MICOTECA DA UNIVERSIDADE DO MINHO (MUM): IMPLEMENTATION OF A QUALITY MANAGEMENT SYSTEM BASED ON ISO 9001:2008
|
|
|
- Caroline Singleton
- 6 years ago
- Views:
Transcription
1 MICOTECA DA UNIVERSIDADE DO MINHO (MUM): IMPLEMENTATION OF A QUALITY MANAGEMENT SYSTEM BASED ON ISO 9001:2008 Marta F. Simões, Anabela Martins, Nicolina Dias, Cledir Santos, Nelson Lima
2 MUM: established in 1996 holds a filamentous fungi culture collection share the facilities of the DEB at Minho University Develop and implement a Quality Management System - QMS: Based on: NP EN ISO 9001:2008
3 MISSION Provide the highest quality services to our customers, collecting, maintaining and supplying fungal strains and their associated information for teaching and research in biotechnology and life sciences, and to be a centre of knowledge, information and training in mycology, operating at a global level and under national and international regulations.
4 Quality Policy of MUM MUM is committed to Quality by valuing costumer s needs and supplying high quality products and services To achieve this, the objective of MUM is the ongoing development of its management processes to continually improve services Using and developing key performance indicators Creating a dynamic work environment by being innovative and performing a high-quality research Following the technological and scientific development of the sector Continuously developing the expertise, professionalism and integrity of the collaborators of MUM.
5 GENERAL QUALITY OBJECTIVES: Strive for complete understanding and meet the standards of customer Continually improve MUM process performance developing key indicators by identification of critical success factors Following the technological and scientific development of the sector by being present in a regular basis on relevant scientific events Continually developing the expertise, professionalism and integrity of MUM collaborators by appropriate training Maintain a dynamic work team performing a innovative high-quality research Foster a team approach to detect prevention and problem solving
6 MUM has identified its processes: MATERIAL RECEPTION PROCESS (MRP) MATERIAL PRESERVATION PROCESS (MPP) MATERIAL SUPPLY PROCESS (MSP)
7
8 CUSTOMER REQUIREMENTS Quality Policy Quality Objectives Human Resources Internal Communication Quality System Revision Claims Suggestions Preventive Actions MANAGEMENT RESPONSABILITY COUNTINOUS IMPROVEMENT Corrective Actions MRP MPP MSP RESOURCES MANAGEMENT MEASUREMENT AND MONITORIZATION Infrastructure Work Environment Internal Audit Customer Satisfaction Process Performance CUSTOMER SATISFACTION
9 Areas of intervention: 1. Creation of a documental structure Legal and regulatory requirements OECD best practices
10
11 Areas of intervention: 2. Description and implementation of processes, procedures and SOPs 3. Selection, evaluation and qualification of suppliers (reception of materials, non-conformities record, corrective actions request) 4. Infra-structure and work environment maintenance 5. Measurement and monitoring equipment (inventory, equipment charts, Control and Maintenance plan) 6. Human resources management (minimum skills required, responsabilities and job description) 7. Customer s satisfaction evaluation (satisfaction inquiry, complaints evaluation, suggestions)
12 CUSTOMER SATISFACTION INQUIRY QUESTION VP P A S VS DID YOU RECEIVE ALL THE PRODUCTS REQUESTED ON TIME? DID THE STRAINS ARRIVE IN GOOD CONDITION? WERE THE INSTRUCTIONS CLEAR AND EFFECTIVE? HOW WAS THE IDENTIFICATION SERVICE? HOW WAS THE TRAINING COURSE OFFERED BY THE MUM? HOW WAS THE STRAINS DEPOSIT SERVICE? HOW WAS THE WEB PAGE ACCESS? DID YOU FEEL PROPERLY ATTENDED TO BY OUR STAFF? HOW WAS THE INFORMATION PROVIDED? HOW WAS THE QUALITY/COST RATIO? WHAT IS YOUR FINAL EVALUATION?
13 Areas of intervention: 8. Continuous improvement Internal audit Corrective and preventive actions Action planning Planning review
14 SYSTEEM IMPLEMENTATI SYSTEM DESIGN AND DOCUMENTATION PLANNING IMPLEMENTATION STEPS M A M J J A S O N D WORK TEAM AND TASKS TEAMS DEFINITION SCHEDULE DEFINITION QUALITY POLICY AND QUALITY OBJECTIVES DEFINITION BUILDING SYSTEM STRUCTURE PROCESS DOCUMENTATION AND CONTROL RESOURCE MANAGEMENT SYSTEM PLANNING INTERNAL AND CUSTOMER COMMUNICATION ANALYSIS AND APPROVALL OF DOCUMENTS RECORDING THE SYSTEM INTERNAL AUDITS SYSTEM REVISION AND IMPROVEMENT OPPORTUNITIES
15 Benefits fom the QMS implementation Improved control and planning Improved efficiency and productivity Consistency in products and services Reduced waste of time and resources Reduced costs Improved collaborators retention/motivation
16 QMS implementation Requalification of MUM Certification Customer satisfaction
17 Thank you for your attention!
PROACTIVE APPROACHES. for QUALITY MANAGEMENT. Anabela Martins Micoteca da Universidade do Minho
 PROACTIVE APPROACHES for QUALITY MANAGEMENT Anabela Martins Micoteca da Universidade do Minho 1 Standard development procedure Reference sources Structure Interpreting the provisions Recognition scheme
PROACTIVE APPROACHES for QUALITY MANAGEMENT Anabela Martins Micoteca da Universidade do Minho 1 Standard development procedure Reference sources Structure Interpreting the provisions Recognition scheme
INTEGRATING ISO 9000 METHODOLOGIES WITH PROJECT QUALITY MANAGEMENT
 INTEGRATING ISO 9000 METHODOLOGIES WITH PROJECT QUALITY MANAGEMENT M a r ch 2015 OBJECTIVE ISO and Project Quality Management Process Are they different or the same? ISO 9000 QMS FAMILY ISO 9000:2005 Vocabulary
INTEGRATING ISO 9000 METHODOLOGIES WITH PROJECT QUALITY MANAGEMENT M a r ch 2015 OBJECTIVE ISO and Project Quality Management Process Are they different or the same? ISO 9000 QMS FAMILY ISO 9000:2005 Vocabulary
Persian Type Culture Collection (WDCM 124)
 Persian Type Culture Collection (WDCM 124) Training Course of Microbial Resources Information Management and Utilization for Developing Countries 2-15 September 2014, WDCM Institute of Microbiology, CAS,
Persian Type Culture Collection (WDCM 124) Training Course of Microbial Resources Information Management and Utilization for Developing Countries 2-15 September 2014, WDCM Institute of Microbiology, CAS,
ASA-100 QUALITY MANUAL
 ASA-100 QUALITY MANUAL Origination Date: XXXX Document Identifier: Date: Document Revision: Latest Revision Date Abstract: This quality manual describes (your Company's) quality management system policies
ASA-100 QUALITY MANUAL Origination Date: XXXX Document Identifier: Date: Document Revision: Latest Revision Date Abstract: This quality manual describes (your Company's) quality management system policies
APS Cleaning Quality Management System Scope of Certification The provision of commercial and industrial cleaning services throughout Queensland.
 Quality Management System Scope of Certification The provision of commercial and industrial cleaning services throughout Queensland. Table of Contents Contents 1. Introduction... 3 1.1. Process Approach...
Quality Management System Scope of Certification The provision of commercial and industrial cleaning services throughout Queensland. Table of Contents Contents 1. Introduction... 3 1.1. Process Approach...
QMS CO-ORDINATOR & GENERAL PROCEDURES:-
 1. QMS CO-ORDINATOR & GENERAL PROCEDURES:- (1) How do you measure effectiveness of system for working of your company? How do you collect necessary information for the same? Are you getting information
1. QMS CO-ORDINATOR & GENERAL PROCEDURES:- (1) How do you measure effectiveness of system for working of your company? How do you collect necessary information for the same? Are you getting information
Quality Manual. This manual complies with the requirements of the ISO 9001:2015 International Standard. AW2 Logistics, Inc Ace Industrial Dr.
 Quality Manual This manual complies with the requirements of the ISO 9001:2015 International Standard. AW2 Logistics, Inc. 6001 Ace Industrial Dr. Cudahy, WI 53210 Quality Manual Rev 3 Page 1 of 30 Table
Quality Manual This manual complies with the requirements of the ISO 9001:2015 International Standard. AW2 Logistics, Inc. 6001 Ace Industrial Dr. Cudahy, WI 53210 Quality Manual Rev 3 Page 1 of 30 Table
Integrated Management Systems. John Marsden Marsden International (UK) Ltd
 Integrated Management Systems John Marsden Marsden International (UK) Ltd 1 Why develop an IMS? Streamline management systems Use of common elements Ease of implementation of systems Better control and
Integrated Management Systems John Marsden Marsden International (UK) Ltd 1 Why develop an IMS? Streamline management systems Use of common elements Ease of implementation of systems Better control and
Fitting ISO 9001:2000 into a 20 Element Quality System
 Fitting ISO 9001:2000 into a 20 Element Quality System ASQ Washington DC Section 0509 Rockville Maryland Presented by Norman P. Moreau, P.E., CSQE, CQA President Theseus Professional Services 410-857-0023
Fitting ISO 9001:2000 into a 20 Element Quality System ASQ Washington DC Section 0509 Rockville Maryland Presented by Norman P. Moreau, P.E., CSQE, CQA President Theseus Professional Services 410-857-0023
ISO 9001:2000 Gap Analysis Report
 The 9000 Advisers ISO 9001:2000 Gap Analysis Report Presented by The 9000 Advisers 290 Edgewater Drive Middleton, TN 38052 731-376-8558 www.9000advisers.com The 9000 Advisers ISO 9001:2000 Gap Analysis
The 9000 Advisers ISO 9001:2000 Gap Analysis Report Presented by The 9000 Advisers 290 Edgewater Drive Middleton, TN 38052 731-376-8558 www.9000advisers.com The 9000 Advisers ISO 9001:2000 Gap Analysis
Stanley Industries, Inc. ISO 9001:2008 Quality Policy Manual
 Stanley ISO 9001:2008 Table of Contents and STANLEY Document Reference Related STANLEY Section Page Procedure(s) 1. Introduction 1 None 2. Scope 1 None 3. Organizational Structure & 1 STANLEY Company History
Stanley ISO 9001:2008 Table of Contents and STANLEY Document Reference Related STANLEY Section Page Procedure(s) 1. Introduction 1 None 2. Scope 1 None 3. Organizational Structure & 1 STANLEY Company History
Quality Assurance Manual, Revision F Page 1 of 20
 Quality Assurance Manual, Revision F Page 1 of 20 AUTOMATION TECHNOLOGY QUALITY ASSURANCE MANUAL Prepared By: Joseph Pollard Date: 7/6/2015 Quality Assurance Manager Quality Management System Representative
Quality Assurance Manual, Revision F Page 1 of 20 AUTOMATION TECHNOLOGY QUALITY ASSURANCE MANUAL Prepared By: Joseph Pollard Date: 7/6/2015 Quality Assurance Manager Quality Management System Representative
Quality Manual. Index
 This quality policy manual is for the use of, its Clients, Vendors, and the appropriate Regulatory Agencies. Unauthorized duplication or transfer to others is prohibited. Index Authorization... 3 Introduction...
This quality policy manual is for the use of, its Clients, Vendors, and the appropriate Regulatory Agencies. Unauthorized duplication or transfer to others is prohibited. Index Authorization... 3 Introduction...
Clause Map IATF 16949:2016 to ISO/TS 16949:2009
 Table of Contents Table of Contents Foreword Foreword + Foreword Automotive QMS Standard + History + Goal + 0.5 Goal of this Technical Specification + Remarks for Certification + Remarks for Certification
Table of Contents Table of Contents Foreword Foreword + Foreword Automotive QMS Standard + History + Goal + 0.5 Goal of this Technical Specification + Remarks for Certification + Remarks for Certification
Quality Assurance. QA Programs for Local Health Departments
 Quality Assurance QA Programs for Local Health Departments Objectives of this course are: Define Quality Know 6 Main Causes of Poor Quality Know 6 Service Quality Dimensions Know Characteristics of a Quality
Quality Assurance QA Programs for Local Health Departments Objectives of this course are: Define Quality Know 6 Main Causes of Poor Quality Know 6 Service Quality Dimensions Know Characteristics of a Quality
Risk Based Thinking & QMS Risk Management as per ISO
 Risk Based Thinking & QMS Risk Management as per ISO 9001-2015 PMI, PMP, PMBOK and the PMI Registered Education Provider logo are registered marks of the Project Management Institute, Inc. At the end of
Risk Based Thinking & QMS Risk Management as per ISO 9001-2015 PMI, PMP, PMBOK and the PMI Registered Education Provider logo are registered marks of the Project Management Institute, Inc. At the end of
The Skyworks Quality Management System strives to:
 Skyworks has embraced a workplace where Quality is the number one differentiator to achieve Customer Loyalty. Skyworks has adopted a single Quality Management System which drives efficiency, consistency
Skyworks has embraced a workplace where Quality is the number one differentiator to achieve Customer Loyalty. Skyworks has adopted a single Quality Management System which drives efficiency, consistency
POLICY MANUAL FOR ISO 9001:2008. Document: PM-9001:2008 Date: April 7, Uncontrolled Copy
 POLICY MANUAL FOR ISO 9001:2008 Document: PM-9001:2008 Date: April 7, 2015 REVIEWED BY: Tim Powers DATE: 4-7-2015 APPROVED BY: C._Bickford Uncontrolled Copy DATE: 4-7-2015 1.0 GENERAL ISS: 1 REV: E Page:
POLICY MANUAL FOR ISO 9001:2008 Document: PM-9001:2008 Date: April 7, 2015 REVIEWED BY: Tim Powers DATE: 4-7-2015 APPROVED BY: C._Bickford Uncontrolled Copy DATE: 4-7-2015 1.0 GENERAL ISS: 1 REV: E Page:
Quality System Manual - Section 00
 Quality System Manual - Section 00 INDEX AND REVISION STATUS Issued by: Quality Assurance Eff. Date: 00/00/00 Rev.: A Pg. 1 of 4 Organization of this manual is the same as the sectional organization of
Quality System Manual - Section 00 INDEX AND REVISION STATUS Issued by: Quality Assurance Eff. Date: 00/00/00 Rev.: A Pg. 1 of 4 Organization of this manual is the same as the sectional organization of
Luminus Devices, Inc Quality Management Systems Manual ISO 9001:2008
 Luminus Devices, Inc Quality Management Systems Manual ISO 9001:2008 This document belongs to Luminus Devices, Inc. It cannot be reproduced without authorized authority. Area: Quality System Document Page
Luminus Devices, Inc Quality Management Systems Manual ISO 9001:2008 This document belongs to Luminus Devices, Inc. It cannot be reproduced without authorized authority. Area: Quality System Document Page
A Division of Research Products Corporation
 A Division of Research Products Corporation Quality Manual ISO 9001:2008 Table of Contents Page Number Approvals 2 Table of Contents 3 Revision History 4 Distribution List - Controlled Copies 5 Introduction,
A Division of Research Products Corporation Quality Manual ISO 9001:2008 Table of Contents Page Number Approvals 2 Table of Contents 3 Revision History 4 Distribution List - Controlled Copies 5 Introduction,
Employee Orientation to ISO 9001
 Employee Orientation to ISO 9001 Sygnetics, Inc. is committed to quality. Quality is the ability to consistently produce a product or service that satisfies your customers needs. This presentation is designed
Employee Orientation to ISO 9001 Sygnetics, Inc. is committed to quality. Quality is the ability to consistently produce a product or service that satisfies your customers needs. This presentation is designed
Summary of ISO 9001:2015 New and Changed Requirements
 This is a summary of the new and changed ISO 9001:2015 requirements compared to ISO 9001:2008. 4. Context of the Organization 4.1 Changes Understanding the Organization and its Context New requirement
This is a summary of the new and changed ISO 9001:2015 requirements compared to ISO 9001:2008. 4. Context of the Organization 4.1 Changes Understanding the Organization and its Context New requirement
QUALITY SYSTEM MANUAL
 QUALITY SYSTEM MANUAL This Manual is a Proprietary Document and any Unauthorized Reproduction is prohibited. ISSUE DATE July 26, 2012 AUTHORIZED BY: Quality Management Representative Eric Hoff Managing
QUALITY SYSTEM MANUAL This Manual is a Proprietary Document and any Unauthorized Reproduction is prohibited. ISSUE DATE July 26, 2012 AUTHORIZED BY: Quality Management Representative Eric Hoff Managing
Operations Management
 Name: ID number: A PREVIOUS EXAM EXAMPLE A PREVIOUS EXAM EXAMPLE UNIVERSIDADE CATÓLICA PORTUGUESA F A C U L D A D E D E C I Ê N C I A S E C O N Ó M I C A S E E M P R E S A R I A I S Operations Management
Name: ID number: A PREVIOUS EXAM EXAMPLE A PREVIOUS EXAM EXAMPLE UNIVERSIDADE CATÓLICA PORTUGUESA F A C U L D A D E D E C I Ê N C I A S E C O N Ó M I C A S E E M P R E S A R I A I S Operations Management
Correlation matrices between ISO 9001:2008 and ISO 9001:2015
 Correlation matrices between ISO 9001:2008 and ISO 9001:2015 ISO 9001:2015 ISO 9001:2008 1 Scope 1 Scope 1.1 General 4 Context of the organization 4 Quality management system 4.1 Understanding the organization
Correlation matrices between ISO 9001:2008 and ISO 9001:2015 ISO 9001:2015 ISO 9001:2008 1 Scope 1 Scope 1.1 General 4 Context of the organization 4 Quality management system 4.1 Understanding the organization
Type Your Company Name Here. Quality Manual. AS9100 Rev C
 Blue text throughout the manual highlight areas for customization Type Your Company Name Here AS9100 Rev C Documents are in Microsoft Word for ease of editing Provides P general purpose and description
Blue text throughout the manual highlight areas for customization Type Your Company Name Here AS9100 Rev C Documents are in Microsoft Word for ease of editing Provides P general purpose and description
HACCPEUROPA PUBLICATIONS ISO 22000:2005 FOOD SAFETY QUALITY MANUAL. ISO 22000:2005 Quality Manual
 HACCPEUROPA PUBLICATIONS ISO 22000:2005 FOOD SAFETY QUALITY MANUAL ISO 22000:2005 Quality Manual QUALITY MANUAL ISO 22000:2005 Food Safety Management HACCPEuropa Publications 2012 Table of Contents Introduction...
HACCPEUROPA PUBLICATIONS ISO 22000:2005 FOOD SAFETY QUALITY MANUAL ISO 22000:2005 Quality Manual QUALITY MANUAL ISO 22000:2005 Food Safety Management HACCPEuropa Publications 2012 Table of Contents Introduction...
SUPPLIER QUALITY SYSTEM SURVEY
 SUPPLIER QUALITY SYSTEM SURVEY DATE (M/d/yy): 55 Dragon Court Woburn, MA 01801 Tel: (781) 933-7300 Fax: (781) 935-4529 GENERAL INFORMATION Supplier Name: Physical Address: City: State: Zip: Sr. Company
SUPPLIER QUALITY SYSTEM SURVEY DATE (M/d/yy): 55 Dragon Court Woburn, MA 01801 Tel: (781) 933-7300 Fax: (781) 935-4529 GENERAL INFORMATION Supplier Name: Physical Address: City: State: Zip: Sr. Company
Laboratory Quality Assurance Manager & Laboratory Assessor RULES & HANDBOOK
 EOQ Personnel Registration Scheme Laboratory Quality Assurance Manager & RULES & HANDBOOK Prepared by: Dr. Eugenia Soboleva, Quality Austria In accordance with the working group on EOQ product development
EOQ Personnel Registration Scheme Laboratory Quality Assurance Manager & RULES & HANDBOOK Prepared by: Dr. Eugenia Soboleva, Quality Austria In accordance with the working group on EOQ product development
Quality Management. Carlos Bachier, MD
 Quality Management Carlos Bachier, MD Goals of Quality Management Ensure credibility of outcomes Improve patient safety and quality of processes Significantly reduce errors Quality Medical and Laboratory
Quality Management Carlos Bachier, MD Goals of Quality Management Ensure credibility of outcomes Improve patient safety and quality of processes Significantly reduce errors Quality Medical and Laboratory
Quality Management Manual
 Quality Management Manual QUALITY MANAGEMENT MANUAL SCOPE..... This manual captures the quality procedures and methods employed by Apex Translations, Inc. All of these procedures and methods are strictly
Quality Management Manual QUALITY MANAGEMENT MANUAL SCOPE..... This manual captures the quality procedures and methods employed by Apex Translations, Inc. All of these procedures and methods are strictly
ISO 9001:2015. Quality Management System. Manual
 ISO 9001:2015 Quality Management System Manual Introduction Company has made the Strategic Business Decision to develop and implement an effective Quality Management Systems (QMS) across all areas of the
ISO 9001:2015 Quality Management System Manual Introduction Company has made the Strategic Business Decision to develop and implement an effective Quality Management Systems (QMS) across all areas of the
What is ISO 9001 QMS? Business Beam
 1 What is ISO 9001 QMS? Business Beam Contents 2 What is Quality About Quality Management System About ISO 9001 ISO 9001 Certification Process 3 What is Quality? What is ISO 9001 QMS? What is Quality?
1 What is ISO 9001 QMS? Business Beam Contents 2 What is Quality About Quality Management System About ISO 9001 ISO 9001 Certification Process 3 What is Quality? What is ISO 9001 QMS? What is Quality?
QUALITY SYSTEM MANUAL
 TITLE: QUALITY SYSTEM MANUAL Page 1 of 15 QUALITY SYSTEM MANUAL TITLE: QUALITY SYSTEM MANUAL Page 2 of 15 Index PARAGRAPH TITLE... PAGE 1.0 GENERAL INFORMATION... 3 2.0 DEFINITIONS... 5 3.0 RELATED DOCUMENTS...
TITLE: QUALITY SYSTEM MANUAL Page 1 of 15 QUALITY SYSTEM MANUAL TITLE: QUALITY SYSTEM MANUAL Page 2 of 15 Index PARAGRAPH TITLE... PAGE 1.0 GENERAL INFORMATION... 3 2.0 DEFINITIONS... 5 3.0 RELATED DOCUMENTS...
ISO 9001:2015 Quality Management System. New/Revised Requirements
 ISO 9001:2015 New/Revised The Quality System Checklist is intended to only identify the new/revised requirements. ISO 9001:2015 requires the adoption of the process approach which extends to internal quality
ISO 9001:2015 New/Revised The Quality System Checklist is intended to only identify the new/revised requirements. ISO 9001:2015 requires the adoption of the process approach which extends to internal quality
Quality Management System MANUAL. SDIX, LLC Headquarters: 111 Pencader Drive Newark, Delaware 19702
 Quality Management System MANUAL SDIX, LLC Headquarters: 111 Pencader Drive Newark, Delaware 19702 Doc. No. G5500 Rev. 11 Status : APPROVED Effective: 09/07/2017 Page 2 of 32 Quality Manual Table of Contents
Quality Management System MANUAL SDIX, LLC Headquarters: 111 Pencader Drive Newark, Delaware 19702 Doc. No. G5500 Rev. 11 Status : APPROVED Effective: 09/07/2017 Page 2 of 32 Quality Manual Table of Contents
RELATIONSHIP CLIENT IMPROVEMENT LEADERSHIP PROCESSES INFORMATION PEOPLE
 RELATIONSHIP CLIENT IMPROVEMENT LEADERSHIP PROCESSES INFORMATION PEOPLE Created by Richard Kill Analyzed by Olcinio Santos Vieira Approved by Gustavo Carvalhinho Term 2017 Revision 00 Updated May/10/2017
RELATIONSHIP CLIENT IMPROVEMENT LEADERSHIP PROCESSES INFORMATION PEOPLE Created by Richard Kill Analyzed by Olcinio Santos Vieira Approved by Gustavo Carvalhinho Term 2017 Revision 00 Updated May/10/2017
Sections of the Standard. Evidence / Comments. (Y) / Nonconforming (NC)
 Date: Sections of the Standard 4 Context of the Organization 4.1 Understanding the organization and its context 4.2 Understanding the needs and expectations of interested parties 4.3 Determining the scope
Date: Sections of the Standard 4 Context of the Organization 4.1 Understanding the organization and its context 4.2 Understanding the needs and expectations of interested parties 4.3 Determining the scope
0. 0 TABLE OF CONTENTS
 QUALITY MANUAL Conforming to ISO 9001:2008 0. 0 TABLE OF CONTENTS Section Description ISO 9001 Clause Page 0 TABLE OF CONTENTS n/a 2 1 PIMA VALVE, INC. DESCRIPTION n/a 3 2 QUALITY MANUAL DESCRIPTION 4.2.2
QUALITY MANUAL Conforming to ISO 9001:2008 0. 0 TABLE OF CONTENTS Section Description ISO 9001 Clause Page 0 TABLE OF CONTENTS n/a 2 1 PIMA VALVE, INC. DESCRIPTION n/a 3 2 QUALITY MANUAL DESCRIPTION 4.2.2
Machined Integrations, LLC
 QUALITY MANUAL Machined Integrations, LLC ISO9001: 2008 Electronically Controlled by Quality Representative, Rev2, January 2014 Page 2 of 25 TABLE OF CONTENTS SECTION ELEMENT PAGE No A Revision and Approval
QUALITY MANUAL Machined Integrations, LLC ISO9001: 2008 Electronically Controlled by Quality Representative, Rev2, January 2014 Page 2 of 25 TABLE OF CONTENTS SECTION ELEMENT PAGE No A Revision and Approval
AB. OUR ISO CONFORMANCE AUDIT QUESTIONNAIRES 8. ASSESS HOW WELL YOU CONFORM TO ISO S REMEDIAL REQUIREMENTS
 8.1 PLANNING REQUIREMENTS 1 Do you plan monitoring, measurement, and analytical processes? 2 Do you plan how monitoring will be used to ensure conformity and effectiveness? 3 Do you plan how it will be
8.1 PLANNING REQUIREMENTS 1 Do you plan monitoring, measurement, and analytical processes? 2 Do you plan how monitoring will be used to ensure conformity and effectiveness? 3 Do you plan how it will be
Quality Management in the ART Laboratory Everyone s Responsibility
 Quality Management in the ART Laboratory Everyone s Responsibility Sandro Esteves, MD, PhD Medical and Scientific Director of ANDROFERT Andrology and Human Reproduction Clinic Campinas, Brazil Learning
Quality Management in the ART Laboratory Everyone s Responsibility Sandro Esteves, MD, PhD Medical and Scientific Director of ANDROFERT Andrology and Human Reproduction Clinic Campinas, Brazil Learning
SKYCOAT, LLC QUALITY CONTROL MANUAL
 QUALITY MANUAL SKYCOAT, LLC QUALITY CONTROL MANUAL Rev. 4 Rev. 2 1 TABLE OF CONTENTS Introduction 1 Organization 1.1 Scope 1.2 Scope Description 1.2.1 Scope Exclusions 1.2.2 Normative References 2.0 Terms
QUALITY MANUAL SKYCOAT, LLC QUALITY CONTROL MANUAL Rev. 4 Rev. 2 1 TABLE OF CONTENTS Introduction 1 Organization 1.1 Scope 1.2 Scope Description 1.2.1 Scope Exclusions 1.2.2 Normative References 2.0 Terms
Atlantic Technical Systems, Inc. 415 Headquarters Drive # 2 Millersville, MD USA Office: (410) Fax: (410)
 ISO 9001 Page: 1 of 29 Quality Assurance Manual ISO 9001:2008 Atlantic Technical Systems, Inc. 415 Headquarters Drive # 2 Millersville, MD 21108 USA Office: (410) 507-2779 Fax: (410) 451-9609 APPROVALS
ISO 9001 Page: 1 of 29 Quality Assurance Manual ISO 9001:2008 Atlantic Technical Systems, Inc. 415 Headquarters Drive # 2 Millersville, MD 21108 USA Office: (410) 507-2779 Fax: (410) 451-9609 APPROVALS
Quality Manual. This manual complies with the requirements of the ISO 9001:2015 International Standard.
 Quality Manual This manual complies with the requirements of the ISO 9001:2015 International Standard. Northeast Power Systems, Inc. 66 Carey Road Queensbury, New York 12804 Quality Manual Rev 0 Printed
Quality Manual This manual complies with the requirements of the ISO 9001:2015 International Standard. Northeast Power Systems, Inc. 66 Carey Road Queensbury, New York 12804 Quality Manual Rev 0 Printed
QUALITY ASSURANCE GUIDE FOR SUPPLIERS
 QUALITY ASSURANCE GUIDE FOR SUPPLIERS Version of 01/03/2017 Index INDEX 2 INTRODUCTION 3 GOAL 3 1. Request for Quotation RFQ 4 2. Selection and evaluation 5 3. Development of New Products 7 3.1. Inputs
QUALITY ASSURANCE GUIDE FOR SUPPLIERS Version of 01/03/2017 Index INDEX 2 INTRODUCTION 3 GOAL 3 1. Request for Quotation RFQ 4 2. Selection and evaluation 5 3. Development of New Products 7 3.1. Inputs
ISO Food Safety Management System Compliance Summary
 ISO 22000 Clause ISO 9001 Clause FSMS Manual Reference Policy / Procedure Title 4. Quality Management System (ISO 9001) 4. Food Safety Quality Management System (ISO 22000) 4.1 General Requirements 4.1
ISO 22000 Clause ISO 9001 Clause FSMS Manual Reference Policy / Procedure Title 4. Quality Management System (ISO 9001) 4. Food Safety Quality Management System (ISO 22000) 4.1 General Requirements 4.1
QUALITY MANUAL. This document defines the requirements, processes, structure and documentation for the Teledyne DGO Quality Management System.
 Page 1 of 13 1. PURPOSE This document defines the requirements, processes, structure and documentation for the Teledyne DGO Quality Management System. 2. SCOPE The Teledyne DGO Quality Management System
Page 1 of 13 1. PURPOSE This document defines the requirements, processes, structure and documentation for the Teledyne DGO Quality Management System. 2. SCOPE The Teledyne DGO Quality Management System
TL 9000 R6.0 REQUIREMENTS
 Logo or heading here TL 9000 R6.0 REQUIREMENTS Nancy Patterson Nokia Tom Yohe - Consultant TL 9000 CONTAINS ALL OF THE AUDITABLE CLAUSES OF ISO 9001:2008 http://tl9000.org/alerts/alerts.html#10 QUEST FORUM
Logo or heading here TL 9000 R6.0 REQUIREMENTS Nancy Patterson Nokia Tom Yohe - Consultant TL 9000 CONTAINS ALL OF THE AUDITABLE CLAUSES OF ISO 9001:2008 http://tl9000.org/alerts/alerts.html#10 QUEST FORUM
Correlation Matrix & Change Summary
 The correlation matrix compares the new requirements of ISO 9001:2015 to the requirements of ISO 9001:2008, and provides a summary of the changes. Correlation Matrix & Change Summary Introduction Correlation
The correlation matrix compares the new requirements of ISO 9001:2015 to the requirements of ISO 9001:2008, and provides a summary of the changes. Correlation Matrix & Change Summary Introduction Correlation
QUALITY MANAGEMENT SYSTEM QUALITY MANUAL ISO 9001:2008
 QUALITY MANAGEMENT SYSTEM QUALITY MANUAL ISO 9001:2008 EXPRESS CONTRACTING SERVICES PTY LTD TRADING AS GOLDEN BROWN CLEANING SERVICES Unit 8/217 Mickleham Road Tullamarine VIC 3043 www.goldenbrown.com.au
QUALITY MANAGEMENT SYSTEM QUALITY MANUAL ISO 9001:2008 EXPRESS CONTRACTING SERVICES PTY LTD TRADING AS GOLDEN BROWN CLEANING SERVICES Unit 8/217 Mickleham Road Tullamarine VIC 3043 www.goldenbrown.com.au
ISO 9001 Quality Management Systems
 ISO 9001 Quality Management Systems INFORMATION GUIDE ISO 9001 Background ISO 9001:2015 is the world s foremost quality management standard, used by hundreds of thousands of organisations in over 170 countries
ISO 9001 Quality Management Systems INFORMATION GUIDE ISO 9001 Background ISO 9001:2015 is the world s foremost quality management standard, used by hundreds of thousands of organisations in over 170 countries
ISO 9001: 2015 Quality Management System Certification. Awareness Training
 ISO 9001: 2015 Quality Management System Certification Awareness Training ISO 9001: 2015 STRUCTURE The new standard is modeled around the ISO Directive Annex SL, a high level structure (HSL) based on the
ISO 9001: 2015 Quality Management System Certification Awareness Training ISO 9001: 2015 STRUCTURE The new standard is modeled around the ISO Directive Annex SL, a high level structure (HSL) based on the
EUROPASS DIPLOMA SUPPLEMENT
 EUROPASS DIPLOMA SUPPLEMENT TITLE OF THE DIPLOMA (ES) Técnico Superior en Administración y Finanzas TRANSLATED TITLE OF THE DIPLOMA (EN) (1) Higher Technician in Administration and Finance ------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------
EUROPASS DIPLOMA SUPPLEMENT TITLE OF THE DIPLOMA (ES) Técnico Superior en Administración y Finanzas TRANSLATED TITLE OF THE DIPLOMA (EN) (1) Higher Technician in Administration and Finance ------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------
Rockwell Automation Quality Management System Overview and Answers
 Rockwell Automation Quality and s Quality and s 1.0 PURPOSE To demonstrate and provide a public summary of the top level Rockwell Automation (RA) Quality (QMS) and RA internal documents (primarily the
Rockwell Automation Quality and s Quality and s 1.0 PURPOSE To demonstrate and provide a public summary of the top level Rockwell Automation (RA) Quality (QMS) and RA internal documents (primarily the
Position Description
 Position Description Position: Reports To: Direct Reports: Chief Executive Officer IFL- 247 Board of Directors Corporate Services Manager Health Services Manager Senior Medical Officer Purpose of the Role
Position Description Position: Reports To: Direct Reports: Chief Executive Officer IFL- 247 Board of Directors Corporate Services Manager Health Services Manager Senior Medical Officer Purpose of the Role
ISO/DIS9001:2008 VS ISO9001:2000. Speaker: Ir. Dr. Aaron TONG Date:
 ISO/DIS9001:2008 VS ISO9001:2000 Speaker: Ir. Dr. Aaron TONG Date:2008-08-01 1 Outline Background Information of ISO/DIS 9001 : 2008 Main Changes of 2008 Edition What we should do? Q & A Section 2 Ir.
ISO/DIS9001:2008 VS ISO9001:2000 Speaker: Ir. Dr. Aaron TONG Date:2008-08-01 1 Outline Background Information of ISO/DIS 9001 : 2008 Main Changes of 2008 Edition What we should do? Q & A Section 2 Ir.
25 D.L. Martin Drive Mercersburg, PA (717)
 QUALITY MANUAL D. L. MARTIN CO. 25 D.L. Martin Drive Mercersburg, PA 17236 (717) 328-2141 Revision 14 August 2012 Michael A. White Manager, QA & Engineering D.L. Martin Co. Quality Manual UNCONTROLLED
QUALITY MANUAL D. L. MARTIN CO. 25 D.L. Martin Drive Mercersburg, PA 17236 (717) 328-2141 Revision 14 August 2012 Michael A. White Manager, QA & Engineering D.L. Martin Co. Quality Manual UNCONTROLLED
Quality Manual QM-9001 Revision 25 May 2013
 Lion Precision 563 Shoreview Park Road Shoreview, MN 55126 Phone: (651) 484-6544 Fax: (651) 484-6824 info@lionprecision.com Quality Manual QM-9001 Revision 25 May 2013 Approved by: Don Martin, President
Lion Precision 563 Shoreview Park Road Shoreview, MN 55126 Phone: (651) 484-6544 Fax: (651) 484-6824 info@lionprecision.com Quality Manual QM-9001 Revision 25 May 2013 Approved by: Don Martin, President
Electronics. Document: QUALITY MANUAL. Revision: W. Date: 08/22/2016
 Document: QUALITY MANUAL Revision: W Date: 08/22/2016 Electronics Gowanda Electronics One Magnetics Parkway Gowanda, NY 14070 Ph: 716-532-2234 Fax: 716-532-2702 www.gowanda.com Table of Contents Table
Document: QUALITY MANUAL Revision: W Date: 08/22/2016 Electronics Gowanda Electronics One Magnetics Parkway Gowanda, NY 14070 Ph: 716-532-2234 Fax: 716-532-2702 www.gowanda.com Table of Contents Table
Quality Manual. Specification No.: Q Revision 07 Page 1 of 14
 Page 1 of 14 Quality Manual This Quality Manual provides the overall quality strategy and objectives of Pyramid Semiconductor s quality system. It is based on the requirements of ISO 9000. This manual
Page 1 of 14 Quality Manual This Quality Manual provides the overall quality strategy and objectives of Pyramid Semiconductor s quality system. It is based on the requirements of ISO 9000. This manual
Title: ISO 9001 Quality Manual Doc. No: CAP-1004 Page 1 of 22
 Doc. No: CAP-1004 Page 1 of 22 Doc. No: CAP-1004 Page 2 of 22 Table of Contents 1. ABOUT THE ORGANIZATION... 4 1.1. ORGANIZATIONAL STRUCTURE... 4 2. PURPOSE, SCOPE AND USERS... 5 3. TERMS AND DEFINITIONS...
Doc. No: CAP-1004 Page 1 of 22 Doc. No: CAP-1004 Page 2 of 22 Table of Contents 1. ABOUT THE ORGANIZATION... 4 1.1. ORGANIZATIONAL STRUCTURE... 4 2. PURPOSE, SCOPE AND USERS... 5 3. TERMS AND DEFINITIONS...
CRITERIA FOR EQUASS ASSURANCE (SSGI)
 CRITERIA FOR EQUASS ASSURANCE (SSGI) 2008 by European Quality for Social Services (EQUASS) All rights reserved. No part of this document may be reproduced in any form or by any means, electronic, mechanical,
CRITERIA FOR EQUASS ASSURANCE (SSGI) 2008 by European Quality for Social Services (EQUASS) All rights reserved. No part of this document may be reproduced in any form or by any means, electronic, mechanical,
ISO 9001 QUALITY MANUAL
 ISO 9001 QUALITY MANUAL Origination Date: 10/01/14 Document Identifier: AIF quality control manual Date: 10/01/14 Project: Customer review Document Status: Released Document Link: www.aeroindfast.com Abstract:
ISO 9001 QUALITY MANUAL Origination Date: 10/01/14 Document Identifier: AIF quality control manual Date: 10/01/14 Project: Customer review Document Status: Released Document Link: www.aeroindfast.com Abstract:
PRINTED COPY IS NOT CONTROLLED
 Quality Manual We Deliver Quality on Time Form 1217 (01.16.2018) Rev. 12 SOR Inc. sorinc.com SOR QUALITY MANUAL Page 1 of 18 Table of Contents Quality Manual 8701-001 Revision 12 Introduction....4 1.0
Quality Manual We Deliver Quality on Time Form 1217 (01.16.2018) Rev. 12 SOR Inc. sorinc.com SOR QUALITY MANUAL Page 1 of 18 Table of Contents Quality Manual 8701-001 Revision 12 Introduction....4 1.0
ISO 9001:2000 What does it mean in the supply chain?
 ISO 9001:2000 What does it mean in the supply chain? What is ISO 9001:2000? ISO 9001:2000 is an international standard that gives requirements for an organization s Quality Management System ( QMS ). It
ISO 9001:2000 What does it mean in the supply chain? What is ISO 9001:2000? ISO 9001:2000 is an international standard that gives requirements for an organization s Quality Management System ( QMS ). It
QMS Team: MR and all HODs (Internal Auditors) MR March 10. Quality policy Define quality policy The Steering committee Objectives and targets
 QMS Roles, Responsibility and Authority Process Clause Activities Records Required Responsibility Authority Deadline Clause 4: Process Development 4.1 Develop processes and sequence, operation controls
QMS Roles, Responsibility and Authority Process Clause Activities Records Required Responsibility Authority Deadline Clause 4: Process Development 4.1 Develop processes and sequence, operation controls
Technical Manufacturing Corporation (TMC)
 Revision: 21 Pages: 1 of 14 Date Issued: 05/11/2016 Technical Manufacturing Corporation (TMC) Quality Manual 15 Centennial Drive Peabody MA 01960 Revision: 21 Pages: 2 of 14 Date Issued: 05/11/2016 1.
Revision: 21 Pages: 1 of 14 Date Issued: 05/11/2016 Technical Manufacturing Corporation (TMC) Quality Manual 15 Centennial Drive Peabody MA 01960 Revision: 21 Pages: 2 of 14 Date Issued: 05/11/2016 1.
ISO 9001:2015. Quality Management Systems Documentation. Quality Manual / Documented Information. Document No. QM-001
 ISO 9001:2015 Quality Management Systems Documentation / Documented Information Document No. QM-001 Mailing Address: PO Box 149 Flippin, AR 72634 1-870-453-2261 www.microplastics.com 1 P a g e Table of
ISO 9001:2015 Quality Management Systems Documentation / Documented Information Document No. QM-001 Mailing Address: PO Box 149 Flippin, AR 72634 1-870-453-2261 www.microplastics.com 1 P a g e Table of
ISO 9001:2008 Quality Management System QMS Manual
 2501 Kutztown Road Reading, PA, 19605 Tel. 610-929-3330 Fax 610-921-6861 ISO 9001:2008 Quality Management System QMS Manual The information contained in this document is Fidelity Technologies Corporation
2501 Kutztown Road Reading, PA, 19605 Tel. 610-929-3330 Fax 610-921-6861 ISO 9001:2008 Quality Management System QMS Manual The information contained in this document is Fidelity Technologies Corporation
Quality Manual ISO 9001:2015 Quality Management System
 Quality management input comprises the standard requirements from ISO 9001:2015 which are deployed by our organization to achieve customer satisfaction through process control. Quality Manual ISO 9001:2015
Quality management input comprises the standard requirements from ISO 9001:2015 which are deployed by our organization to achieve customer satisfaction through process control. Quality Manual ISO 9001:2015
Quality Manual Revision: C Effective: 03/01/10
 TABLE OF CONTENTS DESCRIPTION SECTION PAGE INTRODUCTION 1.0 1 APPROVAL SIGNATURE PAGE 1.1 1 AMENDMENT RECORD 1.2 2 SCOPE 2.0 3 EXCLUSIONS 2.1 3 CORPORATE POLICY 3.0 3 QUALITY MANAGEMENT SYSTEM 4.0 4 GENERAL
TABLE OF CONTENTS DESCRIPTION SECTION PAGE INTRODUCTION 1.0 1 APPROVAL SIGNATURE PAGE 1.1 1 AMENDMENT RECORD 1.2 2 SCOPE 2.0 3 EXCLUSIONS 2.1 3 CORPORATE POLICY 3.0 3 QUALITY MANAGEMENT SYSTEM 4.0 4 GENERAL
INTERNATIONAL ORGANIZATION FOR MIGRATION. Keywords: internal audit, evaluation, investigation, inspection, monitoring, internal oversight
 INTERNATIONAL ORGANIZATION FOR MIGRATION Document Title: Charter of the Office of the Inspector General (OIG) Document Type: Instruction Character: Compliance with this Instruction is mandatory Control
INTERNATIONAL ORGANIZATION FOR MIGRATION Document Title: Charter of the Office of the Inspector General (OIG) Document Type: Instruction Character: Compliance with this Instruction is mandatory Control
FDA 21 CFR Part 820 vs. ISO 13485:2016 Comparison Table created by greenlight.guru
 FDA 21 CFR Part 820 vs. ISO 13485:2016 Comparison Table created by greenlight.guru FDA QSR (21 CFR Part 820) ISO 13485:2016 820.1 Scope 1 Scope 2 Normative References 820.3 Definitions 3 Terms and Definitions
FDA 21 CFR Part 820 vs. ISO 13485:2016 Comparison Table created by greenlight.guru FDA QSR (21 CFR Part 820) ISO 13485:2016 820.1 Scope 1 Scope 2 Normative References 820.3 Definitions 3 Terms and Definitions
ISO 9001 QUALITY MANUAL
 E-mail : info@pai-france.com A. GENERALITIES Page : 1 / 19 ISO 9001 QUALITY MANUAL Field of activity: Supply of equipment for the oil, gas, petrochemical, food and pharmaceutical industries VALIDATION
E-mail : info@pai-france.com A. GENERALITIES Page : 1 / 19 ISO 9001 QUALITY MANUAL Field of activity: Supply of equipment for the oil, gas, petrochemical, food and pharmaceutical industries VALIDATION
Supplier Management. hago Automotive Corp.
 Supplier Management of hago Automotive Corp. Change 24/11/2016 L. Bendel 24/11/2016 J. Blumenstock 9999/ / 00 1 of 13 Table of Contents Table of Contents... 2 Table of Figures... 3 Abbreviations... 3 1
Supplier Management of hago Automotive Corp. Change 24/11/2016 L. Bendel 24/11/2016 J. Blumenstock 9999/ / 00 1 of 13 Table of Contents Table of Contents... 2 Table of Figures... 3 Abbreviations... 3 1
Scientific Working Group on Digital Evidence
 SWGDE Framework of a Quality Management System for Digital and Multimedia Disclaimer: As a condition to the use of this document and the information contained therein, the SWGDE requests notification by
SWGDE Framework of a Quality Management System for Digital and Multimedia Disclaimer: As a condition to the use of this document and the information contained therein, the SWGDE requests notification by
EHQMS Manual & Policy Document
 Quality management input comprises the standard requirements from ISO 9001:2015 which are strategically deployed by our organization to achieve customer satisfaction through process control. Environmental
Quality management input comprises the standard requirements from ISO 9001:2015 which are strategically deployed by our organization to achieve customer satisfaction through process control. Environmental
Vendor Qualification Survey
 1200 West 96 th St Minneapolis, MN 55431 Ph: 952-888-7900 Fax: 952-888-2719 Vendor Qualification Survey Vendor Information Company Name: Date: Address: City: Phone Number: email address: Product or Service
1200 West 96 th St Minneapolis, MN 55431 Ph: 952-888-7900 Fax: 952-888-2719 Vendor Qualification Survey Vendor Information Company Name: Date: Address: City: Phone Number: email address: Product or Service
Quality Management Evaluation & Audit Policy
 Title Quality Management Evaluation & Audit policy Document ID Director Mark Reynolds Status Final Owner Neil McCrirrick Version 1.1 Author Mark Reynolds Version Date 07/11/2011 Quality Management Evaluation
Title Quality Management Evaluation & Audit policy Document ID Director Mark Reynolds Status Final Owner Neil McCrirrick Version 1.1 Author Mark Reynolds Version Date 07/11/2011 Quality Management Evaluation
QUALITY MANUAL RED CHECK DENOTES THIS IS A CONTROLLED COPY. APPROVED BY: (President) APPROVED BY: (Production Manager) APPROVED BY: (Quality Manager)
 PNC, Inc. QUALITY MANUAL, Rev. 6 Date: 11-10-08 Page 1 of 17 QUALITY MANUAL RED CHECK DENOTES THIS IS A CONTROLLED COPY APPROVED BY: (President) APPROVED BY: (Production Manager) APPROVED BY: (Quality
PNC, Inc. QUALITY MANUAL, Rev. 6 Date: 11-10-08 Page 1 of 17 QUALITY MANUAL RED CHECK DENOTES THIS IS A CONTROLLED COPY APPROVED BY: (President) APPROVED BY: (Production Manager) APPROVED BY: (Quality
Quality and Medical Devices: ISO 13485:2003
 Quality and Medical Devices: ISO 13485:2003 Quality Management System requirements for Medical Devices manufacturers O3 Enterprise s.r.l. ISO 13485 Quality Management Systems Medical Device O3 Enterprise
Quality and Medical Devices: ISO 13485:2003 Quality Management System requirements for Medical Devices manufacturers O3 Enterprise s.r.l. ISO 13485 Quality Management Systems Medical Device O3 Enterprise
Security Operations. BS EN ISO 9001: 2008 Issue 1.2: 21/10/2016. Quality Manual. Managing Director. Controlled / Uncontrolled when printed
 of Security Operations BS EN ISO 9001: 2008 : 21/10/2016 Manual No. 1 Issued to Managing Director Authorised By Date Issued 21/10/2016 Controlled / Uncontrolled when printed SECTION: 0 Index Page 2 Note
of Security Operations BS EN ISO 9001: 2008 : 21/10/2016 Manual No. 1 Issued to Managing Director Authorised By Date Issued 21/10/2016 Controlled / Uncontrolled when printed SECTION: 0 Index Page 2 Note
Quality Systems Manual
 Quality Systems Manual This manual was developed to support ISO 9001 elements 1445 Oakland Drive, San Jose, CA 95112-2702 - Tel: (408) 294-9897 - Fax: (408) 297-1540 http://www.gorillacircuits.com Reference
Quality Systems Manual This manual was developed to support ISO 9001 elements 1445 Oakland Drive, San Jose, CA 95112-2702 - Tel: (408) 294-9897 - Fax: (408) 297-1540 http://www.gorillacircuits.com Reference
ISO 9001 Auditing Practices Group. Aligning the QMS with the achievement of organizational and business success
 Slide 1 ISO 9001 Auditing Practices Group Guidance on: Effectiveness Aligning the QMS with the achievement of organizational and business success Slide 2 Business, Quality and Excellence Models and Tools
Slide 1 ISO 9001 Auditing Practices Group Guidance on: Effectiveness Aligning the QMS with the achievement of organizational and business success Slide 2 Business, Quality and Excellence Models and Tools
Quality Manual. Print Name Title Date Prepared by L Naughton QA Consultant 9 th April 09. Reviewed by Bernard Lennon Fire and Safety Officer
 Quality Manual Print Name Title Date Prepared by L Naughton QA Consultant 9 th April 09 Reviewed by Bernard Lennon Fire and Safety Officer 9 th April 09 Corporate Authorisation Joe Hoare Estates Officer
Quality Manual Print Name Title Date Prepared by L Naughton QA Consultant 9 th April 09 Reviewed by Bernard Lennon Fire and Safety Officer 9 th April 09 Corporate Authorisation Joe Hoare Estates Officer
AS9003A QUALITY MANUAL
 Your Logo AS9003A QUALITY MANUAL Origination Date: (month/year) Document Identifier: Date: Document Status: Document Link: AS9003A Quality Manual Latest Revision Date Draft, Redline, Released, Obsolete
Your Logo AS9003A QUALITY MANUAL Origination Date: (month/year) Document Identifier: Date: Document Status: Document Link: AS9003A Quality Manual Latest Revision Date Draft, Redline, Released, Obsolete
Quality Management System Manual
 This Page 1 of 47 of the Quality Management System Manual If issued as a controlled copy, the serial number of this copy is Quality Management System Manual Certified to AS9100 Revision C Printed copies
This Page 1 of 47 of the Quality Management System Manual If issued as a controlled copy, the serial number of this copy is Quality Management System Manual Certified to AS9100 Revision C Printed copies
ISO AUDIT PREPARATION KIT 2016
 ISO AUDIT PREPARATION KIT 2016 IQY Technical College Table of Contents ISO... 3 ISO 9001:2008 Fact Sheet... 6 Further Information... 6 Preparing for the ISO 9001:2008 Audit - 2016... 7 ISO 9001:2008 Definitions...
ISO AUDIT PREPARATION KIT 2016 IQY Technical College Table of Contents ISO... 3 ISO 9001:2008 Fact Sheet... 6 Further Information... 6 Preparing for the ISO 9001:2008 Audit - 2016... 7 ISO 9001:2008 Definitions...
BEGINNER S GUIDE TO ISO 9001 : Quality Management System Requirements Explained
 BEGINNER S GUIDE TO ISO 9001 : 2015 Quality Management System Requirements Explained What is ISO 9001 : 2015? Why use it? ISO 9001 is an internationally recognised standard in quality. It is a guide to
BEGINNER S GUIDE TO ISO 9001 : 2015 Quality Management System Requirements Explained What is ISO 9001 : 2015? Why use it? ISO 9001 is an internationally recognised standard in quality. It is a guide to
Quality Manual ISO 9001:2000
 Quality Manual ISO 9001:2000 Page 2 of 23 TABLE OF CONTENTS COVER PAGE...1 TABLE OF CONTENTS...2 SIGNATURES...3 QUALITY POLICY...4 INTRODUCTION...5 CORPORATE PROFILE...9 4.O QUALITY MANAGEMENT SYSTEM...10
Quality Manual ISO 9001:2000 Page 2 of 23 TABLE OF CONTENTS COVER PAGE...1 TABLE OF CONTENTS...2 SIGNATURES...3 QUALITY POLICY...4 INTRODUCTION...5 CORPORATE PROFILE...9 4.O QUALITY MANAGEMENT SYSTEM...10
ROTEK. IIInnInstI Instrument Corp. ISO 9001 Quality System Manual
 ROTEK IIInnInstI Instrument Corp. ISO 9001 Quality System Manual QSM900100-01 REV O MANUAL NO: 10 ISSUED TO: Rotek Web Site DATE OF ISSUE: 17 July 2002 APPROVALS: ORIGINATOR: Lawrence E. Weissbach Lawrence
ROTEK IIInnInstI Instrument Corp. ISO 9001 Quality System Manual QSM900100-01 REV O MANUAL NO: 10 ISSUED TO: Rotek Web Site DATE OF ISSUE: 17 July 2002 APPROVALS: ORIGINATOR: Lawrence E. Weissbach Lawrence
ISO 9001:2015 Readiness Review
 ISO 9001:2015 Readiness Review Company Name Address Certification No. Contact Name Job Title Telephone Email BSI is committed to ensuring a smooth assessment for all clients wishing to certify to ISO 9001:2015,
ISO 9001:2015 Readiness Review Company Name Address Certification No. Contact Name Job Title Telephone Email BSI is committed to ensuring a smooth assessment for all clients wishing to certify to ISO 9001:2015,
JDI Quality Assurance Guideline
 JDI Quality Assurance Guideline For Supplier, 2017 Japan Display Inc. 7/1/2017 - Table of Contents - 1. Introduction... - 7-1.1. Purpose of this Guideline...- 8-1.2. Structure of this Guideline...- 8-1.3.
JDI Quality Assurance Guideline For Supplier, 2017 Japan Display Inc. 7/1/2017 - Table of Contents - 1. Introduction... - 7-1.1. Purpose of this Guideline...- 8-1.2. Structure of this Guideline...- 8-1.3.
QUALITY MANAGEMENT SYSTEM QUALITY MANUAL
 AERO SUPPLY USA 21941 US HWY 19 NORTH Clearwater, FL. 33765 USA P.(727) 754-4915 F.(727) 754-4920 Website: www.aerosupplyusa.com Email: sales@aerosupplyusa.com QUALITY MANAGEMENT SYSTEM QUALITY MANUAL
AERO SUPPLY USA 21941 US HWY 19 NORTH Clearwater, FL. 33765 USA P.(727) 754-4915 F.(727) 754-4920 Website: www.aerosupplyusa.com Email: sales@aerosupplyusa.com QUALITY MANAGEMENT SYSTEM QUALITY MANUAL
Quality Manual. AARD Spring & Stamping Quality Manual
 Quality Manual Page 1 of 25 AARD Spring & Stamping QM/5.0 Title: QM 5.0 AARD Spring & Stamping Quality Manual Copy Approval Date: 1/27/2011 Reviewer(s): Sign / Print Title Date Dan Wheeler Document Originator
Quality Manual Page 1 of 25 AARD Spring & Stamping QM/5.0 Title: QM 5.0 AARD Spring & Stamping Quality Manual Copy Approval Date: 1/27/2011 Reviewer(s): Sign / Print Title Date Dan Wheeler Document Originator
Continual Improvement
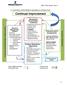 15. CONTINUAL IMPROVEMENT SEQUENCE & INTERACTION Continual Customer and Stakeholders Requirements Resource / Support Finance Document Control Information System Human Resources Competence Training Performance
15. CONTINUAL IMPROVEMENT SEQUENCE & INTERACTION Continual Customer and Stakeholders Requirements Resource / Support Finance Document Control Information System Human Resources Competence Training Performance
Pre Audit Transition Gap Analysis QMS (ISO 9001 Only)
 Pre Audit Transition Gap Analysis QMS (ISO 9001 Only) Company: Contact Name: Certification Number: Email: Contact Number: This document should be used in conjunction with the ISO 9001:2015 standard and
Pre Audit Transition Gap Analysis QMS (ISO 9001 Only) Company: Contact Name: Certification Number: Email: Contact Number: This document should be used in conjunction with the ISO 9001:2015 standard and
Transition Strategy for VDA 6.1, VDA 6.2 and VDA 6.4. Revision January Contents. 1. Foreword
 Transition Strategy for.1,.2 and.4 Revision 1.0 --- 06. January 2017 Contents 1. Foreword 2. Transition timing requirements to the relevant.x standards 3. Transition audit requirements to the relevant.x
Transition Strategy for.1,.2 and.4 Revision 1.0 --- 06. January 2017 Contents 1. Foreword 2. Transition timing requirements to the relevant.x standards 3. Transition audit requirements to the relevant.x
