Oligomerization of µ and δ Opioid Receptors: Generation of Novel Functional Properties
|
|
|
- Oswin Blankenship
- 5 years ago
- Views:
Transcription
1 JBC Papers in Press. Published on June 6, 2 as Manuscript M3452 Oligomerization of µ and δ Opioid Receptors: Generation of Novel Functional Properties Susan R. George 1,2,3, Theresa Fan 3, Zhidong Xie 1, Roderick Tse 1, Vincent Tam 1, George Varghese 1 and Brian F. O Dowd 1,3 Departments of 1 Pharmacology and 2 Medicine, University of Toronto, Toronto and the 3 Centre for Addiction and Mental Health, Toronto, Ontario, Canada Address correspondence to: Dr. Susan R. George Department of Pharmacology University of Toronto Medical Sciences Building, Room King s College Circle Toronto, Ontario M5S 1A8 Canada Tel: (416) Fax: (416) s.george@utoronto.ca Running Title: Oligomerization of mu and delta opioid receptors Copyright 2 by The American Society for Biochemistry and Molecular Biology, Inc.
2 SUMMARY The existence of dimers and oligomers for many G protein coupled receptors (GPCRs) have been described by us and others. Since many GPCR subtypes are highly homologous to each other, we examined whether closely related receptors may interact with each other directly, and thus have the potential to create novel signaling units. Using µ and δ opioid receptors, we show that each receptor expressed individually was pharmacologically distinct and could be visualized following electrophoresis as monomers, homodimers, homotetramers and higher MW oligomers. When µ and δ opioid receptors were coexpressed, the highly selective synthetic agonists for each had reduced potency and altered rank order, whereas endomorphin-1 and Leu-enkephalin had enhanced affinity, suggesting the formation of a novel binding pocket. No heterodimers were visualized in the membranes coexpressing µ and δ receptors by the methods available. However, heterooligomers were identified by the ability to coimmuno-precipitate µ receptors with δ receptors, and vice versa, using differentially epitope tagged receptors. In contrast to the individually expressed µ and δ receptors, the coexpressed receptors showed insensitivity to pertussis toxin and continued signal transduction, likely due to interaction with a different subtype of G protein. In the present study, we provide for the first time, evidence for the direct interaction of µ and δ opioid receptors to form oligomers, with the generation of novel pharmacology and G protein coupling properties. 2
3 INTRODUCTION Opioid receptors have distinct pharmacological profiles and discrete but overlapping distributions in brain. The relatively recent cloning of opioid receptors has established that the products of three genes form the known subtypes, the µ, δ and κ opioid receptors, that interact with the complex family of endogenous opioid peptides (reviewed,1). The endogenous opioid peptide-receptor systems mediate important physiologic functions related to pain perception, locomotion, motivation, reward, autonomic function, immunomodulation and hormone secretion. The analysis of the contribution of each receptor type to the various opioid functions documented has been limited by the selectivity and cross-reactivity of the available opioid ligands and the postulation that multiple receptor subtypes are present. Since the cloning of the opioid receptors, the individual pharmacological and biochemical profiles of the µ, δ and κ opioid receptors have been better defined, however there are many aspects of opioid receptor biology that still remain poorly understood. A major problem that still remains is that the pharmacology of opioid receptors in brain tissue predicts a greater number of receptor subsites than revealed by opioid receptor cloning (1, 2). One possible explanation may be that the precise receptor-effector interactions present in endogenous brain regions may have not been adequately replicated in the heterologous expression systems in which the cloned receptors have been studied, or alternatively, that the relative proportion of the three receptor subtypes, variably expressed endogenously, may predicate the pharmacological profile, possibly by 3
4 receptor-receptor or distal interactions. The existence of such interactions or receptor cross-talk has long been postulated in the field of opioid receptor pharmacology (2,3), with functional (4,5) and biochemical (3,5) evidence for interactions between the µ and δ opioid receptors. Moreover, such interactions may be region-specific, as indicated by their occurrence in brain but not in peripheral locations (6,7). Intriguing observations in µ receptor knockout mice show that δ opioid receptor mediated analgesia was reduced and δ mediated respiratory depression was abolished, whereas κ opioid receptor function was preserved (8). These findings suggest that significant functional interactions may take place between µ and δ opioid receptors in specific neuronal pathways in brain. We and many other groups have described dimers and oligomers for many GPCRs mediating the actions of neurotransmitters, such as dopamine (9-12), serotonin (13), and for neuropeptide receptors such as for vasopressin (14) and opioid peptides (15). Dimerization of the δ opioid receptor has been suggested to have an important role in activation and internalization of the receptor (15). Since homodimerization appears to be an universal occurrence for GPCRs, we examined whether closely related receptors may heterodimerize and thus have the potential to create novel signaling units. For the Family A GPCRs, we showed that the related serotonin 5HT-1B and 5HT-1D receptors (68% identity overall, 77% in transmembrane domains) form homodimers and heterodimers (16). Among the Family B GPCRs, it has been shown that the GABA-B1 and GABA-B2 receptors heterodimerize and were dependent on this process for some aspects of signal transduction and cell surface localization (reviewed, 17). Since the µ and δ opioid receptors are highly homologous (share 65% amino acid 4
5 identity overall), we examined whether these closely related receptors could heterodimerize and whether they exhibited any evidence for direct modification of the distinct pharmacological profile of one when coexpressed with the other. While this work was in progress, a study showing heterodimerization of κ and δ opioid receptors was reported (18). In the present study, we provide for the first time, evidence for the direct interaction of µ and δ opioid receptors to form oligomers, and the generation of novel pharmacology and functional characteristics when µ and δ opioid receptors are expressed together, indicating that such interactions between these receptors may lead to novel signaling properties. 5
6 EXPERIMENTAL PROCEDURES Construction of µ and δ opioid receptor expression vectors cdnas encoding the rat µ and δ opioid receptors were inserted separately into the mammalian expression vector pcdna3. For the immunoprecipitation studies, the receptors were tagged with the c-myc or FLAG epitopes. The receptor cdnas were modified using the Transformer Site Directed Mutagenesis kit (Clontech) to insert after the NH 2 -terminal start methionine, an 11 residue c-myc epitope (EQKLISEEDL) for the µ opioid receptor and an 8 residue FLAG epitope (DYKDDDDK) for the δ opioid receptor. The absence of sequence errors and the correct orientation of the PCR products into the expression vectors were verified by sequencing on both strands. Expression in mammalian cells COS-7 monkey kidney cells and Chinese hamster ovary (CHO-K1) cells (American Type Culture Collection) were maintained as monolayer cultures at 37 C in minimal essential medium supplemented with 1% fetal bovine serum and antibiotics. COS-7 or CHO-K1 cells were transfected with the pcdna3 vectors using Lipofectamine (BRL). An equal amount of pcdna3 vector was co-transfected with each receptor construct so that the total amount of DNA used was consistent with studies involving transfections with two constructs. The receptor expression levels ranged between 7-12 fmol/mg protein. 6
7 Membrane Preparation Cells were washed extensively with PBS. Cell lysate was prepared by polytron disruption in ice-cold 5 mm Tris-HCl, 2 mm EDTA buffer, containing 5 µg/ml leupeptin, 1 µg/ml benzamidine and 5 µg/ml soybean trypsin inhibitor as described previously (13). The lysate was subjected to centrifugation at 1 x g to pellet unbroken cells and nuclei and to recover the supernatant which was centrifuged at high speed (4 x g for 2 min at 4 C) to prepare the crude membrane fraction. Membranes were washed with 5 mm Tris-HCl, 2 mm EDTA buffer containing protease inhibitors and centrifuged at high speed to prepare P2 membranes. Membrane protein was determined by the Bradford assay (BioRad) according to the manufacturer s instructions. Radioligand binding assays Saturation binding experiments were performed with ~2 µg of P2 membrane protein from Cos cells incubated with increasing concentrations of [3H]naloxone or [3H]diprenorphine and used to determine receptor densities (Bmax) and ligand affinities (KD) as previously described (19). Each concentration was examined in duplicate and incubated for 2 hours at 22 C in a total volume of 1 ml binding buffer (5 mm Tris-HCl, 5 mm EDTA, 1.5 mm CaCl2, 5 mm MgCl2, 5 mm KCl, 12 mm NaCl) with protease inhibitors. Nonspecific binding was defined as that not displaced by 1 µm naltrexone. The whole cell binding assays were conducted at 4 C and the incubation period was 4 hours. Competition experiments were performed in triplicate with increasing 7
8 concentrations of competing ligand ( M). The concentration of radioligand used in the competition assays was approximately equivalent to its KD. Bound ligand was isolated by rapid filtration through a 48-well cell harvester (Brandel) using GF/C filters (Whatman). Filters were washed with 1 ml of cold 5 mm Tris-HCl buffer (ph 7.4) and placed in glass vials with scintillation fluid (Universol) and counted for tritium. Data were analysed by nonlinear least-squares regression, using the computer-fitting program Prism (GraphPad). A two-site fit was designated only when a statistically significant improvement of the fit over a one-site model was obtained (by comparison of the coefficients of the goodness-of-fit by an F test). Adenylyl Cyclase Activity Adenylyl cyclase assays were conducted essentially as described (13). The assay mix contained.2 ml of membrane suspension from CHO cells (1-25 µg of protein),.12 mm ATP,.1 mm camp,.53 mm GTP, 2.7 mm phosphoenolpyruvate,.2 U pyruvate kinase, 1 U myokinase, 1 µm forskolin and.13 µci of [ 32 P]ATP in a final volume of.5 ml. The mixture was incubated with M agonist at 22 C for 2 min and enzyme activities were determined. Reactions were stopped by the addition of 1 ml of an ice-cold solution containing.4 mm ATP,.3 mm camp and [3H]-cAMP (25, cpm). Cyclic AMP was isolated by sequential column chromatography using Dowex cation exchange resin and aluminum oxide. Data were analyzed by computer fitted nonlinear least-squares regression. 8
9 Gel electrophoresis and immunoblotting The membrane samples were subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) under reducing conditions using 1% or 12% precast acrylamide gels (Novex) and transferred to nitrocellulose as described previously (16). Immunoreactivity was revealed with antibodies raised against the c-myc epitope (Santa Cruz) and FLAG epitope (Sigma) and HRP-conjugated goat antimouse IgG (BioRad) and the Enhanced Chemiluminescence Detection kit (Amersham). Immunoprecipitation Studies The P2 membrane pellet was resuspended and stirred at 4 C overnight in 5 ml of freshly prepared solubilization buffer, consisting of 1 mm NaCl, 1 mm Tris-HCl ph 7.4, 2% digitonin and 5 mm EDTA with protease inhibitors. The homogenate was centrifuged at 27 g for 2 min, and the solubilized fraction was washed and concentrated in Centriprep-3 four times with 2 ml cold Buffer A: 1 mm NaCl, 1 mm Tris-HCl ph 7.4, with protease inhibitors. The washed fraction was precleared at 4 C with a 1/4 of agarose-fixed goat anti-mouse IgG overnight. The solubilized receptors were immunoprecipitated with the mouse monoclonal anti-myc or anti-flag antibody at a 1/5 dilution in buffer A for 6 h and agitated gently overnight with a 1/4 dilution of agarose-fixed goat anti-mouse IgG. The immunoprecipitate was washed with cold buffer A, solubilized in SDS sample buffer, and electrophoresed by SDS-PAGE. 9
10 RESULTS Interactions between µ and δ receptors Membranes expressing epitope-tagged µ or δ opioid receptors subjected to electrophoresis revealed receptor species detected as monomers, dimers, tetramers and higher order oligomers (Fig. 1A). The approximate molecular weights of the specific bands detected were as follows: for the µ opioid receptor, 9 kda (monomer), 18 kda (dimer), >3 kda (tetramer), and for the δ opioid receptor, 6 kda (monomer), 12 kda (dimer) and ~24 kda (tetramer). The addition of an epitope to the NH 2 -terminus had no effect on the electrophoretic mobility of the receptor species visualized on SDS- PAGE (data not shown). Also, the c-myc-µ receptor could not be immunoprecipitated by the FLAG antibody or vice versa for the FLAG-δ receptor from membranes expressing only one receptor subtype. No distinct heterodimer or heterooligomer bands could be discerned in membranes from cells coexpressing both receptors, whereas the monomers, homodimers and tetramers of each were easily discerned. When the receptors were coexpressed, both µ and δ receptors were detectable by Western blotting in the same membranes (Fig. 1B). In membranes coexpressing both receptors, using differential immunoprecipitation, c-myc-µ receptors were co-precipitated by the FLAG antibody, and FLAG-δ receptors by the c-myc antibody (Fig.1C), indicating an interaction between the µ and δ receptors in the cell, probably at an oligomeric level. The co- 1
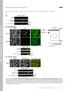
11 immunoprecipitation was specific and did not occur for other GPCRs epitope-tagged similarly and expressed with the c-myc-µ opioid receptor or when membranes expressing the receptors individually were mixed together (data not shown). FIGURE 1 Pharmacological profile of coexpressed µ and δ receptors Membranes from Cos cells transfected with cdna encoding µ, δ or both receptors bound the non-selective [ 3 H]-naloxone and [ 3 H]-diprenorphine with high affinity, which was not altered under the coexpression conditions. The ability of highly selective µ and δ opioid ligands to interact with the receptors expressed separately or coexpressed was examined. The µ- and δ-selective synthetic opioid agonists had high affinity for the receptors expressed alone, but had altered affinity and a different rank order of affinity for the coexpressed µ-δ receptors. The µ-selective agonist DAMGO exhibited >16 fold greater affinity for the high affinity state of the µ receptor (indicated by the lower K H ) compared to the δ receptor high affinity state, and the δ-selective agonist DPDPE had >1, fold greater affinity for the δ receptor over µ (Table I). TABLE I When µ and δ receptors were coexpressed, there was a 1 fold reduction in affinity for DAMGO, DPDPE and morphine, as indicated by the higher affinity constant KH detected (Fig. 2A, B, C). To confirm that these findings did not result from the simple admixture of discrete µ and δ binding sites, these studies were repeated in membranes pooled together from cells separately expressing either µ or δ receptors. As shown in Fig. 3, recognition of µ opioid receptors by the µselective ligand DAMGO was not affected by 11
12 the mere presence of δ receptors in the membrane mixture (Fig. 3A), and similarly for DPDPE recognition of δ opioid receptors (Fig. 3B), as the respective KH values and receptor fractions in the high and low affinity states remained unchanged. FIGURE 2, FIGURE 3 The rank order of the affinities of a series of compounds at the agonist-detected high and low affinity states of the receptor was examined as shown in Table I. For the coexpressed µ and δ opioid receptors, a comparison of the binding profiles of several of the endogenous opioid peptides revealed ~2-3 fold enhanced potencies of one of the endomorphin peptides and Leu-enkephalin at the high affinity site, with a small reduction in the affinity for Met-enkephalin. For the coexpressed µ-δ receptors the affinities for DAMGO and DPDPE were equivalent. In order to confirm that the observed alterations in pharmacology resulted entirely from µ and δ receptor interactions at the cell surface, radioligand binding studies were repeated in whole cells. The effects of receptor coexpression to result in reduced affinity for DAMGO and DPDPE were replicated in a manner identical to that observed in the membrane assays, as shown in Fig. 4. FIGURE 4 Sensitivity to guanine nucleotides Incubation of membranes expressing µ opioid receptors with GTPγS 5-8 µm resulted in a shift of DAMGO-detected affinities to 1 fold lower values, with no change in the proportion of receptors in the high (RH 64% in control vs. 7% with GTPγS 8 µm) and low affinity states. We have previously reported partial desensitization of 12
13 µ opioid receptors occurring with guanine nucleotide analogs, with a shift of receptor affinity to the right but without receptor uncoupling from G protein (19). In contrast to membranes exclusively expressing µ (Fig. 5A) and δ (Fig. 5B) opioid receptors, GTPγS had no effect on the two affinity states detected by DAMGO when the µ and δ receptors were coexpressed (Fig. 5C), suggesting insensitivity to guanine nucleotides (RH 6 % in control vs. 72% with GTPγS 8 µm). FIGURE 5 Sensitivity to pertussis toxin To determine the involvement of G proteins in the generation of the agonistdetected high affinity states, cells expressing the µ or δ receptor or coexpressing µ and δ opioid receptors were treated with pertussis toxin 1 µg/ml for 24 hrs and the membranes harvested. Pertussis toxin treatment resulted in a complete loss of the DAMGO-detected high affinity state of the µ opioid receptor (RH 5% in control and % with PTX) as reported previously (2), with 1% of the receptors existing in a single low affinity state, indicating uncoupling from G protein (Fig. 6A). Similarly in cells expressing the δ receptor, pertussis toxin treatment resulted in complete loss of the DPDPE-detected high affinity sites (RH 48% in control and % with PTX) (Fig. 6B). In the µ-δ expressing membranes, pertussis toxin treatment had no effect on the two affinity states detected by either DAMGO or DPDPE (Fig. 6C, D). These results suggest that the affinity states detected when the receptors are coexpressed, are either not indicative of subpopulations of receptors coupled to or uncoupled from G protein, or alternatively, may result from 13
14 coupling of the receptors to a pertussis toxin-insensitive G protein. FIGURE 6 Desensitization and internalization The effect of agonist activation on receptor desensitization and internalization were assessed for µ and δ opioid receptors expressed separately and together. In cells expressing µ opioid receptors, 74% of receptors were in the agonist-detected high affinity state and 26% in the low affinity state, and following exposure to DAMGO for 1 hr, receptor density on the cell surface was reduced by 3%, but the affinities for agonist (KH and KL) and the relative proportion of receptors in the two states was unchanged (Fig. 7A). For the δ opioid receptor, exposure to DPDPE for 1 hr resulted in a reduction of agonist potency and a 23% reduction in cell surface receptor density (Fig. 7B). In contrast, in cells coexpressing µ and δ opioid receptors no desensitization was evident, however, agonist-induced internalization was preserved and even enhanced with DAMGO but abolished with DPDPE with no changes in affinity for either agonist detected (Fig. 7C and D). FIGURE 7 Effect of µ and δ receptor blockade In order to determine whether the pharmacological profile of the coexpressed µ-δ opioid receptors could be affected by blockade of the component µ and δ opioid receptors, the agonist competition studies were performed in the presence of selective antagonists of each receptor subtype. FIGURE 8 14
15 In the presence of naltrindole, a selective antagonist of the δ opioid receptor, the overall density of the coexpressed receptors detected was reduced by 4-5 %, however the K H for DAMGO and the proportion of receptors in the two affinity states were unaffected, although the K L was shifted to the left (Fig. 8A). Treatment of the coexpressed receptors with the irreversible µ opioid receptor antagonist β FNA also reduced apparent receptor density by 5-7%, and revealed no changes in K H for DPDPE (Fig. 8C). In separate experiments, the effects of the naltrindole concentration on DAMGO competition of µ opioid receptor binding (Fig. 8B), and of β FNA concentration on DPDPE competition of δ opioid receptor binding (Fig. 8D) were assessed and found to have no significant effect on the agonist-detected sites detected. Adenylyl Cyclase Activity Adenylyl cyclase activity was assessed in membranes from cells expressing the µ and δ receptors separately or coexpressing them. Membranes expressing µ opioid receptors revealed inhibition of forskolin-stimulated cyclic AMP production by DAMGO in a dose-responsive manner, with EC nm; this activity was completely abolished by treatment of the cells with pertussis toxin (Fig. 9A). Similarly, in membranes expressing δ opioid receptors, DPDPE inhibited forskolin-stimulated adenylyl cyclase activity with an EC nm, also abolished by pertussis toxin treatment (Fig. 9B). In membranes coexpressing µ and δ receptors, following pertussis 15
16 toxin treatment, there was continued ability of DAMGO (Fig. 9C) and DPDPE (data not shown) to inhibit forskolin stimulated camp production at concentrations ~1 µm (n=3), suggesting the involvement of a pertussis toxin-insensitive G protein. FIGURE 9 DISCUSSION In this study we report that µ opioid receptors heterooligomerize with δ opioid receptors to form a novel signaling entity with distinct radioligand binding and functional properties, different from either the µ or δ opioid receptors. When µ opioid receptors were coexpressed with δ opioid receptors, the resulting µ-δ oligomeric complex had 1 fold lower affinity for the µ-selective and δ-selective agonists DAMGO and DPDPE respectively. The rank order of agonist affinities for a series of selected agonists were different for the µ-δ receptors compared to the profiles for µ or δ receptors, suggesting the formation of a binding pocket different from that present when µ or δ receptors were expressed singly. In addition, there was a small increase in the affinities of some of the endogenous opioid peptides for the high affinity site of the µ-δ receptor complex when compared to the µ opioid receptor by a factor of 2 to 3, suggesting a possible preferential 16
17 interaction with this entity over µ receptors. The novel binding site was dependent on the coexpression of the two receptors and could not be reproduced by simple admixture of membranes expressing each receptor separately. The preservation of the binding characteristics of DAMGO-detected µ opioid sites and of DPDPE-detected δ opioid sites in the mixed membrane preparation adds credence to the notion that a novel µ-δ binding site has been created in the coexpressing membranes. The agonist affinities for the µ-δ receptor were not affected by addition of guanine nucleotide, or pertussis toxin treatment of the cells, suggesting that the agonistdetected binding sites may not result from coupling to a G protein, or alternatively, may result from coupling to a pertussis toxin-insensitive G protein. Precedence for the latter possibility has been established previously (21, 22), with evidence that opioid receptors can couple to multiple G proteins, which in turn may couple to one or more effectors in different experimental models. More specifically, opioid peptide activation of pertussis toxin-insensitive G proteins such as Gz and possibly others, has been shown in physiological settings, such as mouse brain (21) and in a neuroblastoma cell line (22). The ability of the µ-δ receptor complex to inhibit adenylyl cyclase activity following pertussis toxin treatment suggests coupling to a G protein which is pertussis toxin insensitive, such as Gz, which has been shown to link to inhibition of adenylyl cyclase activity (23). The involvement of other effector pathways by the µ-δ complex remains to be established. Agonist treatment of the coexpressed µ-δ receptors revealed significant differences compared to µ or δ receptors expressed alone. In the combined presence of µ 17
18 and δ opioid receptors, there was resistance to desensitization and internalization upon exposure to DPDPE, although exposure to DAMGO resulted in an accentuated loss of receptors from the membrane. The ability to immunoprecipitate the µ opioid receptor using the epitope on the δ opioid receptor, and vice versa, suggests a very close interaction between these receptors in the cell membrane. The relative SDS-resistance of the homodimers of µ and δ receptors seen when each was expressed separately, suggests the presence of covalent and/or strong hydrophobic interactions between receptor monomers to form homodimers, however no heterodimers were visualized. If indeed heterodimers of µ and δ opioid receptors were present in the cell, the inability of the heterodimers to survive SDS-PAGE may indicate the presence of a less stable interaction, or alternatively, may indicate that the homodimers of each may participate in the formation of heterotetramers and heterooligomers. The precise nature of the structural determinants resulting in the interactions between µ and δ receptors remains to be elucidated. That the interaction between µ and δ opioid receptors resulted in the creation of a unique binding site is evident from the pharmacological profile of the coexpressed receptors. This interaction is present at the cell surface, as indicated by the identical radioligand binding parameters in whole cell binding as in membranes. This would suggest that distinct conformational changes occurred, altering the original binding pockets of the µ and δ receptors and even altering the conformation of the G protein interacting intracellular domains. The finding that blockade of one receptor with a selective antagonist did not restore binding of the other suggests that the binding site is 18
19 indeed novel, rather than occurring as a result of altered cooperativity between ligand binding sites on adjacent µ and δ receptors. Several studies have attempted to delineate the sites of interaction of receptors with G proteins, which although not identified, have been shown to depend on agonist mediated conformational changes. For the opioid receptor, different agonists have been shown to promote the exposure of distinct intracellular regions that activate specific G proteins, such as Gi, Go or Gz (22). In an analogous manner, conformational changes resulting from receptor:receptor interactions may also present a different aspect of the intracellular domains to interacting proteins and thus may alter the specificity of these interactions. In summary, we have provided evidence for the heterooligomerization of µ and δ opioid receptors, which results in the generation of a novel binding site with a significantly altered pharmacological profile compared to the receptors expressed individually. This novel binding entity may interact with a different complement of signal transducing G proteins and appears to have distinct patterns of desensitization and internalization upon exposure to µ and δ agonists. These data indicate that such interactions between the µ and δ receptors, and between the κ and δ receptors described recently (18), will enable the generation of a greater diversity of opioid signaling receptor units than predicted by the three cloned opioid receptor genes, and provides greater physiological significance for the overlapping distributions of the closely related opioid receptor subtypes present in brain. 19
20 Acknowledgements: This work was supported by grants from the Medical Research Council of Canada, the National Institutes of Health NIDA, and the Smokeless Tobacco Research Council, Inc. REFERENCES 1. Kieffer, B. L. Opioids: first lessons from knockout mice. Trends Pharmacol. Sci. (1999) 2, Traynor, J. R. and Elliott, J. delta-opioid receptor subtypes and cross-talk with mu-receptors. Trends Pharmacol Sci. (1993) 14, Rothman, R. B., Holaday, J. W. and Porreca, F. Allosteric coupling among opioid receptors: evidence for an opioid receptor complex. (1992) In: Herz A., Ed. Handbook of experimental pharmacology, Vol. 14 pp Springer Verlag, Berlin 2
21 4. Porreca, F., Takemori, A. E., Sultana, M., Portoghese, P. S., Bowen, W. D and, Mosberg, H. I. Modulation of mu-mediated antinociception in the mouse involves opioid delta-2 receptors. J. Pharmacol. Exp. Ther. (1992) 263, Rothman. R. B., Long, J. B., Bykov, V., Jacobson, A. E., Rice, K. C. and Holaday, J. W. Beta-FNA binds irreversibly to the opiate receptor complex: in vivo and in vitro evidence. J Pharmacol Exp Ther (1988) 247, Franklin, T. G. and Traynor, J. R. Alkylation with beta-funaltrexamine suggests differences between mu-opioid receptor systems in guinea-pig brain and myentericplexus. Br J Pharmacol. (1991) 12, Elliott, J. and Traynor, J. R. Evidence for lack of modulation of mu-opioid agonist action by delta-opioid agonists in the mouse vas deferens and guinea-pig ileum. Br J Pharmacol. (1995) 114, Matthes, H. W., Smadja, C., Valverde, O., Vonesch, J. L., Foutz, A. S., Boudinot, E., Denavit-Saubie, M., Severini, C., Negri, L., Roques, B. P., Maldonado, R. and Kieffer, B. L. Activity of the delta-opioid receptor is partially reduced, whereas activity of the kappa-receptor is maintained in mice lacking the mu-receptor. J Neurosci. (1998) 18, Zawarynski, P., Tallerico, T., Seeman, P., Lee, S. P., O Dowd, B. F. and George, S. R. Dopamine D2 receptor dimers in human and rat brain. FEBS Lett. (1998) 441, Ng, G. Y., Mouillac, B., George, S. R., Caron, M., Dennis, M., Bouvier, M. and O Dowd B. F. Desensitization, phosphorylation and palmitoylation of the human 21
22 dopamine D1 receptor. Eur J Pharmacol. (1994) 267, George, S. R., Lee, S., Varghese, G., Zeman, P., Seeman, P., Ng, G. Y. K. and O Dowd, B. F. A transmembrane domain-derived peptide inhibits D1 dopamine receptor function without affecting receptor oligomerization. J. Biol. Chem. (1998) 273, Nimchinsky, E. A., Hof, P. R., Janssen, W. G. M., Morrison, J. H. and Schmauss, C. Expression of dopamine D3 receptor dimers and tetramers in brain and in transfected cells. J Biol Chem. (1997) 272, Ng, G. Y., George, S. R., Zastawny, R. L., Caron, M., Bouvier, M., Dennis, M., O Dowd, B. F. Human serotonin1b receptor expression in Sf9 cells: phosphorylation, palmitoylation, and adenylyl cyclase inhibition. Biochem. (1993) 32, Zhu, X. and Wess, J. Truncated V2 vasopressin receptors as negative regulators of wild-type V2 receptor function. Biochem.. (1998) 37, Cvejic, S., Devi, L. A. Dimerization of the delta opioid receptor: implication for a role in receptor internalization. J Biol. Chem. (1997) 272, Xie, Z., Lee, S. P., O Dowd, B. F. and George, S. R. Serotonin 5HT-1B and 5HT-1D receptors form homodimers when expressed alone and heterodimers when coexpressed. (1999) FEBS Lett. 456, Marshall, F. H., Jones, K. A., Kaupmann, K. and Bettler, B. GABAB receptors - the first 7TM heterodimers. (1999) Trends Pharmacol Sci. 2, , 18. Jordan, B. A., Devi, L. A. G-protein-coupled receptor heterodimerization modulates receptor function. (1999) Nature. 399,
23 19. Pak, Y. S., Kouvelas, A., Schiedeler, M., Rassmussen, J., O Dowd, B. F. and George, S. R. Functional desensitization of the mu opioid receptor is mediated by loss of receptors from the membrane rather than uncoupling from G protein. (1996) Mol. Pharm. 5, Pak, Y., O Dowd, B.F. and George, S.R. Agonist-induced, G protein-dependent and -independent internalization of the mu opioid receptor. (1999) J. Biol. Chem. 274, Garzon, J., Castro, M. and Sanchez-Blazquez, P. Influence of Gz and Gi2 transducer proteins in the affinity of opioid agonists to µ receptors. (1998) Eur. J. Neurosci. 1, Allouche, S., Polastron, J., Hasbi, A., Homberger, V. and Jauzac, P. Differential G-protein activation by alkaloid and peptide opioid agonists in the human neuroblastoma cell line SK-N-BE. (1999) Biochem. J. 342, Obadiah, J., Avidor-Reiss, T., Fishburn, C.S., Carmon, S., Bayewitch, M., Vogel, Z., Fuchs, S. and Levevi-Sivan, B. Adenylyl cyclase interaction with the D2 dopamine receptor family; differential coupling to Gi, Gz and Gs. (1999) Cell. Mol. Neurobiol. 19,
24 Footnote: Abbreviations: K H = affinity constant of the agonist-detected high affinity site; DADLE = [D-Ala 2, D-Leu 5 ]-enkephalin; DAMGO = [D-Ala 2, N-Me-Phe 4, Gly 5 -ol]- enkephalin; DPDPE = [D-Pen 2,5 ]-enkephalin. 24
25 TABLE I AGONIST µ δ µ-δ K H / K L (nm) K H / K L (nm) K H / K L (nm) DADLE.26/536.7/125.5/167 DAMGO / / / / / / DPDPE Morphine.22/ / /262 Endomorphin-2 (YPFF) 4.45/ /1969 Endomorphin-1 (YPWF) Met-enkephalin 6.3/ / / / /54 Leu-enkephalin 6.3/ / /653 β-endorphin Dynorphin A 17./47 22./ / / / /122 Values shown are the mean and S.E.M. of n=7-8 determinations, or the average of n=2 determinations. Abbreviations: K H/L = affinity constant of the agonist-detected high (H) or low (L) affinity sites or the single site detected; DADLE = [D-Ala 2, D-Leu 5 ]-enkephalin; DAMGO = [D-Ala 2, N-Me-Phe 4, Gly 5 -ol]-enkephalin; DPDPE = [D-Pen 2,5 ]- enkephalin. FIGURE LEGENDS Figure 1: Western blots of membranes from Cos cells (A) expressing either µ or δ 25
26 opioid receptors, (B) coexpressing µ and δ receptors and (C) immunoprecipitated from membranes coexpressing both receptors. Epitope-tagged c-myc-µ and FLAG-δ were immunoblotted (i.b.) with the c-myc and FLAG antibodies in membranes from cells expressing each receptor singly (Lanes 2, 4), from untransfected cells (Lanes 1, 3) and from cells coexpressing both receptors (Lanes 5, 6). Coexpressed receptors were immunoprecipitated (i.p.) with FLAG antibody and immunoblotted with c-myc antibody (Lane 7) and immunoprecipitated with c-myc antibody and immunoblotted with FLAG antibody (Lane 8). Figure 2: Competition of 3 H-naloxone binding to membranes from Cos cells expressing µ or δ opioid receptors singly (clear symbols) or coexpressing them (solid symbols), by (A) DAMGO, (B) DPDPE and (C) morphine. Each assay was performed in triplicate, and the data shown are representative of n=8 experiments for DAMGO and DPDPE, and of n=2 for morphine. Figure 3: Competition by (A) DAMGO and (B) DPDPE of 3 H-naloxone binding to membranes from Cos cells expressing µ opioid receptors or δ opioid receptors singly (clear symbols) or mixed together (solid symbols). Each assay was performed in triplicate, and the data shown are representative of n=3 experiments. Figure 4: Competition by (A) DAMGO and (B) DPDPE of 3 H-naloxone binding to 26
27 whole cells (Cos) expressing µ opioid or δ opioid receptors singly or coexpressing them. Each assay was performed in triplicate and the data shown are representative of n=2 experiments. Figure 5: Effect of GTPγS (8 µm) on DAMGO competition of 3 H-naloxone binding to membranes from Cos cells (A) expressing µ opioid receptors and (B) expressing δ opioid receptors, and (C) coexpressing µ and δ opioid receptors. Figure 6: Effect of pertussis toxin (PTX) treatment of Cos cells expressing µ or δ opioid receptors separately, or coexpressing µ and δ opioid receptors. Competition of 3H-naloxone binding by (A, C) DAMGO and (B, D) DPDPE are shown. Figure 7: Effect of selective agonist treatment for 1 hour on competition of 3 H- naloxone binding by (A) DAMGO in membranes from cells expressing µ receptors, (B) DPDPE in membranes from cells expressing δ receptors, (C) DAMGO and (D) DPDPE in membranes from cells coexpressing µ and δ receptors. Figure 8: Effect of selective antagonist treatment on competition of 3 H-naloxone binding to membranes from Cos cells expressing µ opioid or δ opioid receptors singly or together. Shown is DAMGO competition with and without naltrindole 5nM in membranes from cells (A) coexpressing µ and δ receptors and in (B) expressing µ 27
28 receptors, and DPDPE competition with and without βfna 5nM in membranes from cells (C) coexpressing µ and δ receptors and in (D) expressing δ receptors. Figure 9: Effect of DAMGO and DPDPE on forskolin-stimulated adenylyl cyclase activity in membranes from CHO cells expressing (A) µ opioid receptors, (B) δ opioid receptors, and (C) coexpressing µ and δ receptors following vehicle treatment (control) or pertussis toxin treatment (PTX). Values shown are representative of 3 separate experiments. 28
29 29
30 B. myc-µ/ flag-δ myc-µ/ flag-δ - myc - flag kda C. myc-µ/ flag-δ myc-µ/ flag-δ flag myc myc flag kda A. myc-µ flag-δ i.p. i.b. - myc - flag kda myc UT UT - flag kda -25
31 % bound A. B. C µ µ-δ [D AM GO] M % bound δ µ-δ [DPD PE]M % bound µ µ-δ [MOR PHINE]M Fig.2 George et al.
32 A. B. % bound MIXED µ µ+δ [DAM GO ]M % bound δ MIXED µ+δ [DPD PE] M Fig.3 George etal.
33 A. B. % bound µ µ-δ [D AM GO] M δ % bound δ µ-δ [DPD PE]M µ Fig.4 G eorge etal.
34 % bound A µ +GTPγS [DA M GO] M % bound B. C δ [DPD PE] M +GTPγS % bound µ-δ +GTPγS [DA M GO] M Fig.5 Ge orge etal.
35 A. B. % bound µ +PTX % bound δ +PTX 2 2 % bound [DAM G O ] M C. D. µ-δ +PTX [D AM GO] M % bound [DPDPE] M µ-δ +PTX [DPD PE]M Fig.6 George et al.
36 A. B. receptor density fmol/mg receptor density fmol/mg DA MGO treated µ control [DAM GO] M 8 µ δ DA M G O treated µ δ control receptor density fmol/mg C. D. receptor density fmol/mg 1 DP DPE treated δ control [DPD PE] M µ δ control DP DP E treated [DAM GO] M [DPD PE] M Fig.7 Ge orge etal.
37 A. B. % bound % bound NTD µ-δ [D AM GO] M µ-δ +βfna [DPD PE] M % bound 1 C. D. % bound µ +NTD [D AM GO] M δ +βfna [DPD PE]M Fig.8 George et al.
38 Fig.9 George etal. % forskolin-s timulated cam P accum ulation 1 9 control µ [DAM G O ] M PTX %forskolin-stimulated cam P accum ulation 1 9 control δ PTX [DPDPE]M %forskolin-stimulated cam P accum ulation 1 9 control µ-δ PTX [DAM GO] M
39 Oligomerization of mu and delta opioid receptors: generation of novel functional properties Susan R. George, Theresa Fan, Zhidong Xie, Roderick Tse, Vincent Tam, George Varghese and Brian F. O'Dowd J. Biol. Chem. published online June 6, 2 Access the most updated version of this article at doi: 1.174/jbc.M3452 Alerts: When this article is cited When a correction for this article is posted Click here to choose from all of JBC's alerts
Gα i Activation Assay Kit
 A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Gα i Activation Assay Kit Catalog Number 80301 20 assays NewEast Biosciences, Inc 1 Table of Content Product
A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Gα i Activation Assay Kit Catalog Number 80301 20 assays NewEast Biosciences, Inc 1 Table of Content Product
Gα 13 Activation Assay Kit
 A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Gα 13 Activation Assay Kit Catalog Number: 80401 20 assays NewEast Biosciences 1 Table of Content Product
A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Gα 13 Activation Assay Kit Catalog Number: 80401 20 assays NewEast Biosciences 1 Table of Content Product
ab G alpha i Activation Assay Kit
 ab173234 G alpha i Activation Assay Kit Instructions for Use For the simple and fast measurement of G alpha i activation. This product is for research use only and is not intended for diagnostic use. Version
ab173234 G alpha i Activation Assay Kit Instructions for Use For the simple and fast measurement of G alpha i activation. This product is for research use only and is not intended for diagnostic use. Version
Cdc42 Activation Assay Kit
 A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Cdc42 Activation Assay Kit Catalog Number: 80701 20 assays 1 Table of Content Product Description 3 Assay
A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Cdc42 Activation Assay Kit Catalog Number: 80701 20 assays 1 Table of Content Product Description 3 Assay
Arf6 Activation Assay Kit
 A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Arf6 Activation Assay Kit Catalog Number: 82401 20 assays NewEast Biosciences 1 Table of Content Product
A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Arf6 Activation Assay Kit Catalog Number: 82401 20 assays NewEast Biosciences 1 Table of Content Product
Rab5 Activation Assay Kit
 A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Rab5 Activation Assay Kit Catalog Number: 83701 20 assays 24 Whitewoods Lane 1 Table of Content Product
A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based Rab5 Activation Assay Kit Catalog Number: 83701 20 assays 24 Whitewoods Lane 1 Table of Content Product
RheB Activation Assay Kit
 A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based RheB Activation Assay Kit Catalog Number: 81201 20 assays NewEast Biosciences 1 FAX: 610-945-2008 Table
A helping hand for your research Product Manual Configuration-specific Monoclonal Antibody Based RheB Activation Assay Kit Catalog Number: 81201 20 assays NewEast Biosciences 1 FAX: 610-945-2008 Table
LABORATÓRIUMI GYAKORLAT SILLABUSZ SYLLABUS OF A PRACTICAL DEMOSTRATION. Financed by the program
 TÁMOP-4.1.1.C-13/1/KONV-2014-0001 projekt Az élettudományi-klinikai felsőoktatás gyakorlatorientált és hallgatóbarát korszerűsítése a vidéki képzőhelyek nemzetközi versenyképességének erősítésére program
TÁMOP-4.1.1.C-13/1/KONV-2014-0001 projekt Az élettudományi-klinikai felsőoktatás gyakorlatorientált és hallgatóbarát korszerűsítése a vidéki képzőhelyek nemzetközi versenyképességének erősítésére program
Anti-HB-EGF (Human) mab
 Page 1 For Research Use Only. Not for use in diagnostic procedures. CODE No. D308-3 Anti-HB-EGF (Human) mab CLONALITY CLONE ISOTYPE QUANTITY SOURCE IMMUNOGEN FORMURATION STORAGE Monoclonal 3H4 Mouse IgG1
Page 1 For Research Use Only. Not for use in diagnostic procedures. CODE No. D308-3 Anti-HB-EGF (Human) mab CLONALITY CLONE ISOTYPE QUANTITY SOURCE IMMUNOGEN FORMURATION STORAGE Monoclonal 3H4 Mouse IgG1
ab Ran Activation Assay Kit
 ab173247 Ran Activation Assay Kit Instructions for Use For the simple and fast measurement of Ran activation. This product is for research use only and is not intended for diagnostic use. Version 1 Last
ab173247 Ran Activation Assay Kit Instructions for Use For the simple and fast measurement of Ran activation. This product is for research use only and is not intended for diagnostic use. Version 1 Last
Anti-CLOCK (Mouse) mab
 Page 1 For Research Use Only. Not for use in diagnostic procedures. Anti-CLOCK (Mouse) mab CODE No. D349-3 CLONALITY CLONE ISOTYPE QUANTITY SOURCE IMMUNOGEN FORMURATION STORAGE Monoclonal CLSP4 Mouse IgG1
Page 1 For Research Use Only. Not for use in diagnostic procedures. Anti-CLOCK (Mouse) mab CODE No. D349-3 CLONALITY CLONE ISOTYPE QUANTITY SOURCE IMMUNOGEN FORMURATION STORAGE Monoclonal CLSP4 Mouse IgG1
Protein A Agarose Immunoprecipitation Kit
 Protein A Agarose Immunoprecipitation Kit Catalog Number KA0568 20 Reactions Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information...
Protein A Agarose Immunoprecipitation Kit Catalog Number KA0568 20 Reactions Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information...
1. Cross-linking and cell harvesting
 ChIP is a powerful tool that allows the specific matching of proteins or histone modifications to regions of the genome. Chromatin is isolated and antibodies to the antigen of interest are used to determine
ChIP is a powerful tool that allows the specific matching of proteins or histone modifications to regions of the genome. Chromatin is isolated and antibodies to the antigen of interest are used to determine
Supplementary data. sienigma. F-Enigma F-EnigmaSM. a-p53
 Supplementary data Supplemental Figure 1 A sienigma #2 sienigma sicontrol a-enigma - + ++ - - - - - - + ++ - - - - - - ++ B sienigma F-Enigma F-EnigmaSM a-flag HLK3 cells - - - + ++ + ++ - + - + + - -
Supplementary data Supplemental Figure 1 A sienigma #2 sienigma sicontrol a-enigma - + ++ - - - - - - + ++ - - - - - - ++ B sienigma F-Enigma F-EnigmaSM a-flag HLK3 cells - - - + ++ + ++ - + - + + - -
Solutions to 7.02 Quiz II 10/27/05
 Solutions to 7.02 Quiz II 10/27/05 Class Average = 83 Standard Deviation = 9 Range Grade % 87-100 A 43 74-86 B 39 55-73 C 17 > 54 D 1 Question 1 (56 points) While studying deep sea bacteria, you discover
Solutions to 7.02 Quiz II 10/27/05 Class Average = 83 Standard Deviation = 9 Range Grade % 87-100 A 43 74-86 B 39 55-73 C 17 > 54 D 1 Question 1 (56 points) While studying deep sea bacteria, you discover
Supplementary Information: Materials and Methods. Immunoblot and immunoprecipitation. Cells were washed in phosphate buffered
 Supplementary Information: Materials and Methods Immunoblot and immunoprecipitation. Cells were washed in phosphate buffered saline (PBS) and lysed in TNN lysis buffer (50mM Tris at ph 8.0, 120mM NaCl
Supplementary Information: Materials and Methods Immunoblot and immunoprecipitation. Cells were washed in phosphate buffered saline (PBS) and lysed in TNN lysis buffer (50mM Tris at ph 8.0, 120mM NaCl
Supplemental Online Material. The mouse embryonic fibroblast cell line #10 derived from β-arrestin1 -/- -β-arrestin2 -/-
 #1074683s 1 Supplemental Online Material Materials and Methods Cell lines and tissue culture The mouse embryonic fibroblast cell line #10 derived from β-arrestin1 -/- -β-arrestin2 -/- knock-out animals
#1074683s 1 Supplemental Online Material Materials and Methods Cell lines and tissue culture The mouse embryonic fibroblast cell line #10 derived from β-arrestin1 -/- -β-arrestin2 -/- knock-out animals
FlashPlate File #15. High Throughput Screening. J. Watson SmithKline Beecham Pharmaceuticals, UK.
 Drug Discovery Research Clinical Screening High Throughput Screening FlashPlate File #15 Use of Novel FlashPlate Technology to Measure camp Accumulation in Chinese Hamster Ovary Cells Expressing Human
Drug Discovery Research Clinical Screening High Throughput Screening FlashPlate File #15 Use of Novel FlashPlate Technology to Measure camp Accumulation in Chinese Hamster Ovary Cells Expressing Human
Homogeneous GTPγS Assay for High Throughput Screening of GPCRs
 Homogeneous GTPγS Assay for High Throughput Screening of GPCRs Gregory Warner, Ph.D.*, Patricia Kasila, and Harry Harney * Enanta Pharmaceuticals Inc., 750 Main Street, Cambridge, MA 02139, 549 Albany
Homogeneous GTPγS Assay for High Throughput Screening of GPCRs Gregory Warner, Ph.D.*, Patricia Kasila, and Harry Harney * Enanta Pharmaceuticals Inc., 750 Main Street, Cambridge, MA 02139, 549 Albany
IgG TrueBlot Protocol for Mouse, Rabbit or Goatderived Antibodies - For Research Use Only
 IgG TrueBlot Protocol for Mouse, Rabbit or Goatderived Antibodies - For Research Use Only Introduction The IgG TrueBlot for mouse, rabbit, or goat-derived antibodies represents unique series of respective
IgG TrueBlot Protocol for Mouse, Rabbit or Goatderived Antibodies - For Research Use Only Introduction The IgG TrueBlot for mouse, rabbit, or goat-derived antibodies represents unique series of respective
Agonists at the d-opioid receptor modify the binding of m-receptor agonists to the m d receptor hetero-oligomer
 1122..1136 British Journal of Pharmacology DOI:1.1111/j.1476-5381.1.944.x www.brjpharmacol.org RESEARCH PAPERbph_944 Agonists at the d-opioid receptor modify the binding of m-receptor agonists to the m
1122..1136 British Journal of Pharmacology DOI:1.1111/j.1476-5381.1.944.x www.brjpharmacol.org RESEARCH PAPERbph_944 Agonists at the d-opioid receptor modify the binding of m-receptor agonists to the m
For Research Use Only. Not for use in diagnostic procedures.
 Printed December 13, 2011 Version 1.0 For Research Use Only. Not for use in diagnostic procedures. DDDDK-tagged Protein PURIFICATION GEL with Elution Peptide (MoAb. clone FLA-1) CODE No. 3326 / 3327 PURIFICATION
Printed December 13, 2011 Version 1.0 For Research Use Only. Not for use in diagnostic procedures. DDDDK-tagged Protein PURIFICATION GEL with Elution Peptide (MoAb. clone FLA-1) CODE No. 3326 / 3327 PURIFICATION
Analysing protein protein interactions using a GST-fusion protein to pull down the interacting target from the cell lysate Hong Wang and Xin Zeng
 Analysing protein protein interactions using a GST-fusion protein to pull down the interacting target from the cell lysate Hong Wang and Xin Zeng Department of Molecular Genetics, Biochemistry and Microbiology,
Analysing protein protein interactions using a GST-fusion protein to pull down the interacting target from the cell lysate Hong Wang and Xin Zeng Department of Molecular Genetics, Biochemistry and Microbiology,
Tumor tissues or cells were homogenized and proteins were extracted using
 SUPPLEMENTAL MATERIALS AND METHODS Western Blotting Tumor tissues or cells were homogenized and proteins were extracted using T-PER tissue protein extraction buffer. Protein concentrations were determined
SUPPLEMENTAL MATERIALS AND METHODS Western Blotting Tumor tissues or cells were homogenized and proteins were extracted using T-PER tissue protein extraction buffer. Protein concentrations were determined
Sarker et al. Supplementary Material. Subcellular Fractionation
 Supplementary Material Subcellular Fractionation Transfected 293T cells were harvested with phosphate buffered saline (PBS) and centrifuged at 2000 rpm (500g) for 3 min. The pellet was washed, re-centrifuged
Supplementary Material Subcellular Fractionation Transfected 293T cells were harvested with phosphate buffered saline (PBS) and centrifuged at 2000 rpm (500g) for 3 min. The pellet was washed, re-centrifuged
Science June 3, 1988 v240 n4857 p1310(7) Page 1
 Science June 3, 1988 v240 n4857 p1310(7) Page 1 by Brian K. Kobilka, Tong Sun Kobilka, Kiefer Daniel, John W. Regan, Marc G. Caron and Robert J. Lefkowitz COPYRIGHT 1988 American Association for the Advancement
Science June 3, 1988 v240 n4857 p1310(7) Page 1 by Brian K. Kobilka, Tong Sun Kobilka, Kiefer Daniel, John W. Regan, Marc G. Caron and Robert J. Lefkowitz COPYRIGHT 1988 American Association for the Advancement
Kinase Reaction and Alkylation Protocol
 Kinase Reaction and Alkylation Protocol Protocol for the treatment of substrates prior to detection by Thiophosphate Ester antibodies This product is for research use only and is not intended for diagnostic
Kinase Reaction and Alkylation Protocol Protocol for the treatment of substrates prior to detection by Thiophosphate Ester antibodies This product is for research use only and is not intended for diagnostic
Immunoprecipitation Protocol
 Immunoprecipitation Protocol Immunoprecipitation is a general method to obtain the enrichment of a specific protein from tissue lysate and cell lysate. It can be used to purify a specific protein, to identify
Immunoprecipitation Protocol Immunoprecipitation is a general method to obtain the enrichment of a specific protein from tissue lysate and cell lysate. It can be used to purify a specific protein, to identify
RhoC Activation Assay Kit
 Product Manual RhoC Activation Assay Kit Catalog Number STA-403-C 20 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Small GTP-binding proteins (or GTPases) are a family
Product Manual RhoC Activation Assay Kit Catalog Number STA-403-C 20 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Small GTP-binding proteins (or GTPases) are a family
Rapid and sensitive determination of recombinant protein expression
 APPLIAION NOE Pro-Detect Rapid assays Rapid and sensitive determination of recombinant protein expression Introduction Recombinant protein expression and purification is a multistep process that includes:
APPLIAION NOE Pro-Detect Rapid assays Rapid and sensitive determination of recombinant protein expression Introduction Recombinant protein expression and purification is a multistep process that includes:
Protocol(Research use only)
 Immunohistochemistry (without pretreatment) p2 Immunohistochemistry (Microwave pretreatment) p3 Immunohistochemistry (Autoclave pretreatment) p4 Immunohistochemistry (Trypsin pretreatment) p5 Immunohistochemistry
Immunohistochemistry (without pretreatment) p2 Immunohistochemistry (Microwave pretreatment) p3 Immunohistochemistry (Autoclave pretreatment) p4 Immunohistochemistry (Trypsin pretreatment) p5 Immunohistochemistry
His Tag Western and LumiBlot Reagents Sensitive detection of His Tag fusion proteins
 His Tag Western and LumiBlot Reagents Sensitive detection of His Tag fusion proteins The His Tag Reagents are kits containing optimized components for blot detection using the His Tag Monoclonal Antibody.
His Tag Western and LumiBlot Reagents Sensitive detection of His Tag fusion proteins The His Tag Reagents are kits containing optimized components for blot detection using the His Tag Monoclonal Antibody.
BACTERIAL PRODUCTION EXPRESSION METHOD OVERVIEW: PEF # GENE NAME EXPRESSION VECTOR MOLECULAR WEIGHT kda (full-length) 34.
 BACTERIAL PRODUCTION PEF # GENE NAME EXPRESSION VECTOR MOLECULAR WEIGHT 2015-XXXX XXXX pet-32a 50.9 kda (full-length) 34.0 kda (cleaved) EXPRESSION METHOD OVERVIEW: Plasmid DNA was transformed into BL21
BACTERIAL PRODUCTION PEF # GENE NAME EXPRESSION VECTOR MOLECULAR WEIGHT 2015-XXXX XXXX pet-32a 50.9 kda (full-length) 34.0 kda (cleaved) EXPRESSION METHOD OVERVIEW: Plasmid DNA was transformed into BL21
For Research Use Only. Not for use in diagnostic procedures. Anti-NRF2 mab
 Page 1 For Research Use Only. Not for use in diagnostic procedures. Anti-NRF2 mab CODE No. M200-3 CLONALITY CLONE ISOTYPE QUANTITY SOURCE IMMUNOGEN FORMURATION STORAGE Monoclonal 1F2 Mouse IgG1 100 L,
Page 1 For Research Use Only. Not for use in diagnostic procedures. Anti-NRF2 mab CODE No. M200-3 CLONALITY CLONE ISOTYPE QUANTITY SOURCE IMMUNOGEN FORMURATION STORAGE Monoclonal 1F2 Mouse IgG1 100 L,
Phos-tag beads as an immunoblotting enhancer for selective detection of phosphoproteins in cell lysates
 Notes & Tips Phos-tag beads as an immunoblotting enhancer for selective detection of phosphoproteins in cell lysates Emiko Kinoshita-Kikuta, Eiji Kinoshita *, and Tohru Koike Department of Functional Molecular
Notes & Tips Phos-tag beads as an immunoblotting enhancer for selective detection of phosphoproteins in cell lysates Emiko Kinoshita-Kikuta, Eiji Kinoshita *, and Tohru Koike Department of Functional Molecular
Technical Note Detection of post-immunoprecipitation proteins by Western blot using the Quick Western Kit IRDye 680RD
 Technical Note Detection of post-immunoprecipitation proteins by Western blot using the Quick Western Kit IRDye 680RD Developed for: Aerius, Odyssey Classic, Odyssey CLx and Odyssey Sa Imaging Systems
Technical Note Detection of post-immunoprecipitation proteins by Western blot using the Quick Western Kit IRDye 680RD Developed for: Aerius, Odyssey Classic, Odyssey CLx and Odyssey Sa Imaging Systems
TECHNICAL BULLETIN. MEK Activity Assay Kit. Product Code CS0490 Storage Temperature 20 C
 MEK Activity Assay Kit Product Code CS0490 Storage Temperature 20 C TECHNICAL BULLETIN Product Description The MAP kinase kinases (MAPKK, mitogen-activated protein kinase kinase, also termed MEK) are a
MEK Activity Assay Kit Product Code CS0490 Storage Temperature 20 C TECHNICAL BULLETIN Product Description The MAP kinase kinases (MAPKK, mitogen-activated protein kinase kinase, also termed MEK) are a
Strep-Spin Protein Miniprep Kit Catalog No. P2004, P2005
 INSTRUCTION MANUAL Strep-Spin Protein Miniprep Kit Catalog No. P2004, P2005 Highlights Fast protocol to purify Strep-tagged proteins from cell-free extracts Screen your recombinant colonies directly for
INSTRUCTION MANUAL Strep-Spin Protein Miniprep Kit Catalog No. P2004, P2005 Highlights Fast protocol to purify Strep-tagged proteins from cell-free extracts Screen your recombinant colonies directly for
human Angiotensin AT 2 Receptor Cell Line
 TECHNICAL DATA SHEET ValiScreen GPCR Cell Line Caution: For Laboratory Use. A research product for research purposes only human Angiotensin AT 2 Receptor Cell Line Product No.: ES-070-C Lot No.: M1W-C1
TECHNICAL DATA SHEET ValiScreen GPCR Cell Line Caution: For Laboratory Use. A research product for research purposes only human Angiotensin AT 2 Receptor Cell Line Product No.: ES-070-C Lot No.: M1W-C1
PROCEDURE FOR USE NICKEL NTA Magnetic Agarose Beads (5%)
 1 AFFINITY HIS-TAG PURIFICATION PROCEDURE FOR USE NICKEL NTA Magnetic Agarose Beads (5%) DESCRIPTION Nickel NTA Magnetic Agarose Beads are products that allow rapid and easy small-scale purification of
1 AFFINITY HIS-TAG PURIFICATION PROCEDURE FOR USE NICKEL NTA Magnetic Agarose Beads (5%) DESCRIPTION Nickel NTA Magnetic Agarose Beads are products that allow rapid and easy small-scale purification of
Figure 1: TDP-43 is subject to lysine acetylation within the RNA-binding domain a) QBI-293 cells were transfected with TDP-43 in the presence or
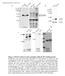 Figure 1: TDP-43 is subject to lysine acetylation within the RNA-binding domain a) QBI-293 cells were transfected with TDP-43 in the presence or absence of the acetyltransferase CBP and acetylated TDP-43
Figure 1: TDP-43 is subject to lysine acetylation within the RNA-binding domain a) QBI-293 cells were transfected with TDP-43 in the presence or absence of the acetyltransferase CBP and acetylated TDP-43
Data Sheet. CRE/CREB Reporter Assay Kit (camp/pka Cell Signaling Pathway) Catalog #: 60611
 Data Sheet CRE/CREB Reporter Assay Kit (camp/pka Cell Signaling Pathway) Catalog #: 60611 Background The main role of the camp response element, or CRE, is mediating the effects of Protein Kinase A (PKA)
Data Sheet CRE/CREB Reporter Assay Kit (camp/pka Cell Signaling Pathway) Catalog #: 60611 Background The main role of the camp response element, or CRE, is mediating the effects of Protein Kinase A (PKA)
LINGO-1, A TRANSMEMBRANE SIGNALING PROTEIN, INHIBITS OLIGODENDROCYTE DIFFERENTIATION AND MYELINATION THROUGH INTERCELLULAR SELF- INTERACTIONS.
 Supplemental Data: LINGO-1, A TRANSMEMBRANE SIGNALING PROTEIN, INHIBITS OLIGODENDROCYTE DIFFERENTIATION AND MYELINATION THROUGH INTERCELLULAR SELF- INTERACTIONS. Scott Jepson, Bryan Vought, Christian H.
Supplemental Data: LINGO-1, A TRANSMEMBRANE SIGNALING PROTEIN, INHIBITS OLIGODENDROCYTE DIFFERENTIATION AND MYELINATION THROUGH INTERCELLULAR SELF- INTERACTIONS. Scott Jepson, Bryan Vought, Christian H.
For Research Use Only. Not for use in diagnostic procedures. Anti-NRF2 mab
 Page 1 For Research Use Only. Not for use in diagnostic procedures. Anti-NRF2 mab CODE No. M200-3 CLONALITY CLONE ISOTYPE QUANTITY SOURCE IMMUNOGEN FORMURATION STORAGE Monoclonal 1F2 Mouse IgG1 κ 100 µl,
Page 1 For Research Use Only. Not for use in diagnostic procedures. Anti-NRF2 mab CODE No. M200-3 CLONALITY CLONE ISOTYPE QUANTITY SOURCE IMMUNOGEN FORMURATION STORAGE Monoclonal 1F2 Mouse IgG1 κ 100 µl,
SensoLyte Anti-alpha-Synuclein Quantitative ELISA Kit (Human/Mouse/Rat) *Colorimetric*
 Catalog # Kit Size SensoLyte Anti-alpha-Synuclein Quantitative ELISA Kit (Human/Mouse/Rat) *Colorimetric* AS-55550 One 96-well strip plate This kit is optimized to detect human/mouse/rat alpha-synuclein
Catalog # Kit Size SensoLyte Anti-alpha-Synuclein Quantitative ELISA Kit (Human/Mouse/Rat) *Colorimetric* AS-55550 One 96-well strip plate This kit is optimized to detect human/mouse/rat alpha-synuclein
Attenuation of synaptic toxicity and MARK4/PAR1-mediated Tau phosphorylation by
 Supplementary Methods and Figures Attenuation of synaptic toxicity and MARK4/PAR1-mediated Tau phosphorylation by methylene blue for Alzheimer s disease treatment Wenchao Sun 1, Seongsoo Lee 1,2, Xiaoran
Supplementary Methods and Figures Attenuation of synaptic toxicity and MARK4/PAR1-mediated Tau phosphorylation by methylene blue for Alzheimer s disease treatment Wenchao Sun 1, Seongsoo Lee 1,2, Xiaoran
Supplemental Data. LMO4 Controls the Balance between Excitatory. and Inhibitory Spinal V2 Interneurons
 Neuron, Volume 61 Supplemental Data LMO4 Controls the Balance between Excitatory and Inhibitory Spinal V2 Interneurons Kaumudi Joshi, Seunghee Lee, Bora Lee, Jae W. Lee, and Soo-Kyung Lee Supplemental
Neuron, Volume 61 Supplemental Data LMO4 Controls the Balance between Excitatory and Inhibitory Spinal V2 Interneurons Kaumudi Joshi, Seunghee Lee, Bora Lee, Jae W. Lee, and Soo-Kyung Lee Supplemental
ONE-HOUR Western TM Multiplex Kit II
 ONE-HOUR Western TM Multiplex Kit II Technical Manual No. 0256 Version 06192009 I Description... 1 II Kit Contents.. 2 III Related Products 2 IV Key Features. 2 V Storage... 2 VI ONE-HOUR Multiplex Western
ONE-HOUR Western TM Multiplex Kit II Technical Manual No. 0256 Version 06192009 I Description... 1 II Kit Contents.. 2 III Related Products 2 IV Key Features. 2 V Storage... 2 VI ONE-HOUR Multiplex Western
Cross Linking Immunoprecipitation
 301PR 03 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Cross Linking Immunoprecipitation Utilizes Protein A/G Agarose& DSS for Antibody
301PR 03 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Cross Linking Immunoprecipitation Utilizes Protein A/G Agarose& DSS for Antibody
CRE/CREB Reporter Assay Kit camp/pka Cell Signaling Pathway Catalog #: 60611
 Data Sheet CRE/CREB Reporter Assay Kit camp/pka Cell Signaling Pathway Catalog #: 60611 Background The main role of the camp response element, or CRE, is mediating the effects of Protein Kinase A (PKA)
Data Sheet CRE/CREB Reporter Assay Kit camp/pka Cell Signaling Pathway Catalog #: 60611 Background The main role of the camp response element, or CRE, is mediating the effects of Protein Kinase A (PKA)
Strep-Spin Protein Miniprep Kit Catalog No. P2004 & P2005
 INSTRUCTION MANUAL Strep-Spin Protein Miniprep Kit Catalog No. P2004 & P2005 Highlights Fast & Simple: Purify Strep-tagged proteins from cell-free extracts using a spin-column in 7 minutes High-Quality:
INSTRUCTION MANUAL Strep-Spin Protein Miniprep Kit Catalog No. P2004 & P2005 Highlights Fast & Simple: Purify Strep-tagged proteins from cell-free extracts using a spin-column in 7 minutes High-Quality:
3. Results. 3.1 Generation of HEK293 cell clones stably expressing ETA and ETB receptors
 3. Results 3.1 Generation of HEK293 cell clones stably expressing ETA and ETB receptors To investigate the dimerisation of the endothelin receptor subtypes HEK293 cells were stably transfected with plasmids
3. Results 3.1 Generation of HEK293 cell clones stably expressing ETA and ETB receptors To investigate the dimerisation of the endothelin receptor subtypes HEK293 cells were stably transfected with plasmids
Accelerating Scientific Discovery
 Elite TM Fluorescent Membrane Potential Dye Kit CATALOG NUMBER: CA-M165 Description camp is a key second messenger involved extensively in cellular signal transduction pathways associated with the majority
Elite TM Fluorescent Membrane Potential Dye Kit CATALOG NUMBER: CA-M165 Description camp is a key second messenger involved extensively in cellular signal transduction pathways associated with the majority
Anti-p62 C-terminal pab
 Page 1 For Research Use Only. Not for use in diagnostic procedures. Anti-p62 C-terminal pab CODE No. CLONALITY Polyclonal ISOTYPE Guinea pig Ig, affinity purified QUANTITY 100 µl SOURCE IMMUNOGEN FORMURATION
Page 1 For Research Use Only. Not for use in diagnostic procedures. Anti-p62 C-terminal pab CODE No. CLONALITY Polyclonal ISOTYPE Guinea pig Ig, affinity purified QUANTITY 100 µl SOURCE IMMUNOGEN FORMURATION
Ral Activation Assay Kit
 Product Manual Ral Activation Assay Kit Catalog Number STA-408 20 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Small GTP-binding proteins (or GTPases) are a family of
Product Manual Ral Activation Assay Kit Catalog Number STA-408 20 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Small GTP-binding proteins (or GTPases) are a family of
Supporting Information
 Supporting Information Drugs Modulate Interactions Between the First Nucleotide-Binding Domain and the Fourth Cytoplasmic Loop of Human P-glycoprotein Tip W. Loo and David M. Clarke Department of Medicine
Supporting Information Drugs Modulate Interactions Between the First Nucleotide-Binding Domain and the Fourth Cytoplasmic Loop of Human P-glycoprotein Tip W. Loo and David M. Clarke Department of Medicine
SUPPLEMENTARY INFORMATION
 The Supplementary Information (SI) Methods Cell culture and transfections H1299, U2OS, 293, HeLa cells were maintained in DMEM medium supplemented with 10% fetal bovine serum. H1299 and 293 cells were
The Supplementary Information (SI) Methods Cell culture and transfections H1299, U2OS, 293, HeLa cells were maintained in DMEM medium supplemented with 10% fetal bovine serum. H1299 and 293 cells were
Glutathione Agarose Resin User s Guide
 Glutathione Agarose Resin User s Guide DESCRIPTION Glutathione Agarose Resin is used to purify recombinant derivatives of glutathione S-transferases or glutathione binding proteins. Resins are products
Glutathione Agarose Resin User s Guide DESCRIPTION Glutathione Agarose Resin is used to purify recombinant derivatives of glutathione S-transferases or glutathione binding proteins. Resins are products
Transcriptional regulation of BRCA1 expression by a metabolic switch: Di, Fernandez, De Siervi, Longo, and Gardner. H3K4Me3
 ChIP H3K4Me3 enrichment.25.2.15.1.5 H3K4Me3 H3K4Me3 ctrl H3K4Me3 + E2 NS + E2 1. kb kb +82 kb Figure S1. Estrogen promotes entry of MCF-7 into the cell cycle but does not significantly change activation-associated
ChIP H3K4Me3 enrichment.25.2.15.1.5 H3K4Me3 H3K4Me3 ctrl H3K4Me3 + E2 NS + E2 1. kb kb +82 kb Figure S1. Estrogen promotes entry of MCF-7 into the cell cycle but does not significantly change activation-associated
Checkpoint Kinase Activity Immunoblot Kit
 Product Manual Checkpoint Kinase Activity Immunoblot Kit Catalog Number STA- 413 20 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cdc25C is a protein phosphatase responsible
Product Manual Checkpoint Kinase Activity Immunoblot Kit Catalog Number STA- 413 20 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cdc25C is a protein phosphatase responsible
Supplementary information to accompany: A novel role for the DNA repair gene Rad51 in Netrin-1 signalling
 Supplementary information to accompany: A novel role for the DNA repair gene Rad51 in Netrin-1 signalling Glendining KA 1, Markie D 2, Gardner RJM 4, Franz EA 3, Robertson SP 4, Jasoni CL 1 Supplementary
Supplementary information to accompany: A novel role for the DNA repair gene Rad51 in Netrin-1 signalling Glendining KA 1, Markie D 2, Gardner RJM 4, Franz EA 3, Robertson SP 4, Jasoni CL 1 Supplementary
OPPF-UK Standard Protocols: Mammalian Expression
 OPPF-UK Standard Protocols: Mammalian Expression Joanne Nettleship joanne@strubi.ox.ac.uk Table of Contents 1. Materials... 3 2. Cell Maintenance... 4 3. 24-Well Transient Expression Screen... 5 4. DNA
OPPF-UK Standard Protocols: Mammalian Expression Joanne Nettleship joanne@strubi.ox.ac.uk Table of Contents 1. Materials... 3 2. Cell Maintenance... 4 3. 24-Well Transient Expression Screen... 5 4. DNA
Supplementary Figure S1 Purification of deubiquitinases HEK293 cells were transfected with the indicated DUB-expressing plasmids.
 Supplementary Figure S1 Purification of deubiquitinases HEK293 cells were transfected with the indicated DUB-expressing plasmids. The cells were harvested 72 h after transfection. FLAG-tagged deubiquitinases
Supplementary Figure S1 Purification of deubiquitinases HEK293 cells were transfected with the indicated DUB-expressing plasmids. The cells were harvested 72 h after transfection. FLAG-tagged deubiquitinases
pt7ht vector and over-expressed in E. coli as inclusion bodies. Cells were lysed in 6 M
 Supplementary Methods MIG6 production, purification, inhibition, and kinase assays MIG6 segment 1 (30mer, residues 334 364) peptide was synthesized using standard solid-phase peptide synthesis as described
Supplementary Methods MIG6 production, purification, inhibition, and kinase assays MIG6 segment 1 (30mer, residues 334 364) peptide was synthesized using standard solid-phase peptide synthesis as described
WesternMAX Alkaline Phosphatase Chemiluminescent Detection Kits
 WesternMAX Alkaline Phosphatase Chemiluminescent Detection Kits Code N221-KIT N220-KIT Description WesternMAX Chemiluminescent AP Kit, Anti-Mouse Includes: Alkaline Phosphatase (AP) Conjugated Anti-Mouse
WesternMAX Alkaline Phosphatase Chemiluminescent Detection Kits Code N221-KIT N220-KIT Description WesternMAX Chemiluminescent AP Kit, Anti-Mouse Includes: Alkaline Phosphatase (AP) Conjugated Anti-Mouse
Protocol Immunprecipitation
 1 Protocol Immunprecipitation IP Juni 08, S.L. Before starting an IP on should be certain, that one can clearly detect the Protein to precipitate in a standard Western Blot with the lysis conditions used
1 Protocol Immunprecipitation IP Juni 08, S.L. Before starting an IP on should be certain, that one can clearly detect the Protein to precipitate in a standard Western Blot with the lysis conditions used
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Nakajima and Tanoue, http://www.jcb.org/cgi/content/full/jcb.201104118/dc1 Figure S1. DLD-1 cells exhibit the characteristic morphology
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Nakajima and Tanoue, http://www.jcb.org/cgi/content/full/jcb.201104118/dc1 Figure S1. DLD-1 cells exhibit the characteristic morphology
Supplemental Information
 Supplemental Information Intrinsic protein-protein interaction mediated and chaperonin assisted sequential assembly of a stable Bardet Biedl syndome protein complex, the BBSome * Qihong Zhang 1#, Dahai
Supplemental Information Intrinsic protein-protein interaction mediated and chaperonin assisted sequential assembly of a stable Bardet Biedl syndome protein complex, the BBSome * Qihong Zhang 1#, Dahai
SensoLyte Anti-alpha-Synuclein Quantitative ELISA Kit (Rat) *Colorimetric*
 SensoLyte Anti-alpha-Synuclein Quantitative ELISA Kit (Rat) *Colorimetric* Revision number: 1.3 Last updated: 1/15/18 Catalog # AS-55550-R Kit Size One 96-well strip plate This kit is optimized to detect
SensoLyte Anti-alpha-Synuclein Quantitative ELISA Kit (Rat) *Colorimetric* Revision number: 1.3 Last updated: 1/15/18 Catalog # AS-55550-R Kit Size One 96-well strip plate This kit is optimized to detect
Single cell imaging of Bruton's Tyrosine Kinase using an irreversible inhibitor
 SUPPLEMENTARY INFORMATION Single cell imaging of Bruton's Tyrosine Kinase using an irreversible inhibitor Anna Turetsky 1,a, Eunha Kim 1,a, Rainer H. Kohler 1, Miles A. Miller 1, Ralph Weissleder 1,2,
SUPPLEMENTARY INFORMATION Single cell imaging of Bruton's Tyrosine Kinase using an irreversible inhibitor Anna Turetsky 1,a, Eunha Kim 1,a, Rainer H. Kohler 1, Miles A. Miller 1, Ralph Weissleder 1,2,
RNA oligonucleotides and 2 -O-methylated oligonucleotides were synthesized by. 5 AGACACAAACACCAUUGUCACACUCCACAGC; Rand-2 OMe,
 Materials and methods Oligonucleotides and DNA constructs RNA oligonucleotides and 2 -O-methylated oligonucleotides were synthesized by Dharmacon Inc. (Lafayette, CO). The sequences were: 122-2 OMe, 5
Materials and methods Oligonucleotides and DNA constructs RNA oligonucleotides and 2 -O-methylated oligonucleotides were synthesized by Dharmacon Inc. (Lafayette, CO). The sequences were: 122-2 OMe, 5
1. QUANTITY OF LYSATE 2. LYSIS BUFFER
 SAMPLE PREPARATION 1. QUANTITY OF LYSATE The amount of protein requested for the Kinex KAM-880 Antibody Microarray service is 100 µg per sample at an approximate concentration of 2 mg/ml. If your samples
SAMPLE PREPARATION 1. QUANTITY OF LYSATE The amount of protein requested for the Kinex KAM-880 Antibody Microarray service is 100 µg per sample at an approximate concentration of 2 mg/ml. If your samples
Supplementary Material
 Supplementary Material Supplementary Methods Cell synchronization. For synchronized cell growth, thymidine was added to 30% confluent U2OS cells to a final concentration of 2.5mM. Cells were incubated
Supplementary Material Supplementary Methods Cell synchronization. For synchronized cell growth, thymidine was added to 30% confluent U2OS cells to a final concentration of 2.5mM. Cells were incubated
Transfection of CRISPR/Cas9 Nuclease NLS ribonucleoprotein (RNP) into adherent mammalian cells using Lipofectamine RNAiMAX
 Transfection of CRISPR/Cas9 Nuclease NLS ribonucleoprotein (RNP) into adherent mammalian cells using Lipofectamine RNAiMAX INTRODUCTION The CRISPR/Cas genome editing system consists of a single guide RNA
Transfection of CRISPR/Cas9 Nuclease NLS ribonucleoprotein (RNP) into adherent mammalian cells using Lipofectamine RNAiMAX INTRODUCTION The CRISPR/Cas genome editing system consists of a single guide RNA
SUPPLEMENTAL MATERIAL. Supplemental Methods:
 SUPPLEMENTAL MATERIAL Supplemental Methods: Immunoprecipitation- As we described but with some modifications [22]. As part of another ongoing project, lysate from human umbilical vein endothelial cells
SUPPLEMENTAL MATERIAL Supplemental Methods: Immunoprecipitation- As we described but with some modifications [22]. As part of another ongoing project, lysate from human umbilical vein endothelial cells
Minicollagen cysteine-rich domains encode distinct modes of. Anja Tursch, Davide Mercadante, Jutta Tennigkeit, Frauke Gräter and Suat
 Supplementary Figure S1. Conformational dynamics of partially reduced CRDs. (A) Root mean square deviation (RMSD) and (B) radius of gyration (R G ) distributions for the N-CRD (green) and C-CRD (red) domains,
Supplementary Figure S1. Conformational dynamics of partially reduced CRDs. (A) Root mean square deviation (RMSD) and (B) radius of gyration (R G ) distributions for the N-CRD (green) and C-CRD (red) domains,
Nature Medicine doi: /nm.3554
 SUPPLEMENTARY FIGURES LEGENDS Supplementary Figure 1: Generation, purification and characterization of recombinant mouse IL-35 (ril-35). High-Five insect cells expressing high levels of the bicistronic
SUPPLEMENTARY FIGURES LEGENDS Supplementary Figure 1: Generation, purification and characterization of recombinant mouse IL-35 (ril-35). High-Five insect cells expressing high levels of the bicistronic
Index 1. Product Description 2. Purification Procedure 3. Troubleshooting 4. Ordering Information
 High Affinity Ni-Charged Resin Cat. No. L00223 Technical Manual No. TM0217 Version 07132010 Index 1. Product Description 2. Purification Procedure 3. Troubleshooting 4. Ordering Information 1. Product
High Affinity Ni-Charged Resin Cat. No. L00223 Technical Manual No. TM0217 Version 07132010 Index 1. Product Description 2. Purification Procedure 3. Troubleshooting 4. Ordering Information 1. Product
Post-translational modification
 Protein expression Western blotting, is a widely used and accepted technique to detect levels of protein expression in a cell or tissue extract. This technique measures protein levels in a biological sample
Protein expression Western blotting, is a widely used and accepted technique to detect levels of protein expression in a cell or tissue extract. This technique measures protein levels in a biological sample
DuoSet IC. Human Phospho-DDR1. Catalog Number DYC Catalog Number DYC5859-5
 DuoSet IC Human Phospho-DDR1 Catalog Number DYC5859-2 Catalog Number DYC5859-5 For the development of sandwich ELISAs to measure phosphorylated human Discoidin Domain Receptor 1 (DDR1) in cell lysates.
DuoSet IC Human Phospho-DDR1 Catalog Number DYC5859-2 Catalog Number DYC5859-5 For the development of sandwich ELISAs to measure phosphorylated human Discoidin Domain Receptor 1 (DDR1) in cell lysates.
ab Ral Activation Assay Kit
 Version 1 Last updated 7 July 2016 ab212012 Ral Activation Assay Kit For the rapid, sensitive and accurate measurement of active Ral in cell or tissue lysates from human, mouse, rat, chicken or dog. This
Version 1 Last updated 7 July 2016 ab212012 Ral Activation Assay Kit For the rapid, sensitive and accurate measurement of active Ral in cell or tissue lysates from human, mouse, rat, chicken or dog. This
Supplemental Figure 1 Human REEP family of proteins can be divided into two distinct subfamilies. Residues (single letter amino acid code) identical
 Supplemental Figure Human REEP family of proteins can be divided into two distinct subfamilies. Residues (single letter amino acid code) identical in all six REEPs are highlighted in green. Additional
Supplemental Figure Human REEP family of proteins can be divided into two distinct subfamilies. Residues (single letter amino acid code) identical in all six REEPs are highlighted in green. Additional
GST Fusion Protein Purification Kit
 Glutathione Resin GST Fusion Protein Purification Kit Cat. No. L00206 Cat. No. L00207 Technical Manual No. TM0185 Version 01042012 Index 1. Product Description 2. Related Products 3. Purification Procedure
Glutathione Resin GST Fusion Protein Purification Kit Cat. No. L00206 Cat. No. L00207 Technical Manual No. TM0185 Version 01042012 Index 1. Product Description 2. Related Products 3. Purification Procedure
Supplementary information
 Supplementary information Table of Content: Supplementary Results... 2 Supplementary Figure S1: Experimental validation of AP-MS results by coimmunprecipitation Western blot analysis.... 3 Supplementary
Supplementary information Table of Content: Supplementary Results... 2 Supplementary Figure S1: Experimental validation of AP-MS results by coimmunprecipitation Western blot analysis.... 3 Supplementary
FIVEphoton Biochemicals
 Human Cyclophilin B (CYPB) ELISA Kit Protocol Protocol for other species is identical except for dilutions of species specific standard. Use the protocol shipped with the kit for your experiment. FIVEphoton
Human Cyclophilin B (CYPB) ELISA Kit Protocol Protocol for other species is identical except for dilutions of species specific standard. Use the protocol shipped with the kit for your experiment. FIVEphoton
No cgmp Maximum HRP activity. Increasing cgmp Decreasing HRP activity. Product Insert. Product # R8065 Quantity: 768, 60 µl reactions
 Product Insert CatchPoint Cyclic-GMP Fluorescent Assay Kit Product # R8065 Quantity: 768, 60 µl reactions Introduction About the cgmp Assay The CatchPoint cyclic-gmp Fluorescent Assay Kit measures levels
Product Insert CatchPoint Cyclic-GMP Fluorescent Assay Kit Product # R8065 Quantity: 768, 60 µl reactions Introduction About the cgmp Assay The CatchPoint cyclic-gmp Fluorescent Assay Kit measures levels
AFFINITY GST PURIFICATION
 DESCRIPTION Glutathione Agarose Resin is used to purify recombinant derivatives of glutathione S-transferases or glutathione binding proteins. are products that allow batch or column purifications. Purification
DESCRIPTION Glutathione Agarose Resin is used to purify recombinant derivatives of glutathione S-transferases or glutathione binding proteins. are products that allow batch or column purifications. Purification
10X ACTOne Membrane Potential Dye Solution, 10 ml each bottle, 10 bottles 10X ACTOne Dye Dilution Buffer, 100 ml
 Codex Technical Data Sheet Codex ACTOne TM Membrane Potential Dye Bulk Kit Product Information Catalog Number: Components: CB-80500-211 10X ACTOne Membrane Potential Dye Solution, 10 ml each bottle, 10
Codex Technical Data Sheet Codex ACTOne TM Membrane Potential Dye Bulk Kit Product Information Catalog Number: Components: CB-80500-211 10X ACTOne Membrane Potential Dye Solution, 10 ml each bottle, 10
with pentaglycine (GGGGG) peptide motifs. Reaction conditions: (1) hexamethylene
 Supplementary Figure 1. Sequential surface reaction scheme to modify polyurethane catheters with pentaglycine (GGGGG) peptide motifs. Reaction conditions: (1) hexamethylene diisocyanate/triethylamine;
Supplementary Figure 1. Sequential surface reaction scheme to modify polyurethane catheters with pentaglycine (GGGGG) peptide motifs. Reaction conditions: (1) hexamethylene diisocyanate/triethylamine;
Western blotting technique: principle, procedure and application
 Western blotting technique: principle, procedure and application The term blotting refers to the transfer of biological samples from a gel to a membrane and their subsequent detection on the surface of
Western blotting technique: principle, procedure and application The term blotting refers to the transfer of biological samples from a gel to a membrane and their subsequent detection on the surface of
Many G protein-coupled receptors (GPCRs) have been shown
 A heterodimer-selective agonist shows in vivo relevance of G protein-coupled receptor dimers Maria Waldhoer*, Jamie Fong*, Robert M. Jones, Mary M. Lunzer, Shiv K. Sharma, Evi Kostenis, Philip S. Portoghese,
A heterodimer-selective agonist shows in vivo relevance of G protein-coupled receptor dimers Maria Waldhoer*, Jamie Fong*, Robert M. Jones, Mary M. Lunzer, Shiv K. Sharma, Evi Kostenis, Philip S. Portoghese,
Supplementary Figure 1 - Characterization of rbag3 binding on macrophages cell surface.
 Supplementary Figure 1 - Characterization of rbag3 binding on macrophages cell surface. (a) Human PDAC cell lines were treated as indicated in Figure 1 panel F. Cells were analyzed for FITC-rBAG3 binding
Supplementary Figure 1 - Characterization of rbag3 binding on macrophages cell surface. (a) Human PDAC cell lines were treated as indicated in Figure 1 panel F. Cells were analyzed for FITC-rBAG3 binding
AFFINITY GST PURIFICATION
 DESCRIPTION Glutathione Agarose Resin is used to purify recombinant derivatives of glutathione S-transferases or glutathione binding proteins. are products that allow batch or column purifications. Purification
DESCRIPTION Glutathione Agarose Resin is used to purify recombinant derivatives of glutathione S-transferases or glutathione binding proteins. are products that allow batch or column purifications. Purification
Supporting Information
 Supporting Information He et al. 10.1073/pnas.1116302108 SI Methods Cell Culture. Mouse J774A.1 and RAW 264.7 macrophages were obtained from ATCC and were cultured in MEM supplemented with 10% FS (Sigma)
Supporting Information He et al. 10.1073/pnas.1116302108 SI Methods Cell Culture. Mouse J774A.1 and RAW 264.7 macrophages were obtained from ATCC and were cultured in MEM supplemented with 10% FS (Sigma)
SUMOylated protein capture kit
 SUMOylated protein capture kit Cat no. A010-100 Capture and detect SUMOylated proteins High capacity, high specificity SUMO binding matrix Fast, convenient protein isolation using purification system provided
SUMOylated protein capture kit Cat no. A010-100 Capture and detect SUMOylated proteins High capacity, high specificity SUMO binding matrix Fast, convenient protein isolation using purification system provided
A sensitive direct human telomerase activity assay Scott B Cohen & Roger R Reddel
 A sensitive direct human telomerase activity assay Scott B Cohen & Roger R Reddel Supplementary figure and text: Supplementary Figure 1 Titration of the sheep polyclonal htert antibody. Supplementary Methods
A sensitive direct human telomerase activity assay Scott B Cohen & Roger R Reddel Supplementary figure and text: Supplementary Figure 1 Titration of the sheep polyclonal htert antibody. Supplementary Methods
Functional GPCR Studies Using AlphaScreen camp Detection Kit
 PPLICTION NOTE lpha Technology uthors: Jen Carlstrom Dawn Nida PerkinElmer, Inc. Hopkinton, M Functional GPCR Studies Using lphascreen cmp Detection Kit Introduction G protein-coupled receptors (GPCRs)
PPLICTION NOTE lpha Technology uthors: Jen Carlstrom Dawn Nida PerkinElmer, Inc. Hopkinton, M Functional GPCR Studies Using lphascreen cmp Detection Kit Introduction G protein-coupled receptors (GPCRs)
Chapter One. Construction of a Fluorescent α5 Subunit. Elucidation of the unique contribution of the α5 subunit is complicated by several factors
 4 Chapter One Construction of a Fluorescent α5 Subunit The significance of the α5 containing nachr receptor (α5* receptor) has been a challenging question for researchers since its characterization by
4 Chapter One Construction of a Fluorescent α5 Subunit The significance of the α5 containing nachr receptor (α5* receptor) has been a challenging question for researchers since its characterization by
Orexin A (HUMAN, MOUSE, RAT, PORCINE, OVINE,
 Orexin A (HUMAN, MOUSE, RAT, PORCINE, OVINE, BOVINE) Western Blot Kit Protocol (Catalog #WBK-003-30) PHOENIX PHARMACEUTICALS, INC. TABLE OF CONTENTS 1. Kit Contents...2 2. Storage...2 3. Introduction...3
Orexin A (HUMAN, MOUSE, RAT, PORCINE, OVINE, BOVINE) Western Blot Kit Protocol (Catalog #WBK-003-30) PHOENIX PHARMACEUTICALS, INC. TABLE OF CONTENTS 1. Kit Contents...2 2. Storage...2 3. Introduction...3
Data Sheet CD137/NF-κB Reporter - HEK293 Recombinant Cell Line Catalog # 79289
 Data Sheet CD137/NF-κB Reporter - HEK293 Recombinant Cell Line Catalog # 79289 Background Human CD137 (4-1BB; TNFRS9) is an inducible co-stimulatory molecule that activates T cells. CD137:CD137L-mediated
Data Sheet CD137/NF-κB Reporter - HEK293 Recombinant Cell Line Catalog # 79289 Background Human CD137 (4-1BB; TNFRS9) is an inducible co-stimulatory molecule that activates T cells. CD137:CD137L-mediated
