Density Answers to the End of module questions
|
|
|
- Bryan Wheeler
- 6 years ago
- Views:
Transcription
1 IDS 101 Density Answers to the End of module questions 1. You are experimenting with the mysterious substance X. You carefully measure the mass of a uniform 2 ml sample of pure substance X. You slice it into two identical pieces with a volume of 1 ml each. You then measure the density of one of the pieces. a) You would find that the density of this 1 ml piece is (choose one): i) Less than the density of the 2 ml piece i The same as the density of the 2 ml piece Greater than the density of the 2 ml piece The density of a substance is not dependent on the size of the piece, so answer ii is correct. b) You then discover that substance X has the wonderful property that you can change its volume without changing its mass. You squish one of the 1 ml pieces down so that it has a smaller volume but still has its original mass. In the process, the density of this piece (choose one): i) Decreased i Stayed the same Increased iii is correct If the mass of the object is the same, but the volume decreased, the density would increase. c) You then take the other 1 ml piece of substance X and roll it out into a large flat sheet. The sheet still has a volume of 1 ml, but now has a much larger surface area. In the course of rolling out this large flat piece, the density of this piece (choose one): i) Decreased i Stayed the same Increased ii is correct- if the mass and the volume stayed the same, the density is the same. Changing the surface area does not change the density.
2 2. You are on an oceanography cruise that has taken some water samples of fresh water from a river and some water from Puget Sound. Unfortunately, the samples were not labeled properly so we can t tell which samples are the salt water and which are the freshwater. We do not want to open the containers for fear of contamination, so let s use the density of the water as a way to tell them apart. If the containers are exactly one liter and we ignore the weight of the plastic in the bottles and the lids, what would be the mass of the freshwater samples if the density of freshwater is about 1000 kg/ m? If saltwater is about 1021 kg/ m, what will be the mass? 1 liter = 1000 ml= 1000 cm 1000Kg 1000cm cm = 1021Kg 1000cm 1m cm = 1Kg 1.021Kg. You have a block of unknown metal it is heavy, it looks like gold, could it be gold? If it is gold, the density should be 19. grams per cubic cm. When you put the metallic block into a graduated cylinder it raises the water level 12 ml. What should the mass of the block be, if it is gold? Density = mass/volume 12 ml of water displaced = 12 cm Mass = volume X density = 12 cm x 19. g/cm = 21.6 grams 4. In a previous class we collected the data for the mass and volume of various metals shown in the graph on the last page of this handout. You will be asked to refer to this graph in the questions that follow. a. Use the graph to find the mass of a piece of copper with a volume of 14 ml. Explain in words how you found the answer. There are many ways to do this. One way is to use the trend line for copper and find the mass value that corresponds to a volume of 14 ml. You could also use the equation of the trend line y = 8.55x and calculate the value of y for x= 14 ml. Either way, your answer should be close to 120 grams. b. Use the graph to find the volume, in cm, of a piece of aluminum with a mass of 50 g. Explain in words how you found the answer. Once again, use the trend line and read off the value of the volume for a mass of 50 g. Or, use the equation of the trend line, y = 2.78x. Now solve for x, with y = 50 g. Either way, your answer should be close to 18 cm.
3 c. Imagine that you have been handed two blocks of metal that have the exact same dimensions. The blocks have been painted black so that they look identical. If you were told that one block was copper and one was zinc, how would you determine which was which? Explain how your graph helps you answer this question. The graph tells you that copper (steeper trend line) has a greater density than zinc. Therefore the mass with the greater mass will be made of copper since M= D V. The graph shows that for a given volume (pick any number you wish), the mass value for zinc will be less than for copper. d. Imagine that you have been handed two blocks of metal that have the exact same mass. The blocks have been painted black so that they look identical. If you were told that one block was lead and one was aluminum, how would you determine which was which? Explain how your graph helps you answer this question. Once again, the graph tells you that lead (steeper trend line) has a greater density than aluminum. The graph shows that for a given mass (pick any value you wish), aluminum occupies a greater volume than lead. 5. In an experiment to investigate sinking and floating behavior, four solid objects (solids # 1, 2, and 4) are tested using three different liquids (liquids A, B and C). The results are shown below: i) Solid # 1 sinks in all three liquids Solids #2 floats in all three liquids i Solid # sinks in Liquid B but floats in liquids A and C. iv) Solid #4 sinks in liquids B and C, but floats in liquid A Based on this information, rank the three liquids and four solids in order of increasing density. Explain your reasoning. DENSE NOT SO DENSE solid #1 liquid A solid #4 liquid C solid # liquid B solid #2 Reasoning: Solid #1 sinks in all liquids and no other solid does, so #1 is the most dense. Most solids float on liquid A, so they are not as dense as liquid A. The only thing that sinks in liquid A is solid #1, so liquid A is slightly less dense than solid #1. Solid #4 sinks in the other liquids but floats on liquid A, so it is next in density. Solid #4 sinks in liquid C but # and #2 float on it, so liquid C is next in density.
4 Solid # only sinks in liquid B and floats in liquid C, so it is next in density. Only solid #2 floats on liquid B, so liquid B is second to lowest in density. Solid #2 floats on everything to it is the least dense. 6. In the table below we have listed five solid compounds and four liquids. We will put a sample of each solid in each liquid. Please help us predict which ones will float and which ones will sink. Place an X in the spaces for the solids that will float and predict the percentage of the solid that will be above the liquid. (For example coal will float in water and 17% of the coal will be above the surface of the water. This is an important process because this is how we separate coal from sandstone in mining!) water (1.0 g/cm ) coal (0.8 g/cm ) X (17%) alcohol salt water brine mercury (1.6 (0.79 g/cm ) (1.2 g/cm ) g/cm ) sink X (1%) X(94%) basalt (.0 g/cm ) beeswax (0.96 g/cm ) iron (7.8 g/cm ) magnesium (1.7 g/cm ) sink sink sink X(78%) X(4%) sink X(20%) X(9%) sink sink sink X(4%) sink sink sink X(88%) 7. Along the eastern part of North America, the Appalachian Mountains were formed more than 250 million years ago as North America and Africa converged to form a large supercontinent called Pangaea (more about this in IDS 102!). The rate of erosion for mountain ranges is such that the Appalachians should have been eroded many years ago. Using isostasy, suggest a reason why we still see the Appalachians today assuming the rates of erosion in the past were equivalent to today s rates. Isostasy is the application of buoyancy to the Earth s crust. The erosion of the Appalachians could be compared to removing the top part of an ice berg. The ice berg would rise to be in balance. The same has happened with the Earth s crust. As erosion has stripped away the mountains, the crust has uplifted to be in balance.
5 Mass (g) copper aluminum zinc lead Mass as a Function of Volume for Various Metals y = x y = 8.55x y = x Volume (ml)
1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter.
 1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter. What is the mass of this object? A) 0.5 g B) 2 g C) 18 g D) 36 g Base your answers to questions 2 through
1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter. What is the mass of this object? A) 0.5 g B) 2 g C) 18 g D) 36 g Base your answers to questions 2 through
Calculating Ballast By Troy Averill
 Calculating Ballast By Troy Averill Lesson Overview: Students use the dimensions of the cargo hold of a ship s hull to calculate the volume of material that can be contained. Students will then use the
Calculating Ballast By Troy Averill Lesson Overview: Students use the dimensions of the cargo hold of a ship s hull to calculate the volume of material that can be contained. Students will then use the
2.3 Density and Density Calculations
 2.3 Density and Density Calculations When people say that lead is heavier than wood, they do not mean that a pea sized piece of lead weighs more than a truckload of pine logs. What they mean is that a
2.3 Density and Density Calculations When people say that lead is heavier than wood, they do not mean that a pea sized piece of lead weighs more than a truckload of pine logs. What they mean is that a
SIGNIFICANT FIGURES WORKSHEET
 SIGNIFICANT FIGURES WORKSHEET PART 1 - Determine the number of significant figures in the following numbers. 1.) 0.02 2.) 0.020 3.) 501 4.) 501.0 5.) 5,000 6.) 5,000. 7.) 6,051.00 8.) 0.0005 9.) 0.1020
SIGNIFICANT FIGURES WORKSHEET PART 1 - Determine the number of significant figures in the following numbers. 1.) 0.02 2.) 0.020 3.) 501 4.) 501.0 5.) 5,000 6.) 5,000. 7.) 6,051.00 8.) 0.0005 9.) 0.1020
SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example,
 SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example, density, thermal or electrical conductivity, solubility,
SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example, density, thermal or electrical conductivity, solubility,
Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3
 Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3 Directions to Students: Do not open this booklet until you are told to do so. Please respond to the following items by marking the best answer
Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3 Directions to Students: Do not open this booklet until you are told to do so. Please respond to the following items by marking the best answer
Experiment 1: The Densities of Liquids and Solids (from Masterson & Hurley)
 Experiment 1: The Densities of Liquids and Solids (from Masterson & Hurley) One of the fundamental properties of any sample of matter is its density, which is its mass per unit of volume. The density of
Experiment 1: The Densities of Liquids and Solids (from Masterson & Hurley) One of the fundamental properties of any sample of matter is its density, which is its mass per unit of volume. The density of
Oceanography Page 1 of 6 Lab: Ocean Salinity and Density M.Sewell rm #70
 Oceanography Page 1 of 6 Salty Water! Description: This lab is designed to demonstrate the formation of the world s oceans and why the oceans are salty, as well as the changes that take place in density
Oceanography Page 1 of 6 Salty Water! Description: This lab is designed to demonstrate the formation of the world s oceans and why the oceans are salty, as well as the changes that take place in density
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
Name # Date Period! FINDING&VOLUME&AND&DENSITY& !!! !!!!!!!
 Name # Date Period FINDING&VOLUME&AND&DENSITY& TherectangularshapedboxinFigure1isfilledwithdrysand.Findthe volumeanddensityofthesand. 1.Firstfindthevolumeofthebox.Thisisalsothevolumeofthesand. Usethediagramtofindthedimensionsofthebox.
Name # Date Period FINDING&VOLUME&AND&DENSITY& TherectangularshapedboxinFigure1isfilledwithdrysand.Findthe volumeanddensityofthesand. 1.Firstfindthevolumeofthebox.Thisisalsothevolumeofthesand. Usethediagramtofindthedimensionsofthebox.
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 3 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 3 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
IDS 102 Answers for Specific Heat End of Module Questions
 IDS 102 Answers for Specific Heat End of Module Questions 1. A cup with 40 grams of 90 C water is mixed with a cup with 70 grams of water at 10 C. a. Predict the final temperature of the water. Explain
IDS 102 Answers for Specific Heat End of Module Questions 1. A cup with 40 grams of 90 C water is mixed with a cup with 70 grams of water at 10 C. a. Predict the final temperature of the water. Explain
In a normal year, where would you expect to see the lowest temperatures: east or west of the Cascades?
 IDS 102 Winter 2011 Heat and Temperature, Part IV Specific Heat and the Importance of Water The weather pattern around Puget Sound, in the region west of the Cascade Mountains, is very different from the
IDS 102 Winter 2011 Heat and Temperature, Part IV Specific Heat and the Importance of Water The weather pattern around Puget Sound, in the region west of the Cascade Mountains, is very different from the
The table below shows some of the properties of the elements cobalt and nickel.
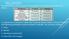 BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
Density What is density? How do you measure density?
 Density What is density? How do you measure density? What is density? Density describes how much mass is in a given volume of a material. Mass: the amount of matter in an object. Volume: the amount of
Density What is density? How do you measure density? What is density? Density describes how much mass is in a given volume of a material. Mass: the amount of matter in an object. Volume: the amount of
OCN 201 Chemical Oceanography Class Notes, Fall 2014 The origin of sea salt Chris Measures, Department of Oceanography
 OCN 201 Chemical Oceanography Class Notes, Fall 2014 The origin of sea salt Chris Measures, Department of Oceanography 1 Introduction Everyone knows that the sea is salty but what exactly is the salt in
OCN 201 Chemical Oceanography Class Notes, Fall 2014 The origin of sea salt Chris Measures, Department of Oceanography 1 Introduction Everyone knows that the sea is salty but what exactly is the salt in
COMPOSITION OF SEAWATER
 COMPOSITION OF SEAWATER Ocean water is a combination of freshwater and a variety of dissolved substances. Salinity is a measure of the amount of dissolved salts in seawater, measured in parts per thousand
COMPOSITION OF SEAWATER Ocean water is a combination of freshwater and a variety of dissolved substances. Salinity is a measure of the amount of dissolved salts in seawater, measured in parts per thousand
A TEACHER-DIRECTED, GUIDED DISCOVERY ACTIVITY FOR ADVANCED PHYSICAL SCIENCE STUDENTS
 A TEACHER-DIRECTED, GUIDED DISCOVERY ACTIVITY FOR ADVANCED PHYSICAL SCIENCE STUDENTS 1 2 FEATURES OF THIS ACTIVITY Problem solving Cooperative learning Requires student reasoning Requires student initiative
A TEACHER-DIRECTED, GUIDED DISCOVERY ACTIVITY FOR ADVANCED PHYSICAL SCIENCE STUDENTS 1 2 FEATURES OF THIS ACTIVITY Problem solving Cooperative learning Requires student reasoning Requires student initiative
Watershed Models. Investigative Question: What is a watershed?
 Investigative Question: What is a watershed? Watershed Models Goal: Students build a model to explore watershed characteristics and use the model to learn how landscape characteristics (such as elevation,
Investigative Question: What is a watershed? Watershed Models Goal: Students build a model to explore watershed characteristics and use the model to learn how landscape characteristics (such as elevation,
1. Hardness 2. Streak 3. Density 4. Crystal structure 5. A mineral must be a naturally occurring, inorganic. 7. c 8. f 9. a 10. d 11. g 12. e 13.
 Properties of Guided Reading and Study Use Target Reading Skills Properties of I. What Is a Mineral? A. Naturally Occurring B. Inorganic C. Solid D. Crystal Structure E. Definite Chemical Composition II.
Properties of Guided Reading and Study Use Target Reading Skills Properties of I. What Is a Mineral? A. Naturally Occurring B. Inorganic C. Solid D. Crystal Structure E. Definite Chemical Composition II.
LESSON 3 OTHER LAND RESOURCES C H A P T E R 6, C O N S E R V I N G O U R R E S O U R C E S
 LESSON 3 OTHER LAND RESOURCES C H A P T E R 6, C O N S E R V I N G O U R R E S O U R C E S OBJECTIVES Compare renewable and nonrenewable resources. Describe how human activities affect the environment.
LESSON 3 OTHER LAND RESOURCES C H A P T E R 6, C O N S E R V I N G O U R R E S O U R C E S OBJECTIVES Compare renewable and nonrenewable resources. Describe how human activities affect the environment.
EOC 7th Grade District Common Assessment
 EOC 7th Grade District Common Assessment 1. Todd needs to measure 50 grams of table salt for an experiment. Which measuring device will most accurately measure 50 grams? 2. What is the length (in millimeters)
EOC 7th Grade District Common Assessment 1. Todd needs to measure 50 grams of table salt for an experiment. Which measuring device will most accurately measure 50 grams? 2. What is the length (in millimeters)
TEKS Lesson 7.8C: Effects of Human Activity on Surface Water and Groundwater
 Class ----- Date -------- TEKS Lesson 7.SC TEKS 7.8e Model the effects of human activity on groundwater surface water in a watershed. and TEKS Lesson 7.8C: Effects of Human Activity on Surface Water and
Class ----- Date -------- TEKS Lesson 7.SC TEKS 7.8e Model the effects of human activity on groundwater surface water in a watershed. and TEKS Lesson 7.8C: Effects of Human Activity on Surface Water and
Chapter 2: Properties of Ocean Water
 Chapter 2: Properties of Ocean Water Oceanography, Emmaus High School Mr. Mihalik Name: Period: CHAPTER 2 - OCEAN WATER 1 MR. MIHALIK, ROOM 442 DENSITY-IN-A-BOTTLE DESIGN During the Golf Ball Density lab,
Chapter 2: Properties of Ocean Water Oceanography, Emmaus High School Mr. Mihalik Name: Period: CHAPTER 2 - OCEAN WATER 1 MR. MIHALIK, ROOM 442 DENSITY-IN-A-BOTTLE DESIGN During the Golf Ball Density lab,
Water: A Valuable, Yet Limited Resource
 Water: A Valuable, Yet Limited Resource Subject: Science Target Grades 4-5 Duration: One class period Materials per class 3 100-ml graduated cylinders container of water (10-ml/student) Water Cycle with
Water: A Valuable, Yet Limited Resource Subject: Science Target Grades 4-5 Duration: One class period Materials per class 3 100-ml graduated cylinders container of water (10-ml/student) Water Cycle with
EXPERIMENT 3: Identification of a Substance by Physical Properties
 EXPERIMENT 3: Identification of a Substance by Physical Properties Materials: Hot plate Digital balance Capillary tubes (3) Thermometer Beakers (250 ml) Watch glass Graduated Cylinder (10 ml) Mel-Temp
EXPERIMENT 3: Identification of a Substance by Physical Properties Materials: Hot plate Digital balance Capillary tubes (3) Thermometer Beakers (250 ml) Watch glass Graduated Cylinder (10 ml) Mel-Temp
PHASE CHANGES. Time Temperature Observations. Name(s)
 3 5 PHASE CHANGES PHASE CHANGES Name(s) The activities presented here focus on the energy changes that occur in substances undergoing a phase change. The first activity will take the most time to complete.
3 5 PHASE CHANGES PHASE CHANGES Name(s) The activities presented here focus on the energy changes that occur in substances undergoing a phase change. The first activity will take the most time to complete.
Module 2, Add on Lesson Turbidity Sensor. Student. 90 minutes
 Module 2, Add on Lesson Turbidity Sensor Student 90 minutes Purpose Construct a sensor to measure the turbidity of water Graph data and reason about curves and linear relationships Calibrate the turbidity
Module 2, Add on Lesson Turbidity Sensor Student 90 minutes Purpose Construct a sensor to measure the turbidity of water Graph data and reason about curves and linear relationships Calibrate the turbidity
WATER. Name Date. Survey/Posttest
 WATER Date 1. What happens to the level of the water in the straw when the water in the bottle is heated? A. The water level goes down. B. The water level stays the same. C. The water level goes up. Why
WATER Date 1. What happens to the level of the water in the straw when the water in the bottle is heated? A. The water level goes down. B. The water level stays the same. C. The water level goes up. Why
Art caption: Natural resources such as rocks are mined in rock quarries (KWOR-eez) like this one. DRAFT
 1 OBSERVING EARTH S RESOURCES INVESTIGATION Humans use a lot of materials found naturally on Earth. There are many materials, including metals such as copper and woods such as pine. Materials that are
1 OBSERVING EARTH S RESOURCES INVESTIGATION Humans use a lot of materials found naturally on Earth. There are many materials, including metals such as copper and woods such as pine. Materials that are
yk4lm (1:26) vd_90 (4:37)
 Properties of Water Video Notes http://www.youtube.com/watch?v=2vfld5 yk4lm (1:26) http://www.youtube.com/watch?v=ivu4nl vd_90 (4:37) https://www.youtube.com/watch?v=0ens nj4zfz8#t=4.4600086 (4:58) new
Properties of Water Video Notes http://www.youtube.com/watch?v=2vfld5 yk4lm (1:26) http://www.youtube.com/watch?v=ivu4nl vd_90 (4:37) https://www.youtube.com/watch?v=0ens nj4zfz8#t=4.4600086 (4:58) new
Science Supply List Earth Science
 2017-2018 Science Supply List Earth Science 2017 Glynlyon, Inc. Table of Contents UNIT 1: DYNAMIC STRUCTURE OF EARTH... 1 UNIT 2: FORCES AND FEATURES OF EARTH... 1 UNIT 3: FEATURES OF EARTH'S CRUST...
2017-2018 Science Supply List Earth Science 2017 Glynlyon, Inc. Table of Contents UNIT 1: DYNAMIC STRUCTURE OF EARTH... 1 UNIT 2: FORCES AND FEATURES OF EARTH... 1 UNIT 3: FEATURES OF EARTH'S CRUST...
EXTRA CREDIT - EXPERIMENT G ELECTROCHEMISTRY ACTIVITY OF METALS
 EXTRA CREDIT - EXPERIMENT G ELECTROCHEMISTRY ACTIVITY OF METALS INTRODUCTION The objective of this experiment is to develop an abbreviated activity series of metals using: 1. Displacement reactions 2.
EXTRA CREDIT - EXPERIMENT G ELECTROCHEMISTRY ACTIVITY OF METALS INTRODUCTION The objective of this experiment is to develop an abbreviated activity series of metals using: 1. Displacement reactions 2.
Physical Geology, 15/e
 Lecture Outlines Physical Geology, 15/e Plummer, Carlson & Hammersley Resources Physical Geology 15/e, Chapter 22 Types of Geologic Resources Energy resources petroleum (oil and natural gas), coal, uranium,
Lecture Outlines Physical Geology, 15/e Plummer, Carlson & Hammersley Resources Physical Geology 15/e, Chapter 22 Types of Geologic Resources Energy resources petroleum (oil and natural gas), coal, uranium,
Estuary Adventures. Background. Objective
 Estuary Adventures Objective Students will work in groups to understand the concept of estuaries, their importance, and the role that density plays in the mixing of fresh and salt water. Students will
Estuary Adventures Objective Students will work in groups to understand the concept of estuaries, their importance, and the role that density plays in the mixing of fresh and salt water. Students will
esources / ^ Vocabulary Process Skill
 / ^ Vocabulary natural resource, C38 renewable resource, C40 nonrenewable resource, C41 pollution, C42 esources Why do miners dig deep into Earth? They are looking for useful materials, such as metals,
/ ^ Vocabulary natural resource, C38 renewable resource, C40 nonrenewable resource, C41 pollution, C42 esources Why do miners dig deep into Earth? They are looking for useful materials, such as metals,
Chapter 3: Ocean Water
 Chapter 3: Ocean Water CHAPTER 3 PROJECT - DENSITY BOTTLES During the Golf Ball Density lab, you will see the ways in which fresh water interacts with salt water when the two are mixed. The two forms of
Chapter 3: Ocean Water CHAPTER 3 PROJECT - DENSITY BOTTLES During the Golf Ball Density lab, you will see the ways in which fresh water interacts with salt water when the two are mixed. The two forms of
OPERATING INSTRUCTIONS
 Water Sampler # 15010 OPERATING INSTRUCTIONS Purpose: Contents: For the collection of water samples at varying depths or distances away from a shoreline. One (1) Assembled Water Sampler One (1) Cord (15
Water Sampler # 15010 OPERATING INSTRUCTIONS Purpose: Contents: For the collection of water samples at varying depths or distances away from a shoreline. One (1) Assembled Water Sampler One (1) Cord (15
2.3 Chemical Changes corrosion Kinds of Corrosion
 2.3 Chemical Changes Have you ever wondered why metal car bodies rust but plastic bumpers do not? As you know, different substances have different physical properties, such as colour and hardness, and
2.3 Chemical Changes Have you ever wondered why metal car bodies rust but plastic bumpers do not? As you know, different substances have different physical properties, such as colour and hardness, and
THE NUCLEAR FUEL CYCLE
 Getting to the Core of THE NUCLEAR FUEL CYCLE From the mining of uranium to the disposal of nuclear waste @ Getting to the Core of the Nuclear Fuel Cycle The various activities associated with the production
Getting to the Core of THE NUCLEAR FUEL CYCLE From the mining of uranium to the disposal of nuclear waste @ Getting to the Core of the Nuclear Fuel Cycle The various activities associated with the production
Water Pollution: Sediment
 Invisible Passengers Students perform an experiment to investigate the characteristics of sediment in water. They will also discuss the effects of various types of pollution on aquatic organisms. Level(s):
Invisible Passengers Students perform an experiment to investigate the characteristics of sediment in water. They will also discuss the effects of various types of pollution on aquatic organisms. Level(s):
Part I: Salish Sea Introduction. Review:
 Part I: Salish Sea Introduction Review: The Salish Sea The Salish Sea was formed about 20,000 years ago during the last ice age by the carving action of glaciers. There are 3 major parts of the Salish
Part I: Salish Sea Introduction Review: The Salish Sea The Salish Sea was formed about 20,000 years ago during the last ice age by the carving action of glaciers. There are 3 major parts of the Salish
WONDERFUL, WATERFUL WETLANDS
 WONDERFUL, WATERFUL WETLANDS OBJECTIVES The student will do the following: 1. List characteristics of wetlands. SUBJECTS: Science, Language Arts TIME: 60 minutes 2. Describe the functions of a wetland.
WONDERFUL, WATERFUL WETLANDS OBJECTIVES The student will do the following: 1. List characteristics of wetlands. SUBJECTS: Science, Language Arts TIME: 60 minutes 2. Describe the functions of a wetland.
Properties of Minerals
 What is a mineral? Properties of Minerals Mineral 5 characteristics: naturally occurring (not made by people) inorganic (cannot come from living things) solid (definite shape and volume) crystal structure
What is a mineral? Properties of Minerals Mineral 5 characteristics: naturally occurring (not made by people) inorganic (cannot come from living things) solid (definite shape and volume) crystal structure
CHAPTER 1. Conversions. General Plan for Converting Measurements. State the relationship between the unit given and the unit sought as an equality.
 CHAPTER 1 Conversions One of the aims of chemistry is to describe changes to tell what changed, how it changed, and what it changed into. Another aim of chemistry is to look at matter and its changes and
CHAPTER 1 Conversions One of the aims of chemistry is to describe changes to tell what changed, how it changed, and what it changed into. Another aim of chemistry is to look at matter and its changes and
water, forestry, fossil fuels, metallic and non-metallic minerals
 water, forestry, fossil fuels, metallic and non-metallic minerals Faith / spirituality (water baptisms, FNMI water stories, etc) Sustains us / necessary for life Sanitation Transportation (canoe, boat,
water, forestry, fossil fuels, metallic and non-metallic minerals Faith / spirituality (water baptisms, FNMI water stories, etc) Sustains us / necessary for life Sanitation Transportation (canoe, boat,
Iron filings (Fe) 56g IRON + SULPHUR IRON SULPHIDE
 W.S.51. Chemical reactions. All of the different materials around us have been formed by chemical reactions from about one hundred simple elements. The diagram below shows a chemical reaction between the
W.S.51. Chemical reactions. All of the different materials around us have been formed by chemical reactions from about one hundred simple elements. The diagram below shows a chemical reaction between the
85 Q.51 Which of the following carbonates would give the metal when heated with carbon? (1) MgCO 3 (2) PbCO 3 (3) K 2 CO 3 (4) CuCO 3
 Metal and metal reactivity / Section 2 / Sect2pp.doc / S. W. Tse / P.1 85 Q.51 Which of the following carbonates would give the metal when heated with carbon? (1) MgCO 3 (2) PbCO 3 (3) K 2 CO 3 (4) CuCO
Metal and metal reactivity / Section 2 / Sect2pp.doc / S. W. Tse / P.1 85 Q.51 Which of the following carbonates would give the metal when heated with carbon? (1) MgCO 3 (2) PbCO 3 (3) K 2 CO 3 (4) CuCO
3 The Formation, Mining, and Use of Minerals
 CHAPTER 3 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
CHAPTER 3 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
Chapter 16 Minerals: A Nonrenewable Resource
 Lecture Outline: Chapter 16 Minerals: A Nonrenewable Resource I. Introduction to Minerals A. Minerals are elements or compounds of elements that occur naturally in Earth s crust and have precise chemical
Lecture Outline: Chapter 16 Minerals: A Nonrenewable Resource I. Introduction to Minerals A. Minerals are elements or compounds of elements that occur naturally in Earth s crust and have precise chemical
Covered with a thin layer of oxide at ordinary temperatures.
 1 More about Metals Physical properties of metals In general metals have luster, are malleable and ductile, good conductors of heat and electricity and have high boiling and melting points and nonmetals
1 More about Metals Physical properties of metals In general metals have luster, are malleable and ductile, good conductors of heat and electricity and have high boiling and melting points and nonmetals
Materials. Materials. NOTE Delta Education Customer Service can be reached at
 w Weather and Water Measuring Matter Materials Materials Contents Kit Inventory List... 42 Materials Supplied by the Teacher... 45 Preparing the Kit for Your Classroom... 47 Care, Reuse, and Recycling...
w Weather and Water Measuring Matter Materials Materials Contents Kit Inventory List... 42 Materials Supplied by the Teacher... 45 Preparing the Kit for Your Classroom... 47 Care, Reuse, and Recycling...
Temperature: Air vs. Water vs. More Water
 Temperature: Air vs. Water vs. More Water Temperature Adapted from: A Change in the Weather? in Living in Water. National Aquarium in Baltimore, 1997. Grade Level: basic Duration: 1 class Setting: classroom
Temperature: Air vs. Water vs. More Water Temperature Adapted from: A Change in the Weather? in Living in Water. National Aquarium in Baltimore, 1997. Grade Level: basic Duration: 1 class Setting: classroom
Curriculum Map: Elementary Science Grade 3 Course: Gr 3 Science
 Curriculum Map: Elementary Science Grade 3 Course: Gr 3 Science Course Description: Science & Technology & Engineering Education: Science & Technology & Engineering Education is taught through the inquiry
Curriculum Map: Elementary Science Grade 3 Course: Gr 3 Science Course Description: Science & Technology & Engineering Education: Science & Technology & Engineering Education is taught through the inquiry
GROUNDWATER. Narrative
 GROUNDWATER Narrative BIG IDEAS: Groundwater. is between the grains of sand and gravel.. moves because gravity works underground just like it does above ground.. is connected to surface water. 4. is part
GROUNDWATER Narrative BIG IDEAS: Groundwater. is between the grains of sand and gravel.. moves because gravity works underground just like it does above ground.. is connected to surface water. 4. is part
Edexcel GCSE Chemistry. Topic 4: Extracting metals and equilibria. Obtaining and using metals. Notes.
 Edexcel GCSE Chemistry Topic 4: Extracting metals and equilibria Obtaining and using metals Notes 4.1 Deduce the relative reactivity of some metals, by their reactions with water, acids and salt solutions
Edexcel GCSE Chemistry Topic 4: Extracting metals and equilibria Obtaining and using metals Notes 4.1 Deduce the relative reactivity of some metals, by their reactions with water, acids and salt solutions
POLLUTANTS: HOW MUCH TOTAL OR HOW MUCH PER UNIT OF WATER?
 POLLUTANTS: HOW MUCH TOTAL OR HOW MUCH PER UNIT OF WATER? 9-12 OBJECTIVES The student will do the following: 1. Define the concept of concentration as different from an expression of total amount. 2. Explain
POLLUTANTS: HOW MUCH TOTAL OR HOW MUCH PER UNIT OF WATER? 9-12 OBJECTIVES The student will do the following: 1. Define the concept of concentration as different from an expression of total amount. 2. Explain
BUILDING A WATERSHED MODEL
 BUILDING A WATERSHED MODEL OBJECTIVES Define a watershed and describe how it functions Identify that students live in a watershed within the San Antonio River Basin TOPICS Watersheds Runoff TEKS ALIGNMENT
BUILDING A WATERSHED MODEL OBJECTIVES Define a watershed and describe how it functions Identify that students live in a watershed within the San Antonio River Basin TOPICS Watersheds Runoff TEKS ALIGNMENT
Cadmium Testing and On Site Calibration for Water Testing Detection Range: ppm
 Document: AND016 04 2014 Cadmium Testing and On Site Calibration for Water Testing Detection Range: 0.1 1.0ppm April, 2014 Edition 3 ANDalyze, Inc., 2012. All rights reserved. Printed in USA. Table of
Document: AND016 04 2014 Cadmium Testing and On Site Calibration for Water Testing Detection Range: 0.1 1.0ppm April, 2014 Edition 3 ANDalyze, Inc., 2012. All rights reserved. Printed in USA. Table of
look down at cross on paper paper cross on paper
 1 The equation for the reaction between sodium thiosulfate and hydrochloric acid is given below. Na 2 S 2 O 3 (aq) + 2HCl (aq) 2NaCl (aq) + S(s) + SO 2 (g) + H 2 O(l) The speed of this reaction was investigated
1 The equation for the reaction between sodium thiosulfate and hydrochloric acid is given below. Na 2 S 2 O 3 (aq) + 2HCl (aq) 2NaCl (aq) + S(s) + SO 2 (g) + H 2 O(l) The speed of this reaction was investigated
PLANET EARTH: Fresh Water
 PLANET EARTH: Fresh Water Teacher s Guide Grade Level: 9-12 Running Time: 42 minutes Program Description Follow rivers as they course from mountain to the sea, nourishing unique and dramatic wildlife and
PLANET EARTH: Fresh Water Teacher s Guide Grade Level: 9-12 Running Time: 42 minutes Program Description Follow rivers as they course from mountain to the sea, nourishing unique and dramatic wildlife and
Description: Students build models of sinkholes to gain an intuitive knowledge of their physical aspects.
 A Hole in the Ground Description: Students build models of sinkholes to gain an intuitive knowledge of their physical aspects. Learning Objectives: Students will learn how groundwater creates sinkholes.
A Hole in the Ground Description: Students build models of sinkholes to gain an intuitive knowledge of their physical aspects. Learning Objectives: Students will learn how groundwater creates sinkholes.
Significance of Water Observation Lab
 Significance of Water Observation Lab It has been said that the chemistry of life is water chemistry. Because of its chemical properties, water is the medium in which most of life s chemical reactions
Significance of Water Observation Lab It has been said that the chemistry of life is water chemistry. Because of its chemical properties, water is the medium in which most of life s chemical reactions
Water, Water, Everywhere? Students explore the availability of water for humans and wildlife use.
 Students explore the availability of water for humans and wildlife use. Grade Level: 3-4, K-2, 5-8 Time: Two 30-45 minute periods Learning Objectives: Students will be able to: Explain how much water is
Students explore the availability of water for humans and wildlife use. Grade Level: 3-4, K-2, 5-8 Time: Two 30-45 minute periods Learning Objectives: Students will be able to: Explain how much water is
Calcium and Magnesium; Chlorophosphonazo Rapid Liquid Method Method to 1000 µg/l Ca and Mg as CaCO 3 (ULR) Pour-Thru Cell
 Hardness, Total DOC316.53.01045 Calcium and Magnesium; Chlorophosphonazo Rapid Liquid Method Method 8374 4 to 1000 µg/l Ca and Mg as CaCO 3 (ULR) Pour-Thru Cell Scope and application: For boiler and ultrapure
Hardness, Total DOC316.53.01045 Calcium and Magnesium; Chlorophosphonazo Rapid Liquid Method Method 8374 4 to 1000 µg/l Ca and Mg as CaCO 3 (ULR) Pour-Thru Cell Scope and application: For boiler and ultrapure
Recrystallization with a Single Solvent
 Experiment: Recrystallization Part II: Purification of Solids In Part I of the recrystallization experiment, you learned about the factors which make a good recrystallization solvent, and you learned how
Experiment: Recrystallization Part II: Purification of Solids In Part I of the recrystallization experiment, you learned about the factors which make a good recrystallization solvent, and you learned how
Marbury Water System Lead and Copper Sampling Plan
 Marbury Water System Lead and Copper Sampling Plan Page 1 Table of Contents Section 1 Marbury Water System Material List... Page 3 Lead & Copper Sample Site Plan Selection Criteria for Community Systems.
Marbury Water System Lead and Copper Sampling Plan Page 1 Table of Contents Section 1 Marbury Water System Material List... Page 3 Lead & Copper Sample Site Plan Selection Criteria for Community Systems.
Chapter 6, Lesson 11: Chemical Reactions & Engineering Design
 Chapter 6, Lesson 11: Chemical Reactions & Engineering Design NGSS Standard: MS-PS1-6 Undertake a design project to construct, test, and modify a device that either releases or absorbs thermal energy by
Chapter 6, Lesson 11: Chemical Reactions & Engineering Design NGSS Standard: MS-PS1-6 Undertake a design project to construct, test, and modify a device that either releases or absorbs thermal energy by
Determination of the Empirical Formula of Magnesium Oxide
 Determination of the Empirical Formula of Magnesium Oxide The quantitative stoichiometric relationships governing mass and amount will be studied using the combustion reaction of magnesium metal. Magnesium
Determination of the Empirical Formula of Magnesium Oxide The quantitative stoichiometric relationships governing mass and amount will be studied using the combustion reaction of magnesium metal. Magnesium
There are 2 different types of numbers Exact -Measured
 Significant Figures When measuring or using our calculators we must determine the correct answer; our calculators are mindless drones and don t know the correct answer. There are 2 different types of numbers
Significant Figures When measuring or using our calculators we must determine the correct answer; our calculators are mindless drones and don t know the correct answer. There are 2 different types of numbers
INVESTIGATION: Flour vs. Bread: How Soil Aggregate Structure Influences Water Flows. Time: minutes, depending on the amount of discussion.
 INVESTIGATION: Flour vs. Bread: How Soil Aggregate Structure Influences Water Flows Time: 15-60 minutes, depending on the amount of discussion. Grade Level: appropriate for any age. Summary: If I only
INVESTIGATION: Flour vs. Bread: How Soil Aggregate Structure Influences Water Flows Time: 15-60 minutes, depending on the amount of discussion. Grade Level: appropriate for any age. Summary: If I only
Which 750" ml bottle of coke would you pick?
 Which 750" ml bottle of coke would you pick? You have recognized the variability of the 750" ml coke pop bottle process! What are the sources of the variability? Which process is the more variable? You
Which 750" ml bottle of coke would you pick? You have recognized the variability of the 750" ml coke pop bottle process! What are the sources of the variability? Which process is the more variable? You
What is Purified Water?
 What is Purified Water? The majority of the human body is comprised of water and human beings can live only a very short time without water. Water is a chemical substance that is essential to all known
What is Purified Water? The majority of the human body is comprised of water and human beings can live only a very short time without water. Water is a chemical substance that is essential to all known
Curriculum Guide. National Science Education Standards Grades 5-8 Earth and Space Science
 National Science Education Standards Grades 5-8 Earth and Space Science Curriculum Guide Structure of the Earth system: Global patterns of atmospheric movement influence local weather. Oceans has a major
National Science Education Standards Grades 5-8 Earth and Space Science Curriculum Guide Structure of the Earth system: Global patterns of atmospheric movement influence local weather. Oceans has a major
Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet
 Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet Name: Sec: Date: Q1.Choose the best answer. 1. Which of the following is an element? a) steam b) sugar c)dry ice d) sulphur 2. Which
Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet Name: Sec: Date: Q1.Choose the best answer. 1. Which of the following is an element? a) steam b) sugar c)dry ice d) sulphur 2. Which
Teaching Guidelines. What do you think are the most important properties of a fuel?
 ENERGY IN A BOTTLE Teaching Guidelines Summary: Students work in teams to carry out and present research concerning the chemistry of energy storage in fuels and batteries. Subject: Science Topics: Chemistry
ENERGY IN A BOTTLE Teaching Guidelines Summary: Students work in teams to carry out and present research concerning the chemistry of energy storage in fuels and batteries. Subject: Science Topics: Chemistry
Determination of the Empirical Formula of an Unknown Lead Oxide
 Determination of the Empirical Formula of an Unknown Lead Oxide by: Student Name Abstract Using a process known as smelting, 1 the empirical formula of an unknown lead oxide was determined 2 to be PbO
Determination of the Empirical Formula of an Unknown Lead Oxide by: Student Name Abstract Using a process known as smelting, 1 the empirical formula of an unknown lead oxide was determined 2 to be PbO
An Oxidation-Reduction Titration: The Reaction of Fe 2+ and Ce 4+
 An Oxidation-Reduction Titration: The Reaction of Fe 2+ and Ce 4+ LAB ADV COMP 8 From Advanced Chemistry with Vernier, Vernier Software & Technology, 2004 INTRODUCTION A titration, as you recall, is a
An Oxidation-Reduction Titration: The Reaction of Fe 2+ and Ce 4+ LAB ADV COMP 8 From Advanced Chemistry with Vernier, Vernier Software & Technology, 2004 INTRODUCTION A titration, as you recall, is a
WHAT TO DO? ELECTROPLATING OR METAL SPRAYING?
 WHAT TO DO? ELECTROPLATING OR METAL SPRAYING? S. de Vries, J.W. Rauhorst, B. Plasse & F. Kemeling The first step to improve and control the quality of a metal product starts at the desk of the designer.
WHAT TO DO? ELECTROPLATING OR METAL SPRAYING? S. de Vries, J.W. Rauhorst, B. Plasse & F. Kemeling The first step to improve and control the quality of a metal product starts at the desk of the designer.
Metal and Non Metals
 Metal and Non Metals Malleable Ductile Sonorous Conductor Insulator KEYWORDS Rusting Brass Calcium Chloride Galvanising Reactivity Metals on the Periodic table Metals Non Metals Metals and their properties
Metal and Non Metals Malleable Ductile Sonorous Conductor Insulator KEYWORDS Rusting Brass Calcium Chloride Galvanising Reactivity Metals on the Periodic table Metals Non Metals Metals and their properties
HOW TO BUILD A PIZZA BOX SOLAR OVEN
 HOW TO BUILD A PIZZA BOX SOLAR OVEN ACTIVITY Get cooking with the sun! Use one of our MANY sunny days to learn how you can trap the sun s natural heat to make a homemade oven. KEY CONCEPTS ENERGY SOLAR
HOW TO BUILD A PIZZA BOX SOLAR OVEN ACTIVITY Get cooking with the sun! Use one of our MANY sunny days to learn how you can trap the sun s natural heat to make a homemade oven. KEY CONCEPTS ENERGY SOLAR
Experiment 13: Determination of Molecular Weight by Freezing Point Depression
 1 Experiment 13: Determination of Molecular Weight by Freezing Point Depression Objective: In this experiment, you will determine the molecular weight of a compound by measuring the freezing point of a
1 Experiment 13: Determination of Molecular Weight by Freezing Point Depression Objective: In this experiment, you will determine the molecular weight of a compound by measuring the freezing point of a
Close Reading and Text Dependent Questions in Science Thermal Equilibrium (Physics HS)
 Close Reading and Text Dependent Questions in Science Thermal Equilibrium (Physics HS) The text selection, Thermal Equilibrium, can be found at the following link: http://www.buzzle.com/articles/thermal-
Close Reading and Text Dependent Questions in Science Thermal Equilibrium (Physics HS) The text selection, Thermal Equilibrium, can be found at the following link: http://www.buzzle.com/articles/thermal-
1 Magnets and Magnetic Fields
 CHAPTER 18 1 Magnets and Magnetic Fields ECTIO Magnetism KEY IDEA As you read this section, keep these questions in mind: What happens when the s of two magnets come close together? What causes a magnet
CHAPTER 18 1 Magnets and Magnetic Fields ECTIO Magnetism KEY IDEA As you read this section, keep these questions in mind: What happens when the s of two magnets come close together? What causes a magnet
Climate Change Connected Curriculum
 Climate Change Connected Curriculum Climate change is happening, humans are causing it, and I think this is perhaps the most serious environmental issue facing us. - - - - -Bill Nye Climate Change across
Climate Change Connected Curriculum Climate change is happening, humans are causing it, and I think this is perhaps the most serious environmental issue facing us. - - - - -Bill Nye Climate Change across
MINING IN A NUTSHELL - Advanced Version
 INTRODUCTION MINING IN A NUTSHELL - Advanced Version Mining in a nutshell, advanced 1 Minerals are used by people and societies to produce goods. These minerals are found in the Earth and must be extracted
INTRODUCTION MINING IN A NUTSHELL - Advanced Version Mining in a nutshell, advanced 1 Minerals are used by people and societies to produce goods. These minerals are found in the Earth and must be extracted
7.9.6 Magnetic Poles. 85 minutes. 117 marks. Page 1 of 37
 7.9.6 Magnetic Poles 85 minutes 117 marks Page 1 of 37 Q1. The diagram shows an electromagnet used in a door lock. (a) The push switch is closed and the door unlocks. Explain in detail how this happens.
7.9.6 Magnetic Poles 85 minutes 117 marks Page 1 of 37 Q1. The diagram shows an electromagnet used in a door lock. (a) The push switch is closed and the door unlocks. Explain in detail how this happens.
Lab Question from Board papers Class-09[2018] 1. How will you separate a mixture of iron filings, iodine and common salt?
![Lab Question from Board papers Class-09[2018] 1. How will you separate a mixture of iron filings, iodine and common salt? Lab Question from Board papers Class-09[2018] 1. How will you separate a mixture of iron filings, iodine and common salt?](/thumbs/77/74991575.jpg) Lab Question from Board papers Class-09[2018] SET-1 Short Answer Type Questions (2 marks) 1. How will you separate a mixture of iron filings, iodine and common salt? Ans. (i) Remove iron filings with the
Lab Question from Board papers Class-09[2018] SET-1 Short Answer Type Questions (2 marks) 1. How will you separate a mixture of iron filings, iodine and common salt? Ans. (i) Remove iron filings with the
EXPERIMENT 6. Determination of the Ideal Gas Law Constant - R. Magnesium metal reacts with hydrochloric acid according to the following reaction,
 EXPERIMENT 6 Determination of the Ideal Gas Law Constant - R Magnesium metal reacts with hydrochloric acid according to the following reaction, Mg + 2 HCl MgCl 2 + H 2 (g) In this experiment you will use
EXPERIMENT 6 Determination of the Ideal Gas Law Constant - R Magnesium metal reacts with hydrochloric acid according to the following reaction, Mg + 2 HCl MgCl 2 + H 2 (g) In this experiment you will use
Make Five. A game about the chemical formulas of some common minerals
 Make Five A game about the chemical formulas of some common minerals By defi nition, a mineral has a defi nite chemical composition. It s sort of like a recipe. This game is for students who want to (or
Make Five A game about the chemical formulas of some common minerals By defi nition, a mineral has a defi nite chemical composition. It s sort of like a recipe. This game is for students who want to (or
Marginal Costing Q.8
 Marginal Costing. 2008 Q.8 Break-Even Point. Before tackling a marginal costing question, it s first of all crucial that you understand what is meant by break-even point. What this means is that a firm
Marginal Costing. 2008 Q.8 Break-Even Point. Before tackling a marginal costing question, it s first of all crucial that you understand what is meant by break-even point. What this means is that a firm
Student Exploration: Mineral Identification
 Name: Date: Student Exploration: Identification Vocabulary: crystal, density, hardness, homogeneous, luster, mass, mineral, streak, volume Prior Knowledge Questions (Do these BEFORE using the Gizmo.) 1.
Name: Date: Student Exploration: Identification Vocabulary: crystal, density, hardness, homogeneous, luster, mass, mineral, streak, volume Prior Knowledge Questions (Do these BEFORE using the Gizmo.) 1.
DIRTY WATER? Identifying Bad Water Quality Problems. by Richard Gellert
 DIRTY WATER? Identifying Bad Water Quality Problems 92 MAXIMUM YIELD USA November / December 2007 The quality of the water you are feeding your plants is directly correlated to the quality of your harvest.
DIRTY WATER? Identifying Bad Water Quality Problems 92 MAXIMUM YIELD USA November / December 2007 The quality of the water you are feeding your plants is directly correlated to the quality of your harvest.
R = kc Eqn 5.1. R = k C Eqn 5.2. R = kc + B Eqn 5.3
 5 Standard addition 5.1 Introduction An interference is anything that causes an analysis to be incorrect, which means in practice, that the measured concentration for the presented sample is wrong. There
5 Standard addition 5.1 Introduction An interference is anything that causes an analysis to be incorrect, which means in practice, that the measured concentration for the presented sample is wrong. There
Technical Process Bulletin
 ALODINE 1600 Technical Process Bulletin Technical Process Bulletin No. This Revision: 02/12/2007 1. Introduction: ALODINE 1600 is a concentrated two-package liquid chemical used to produce a hexavalent
ALODINE 1600 Technical Process Bulletin Technical Process Bulletin No. This Revision: 02/12/2007 1. Introduction: ALODINE 1600 is a concentrated two-package liquid chemical used to produce a hexavalent
Salinity in Seawater
 Salinity in Seawater Objective To familiarize students with the different methods used for measuring salinity of water. Introduction: Salinity exerts profound impacts on the marine environment. It controls
Salinity in Seawater Objective To familiarize students with the different methods used for measuring salinity of water. Introduction: Salinity exerts profound impacts on the marine environment. It controls
Name: 2017 AP Environmental Science Summer Assignment
 P a g e 1 Name: 2017 AP Environmental Science Summer Assignment The Summer Assignment is due on the first day of class. P a g e 2 AP Environmental Science Summer Assignment (This assignment is due on the
P a g e 1 Name: 2017 AP Environmental Science Summer Assignment The Summer Assignment is due on the first day of class. P a g e 2 AP Environmental Science Summer Assignment (This assignment is due on the
ALE 20. Crystalline Solids, Unit Cells, Liquids and the Uniqueness of Water
 Name Chem 162, Section: Group Number: ALE 20. Crystalline Solids, Unit Cells, Liquids and the Uniqueness of Water (Reference: pp. 463 473 of Sec. 12.6 Silberberg 5 th edition) How are the particles within
Name Chem 162, Section: Group Number: ALE 20. Crystalline Solids, Unit Cells, Liquids and the Uniqueness of Water (Reference: pp. 463 473 of Sec. 12.6 Silberberg 5 th edition) How are the particles within
PE PLE WHO POLLUTED THE RIVER?
 UNIT 7 PEOPLE AND WASTE WHO POLLUTED THE RIVER? COUNTING ON PE PLE K-5 Activities for Global Citizenship METHOD Through an interactive story, K-2 students experience the pollution of a local river over
UNIT 7 PEOPLE AND WASTE WHO POLLUTED THE RIVER? COUNTING ON PE PLE K-5 Activities for Global Citizenship METHOD Through an interactive story, K-2 students experience the pollution of a local river over
