Laboratory Study on Precipitation of Barium Sulphate in Malaysia Sandstone Cores
|
|
|
- Melanie Barrett
- 6 years ago
- Views:
Transcription
1 The Open Petroleum Engineering Journal, 2009, 2, - Open Access Laboratory Study on Precipitation of Barium Sulphate in Malaysia Sandstone Cores Amer Badr Bin Merdhah * and Abu Azam Mohd Yassin Faculty of Chemical and Natural Resources Engineering, Universiti Teknologi Malaysia. 830 Skudai, Johor, Malaysia Abstract: Scale formation is one of the most serious oil field problems that inflict water injection systems primarily when two incompatible waters are involved. Two waters are incompatible if they interact chemically and precipitate minerals when mixed. Due to the lack of reaction kinetics data, the rate of barium sulphate deposition in porous rock was measured through flooding sandstone core samples of uniform properties with supersaturated brine. The brine was formulated at the core inlet by mixing of injected sea water and formation water that contained high concentration of barium ion at various temperatures (50-80 C) and differential pressures ( psig). The rate of BaSO 4 scale formation was estimated by monitoring the core effluent s barium ion concentration. The solubility of barium sulphate scale formed and how its solubility was affected by changes in salinity and temperatures (40-90 C) were also studied. Scanning Electron Microscopy analysis was also used to examine the nature of scale deposition throughout the core. The results indicated increased rate of BaSO 4 precipitation at higher temperatures and greater brine super-saturation. The results were utilized to build a general reaction rate equation to predict BaSO 4 deposition in sandstone cores for a given temperature, brine super-saturation and differential pressures. Keywords: Scale deposition, scale solubility, concentration of barium ion, temperature and pressure effects. INTRODUCTION The injection of seawater into oilfield reservoirs to maintain reservoir pressure and improve secondary recovery is a well-established and mature operation. Moreover, the degree of risk posed by deposition of mineral scales to the injection and production wells during such operations has been much studied. Scale deposition is one of the most serious oil field problems that inflict water injection systems primarily when two incompatible waters are involved. Due to the limited availability of reaction kinetics data in the literature, especially for barium sulfate precipitation within porous media, this study was conducted to measure and model the rate of this reaction. Since this was intended to be the first in a series of progressively elaborate studies, investigation was focused on the brine s concentration and flow conditions rather than the porous medium. OILFIELD SCALE TYPES The most common oil field scales are listed in Table, along with the primary variables that affect their solubility []. These scales are sulfates such as calcium sulfate (anhydrite, gypsum), barium sulfate (barite), Strontium sulfate (celestite) and calcium carbonate. Other less common scales have also been reported such as iron oxides, iron sulfides and iron carbonate. Lead and zinc sulfide scale has recently become a concern in a number of North Sea oil and gas fields [2]. *Address correspondence to this author at the Faculty of Chemical and Natural Resources Engineering, Universiti Teknologi Malaysia. 830 Skudai, Johor, Malaysia; amer0227@yahoo.com SCALE DEPOSITION MECHANISMS Scale deposition in surface and subsurface oil and gas production equipment has been recognized. Scale deposition is one of the most important and serious problems that inflict oil field water injection systems. Scale limits and sometimes blocks oil and gas production by plugging the oilproducing formation matrix or fractures and perforated intervals. It can also plug production lines and equipment and impair fluid flow. Scale also deposited in down-hole pumps, tubing, casing flow-lines, heater treaters, tanks and other production equipment and facilities. The consequence could be production-equipment failure, emergency shutdown, increased maintenance cost, and overall decrease in production efficiency. In case of water injection systems, scale could plug the pores of the formation and results in injectivity decline with time [3-8]. Scale also can deposit when two incompatible waters are mixed and supersaturation is reached [3, 9-3]. SOURCE OF OIL FIELD SCALE The chief source of oil field scale is mixing of incompatible waters. Two waters are called incompatible if they interact chemically and precipitate minerals when mixed. A typical example of incompatible waters is sea water with high concentration of SO 4-2 and low concentrations of Ca +2, Ba +2 /Sr +2, and formation waters with very low concentrations of SO 4-2 but high concentrations of Ca +2, Ba +2 and Sr +2. Mixing of these waters, therefore, causes precipitation of CaSO 4, BaSO 4, and/or SrSO 4. Field produced water (disposal water) can also be incompatible with seawater. In cases where disposal water is mixed with seawater for re-injection, scale deposition is possible [4, 6, 7, 4, 5] / Bentham Open
2 2 The Open Petroleum Engineering Journal, 2009, Volume 2 Merdhah and Yassin Table. Most Common Oilfield Scales Name Chemical Formula Primary Variables Calcium Carbonate CaCO 3 Partial pressure of CO 2, temperature, total dissolved salts, ph Calcium Sulfate: Gypsum Hemihydrate Anhydrite CaSO 4.2H 2O CaSO 4.H 2O CaSO 4 Temperature, total dissolved salts, pressure Barium Sulfate BaSO 4 Temperature, pressure Strontium Sulfate SrSO 4 Temperature, pressure, total dissolved salts Iron Compounds: Ferrous Carbonate Ferrous Sulfide Ferrous Hydroxide Ferrous Hydroxide FeCO 3 FeS Fe(OH) 2 Fe(OH) 3 Corrosion, dissolved gases, ph SCALE FORMATION ALONG THE INJECTION- WATER PATH IN WATER-FLOOD OPERATIONS At the injection wellhead, injection water temperature is usually much lower than reservoir temperature. When it travels down the injection well-string, the water cools the surrounding formations, and its temperature and pressure increase. If the water is saturated at surface conditions with salts whose solubility decreases with increasing temperatures (e.g. anhydrite), scale may form along the well-string. Scale precipitation from the injection water may happen behind the mixing zone as a consequence of temperature and pressure changes. This is particularly true of waters containing salts whose solubility decreases with increasing temperature and decreasing pressure. Forward of the mixing zone only reservoir brine (with oil) is present in the rock pores. Behind the mixing zone, only injected water in equilibrium at local temperature and pressure (with residual oil) exists. In the mixing zone, precipitation of insoluble salts may occur due to the interaction, at local temperature and pressure, of chemical species contained in the injection water with chemical species present in the reservoir brine. Nevertheless, at a different pressure, the remaining clear water moves ahead mix again with reservoir brine and scale precipitation may again take place. This cycle is repeated until the remaining clear water reaches a production well. Pressure and temperature decrease along the flow string up to the surface in the production well, and further changes in thermodynamic conditions occur in the surface equipment. This may again result in scale formation. Normally, these scales do the most damage in the well-bore when there are major falls in pressure but hardly any temperature changes [6]. There are three principal mechanisms by which scales form in both offshore and onshore oil field system [7, 8]: a) Decrease in pressure and/or increase in temperature of a brine, goes to a reduction in the solubility of the salt (most commonly these lead to precipitation of carbonate scales, such as CaCO 3 ). Ca (HCO 3 ) 2 CaCO 3 + CO 2 + H 2 O () b) Mixing of two incompatible brines (most commonly formation water rich in cations such as barium, calcium and/or strontium, mixing with sulfate rich seawater, goes to the precipitation of sulfate scales, such as BaSO 4 ). Ba 2+ (or Sr 2+ or Ca ) + SO 4 BaSO 4 (or SrSO 4 or CaSO 4 ) (2) c) Other fluid incompatibilities include sulfide scale where hydrogen sulfide gas mixes with iron, zinc or lead rich formation waters: Zn 2+ + H 2 S ZnS + 2H 2+ (3) brine evaporation, resulting in salt concentration increasing above the solubility limit and goes to salt precipitation (as may occur in HP/HT gas wells where a dry gas stream may mix with a low rate brine stream resulting in dehydration and most commonly the precipitation of NaCl). THE SCALING PROBLEM IN OIL FIELDS A scale problem will occur, if at a high water cut part of the water is present as free water. The rate of scale deposition will then be approximately proportional to the rate of free water production. Depending upon where the formation water becomes supersaturated, scale may be deposited in the flow line only, in both flow line and tubing, and in some cases even in the perforations and in the formation near the wellbore. Scale formation is a major problem in the oil industry. They may occur down-hole or in surface facilities. The formations of these scales plug production lines and equipment and impair fluid flow. Their consequence could be production-equipment failure, emergency shutdown, increased maintenance cost, and an overall decrease in production efficiency. The failure of production equipment and instruments could result in safety hazards [9]. Many case histories of oil well scaling by calcium carbonate, calcium sulfate, strontium sulfate and barium sulfate have been reported [20-23]. Problems in connection to oil
3 Laboratory Study on Precipitation of Barium Sulphate in Malaysia The Open Petroleum Engineering Journal, 2009, Volume 2 3 well scaling in the Russia where scale has seriously plugged wells and are similar to cases in North Sea fields have been reported [20]. Oilfields scale problems have occurred because of water flooding in Saudi oil fields, Algeria, Indonesia in south Sumatra oilfields, and Egypt in el-morgan oilfield where calcium and strontium sulfate scales have been found in surface and subsurface production equipment [24]. SOLUBILITY OF SCALES Solubility is defined as the limiting amount of solute that can dissolve in a solvent under a given set of physical conditions. The chemical species of interest to us are present in aqueous solutions as ions. Certain combinations of these ions lead to compounds, which have low solubility. Once this capacity or solubility is exceeded the compounds precipitate from solution as solids. Therefore, precipitation of solid materials, which may form scale, will occur if: (i) The water contains ions, which are capable of forming compounds of limited solubility. (ii) There is a change in the physical conditions or water composition, lowering the solubility. Factors that affect scale precipitation, deposition and crystal growth can be summarized as: super-saturation, temperature, pressure, ionic strength, evaporation, contact time and ph. Effective scale control should be one of the primary objectives of any efficient water injection and normal production operation in oil and gas fields. Barium sulfate scale (barite) in oil fields can be precipitated easily on the basis of already available information relating to thermodynamic condition and the kinetics of precipitation [20, 25]. Barium sulfate solubility increased with temperature increase, with increase ionic strength of brine, and with pressure. Barium sulfate precipitation was affected most strongly by temperature []. REACTION KINETICS For a homogenous simple chemical reaction, A + B C The reaction rate (R) is defined as the change in the amount of a reaction per unit time per unit volume of reaction mixture. If the amount is measured in moles, then R becomes R = dc A dt = dc B dt = dc C dt where: C A, C B, and C C are the molar concentration (m) of species A, B and C, respectively. For a first order reaction, the rate of the reaction is proportional to the product of the concentrations of the reactants: R = K C A C B (4) where: K is the proportionality constant, also known as the constant reaction rate. Equation (4) is called the rate law equation for the reaction. The rate constant of most reactions is related to the absolute temperature by the Arrhenius equation: K = Ae E A RT where, A: frequency factor E A : reaction activation energy, J/mole R: Universal gas constant = 8.34 J mole - K - T: absolute temperature, ºK. If the Arrhenius equation applies, a plot of ln K versus /T should given a straight line of slope (-E/R) and intercept ln A. The frequency factor could depend on temperature, pressure and ionic strength of the solution. For BaSO 4 Precipitation Reaction: Ba SO 4 BaSO 4 Many rate laws have been proposed in the literature [26, 27]. But we used one of the rate laws was: K = RK sp / C Ba C SO4 (6) where: K: kinetic rate constant (m. min - ) R: rate of the BaSO 4 precipitation reaction (m. min - ) C Ba, C SO4: average steady-state concentrations of the ions across the core (m) K sp: solubility product of BaSO 4 in solution under the conditions of the reaction. MATERIALS AND METHODS Core Material In all flooding experiments, sandstone cores from Malaysia with 3inch length and of diameter inch with average porosity of 32% and of absolute permeability varied from md. No oil was present in the cores. All the cores were cleaned using methanol in Soxhlet extractor and dried in a Memmert Universal Oven at 00 C for overnight before use. Brines The ionic compositions of synthetic formation water and water injection (Angsi and Barton seawaters) are given in Table 2. Note the formation water has barium ion, and the sea water contains sulphate ion. It is clear that the mixing of these waters can lead to barium sulphates precipitation. Seven salts used for the preparation of synthetic formation water and water injections, the description of these salts are as follow: () Sodium Chloride grade (AR) NaCl (M.Wt. = g/mol, 99.8% purity) supplied by QReCTM. (2) Potassium Sulphate K 2 SO 4 (M.Wt. = g/mol, 99% purity) supplied by BHD chemicals Ltd Pool England. (3) Magnesium Chloride MgCl 2.6H 2 O (M.Wt. = g/mol, 98% purity) supplied by R&M Chemicals. (5)
4 4 The Open Petroleum Engineering Journal, 2009, Volume 2 Merdhah and Yassin (4) Calcium Chloride (dihydrate) grade (AR) CaCl 2.2H 2 O (M.Wt. = g/mol, 78% purity) supplied by QReCTM. (5) Sodium Bicarbonate NaHCO 3 (M.Wt. = 84.0 g/mol, 99.5% purity) supplied by GCE Laboratory Chemicals. (6) Strontium Chloride (6-hydrate) SrCl 2.6H 2 O (M.Wt. = g/mol, 99% purity) supplied by GCE Laboratory Chemicals. (7) Barium Chloride (dihydrate) grade (AR) BaCl 2.2H 2 O (M.Wt. = g/mol, 99% purity) supplied by QReCTM. Scaling Test Rig Experiments were carried out using a test rig, which is schematically shown in Fig. (). The core test equipment consists of five parts: constant pressure pump, transfer cell, oven, pressure transducer and core holder. There follows a brief description of each part. Constant pressure pump: Double-piston plunger pump manufactured by Lushyong Machiney Industry Limited, with.5 horse power motor, maximum design pressure of 35 bars and approximate flow rate of 20 L/min was used to inject the brines during flooding at different pressures. Transfer cell: Stainless steel transfer cell manufactured by TEMCO, Inc., USA which can withstand pressures up to 0,000 psia was used to store and pump the injected brine to the core holder. The cell with a capacity of 000 ml has a free-floating piston, which separates the pump fluid (distilled water) from the injection brine. The pump fluid was pumped into a transfer cell to displace the brine into the core. Oven: During all flooding runs, the core holder is placed inside a temperature controlled oven. Pressure transducer: The differential pressure across the core during flooding runs was measured by using a pressure transducer (model E B29) manufactured by Lushyong Machiney Industry Limited, with a digital display. Core holder: A Hassler type, stainless steel core holder designed for consolidated core samples, 3 inch length and inch diameter, was used. The holder was manufactured by TEMCO, Inc., USA and could withstand pressures up to 0,000 psia. A rubber sleeved core holder, subjected to an external confining pressure, into which a sandstone core is placed. TEST PROCEDURES Beaker Test The intent of this study was to determine solubility of barium sulphate scale from mixing synthetic brines (formation water and sea waters) at various temperatures 40 to 90 C. For each experiment of barium sulphate scale, 00 ml of each filtered opposite waters were poured simultaneously into a beaker. The synthetic brines were heated on hot plate and the solution was stirred by magnetic stirrer and after that the solution was filtered through 0.45-m filter paper. After filtration, 5 ml of the filtrate was taken into a 50 ml volumetric flask and was diluted with distilled water to make up to 50 ml of solution. This instantaneous dilution of BaSO 4 containing brines was performed in order to prevent BaSO 4 precipitation between filtering and analytical determination of the barium concentration. The barium determination was calibrated by measuring BaCl 2 standard solution. A barium concentration in the diluted filtrates was determined by atomic absorption spectrometry. After multiplying with the dilution factor, the exact concentration of barium was computed. Core Test Core Saturation: A schematic diagram of core saturation used in this study was shown in Fig. (2). Before each run, the core sample was dried in a Memmert Universal Oven at 00 C for overnight. The core sample was prepared for installation in the core-holder. A vacuum was drawn on the core sample for several hours to remove all air from the core. The core was saturated with formation water at room temperature. After the appearance of formation water at the outlet flooding was continued long enough to ensure 00% saturation. Flooding Experiment: As shown in Fig. (), the system consisting of the core holder assembly placed inside the oven and transfer cell containing sea water was then placed inside the water bath and heated to the desired temperature of the run. The required confining pressure was then adjusted to be approximately at double inlet pressure. A flooding run was Table 2. Ions of Synthetic Formation and Injection Waters Ionic Normal Barium Formation Water (ppm) High barium Formation Water (ppm) Barton Seawater (ppm) Angsi Seawater (ppm) Sodium 42,707 42,707 9,749 0,804 Potassium,972, Magnesium 02 02,060,295 Calcium Strontium Barium 250 2,200 <0.2 - Chloride 66,706 67,73 7,28 9,307 Sulphate 5 5 2,960 2,750 Bicarbonate 2,40 2,
5 Laboratory Study on Precipitation of Barium Sulphate in Malaysia The Open Petroleum Engineering Journal, 2009, Volume 2 5 Pressure Transducer Digital Readout Water Tank Oven S.W Valve Core Holder Pump Water Bath Transfer Cell Brine Collection To Nitrogen Cylinder Fig. (). Schematic of the core flooding apparatus. Pressure gage Water Tank F.W Core Holder Valve Vacuum pump Pump Air evacuation Fig. (2). Schematic of the core saturation apparatus. started by setting plunger pump at different pressures. Thus, the sea water was injected into the core and mixed with formation water inside porous media. The inlet pressure was measured by pressure transducer while the outlet pressure was atmospheric pressure. During each run, the flow rate across the core was recorded continuously and the permeability of core was calculated with Darcy s linear flow equation before and after scale deposition. scale deposition have been observed, the core sample was removed at the end of flooding then dried and cut into sections for scanning electron microscopy (SEM). RESULTS AND DISCUSSION Beaker Test The barium concentration in the diluted filtrates was determined by atomic absorption spectrometry. The solubility of BaSO 4 at various temperatures of this study was calculated. Graphical presentations are given in Fig. (3). Solubility of BaSO 4 (ppm) Ba = 250 ppm Temperature ( o C) Fig. (3). BaSO 4 solubility is dependent on temperature. The expected trend in this temperature range is an increase in BaSO 4 solubility because the dissociation of BaSO 4
6 6 The Open Petroleum Engineering Journal, 2009, Volume 2 Merdhah and Yassin is endothermic reaction. A graphical presentation of the experimental results (Fig. 3) illustrates this trend in these experiments. The sulphate ion content in the sea water brine was reacted with barium ion during heating. The more precipitation of BaSO 4 results from the presence of a large concentration of barium ion as compare to less precipitation at normal concentrations of barium ion. Core Test The main objective of this part of the investigation is to build a general reaction rate equation to predict BaSO 4 deposition in sandstone cores and study permeability reduction caused by BaSO 4 scale deposition in porous media. During each run, the flow rate across the core was recorded continuously and the permeability of core was calculated using Darcy s linear- flow equation. The flow rate decreased during the experiments only when a super-saturated solution was flowing through the cores. This confirms that the decrease of flow rate is due to precipitation of the barium sulphate inside the core with the consequent reduction in its permeability and porosity. In the following, extend of permeability damage and the results for various temperatures, differential pressure, and super-saturation are discussed: Extend of Permeability Reduction: Extend of permeability reduction caused by BaSO 4 scaling in the rock pores varied in different situations. Fig. (4a) shows the permeability change of a less damaged core at temperature (80 C) and differential pressure (00 psig); Fig. (4b) shows that of a severely damaged core after BaSO 4 scaling at temperature (50 C) and differential pressure (200 psig). About 5% - 3% permeability reduction is observed in Fig. (4a), but more than 9% - 9% initial permeability reduction could occur in a heavily scaled core, as Fig. (4b) indicates. The reduction in permeability is possibly caused by crystals blocking the pore throats as shown in the SEM view of Fig. (). The amount of precipitation varied within the sandstone cores, there being more scale near the formation water inlets and least scale was observed furthest from the inlet parts. Effect of Temperature: Temperature has a significant influence on solubility and crystal growth of barium sulphate. To study its effect on the reaction rate constant and permeability reduction, a number of runs were carried out where concentration of injected brine and differential pressure were kept constant and temperatures were varied from 50 to 80 C. Fig. (5) shows variation of permeability reduction with time at different temperatures. As temperature rises, the rate of nucleation and crystal growth and plugging are decreased. The permeability decline is less rapid at higher temperature, since the rate of BaSO 4 precipitation decrease with temperature. This is because the solubility of BaSO 4 increases with temperature. Fig. (6) shows variation of reaction rate constant with differential pressure at different temperatures. It also shows the effect of temperature on reaction rate constant. The reaction rate constant increases as the temperature is decreased. Effect of Differential Pressure: To investigate the effect of differential pressure on the reaction rate constant and permeability reduction a number of runs were carried out. In these experiments, the concentration of brine and temperature were kept constant and differential pressure varied from 00 Permeability ratio (kd/ki) Permeability ratio (Kd/ki) Ba = 250 ppm Ba = 250 ppm Fig. (4). Variation of permeability ratio as a function of time showing the effect of concentration at 00 psig and 80 C 200 psig and 50 C. to 200 psig. The variation of permeability reduction with time at different differential pressures is show in Fig. (7). From this figure, the permeability decline of porous medium is evident, even at such low differential pressures. The results illustrate that at low differential pressure, scale formation has already as significant effect on the permeability decline. As, the differential pressure was increased, the rate of permeability decline becomes more rapid. Moreover, at higher differential pressure more sulphate ions will pass through the porous medium in a given interval of time. Fig. (8) shows a variation of reaction rate constant with temperature at different differential pressures. This figure shows the effect of differential pressure on reaction rate constant. The reaction rate constant increases with increasing differential pressure. Effect of Super-Saturation: A number of runs were carried out to study the effect of barium and sulphate concentrations on the precipitation reaction. These runs were performed at differential pressure from 00 to 200 psig and temperatures of C with two different brine concentrations (see Table 2). Fig. (9) shows the increase in temperature causes a decrease in super-saturation, because the solubility of BaSO 4 increases with temperature. This must have
7 Laboratory Study on Precipitation of Barium Sulphate in Malaysia The Open Petroleum Engineering Journal, 2009, Volume 2 7 Permeability ratio (Kd/ki) Permeability ratio (Kd/ki) T = 70 C T = 70 C Fig. (5). Variation of permeability ratio as a function of time showing the effect of temperature at 00 psig and 200 psig. led to the permeability decline is less rapid at higher temperature, since the rate of BaSO 4 precipitation decrease with temperature. Scanning Electron Microscopy Analysis: The scaled core samples were examined by scanning electron microscopy (SEM) to observe the particle size and morphology of the precipitates. The formation of BaSO 4 during the flow of injection and formation waters in the porous media was observed by SEM micrographs. Fig. () shows the SEM image of the BaSO 4 scaling crystals in rock pores precipitated from mixed seawater with formation water inside the cores. The average size of BaSO 4 crystals precipitated from mixed brines was about 2.5 m. In all core tests, the abundance of scale reduced significantly from the front of the core to the rear indicating that scale formation in the porous media was rapid with the observation that the flow rate decreased soon after two incompatible waters were mixed into a core. In general, Fig. () indicates that the front sections of a core suffered considerable greater scaling damage. The reason the scaling decreased downstream of a core is clear, most of the scaling ions had deposited within the front sections as soon as they were mixed and left few ions to precipitate from the flow stream in the rear sections. Fig. (0) shows a SEM image of an unscaled core samples. Reaction Rate Constant (K) m min - Reaction Rate Constant (K) m min -.50E-02.00E E Fig. (6). Variation of reaction rate constant as a function of differential pressure showing the effect of temperature at a) Ba = 2200 ppm and b) Ba = 250 ppm. Rate Constant (K) Calculations Since barium concentration profile across the core is not available, the average reaction rate across the core is calculated by: Rate (R) = -d C Ba dt = C Ba t = C Ba out C Ba in t = C Ba in C Ba out (m min - ) t t is the residence time of the brine in the core as given by: t= V p Q 2.50E E-03.50E-03.00E E-04 T = 70 C Ba = 2200 T = 70 C Ba = 250 ppm Differential Pressure (psig) Differential Pressure (psig) where, Q: brine injection flow rate (m min - ) V p : pore volume of the core sample (ml) A total of 7 runs were performed, giving, e.g., the following data of run 6:
8 8 The Open Petroleum Engineering Journal, 2009, Volume 2 Merdhah and Yassin C Ba = m and t = min. Thus, R = Permeability ratio (Kd/ki) 5 5 = m min - ΔP = 50 psig Reaction Rate Constant (K) m min -.50E-02 ΔP = 50 psig.00e E Temperature (K) Permeability ratio (Kd/ki) 5 5 Fig. (7). Variation of permeability ratio as a function of time showing the effect of differential pressure at 50 C and 80 C. To compute the reaction rate constant for run 6, a rate law is employed; however, with average values of C Ba, C SO4, and K sp as demonstrated below using Equ. 6: K = RK sp / C Ba C SO4 ΔP = 50 psig where, C Ba, C SO4: average steady-state concentrations of the ions across the core (m). K sp: solubility product of BaSO 4 in solution under the conditions of the reaction (m). Given the following data for run 6: C Ba = ( )/ 2 = PPM = m C SO4 = ( )/ 2 = PPM = m (outlet value estimated from material balance) K sp = [Ca 2+ ] [SO 2-4 ] = * = m 2 The reaction rate constant becomes K = m min - Reaction Rate Constant (K) m min E E-03.50E-03.00E E-04 Ba = 250 ppm ΔP = 50 psig Temperature (K) Fig. (8). Variation of reaction rate constant as a function of temperature showing the effect of differential pressure at Ba = 2200 ppm and Ba = 250 ppm. Reaction Rate Constant (K) m min -.20E-02.00E E E E E-03 T = 70 C supersaturation Fig. (9). Reaction rate constant vs super-saturation. Kinetic Model The Arrhenius equation (Equ. 5) stipulates that K varies linearly with /T when all other reaction parameters are fixed. A plot of K (computed by Equ.6) versus /T for 7 runs reveals linear trends as shown in Fig. (2). The slope of lin-
9 Laboratory Study on Precipitation of Barium Sulphate in Malaysia The Open Petroleum Engineering Journal, 2009, Volume 2 9 ear fits indicates the reaction s activation energy (E A ) is kj /mol. Employing the Arrhenius equation with E A = kj/mol, values of A for those runs were computed and plotted in Fig. (3). The trend is described by: Fig. (0). SEM image of an unscaled sandstone core. A= 2*0 - *P.6567 (7) Combining Equs.5 and 7, the general equation for the constant reaction rate should have the following form: K = 2*0 - *P.6567 * e *T The values of kinetic rate constant for 7 runs were obtained by substituting the operating parameters of each run (P and T) into that equation. A plot of the K values predicted by Equ.8 versus the experimentally determined values is shown in Fig. (4). The points show absolute percent errors averaging 0.23%. (8) Fig. (). SEM image of BaSO 4 scales in sandstone cores. CONCLUSIONS The experimental results confirm the general trend in solubility dependencies for common oil field scales, determined at various temperatures. A temperature rise from 40 to 90 C causes an increase in BaSO 4 solubility Permeability decline caused by BaSO 4 scale formation in the porous media ranged from 5% to 9% of the initial permeability, depending on brine composition, initial permeability, temperature, differential pressure, and brine injection period. The pattern of permeability decline in a porous medium due to scaling injection was characterized by a concave curve with a steep initial decline which gradually slowed down to a lower. The initial
10 0 The Open Petroleum Engineering Journal, 2009, Volume 2 Merdhah and Yassin ln K (Reaction Rate Constant ) /Temperature (K - ) y = x R 2 = Fig. (2). Variation of rate constant with temperature. 2.00E-07 Pre exponential Factor A y = 2 E -x e-07 R 2 = E E Fig. (3). Pre-exponential factor (A) with differential pressure. Reaction Rate (K) from Model m min E E-03.50E-03.00E E-04 Differential Pressure (psig) y =.0548x R 2 = E-04.00E-03.50E E E-03 Reaction Rate (K) from Experimental Data m min - Fig. (4). Comparison between experimental and predicted kinetic rate constants for all 7 runs. steepness of these curves generally decreased with increasing distance from the point of mixing of the incompatible brines. The concave shape of the permeability-time curves was common to the majority of the porous medium flow tests. Several factors influencing scale formation had been examined. Increasing temperature, super-saturation, and differential pressure had a detrimental effect on the permeability reduction and constant reaction rate. The formation of BaSO 4 during flow of injection and formation waters in porous media have been proved by Scanning Electron Microscopy (SEM) micrographs show BaSO 4 crystals formation in porous space The constant reaction rate (K) varies with temperature according to Arrhenius equation. The reaction s activation energy was estimated at kj /mol. The following kinetic rate constant equation for BaSO 4 scale precipitation in sandstone cores fitted the experimental data rather well:
11 Laboratory Study on Precipitation of Barium Sulphate in Malaysia The Open Petroleum Engineering Journal, 2009, Volume 2 K = 2*0 - *P.6567 * e *T This equation yielded a mean absolute percent error of 0.23%. ACKNOWLEDGEMENTS The authors would like to thank the University Technology Malaysia and Mosti for paid fees to publish this paper. REFERENCES [] J. Moghadasi, M. Jamialahmadi, H. Muller-Steinhagen, A. Sharif, A. Ghalambor, R. M. Izadpanah, and E. Motaie, Scale Formation in Iranian Oil Reservoir and Production Equipment during Water Injection, The 5th international oilfield scale symposium and exhibition, January 29-30, Aberdeen, UK, SPE 80406, 2003, pp.-4. [2] R. I. Collins and M. M. Jordan, Occurrence, Prediction and Prevention of Zinc Sulfide Scale within Gulf Coast and North Sea High Temperature/High Salinity Production Wells, The SPE third international symposium on oilfield scale, January 30-3, Aberdeen, UK, SPE 6837, 200, pp.-7. [3] C. A. Todd and D. M. Yuan, Barium and Strontium Sulfate Solid- Solution Scale Formation at Elevated Temperatures, SPE Production Engineering, SPE 9762, pp , 992. [4] G.H. Bayona, A Review of Well Injectivity Performance in Saudi Arabia s Ghawar Field Seawater Injection Program, The SPE middle east oil technical conference and exhibition, April 3-6, Bahrain, SPE 2553, 993, pp [5] K. Asghari and R. Kharrat, Alteration of Permeability by Fine Particle Movement-A Water Injectivity Problem, The SPE international symposium on oilfield chemistry, February 4-7, San Antonio, Texas, SPE 29006, 995, pp [6] I. K. Andersen, E. Halvorsen, T. Saelensminde, and O. N. Ostbye, Water Management in a Closed Loop - Problems and Solutions at Brage Field, The SPE european petroleum conference, October 24-25, Paris, France, SPE 6562, 2000, pp. -2. [7] J. Paulo, J. E. Mackay, N. Menzies, and N. Poynton, Implications of Brine Mixing in the Reservoir for Scale Management in the Alba Field, The 200 SPE international symposium on oilfield scale, January 30-3, Aberdeen, UK, SPE 6830, 200, pp.-3. [8] I. A. Voloshin, V. V. Ragulin, E. N. Tyabayeva, I. I. Diakonov, and J. E. Mackay, Scaling Problems in Western Siberia, The SPE fifth international symposium on oilfield scale, January 29-30, Aberdeen, UK, SPE 80407, 2003, pp.-8. [9] M. Nassivera and A. Essel, Fateh field sea water injection Water Treatment, Corrosion, And scale control, The middle east oil technical conference of the society of petroleum Engineers, March 25-29, Manama, Bahrain, SPE 7765, 979, pp [0] A. P. Read and K. J. Ringen, The Use of Laboratory Tests to Evaluate Scaling Problems during Water Injection, The SPE sixth international symposium on oilfield and geothermal chemistry, January 25-27, Dallas, Texas, SPE0593, 983, pp [] J. O. Vetter, V. Kandarpa, and A. Harouaka, Prediction of Scale Problems Due To Injection of Incompatible Waters, J. Pet. Technol., pp , February 982. [2] J. Moghadasi, M. Jamialahmadi, H. Muller-Steinhagen, and A. Sharif, Scale Formation in Oil Reservoir and Production Equipment during Water Injection (Kinetics of CaSO 4and CaCO 3 Crystal Growth and Effect on Formation Damage), The SPE European Formation Damage Conference, May 3-4, Hague, Netherlands, SPE 82233, 2003, pp.-2. [3] J. Moghadasi, H. Muller-Steinhagen, M. Jamialahmadi, and A. Sharif, Model study on the kinetics of oil field formation damage due to salt precipitation from injection, J. Petrol. Sci. Eng., vol.43, pp , [4] P. Bedrikovistsky, D. Marchesin, F. Shecaira, L. A. Serra, A. Marchesin, E. Reszende, and G. Hime, Well Impairment During Sea/Produced Water Flooding: Treatment of Laboratory Data, The SPE Latin American and Caribbean petroleum engineering conference, March Buenos Aires, Argentina, SPE 69546, 200, pp.-9. [5] R. Stalker, R. I. Collins, and M. G. Graham, The Impact of Chemical Incompabilities in Commingled Fluids on the Efficiency of a Produced Water Reinjection System: A North Sea Example, The SPE international symposium on oilfield chemistry, February 5-7, Houston, Texas, SPE 80257, 2003, pp. -3. [6] C. Khelil, A. Harouaka, and A. Delhoume, Water Injection in Algeria-Problems and Solutions, The middle east oil technical conference of the Society of petroleum engineers, March 25-29, Manama, Bahrain: SPE 7762, 979, pp [7] J. E. Mackay, Scale Inhibitor Application in Injection Wells to Protect Against Damage to Production Wells, The SPE european formation damage conference, May Scheveningen, Netherlands, SPE 95022, 2005, pp.-9. [8] M. M. Jordan and J. E. Mackay, Integrated Field Development for Effective Scale Control Throughout the Water Cycle in Deep Water Subsea Fields, The SPE Europe/EAGE annual conference, June 3-6, Madrid, Spain, SPE 94052, 2005, pp.-9. [9] D. Y. Yeboah, K. S. Somuah, and R. M. Saeed, Anew and Reliable Model for Predicting Oilfield Scale Formation, The SPE international symposium on oilfield chemistry, March 2-5. New Orieans, USA., SPE 2566, 993, pp.-0. [20] W. R., Mitchell, M. D. Grist, and J. M. Boyle, Chemical Treatments Associated With North Sea Projects, J. Petrol. Technol., SPE 7880, pp , 980. [2] C. J. Lindlof, and G. K. Stoffer, A case study of sea water injection incompatibility, J. Pet. Technol., pp , July 983. [22] J. O. Vetter, A. W. Farone, E. Veith, and S. Lankford, Calcium Carbonate Scale Considerations: A Practical Approach, The SPE production technology symposium, November 6-7, Lubbock, Texas, SPE 7009, 987, pp.-4. [23] J. P. Shuler, A. E. Freitas, and A. K. Bowker, Selection and Application of BaSO 4 Scale Inhibitors for a CO 2 Flood, Rangely Weber Sand Unit, Colorado, Soc. Petrol. Eng., SPE 8973, pp. -8, 99. [24] I. M. El-Hattab, GUPCO S Experience in Treating Gulf of Suez Seawater for Waterflooding the El Morgan Oil Field, J. Petrol. Technol., pp , July 982. [25] H. G. Nancollas and T. S. Liu, Crystal Growth and Dissolution of Barium Sulfate, Soc. Petrol. Eng. J., pp , December 975. [26] P. G. Bedrikovistsky, R. P. Lopes, F. F. Rosario, M. C. Bezerra, and E. A. Lima, Oilfield scaling- part : mathematical and laboratory modelling, The SPE Latin American and Caribbean petroleum engineering conference., April 27-30, Trinidad, West Indies, SPE 827, 2003, pp. -3. [27] P. G. Bedrikovistsky, R. P. Lopes, F. F. Rosario, M. C. Bezerra, and E. A. Lima, Barium sulphate oilfield Scaling: Mathematical and laboratory Modelling, The SPE 6 th international symposium on oilfield scale, May 26-27, Aberdeen, UK, SPE 87457, 2004, pp. -3. Received: November 24, 2008 Revised: November 28, 2008 Accepted: December, 2008 Merdhah and Yassin; Licensee Bentham Open. This is an open access article licensed under the terms of the Creative Commons Attribution Non-Commercial License ( which permits unrestricted, non-commercial use, distribution and reproduction in any medium, provided the work is properly cited.
Scale Formation Due to Water Injection in Malaysian Sandstone Cores
 American Journal of Applied Sciences 6 (8): 1531-1538, 2009 ISSN 1546-9239 2009 Science Publications Scale Formation Due to Water Injection in Malaysian Sandstone Cores Amer Badr Bin Merdhah and Abu Azam
American Journal of Applied Sciences 6 (8): 1531-1538, 2009 ISSN 1546-9239 2009 Science Publications Scale Formation Due to Water Injection in Malaysian Sandstone Cores Amer Badr Bin Merdhah and Abu Azam
Determining the Rates for Scale Formation in Oil Wells
 ISSN : 8-96, Vol. 6, Issue 9, ( Part -1) September 016, pp.6-66 RESEARCH ARTICLE OPEN ACCESS Determining the Rates for Scale Formation in Oil Wells Otumudia Ephraim* 1 Ujile Awajiogak* * 1 (Department
ISSN : 8-96, Vol. 6, Issue 9, ( Part -1) September 016, pp.6-66 RESEARCH ARTICLE OPEN ACCESS Determining the Rates for Scale Formation in Oil Wells Otumudia Ephraim* 1 Ujile Awajiogak* * 1 (Department
Electrochemical impedance spectroscopy as a mean of measuring film thickness of calcium sulfate precipitate on an XC70 carbon steel
 International Letters of Chemistry, Physics and Astronomy Online: 2014-04-22 ISSN: 2299-3843, Vol. 32, pp 54-61 doi:10.18052/www.scipress.com/ilcpa.32.54 2014 SciPress Ltd., Switzerland Electrochemical
International Letters of Chemistry, Physics and Astronomy Online: 2014-04-22 ISSN: 2299-3843, Vol. 32, pp 54-61 doi:10.18052/www.scipress.com/ilcpa.32.54 2014 SciPress Ltd., Switzerland Electrochemical
SPE DISTINGUISHED LECTURER SERIES is funded principally through a grant of the SPE FOUNDATION
 SPE DISTINGUISHED LECTURER SERIES is funded principally through a grant of the SPE FOUNDATION The Society gratefully acknowledges those companies that support the program by allowing their professionals
SPE DISTINGUISHED LECTURER SERIES is funded principally through a grant of the SPE FOUNDATION The Society gratefully acknowledges those companies that support the program by allowing their professionals
ACIDIZING DOLOMITE RESERVOIRS USING HCL ACID PREPARED WITH SEAWATER: PROBLEMS AND SOLUTIONS. A Thesis DENNIS GEORGE ARENSMAN
 ACIDIZING DOLOMITE RESERVOIRS USING HCL ACID PREPARED WITH SEAWATER: PROBLEMS AND SOLUTIONS A Thesis by DENNIS GEORGE ARENSMAN Submitted to the Office of Graduate and Professional Studies of Texas A&M
ACIDIZING DOLOMITE RESERVOIRS USING HCL ACID PREPARED WITH SEAWATER: PROBLEMS AND SOLUTIONS A Thesis by DENNIS GEORGE ARENSMAN Submitted to the Office of Graduate and Professional Studies of Texas A&M
PHYSICOCHEMICAL CHARACTERIZATION AND ANALYSIS OF INJECTION WATER QUALITY DURING WATERFLOODING AT OFFSHORE PETROLEUM FACILITIES
 Int. J. Chem. Sci.: 14(4), 2016, 2867-2876 ISSN 0972-768X www.sadgurupublications.com PHYSICOCHEMICAL CHARACTERIZATION AND ANALYSIS OF INJECTION WATER QUALITY DURING WATERFLOODING AT OFFSHORE PETROLEUM
Int. J. Chem. Sci.: 14(4), 2016, 2867-2876 ISSN 0972-768X www.sadgurupublications.com PHYSICOCHEMICAL CHARACTERIZATION AND ANALYSIS OF INJECTION WATER QUALITY DURING WATERFLOODING AT OFFSHORE PETROLEUM
An experimental study of permeability determination in the lab
 Petroleum and Mineral Resources 221 An experimental study of permeability determination in the lab E. Lock, M. Ghasemi, M. Mostofi & V. Rasouli Department of Petroleum Engineering, Curtin University, Australia
Petroleum and Mineral Resources 221 An experimental study of permeability determination in the lab E. Lock, M. Ghasemi, M. Mostofi & V. Rasouli Department of Petroleum Engineering, Curtin University, Australia
Evaluation and prevention of formation damage in offshore sandstone reservoirs in China
 Pet.Sci.():- DOI./s--- Evaluation and prevention of formation damage in offshore sandstone reservoirs in China Yang Shenglai, Sheng Zhichao, Liu Wenhui, Song Zhixue, Wu Ming and Zhang Jianwei Key Laboratory
Pet.Sci.():- DOI./s--- Evaluation and prevention of formation damage in offshore sandstone reservoirs in China Yang Shenglai, Sheng Zhichao, Liu Wenhui, Song Zhixue, Wu Ming and Zhang Jianwei Key Laboratory
STUDYING THE EFFECT OF CALCIUM CARBONATE SCALE DEPOSIT ON CRUDE OIL FLOW CHARACTERISTICS
 STUDYING THE EFFECT OF CALCIUM CARBONATE SCALE DEPOSIT ON CRUDE OIL FLOW CHARACTERISTICS UKPAKA C.P., IMINABO J.T., AND IMINABO, M. ABSTRACT This paper describes detailed study of calcium carbonate scale
STUDYING THE EFFECT OF CALCIUM CARBONATE SCALE DEPOSIT ON CRUDE OIL FLOW CHARACTERISTICS UKPAKA C.P., IMINABO J.T., AND IMINABO, M. ABSTRACT This paper describes detailed study of calcium carbonate scale
Research Article Performance Comparison of New Combinations of Acids with Mud Acid in Sandstone Acidizing
 Research Journal of Applied Sciences, Engineering and Technology 7(2): 323-328, 2014 DOI:10.19026/rjaset.7.258 ISSN: 2040-7459; e-issn: 2040-7467 2014 Maxwell Scientific Publication Corp. Submitted: April
Research Journal of Applied Sciences, Engineering and Technology 7(2): 323-328, 2014 DOI:10.19026/rjaset.7.258 ISSN: 2040-7459; e-issn: 2040-7467 2014 Maxwell Scientific Publication Corp. Submitted: April
Aegis Tech Line Aegis Chemical Solutions Technical Newsletter Volume 07, September 2018
 SCALE IN OIL & GAS PRODUCTION Presented below are some definitions that apply to scale in oil & gas production. components to become supersaturated (equilibrium shift). Chemically, the following can be
SCALE IN OIL & GAS PRODUCTION Presented below are some definitions that apply to scale in oil & gas production. components to become supersaturated (equilibrium shift). Chemically, the following can be
Membrane Technology: From Manufacture to. May Production
 Membrane Technology: From Manufacture to May 2018 Production Osmosis Graphic pulled from https://earthobservatory.nasa.gov/features/water/page2.php Water Sources Surface Waters Ground Water Seawater Lakes
Membrane Technology: From Manufacture to May 2018 Production Osmosis Graphic pulled from https://earthobservatory.nasa.gov/features/water/page2.php Water Sources Surface Waters Ground Water Seawater Lakes
FORMATION DAMAGE DUE TO SCALE FORMATION IN POROUS MEDIA RESULTING WATER INJECTION
 Emirates Journal for Engineering Research, 3 (3), 69-79 (2008) (Regular Paper) FORMATION DAMAGE DUE TO SCALE FORMATION IN POROUS MEDIA RESULTING WATER INJECTION A.B. Bin Merdhah and A.M. Yassin Faculty
Emirates Journal for Engineering Research, 3 (3), 69-79 (2008) (Regular Paper) FORMATION DAMAGE DUE TO SCALE FORMATION IN POROUS MEDIA RESULTING WATER INJECTION A.B. Bin Merdhah and A.M. Yassin Faculty
Research Article Experimental Study on the Cause of Inorganic Scale Formation in the Water Injection Pipeline of Tarim Oilfield
 Chemistry, Article ID 619834, 4 pages http://dx.doi.org/10.1155/2014/619834 Research Article Experimental Study on the Cause of Inorganic Scale Formation in the Water Injection Pipeline of Tarim Oilfield
Chemistry, Article ID 619834, 4 pages http://dx.doi.org/10.1155/2014/619834 Research Article Experimental Study on the Cause of Inorganic Scale Formation in the Water Injection Pipeline of Tarim Oilfield
IN THE APPLICATION INVENTOR(S) IBNELWALEED A. HUSSEIN, MOHAMMED ABDULLAH HUSSEIN AL-YAARI, AND ABDELSALAM AL-SARKHI FOR AN
 Attorney Docket No. 35000.74 IN THE APPLICATION OF INVENTOR(S) IBNELWALEED A. HUSSEIN, MOHAMMED ABDULLAH HUSSEIN AL-YAARI, AND ABDELSALAM AL-SARKHI FOR AN ACID EMULSION FOR ACID TREATMENT OF OIL RESERVOIRS
Attorney Docket No. 35000.74 IN THE APPLICATION OF INVENTOR(S) IBNELWALEED A. HUSSEIN, MOHAMMED ABDULLAH HUSSEIN AL-YAARI, AND ABDELSALAM AL-SARKHI FOR AN ACID EMULSION FOR ACID TREATMENT OF OIL RESERVOIRS
1. Which of the given statements about the reaction below are incorrect?
 1. Which of the given statements about the reaction below are incorrect? 2PbO(s) + C(s) 2Pb(s) + CO 2 (g) a. Lead is getting reduced b. Carbon dioxide is getting oxidised c. Carbon is getting oxidised
1. Which of the given statements about the reaction below are incorrect? 2PbO(s) + C(s) 2Pb(s) + CO 2 (g) a. Lead is getting reduced b. Carbon dioxide is getting oxidised c. Carbon is getting oxidised
DEVELOPMENT OF A PHOSPHONATE BASED NANOFLUID FOR THE INHIBITION OF CALCIUM CARBONATE PRECIPITATION
 DEVELOPMENT OF A PHOSPHONATE BASED NANOFLUID FOR THE INHIBITION OF CALCIUM CARBONATE PRECIPITATION DESARROLLO DE UN NANOFLUIDO BASE FOSFONATO PARA LA INHIBICIÓN DE LA PRECIPITACIÓN DE CARBONATO DE CALCIO
DEVELOPMENT OF A PHOSPHONATE BASED NANOFLUID FOR THE INHIBITION OF CALCIUM CARBONATE PRECIPITATION DESARROLLO DE UN NANOFLUIDO BASE FOSFONATO PARA LA INHIBICIÓN DE LA PRECIPITACIÓN DE CARBONATO DE CALCIO
Predicting Injectivity Decline in Water Injection Wells by Upscaling On-Site Core Flood Data
 Predicting Injectivity Decline in Water Injection Wells by Upscaling On-Site Core Flood Data Paul Evans Oil Plus Ltd. Hambridge Road, Newbury Berkshire, RG14 5SS, England Fax: +44 (0) 1635 49618 E-mail:
Predicting Injectivity Decline in Water Injection Wells by Upscaling On-Site Core Flood Data Paul Evans Oil Plus Ltd. Hambridge Road, Newbury Berkshire, RG14 5SS, England Fax: +44 (0) 1635 49618 E-mail:
Well Stimulation and Sand Production Management (PGE 489 ) Sandstone Acidizing. By Dr. Mohammed A. Khamis
 Well Stimulation and Sand Production Management (PGE 489 ) Sandstone Acidizing By Dr. Mohammed A. Khamis 23-02-2016 Sandstones Acidizing The goal of sandstone matrix acidizing is to remove siliceous particles
Well Stimulation and Sand Production Management (PGE 489 ) Sandstone Acidizing By Dr. Mohammed A. Khamis 23-02-2016 Sandstones Acidizing The goal of sandstone matrix acidizing is to remove siliceous particles
Geological sequestration. or storage of CO 2
 Geological sequestration 10-100 mm or storage of CO 2 Porous matrix Berea sandstone Dmitriy Silin - UC Berkley, 2005 Fluids (oil, natural gas, CO 2 and/or water) are contained in tiny pore spaces in porous
Geological sequestration 10-100 mm or storage of CO 2 Porous matrix Berea sandstone Dmitriy Silin - UC Berkley, 2005 Fluids (oil, natural gas, CO 2 and/or water) are contained in tiny pore spaces in porous
SQUEEZING THE BAKKEN: SUCCESSFUL SQUEEZE PROGRAMS LEAD TO SHIFT CHANGE IN BAKKEN SCALE CONTROL
 SQUEEZING THE BAKKEN: SUCCESSFUL SQUEEZE PROGRAMS LEAD TO SHIFT CHANGE IN BAKKEN SCALE CONTROL WILLISTON BASIN PETROLEUM CONFERENCE KEVIN SPICKA, PHD May 3, 2017 1 Outline Background Product Qualification
SQUEEZING THE BAKKEN: SUCCESSFUL SQUEEZE PROGRAMS LEAD TO SHIFT CHANGE IN BAKKEN SCALE CONTROL WILLISTON BASIN PETROLEUM CONFERENCE KEVIN SPICKA, PHD May 3, 2017 1 Outline Background Product Qualification
The Pennsylvania State University. The Graduate School. Department of Energy and Mineral Engineering
 The Pennsylvania State University The Graduate School Department of Energy and Mineral Engineering ANALYSIS OF SCALING POTENTIALS IN MARCELLUS SHALE GAS WELLS A Thesis in Energy & Mineral Engineering by
The Pennsylvania State University The Graduate School Department of Energy and Mineral Engineering ANALYSIS OF SCALING POTENTIALS IN MARCELLUS SHALE GAS WELLS A Thesis in Energy & Mineral Engineering by
Gravimetric Analysis: Determination of % Sulfur in Fertilizer
 Gravimetric Analysis: Determination % Sulfur in Fertilizer This is another "real world" sample experiment in this case we will analyze a fertilizer sample for the sulfate content and express the result
Gravimetric Analysis: Determination % Sulfur in Fertilizer This is another "real world" sample experiment in this case we will analyze a fertilizer sample for the sulfate content and express the result
Chapter 17 Solubility and Complex Ion Equilibria
 Chem 1046 General Chemistry by Ebbing and Gammon, 8th Edition Prof George W.J. Kenney, Jr Last Update: 27Nov2008 Chapter 17 Solubility and Complex Ion Equilibria These Notes are to SUPPLIMENT the Text,
Chem 1046 General Chemistry by Ebbing and Gammon, 8th Edition Prof George W.J. Kenney, Jr Last Update: 27Nov2008 Chapter 17 Solubility and Complex Ion Equilibria These Notes are to SUPPLIMENT the Text,
THE EFFECT OF TEMPERATURE AND BRINE COMPOSITION ON THE MECHANICAL STRENGTH OF KANSAS CHALK
 SCA2008-55 1/6 THE EFFECT OF TEMPERATURE AND BRINE COMPOSITION ON THE MECHANICAL STRENGTH OF KANSAS CHALK Madland, M.V.*, Midtgarden, K*, Manafov, R.*, Korsnes, R.I.*, Kristiansen, T.G. BP Norway, Hiorth,
SCA2008-55 1/6 THE EFFECT OF TEMPERATURE AND BRINE COMPOSITION ON THE MECHANICAL STRENGTH OF KANSAS CHALK Madland, M.V.*, Midtgarden, K*, Manafov, R.*, Korsnes, R.I.*, Kristiansen, T.G. BP Norway, Hiorth,
EFFECT OF TEMPERATURE AND SOLUTION COMPOSITION ON THE PERMEABILITY OF ST. PETERS SANDSTONE: ROLE OF IRON (111)
 -31 6- EFFECT OF TEMPERATURE AND SOLUTON COMPOSTON ON THE PERMEABLTY OF ST. PETERS SANDSTONE: ROLE OF RON (111) J.M. Potter, A. Nur, Stanford Rock Physics Project W.E. Dibble Jr., Dept. of Geology, Stanford
-31 6- EFFECT OF TEMPERATURE AND SOLUTON COMPOSTON ON THE PERMEABLTY OF ST. PETERS SANDSTONE: ROLE OF RON (111) J.M. Potter, A. Nur, Stanford Rock Physics Project W.E. Dibble Jr., Dept. of Geology, Stanford
COPPER CYCLE EXPERIMENT 3
 COPPER CYCLE EXPERIMENT 3 INTRODUCTION One simple way to state the aim of chemistry is: The study of matter and its transformations. In this experiment, a copper sample will appear in five different forms
COPPER CYCLE EXPERIMENT 3 INTRODUCTION One simple way to state the aim of chemistry is: The study of matter and its transformations. In this experiment, a copper sample will appear in five different forms
Utilization of Calcium Carbonate (Clam Shell) Universities Research Journal 2013, Vol. 6
 Title All Authors Publication Type Publisher (Journal name, issue no., page no etc.) Abstract Keywords Utilization of Calcium Carbonate (Clam Shell) Aye Aye Mar Local Publication Universities Research
Title All Authors Publication Type Publisher (Journal name, issue no., page no etc.) Abstract Keywords Utilization of Calcium Carbonate (Clam Shell) Aye Aye Mar Local Publication Universities Research
Analysis Fraction Flow of Water versus Cumulative Oil Recoveries Using Buckley Leverett Method
 Analysis Fraction Flow of Water versus Cumulative Oil Recoveries Using Buckley Leverett Method Reza Cheraghi Kootiani, and Ariffin Bin Samsuri International Science Index, Physical and Mathematical Sciences
Analysis Fraction Flow of Water versus Cumulative Oil Recoveries Using Buckley Leverett Method Reza Cheraghi Kootiani, and Ariffin Bin Samsuri International Science Index, Physical and Mathematical Sciences
NIOR Core plug preparation procedures
 NIOR Core plug preparation procedures Introduction Reservoir rock state changed during sampling, mud contamination, storage and cleaning Representative wettability conditions in SCAL and EOR-experiments
NIOR Core plug preparation procedures Introduction Reservoir rock state changed during sampling, mud contamination, storage and cleaning Representative wettability conditions in SCAL and EOR-experiments
Solubility Equilibrium
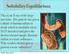 Solubility Equilibrium This is an X-ray of the large intestine. The patient was given a drink of barium sulfate to drink which is insoluble (does NOT dissolve) and gives the doctor a better image. Barium
Solubility Equilibrium This is an X-ray of the large intestine. The patient was given a drink of barium sulfate to drink which is insoluble (does NOT dissolve) and gives the doctor a better image. Barium
Compiled by Rahul Arora What do you mean by corrosion? How can you prevent it?
 Rahul Arora 12. What do you mean by corrosion? How can you prevent it? 13. MnO2 + 4HCl MnCl2 + 2H2O + Cl2 In the above equation, name the compound which is oxidized and which is reduced? 14. Match the
Rahul Arora 12. What do you mean by corrosion? How can you prevent it? 13. MnO2 + 4HCl MnCl2 + 2H2O + Cl2 In the above equation, name the compound which is oxidized and which is reduced? 14. Match the
Core Analysis of the Round Tank Queen Reservoir, Chaves County, New Mexico. By: Garrett Wilson
 Core Analysis of the Round Tank Queen Reservoir, Chaves County, New Mexico By: Garrett Wilson 1. Introduction 2. Apparatus 3. Procedure 4. Results and Calculations 1. Porosity 2. Permeability 3. Fluid
Core Analysis of the Round Tank Queen Reservoir, Chaves County, New Mexico By: Garrett Wilson 1. Introduction 2. Apparatus 3. Procedure 4. Results and Calculations 1. Porosity 2. Permeability 3. Fluid
Down Hole Flow Assurance
 Down Hole Flow Assurance Ferhat Erdal Senior Advisor - Flow Assurance Steve Cochran Senior Advisor - Flow Assurance SPE GCS Annual Drilling Symposium Spring, TX April 13, 2017 Flow Assurance Key Words
Down Hole Flow Assurance Ferhat Erdal Senior Advisor - Flow Assurance Steve Cochran Senior Advisor - Flow Assurance SPE GCS Annual Drilling Symposium Spring, TX April 13, 2017 Flow Assurance Key Words
FORMATION DAMAGE TESTS OF SOME COMPLETION FLUIDS
 FORMATION DAMAGE TESTS OF SOME COMPLETION FLUIDS Adel M. Hemeida, Ahmed A. Gawish King Saud University, Petroleum and Natural Gas Engineering, P.O. Box 800, Riyadh 11421, Saudi Arabia Phone 00966014676889
FORMATION DAMAGE TESTS OF SOME COMPLETION FLUIDS Adel M. Hemeida, Ahmed A. Gawish King Saud University, Petroleum and Natural Gas Engineering, P.O. Box 800, Riyadh 11421, Saudi Arabia Phone 00966014676889
INVESTIGATION OF CO- AND COUNTER CURRENT FLOW BEHAVIOR IN CARBONATE ROCK CORES
 SCA2017-061 1/8 INVESTIGATION OF CO- AND COUNTER CURRENT FLOW BEHAVIOR IN CARBONATE ROCK CORES Pouyan Ahmadi 1, Masoud Riazi 1, Mohammad Reza Malayeri 1 1 EOR Research Center, School of Chemical and Petroleum
SCA2017-061 1/8 INVESTIGATION OF CO- AND COUNTER CURRENT FLOW BEHAVIOR IN CARBONATE ROCK CORES Pouyan Ahmadi 1, Masoud Riazi 1, Mohammad Reza Malayeri 1 1 EOR Research Center, School of Chemical and Petroleum
Experiment 3: Determination of an Empirical Formula
 Background Information The composition of a compound is defined by its chemical formula, which gives the number ratio of the different elements in the compound. For example, water has a fixed composition
Background Information The composition of a compound is defined by its chemical formula, which gives the number ratio of the different elements in the compound. For example, water has a fixed composition
Modeling Mutual Solvent Preflush -The Case of Wettability
 Modeling Mutual Solvent Preflush -The Case of Wettability W. N. Aggrey S. Afari K. Sarkodie Petroleum Engineering Programme Department of Petroleum Engineering College of Engineering Kwame Nkrumah University
Modeling Mutual Solvent Preflush -The Case of Wettability W. N. Aggrey S. Afari K. Sarkodie Petroleum Engineering Programme Department of Petroleum Engineering College of Engineering Kwame Nkrumah University
Study on the Workover Fluid Formula and Performance of the Prevention Reservoir
 Study on the Workover Fluid Formula and Performance of the Prevention Reservoir Fengying Zhang, Jienian Yan & Zhiyong Li MOE Key Laboratory of Petroleum Engineering China University of Petroleum Beijing
Study on the Workover Fluid Formula and Performance of the Prevention Reservoir Fengying Zhang, Jienian Yan & Zhiyong Li MOE Key Laboratory of Petroleum Engineering China University of Petroleum Beijing
Heriot-Watt University
 Heriot-Watt University Heriot-Watt University Research Gateway Reservoir simulation and near-well bore modelling to aid scale management in a low temperature development with multilateral wells Ishkov,
Heriot-Watt University Heriot-Watt University Research Gateway Reservoir simulation and near-well bore modelling to aid scale management in a low temperature development with multilateral wells Ishkov,
1
 1 TENORM = Technologically Enhanced NORM. TENORM is encountered in the petroleum & gas industry. Enhanced levels of natural radioactivity in scale, sand and sludge of oil & gas production was first discovered
1 TENORM = Technologically Enhanced NORM. TENORM is encountered in the petroleum & gas industry. Enhanced levels of natural radioactivity in scale, sand and sludge of oil & gas production was first discovered
MultiScale Simulation Software
 MultiScale Simulation Software Simulation software for prediction of mineral scaling 2 MultiScale simulation software Over the years, oilfields will experience an increase in mineral scale production due
MultiScale Simulation Software Simulation software for prediction of mineral scaling 2 MultiScale simulation software Over the years, oilfields will experience an increase in mineral scale production due
Preparation of potassium nitrate from potassium chloride and magnesium nitrate in a laboratory scale using industrial raw materials
 Preparation of potassium nitrate from potassium chloride and magnesium nitrate in a laboratory scale using industrial raw materials Jana Jurišová a, Pavel Fellner a, Vladimír Danielik a, Marek Lencsés
Preparation of potassium nitrate from potassium chloride and magnesium nitrate in a laboratory scale using industrial raw materials Jana Jurišová a, Pavel Fellner a, Vladimír Danielik a, Marek Lencsés
Sodium Peroxides (Na 2 O 2 ): Preparation: It is formed by heating the metal in excess of air or oxygen at 300, which is free from
 S-Block Elements Generally one question was asked every year from this topic. This is completely theoretical and little memory based. Last minute revision generally helps. The general trends in the properties
S-Block Elements Generally one question was asked every year from this topic. This is completely theoretical and little memory based. Last minute revision generally helps. The general trends in the properties
A STUDY OF PROTECTIVE IRON CARBONATE SCALE FORMATION IN CO 2 CORROSION
 A STUDY OF PROTECTIVE IRON CARBONATE SCALE FORMATION IN CO CORROSION Wei Sun, Kunal Chokshi, Srdjan Nesic, and Daniel A. Gulino Institute for Corrosion and Multiphase Technology 4 West State Street Ohio
A STUDY OF PROTECTIVE IRON CARBONATE SCALE FORMATION IN CO CORROSION Wei Sun, Kunal Chokshi, Srdjan Nesic, and Daniel A. Gulino Institute for Corrosion and Multiphase Technology 4 West State Street Ohio
EXPERIMENTAL INVESTIGATION OF COOLING EFFECTS RESULTING FROM INJECTING HIGH PRESSURE LIQUID OR SUPERCRITICAL CO 2 INTO A LOW PRESSURE GAS RESERVOIR
 SCA28-4 1/12 EXPERIMENTAL INVESTIGATION OF COOLING EFFECTS RESULTING FROM INJECTING HIGH PRESSURE LIQUID OR SUPERCRITICAL CO 2 INTO A LOW PRESSURE GAS RESERVOIR Daniel R. Maloney and Marcos Briceno, ConocoPhillips
SCA28-4 1/12 EXPERIMENTAL INVESTIGATION OF COOLING EFFECTS RESULTING FROM INJECTING HIGH PRESSURE LIQUID OR SUPERCRITICAL CO 2 INTO A LOW PRESSURE GAS RESERVOIR Daniel R. Maloney and Marcos Briceno, ConocoPhillips
Water supplied by Marafiq does not meet the process requirements.
 WATER TREATMENT In Industries Water is used for: a. Drinking b. Cleaning c. Cooling d. Producing Steam e. Process requirement Why we need to treat water? For human consumption a. Water to be purified (Make
WATER TREATMENT In Industries Water is used for: a. Drinking b. Cleaning c. Cooling d. Producing Steam e. Process requirement Why we need to treat water? For human consumption a. Water to be purified (Make
MEASUREMENTS OF REMAINING OIL SATURATION IN MIXED-WET CARBONATES
 SCA2013-050 1/6 MEASUREMENTS OF REMAINING OIL SATURATION IN MIXED-WET CARBONATES Nayef Al-Ansi and Martin J. Blunt, Imperial College London This paper was prepared for presentation at the International
SCA2013-050 1/6 MEASUREMENTS OF REMAINING OIL SATURATION IN MIXED-WET CARBONATES Nayef Al-Ansi and Martin J. Blunt, Imperial College London This paper was prepared for presentation at the International
Chemical reactions and electrolysis
 Chemical reactions and electrolysis Higher Revision Questions Name: Class: Date: Time: 95 minutes Marks: 95 marks Comments: Page of 29 (a) Magnesium metal is shaped to make magnesium ribbon. Explain why
Chemical reactions and electrolysis Higher Revision Questions Name: Class: Date: Time: 95 minutes Marks: 95 marks Comments: Page of 29 (a) Magnesium metal is shaped to make magnesium ribbon. Explain why
Bucking the Industry Trend. Simple Change to Water System Saves New Jersey Manufacturer $23,000 Annually
 Simple Change to Water System Saves New Jersey Manufacturer by Kevin Preising Sales Engineer . Summary: Over the past two decades, the trend in the water treatment industry has been towards more and more
Simple Change to Water System Saves New Jersey Manufacturer by Kevin Preising Sales Engineer . Summary: Over the past two decades, the trend in the water treatment industry has been towards more and more
IOP Conference Series: Materials Science and Engineering PAPER OPEN ACCESS
 IOP Conference Series: Materials Science and Engineering PAPER OPEN ACCESS Enhanced oil recovery by nitrogen and carbon dioxide injection followed by low salinity water flooding for tight carbonate reservoir:
IOP Conference Series: Materials Science and Engineering PAPER OPEN ACCESS Enhanced oil recovery by nitrogen and carbon dioxide injection followed by low salinity water flooding for tight carbonate reservoir:
1. Scaling. H.O.: H-5/21, KRISHNA NAGAR, DELHI Tel.: , Fax:
 Boiler Water Problems and Its Causes Water is the essential medium for steam generation. Conditioning it properly can increase the efficiency of boiler and as well as extend the boiler s life. Treating
Boiler Water Problems and Its Causes Water is the essential medium for steam generation. Conditioning it properly can increase the efficiency of boiler and as well as extend the boiler s life. Treating
Total Dissolved Solids
 Total Dissolved Solids LabQuest 12 INTRODUCTION Solids are found in streams in two forms, suspended and dissolved. Suspended solids include silt, stirred-up bottom sediment, decaying plant matter, or sewage-treatment
Total Dissolved Solids LabQuest 12 INTRODUCTION Solids are found in streams in two forms, suspended and dissolved. Suspended solids include silt, stirred-up bottom sediment, decaying plant matter, or sewage-treatment
Effects of CO 2 and acid gas injection on enhanced gas recovery and storage
 J Petrol Explor Prod Technol (2013) 3:55 60 DOI 10.1007/s13202-012-0044-8 ORIGINAL PAPER - PRODUCTION ENGINEERING Effects of CO 2 and acid gas injection on enhanced gas recovery and storage Chawarwan Khan
J Petrol Explor Prod Technol (2013) 3:55 60 DOI 10.1007/s13202-012-0044-8 ORIGINAL PAPER - PRODUCTION ENGINEERING Effects of CO 2 and acid gas injection on enhanced gas recovery and storage Chawarwan Khan
1001 Nitration of toluene to 4-nitrotoluene, 2-nitrotoluene and 2,4-dinitrotoluene
 1001 Nitration of toluene to 4-nitrotoluene, 2-nitrotoluene and 2,4-dinitrotoluene CH 3 CH 3 CH 3 NO 2 CH 3 NO 2 HNO 3 /H 2 SO 4 + + + side products NO 2 NO 2 C 7 H 8 (92.1) HNO 3 (63.0) H 2 SO 4 (98.1)
1001 Nitration of toluene to 4-nitrotoluene, 2-nitrotoluene and 2,4-dinitrotoluene CH 3 CH 3 CH 3 NO 2 CH 3 NO 2 HNO 3 /H 2 SO 4 + + + side products NO 2 NO 2 C 7 H 8 (92.1) HNO 3 (63.0) H 2 SO 4 (98.1)
Duncan. UNIT 8 - Chemical Equations BALANCING EQUATIONS PRACTICE WORKSHEET 14.) C2H6 + O2 CO2 + H2O. 2.) Na + I2 NaI 3.) N2 + O2 N2O 4.
 BALANCING EQUATIONS PRACTICE WORKSHEET 1.) CH4 + O2 CO2 + H2O 2.) Na + I2 NaI 3.) N2 + O2 N2O 4.) N2 + H2 NH3 5.) KI + Cl2 KCl + I2 6.) HCl + Ca(OH)2 CaCl2 + H2O 7.) KClO3 KCl + O2 8.) K3PO4 + HCl KCl
BALANCING EQUATIONS PRACTICE WORKSHEET 1.) CH4 + O2 CO2 + H2O 2.) Na + I2 NaI 3.) N2 + O2 N2O 4.) N2 + H2 NH3 5.) KI + Cl2 KCl + I2 6.) HCl + Ca(OH)2 CaCl2 + H2O 7.) KClO3 KCl + O2 8.) K3PO4 + HCl KCl
PREPARATION & ANALYSIS OF AN IRON COORDINATION COMPOUND PART A: PREPARATION OF AN IRON COORDINATION COMPOUND
 Chemistry 112 PREPARATION & ANALYSIS OF AN IRON COORDINATION COMPOUND PART A: PREPARATION OF AN IRON COORDINATION COMPOUND A. INTRODUCTION In this experiment you will synthesize the iron coordination compound,
Chemistry 112 PREPARATION & ANALYSIS OF AN IRON COORDINATION COMPOUND PART A: PREPARATION OF AN IRON COORDINATION COMPOUND A. INTRODUCTION In this experiment you will synthesize the iron coordination compound,
Starting Out in the Oil Industry
 Starting Out in the Oil Industry DR. JAMIE KERR, CLARIANT OIL SERVICES Public Dr. Jamie Kerr 01.06.2015 2 Public, Starting Out in the Oil Industry Table of Contents Oil Recovery Problems (and Opportunities)
Starting Out in the Oil Industry DR. JAMIE KERR, CLARIANT OIL SERVICES Public Dr. Jamie Kerr 01.06.2015 2 Public, Starting Out in the Oil Industry Table of Contents Oil Recovery Problems (and Opportunities)
SILICA SCALE PREVENTION METHOD USING SEED MADE FROM GEOTHERMAL BRINE
 SILICA SCALE PREVENTION METHOD USING SEED MADE FROM GEOTHERMAL BRINE Hajime Sugita, Isao Matsunaga, Tsutomu Yamaguchi Geo-Energy Division, Geotechnology Department, National Institute for Resources and
SILICA SCALE PREVENTION METHOD USING SEED MADE FROM GEOTHERMAL BRINE Hajime Sugita, Isao Matsunaga, Tsutomu Yamaguchi Geo-Energy Division, Geotechnology Department, National Institute for Resources and
CARBONATION OF WOLLASTONITE USING SUPERCRITICAL CARBON DIOXIDE
 CARBONATION OF WOLLASTONITE USING SUPERCRITICAL CARBON DIOXIDE Clifford Y. Tai*, W.-R. Chen and Shin-Min Shih Department of Chemical Engineering National Taiwan University Email: cytai@ntu.edu.tw The feasibility
CARBONATION OF WOLLASTONITE USING SUPERCRITICAL CARBON DIOXIDE Clifford Y. Tai*, W.-R. Chen and Shin-Min Shih Department of Chemical Engineering National Taiwan University Email: cytai@ntu.edu.tw The feasibility
CHLOR-ALKALI INDUSTRY
 CHLOR-ALKALI INDUSTRY The chlor-alkali industry represents of three major industrial chemicals: Soda ash (sodium carbonate-na 2 CO 3 ) Caustic soda (sodium hydroxide-naoh) Chlorine (Cl 2 ) These chemicals
CHLOR-ALKALI INDUSTRY The chlor-alkali industry represents of three major industrial chemicals: Soda ash (sodium carbonate-na 2 CO 3 ) Caustic soda (sodium hydroxide-naoh) Chlorine (Cl 2 ) These chemicals
Membrane Desalination Technology
 Membrane Desalination Technology Desalination, or demineralization is a treatment process that removes salt and other minerals from brackish water and seawater to produce high quality drinking water. Various
Membrane Desalination Technology Desalination, or demineralization is a treatment process that removes salt and other minerals from brackish water and seawater to produce high quality drinking water. Various
Microbial Fluid-Rock Interactions in Chalk Samples and Salinity Factor in Divalent Ca 2+ ions Release for Microbial Enhanced Oil Recovery Purposes
 Microbial Fluid-Rock Interactions in Chalk Samples and Salinity Factor in Divalent Ca 2+ ions Release for Microbial Enhanced Oil Recovery Purposes Ismaila A. Jimoh, Svetlana N. Rudyk and Erik G. Søgaard
Microbial Fluid-Rock Interactions in Chalk Samples and Salinity Factor in Divalent Ca 2+ ions Release for Microbial Enhanced Oil Recovery Purposes Ismaila A. Jimoh, Svetlana N. Rudyk and Erik G. Søgaard
Solutions Unit Exam Name Date Period
 Name Date Period Ms. Roman Page 1 Regents Chemistry 1. Which mixture can be separated by using the equipment shown below? 6. Which ion, when combined with chloride ions, Cl, forms an insoluble substance
Name Date Period Ms. Roman Page 1 Regents Chemistry 1. Which mixture can be separated by using the equipment shown below? 6. Which ion, when combined with chloride ions, Cl, forms an insoluble substance
Equilibria of Slightly Soluble Ionic Compounds: We lied to you in chem 115
 Precipitation Reactions Predicting Whether a Precipitate Will Form Note the ions present in the reactants Consider all possible cation-anion combinations Use the solubility rules to decide whether any
Precipitation Reactions Predicting Whether a Precipitate Will Form Note the ions present in the reactants Consider all possible cation-anion combinations Use the solubility rules to decide whether any
Hard water. Hard and Soft Water. Hard water. Hard water 4/2/2012
 Hard and Soft Water is the type of water that has high mineral content (in contrast with soft water). minerals primarily consist of calcium (Ca 2+ ), and magnesium (Mg 2+ ) metal cations, and sometimes
Hard and Soft Water is the type of water that has high mineral content (in contrast with soft water). minerals primarily consist of calcium (Ca 2+ ), and magnesium (Mg 2+ ) metal cations, and sometimes
A New Approach to Simultaneously Enhancing Heavy Oil Recovery and Hindering Asphaltene Precipitation
 Iranian Journal of Oil & Gas Science and Technology, Vol. 1 (2012), No. 1, pp. 37-42 http://ijogst.put.ac.ir A New Approach to Simultaneously Enhancing Heavy Oil Recovery and Hindering Asphaltene Precipitation
Iranian Journal of Oil & Gas Science and Technology, Vol. 1 (2012), No. 1, pp. 37-42 http://ijogst.put.ac.ir A New Approach to Simultaneously Enhancing Heavy Oil Recovery and Hindering Asphaltene Precipitation
SPE Abstract. Copyright 2012, Society of Petroleum Engineers
 SPE 155124 Prediction of Mineral Scaling in a MEG Loop System of a Gas Production Offshore Baraka-Lokmane, S. 1, Hurtevent, C. 1, Ohanessian, J-L. 1, Rousseau, G. 1, Seiersten, M. 2, Deshmush, S. 3, 1
SPE 155124 Prediction of Mineral Scaling in a MEG Loop System of a Gas Production Offshore Baraka-Lokmane, S. 1, Hurtevent, C. 1, Ohanessian, J-L. 1, Rousseau, G. 1, Seiersten, M. 2, Deshmush, S. 3, 1
Evaluation copy. Total Dissolved Solids. Computer INTRODUCTION
 Total Dissolved Solids Computer 12 INTRODUCTION Solids are found in streams in two forms, suspended and dissolved. Suspended solids include silt, stirred-up bottom sediment, decaying plant matter, or sewage-treatment
Total Dissolved Solids Computer 12 INTRODUCTION Solids are found in streams in two forms, suspended and dissolved. Suspended solids include silt, stirred-up bottom sediment, decaying plant matter, or sewage-treatment
Improved Oil Recovery by Injection of Water and Gas
 Improved Oil Recovery by Injection of Water and Gas Dr. Amin Azhdarpour Department Of Petroleum Engineering, Marvdasht Branch, Islamic Azad University, Marvdasht, Iran. E-mail: aminazh22@gmail.com;amin.azhdarpour@miau.ac.ir
Improved Oil Recovery by Injection of Water and Gas Dr. Amin Azhdarpour Department Of Petroleum Engineering, Marvdasht Branch, Islamic Azad University, Marvdasht, Iran. E-mail: aminazh22@gmail.com;amin.azhdarpour@miau.ac.ir
EFFECT OFTHE TYPE OFCARBON MATERIALON THE REDUCTION KINETICS OF BARIUM SULFATE
 EFFECT OFTHE TYPE OFCARBON MATERIALON THE REDUCTION KINETICS OF BARIUM SULFATE M. Sh. Bafghi 1,*, A. Yarahmadi 2, A. Ahmadi 3 and H. Mehrjoo 3 * msbafghi@iust.ac.ir Received: April 2011 Accepted: July
EFFECT OFTHE TYPE OFCARBON MATERIALON THE REDUCTION KINETICS OF BARIUM SULFATE M. Sh. Bafghi 1,*, A. Yarahmadi 2, A. Ahmadi 3 and H. Mehrjoo 3 * msbafghi@iust.ac.ir Received: April 2011 Accepted: July
UTILIZATION OF CALCIUM SULFIDE DERIVED FROM WASTE GYPSUM BOARD FOR METAL-CONTAINING WASTEWATER TREATMENT
 Global NEST Journal, Vol 1, No 1, pp 11-17, 28 Copyright 28 Global NEST Printed in Greece. All rights reserved UTILIZATION OF CALCIUM SULFIDE DERIVED FROM WASTE GYPSUM BOARD FOR METAL-CONTAINING WASTEWATER
Global NEST Journal, Vol 1, No 1, pp 11-17, 28 Copyright 28 Global NEST Printed in Greece. All rights reserved UTILIZATION OF CALCIUM SULFIDE DERIVED FROM WASTE GYPSUM BOARD FOR METAL-CONTAINING WASTEWATER
FACTFILE: GCE CHEMISTRY
 FACTFILE: GCE CHEMISTRY 2.11 GROUP II ELEMENTS AND THEIR COMPOUNDS Learning Outcomes Students should be able to: 2.11.1 explain why these are regarded as s-block elements; 2.11.2 recall and explain the
FACTFILE: GCE CHEMISTRY 2.11 GROUP II ELEMENTS AND THEIR COMPOUNDS Learning Outcomes Students should be able to: 2.11.1 explain why these are regarded as s-block elements; 2.11.2 recall and explain the
5-4 Chemical changes Trilogy
 5-4 Chemical changes Trilogy.0 A student investigated the reaction of sodium carbonate with dilute hydrochloric acid. The student used the apparatus shown in Figure. Figure Sodium carbonate This is the
5-4 Chemical changes Trilogy.0 A student investigated the reaction of sodium carbonate with dilute hydrochloric acid. The student used the apparatus shown in Figure. Figure Sodium carbonate This is the
Trace Elements in. Elements. Trace Elements and Minor. Elements 2/25/2009
 Trace Elements in Carbonates A Short Course VU March, 2009 Peter Swart University of Miami Trace Elements and Minor Elements Minor Elements (Usually concentrations over 1000 ppm) Strontium Aragonite-7000
Trace Elements in Carbonates A Short Course VU March, 2009 Peter Swart University of Miami Trace Elements and Minor Elements Minor Elements (Usually concentrations over 1000 ppm) Strontium Aragonite-7000
New Mechanisms of Iron Sulfide Deposition during Acid Stimulation in Sour Gas Carbonate Reservoir
 New Mechanisms of Iron Sulfide Deposition during Acid Stimulation in Sour Gas Carbonate Reservoir Dr. Tao Chen, Dr. Qiwei Wang, and Dr. Fakuen F. Chang ABSTRACT (a) Iron sulfide deposition in sour gas
New Mechanisms of Iron Sulfide Deposition during Acid Stimulation in Sour Gas Carbonate Reservoir Dr. Tao Chen, Dr. Qiwei Wang, and Dr. Fakuen F. Chang ABSTRACT (a) Iron sulfide deposition in sour gas
1. The equation that represents the equilibrium in a saturated solution of Fe 2 (SO 4 ) 3 is.. The precipitate which forms first is
 CHEMISTRY 12 UNIT 3 - REVIEW Ksp Part A - Multiple Choice 1. The equation that represents the equilibrium in a saturated solution of Fe 2 (SO 4 ) 3 is A. Fe 2 (SO 4 ) 3 (s) 3 Fe 2+ (aq) + 2 SO 4 3 (aq)
CHEMISTRY 12 UNIT 3 - REVIEW Ksp Part A - Multiple Choice 1. The equation that represents the equilibrium in a saturated solution of Fe 2 (SO 4 ) 3 is A. Fe 2 (SO 4 ) 3 (s) 3 Fe 2+ (aq) + 2 SO 4 3 (aq)
EXPERIMENTAL INVESTIGATION OF THE IMPACT OF SALT PRECIPITATION ON CO2 INJECTIVITY
 SCA2017-044 1/9 EXPERIMENTAL INVESTIGATION OF THE IMPACT OF SALT PRECIPITATION ON CO2 INJECTIVITY Yen Adams Sokama-Neuyam, Jann Rune Ursin Department of Petroleum Engineering, University of Stavanger,
SCA2017-044 1/9 EXPERIMENTAL INVESTIGATION OF THE IMPACT OF SALT PRECIPITATION ON CO2 INJECTIVITY Yen Adams Sokama-Neuyam, Jann Rune Ursin Department of Petroleum Engineering, University of Stavanger,
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education
 UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education *7279389372* CHEMISTRY 06/06 Paper 6 Alternative to Practical October/November 07 1 hour Candidates
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education *7279389372* CHEMISTRY 06/06 Paper 6 Alternative to Practical October/November 07 1 hour Candidates
CO 2 -Brine Relative Permeability Characteristics of Low Permeable Sandstones in Svalbard
 SCA22-42 /6 CO 2 -Brine Relative Permeability Characteristics of Low Permeable Sandstones in Svalbard Raheleh Farokhpoor, Erik Lindeberg 2, Mai Britt E. Mørk, Ole Torsæter, NTNU, 2, SINTEF This paper was
SCA22-42 /6 CO 2 -Brine Relative Permeability Characteristics of Low Permeable Sandstones in Svalbard Raheleh Farokhpoor, Erik Lindeberg 2, Mai Britt E. Mørk, Ole Torsæter, NTNU, 2, SINTEF This paper was
Revolution. NON-Chemical Treatment. Technology. for Scale Control. 500 different Applications NON TOXIC FOR POLLUTION PREVENTION AND GREEN TECHNOLOGY
 Revolution of NON-Chemical Treatment Technology for Scale Control in 500 different Applications NON TOXIC FOR POLLUTION PREVENTION AND GREEN TECHNOLOGY FILTERSORB SP3 Table of contents 1. Glossary of terms
Revolution of NON-Chemical Treatment Technology for Scale Control in 500 different Applications NON TOXIC FOR POLLUTION PREVENTION AND GREEN TECHNOLOGY FILTERSORB SP3 Table of contents 1. Glossary of terms
Capture of Carbon Dioxide Using Aqueous Ammonia in a Lab-Scale Stirred-Tank Scrubber
 Advanced Materials Research Online: 204-06-8 ISSN: 662-8985, Vols. 955-959, pp 927-934 doi:0.4028/www.scientific.net/amr.955-959.927 204 Trans Tech Publications, Switzerland Capture of Carbon Dioxide Using
Advanced Materials Research Online: 204-06-8 ISSN: 662-8985, Vols. 955-959, pp 927-934 doi:0.4028/www.scientific.net/amr.955-959.927 204 Trans Tech Publications, Switzerland Capture of Carbon Dioxide Using
OXFORD CAMBRIDGE AND RSA EXAMINATIONS GCSE A172/02. TWENTY FIRST CENTURY SCIENCE CHEMISTRY A/ADDITIONAL SCIENCE A Modules C4 C5 C6 (Higher Tier)
 OXFORD CAMBRIDGE AND RSA EXAMINATIONS GCSE A172/02 TWENTY FIRST CENTURY SCIENCE CHEMISTRY A/ADDITIONAL SCIENCE A Modules C4 C5 C6 (Higher Tier) TUESDAY 10 JUNE 2014: Afternoon DURATION: 1 hour plus your
OXFORD CAMBRIDGE AND RSA EXAMINATIONS GCSE A172/02 TWENTY FIRST CENTURY SCIENCE CHEMISTRY A/ADDITIONAL SCIENCE A Modules C4 C5 C6 (Higher Tier) TUESDAY 10 JUNE 2014: Afternoon DURATION: 1 hour plus your
BENEFICIAL USE OF PRODUCED WATER SOUNDS SIMPLE ENOUGH. Rick McCurdy, Ground Water Protection Council s Annual UIC Conference February 21-23, 2017
 BENEFICIAL USE OF PRODUCED WATER SOUNDS SIMPLE ENOUGH Rick McCurdy, Ground Water Protection Council s Annual UIC Conference February 21-23, 2017 BUT FIRST A MOST PRECIOUS COMMODITY AGENDA What s In Produced
BENEFICIAL USE OF PRODUCED WATER SOUNDS SIMPLE ENOUGH Rick McCurdy, Ground Water Protection Council s Annual UIC Conference February 21-23, 2017 BUT FIRST A MOST PRECIOUS COMMODITY AGENDA What s In Produced
The study of local polymers on enhance oil recovery
 Available online atwww.scholarsresearchlibrary.com Archives of Applied Science Research, 2015, 7 (6):48-55 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-508X CODEN (USA) AASRC9 The study
Available online atwww.scholarsresearchlibrary.com Archives of Applied Science Research, 2015, 7 (6):48-55 (http://scholarsresearchlibrary.com/archive.html) ISSN 0975-508X CODEN (USA) AASRC9 The study
XII-EROSION-CORROSION MODELING AND EXPERIMENTS. Introduction
 EROSION-CORROSION MODELING AND EXPERIMENTS XII-1 XII-EROSION-CORROSION MODELING AND EXPERIMENTS Introduction In the oil and gas production industry, when sand is produced, carbon steel tubing and piping
EROSION-CORROSION MODELING AND EXPERIMENTS XII-1 XII-EROSION-CORROSION MODELING AND EXPERIMENTS Introduction In the oil and gas production industry, when sand is produced, carbon steel tubing and piping
NANDI NORTH DISTRICT JOINT MOCK EVALUATION TEST 2013
 NAME:. SIGNATURE: INDEX NO:. DATE :.. 233/1 CHEMISTRY PAPER 1 THEORY JULY / AUGUST 2013 TIME: 2 HOURS NANDI NORTH DISTRICT JOINT MOCK EVALUATION TEST 2013 Kenya Certificate of Secondary Education (K.C.S.E.)
NAME:. SIGNATURE: INDEX NO:. DATE :.. 233/1 CHEMISTRY PAPER 1 THEORY JULY / AUGUST 2013 TIME: 2 HOURS NANDI NORTH DISTRICT JOINT MOCK EVALUATION TEST 2013 Kenya Certificate of Secondary Education (K.C.S.E.)
Effect of ph and Slug Ratio of Alkaline Surfactant Polymer Alternating Gas Flooding on Oil Recovery
 Engineering and Technology 2016; 3(2): 47-52 http://www.aascit.org/journal/et ISSN: 2381-1072 (Print); ISSN: 2381-1080 (Online) Effect of ph and Slug Ratio of Alkaline Surfactant Polymer Alternating Gas
Engineering and Technology 2016; 3(2): 47-52 http://www.aascit.org/journal/et ISSN: 2381-1072 (Print); ISSN: 2381-1080 (Online) Effect of ph and Slug Ratio of Alkaline Surfactant Polymer Alternating Gas
Quantitive Chemistry Question paper
 Quantitive Chemistry Question paper Level Subject Exam Board Topic Sub-Topic Booklet GCSE Chemistry CCEA Quantitative Chemistry Quantitive Chemistry Question paper Time Allowed: 93 minutes Score: /77 Percentage:
Quantitive Chemistry Question paper Level Subject Exam Board Topic Sub-Topic Booklet GCSE Chemistry CCEA Quantitative Chemistry Quantitive Chemistry Question paper Time Allowed: 93 minutes Score: /77 Percentage:
Introduction. Hussein B. Ghalib 1 Inass Abdal Razaq Almallah 1
 https://doi.org/10.1007/s40808-017-0384-y ORIGINAL ARTICLE Scaling simulation resulting from mixing predicted model between Mishrif formation and different s injection in Basrah oil field, southern Iraq
https://doi.org/10.1007/s40808-017-0384-y ORIGINAL ARTICLE Scaling simulation resulting from mixing predicted model between Mishrif formation and different s injection in Basrah oil field, southern Iraq
Experiment 18 Determination of Iron by Visible Spectrophotometry
 Experiment 18 Determination of Iron by Visible Spectrophotometry PRELAB DISCUSSION Visible spectrophotometry (spectral analysis) is a method of measuring the concentration of colored solutions by the amount
Experiment 18 Determination of Iron by Visible Spectrophotometry PRELAB DISCUSSION Visible spectrophotometry (spectral analysis) is a method of measuring the concentration of colored solutions by the amount
NEW METHOD TO PREPARE OUTCROP CHALK CORES FOR WETTABILITY AND OIL RECOVERY STUDIES AT LOW INITIAL WATER SATURATION
 SCA2007-37 1/12 NEW METHOD TO PREPARE OUTCROP CHALK CORES FOR WETTABILITY AND OIL RECOVERY STUDIES AT LOW INITIAL WATER SATURATION Tina Puntervold, Skule Strand and Tor Austad University of Stavanger,
SCA2007-37 1/12 NEW METHOD TO PREPARE OUTCROP CHALK CORES FOR WETTABILITY AND OIL RECOVERY STUDIES AT LOW INITIAL WATER SATURATION Tina Puntervold, Skule Strand and Tor Austad University of Stavanger,
FilterSorb SP3 Treatment Method: Part II
 FilterSorb SP3 Treatment Method: Part II By Deepak Chopra WatchvWater GmbH Fahrlachstraße 14 68165 Mannheim Germany May 2013 Web: www.watchwater.de email: info@watchwater.de Telefon: +49 (0) 621 87951-0
FilterSorb SP3 Treatment Method: Part II By Deepak Chopra WatchvWater GmbH Fahrlachstraße 14 68165 Mannheim Germany May 2013 Web: www.watchwater.de email: info@watchwater.de Telefon: +49 (0) 621 87951-0
INDIAN SCHOOL MUSCAT SENIOR SECTION DEPARTMENT OF CHEMISTRY CLASS X- PRACTICAL WORKSHEET
 INDIAN SCHOOL MUSCAT SENIOR SECTION DEPARTMENT OF CHEMISTRY CLASS X- PRACTICAL WORKSHEET Different types of chemical reactions Experiment No: 1(a) Combination reaction Objectives: To study the Combination
INDIAN SCHOOL MUSCAT SENIOR SECTION DEPARTMENT OF CHEMISTRY CLASS X- PRACTICAL WORKSHEET Different types of chemical reactions Experiment No: 1(a) Combination reaction Objectives: To study the Combination
Valve Nest Filter with Time Clock Control Installation and Operation Manual
 Valve Nest Filter with Time Clock Control Installation and Operation Manual Effective 5/24/12 Effective 5/24/12 Table of Contents Operating Data. 1 Pre-Installation Data... 2 Filter Parameters Installation
Valve Nest Filter with Time Clock Control Installation and Operation Manual Effective 5/24/12 Effective 5/24/12 Table of Contents Operating Data. 1 Pre-Installation Data... 2 Filter Parameters Installation
Chemical Flood Design
 Chemical Flood Design V L A D I M I R A L V A R A D O C H E M I C A L A N D P E T R O L E U M E N G I N E E R I N G J A N U A R Y 2 0 1 2 Outline Project objective Fluid characterization Design challenge
Chemical Flood Design V L A D I M I R A L V A R A D O C H E M I C A L A N D P E T R O L E U M E N G I N E E R I N G J A N U A R Y 2 0 1 2 Outline Project objective Fluid characterization Design challenge
How Salty Is Our Water?
 Title: How Salty Is Our Water? (Water Chemistry) Grade(s): 6-7 Introduction: Corals require a relatively constant environment. However, due to the proximity to the surface of the ocean, the salinity of
Title: How Salty Is Our Water? (Water Chemistry) Grade(s): 6-7 Introduction: Corals require a relatively constant environment. However, due to the proximity to the surface of the ocean, the salinity of
Evaporative Condenser Passivation. Cameron Klein Strand Associates, Inc.
 Evaporative Condenser Passivation Cameron Klein Strand Associates, Inc. Evaporative Condenser Wetted Surface Materials of Construction Galvanized Steel Protective zinc coating fused to a steel substrate
Evaporative Condenser Passivation Cameron Klein Strand Associates, Inc. Evaporative Condenser Wetted Surface Materials of Construction Galvanized Steel Protective zinc coating fused to a steel substrate
Reactivity Series. Question Paper. Save My Exams! The Home of Revision. Module Double Award (Paper 1C) Chemistry of the Elements.
 Save My Exams! The Home of Revision For more awesome GCSE and A level resources, visit us at www.savemyexams.co.uk Reactivity Series Question Paper Level GCSE Subject Chemistry Exam Board Edexcel IGCSE
Save My Exams! The Home of Revision For more awesome GCSE and A level resources, visit us at www.savemyexams.co.uk Reactivity Series Question Paper Level GCSE Subject Chemistry Exam Board Edexcel IGCSE
London Examinations IGCSE
 Centre No. Paper Reference Surname Initial(s) Candidate No. Signature Paper Reference(s) 4335/03 4437/08 London Examinations IGCSE Chemistry 4335 Paper 3 Science (Double Award) 4437 Paper 8 Foundation
Centre No. Paper Reference Surname Initial(s) Candidate No. Signature Paper Reference(s) 4335/03 4437/08 London Examinations IGCSE Chemistry 4335 Paper 3 Science (Double Award) 4437 Paper 8 Foundation
