Right and wrong in antinuclear antibody tests. Timo Walle HUSLAB, Department of Immunology
|
|
|
- Bernadette Melton
- 6 years ago
- Views:
Transcription
1 Right and wrong in antinuclear antibody tests Timo Walle HUSLAB, Department of Immunology
2 Antinuclear antibodies (ANA) Autoantibodies reacting with nuclear antigens Nucleoplasm Nuclear membrane Nucleoli (Mitotic spindle) Today, all antibodies reacting with Hep2 cells are often called ANA including antibodies to cytoplasmic structures Ribosomes Mitochondria Lysosomes Golgi Cytoskeleton Centrioles
3 Antinuclear antibodies in diagnostics Tentative diagnosis ANA screening IFA / EIA Staining pattern Titer/Units + - ANA to specific antigens (EIA screen?) SSA Jo-1 Ribosomal P prot dsdna, histone, nucleosome Sm, U1-RNP, SS-A, -B, Scl-70/Topo I, Jo-1 (Histidyl trna synthetase), Ribosomal P proteins etc.
4 Conditions associated with a positive ANA ANA very useful for diagnosis Systemic lupus erythematosus Systemic sclerosis ANA somewhat useful for diagnosis Sjögren's syndrome Polymyositis-dermatomyositis ANA very useful for monitoring or prognosis Juvenile chronic arthritis Raynaud's phenomenon ANA is a critical part of the diagnostic criteria Drug-associated lupus Mixed connective tissue disease Autoimmune hepatitis Solomon DH et al. Evidence-based guidelines for the use of immunologic tests: Antinuclear antibody testing. Arthr Rheum 2002;47,4,434
5 ANA not useful or has no proven value for diagnosis, monitoring or prognosis Rheumatoid arthritis Multiple sclerosis Thyroid disease Infectious disease Idiopathic thrombocytopenic purpura Fibromyalgia
6 ANA screening methods Indirect immunofluorescence assay (IFA) Substrate Secondary antibody Hep-2 Hep-2 cells + SSA/Ro60 Other cell lines (rarely used) Tissue sections Anti-human-IgG Anti-human-Ig
7 ANA screening methods EIA and other solid phase assays Antigens Nuclear extracts Purified nuclear antigens Recombinant proteins
8 IFA with Hep-2 substrate as screening test Today considered as the standard method for ANA Human laryngeal carcinoma (epithelial) cell line A vast array of different autoantigens in relatively native conformation Fixation/handling of the cells may affect by denaturing antigen determinants or extracting antigens (SSA) Sometimes allows presumptive estimate of the antibody specificity (centromere, nucleolus antigens, mitochondria etc.) In most cases further testing is needed to find the specificity of the ANA reaction Labor intensive, subjective (microscopy) Specificity should be set to an acceptable level, 5 % positive results in healthy population
9 ANA in healthy individuals Tan et al, Arthr Rheum 40: 1601, 1997 International units (WHO, IU/ml) can be used
10 Solid phase screening methods Easy to automate, objective results Able to detect only a limited number of ANA specificities Abs to rare antigens are often missed (nucleolus, nuclear membrane, mitotic spindle etc.) Mixtures of purified antigens are best suited to screening before specific ANA tests Nuclear extracts combined with defined antigens correlate best with IFA No information about the ANA specificity Is it right to call these tests ANA?
11 Antibodies to specific nuclear antigens ENA (extractable nuclear antigens) Original antigens saline extracts of nuclear proteins or RNAprotein complexes Sm (B/B, D1/2/3) RNP (70k, A, C) SS-A/Ro (Ro52, Ro60) SS-B/La Scl70/Topo I Jo-1 (and other aminoacyl trna synthetases) Ribosomal P proteins Original methods: Double immunodiffusion Counter-immunoelectrophoresis (Passive hemagglutination, Western blotting)
12 Solid phase techniques for specific ANA Methods EIA Bead arrays Line/dot blotting Microchip technology Antigens Purified proteins Recombinant proteins E. coli Baculovirus Synthetic peptides (immunodominant peptide)
13 Anti-dsDNA antibodies ANA and anti-dsdna (or anti-sm) antibodies are included in the ACR classification criteria for SLE More often present in active disease and in cases with renal involvment In some patients rising levels indicate impending disease flares may be useful in longitudinal assessment
14 RIA Anti-dsDNA techniques Farr assay (precipitation with ammonium sulphate) Detects high avidity Abs, all Ig isotypes Quantitative PEG RIA (precipitation with polyethylene glycol) Detects also antibodies with lower avidity IFA Crithidia luciliae kinetoplast Detects medium to high avidity Abs High disease specificity for SLE, low sensitivity Usually IgG (isotype can be determined) Semiquantitative
15 Anti-dsDNA techniques EIA (and other solid phase techniques) Detect also low avidity Abs Sensitive Lower disease specificity for SLE Quantitative Easy to automate Modification for analysis of high avidity Abs possible
16 dsdna antigens Bacterial Mammalian (calf thymus) Plasmid Bacteriophage Defined human oligonucleotides Denaturation of dsdna to ssdna may lead to unspecific reactions circular dsdna (plasmid or bacteriophage) is preferred Protein contamination
17 Anti-dsDNA Discrepant results with different techniques Avidity, Ig isotype, antigen At population level relatively good correlation, for individual patient sera discrepancies (up to 30 %) Different tests for different applications: Screening High sensitivity (EIA) Diagnosis High specificity (IFA, Farr, EIA with higher cut off?) Monitoring disease activity (Farr, EIA?) Cooperation between clinicians and laboratories
18 External quality assessment in ANA tests EQA schemes are usually able to provide consensus reports only Ideal samples - Single donor serum/plasma (not diluted) - Clinically diagnosed donors - Reactivity tested in a few specialized laboratories using several methods including conceivable golden standards Ideal samples are not readily available
19 Labquality ANA, ENA, dsdna Methods used for selecting samples Test ANA (titer, staining pattern) Anti-dsDNA Anti-Sm (B, D) Anti-U1-RNP (70k, A, C) Anti-SSA (Ro52, Ro60) Anti-SSB Anti-Scl-70/Topo I Anti-Jo1 (histidyl trna synthetase) Anti-Cenp-B Method IFA (Hep2) IFA (Crithidia luciliae), FEIA FEIA, Line blotting FEIA, Line blotting FEIA, Line blotting FEIA, Line blotting FEIA, Line blotting FEIA, Line blotting FEIA, Line blotting
20 Results: Right or wrong? Policy of Labquality Informative reports, no scoring Expected/intended results = results in the reference laboratory - should not be taken as the only right response Educational challenge Comparison between other laboratories (own method, all methods) Comparison between methods Before international standards for the specific ANAs are available the results can be considered right or wrong according to participant consensus The qualitative results for each ANA test can be evaluated when 80 % participant consensus is reached?
21 Use of international serum standards Standards/International units (IU) accepted by WHO WHO 66/233 (anti-nuclear factor) for homogeneous pattern in IFA WHO Wo/80 for anti-dsdna antibodies Other reference sera available for quality assurance Arthritis Foundation/Centers of Disease Control and Prevention (CDC) antinuclear antibody reference sera for IFA patterns and for defined ANA specificities Association of Medical Laboratory Immunologists (AMLI) consensus reference panel for defined ANA specificities
immuno concepts Immuno Concepts is proud to announce that when present the characteristic SS-A/Ro pattern detected by the HEp-2000 substrate
 immuno concepts Immuno Concepts is proud to announce that when present the characteristic SS-A/Ro pattern detected by the HEp-2000 substrate is now considered confirmatory for the presence of antibodies
immuno concepts Immuno Concepts is proud to announce that when present the characteristic SS-A/Ro pattern detected by the HEp-2000 substrate is now considered confirmatory for the presence of antibodies
AUTOIMMUNE DISEASE ASSAYS
 product range 1 Corgenix PRODUCT RANGE 04-05 Antiphospholipid Syndrome Assays 06-10 AUTOIMMUNE DISEASE ASSAYS 11 Liver Disease Assays 11 Liver Fibrosis Markers 12 Cardiovascular Markers 12-13 Platelet
product range 1 Corgenix PRODUCT RANGE 04-05 Antiphospholipid Syndrome Assays 06-10 AUTOIMMUNE DISEASE ASSAYS 11 Liver Disease Assays 11 Liver Fibrosis Markers 12 Cardiovascular Markers 12-13 Platelet
ANA Screen 8 ELISA KIT
 ANA Screen 8 ELISA KIT Cat. No.:DEIA6289 Pkg.Size:96T Intended use ANA Screen 8 ELISA is for the qualitative screening of IgG antibodies against dsdna, RNP, Sm, SS-A/Ro, SS-B/La, Scl-70, CENP-B and Jo-1
ANA Screen 8 ELISA KIT Cat. No.:DEIA6289 Pkg.Size:96T Intended use ANA Screen 8 ELISA is for the qualitative screening of IgG antibodies against dsdna, RNP, Sm, SS-A/Ro, SS-B/La, Scl-70, CENP-B and Jo-1
Detection and identification of antinuclear antibodies (ANA) in a large and consecutive cohort of serum samples referred for ANA testing
 Ann Rheum Dis 2001;60:1131 1136 1131 Department of Rheumatology, Ghent University Hospital, Belgium I Peene EMVeys F De Keyser Innogenetics NV, Ghent, Belgium L Meheus Correspondence to: Dr F De Keyser,
Ann Rheum Dis 2001;60:1131 1136 1131 Department of Rheumatology, Ghent University Hospital, Belgium I Peene EMVeys F De Keyser Innogenetics NV, Ghent, Belgium L Meheus Correspondence to: Dr F De Keyser,
Bio-Rad Laboratories BIOPLEX 2200 SYSTEM. BioPlex 2200 ANA Screen with MDSS. The first and only fully-automated, multiplexed ANA Screen
 Bio-Rad Laboratories BIOPLEX 2200 SYSTEM BioPlex 2200 ANA Screen with MDSS The first and only fully-automated, multiplexed ANA Screen Bio-Rad Laboratories BIOPLEX 2200 SYSTEM Like no other The BioPlex
Bio-Rad Laboratories BIOPLEX 2200 SYSTEM BioPlex 2200 ANA Screen with MDSS The first and only fully-automated, multiplexed ANA Screen Bio-Rad Laboratories BIOPLEX 2200 SYSTEM Like no other The BioPlex
Screening for IgG Antinuclear Autoantibodies by HEp-2 Indirect Fluorescent Antibody Assays and the Need for Standardization
 Immunopathology / HEp-2 Immunofluorescence Standardization Screening for IgG Antinuclear Autoantibodies by HEp-2 Indirect Fluorescent Antibody Assays and the Need for Standardization Susan S. Copple, MS,
Immunopathology / HEp-2 Immunofluorescence Standardization Screening for IgG Antinuclear Autoantibodies by HEp-2 Indirect Fluorescent Antibody Assays and the Need for Standardization Susan S. Copple, MS,
INTERPRETIVE INFORMATION: Jo-1 Antibody, IgG 29 AU/mL or less...negative AU/mL...Equivocal 41 AU/mL or greater...positive
 Client: Example Client ABC123 123 Test Drive Salt Lake City, UT 84108 UNITED STATES Physician: Doctor, Example DOB Unknown Gender: Unknown Collection Date: 00/00/0000 00:00 Myositis Extended Panel ARUP
Client: Example Client ABC123 123 Test Drive Salt Lake City, UT 84108 UNITED STATES Physician: Doctor, Example DOB Unknown Gender: Unknown Collection Date: 00/00/0000 00:00 Myositis Extended Panel ARUP
ANA and Antibody Series
 ANA and Antibody Series How to Do Scleroderma ANA and Antibody Testing Correctly (A Practical Guide for Clinicians) Background When a clinician has a reason to suspect that a patient may have an autoimmune
ANA and Antibody Series How to Do Scleroderma ANA and Antibody Testing Correctly (A Practical Guide for Clinicians) Background When a clinician has a reason to suspect that a patient may have an autoimmune
SAMPLE I/LA02-A2. Archived Document
 Archived Document This archived document is no longer being reviewed through the CLSI Consensus Document Development Process. However, this document is technically valid as of September 2016. Because of
Archived Document This archived document is no longer being reviewed through the CLSI Consensus Document Development Process. However, this document is technically valid as of September 2016. Because of
CERTIFICATE OF ANALYSIS
 Catalog #: K07110H Lot #: 17A01918 Host Animal: Positive Control Set For Autoimmune Positive Control Set (SS-A, SS-B, Sm, nrnp, Scl-70, Jo-1) Set Includes: Catalog# K07103H Human anti Human Ro (SS-A) Antigen
Catalog #: K07110H Lot #: 17A01918 Host Animal: Positive Control Set For Autoimmune Positive Control Set (SS-A, SS-B, Sm, nrnp, Scl-70, Jo-1) Set Includes: Catalog# K07103H Human anti Human Ro (SS-A) Antigen
Carmen Gelpí Elena Pérez Cristina Roldan
 Autoimmun Highlights (2014) 5:47 54 DOI 10.1007/s13317-014-0059-x REVIEW ARTICLE Efficiency of a solid-phase chemiluminescence immunoassay for detection of antinuclear and cytoplasmic autoantibodies compared
Autoimmun Highlights (2014) 5:47 54 DOI 10.1007/s13317-014-0059-x REVIEW ARTICLE Efficiency of a solid-phase chemiluminescence immunoassay for detection of antinuclear and cytoplasmic autoantibodies compared
Correlation of Line Immuno Assay with Indirect Immunofluoresence Assay for the Detection of Anti-Nuclear Antibodies in Various Autoimmune Disorders
 Research Article imedpub Journals http://www.imedpub.com/ Journal of Autoimmune Disorders Correlation of Line Immuno Assay with Indirect Immunofluoresence Assay for the Detection of Anti-Nuclear Antibodies
Research Article imedpub Journals http://www.imedpub.com/ Journal of Autoimmune Disorders Correlation of Line Immuno Assay with Indirect Immunofluoresence Assay for the Detection of Anti-Nuclear Antibodies
Development of the Antinuclear and Anticytoplasmic Antibody Consensus Panel by the Association of Medical Laboratory Immunologists
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, May 2000, p. 436 443 Vol. 7, No. 3 1071-412X/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Development of the Antinuclear
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, May 2000, p. 436 443 Vol. 7, No. 3 1071-412X/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Development of the Antinuclear
Practical Evaluation of Methods for Detection and Specificity of Autoantibodies to Extractable Nuclear Antigens
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Mar. 2004, p. 297 301 Vol. 11, No. 2 1071-412X/04/$08.00 0 DOI: 10.1128/CDLI.11.2.297 301.2004 Copyright 2004, American Society for Microbiology. All Rights
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Mar. 2004, p. 297 301 Vol. 11, No. 2 1071-412X/04/$08.00 0 DOI: 10.1128/CDLI.11.2.297 301.2004 Copyright 2004, American Society for Microbiology. All Rights
Immunofluorescence Assays for Autoimmune and Infectious Disease
 life.science.discovery. Immunofluorescence Assays for Autoimmune and Infectious Disease PRODUCT CATALOG COMPANY MBL Bion, part of the MBL, has been manufacturing high-quality kits and components for autoimmune,
life.science.discovery. Immunofluorescence Assays for Autoimmune and Infectious Disease PRODUCT CATALOG COMPANY MBL Bion, part of the MBL, has been manufacturing high-quality kits and components for autoimmune,
NOVA Lite HEp-2 External EB Kits For In Vitro Diagnostic Use
 NOVA Lite HEp-2 External EB Kits For In Vitro Diagnostic Use Product Code: 704230, 704235 CLIA Complexity: High Intended Use This product (kit or substrate slides) is intended for use in the screening
NOVA Lite HEp-2 External EB Kits For In Vitro Diagnostic Use Product Code: 704230, 704235 CLIA Complexity: High Intended Use This product (kit or substrate slides) is intended for use in the screening
Enzyme-Linked Immunosorbent Assay Screening Then Indirect Immunofluorescence Confirmation of Antinuclear Antibodies A Statistical Analysis
 Immunopathology / Screening Antinuclear Antibodies Enzyme-Linked Immunosorbent Assay Screening Then Indirect Immunofluorescence Confirmation of Antinuclear Antibodies A Statistical Analysis Susan S. Copple,
Immunopathology / Screening Antinuclear Antibodies Enzyme-Linked Immunosorbent Assay Screening Then Indirect Immunofluorescence Confirmation of Antinuclear Antibodies A Statistical Analysis Susan S. Copple,
ENA-6 Screen ELISA KIT
 ENA-6 Screen ELISA KIT Cat. No.:DEIA05732 Pkg.Size:96T Intended use For the qualitative screening of human IgG antibodies to extractable nuclear antigens (ENA) in human serum by indirect enzyme immunoassay
ENA-6 Screen ELISA KIT Cat. No.:DEIA05732 Pkg.Size:96T Intended use For the qualitative screening of human IgG antibodies to extractable nuclear antigens (ENA) in human serum by indirect enzyme immunoassay
EL-ANAScr. An enzyme immunoassay for the screening of human serum to detect antinuclear antibodies (ANAs) Instruction Manual
 EL-ANAScr An enzyme immunoassay for the screening of human serum to detect antinuclear antibodies (ANAs) Instruction Manual Catalog Nos.: 100-002, 100-004 TheraTest Laboratories, Inc. 1111 North Main Street
EL-ANAScr An enzyme immunoassay for the screening of human serum to detect antinuclear antibodies (ANAs) Instruction Manual Catalog Nos.: 100-002, 100-004 TheraTest Laboratories, Inc. 1111 North Main Street
AUTOIMMUNITY CATALOGUE
 AUTOIMMUNITY Autoimmune Rheumatic Disease Antiphospholipid Syndrome ANCA & Vasculitis Hashimoto Thyroiditis Celiac Disease Autoimmune Gastroenteritis Disease Inflammatory Bowel Disease Autoimmune Diabetes
AUTOIMMUNITY Autoimmune Rheumatic Disease Antiphospholipid Syndrome ANCA & Vasculitis Hashimoto Thyroiditis Celiac Disease Autoimmune Gastroenteritis Disease Inflammatory Bowel Disease Autoimmune Diabetes
3. REAGENTS, MATERIALS AND INSTRUMENTATION
 DCM9-0 Ed. 03/202 ENA Profile for routine analysis Indirect immunoenzymatic determination of IgG class autoantibodies against ENA (Extractable Nuclear s) in human serum or plasma. IVD LOT See external
DCM9-0 Ed. 03/202 ENA Profile for routine analysis Indirect immunoenzymatic determination of IgG class autoantibodies against ENA (Extractable Nuclear s) in human serum or plasma. IVD LOT See external
ANA ELISA Screen Enzyme Immunoassay Test Kit For In Vitro Diagnostic Use Catalog No Tests
 SUMMARY OF PROCEDURE 1. Prepare 1:101 dilutions of samples in Sample Diluent. Mix well. 2. Add 100 µl of diluted samples into the antigen wells. 3. Incubate at room temperature (18-30 o C) for 30 + 5 min.
SUMMARY OF PROCEDURE 1. Prepare 1:101 dilutions of samples in Sample Diluent. Mix well. 2. Add 100 µl of diluted samples into the antigen wells. 3. Incubate at room temperature (18-30 o C) for 30 + 5 min.
ANA Diagnostics Using Indirect Immunofluorescence
 ANA Diagnostics Using Indirect Immunofluorescence EUROIMMUN D-23560 Luebeck (Germany) Seekamp 31 Tel +49 45158550 Fax 5855591 E-mail euroimmun@euroimmun.de Table of contents Autoantibodies against cell
ANA Diagnostics Using Indirect Immunofluorescence EUROIMMUN D-23560 Luebeck (Germany) Seekamp 31 Tel +49 45158550 Fax 5855591 E-mail euroimmun@euroimmun.de Table of contents Autoantibodies against cell
(Enzyme immunoassay for the qualitative detection of auto-antibodies to extractable nuclear antigens) Codes: Well packs: Instructions for Use
 AUTOZYME TM ENA (Enzyme immunoassay for the qualitative detection of auto-antibodies to extractable nuclear antigens) Codes: Bulk pack: Well packs: Anti-Ro: Anti-La: Anti-Sm: Anti-Sm/RNP: Anti-Jo-1: Anti-Scl-70:
AUTOZYME TM ENA (Enzyme immunoassay for the qualitative detection of auto-antibodies to extractable nuclear antigens) Codes: Bulk pack: Well packs: Anti-Ro: Anti-La: Anti-Sm: Anti-Sm/RNP: Anti-Jo-1: Anti-Scl-70:
Detection of Anti-SSA Antibodies by Indirect Immunofluorescence
 Clinical Chemistry 50:12 2361 2369 (2004) Clinical Immunology Detection of Anti-SSA Antibodies by Indirect Immunofluorescence Xavier Bossuyt, 1* Johan Frans, 1 Ann Hendrickx, 1 Godelieve Godefridis, 1
Clinical Chemistry 50:12 2361 2369 (2004) Clinical Immunology Detection of Anti-SSA Antibodies by Indirect Immunofluorescence Xavier Bossuyt, 1* Johan Frans, 1 Ann Hendrickx, 1 Godelieve Godefridis, 1
ENA-6 Screen Enzyme Immunoassay Test Kit For In Vitro Diagnostic Use Catalog No Tests
 SUMMARY OF PROCEDURE ENA-6 Screen Enzyme Immunoassay Test Kit For In Vitro Diagnostic Use Catalog No. 720-230 96 Tests 1. Prepare a 1:101 dilution of samples in Sample Diluent. Mix well. 2. Add 100 µl
SUMMARY OF PROCEDURE ENA-6 Screen Enzyme Immunoassay Test Kit For In Vitro Diagnostic Use Catalog No. 720-230 96 Tests 1. Prepare a 1:101 dilution of samples in Sample Diluent. Mix well. 2. Add 100 µl
Research Article Evaluation of a Multiplex ELISA for Autoantibody Profiling in Patients with Autoimmune Connective Tissue Diseases
 Hindawi Publishing Corporation Autoimmune Diseases Volume 2014, Article ID 896787, 6 pages http://dx.doi.org/10.1155/2014/896787 Research Article Evaluation of a Multiplex ELISA for Autoantibody Profiling
Hindawi Publishing Corporation Autoimmune Diseases Volume 2014, Article ID 896787, 6 pages http://dx.doi.org/10.1155/2014/896787 Research Article Evaluation of a Multiplex ELISA for Autoantibody Profiling
IMMUNOASSAYS IMMUNOFLUORESCENCE RADIOIMMUNOLOGY ANTIGENS ENZYMOIMMUNOLOGY COLORIMETRY : ELISA,DOT FLUORIMETRY CHEMILUMINESCENCE LUMINOMETRY
 IMMUNOASSAYS IMMUNOFLUORESCENCE RADIOIMMUNOLOGY ENZYMOIMMUNOLOGY ANTIGENS COLORIMETRY : ELISA,DOT FLUORIMETRY CHEMILUMINESCENCE LUMINOMETRY BIOCHIPS ANTIGENS IMMUNOASSAYS IMMUNOFLUORESCENCE Natural Antigens
IMMUNOASSAYS IMMUNOFLUORESCENCE RADIOIMMUNOLOGY ENZYMOIMMUNOLOGY ANTIGENS COLORIMETRY : ELISA,DOT FLUORIMETRY CHEMILUMINESCENCE LUMINOMETRY BIOCHIPS ANTIGENS IMMUNOASSAYS IMMUNOFLUORESCENCE Natural Antigens
Interpath Laboratory, Inc. Test File Update
 As an Interpath customer who receives electronic results or sends electronic orders you may need to be notified when we update our Service Manual. Although we try to keep these changes to a minimum, laboratory
As an Interpath customer who receives electronic results or sends electronic orders you may need to be notified when we update our Service Manual. Although we try to keep these changes to a minimum, laboratory
Antinuclear antibody determination in a routine
 Ann Rheum Dis 1996;55:723-727 723 NOW AND THEN Arthron, Research Centre for Rheumatic Diseases Amsterdam, Amsterdam, The Netherlands T E W Feltkamp Correspondence to: Professor Dr T E W Feltkamp, Arthron,
Ann Rheum Dis 1996;55:723-727 723 NOW AND THEN Arthron, Research Centre for Rheumatic Diseases Amsterdam, Amsterdam, The Netherlands T E W Feltkamp Correspondence to: Professor Dr T E W Feltkamp, Arthron,
RNA polymerase II precursor mrna (hnrna) introns introns U1RNP. splicing
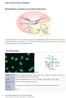 DNA RNA polymerase II precursor mrna (hnrna) introns introns U2RNP splicing mrna U1RNP U5RNP U4/U6RNP splicing The genetic information of DNA is transcribed to precursor mrna (heterogeneous nuclear RNA,
DNA RNA polymerase II precursor mrna (hnrna) introns introns U2RNP splicing mrna U1RNP U5RNP U4/U6RNP splicing The genetic information of DNA is transcribed to precursor mrna (heterogeneous nuclear RNA,
ENA Combined SCREEN (6 Antigen)
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Comparison of assay systems for detecting
 J Clin Pathol 1991;44:685-689 Department of Medicine, Divisions of Rheumatology and Clinical Immunology, Hospital for Joint Diseases Orthopaedic Institute, New York University Medical Center, 301 East
J Clin Pathol 1991;44:685-689 Department of Medicine, Divisions of Rheumatology and Clinical Immunology, Hospital for Joint Diseases Orthopaedic Institute, New York University Medical Center, 301 East
DRG International, Inc., USA Fax: (908)
 Please use only the valid version of the package insert provided with the kit. 1 NAME AND INTENDED USE ANA Screen Blot is a membrane based enzyme immunoassay for determination of IgG class autoantibodies
Please use only the valid version of the package insert provided with the kit. 1 NAME AND INTENDED USE ANA Screen Blot is a membrane based enzyme immunoassay for determination of IgG class autoantibodies
AccuDiag. SSA (Ro) ELISA INTENDED USE
 Test Method Principle Detection Range Sample Total Time Shelf Life INTENDED USE AccuDiag SSA (Ro) Kit Cat# 2555-2 See external Label 96 Tests SSA (Ro) Enzyme Linked Immunosorbent Assay - Indirect; Antigen
Test Method Principle Detection Range Sample Total Time Shelf Life INTENDED USE AccuDiag SSA (Ro) Kit Cat# 2555-2 See external Label 96 Tests SSA (Ro) Enzyme Linked Immunosorbent Assay - Indirect; Antigen
A nti-ssa/ro antibodies are the most frequently identified
 929 CONCISE REPORT Anti-Ro52 reactivity is an independent and additional serum marker in connective tissue disease I Peene, L Meheus, S De Keyser, R Humbel, E M Veys, F De Keyser... Ann Rheum Dis 2002;61:929
929 CONCISE REPORT Anti-Ro52 reactivity is an independent and additional serum marker in connective tissue disease I Peene, L Meheus, S De Keyser, R Humbel, E M Veys, F De Keyser... Ann Rheum Dis 2002;61:929
Anti-Histone (H1) (IgG) ELISA
 Anti-Histone (H1) (IgG) ELISA For the quantitative and qualitative detection of antibodies against histone H1 in serum. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 35-HH1HU-E01
Anti-Histone (H1) (IgG) ELISA For the quantitative and qualitative detection of antibodies against histone H1 in serum. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 35-HH1HU-E01
dsdna Ab ELISA Kit Catalog Number KA assays Version: 01 Intended for research use only
 dsdna Ab ELISA Kit Catalog Number KA3171 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
dsdna Ab ELISA Kit Catalog Number KA3171 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
anti-ssb Enzyme Immunoassay
 anti-ssb Enzyme Immunoassay For Individual Laboratory to Complete: Laboratory Name Adopted Reviewed Reviewed Revised Supercedes Method: Diamedix Corp., Immunosimplicity Manual or in conjunction with one
anti-ssb Enzyme Immunoassay For Individual Laboratory to Complete: Laboratory Name Adopted Reviewed Reviewed Revised Supercedes Method: Diamedix Corp., Immunosimplicity Manual or in conjunction with one
AccuDiag. SSB (La) ELISA
 AccuDiag SSB (La) ELISA Kit Cat# 2556-2Z See external Label 2-8 C = 96 Tests Test SSB (La) ELISA Method Enzyme Linked Immunosorbent Assay Principle Indirect; Antigen Coated Plate Detection Range Qualitative:
AccuDiag SSB (La) ELISA Kit Cat# 2556-2Z See external Label 2-8 C = 96 Tests Test SSB (La) ELISA Method Enzyme Linked Immunosorbent Assay Principle Indirect; Antigen Coated Plate Detection Range Qualitative:
COMPARATIVE STUDY OF ANTI-DOUBLE STRANDED DNA DETECTION BY ELISA AND CRITHIDIA LUCILIAE IMMUNOFLUORESCENCE
 COMPARATIVE STUDY OF ANTI-DOUBLE STRANDED DNA DETECTION BY ELISA AND CRITHIDIA LUCILIAE IMMUNOFLUORESCENCE Kritsana Janyapoon, Pantakit Jivakanont, Kantima Choosang, Rungkarn Surbrsing, Vannapa Charoenying
COMPARATIVE STUDY OF ANTI-DOUBLE STRANDED DNA DETECTION BY ELISA AND CRITHIDIA LUCILIAE IMMUNOFLUORESCENCE Kritsana Janyapoon, Pantakit Jivakanont, Kantima Choosang, Rungkarn Surbrsing, Vannapa Charoenying
Anti ENA Screen ELISA
 Anti ENA Screen ELISA GWB-521218 www.genwaybio.com FOR RESEARCH USE ONLY 1. INTRODUCTION Enzyme immunoassay for the quantitative determination of anti ENA in human serum or plasma Autoimmune diseases are
Anti ENA Screen ELISA GWB-521218 www.genwaybio.com FOR RESEARCH USE ONLY 1. INTRODUCTION Enzyme immunoassay for the quantitative determination of anti ENA in human serum or plasma Autoimmune diseases are
AccuDiag ENA Combined Screen (6 Antigen) ELISA Kit
 AccuDiag ENA Combined Screen (6 Antigen) ELISA Kit Test Method Principle Detection Range Sample Total Time Shelf Life INTENDED USE Cat# 2552-2 See external Label 96 Tests ENA Combined Screen ELISA Enzyme
AccuDiag ENA Combined Screen (6 Antigen) ELISA Kit Test Method Principle Detection Range Sample Total Time Shelf Life INTENDED USE Cat# 2552-2 See external Label 96 Tests ENA Combined Screen ELISA Enzyme
Human ANA IgG ELISA Kit
 Human ANA IgG ELISA Kit Cat.No:DEIA1695 Lot. No. (See product label) PRODUCT INFOMATION Storage specificity 1. Store the unopened kit at 2-8 C. 2. Coated microwell strips: Store between 2 and 8 C. Extra
Human ANA IgG ELISA Kit Cat.No:DEIA1695 Lot. No. (See product label) PRODUCT INFOMATION Storage specificity 1. Store the unopened kit at 2-8 C. 2. Coated microwell strips: Store between 2 and 8 C. Extra
Immunometric Enzyme Immunoassay for the quantitative determination of IgG autoantibodies to double-stranded DNA
 ORGENTEC Diagnostika GmbH Carl-Zeiss-Straße 49 55129 Mainz - Germany Phone: +496131/92580 Fax: +496131/925858 Internet : www. orgentec.com Instruction for use August 2005 ORG 604 CONTENTS Anti-dsDNA Immunometric
ORGENTEC Diagnostika GmbH Carl-Zeiss-Straße 49 55129 Mainz - Germany Phone: +496131/92580 Fax: +496131/925858 Internet : www. orgentec.com Instruction for use August 2005 ORG 604 CONTENTS Anti-dsDNA Immunometric
AccuDiag ENA Combined Screen (6 Antigen) ELISA Kit
 AccuDiag ENA Combined Screen (6 Antigen) ELISA Kit Test Method Principle Detection Range Sample Total Time Shelf Life INTENDED USE Cat# 2552-2 See external Label 96 Tests ENA Combined Screen ELISA Enzyme
AccuDiag ENA Combined Screen (6 Antigen) ELISA Kit Test Method Principle Detection Range Sample Total Time Shelf Life INTENDED USE Cat# 2552-2 See external Label 96 Tests ENA Combined Screen ELISA Enzyme
AccuDiag ENA Profile-6 SM/RNP, Sm, Jo-1, Scl-70, SS-A, SS-B ELISA Kit
 AccuDiag ENA Profile-6 SM/RNP, Sm, Jo-1, Scl-70, SS-A, SS-B Kit with a homozygous C2 deficiency (13), and in a subset of patients who lack antidsdna antibodies (11). Scl-70 antibodies are highly specific
AccuDiag ENA Profile-6 SM/RNP, Sm, Jo-1, Scl-70, SS-A, SS-B Kit with a homozygous C2 deficiency (13), and in a subset of patients who lack antidsdna antibodies (11). Scl-70 antibodies are highly specific
Original papers. Introduction. Andrea Tešija Kuna* 1, Lovorka Đerek 2, Ana Kozmar 3, Vedrana Drvar 4
 Original papers Current practice in laboratory diagnostics of autoimmune diseases in Croatia. Survey of the Working group for laboratory diagnostics of autoimmune diseases of the Croatian Society of Medical
Original papers Current practice in laboratory diagnostics of autoimmune diseases in Croatia. Survey of the Working group for laboratory diagnostics of autoimmune diseases of the Croatian Society of Medical
Diagnostic Automation, Inc Craftsman Road, Suite E/F Calabasas, CA Tel: , Fax:
 Diagnostic Automation, Inc. 23961 Craftsman Road, Suite E/F Calabasas, CA 91302 Tel:818-591-3030, Fax:818-591-8383 RNP IgG,A,M ELISA For in vitro diagnostic use. Catalog No. 2558-1 RNP IgG, A, M; Page
Diagnostic Automation, Inc. 23961 Craftsman Road, Suite E/F Calabasas, CA 91302 Tel:818-591-3030, Fax:818-591-8383 RNP IgG,A,M ELISA For in vitro diagnostic use. Catalog No. 2558-1 RNP IgG, A, M; Page
SSA (Ro) IgG,A,M ELISA For in vitro diagnostic use Catalog No
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
!""#$%&'(#)'%$*+!,-*
 ! How does methodology influence autoantibody test results? Johan Rönnelid MD, PhD Unit of Clinical Immunology and Transfusion medicine Uppsala University Hospital Department of Immunology, Genetics and
! How does methodology influence autoantibody test results? Johan Rönnelid MD, PhD Unit of Clinical Immunology and Transfusion medicine Uppsala University Hospital Department of Immunology, Genetics and
immunofluorescence tests in routine laboratories Dr. Dirk Roggenbuck
 Standardization di ti of cellbased immunofluorescence tests in routine laboratories Dr. Dirk Roggenbuck Overview Automatic interpretation system AKLIDES - basis for standardization Cell-based immunofluorescence
Standardization di ti of cellbased immunofluorescence tests in routine laboratories Dr. Dirk Roggenbuck Overview Automatic interpretation system AKLIDES - basis for standardization Cell-based immunofluorescence
ENA-6-Profile ORG x 16 Tests. Immunometric Enzyme Immunoassay for the semiquantitative. of anti-ena-antibodies. Instruction for use
 ORGENTEC Diagnostika GmbH Carl-Zeiss-Straße 49 55129 Mainz Tel.: 06131-9258-0 Fax: 06131-9258-58 ENA-6-Profile ORG 546 6 x 16 Tests Immunometric Enzyme Immunoassay for the semiquantitative determination
ORGENTEC Diagnostika GmbH Carl-Zeiss-Straße 49 55129 Mainz Tel.: 06131-9258-0 Fax: 06131-9258-58 ENA-6-Profile ORG 546 6 x 16 Tests Immunometric Enzyme Immunoassay for the semiquantitative determination
SUMMARY AND EXPLANATION OF THE TEST
 RELISA ENA SINGLE WELL SCREENING TEST FOR ANTIBODIES TO EXTRACTABLE NUCLEAR ANTIGENS For in vitro Diagnostic Use For Professional Use Catalog numbers: 7096-10 (96 wells) and 7696-10 (576 wells) INTENDED
RELISA ENA SINGLE WELL SCREENING TEST FOR ANTIBODIES TO EXTRACTABLE NUCLEAR ANTIGENS For in vitro Diagnostic Use For Professional Use Catalog numbers: 7096-10 (96 wells) and 7696-10 (576 wells) INTENDED
510(k) SUBSTANTIAL EQUIVALENCE DETERMINATION DECISION SUMMARY ASSAY ONLY TEMPLATE
 510(k) SUBSTANTIAL EQUIVALENCE DETERMINATION DECISION SUMMARY ASSAY ONLY TEMPLATE A. 510(k) Number: k100499 B. Purpose for Submission: New Device C. Measurand: Rheumatoid Factors IgG, IgM and IgA and Rheumatoid
510(k) SUBSTANTIAL EQUIVALENCE DETERMINATION DECISION SUMMARY ASSAY ONLY TEMPLATE A. 510(k) Number: k100499 B. Purpose for Submission: New Device C. Measurand: Rheumatoid Factors IgG, IgM and IgA and Rheumatoid
dsdna Screen ELISA Kit
 dsdna Screen ELISA Kit Catalog Number KA1101 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
dsdna Screen ELISA Kit Catalog Number KA1101 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
Evaluation of Multiplexed Fluorescent Microsphere Immunoassay for Detection of Autoantibodies to Nuclear Antigens
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Nov. 2004, p. 1054 1059 Vol. 11, No. 6 1071-412X/04/$08.00 0 DOI: 10.1128/CDLI.11.6.1054 1059.2004 Copyright 2004, American Society for Microbiology. All
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Nov. 2004, p. 1054 1059 Vol. 11, No. 6 1071-412X/04/$08.00 0 DOI: 10.1128/CDLI.11.6.1054 1059.2004 Copyright 2004, American Society for Microbiology. All
Anti-Sm ORG Tests. Immunometric Enzyme Immunoassay for the quantitative determination of anti-sm Antibodies. Instruction for use
 ORGENTEC Diagnostika GmbH Carl-Zeiss-Straße 49 55129 Mainz Tel.: 06131-9258-0 Fax: 06131-9258-58 Anti-Sm ORG 510 96 Tests Immunometric Enzyme Immunoassay for the quantitative determination of anti-sm Antibodies
ORGENTEC Diagnostika GmbH Carl-Zeiss-Straße 49 55129 Mainz Tel.: 06131-9258-0 Fax: 06131-9258-58 Anti-Sm ORG 510 96 Tests Immunometric Enzyme Immunoassay for the quantitative determination of anti-sm Antibodies
Evaluation of five commercial kits to detect dsdna
 J Clin Pathol 1988;41:1223-1228 Evaluation of five commercial kits to detect dsdna antibodies S A McMILLAN, ANNE C FAY Northern Ireland Regional Immunology Laboratory, Belfast City Hospital, Belfast, summary
J Clin Pathol 1988;41:1223-1228 Evaluation of five commercial kits to detect dsdna antibodies S A McMILLAN, ANNE C FAY Northern Ireland Regional Immunology Laboratory, Belfast City Hospital, Belfast, summary
Routine use of Zenit RA, a novel chemiluminescent immunoanalyzer in autoimmune disease diagnosis
 Autoimmun Highlights (2012) 3:27 31 DOI 10.1007/s13317-012-0032-5 ORIGINAL ARTICLE Routine use of Zenit RA, a novel chemiluminescent immunoanalyzer in autoimmune disease diagnosis P. Ghillani L. Dufat
Autoimmun Highlights (2012) 3:27 31 DOI 10.1007/s13317-012-0032-5 ORIGINAL ARTICLE Routine use of Zenit RA, a novel chemiluminescent immunoanalyzer in autoimmune disease diagnosis P. Ghillani L. Dufat
Biohit Oyj. Anti-Telomere IgG. ELISA kit for the detection of antitelomere IgG antibodies based on the double-stranded telomeric DNA recognition
 Biohit Oyj Anti-Telomere IgG ELISA kit for the detection of antitelomere IgG antibodies based on the double-stranded telomeric DNA recognition For in vitro Diagnostic Use Store at 2-8 o C Upon Receipt
Biohit Oyj Anti-Telomere IgG ELISA kit for the detection of antitelomere IgG antibodies based on the double-stranded telomeric DNA recognition For in vitro Diagnostic Use Store at 2-8 o C Upon Receipt
Selective Detection of Autoimmune Antibodies to Singleand Double-Stranded DNA by Enzyme Immunoassay
 142 Annals of Clinical & Laboratory Science, vol. 33, no. 2, 2003 Selective Detection of Autoimmune Antibodies to Singleand Double-Stranded DNA by Enzyme Immunoassay Gregory S. Makowski 1 and Melinda L.
142 Annals of Clinical & Laboratory Science, vol. 33, no. 2, 2003 Selective Detection of Autoimmune Antibodies to Singleand Double-Stranded DNA by Enzyme Immunoassay Gregory S. Makowski 1 and Melinda L.
ANA Screening IgG. Enzyme Immunoassay (ELISA) for the qualitative determination of IgG antibodies to anti Nuclear Antigens in human serum and plasma
 Doc.: INS ANAS.CE/eng Page 1 of 7 Rev.: 0 Date: 0911 ANA Screening IgG Enzyme Immunoassay (ELISA) for the qualitative determination of IgG antibodies to anti Nuclear Antigens in human serum and plasma
Doc.: INS ANAS.CE/eng Page 1 of 7 Rev.: 0 Date: 0911 ANA Screening IgG Enzyme Immunoassay (ELISA) for the qualitative determination of IgG antibodies to anti Nuclear Antigens in human serum and plasma
ab Anti-ENA Screen Human ELISA Kit
 ab178619 Anti-ENA Screen Human ELISA Kit Instructions for Use For the semi-quantitative measurement of IgG class antibodies against extractable nuclear antigens (ENA) in Human serum and plasma. This product
ab178619 Anti-ENA Screen Human ELISA Kit Instructions for Use For the semi-quantitative measurement of IgG class antibodies against extractable nuclear antigens (ENA) in Human serum and plasma. This product
BCH 462. Single Radial Immunodiffusion and Immuno-electrophoresis
 BCH 462 Single Radial Immunodiffusion and Immuno-electrophoresis Immunoassays tests include: 1. Precipitation. 2. Agglutination. 3. Immunofluorescence. 4. Radioimmunoassay (RIA). 5. Enzyme-Linked Immuno
BCH 462 Single Radial Immunodiffusion and Immuno-electrophoresis Immunoassays tests include: 1. Precipitation. 2. Agglutination. 3. Immunofluorescence. 4. Radioimmunoassay (RIA). 5. Enzyme-Linked Immuno
IMMUNOCHEMICAL TECHNIQUES
 24 IMMUNOCHEMICAL TECHNIQUES 24.1 INTRODUCTION All vertebrates have advanced immune system. The more complex the organism the more advanced the immune system. The immune system of mammals has evolved over
24 IMMUNOCHEMICAL TECHNIQUES 24.1 INTRODUCTION All vertebrates have advanced immune system. The more complex the organism the more advanced the immune system. The immune system of mammals has evolved over
Assessment Schemes in Immunology, Allergy and Immunochemistry: Report Interpretation/Scoring th November 2006 Portugal
 Assessment Schemes in Immunology, Allergy and Immunochemistry: Report Interpretation/Scoring 13-14 th November 2006 Portugal Dina Patel UKNEQAS Immunology & Immunochemistry Operations Manager In UK NEQAS
Assessment Schemes in Immunology, Allergy and Immunochemistry: Report Interpretation/Scoring 13-14 th November 2006 Portugal Dina Patel UKNEQAS Immunology & Immunochemistry Operations Manager In UK NEQAS
AccuDiag dsdna ELISA Kit
 AccuDiag dsdna ELISA Kit 2553-1 The first quantitative assay for DNA is the Farr assay.4 The original procedure used precipitation, radio-labeled DNA and separation techniques. The difficulties in obtaining
AccuDiag dsdna ELISA Kit 2553-1 The first quantitative assay for DNA is the Farr assay.4 The original procedure used precipitation, radio-labeled DNA and separation techniques. The difficulties in obtaining
Catalog. Immunofluorescence assays (IFA) The IFA-Galaxy Instruments & Assays
 Catalog Immunofluorescence assays (IF) The IF-Galaxy Instruments & ssays Welcome to our galaxy of automated immunofluorescence assays (IF) Reducing manual routines is an effective step to save time and
Catalog Immunofluorescence assays (IF) The IF-Galaxy Instruments & ssays Welcome to our galaxy of automated immunofluorescence assays (IF) Reducing manual routines is an effective step to save time and
Prevalence and Clinical Significance of anti-dfs70 in ANA Positive Patients Undergoing Routine ANA Testing in a New Zealand Public Hospital.
 Prevalence and Clinical Significance of anti-dfs70 in ANA Positive Patients Undergoing Routine ANA Testing in a New Zealand Public Hospital. Stacey Lucas A thesis submitted to Auckland University of Technology
Prevalence and Clinical Significance of anti-dfs70 in ANA Positive Patients Undergoing Routine ANA Testing in a New Zealand Public Hospital. Stacey Lucas A thesis submitted to Auckland University of Technology
AccuDiag. SSB (La) ELISA TEST PRINCIPLE INTENDED USE SUMMARY AND EXPLANATION SPECIMEN COLLECTION AND PREPARATION See external Label
 Test Method Principle Detection Range Sample Total Time Shelf Life INTENDED USE AccuDiag SSB (La) Kit See external Label 2556-2 96 Tests SSB (La) Enzyme Linked Immunosorbent Assay Indirect; Antigen Coated
Test Method Principle Detection Range Sample Total Time Shelf Life INTENDED USE AccuDiag SSB (La) Kit See external Label 2556-2 96 Tests SSB (La) Enzyme Linked Immunosorbent Assay Indirect; Antigen Coated
QUANTA Lite SS-A ELISA For In Vitro Diagnostic Use CLIA Complexity: High
 Format to CLSI Standards GP2-A5 (Formerly NCCLS) Vol. 26 No. 12 Issue Date: 12/05/11 QUANTA Lite SS-A ELISA 708570 For In Vitro Diagnostic Use CLIA Complexity: High Principles of the Procedure Purified
Format to CLSI Standards GP2-A5 (Formerly NCCLS) Vol. 26 No. 12 Issue Date: 12/05/11 QUANTA Lite SS-A ELISA 708570 For In Vitro Diagnostic Use CLIA Complexity: High Principles of the Procedure Purified
anti-dsdna Enzyme Immunoassay Test Kit For In Vitro Diagnostic Use Catalog No Tests
 SUMMARY OF PROCEDURE anti-dsdna Enzyme Immunoassay Test Kit For In Vitro Diagnostic Use Catalog No. 72-7 96 Tests 1. Prepare a 1:11 dilution of Standards, controls and samples in Sample Diluent. Mix well.
SUMMARY OF PROCEDURE anti-dsdna Enzyme Immunoassay Test Kit For In Vitro Diagnostic Use Catalog No. 72-7 96 Tests 1. Prepare a 1:11 dilution of Standards, controls and samples in Sample Diluent. Mix well.
Performance of the BMD FIDIS Connective 10 ENA luminex method in external quality assurance proficiency programmes in a diagnostic laboratory setting
 Performance of the BMD FIDIS Connective 10 ENA luminex method in external quality assurance proficiency programmes in a diagnostic laboratory setting Paul M Austin, Helena T Thompson and Anthony R Brown
Performance of the BMD FIDIS Connective 10 ENA luminex method in external quality assurance proficiency programmes in a diagnostic laboratory setting Paul M Austin, Helena T Thompson and Anthony R Brown
AccuDiag. SSB (La) ELISA
 Test Method Principle Detection Range Sample Total Time Shelf Life INTENDED USE AccuDiag SSB (La) ELISA Kit Cat# 2556-2 See external Label 96 Tests SSB (La) ELISA Enzyme Linked Immunosorbent Assay Indirect;
Test Method Principle Detection Range Sample Total Time Shelf Life INTENDED USE AccuDiag SSB (La) ELISA Kit Cat# 2556-2 See external Label 96 Tests SSB (La) ELISA Enzyme Linked Immunosorbent Assay Indirect;
ANTINUCLEAR ANTIBODIES IN BULGARIAN PATIENTS WITH CHRONIC INFECTIONS
 PROCEEDINGS OF THE BALKAN SCIENTIFIC CONFERENCE OF BIOLOGY IN PLOVDIV (BULGARIA) FROM 19 TH TILL 21 ST OF MAY 2005 (EDS B. GRUEV, M. NIKOLOVA AND A. DONEV), 2005 (P. 246 252) ANTINUCLEAR ANTIBODIES IN
PROCEEDINGS OF THE BALKAN SCIENTIFIC CONFERENCE OF BIOLOGY IN PLOVDIV (BULGARIA) FROM 19 TH TILL 21 ST OF MAY 2005 (EDS B. GRUEV, M. NIKOLOVA AND A. DONEV), 2005 (P. 246 252) ANTINUCLEAR ANTIBODIES IN
Anti-SS-A-52 (IgG) ELISA
 Anti-SS-A-52 (IgG) ELISA For the quantitative and qualitative detection of antibodies against the 52 kda Ro/ SS-A in serum. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number:
Anti-SS-A-52 (IgG) ELISA For the quantitative and qualitative detection of antibodies against the 52 kda Ro/ SS-A in serum. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number:
Anti-dsDNA ELISA. Anti-dsDNA ELISA Assay Kit 1/7 Catalog Number: DSD31-K01. INTENDED USE
 INTENDED USE Anti-dsDNA ELISA Catalog Number: DSD31-K01 Enzyme immunoassay for the determination of IgG antibodies to dsdna in human serum and plasma The Eagle Biosciences Anti-dsDNA ELISA Assay Kit is
INTENDED USE Anti-dsDNA ELISA Catalog Number: DSD31-K01 Enzyme immunoassay for the determination of IgG antibodies to dsdna in human serum and plasma The Eagle Biosciences Anti-dsDNA ELISA Assay Kit is
BIMM18 Dec 20 th - Flow cytometry in clinical diagnostics
 BIMM18 Dec 20 th - Flow cytometry in clinical diagnostics I. B cell leukemia and lymphomas Immunophenotyping as part of the diagnostic work- up of hematologic malignancies offers a rapid and effective
BIMM18 Dec 20 th - Flow cytometry in clinical diagnostics I. B cell leukemia and lymphomas Immunophenotyping as part of the diagnostic work- up of hematologic malignancies offers a rapid and effective
Chapter 17: Immunization & Immune Testing. 1. Immunization 2. Diagnostic Immunology
 Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
1. Immunization. What is Immunization? 12/9/2016. Chapter 17: Immunization & Immune Testing. 1. Immunization 2. Diagnostic Immunology
 Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
Structure of IgG and IgM
 Structure of IgG and IgM Fig. 5-1 A,B Crystal Structure of Secreted IgG Fig. 5-1 C Structure of an Ig Domain Fig. 5-2 Proteolytic Fragments of IgG (1) Fig. 5-3A Proteolytic Fragments of IgG (2) Fig. 5-3B
Structure of IgG and IgM Fig. 5-1 A,B Crystal Structure of Secreted IgG Fig. 5-1 C Structure of an Ig Domain Fig. 5-2 Proteolytic Fragments of IgG (1) Fig. 5-3A Proteolytic Fragments of IgG (2) Fig. 5-3B
Diversity and Origin of Rheumatologic Autoantibodies
 CLINICAL MICROBIOLOGY REVIEWS, JUIY 1991, p. 256-269 Vol. 4, No. 3 0893-8512/91/030256-14$02.00/0 Copyright 1991, American Society for Microbiology Diversity and Origin of Rheumatologic Autoantibodies
CLINICAL MICROBIOLOGY REVIEWS, JUIY 1991, p. 256-269 Vol. 4, No. 3 0893-8512/91/030256-14$02.00/0 Copyright 1991, American Society for Microbiology Diversity and Origin of Rheumatologic Autoantibodies
So we can separate antigens into their components and allow them to react with their antibodies
 Ag-ab reactions As for single immunodiffusion, double immunodiffusion can be also combined with electrophoresis to speed up the reaction, and in this case the test is called immunoelectrophoresis. So electrophoresis
Ag-ab reactions As for single immunodiffusion, double immunodiffusion can be also combined with electrophoresis to speed up the reaction, and in this case the test is called immunoelectrophoresis. So electrophoresis
Lecture 24. Autoimmunity. Origins of autoimmunity. Diseases. Immune Diseases - Chapter 16 - Allergies - Autoimmunity - Immunodeficiency
 Lecture 24 Immune Diseases - Chapter 16 - Allergies - Autoimmunity - Immunodeficiency Disease Diagnostics - Chapter 17 - Sample Collections - Phenotypic Method - Genotypic Method - Immunological Method
Lecture 24 Immune Diseases - Chapter 16 - Allergies - Autoimmunity - Immunodeficiency Disease Diagnostics - Chapter 17 - Sample Collections - Phenotypic Method - Genotypic Method - Immunological Method
Next-Generation Autoantibody Testing by Combination of Screening and Confirmation the CytoBead Technology
 Clinic Rev Allerg Immunol (2017) 53:87 104 DOI 10.1007/s12016-016-8574-3 Next-Generation Autoantibody Testing by Combination of Screening and Confirmation the CytoBead Technology Mandy Sowa 1 & Rico Hiemann
Clinic Rev Allerg Immunol (2017) 53:87 104 DOI 10.1007/s12016-016-8574-3 Next-Generation Autoantibody Testing by Combination of Screening and Confirmation the CytoBead Technology Mandy Sowa 1 & Rico Hiemann
ANA Screen ELISA Cat # Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
THE QUALITY ASSURANCE AND ORGANIZATION OF AUTOANTIBODY LABORATORY
 THE QUALITY ASSURANCE AND ORGANIZATION OF AUTOANTIBODY LABORATORY Tanja Kveder, Ph.D.; Assist. Prof. Borut Božič, Ph.D. Department of Rheumatology, University Medical Centre, 1000 Ljubljana, Slovenia University
THE QUALITY ASSURANCE AND ORGANIZATION OF AUTOANTIBODY LABORATORY Tanja Kveder, Ph.D.; Assist. Prof. Borut Božič, Ph.D. Department of Rheumatology, University Medical Centre, 1000 Ljubljana, Slovenia University
Prediction and Evaluation of Diagnostic Assays for Autoimmune Disorders
 International Journal of Genetic Engineering and Biotechnology. ISSN 0974-3073 Volume 2, Number 3 (2011), pp. 215-224 International Research Publication House http://www.irphouse.com Prediction and Evaluation
International Journal of Genetic Engineering and Biotechnology. ISSN 0974-3073 Volume 2, Number 3 (2011), pp. 215-224 International Research Publication House http://www.irphouse.com Prediction and Evaluation
Reagents 1. dsdna (Crithidia luciliae) Slide, 6 wells or 12 wells/slide, with desiccant
 NOVA Lite dsdna Crithidia luciliae Kits/Substrate Slides For In Vitro Diagnostic Use CLIA Complexity: High REF 708200, 708205, 508200.10, 508205.20, 508205.80 Rx Only Intended Use NOVA Lite dsdna Crithidia
NOVA Lite dsdna Crithidia luciliae Kits/Substrate Slides For In Vitro Diagnostic Use CLIA Complexity: High REF 708200, 708205, 508200.10, 508205.20, 508205.80 Rx Only Intended Use NOVA Lite dsdna Crithidia
Scl-86, A Marker Antigen for Diffuse Scleroderma
 Scl-86, A Marker Antigen for Diffuse Scleroderma W. J. van Venrooij, S. 0. Stapel, H. Houben, W. J. Habets, C. G. M. Kallenberg, E. Penner, and L. B. van de Putte Department of Biochemistry and Department
Scl-86, A Marker Antigen for Diffuse Scleroderma W. J. van Venrooij, S. 0. Stapel, H. Houben, W. J. Habets, C. G. M. Kallenberg, E. Penner, and L. B. van de Putte Department of Biochemistry and Department
A 52-kD PROTEIN IS A NOVEL COMPONENT OF THE SS-A/Ro ANTIGENIC PARTICLE
 A 52-kD PROTEIN IS A NOVEL COMPONENT OF THE SS-A/Ro ANTIGENIC PARTICLE By ELDAD BEN-CHETRIT, EDWARD K. L. CHAN, KEVIN F. SULLIVAN, AND ENG M. TAN From the W. M. Keck Autoimmune Disease Center, Scripps
A 52-kD PROTEIN IS A NOVEL COMPONENT OF THE SS-A/Ro ANTIGENIC PARTICLE By ELDAD BEN-CHETRIT, EDWARD K. L. CHAN, KEVIN F. SULLIVAN, AND ENG M. TAN From the W. M. Keck Autoimmune Disease Center, Scripps
Epidermal nuclear immunoglobulin deposits in some connective tissue diseases: correlation with ENA antibodies
 Annals of the Rheumatic Diseases, 1983, 42, 163-167 Epidermal nuclear immunoglobulin deposits in some connective tissue diseases: correlation with ENA antibodies IRENE ANDERSEN,1 HANS GRAUDAL' PAUL ANDERSEN,2
Annals of the Rheumatic Diseases, 1983, 42, 163-167 Epidermal nuclear immunoglobulin deposits in some connective tissue diseases: correlation with ENA antibodies IRENE ANDERSEN,1 HANS GRAUDAL' PAUL ANDERSEN,2
October Monthly Update, Quest Diagnostics Nichols Institute, Valencia
 October 2014 - Monthly Update, NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated.
October 2014 - Monthly Update, NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated.
QUANTA Lite SS-A ELISA For In Vitro Diagnostic Use CLIA Complexity: High
 QUANTA Lite SS-A ELISA 708570 For In Vitro Diagnostic Use CLIA Complexity: High Intended Use QUANTA Lite SS-A is an enzyme-linked immunosorbent assay (ELISA) for the semi-quantitative detection of SS-A
QUANTA Lite SS-A ELISA 708570 For In Vitro Diagnostic Use CLIA Complexity: High Intended Use QUANTA Lite SS-A is an enzyme-linked immunosorbent assay (ELISA) for the semi-quantitative detection of SS-A
Immunodiagnosis SCBM 343 CLINICAL PATHOLOGY. Lect. Dr. Witchuda Payuhakrit
 Immunodiagnosis SCBM 343 CLINICAL PATHOLOGY Lect. Dr. Witchuda Payuhakrit witchuda.pay@mahidol.ac.th Objectives 2 Understand the antigen and antibody interaction Understand the principle of immunoassay
Immunodiagnosis SCBM 343 CLINICAL PATHOLOGY Lect. Dr. Witchuda Payuhakrit witchuda.pay@mahidol.ac.th Objectives 2 Understand the antigen and antibody interaction Understand the principle of immunoassay
SUMMARY AND EXPLANATION OF THE TEST
 HEp-2000 COLORZYME ANA-Ro TEST SYSTEM For in vitro Diagnostic Use For Professional Use INTENDED USE: This is an indirect enzyme antibody test for the semi-quantitative detection of antinuclear antibody
HEp-2000 COLORZYME ANA-Ro TEST SYSTEM For in vitro Diagnostic Use For Professional Use INTENDED USE: This is an indirect enzyme antibody test for the semi-quantitative detection of antinuclear antibody
ZENIT RA Jo-1 REF ZENIT RA Jo-1
 REF 41419 ZENIT RA Jo-1 Distributed by INSTRUCTIONS FOR USE 50 INTENDED USE The ZENIT RA Jo-1 test is a chemiluminescent immunoassay (CLIA) for use on the dedicated ZENIT RA Analyzer for quantitative determination
REF 41419 ZENIT RA Jo-1 Distributed by INSTRUCTIONS FOR USE 50 INTENDED USE The ZENIT RA Jo-1 test is a chemiluminescent immunoassay (CLIA) for use on the dedicated ZENIT RA Analyzer for quantitative determination
Cellular RNA-Protein Particles
 Antibodies from Patients with Connective Tissue Diseases Bind Specific Subsets of Cellular RNA-Protein Particles JOHN A. HARDIN, DANIEL R. RAHN, CALVIN SHEN, MICHAEL R. LERNER, SANDRA L. WOLIN, MARGARET
Antibodies from Patients with Connective Tissue Diseases Bind Specific Subsets of Cellular RNA-Protein Particles JOHN A. HARDIN, DANIEL R. RAHN, CALVIN SHEN, MICHAEL R. LERNER, SANDRA L. WOLIN, MARGARET
anti-dsdna assays has been carried out by testing panels of serum samples from selected patients with
 nnals of the Rheumatic Diseases, 1985, 44, 245-251 Detection of anti-dsdn as a diagnostic tool: a prospective study in 441 non-systemic lupus erythematosus patients with anti-dsdn antibody (anti-dsdn)
nnals of the Rheumatic Diseases, 1985, 44, 245-251 Detection of anti-dsdn as a diagnostic tool: a prospective study in 441 non-systemic lupus erythematosus patients with anti-dsdn antibody (anti-dsdn)
