PROPERTIES OF MATERIALS PART HARDNESS
|
|
|
- Nathan Ellis
- 6 years ago
- Views:
Transcription
1 CHAPTER 3 PROPERTIES OF MATERIALS PART HARDNESS A Resistance to permanently indenting the surface Large hardness means resistance to plastic deformation or cracking In compression, better wear properties
2 3.2 ELECTRICAL PROPERTIES Electrical Conductivity is a function of a material s ability to carry an electrical current. Conduction requires electron transport. The conductivity (σ) of a material is determined by taking the reciprocal of the measured electrical resistance (R) to the flow of electricity in a length (L) of material divided by the cross-sectional area (A). σ = 1 R A L ELECTRICAL CONDUCTIVITY
3 EXAMPLES Electrical conductivity order of the first four metals: Silver >Copper>Gold >Aluminum The electrical properties of ceramic materials vary greatly, with characteristic measures spanning over many orders of magnitude.ceramics are probably best known as electrical insulators. Some ceramic insulators (such as BaTiO3) can be polarized and used as capacitors. Other ceramics conduct electrons when threshold energy is reached, and are thus called semiconductors. In 1986, a new class of ceramics was discovered, the high Tc superconductors. These materials conduct electricity with essentially zero resistance TEMPERATURE DEPENDENCE Conductivity is temperature dependent. As the temperature increase, the conductivity of a metal decrease. In contrast, the conductivity of pure semiconductors and insulators increases as the temperature increases
4 3.2.2 TEMPERATURE DEPENDENCE Electrical conductivities of some common materials insulators semiconductors metals Conductivity ohm -1 cm -1 ( ) TEMPERATURE DEPENDENCE The energy bands of metals, semiconductors and insulators
5 3.3 OPTICAL PROPERTIES An optical property describes the way a material reacts to exposure to light. Visible light is a form of electromagnetic radiation with wavelengths in the range of 400 to 700 nm corresponding to an energy range of 3.1 to 1.8 electron volts (ev) (From E = hc/λ, where c = 3 x nm/s and h = 4.13 x ev.s) LIGHT INTERACTION WITH SOLIDS When light strikes an object it may be transmitted, absorbed, or reflected
6 3.3.2 OPTICAL CLASSIFICATION Materials vary in their ability to transmit light, and are usually described as transparent, translucent, or opaque. Transparent materials, such as glass, transmit light with little absorption or reflection. Materials that transmit light diffusely, such as frosted glass, are translucent. Opaque materials do not transmit light Applications: Fiber Optics
7 PHOTOCONDUCTIVITY MAGNETIC PROPERTIES
8 3.4.1 MAGNETIC STORAGE THERMAL PROPERTIES HEAT CAPACITY In general heat capacity is the ability of a material to absorb heat SPECIFIC HEAT CAPACITY Specific heat capacity is the amount of heat energy required to raise the temperature of a unit mass by 1 unit
9 3.5.2 SPECIFIC HEAT CAPACITY Comparison: Heat Capacity Values THERMAL PROPERTIES SPECIFIC LATENT HEAT Specific latent heat is the energy per unit mass absorbed or evolved when a substance changes phase. There are 2 types: Latent heat of fusion the heat given When a liquid changes into a solid. Latent heat of vaporization the heat absorbed when a liquid turns into a gas THERMAL CONDUCTIVITY The ability of a material to transfer heat
10 3.5.4 THERMAL CONDUCTIVITY Comparison: Thermal Conductivities THERMAL EXPANSION Materials change size when heating. Comparison: Thermal Expansion Values
11 3.5.6 THERMAL PROTECTION SYSTEM Comparison of thermal properties of different ceramic materials Material Melting Temp. ( o C) Heat Capacity (J/kgK) Coef. of Linear Expansion 1/ o Cx10-6 Thermal Conductivity (W/mK) Aluminum Metal Copper Metal Alumina Fused Silica Soda-lime glass Polyethylene Polystyrene
12 3.6 CORROSION PROPERTIES Most materials experience some type of interaction with a large number of diverse environments. Such interactions impair a material s usefulness as a result of decrease in mechanical property qualities (ductility and strength), other physical properties, or appearance. In metals there is actual material loss by dissolution : Corrosion Oxidation of metals by exposure to air/moisture/salts Ceramic materials are relatively resistant to deterioration, usually occurs at elevated temperatures or in rather extreme environments : Corrosion CORROSION PROPERTIES For polymers mechanisms are different from metals and ceramics: Degradation Fe OH Fe(OH) 2 (s) Fe 2 O 3 O 2 + 2H 2 O + 4e 4OH Fe 2+ cathode anode e Iron Iron Fe Fe e
13 3.6.1 CORROSION ENVIRONMENTS Atmospheric corrosion accounts for the greatest losses. Moisture containing dissolved oxygen is the primary corrosive agent and also sulfur compounds and NaCl. Marine atmospheres are highly corrosive because of NaCl. Acid rain (dilute sulfuric acid solutions) in industrial environments can also cause corrosion problems PREVENTING CORROSION For protection, alloys of Al and Cu, galvanized steel are applied. Coatings or Cathodic Protection Coat with Zn to galvanize it. Zn reacts immediately with oxygen to form a hard protective coating. The coating is ZnO and completely seals the surface. If the surface is scratched, any exposed Zn will react to seal it again. Only if the steel core is exposed will the sample oxidize. ZnO ZnO Zn Zn Steel
14 3.6.3 CATHODIC PROTECTION Use a piece of metal that is more reactive and unimportant. Here, the item cannot be physically galvanized due to size. Zn will oxidize before the ship s hull will. Zn
Chapter 16 Corrosion and Degradation of Materials
 Chapter 16 Corrosion and Degradation of Materials Concept Check 16.1 Question: Would you expect iron to corrode in water of high purity? Why or why not? Answer: Iron would not corrode in water of high
Chapter 16 Corrosion and Degradation of Materials Concept Check 16.1 Question: Would you expect iron to corrode in water of high purity? Why or why not? Answer: Iron would not corrode in water of high
MATERIALS SCIENCE AND ENGINEERING
 MATERIALS SCIENCE AND ENGINEERING materials science - the discipline that involves investigating the relationships that exist between the structures and properties of materials materials engineering -
MATERIALS SCIENCE AND ENGINEERING materials science - the discipline that involves investigating the relationships that exist between the structures and properties of materials materials engineering -
Materials of Engineering ENGR 151 CORROSION ELECTRICAL PROPERTIES
 Materials of Engineering ENGR 151 CORROSION ELECTRICAL PROPERTIES more anodic (active) more cathodic (inert) GALVANIC SERIES Ranking of the reactivity of metals/alloys in seawater Platinum Gold Graphite
Materials of Engineering ENGR 151 CORROSION ELECTRICAL PROPERTIES more anodic (active) more cathodic (inert) GALVANIC SERIES Ranking of the reactivity of metals/alloys in seawater Platinum Gold Graphite
Electrical conductivity
 Electrical conductivity Ohm's Law: voltage drop (volts = J/C) C = Coulomb A (cross sect. area) ΔV = I R Resistivity, ρ and Conductivity, σ: -- geometry-independent forms of Ohm's Law resistance (Ohms)
Electrical conductivity Ohm's Law: voltage drop (volts = J/C) C = Coulomb A (cross sect. area) ΔV = I R Resistivity, ρ and Conductivity, σ: -- geometry-independent forms of Ohm's Law resistance (Ohms)
Introduction to Materials Science & Engineering
 Introduction to Materials Science & Engineering Course Objective... Introduce fundamental concepts in Materials Science You will learn about: material structure how structure dictates properties how processing
Introduction to Materials Science & Engineering Course Objective... Introduce fundamental concepts in Materials Science You will learn about: material structure how structure dictates properties how processing
Electrical conductivity
 Electrical conductivity Ohm's Law: voltage drop (volts = J/C) C = Coulomb A (cross sect. area) ΔV = I R Resistivity, ρ and Conductivity, σ: -- geometry-independent forms of Ohm's Law resistance (Ohms)
Electrical conductivity Ohm's Law: voltage drop (volts = J/C) C = Coulomb A (cross sect. area) ΔV = I R Resistivity, ρ and Conductivity, σ: -- geometry-independent forms of Ohm's Law resistance (Ohms)
Rusting is an example of corrosion, which is a spontaneous redox reaction of materials with substances in their environment.
 CORROSION WHAT IS CORROSION? Corrosion is the deterioration of a metal as a result of chemical reactions between it and the surrounding environment. Rusting is an example of corrosion, which is a spontaneous
CORROSION WHAT IS CORROSION? Corrosion is the deterioration of a metal as a result of chemical reactions between it and the surrounding environment. Rusting is an example of corrosion, which is a spontaneous
Chapter 1 - Introduction
 Chapter 1 - Introduction What is materials science? Why should we know about it? Materials drive our society Stone Age Bronze Age Iron Age Now? Silicon Age? Polymer Age? Chapter 1-1 Chapter 1-2 Hardness
Chapter 1 - Introduction What is materials science? Why should we know about it? Materials drive our society Stone Age Bronze Age Iron Age Now? Silicon Age? Polymer Age? Chapter 1-1 Chapter 1-2 Hardness
Chemistry 145 Exam number 4 name 11/19/98 # Faraday s constant is 96,500 c/mole of electrons.
 Chemistry 145 Exam number 4 name 11/19/98 # Faraday s constant is 96,500 c/mole of electrons. A.(16) An electrochemical cell is prepared with a strip of manganese metal dipping in to a 1.0 M MnSO 4 solution
Chemistry 145 Exam number 4 name 11/19/98 # Faraday s constant is 96,500 c/mole of electrons. A.(16) An electrochemical cell is prepared with a strip of manganese metal dipping in to a 1.0 M MnSO 4 solution
Introduction to Material Science and Engineering
 Introduction to Material Science and Engineering Introduction What is materials engineering? the understanding and modification of the structure and properties of materials to improve the performance and
Introduction to Material Science and Engineering Introduction What is materials engineering? the understanding and modification of the structure and properties of materials to improve the performance and
ME 206: Materials Science
 ME 206: Materials Science Course Objective... Introduce fundamental concepts in Materials Science You will learn about: material structure how structure dictates properties how processing can change structure
ME 206: Materials Science Course Objective... Introduce fundamental concepts in Materials Science You will learn about: material structure how structure dictates properties how processing can change structure
Properties of Materials
 Properties of Materials Thermal Properties Thermal Conductivity Temperature Wall The Thermal Conductivity (k) is the measure of the ability of a material to transmit heat by conduction. The heat (Q) is
Properties of Materials Thermal Properties Thermal Conductivity Temperature Wall The Thermal Conductivity (k) is the measure of the ability of a material to transmit heat by conduction. The heat (Q) is
Materials Science and Engineering: An Introduction
 Materials Science and Engineering: An Introduction Callister, William D. ISBN-13: 9780470419977 Table of Contents List of Symbols. 1 Introduction. 1.1 Historical Perspective. 1.2 Materials Science and
Materials Science and Engineering: An Introduction Callister, William D. ISBN-13: 9780470419977 Table of Contents List of Symbols. 1 Introduction. 1.1 Historical Perspective. 1.2 Materials Science and
Chapter 18: Electrical Properties
 Chapter 18: Electrical Properties ISSUES TO ADDRESS... How are electrical conductance and resistance characterized? What are the physical phenomena that distinguish conductors, semiconductors, and insulators?
Chapter 18: Electrical Properties ISSUES TO ADDRESS... How are electrical conductance and resistance characterized? What are the physical phenomena that distinguish conductors, semiconductors, and insulators?
CHAPTER 9. The Uses of Sulphuric Acid
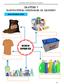 CHAPTER 9 MANUFACTURED SUBSTANCES IN INDUSTRY SULPHURIC ACID The Uses of Sulphuric Acid 175 POLLUTION of Sulphur DIOXIDE Formation of Acid Rain Burning of Sulphur Burning of Sulphur Dioxide 176 Effect
CHAPTER 9 MANUFACTURED SUBSTANCES IN INDUSTRY SULPHURIC ACID The Uses of Sulphuric Acid 175 POLLUTION of Sulphur DIOXIDE Formation of Acid Rain Burning of Sulphur Burning of Sulphur Dioxide 176 Effect
2) Galvanic or Two-Metal corrosion
 1 2) Galvanic or Two-Metal corrosion Galvanic corrosion occurs when two metals or alloys are electrically coupled while exposed to an electrolyte. The more reactive metal in the particular environment
1 2) Galvanic or Two-Metal corrosion Galvanic corrosion occurs when two metals or alloys are electrically coupled while exposed to an electrolyte. The more reactive metal in the particular environment
Materials Engineering PTT 110
 By: Pn. Nurul Ain Harmiza Abdullah Materials Engineering PTT 110 SEMESTER 1 (2013/2014) PowerPoint Lecture Slides for Foundations of Materials Science and Engineering Fifth Edition William F. Smith Javad
By: Pn. Nurul Ain Harmiza Abdullah Materials Engineering PTT 110 SEMESTER 1 (2013/2014) PowerPoint Lecture Slides for Foundations of Materials Science and Engineering Fifth Edition William F. Smith Javad
What is Materials Science and Engineering??
 Materials Science and Engineering What is Materials Science and Engineering?? Engineering is Doing? Engineers use science and math to design new products and services Engineers solve problems They make
Materials Science and Engineering What is Materials Science and Engineering?? Engineering is Doing? Engineers use science and math to design new products and services Engineers solve problems They make
CORROSION of Metals CORROSION CORROSION. Outline ISSUES TO ADDRESS... Why does corrosion occur? What metals are most likely to corrode?
 Outline Corrosion - Introduction Corrosion of Metals - e.g. Rusting of iron in water Electrochemical Cell Electrode Potential in Electrochemical Cell Standard Electromotive Force Example Relative Corrosion
Outline Corrosion - Introduction Corrosion of Metals - e.g. Rusting of iron in water Electrochemical Cell Electrode Potential in Electrochemical Cell Standard Electromotive Force Example Relative Corrosion
One of the main ores of zinc is zinc blende, ZnS. There are two stages in the extraction of zinc from this ore.
 1 Two of the main uses of zinc are for galvanising and for making alloys. One of the main ores of zinc is zinc blende, ZnS. There are two stages in the extraction of zinc from this ore. (a) Stage 1 Zinc
1 Two of the main uses of zinc are for galvanising and for making alloys. One of the main ores of zinc is zinc blende, ZnS. There are two stages in the extraction of zinc from this ore. (a) Stage 1 Zinc
Mark schemes. for 1 mark. for 1 mark. An arrow indicating a position between aluminium and zinc. electrolysis
 Page of 2 Mark schemes hydrogen for mark oxygen for mark [2] 2 An arrow indicating a position between aluminium and zinc. electrolysis because calcium is more reactive (than aluminium or carbon) accept
Page of 2 Mark schemes hydrogen for mark oxygen for mark [2] 2 An arrow indicating a position between aluminium and zinc. electrolysis because calcium is more reactive (than aluminium or carbon) accept
Corrosion of Metals. Industrial Metallurgists, LLC Northbrook, IL Copyright 2013 Industrial Metallurgists, LLC
 Corrosion of Metals Industrial Metallurgists, LLC Northbrook, IL 60062 847.528.3467 www.imetllc.com Copyright 2013 Industrial Metallurgists, LLC Course structure Learning modules 1. Introduction to corrosion
Corrosion of Metals Industrial Metallurgists, LLC Northbrook, IL 60062 847.528.3467 www.imetllc.com Copyright 2013 Industrial Metallurgists, LLC Course structure Learning modules 1. Introduction to corrosion
1-Materials Science & Materials Engineering
 1-Materials Science & Materials Engineering 1-1-Structure & Properties Relationship (Materials Science or Materials Engineering) Processing Structure Properties Performance Sub Atomic Atomic Sub Atomic
1-Materials Science & Materials Engineering 1-1-Structure & Properties Relationship (Materials Science or Materials Engineering) Processing Structure Properties Performance Sub Atomic Atomic Sub Atomic
Materials Science. Introduce fundamental concepts in Materials Science
 Materials Science Course Objective... Introduce fundamental concepts in Materials Science You will learn about: material structure how structure dictates properties how processing can change structure
Materials Science Course Objective... Introduce fundamental concepts in Materials Science You will learn about: material structure how structure dictates properties how processing can change structure
Corrosion and batteries
 Corrosion and batteries Corrosion is a process of gradual destruction of metal from its surface due to its unwanted chemical or electrochemical interaction of metal. Ore of metal which is stable and is
Corrosion and batteries Corrosion is a process of gradual destruction of metal from its surface due to its unwanted chemical or electrochemical interaction of metal. Ore of metal which is stable and is
METAL FINISHING. (As per revised VTU syllabus: )
 METAL FINISHING (As per revised VTU syllabus: 2015-16) Definition: It is a process in which a specimen metal (article) is coated with another metal or a polymer in order to modify the surface properties
METAL FINISHING (As per revised VTU syllabus: 2015-16) Definition: It is a process in which a specimen metal (article) is coated with another metal or a polymer in order to modify the surface properties
Module-13. Corrosion and Degradation of materials
 Module-13 Corrosion and Degradation of materials Contents 1) Corrosion of metals 2) Corrosion of ceramics 3) Degradation of polymers Deterioration of materials Conventional engineering materials are not
Module-13 Corrosion and Degradation of materials Contents 1) Corrosion of metals 2) Corrosion of ceramics 3) Degradation of polymers Deterioration of materials Conventional engineering materials are not
Mat E 272 Lecture 26: Oxidation and Corrosion
 Mat E 272 Lecture 26: Oxidation and Corrosion December 11, 2001 Introduction: Environmental degradation of materials is one of the most costly failure modes, accounting for over 5 percent of the total
Mat E 272 Lecture 26: Oxidation and Corrosion December 11, 2001 Introduction: Environmental degradation of materials is one of the most costly failure modes, accounting for over 5 percent of the total
Chapter 3 Metals and Non-metals
 Chapter 3 Metals and Non-metals Intext Questions On Page 40 Question 1: Give an example of a metal which (i) (ii) (iii) (iv) Is a liquid at room temperature Can be easily cut with a knife. Is the best
Chapter 3 Metals and Non-metals Intext Questions On Page 40 Question 1: Give an example of a metal which (i) (ii) (iii) (iv) Is a liquid at room temperature Can be easily cut with a knife. Is the best
OFFSHORE CATHODIC PROTECTION 101: WHAT IS IT AND HOW DOES IT WORK? Dick Baxter & Jim Britton
 This PDF document contains Live Web Links Click them for more information from the web OFFSHORE CATHODIC PROTECTION 101: WHAT IS IT AND HOW DOES IT WORK? Dick Baxter & Jim Britton www.cathodicprotection101.com
This PDF document contains Live Web Links Click them for more information from the web OFFSHORE CATHODIC PROTECTION 101: WHAT IS IT AND HOW DOES IT WORK? Dick Baxter & Jim Britton www.cathodicprotection101.com
Electricity and Chemistry
 Electricity and Chemistry Electrochemistry: It is a branch of chemistry that deals with the reactions involving the conversion of chemical energy into electrical energy and vice-versa. Electrochemical
Electricity and Chemistry Electrochemistry: It is a branch of chemistry that deals with the reactions involving the conversion of chemical energy into electrical energy and vice-versa. Electrochemical
Fundamentals of Sealing and Encapsulation
 Fundamentals of Sealing and Encapsulation Sealing and Encapsulation Encapsulation and sealing are two of the major protecting functions of IC packaging. They are used to protect IC devices from adverse
Fundamentals of Sealing and Encapsulation Sealing and Encapsulation Encapsulation and sealing are two of the major protecting functions of IC packaging. They are used to protect IC devices from adverse
Types of Glasses : 1. Soda lime Glass: (Soda glass or Normal Glass or Soft Glass)
 Introduction: Phosphate glasses and glass ceramics are useful for applications such as bone transplantation, glass to metal seals, containment of radioactive wastes, fast ion conductors, laser host materials
Introduction: Phosphate glasses and glass ceramics are useful for applications such as bone transplantation, glass to metal seals, containment of radioactive wastes, fast ion conductors, laser host materials
Manufacturing A Unit 5, Lesson 2 Explanation
 Manufacturing 5.2.1A Unit 5, Lesson 2 Explanation The Unit Big Idea The designed world is the product of a design process, which provides ways to turn resources - materials, tools and machines, people,
Manufacturing 5.2.1A Unit 5, Lesson 2 Explanation The Unit Big Idea The designed world is the product of a design process, which provides ways to turn resources - materials, tools and machines, people,
ME -215 ENGINEERING MATERIALS AND PROCESES
 ME -215 ENGINEERING MATERIALS AND PROCESES Instructor: Office: MEC325, Tel.: 973-642-7455 E-mail: samardzi@njit.edu PROPERTIES OF MATERIALS Chapter 3 Materials Properties STRUCTURE PERFORMANCE PROCESSING
ME -215 ENGINEERING MATERIALS AND PROCESES Instructor: Office: MEC325, Tel.: 973-642-7455 E-mail: samardzi@njit.edu PROPERTIES OF MATERIALS Chapter 3 Materials Properties STRUCTURE PERFORMANCE PROCESSING
SAMPLE PAGES PAGES. Extraction of metals from metal oxides. mixture of iron sand and coal are heated as they move down kiln, by force of gravity
 Unit 11.5 Metals and Non-metals Topic 3: Extraction of metals and corrosion In the previous two Topics we looked at the physical and chemical properties of metals. In Topic 3 we now examine how metals
Unit 11.5 Metals and Non-metals Topic 3: Extraction of metals and corrosion In the previous two Topics we looked at the physical and chemical properties of metals. In Topic 3 we now examine how metals
Alloys and Solid Solutions
 Alloys and Solid Solutions Chemistry 123 Spring 2008 Dr. Woodward Solutions Solid Solution 14 Carat Gold Liquid Solution Vodka Gaseous Solution Air Solution = A homogeneous mixture 1 Alloys An alloy is
Alloys and Solid Solutions Chemistry 123 Spring 2008 Dr. Woodward Solutions Solid Solution 14 Carat Gold Liquid Solution Vodka Gaseous Solution Air Solution = A homogeneous mixture 1 Alloys An alloy is
Chapter 4: Theoretical background for the
 Chapter 4: Theoretical background for the hydraulic system Theoretical information on the design of a hydraulic system is provided in this chapter. An overview of Bernoulli s equation to determine pump
Chapter 4: Theoretical background for the hydraulic system Theoretical information on the design of a hydraulic system is provided in this chapter. An overview of Bernoulli s equation to determine pump
Group A Good Earth School Naduveerapattu Chemistry Worksheet Class X Metallurgy (1) Name a metal which has the following properties.
 Group A Good Earth School Naduveerapattu Chemistry Worksheet Class X Metallurgy (1) Name a metal which has the following properties. 1. Is a liquid at ordinary temperatures. 2. Has a low boiling point
Group A Good Earth School Naduveerapattu Chemistry Worksheet Class X Metallurgy (1) Name a metal which has the following properties. 1. Is a liquid at ordinary temperatures. 2. Has a low boiling point
Chapter 17: Corrosion and Degradation of Materials
 Chapter 17: Corrosion and Degradation of Materials ISSUES TO ADDRESS... How does corrosion of metals occur? Which metals are more likely to corrode and which less? How do we prevent or control corrosion?
Chapter 17: Corrosion and Degradation of Materials ISSUES TO ADDRESS... How does corrosion of metals occur? Which metals are more likely to corrode and which less? How do we prevent or control corrosion?
2.3 Chemical Changes corrosion Kinds of Corrosion
 2.3 Chemical Changes Have you ever wondered why metal car bodies rust but plastic bumpers do not? As you know, different substances have different physical properties, such as colour and hardness, and
2.3 Chemical Changes Have you ever wondered why metal car bodies rust but plastic bumpers do not? As you know, different substances have different physical properties, such as colour and hardness, and
CORROSION & ITS CONTROL -Content. Introduction Classification Galvanic series Factors affecting Protection methods Summary
 CORROSION & ITS CONTROL -Content Introduction Classification Galvanic series Factors affecting Protection methods Summary 3 Learning Objectives At the end of the chapter the learner will be able to; 1.
CORROSION & ITS CONTROL -Content Introduction Classification Galvanic series Factors affecting Protection methods Summary 3 Learning Objectives At the end of the chapter the learner will be able to; 1.
MECHANICAL AND PHYSICAL PROPRIETIES
 Kera-Coat Ceramic Coatings are Special Ceramic Coating defined as a substantially vitreous or glassy inorganic coating bonded to metal by fusion at a temperature above 800 F. In this short abstract we
Kera-Coat Ceramic Coatings are Special Ceramic Coating defined as a substantially vitreous or glassy inorganic coating bonded to metal by fusion at a temperature above 800 F. In this short abstract we
Atomic structure, arrangement, and movement Introduction to Materials Introduction Types of Materials Structure-Property-Processing Relationship
 Atomic structure, arrangement, and movement to Materials Types of Materials Structure-Property-Processing Relationship Environmental Effects on Material Behavior Materials Design and Selection Atomic Structure
Atomic structure, arrangement, and movement to Materials Types of Materials Structure-Property-Processing Relationship Environmental Effects on Material Behavior Materials Design and Selection Atomic Structure
Methods of Corrosion Control. Corrosion Control or Corrosion Management?
 Corrosion Control or Corrosion Management? Corrosion control is a process aimed at reducing the corrosion rate to a tolerable level (or predictable limits) Corrosion control focuses mainly on (i) materials
Corrosion Control or Corrosion Management? Corrosion control is a process aimed at reducing the corrosion rate to a tolerable level (or predictable limits) Corrosion control focuses mainly on (i) materials
The Technological World. Chapter 12
 The Technological World Chapter 12 Chapter 12 Manufacturing Technical Objects There have been many inventions that have improved the quality of our lives. We will refer to these inventions as technical
The Technological World Chapter 12 Chapter 12 Manufacturing Technical Objects There have been many inventions that have improved the quality of our lives. We will refer to these inventions as technical
NEW MATERIALS CREATING OPTIONS FOR OUR FUTURE
 NEW MATERIALS CREATING OPTIONS FOR OUR FUTURE SC 208 APRIL 5, 2016 JOHN BUSH OUTLINE OF CLASS CONTENT Introduction to material science/engineering Some materials frontiers Logic of materials development
NEW MATERIALS CREATING OPTIONS FOR OUR FUTURE SC 208 APRIL 5, 2016 JOHN BUSH OUTLINE OF CLASS CONTENT Introduction to material science/engineering Some materials frontiers Logic of materials development
Module 2 Selection of Materials and Shapes. IIT, Bombay
 Module 2 Selection of Materials and Shapes Lecture 1 Physical and Mechanical Properties of Engineering Materials Instructional objectives At the of this lecture, the student should be able to appreciate
Module 2 Selection of Materials and Shapes Lecture 1 Physical and Mechanical Properties of Engineering Materials Instructional objectives At the of this lecture, the student should be able to appreciate
Engineering Materials & Minerals
 Course Book Engineering Materials & Minerals Lecturer: Dr.Payman Suhbat Ahmed E-mail: payman.suhbat@koyauniversity.org Coordinator: Nawzat Rashad Ismail E-mail: nawzat.rashad@koyauniversity.org 2 nd Stage
Course Book Engineering Materials & Minerals Lecturer: Dr.Payman Suhbat Ahmed E-mail: payman.suhbat@koyauniversity.org Coordinator: Nawzat Rashad Ismail E-mail: nawzat.rashad@koyauniversity.org 2 nd Stage
Corrosion Issues & Test Methods. Jerry Byers
 Corrosion Issues & Test Methods Jerry Byers Cost of Corrosion to US Industries Corrosion is the deterioration of a material (often a metal) due to interaction with its environment. A study in 1998 estimated
Corrosion Issues & Test Methods Jerry Byers Cost of Corrosion to US Industries Corrosion is the deterioration of a material (often a metal) due to interaction with its environment. A study in 1998 estimated
Properties of Dental Materials
 Properties of Dental Materials Importance of Studying Properties of Materials Materials used to replace teeth or parts of teeth must withstand oral environment and forces of mastication Materials must
Properties of Dental Materials Importance of Studying Properties of Materials Materials used to replace teeth or parts of teeth must withstand oral environment and forces of mastication Materials must
NCERT solutions for Metals and Non Metals
 NCERT solutions for Metals and Non Metals 1 Question 1 Give an example of a metal which (i) is a liquid at room temperature. (ii) can be easily cut with a knife. (iii) is the best conductor of heat. (iv)
NCERT solutions for Metals and Non Metals 1 Question 1 Give an example of a metal which (i) is a liquid at room temperature. (ii) can be easily cut with a knife. (iii) is the best conductor of heat. (iv)
Selection of Engineering Materials
 Selection of Engineering IM 515E Dr Yehia M. Youssef 1 Textbook: Budinski, K.G. and Budinski, M.K., Engineering : Properties and selection, 8 th ed., Prentice Hall, 2005. Other References: 1) Ashby, M.,
Selection of Engineering IM 515E Dr Yehia M. Youssef 1 Textbook: Budinski, K.G. and Budinski, M.K., Engineering : Properties and selection, 8 th ed., Prentice Hall, 2005. Other References: 1) Ashby, M.,
Nanodiamond-Polymer Composite Fibers and Coatings
 Nanodiamond-Polymer Composite Fibers and Coatings Yury Gogotsi et al. A.J. Drexel Nanotechnology Institute and Department of Materials Science and Engineering Drexel University, Philadelphia, Pennsylvania
Nanodiamond-Polymer Composite Fibers and Coatings Yury Gogotsi et al. A.J. Drexel Nanotechnology Institute and Department of Materials Science and Engineering Drexel University, Philadelphia, Pennsylvania
Engineering Materials
 Engineering Materials Lecture 2 MEL120: Manufacturing Practices 1 Selection of Material A particular material is selected is on the basis of following considerations 1. Properties of material 1. Properties
Engineering Materials Lecture 2 MEL120: Manufacturing Practices 1 Selection of Material A particular material is selected is on the basis of following considerations 1. Properties of material 1. Properties
Turn off all electronic devices
 Knives and Steel 1 Knives and Steel Observations about Knives and Steel Knives and Steel 2 Some knives can t keep their cutting edges Some knives bend while others break Making good knives involves heat
Knives and Steel 1 Knives and Steel Observations about Knives and Steel Knives and Steel 2 Some knives can t keep their cutting edges Some knives bend while others break Making good knives involves heat
What happens if we connect Zn and Pt in HCl solution? Corrosion of platinum (Pt) in HCl. 1. If Zn and Pt are not connected
 Corrosion of platinum (Pt) in HCl Now if we place a piece of Pt in HCl, what will happen? Pt does not corrode does not take part in the electrochemical reaction Pt is a noble metal Pt acts as a reference
Corrosion of platinum (Pt) in HCl Now if we place a piece of Pt in HCl, what will happen? Pt does not corrode does not take part in the electrochemical reaction Pt is a noble metal Pt acts as a reference
Offshore Wind Turbines Power Electronics Design and Reliability Research
 Offshore Wind Turbines Power Electronics Design and Reliability Research F. P. McCluskey CALCE/Dept. Of Mechanical Engineering University of Maryland, College Park, MD (301) 405-0279 mcclupa@umd.edu 1
Offshore Wind Turbines Power Electronics Design and Reliability Research F. P. McCluskey CALCE/Dept. Of Mechanical Engineering University of Maryland, College Park, MD (301) 405-0279 mcclupa@umd.edu 1
Chapter 3: Metals and Non-metals Question 1: Define amphoteric oxides. Give two examples. Answer: Oxides that react with both acids and bases to form
 Chapter 3: Metals and Non-metals Question 1: Define amphoteric oxides. Give two examples. Oxides that react with both acids and bases to form salt and water are known as amphoteric oxides. Examples: PbO
Chapter 3: Metals and Non-metals Question 1: Define amphoteric oxides. Give two examples. Oxides that react with both acids and bases to form salt and water are known as amphoteric oxides. Examples: PbO
Corrosion. Cause of Corrosion: Electrochemical Mechanism of Corrosion (Rusting of Iron)
 Corrosion Any process of deterioration (or destruction) and consequent loss of a solid metallic material, through an unwanted (or unintentional) chemical or electrochemical attack by its environment, starting
Corrosion Any process of deterioration (or destruction) and consequent loss of a solid metallic material, through an unwanted (or unintentional) chemical or electrochemical attack by its environment, starting
USNA Chemistry Department CORROSION LECTURE. USNA Chemistry Dept.
 USNA Chemistry Department CORROSION LECTURE Corrosion Why is Corrosion Important? Corrosion is the destructive attack of a metal due to an ELECTROCHEMICAL reaction. Attack of Elements on Structural Metals
USNA Chemistry Department CORROSION LECTURE Corrosion Why is Corrosion Important? Corrosion is the destructive attack of a metal due to an ELECTROCHEMICAL reaction. Attack of Elements on Structural Metals
AERO 214. Introduction to Aerospace Mechanics of Materials. Lecture 2
 AERO 214 Introduction to Aerospace Mechanics of Materials Lecture 2 Materials for Aerospace Structures Aluminum Titanium Composites: Ceramic Fiber-Reinforced Polymer Matrix Composites High Temperature
AERO 214 Introduction to Aerospace Mechanics of Materials Lecture 2 Materials for Aerospace Structures Aluminum Titanium Composites: Ceramic Fiber-Reinforced Polymer Matrix Composites High Temperature
Properties of Engineering Materials
 Properties of Engineering Materials Syllabus Mechanical Properties, Tensile, Fatigue, Creep, Impact, Hardness, Chemical Properties, Physical properties, Corrosion and Cathodic Protection, Carbon Steel,
Properties of Engineering Materials Syllabus Mechanical Properties, Tensile, Fatigue, Creep, Impact, Hardness, Chemical Properties, Physical properties, Corrosion and Cathodic Protection, Carbon Steel,
Thermodynamics and Electrode Potential ME Dr. Zuhair M. Gasem
 Thermodynamics and Electrode Potential ME 472-062 Copyright Dr. Zuhair M. Gasem Corrosion Science and Engineering 2 Corrosion Science Engineering: corrosion forms, and controlling methods Chpater2 Thermodynamics
Thermodynamics and Electrode Potential ME 472-062 Copyright Dr. Zuhair M. Gasem Corrosion Science and Engineering 2 Corrosion Science Engineering: corrosion forms, and controlling methods Chpater2 Thermodynamics
 CHAPTER 3 METALS AND NON-METALS About 118 elements are known today. There are more than 90 metals, 22 non metals and a few metalloids. Sodium (Na), potassium (K), magnesium(mg), aluminium(al), calcium(ca),
CHAPTER 3 METALS AND NON-METALS About 118 elements are known today. There are more than 90 metals, 22 non metals and a few metalloids. Sodium (Na), potassium (K), magnesium(mg), aluminium(al), calcium(ca),
Types of corrosion. 1)Uniform or General Corrosion
 Types of corrosion It is convenient to classify corrosion by the forms in which it manifests itself, the basis for this classification being the appearance of the corroded metal. In most cases the naked
Types of corrosion It is convenient to classify corrosion by the forms in which it manifests itself, the basis for this classification being the appearance of the corroded metal. In most cases the naked
Project: Corrosion of Iron
 Project: Corrosion of Iron Potential Credits: /15 Name: Goal: The goal of this lab is to help you acquaint you with the process of corrosion and to investigate how it can be slowed. You will be provided
Project: Corrosion of Iron Potential Credits: /15 Name: Goal: The goal of this lab is to help you acquaint you with the process of corrosion and to investigate how it can be slowed. You will be provided
Corrosion Rate Measurement on C-Steel
 Measurements of corrosion rate on Carbon-steel using Electrochemical (potentiodynamic Polarization, EIS etc.) technique. Corrosion Rate Measurement on C-Steel Abdullah Al Ashraf 1. Introduction: The degradation
Measurements of corrosion rate on Carbon-steel using Electrochemical (potentiodynamic Polarization, EIS etc.) technique. Corrosion Rate Measurement on C-Steel Abdullah Al Ashraf 1. Introduction: The degradation
PROCON I N T E R N A T I O N A L, L L C. CONSULTING ARCHITECTS & ENGINEERS CONSTRUCTION TECHNOLOGY
 PROCON P R O F E S S I O N A L C O N S U L T A N T S I N T E R N A T I O N A L, L L C CONSULTING ARCHITECTS & ENGINEERS CONSTRUCTION TECHNOLOGY www.proconweb.com Assessing the Performance of Metal Trim
PROCON P R O F E S S I O N A L C O N S U L T A N T S I N T E R N A T I O N A L, L L C CONSULTING ARCHITECTS & ENGINEERS CONSTRUCTION TECHNOLOGY www.proconweb.com Assessing the Performance of Metal Trim
Iron is found in an oxidized state and is mined from the ground as an iron ore.
 Iron is found in an oxidized state and is mined from the ground as an iron ore. Energy is used to convert this iron oxide into steel. Ore is loaded into a blast furnace where heat energy removes the oxygen
Iron is found in an oxidized state and is mined from the ground as an iron ore. Energy is used to convert this iron oxide into steel. Ore is loaded into a blast furnace where heat energy removes the oxygen
UNIT-I ELECTROCHEMISTRY PART-A
 UNIT-I ELECTROCHEMISTRY PART-A 1. What is electrochemistry? 2. What do you understand by electrode potential? 3. Define E.M.F of an electrochemical cell? 4. Define (a) Single electrode potential (b) Standard
UNIT-I ELECTROCHEMISTRY PART-A 1. What is electrochemistry? 2. What do you understand by electrode potential? 3. Define E.M.F of an electrochemical cell? 4. Define (a) Single electrode potential (b) Standard
I. PHYSICAL PROPERTIES. PROPERTY METALS NON-METALS 1.Lustre Metals have shining surface. They do not have shining surface.
 Elements can be classified as metals and non-metals on the basis of their properties. Example of some metals are : Iron (Fe), Aluminium (Al), Silver (Ag), Copper (Cu) Examples of some non-metals are :
Elements can be classified as metals and non-metals on the basis of their properties. Example of some metals are : Iron (Fe), Aluminium (Al), Silver (Ag), Copper (Cu) Examples of some non-metals are :
I. PHYSICAL PROPERTIES PROPERTY METALS NON-METALS
 Elements can be classified as metals and non-metals on the basis of their properties. Example of some metals are : Iron (Fe), Aluminium (Al), Silver (Ag), Copper (Cu) Examples of some non-metals are :
Elements can be classified as metals and non-metals on the basis of their properties. Example of some metals are : Iron (Fe), Aluminium (Al), Silver (Ag), Copper (Cu) Examples of some non-metals are :
1.6.3 Measurement of Linear Coefficients of Thermal Expansion Thermal Strains Thermal Shock
 1 The Properties of Refractory and Heat Insulation Materials... 1 1.1 Classifications and Some Words About Quality Control... 1 1.2 Density, Porosity, and Related Characteristics; Types of Porosity....................................
1 The Properties of Refractory and Heat Insulation Materials... 1 1.1 Classifications and Some Words About Quality Control... 1 1.2 Density, Porosity, and Related Characteristics; Types of Porosity....................................
Corrosion of metals and their protection
 Chapter 13 Corrosion of metals and their protection 13.1 Corrosion of metals 13.2 Corrosion of iron rusting 13.3 Factors that speed up rusting 13.4 To observe the rusting of iron using rust indicator 13.5
Chapter 13 Corrosion of metals and their protection 13.1 Corrosion of metals 13.2 Corrosion of iron rusting 13.3 Factors that speed up rusting 13.4 To observe the rusting of iron using rust indicator 13.5
Material Science. Prof. Satish V. Kailas Associate Professor Dept. of Mechanical Engineering, Indian Institute of Science, Bangalore India
 Material Science Prof. Satish V. Kailas Associate Professor Dept. of Mechanical Engineering, Indian Institute of Science, Bangalore 560012 India Chapter 13. Corrosion and Degradation of Materials All engineering
Material Science Prof. Satish V. Kailas Associate Professor Dept. of Mechanical Engineering, Indian Institute of Science, Bangalore 560012 India Chapter 13. Corrosion and Degradation of Materials All engineering
Fundamentals of Corrosion and Their Application to Coil-Coated Metal
 Fundamentals of Corrosion and Their Application to Coil-Coated Metal INTRODUCTION This document provides an introduction to the fundamentals of corrosion and its relevance to coil-coated metal. The intent
Fundamentals of Corrosion and Their Application to Coil-Coated Metal INTRODUCTION This document provides an introduction to the fundamentals of corrosion and its relevance to coil-coated metal. The intent
REFRACTORY CERAMIC FIBER TEXTILE
 REFRACTORY CERAMIC FIBER TEXTILE UNITECO Refractory ceramic fiber textile is made from ceramic fiber raw material with glasses fiber or refractory stainless steel wire as strengthening stuff, the fiber
REFRACTORY CERAMIC FIBER TEXTILE UNITECO Refractory ceramic fiber textile is made from ceramic fiber raw material with glasses fiber or refractory stainless steel wire as strengthening stuff, the fiber
Topic: Gases in the Atmosphere
 Topic: Gases in the Atmosphere Composition of the atmosphere The two main gases making up 99% of dry air are nitrogen and oxygen. Our air also contains varying amounts of water vapour, which we refer to
Topic: Gases in the Atmosphere Composition of the atmosphere The two main gases making up 99% of dry air are nitrogen and oxygen. Our air also contains varying amounts of water vapour, which we refer to
Materials Science Applied to Household Appliances
 Abstract Materials Science Applied to Household Appliances Adapted from PNNL Teachers Handbook* This exercise gives students experience with materials used in products that they are around every day. It
Abstract Materials Science Applied to Household Appliances Adapted from PNNL Teachers Handbook* This exercise gives students experience with materials used in products that they are around every day. It
أت ارش. Dr. Abdel-Wahab El-Morsy Faculty of Engineering - Rabigh
 Basic Workshop 1 أت ارش There are thousands of materials available for use in engineering applications. Most materials fall into one of three classes that are based on the atomic bonding forces of a particular
Basic Workshop 1 أت ارش There are thousands of materials available for use in engineering applications. Most materials fall into one of three classes that are based on the atomic bonding forces of a particular
Environmental Interactions
 Environmental Interactions Chemical reaction between the material and its environment Beneficial interactions: materials processing Carburization and nitriding hardens for wear resistance Doping adds electrically
Environmental Interactions Chemical reaction between the material and its environment Beneficial interactions: materials processing Carburization and nitriding hardens for wear resistance Doping adds electrically
APPLICATIONS OF ELECTROCHEMISTRY
 APPLICATIONS OF ELECTROCHEMISTRY SPONTANEOUS REDOX REACTIONS APPLICATIONS OF ELECTROCHEMICAL CELLS BATTERIES A galvanic cell, or series of combined galvanic cells, that can be used as a source of direct
APPLICATIONS OF ELECTROCHEMISTRY SPONTANEOUS REDOX REACTIONS APPLICATIONS OF ELECTROCHEMICAL CELLS BATTERIES A galvanic cell, or series of combined galvanic cells, that can be used as a source of direct
Synthesis and Characterization of Zinc Iron Sulphide (ZnFeS) Of Varying Zinc Ion Concentration
 International Journal of Science and Technology Volume 5 No. 5, May, 2016 Synthesis and Characterization of Zinc Iron Sulphide (ZnFeS) Of Varying Zinc Ion Concentration I. B. Obasi 1 and J. C. Osuwa 2
International Journal of Science and Technology Volume 5 No. 5, May, 2016 Synthesis and Characterization of Zinc Iron Sulphide (ZnFeS) Of Varying Zinc Ion Concentration I. B. Obasi 1 and J. C. Osuwa 2
CO forms CO 2. forms. (a) The coke reacts with the oxygen in the air to form carbon dioxide. C + O 2
 1 Iron is extracted from the ore hematite in the Blast Furnace. waste gases firebrick lining raw materials: coke, C iron ore, Fe 2 O 3 limestone, CaCO 3 CO forms air slag molten iron CO 2 forms (a) The
1 Iron is extracted from the ore hematite in the Blast Furnace. waste gases firebrick lining raw materials: coke, C iron ore, Fe 2 O 3 limestone, CaCO 3 CO forms air slag molten iron CO 2 forms (a) The
There s also got to be a wire, but that s kind of taken for granted.
 RedOx Pt 2 Electrochemical Cells (AKA simple batteries) An electrochemical cell has 3 major components: 1. The Cathode (and it s corresponding solution) 2. The Anode (and it s corresponding solution) 3.
RedOx Pt 2 Electrochemical Cells (AKA simple batteries) An electrochemical cell has 3 major components: 1. The Cathode (and it s corresponding solution) 2. The Anode (and it s corresponding solution) 3.
Environmental Degradation of Micro- & Nano-Composites
 Environmental Degradation of Micro- & Nano-Composites Dr. Muhammad Shahid Dept of Mechanical Engineering, College of EME, NUST mshahid@ceme.edu.pk www.ceme.edu.pk Corrosion Deterioration of materials (generally
Environmental Degradation of Micro- & Nano-Composites Dr. Muhammad Shahid Dept of Mechanical Engineering, College of EME, NUST mshahid@ceme.edu.pk www.ceme.edu.pk Corrosion Deterioration of materials (generally
Material Science. Prof. Satish V. Kailas Associate Professor Dept. of Mechanical Engineering, Indian Institute of Science, Bangalore India
 Material Science Prof. Satish V. Kailas Associate Professor Dept. of Mechanical Engineering, Indian Institute of Science, Bangalore 560012 India Chapter 13. Corrosion and Degradation of Materials What
Material Science Prof. Satish V. Kailas Associate Professor Dept. of Mechanical Engineering, Indian Institute of Science, Bangalore 560012 India Chapter 13. Corrosion and Degradation of Materials What
Chapter 20 CHEMISTRY. Metallurgy and the Chemistry of Metals. Dr. Ibrahim Suleiman
 CHEMISTRY Chapter 20 Metallurgy and the Chemistry of Metals Dr. Ibrahim Suleiman GENERAL PROPERTIES AND STRUCTURE OF METALS opaque good conductors of heat and electricity high malleability and ductility
CHEMISTRY Chapter 20 Metallurgy and the Chemistry of Metals Dr. Ibrahim Suleiman GENERAL PROPERTIES AND STRUCTURE OF METALS opaque good conductors of heat and electricity high malleability and ductility
BASICS OF CORROSION. Dr. Ramazan Kahraman
 BASICS OF CORROSION Reading Material: Chapter 1 in Principles and Prevention of Corrosion, Denny Jones, Prentice-Hall, 1996. Dr. Ramazan Kahraman Chemical Engineering Department King Fahd University of
BASICS OF CORROSION Reading Material: Chapter 1 in Principles and Prevention of Corrosion, Denny Jones, Prentice-Hall, 1996. Dr. Ramazan Kahraman Chemical Engineering Department King Fahd University of
Metals and Non-metals
 Metals and Non-metals Q.1. Define amphoteric oxides. Give two examples. Oxides that react with both acids and bases to form salt and water are known as amphoteric oxides. Examples: PbO and Al2O3. Q.2.
Metals and Non-metals Q.1. Define amphoteric oxides. Give two examples. Oxides that react with both acids and bases to form salt and water are known as amphoteric oxides. Examples: PbO and Al2O3. Q.2.
Free Electron Model What kind of interactions hold metal atoms together? How does this explain high electrical and thermal conductivity?
 Electrical Good conductors of heat & electricity Create semiconductors Oxides are basic ionic solids Aqueous cations (positive charge, Lewis acids) Reactivity increases downwards in family Mechanical Lustrous
Electrical Good conductors of heat & electricity Create semiconductors Oxides are basic ionic solids Aqueous cations (positive charge, Lewis acids) Reactivity increases downwards in family Mechanical Lustrous
What is Electroplating?
 Electroplating 1 What is Electroplating? An electrochemical process where metal ions are transferred from a solution and are deposited as a thin layer onto surface of a cathode. The setup is composed DC
Electroplating 1 What is Electroplating? An electrochemical process where metal ions are transferred from a solution and are deposited as a thin layer onto surface of a cathode. The setup is composed DC
TWEED RIVER HIGH SCHOOL 2006 PRELIMINARY CHEMISTRY. Unit 2 Metals
 TWEED RIVER HIGH SCHOOL 2006 PRELIMINARY CHEMISTRY Unit 2 Metals Part 2 Metals differ in their reactivity with other chemicals and this influences their uses. Describe observable changes when metals react
TWEED RIVER HIGH SCHOOL 2006 PRELIMINARY CHEMISTRY Unit 2 Metals Part 2 Metals differ in their reactivity with other chemicals and this influences their uses. Describe observable changes when metals react
CORROSION AND CORROSION CONTROL An Introduction to Corrosion Science and Engineering
 CORROSION AND CORROSION CONTROL An Introduction to Corrosion Science and Engineering FOURTH EDITION R. Winston Revie Senior Research Scientist CANMET Materials Technology Laboratory Natural Resources Canada
CORROSION AND CORROSION CONTROL An Introduction to Corrosion Science and Engineering FOURTH EDITION R. Winston Revie Senior Research Scientist CANMET Materials Technology Laboratory Natural Resources Canada
CHAPTER INTRODUCTION
 1 CHAPTER-1 1.0 INTRODUCTION Contents 1.0 Introduction 1 1.1 Aluminium alloys 2 1.2 Aluminium alloy classification 2 1.2.1 Aluminium alloys (Wrought) 3 1.2.2 Heat treatable alloys (Wrought). 3 1.2.3 Aluminum
1 CHAPTER-1 1.0 INTRODUCTION Contents 1.0 Introduction 1 1.1 Aluminium alloys 2 1.2 Aluminium alloy classification 2 1.2.1 Aluminium alloys (Wrought) 3 1.2.2 Heat treatable alloys (Wrought). 3 1.2.3 Aluminum
Slide 1. Slide 2. Slide 3. Chapter 19: Electronic Materials. Learning Objectives. Introduction
 Slide 1 Chapter 19: Electronic Materials 19-1 Slide 2 Learning Objectives 1. Ohm s law and electrical conductivity 2. Band structure of solids 3. Conductivity of metals and alloys 4. Semiconductors 5.
Slide 1 Chapter 19: Electronic Materials 19-1 Slide 2 Learning Objectives 1. Ohm s law and electrical conductivity 2. Band structure of solids 3. Conductivity of metals and alloys 4. Semiconductors 5.
Electrochemical Considerations
 17-1 CHAPTER 17 CORROSION AND DEGRADATION OF MATERIALS PROBLEM SOLUTIONS Electrochemical Considerations 17.1 (a) Oxidation is the process by which an atom gives up an electron (or electrons) to become
17-1 CHAPTER 17 CORROSION AND DEGRADATION OF MATERIALS PROBLEM SOLUTIONS Electrochemical Considerations 17.1 (a) Oxidation is the process by which an atom gives up an electron (or electrons) to become
PHYSICAL AND CHEMICAL CHANGES CLASS 7. Types of changes: The changes are of two kinds, physical and chemical..
 PHYSICAL AND CHEMICAL CHANGES CLASS 7 Types of changes: The changes are of two kinds, physical and chemical.. Physical Properties of Substances Properties such as shape, size, colour and state of a substance
PHYSICAL AND CHEMICAL CHANGES CLASS 7 Types of changes: The changes are of two kinds, physical and chemical.. Physical Properties of Substances Properties such as shape, size, colour and state of a substance
Extracting a metal from its ore 2004 by David A. Katz. All rights reserved.
 Extracting a metal from its ore 2004 by David A. Katz. All rights reserved. Minerals are solid elements or compounds found naturally in the Earth's crust. Those minerals that contain sufficient metal to
Extracting a metal from its ore 2004 by David A. Katz. All rights reserved. Minerals are solid elements or compounds found naturally in the Earth's crust. Those minerals that contain sufficient metal to
