Pharmacokinetics-pharmacodynamics analysis of spiroindolone analogs and KAE609 in. a murine malaria model
|
|
|
- Deirdre Carson
- 6 years ago
- Views:
Transcription
1 Pharmacokinetics-pharmacodynamics analysis of spiroindolone analogs and KAE609 in a murine malaria model Suresh B. Lakshminarayana 1#, Céline reymond 2,3, Christoph ischli 2,3, Jing Yu 4, Sebastian Weber 5, Anne Goh 1, Bryan K. S. Yeung 1, Paul C. o 6, Véronique Dartois 1,a, Thierry T. Diagana 1, Matthias Rottmann 2,3 and rancesca Blasco 1# 6 7 (1) ovartis Institute for Tropical Diseases, Singapore 8 (2) Swiss Tropical and Public ealth Institute, Basel, Switzerland 9 (3) University of Basel, Switzerland 10 (4) ovartis Institutes for Biomedical Research, Cambridge, MA, USA 11 (5) ovartis Pharma, Basel, Switzerland 12 (6) Department of Pharmacy, ational University of Singapore, Singapore # Corresponding Authors SBL & B Suresh B. Lakshminarayana ovartis Institute for Tropical Diseases, 10 Biopolis Road, #05-01 Chromos Singapore, suresh.b_lakshminarayana@novartis.com; Phone: rancesca Blasco ovartis Institute for Tropical Diseases, 10 Biopolis Road, #05-01 Chromos 22 Singapore,
2 23 Phone: Present address: (a) Public ealth Research Institute Center, ew Jersey, USA
3 43 Supplementary material Table S1. In vitro PK properties of spiroindolone analogs igure S1: Structure of spiroindolone analogs igure S2. Goodness-of-fit plots for dose response relationship 49 igure S3. Goodness-of-fit plots for pharmacokinetic modeling of KAE igure S4. Dose proportionality for KAE609 in CD-1 mouse
4 67 Table S1: In vitro PK properties of spiroindolone analogs 68 Caco-2 (c) epatic CL int Compound Solubility (p 6.8, µm) (a) PAMPA (b) %A P app (A-B) 10-6 cm/sec in mouse liver microsomes (d) (µl/min/mg) Mouse PPB (e) (%) RBC to Plasma Ratio (f) (+) (+) n.t (+) (+) (+) (+) (+)-7 (KAE609) (-) (-) In vitro PK parameters like solubility, PAMPA, Caco-2 permeability, hepatic clearance, mouse plasma protein binding and RBC to plasma partitioning were determined in a medium 4
5 to high through put format using in-house standard assay protocols that are described in the literature. (a) T-Equilibrium solubility at p 6.8 (1); (b) Parallel Artificial Membrane Permeability Assay expressed in % fraction absorbed (A) (2, 3); (c) Apparent permeability from apical to basolateral in Caco-2 cells expressed as P app (A-B) 10-6 cm/sec (4, 5), n.t, not tested; (d) intrinsic clearance in mouse liver microsomes expressed as µl/min/mg protein, in the presence of liver enzymes (6); (e) Mouse plasma protein binding using rapid equilibrium dialysis method (7, 8), (f) RBC to plasma ratio, mean from 1 µm & 10 µm (9)
6 99 igure S1. Structure of spiroindolone analogs. Active spiroindolones O O ( + ) -1 ( + ) -2 O O O ( + ) -3 (+ ) -4 (+ ) -5 O O ( + ) -6 (+ ) -7 (ITD609) Inactive spiroindolones O O ( - ) -6 ( - ) Active spiroindolones have the 1R, 3S configuration while inactive spiroindolones have the 1S, 3R configuration (10)
7 111 igure S2. Goodness-of-fit plots for dose response relationship Goodness-of-fit plots for a representative compound [(+)-6] (A) observed data (DV) vs populations predictions (PRED) (B) weighted residuals (WRES) vs PRED (C) WRES vs Dose
8 128 igure S3. Goodness-of-fit plots for pharmacokinetic modeling of KAE Goodness-of-fit plots for KAE609 pharmacokinetic modeling (A) observed data (DV) vs populations predictions (PRED) (B) weighted residuals (WRES) vs PRED (C) WRES vs Time
9 145 igure S4. Dose proportionality for KAE609 in CD-1 mouse 100 A 1000 B C max (µg/ml) AUC (µg*h/ml) Dose (mg/kg) Dose (mg/kg) Dose linearity tests on C max and AUC 0-24 were carried out by the regression of logtransformed data (power regression model) (11, 12). Doses ( mg/kg) and PK parameters (C max and AUC 0-24 ) were log-transformed, and correlation coefficient (R 2 ), slope, and 95 % confidence intervals of slope were calculated using the GraphPad Prism version 5.02 for windows (GraphPad software, San Diego, California USA). The system was considered to be linear when R 2 ~ 1, Slope 95 % CI lower > 0.8, and CI upper < 1.25 (11, 12). (A) Dose vs C max (R 2 = 0.97, Slope = 1.18, 95 % CI lower = 0.52 and CI upper = 1.83); (B) Dose vs AUC 0-24 (R 2 = 0.97, Slope = 1.30, 95 % CI lower = 0.65 and CI upper = 1.96). Based on the above observations, KAE609 shows nonlinear behavior in CD-1 mouse between the doses studied
10 163 Reference List Zhou, L., L. Yang, S. Tilton, and J. Wang Development of a high throughput equilibrium solubility assay using miniaturized shake-flask method in early drug discovery. J.Pharm.Sci. 96: doi: /jps [doi] Avdeef, A The rise of PAMPA. Expert.Opin.Drug Metab Toxicol. 1: doi: / [doi] Avdeef, A., S. Bendels, L. Di, B. aller, M. Kansy, K. Sugano, and Y. Yamauchi PAMPA--critical factors for better predictions of absorption. J.Pharm.Sci. 96: doi: /jps [doi] Balimane, P. V. and S. Chong Cell culture-based models for intestinal permeability: a critique. Drug Discov.Today 10: doi:s [pii]; /s (04) [doi] Marino, A. M., M. Yarde,. Patel, S. Chong, and P. V. Balimane Validation of the 96 well Caco-2 cell culture model for high throughput permeability assessment of discovery compounds. Int.J.Pharm. 297: doi:s (05) [pii]; /j.ijpharm [doi] Obach, R. S Prediction of human clearance of twenty-nine drugs from hepatic microsomal intrinsic clearance data: An examination of in vitro half-life approach and nonspecific binding to microsomes. Drug Metab Dispos. 27:
11 Banker, M. J., T.. ark, and J. A. Williams Development and validation of a 96-well equilibrium dialysis apparatus for measuring plasma protein binding. J.Pharm.Sci. 92: doi: /jps [doi] Kariv, I.,. Cao, and K. R. Oldenburg Development of a high throughput equilibrium dialysis method. J.Pharm.Sci. 90: doi: / (200105)90:5<580::aid-jps1014>3.0.co;2-4 [pii] Yu, S., S. Li,. Yang,. Lee, J. T. Wu, and M. G. Qian A novel liquid chromatography/tandem mass spectrometry based depletion method for measuring red blood cell partitioning of pharmaceutical compounds in drug discovery. Rapid Commun.Mass Spectrom. 19: doi: /rcm.1777 [doi] Yeung, B. K., B. Zou, M. Rottmann, S. B. Lakshminarayana, S.. Ang, S. Y. Leong, J. Tan, J. Wong, S. Keller-Maerki, C. ischli, A. Goh, E. K. Schmitt, P. Krastel, E. rancotte, K. Kuhen, D. Plouffe, K. enson, T. Wagner, E. A. Winzeler,. Petersen, R. Brun, V. Dartois, T. T. Diagana, and T.. Keller Spirotetrahydro beta-carbolines (spiroindolones): a new class of potent and orally efficacious compounds for the treatment of malaria. J.Med.Chem. 53: doi: /jm100410f [doi] ummel, J., S. McKendrick, C. Brindley, and R. rench Exploratory assessment of dose proportionality: review of current approaches and proposal for a practical criterion. Pharm.Stat. 8: doi: /pst.326 [doi] Smith, B. P.,. R. Vandenhende, K. A. DeSante,. A. arid, P. A. Welch, J. T. Callaghan, and S. T. orgue Confidence interval criteria for assessment of dose proportionality. Pharm.Res. 17:
the treatment of malaria The NGBS consortium Thierry Diagana MMV stakeholder meeting, Barcelona- March 15 th 2010
 Spiroindolones: a new chemotype for the treatment of malaria The NGBS consortium Thierry Diagana MMV stakeholder meeting, Barcelona- March 15 th 2010 The NGBS consortium A public-private i partnership
Spiroindolones: a new chemotype for the treatment of malaria The NGBS consortium Thierry Diagana MMV stakeholder meeting, Barcelona- March 15 th 2010 The NGBS consortium A public-private i partnership
Using WinNonlin with UNIFI: Enhanced Export for the Metabolite Identification Application Solution
 Using WinNonlin with UNIFI: Enhanced Export for the Metabolite Identification Application Solution Yun Alelyunas, Paul Rainville, and Mark Wrona; Waters Corporation GOAL To demonstrate a simple workflow
Using WinNonlin with UNIFI: Enhanced Export for the Metabolite Identification Application Solution Yun Alelyunas, Paul Rainville, and Mark Wrona; Waters Corporation GOAL To demonstrate a simple workflow
Drug Discovery at the Swiss TPH
 Medical Parasitology & Infection Biology Parasite Chemotherapy Unit Building on Success Malaria Control and Elimination Swiss TPH Winter Symposium 2016 Drug Discovery at the Swiss TPH Matthias Rottmann
Medical Parasitology & Infection Biology Parasite Chemotherapy Unit Building on Success Malaria Control and Elimination Swiss TPH Winter Symposium 2016 Drug Discovery at the Swiss TPH Matthias Rottmann
SUPPLEMENTARY INFORMATION
 In format provided by Orchard et al. (SEPTEMBER 2011) Supplementary information S1 Sample MIABE file exemplifying data extracted from PMID: 20568778 Responsible Person or Role Contact Person Bryan K. S.
In format provided by Orchard et al. (SEPTEMBER 2011) Supplementary information S1 Sample MIABE file exemplifying data extracted from PMID: 20568778 Responsible Person or Role Contact Person Bryan K. S.
NITD609: a novel and potent drug candidate for the treatment of uncomplicated malaria
 NITD609: a novel and potent drug candidate for the treatment of uncomplicated malaria Thierry Diagana, Novartis Institute for Tropical Diseases New Delhi, November 7 th 2012 MMV stakeholders meeting Treatment
NITD609: a novel and potent drug candidate for the treatment of uncomplicated malaria Thierry Diagana, Novartis Institute for Tropical Diseases New Delhi, November 7 th 2012 MMV stakeholders meeting Treatment
Preclinical pharmacokinetics and pharmacodynamics of AG-519, an allosteric pyruvate kinase activator
 S830 Preclinical pharmacokinetics and pharmacodynamics of AG-519, an allosteric pyruvate kinase activator Yue Chen, Raj Nagaraja, Kha Le, Penelope A Kosinski, Gavin Histen, Charles Kung, Hyeryun Kim, Chandra
S830 Preclinical pharmacokinetics and pharmacodynamics of AG-519, an allosteric pyruvate kinase activator Yue Chen, Raj Nagaraja, Kha Le, Penelope A Kosinski, Gavin Histen, Charles Kung, Hyeryun Kim, Chandra
Information Sharing and Automation of PK Calculations for Efficient Project Support
 Information Sharing and Automation of PK Calculations for Efficient Project Support Tibco Spotfire UM, Basel, Nov 3 rd, 2016 Stefanie Bendels Quantitative Systems Pharmacology Pharmaceutical Sciences,
Information Sharing and Automation of PK Calculations for Efficient Project Support Tibco Spotfire UM, Basel, Nov 3 rd, 2016 Stefanie Bendels Quantitative Systems Pharmacology Pharmaceutical Sciences,
Informatics challenges in data management, high-throughput screening and development of predictive models of ADME properties
 Informatics challenges in data management, high-throughput screening and development of predictive models of ADME properties Peter Gund 1, Janet Cohen 1, William J. Egan 1,2, Osman F. Güner 3, and Kirk
Informatics challenges in data management, high-throughput screening and development of predictive models of ADME properties Peter Gund 1, Janet Cohen 1, William J. Egan 1,2, Osman F. Güner 3, and Kirk
Supporting information. Single-cell and subcellular pharmacokinetic imaging allows insight into drug action in vivo
 Supporting information Single-cell and subcellular pharmacokinetic imaging allows insight into drug action in vivo Greg Thurber 1, Katy Yang 1, Thomas Reiner 1, Rainer Kohler 1, Peter Sorger 2, Tim Mitchison
Supporting information Single-cell and subcellular pharmacokinetic imaging allows insight into drug action in vivo Greg Thurber 1, Katy Yang 1, Thomas Reiner 1, Rainer Kohler 1, Peter Sorger 2, Tim Mitchison
Handbook of Essential Pharmacokinetics, Pharmacodynamics and Drug Metabolism for Industrial Scientists
 Handbook of Essential Pharmacokinetics, Pharmacodynamics and Drug Metabolism for Industrial Scientists Handbook of Essential Pharmacokinetics, Pharmacodynamics and Drug Metabolism for Industrial Scientists
Handbook of Essential Pharmacokinetics, Pharmacodynamics and Drug Metabolism for Industrial Scientists Handbook of Essential Pharmacokinetics, Pharmacodynamics and Drug Metabolism for Industrial Scientists
Pharmacokinetics. Processes, Mathematics, and Applications. Second Edition. Peter G. Welling. Institut de Recherche Jouveinal
 Pharmacokinetics Processes, Mathematics, and Applications Second Edition Peter G. Welling Institut de Recherche Jouveinal ACS Professional Reference Book American Chemical Society Washington, DC Contents
Pharmacokinetics Processes, Mathematics, and Applications Second Edition Peter G. Welling Institut de Recherche Jouveinal ACS Professional Reference Book American Chemical Society Washington, DC Contents
Biopharmaceutics Applications in Drug Development
 Biopharmaceutics Applications in Drug Development Edited by Rajesh Krishna Merck & Co., Inc. Rahwav, NJ. USA and Lawrence Yu Center for Drug Evaluation & Research Rockville, MD, USA 4y Springer Contents
Biopharmaceutics Applications in Drug Development Edited by Rajesh Krishna Merck & Co., Inc. Rahwav, NJ. USA and Lawrence Yu Center for Drug Evaluation & Research Rockville, MD, USA 4y Springer Contents
Lipoidal permeability central to drug ADME Major barriers to drug discovery: dogma surrounding plasma protein binding and tissue concentration
 Lipoidal permeability central to drug ADME Major barriers to drug discovery: dogma surrounding plasma protein binding and tissue concentration Lipoidal permeability of typical antagonists / inhibitors
Lipoidal permeability central to drug ADME Major barriers to drug discovery: dogma surrounding plasma protein binding and tissue concentration Lipoidal permeability of typical antagonists / inhibitors
Workshop: Applying Exposure Science to Increase the Utility of Non-Animal Data in Efficacy and Safety Testing, 16 Feb. 2017
 Workshop: Applying Exposure Science to Increase the Utility of Non-Animal Data in Efficacy and Safety Testing, 16 Feb. 2017 Presentation Outline Should we account for kinetics in vitro and in vivo for
Workshop: Applying Exposure Science to Increase the Utility of Non-Animal Data in Efficacy and Safety Testing, 16 Feb. 2017 Presentation Outline Should we account for kinetics in vitro and in vivo for
drug discovery: Where are we now? How did we RSC February 2013
 The role of pharmacokinetics in drug discovery: Where are we now? How did we get th here??where are we going? Peter Webborn RSC February 2013 Why conduct PK studies in animals? The primary purpose of pre-clinical
The role of pharmacokinetics in drug discovery: Where are we now? How did we get th here??where are we going? Peter Webborn RSC February 2013 Why conduct PK studies in animals? The primary purpose of pre-clinical
PK and PK/PD Guided Starting Dose Selection for First-In-Human Trials. Sylvia Zhao ( 赵子微 ) Translational Clinical Oncology Novartis
 PK and PK/PD Guided Starting Dose Selection for First-In-Human Trials Sylvia Zhao ( 赵子微 ) Translational Clinical Oncology Novartis Disclaimer Contents are the opinion of the author and not that of Novartis
PK and PK/PD Guided Starting Dose Selection for First-In-Human Trials Sylvia Zhao ( 赵子微 ) Translational Clinical Oncology Novartis Disclaimer Contents are the opinion of the author and not that of Novartis
Use of Mechanistic Population Based PBPK Models in the Establishment and Application of IVIVCs. Nikunjkumar Patel, Simcyp Limited
 Use of Mechanistic Population Based PBPK Models in the Establishment and Application of IVIVCs Nikunjkumar Patel, Simcyp Limited IVIVC and Its Components CONVOLUTION VALIDATION DECONVOLUTION MODELS IVIVC
Use of Mechanistic Population Based PBPK Models in the Establishment and Application of IVIVCs Nikunjkumar Patel, Simcyp Limited IVIVC and Its Components CONVOLUTION VALIDATION DECONVOLUTION MODELS IVIVC
In silico Prediction of Bioavailability of Pharmaceutical Formulations Using Population Pharmacokinetic Model Simulation
 In silico Prediction of Bioavailability of Pharmaceutical Formulations Using Population Pharmacokinetic Model Simulation Uthpali Mannapperuma Mahidol University Department of Pharmacy, Faculty of Pharmacy,
In silico Prediction of Bioavailability of Pharmaceutical Formulations Using Population Pharmacokinetic Model Simulation Uthpali Mannapperuma Mahidol University Department of Pharmacy, Faculty of Pharmacy,
Simultaneous in vivo Quantification and Metabolite Identification of Plasma Samples Using High Resolution QTof and Routine MS E Data Analysis
 Simultaneous in vivo Quantification and Metabolite Identification of Plasma Samples Using High Resolution QTof and Routine MS E Data Analysis Mark Wrona, 1 Paul Rainville, 1 Eric Langlois, 2 Nigel Ewing,
Simultaneous in vivo Quantification and Metabolite Identification of Plasma Samples Using High Resolution QTof and Routine MS E Data Analysis Mark Wrona, 1 Paul Rainville, 1 Eric Langlois, 2 Nigel Ewing,
The relevance of dissolution testing for trimetazidine
 Vol XV, Number 4, December 211 Pages 274-278 Copyright reserved 211 ORIGINAL PAPER Mourad A. 1, Miron Dalia Simona 2, Lupuleasa D. 1, Budura Emma Adriana 1, Voicu A. V. 3 1. University of Medicine and
Vol XV, Number 4, December 211 Pages 274-278 Copyright reserved 211 ORIGINAL PAPER Mourad A. 1, Miron Dalia Simona 2, Lupuleasa D. 1, Budura Emma Adriana 1, Voicu A. V. 3 1. University of Medicine and
Introduction to Drug Development in Commercializing Biomedical Technology
 Introduction to Drug Development in Commercializing Biomedical Technology Kevin W. Hunt, Ph.D. Director of Biopharmaceutical Product Development Office of Technology Development and Head of Translational
Introduction to Drug Development in Commercializing Biomedical Technology Kevin W. Hunt, Ph.D. Director of Biopharmaceutical Product Development Office of Technology Development and Head of Translational
Value Scale (1-5) Summary of Results
 In this example, the set of compounds that were studied satisfied many of the criteria for "drug likeness". However, in this program, there were issues with potency, novelty, and metabolic stability. This
In this example, the set of compounds that were studied satisfied many of the criteria for "drug likeness". However, in this program, there were issues with potency, novelty, and metabolic stability. This
Value Scale (1-5) 1 I. Final Product Profile Characteristics of the product required for patient care
 In this example, the majority of the compounds was active in the xenograft model and showed "drug-like" properties from in-silico and experimental data. However, the animal studies showed moderate toxicity
In this example, the majority of the compounds was active in the xenograft model and showed "drug-like" properties from in-silico and experimental data. However, the animal studies showed moderate toxicity
CHAPTER 4. Milestones of the drug discovery
 CHAPTER 4 Milestones of the drug discovery 4.Milestones of the drug discovery: Highlights the importance of the below critical milestones of the drug discovery and correlated to the current research. 1.
CHAPTER 4 Milestones of the drug discovery 4.Milestones of the drug discovery: Highlights the importance of the below critical milestones of the drug discovery and correlated to the current research. 1.
Abstract. Technical Aspects. Applying GastroPlus for Extensions of Biowaivers for BCS Class II Compounds 2
 Abstract GastroPlus is a mechanistically based simulation software package that predicts absorption, pharmacokinetics, and pharmacodynamics in humans and animals. GastroPlus modeling and simulation has
Abstract GastroPlus is a mechanistically based simulation software package that predicts absorption, pharmacokinetics, and pharmacodynamics in humans and animals. GastroPlus modeling and simulation has
Injectable modified release products
 Guideline on the pharmacokinetic and clinical evaluation of modified release dosage forms (EMA/CPMP/EWP/280/96 Corr1) Injectable modified release products Dr Sotiris Michaleas, National Expert for the
Guideline on the pharmacokinetic and clinical evaluation of modified release dosage forms (EMA/CPMP/EWP/280/96 Corr1) Injectable modified release products Dr Sotiris Michaleas, National Expert for the
Chagas Disease Drug Discovery Entering a New Era. Eric Chatelain, PhD Head of Drug Discovery
 Chagas Disease Drug Discovery Entering a New Era Eric Chatelain, PhD Head of Drug Discovery ICOPA Meeting, Mexico, 12 th August 2014 Chagas Disease Effective immune responses provide control of the infection
Chagas Disease Drug Discovery Entering a New Era Eric Chatelain, PhD Head of Drug Discovery ICOPA Meeting, Mexico, 12 th August 2014 Chagas Disease Effective immune responses provide control of the infection
Σχεδιασμός κλινικών μελετών βιοϊσοδυναμίας (DESIGN OF BIOEQUIVALENCE STUDIES) To demonstrate that two (or more) medicinal products are bioequivalent
 Σχεδιασμός κλινικών μελετών βιοϊσοδυναμίας (DESIGN OF BIOEQUIVALENCE STUDIES) AIM OF BIOEQUIVALENCE STUDIES To demonstrate that two (or more) medicinal products are bioequivalent AND Two medicinal products
Σχεδιασμός κλινικών μελετών βιοϊσοδυναμίας (DESIGN OF BIOEQUIVALENCE STUDIES) AIM OF BIOEQUIVALENCE STUDIES To demonstrate that two (or more) medicinal products are bioequivalent AND Two medicinal products
Fig. 4. A two-compartment pharmacokinetic model.
 3.4 Pharmacokinetic (PK) modeling and simulations Noora Sjöstedt, Wilma Kiander, Heidi Kidron 3.4.1 Introduction (Hanna Kortejärvi) Pharmacokinetic modeling and simulations can be performed with different
3.4 Pharmacokinetic (PK) modeling and simulations Noora Sjöstedt, Wilma Kiander, Heidi Kidron 3.4.1 Introduction (Hanna Kortejärvi) Pharmacokinetic modeling and simulations can be performed with different
KING KHALID UNIVERSITY COLLEGE OF PHARMACY DEPARTMENT OF PHARMACEUTICS COURSE SPECIFICTION FOR PHARMACEUTICAL SCIENCES/PHARM-D
 KING KHALID UNIVERSITY COLLEGE OF PHARMACY DEPARTMENT OF PHARMACEUTICS COURSE SPECIFICTION FOR PHARMACEUTICAL SCIENCES/PHARM-D BY Mohammed Mahmoud Academic Session 147-8 1 st Semester (Semester No 71)
KING KHALID UNIVERSITY COLLEGE OF PHARMACY DEPARTMENT OF PHARMACEUTICS COURSE SPECIFICTION FOR PHARMACEUTICAL SCIENCES/PHARM-D BY Mohammed Mahmoud Academic Session 147-8 1 st Semester (Semester No 71)
From Candidate to Clinic: Strategies to Select, Assess, Formulate, and Deliver the Right Drug Candidate in the Early Phase
 An Executive Summary From Candidate to Clinic: Strategies to Select, Assess, Formulate, and Deliver the Right Drug Candidate in the Early Phase Leveraging knowledge about an API s physicochemical properties
An Executive Summary From Candidate to Clinic: Strategies to Select, Assess, Formulate, and Deliver the Right Drug Candidate in the Early Phase Leveraging knowledge about an API s physicochemical properties
Oral Delivery of Drugs
 Oral Delivery of Drugs 1 S E S S I O N O N E O F T I P P R O J E C T Advantages of taking oral drugs Convenient (storage, portability, premeasured dose) economical non-invasive, often safer route requires
Oral Delivery of Drugs 1 S E S S I O N O N E O F T I P P R O J E C T Advantages of taking oral drugs Convenient (storage, portability, premeasured dose) economical non-invasive, often safer route requires
Screening of the Malaria Box against kinetoplastids: a concrete example of Open Innovation collaborative research
 Screening of the Malaria Box against kinetoplastids: a concrete example of Open Innovation collaborative research Meetings of Stakeholders for Selected Health R&D Demonstration Projects, Exploiting the
Screening of the Malaria Box against kinetoplastids: a concrete example of Open Innovation collaborative research Meetings of Stakeholders for Selected Health R&D Demonstration Projects, Exploiting the
TB-Epidemiology. Tuberculosis Drug Development: PK/PD Challenges
 7 th Symposium on ew Developments in 7 th Global Meeting, Tuberculosis Drug Development: PK/PD Challenges Bernd Meibohm, PhD, FCP Professor & Associate Dean for Graduate Programs and Research College of
7 th Symposium on ew Developments in 7 th Global Meeting, Tuberculosis Drug Development: PK/PD Challenges Bernd Meibohm, PhD, FCP Professor & Associate Dean for Graduate Programs and Research College of
Guideline for Bioequivalence Studies of Generic Products. December 22, 1997
 Guideline for Bioequivalence Studies of Generic Products December 22, 1997 Index Section 1: Introduction Section 2: Terminology Section 3: Tests A. Oral conventional dosage forms and enteric coated products
Guideline for Bioequivalence Studies of Generic Products December 22, 1997 Index Section 1: Introduction Section 2: Terminology Section 3: Tests A. Oral conventional dosage forms and enteric coated products
Real World Drug Discovery A Chemist's Guide to Biotech and Pharmaceutical Research
 Real World Drug Discovery A Chemist's Guide to Biotech and Pharmaceutical Research Robert M. Rydzewski ELSEVIER Amsterdam Boston Heidelberg London New York Oxford Paris San Diego San Francisco Singapore
Real World Drug Discovery A Chemist's Guide to Biotech and Pharmaceutical Research Robert M. Rydzewski ELSEVIER Amsterdam Boston Heidelberg London New York Oxford Paris San Diego San Francisco Singapore
In silico tools to study food-drug interactions, an Industry Perspective
 Paris, April 4th, 2018 Physiologically Based PharmacoKinetic modeling (PBPK): A new Paradigm in Drug Development In silico tools to study food-drug interactions, an Industry Perspective Neil Parrott, Pharmaceutical
Paris, April 4th, 2018 Physiologically Based PharmacoKinetic modeling (PBPK): A new Paradigm in Drug Development In silico tools to study food-drug interactions, an Industry Perspective Neil Parrott, Pharmaceutical
PHARMACOKINETICS. Dr. M.Mothilal Assistant professor
 PHARMACOKINETICS Dr. M.Mothilal Assistant professor 1 OVERVIEW Basic considerations in pharmacokinetics Compartment models One compartment model Assumptions Intravenous bolus administration Intravenous
PHARMACOKINETICS Dr. M.Mothilal Assistant professor 1 OVERVIEW Basic considerations in pharmacokinetics Compartment models One compartment model Assumptions Intravenous bolus administration Intravenous
Predicting Tissue Distribution & Clearance of Antibody Formats to Improve Selectivity of Targeted Therapies
 Predicting Tissue Distribution & Clearance of Antibody Formats to Improve Selectivity of Targeted Therapies Ben-Fillippo Krippendorff, Roche Innovation Center Basel ben-fillippo.krippendorff@roche.com
Predicting Tissue Distribution & Clearance of Antibody Formats to Improve Selectivity of Targeted Therapies Ben-Fillippo Krippendorff, Roche Innovation Center Basel ben-fillippo.krippendorff@roche.com
The Challenges of Designing Cyclic Prodrugs of Opioid Peptides that Permeate the Intestinal Mucosa and the Blood-Brain Barrier
 The Challenges of Designing Cyclic Prodrugs of pioid Peptides that Permeate the Intestinal Mucosa and the Blood-Brain Barrier Ronald T. Borchardt Department of Pharmaceutical Chemistry The University of
The Challenges of Designing Cyclic Prodrugs of pioid Peptides that Permeate the Intestinal Mucosa and the Blood-Brain Barrier Ronald T. Borchardt Department of Pharmaceutical Chemistry The University of
Guideline on the reporting of physiologically based pharmacokinetic (PBPK) modelling and simulation
 13 December 2018 EMA/CHMP/458101/2016 Committee for Medicinal Products for Human Use (CHMP) Guideline on the reporting of physiologically based pharmacokinetic (PBPK) modelling and simulation Draft agreed
13 December 2018 EMA/CHMP/458101/2016 Committee for Medicinal Products for Human Use (CHMP) Guideline on the reporting of physiologically based pharmacokinetic (PBPK) modelling and simulation Draft agreed
INTRODUCTION TO PHARMACOLOGY
 INTRODUCTION TO PHARMACOLOGY Pharmacology is the study of how chemicals interact with the body Endogenous hormones, growth factors, etc Exogenous drugs Two areas of study Pharmacodynamics Interaction of
INTRODUCTION TO PHARMACOLOGY Pharmacology is the study of how chemicals interact with the body Endogenous hormones, growth factors, etc Exogenous drugs Two areas of study Pharmacodynamics Interaction of
BASIC PHARMACOKINETICS AND PHARMACODYNAMICS
 BASIC PHARMACOKINETICS AND PHARMACODYNAMICS An Integrated Textbook and Computer Simulations SARA ROSENBAUM ~ WILEY A lohn WILEY & SONS, INC., PUBLICATION CONTENTS Preface 1 Introduction to Pharmacokinetics
BASIC PHARMACOKINETICS AND PHARMACODYNAMICS An Integrated Textbook and Computer Simulations SARA ROSENBAUM ~ WILEY A lohn WILEY & SONS, INC., PUBLICATION CONTENTS Preface 1 Introduction to Pharmacokinetics
From Discovery to Development of new Drugs. and pitfalls along the way. by Kim Dekermendjian, PhD in Medicine BD & Key Account manager
 From Discovery to Development of new Drugs. and pitfalls along the way by Kim Dekermendjian, PhD in Medicine BD & Key Account manager The roots of Drug Discovery Before 20 th century the term didn't exists,
From Discovery to Development of new Drugs. and pitfalls along the way by Kim Dekermendjian, PhD in Medicine BD & Key Account manager The roots of Drug Discovery Before 20 th century the term didn't exists,
Discovery and Development of Highly Selective and Orally Bioavailable Macrocyclic IDO-1 Inhibitors
 Discovery and Development of Highly Selective and Orally Bioavailable Macrocyclic IDO-1 Inhibitors 1 Tim Briggs Senior Director, Medicinal Chemistry Ensemble Therapeutics Corp. Ensemble Therapeutics Corp.
Discovery and Development of Highly Selective and Orally Bioavailable Macrocyclic IDO-1 Inhibitors 1 Tim Briggs Senior Director, Medicinal Chemistry Ensemble Therapeutics Corp. Ensemble Therapeutics Corp.
PBPK A Platform for Bridging People & Knowledge in Early Drug Development Club Phase I workshop PBPK: A new Paradigm in Drug Development Neil Miller
 PBPK A Platform for Bridging People & Knowledge in Early Drug Development Club Phase I workshop PBPK: A new Paradigm in Drug Development Neil Miller 27 th April 2017 Summary Take home messages PBPK has
PBPK A Platform for Bridging People & Knowledge in Early Drug Development Club Phase I workshop PBPK: A new Paradigm in Drug Development Neil Miller 27 th April 2017 Summary Take home messages PBPK has
Mechanistic IVIVC Using the Simcyp ADAM Model. Make SCIENCE out of IVIVC
 Mechanistic IVIVC Using the Simcyp ADAM Model Make SCIENCE out of IVIVC %Dissolved in vivo % Dissolved/Absorbed %Dissolved/Absorbed Plasma Conc (ng/ml) IVIVC and Its Components IVIVC: A predictive mathematical
Mechanistic IVIVC Using the Simcyp ADAM Model Make SCIENCE out of IVIVC %Dissolved in vivo % Dissolved/Absorbed %Dissolved/Absorbed Plasma Conc (ng/ml) IVIVC and Its Components IVIVC: A predictive mathematical
DRUG SOLUBILITY EXECUTIVE SUMMARY
 DRUG SOLUBILITY Utilising a high throughput screening platform to determine the optimum pharmaceutical excipients in the development of liquid formulations EXECUTIVE SUMMARY Drugs are small molecules or
DRUG SOLUBILITY Utilising a high throughput screening platform to determine the optimum pharmaceutical excipients in the development of liquid formulations EXECUTIVE SUMMARY Drugs are small molecules or
In vitro in vivo correlations (IVIVC) can be summarized
 dx.doi.org/10.14227/dt220215p44 Use of IVIVC to Optimize Generic Development J.-M. Cardot *, G. Garrait, and E. Beyssac e-mail: j-michel.cardot@udamail.fr Auvergne University, UFR Pharmacie, EA4678, Biopharmaceutical
dx.doi.org/10.14227/dt220215p44 Use of IVIVC to Optimize Generic Development J.-M. Cardot *, G. Garrait, and E. Beyssac e-mail: j-michel.cardot@udamail.fr Auvergne University, UFR Pharmacie, EA4678, Biopharmaceutical
Supplementary Figure 1. Determination of the purity of CP. a, SDS-PAGE of CP and CP- PTX conjugate, and b, HPLC trace of purified CP.
 Supplementary Figure 1. Determination of the purity of CP. a, SDS-PAGE of CP and CP- PTX conjugate, and b, HPLC trace of purified CP. Supplementary Figure 2. Synthesis of CP-PTX conjugate. Supplementary
Supplementary Figure 1. Determination of the purity of CP. a, SDS-PAGE of CP and CP- PTX conjugate, and b, HPLC trace of purified CP. Supplementary Figure 2. Synthesis of CP-PTX conjugate. Supplementary
Guideline on the qualification and reporting of physiologically based pharmacokinetic (PBPK) modelling and simulation
 1 2 3 21 July 2016 EMA/CHMP/458101/2016 Committee for Medicinal Products for Human Use (CHMP) 4 5 6 7 Guideline on the qualification and reporting of physiologically based pharmacokinetic (PBPK) modelling
1 2 3 21 July 2016 EMA/CHMP/458101/2016 Committee for Medicinal Products for Human Use (CHMP) 4 5 6 7 Guideline on the qualification and reporting of physiologically based pharmacokinetic (PBPK) modelling
High throughput ADME screening systems Enabling walkaway ADME solutions
 High throughput ADME screening systems Enabling walkaway ADME solutions The ADME product suite The current trend within the drug discovery arena is to incorporate ADME (absorption, distribution, metabolism
High throughput ADME screening systems Enabling walkaway ADME solutions The ADME product suite The current trend within the drug discovery arena is to incorporate ADME (absorption, distribution, metabolism
High affinity target binding: a cause of non-linear Pharmacokinetics of drugs (Target mediated drug disposition: TMDD)
 High affinity target binding: a cause of non-linear Pharmacokinetics of drugs (Target mediated drug disposition: TMDD) Devang Shah Senior Principal Investigator Metabolism and Pharmacokinetics, Pharmaceutical
High affinity target binding: a cause of non-linear Pharmacokinetics of drugs (Target mediated drug disposition: TMDD) Devang Shah Senior Principal Investigator Metabolism and Pharmacokinetics, Pharmaceutical
Delivering in vitro DM assay services
 Delivering in vitro DM assay services UGM_BIORAILS Copenhagen, 19-20 Oct 2017 Annalisa Pellacani An Integrated Approach to Drug Discovery: from Target to Clinic Target ID and Validation Screening and Hit
Delivering in vitro DM assay services UGM_BIORAILS Copenhagen, 19-20 Oct 2017 Annalisa Pellacani An Integrated Approach to Drug Discovery: from Target to Clinic Target ID and Validation Screening and Hit
Comparative Assessment of In Vitro In Vivo Extrapolation Methods used for Predicting Hepatic Metabolic Clearance of Drugs
 Comparative Assessment of In Vitro In Vivo Extrapolation Methods used for Predicting Hepatic Metabolic Clearance of Drugs PATRICK POULIN, 1 CORNELIS E. C. A. HOP, 2 QUYNH HO, 2 JASON S. HALLADAY, 2 SAMI
Comparative Assessment of In Vitro In Vivo Extrapolation Methods used for Predicting Hepatic Metabolic Clearance of Drugs PATRICK POULIN, 1 CORNELIS E. C. A. HOP, 2 QUYNH HO, 2 JASON S. HALLADAY, 2 SAMI
Compartmental Pharmacokinetic Analysis. Dr Julie Simpson
 Compartmental Pharmacokinetic Analysis Dr Julie Simpson Email: julieas@unimelb.edu.au BACKGROUND Describes how the drug concentration changes over time using physiological parameters. Gut compartment Absorption,
Compartmental Pharmacokinetic Analysis Dr Julie Simpson Email: julieas@unimelb.edu.au BACKGROUND Describes how the drug concentration changes over time using physiological parameters. Gut compartment Absorption,
Applied Biosystems/MDS Sciex LC/MS Users Meeting Daiichi Pure Chemicals Co., Ltd.
 Introduction ADMET screening Drug-drug interaction Introduction ADMET screening 40% ADMET screening in vitro Absorption : Caco-2 Permeability, Transporter assay Distribution : Protein binding, Transporter
Introduction ADMET screening Drug-drug interaction Introduction ADMET screening 40% ADMET screening in vitro Absorption : Caco-2 Permeability, Transporter assay Distribution : Protein binding, Transporter
BIOSTATISTICAL METHODS
 BIOSTATISTICAL METHODS FOR TRANSLATIONAL & CLINICAL RESEARCH Phase 0 Trials: EARLY-PHASE CLINICAL TRIALS Steps to New Drug Discovery Get idea for drug target Develop a bioassay Screen chemical compounds
BIOSTATISTICAL METHODS FOR TRANSLATIONAL & CLINICAL RESEARCH Phase 0 Trials: EARLY-PHASE CLINICAL TRIALS Steps to New Drug Discovery Get idea for drug target Develop a bioassay Screen chemical compounds
Pharmacokinetic & Pharmacodynamic Data Analysis
 Pharmacokinetic & Pharmacodynamic Data Analysis CONCEPTS AND APPLICATIONS 4TH EDITION REVISED AND EXPANDED Table of Contents 1. 1.1 1.2 1.3 1.4 1.5 2. 2.1 2.2 2.2.1 2.2.2 2.2.3 2.2.4 2.2.5 2.2.6 2.2.7
Pharmacokinetic & Pharmacodynamic Data Analysis CONCEPTS AND APPLICATIONS 4TH EDITION REVISED AND EXPANDED Table of Contents 1. 1.1 1.2 1.3 1.4 1.5 2. 2.1 2.2 2.2.1 2.2.2 2.2.3 2.2.4 2.2.5 2.2.6 2.2.7
BIOSTATISTICAL METHODS FOR TRANSLATIONAL & CLINICAL RESEARCH
 BIOSTATISTICAL METHODS FOR TRANSLATIONAL & CLINICAL RESEARCH Phase 0 Trials: EARLY-PHASE CLINICAL TRIALS CLINICAL PHASE Clinical Studies: Class of all scientific approaches to evaluate Disease Prevention,
BIOSTATISTICAL METHODS FOR TRANSLATIONAL & CLINICAL RESEARCH Phase 0 Trials: EARLY-PHASE CLINICAL TRIALS CLINICAL PHASE Clinical Studies: Class of all scientific approaches to evaluate Disease Prevention,
Official Letter from the DOH
 Issued Date 2009/04/02 Issued by DOH Ref. No 0980316268 RE The DOH issued an official letter to announce the implementation of the Guideline for BA/BE Studies on April 2, 2009 (Ref. No. 0980316265). Please
Issued Date 2009/04/02 Issued by DOH Ref. No 0980316268 RE The DOH issued an official letter to announce the implementation of the Guideline for BA/BE Studies on April 2, 2009 (Ref. No. 0980316265). Please
VELTIS : INNOVATIVE ALBUMIN BASED TECHNOLOGY FOR HALF- LIFE EXTENSION AND OPTIMIZATION OF BIOTHERAPEUTICS
 VELTIS : INNOVATIVE ALBUMIN BASED TECHNOLOGY FOR HALF- LIFE EXTENSION AND OPTIMIZATION OF BIOTHERAPEUTICS Dr Mikael Bjerg Caspersen Industrial Biotechnology Conference August 10 th 2015 INNOVATIVE TECHNOLOGY
VELTIS : INNOVATIVE ALBUMIN BASED TECHNOLOGY FOR HALF- LIFE EXTENSION AND OPTIMIZATION OF BIOTHERAPEUTICS Dr Mikael Bjerg Caspersen Industrial Biotechnology Conference August 10 th 2015 INNOVATIVE TECHNOLOGY
ATPlite Assay Performance in Human Primary Cells
 A P P L I C AT I O N N O T E Cell Viability Assays Author: Verena Brucklacher-Waldert Crescendo Biologics Cambridge, UK Assay Performance in Human Primary Cells Introduction In vitro assays using primary
A P P L I C AT I O N N O T E Cell Viability Assays Author: Verena Brucklacher-Waldert Crescendo Biologics Cambridge, UK Assay Performance in Human Primary Cells Introduction In vitro assays using primary
Samatasvir (IDX719), a Potent Pan-Genotypic NS5A Inhibitor, for the Treatment of Hepatitis C Virus (HCV) Infection
 Samatasvir (IDX719), a Potent Pan-Genotypic NS5A Inhibitor, for the Treatment of Hepatitis C Virus (HCV) Infection Douglas Mayers, MD December 11, 2013 1 Idenix: Advancing All-Oral, Pan-Genotypic Combination
Samatasvir (IDX719), a Potent Pan-Genotypic NS5A Inhibitor, for the Treatment of Hepatitis C Virus (HCV) Infection Douglas Mayers, MD December 11, 2013 1 Idenix: Advancing All-Oral, Pan-Genotypic Combination
The ENABLE project: An antibiotic discovery platform
 The ENABLE project: An antibiotic discovery platform Anders Karlén Putting open innovation into practice case studies from Europe 23.05.2017 PSWC Stockholm, Sweden The rising threat of antimicrobial resistance
The ENABLE project: An antibiotic discovery platform Anders Karlén Putting open innovation into practice case studies from Europe 23.05.2017 PSWC Stockholm, Sweden The rising threat of antimicrobial resistance
Discovery of Novel, Drug-like Ferroptosis Inhibitors with
 Supporting Information Discovery of Novel, Drug-like Ferroptosis Inhibitors with In Vivo Efficacy Lars Devisscher # a, Samya Van Coillie # b,c, Sam Hofmans a, Dries Van Rompaey a, Kenneth Goossens a, Eline
Supporting Information Discovery of Novel, Drug-like Ferroptosis Inhibitors with In Vivo Efficacy Lars Devisscher # a, Samya Van Coillie # b,c, Sam Hofmans a, Dries Van Rompaey a, Kenneth Goossens a, Eline
Conventional versus Physiologically-Based (PB)-IVIVC: Revisiting Some Successful and Failed Conventional IVIVC Cases with PB-IVIVC
 Conventional versus Physiologically-Based (PB)-IVIVC: Revisiting Some Successful and Failed Conventional IVIVC Cases with PB-IVIVC Nikunj Patel, Senior Research Scientist Simcyp (a Certara Company) AAPS
Conventional versus Physiologically-Based (PB)-IVIVC: Revisiting Some Successful and Failed Conventional IVIVC Cases with PB-IVIVC Nikunj Patel, Senior Research Scientist Simcyp (a Certara Company) AAPS
Pharmaceutical LC/MS Solutions from Agilent Technologies
 Pharmaceutical LC/MS Solutions from Agilent Technologies Application Compendium Overview LC/MS plays a key role in the drug discovery and drug development process. Since the introduction of electrospray
Pharmaceutical LC/MS Solutions from Agilent Technologies Application Compendium Overview LC/MS plays a key role in the drug discovery and drug development process. Since the introduction of electrospray
How do drugs work? Part 1. PK: Principles & Processes. PK vs. PD. Pharmacokinetics : Basic Principles Overview. What does the body do to a drug?
 Pharmacokinetics : Basic Principles verview 1. PK : Basic principles and processes 2. Important PK Parameters 3. Modulating PK Parameters 4. PK : Drugs vs. contrast agents Twan Lammers Dept. of Experimental
Pharmacokinetics : Basic Principles verview 1. PK : Basic principles and processes 2. Important PK Parameters 3. Modulating PK Parameters 4. PK : Drugs vs. contrast agents Twan Lammers Dept. of Experimental
Pharmacokinetics in clinical oncology
 Pharmacokinetics in clinical oncology Carlos Ochoa Biostatistics Journal Club Zurich, 27 March 2013 Pharmacokinetics in clinical oncology Anticancer drug effects: Shrink tumour size Slow tumour growth
Pharmacokinetics in clinical oncology Carlos Ochoa Biostatistics Journal Club Zurich, 27 March 2013 Pharmacokinetics in clinical oncology Anticancer drug effects: Shrink tumour size Slow tumour growth
06/03/2009. Overview. Preclinical Support for Exploratory Phase I Clinical Trials. Micro-dosing IND. Pharmacological Active Single Dose IND
 Preclinical Support for Exploratory Phase I Clinical Trials Clive Joseph, DSRD Sandwich Overview Identify the most appropriate development paradigm - traditional vs alternative IND approach Confidence
Preclinical Support for Exploratory Phase I Clinical Trials Clive Joseph, DSRD Sandwich Overview Identify the most appropriate development paradigm - traditional vs alternative IND approach Confidence
Cytotoxicity assays: Principles and Applications
 Cytotoxicity assays: Principles and Applications Dr. Markus Kamber Xenometrix AG Gewerbestr. 25 CH-4123 Allschwil Switzerland Overview 1. Major Applications 2. Why perform cytotoxicity assays? 3. What
Cytotoxicity assays: Principles and Applications Dr. Markus Kamber Xenometrix AG Gewerbestr. 25 CH-4123 Allschwil Switzerland Overview 1. Major Applications 2. Why perform cytotoxicity assays? 3. What
COMMITTEE FOR VETERINARY MEDICINAL PRODUCTS GUIDELINES FOR THE CONDUCT OF BIOEQUIVALENCE STUDIES FOR VETERINARY MEDICINAL PRODUCTS
 The European Agency for the Evaluation of Medicinal Products Veterinary Medicines and Information Technology EMEA/CVMP/016/00-corr-FINAL COMMITTEE FOR VETERINARY MEDICINAL PRODUCTS GUIDELINES FOR THE CONDUCT
The European Agency for the Evaluation of Medicinal Products Veterinary Medicines and Information Technology EMEA/CVMP/016/00-corr-FINAL COMMITTEE FOR VETERINARY MEDICINAL PRODUCTS GUIDELINES FOR THE CONDUCT
Optimal experimental design for assessment of enzyme kinetics in a drug discovery screening environment
 DMD Fast This Forward. article has not Published been copyedited on and February formatted. The 2, 2011 final version as doi:10.1124/dmd.110.037309 may differ from this version. Optimal experimental design
DMD Fast This Forward. article has not Published been copyedited on and February formatted. The 2, 2011 final version as doi:10.1124/dmd.110.037309 may differ from this version. Optimal experimental design
On applications of conservation laws in pharmacokinetics
 On applications of conservation laws in pharmacokinetics S.Piekarski (1), M.Rewekant (2) Institute of Fundamental Technological Research Polish Academy of Sciences (1), Medical University of Warsaw, Poland
On applications of conservation laws in pharmacokinetics S.Piekarski (1), M.Rewekant (2) Institute of Fundamental Technological Research Polish Academy of Sciences (1), Medical University of Warsaw, Poland
Optimizing the Development of Biosimilars Using PK/PD: Recent Scientific and Regulatory Advances
 Optimizing the Development of Biosimilars Using PK/PD: Recent Scientific and Regulatory Advances Jian Wang, MD, PhD Chief, Clinical Evaluation Division Biologics and Genetic Therapies Directorate Health
Optimizing the Development of Biosimilars Using PK/PD: Recent Scientific and Regulatory Advances Jian Wang, MD, PhD Chief, Clinical Evaluation Division Biologics and Genetic Therapies Directorate Health
Application of PBPK Models in Assessment of Bioequivalence
 Application of PBPK Models in Assessment of Bioequivalence Robert Lionberger, Ph.D. Acting Director Office of Research and Standards Office of Generic Drugs Center for Drug Evaluation and Research, FDA
Application of PBPK Models in Assessment of Bioequivalence Robert Lionberger, Ph.D. Acting Director Office of Research and Standards Office of Generic Drugs Center for Drug Evaluation and Research, FDA
The impact of apical and basolateral albumin on intestinal zinc resorption in the Caco-2/HT-29-MTX co-culture model
 Electronic Supplementary Material (ESI) for Metallomics. This journal is The Royal Society of Chemistry 2018 Supplemental Material The impact of apical and basolateral albumin on intestinal zinc resorption
Electronic Supplementary Material (ESI) for Metallomics. This journal is The Royal Society of Chemistry 2018 Supplemental Material The impact of apical and basolateral albumin on intestinal zinc resorption
Utility of preclinical PKPD modeling in QT safety testing
 Utility of preclinical PKPD modeling in QT safety testing Sandra Visser & Piet van der Graaf EMA/EFPIA M&S Workshop on the role and scope of modelling and simulation in drug development BOS1, London 1
Utility of preclinical PKPD modeling in QT safety testing Sandra Visser & Piet van der Graaf EMA/EFPIA M&S Workshop on the role and scope of modelling and simulation in drug development BOS1, London 1
Eurofins ADME BIOANALYSES Your partner in drug development
 Eurofins ADME BIOANALYSES Your partner in drug development Since 1987, you have placed your confidence in us. Our responsibility, to which my staff and I commit, is to consistently provide you with relevant
Eurofins ADME BIOANALYSES Your partner in drug development Since 1987, you have placed your confidence in us. Our responsibility, to which my staff and I commit, is to consistently provide you with relevant
Informatics and High Resolution QTof MS. How can we ask better questions and get better answers in DMPK? Mark D. Wrona
 Informatics and High Resolution QTof MS How can we ask better questions and get better answers in DMPK? Mark D. Wrona Principal Scientist Pharmaceutical & Life Sciences, Waters Corporation Pacific Northwest
Informatics and High Resolution QTof MS How can we ask better questions and get better answers in DMPK? Mark D. Wrona Principal Scientist Pharmaceutical & Life Sciences, Waters Corporation Pacific Northwest
Course Curriculum for Master Degree in Pharmaceutical Technology
 Course Curriculum for Master Degree in Pharmaceutical Technology The Master Degree in Pharmaceutical Technology is awarded by the Faculty of Graduate studies at Jordan University of Science and Technology
Course Curriculum for Master Degree in Pharmaceutical Technology The Master Degree in Pharmaceutical Technology is awarded by the Faculty of Graduate studies at Jordan University of Science and Technology
Mechanistic bioaccumulation model(s) for ionogenic organic substances in fish
 LRI-ECO21 Mechanistic bioaccumulation model(s) for ionogenic organic substances in fish Cefic-LRI Annual workshop Brussels, Nov. 2015 Partner 1 Partner 2 Partner 3 Partner 4 Partner 5 Rationale ECO21 1.
LRI-ECO21 Mechanistic bioaccumulation model(s) for ionogenic organic substances in fish Cefic-LRI Annual workshop Brussels, Nov. 2015 Partner 1 Partner 2 Partner 3 Partner 4 Partner 5 Rationale ECO21 1.
In Vitro-In Vivo Correlation:
 In Vitro-In Vivo Correlation: Linking Drug Release to Clinical Performance Yihong Qiu, Ph.D. Abbott Laboratories 51st Land O' Lakes Conference: Bridging Material and Product Quality in Developing Tablet
In Vitro-In Vivo Correlation: Linking Drug Release to Clinical Performance Yihong Qiu, Ph.D. Abbott Laboratories 51st Land O' Lakes Conference: Bridging Material and Product Quality in Developing Tablet
Drug Metabolism and Disposition. Supplement Materials
 Drug Metabolism and Disposition Supplement Materials Inhibition of P-glycoprotein leads to improved oral bioavailability of Compound K, an anti-cancer metabolite of red ginseng extract produced by gut
Drug Metabolism and Disposition Supplement Materials Inhibition of P-glycoprotein leads to improved oral bioavailability of Compound K, an anti-cancer metabolite of red ginseng extract produced by gut
! Background. ! What is really new?! The new Section 7: Explorative Clinical Trials (ECTs) ! Consequences in General
 ! Background! What is really new?! The new Section 7: Explorative Clinical Trials (ECTs)! 5 Approaches (Table 3) for:! Microdose trials (7.1)! Single-Dose (SD) Trials at Sub-therapeutic Doses or Into the
! Background! What is really new?! The new Section 7: Explorative Clinical Trials (ECTs)! 5 Approaches (Table 3) for:! Microdose trials (7.1)! Single-Dose (SD) Trials at Sub-therapeutic Doses or Into the
Miniaturized Whole Cell-based GPCR camp Assay Using a Novel Detection System
 Miniaturized Whole Cell-based GPCR camp Assay Using a Novel Detection System Geetha Shankar, Ph.D. Associate Director New Lead Discovery Exelixis, Inc South San Francisco, CA Overview GPCR HTS assays ACT:One
Miniaturized Whole Cell-based GPCR camp Assay Using a Novel Detection System Geetha Shankar, Ph.D. Associate Director New Lead Discovery Exelixis, Inc South San Francisco, CA Overview GPCR HTS assays ACT:One
Passive Dosing of hydrophobic organic chemicals (HOCs) to in vitro assays controlling, defining and linking exposure
 Passive Dosing of hydrophobic organic chemicals (HOCs) to in vitro assays controlling, defining and linking exposure Philipp Mayer Contributions from Dorothea Gilbert, Stine Schmidt, Kilian Smith and more
Passive Dosing of hydrophobic organic chemicals (HOCs) to in vitro assays controlling, defining and linking exposure Philipp Mayer Contributions from Dorothea Gilbert, Stine Schmidt, Kilian Smith and more
Modeling for decision making in clinical programs - Case Studies Rolf Burghaus Bayer Schering Pharma Clinical Pharmacology / Modeling & Simulation
 Modeling for decision making in clinical programs - Case Studies Rolf Burghaus Bayer Schering Pharma Clinical Pharmacology / Modeling & Simulation Modeling & Simulation - Positioning Modeling & Simulation
Modeling for decision making in clinical programs - Case Studies Rolf Burghaus Bayer Schering Pharma Clinical Pharmacology / Modeling & Simulation Modeling & Simulation - Positioning Modeling & Simulation
Biowaivers and Harmonization Guidelines for Class I and 3 Drugs: Biowaiver Case Studies
 Biowaivers and Harmonization Guidelines for Class I and 3 Drugs: Biowaiver Case Studies Barbara M. Davit, Merck & Co., Inc. 3 rd FDA/PQRI Conference on Advancing Product Quality March 22, 2017 Disclaimer
Biowaivers and Harmonization Guidelines for Class I and 3 Drugs: Biowaiver Case Studies Barbara M. Davit, Merck & Co., Inc. 3 rd FDA/PQRI Conference on Advancing Product Quality March 22, 2017 Disclaimer
The Prediction of Human Pharmacokinetic Parameters from Preclinical and In Vitro Metabolism Data
 0022-3565/97/2831-0046$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 283, No. 1 Copyright 1997 by The American Society for Pharmacology and Experimental Therapeutics Printed in
0022-3565/97/2831-0046$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 283, No. 1 Copyright 1997 by The American Society for Pharmacology and Experimental Therapeutics Printed in
Identification of the In Vivo Pharmacokinetics and Pharmacodynamic Drivers of Iclaprim
 AAC Accepted Manuscript Posted Online 29 January 2018 Antimicrob. Agents Chemother. doi:10.1128/aac.02550-17 Copyright 2018 American Society for Microbiology. All Rights Reserved. 1 Identification of the
AAC Accepted Manuscript Posted Online 29 January 2018 Antimicrob. Agents Chemother. doi:10.1128/aac.02550-17 Copyright 2018 American Society for Microbiology. All Rights Reserved. 1 Identification of the
Medicines Control Authority Of Zimbabwe
 Medicines Control Authority Of Zimbabwe BIOEQUIVALENCE APPLICATION FORM Form: EVF03 This application form is designed to facilitate information exchange between the Applicant and the MCAZ for bioequivalence
Medicines Control Authority Of Zimbabwe BIOEQUIVALENCE APPLICATION FORM Form: EVF03 This application form is designed to facilitate information exchange between the Applicant and the MCAZ for bioequivalence
In vitro to in vivo extrapolation of hepatic metabolism in fish: an inter laboratory comparison of in vitro methods
 ILSI Health and Environmental Sciences Institute In vitro to in vivo extrapolation of hepatic metabolism in fish: an inter laboratory comparison of in vitro methods Kellie Fay, USEPA HESI Annual Meeting
ILSI Health and Environmental Sciences Institute In vitro to in vivo extrapolation of hepatic metabolism in fish: an inter laboratory comparison of in vitro methods Kellie Fay, USEPA HESI Annual Meeting
Dr. M.Mothilal Assistant professor
 Dr. M.Mothilal Assistant professor Complex picture of drug interactions in the body. This slide gives an idea of the complexity of drug disposition. Shown in this slide are many of the steps to getting
Dr. M.Mothilal Assistant professor Complex picture of drug interactions in the body. This slide gives an idea of the complexity of drug disposition. Shown in this slide are many of the steps to getting
Public Assessment Report Scientific discussion. Naproxen ABECE (naproxen) Asp no:
 Public Assessment Report Scientific discussion Naproxen ABECE (naproxen) Asp no: 2016-1607-08 This module reflects the scientific discussion for the approval of Naproxen ABECE. The procedure was finalised
Public Assessment Report Scientific discussion Naproxen ABECE (naproxen) Asp no: 2016-1607-08 This module reflects the scientific discussion for the approval of Naproxen ABECE. The procedure was finalised
Current Guidelines for Drug-drug Interaction Evaluation
 Current Guidelines for Drug-drug Interaction Evaluation Odette A Fahmi, PhD DDI-Edge Consulting, LLC June 2018 21 ST International Conference on Drug-Drug Interactions OUTLINE Recommended DDI approaches
Current Guidelines for Drug-drug Interaction Evaluation Odette A Fahmi, PhD DDI-Edge Consulting, LLC June 2018 21 ST International Conference on Drug-Drug Interactions OUTLINE Recommended DDI approaches
Single cell resolution in vivo imaging of DNA damage following PARP inhibition. Supplementary Data
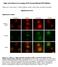 Single cell resolution in vivo imaging of DNA damage following PARP inhibition Katherine S. Yang, Rainer H. Kohler, Matthieu Landon, Randy Giedt, and Ralph Weissleder Supplementary Data Supplementary Figures
Single cell resolution in vivo imaging of DNA damage following PARP inhibition Katherine S. Yang, Rainer H. Kohler, Matthieu Landon, Randy Giedt, and Ralph Weissleder Supplementary Data Supplementary Figures
Model based approaches to target special populations with rational formulation and clinical design strategies
 Model based approaches to target special populations with rational formulation and clinical design strategies David Good 3 rd FDA/PQRI Conference on Advancing Product Quality Modeling for Oral and Non-Oral
Model based approaches to target special populations with rational formulation and clinical design strategies David Good 3 rd FDA/PQRI Conference on Advancing Product Quality Modeling for Oral and Non-Oral
PIONEERING WORLD-CLASS DRUG DISCOVERY IN AFRICA
 PIONEERING WORLD-CLASS DRUG DISCOVERY IN AFRICA MISSION & VISION Prof. Kelly Chibale H3D Director Vision To be the leading organisation for integrated drug discovery and development on the African continent.
PIONEERING WORLD-CLASS DRUG DISCOVERY IN AFRICA MISSION & VISION Prof. Kelly Chibale H3D Director Vision To be the leading organisation for integrated drug discovery and development on the African continent.
