Ozone Measurements in South Carolina Using Passive Samplers
|
|
|
- Cassandra Hutchinson
- 6 years ago
- Views:
Transcription
1 Journal of the Air & Waste Management Association ISSN: (Print) (Online) Journal homepage: Ozone Measurements in South Carolina Using Passive Samplers Anna C. Franklin, Lynn G. Salmon, Jack M. Wolfson & Christos S. Christoforou To cite this article: Anna C. Franklin, Lynn G. Salmon, Jack M. Wolfson & Christos S. Christoforou (2004) Ozone Measurements in South Carolina Using Passive Samplers, Journal of the Air & Waste Management Association, 54:10, , DOI: / To link to this article: Published online: 22 Feb Submit your article to this journal Article views: 125 View related articles Citing articles: 5 View citing articles Full Terms & Conditions of access and use can be found at Download by: [ ] Date: 31 March 2017, At: 08:52
2 TECHNICAL PAPER ISSN J. Air & Waste Manage. Assoc. 54: Copyright 2004 Air & Waste Management Association Ozone Measurements in South Carolina Using Passive Samplers Anna C. Franklin Environmental Engineering and Science Department, School of the Environment, Clemson University, Clemson, SC Lynn G. Salmon Environmental Quality Laboratory, California Institute of Technology, Pasadena, CA Jack M. Wolfson Harvard School of Public Health, Environmental Science and Engineering Program, Boston, MA Christos S. Christoforou Environmental Engineering and Science Department, School of the Environment, Clemson University, Clemson, SC ABSTRACT Passive samplers with two different collection substrates were used to obtain an average ozone concentration for 1 month during the summer of 2002 for each South Carolina county. One sampler contained a filter coated with indigo carmine, whose color fades when exposed to ozone. The fading was measured by reflectance spectroscopy. The other sampler contained filters that were coated with nitrite, which is oxidized to nitrate when exposed to ozone. The nitrate was measured by ion chromatography. Calibration curves were developed for the two methods by comparing color fading from indigo carmine and nitrate ion concentration from the nitrite filter with ambient ozone concentration measured by a co-located reference continuous UV ozone analyzer. These curves were used to calculate integrated ozone concentrations for samplers distributed across South Carolina. IMPLICATIONS Two passive sampling methods have been calibrated and used to measure simultaneously ambient ozone concentrations in each county in South Carolina. The nitrite-to-nitrate method yielded better results than the fading of indigo carmine. Such easy-to-analyze methods serve as useful screening techniques to find areas of high ozone and to estimate inexpensively background ozone concentrations for setting boundary conditions. Using the indigo carmine method, the average ozone concentrations ranged from 21 to 64 ppb (average ppb, n 58) across the 46 counties in the state during one summer month of Concentrations for the same time period from the nitrite-coated filters ranged from 23 to 62 ppb (average ppb, n 58). Also for the same time period, the 23 continuous UV photometric ozone monitors operated by the South Carolina Department of Health and Environmental Control at sites within 10 miles of some of the passive monitors showed ozone concentrations ranging from 28 to 50 ppb (average ppb, n 22). INTRODUCTION Ground-level ozone (O 3 ), a secondary pollutant, is formed through photochemical reactions between precursor emissions of volatile organic compounds (VOCs) and nitrogen oxides (NO x ). With elevated temperatures, sunlight, and levels of NO x and VOCs present, ozone will form more rapidly than it decomposes, causing it to build up in the atmosphere. Although NO x is mainly emitted from transportation and industrial sources, VOCs can also be emitted from chemical manufacturing, paint shops, and other solventusing sources. Natural sources such as pine trees can produce VOCs as well. Elevated levels of ground-level ozone are capable of causing adverse health effects. Ozone can affect the ability of the immune system to defend against lung infections. It also causes acute respiratory problems and inflammation of 1312 Journal of the Air & Waste Management Association Volume 54 October 2004
3 lung tissue, which can lead to the aggravation of asthma in susceptible people. Symptoms related to ozone exposure include cough, shortness of breath, pain at deep inspiration, malaise, headache, and nausea. Although small children and the elderly are at greatest risk, anyone could be potentially affected by ground-level ozone. 1 As well as being a threat to human health, groundlevel ozone also causes harm to the environment. It can interfere with the ability of plants to produce and store food, compromising growth, reproduction, and overall plant health. Ozone also causes damage to natural rubber and synthetic polymers. The strength of textiles, carpets, curtains, and shop canopies is reduced by exposure to ozone. 2 Annually in South Carolina 204,000 adults and 77,000 children suffer from asthma. In 2000, 1.2% of all visits to the emergency room were because of asthma and more than 27% of all those hospitalizations were among children. In northern South Carolina (Greenville, Spartanburg, Anderson, Pickens, Cherokee, Oconee, and Abbeville counties) 52,835 (6.6%) adults suffer annually from asthma. In 2000, there were 1,252 hospitalizations because of asthma in northern South Carolina for all ages. Two thousand seven people under the age of 18 visited the emergency room because of asthma, and 518 people under the age of 18 were hospitalized because of asthma. 3 In 1971, the U.S. Environmental Protection Agency (EPA) set the National Ambient Air Quality Standard for several air pollutants, including photochemical oxidants. In 1979, the National Ambient Air Quality Standard for ozone was revised and set at 0.12 ppm, measured as a 1-hr average concentration. However, the EPA decided that this ozone standard did not adequately protect human health, so in 1997 the 8-hr standard was established. The new standard, set at 0.08 ppm, is based on a 3-yr average of the annual fourth highest daily maximum 8-hr ozone concentration. 4 South Carolina is currently designated as being in attainment for ozone in all counties. However, under the new 8-hr standard, areas of the state are threatened with possible non-attainment status. Consequences from a non-attainment designation include extensive permitting requirements for industry and possible limitations on the way federal funds may be spent on road projects in non-attainment areas. The objective of this study was to obtain calibration curves for the two different passive ozone methods indigo carmine and nitrite ion by comparison with a reference ambient continuous UV photometric monitor, and then to use these two calibration curves to obtain an average ozone concentration for each passive method for 1 month during the summer of 2002 for each South Carolina county. Passive Ozone Samplers Ozone monitoring traditionally has required large and expensive laboratory instruments. A simpler, inexpensive device was needed to measure indoor and personal exposure. The solution to this dilemma was a passive sampling device, which could easily and reliably monitor ozone in any location without a power supply. These samplers are based on the principle of passive diffusion of a pollutant to an adsorbing collection medium. Various types of passive samplers have been developed with different types of samplers as well as different collection mediums. Although passive sampling can be an efficient method of measuring ozone, certain aspects may affect the ability of the sampler to produce the desired results. Interference from oxidants such as peroxyacetyl nitrates (PAN), as well as oxides of nitrogen and sulfur, may cause an overestimation of the cumulative ozone concentration. Wind velocity and the associated air turbulence may also contribute to inaccurate ozone measurements. Finally, solar radiation, air temperature, and relative humidity can also have an adverse impact on passive sampling. These interfering factors are usually specific to the sampler design and collection medium used. Grosjean and Hisham 5 developed a passive sampler consisting of a plastic filter holder, a Teflon membrane filter mounted upstream as the diffusion barrier, and a coated cellulose paper filter downstream. A diffusion gap was created between the two filters. Variations in the diffusion rate of the passive sampler were tested by changing the pore size of the Teflon filter, by changing the Teflon filter to a different plastic grid support, and by using a rectangular badge. An increase in the diffusion rate allows short-term samples to be collected for measuring peak ozone levels, whereas a decrease in the diffusion rate can be used for collecting longer sample runs. The following three different ozone-trapping reagents were examined: indigo carmine, curcumin, and phenoxazine. Indigo carmine and curcumin fade when exposed to ozone, whereas phenoxazine yields a colored product. The color changes or fading, measured by reflectance spectroscopy, are proportional to the product of ozone concentration and the sampling duration. Indigo carmine proved to be the most effective for ozone. As an alternative to colorant methods, Koutrakis et al. 6 developed a method in which a filter is coated with nitrite ion. In the presence of ozone, nitrite is oxidized to nitrate on the filter. To determine the amount of nitrate present, the filters are extracted with ultra-pure water and analyzed by ion chromatography. The laboratory and field tests present good agreement between the passive method and a reference continuous UV photometric ozone analyzer (coefficient of variance 8.7%, n 44). However, face velocity and wind direction with respect to Volume 54 October 2004 Journal of the Air & Waste Management Association 1313
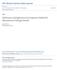
4 the samplers placement did affect the collection rate of the samplers. An unpublished report 7 showed negligible interferences for the nitrite method for typical concentrations of PAN, H 2 O 2,NO 2, HONO, and SO 2. Other researchers also developed samplers for passive collection of ozone Based on a review of all of these passive ozone techniques, it was concluded that the methods of Grosjean and Hisham 5 and Koutrakis et al. 6 would provide the most accurate results and still offer ease of sampling and analysis. MATERIALS AND METHODS Filter Preparation Indigo Carmine. To prepare these filters, 400 ml of methanol was measured with a 100-mL graduated cylinder. Indigo carmine, 3.25 g (certified, ph , dye content 94%, Lot No ks, Catalog No. 13,116-4, Aldrich, St. Louis, MO), was measured on a Sartorius (Edgewood, NY) balance and then dissolved in a dark colored glass bottle with the methanol (400 ml). The bottle was shaken 100 times, and then the mixed solution was placed in a plastic box large enough to hold the filters under the hood. Whatman 41 cellulose filter paper (24 cm, Catalog No , Whatman 41, Whatman, Hillsboro, OR) was placed with the mixed solution for 3 min or until covered with the dye. The filters were hung in the hood and allowed to dry for 24 hr. Filters were then cut into 3-cm diameter circles and measured by reflectance spectroscopy before outdoor exposure. Color changes are measured by reflectance spectroscopy with a MacBeth ColorEye 3000 Spectrophotometer (division of Kollmorgen Instruments Corp., Northampton, MA). The spectral response of this instrument closely approximates the Commission Internationale de l Eclairage colorimetric Standard Observer curves. The color change measured, E, is given by: E ( L 2 a 2 b 2 ) 1/2 (1) where L, a, and b are the standard the Commission Internationale de l Eclairage coordinates for hue and saturation a and b (which taken together are called chromaticity), and brightness (L). These coordinates distinguish one color from another. Chromaticity is the quality of a color that is the combination of hue and brightness. Hue is the color reflected from, or transmitted through, an object. In common use, hue refers to the name of the color, such as red, orange, or green. Saturation is the strength or purity of a color, or the amount of white light mixed with a hue. An item that is highly saturated has a very intense color. An item that is less intense would be less saturated or have less chromaticity. The brightness of a colored surface depends upon the illuminance and upon its reflectivity. 13 The instrument is calibrated by using a white reflector plate standard, and the light source for all measurements is illuminant D 65 (daylight, with a color temperature of 6,500 K). After the filters are painted, dried, and cut into 30- mm-diameter circles, each filter is assigned a labeled number. Each filter is analyzed after being painted with indigo carmine and before ozone exposure ( E 0 ), then again after exposure ( E). The difference between the measurements, E, was determined by: E E 0 E (2) Nitrite Ion The nitrite method used 15-mm-diameter glass fiber filters coated with nitrite ion (obtained from Harvard University School of Public Health, Cambridge, MA), which in the presence of ozone (O 3 ) is oxidized to nitrate ion. The solution to coat the collecting filters included sodium and potassium salts of nitrite and carbonate in a solution of glycerol, methanol, and water, as described by Koutrakis et al. 6 After exposure the filters were extracted by using the following procedure. Ultra-pure water, in aliquots of 2 ml, was placed in a clean cup along with the two filters. The cups were then placed on a shaker table for 3 hr at 150 rpm. After shaking, 1-mL aliquots of each sample were placed in labeled ion chromatography (IC) glass vials. The samples were then placed in the refrigerator until analyzed. A Dionex ion chromatography system (DX-100 Ion Chromatograph, Dionex Corp., Sunnyvale, CA) with an IonPac column (IonPac AG4A 4 mm [10 32] P/N 37042) was used for analysis of the anion, NO 3. Carbonate/bicarbonate eluent was used for the IC (233 mg/l Na 2 CO 3 and 235 mg/l NaHCO 3 ). Two replicates of each of the five standards chosen were prepared from UltraGrade Nitrate Ion Chromatography Solution (UltraScientific, North Kingstown, RI, 1,000 g/l nitrate in water, Lot No. IG-0382) and run on the IC. The results from the standards were used to create a calibration curve, which was necessary to convert the sample peak area to a concentration of NO 3 in the samples. A new calibration curve was constructed each time the IC was run. Passive Sampler Preparation Passive ozone samplers were used for the collection of data. The samplers produced by 3M for assessing occupational exposure to volatile organic compounds were modified to use filter substrates, as shown in Figure 1. Parts of the sampler include the following: front cap, diffusion membrane or barrier, spacer, collection filter, and badge body Journal of the Air & Waste Management Association Volume 54 October 2004
5 Figure 1. Diagram of a passive badge sampler with the following parts, right to left: (1) front cap; (2) diffusion membrane or barrier; (3) spacer; (4) collection filter (one indigo carmine or two nitrite filters); and (5) badge body. The polymer badge body is 3.3 cm in diameter and 1 cm deep. The spacer sets the diffusion distance at 10 mm and holds the collection filter in place. The diffusion barrier, comprised of two filters, attaches to the badge body with the front cap by friction fit. Two filters were used for the diffusion barrier. A quartz filter (Gelman Sciences Pallflex membrane filter Tissuequartz 2500QAO- UP) and a track-etched polycarbonate membrane filter (Whatman Nuclepore, Ann Arbor, MI, 47 mm pore size 3 m). Two nitrite-coated filters were placed in the sampler to provide maximum capacity for O 3. The two filters were extracted together. A single filter painted with indigo carmine was used per sampler. Experimental Design Sampling Sites and Events. One of the main objectives of this project was to determine the average ozone concentration for 1 month in each of 46 counties in South Carolina. To accomplish this, high school students were asked to help obtain the ozone data. Lead high school science teachers in every county were sent a letter requesting addresses for two motivated students to whom a passive sampler would be sent. South Carolina Department of Health and Environmental Control (SCDHEC) district health directors were also contacted about placing the samplers at their county health departments. The participants were asked to hang the sampler outside for 30 days from June 24, 2002 to July 24, At the end of the time period, the samplers were returned for analysis. The following instructions were sent to the participants: The sampler was to be placed outside where it would remain dry. The chosen location was not to be exposed to direct sunlight, rain, or other weather conditions that could damage the sampler. The instructions suggested that an ideal location would be to hang the sampler under the eaves of a roof on the outside of a building (on a balcony or porch, for example). It was important that air was able to reach the sampler on all sides. The sampler was not to be attached directly to a wall or placed near the ground. The sampler should have hung at least 0.5 m ( 1.65 ft) from the nearest wall and 1.5 m ( 5 ft) or more above ground level. The desired method of hanging the filter was horizontally, with the filter facing downwards. The sampler was sent to participants with the strings attached. RESULTS AND DISCUSSION Nitrite Method Calibration Curve Calibration data were collected over 4 weeks in May and June The passive samplers were exposed for times ranging from 1 week to 1 month. At the same time, ambient ozone concentrations as measured by an automatic ozone analyzer were recorded and averaged over the corresponding exposure interval of each passive sampler. Figure 2 shows the calibration data for the passive nitrite method versus continuous monitor, with 30 data points. The graph shows the nitrate as measured by ion chromatography against the average continuous ozone concentration in parts per billion multiplied by the exposure time in hours. These results indicate that the best fit is a linear one between the nitrate concentration and the ppb ozone*hr, with a correlation coefficient value (R 2 )of The least squares fit was used to derive eq 3: ozone ppb nitrate ppm hr exposed (3) Indigo Carmine Calibration Curve Figure 3 shows the calibration data from the indigo carmine experiments, which were carried out by using the Figure 2. Calibration curve of the nitrite method. Volume 54 October 2004 Journal of the Air & Waste Management Association 1315
6 Figure 3. Calibration curve of the indigo carmine method. same method as the nitrite samples. There are 55 data points shown on the graph, collected over the same period of time and at the same location as the nitrite samples. The graph shows the fading as measured by reflectance spectroscopy (in units of E) versus the average continuous ozone concentration in parts per billion multiplied by the filter exposure time in hours. The linear correlation for indigo carmine method is not as strong as for the nitrite method, with the R 2 value of This is consistent with a study from Zhou and Smith 10 that reported that the correlation of the indigo carmine was weaker than that of the other trapping reagents tested in their study, among which was the nitrite method. Although Grosjean and Hisham 5 reported that color change increased linearly with ozone dose for indigo carmine, their experiments were conducted in an artificial test atmosphere without exposure to possible interferences from ambient air. However, in their research, when indigo carmine was exposed to ambient levels of PAN and NO 2 equal to those of ozone, a positive interference of 16% and 15%, respectively, was found. 5 Table 1. Average ozone concentrations for counties in South Carolina. Dates Exposed City County Location Ozone (ppb) from Nitrite Ozone (ppb) from Indigo Carmine Dates Exposed City County Location Ozone (ppb) from Nitrite Ozone (ppb) from Indigo Carmine 6/24 8/09 Calhoun Falls Abbeville /24 7/24 Aiken Aiken /25 7/26 Aiken Aiken /12 8/09 Allendale Allendale /24 7/24 Powdersville Anderson /14 8/14 Bamberg Bamberg /24 7/24 Denmark Bamberg /11 8/14 Barnwell Barnwell /24 7/24 Beaufort Beaufort /24 7/24 Goose Creek Berkeley 30 NA 7/19 8/19 St. Matthews Calhoun /24 7/24 Charleston Charleston /24 7/24 Gaffney Cherokee /24 7/24 Chester Chester /24 7/24 Chesterfield Chesterfield /24 7/24 Manning Clarendon /24 7/24 Walterboro Colleton /24 7/24 Hartsville Darlington /24 7/24 Dillon Dillon /25 7/29 Summerville Dorchester /24 7/24 Edgefield Edgefield /24 7/24 Johnston Edgefield /24 7/24 Winnsboro Fairfield /25 7/25 Florence Florence /2 8/10 Traveler s Rest Greenville /24 7/24 Greenville Greenville /4 8/11 Taylors Greenville /2 8/2 Traveler s Rest Greenville /25 7/24 Greenwood Greenwood NA 45 6/24 7/24 Ninety Six Greenwood /24 7/24 Greenwood Greenwood /25 7/26 Greenwood Greenwood /24 7/24 Varnville Hampton /24 7/24 Little River Horry /24 7/24 Ridgeland Jasper /24 7/24 Camden Kershaw 23 NA 6/24 7/24 Lancaster Lancaster /25 7/25 Laurens Laurens /24 7/24 Bishopville Lee /24 7/24 Lexington Lexington /24 7/24 Mullins Marion /24 7/24 Bennettsville Marlboro /28 7/24 McCormick McCormick /24 7/25 Newberry Newberry /24 7/26 Newberry Newberry /25 7/25 Walhalla Oconee /27 7/29 Bowman Orangeburg /24 7/25 Orangeburg Orangeburg NA 39 6/23 7/26 Easley Pickens /24 7/24 Columbia Richland /21 7/22 Columbia Richland /25 7/25 Blythewood Richland /25 7/25 Columbia Richland /28 7/24 Saluda Saluda /25 7/25 Roebuck Spartanburg /24 7/24 Sumter Sumter /2 8/2 Sumter Sumter /24 7/24 Union Union /26 7/24 Kingstree Williamsburg /24 7/24 Clover York Journal of the Air & Waste Management Association Volume 54 October 2004
7 The least squares fit was used to derive eq 4: ozone ppb E hr exposed (4) South Carolina Ozone Concentrations Equations 3 and 4 were used to obtain the average ozone concentrations for the samplers sent out to participants in the different counties. Of the 69 packages sent out, 60 packages were returned and then analyzed. Because the samplers were sent out to volunteers who assisted in data collection, it was difficult to control sampler exposure time. Some samplers were exposed from June 24 to July 24, 2002, as instructed. However, 28 of the 60 samplers returned had exposure times a little different than the specified time period. Table 1 shows the resulting average ozone concentrations from the nitrite and fading technique for each county in South Carolina. Data were collected in every South Carolina county except Georgetown. Some counties had several sampling sites, whereas some counties only had one sampling site. Nitrite method data was not available for Berkeley and Kershaw Counties, and Orangeburg and Greenwood Counties did not have indigo carmine method data. Generally, during our experiments the average ozone concentrations were relatively low, and below the level of concern (85 ppb). Using the nitrite method, Charleston County had the highest average ozone concentration at 62 ppb, whereas Kershaw County had the lowest value at 23 ppb. The average value for the whole state was 41 ppb ( 8.1, n 58). For the indigo carmine method, McCormick County had the highest concentration at 64 ppb, whereas Sumter County had the lowest concentration at 21 ppb. The average value for the whole state was 46 ppb ( 7.9, n 58). Figures 4 and 5 show the distribution in South Carolina of ozone concentrations using the nitrite and indigo carmine methods, respectively. Figure 4 shows a cluster of concentrations within the ppb range along the Midlands (Richland, Sumter, Lee, Clarendon, Orangeburg, Williamsburg, etc.) and again in the North (Oconee, Pickens, Greenville, Spartanburg). The only concentration within the ppb range is located in Charleston County. Generally, the coastal counties have good atmospheric mixing because of the sea breeze, which keeps the ozone concentrations Figure 4. Average ozone concentrations from samplers containing nitrite-impregnated filters. Volume 54 October 2004 Journal of the Air & Waste Management Association 1317
8 Figure 5. Average ozone concentrations from samplers containing indigo carmine-painted filters. lower relative to other areas. The location of the sampler in a heavily urban area in Charleston could have contributed to this high ozone value. Samplers containing the indigo filters, in general, had higher concentrations than those containing the nitritecoated filters (this may be because of small positive interferences to the indigo method from other pollutant gases, as mentioned above). The values were scattered across the state with no apparent trends. For ranges of , , , , and ppb, there were 3, 7, 27, 19, and 2 samples, respectively. For the nitritecoated filters, for ranges of , , , , and ppb, there were 5, 22, 24, 6, and 1 sample, respectively. Ozone concentrations from 22 SCDHEC automated ozone analyzers located in counties across South Carolina were averaged for the time period of June 24 to July 24, 2002, and are shown in Figure 6. Generally higher concentrations were found Upstate, but the highest value was observed at the Sandhill monitor in Richland County. The lowest values were found along the coastal region, as expected. The ozone concentrations ranged from 28 ppb in Williamsburg County to 51 ppb in Richland County. The average value was 39 ppb ( 6.3, n 22). The data from the two methods evaluated in this project were compared with the data from ozone monitors operated by SCDHEC located in the same county that were at a distance of less than 10 miles from the passive sampler location. Even though 21 passive samplers were in the same county as a monitor operated by SCDHEC, in 11 instances the distance between the two was too large to be suitable for comparison. The ozone concentrations from the SCDHEC monitors were averaged for the same time periods that the passive samplers were exposed. Figures 7 and 8 show a comparison of the ozone concentration as measured by the nitrite and indigo passive samplers, respectively, and the continuous monitors operated by SCDHEC. The counties in Figures 7 and 8 are arranged in order of increasing distance from the continuous monitors, with co-located samplers at Anderson County and a distance of 10 miles between the passive sampler and the SCDHEC monitor at Oconee. The error bars on the ozone concentration as estimated by our experiments represent the residual of the experimental data as shown in the calibration curves (Figures 2 and 3). These were found to be 6 and 25.5 ppb for the nitrate and indigo carmine methods, respectively. The larger error bar on the ozone concentrations estimated with the indigo 1318 Journal of the Air & Waste Management Association Volume 54 October 2004
9 Figure 6. Ozone concentrations in South Carolina as determined from automatic ozone analyzers operated by SCDHEC. carmine method than the nitrate method is a result of the greater scatter of data on the calibration curve (Figure 3). It is seen that there is good agreement, in general, between the results from passive sampling and SCDHEC ozone monitors. Both methods may be used to assess ambient ozone concentrations. The nitrite method has yielded better results than the indigo carmine method (R versus R , respectively). On the other hand, the implementation of the indigo carmine method is simpler because it does not require knowledge of an IC, depending instead on the use of a spectrophotometer, which is more straightforward to operate. Figure 7. Comparison of ozone data as measured by the nitrite method and the SCDHEC ozone monitor within 10 miles of the sampler. Error bars on the ozone concentration as estimated by the nitrate method represent the residual error calculated from the calibration line shown in Figure 2 (6 ppb). Figure 8. Comparison of ozone data as measured by the indigo carmine method and the SCDHEC ozone monitor within 10 miles of the sampler. Error bars on the ozone concentration as with the indigo carmine method represent the residual error calculated from the calibration line shown in Figure 3 (25.5 ppb). Volume 54 October 2004 Journal of the Air & Waste Management Association 1319
10 CONCLUSIONS Passive samplers using two types of coated filters as collection substrates were evaluated for measuring ambient ozone concentrations. One filter was coated with indigo carmine and the other filter was coated with a nitrite salt. Calibration curves were developed for the two methods by comparing fading of the indigo carmine and nitrate ion concentration from the nitrite filters with ambient ozone concentration as determined by a reference continuous UV photometric ozone analyzer. These curves were used to calculate the average ozone concentrations for samplers distributed across South Carolina. The passive sampler measurements were in good agreement with measurements made by continuous instruments at locations within 10 miles of the sampling sites. Passive sampling is not generally suitable for shortterm measurements, but can be appropriate for determining long-term effects, such as human exposure evaluations and assessment of ozone dose to determine the long-term effects on the environment. The new EPA 8-hr standard for ozone of 0.08 ppm, together with its averaging method (the 3-yr average of the fourth highest annual 8-hr ozone concentration) may be seen as placing more importance on long-term average ozone concentrations, as opposed to the previous 1-hr maximum ozone concentration of 0.12 ppm. Passive sampling is useful because measurements at many sampling locations can be carried out simultaneously at low cost. Results from such measurements can be used to estimate the spatial distribution of ozone, and identify those areas with potentially high ozone concentrations where more intensive sampling with continuous analyzers would be warranted. REFERENCES 1. Lippman, M. Health Effects of Ozone, A Critical Review; J. Air Pollut. Control Assoc. 1989, 39, Lee, D.S.; Holland, M.R.; Falla, N. The Potential Impact of Ozone on Materials in the UK; Atmosph. Environ. 1996, 30, South Carolina Department of Health and Environmental Control. PowerPoint presentation from Upstate Air Quality Summit II, November 21, 2002, (accessed January 2003). 4. United States Environmental Protection Agency. Fact Sheet on EPA s Revised Ozone Standard, o3fact.html (accessed November 2002). 5. Grosjean, D.; Hisham, M.W.M. A Passive Sampler for Atmospheric Ozone; J. Air Waste Mgmt. Assoc. 1992, 42, Koutrakis, P.; Wolfson, J.M.; Bunyaviroch, A.; Froehlich, S.E.; Hirano, K.; Milik, J.D. Measurement of Ambient Ozone Using a Nitrite-Coated Filter; Analytic. Chem. 1993, 65, Lurmann, F.W.; Roberts, P.T.; Main, H.H.; Hering, S.V.; Avol, E.L.; Colome, S.D. Exposure Assessment Methodology; Phase II Report, Appendix A; Draft final report from Sonoma Technology, Inc., to the California Air Resources Board, August Monn, C.; Hangartner, M. Passive Sampling for Ozone; J. Air & Waste Mgmt. Assoc. 1990, 40, Scheeren, B.A.; Adema, E.H. Monitoring Ambient Ozone With a Passive Measurement Technique Method, Field Results and Strategy; Water, Air, Soil Pollut. 1996, 91, Zhou, J.; Smith, S. Measurement of Ozone Concentrations in Ambient air Using a Badge-Type Passive Monitor; J. Air & Waste Mgmt. Assoc. 1997, 47, Helaleh, M.; Suharto, N.; Takashi, K.; Tanaka, K. Development of Passive Sampler Technique for Ozone Monitoring. Estimation of Indoor and Outdoor Ozone Concentration. Talanta. 2002, 58, Tang, H.; Lau, T. A New All Season Passive Sampling System for Monitoring Ozone in Air. Environ. Monitor. Assess. 2000, 65, Billmeyer, F.W.; Saltzman, M. Principles of Color Technology, 2nd ed.; John Wiley and Sons: New York, About the Authors Anna C. Franklin is currently an engineer with Rogers and Callcott Engineers in Greenville, SC. Lynn G. Salmon is an engineer with Environmental Quality Laboratory in Pasadena, CA. Jack M. Wolfson is a chemist at the Harvard School of Public Health in Boston, MA. Christos S. Christoforou is an assistant professor at the School of the Environment, Clemson University, Clemson, SC. Address correspondence to: Christos S. Christoforou, Environmental Engineering and Science Department, School of the Environment, Clemson University, P.O. Box , Clemson, SC; csc@clemson.edu Journal of the Air & Waste Management Association Volume 54 October 2004
We have completed the 2016 Southern Pine Beetle (SPB) pheromone trapping. The results and counties of this survey are attached.
 Memo to: Through: From: SCFC Employees Registered Foresters in South Carolina D. Jones F. Chisolm Beckham, David Jenkins and Kevin Douglas Date: May 22, 2016 Subject: Results of 2016 SPB Pheromone Trapping
Memo to: Through: From: SCFC Employees Registered Foresters in South Carolina D. Jones F. Chisolm Beckham, David Jenkins and Kevin Douglas Date: May 22, 2016 Subject: Results of 2016 SPB Pheromone Trapping
Economic Characteristics of South Carolina Manufacturing Sector Exporters
 Economic Characteristics of South Carolina Manufacturing Sector Exporters John D. Mittelstaedt Clemson University George N. Harben South Carolina Department of Commerce & William A. Ward Clemson University
Economic Characteristics of South Carolina Manufacturing Sector Exporters John D. Mittelstaedt Clemson University George N. Harben South Carolina Department of Commerce & William A. Ward Clemson University
DIRECT VACANT (SF) SUBLEASE VACANT (SF)
 ABBEVILLE ABSORPTION Manufacturing 11 1,791,876 141,000-141,000 7.87% - $3.75 Warehouse 7 346,350 60,000-60,000 17.32% - - Abbeville Total 18 2,138,226 201,000-201,000 9.40% - $3.75 AIKEN Flex/R&D 1 14,250
ABBEVILLE ABSORPTION Manufacturing 11 1,791,876 141,000-141,000 7.87% - $3.75 Warehouse 7 346,350 60,000-60,000 17.32% - - Abbeville Total 18 2,138,226 201,000-201,000 9.40% - $3.75 AIKEN Flex/R&D 1 14,250
FALL SEMESTER 2006 PROFILE FOR THE CORPS OF CADETS IN-STATE/OUT-OF-STATE RESIDENCY STATUS
 PROFILE FOR THE CORPS OF CADETS IN-STATE/OUT-OF-STATE RESIDENCY STATUS Note: In-State Residency includes (1) residents, (2) military and their dependents, (3) faculty and administration employees and their
PROFILE FOR THE CORPS OF CADETS IN-STATE/OUT-OF-STATE RESIDENCY STATUS Note: In-State Residency includes (1) residents, (2) military and their dependents, (3) faculty and administration employees and their
Drought Tabletop/ Water Shortage Exercise
 Drought Tabletop/ Water Shortage Exercise September 27 th, 2017 Acknowledgements Planning Committee Hope Mizzell Department of Natural Resources, State Climatology Office Ekaterina Altman, Amanda Farris,
Drought Tabletop/ Water Shortage Exercise September 27 th, 2017 Acknowledgements Planning Committee Hope Mizzell Department of Natural Resources, State Climatology Office Ekaterina Altman, Amanda Farris,
FALL SEMESTER 2005 PROFILE FOR THE CORPS OF CADETS IN-STATE/OUT-OF-STATE RESIDENCY STATUS
 PROFILE FOR THE CORPS OF CADETS IN-STATE/OUT-OF-STATE RESIDENCY STATUS Note: In-State Residency includes (1) residents, (2) military and their dependents, (3) faculty and administration employees and their
PROFILE FOR THE CORPS OF CADETS IN-STATE/OUT-OF-STATE RESIDENCY STATUS Note: In-State Residency includes (1) residents, (2) military and their dependents, (3) faculty and administration employees and their
Contributions of the Ornamental Horticulture and Turfgrass Industry to the South Carolina Economy, 1999
 EER 194 April 01 Contributions of the Ornamental Horticulture and Turfgrass Industry to the South Carolina Economy, 1999 by P. James Rathwell Michalann G. Evatt Mark S. Henry Department of Agricultural
EER 194 April 01 Contributions of the Ornamental Horticulture and Turfgrass Industry to the South Carolina Economy, 1999 by P. James Rathwell Michalann G. Evatt Mark S. Henry Department of Agricultural
Infrastructure Recovery Daily Report Saturday, October 17, 2015
 Resources Currently Deployed on Recovery Operations 2222 Highway Workers are actively working. 249 crews are deployed statewide focused on Recovery Efforts. Of the 249 crews, 44 are working in Richland
Resources Currently Deployed on Recovery Operations 2222 Highway Workers are actively working. 249 crews are deployed statewide focused on Recovery Efforts. Of the 249 crews, 44 are working in Richland
SCDOT Consolidated Program Delivery Plan
 SCDOT Consolidated Program Delivery Plan 8 Years 11/1/2016 A Complex Financial Picture State Infrastructure Bank State of SC General Fund $50M Act 98 Reimbursements Annual Appropriation $84M Department
SCDOT Consolidated Program Delivery Plan 8 Years 11/1/2016 A Complex Financial Picture State Infrastructure Bank State of SC General Fund $50M Act 98 Reimbursements Annual Appropriation $84M Department
South Carolina. Annual Fertilizer Report Clemson University~ Plant Industry~ 511 Westinghouse Road~ Pendleton~ SC ~29670
 South Carolina Annual Fertilizer Report 2015-2016 Clemson University~ Plant Industry~ 511 Westinghouse Road~ Pendleton~ SC ~29670 Clemson University Administration Program Administration and Staff James
South Carolina Annual Fertilizer Report 2015-2016 Clemson University~ Plant Industry~ 511 Westinghouse Road~ Pendleton~ SC ~29670 Clemson University Administration Program Administration and Staff James
Technical Memorandum EXISTING CONDITIONS. Prepared for: Prepared by:
 Technical Memorandum EXISTING CONDITIONS Prepared for: Prepared by: April 2013 TABLE OF CONTENTS 1. Introduction... 1 1.1 Report Purpose... 1 1.2 Report Structure... 1 1.3 Strategic Partnerships... 1 2.
Technical Memorandum EXISTING CONDITIONS Prepared for: Prepared by: April 2013 TABLE OF CONTENTS 1. Introduction... 1 1.1 Report Purpose... 1 1.2 Report Structure... 1 1.3 Strategic Partnerships... 1 2.
Validation of Sulfur Dioxide Using. SKC UME x 200 Passive Sampler Cat. No Page 1 of 14 Publication 1781 Rev 1607
 Validation of Sulfur Dioxide Using SKC UME x 200 Passive Sampler Cat. No. 500-200 Page 1 of 14 Publication 1781 Rev 1607 Research Report Validation of Sulfur Dioxide Using SKC UME x 200 Passive Sampler
Validation of Sulfur Dioxide Using SKC UME x 200 Passive Sampler Cat. No. 500-200 Page 1 of 14 Publication 1781 Rev 1607 Research Report Validation of Sulfur Dioxide Using SKC UME x 200 Passive Sampler
SOUTHERN PINE BEETLE PHEROMONE TRAPPING REPORT, 2018
 SOUTHERN PINE BEETLE PHEROMONE TRAPPING REPORT, 2018 1 The SCFC insect and disease staff discovered this southern pine beetle spot in Oconee County in September. EXECUTIVE SUMMARY Southern pine beetle
SOUTHERN PINE BEETLE PHEROMONE TRAPPING REPORT, 2018 1 The SCFC insect and disease staff discovered this southern pine beetle spot in Oconee County in September. EXECUTIVE SUMMARY Southern pine beetle
Forest Inventory Data
 Forest Inventory Data Greg Reams FIA National Program Leader Washington, DC Jeff Turner Forester Southern Research Station Knoxville, TN Agenda Brief History of FIA Current FIA Program Phase 1: Stratification
Forest Inventory Data Greg Reams FIA National Program Leader Washington, DC Jeff Turner Forester Southern Research Station Knoxville, TN Agenda Brief History of FIA Current FIA Program Phase 1: Stratification
SOUTH CAROLINA'S Forest Products Industry: Performance and Contribution to the State's Economy, 1970 to 1980
 United States Department of Agriculture Forest Service Pacific Northwest Research Station Research Paper PNW-351 February 1986 EDITOR'S FILE COPY SOUTH CAROLINA'S Forest Products Industry: Performance
United States Department of Agriculture Forest Service Pacific Northwest Research Station Research Paper PNW-351 February 1986 EDITOR'S FILE COPY SOUTH CAROLINA'S Forest Products Industry: Performance
Air Pollution. tutorial by Paul Rich. Brooks/Cole Publishing Company / ITP
 Air Pollution tutorial by Paul Rich Outline 1. The Atmosphere layers, some major processes 2. Urban Air Pollution photochemical & industrial smog 3. Regional Air Pollution from Acid Deposition acid deposition,
Air Pollution tutorial by Paul Rich Outline 1. The Atmosphere layers, some major processes 2. Urban Air Pollution photochemical & industrial smog 3. Regional Air Pollution from Acid Deposition acid deposition,
BULLETIN 603 October 1977 OPTIMUM NUMBER, SIZE, AND LOCATION OF FLUID MILK PROCESSING PLANTS IN SOUTH CAROLINA
 BULLETIN 603 October 1977 OPTIMUM NUMBER, SIZE, AND LOCATION OF FLUID MILK PROCESSING PLANTS IN SOUTH CAROLINA ABSTRACT This study was concerned with determining the optimum number, size, and location
BULLETIN 603 October 1977 OPTIMUM NUMBER, SIZE, AND LOCATION OF FLUID MILK PROCESSING PLANTS IN SOUTH CAROLINA ABSTRACT This study was concerned with determining the optimum number, size, and location
School Calendar Information
 2015-2016 School Calendar Information State County First Day Spring Break Last Day Georgia Kentucky New York Georgia Atlanta 8/5/2015 4/4/2016-4/8/2016 5/25/2016 Augusta 8/7/2015 4/4/2016-4/8/2016 5/27/2016
2015-2016 School Calendar Information State County First Day Spring Break Last Day Georgia Kentucky New York Georgia Atlanta 8/5/2015 4/4/2016-4/8/2016 5/25/2016 Augusta 8/7/2015 4/4/2016-4/8/2016 5/27/2016
Experiment # 2 Aerosol and Trace Gas Sampling
 Experiment # 2 Aerosol and Trace Gas Sampling The purpose of this experiment is to teach you a common method for sampling inorganic atmospheric aerosols and trace gases for chemical analysis. Ambient air
Experiment # 2 Aerosol and Trace Gas Sampling The purpose of this experiment is to teach you a common method for sampling inorganic atmospheric aerosols and trace gases for chemical analysis. Ambient air
SOUTHERN PINE BEETLE PHEROMONE TRAPPING REPORT, 2017
 SOUTHERN PINE BEETLE PHEROMONE TRAPPING REPORT, 2017 1 EXECUTIVE SUMMARY Photo by Erich G. Vallery (USDA Forest Service - SRS-4552, Bugwood.org) SPB is one of the most destructive insects to southern yellow
SOUTHERN PINE BEETLE PHEROMONE TRAPPING REPORT, 2017 1 EXECUTIVE SUMMARY Photo by Erich G. Vallery (USDA Forest Service - SRS-4552, Bugwood.org) SPB is one of the most destructive insects to southern yellow
Air Pollution Chapter 21. Atmosphere as a Resource
 Air Pollution Chapter 21 Atmosphere as a Resource Atmospheric Composition Nitrogen 78.08% Oxygen 20.95% Argon 0.93% Carbon dioxide 0.04% Ecosystem services Blocks UV radiation Moderates the climate Redistributes
Air Pollution Chapter 21 Atmosphere as a Resource Atmospheric Composition Nitrogen 78.08% Oxygen 20.95% Argon 0.93% Carbon dioxide 0.04% Ecosystem services Blocks UV radiation Moderates the climate Redistributes
2016 Nitrogen Dioxide Summary
 2016 Nitrogen Dioxide Summary New Jersey Department of Environmental Protection SOURCES Nitrogen dioxide (NO2) is a reddish-brown highly reactive gas that is formed in the air through the oxidation of
2016 Nitrogen Dioxide Summary New Jersey Department of Environmental Protection SOURCES Nitrogen dioxide (NO2) is a reddish-brown highly reactive gas that is formed in the air through the oxidation of
Helping people with disabilities become and stay employed. Helping businesses find and keep talent. scvrd.net
 Helping people with disabilities become and stay employed. Helping businesses find and keep talent. scvrd.net Through hard work and timely delivery of individualized services, VR prepares and assists eligible
Helping people with disabilities become and stay employed. Helping businesses find and keep talent. scvrd.net Through hard work and timely delivery of individualized services, VR prepares and assists eligible
2017 Ozone Summary SOURCES. New Jersey Department of Environmental Protection
 2017 Ozone Summary New Jersey Department of Environmental Protection SOURCES Ozone (O3) is a gas consisting of three oxygen atoms. It occurs naturally in the upper atmosphere (stratospheric ozone) where
2017 Ozone Summary New Jersey Department of Environmental Protection SOURCES Ozone (O3) is a gas consisting of three oxygen atoms. It occurs naturally in the upper atmosphere (stratospheric ozone) where
Advanced Monitoring Method for Air Environment by Ogawa Passive Sampler
 Advanced Monitoring Method for Air Environment by Ogawa Passive Sampler Keiro Higuchi 1, D. R. Schaeffer 2 *, Koichiro Hirano 3 1 Ogawa & Co., Ltd. 3-1-25-501 Hio-cho, Nada-ku, Kobe 657-0029 Japan 2 Ogawa
Advanced Monitoring Method for Air Environment by Ogawa Passive Sampler Keiro Higuchi 1, D. R. Schaeffer 2 *, Koichiro Hirano 3 1 Ogawa & Co., Ltd. 3-1-25-501 Hio-cho, Nada-ku, Kobe 657-0029 Japan 2 Ogawa
SOUTH CAROLINA FORESTRY COMMISSION. Fire Report Instruction Manual
 SOUTH CAROLINA FORESTRY COMMISSION Fire Report Instruction Manual Rev. March 2013 SOUTH CAROLINA FORESTRY COMMISSION Fire Report Instruction Manual March 19, 2013 GENERAL INSTRUCTIONS Requirements A complete
SOUTH CAROLINA FORESTRY COMMISSION Fire Report Instruction Manual Rev. March 2013 SOUTH CAROLINA FORESTRY COMMISSION Fire Report Instruction Manual March 19, 2013 GENERAL INSTRUCTIONS Requirements A complete
Pollution: Los Angeles: Worst Air Quality in USA
 Pollution: Contamination of natural environment w/ harmful substances as a consequence of human activities High enough concentration to interfere w/ Health (human & wildlife) Ecosystem processes (Nutrient
Pollution: Contamination of natural environment w/ harmful substances as a consequence of human activities High enough concentration to interfere w/ Health (human & wildlife) Ecosystem processes (Nutrient
Regional Transit & Coordination Plan CATAWBA REGION. Prepared for: Prepared by:
 CATAWBA REGION Prepared for: Prepared by: November 2014 TABLE OF CONTENTS 1. Introduction... 1 1.1 Overview... 1 1.2 Community Summary... 2 1.2.1 Population Trends... 3 1.2.2 Economic Summary... 6 1.2.3
CATAWBA REGION Prepared for: Prepared by: November 2014 TABLE OF CONTENTS 1. Introduction... 1 1.1 Overview... 1 1.2 Community Summary... 2 1.2.1 Population Trends... 3 1.2.2 Economic Summary... 6 1.2.3
Regional Transit & Coordination Plan LOWCOUNTRY REGION. Prepared for: Prepared by:
 LOWCOUNTRY REGION Prepared for: Prepared by: November 2014 TABLE OF CONTENTS 1. Introduction... 1 1.1 Overview... 1 1.2 Community Summary... 2 1.2.1 Population Trends... 3 1.2.2 Economic Summary... 5 1.2.3
LOWCOUNTRY REGION Prepared for: Prepared by: November 2014 TABLE OF CONTENTS 1. Introduction... 1 1.1 Overview... 1 1.2 Community Summary... 2 1.2.1 Population Trends... 3 1.2.2 Economic Summary... 5 1.2.3
2005 Ozone Summary. Since ground-level ozone needs sunlight to form, it is mainly a Ozone is bad down here Cars, trucks,
 2005 Ozone Summary New Jersey Department of Environmental Protection NATURE AND SOURCES Ozone (O 3 ) is a gas consisting of three oxygen atoms. It occurs naturally in the upper atmosphere (stratospheric
2005 Ozone Summary New Jersey Department of Environmental Protection NATURE AND SOURCES Ozone (O 3 ) is a gas consisting of three oxygen atoms. It occurs naturally in the upper atmosphere (stratospheric
Final Revisions to the Primary National Ambient Air Quality Standard for Nitrogen Dioxide (NO 2 )
 Final Revisions to the Primary National Ambient Air Quality Standard for Nitrogen Dioxide (NO 2 ) General Overview Office of Air and Radiation Office of Air Quality Planning and Standards January 2010
Final Revisions to the Primary National Ambient Air Quality Standard for Nitrogen Dioxide (NO 2 ) General Overview Office of Air and Radiation Office of Air Quality Planning and Standards January 2010
Oregon. 800 NE Oregon St. #640 Portland, OR (971)
 Air Quality Frequently Asked Questions Oregon Environmental Public Health Tracking (EPHT) Oregon ENVIRONMENTAL PUBLIC HEALTH TRACKING 800 NE Oregon St. #640 Portland, OR 97232 www.oregon.gov/dhs/ph/epht
Air Quality Frequently Asked Questions Oregon Environmental Public Health Tracking (EPHT) Oregon ENVIRONMENTAL PUBLIC HEALTH TRACKING 800 NE Oregon St. #640 Portland, OR 97232 www.oregon.gov/dhs/ph/epht
Air Quality Monitoring: Ozone Attainment Status
 As of April 13, 2015 (#C117-1002) - Four Corners (#C097-2002) 2015 17-Mar 2015 56 16-Mar 2014 4-May 2014 64 1-Apr 2013 61 15-Mar 2013 16-Mar 2012 71 28-Jun 2012 22-May 2014 3- Attainment Average: 2015
As of April 13, 2015 (#C117-1002) - Four Corners (#C097-2002) 2015 17-Mar 2015 56 16-Mar 2014 4-May 2014 64 1-Apr 2013 61 15-Mar 2013 16-Mar 2012 71 28-Jun 2012 22-May 2014 3- Attainment Average: 2015
South Carolina Water Use Report 2009 Annual Summary
 Bureau of Water South Carolina Department of Health and Environmental Control South Carolina Water Use Report 2009 Annual Summary www.scdhec.net/water South Carolina Water Use Report 2009 Summary South
Bureau of Water South Carolina Department of Health and Environmental Control South Carolina Water Use Report 2009 Annual Summary www.scdhec.net/water South Carolina Water Use Report 2009 Summary South
Ozone 101. Maricopa County Air Quality Department. September 4, Tom Moore WRAP Air Quality Program Manager WESTAR Council
 Ozone 101 Maricopa County Air Quality Department September 4, 2014 Tom Moore WRAP Air Quality Program Manager WESTAR Council 1 Ozone is an air pollutant of concern, regulated under the Clean Air Act High
Ozone 101 Maricopa County Air Quality Department September 4, 2014 Tom Moore WRAP Air Quality Program Manager WESTAR Council 1 Ozone is an air pollutant of concern, regulated under the Clean Air Act High
State Implementation Plans for Federal 8-Hour Ozone and PM2.5 Standards San Joaquin Valley Eastern Kern County
 State Implementation Plans for Federal 8-Hour Ozone and PM2.5 Standards San Joaquin Valley Eastern Kern County Public Meeting January 4, 2005 Fresno, Bakersfield, Modesto 1 San Joaquin Valley Air Pollution
State Implementation Plans for Federal 8-Hour Ozone and PM2.5 Standards San Joaquin Valley Eastern Kern County Public Meeting January 4, 2005 Fresno, Bakersfield, Modesto 1 San Joaquin Valley Air Pollution
Frumkin, 2e Part Three: Environmental Health on the Regional Scale. Chapter 12: Air Pollution
 Frumkin, 2e Part Three: Environmental Health on the Regional Scale Chapter 12: Air Pollution History of Air Pollution Since human beings discovered fire, they began to pollute the air. At first, air pollution
Frumkin, 2e Part Three: Environmental Health on the Regional Scale Chapter 12: Air Pollution History of Air Pollution Since human beings discovered fire, they began to pollute the air. At first, air pollution
Regional Transit & Coordination Plan WACCAMAW REGION. Prepared for: Prepared by:
 WACCAMAW REGION Prepared for: Prepared by: November 2014 TABLE OF CONTENTS 1. Introduction... 1 1.1 Overview... 1 1.2 Community Summary... 2 1.2.1 Population Trends... 3 1.2.2 Economic Summary... 5 1.2.3
WACCAMAW REGION Prepared for: Prepared by: November 2014 TABLE OF CONTENTS 1. Introduction... 1 1.1 Overview... 1 1.2 Community Summary... 2 1.2.1 Population Trends... 3 1.2.2 Economic Summary... 5 1.2.3
SPATIAL AND SEASONAL DISTRIBUTION OF VOLATILE ORGANIC COMPOUNDS (VOCs) IN THERMAL POWER PLANT REGION IN KUTAHYA, TURKEY
 Proceedings of the 14 th International Conference on Environmental Science and Technology Rhodes, Greece, 3-5 September 2015 SPATIAL AND SEASONAL DISTRIBUTION OF VOLATILE ORGANIC COMPOUNDS (VOCs) IN THERMAL
Proceedings of the 14 th International Conference on Environmental Science and Technology Rhodes, Greece, 3-5 September 2015 SPATIAL AND SEASONAL DISTRIBUTION OF VOLATILE ORGANIC COMPOUNDS (VOCs) IN THERMAL
Health Effects Associated with Stack Chemical Emissions from NYS Natural Gas Compressor Stations:
 Health Effects Associated with Stack Chemical Emissions from NYS Natural Gas Compressor Stations: 2008-2014 A Technical Report Prepared for the Southwest Pennsylvania Environmental Health Project D.O.
Health Effects Associated with Stack Chemical Emissions from NYS Natural Gas Compressor Stations: 2008-2014 A Technical Report Prepared for the Southwest Pennsylvania Environmental Health Project D.O.
SC Fruit, Vegetable, and Specialty Crop Association
 SC FreshXpress a publication of the SC Fruit, Vegetable, & Specialty Crop Association July 2008 SC Fruit, Vegetable, and Specialty Crop Association Purpose The SC Fruit, Vegetable, and Specialty Crop Association
SC FreshXpress a publication of the SC Fruit, Vegetable, & Specialty Crop Association July 2008 SC Fruit, Vegetable, and Specialty Crop Association Purpose The SC Fruit, Vegetable, and Specialty Crop Association
SANTEE-LYNCHES REGION
 SANTEE-LYNCHES REGION Prepared for: Prepared by: November 2014 TABLE OF CONTENTS 1. Introduction... 1 1.1 Overview... 1 1.2 Community Summary... 2 1.2.1 Population Trends... 2 1.2.2 Economic Summary...
SANTEE-LYNCHES REGION Prepared for: Prepared by: November 2014 TABLE OF CONTENTS 1. Introduction... 1 1.1 Overview... 1 1.2 Community Summary... 2 1.2.1 Population Trends... 2 1.2.2 Economic Summary...
2011 Ozone Summary NATURE AND SOURCES ENVIRONMENTAL EFFECTS. New Jersey Department of Environmental Protection
 2011 Ozone Summary New Jersey Department of Environmental Protection NATURE AND SOURCES Figure 1 Good and Bad Ozone Ozone (O3) is a gas consisting of three oxygen atoms. It occurs naturally in the upper
2011 Ozone Summary New Jersey Department of Environmental Protection NATURE AND SOURCES Figure 1 Good and Bad Ozone Ozone (O3) is a gas consisting of three oxygen atoms. It occurs naturally in the upper
Performance and Application of an Inexpensive Method for Measurement of Nitrogen Dioxide
 PSU McNair Scholars Online Journal Volume 1 Issue 1 Transformative Possibilities: Transcending Interlocking Boundaries Article 10 2004 Performance and Application of an Inexpensive Method for Measurement
PSU McNair Scholars Online Journal Volume 1 Issue 1 Transformative Possibilities: Transcending Interlocking Boundaries Article 10 2004 Performance and Application of an Inexpensive Method for Measurement
2010 Ozone Summary NATURE AND SOURCES ENVIRONMENTAL EFFECTS. Figure 2. New Jersey Department of Environmental Protection.
 2010 Ozone Summary New Jersey Department of Environmental Protection NATURE AND SOURCES Ozone (O 3 ) is a gas consisting of three oxygen atoms. It occurs naturally in the upper atmosphere (stratospheric
2010 Ozone Summary New Jersey Department of Environmental Protection NATURE AND SOURCES Ozone (O 3 ) is a gas consisting of three oxygen atoms. It occurs naturally in the upper atmosphere (stratospheric
CHAPTER 2 - Air Quality Trends and Comparisons
 CHAPTER 2 - Air Quality Trends and Comparisons Particulate Sampling Total Suspended Particulate Matter With the monitoring for PM 2.5 particulate matter being labor intensive, DEP reduced the number of
CHAPTER 2 - Air Quality Trends and Comparisons Particulate Sampling Total Suspended Particulate Matter With the monitoring for PM 2.5 particulate matter being labor intensive, DEP reduced the number of
Overview of Chapter 19
 19 Air Pollution Overview of Chapter 19 Atmosphere as a Resource Types and Sources of Air Pollution Effects of Air Pollution Controlling Air Pollution in the US Ozone Depletion in the Stratosphere Acid
19 Air Pollution Overview of Chapter 19 Atmosphere as a Resource Types and Sources of Air Pollution Effects of Air Pollution Controlling Air Pollution in the US Ozone Depletion in the Stratosphere Acid
South Carolina Drought and Water Shortage Tabletop Exercise
 South Carolina Drought and Water Shortage Tabletop Exercise Summary Report September 27, 2017 South Carolina Emergency Operations Center West Columbia, SC Prepared by Ekaterina Altman and Kirsten Lackstrom
South Carolina Drought and Water Shortage Tabletop Exercise Summary Report September 27, 2017 South Carolina Emergency Operations Center West Columbia, SC Prepared by Ekaterina Altman and Kirsten Lackstrom
APPENDIX 10 (SOUTH CAROLINA DROUGHT RESPONSE PLAN) TO THE SOUTH CAROLINA EMERGENCY OPERATIONS PLAN
 I. INTRODUCTION A. A drought is a slowly developing disaster that may occur over several months or years. Impacts from drought may occur quickly for some sectors while for others it may take years to have
I. INTRODUCTION A. A drought is a slowly developing disaster that may occur over several months or years. Impacts from drought may occur quickly for some sectors while for others it may take years to have
Knocking Over Pollutants
 Lesson 5- Air Pollution Knocking Over Pollutants Background Information The U.S. Environmental Protection Agency (EPA) has established national ambient air quality standards for six air pollutants- ozone,
Lesson 5- Air Pollution Knocking Over Pollutants Background Information The U.S. Environmental Protection Agency (EPA) has established national ambient air quality standards for six air pollutants- ozone,
Lab 6 Measurement of Ozone
 Georgia Institute of Technology School of Earth and Atmospheric Sciences EAS 4641 Spring 2008 Lab 6 Measurement of Ozone Purpose of Lab 6: In this lab you will measure the ambient concentration of ozone
Georgia Institute of Technology School of Earth and Atmospheric Sciences EAS 4641 Spring 2008 Lab 6 Measurement of Ozone Purpose of Lab 6: In this lab you will measure the ambient concentration of ozone
ATMOSPHERIC CHEMISTRY NITROGEN DIOXIDE Version 1.1 (previously AC Protocol, updated Jan 2001)
 AN Protocol Aim Rationale Method ATMOSPHERIC CHEMISTRY NITROGEN DIOXIDE Version 1.1 (previously AC Protocol, updated Jan 2001) To detect and define changes in periodic mean atmospheric concentrations of
AN Protocol Aim Rationale Method ATMOSPHERIC CHEMISTRY NITROGEN DIOXIDE Version 1.1 (previously AC Protocol, updated Jan 2001) To detect and define changes in periodic mean atmospheric concentrations of
Section II. The Physical Geography and Growth Patterns of the Counties and Municipalities of the Central Midlands Region of South Carolina
 Section II The Physical Geography and Growth Patterns of the Counties and Municipalities of the Central Midlands Region of South Carolina The Central Midlands region is comprised of the four-county area
Section II The Physical Geography and Growth Patterns of the Counties and Municipalities of the Central Midlands Region of South Carolina The Central Midlands region is comprised of the four-county area
2012 Sulfur Dioxide Summary
 2012 Sulfur Dioxide Summary New Jersey Department of Environmental Protection NATURE AND SOURCES Sulfur dioxide (SO 2 ) is a heavy, colorless gas with a suffocating odor that easily dissolves in water
2012 Sulfur Dioxide Summary New Jersey Department of Environmental Protection NATURE AND SOURCES Sulfur dioxide (SO 2 ) is a heavy, colorless gas with a suffocating odor that easily dissolves in water
2010 Sulfur Dioxide Summary
 2010 Sulfur Dioxide Summary New Jersey Department of Environmental Protection NATURE AND SOURCES Sulfur dioxide (SO 2 ) is a heavy, colorless gas with a suffocating odor that easily dissolves in water
2010 Sulfur Dioxide Summary New Jersey Department of Environmental Protection NATURE AND SOURCES Sulfur dioxide (SO 2 ) is a heavy, colorless gas with a suffocating odor that easily dissolves in water
ECONOMIC DEVELOPMENT OF SOUTH CAROLINA S I-95 COUNTIES: INFRASTRUCTURE DEVELOPMENT AND PROJECT POSSIBILITIES
 ECONOMIC DEVELOPMENT OF SOUTH CAROLINA S I-95 COUNTIES: INFRASTRUCTURE DEVELOPMENT AND PROJECT POSSIBILITIES Robert T. Barrett, SC State University, Orangeburg, SC, 29117, 803-536-8186 rbarret1@scsu.edu
ECONOMIC DEVELOPMENT OF SOUTH CAROLINA S I-95 COUNTIES: INFRASTRUCTURE DEVELOPMENT AND PROJECT POSSIBILITIES Robert T. Barrett, SC State University, Orangeburg, SC, 29117, 803-536-8186 rbarret1@scsu.edu
2011 Particulate Summary
 2011 Particulate Summary New Jersey Department of Environmental Protection NATURE AND SOURCES Particulate air pollution is a complex mixture of organic and inorganic substances present in the atmosphere
2011 Particulate Summary New Jersey Department of Environmental Protection NATURE AND SOURCES Particulate air pollution is a complex mixture of organic and inorganic substances present in the atmosphere
Fine Particles in the Air
 Fine Particles in the Air Presentation for STAPPA Outreach Committee Rich Damberg, EPA Office of Air Quality Planning and Standards April 6, 2004 Particulate Matter: What is It? A complex mixture of extremely
Fine Particles in the Air Presentation for STAPPA Outreach Committee Rich Damberg, EPA Office of Air Quality Planning and Standards April 6, 2004 Particulate Matter: What is It? A complex mixture of extremely
2014 Ozone Summary. Figure 1: Good and Bad Ozone. POLLUTION THAT FORMS GROUND-LEVEL OZONE, A PRIMARY COMPONENT OF SMOG.
 2014 Ozone Summary New Jersey Department of Environmental Protection NATURE AND SOURCES Ozone (O 3 ) is a gas consisting of three oxygen atoms. It occurs naturally in the upper atmosphere (stratospheric
2014 Ozone Summary New Jersey Department of Environmental Protection NATURE AND SOURCES Ozone (O 3 ) is a gas consisting of three oxygen atoms. It occurs naturally in the upper atmosphere (stratospheric
Passive Monitoring: A Guide to Sorbent Tube Sampling for EPA Method 325. Nicola Watson July 2015
 Passive Monitoring: A Guide to Sorbent Tube Sampling for EPA Method 325 Nicola Watson July 2015 EPA 325 - Refinery perimeter monitoring Revised Federal regulation (CFR 40, part 60 and 63) to be implemented
Passive Monitoring: A Guide to Sorbent Tube Sampling for EPA Method 325 Nicola Watson July 2015 EPA 325 - Refinery perimeter monitoring Revised Federal regulation (CFR 40, part 60 and 63) to be implemented
MODELING GROWTH AND PREDICTING FUTURE DEVELOPED LAND IN THE UPSTATE OF SOUTH CAROLINA
 MODELING GROWTH AND PREDICTING FUTURE DEVELOPED LAND IN THE UPSTATE OF SOUTH CAROLINA Craig E. Campbell, Jeffery Allen, and Kang Shou Lu AUTHORS: Craig E. Campbell, Ph.D., Research Associate, Strom Thurmond
MODELING GROWTH AND PREDICTING FUTURE DEVELOPED LAND IN THE UPSTATE OF SOUTH CAROLINA Craig E. Campbell, Jeffery Allen, and Kang Shou Lu AUTHORS: Craig E. Campbell, Ph.D., Research Associate, Strom Thurmond
A Spatial Investigation of Highly-Resolved Environmental Benzene Concentrations Using Passive Sampling
 A Spatial Investigation of Highly-Resolved Environmental Benzene Concentrations Using Passive Sampling 2011-A-72-SP-AWMA Samantha C. Fridh, Amy L. Stuart University of South Florida, Department of Environmental
A Spatial Investigation of Highly-Resolved Environmental Benzene Concentrations Using Passive Sampling 2011-A-72-SP-AWMA Samantha C. Fridh, Amy L. Stuart University of South Florida, Department of Environmental
Methods and Materials Manual Of Ion Chromatography Using DX-100 IC. Collecting Samples. LDPE bottle with cap (pre-rinsed with DI water)
 Methods and Materials Manual Of Ion Chromatography Using DX-100 IC Collecting Samples Field Material: LDPE bottle with cap (pre-rinsed with DI water) permanent marker thermometer field notebook 1. Record
Methods and Materials Manual Of Ion Chromatography Using DX-100 IC Collecting Samples Field Material: LDPE bottle with cap (pre-rinsed with DI water) permanent marker thermometer field notebook 1. Record
MERCAPTANS, METHYL-, ETHYL-, and n-butyl- 2542
 MERCAPTANS, METHYL-, ETHYL-, and n-butyl- 2542 SH MW: 48.11 CAS: 74-93-1 RTECS: PB4375000 SH 62.13 75-08-1 KI9625000 SH 90.19 109-79-5 EK6300000 METHOD: 2542, Issue 1 EVALUATION: PARTIAL Issue 1: 15 August
MERCAPTANS, METHYL-, ETHYL-, and n-butyl- 2542 SH MW: 48.11 CAS: 74-93-1 RTECS: PB4375000 SH 62.13 75-08-1 KI9625000 SH 90.19 109-79-5 EK6300000 METHOD: 2542, Issue 1 EVALUATION: PARTIAL Issue 1: 15 August
Overview of the Human Health and Environmental Effects of Power Generation: Focus on Sulfur Dioxide (SO 2 ), Nitrogen Oxides (NO X ) and Mercury (Hg)
 The information presented here reflects EPA's modeling of the Clear Skies Act of 2002. The Agency is in the process of updating this information to reflect modifications included in the Clear Skies Act
The information presented here reflects EPA's modeling of the Clear Skies Act of 2002. The Agency is in the process of updating this information to reflect modifications included in the Clear Skies Act
Operation and Performance of the Gas In-Vitro Exposure System (GIVES)
 Operation and Performance of the Gas In-Vitro Exposure System (GIVES) Supplement to manuscript APC-2012-1: Gaseous VOCs Rapidly Modify Particulate Matter and Its Biological Effects: Part 1, Simple VOCs
Operation and Performance of the Gas In-Vitro Exposure System (GIVES) Supplement to manuscript APC-2012-1: Gaseous VOCs Rapidly Modify Particulate Matter and Its Biological Effects: Part 1, Simple VOCs
Changes in the Ozone Standard: What will the impact be? February 9, 2016 Sara G. Smith
 Changes in the Ozone Standard: What will the impact be? February 9, 2016 Sara G. Smith What is Ozone? Ozone is a colorless, odorless gas composed of three oxygen atoms joined together. Most oxygen in the
Changes in the Ozone Standard: What will the impact be? February 9, 2016 Sara G. Smith What is Ozone? Ozone is a colorless, odorless gas composed of three oxygen atoms joined together. Most oxygen in the
INTERDISCIPLINARY INVESTIGATION (IDI)-LAB LABORATORY HANDOUT
 INTERDISCIPLINARY INVESTIGATION (IDI)-LAB LABORATORY HANDOUT Research-based Interdisciplinary Science Education ION CHROMATOGRAPHY TO INVESTIGATE WATER QUALITY ISSUES: FLUORIDE IN DRINKING WATER Introduction
INTERDISCIPLINARY INVESTIGATION (IDI)-LAB LABORATORY HANDOUT Research-based Interdisciplinary Science Education ION CHROMATOGRAPHY TO INVESTIGATE WATER QUALITY ISSUES: FLUORIDE IN DRINKING WATER Introduction
Diesel health effects West Coast Collaborative Hawai'i Partners Meeting June 25, 2008
 Diesel health effects West Coast Collaborative Hawai'i Partners Meeting June 25, 2008 Sumeet Saksena, Ph.D. The East-West Center Honolulu What is diesel exhaust (DE)? Most important: fine particulate matter,
Diesel health effects West Coast Collaborative Hawai'i Partners Meeting June 25, 2008 Sumeet Saksena, Ph.D. The East-West Center Honolulu What is diesel exhaust (DE)? Most important: fine particulate matter,
The Good, the Bad, and the Ugly of Ozone
 Name: Date: Period: Background The Good, the Bad, and the Ugly of Ozone Ozone (O 3 ) is colorless and consists of three oxygen atoms (the third oxygen makes it highly reactive). There are two main types
Name: Date: Period: Background The Good, the Bad, and the Ugly of Ozone Ozone (O 3 ) is colorless and consists of three oxygen atoms (the third oxygen makes it highly reactive). There are two main types
Chapter Introduction. 1.2 Purpose. 1.3 Background. 1.4 Ozone Production and Health Effects. 1.5 Responsibilities of Agencies
 Chapter 1 1.1 Introduction 1.2 Purpose 1.3 Background 1.4 Ozone Production and Health Effects 1.5 Responsibilities of Agencies 1.1 INTRODUCTION On July 18, 1997, the United States Environmental Protection
Chapter 1 1.1 Introduction 1.2 Purpose 1.3 Background 1.4 Ozone Production and Health Effects 1.5 Responsibilities of Agencies 1.1 INTRODUCTION On July 18, 1997, the United States Environmental Protection
Air quality in the vicinity of a governmental school in Kuwait
 Air Pollution XVI 237 Air quality in the vicinity of a governmental school in Kuwait E. Al-Bassam 1, V. Popov 2 & A. Khan 1 1 Environment and Urban Development Division, Kuwait Institute for Scientific
Air Pollution XVI 237 Air quality in the vicinity of a governmental school in Kuwait E. Al-Bassam 1, V. Popov 2 & A. Khan 1 1 Environment and Urban Development Division, Kuwait Institute for Scientific
Chapter 17 Air and Air Pollution
 Chapter 17 Air and Air Pollution The Atmosphere Each layer characterized by abrupt changes in temperature, the result of differences in the absorption of incoming solar energy. Troposphere: Weather Breeder
Chapter 17 Air and Air Pollution The Atmosphere Each layer characterized by abrupt changes in temperature, the result of differences in the absorption of incoming solar energy. Troposphere: Weather Breeder
Guidance on a laboratory leaching test method for materials that are treated with biocides
 Final version 12 October 2015 Guidance on a laboratory leaching test method for materials that are treated with biocides to fulfil requirements of the European Biocidal Products Regulation This guidance
Final version 12 October 2015 Guidance on a laboratory leaching test method for materials that are treated with biocides to fulfil requirements of the European Biocidal Products Regulation This guidance
EFFECT OF OZONE AND DISTANCE FROM A MAJOR ROADWAY ON NITROGEN OXIDES CONCENTRATIONS
 HAMPTON UNIVERSITY EASTERN SEABOARD INTERMODAL TRANSPORTATION APPLICATIONS CENTER EFFECT OF OZONE AND DISTANCE FROM A MAJOR ROADWAY ON NITROGEN OXIDES CONCENTRATIONS Dr. Ates Akyurtlu Professor of Chemical
HAMPTON UNIVERSITY EASTERN SEABOARD INTERMODAL TRANSPORTATION APPLICATIONS CENTER EFFECT OF OZONE AND DISTANCE FROM A MAJOR ROADWAY ON NITROGEN OXIDES CONCENTRATIONS Dr. Ates Akyurtlu Professor of Chemical
THE. Where are VOCs regulated?
 THE In markets subject to volatile organic compound (VOC) regulations, single-ply roof membrane manufacturers supply alternative materials for solvent-based adhesives and primers. Among the VOC regulation-compliant
THE In markets subject to volatile organic compound (VOC) regulations, single-ply roof membrane manufacturers supply alternative materials for solvent-based adhesives and primers. Among the VOC regulation-compliant
Ambient Air Quality Monitoring
 Ambient Air Quality Monitoring Ambient Air Quality Division Air Quality and Noise Management Bureau Pollution Control Department 7/31/2012 2:09 PM 1 Carbon Monoxide (CO) Nitrogen Dioxide (NO 2 ) Ozone
Ambient Air Quality Monitoring Ambient Air Quality Division Air Quality and Noise Management Bureau Pollution Control Department 7/31/2012 2:09 PM 1 Carbon Monoxide (CO) Nitrogen Dioxide (NO 2 ) Ozone
Chapter 20 Air Pollution
 Chapter 20 Air Pollution Overview of Chapter 20 Atmosphere as a Resource Types and Sources of Air Pollution Effects of Air Pollution Controlling Air Pollution in the US Ozone Depletion in the Stratosphere
Chapter 20 Air Pollution Overview of Chapter 20 Atmosphere as a Resource Types and Sources of Air Pollution Effects of Air Pollution Controlling Air Pollution in the US Ozone Depletion in the Stratosphere
An Introduction to Air Quality
 An Introduction to Air Quality Learning Goals "We came all this way to explore the Moon, and the most important thing is that we discovered the Earth - William Anders After this lesson, you will be able
An Introduction to Air Quality Learning Goals "We came all this way to explore the Moon, and the most important thing is that we discovered the Earth - William Anders After this lesson, you will be able
Air pollution is caused by high concentrations of gases and particles emitted form combustion sources (vehicles, power plants, industries)
 AIR POLLUTION Clean Air Act & Criteria Pollutants 7.1-7.5 Motor Vehicle Emissions: 7.8 Indoor Air and Box Models: 7.12 Air Pollution Meteorology: 7.10 Gaussian Plume Modeling: 7.11 1 Air pollution is caused
AIR POLLUTION Clean Air Act & Criteria Pollutants 7.1-7.5 Motor Vehicle Emissions: 7.8 Indoor Air and Box Models: 7.12 Air Pollution Meteorology: 7.10 Gaussian Plume Modeling: 7.11 1 Air pollution is caused
NO 2 Primary NAAQS Final Rulemaking
 NO 2 Primary NAAQS Final Rulemaking NO2 Briefing February 10, 2011 Prepared for WILMAPCO By Gregory A. Becoat Air Program Planning 1 Schedule Action Date Revised NO2 NAAQS signed January 22, 2010 State/Tribe
NO 2 Primary NAAQS Final Rulemaking NO2 Briefing February 10, 2011 Prepared for WILMAPCO By Gregory A. Becoat Air Program Planning 1 Schedule Action Date Revised NO2 NAAQS signed January 22, 2010 State/Tribe
Collection of Samples for Analysis of Volatile Organic Compounds (VOCs) in Indoor Air Using Sorbent Tubes. BAA Guide October 1, 2007
 BERKELEY ANALYTICAL ASSOCIATES 815 Harbour Way South, Unit 6 Richmond, CA 94804-3612 Tel: 510-236-2325 Fax: 510-236-2335 Email: baalab@berkeleyanalytical.com www.berkeleyanalytical.com Collection of Samples
BERKELEY ANALYTICAL ASSOCIATES 815 Harbour Way South, Unit 6 Richmond, CA 94804-3612 Tel: 510-236-2325 Fax: 510-236-2335 Email: baalab@berkeleyanalytical.com www.berkeleyanalytical.com Collection of Samples
Lab 7 Measurement of Ozone
 Georgia Institute of Technology School of Earth and Atmospheric Sciences EAS 4641 Spring 2007 Lab 7 Measurement of Ozone Purpose of Lab 7: In this lab you will measure the ambient concentration of ozone
Georgia Institute of Technology School of Earth and Atmospheric Sciences EAS 4641 Spring 2007 Lab 7 Measurement of Ozone Purpose of Lab 7: In this lab you will measure the ambient concentration of ozone
Existing Conditions and Environmental Consequences Water Quality
 3.6 3.6.1 HOW IS WATER QUALITY ASSESSED? The Clean Water Act (CWA) of 1972 requires that each state set water quality standards for all contaminants in surface waters. These standards are typically based
3.6 3.6.1 HOW IS WATER QUALITY ASSESSED? The Clean Water Act (CWA) of 1972 requires that each state set water quality standards for all contaminants in surface waters. These standards are typically based
X X. From the Office of Governor Nikki Haley: 2011 Legislative Report Card for the South Carolina General Assembly LEGISLATION SENATE HOUSE 50% 100%
 From the Office of Governor Nikki Haley: 2011 Legislative Report Card for the South Carolina General Assembly LEGISLATION SENATE On the Record Voting : Requires legislators votes to be recorded, which
From the Office of Governor Nikki Haley: 2011 Legislative Report Card for the South Carolina General Assembly LEGISLATION SENATE On the Record Voting : Requires legislators votes to be recorded, which
2012 Nitrogen Dioxide Summary
 2012 Nitrogen Dioxide Summary New Jersey Department of Environmental Protection NATURE AND SOURCES Nitrogen Dioxide (NO 2 ) is a reddishbrown, highly reactive gas that is formed in the air through the
2012 Nitrogen Dioxide Summary New Jersey Department of Environmental Protection NATURE AND SOURCES Nitrogen Dioxide (NO 2 ) is a reddishbrown, highly reactive gas that is formed in the air through the
GE 2211 Environmental Science and Engineering Unit III Air Pollution. M. Subramanian
 GE 2211 Environmental Science and Engineering Unit III Air Pollution M. Subramanian Assistant Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering Kalavakkam 603
GE 2211 Environmental Science and Engineering Unit III Air Pollution M. Subramanian Assistant Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering Kalavakkam 603
Earth s Atmosphere. Air Quality. 7. If no humans lived on Earth, there would be no air pollution.
 CHAPTER 4 LESSON 4 Earth s Atmosphere Air Quality Key Concepts How do humans impact air quality? Why do humans monitor air quality standards? What do you think? Read the two statements below and decide
CHAPTER 4 LESSON 4 Earth s Atmosphere Air Quality Key Concepts How do humans impact air quality? Why do humans monitor air quality standards? What do you think? Read the two statements below and decide
High-Quality Passive Sampling of NO-NO x, NO 2, SO 2, O 3 and NH 3 in Indoor or Outdoor Environments
 High-Quality Passive Sampling of NO-NO x, NO 2, SO 2, O 3 and NH 3 in Indoor or Outdoor Environments 1230 S.E. 7th Avenue Pompano Beach, FL 33060 USA Phone +1 (954) 781-6223 Fax +1 (954) 781-6224 E-Mail
High-Quality Passive Sampling of NO-NO x, NO 2, SO 2, O 3 and NH 3 in Indoor or Outdoor Environments 1230 S.E. 7th Avenue Pompano Beach, FL 33060 USA Phone +1 (954) 781-6223 Fax +1 (954) 781-6224 E-Mail
Surface ozone measurements in mountain forested areas using diffusive samplers
 Air Pollution XIV 585 Surface ozone measurements in mountain forested areas using diffusive samplers I. Hůnová Czech Hydrometeorological Institute in Prague, Czech Republic Institute for Environmental
Air Pollution XIV 585 Surface ozone measurements in mountain forested areas using diffusive samplers I. Hůnová Czech Hydrometeorological Institute in Prague, Czech Republic Institute for Environmental
OUR NATION'S AIR STATUS AND TRENDS THROUGH 2015 WELCOME!
 Page 1 of 14 OUR NATION'S AIR STATUS AND TRENDS THROUGH 2015 WELCOME! The U.S. Environmental Protection Agency (EPA) is committed to protecting public health by improving air quality and reducing air pollution.
Page 1 of 14 OUR NATION'S AIR STATUS AND TRENDS THROUGH 2015 WELCOME! The U.S. Environmental Protection Agency (EPA) is committed to protecting public health by improving air quality and reducing air pollution.
Indoor Air Pollution. Indoor air pollution can be an even greater health threat than outdoor air pollution. Common pollutants often
 Indoor Air Pollution Indoor air pollution can be an even greater health threat than outdoor air pollution. Common pollutants often. Smoke 4 Most Hazardous Indoor Pollutants Formaldehyde: Asbestos: Outgassed
Indoor Air Pollution Indoor air pollution can be an even greater health threat than outdoor air pollution. Common pollutants often. Smoke 4 Most Hazardous Indoor Pollutants Formaldehyde: Asbestos: Outgassed
Tropospheric Ozone Pollution and Personal Exposure
 Tropospheric Ozone Pollution and Personal Exposure Lindsay Willman ATOC 3500 22 April 2010 Photo: NREL Tropospheric Ozone Formation http://www.derm.qld.gov.au/images/environmental_management/air/ozone-formation.jpg
Tropospheric Ozone Pollution and Personal Exposure Lindsay Willman ATOC 3500 22 April 2010 Photo: NREL Tropospheric Ozone Formation http://www.derm.qld.gov.au/images/environmental_management/air/ozone-formation.jpg
Approved for NPDES (Editorial Revision 1978) Silica, Dissolved (Colorimetric)
 METHOD #: 370.1 TITLE: Approved for NPDES (Editorial Revision 1978) Silica, Dissolved (Colorimetric) ANALYTE: Silica, SiO 2 INSTRUMENTATION: Spectrophotometer STORET No. Dissolved 00955 1.0 Scope and Application
METHOD #: 370.1 TITLE: Approved for NPDES (Editorial Revision 1978) Silica, Dissolved (Colorimetric) ANALYTE: Silica, SiO 2 INSTRUMENTATION: Spectrophotometer STORET No. Dissolved 00955 1.0 Scope and Application
High-Quality Passive Sampling of NO-NO x, NO 2, SO 2, O 3 and NH 3 in Indoor or Outdoor Environments
 High-Quality Passive Sampling of NO-NO x, NO 2, SO 2, O 3 and NH 3 in Indoor or Outdoor Environments 1230 S.E. 7th Avenue Pompano Beach, FL 33060 USA Phone +1 (954) 781-6223 Fax +1 (954) 781-6224 E-Mail
High-Quality Passive Sampling of NO-NO x, NO 2, SO 2, O 3 and NH 3 in Indoor or Outdoor Environments 1230 S.E. 7th Avenue Pompano Beach, FL 33060 USA Phone +1 (954) 781-6223 Fax +1 (954) 781-6224 E-Mail
Effect of PM2.5 on AQI in Taiwan
 Environmental Modelling & Software 17 (2002) 29 37 www.elsevier.com/locate/envsoft Effect of PM2.5 on AQI in Taiwan Chung-Ming Liu * Department of Atmospheric Sciences, National Taiwan University, Taipei,
Environmental Modelling & Software 17 (2002) 29 37 www.elsevier.com/locate/envsoft Effect of PM2.5 on AQI in Taiwan Chung-Ming Liu * Department of Atmospheric Sciences, National Taiwan University, Taipei,
Section 4 The Air We Breathe
 Section 4 The Air We Breathe Key Concept Air is an important natural resource that is affected by human activities. What You Will Learn Air pollution is caused by human activities, such as burning fossil
Section 4 The Air We Breathe Key Concept Air is an important natural resource that is affected by human activities. What You Will Learn Air pollution is caused by human activities, such as burning fossil
Practical guidance for switching between anion and cation analysis systems
 TECHNICAL NOTE 729 Practical guidance for switching between anion and cation analysis systems Authors Introduction Manali Aggrawal and Jeffrey Rohrer Thermo Fisher Scientific, Sunnyvale, CA, USA Ion chromatography
TECHNICAL NOTE 729 Practical guidance for switching between anion and cation analysis systems Authors Introduction Manali Aggrawal and Jeffrey Rohrer Thermo Fisher Scientific, Sunnyvale, CA, USA Ion chromatography
Prepared for The South Carolina General Assembly and The South Carolina Department of Labor, Licensing and Regulation (LLR)
 Assessment of the Seismic Provisions in the 2015 International Residential Code, the 2014 USGS Seismic Hazard Maps for South Carolina, and the Likely Impact of Elevated Seismic Hazard on the 2018 International
Assessment of the Seismic Provisions in the 2015 International Residential Code, the 2014 USGS Seismic Hazard Maps for South Carolina, and the Likely Impact of Elevated Seismic Hazard on the 2018 International
So What s the Matter With Ozone?
 So What s the Matter With Ozone? Activity 3D Activity Objectives: This activity is written in five parts so students can conduct an in depth study of ozone and its effects on our health or can do each
So What s the Matter With Ozone? Activity 3D Activity Objectives: This activity is written in five parts so students can conduct an in depth study of ozone and its effects on our health or can do each
