TABLE. Page 1. Annual Percentage Increase in the Consumer Price Indexand Health Care. Expenditures in Eight Industrialized Countries...
|
|
|
- Beatrix Kennedy
- 5 years ago
- Views:
Transcription
1 Introduction
2 Page Rising Medical Care Expenditures The Diffusion of Medical Technologies Description of This Volume Description of Five Medical Technologies Chapter l References TABLE Table No. Page 1. Annual Percentage Increase in the Consumer Price Indexand Health Care Expenditures in Eight Industrialized Countries FIGURE Figure No. Page l. Stagesin the Development and Diffusion of Medical Technologies
3 1 Introduction; RISING MEDICAL CARE EXPENDITURES the rapid rate of growth in health expenditures can be stemmed simply by eliminating technologies and services that do not provide any benefit. Unfortunately, however, this is not an adequate solution to the problem of rising costs. The reason is that most new technologies do appear to have at least some benefit, however small or costly. Examples of technologies that fall into this category are halfway technologies such as organ transplantation, artificial organs, many cancer therapies, and current treatment for coronary artery disease (11). Since the growth of resources used for medical care, over and above the effects of economywide inflation, is the primary reason for rapidly rising costs, nations seeking to control health care costs must effectively control the growth and/or use of new resources. Inevitably, this effort will involve them in controlling the processes by which technologies are developed, evaluated, adopted, and used. And fundamentally, this means that they will be forced to choose among beneficial technologies, providing some to the fullest extent, others to a limited extent, and still others not at all.
4 4 Backround Paper #4: The Management of Health Care Technology it Ten Countries THE DIFFUSION OF MEDICAL TECHNOLOGIES Public intervention to control the diffusion and use of specific technologies is likely to be related to one or another of the four theoretical stages in the process of development and diffusion of medical technologies shown in figure 1. The first stage, basic research, produces new knowledge about the biological mechanisms underlying the normal functioning of the human body and its malfunction in disease (6). In the second stage, applied R&D, this basic information is used to create new solutions to problems in the prevention, treatment, or cure of disease. The next stage, clinical investigation and testing, involves the testing of new medical technologies in human subjects. This stage encompasses a range of activities from first human use to large-scale clinical trials and demonstration projects to demonstrate efficacy and safety (s). Efficacy is the benefit from use of a technology; safety is a measure of the risk of a technology. Finally, as a new technology appears to be of value, clinicians begin to use it and patients begin to ask for it. As more and more physicians use the technology on more and more patients, the extent of its use increases. This is the process of diffusion, Diffusion may end with the technology s attainment of an appropriate level of use. Alternatively, it may end with the technology s being abandoned, either because it was of no value or because a more effective technology Figure 1. Stages in the Development and Diffusion of Medical Technologies SOURCE. Office of Technology Assessment, U S Congress, Development of Medical Technology Opportunities for Assessment (Washington, D C U S Government Printing Office, 1976) (6)
5 has been developed, or with its being used too much or too little. The model sequence depicted in figure 1 is attractive because it offers a way to understand the process of development and diffusion of medical technologies such as drugs, devices, and procedures. In reality, however, medical technologies emerge from a process that is far less systematic than the model implies. In the ideal model of diffusion, for example, scientific evaluation of efficacy and safety is an integral part of the diffusion process. In the real world of medical care and health care policy, however, such evaluation is often not done (5). Epidemiological and statistical methods have been developed to measure scientifically the benefits and risks of a technolog y under controlled conditions. Increasingly, these methods, and especially the controlled clinical trial, have been proposed as the basis for decisions concerning medical technology. DESCRIPTION OF THIS VOLUME All industrialized countries have begun to experiment with the kinds of mechanisms that will be necessary to effect changes in development, diffusion, and use of medical technologies. The general and specific public policies that affect the development and diffusion of medical technologies in nine industrialized countries are discussed in chapters 2 through 10 of this volume: the United Kingdom (ch. 2), Canada (ch. 3), Australia (ch. 4), Japan (ch. 5), France (ch. 6), West Germany (ch. 7), the Netherlands (ch. 8), Iceland (ch. 9), and Sweden (ch. 10). In chapter 11, U.S. policies pertaining to the development and diffusion of medical technologies are compared to the policies of the other nine countries. Also compared are the United States and other countries experience with five specific technologies: 1 ) computed tomograph y scanners,2) renal dialysis, 3) coronary bypass surgery, 4) cobalt therapy, and 5) automated clinical laboratories. Generally, each chapter begins with an introductory section in which the author briefly describes the country s form of government and economy. Following this is a section in which the country s medical care system is discussed. In the third section of each chapter, the country s policies concerning the R&D, evaluation, and regulation of medical technologies are examined. Along with institutions for biomedical research, government funding of research, and priority areas of research, government policies toward and support of the evaluation of medical technologies are discussed. Also covered are safety and efficacy regulation, health planning and related investment controls, utilization review, and both general health care financing arrangements and financing arrangements specific to technologies. To help illustrate the application of the country s general policies, in the fourth section of each chapter the country s treatment of five specific technologies is examined. As background for the policy discussions in the remaining chapters of this volume, these five technologies are defined below, and their uses and costs briefly identified. DESCRIPTION OF FIVE MEDICAL TECHNOLOGIES Computed tomography (CT) scanners. The CT scanner is a diagnostic device that combines X-ray equipment with a computer and a cathode ray tube (television-like device) to produce images of cross-sections of the human body (7). The first machines were head scanners, designed to produce images of abnormalities within the skull (e. g., brain tumors). These machines were developed in Britain in the late 1960 s. Body scanners able to scan the rest of the body as well as the head have been developed more recentlv. - J
6 6. Backround Paper #4: The Management of Health Cure Technology it Ten Countries Following its development, the CT scanner was quickly hailed as the greatest advance in radiology since the discovery of X-rays. CT scanning was rapidly and enthusiastically accepted by the medical community. More recently, however, three factors the rapid spread of CT scanners, the frequency of their use, and the expenditures associated with them have combined to focus attention on the contribution of CT and other diagnostic medical technologies to the recent growth of medical care expenditures. The concern over expenditures has also caused decisionmakers to examine policies pertaining to other medical technologies. In 1979, a CT scanner cost, on average, more than $500,000 to buy and $400,000 to $500,000 a year to operate. That year, the United States had more than 1,200 scanners, so the cost of scanning in 1979 was more than $500 million. Renal dialysis. Hemodialysis and renal transplantation are two life-extending therapies that were developed in the early 1960 s for victims of end-stage renal disease. End-stage renal disease is a clinical condition reached when a person has such a degree of deterioration of kidney function that without treatment he or she will soon die. Hemodialysis is the process of removing toxic waste products from the blood by means of an artificial kidney. The first dialysis machine was built in Holland in the early 1940 s, but could be used only for short periods of time (6). Longterm dialysis became possible when Scribner and his colleagues developed the Scribner shunt. This device, a semipermanent apparatus that linked an artery to a vein, could be used to connect a patient to a dialysis machine, without surgery for each session of dialysis. A patient generally requires dialysis about three times a week. Renal transplantation is a surgical procedure whereby a healthy kidney from a living person or a person who has recently died is substituted for an individual s nonfunctioning kidney. Transplantation has become more and more reliable, but is still in a somewhat experimental stage. The recipient s body tends to reject the kidney graft, and drugs are necessary to suppress this rejection. Concerns about the treatment of end-stage renal disease in both the United States and other countries have focused on costs. In the United States, it was estimated in 1975 that the average annual charge for dialysis received in a hospital was $30,500; $27,500 for nonhospital dialysis; $14,000 for the first year of dialysis at home, and $7,000 for successive years (8). Transplantation charges averaged about $12,000. The treatment of end-stage renal disease has been covered under the medicaid program since 1972, and cost the program $573 million in Costs in 1979 were expected to exceed $1 billion. Coronary bypass surgery. Coronary bypass surgery is a surgical procedure in which a graft is placed between the aorta and a coronary artery to bypass a constricted portion of the artery and thus improve oxygen supply to the heart muscle (5). The surgery is used as a treatment of coronary artery disease, a disease caused by narrowing and blocking of the arteries that supply blood to the heart. This disease is the number one cause of death in the United States. In 1975, it caused 642,719 deaths. Coronary bypass surgery came into practice in the early 1970 s. Approximately 25,000 operations were performed in the United States in 1973, and perhaps 100,000 in In 1977, the total cost of coronary bypass surgery in the United States averaged $15,000 per patient. If 100,000 operations were performed in 1978, the aggregate costs to the Nation were more than $1.5 billion. The benefits of coronary bypass surgery for all classes of patients with coronary artery disease have not been clearly demonstrated. Cobalt therapy. Cobalt therapy is a form of radiation therapy (9). Radiation therapy is used almost exclusively for the treatment of cancer, either to cure it or to alleviate its symptoms. In the United States, there are approximately 300 new cases of cancer per 100,000 population each year. Including both new and previously discovered cases, 430 people per 100,000 population are treated for cancer each year. About 70 percent of those who are treated for cancer receive radiation therapy at some point
7 during their illness. It is difficult to evaluate the benefits of radiation therapy. Not only is it generally used in combination with other therapies, but its benefits must be weighed against sometimes serious side effects. Furthermore, the therapeutic goal is often to alleviate rather than to cure. In 1975, the cost of purchasing a cobalt therapy unit was about $90,000 to $125,000. Construction costs are high because of the need to shield staff and the surrounding population from dangerous radiation. The issue with cobalt therapy, as with many other large and expensive technologies, concerns the number and distribution of units. Most experts believe that, like many expensive technologies, cobalt therapy should be centrally located to ensure access and located in a specialized medical center to permit optimal use. Automated clinical laboratories. The primary function of the clinical laboratory is to analyze and provide data on samples of body tissues or fluids. By correlating these data with firsthand observations and results of other tests, physicians are better able to make accurate diagnoses and to determine the proper therapy for their patients. Appropriate and reliable data from clinical laboratories are essential for current medical practice. The automation of clinical laboratories began in the late 1950 s with the marketing of the continuous-flow blood analyzer, a machine that performs multiple tests on a single sample of blood. Newer machines have improved on the original blood analyzer, and clinical laboratory functions in addition to blood analysis have since been automated. By 1972, more than 50 percent of U.S. hospitals had automated their hematology and/or chemis try laboratories. Automating clinical laboratory functions both lowered unit costs for laboratory tests improved the reliability and validit y of the has and test results. At the same time, however, the ready availability of automated equipment has stimulated the use of laboratory tests and increased the total volume of tests performed to such an extent, in fact, that the value of much of this testing is now in question. In 1975, 5 billion laboratory tests were done in the United States at an estimated cost of $15 billion (6). The number of tests was increasing both for hospitalized and ambulatory patients. Between 1969 and 1976, the average number of tests provided per patient per day in the hospital rose from 2.3 to 5,0, an average annual increase of 11.1 percent (1). During the same period, the average cost per test rose by $0.22, from $1.34 to $1.56. In 1976, it was projected that the total volume of laboratory tests nationally would rise at the rate of 11 percent a year (6). CHAPTER 1 REFERENCES 1. AHA Indexes, Growth in Laboratory Tests Indicative of Increase in Service Intensity, Hospztak 53:46, Altman, S., and Wallack, S., Technology on Trial Is It the Culprit Behind Rising Health Costs? The Case For and Against, in Medical Tech )Iology: Tile Culprit Belli)ld Health Care Costs? edited by S. Altman and R. Blendon, DHEW publication No. (PHS) (Hyattsville, Md: National Center for Health Services Research, and Bureau of Health Planning, 1979). 3. Cohn, V., and Miluis, P., They Make Good by Making Well, Waslzi)lgto)z Post, January 7, Fineberg, H. V., Gastric Freezing A Study of Diffusion of a Medical Innovation, in Medical Teclz~lology and tlze Healt)l Care Systet}l (Washington, D. C.: National Academy of Sciences, Committee on Technology and Health Care, 1979). 5. Office of Technology Assessment, U.S. Congress, Assessing the Efficacy and Safety of Medical Technologies, GPO stock No o (Washington, D. C.: U.S. Government Printing Office, September 1978). 6., Dezlelopment of Medical Techtlology: Opportu)lities for Assessment, GPO stock No (Washington, D. C.: U.S. Gov-
8 7 < 8. ernment Printing Office, August 1976). 9. Policy Implications of t}?e Computed To)nog raphy (CT) Scar?ner, GPO stock No (Washington, D. C.: U.S. Government Printing Office, August 1978). Rettig, R., ln?plet~le)ltitlg the E) Id-Stage Re/zal Disease Program of Medicare, draft report for 11. Health Care Financing Administration, grant No. 18-P-90742/9-01 (Washington, D. C.: RAND Corp., 1979). Russell, L., Tech) zology i}z Hospitals (Washington, D. C.: Brookings Institution, 1979). Simanis, J. G., and Coleman, J. R., Health Care Expenditures in Nine Industrialized Countries, , social Secz{rity Blllleti)l 43:3, Thomas, L., T//e l.i~~es of a Cell (New York, N. Y.: Viking Press, 1974).
A Coordinated Registry Network Based on the Vascular Quality Initiative: VISION. Vascular Implant Surveillance & Interventional Outcomes Network
 A Coordinated Registry Network Based on the Vascular Quality Initiative: VISION Vascular Implant Surveillance & Interventional Outcomes Network Jack L. Cronenwett, MD Medical Director, Society for Vascular
A Coordinated Registry Network Based on the Vascular Quality Initiative: VISION Vascular Implant Surveillance & Interventional Outcomes Network Jack L. Cronenwett, MD Medical Director, Society for Vascular
characteristics of living things.notebook September 08, 2017
 CHARACTERISTICS OF LIVING THINGS Create a list of things that all living things have in common. All living things: 1. Are composed of cells. 2. Use energy Living things take in energy and use it for maintenance
CHARACTERISTICS OF LIVING THINGS Create a list of things that all living things have in common. All living things: 1. Are composed of cells. 2. Use energy Living things take in energy and use it for maintenance
Ethics of Radiation Protection in Medicine
 Ethics of Radiation Protection in Medicine Richard J. Vetter, Ph.D. CHP Professor Emeritus Mayo Clinic First IRPA North American Workshop on the Ethics of Radiological Protection Baltimore, Maryland, USA
Ethics of Radiation Protection in Medicine Richard J. Vetter, Ph.D. CHP Professor Emeritus Mayo Clinic First IRPA North American Workshop on the Ethics of Radiological Protection Baltimore, Maryland, USA
Brain Tumour Australia Information FACT SHEET 22 Clinical Trials: Questions and Answers
 FACT SHEET 22 Clinical Trials: Questions and Answers What is a clinical trial? Clinical trials are research studies that answer scientific questions and try to find better ways to prevent, screen for,
FACT SHEET 22 Clinical Trials: Questions and Answers What is a clinical trial? Clinical trials are research studies that answer scientific questions and try to find better ways to prevent, screen for,
social, and population-based research conducted and supported by the National Institutes of Health (NIH). The
 Statement by the Ad Hoc Group for Medical Research on FY 2015 Appropriations for the National Institutes of Health Submitted for the record on March 28, 2014, to the Subcommittee on Labor, Health and Human
Statement by the Ad Hoc Group for Medical Research on FY 2015 Appropriations for the National Institutes of Health Submitted for the record on March 28, 2014, to the Subcommittee on Labor, Health and Human
National Museum of Health and Medicine
 National Museum of Health and Medicine Accession Number: 2014.0006 Date of Records: 1951-1955 Size: 2 boxes Finding Aid by: Eric W. Boyle (2014) Otis Historical Archives MacNeill Collection Background
National Museum of Health and Medicine Accession Number: 2014.0006 Date of Records: 1951-1955 Size: 2 boxes Finding Aid by: Eric W. Boyle (2014) Otis Historical Archives MacNeill Collection Background
Medical Research: Participation Issues
 Medical Research: Participation Issues Key Points Types of medical solutions vary Various factors influence support & adoption Clinical trials are a critical, expensive piece of the approval process Threat
Medical Research: Participation Issues Key Points Types of medical solutions vary Various factors influence support & adoption Clinical trials are a critical, expensive piece of the approval process Threat
Testimony of Christopher Newton-Cheh, MD, MPH Volunteer for the American Heart Association
 Testimony of Christopher Newton-Cheh, MD, MPH Volunteer for the American Heart Association Before the House Energy and Commerce Subcommittee on Health 21st Century Cures: Examining the Regulation of Laboratory
Testimony of Christopher Newton-Cheh, MD, MPH Volunteer for the American Heart Association Before the House Energy and Commerce Subcommittee on Health 21st Century Cures: Examining the Regulation of Laboratory
External Defibrillator Improvement Initiative
 External Defibrillator Improvement Initiative November 2010 Center for Devices and Radiological Health U.S. Food and Drug Administration External Defibrillator Improvement Initiative Table of Contents
External Defibrillator Improvement Initiative November 2010 Center for Devices and Radiological Health U.S. Food and Drug Administration External Defibrillator Improvement Initiative Table of Contents
Achievement Level Descriptors for Medical Interventions
 Achievement Level Descriptors for Medical Interventions Achievement Level Descriptors (ALDs) are statements of what students should know and be able to do in a PLTW classroom. The ALDs can be used to better
Achievement Level Descriptors for Medical Interventions Achievement Level Descriptors (ALDs) are statements of what students should know and be able to do in a PLTW classroom. The ALDs can be used to better
INDIVIDUALIZED TREATMENT
 1 INDIVIDUALIZED TREATMENT The healthcare system is under pressure: better budgetary compliance, quicker treatment, more equitable patient rights, and higher expectations from patients and relatives. These
1 INDIVIDUALIZED TREATMENT The healthcare system is under pressure: better budgetary compliance, quicker treatment, more equitable patient rights, and higher expectations from patients and relatives. These
Medical instrumentationi 11/19/2010
 Medical instrumentationi BIOEN 302 11/19/2010 Medical instrumentation Definition: instrument for sensing, diagnostics, therapeutics or surgery of human being. 2 Medical instrumentation Definition: instrument
Medical instrumentationi BIOEN 302 11/19/2010 Medical instrumentation Definition: instrument for sensing, diagnostics, therapeutics or surgery of human being. 2 Medical instrumentation Definition: instrument
First Place Essay. Katie P.
 First Place Essay Katie P. Eighth Grade West Reading, PA 19611 Bioscience research is seeking information by exploring life's processes and diseases. It has helped many scientists understand how the body
First Place Essay Katie P. Eighth Grade West Reading, PA 19611 Bioscience research is seeking information by exploring life's processes and diseases. It has helped many scientists understand how the body
The Path to Lung Transplantation: Roles of Extracorporeal Membrane Oxygenation (ECMO)
 Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/medical-breakthroughs-from-penn-medicine/path-lung-transplantationroles-extracorporeal-membrane-oxygenation-ecmo/7851/
Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/medical-breakthroughs-from-penn-medicine/path-lung-transplantationroles-extracorporeal-membrane-oxygenation-ecmo/7851/
ORGANIZATION AND ROLE OF A PHASE I ONCOLOGY UNIT. Dr Philippe CASSIER Centre Léon Bérard, Lyon
 ORGANIZATION AND ROLE OF A PHASE I ONCOLOGY UNIT Dr Philippe CASSIER Centre Léon Bérard, Lyon Outline Cancer & Oncology Drug development in oncology Specificity of phase I trials in oncology Study design
ORGANIZATION AND ROLE OF A PHASE I ONCOLOGY UNIT Dr Philippe CASSIER Centre Léon Bérard, Lyon Outline Cancer & Oncology Drug development in oncology Specificity of phase I trials in oncology Study design
Statements on the Regulation of Laboratory Developed Tests
 Statements on the Regulation of Laboratory Developed Tests Current Regulatory Gaps and Perspectives on Oversight of LDTs American Cancer Society Cancer Action Network says Molecular tests, in particular,
Statements on the Regulation of Laboratory Developed Tests Current Regulatory Gaps and Perspectives on Oversight of LDTs American Cancer Society Cancer Action Network says Molecular tests, in particular,
Insight to Gene Techno Science Co.,Ltd
 Unlimited drug discovery from the beginning Ticker symbol: 4584 Insight to Gene Techno Science Co.,Ltd November 2017 2 Corporate Overview Chief Executive Founded March 2001 Masaharu Tani, President Listed
Unlimited drug discovery from the beginning Ticker symbol: 4584 Insight to Gene Techno Science Co.,Ltd November 2017 2 Corporate Overview Chief Executive Founded March 2001 Masaharu Tani, President Listed
Unit 1 Wellness and Homeostasis
 Unit 1 Wellness and Homeostasis Organs and Tissues Available for Transplant Lesson Plan Objectives Connections to Curriculum Personal Perspectives/ Reflection Decision Making Working in Science B11-1-03
Unit 1 Wellness and Homeostasis Organs and Tissues Available for Transplant Lesson Plan Objectives Connections to Curriculum Personal Perspectives/ Reflection Decision Making Working in Science B11-1-03
THE MEDICAL DIRECTIVE
 THE MEDICAL DIRECTIVE As part of a person s right to self-determination, every adult may accept or refuse any recommended medical treatment. This is relatively easy when people are well and can speak.
THE MEDICAL DIRECTIVE As part of a person s right to self-determination, every adult may accept or refuse any recommended medical treatment. This is relatively easy when people are well and can speak.
COPE. FAQs CORPORATE ONCOLOGY PROGRAM FOR EMPLOYEES
 COPE CORPORATE ONCOLOGY PROGRAM FOR EMPLOYEES FAQs This brochure addresses FAQ and is prepared solely for the German market. Local adjustments may be necessary. Please contact us if you require additional
COPE CORPORATE ONCOLOGY PROGRAM FOR EMPLOYEES FAQs This brochure addresses FAQ and is prepared solely for the German market. Local adjustments may be necessary. Please contact us if you require additional
New test for transplant rejection on the horizon
 December 6, 2004 New test for transplant rejection on the horizon Vancouver Researchers at St. Paul s Hospital and Vancouver General Hospital are developing a revolutionary new test to diagnose and facilitate
December 6, 2004 New test for transplant rejection on the horizon Vancouver Researchers at St. Paul s Hospital and Vancouver General Hospital are developing a revolutionary new test to diagnose and facilitate
HEALTH TECHNOLOGY ASSESSMENTS IN KOREA
 HEALTH TECHNOLOGY ASSESSMENTS IN KOREA July 24, 2012 HTA and Coverage Decisions Conference Taipei, Taiwan Jeonghoon Ahn, PhD Senior Director National Evidence-based healthcare Collaborating Agency (NECA)
HEALTH TECHNOLOGY ASSESSMENTS IN KOREA July 24, 2012 HTA and Coverage Decisions Conference Taipei, Taiwan Jeonghoon Ahn, PhD Senior Director National Evidence-based healthcare Collaborating Agency (NECA)
April 15, 2015 VIA ELECTRONIC MAIL
 April 15, 2015 VIA ELECTRONIC MAIL Patricia Brooks, RHIA Senior Technical Advisor Centers for Medicare and Medicaid Services Hospital and Ambulatory Policy Group Mail Stop C4-08-06 7500 Security Boulevard
April 15, 2015 VIA ELECTRONIC MAIL Patricia Brooks, RHIA Senior Technical Advisor Centers for Medicare and Medicaid Services Hospital and Ambulatory Policy Group Mail Stop C4-08-06 7500 Security Boulevard
MAYO CLINIC CENTER FOR BIOMEDICAL DISCOVERY EXCEPTIONAL RESEARCH LEADS TO EXCEPTIONAL PATIENT CARE
 MAYO CLINIC CENTER FOR BIOMEDICAL DISCOVERY EXCEPTIONAL RESEARCH LEADS TO EXCEPTIONAL PATIENT CARE THE RESEARCH WE DO TODAY WILL DETERMINE THE TYPE OF MEDICAL AND SURGICAL PRACTICE WE CARRY ON AT THE CLINIC
MAYO CLINIC CENTER FOR BIOMEDICAL DISCOVERY EXCEPTIONAL RESEARCH LEADS TO EXCEPTIONAL PATIENT CARE THE RESEARCH WE DO TODAY WILL DETERMINE THE TYPE OF MEDICAL AND SURGICAL PRACTICE WE CARRY ON AT THE CLINIC
Current Market Dynamics and Future Vision of the Care Cycle
 Current Market Dynamics and Future Vision of the Care Cycle Tim Irish Analysts Meeting June 15 th, 2005 Overview Market and Customer trends The Care Cycle and Molecular Medicine 2 Healthcare is the world
Current Market Dynamics and Future Vision of the Care Cycle Tim Irish Analysts Meeting June 15 th, 2005 Overview Market and Customer trends The Care Cycle and Molecular Medicine 2 Healthcare is the world
Nanotechnology and Advanced Materials for more effective Healthcare
 Nanotechnology and Advanced Materials for more effective Healthcare This challenge taps into the potential of nanotechnologies and advanced materials to enable more effective therapies and diagnostics
Nanotechnology and Advanced Materials for more effective Healthcare This challenge taps into the potential of nanotechnologies and advanced materials to enable more effective therapies and diagnostics
Re: Request for Comments regarding the July 30, 2008 CMS Posting of Potential National Coverage Decision Topics
 September 26, 2008 Steve Phurrough, M.D. Coverage and Analysis Group Centers for Medicare & Medicaid Services Department of Health and Human Services Mailstop: C1-13-18 7500 Security Blvd. Baltimore, MD
September 26, 2008 Steve Phurrough, M.D. Coverage and Analysis Group Centers for Medicare & Medicaid Services Department of Health and Human Services Mailstop: C1-13-18 7500 Security Blvd. Baltimore, MD
MRI Systems Market to 2018
 MRI Systems Market to 2018 Technological advancements, Increasing Number of Applications and Advent of MRI Compatible Pacemakers to Drive Future Growth GBI Research Report Guidance GBI Research Report
MRI Systems Market to 2018 Technological advancements, Increasing Number of Applications and Advent of MRI Compatible Pacemakers to Drive Future Growth GBI Research Report Guidance GBI Research Report
The NYSCF Research Institute
 Regenerative Medicine Research Update Susan L. Solomon The NYSCF Research Institute New York Pharma Forum February 25, 2016 Regenerative Medicine Research Regenerative medicine uses laboratory grown human
Regenerative Medicine Research Update Susan L. Solomon The NYSCF Research Institute New York Pharma Forum February 25, 2016 Regenerative Medicine Research Regenerative medicine uses laboratory grown human
THE NEW 640 SLICE CT SCANNER
 THE NEW 640 SLICE CT SCANNER www.ahdubai.com The American Hospital Dubai has recently acquired a state-ofthe-art computerized tomography (CT) scanner that houses a variety of intelligent and industry-leading
THE NEW 640 SLICE CT SCANNER www.ahdubai.com The American Hospital Dubai has recently acquired a state-ofthe-art computerized tomography (CT) scanner that houses a variety of intelligent and industry-leading
Translational & Molecular Imaging Institute
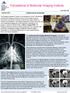 Translational & Molecular Imaging Institute tmii.mssm.edu Summer 2015 CARDIOVASCULAR IMAGING The Imaging Research Center is the backbone of the Translational & Molecular Imaging Institute at Mount Sinai
Translational & Molecular Imaging Institute tmii.mssm.edu Summer 2015 CARDIOVASCULAR IMAGING The Imaging Research Center is the backbone of the Translational & Molecular Imaging Institute at Mount Sinai
ORACLE OTTAWA REGION FOR ADVANCED CARDIOVASCULAR RESEARCH EXCELLENCE STRATEGIC DIRECTIONS
 ORACLE OTTAWA REGION FOR ADVANCED CARDIOVASCULAR RESEARCH EXCELLENCE STRATEGIC DIRECTIONS 2018-2024 ORACLE 2.0: Leading Globally, from Innovation to Impact P1 P2 NOTE FROM OUR LEADERS NOTE FROM VP RESEARCH,
ORACLE OTTAWA REGION FOR ADVANCED CARDIOVASCULAR RESEARCH EXCELLENCE STRATEGIC DIRECTIONS 2018-2024 ORACLE 2.0: Leading Globally, from Innovation to Impact P1 P2 NOTE FROM OUR LEADERS NOTE FROM VP RESEARCH,
IDSA is pleased to offer the following comments on specific priority areas identified by FDA:
 October 31, 2018 Shashi Malhotra Office of Acquisitions & Grants Services (OAGS) Food and Drug Administration Telephone: 240-402-7592 Email: Shashi.Malhotra@fda.hhs.gov SUBJECT: NOT-FD-18-16: Development
October 31, 2018 Shashi Malhotra Office of Acquisitions & Grants Services (OAGS) Food and Drug Administration Telephone: 240-402-7592 Email: Shashi.Malhotra@fda.hhs.gov SUBJECT: NOT-FD-18-16: Development
Stem Cell Research: Identifying emerging high priority policy issues
 The state stem cell agency Stem Cell Research: Identifying emerging high priority policy issues Ellen G. Feigal, M.D. SVP, Research and Development National Cancer Policy Summit Washington, DC November
The state stem cell agency Stem Cell Research: Identifying emerging high priority policy issues Ellen G. Feigal, M.D. SVP, Research and Development National Cancer Policy Summit Washington, DC November
MOSAIQUES DIAGNOSTICS
 MOSAIQUES DIAGNOSTICS CLINICAL PROTEOMICS IN DRUG DEVELOPMENT INFORMATION Clinical Proteomics for early and differential diagnosis FDA Letter of Support Possibilities of a companion test in Drug Development
MOSAIQUES DIAGNOSTICS CLINICAL PROTEOMICS IN DRUG DEVELOPMENT INFORMATION Clinical Proteomics for early and differential diagnosis FDA Letter of Support Possibilities of a companion test in Drug Development
PLTW Biomedical Science Medical Interventions Course Outline
 Follow the fictitious Smith family as you learn about the prevention, diagnosis, and treatment of disease. Play the role of biomedical professionals to analyze case information and diagnose and treat your
Follow the fictitious Smith family as you learn about the prevention, diagnosis, and treatment of disease. Play the role of biomedical professionals to analyze case information and diagnose and treat your
Innovation and Technology in Breast Cancer 2018 A research initiative from Breast Cancer Foundation NZ.
 Request for Proposals Innovation and Technology in Breast Cancer 2018 A research initiative from Breast Cancer Foundation NZ. Overview Breast Cancer Foundation NZ (BCFNZ) is committed to funding research
Request for Proposals Innovation and Technology in Breast Cancer 2018 A research initiative from Breast Cancer Foundation NZ. Overview Breast Cancer Foundation NZ (BCFNZ) is committed to funding research
ADDIS ABABA UNIVERSITY CENTER OF BIOMEDICAL ENGINEERING
 ADDIS ABABA UNIVERSITY CENTER OF BIOMEDICAL ENGINEERING November 2013 History of Biomedical Engineering Definition of Biomedical Engineering Achievements of Biomedical Engineering Streams in Biomedical
ADDIS ABABA UNIVERSITY CENTER OF BIOMEDICAL ENGINEERING November 2013 History of Biomedical Engineering Definition of Biomedical Engineering Achievements of Biomedical Engineering Streams in Biomedical
REIMAGINING DRUG DEVELOPMENT:
 Biology Reconstructed REIMAGINING DRUG DEVELOPMENT: Accurate Disease Modeling To Drive Successful Therapies Julia Kirshner, CEO julia@zpredicta.com 1 SUCCESS RATES OF DRUG DEVELOPMENT ARE LOW, " PARTICULARLY
Biology Reconstructed REIMAGINING DRUG DEVELOPMENT: Accurate Disease Modeling To Drive Successful Therapies Julia Kirshner, CEO julia@zpredicta.com 1 SUCCESS RATES OF DRUG DEVELOPMENT ARE LOW, " PARTICULARLY
Data Mining for Genomic- Phenomic Correlations
 Data Mining for Genomic- Phenomic Correlations Joyce C. Niland, Ph.D. Associate Director & Chair, Information Sciences Rebecca Nelson, Ph.D. Lead, Data Mining Section City of Hope National Medical Center
Data Mining for Genomic- Phenomic Correlations Joyce C. Niland, Ph.D. Associate Director & Chair, Information Sciences Rebecca Nelson, Ph.D. Lead, Data Mining Section City of Hope National Medical Center
Occams Business Research & Consultancy
 Occams Business Research & Consultancy http://www.marketresearch.com/occams Business Research Consultancy v4040/ Publisher Sample Phone: 800.298.5699 (US) or +1.240.747.3093 or +1.240.747.3093 (Int'l)
Occams Business Research & Consultancy http://www.marketresearch.com/occams Business Research Consultancy v4040/ Publisher Sample Phone: 800.298.5699 (US) or +1.240.747.3093 or +1.240.747.3093 (Int'l)
Re: Medicare Program; Hospital Outpatient Prospective Payment and Ambulatory Surgical Center Payment Systems; Proposed Rule; CMS-1656-P
 August 26, 2016 Andrew Slavitt Acting Administrator Centers for Medicare and Medicaid Services Department of Health and Human Services Attention: CMS-1656-P Mail Stop C4-26-05 7500 Security Boulevard Baltimore,
August 26, 2016 Andrew Slavitt Acting Administrator Centers for Medicare and Medicaid Services Department of Health and Human Services Attention: CMS-1656-P Mail Stop C4-26-05 7500 Security Boulevard Baltimore,
HCCA 13 TH ANNUAL COMPLIANCE INSTITUTE LAS VEGAS, NV APRIL 29, 2009
 HCCA 13 TH ANNUAL COMPLIANCE INSTITUTE LAS VEGAS, NV APRIL 29, 2009 MEDICARE COVERAGE ANALYSIS WORKSHOP: THE HOW TO OF MEDICARE COVERAGE IN RESEARCH Suzanne LivPage, J.D. Director, Clinical Research Initiation
HCCA 13 TH ANNUAL COMPLIANCE INSTITUTE LAS VEGAS, NV APRIL 29, 2009 MEDICARE COVERAGE ANALYSIS WORKSHOP: THE HOW TO OF MEDICARE COVERAGE IN RESEARCH Suzanne LivPage, J.D. Director, Clinical Research Initiation
Innovation and Technology in Breast Cancer 2019 A research initiative from Breast Cancer Foundation NZ.
 Request for Proposals Innovation and Technology in Breast Cancer 2019 A research initiative from Breast Cancer Foundation NZ. Overview Breast Cancer Foundation NZ (BCFNZ) is committed to funding research
Request for Proposals Innovation and Technology in Breast Cancer 2019 A research initiative from Breast Cancer Foundation NZ. Overview Breast Cancer Foundation NZ (BCFNZ) is committed to funding research
Computer-Aided Surgical Navigation Coding Guide Neurosurgery. May 1, 2009
 Computer-Aided Surgical Navigation Coding Guide Neurosurgery May 1, 2009 Please direct any questions to: Kim Brew Manager, Reimbursement and Therapy Access Medtronic Surgical Technologies (904) 279-7569
Computer-Aided Surgical Navigation Coding Guide Neurosurgery May 1, 2009 Please direct any questions to: Kim Brew Manager, Reimbursement and Therapy Access Medtronic Surgical Technologies (904) 279-7569
EIGHT BASIC ELEMENTS OF INFORMED CONSENT
 1/6 This guidance addresses: o Eight Basic elements of informed consent o Additional elements of informed consent o Other information required by the IRB EIGHT BASIC ELEMENTS OF INFORMED CONSENT Required
1/6 This guidance addresses: o Eight Basic elements of informed consent o Additional elements of informed consent o Other information required by the IRB EIGHT BASIC ELEMENTS OF INFORMED CONSENT Required
Preparing For A New Era of Medical Product Development
 Latham & Watkins Health Care & Life Sciences Practice Number 1607 November 7, 2013 Preparing For A New Era of Medical Product Development FDA report demonstrates support for personalized medicine and more
Latham & Watkins Health Care & Life Sciences Practice Number 1607 November 7, 2013 Preparing For A New Era of Medical Product Development FDA report demonstrates support for personalized medicine and more
Medical Electronic Devices
 Medical Electronic Devices Basic Electronics (Electronic devices, Amplifiers, A/D, D/A) Electrodes EEG Deep brain stimulator ECG Cardiac Pacemakers External Defibrillators EMG Neuromuscular Electrical
Medical Electronic Devices Basic Electronics (Electronic devices, Amplifiers, A/D, D/A) Electrodes EEG Deep brain stimulator ECG Cardiac Pacemakers External Defibrillators EMG Neuromuscular Electrical
2111: ALL Post-HCT. Add/ Remove/ Modify. Manual Section. Date. Description. Comprehensive Disease- Specific Manuals
 2111: ALL Post-HCT The Acute Lymphoblastic Leukemia Post-HCT Data Form is one of the Comprehensive Report Forms. This form captures ALL-specific post-hct data such as: planned treatments post-hct, the
2111: ALL Post-HCT The Acute Lymphoblastic Leukemia Post-HCT Data Form is one of the Comprehensive Report Forms. This form captures ALL-specific post-hct data such as: planned treatments post-hct, the
Thailand Medical Industry Overview
 Medical Industry Thailand Medical Industry Overview Thailand s Healthcare Expenditure as % of GDP [Unit in %] 20 17,1 15 10 5 11,3 9,7 9,1 7,2 5,6 4,6 4,6 4 3,1 0 USA Germany Sweden UK S.Korea China Thailand
Medical Industry Thailand Medical Industry Overview Thailand s Healthcare Expenditure as % of GDP [Unit in %] 20 17,1 15 10 5 11,3 9,7 9,1 7,2 5,6 4,6 4,6 4 3,1 0 USA Germany Sweden UK S.Korea China Thailand
in94070.txt at Page 1 of 5 2/24/00
 in94070.txt at www.nrc.gov Page 1 of 5 UNITED STATES NUCLEAR REGULATORY COMMISSION OFFICE OF NUCLEAR MATERIAL SAFETY AND SAFEGUARDS WASHINGTON, D.C. 20555 NRC INFORMATION NOTICE 94-70: ISSUES ASSOCIATED
in94070.txt at www.nrc.gov Page 1 of 5 UNITED STATES NUCLEAR REGULATORY COMMISSION OFFICE OF NUCLEAR MATERIAL SAFETY AND SAFEGUARDS WASHINGTON, D.C. 20555 NRC INFORMATION NOTICE 94-70: ISSUES ASSOCIATED
NHS ENGLAND BOARD PAPER
 NHS ENGLAND BOARD PAPER Paper: PB.30.03.2017/06 Title: Creating a genomic medicine service to lay the foundations to deliver personalised interventions and treatments Lead Director: Professor Sir Bruce
NHS ENGLAND BOARD PAPER Paper: PB.30.03.2017/06 Title: Creating a genomic medicine service to lay the foundations to deliver personalised interventions and treatments Lead Director: Professor Sir Bruce
Biotech Patents in Europe
 Biotech Patents in Europe Introduction This circular relates to biotech patent practice in Europe. It is based on our experience of drafting and prosecuting biotech applications. The circular is written
Biotech Patents in Europe Introduction This circular relates to biotech patent practice in Europe. It is based on our experience of drafting and prosecuting biotech applications. The circular is written
THE GLOBAL MARKET FOR STEM CELLS
 THE GLOBAL MARKET FOR STEM CELLS BIO035F May 2016 Paul Evers Project Analyst ISBN: 1-62296-292-3 BCC Research 49 Walnut Park, Building 2 Wellesley, MA 02481 USA 866-285-7215 (toll-free within the USA),
THE GLOBAL MARKET FOR STEM CELLS BIO035F May 2016 Paul Evers Project Analyst ISBN: 1-62296-292-3 BCC Research 49 Walnut Park, Building 2 Wellesley, MA 02481 USA 866-285-7215 (toll-free within the USA),
Japan Update - Implementation of PMD Act - March, 2015
 Japan Update - Implementation of PMD Act - March, 2015 Topics Implementation of PMD Act; Revision of Pharmaceutical Affairs Law (PAL) PMDA Medical Device Training Seminar 2 Implementation of PMD Act: The
Japan Update - Implementation of PMD Act - March, 2015 Topics Implementation of PMD Act; Revision of Pharmaceutical Affairs Law (PAL) PMDA Medical Device Training Seminar 2 Implementation of PMD Act: The
Pilot Socioeconomic Impact Analysis of CFI and CIHR Funding: Medical Imaging R&D
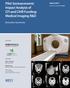 Pilot Socioeconomic Impact Analysis of CFI and CIHR Funding: Medical Imaging R&D March 2013 RTI Project Number 0213097 Executive Summary Prepared for Prepared by Alan C. O Connor RTI International Albert
Pilot Socioeconomic Impact Analysis of CFI and CIHR Funding: Medical Imaging R&D March 2013 RTI Project Number 0213097 Executive Summary Prepared for Prepared by Alan C. O Connor RTI International Albert
Flordeliza Villanueva and colleagues were the first to prove the principle that with the use of ultrasound heart disease could be diagnosed in a
 Flordeliza Villanueva and colleagues were the first to prove the principle that with the use of ultrasound heart disease could be diagnosed in a living being at the cellular level. Here is an early version
Flordeliza Villanueva and colleagues were the first to prove the principle that with the use of ultrasound heart disease could be diagnosed in a living being at the cellular level. Here is an early version
FDA PLANS TO REGULATE TESTS AS DEVICES. August 2010 SPECIAL REPRINT. By Ellen Flannery and Scott Danzis
 August 2010 SPECIAL REPRINT FDA PLANS TO REGULATE LABORATOR ORY DEVELOPED TESTS AS DEVICES By Ellen Flannery and Scott Danzis Reproduced with the kind permission of Global Regulatory Press from the Journal
August 2010 SPECIAL REPRINT FDA PLANS TO REGULATE LABORATOR ORY DEVELOPED TESTS AS DEVICES By Ellen Flannery and Scott Danzis Reproduced with the kind permission of Global Regulatory Press from the Journal
Synagis (Palivizumab) Drug Prior Authorization Protocol
 Line of Business: Medi-Cal Effective Date: August 16, 2017 Revision Date: August 16, 2017 Synagis (Palivizumab) Drug Prior Authorization Protocol This policy has been developed through review of medical
Line of Business: Medi-Cal Effective Date: August 16, 2017 Revision Date: August 16, 2017 Synagis (Palivizumab) Drug Prior Authorization Protocol This policy has been developed through review of medical
Medical Technology: Cost Impact. Medical Technology EBPA 2008 EBPA 2008 EBPA 2008
 Medical Technology: Cost Impact Employee Benefit Planning Association November 6, 2008 Mark Lyons, RPh, MBA Director, Care Facilitation Consulting Healthcare Costs continue to rise Current medical costs
Medical Technology: Cost Impact Employee Benefit Planning Association November 6, 2008 Mark Lyons, RPh, MBA Director, Care Facilitation Consulting Healthcare Costs continue to rise Current medical costs
National MS Society Information Sourcebook
 National MS Society Information Sourcebook www.nationalmssociety.org/sourcebook Interferons The interferons are a group of natural proteins that are produced by human cells in response to viral infection
National MS Society Information Sourcebook www.nationalmssociety.org/sourcebook Interferons The interferons are a group of natural proteins that are produced by human cells in response to viral infection
1. Executive Summary
 1. Executive Summary 1.1 General The fluoroscope is defined as an instrument used chiefly in industry and in the practice of medicine for observing the internal structure of objects (such as the living
1. Executive Summary 1.1 General The fluoroscope is defined as an instrument used chiefly in industry and in the practice of medicine for observing the internal structure of objects (such as the living
Brazil Gastric Balloon Procedures Outlook to 2020
 Brazil Gastric Balloon Procedures Outlook to 2020 Reference Code: GDMECC0472PDB Publication Date: July 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 3 1.2 List of Figures...
Brazil Gastric Balloon Procedures Outlook to 2020 Reference Code: GDMECC0472PDB Publication Date: July 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 3 1.2 List of Figures...
Defining the true market
 Defining the true market Case study: How real-world data and outcomes improved a major pharmaceutical company s launch strategy Cardinal Health Specialty Solutions Competing in a crowded marketplace A
Defining the true market Case study: How real-world data and outcomes improved a major pharmaceutical company s launch strategy Cardinal Health Specialty Solutions Competing in a crowded marketplace A
Investigational New Devices
 Page 1 of 14 Investigational New Devices I. Purpose This document describes the policies and procedures for conducting studies involving investigational new devices at Stanford Hospital & Clinics (SHC)
Page 1 of 14 Investigational New Devices I. Purpose This document describes the policies and procedures for conducting studies involving investigational new devices at Stanford Hospital & Clinics (SHC)
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: clinical_trial_services 3/2002 2/2018 2/2019 8/2018 Description of Procedure or Service Clinical trials are
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: clinical_trial_services 3/2002 2/2018 2/2019 8/2018 Description of Procedure or Service Clinical trials are
United Kingdom Tissue Engineered - Skin Substitutes Market Outlook to 2020
 United Kingdom Tissue Engineered - Skin Substitutes Market Outlook to Reference Code: GDMECC0860DB Publication Date: October 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables...
United Kingdom Tissue Engineered - Skin Substitutes Market Outlook to Reference Code: GDMECC0860DB Publication Date: October 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables...
THERAC-25. Computerized Radiation Therapy. Report by: TROY GALLAGHER
 1 of 5 28/12/2007 9:45 AM THERAC-25 Computerized Radiation Therapy Report by: TROY GALLAGHER Introduction: The use of computers in the medical field is becoming more and more widely used. Computers are
1 of 5 28/12/2007 9:45 AM THERAC-25 Computerized Radiation Therapy Report by: TROY GALLAGHER Introduction: The use of computers in the medical field is becoming more and more widely used. Computers are
FDA s Same Surgical Procedure Draft Guidance Could Stifle Use of Autologous Stem Cell Therapies
 FDA s Same Surgical Procedure Draft Guidance Could Stifle Use of Autologous Stem Cell Therapies Executive Summary, June 2015 Life Sciences Practice Group AUTHOR Areta L. Kupchyk Nixon Peabody LLP Washington,
FDA s Same Surgical Procedure Draft Guidance Could Stifle Use of Autologous Stem Cell Therapies Executive Summary, June 2015 Life Sciences Practice Group AUTHOR Areta L. Kupchyk Nixon Peabody LLP Washington,
Medical Technology: Cost Impact
 Medical Technology: Cost Impact Employee Benefit Planning Association November 6, 2008 Mark Lyons, RPh, MBA Director, Care Facilitation Consulting Healthcare Costs continue to rise Current medical costs
Medical Technology: Cost Impact Employee Benefit Planning Association November 6, 2008 Mark Lyons, RPh, MBA Director, Care Facilitation Consulting Healthcare Costs continue to rise Current medical costs
COVERAGE ELIGIBILITY OF SERVICES ASSOCIATED WITH A CLINICAL TRIAL
 Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline must be read in its
Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline must be read in its
Chapter XIII. USRDS Research Studies. Appreciation Of Renal Community Participation. Ongoing USRDS Special Studies
 Chapter XIII T he USRDS conducts two different kinds of studies: Existing Data Studies and Special Studies. Existing Data Studies analyze data that already have been collected; these studies will be discussed
Chapter XIII T he USRDS conducts two different kinds of studies: Existing Data Studies and Special Studies. Existing Data Studies analyze data that already have been collected; these studies will be discussed
SEMINAR ON HEALTH CARE SYSTEMS INNOVATION. Comparative Effectiveness The Savior"? ESD.69 HST.926. October, 2012
 SEMINAR ON HEALTH CARE ESD.69 HST.926 SYSTEMS INNOVATION Comparative Effectiveness The Savior"? Stan N. Finkelstein, M.D. Massachusetts Institute of Technology October, 2012 1 Patient Centered Outcomes
SEMINAR ON HEALTH CARE ESD.69 HST.926 SYSTEMS INNOVATION Comparative Effectiveness The Savior"? Stan N. Finkelstein, M.D. Massachusetts Institute of Technology October, 2012 1 Patient Centered Outcomes
Clinical Commissioning Policy: Eculizumab for the treatment of refractory antibody mediated rejection post kidney transplant
 Clinical Commissioning Policy: Eculizumab for the treatment of refractory antibody mediated rejection post kidney transplant January 2014 Reference: NHS ENGLAND A07/P/c NHS England Clinical Commissioning
Clinical Commissioning Policy: Eculizumab for the treatment of refractory antibody mediated rejection post kidney transplant January 2014 Reference: NHS ENGLAND A07/P/c NHS England Clinical Commissioning
Clarifying digital health and software regulation: FDA releases three new guidance documents
 Clarifying digital health and software regulation: FDA releases three new guidance documents December 15, 2017 On December 7, 2017, the Food and Drug Administration (FDA or the Agency) released three guidance
Clarifying digital health and software regulation: FDA releases three new guidance documents December 15, 2017 On December 7, 2017, the Food and Drug Administration (FDA or the Agency) released three guidance
Information Technology for Genetic and Genomic Based Personalized Medicine. Submitted. April 23, 2008
 Information Technology for Genetic and Genomic Based Personalized Medicine Submitted April 23, 2008 By The Harvard Medical School Partners HealthCare Center for Genetics and Genomics in Partnership with
Information Technology for Genetic and Genomic Based Personalized Medicine Submitted April 23, 2008 By The Harvard Medical School Partners HealthCare Center for Genetics and Genomics in Partnership with
Genitope Corporation. Summary of MyVax Personalized Immunotherapy Phase 3 Clinical Trial Results
 Genitope Corporation Summary of MyVax Personalized Immunotherapy Phase 3 Clinical Trial Results Introduction In December 27, Genitope Corporation ( Genitope ) obtained data indicating that its pivotal
Genitope Corporation Summary of MyVax Personalized Immunotherapy Phase 3 Clinical Trial Results Introduction In December 27, Genitope Corporation ( Genitope ) obtained data indicating that its pivotal
Towards a P4 Healthcare System: Predictive, Preventive, Personalized & Participatory
 Towards a P4 Healthcare System: Predictive, Preventive, Personalized & Participatory Bull, 2012 Natalia Jiménez Lozano Business Development Manager BULL e-health Consultancy & Business Solutions Tlf: 913939253
Towards a P4 Healthcare System: Predictive, Preventive, Personalized & Participatory Bull, 2012 Natalia Jiménez Lozano Business Development Manager BULL e-health Consultancy & Business Solutions Tlf: 913939253
Manitoba Centre for Proteomics and Systems Biology 10th Anniversary
 Manitoba Centre for Proteomics and Systems Biology 10th Anniversary Originally published in HSC Focus Volume 23, Number 6 September 23, 2016 Collaborative and Patient-Oriented Research At HSC Winnipeg,
Manitoba Centre for Proteomics and Systems Biology 10th Anniversary Originally published in HSC Focus Volume 23, Number 6 September 23, 2016 Collaborative and Patient-Oriented Research At HSC Winnipeg,
A GUIDE TO THIS REFLECTIONS B RESEARCH STUDY IF YOU RE FIGHTING BREAST CANCER, YOU RE NOT ALONE
 A GUIDE TO THIS REFLECTIONS B327-02 RESEARCH STUDY IF YOU RE FIGHTING BREAST CANCER, YOU RE NOT ALONE Do you have breast cancer that has spread to outside the breast? Has your tumor tested positive for
A GUIDE TO THIS REFLECTIONS B327-02 RESEARCH STUDY IF YOU RE FIGHTING BREAST CANCER, YOU RE NOT ALONE Do you have breast cancer that has spread to outside the breast? Has your tumor tested positive for
Realizing the Future that Regenerative Medicine Will Open
 470 Hitachi Review Vol. 65 (2016), No. 9 Featured Articles Realizing the Future that Regenerative Medicine Will Open Shizu Takeda, Ph.D. OVERVIEW: Regenerative medicine is an innovative approach to medicine
470 Hitachi Review Vol. 65 (2016), No. 9 Featured Articles Realizing the Future that Regenerative Medicine Will Open Shizu Takeda, Ph.D. OVERVIEW: Regenerative medicine is an innovative approach to medicine
Bioresearch Monitoring Inspections in Vitro Diagnostics Devices
 Seite 1 von 7 U.S. Food and Drug Administration Protecting and Promoting Your Health Bioresearch Monitoring Inspections in Vitro Diagnostics Devices TABLE OF CONTENTS Introduction Nature, Scope, & Purpose
Seite 1 von 7 U.S. Food and Drug Administration Protecting and Promoting Your Health Bioresearch Monitoring Inspections in Vitro Diagnostics Devices TABLE OF CONTENTS Introduction Nature, Scope, & Purpose
Weighing the Impact of FDA Regulatory Uncertainty on Clinical Decision Support Development
 Executive Summary Weighing the Impact of FDA Regulatory Uncertainty on Clinical Decision Support Development February 25, 2016 Clinical decision support ( CDS ) software is not getting the regulatory clarity
Executive Summary Weighing the Impact of FDA Regulatory Uncertainty on Clinical Decision Support Development February 25, 2016 Clinical decision support ( CDS ) software is not getting the regulatory clarity
Prescription Medicines: Costs in Context
 Prescription Medicines: Costs in Context 2017 We are in a new era of medicine where breakthrough science is transforming care with innovative treatment approaches... Then Now Medicines made of chemical
Prescription Medicines: Costs in Context 2017 We are in a new era of medicine where breakthrough science is transforming care with innovative treatment approaches... Then Now Medicines made of chemical
Epoetin alfa. Epogen, Procrit (epoetin alfa), Retacrit (epoetin alfa epbx) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.85.06 Subject: Epoetin alfa Page: 1 of 8 Last Review Date: September 20, 2018 Epoetin alfa Description
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.85.06 Subject: Epoetin alfa Page: 1 of 8 Last Review Date: September 20, 2018 Epoetin alfa Description
Germany Tissue Engineered - Skin Substitutes Market Outlook to 2020
 Germany Tissue Engineered - Skin Substitutes Market Outlook to 2020 Reference Code: GDMECC0884DB Publication Date: October 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables...
Germany Tissue Engineered - Skin Substitutes Market Outlook to 2020 Reference Code: GDMECC0884DB Publication Date: October 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables...
Gene Therapy and Ethics: the Patient View. A tool for public dialogue
 Gene Therapy and Ethics: the Patient View A tool for public dialogue Colofon This brochure has been developed by the European Genetic Alliances' Network EGAN. You are allowed to download, print or copy
Gene Therapy and Ethics: the Patient View A tool for public dialogue Colofon This brochure has been developed by the European Genetic Alliances' Network EGAN. You are allowed to download, print or copy
Whitepaper. Tackling Complex Data Challenges in Healthcare Analytics.
 Whitepaper Tackling Complex Data Challenges in Healthcare Analytics Introduction: In recent years, healthcare organizations have grown increasingly aware of the potential beneficiary uses of the data which
Whitepaper Tackling Complex Data Challenges in Healthcare Analytics Introduction: In recent years, healthcare organizations have grown increasingly aware of the potential beneficiary uses of the data which
Experimental Medicine: A Platform for Cooperation between Medicine and Veterinary
 Experimental Medicine: A Platform for Cooperation between Medicine and Veterinary Eiji Kobayashi M.D. Ph.D. Departement of Organ Fabrication, Keio University Shool of Medicine Opening Remarks Experimental
Experimental Medicine: A Platform for Cooperation between Medicine and Veterinary Eiji Kobayashi M.D. Ph.D. Departement of Organ Fabrication, Keio University Shool of Medicine Opening Remarks Experimental
The European Medical Technology Industry. in figures / 2018
 The European Medical Technology Industry in figures / 2018 Table of contents What is Medical Technology? 4 Innovation 10 Employment 14 Companies 18 Expenditure on Medical Technology 20 MedTech Market in
The European Medical Technology Industry in figures / 2018 Table of contents What is Medical Technology? 4 Innovation 10 Employment 14 Companies 18 Expenditure on Medical Technology 20 MedTech Market in
Immunizations, injections and infusions (including triggerpoint injections), skin substitutes, and provider-administered pharmaceuticals.
 ACTION: Final DATE: 10/12/2017 3:11 PM 5160-4-12 Immunizations, injections and infusions (including triggerpoint injections), skin substitutes, and provider-administered pharmaceuticals. (A) General provisions.
ACTION: Final DATE: 10/12/2017 3:11 PM 5160-4-12 Immunizations, injections and infusions (including triggerpoint injections), skin substitutes, and provider-administered pharmaceuticals. (A) General provisions.
The Cost-Benefit Calculus of CT Angiograms
 Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/heart-matters/the-cost-benefit-calculus-of-ct-angiograms/4047/
Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/heart-matters/the-cost-benefit-calculus-of-ct-angiograms/4047/
HEALTH POLICY AND DRUG PRICING: A RESEARCHER S PERSPECTIVE
 HEALTH POLICY AND DRUG PRICING: A RESEARCHER S PERSPECTIVE NATASHA KEKRE, MD, MPH, FRCPC NOV 16, 2016 www.ohri.ca WHY THIS MATTERS TO ME I am a hematologist in Ottawa with 75% dedicated time for research
HEALTH POLICY AND DRUG PRICING: A RESEARCHER S PERSPECTIVE NATASHA KEKRE, MD, MPH, FRCPC NOV 16, 2016 www.ohri.ca WHY THIS MATTERS TO ME I am a hematologist in Ottawa with 75% dedicated time for research
closing the price gap for commodity services
 REPRINT August 2017 Jamie Cleverley healthcare financial management association hfma.org closing the price gap for commodity services The competitive pricing offered by freestanding clinics has many hospitals
REPRINT August 2017 Jamie Cleverley healthcare financial management association hfma.org closing the price gap for commodity services The competitive pricing offered by freestanding clinics has many hospitals
Federal Funding for Brain Research. Congressional Support Accelerates Discovery
 The American Brain Coalition (ABC) is a non-profit organization that brings together people with disabling brain disorders, the families of those that are affected, and the professionals who research and
The American Brain Coalition (ABC) is a non-profit organization that brings together people with disabling brain disorders, the families of those that are affected, and the professionals who research and
Special Issue. Mesoblast Limited
 Overview is an Australian biotechnology company committed to the commercialization of novel treatments for orthopedic conditions by using its unique adult stem cell technology for the regeneration and
Overview is an Australian biotechnology company committed to the commercialization of novel treatments for orthopedic conditions by using its unique adult stem cell technology for the regeneration and
NEWS RELEASE For Immediate Release LIFE SCIENCES DISCOVERY FUND ANNOUNCES GRANTS TO COMMERCIALIZE HEALTH-RELATED PRODUCTS AND SERVICES
 NEWS RELEASE For Immediate Release Media contact: Cathyryne Manner Tel: 206-456-9583 manner@lsdfa.org LIFE SCIENCES DISCOVERY FUND ANNOUNCES GRANTS TO COMMERCIALIZE HEALTH-RELATED PRODUCTS AND SERVICES
NEWS RELEASE For Immediate Release Media contact: Cathyryne Manner Tel: 206-456-9583 manner@lsdfa.org LIFE SCIENCES DISCOVERY FUND ANNOUNCES GRANTS TO COMMERCIALIZE HEALTH-RELATED PRODUCTS AND SERVICES
Pluristem Issues Letter to Shareholders
 Pluristem Issues Letter to Shareholders HAIFA, Israel, January 30, 2014 -- Pluristem Therapeutics Inc. (NASDAQCM: PSTI) (TASE:PLTR), a leading developer of placenta-based cell therapies, today issued a
Pluristem Issues Letter to Shareholders HAIFA, Israel, January 30, 2014 -- Pluristem Therapeutics Inc. (NASDAQCM: PSTI) (TASE:PLTR), a leading developer of placenta-based cell therapies, today issued a
Restrooms Cell phones on silent/vibrate. Refreshments. Patient resource materials
 Restrooms Cell phones on silent/vibrate Refreshments Patient resource materials This presentation should not replace discussions with your healthcare provider, but seeks to provide information and resources
Restrooms Cell phones on silent/vibrate Refreshments Patient resource materials This presentation should not replace discussions with your healthcare provider, but seeks to provide information and resources
Animal Research: The Benefits Outweigh the Costs. Alexis Miller. Loras College
 Miller 1 Animal Research: The Benefits Outweigh the Costs Alexis Miller Loras College Miller 2 Animal research has become a controversial ethical topic in psychology today. There are many different opinions
Miller 1 Animal Research: The Benefits Outweigh the Costs Alexis Miller Loras College Miller 2 Animal research has become a controversial ethical topic in psychology today. There are many different opinions
