D =? D = M = 10 g = 5 g/cm 3 M = 10 g V 2 cm 3 V = 2 cm 3 5 g/cm 3
|
|
|
- Osborn Blair
- 6 years ago
- Views:
Transcription
1 Using the formula for density, answer the following calculation problems. M D = (g/ml or g/cm 3 ) Density = Mass D = M M = (g) Volume V D V V = (ml or cm 3 ) For all of your problems, please complete them with the following methods to obtain full marks: 1 mark for LISTING your variables (including units and the unknown) 1 mark for choosing the right FORMULA (write out the entire formula) 1 mark for the WORK shown (including units) 1 mark for the correct ANSWER (including the units) **Sometimes, the object s volume is noted in ml. Please be aware that in most cases: 1 cm 3 (ice) = 1 ml (pure water) Solids, Gases are measured in cm 3, Liquids in ml Part A: Calculations (Eg 1) Calculate the density of an object having a mass of 10 g and a volume of 2 cm 3. D =? D = M = 10 g = 5 g/cm 3 M = 10 g V 2 cm 3 V = 2 cm 3 5 g/cm 3 (Eg 2) Calculate the density of a 120 g object with a volume of 60 ml. D =? D = M = 120 g = 2 g/ml M = 120 g V 60 ml V = 60 ml 2 g/ml (Eg 3) If an object has a density of 0.35 g/cm 3, what is the mass if it s volume is 350 cm 3? D = 0.35 g/cm 3 M = D V M =? = (0.35 g/cm 3 )(350 cm 3 ) V = 350 cm 3 = g g (Eg 4) Given a density of 2.25 g/cm 3, what is the volume if an object s mass is 675 g? D = 2.25 g/cm 3 V = M = 675 g = 300 cm 3 M = 675 g D 2.25 g/cm 3 V =? 300 cm 3
2 1. A sample of quartz was found to have a mass of 130 g and to occupy a volume of 50 cm 3. What is the density of quartz? (1 decimal) D =? D = M = 130 g = 2.6 g/cm 3 M = 130 g V 50 cm 3 V = 50 cm g/cm 3 2. A substance has a mass of 42 g and occupies 294 cm 3. What is its density? (2 decimals) D =? D = M = 42 g = 0.14 g/cm 3 M = 42 g V 294 cm 3 V = 294 cm g/cm 3 3. If the volume of an object is 100 cm 3 and its density is 1.20 g/cm 3, what is its mass? D = 1.20 g/cm 3 M = D V M =? = (1.20 g/cm 3 )(100 cm 3 ) V = 100 cm 3 = 120 g 120 g 4. The average human body has 6820 ml of blood that has a mass of g. What is the density of human blood? (2 decimals) D =? D = M = g = 1.06 g/ml M = g V 6820 ml V = ml 1.06 g/ml 5. Find the volume of piece of brass (density 8.4 g/cm 3 ) and a mass of 600 g. (1 decimal) D = 8.4 g/cm 3 V = M = 600 g = 71.4 cm 3 M = 600 g D 8.4 g/cm 3 V =? 71.4 cm 3 6. Calculate the density of a solid block that has a volume of 600 cm 3 and a mass of 150 g. D =? D = M = 150 g = 0.25 g/ cm 3 M = 150 g V 600 cm 3 V = 600 cm g/cm 3 7. Calculate the volume of carbon dioxide gas (density = g/cm 3 ) and a mass of 500 g. D = g/cm 3 V = M = 500 g = cm 3 M = 500 g D g/cm 3 V =? cm 3
3 Part B: Use the Density Chart on the back to complete the following questions. 11. If the following liquids were mixed in a cylinder and allowed to settle out, state the order of liquids from bottom to top. Rank starting from 1 as being the bottom. Assume they will NOT dissolve in each other. a) _2 anti-freeze, _1 glycerin, _3 turpentine b) _1 mercury, _4 gasoline, _2 sea water, _3 ethyl alcohol c) _3 sea water, _4 oil, _5 kerosene, _1 molasses, _2 saturated salt water d) _5 ethyl alcohol, _4 kerosene, _1 molasses, _3 water (4 C), _2 anti-freeze 12. List all the solids (from the chart on the back page) that would sink in mercury (13.5). Gold (19.3) Osmium (22.7) Platinum (21.5) 13. Why does a marshmallow float on top of hot chocolate? Marshmallows are less dense (lots of air) than water 14. Why does a melted marshmallow sink to the bottom of hot chocolate? Melted marshmallows have no air and is more dense than water 15. Why must you always shake a mix of oil and vinegar dressing before serving it? Oil and vinegar don t mix due their different densities (and strong attraction for their own type of particles). After sitting, the two substances separate and so it needs to be shaken first. 16. Why does a hot air balloon rise? (Hint: List the densities of air and helium) How air balloons are filled with helium (which is less denser than air). When filled, the balloon becomes less dense than air and rises. 17. Why do bubbles rise to the top of a bottle of pop (Hint: List the densities of water and carbon dioxide) Carbon dioxide gas is less dense than the water (solvent) in the pop. (0.002 g/cm 3 ) (1.0 g/ml)
4 SOLIDS 20 0 C g/cm 3 Density Information LIQUIDS g/ml Aluminum 2.7 Amber 1.1 Ash wood Balsa wood 0.34 Beech wood Birch wood Brass 8.44 Butter 0.86 Cast iron 7.0 Cedar Chalk 2.0 Cork 0.25 Bone 1.8 Copper 8.9 Diamond 3.5 Eggs 1.02 Glass 2.4 Gold 19.3 Granite Graphite 2.2 Ice (0 0 C) 0.92 Iron 7.9 Lead 11.4 Magnesium 1.7 Manganese 7.2 Nickel 8.9 Oak Osmium 22.7 Pine Plastic Platinum 21.5 Quartz 2.65 Rubber 1.2 Salt 2.18 Silicon 2.3 Silver 10.5 Sodium 0.97 Spruce Steel 7.8 Styrofoam Tin 7.3 Uranium 19.1 Vinyl Zinc 7.1 Anti-freeze 1.1 Ethyl alcohol 0.79 Gasoline 0.70 Glycerin 1.26 Kerosene 0.82 Mercury 13.5 Methyl alcohol 0.79 Molasses 1.4 Oil 0.9 Saturated salt water 1.1 Sea water Turpentine 0.87 Water (4 0 C) 1.0 GASES (0 0 C / sea level) g/cm 3 Air Carbon dioxide Helium Hydrogen Neon Nitrogen Oxygen
5
Name # Date Period! FINDING&VOLUME&AND&DENSITY& !!! !!!!!!!
 Name # Date Period FINDING&VOLUME&AND&DENSITY& TherectangularshapedboxinFigure1isfilledwithdrysand.Findthe volumeanddensityofthesand. 1.Firstfindthevolumeofthebox.Thisisalsothevolumeofthesand. Usethediagramtofindthedimensionsofthebox.
Name # Date Period FINDING&VOLUME&AND&DENSITY& TherectangularshapedboxinFigure1isfilledwithdrysand.Findthe volumeanddensityofthesand. 1.Firstfindthevolumeofthebox.Thisisalsothevolumeofthesand. Usethediagramtofindthedimensionsofthebox.
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example,
 SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example, density, thermal or electrical conductivity, solubility,
SC.8.P.8.4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example, density, thermal or electrical conductivity, solubility,
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 3 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 3 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet
 Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet Name: Sec: Date: Q1.Choose the best answer. 1. Which of the following is an element? a) steam b) sugar c)dry ice d) sulphur 2. Which
Science Class 8 Topic: Elements And Compounds Reinforcement Worksheet Name: Sec: Date: Q1.Choose the best answer. 1. Which of the following is an element? a) steam b) sugar c)dry ice d) sulphur 2. Which
2.3 Density and Density Calculations
 2.3 Density and Density Calculations When people say that lead is heavier than wood, they do not mean that a pea sized piece of lead weighs more than a truckload of pine logs. What they mean is that a
2.3 Density and Density Calculations When people say that lead is heavier than wood, they do not mean that a pea sized piece of lead weighs more than a truckload of pine logs. What they mean is that a
THE PERIODIC TABLE. Chapter 15
 THE PERIODIC TABLE Chapter 15 MENDELEEV 1869 The first version of the periodic table was published by Dmitri Mendeleev, a Russian chemist He arranged the elements in order of increasing atomic mass This
THE PERIODIC TABLE Chapter 15 MENDELEEV 1869 The first version of the periodic table was published by Dmitri Mendeleev, a Russian chemist He arranged the elements in order of increasing atomic mass This
Density Answers to the End of module questions
 IDS 101 Density Answers to the End of module questions 1. You are experimenting with the mysterious substance X. You carefully measure the mass of a uniform 2 ml sample of pure substance X. You slice it
IDS 101 Density Answers to the End of module questions 1. You are experimenting with the mysterious substance X. You carefully measure the mass of a uniform 2 ml sample of pure substance X. You slice it
The table below shows some of the properties of the elements cobalt and nickel.
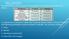 BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
Density What is density? How do you measure density?
 Density What is density? How do you measure density? What is density? Density describes how much mass is in a given volume of a material. Mass: the amount of matter in an object. Volume: the amount of
Density What is density? How do you measure density? What is density? Density describes how much mass is in a given volume of a material. Mass: the amount of matter in an object. Volume: the amount of
Covered with a thin layer of oxide at ordinary temperatures.
 1 More about Metals Physical properties of metals In general metals have luster, are malleable and ductile, good conductors of heat and electricity and have high boiling and melting points and nonmetals
1 More about Metals Physical properties of metals In general metals have luster, are malleable and ductile, good conductors of heat and electricity and have high boiling and melting points and nonmetals
(A) Copper. (B) Zinc. (C) Iron. (D) Wood. (A) Zinc. (B) Wood. (C) Rubber. (D) Plastic.
 Downloaded from MATERIALS : METALS AND NON-METALS 1.Give one example of each: metals and non-metals. 2.Name the metal, which is the best conductor of heat and electricity. 3.Name the property by which
Downloaded from MATERIALS : METALS AND NON-METALS 1.Give one example of each: metals and non-metals. 2.Name the metal, which is the best conductor of heat and electricity. 3.Name the property by which
Metal and Non Metals
 Metal and Non Metals Malleable Ductile Sonorous Conductor Insulator KEYWORDS Rusting Brass Calcium Chloride Galvanising Reactivity Metals on the Periodic table Metals Non Metals Metals and their properties
Metal and Non Metals Malleable Ductile Sonorous Conductor Insulator KEYWORDS Rusting Brass Calcium Chloride Galvanising Reactivity Metals on the Periodic table Metals Non Metals Metals and their properties
Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3
 Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3 Directions to Students: Do not open this booklet until you are told to do so. Please respond to the following items by marking the best answer
Chemistry Attitudes, Skills, & Knowledge Survey (CASKS) Form 3 Directions to Students: Do not open this booklet until you are told to do so. Please respond to the following items by marking the best answer
Appendix 1 PROPERTY TABLES AND CHARTS (SI UNITS)
 23-R4232-APP1 6/8/07 5:09 PM Page 987 Appendix 1 PROPERTY TABLES AND CHARTS (SI UNITS) Table A 1 Molar mass, gas constant, and critical-point properties 988 Table A 2 Ideal-gas specific heats of various
23-R4232-APP1 6/8/07 5:09 PM Page 987 Appendix 1 PROPERTY TABLES AND CHARTS (SI UNITS) Table A 1 Molar mass, gas constant, and critical-point properties 988 Table A 2 Ideal-gas specific heats of various
1. Elemental mercury is a shiny, silver-colored, dense liquid that flows easily. Are these characteristics of mercury physical or chemical properties?
 Chemistry Dr. Saulmon 2014-15 School Year Unit 2: Basics of Chemistry Problem Set 3 Wednesday, September 3, 2014 1. Elemental mercury is a shiny, silver-colored, dense liquid that flows easily. Are these
Chemistry Dr. Saulmon 2014-15 School Year Unit 2: Basics of Chemistry Problem Set 3 Wednesday, September 3, 2014 1. Elemental mercury is a shiny, silver-colored, dense liquid that flows easily. Are these
EXPERIMENT 3: Identification of a Substance by Physical Properties
 EXPERIMENT 3: Identification of a Substance by Physical Properties Materials: Hot plate Digital balance Capillary tubes (3) Thermometer Beakers (250 ml) Watch glass Graduated Cylinder (10 ml) Mel-Temp
EXPERIMENT 3: Identification of a Substance by Physical Properties Materials: Hot plate Digital balance Capillary tubes (3) Thermometer Beakers (250 ml) Watch glass Graduated Cylinder (10 ml) Mel-Temp
Chapter: The Periodic Table
 Table of Contents Chapter: The Periodic Table Section 1: Introduction to the Periodic Table Section 2: Representative Elements Section 3: Transition Elements 1 Introduction to the Periodic Table Development
Table of Contents Chapter: The Periodic Table Section 1: Introduction to the Periodic Table Section 2: Representative Elements Section 3: Transition Elements 1 Introduction to the Periodic Table Development
Copper Odyssey. Chemical Reactions of Copper
 Name Lab Partner(s) Copper Odyssey Chemical Reactions of Copper Date Period Elemental copper metal will be converted into copper (II) ion and then brought through a series of compound conversions until
Name Lab Partner(s) Copper Odyssey Chemical Reactions of Copper Date Period Elemental copper metal will be converted into copper (II) ion and then brought through a series of compound conversions until
Electricity and Chemistry
 Electricity and Chemistry Electrochemistry: It is a branch of chemistry that deals with the reactions involving the conversion of chemical energy into electrical energy and vice-versa. Electrochemical
Electricity and Chemistry Electrochemistry: It is a branch of chemistry that deals with the reactions involving the conversion of chemical energy into electrical energy and vice-versa. Electrochemical
2.2 Physical Properties. Physical Properties. Notes due EOC
 Physical Properties Notes due EOC There are pitchers of ice water and lemonade on a picnic table. How do you know which liquid is in each pitcher? It s easy! Lemonade is yellow and has a tart taste that
Physical Properties Notes due EOC There are pitchers of ice water and lemonade on a picnic table. How do you know which liquid is in each pitcher? It s easy! Lemonade is yellow and has a tart taste that
A TEACHER-DIRECTED, GUIDED DISCOVERY ACTIVITY FOR ADVANCED PHYSICAL SCIENCE STUDENTS
 A TEACHER-DIRECTED, GUIDED DISCOVERY ACTIVITY FOR ADVANCED PHYSICAL SCIENCE STUDENTS 1 2 FEATURES OF THIS ACTIVITY Problem solving Cooperative learning Requires student reasoning Requires student initiative
A TEACHER-DIRECTED, GUIDED DISCOVERY ACTIVITY FOR ADVANCED PHYSICAL SCIENCE STUDENTS 1 2 FEATURES OF THIS ACTIVITY Problem solving Cooperative learning Requires student reasoning Requires student initiative
(a) To find out which is the more reactive metal, zinc or tin, the following experiment could be carried out. piece of zinc shiny surface
 1 The reactivity series lists metals in order of reactivity. (a) To find out which is the more reactive metal, zinc or tin, the following experiment could be carried out. piece of zinc shiny surface tin(ii)
1 The reactivity series lists metals in order of reactivity. (a) To find out which is the more reactive metal, zinc or tin, the following experiment could be carried out. piece of zinc shiny surface tin(ii)
Determining the Coefficient of Friction. Part by by Ron Kurtus
 Determining the Coefficient of Part by by Ron Kurtus Different types of coefficient The different types of friction are static, kinetic, deformation, molecular and rolling. Each has its own coefficient
Determining the Coefficient of Part by by Ron Kurtus Different types of coefficient The different types of friction are static, kinetic, deformation, molecular and rolling. Each has its own coefficient
OC30 Conduct a qualitative experiment to detect the presence of dissolved solids in water samples, and test water for hardness (soap test)
 Chemistry: 6. Water Please remember to photocopy 4 pages onto one sheet by going A3 A4 and using back to back on the photocopier Syllabus OC14 Use cobalt chloride paper or anhydrous copper sulfate to test
Chemistry: 6. Water Please remember to photocopy 4 pages onto one sheet by going A3 A4 and using back to back on the photocopier Syllabus OC14 Use cobalt chloride paper or anhydrous copper sulfate to test
Lab Question from Board papers Class-09[2018] 1. How will you separate a mixture of iron filings, iodine and common salt?
![Lab Question from Board papers Class-09[2018] 1. How will you separate a mixture of iron filings, iodine and common salt? Lab Question from Board papers Class-09[2018] 1. How will you separate a mixture of iron filings, iodine and common salt?](/thumbs/77/74991575.jpg) Lab Question from Board papers Class-09[2018] SET-1 Short Answer Type Questions (2 marks) 1. How will you separate a mixture of iron filings, iodine and common salt? Ans. (i) Remove iron filings with the
Lab Question from Board papers Class-09[2018] SET-1 Short Answer Type Questions (2 marks) 1. How will you separate a mixture of iron filings, iodine and common salt? Ans. (i) Remove iron filings with the
Distribution Review. Corrosion Control. Corrosion Control Vocabulary. American Water College 1. Corrosion Control Training Objectives
 Distribution Review Corrosion Control Corrosion Control Training Objectives To gain an understanding of: Corrosion control vocabulary The factors affecting corrosion Methods used to control corrosion Alkalinity
Distribution Review Corrosion Control Corrosion Control Training Objectives To gain an understanding of: Corrosion control vocabulary The factors affecting corrosion Methods used to control corrosion Alkalinity
4. Where do the names of the elements come from? Some were named as substances before they were known to be elements. sulfur 16
 4. Where do the names of the elements come from? Some were named as substances before they were known to be elements. silver 47 sulfur 16 copper 29 Some are named from the natural substance that they are
4. Where do the names of the elements come from? Some were named as substances before they were known to be elements. silver 47 sulfur 16 copper 29 Some are named from the natural substance that they are
ALE 20. Crystalline Solids, Unit Cells, Liquids and the Uniqueness of Water
 Name Chem 162, Section: Group Number: ALE 20. Crystalline Solids, Unit Cells, Liquids and the Uniqueness of Water (Reference: pp. 463 473 of Sec. 12.6 Silberberg 5 th edition) How are the particles within
Name Chem 162, Section: Group Number: ALE 20. Crystalline Solids, Unit Cells, Liquids and the Uniqueness of Water (Reference: pp. 463 473 of Sec. 12.6 Silberberg 5 th edition) How are the particles within
Physical Science Chapter 19. Elements and Their Properties Quick Notes
 Physical Science Chapter 19 Elements and Their Properties Quick Notes 1 19:1 A.Metals conduct heat and electricity, reflect light (luster); are malleable ( ) Can be hammered or rolled into sheets are ductile
Physical Science Chapter 19 Elements and Their Properties Quick Notes 1 19:1 A.Metals conduct heat and electricity, reflect light (luster); are malleable ( ) Can be hammered or rolled into sheets are ductile
SIGNIFICANT FIGURES WORKSHEET
 SIGNIFICANT FIGURES WORKSHEET PART 1 - Determine the number of significant figures in the following numbers. 1.) 0.02 2.) 0.020 3.) 501 4.) 501.0 5.) 5,000 6.) 5,000. 7.) 6,051.00 8.) 0.0005 9.) 0.1020
SIGNIFICANT FIGURES WORKSHEET PART 1 - Determine the number of significant figures in the following numbers. 1.) 0.02 2.) 0.020 3.) 501 4.) 501.0 5.) 5,000 6.) 5,000. 7.) 6,051.00 8.) 0.0005 9.) 0.1020
CHAPTER 6. Natural Mineral Mineral Content Elements In The Minerals Bauxite Aluminium oxide Aluminium, oxygen. Cassiterite Tin oxide Tin, oxygen
 CHAPTER 6 6.1 Minerals Found In The Earth s Crust mineral : is a naturally occurring solid element or compound with a definite crystalline structure and chemical composition. natural elements : gold, silver
CHAPTER 6 6.1 Minerals Found In The Earth s Crust mineral : is a naturally occurring solid element or compound with a definite crystalline structure and chemical composition. natural elements : gold, silver
Calculating Ballast By Troy Averill
 Calculating Ballast By Troy Averill Lesson Overview: Students use the dimensions of the cargo hold of a ship s hull to calculate the volume of material that can be contained. Students will then use the
Calculating Ballast By Troy Averill Lesson Overview: Students use the dimensions of the cargo hold of a ship s hull to calculate the volume of material that can be contained. Students will then use the
Skills in Science. Lab equipment. (Always draw 2D) Drawings below are NOT to scale. Beaker - A general purpose container with a pouring lip.
 Skills in Science Safety: Do NOT enter or leave the lab without permission from a teacher. Keep the gaps between tables clear of stools and bags. Never run in the lab. Do not throw things around in the
Skills in Science Safety: Do NOT enter or leave the lab without permission from a teacher. Keep the gaps between tables clear of stools and bags. Never run in the lab. Do not throw things around in the
1. Hardness 2. Streak 3. Density 4. Crystal structure 5. A mineral must be a naturally occurring, inorganic. 7. c 8. f 9. a 10. d 11. g 12. e 13.
 Properties of Guided Reading and Study Use Target Reading Skills Properties of I. What Is a Mineral? A. Naturally Occurring B. Inorganic C. Solid D. Crystal Structure E. Definite Chemical Composition II.
Properties of Guided Reading and Study Use Target Reading Skills Properties of I. What Is a Mineral? A. Naturally Occurring B. Inorganic C. Solid D. Crystal Structure E. Definite Chemical Composition II.
Chapter 11 The Periodic Table
 Chapter 11 The Periodic Table Lesson 1 Using the Periodic Table Textbook pages 391 397 Lesson 2 Metals Textbook pages 401 402 Lesson 3 Nonmetals and Metalloids Textbook pages 409 410 & 413-414 1 Chapter
Chapter 11 The Periodic Table Lesson 1 Using the Periodic Table Textbook pages 391 397 Lesson 2 Metals Textbook pages 401 402 Lesson 3 Nonmetals and Metalloids Textbook pages 409 410 & 413-414 1 Chapter
Iron filings (Fe) 56g IRON + SULPHUR IRON SULPHIDE
 W.S.51. Chemical reactions. All of the different materials around us have been formed by chemical reactions from about one hundred simple elements. The diagram below shows a chemical reaction between the
W.S.51. Chemical reactions. All of the different materials around us have been formed by chemical reactions from about one hundred simple elements. The diagram below shows a chemical reaction between the
Galvanic Corrosion Prevention Guide for Water Cooling Systems
 WATER COOLED DEVICES Galvanic Corrosion Prevention Guide for Water Cooling Systems November 2017 White Paper Created by Helen E. Kane, Advanced Energy Industries, Inc. Water Cooled Devices Abstract This
WATER COOLED DEVICES Galvanic Corrosion Prevention Guide for Water Cooling Systems November 2017 White Paper Created by Helen E. Kane, Advanced Energy Industries, Inc. Water Cooled Devices Abstract This
(3) The compound boron nitride (BN) has a high melting point (2967 ºC), high density, and is very hard. What is the best classification of this solid?
 Solids and Liquids Name: Period: (1) Identify the type of solid formed by each compound. (a) Ag (b) CO 2 (c) SiO 2 (d) wax (e) MgCl 2 (f) Fe (g) graphite (h) SO 2 (i) CaCO 3 (j) I 2 (k) rubber (l) SiC
Solids and Liquids Name: Period: (1) Identify the type of solid formed by each compound. (a) Ag (b) CO 2 (c) SiO 2 (d) wax (e) MgCl 2 (f) Fe (g) graphite (h) SO 2 (i) CaCO 3 (j) I 2 (k) rubber (l) SiC
Saskatchewan s Mineral Resources Lesson: Rocks and Minerals in Your Life
 Saskatchewan s Mineral Resources Lesson: Rocks and Minerals in Your Life Overview Students identify products they use in their daily activities, and discover the rocks and minerals used to make them. Source:
Saskatchewan s Mineral Resources Lesson: Rocks and Minerals in Your Life Overview Students identify products they use in their daily activities, and discover the rocks and minerals used to make them. Source:
Properties A Metal B Non- metal Electronic configuration?? Nature of oxides?? Oxidizing or reducing action?? Conduction of heat and electricity??
 CLASS: X NCERT (CBSE) SCIENCE: Chemistry Page: 1 Question 1: Compare the properties of a typical metal and a non-metal on the basis of the following. Fill in Column A, B. Properties A Metal B Non- metal
CLASS: X NCERT (CBSE) SCIENCE: Chemistry Page: 1 Question 1: Compare the properties of a typical metal and a non-metal on the basis of the following. Fill in Column A, B. Properties A Metal B Non- metal
3 The Formation, Mining, and Use of Minerals
 CHAPTER 3 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
CHAPTER 3 3 The Formation, Mining, and Use of Minerals SECTION Minerals of the Earth s Crust BEFORE YOU READ After you read this section, you should be able to answer these questions: How do minerals form?
Warm Up (Sept 12) How will an atom change if you change the number of: a) Protons?
 Warm Up (Sept 12) How will an atom change if you change the number of: a) Protons? b) Electrons? c) Neutrons? 1 CH3OS Warm Up (Sept 13) 1. What is an isotope? 2. Neon has two major isotopes, Neon 20 and
Warm Up (Sept 12) How will an atom change if you change the number of: a) Protons? b) Electrons? c) Neutrons? 1 CH3OS Warm Up (Sept 13) 1. What is an isotope? 2. Neon has two major isotopes, Neon 20 and
Compounds & Reactions Week 1. Writing Formulas & Balancing Equations. Write the chemical formula for each molecular (covalent) compound.
 Compounds & Reactions Week 1 Name Writing Formulas & Balancing Equations Write the chemical formula for each ionic compound. 1. Lithium fluoride 2. Copper (II) chloride 3. Manganese (II) oxide 4. Potassium
Compounds & Reactions Week 1 Name Writing Formulas & Balancing Equations Write the chemical formula for each ionic compound. 1. Lithium fluoride 2. Copper (II) chloride 3. Manganese (II) oxide 4. Potassium
Experiment 1: The Densities of Liquids and Solids (from Masterson & Hurley)
 Experiment 1: The Densities of Liquids and Solids (from Masterson & Hurley) One of the fundamental properties of any sample of matter is its density, which is its mass per unit of volume. The density of
Experiment 1: The Densities of Liquids and Solids (from Masterson & Hurley) One of the fundamental properties of any sample of matter is its density, which is its mass per unit of volume. The density of
One of the main ores of zinc is zinc blende, ZnS. There are two stages in the extraction of zinc from this ore.
 1 Two of the main uses of zinc are for galvanising and for making alloys. One of the main ores of zinc is zinc blende, ZnS. There are two stages in the extraction of zinc from this ore. (a) Stage 1 Zinc
1 Two of the main uses of zinc are for galvanising and for making alloys. One of the main ores of zinc is zinc blende, ZnS. There are two stages in the extraction of zinc from this ore. (a) Stage 1 Zinc
calcium oxide removes acidic oxides basic oxides acidic oxides basic oxides Iron obtained from the blast furnace is contaminated with
 1 Iron from a blast furnace is treated with and with calcium oxide to make steel. Which substances in the are removed? removes calcium oxide removes acidic oxides basic oxides acidic oxides basic oxides
1 Iron from a blast furnace is treated with and with calcium oxide to make steel. Which substances in the are removed? removes calcium oxide removes acidic oxides basic oxides acidic oxides basic oxides
Physical Behavior of Metals
 Activity 4 Physical Behavior of Metals GOALS In this activity you will: Discover what an alloy is. Make a brass-coated penny. Determine how the properties of a metal are affected by making it into an alloy.
Activity 4 Physical Behavior of Metals GOALS In this activity you will: Discover what an alloy is. Make a brass-coated penny. Determine how the properties of a metal are affected by making it into an alloy.
Equation Writing and Predicting Products Chemistry I Acc
 Introduction: Equation Writing and Predicting Products Chemistry I Acc If you examine your bicycle after it has been left out in the rain a number of times you will find that it has begun to rust. Rust
Introduction: Equation Writing and Predicting Products Chemistry I Acc If you examine your bicycle after it has been left out in the rain a number of times you will find that it has begun to rust. Rust
TYPES OF CHEMICAL REACTIONS PART I INTRODUCTION
 EXPERIMENT 10 (2 Weeks) Chemistry 100 Laboratory TYPES OF CHEMICAL REACTIONS PART I INTRODUCTION It is useful to classify reactions into different types, because products of reactions can be predicted.
EXPERIMENT 10 (2 Weeks) Chemistry 100 Laboratory TYPES OF CHEMICAL REACTIONS PART I INTRODUCTION It is useful to classify reactions into different types, because products of reactions can be predicted.
5072 CHEMISTRY (NEW PAPERS WITH SPA) TOPIC 9: METALS 5067 CHEMISTRY (NEW PAPERS WITH PRACTICAL EXAM) TOPIC 9: METALS
 5072 CHEMISTRY (NEW PAPERS WITH SPA) TOPIC 9: METALS 5067 CHEMISTRY (NEW PAPERS WITH PRACTICAL EXAM) TOPIC 9: METALS SUB-TOPIC 9.3 TO 5 EXTRACTION OF METALS; RECYLING OF METALS; IRON LEARNING OUTCOMES
5072 CHEMISTRY (NEW PAPERS WITH SPA) TOPIC 9: METALS 5067 CHEMISTRY (NEW PAPERS WITH PRACTICAL EXAM) TOPIC 9: METALS SUB-TOPIC 9.3 TO 5 EXTRACTION OF METALS; RECYLING OF METALS; IRON LEARNING OUTCOMES
ADVANTAGES OF POLYETHYLENE PIPES COMPARED TO PVC PIPES
 1 ADVANTAGES OF POLYETHYLENE PIPES COMPARED TO PVC PIPES The main advantages and improvements in drainage networks that will be obtained by the use of PE MAGNUM pipes, in comparison to PVC pipes are: 1.
1 ADVANTAGES OF POLYETHYLENE PIPES COMPARED TO PVC PIPES The main advantages and improvements in drainage networks that will be obtained by the use of PE MAGNUM pipes, in comparison to PVC pipes are: 1.
New GCSE 4462/02 SCIENCE A HIGHER TIER CHEMISTRY 1
 Surname Other Names Centre Number 0 Candidate Number New GCSE 4462/02 SCIENCE A HIGHER TIER CHEMISTRY 1 ADDITIONAL MATERIALS A.M. WEDNESDAY, 18 January 2012 1 hour In addition to this paper you may require
Surname Other Names Centre Number 0 Candidate Number New GCSE 4462/02 SCIENCE A HIGHER TIER CHEMISTRY 1 ADDITIONAL MATERIALS A.M. WEDNESDAY, 18 January 2012 1 hour In addition to this paper you may require
Draw a ring around the correct word in the box to complete the sentence.
 Q. Iron is the main structural metal used in the world. (a) The diagram represents the particles in iron, Fe. Draw a ring around the correct word in the box to complete the sentence. Iron is described
Q. Iron is the main structural metal used in the world. (a) The diagram represents the particles in iron, Fe. Draw a ring around the correct word in the box to complete the sentence. Iron is described
7.3 Bonding in Metals > Chapter 7 Ionic and Metallic Bonding. 7.3 Bonding in Metals. 7.1 Ions 7.2 Ionic Bonds and Ionic Compounds
 Chapter 7 Ionic and Metallic Bonding 7.1 Ions 7.2 Ionic Bonds and Ionic Compounds 7.3 Bonding in Metals 1 Copyright Pearson Education, Inc., or its affiliates. All Rights Reserved. CHEMISTRY & YOU What
Chapter 7 Ionic and Metallic Bonding 7.1 Ions 7.2 Ionic Bonds and Ionic Compounds 7.3 Bonding in Metals 1 Copyright Pearson Education, Inc., or its affiliates. All Rights Reserved. CHEMISTRY & YOU What
Douglas G. Brookins. Eh-pH Diagrams. for. Geochemistry. With 98 Figures and 61 Tables. Springer -Verlag Berlin Heidelberg New York London Paris Tokyo
 Douglas G. Brookins Eh-pH Diagrams for Geochemistry With 98 Figures and 61 Tables Springer -Verlag Berlin Heidelberg New York London Paris Tokyo Dr. Douglas G. Brookins Department of Geology University
Douglas G. Brookins Eh-pH Diagrams for Geochemistry With 98 Figures and 61 Tables Springer -Verlag Berlin Heidelberg New York London Paris Tokyo Dr. Douglas G. Brookins Department of Geology University
Preferred. Steel, Epoxy Powder Coated. Satisfactory. Limited
 Corrosion Resistance We manufactures enclosures from a wide range of materials, offering varying degrees of resistance against chemical corrosion. All materials are affected by corrosion in some manner.
Corrosion Resistance We manufactures enclosures from a wide range of materials, offering varying degrees of resistance against chemical corrosion. All materials are affected by corrosion in some manner.
Extracting and using metals. ores. native. Only the most unreactive metals such as gold and platinum are found as native metals.
 Extracting and using metals Only the most unreactive metals such as gold and platinum are found as native metals. ores All the other metals we use are extracted from their ores by chemical processes. native
Extracting and using metals Only the most unreactive metals such as gold and platinum are found as native metals. ores All the other metals we use are extracted from their ores by chemical processes. native
IDENTIFYING UNKNOWN SUBSTANCES
 IDENTIFYING UNKNOWN SUBSTANCES LAB 15 EXPERIMENT STUDENT BOOK Chapter 1, page 25 TOOLBOX Page 4 and 36 Goal Identify unknown substances with the help of different tests. 1. What is the independent variable
IDENTIFYING UNKNOWN SUBSTANCES LAB 15 EXPERIMENT STUDENT BOOK Chapter 1, page 25 TOOLBOX Page 4 and 36 Goal Identify unknown substances with the help of different tests. 1. What is the independent variable
Packaged gases Applications and services
 SIAD S.p.A. I-24126 Bergamo - Via San Bernardino, 92 Tel. +39 035 328111 - Fax +39 035 315486 www.siad.com - siad@siad.com Packaged gases and services SIAD PACKAGED GASES ACETYLENE Oxygen, nitrogen, argon,
SIAD S.p.A. I-24126 Bergamo - Via San Bernardino, 92 Tel. +39 035 328111 - Fax +39 035 315486 www.siad.com - siad@siad.com Packaged gases and services SIAD PACKAGED GASES ACETYLENE Oxygen, nitrogen, argon,
Degassing Components Unique Oxidation Resistance Treatment
 Page 1/7 CGT Carbon is supplier of Carbon Graphite Materials and their Components including carbon graphite felt, rigid graphite felt, carbon-carbon composite and also quartz crucible. We offer these products
Page 1/7 CGT Carbon is supplier of Carbon Graphite Materials and their Components including carbon graphite felt, rigid graphite felt, carbon-carbon composite and also quartz crucible. We offer these products
Pure Substances and Mixtures
 Unit 1 Pure Substances and Mixtures Unit I Overview Getting Started: What Are Things Made Of? I. I Inquiry Investigation: Classifying Substances 1.2 Pure Substances and Mixtures I.3 I.4 I.5 I.6 Inquiry
Unit 1 Pure Substances and Mixtures Unit I Overview Getting Started: What Are Things Made Of? I. I Inquiry Investigation: Classifying Substances 1.2 Pure Substances and Mixtures I.3 I.4 I.5 I.6 Inquiry
CHAPTER 1. Conversions. General Plan for Converting Measurements. State the relationship between the unit given and the unit sought as an equality.
 CHAPTER 1 Conversions One of the aims of chemistry is to describe changes to tell what changed, how it changed, and what it changed into. Another aim of chemistry is to look at matter and its changes and
CHAPTER 1 Conversions One of the aims of chemistry is to describe changes to tell what changed, how it changed, and what it changed into. Another aim of chemistry is to look at matter and its changes and
2.3 Chemical Changes corrosion Kinds of Corrosion
 2.3 Chemical Changes Have you ever wondered why metal car bodies rust but plastic bumpers do not? As you know, different substances have different physical properties, such as colour and hardness, and
2.3 Chemical Changes Have you ever wondered why metal car bodies rust but plastic bumpers do not? As you know, different substances have different physical properties, such as colour and hardness, and
Edexcel GCSE Chemistry. Topic 4: Extracting metals and equilibria. Obtaining and using metals. Notes.
 Edexcel GCSE Chemistry Topic 4: Extracting metals and equilibria Obtaining and using metals Notes 4.1 Deduce the relative reactivity of some metals, by their reactions with water, acids and salt solutions
Edexcel GCSE Chemistry Topic 4: Extracting metals and equilibria Obtaining and using metals Notes 4.1 Deduce the relative reactivity of some metals, by their reactions with water, acids and salt solutions
1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter.
 1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter. What is the mass of this object? A) 0.5 g B) 2 g C) 18 g D) 36 g Base your answers to questions 2 through
1. The diagram below represents a solid object with a density of 3 grams per cubic centimeter. What is the mass of this object? A) 0.5 g B) 2 g C) 18 g D) 36 g Base your answers to questions 2 through
Colourants and pigments
 Colourants and pigments (including printing inks) provisions in NL Bianca van de Ven RIVM Content 1. Regulation of FCM in NL 2. Provisions on colourants and pigments FCM Regulation in NL - Packaging and
Colourants and pigments (including printing inks) provisions in NL Bianca van de Ven RIVM Content 1. Regulation of FCM in NL 2. Provisions on colourants and pigments FCM Regulation in NL - Packaging and
SILLIKER, Inc. Southern California Laboratory 6360 Gateway Drive, Cypress, CA Tel. 209/ Fax. 714/
 6360 Gateway Drive, 90630 95073 Page 1 of 8 CI-516 Cistanche 361439388 ICP MS Full Mineral Screen EPA 3050/6020 USP730 BRN Aluminum 2.2 ppm (w/w) Antimony Arsenic 0.06 ppm (w/w) Barium 0.09 ppm (w/w) Beryllium
6360 Gateway Drive, 90630 95073 Page 1 of 8 CI-516 Cistanche 361439388 ICP MS Full Mineral Screen EPA 3050/6020 USP730 BRN Aluminum 2.2 ppm (w/w) Antimony Arsenic 0.06 ppm (w/w) Barium 0.09 ppm (w/w) Beryllium
Element, Mixture, Compound Lab
 Element, Mixture, Compound Lab Background: Material engineers and mechanical engineers are focused on understanding different materials so that they can create new materials with desired properties. For
Element, Mixture, Compound Lab Background: Material engineers and mechanical engineers are focused on understanding different materials so that they can create new materials with desired properties. For
Chemistry CH1HP. (Jan13CH1Hp01) General Certificate of Secondary Education Higher Tier January Unit Chemistry C1. Unit Chemistry C1 TOTAL
 Centre Number Surname Candidate Number For Examiner s Use Other Names Candidate Signature Examiner s Initials Science A Unit Chemistry C1 Chemistry Unit Chemistry C1 Monday 14 January 2013 For this paper
Centre Number Surname Candidate Number For Examiner s Use Other Names Candidate Signature Examiner s Initials Science A Unit Chemistry C1 Chemistry Unit Chemistry C1 Monday 14 January 2013 For this paper
CHEMICAL RESISTANCE TABLE
 CHEMICAL RESISTANCE TABLE The chemical medias being handled by elastomer tank linings are increasing in number and diversity. To assist in the selection of the proper elastomeric lining for various chemicals,
CHEMICAL RESISTANCE TABLE The chemical medias being handled by elastomer tank linings are increasing in number and diversity. To assist in the selection of the proper elastomeric lining for various chemicals,
أت ارش. Dr. Abdel-Wahab El-Morsy Faculty of Engineering - Rabigh
 Basic Workshop 1 أت ارش There are thousands of materials available for use in engineering applications. Most materials fall into one of three classes that are based on the atomic bonding forces of a particular
Basic Workshop 1 أت ارش There are thousands of materials available for use in engineering applications. Most materials fall into one of three classes that are based on the atomic bonding forces of a particular
Methods for determination of mercury in LP gas
 (JLPGA-S-07) Methods for determination of mercury in LP gas 1. Scope This standard specifies the methods for determination of gaseous mercury in vaporized liquid liquefied petroleum (LP) gas. Remarks 1
(JLPGA-S-07) Methods for determination of mercury in LP gas 1. Scope This standard specifies the methods for determination of gaseous mercury in vaporized liquid liquefied petroleum (LP) gas. Remarks 1
REPORT NUMBER REPORT DATE SEND TO ISSUE DATE Apr 18, Apr 18, 2017 RECEIVED DATE Apr 05, 2017
 PAGE 1/7 Sample ID: EWA PELLETS Lab Number: 2653281 Date Sampled: 2017-04-04 0850 Carbon nitrogen ratio C/N 6 : 1 0.1 Calculation Auto-2017/04/12 Auto-2017/04/18 Carbon (total) 38.53 % 0.050 ASTM D 5373
PAGE 1/7 Sample ID: EWA PELLETS Lab Number: 2653281 Date Sampled: 2017-04-04 0850 Carbon nitrogen ratio C/N 6 : 1 0.1 Calculation Auto-2017/04/12 Auto-2017/04/18 Carbon (total) 38.53 % 0.050 ASTM D 5373
Properties of Minerals
 What is a mineral? Properties of Minerals Mineral 5 characteristics: naturally occurring (not made by people) inorganic (cannot come from living things) solid (definite shape and volume) crystal structure
What is a mineral? Properties of Minerals Mineral 5 characteristics: naturally occurring (not made by people) inorganic (cannot come from living things) solid (definite shape and volume) crystal structure
Drinking Water Corrosion Control and POU/POE Treatment
 Drinking Water Corrosion Control and POU/POE Treatment Greg Reyneke CWS-VI Learning Objectives Symptoms of Corrosion Types & Causes of Corrosion Lead & Copper Rule Regulatory Environment Corrosion Prevention
Drinking Water Corrosion Control and POU/POE Treatment Greg Reyneke CWS-VI Learning Objectives Symptoms of Corrosion Types & Causes of Corrosion Lead & Copper Rule Regulatory Environment Corrosion Prevention
Mathematics and Science in Schools in Sub-Saharan Africa
 Mathematics and Science in Schools in Sub-Saharan Africa MATERIAL SCIENCE METAL ALLOYS Metals have many advantages Strong Malleable Conductors Shiny Metal Customizing Cold-working and heat treating metals
Mathematics and Science in Schools in Sub-Saharan Africa MATERIAL SCIENCE METAL ALLOYS Metals have many advantages Strong Malleable Conductors Shiny Metal Customizing Cold-working and heat treating metals
HAND BOOK OF ELECTROPLATING ANODIZING & SURFACE FINISHING TECHNOLOGY
 HAND BOOK OF ELECTROPLATING ANODIZING & SURFACE FINISHING TECHNOLOGY Principles of Electroplating The Electro Chemical Series Electrodeposition Electrochemical Terms Using a Copper Anode Using an Inert
HAND BOOK OF ELECTROPLATING ANODIZING & SURFACE FINISHING TECHNOLOGY Principles of Electroplating The Electro Chemical Series Electrodeposition Electrochemical Terms Using a Copper Anode Using an Inert
Downloaded from
 Fun with Magnets 1 1.What happens when the north pole of a magnet is placed near the north pole of another magnet? 2.Which of the following cannot be attracted by a magnet? (A) Wood. (B) Nickel. (C)
Fun with Magnets 1 1.What happens when the north pole of a magnet is placed near the north pole of another magnet? 2.Which of the following cannot be attracted by a magnet? (A) Wood. (B) Nickel. (C)
Name Lab Section Date. Sediment Lab
 Name Lab Section Date. Investigating Stokes Law Sediment Lab ds = density of solid, g/cm dw = density of water, g/cm g = gravity, 980 cm/second 2 D = particle diameter in centimeters μ = molecular viscosity,
Name Lab Section Date. Investigating Stokes Law Sediment Lab ds = density of solid, g/cm dw = density of water, g/cm g = gravity, 980 cm/second 2 D = particle diameter in centimeters μ = molecular viscosity,
A STUDY OF CASTING CHARACTERISTICS FOR DIE-CAST ALUMINUM ALLOY
 ME8109: Casting And Solidification of Material A STUDY OF CASTING CHARACTERISTICS FOR DIE-CAST ALUMINUM ALLOY Department of Mechanical & Industrial Engineering Graduate Program in Mechanical Engineering
ME8109: Casting And Solidification of Material A STUDY OF CASTING CHARACTERISTICS FOR DIE-CAST ALUMINUM ALLOY Department of Mechanical & Industrial Engineering Graduate Program in Mechanical Engineering
Make Five. A game about the chemical formulas of some common minerals
 Make Five A game about the chemical formulas of some common minerals By defi nition, a mineral has a defi nite chemical composition. It s sort of like a recipe. This game is for students who want to (or
Make Five A game about the chemical formulas of some common minerals By defi nition, a mineral has a defi nite chemical composition. It s sort of like a recipe. This game is for students who want to (or
WELDING Topic and Contents Hours Marks
 Topic and Contents Hours Marks 3.1 Introduction 04 Marks Classification and selection of welding process. Working principle of Gas welding and types of flames. 3.2 Arc welding process 08 Marks Metal arc,
Topic and Contents Hours Marks 3.1 Introduction 04 Marks Classification and selection of welding process. Working principle of Gas welding and types of flames. 3.2 Arc welding process 08 Marks Metal arc,
Designer s Guide to the Cypress PSoC
 Designer s Guide to the Cypress PSoC By Robert Ashby July 2005 ISBN 978-0-7506-7780-6 Paperback 272 Pages $51.95 Features: The first independent technical reference available on the PSoC, a product line
Designer s Guide to the Cypress PSoC By Robert Ashby July 2005 ISBN 978-0-7506-7780-6 Paperback 272 Pages $51.95 Features: The first independent technical reference available on the PSoC, a product line
AIM: SWBAT determine the location of metals, nonmetals and metalloids on the periodic table. What is another name for a column in the periodic table?
 th Grade Science Metals, nonmetals and metalloids Name Date: UNIT 1 LESSON 1 Do Now AIM: SWBAT determine the location of metals, nonmetals and metalloids on the periodic table Directions: Answer the following
th Grade Science Metals, nonmetals and metalloids Name Date: UNIT 1 LESSON 1 Do Now AIM: SWBAT determine the location of metals, nonmetals and metalloids on the periodic table Directions: Answer the following
Resource Guide. Section 4: Ni-Resist
 Resource Guide Section 4: Ni-Resist Section 4 Ni-Resist Description of Grades... 4-3 201 (Type 1) Ni-Resist... 4-4 202 (Type 2) Ni-Resist... 4-6 Stock Listings... 4-8 4-2 Ni-Resist Description of Grades
Resource Guide Section 4: Ni-Resist Section 4 Ni-Resist Description of Grades... 4-3 201 (Type 1) Ni-Resist... 4-4 202 (Type 2) Ni-Resist... 4-6 Stock Listings... 4-8 4-2 Ni-Resist Description of Grades
MINERALS MR. WHITE ADVANCED CONTENT EARTH SCIENCE
 MINERALS MR. WHITE ADVANCED CONTENT EARTH SCIENCE WHO STUDIES THE EARTH S SURFACE & ITS CHANGING SHAPE? Geologists! GEOLOGISTS STUDY minerals rocks earthquakes volcanoes plate tectonics WHAT IS A MINERAL?
MINERALS MR. WHITE ADVANCED CONTENT EARTH SCIENCE WHO STUDIES THE EARTH S SURFACE & ITS CHANGING SHAPE? Geologists! GEOLOGISTS STUDY minerals rocks earthquakes volcanoes plate tectonics WHAT IS A MINERAL?
Chapter - 3. Mineral and Power Resources
 Chapter - 3 Mineral and Power Resources What are Minerals? A naturally occurring substance that has a definite chemical composition is a mineral Minerals Not distributed properly concentrated in a particular
Chapter - 3 Mineral and Power Resources What are Minerals? A naturally occurring substance that has a definite chemical composition is a mineral Minerals Not distributed properly concentrated in a particular
METALS AND THEIR COMPOUNDS
 METALS AND THEIR COMPOUNDS Metals are elements whose atoms ionize by electron loss, while non-metals are elements whose atoms ionize by electron gain. Metals are in groups 1, 2 and 3 of the periodic table.
METALS AND THEIR COMPOUNDS Metals are elements whose atoms ionize by electron loss, while non-metals are elements whose atoms ionize by electron gain. Metals are in groups 1, 2 and 3 of the periodic table.
Metropolitan Water Reclamation District of Greater Chicago 10/19/ Mark Joyce Senior Mechanical Engineer Engineering Department
 Metropolitan Water Reclamation District of Greater Chicago 10/19/201 2 Mark Joyce Senior Mechanical Engineer Engineering Department 1 Definition of Corrosion Corrosion is the deterioration of a substance
Metropolitan Water Reclamation District of Greater Chicago 10/19/201 2 Mark Joyce Senior Mechanical Engineer Engineering Department 1 Definition of Corrosion Corrosion is the deterioration of a substance
Filament wound Plastic Self-lubricating Bearings
 Filament wound Plastic Self-lubricating Bearings Filament wound Plastic Self-lubricating Bearings CSB-CRB: Glass fiber epoxy-impregnated with PTFE and high strength fibers CSB-CRG: Glass fiber epoxy-impregnated
Filament wound Plastic Self-lubricating Bearings Filament wound Plastic Self-lubricating Bearings CSB-CRB: Glass fiber epoxy-impregnated with PTFE and high strength fibers CSB-CRG: Glass fiber epoxy-impregnated
85 Q.51 Which of the following carbonates would give the metal when heated with carbon? (1) MgCO 3 (2) PbCO 3 (3) K 2 CO 3 (4) CuCO 3
 Metal and metal reactivity / Section 2 / Sect2pp.doc / S. W. Tse / P.1 85 Q.51 Which of the following carbonates would give the metal when heated with carbon? (1) MgCO 3 (2) PbCO 3 (3) K 2 CO 3 (4) CuCO
Metal and metal reactivity / Section 2 / Sect2pp.doc / S. W. Tse / P.1 85 Q.51 Which of the following carbonates would give the metal when heated with carbon? (1) MgCO 3 (2) PbCO 3 (3) K 2 CO 3 (4) CuCO
TECHNICAL SPECIFICATION Conductive Elastomer Molded Gaskets
 Pag. 1/6 TECHNICAL SPECIFICATION Conductive Elastomer Molded Gaskets This category of items is in continuous evolution. The raw material from which it is made is the first component that must be analyzed
Pag. 1/6 TECHNICAL SPECIFICATION Conductive Elastomer Molded Gaskets This category of items is in continuous evolution. The raw material from which it is made is the first component that must be analyzed
Sci-Tech. Connections. The Heat's On. Hot Potatoes Grade 7 Sampler
 Sci-Tech Connections The Heat's On Hot Potatoes Grade 7 Sampler HOT POTATOES EXPECTATIONS work cooperatively, respect each other, work safely, and keep a clean work space use the terms heat and cooling
Sci-Tech Connections The Heat's On Hot Potatoes Grade 7 Sampler HOT POTATOES EXPECTATIONS work cooperatively, respect each other, work safely, and keep a clean work space use the terms heat and cooling
look down at cross on paper paper cross on paper
 1 The equation for the reaction between sodium thiosulfate and hydrochloric acid is given below. Na 2 S 2 O 3 (aq) + 2HCl (aq) 2NaCl (aq) + S(s) + SO 2 (g) + H 2 O(l) The speed of this reaction was investigated
1 The equation for the reaction between sodium thiosulfate and hydrochloric acid is given below. Na 2 S 2 O 3 (aq) + 2HCl (aq) 2NaCl (aq) + S(s) + SO 2 (g) + H 2 O(l) The speed of this reaction was investigated
Electron Microscopy (EM) Grid
 Anirban Som 25-01-14 Instrumental technique presentation Electron Microscopy (EM) Grid What I will talk about Some basic topics about EM grid Home-made grid preparation Grid cleaning Carbon coating and
Anirban Som 25-01-14 Instrumental technique presentation Electron Microscopy (EM) Grid What I will talk about Some basic topics about EM grid Home-made grid preparation Grid cleaning Carbon coating and
NONFERROUS METALS AND ALLOYS
 NONFERROUS METALS AND ALLOYS Chapter 7 7.1 Introduction Usage of nonferrous metals and alloys has increased due to technology Possess certain properties that ferrous materials do not have Resistance to
NONFERROUS METALS AND ALLOYS Chapter 7 7.1 Introduction Usage of nonferrous metals and alloys has increased due to technology Possess certain properties that ferrous materials do not have Resistance to
21.The smallest part of an element that keeps the same properties of that element. a. Element b. Compound c. Atom d. quark
 21.The smallest part of an element that keeps the same properties of that element a. Element b. Compound c. Atom d. quark 22.A substance made of 2 or more elements chemically combined a. Compound b. Mixture
21.The smallest part of an element that keeps the same properties of that element a. Element b. Compound c. Atom d. quark 22.A substance made of 2 or more elements chemically combined a. Compound b. Mixture
GCSE BITESIZE Examinations
 GCSE BITESIZE Examinations General Certificate of Secondary Education AQA SCIENCE A CHY1A Unit Chemistry C1a (Products from Rocks) AQA Chemistry Unit Chemistry C1a (Products from Rocks) FOUNDATION TIER
GCSE BITESIZE Examinations General Certificate of Secondary Education AQA SCIENCE A CHY1A Unit Chemistry C1a (Products from Rocks) AQA Chemistry Unit Chemistry C1a (Products from Rocks) FOUNDATION TIER
