the membership
