Tissue Engineering For Regenerating Life. Cryopreserved Products. Tissue Corporation
|
|
|
- Tamsin Kelley
- 6 years ago
- Views:
Transcription
1 Tissue Engineering For Regenerating Life Cryopreserved Products Tissue Corporation
2 Tissue Corporation TRC (Tissue regeneration corporation) is a multi-facility institute specializing in the preparation of a wide range of grafts based on the science of tissue engineering. Tissue engineering is an emerging science that aims to regenerate existing biological tissue and create new tissue using biological cells and biomaterial. Our competitive edge is derived from a strong focus on improving patient outcomes. The TRC team consists of highly dedicated and motivated professionals who are committed to finding solutions in order to achieve the highest standards in our work. 2 Cryopreserved Products Tissue Regeneration Corporation 3
3 STATE-OF-THE-ART PRODUCTION PRODUCTS BENEFITS QUALITY ASSURANCE Tissue Regeneration Corporation adheres to strict policies and procedures that were devised in line with the guidelines and standards of the FDA and UK codes of practice for productions of human derived therapeutic materials. All tissues are procured in a class 1000 environment and processed in sterile class clean rooms. The donor coordinator acquires the necessary consent for donation and interviews the family of each donor to obtain the donor s medical history. TRC will only supply tissue from donors where lawful consent has been established. Where consent has been obtained by TRC, the tissue preparation is undertaken by our highly trained team who are constantly assessed using our specifically developed competency assessment program. Every donated tissue is tasted using several microbiologic and serologic testes such as HBs Ag, HBc Ab, HCV Ab, HIV Ab(1&2), HTLV Ab (I&II), RPR with ELISA method and complementary tests such as PCR method and FTA. Most of the donors are young, additionally our country has one of the lowest HIV infection rates in the world therefore we can provide some of the best quality tissues with minimal risk of AIDS transmission. All our bony grafts have both osteoinductive and osteoconductive properties confirmed by both in vitro and in vivo. Furthermore, the biomechanical properties of both the machined and large bones are routinely tested according to ASTM standards. Our work is consistent with the fundamentals of both national and international quality standards and ethical principles, specifically we obey all AATB and FDA rules in cellular and tissues based products. The services and facilities (including pharmaceutical grade cleanrooms) are all consistent with the current good manufacturing practice (cgmp). Freeze dried bone is lyophilized to measure <0.5 aw (water activity) eliminating the potential for microbial growth and minimizing autodegredative reactions. Irradiation is carried out to an established protocol ensuring a minimum dose of 25KGY is received by the tissue. Processed bone grafts are non cytotoxic as per ISO Final product release is undertaken as an independent function by quality assurance specialist personnel. All microbiology testing is performed internally by accredited laboratories specializing in donation screening. Final donor assessment and selection is undertaken by our own clinical specialist in tissue donation under supervision of coroner specialists. Donations are tracked by barcode including automated test result transfer to the database (the same database used for blood donation, processing and supply). This database has automated controls to prevent release of non-conforming tissue. Processes are validated in-house by the tissue development laboratory. All critical physical/chemical parameters are continuously monitored using a sophisticated IT package with appropriate warning levels and alarm states. This package continuously monitors (where appropriate) temperatures (of rooms, deep freezers, liquid nitrogen tanks etc), clean room pressures, air particles, oxygen levels, etc. Tissues engineered products from allogenic sources are used in many surgical procedures because they are naturally biocompatible and can be remodeled to the patient s own bone. They simplify potential revision procedures, and they eliminate second site morbidity and pain that may result from autograft removal. They are easy to use, take little time to prepare and are available pre-shaped to exact specifications. the end result is a facility, which ranks amongst the best in the world. TRC is staffed with highly trained dedicated doctors, scientists, technicians, nurses and all levels of support staff. This combination of a motivated professional workforce within a state-of-the-art facility ensures our commitment to safety, quality and efficacy of all our tissue grafts. SHIPPING AND INSTRUCTION TO USE ORDERING All shipping arrangements are made and handed on an individual basis. The product is usually delivered by either TRC transport or via the express special mail as special delivery in a padded envelope usually direct to the point of use e.g. theatre. More urgent delivery e.g same day or by specified time can be arranged at additional cost. Where an operation is graft critical, the patient must not be taken to theatre before the graft has arrived and its condition checked. Deep freeze products will be sent packed in dry ice (-70 c). Vapor phase LN2 shipper (-130 c) is also available in the event of extended transport durations. These products should be stored in -60 c or lower temperature freezers as soon as they are received by the client. They should fully thaw prior to transplantation. Thawing protocols are attached to all frozen products. On the other hand, freeze-dried products should be transported and stored at room temperature until they are used. Such grafts should be stored away from direct sunlight at ambient temperature. They must be re-hydrated with a physiologic solution or the recipient s own blood prior to implantation for at least 30 minutes. Extensive inventories are available for allograft tissues by Tissue Regeneration Corporation. Please contact us up to 2 weeks before the required time and specify both the type and specification of your desired product. our staff will contact you as soon as possible and will send you the specifications along with digital photographs of the mentioned tissues and delivery options. We have a strict Surgeon OK program before your request is dispatched. In cases of urgency and special rush orders, we can arrange the shipment of tissues to the client in less than 48 hours. 4 Cryopreserved Products Tissue Regeneration Corporation 5
4 PRODUCTS LIST Cryopreserved Products Note: Tissues listed below don t reflect the complete stock of our products, merely a representation of the types and sample size available in Tissue Regeneration Corporation. Please contact your representative for information pertaining to items not listed below. CenoValve Indication: Allograft bio-implants for cardiac and vascular procedures. Suitable for a wide variety of complex congenital heart defects and adults with extensive valvular disease as well as for managing vascular reconstruction including aortic stenosis or atresia, hypoplastic left heart, repair of RVOTs (Right Ventricular Outflow Tract), pulmonary atresia, truncus arteriosus, Ross procedure, single ventricle/double outlet (Fontan), hypoplastic left heart (modified Fontan), tetralogy of Fallot, RVOT/ root enlargement procedures, ASD/VSD repair, transposition of great vessels, Peripheral Vascular Disease (PVD), Infected Synthentic Graft Replacement etc. CenoValve Aortic heart valve D: 9mm Aortic heart valve D: 11mm Aortic heart valve D: 13mm Aortic heart valve D: 15mm Aortic heart valve D: 17mm Aortic heart valve D: 19mm Aortic heart valve D: 21mm Aortic heart valve D: 23mm Aortic heart valve D: 25mm Aortic heart valve D: 27mm Aortic heart valve D: 29mm Aortic heart valve D: 31mm CenoValve (Continue) Pulmonary heart valve D: 9mm Pulmonary heart valve D: 11mm Pulmonary heart valve D: 13mm Pulmonary heart valve D: 15mm Pulmonary heart valve D: 17mm Pulmonary heart valve D: 19mm Pulmonary heart valve D: 21mm Pulmonary heart valve D: 23mm Pulmonary heart valve D: 25mm Pulmonary heart valve D: 27mm Pulmonary heart valve D: 29mm Pulmonary heart valve D: 31mm Aortic conduit D: 9-31mm Pulmonary conduit D: 9-31mm Hemi-pulmonary artery L=20-80mm Mono cusp patch W/L varies Pulmonary patch 20* Pulmonary patch 30* Pulmonary patch 20* Pulmonary patch 20* Femoral artery L<50mm D: 5-15mm Femoral artery L=50-100mm D: 5-15mm Femoral artery L>100mm D: 5-15mm Saphenous vein L<40cm D: 2-4mm Saphenous vein L=40-80cm D: 2-4mm Saphenous vein L>80cm D: 2-4mm Pericardial patch 20* Pericardial patch 30*30 6 Cryopreserved Products Tissue Regeneration Corporation 7
5 AmniPatch CenoValve (Continue) Pericardial patch 40* Pericardial patch 50* Pericardial patch 60* Pericardial patch 70* Pericardial patch 80* Pericardial patch 90* Pericardial patch 100*100 Indication: In ophthalmology, where it is used to proceed reepithelialization for a variety of procedures in conjunctival and corneal problems; in dermatology, where it is used to treat and protect skin burns or help heal the torpid ulcers; in general surgery, where it prevents post- surgery adhesions; in neurosurgery for brain operations and for the reconstruction of urinary and genital apparatus. AmniPatch Antibiotic Impregnated Amniotic Membrane 20* Antibiotic Impregnated Amniotic Membrane 30* Antibiotic Impregnated Amniotic Membrane 40* Antibiotic Impregnated Amniotic Membrane 50* Antibiotic Impregnated Amniotic Membrane 60* Antibiotic Impregnated Amniotic Membrane 70* Antibiotic Impregnated Amniotic Membrane 80* Antibiotic Impregnated Amniotic Membrane 90* Antibiotic Impregnated Amniotic Membrane 100*100 CenoTendon Indication: Anterior Cruciate Ligament (ACL) reconstruction, Posterior Cruciate Ligament (PCL) reconstruction, Achilles tendon repair, Reconstruction or augmentation of the rotator cuff and any other tendon soft tissue augmentation or repair. CenoTendon Code Item Description Size Patellar Bone + Patellar Tendon + Tibial Bone(BTB) Pre-shaped (W)=17-13 mm (L)= mm Achilles Tendon with Calcaneus Whole (W)=20-10 mm Achilles tendon with Calcaneus Hemi (W)=12-6 mm Achilles Tendon without Calcaneus Whole (W)=20-10 mm Achilles tendon without Calcaneus Hemi (W)=12-6 mm Proneus Longus Tendon Whole (Folded Diameter)=12-6 mm Posterior Tibialis Tendon Whole (Folded Diameter)=12-6 mm Anterior Tibialis Tendon Whole (Folded Diameter)=12-6 mm Semitendinosus Tendon Whole (W)=10-3 mm Gracilis Tendon Whole (W)=10-3 mm Medial Meniscus with Tibial Plateau W/L varies Bone Block Lateral Meniscus with Tibial Plateau Bone Block W/L varies 8 Cryopreserved Products Tissue Regeneration Corporation 9
6 Instructions for thawing contents and dilution of cryoprotectant 1.Two sterile 500 ml basins and one sterile 5000 ml basin are needed. 2.Pour approximately 3000 ml of 40ºc sterile saline into large basin. 3.Pour 300 ml 4-10ºc RPMI or M199 sterile tissue culture medium into one of the 500 ml basins. Note: you can use sterile isotonic fluid instead of culture medium (without adding recipient blood) but if there is more than 5 hours between thawing and implantation time, we recommend using culture medium or adding heparinized recipient blood to your isotonic fluid in thawing process. 4.Remove the cryopreserved CenoValve/CenoTendon pouch form the cryotransport container and dry the outer surface of the pouch thoroughly. 5.Scrub the opening site using proper disinfectants. Circulating nurse then opens the pouch with sterile scissors. Note: Be careful not to contaminate the inner part of the sterile pouch. 6.Contents are presented to the scrubbing nurse who retrieves the inner sterile pouch with a kocher clamp. 7.Place inner pouch in the large basin and gently agitate the pouch for 3-4 minutes. 8.Add 1000 ml of warmed saline to a large basin and gently agitate the pouch for additional 3-4 minutes. 9.Glycerol is not toxic in 37ºc therefore allow medium to completely thaw. Open the pouches with sterile scissors and pour their content into the empty sterile small basin. 10.Add 50 ml of tissue culture medium from its basin to the CenoValve/CenoTendon containing basin. Gently agitate for 2 minutes. 11. Add another 150 ml of tissue culture medium from it s basin to the CenoValve /CenoTendon containing basin. Gently agitate for 2 minutes. Note: use sterile 50 cc syringes for adding medium. 12. The CenoValve/CenoTendon should be removed by the surgeon in sterile condition. Inspect for cracks or any damage and then place CenoValve/CenoTendon in another sterile 100 ml medium containing basin (100 ml remaining). Gently agitate for 2 minutes. Note: Don t use CenoValve/CenoTendon if there is any crack or damage throughout thawing process. 13. Maintain the CenoValve/CenoTendon completely immersed within this basin until needed for implantation. We recommend obtaining approximately 50 ml of recipient s heparinized blood and Add to CenoValve/CenoTendon containing basin. 14. If the prepared CenoValve/CenoTendon is not soon implanted, place the basin of CenoValve/CenoTendon on ice until needed. 15. A sample of the CenoValve /CenoTendon may be obtained for culturing, at surgeons discretion, to ensure sterility. Note: please report any adverse reactions due to CenoValve/CenoTendon to TRC (Tissue Regeneration Corporation). 10 Cryopreserved Products Tissue Regeneration Corporation 11
7 Tissue Engineering For Regenerating Life Tissue Corporation Headquarter & Production complex: No.33, 3rd industrial park, kish free zone, Kish island, Iran Tel: +98(764) Fax: +98(764) Tehran Branch: No.8, eastern 19th Ave, Sarafha St, Saadat Abad, Tehran, Iran Tel: +98(21) (21) (21) (21) (21) Mobile: +98(912) Fax: +98(21) Website:
2015-Tissue Storage. Welcome to the 2015 Tissue Storage survey. Please refer to the following instructions as you complete this section of the survey.
 2015-Tissue Storage Form Approved OMB No. 0990-0457 Exp. Date 06/30/2020 Welcome to the 2015 Tissue Storage survey. Please refer to the following instructions as you complete this section of the survey.
2015-Tissue Storage Form Approved OMB No. 0990-0457 Exp. Date 06/30/2020 Welcome to the 2015 Tissue Storage survey. Please refer to the following instructions as you complete this section of the survey.
TRELLIS COLLAGEN RIBBON
 TRELLIS COLLAGEN RIBBON 147321-1 English (en) The following languages are included in this packet: M Wright Medical Technology, Inc. 5677 Airline Rd. Arlington, TN 38002 USA www.wmt.com August 2012 Printed
TRELLIS COLLAGEN RIBBON 147321-1 English (en) The following languages are included in this packet: M Wright Medical Technology, Inc. 5677 Airline Rd. Arlington, TN 38002 USA www.wmt.com August 2012 Printed
ALLODERM SELECT ALLODERM SELECT RESTORE
 ALLODERM SELECT ALLODERM SELECT RESTORE Regenerative Tissue Matrix Instructions for Use Processed from donated human tissue by: LifeCell Corporation One Millennium Way Branchburg, NJ 08876-3876 DESCRIPTION
ALLODERM SELECT ALLODERM SELECT RESTORE Regenerative Tissue Matrix Instructions for Use Processed from donated human tissue by: LifeCell Corporation One Millennium Way Branchburg, NJ 08876-3876 DESCRIPTION
Repliform. Tissue Regeneration Matrix. Instructions for Use. Distributed by:
 Repliform Tissue Regeneration Matrix Instructions for Use Distributed by: Boston Scientific Corporation 300 Boston Scientific Way Marlborough, MA 01752 Processed from Donated Human Tissue by: LifeCell
Repliform Tissue Regeneration Matrix Instructions for Use Distributed by: Boston Scientific Corporation 300 Boston Scientific Way Marlborough, MA 01752 Processed from Donated Human Tissue by: LifeCell
Tissue Products Catalogue
 Tissue Products Catalogue Tissue Processing: ecoo Technology Our Tissues business segment is focused on processing tissue components such as bones, soft tissues and skin of both human and animal origin.
Tissue Products Catalogue Tissue Processing: ecoo Technology Our Tissues business segment is focused on processing tissue components such as bones, soft tissues and skin of both human and animal origin.
Importing/Exporting Conventional HCT/Ps
 Importing/Exporting Conventional HCT/Ps ISCT/FDA Liaison Meeting January 4, 2007 Scott A. Brubaker, CTBS American Association of Tissue Banks Conventional Tissue Banks! FDA term from the preamble to the
Importing/Exporting Conventional HCT/Ps ISCT/FDA Liaison Meeting January 4, 2007 Scott A. Brubaker, CTBS American Association of Tissue Banks Conventional Tissue Banks! FDA term from the preamble to the
Platelet Concentrate in Total Knees BIOLOGICS. This brochure is for International use only. It is not for distribution in the United States.
 Platelet Concentrate in Total Knees BIOLOGICS This brochure is for International use only. It is not for distribution in the United States. Platelet Concentrate in Total Knees Total Knee Arthroplasty Total
Platelet Concentrate in Total Knees BIOLOGICS This brochure is for International use only. It is not for distribution in the United States. Platelet Concentrate in Total Knees Total Knee Arthroplasty Total
Cell Therapy Services Your Product. Our Passion.
 Pharma&Biotech Cell Therapy Services Your Product. Our Passion. Complete Portfolio of Services for cgmp Manufacturing of Cell-based Products Pharma&Biotech Cell Therapy Services for cgmp Manufacturing
Pharma&Biotech Cell Therapy Services Your Product. Our Passion. Complete Portfolio of Services for cgmp Manufacturing of Cell-based Products Pharma&Biotech Cell Therapy Services for cgmp Manufacturing
COMMISSION OF THE EUROPEAN COMMUNITIES. Proposal for a DIRECTIVE OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL
 COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 19.6.2002 COM(2002) 319 final 2002/0128(COD) Proposal for a DIRECTIVE OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL on setting standards of quality and
COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 19.6.2002 COM(2002) 319 final 2002/0128(COD) Proposal for a DIRECTIVE OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL on setting standards of quality and
Packaging, Labelling and Transport of Organs in Deceased and Living Donation and Transplantation. Summary of Significant Changes
 This National Operating Procedure replaces: NOP003 Summary of Significant Changes Effective: 6 th December 2016 Document updated to reflect changes in organ packaging and labelling including minor changes
This National Operating Procedure replaces: NOP003 Summary of Significant Changes Effective: 6 th December 2016 Document updated to reflect changes in organ packaging and labelling including minor changes
Guide for Cleaning, Sterilization and Storage of Introducers for Caldera Medical Implants
 Guide for Cleaning, Sterilization and Storage of Introducers for Caldera Medical Implants M Manufactured by: Caldera Medical, Inc. 5171 Clareton Drive Agoura Hills, CA 91301 U.S. Toll Free: 866-4-CALDERA
Guide for Cleaning, Sterilization and Storage of Introducers for Caldera Medical Implants M Manufactured by: Caldera Medical, Inc. 5171 Clareton Drive Agoura Hills, CA 91301 U.S. Toll Free: 866-4-CALDERA
Human Adult Mammary Fibroblast Care Manual
 Human Adult Mammary Fibroblast Care Manual INSTRUCTIONAL MANUAL ZBM0062.05 SHIPPING CONDITIONS Human Adult Mammary Fibroblast Cells Orders are delivered via Federal Express courier. All US and Canada orders
Human Adult Mammary Fibroblast Care Manual INSTRUCTIONAL MANUAL ZBM0062.05 SHIPPING CONDITIONS Human Adult Mammary Fibroblast Cells Orders are delivered via Federal Express courier. All US and Canada orders
Agenzia Italiana del Farmaco
 Agenzia Italiana del Farmaco European Regulation on Advanced Therapies Cristina Pintus Head of European Relations Unit and Coordinator of the Advanced Therapy Project Italian Medicines Agency Proposal
Agenzia Italiana del Farmaco European Regulation on Advanced Therapies Cristina Pintus Head of European Relations Unit and Coordinator of the Advanced Therapy Project Italian Medicines Agency Proposal
Section 8. To introduce the basic theory and principles of collecting blood for Intraoperative Cell Salvage (ICS)
 Section 8 Practicalities Blood Collection Aim To introduce the basic theory and principles of collecting blood for Intraoperative Cell Salvage (ICS) Learning Outcomes To identify the equipment used for
Section 8 Practicalities Blood Collection Aim To introduce the basic theory and principles of collecting blood for Intraoperative Cell Salvage (ICS) Learning Outcomes To identify the equipment used for
HCT/P Regulation vs 361 Products
 HCT/P Regulation - 351 vs 361 Products Presented by: Paul Gadiock February 15, 2017 Arent Fox LLP Washington, DC New York, NY Los Angeles, CA San Francisco, CA 1 Presentation Overview Introduction Public
HCT/P Regulation - 351 vs 361 Products Presented by: Paul Gadiock February 15, 2017 Arent Fox LLP Washington, DC New York, NY Los Angeles, CA San Francisco, CA 1 Presentation Overview Introduction Public
SOP 2 Solid Tumor Processing for Cell Culture and Xenografts
 Texas Cancer Cell Repository Cell Culture and Xenograft Repository http://cancer.ttuhsc.edu www.txccr.org www.cogcell.org SOP 2 Solid Tumor Processing for Cell Culture and Xenografts Receiving the Sample:
Texas Cancer Cell Repository Cell Culture and Xenograft Repository http://cancer.ttuhsc.edu www.txccr.org www.cogcell.org SOP 2 Solid Tumor Processing for Cell Culture and Xenografts Receiving the Sample:
HCT/P Deviations & Reporting. 13 th ANNUAL FDA & The Changing Paradigm for HCT/P Regulation February 13-15, 2017
 HCT/P Deviations & Reporting CASE STUDIES FROM TISSUE AND EYE BANKS AND CELLULAR THERAPY. 13 th ANNUAL FDA & The Changing Paradigm for HCT/P Regulation February 13-15, 2017 Tissue Bank Perspective Ashley
HCT/P Deviations & Reporting CASE STUDIES FROM TISSUE AND EYE BANKS AND CELLULAR THERAPY. 13 th ANNUAL FDA & The Changing Paradigm for HCT/P Regulation February 13-15, 2017 Tissue Bank Perspective Ashley
PRO-STIM Injectable Inductive Graft Containing DONATED HUMAN TISSUE
 PRO-STIM Injectable Inductive Graft Containing DONATED HUMAN TISSUE 137835-5 The following languages are included in this packet: English (en) For additional information please contact the manufacturer
PRO-STIM Injectable Inductive Graft Containing DONATED HUMAN TISSUE 137835-5 The following languages are included in this packet: English (en) For additional information please contact the manufacturer
Annual Results Presentation 23 rd May 2016 Ian Jefferson - CFO
 Annual Results Presentation 23 rd May 2016 Ian Jefferson - CFO 1 Financial Overview Selected Highlights Income statement 000 31 Jan 2016 31 Jan 2015 Revenue 816 100 Cost of sales (154) (32) Gross Margin
Annual Results Presentation 23 rd May 2016 Ian Jefferson - CFO 1 Financial Overview Selected Highlights Income statement 000 31 Jan 2016 31 Jan 2015 Revenue 816 100 Cost of sales (154) (32) Gross Margin
Product Size Catalog Number
 Smooth Muscle Cells Instruction Manual Product Size Catalog Number Human Aortic (HAoSMC) Human Coronary Artery Smooth Muscle Cells (HCASMC) Human Pulmonary Artery Smooth Muscle Cells (HPASMC) Human Umbilical
Smooth Muscle Cells Instruction Manual Product Size Catalog Number Human Aortic (HAoSMC) Human Coronary Artery Smooth Muscle Cells (HCASMC) Human Pulmonary Artery Smooth Muscle Cells (HPASMC) Human Umbilical
Liquidmetal Alloys in Minimally Invasive Medical Devices
 Liquidmetal Alloys in Minimally Invasive Medical Devices Industry Medical Challenge Produce a high performance medical device that meets extreme demands in an economically sensitive market. M illions of
Liquidmetal Alloys in Minimally Invasive Medical Devices Industry Medical Challenge Produce a high performance medical device that meets extreme demands in an economically sensitive market. M illions of
Special Specimen Collection Procedures for Coagulation Testing
 Special Specimen Collection Procedures for Coagulation Testing The accuracy of hemostasis testing depends upon the quality of the specimen submitted. Hemostasis specimens must be properly collected, labeled,
Special Specimen Collection Procedures for Coagulation Testing The accuracy of hemostasis testing depends upon the quality of the specimen submitted. Hemostasis specimens must be properly collected, labeled,
An Advancing Allografts Brief October Sterilization of Allografts. Advancing Allografts
 An Advancing Allografts Brief October 24 Sterilization of Allografts Advancing Allografts Ensuring Safety in Tissue Transplantation: The Sterilization of Allografts By Lloyd Wolfinbarger, Jr., PhD Tissue
An Advancing Allografts Brief October 24 Sterilization of Allografts Advancing Allografts Ensuring Safety in Tissue Transplantation: The Sterilization of Allografts By Lloyd Wolfinbarger, Jr., PhD Tissue
MIIG INJECTABLE Graft The following languages are included in this packet:
 MIIG INJECTABLE Graft 128801-10 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese (sch)
MIIG INJECTABLE Graft 128801-10 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese (sch)
CAUTION: U.S. Federal law restricts this device to sale by or on the order of a licensed physician.
 TM CAUTION: U.S. Federal law restricts this device to sale by or on the order of a licensed physician. TABLE OF CONTENTS Section Port Styles 4 Description 5 Indications 5 Contraindications 5-6 Information
TM CAUTION: U.S. Federal law restricts this device to sale by or on the order of a licensed physician. TABLE OF CONTENTS Section Port Styles 4 Description 5 Indications 5 Contraindications 5-6 Information
DuraGen Secure. Dural Regeneration Matrix
 DuraGen Secure Dural Regeneration Matrix DESCRIPTION DuraGen Secure Dural Regeneration Matrix is an absorbable implant for the repair of dural defects. DuraGen Secure Dural Regeneration Matrix is an easy
DuraGen Secure Dural Regeneration Matrix DESCRIPTION DuraGen Secure Dural Regeneration Matrix is an absorbable implant for the repair of dural defects. DuraGen Secure Dural Regeneration Matrix is an easy
Regulating Cell Therapy: An Ex-Regulators Perspective
 Regulating Cell Therapy: An Ex-Regulators Perspective Christopher A Bravery 1 Introduction Regulation of Healthcare products in the EU What is cell therapy? How does it fit into existing framework? Is
Regulating Cell Therapy: An Ex-Regulators Perspective Christopher A Bravery 1 Introduction Regulation of Healthcare products in the EU What is cell therapy? How does it fit into existing framework? Is
Guidelines for the use of blood warming devices(aabb) Reference: 2002 AABB
 Guidelines for the use of blood warming devices(aabb) Reference: 2002 AABB 1 Hypothermia Introduction Induced by rapid, large-volume transfusion of refrigerated blood components A potential source of serious
Guidelines for the use of blood warming devices(aabb) Reference: 2002 AABB 1 Hypothermia Introduction Induced by rapid, large-volume transfusion of refrigerated blood components A potential source of serious
Advancing the Science of Gamma Irradiation Continuous improvement in radiation sterilization
 Advancing the Science of Gamma Irradiation Continuous improvement in radiation sterilization Fatima Hasanain, Polymer Materials Specialist www.nordion.com Why are we here today? 2 Overview Radiation sterilization
Advancing the Science of Gamma Irradiation Continuous improvement in radiation sterilization Fatima Hasanain, Polymer Materials Specialist www.nordion.com Why are we here today? 2 Overview Radiation sterilization
University Health Network UDI Capture Work Group Case Study
 LEARNING UDI COMMUNITY University Health Network UDI Capture Work Group Case Study WWW.AHRMM.ORG / LUC WORK GROUP TITLE: UDI Capture Work Group CASE STUDY PARTICIPANTS Wendy Watson, OR Supply Chain Manager
LEARNING UDI COMMUNITY University Health Network UDI Capture Work Group Case Study WWW.AHRMM.ORG / LUC WORK GROUP TITLE: UDI Capture Work Group CASE STUDY PARTICIPANTS Wendy Watson, OR Supply Chain Manager
New creative textile construction using innovative braiding technology and materials
 White Paper New creative textile construction using innovative braiding technology and materials DSM and Meister Provide Foundation for Medical Device Design Philippe Gédet, Engineer, Meister & Cie AG
White Paper New creative textile construction using innovative braiding technology and materials DSM and Meister Provide Foundation for Medical Device Design Philippe Gédet, Engineer, Meister & Cie AG
UNIVERSITY OF TOLEDO
 UNIVERSITY OF TOLEDO SUBJECT: SHIPPING, PACKAGING AND RECEIPT OF Procedure No: HM-08-031 HAZARDOUS MATERIALS PROCEDURE Employees of the University of Toledo attempting to transport hazardous materials,
UNIVERSITY OF TOLEDO SUBJECT: SHIPPING, PACKAGING AND RECEIPT OF Procedure No: HM-08-031 HAZARDOUS MATERIALS PROCEDURE Employees of the University of Toledo attempting to transport hazardous materials,
Artist Proof: Clotalyst. Autologous Serum Collection System
 Artist Proof: 01-15-10 Clotalyst Autologous Serum Collection System Clotalyst Autologous Serum The kit is designed for the preparation of autologous serum that is to be mixed with PRP and autograft or
Artist Proof: 01-15-10 Clotalyst Autologous Serum Collection System Clotalyst Autologous Serum The kit is designed for the preparation of autologous serum that is to be mixed with PRP and autograft or
GUIDELINES ON MEDICAL DEVICES
 EUROPEAN COMMISSION ENTERPRISE DIRECTORATE-GENERAL Single Market : regulatory environment, standardisation and New Approach Pressure equipment, medical devices, metrology MEDDEV. 2.11/1 rev.1 February
EUROPEAN COMMISSION ENTERPRISE DIRECTORATE-GENERAL Single Market : regulatory environment, standardisation and New Approach Pressure equipment, medical devices, metrology MEDDEV. 2.11/1 rev.1 February
PREVELEAK Surgical sealant
 PREVELEAK Surgical sealant Rx only Instructions for Use DEVICE DESCRIPTION PREVELEAK Surgical sealant (PREVELEAK) is a sealant developed to seal suture holes formed during surgical repair of the circulatory
PREVELEAK Surgical sealant Rx only Instructions for Use DEVICE DESCRIPTION PREVELEAK Surgical sealant (PREVELEAK) is a sealant developed to seal suture holes formed during surgical repair of the circulatory
Case Study: Examples Relating to the Quality Control of Cell-based Products
 Case Study: Examples Relating to the Quality Control of Cell-based Products CMC Strategy Forum Japan 2014 Yuuki Miyatake1), 2) 1) TEIJIN PHARMA LIMITED 2) Japan Pharmaceutical Manufacturers Association
Case Study: Examples Relating to the Quality Control of Cell-based Products CMC Strategy Forum Japan 2014 Yuuki Miyatake1), 2) 1) TEIJIN PHARMA LIMITED 2) Japan Pharmaceutical Manufacturers Association
ISO INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD ISO 3826-3 First edition 2006-09-15 Plastics collapsible containers for human blood and blood components Part 3: Blood bag systems with integrated features Poches en plastique souple
INTERNATIONAL STANDARD ISO 3826-3 First edition 2006-09-15 Plastics collapsible containers for human blood and blood components Part 3: Blood bag systems with integrated features Poches en plastique souple
Development of non-substantially manipulated cell-based ATMPs 1 : flexibility introduced via the application of the risk-based approach
 3 July 2017 EMA/CAT/216556/2017 Inspections, Human Medicines, Pharmacovigilance and Committees Division Development of non-substantially manipulated cell-based ATMPs 1 : flexibility introduced via the
3 July 2017 EMA/CAT/216556/2017 Inspections, Human Medicines, Pharmacovigilance and Committees Division Development of non-substantially manipulated cell-based ATMPs 1 : flexibility introduced via the
Skeletal Muscle Cells
 Skeletal Muscle Cells Instruction Manual Product Size Catalog Number Human Skeletal Muscle Cells (SkMC) 500,000 cryopreserved cells 500,000 proliferating cells C-12530 C-12580 Product Description New skeletal
Skeletal Muscle Cells Instruction Manual Product Size Catalog Number Human Skeletal Muscle Cells (SkMC) 500,000 cryopreserved cells 500,000 proliferating cells C-12530 C-12580 Product Description New skeletal
our history Codman Instrument Portfolio
 our history Symmetry Surgical is dedicated to developing high-quality surgical instruments that respond to the needs of clinicians as they arise, making a real difference in the lives of patients. Our
our history Symmetry Surgical is dedicated to developing high-quality surgical instruments that respond to the needs of clinicians as they arise, making a real difference in the lives of patients. Our
SA 450/60 Material Data Sheet
 General Product Information SA 450/60 Material Data Sheet SA 450/60 is a speciality medical grade platinum cured elastomer, offering high clarity combined with excellent physical properties. It is none
General Product Information SA 450/60 Material Data Sheet SA 450/60 is a speciality medical grade platinum cured elastomer, offering high clarity combined with excellent physical properties. It is none
Antibody-based HLA (Human Leucocyte Antigen ) tissue-typing technologies
 Human AB Serum Product Description Human AB Serum is a vital cell culture reagent for some human cell types providing growth factors, vitamins, nutrients as well as trace elements and transport factors,
Human AB Serum Product Description Human AB Serum is a vital cell culture reagent for some human cell types providing growth factors, vitamins, nutrients as well as trace elements and transport factors,
Discover TruPRP. PRP the way you want it.
 Discover TruPRP PRP the way you want it. Discover TruPRP Discover the quality of Magellan TruPRP. The Magellan technology provides an automated dual spin processing system that can deliver (PRP) Platelet
Discover TruPRP PRP the way you want it. Discover TruPRP Discover the quality of Magellan TruPRP. The Magellan technology provides an automated dual spin processing system that can deliver (PRP) Platelet
Contains Nonbinding Recommendations Draft - Not for Implementation
 Reprinted from FDA s website by EAS Consulting Group, LLC Select Updates for Non-Clinical Engineering Tests and Recommended Labeling for Intravascular Stents and Associated Delivery Systems Draft Guidance
Reprinted from FDA s website by EAS Consulting Group, LLC Select Updates for Non-Clinical Engineering Tests and Recommended Labeling for Intravascular Stents and Associated Delivery Systems Draft Guidance
MDR ID: Definition: Applicable:
 MDR Classification: (Reference Medical Device Regulation EU 2017/745, Annex VIII) Product: Product Name 1. DURATION OF USE MDR ID: Definition: Applicable: - Invasive Device: Continue Go to 2. Invasive
MDR Classification: (Reference Medical Device Regulation EU 2017/745, Annex VIII) Product: Product Name 1. DURATION OF USE MDR ID: Definition: Applicable: - Invasive Device: Continue Go to 2. Invasive
COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP)
 European Medicines Agency Evaluation of Medicines for Human Use London, 29 July 2004 CPMP/BPWG/1089/00 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE ON THE CLINICAL INVESTIGATION OF PLASMA
European Medicines Agency Evaluation of Medicines for Human Use London, 29 July 2004 CPMP/BPWG/1089/00 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE ON THE CLINICAL INVESTIGATION OF PLASMA
Where Quality Meets Flexibility
 Where Quality Meets Flexibility is an industry leading 503B Outsourcing Facility providing sterile and non-sterile compounding services to hospitals, surgery centers, clinics, researchers & patients nationwide.
Where Quality Meets Flexibility is an industry leading 503B Outsourcing Facility providing sterile and non-sterile compounding services to hospitals, surgery centers, clinics, researchers & patients nationwide.
Avalon CUSTOM. Catheter Solutions
 Avalon CUSTOM Catheter Solutions Nordson MEDICAL Nordson MEDICAL is your partner in the global life sciences market providing innovative components, devices and custom OEM solutions for the precision management
Avalon CUSTOM Catheter Solutions Nordson MEDICAL Nordson MEDICAL is your partner in the global life sciences market providing innovative components, devices and custom OEM solutions for the precision management
April 15, 2015 VIA ELECTRONIC MAIL
 April 15, 2015 VIA ELECTRONIC MAIL Patricia Brooks, RHIA Senior Technical Advisor Centers for Medicare and Medicaid Services Hospital and Ambulatory Policy Group Mail Stop C4-08-06 7500 Security Boulevard
April 15, 2015 VIA ELECTRONIC MAIL Patricia Brooks, RHIA Senior Technical Advisor Centers for Medicare and Medicaid Services Hospital and Ambulatory Policy Group Mail Stop C4-08-06 7500 Security Boulevard
Patient Handbook on Stem Cell Therapies
 Patient Handbook on Stem Cell Therapies WWW.ISSCR.ORG WWW.CLOSERLOOKATSTEMCELLS.ORG Patient Handbook on Stem Cell Therapies Introduction We have all heard about the extraordinary promise that stem cell
Patient Handbook on Stem Cell Therapies WWW.ISSCR.ORG WWW.CLOSERLOOKATSTEMCELLS.ORG Patient Handbook on Stem Cell Therapies Introduction We have all heard about the extraordinary promise that stem cell
KEY TO INNOVATION AT VASCUTEK JAPAN MATTERS FEBRUARY 7TH 2014
 KEY TO INNOVATION AT VASCUTEK JAPAN MATTERS FEBRUARY 7TH 2014 VASCUTEK LTD : Background Manufacturer of synthetic vascular grafts In business since 1982 Over 500 employees Turnover around 70 million No.
KEY TO INNOVATION AT VASCUTEK JAPAN MATTERS FEBRUARY 7TH 2014 VASCUTEK LTD : Background Manufacturer of synthetic vascular grafts In business since 1982 Over 500 employees Turnover around 70 million No.
Policies Concerning Survival Surgery of Mice
 Policies Concerning Survival Surgery of Mice Date approved: October 25, 2016 Purpose This document details the minimum standards for survival (recovery) surgery in all laboratory rodent species. Responsibility
Policies Concerning Survival Surgery of Mice Date approved: October 25, 2016 Purpose This document details the minimum standards for survival (recovery) surgery in all laboratory rodent species. Responsibility
Germany Tissue Engineered - Skin Substitutes Market Outlook to 2020
 Germany Tissue Engineered - Skin Substitutes Market Outlook to 2020 Reference Code: GDMECC0884DB Publication Date: October 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables...
Germany Tissue Engineered - Skin Substitutes Market Outlook to 2020 Reference Code: GDMECC0884DB Publication Date: October 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables...
Applying Cleaning Validation Principles in the Manufacture of Medical Devices and Diagnostics August 20, 2014 IVT San Diego
 1 Applying Cleaning Validation Principles in the Manufacture of Medical Devices and Diagnostics August 20, 2014 IVT San Diego Destin A. LeBlanc Cleaning Validation Technologies 2014 Destin A. LeBlanc Focus
1 Applying Cleaning Validation Principles in the Manufacture of Medical Devices and Diagnostics August 20, 2014 IVT San Diego Destin A. LeBlanc Cleaning Validation Technologies 2014 Destin A. LeBlanc Focus
March 6, Division of Dockets Management (HFA-305) Food and Drug Administration 5630 Fishers Lane, Room 1061 Rockville, MD 20852
 701 Pennsylvania Avenue, NW Suite 800 Washington, D.C. 20004 2654 Tel: 202 783 8700 Fax: 202 783 8750 www.advamed.org March 6, 2017 Division of Dockets Management (HFA-305) Food and Drug Administration
701 Pennsylvania Avenue, NW Suite 800 Washington, D.C. 20004 2654 Tel: 202 783 8700 Fax: 202 783 8750 www.advamed.org March 6, 2017 Division of Dockets Management (HFA-305) Food and Drug Administration
UNIT CELL PROCESSES UNDERLYING TISSUE ENGINEERING AND REGENERATIVE MEDICINE
 Massachusetts Institute of Technology Harvard Medical School Brigham and Women s Hospital VA Boston Healthcare System 2.79J/3.96J/20.441/HST522J UNIT CELL PROCESSES UNDERLYING TISSUE ENGINEERING AND REGENERATIVE
Massachusetts Institute of Technology Harvard Medical School Brigham and Women s Hospital VA Boston Healthcare System 2.79J/3.96J/20.441/HST522J UNIT CELL PROCESSES UNDERLYING TISSUE ENGINEERING AND REGENERATIVE
Bill Kabat and Carmen Irizarry of the ACTG/IMPAACT Lab Tech Committee. Network Name, Title Signature Date
 Title: Dried PBMC Pellet Processing Standard Operating Procedure (LDMS specimen code = BLD/EDTA or ACD/PEL) Origination Date: 01 Jun 2004 Total Pages: 6 Effective Date: 29 Nov 2012 SOP Number LTC-SOP-59
Title: Dried PBMC Pellet Processing Standard Operating Procedure (LDMS specimen code = BLD/EDTA or ACD/PEL) Origination Date: 01 Jun 2004 Total Pages: 6 Effective Date: 29 Nov 2012 SOP Number LTC-SOP-59
Update on the IVDR. Sue Spencer
 Update on the IVDR Sue Spencer Caution The new regulations are draft the principles have now been agreed but the Annexes are subject to minor changes Further details will be added later pre and post application
Update on the IVDR Sue Spencer Caution The new regulations are draft the principles have now been agreed but the Annexes are subject to minor changes Further details will be added later pre and post application
HydroSet. Injectable HA Bone Substitute
 HydroSet Injectable HA Bone Substitute The Latest Advancement in Bone Substitute Technology Fast Setting Injectable or Manual Implantation Osteoconductive Excellent Wet-field Characteristics Isothermic
HydroSet Injectable HA Bone Substitute The Latest Advancement in Bone Substitute Technology Fast Setting Injectable or Manual Implantation Osteoconductive Excellent Wet-field Characteristics Isothermic
Palivizumab (Synagis ) Criteria for the Respiratory Syncytial Virus (RSV) Season for Fee-For-Service Legacy Medicaid Recipients
 Palivizumab (Synagis ) Criteria for the 2017-2018 Respiratory Syncytial Virus (RSV) Season for Fee-For-Service Legacy Medicaid Recipients Palivizumab is indicated for the prevention of serious lower respiratory
Palivizumab (Synagis ) Criteria for the 2017-2018 Respiratory Syncytial Virus (RSV) Season for Fee-For-Service Legacy Medicaid Recipients Palivizumab is indicated for the prevention of serious lower respiratory
Freshman/Sophomore Junior Senior
 Freshman/Sophomore Junior Senior Course Recommendations Mathematics (6 courses) MA 1021 MA 1024 (Calculus I) (Calculus IV) MA 1022 MA 2051 (Calculus II) (Differential Equations) MA 1023 MA 2611 (Calculus
Freshman/Sophomore Junior Senior Course Recommendations Mathematics (6 courses) MA 1021 MA 1024 (Calculus I) (Calculus IV) MA 1022 MA 2051 (Calculus II) (Differential Equations) MA 1023 MA 2611 (Calculus
European Regenerative Medicine Firms & Their Strategic Approaches. Michael Morrison University of York
 European Regenerative Medicine Firms & Their Strategic Approaches Michael Morrison University of York OVERVIEW Creating the European RM Universe of firms Characterizing the European RM Universe Strategic
European Regenerative Medicine Firms & Their Strategic Approaches Michael Morrison University of York OVERVIEW Creating the European RM Universe of firms Characterizing the European RM Universe Strategic
Presenting SURGICEL Powder
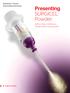 SURGICEL Powder Presenting SURGICEL Powder Built to stop continuous, broad-surface oozing fast1,2 The next generation of SURGICEL Absorbable Hemostats SURGICEL Powder efficiently and effectively controls
SURGICEL Powder Presenting SURGICEL Powder Built to stop continuous, broad-surface oozing fast1,2 The next generation of SURGICEL Absorbable Hemostats SURGICEL Powder efficiently and effectively controls
ROUTINE SPECIMEN COLLECTION BY SPECIMEN TYPE
 1. AUTOPSY EYE ROUTINE SPECIMEN COLLECTION BY SPECIMEN TYPE a. Arrangements: No special arrangements necessary. i. Immerse the specimen in a sufficient quantity of fixative so that the eye is covered completely
1. AUTOPSY EYE ROUTINE SPECIMEN COLLECTION BY SPECIMEN TYPE a. Arrangements: No special arrangements necessary. i. Immerse the specimen in a sufficient quantity of fixative so that the eye is covered completely
Medical Device Regulatory Framework 9 SEPTEMBER 2015 FUNDISA CONFERENCE JANE ROGERS
 Medical Device Regulatory Framework 9 SEPTEMBER 2015 FUNDISA CONFERENCE JANE ROGERS Key Topics Definitions Essential Principles Classification Conformity Assessment Framework License to Manufacture, Import,
Medical Device Regulatory Framework 9 SEPTEMBER 2015 FUNDISA CONFERENCE JANE ROGERS Key Topics Definitions Essential Principles Classification Conformity Assessment Framework License to Manufacture, Import,
Human IL-1 alpha ELISA Kit
 Human IL-1 alpha ELISA Kit Catalog No: CDK025B Quantity: 2 x 96 tests SPECIFICITY : RANGE : SENSITIVITY : INCUBATION : SAMPLE TYPES : Recognizes both natural and recombinant human IL-1 alpha 31.2 pg /
Human IL-1 alpha ELISA Kit Catalog No: CDK025B Quantity: 2 x 96 tests SPECIFICITY : RANGE : SENSITIVITY : INCUBATION : SAMPLE TYPES : Recognizes both natural and recombinant human IL-1 alpha 31.2 pg /
MISSION - Set (align) the arm bone, then apply the blue cast. The cast needs to be all the way down, and it needs to completely cover the break.
 2010 FLL CHALLENGE robot game Missions Biomedical engineering is the use of various engineering disciplines to help doctors and hospitals help patients. The fields of chemical, mechanical, electrical,
2010 FLL CHALLENGE robot game Missions Biomedical engineering is the use of various engineering disciplines to help doctors and hospitals help patients. The fields of chemical, mechanical, electrical,
The largest network of harmonized bio/ pharmaceutical GMP product testing labs worldwide, Eurofins BioPharma Product Testing enables companies to
 The largest network of harmonized bio/ pharmaceutical GMP product testing labs worldwide, Eurofins BioPharma Product Testing enables companies to advance candidates from development through commercialization
The largest network of harmonized bio/ pharmaceutical GMP product testing labs worldwide, Eurofins BioPharma Product Testing enables companies to advance candidates from development through commercialization
Clonetics Skeletal Muscle Myoblast Cell Systems HSMM Instructions for Use
 Lonza Walkersville, Inc. Walkersville, MD 21793-0127 USA U.S. Scientific Support: 800 521 0390 scientific.support@lonza.com EU/ROW Scientific Support: +32 87 321 611 scientific.support.eu@lonza.com Document
Lonza Walkersville, Inc. Walkersville, MD 21793-0127 USA U.S. Scientific Support: 800 521 0390 scientific.support@lonza.com EU/ROW Scientific Support: +32 87 321 611 scientific.support.eu@lonza.com Document
Precision in fixation
 For more information, visit Medartis at www.medartis.com or contact us directly: Medartis AG Hochbergerstrasse 60E CH-4057 Basel P +41 61 633 34 34 F +41 61 633 34 00 www.medartis.com Precision in fixation
For more information, visit Medartis at www.medartis.com or contact us directly: Medartis AG Hochbergerstrasse 60E CH-4057 Basel P +41 61 633 34 34 F +41 61 633 34 00 www.medartis.com Precision in fixation
Porton Biopharma Limited 1/17/17
 Porton Biopharma Limited 1/17/17 10903 New Hampshire Avenue Silver Spring, MD 20993 Via UPS Warning Letter 320 17 19 Return Receipt Requested January 19, 2017 Dr. Roger J. Hinton Managing Director Porton
Porton Biopharma Limited 1/17/17 10903 New Hampshire Avenue Silver Spring, MD 20993 Via UPS Warning Letter 320 17 19 Return Receipt Requested January 19, 2017 Dr. Roger J. Hinton Managing Director Porton
Functional Assessment and Clinical Outcome
 Tissue Engineering Functional Assessment and Clinical Outcome STEVEN A. GOLDSTEIN Orthopaedic Research Laboratories, Department of Orthopaedic Surgery, University of Michigan, Ann Arbor, Michigan 48109,
Tissue Engineering Functional Assessment and Clinical Outcome STEVEN A. GOLDSTEIN Orthopaedic Research Laboratories, Department of Orthopaedic Surgery, University of Michigan, Ann Arbor, Michigan 48109,
FAQ ABOUT. 21CFR Part 1271 FDA REGULATION OF REPRODUCTIVE TISSUE LABS
 FAQ ABOUT 21CFR Part 1271 FDA REGULATION OF REPRODUCTIVE TISSUE LABS XYTEX INTERNATIONAL, LTD. AUGUSTA, GEORGIA NOVEMBER 2005 Xytex International, 2005 Xytex is a registered trademark of Xytex International,
FAQ ABOUT 21CFR Part 1271 FDA REGULATION OF REPRODUCTIVE TISSUE LABS XYTEX INTERNATIONAL, LTD. AUGUSTA, GEORGIA NOVEMBER 2005 Xytex International, 2005 Xytex is a registered trademark of Xytex International,
Quality is Our Promise.
 Quality is Our Promise. Our goal at KRS Global Biotechnology is to provide the highest quality pharmaceutical preparations. We accomplish this with an unrivaled quality assurance and quality control program
Quality is Our Promise. Our goal at KRS Global Biotechnology is to provide the highest quality pharmaceutical preparations. We accomplish this with an unrivaled quality assurance and quality control program
01.48 Receiving and Processing Vendor Owned Surgical Instrumentation in the Sterile Processing Department
 01.48 Receiving and Processing Vendor Owned Surgical Instrumentation in the Purpose To provide effective management of and ensure standardization of processing for all reusable surgical instruments that
01.48 Receiving and Processing Vendor Owned Surgical Instrumentation in the Purpose To provide effective management of and ensure standardization of processing for all reusable surgical instruments that
FREQUENTLY ASKED QUESTIONS FOR THE ADVANTAGE
 FREQUENTLY ASKED QUESTIONS FOR THE ADVANTAGE What is an Automated Endoscope Reprocessor? An Automated Endoscopes Reprocessor (AER) performs all the cycle steps (fresh water preflush, rinsing, disinfection,
FREQUENTLY ASKED QUESTIONS FOR THE ADVANTAGE What is an Automated Endoscope Reprocessor? An Automated Endoscopes Reprocessor (AER) performs all the cycle steps (fresh water preflush, rinsing, disinfection,
RealLine Extraction 100
 Instructions for use EXTRACTION OF RNA AND DNA FROM CLINICAL SPECIMENS In vitro diagnosticum VBC8896 48 Tests valid from October 2016 Rev13102016_EN Page 1 of 12 Explanation of symbols used in labeling:
Instructions for use EXTRACTION OF RNA AND DNA FROM CLINICAL SPECIMENS In vitro diagnosticum VBC8896 48 Tests valid from October 2016 Rev13102016_EN Page 1 of 12 Explanation of symbols used in labeling:
NEXT GENERATION ECM-BASED ALLOGRAFT TECHNOLOGY:
 NEXT GENERATION ECM-BASED ALLOGRAFT TECHNOLOGY: Potent biological scaffolds strategically control stem cell fate and function, allowing our allografts to harness the regenerative potential of patient s
NEXT GENERATION ECM-BASED ALLOGRAFT TECHNOLOGY: Potent biological scaffolds strategically control stem cell fate and function, allowing our allografts to harness the regenerative potential of patient s
NHSBT Portfolio of Blood Components and Guidance for their Clinical Use. Summary of Significant Changes. Purpose. Definitions. Applicable Documents
 This Specification replaces SPN223/7 Copy Number Effective: 21/12/16 Summary of Significant Changes Introduction of HEV testing, and Pooled Platelets in Plasma/Additive mixture Amendment to interruption
This Specification replaces SPN223/7 Copy Number Effective: 21/12/16 Summary of Significant Changes Introduction of HEV testing, and Pooled Platelets in Plasma/Additive mixture Amendment to interruption
Finest Tubes for Medical Devices
 Finest Tubes for Medical Devices Who we are A Cirtec Company For over 30 years, Cirtec Medical has been partnering with medical device manufacturers, providing product design, development and contract
Finest Tubes for Medical Devices Who we are A Cirtec Company For over 30 years, Cirtec Medical has been partnering with medical device manufacturers, providing product design, development and contract
TIME SAVER LIFE SAVER
 TIME SAVER LIFE SAVER FedEx HealthCare Solutions FedEx. Solutions powered by people. FedEx HealthCare Solutions designed to fit your business With time-sensitive, high-value healthcare products such as
TIME SAVER LIFE SAVER FedEx HealthCare Solutions FedEx. Solutions powered by people. FedEx HealthCare Solutions designed to fit your business With time-sensitive, high-value healthcare products such as
Copyright 2017 Aegis Sciences Corporation All Rights Reserved
 Oral Fluid Collection Healthcare Collection Site Preparation 1. The collection site must have all the supplies needed to complete a specimen collection (e.g. collection kits, ink pens, Custody and Control
Oral Fluid Collection Healthcare Collection Site Preparation 1. The collection site must have all the supplies needed to complete a specimen collection (e.g. collection kits, ink pens, Custody and Control
SHIPMENT OF CLINICAL SPECIMENS TO THE IMPAACT SPECIMEN REPOSITORY
 SOP Number: SHP-001.01.02 Title: Shipment of Clinical Specimens to the NICHD Specimen Repository Version: 2.0 Effective Date: September 12, 2012 Approved By: Rohan Hazra, NICHD Last Review Date: September
SOP Number: SHP-001.01.02 Title: Shipment of Clinical Specimens to the NICHD Specimen Repository Version: 2.0 Effective Date: September 12, 2012 Approved By: Rohan Hazra, NICHD Last Review Date: September
VAPR TRIPOLAR 90 Suction Electrode with COOLPULSE Technology VALUE ANALYSIS BRIEF
 VAPR TRIPOLAR 90 Suction Electrode with COOLPULSE Technology VALUE ANALYSIS BRIEF Introduction and Methodology This value analysis brief presents information on the design features and potential procedural
VAPR TRIPOLAR 90 Suction Electrode with COOLPULSE Technology VALUE ANALYSIS BRIEF Introduction and Methodology This value analysis brief presents information on the design features and potential procedural
MALTE Outdoor. the compact and versatile posterior-walker. Instructions for use
 MALTE Outdoor the compact and versatile posterior-walker Instructions for use Dear Customer, At this point we would like to thank you for placing your trust in our company and for purchasing our product.
MALTE Outdoor the compact and versatile posterior-walker Instructions for use Dear Customer, At this point we would like to thank you for placing your trust in our company and for purchasing our product.
BASE 128 and BASE CONTENTS
 BASE 128 and BASE CONTENTS INTRODUCTION RATIONALE FOR PRODUCT DEVELOPMENT & FORMULAS DECONTAMINATION EFFICACY OF BASE 128 RINSING EFFICACY OF BASE TISSUE SAFETY PRODUCT VALIDATION & COLLABORATIONS COMPLIANCE
BASE 128 and BASE CONTENTS INTRODUCTION RATIONALE FOR PRODUCT DEVELOPMENT & FORMULAS DECONTAMINATION EFFICACY OF BASE 128 RINSING EFFICACY OF BASE TISSUE SAFETY PRODUCT VALIDATION & COLLABORATIONS COMPLIANCE
COMPANY WITH QUALITY MANAGEMENT SYSTEM CERTIFIED BY DNV GL = ISO 9001 = Biomaterial Hemolytic Assay. Kit Insert
 COMPANY WITH QUALITY MANAGEMENT SYSTEM CERTIFIED BY DNV GL = ISO 9001 = Biomaterial Hemolytic Assay Kit Insert Version: April 2017 Summary Hemolytic activity is a requirement to be tested for any blood
COMPANY WITH QUALITY MANAGEMENT SYSTEM CERTIFIED BY DNV GL = ISO 9001 = Biomaterial Hemolytic Assay Kit Insert Version: April 2017 Summary Hemolytic activity is a requirement to be tested for any blood
Process validation in medical devices
 Process validation in medical devices Fulfil requirements with expert regulatory guidance 1TÜV SÜD Contents INTRODUCTION 4 VALIDATION PLANNING 5 INSTALLATION QUALIFICATION 7 OPERATIONAL QUALIFICATION 9
Process validation in medical devices Fulfil requirements with expert regulatory guidance 1TÜV SÜD Contents INTRODUCTION 4 VALIDATION PLANNING 5 INSTALLATION QUALIFICATION 7 OPERATIONAL QUALIFICATION 9
TST Medical Devices. company profile
 TST Medical Devices company profile WE ARE LOOKING FORWARD TO THE FUTURE, ANTICIPATING CUSTOMERS NEEDS AND COMING UP WITH SOLUTIONS. We are offering quality and value. We strive for continuous improvment.
TST Medical Devices company profile WE ARE LOOKING FORWARD TO THE FUTURE, ANTICIPATING CUSTOMERS NEEDS AND COMING UP WITH SOLUTIONS. We are offering quality and value. We strive for continuous improvment.
Plastics collapsible containers for human blood and blood components. Part 1: Conventional containers
 Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 3826-1 Second edition 2013-06-01 Plastics collapsible containers for human blood and blood components Part 1: Conventional containers Poches en
Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 3826-1 Second edition 2013-06-01 Plastics collapsible containers for human blood and blood components Part 1: Conventional containers Poches en
Aramus Single-Use 2D Bag Assemblies
 LIFE SCIENCES VALIDATION PACKAGE Aramus Single-Use 2D Bag Assemblies High-grade, gamma-stable fluoropolymer film providing higher purity and greater reliability TABLE OF CONTENTS Section 1: Introduction...
LIFE SCIENCES VALIDATION PACKAGE Aramus Single-Use 2D Bag Assemblies High-grade, gamma-stable fluoropolymer film providing higher purity and greater reliability TABLE OF CONTENTS Section 1: Introduction...
Material Safety Data Sheet
 Material Safety Data Sheet Issue: 8 Date: April 2015 SECTION 1 PRODUCT AND COMPANY IDENTIFICATION PRODUCT NAME: BioBall PROPER SHIPPING NAME: Biological Substance, Category B PRODUCT USE: Used for quantitative
Material Safety Data Sheet Issue: 8 Date: April 2015 SECTION 1 PRODUCT AND COMPANY IDENTIFICATION PRODUCT NAME: BioBall PROPER SHIPPING NAME: Biological Substance, Category B PRODUCT USE: Used for quantitative
