Common Enterobacterial Antigen
|
|
|
- April Caldwell
- 5 years ago
- Views:
Transcription
1 INFECTION AND IMMUNITY, Mar. 1973, p Vol. 7, No. 3 Copyright 1973 American Society for Microbiology Printed in U.S.A. Common Enterobacterial Antigen II. Effect of Immunization on Challenge with Heterologous Bacilli WILLIAM R. McCABE AND ANNA GREELY Departments of Medicine and Microbiology, University Hospital, Boston University School of Medicine, Boston, Massachusetts Rteceived for publication 8 September 1972 Studies were carried out evaluating the protective activity of immunization with common enterobacterial antigen (CA) against challenge with heterologous gram-negative bacilli. Active immunization of mice with Escherichia coli 0:14 elicited titers of antibody to CA of 1: 640 or greater but completely failed to enhance resistance to challenge by mouse virulent strains of Klebsiella pneumoniae or E. coli. Similarly, two lots, 324 and 422, of rabbit antisera to CA failed to afford passive protection to mice challenged with K. pneumoniae or E. coli. A third lot, 166, of rabbit antisera to CA did passively protect mice. The protective activity of antisera 166 was demonstrated to reflect its content of antibody to another crossreactive antigen, Re determinant, of gram-negative bacilli which has previously been shown to protect against infections with heterologous bacilli rather than any protective effect of antibody to CA. These studies failed to demonstrate any protective activity of antibody to CA against challenge with heterologous CA containiing gram-negative bacilli. Most studies of immunity to infection have been concerned primarily with type-specific immune mechanisms. Little attention has been directed toward investigation of any potential protective effects mediated by antibodies to shared, cross-reactive antigens. Historically, cross-reactive antigens have proven important in the development of practical immunizing procedures as evidenced by Jenner's classical demonstration that cowpox protected against small-pox infection. More recently, prior infection with influenza A has been shown to protect mice against infection with influenza A2 (4, 13). Similarly, studies by one of the authors have demonstrated that both active and passive immunization with a rough mutant, Re 595, of Salmonella minnesota afforded significant protection to mice against challenge with heterologous smooth gram-negative bacilli (9). The chemical similarity of the core region of the lipopolysaccharides of most gram-negative bacilli results in their sharing cross-reactive antigens (8). One of the cross-reactive antigens shared by Enterobacteriaceae is the common enterobacterial antigen (CA) first described by Kunin (5-7). These studies demonstrated that, although most enterobacteria contain CA, only a few straiins of Escherichia coli (0:14, 0:56, 0:124, and 0:144) were capable of inducing an antibody response to CA. Although other investigators have elicited an antibody response to CA using semipurified ethanol-soluble fractions from other gram-negative bacilli (14), highly purified CA prepared by both Kunin and in this laboratory have not proven to be immunogenic (5; M. Johns et al., J. Immunol., in press). Antibodies to CA have been demonstrated to opsonize heterologous gramnegative bacilli for phagocytosis (1). In addition, equivocal prolongation of survival of Salmonellachallenged mice after immunzation with CA has suggested that antibody to CA might have protective activity in experimental animals (3). These observations, plus previous demonstrations by the author of protective activity of antibody to other cross-reactive antigens (determinants of Re mutants) of gram-negative bacilli in both experimental animals and man (9, 11), prompted the present studies to determine whether immunization with CA protected experimental animals against challenge with heterologous gramnegative bacilli. MATERIALS AND METHODS Immunizing agents. E. coli 0:14 was supplied through the courtesy of Calvin Kunin. An additional strain of E. coli 0:14 and E. coli 0:56, 0:124, and 0:144 were provided from the Center for Disease Control by John Winn. Immunization of mice or rabbits with these strains of E. coli induces an antibody response to CA as well as the 0-specific antigen of each strain. 386
2 VOL. 7, 1973 IMMUNIZATION WITH CA 387 A strain of Pseudomonas aeruginosa isolated in the Bacteriology Laboratory, University Hospital, from a blood culture was used to provide a nonspecific endotoxin stimulus for control animals as previously described (9). Immunization of mice and rabbits with this strain of P. aeruginosa, which does not contain CA, did not elicit an antibody response to CA or the Re determinant of S. minnesota. Infecting agents. Two species of mouse virulent gram-negative bacilli were used as challenge agents. The Caroli strain of K. pneumoniae type II was obtained from Louis Chedid of the Pasteur Institute. The second challenge strain, a smooth, nontypable E. coli 107, had been isolated from a blood culture of a patient with bacteremia. The characteristics of the infections produced by these bacteria are described in an earlier publication (9). Both of these challenge strains were shown to contain CA (see below). Immunization of mice. Mice were immunized with E. coli 0:14, 0:56, 0:124, and 0:144 to induce antibodies to CA. Single-colony isolates of each strain were inoculated into 10 ml of brain heart infusion (BHI) broth. After overnight incubation at 37 C, this was spread onto BHI agar plates to prepare a lawn. These plates were incubated overnight and the bacterial growth was harvested in 10 ml of 0.9% saline. The bacteria were washed three times in 0.9% saline, resuspended in 10 ml of saline, and boiled for 2.5 h. This antigen preparation was diluted 1:10 with 0.9% saline before immunization (42 X 108 bacteria/ml). Twenty- to 22-g male, CF1 mice were injected intraperitoneally with 0.2 ml of the E. coli 0:14, 0:56, 0:124, and 0:144 antigen suspensions for 3 consecutive days of each week for 2 weeks and then once weekly for 2 weeks. Five days later, lots of 10 mice were bled from the retro-orbital plexus, the blood was pooled, the serum was separated, and antibody titers were determined by indirect hemagglutination. If antibody titers to CA were less than 1:640, weekly injections of antigen were continued until titers of antibody to CA of 1:640 or greater were attained. After final injection of antigen, at least 1 week was allowed to elapse before challenge to minimize any nonspecific protective effect of endotoxin. Immunization of rabbits. Antisera to CA was prepared by immunization of rabbits with E. coli 0:14, 0:56, 0:124, and 0:144 using methods similar to those described for the preparation of antigens for immunization of mice. The bacterial growth was harvested in 10 ml of 0.9% saline, washed three times in saline, resuspended in 10 ml of saline, and killed by the addition of 0.5% Formalin. This material was resuspended in saline to 80%o transmission at 550 nm in a Coleman spectrophotometer prior to rabbit immunization. Groups of three rabbits were immunized by intravenous injection of 0.5, 1.0, 2.0, 4.0, and 4.0 ml at 4- to 5-day intervals. Animals were bled for antibody determination 7 days after the last injection of antigen. If satisfactory titers of antibody had been attained, animals were anesthetized and exsanguinated. After separation of serum, Merthiolate was added to a concentration of 1:10,000 as a preservative, and the serum was stored at -70 C until used. Hemagglutination reaction for CA. Titrations of antibody to CA were performed by modification of the indirect hemagglutination technique of Neter (12) using erythrocytes coated with extracts of a bacillus previously shown to contain large quantities of CA, Proteus rettgeri 6572 (11). P. rettgeri 6572 was grown overnight at 37 C oni BHI agar and harvested in 10 ml of saline. The bacterial suspeusion was boiled for 2.5 h and centrifuged at 6,000 rpm at 4 C, and the supernatant was removed. One milliliter of 95% ethyl alcohol was added to the supernatant. A 1:10 dilution of this crude antigen preparation was incubated with an equal volume of a 2.5% suspeusion of human group O erythrocytes in phosphate-buffered saline (PBS) for 1 h at 37 C in a shaker water bath. The sensitized erythrocytes were washed three times in PBS and resuspended at a 0.5% concentration for immediate use in the hemagglutination test. Serum was serially diluted in PBS in test tubes (13 by 100 mm) at dilutions ranging from 1:10 to 1:163,840, and an equal volume of sensitized erythrocytes was added. Tubes were incubated for 1 h at 37 C, refrigerated overnight at 4 C, and read by pattern and for agglutination after shaking. Hemmagglutination inhibition. Hemagglutination-inhibition studies were performed using the methods described for indirect hemagglutination except that 0.2 ml of boiled extracts of Enterobacter aerogenee, another CA containing strain (11), was added to one series of antiserum dilutions while 0.2 ml of PBS was added to a duplicate set as controls. Antiserum and the inhibiting antigen extract were incubated for 1 h at 37 C prior to addition of sensitized erythrocyes. Antibody titers to CA were considered to be those titers in serum from mice or rabbits immunized with E. coli 0:14 which agglutinated erythrocytes coated with extracts of P. rettgeri and which was completely blocked by extracts of E. aerogenes. Confirmation that hemagglutination inhibition with E. aerogenes accurately reflected inhibition of antibodies to CA was obtained by demonstration of identical results when highly purified CA (M. Johns, et al., J. Immunol., in press) was used for inhibition. CA content of challenge strains. Content of CA of the two challenge strains, K. pneumoniae type II and E. coli 107, was determined by hemagglutination inhibition as described above. The maximal dilution of each organism capable of inhibiting agglutination of erythrocytes coated by P. rettgeri 6572 by 4 to 8 hemagglutinating units of E. coli 0:14 antiserum was considered to represent their CA content and was reported as hemagglutination-inhibiting units. 0-specific and Re antibody. Antibody titers to both challenge strains were determined in serum from immunized mice and rabbits by indirect
3 388 McCABE AND GREELY I NFECT. IMMUNITY hemagglutination of erythrocytes coated with extracts of each organism. Prior inhibition, with E. aerogenes extracts, was carried out as described above to distinguish type-specific antibody from antibody to CA. In addition, since previous investigations have demonstrated that antibodies to the determitlants of Re mutants protect against heterologous gram-negative bacilli, antibody titers to Re were measured in the serum of all immuniized animals by previously described techniques (9). Mercaptoethanol treatment. Treatment with 2-mercaptoethanol was used to distinguish 19s from 7s antibody to CA. A 0.05-ml amount of 1 M 2-mercaptoethanol was added to serial dilutions of serum and incubated at 37 C for 1 h. Sensitized erythrocytes were then added and incubated for 1 h. Concomitant titrations of each serum specimen, untreated with 2-mercaptoethanol, were run for comparison. Adsorption with Re determinants and CA. Adsorption of rabbit antiserum to CA was carried out using the Re 595 mutant of S. minnesota. Bacteria were grown overnight in meat extract broth, removed by centrifugation, washed three times in saline, and killed and dried with acetone. To prevent release of endotoxin and toxicity of the Readsorbed serum, dried bacteria were hydrolyzed in 1% acetic acid at 100 C for 2 h prior to adsorption of CA antiserum. Preliminary investigation revealed that 1 mg of purified CA per ml would remove all the CA antibody from antiserum 166. Six milliliters of 166 antiserum was added to 6 mg of CA dissolved in 0.2 ml of PBS. The mixture was held at room temperature for 1 h and then at 4 C for 3 days with daily shaking to resuspend the precipitate. The precipitate was then removed by centrifugation and the supernatant was tested for CA antibody content prior to injection into mice. Experimental infections. Mice were infected by intravenous injection of the challenge organism into the tail vein. For most experiments, animals were challenged with 100 mean lethal dose units (LDw) of either infecting organism. Maintenance of virulence of the two challenge strains and the characteristics of the experimental infections have been described previously (9). RESULTS Active immunization. Interperitoneal immunization with E. coli 0:14 induced rises in antibody titers to CA from less than 1:20 to 1:640 or greater in mice. In contrast, immunization with E. coli 0:56, 0:124, and 0:144 failed to induce antibody titers to CA in excess of 1:80 to 1:160 even when protracted courses of immunization were given, and studies of animals immunized with these strains were not pursued further. Antibody titers to CA showed only a slight increase ( -1 :80) within the first 3 weeks after immunization with E. coli 0:14. Titers reached levels of 1:320 to 1:640 during the 4th week and 1:1,280 to 1:2,560 in the 5th week of immunization. Mouse antibody to CA appeared to be composed of approximately equal portions of 7s and 19s antibody since treatment with 2-mercaptoethanol consistently produced only a 1-tube (twofold) reduction in antibody titer to CA. Studies of serum from mice with CA titers of 1: 640 or greater after immunization failed to demonstrate any rise in 0-specific antibody to either of the challenge strains or the determinants of Re mutants. Once titers of 1: 640 or greater to CA had been achieved, at least 1 week was allowed to elapse after the last injection of antigen before the immunized mnice and control animals were challenged with 100 LD10 units of K. pneumoniae (106) or E. coli 107 (109) to obviate any nonspecific protective effects of endotoxin. The results of these studies are illustrated in Table 1. Since no significant differences in fatality rates were observed between controls injected with saline or P. aeruginosa, both groups of controls were combined for analysis. Survival rates in animals immunized with CA prior to challenge with K pneumoniae were 23%, whereas 30% of control animals survived. Slightly higher survival rates (49%) were observed in CA-immunized mice than in controls challenged with E. coli 107 (31%), but this difference was not statistically significant TABLE 1. Effect of immunization with common antigen (Escherichia coli 0:14) on intravenous challenge with Klebsiella pneumoniae or E. coli 107 a Common antigen. b Number of survivors per number of animals challenged (percent survivors).
4 VOI,. 7, 1973 (X2 = 3.6; P > 0.05). Additional analysis also failed to demonstrate any relation between antibody titers and survival rates in mice with titers to CA ranging from 1:640 to 1:5,120. Animals with higher titers of antibody to CA did not have greater survival rates after challenge with either K. pneumoniae or E. coli than animals with lower titers. Since the dose of the two challenge strains (100 LD,o) which were used, represented a relatively stringent challenge, additional studies were undertaken to determine whether immunization with CA induced lesser degrees of protection. Table 2 demonstrates the number of bacilli of each of the challenge strains required to produce and LDo in controls, mice immunized with CA, and animals who had received type-specific immunization. The LD,o of K. pneumoniae in control animals was 2.6 X 10' bacilli which was identical with the number of bacilli (2.6 X 10') required to produce an LD,o in CA-immunized mice. In contrast, the number of K. pneumoniae required to produce an LD5o was increased by 1,000-fold in mice who were immunized against the challenge strain. Immunization with CA failed to increase the LD50 (1.3 X 104) in comparison with controls (1.1 X 104), whereas type-specific immunization induced at least a 10-fold increase in protection against E. coli 107. Passive immunization. The ability of passively transferred rabbit antiserum to CA to protect against challenge with. heterologous gramnegative bacilli was also evaluated. Antibody titers to CA in rabbits immunized with E. coli IMMUNIZATION WITH CA 0:56, 0:124, and 0:144 were considerably lower than those in rabbits immunized with E. coli 0:14, similar to the results observed in mice. Because of the low titers of CA in rabbits immunized with E. coli 0: 56, 0:124, and 0: 144, the protective activity of the antisera was not studied. Mice were injected intravenously with 0.3 ml of saline, normal rabbit serum, or serum from rabbits immunized with CA (E. coli 0:14) and challenged 60 minutes later by the injection of 10' K. pneumoniae or 10' E. coli 107 intravenously. The results observed with three different antisera (166, 324, 422), and the titers of antibody to CA and other antigens of these antisera are shown in Table 3. Titers of antibody to CA varied from 1:10,240 to 1:163,840 in the three sera studied. The demonstration that agglutination of erythrocytes coated with extracts of P. rettgeri 6572 was completely inhibited by extracts of an unrelated species, E. aerogenes, as well as purified CA, indicated that these were antibodies to CA. Antibody titers ranging from 1:160 to 1:5,120 to the challenge strains were also observed. However, the ability of extracts of E. aerogenes and purified CA to completely inhibit agglutination of erythrocytes sensitized with extracts of the challenge strains demonstrated that such antibody was directed against CA present in the two challenge strains rather than 0-specific antibody. Measurement of CA content in extracts of the two challenge strains revealed >1,000 hemagglutinationinhibiting units per ml of K. pneumoniae extracts and 320 units/ml of E. coli 107 extracts. One of the CA antisera, 166, also contained antibody TABLE 2. Comparison of type-specific immunization and immunization with commorn antigen (CA) Challenge strain and dose Immunizing agent Saline coi 0:14) Klebiela pneumoniae K. pneumoniae 2.0 X 106 0/10a 0/10 10/ X 10' 0/10 0/10 10/ X 104 4/10 4/10 10/ X 10' 6/10 6/10 10/ X 102 7/10 7/10 10/10 LD5ob 2.6 X 10' 2.6 X 103 >2.0 X 106 E. coli 107 (E. coli 107) 3.0 X 108 0/10 0/10 8/ X 10' 1/10 2/10 10/ X /10 10/10 10/10 LD&o 1.1 X X 107 >3.0 X 10l a Number of survivors 72 h after challenge per total number challenged. b Mean lethal dose. 389
5 390 McCABE AND GREELY INFECT. IMMUNITY TABLE 3. Effect of passive transfer of serum from rabbits immunized with common antigen (CA) on lethal Klebsiella pneumoniae and Escherschia coli infections of mice Antibody titers Determination To CA To challenge strain To Re Survivors/no. of animals Unni- Inhibited Uni Inhibited challenged.uninhib- b by E. itd ie aerogenes aerogenes (K. pneumioniae challenge) Controls. Saline 15/47 (32%) NRSb < 1: 20 < 1: 20 < 1: 20 23/69 (33%) Serum from rabbits immunized with CA 166 1:10,240 < 1:20 1:640 < 1:20 1:160 64/69 (81%) 324 1:40,960 <1:20 1:150 <1:20 <1:20 19/37 (51%) 422 1:163,840 < 1:20 1:1280 <1:20 <1:20 3/30 (10%) (E. coli challenge) Controls Saline 4/40 (10%) NRS <1:20 <1:20 4/40 (10%) Serum from rabbits immunized with CA 166 1:10,240 < 1:20 1:320 < 1:20 1:160 27/30 (90%) 324 1:40,960 < 1:20 1:160 < 1:20 < 1:20 2/40 (05%) 422 1:163,840 < 1:20 1:5,120 < 1: 20 < 1:20 0/40 (00%) a Enterobacter aerogenes. b NRS = Nonimmune rabbit serum. against determinants of Re mutants. Antibody to Re was not inhibited by extracts containing CA. In contrast to mouse antiserum to CA, rabbit antiserum to CA was composed primarily of 198 antibody. Treatment of rabbit antiserum with 2-mercaptoethanol produced a 32- to 256- fold reduction in antibody titer to CA. Studies were carried out to measure persistence of passively injected rabbit antiserum to CA. Serum obtained from four mice, 1 h after passive transfer of 0.3 ml of 324 antiserum, revealed that all had titers of antibody to CA of 1:2,560. Titers of 1:1,280 and 1:320 were present 6 h and 24 h, respectively, after passive transfer of rabbit antiserum 324. Survival rates in mice passively immunized by intravenous injection of 0.3 ml of 324 or 422 antiserum to CA and subsequently challenged with K. pneumoniae or E. coli were not significantly greater than among controls given saline or normal rabbit serum (Table 3). In contrast, survival rates in mice given antiserum 166 and challenged with both K. pneumoniae (81%) and E. coli (90%) were significantly greater (X2 = 70; P < 0.001) than in control animals (10-33%). The demonstration that only one of the three antisera to CA afforded significant protection against challenge with heterologous gram-negative bacilli and that this antiserum contained the lowest titers of antibody to CA suggested that antibody to CA might not be responsible for the protection observed. Since previous investigations have demonstrated that antibody to Re protects against challenge with heterologous bacilli and since only CA antiserum 166 contained antibodies to Re (Table 3), this possibility was evaluated further by determination of the protective activity of 166 antiserum before and after adsorption with Re. Antiserum 166 was adsorbed overnight at 4 C with 100 mg of dead Res59 mutant (previously demonstrated not to contain CA) of S. minnesota per ml of serum. Such adsorption reduced antibody titers to Re from 1:160 to less than 1:20 but did not affect antibody titers to CA. After centrifugation for removal of bacteria, 0.3 ml of adsorbed or unadsorbed 166 antiserum was injected intravenously into each animal of groups of 40 mice 1 h prior to challenge with K. pneumoniae or E. coli. The results of these studies are summarized in Table 4. Although unadsorbed CA antiserum 166 afforded significant protection against challenge
6 VoL. 7, 1973 IMMUNIZATION WITH CA 391 TABLE 4. Effect of passive transfer of rabbit antiserum to common antigen (CA; 166) before and after adsorption with Re Passive transfer of Challenge strain No. of survivors/no. of animals challenged Saline Escherichia coli 107 3/20 NRSG E. coli 107 2/20 CA antiserum 166 (unadsorbed) E. coli /201 X2 = 17; P <0.001 CA antiserum 166 (adsorbed E. coli 107 2/20' with Re) Saline Klebsiella pneumoniae 4/20 NRSa K. pneumoniae 5/20 CA antiserum 166 (unadsorbed) K. pneumoniae 14/201 X2 = 12; P <0.001 CA antiserum 166 (adsorbed K. pneumoniae 3/20 with Re) a Nonimmune rabbit serum. with both K. pneumoniae and E. coli, this protective activity was obliterated by adsorption with Re. Thus, the protective activity of CA antiserum 166 appeared to reflect its content only of antibody to Re rather than antibody to CA Ȧdditional studies were carried out with antiserum 166 after adsorption with purified CA. These studies were limited by the availability of only a small amount of 166 serum remaining after previous studies. Adsorption with purified CA reduced antibody titers from 1:10,240 to less than 1:20 but failed to inhibit its protective activity. Survival rates, 5 of 10 (50%), were signiificantly greater (X2 = 4.03; P < 0.05) in mice injected with 0.3 ml of 166 antiserum adsorbed with CA prior to challenge. In contrast, only 3 of 20 (15%) mice injected with normal rabbit serum survived challenge with E. coli 107. Thus, the protective activity of CA antiserum 166 failed to reflect its content of antibody to CA and reflected, instead, the antibody activity to another crossreacting antigen, determinants of Re mutants, of gram-negative bacilli. DISCUSSION The failure to demonstrate any protective effect of active or passive immunization with CA against lethal infections with either K. pneumoniae or E. coli contrasts with earlier studies by the authors which have shown that immunization with another cross-reactive antigen, determinants of Re mutants, afforded significant protection against challenge with these same strains. The present investigations provided additional confirmation of the protective activity of antibody to Re. Only one of the three rabbit antisera to CA, 166, protected mice against challenge with K. pneumoniae and E. coli, but this antiserum was also the only one of the three which contained antibody against Re. Adsorption of antiserum 166 with purified CA failed to remove its protective activity, whereas adsorption of the same antiserum with killed Re mutants did not affect antibody titers to CA but removed antibody reactive against Re and abolished the protective activity of this serum. This observation clearly emphasizes the problems of interpretation of protective effects observed after immunization with whole bacilli or impure antigen preparations which may produce antibodies against a variety of determinants. If prior studies had not demonstrated that the determinants of Re mutants are shared by most gram-negative bacilli and that antibody to Re protected against challenge with heterologous smooth gram-negative bacilli, the protection observed with the 166 CA antiserum might have been attributed to antibody to CA. Similarly, previous reports of possible protective activity induced by immunization with CA (2, 3) might also have reflected antibody directed against antigens other than CA. The failure to demonstrate protection against challenge with heterologous bacilli in mice actively and passively immunized with CA is consistent with observations in man. Studies of 206 episodes of gram-negative bacteremia demonstrated that bacteremia often developed despite the presence of high titers of antibody to CA and that high titers of CA did not ameliorate the severity of bacteremia. However, despite the lack of protective activity of antibody to CA in man, antibody to another shared, cross-reactive antigen (Re) did appear to be associated with lesser severity and enhanced survival in human gramnegative bacteremia analogous to observations in animals (10).
7 392 McCABE AND GREELY INFECT. IMMUNITY ACKNOWLEDGMENTS These studies were supported by U.S. Public Health Service research grants AI , AI , AI-11,116-01, and training grant 5-TO1-AI from the National Institute of Allergy and Infectious Diseases and grant 5 P01-HE from The National Heart Institute. LITERATURE CITED 1. Domingue, G. J., and E. Neter Opsonizing and bacterial activities of antibodies against common antigen of Enterobacteriaceae. J. Bacteriol. 91: Domingue, G., A. Salki, C. Rountree, and W. Little Prevention of experimental hematogenous and retrograde pyelonephritis by antibodies against enterobacterial common antigen. Infect. Immunity 2: Gorzynski, E. A., J. L. Ambrus, and E. Neter Effect of common enterobacterial antiserum on experimental Salmonella typhimurium infection of mice. Proc. Soc. Exp. Biol. Med. 137: Henle, W., and F. S. Lief The broadening of antibody spectra following multiple exposures to influenza viruses. Amer. Rev. Resp. Dis. 88: Kunin, C. M Separation, characterization and biological significance of a common antigen in Enterobacteriaceae. J. Exp. Med. 118: Kunin, C. M., and M. V. Beard Serological studies of 0 antigens of E8cherichia coli by means of the hemagglutination tests. J. Bacteriol. 85: Kunin, C. M., M. V. Beard, and N. E. Halmagyi Evidence for a common hapten associated with endotoxin fractions of E. coli and other Enterobacteriaceae. Proc. Soc. Exp. Biol. Med. 111: Ltlderitz, O., A. M. Staub, and 0. Westphal Immunochemistry of 0 and R antigens of SalmoneUa and related Enterobacteriaceae. Bacteriol. Rev. 30: McCabe, W. R Immunization with R mutants of S. minne8ota. I. Protection against challenge with heterologous gram-negative bacilli. J. Immunol. 108: McCabe, W. R., M. Johns, and T. DiGenio Common enterobacterial antigen. III. Initial titers and antibody response in bacteremia caused by gram-negative bacilli. Infect. Immunity 7: McCabe, W. R., B. E. Kreger, and M. Johns Type specific and cross-reactive antibodies in gram-negative bacteremia. N. Engl. J. Med. 287: Neter, E., L. F. Bertram, D. A. Zak, M. R. Murdock, and C. E. Arbesman Studies on hemagglutination and hemolysis by Escherichia coli antisera. J. Exp. Med. 96: Schulman, J. L., and E. D. Kilbourne Induction of partial specific heterotypic immunity in mice by a single infection with Influenza A virus. J. Bacteriol. 89: Suzuki, T., E. A. Gorzynski, and E. Neter Separation by ethanol of common and somatic antigens of Enterobacteriaceae. J. Bacteriol. 88: Downloaded from on July 22, 2018 by guest
Grown in a Synthetic Culture Medium
 INFECTION AND IMMUNITY, Dec. 1970, p. 767-771 Copyright 1970 American Society for Microbiology V'ol. 2, No. 6 Printed in U.S.A. Production of Common Antigen by Enteric Bacteria Grown in a Synthetic Culture
INFECTION AND IMMUNITY, Dec. 1970, p. 767-771 Copyright 1970 American Society for Microbiology V'ol. 2, No. 6 Printed in U.S.A. Production of Common Antigen by Enteric Bacteria Grown in a Synthetic Culture
THE OCCURRENCE DURING ACUTE INFECTIONS OF A PROTEIN NOT NORMALLY PRESENT IN THE BLOOD
 Published Online: 1 February, 1941 Supp Info: http://doi.org/10.1084/jem.73.2.191 Downloaded from jem.rupress.org on November 21, 2018 THE OCCURRENCE DURING ACUTE INFECTIONS OF A PROTEIN NOT NORMALLY PRESENT
Published Online: 1 February, 1941 Supp Info: http://doi.org/10.1084/jem.73.2.191 Downloaded from jem.rupress.org on November 21, 2018 THE OCCURRENCE DURING ACUTE INFECTIONS OF A PROTEIN NOT NORMALLY PRESENT
FURTHER OBSERVATIONS ON THE AGGLUTINATION OF BACTERIA IN VIVO.
 FURTHER OBSERVATIONS ON THE AGGLUTINATION OF BACTERIA IN VIVO. BY CARROLL G. BULL, M.D. (From the Laboratories of The Rockefeller Institute for Medical Research.) PLATE 7. (Received for publication, April
FURTHER OBSERVATIONS ON THE AGGLUTINATION OF BACTERIA IN VIVO. BY CARROLL G. BULL, M.D. (From the Laboratories of The Rockefeller Institute for Medical Research.) PLATE 7. (Received for publication, April
(From the Oscar Johnson Institute, Washington University School of Medicine, St. Louis)
 Published Online: 1 July, 1935 Supp Info: http://doi.org/10.1084/jem.62.1.11 Downloaded from jem.rupress.org on April 7, 2018 THE IMMUNOLOGICAL SPECIFICITY OF STAPHYLOCOCCI I. THE OcctrRRENCE OF SEROLOGICAL
Published Online: 1 July, 1935 Supp Info: http://doi.org/10.1084/jem.62.1.11 Downloaded from jem.rupress.org on April 7, 2018 THE IMMUNOLOGICAL SPECIFICITY OF STAPHYLOCOCCI I. THE OcctrRRENCE OF SEROLOGICAL
PASSIVE PROTECTION BY HUMAN SERUM IN MICE INFECTED WITH ENCAPSULATED STAPHYLOCOCCUS A UREUS
 PASSIVE PROTECTION BY HUMAN SERUM IN MICE INFECTED WITH ENCAPSULATED STAPHYLOCOCCUS A UREUS K. YOSHIDA, Y. ICHIMAN, S. NARIKAWA, M. TAKAHASHI, E. KONO* AND C. L. SAN CLEMENTE? Department of Microbiology
PASSIVE PROTECTION BY HUMAN SERUM IN MICE INFECTED WITH ENCAPSULATED STAPHYLOCOCCUS A UREUS K. YOSHIDA, Y. ICHIMAN, S. NARIKAWA, M. TAKAHASHI, E. KONO* AND C. L. SAN CLEMENTE? Department of Microbiology
Demonstration of Serologically Different Capsular
 INFECTION AND IMMUNITY, Apr. 1971, p. 535-539 Copyright 1971 American Society for Microbiology Vol. 3, No. 4 Printed in U.S.A. Demonstration of Serologically Different Capsular Types Among Strains of Staphylococcus
INFECTION AND IMMUNITY, Apr. 1971, p. 535-539 Copyright 1971 American Society for Microbiology Vol. 3, No. 4 Printed in U.S.A. Demonstration of Serologically Different Capsular Types Among Strains of Staphylococcus
QUANTITATIVE EXPERIMENTS WITH ANTIBODIES TO SPECIFIC PRECIPITATES. II*
 Published Online: 1 February, 1941 Supp Info: http://doi.org/1.184/jem.73.2.293 Downloaded from jem.rupress.org on October 7, 218 QUANTITATIVE EXPERIMENTS WITH ANTIBODIES TO SPECIFIC PRECIPITATES. II*
Published Online: 1 February, 1941 Supp Info: http://doi.org/1.184/jem.73.2.293 Downloaded from jem.rupress.org on October 7, 218 QUANTITATIVE EXPERIMENTS WITH ANTIBODIES TO SPECIFIC PRECIPITATES. II*
(From the Hospital of The Rockefeller Institute for Medical Research)
 Published Online: 1 April, 1934 Supp Info: http://doi.org/10.1084/jem.59.4.441 Downloaded from jem.rupress.org on January 13, 2019 A SEROLOGICAL DIFFERENTIATION OF SPECIFIC TYPES OF BOVINE HEMOLYTIC STREPTOCOCCI
Published Online: 1 April, 1934 Supp Info: http://doi.org/10.1084/jem.59.4.441 Downloaded from jem.rupress.org on January 13, 2019 A SEROLOGICAL DIFFERENTIATION OF SPECIFIC TYPES OF BOVINE HEMOLYTIC STREPTOCOCCI
ANTIGENIC RELATIONSHIPS OF TORULOPSIS GLABRATA AND SEVEN SPECIES OF THE GENUS CANDIDA
 ANTIGENIC RELATIONSHIPS OF TORULOPSIS GLABRATA AND SEVEN SPECIES OF THE GENUS CANDIDA H. F. HASENCLEVER AND WILLIAM 0. MITCHELL U. S. Department of Health, Education and Welfare, Public Health Service,
ANTIGENIC RELATIONSHIPS OF TORULOPSIS GLABRATA AND SEVEN SPECIES OF THE GENUS CANDIDA H. F. HASENCLEVER AND WILLIAM 0. MITCHELL U. S. Department of Health, Education and Welfare, Public Health Service,
Separation and Properties of a Red Cell Sensitizing
 JOURNAL OF BACTERIOLOGY, June, 1966 Copyright @ 1966 American Society for Microbiology Vol. 91, No. 6 Printed in U.S.A. Separation and Properties of a Red Cell Sensitizing Substance from Streptococci MERWIN
JOURNAL OF BACTERIOLOGY, June, 1966 Copyright @ 1966 American Society for Microbiology Vol. 91, No. 6 Printed in U.S.A. Separation and Properties of a Red Cell Sensitizing Substance from Streptococci MERWIN
Detection of Antibodies to Brucella abortust
 JOURNAL OF CLINICAL MICROBIOLOGY, Apr. 198, p. 42-48 95-1 137/8/4-42/7$2. Vol. I1, No.4 Conditions for Conducting the Indirect Hemolysis Test for Detection of Antibodies to Brucella abortust EDWARD M.
JOURNAL OF CLINICAL MICROBIOLOGY, Apr. 198, p. 42-48 95-1 137/8/4-42/7$2. Vol. I1, No.4 Conditions for Conducting the Indirect Hemolysis Test for Detection of Antibodies to Brucella abortust EDWARD M.
Transfer Agent of Immunity. VI. Serial Passive Transfers of Cellular Immunity to Salmonella Infection by Immune Ribonucleic Acid
 Japan. J. Microbiol. Vol. 15 (2), 159-168, 1971 Transfer Agent of Immunity VI. Serial Passive Transfers of Cellular Immunity to Salmonella Infection by Immune Ribonucleic Acid Kazuko SAITO, Nobutaka OSAWA,
Japan. J. Microbiol. Vol. 15 (2), 159-168, 1971 Transfer Agent of Immunity VI. Serial Passive Transfers of Cellular Immunity to Salmonella Infection by Immune Ribonucleic Acid Kazuko SAITO, Nobutaka OSAWA,
3.0. Materials and methods
 63 3.0. Materials and methods 3.1. Plant materials and preparation of extracts Salacia oblonga plants were collected from Western Ghats, Karnataka, India. S. oblonga (RRCBI 7881) authentication was done
63 3.0. Materials and methods 3.1. Plant materials and preparation of extracts Salacia oblonga plants were collected from Western Ghats, Karnataka, India. S. oblonga (RRCBI 7881) authentication was done
THE TOXIC ANTIGENIC FACTORS PRODUCED BY CLOSTRIDIUM BOTULINUM TYPES C AND D
 Onderstepoort ]. vet. Res. 38 (2), 93-98 (1971) THE TOXIC ANTIGENIC FACTORS PRODUCED BY CLOSTRIDIUM BOTULINUM TYPES C AND D B. C. JANSEN, Veterinary Research Institute, Onderstepoort ABSTRACT JANSEN, B.
Onderstepoort ]. vet. Res. 38 (2), 93-98 (1971) THE TOXIC ANTIGENIC FACTORS PRODUCED BY CLOSTRIDIUM BOTULINUM TYPES C AND D B. C. JANSEN, Veterinary Research Institute, Onderstepoort ABSTRACT JANSEN, B.
INFLUENCE OF GUINEA PIG PLASMA FACTORS ON PHAGOCYTOSIS
 INFLUENCE OF GUINEA PIG PLASMA FACTORS ON PHAGOCYTOSIS OF PASTEURELLA PESTIS II. PLASMA FROM PLAGUE-INFECTED GUINEA PIGS W. G. STANZIALE1 AND J. D. WHITE U. S. Army Chemical Corps Biological Laboratories,
INFLUENCE OF GUINEA PIG PLASMA FACTORS ON PHAGOCYTOSIS OF PASTEURELLA PESTIS II. PLASMA FROM PLAGUE-INFECTED GUINEA PIGS W. G. STANZIALE1 AND J. D. WHITE U. S. Army Chemical Corps Biological Laboratories,
Supplementary Figure 1: Sequence alignment of partial stem region of flaviviruses
 Supplementary Figure 1: Sequence alignment of partial stem region of flaviviruses E prtoeins. Polyprotein sequences of viruses were downloaded from GenBank and aligned by CLC Sequence Viewer software.
Supplementary Figure 1: Sequence alignment of partial stem region of flaviviruses E prtoeins. Polyprotein sequences of viruses were downloaded from GenBank and aligned by CLC Sequence Viewer software.
Detection of Escherichia coli Antigens by a Latex Agglutination Test
 APPLIED MICROBIOLOGY, Aug. 1974, P. 306-311 Copyright ( 1974 American Society for Microbiology Vol. 28, No. 2 Printed in U.S.A. Detection of Escherichia coli Antigens by a Latex Agglutination Test K. HECHEMY,
APPLIED MICROBIOLOGY, Aug. 1974, P. 306-311 Copyright ( 1974 American Society for Microbiology Vol. 28, No. 2 Printed in U.S.A. Detection of Escherichia coli Antigens by a Latex Agglutination Test K. HECHEMY,
Effect of Hypertonic Sucrose Upon the Immune Bactericidal Reaction
 INFECrION AND IMMUNITY, Jan. 1970, p. 51-55 Copyright 1970 American Society for Microbiology Vol. 1, No. 1 Printed in U.S.A. Effect of Hypertonic Sucrose Upon the Immune Bactericidal Reaction LOUIS H.
INFECrION AND IMMUNITY, Jan. 1970, p. 51-55 Copyright 1970 American Society for Microbiology Vol. 1, No. 1 Printed in U.S.A. Effect of Hypertonic Sucrose Upon the Immune Bactericidal Reaction LOUIS H.
AKIBA, KAZUO IWATA AND SUTEMI INOUYE. Department of Bacteriology, Faculty of Medicine, University of Tokyo, Tokyo
 Japan. J. Microb., Vol. 1, No. 1, 1957 STUDIES ON THE SEROLOGIC DIAGNOSIS OF THE DEEP-SEATED CANDIDIASIS TOMOICHIRO AKIBA, KAZUO IWATA AND SUTEMI INOUYE Department of Bacteriology, Faculty of Medicine,
Japan. J. Microb., Vol. 1, No. 1, 1957 STUDIES ON THE SEROLOGIC DIAGNOSIS OF THE DEEP-SEATED CANDIDIASIS TOMOICHIRO AKIBA, KAZUO IWATA AND SUTEMI INOUYE Department of Bacteriology, Faculty of Medicine,
SKIN INFECTION OF RABBITS WITH HEMOLYTIC STREP- TOCOCCI ISOLATED FROM A PATIENT WITH ERYSIPELAS.
 SKIN INFECTION OF RABBITS WITH HEMOLYTIC STREP- TOCOCCI ISOLATED FROM A PATIENT WITH ERYSIPELAS. I. METHOD OF DEMONSTRATING PROTECTIVE ACTION OF IMMUNE SERA. BY THOMAS M. RIVERS, M.D. (From the Hospital
SKIN INFECTION OF RABBITS WITH HEMOLYTIC STREP- TOCOCCI ISOLATED FROM A PATIENT WITH ERYSIPELAS. I. METHOD OF DEMONSTRATING PROTECTIVE ACTION OF IMMUNE SERA. BY THOMAS M. RIVERS, M.D. (From the Hospital
Test Method of Specified Requirements of Antibacterial Textiles for Medical Use FTTS-FA-002
 Test Method of Specified Requirements of Antibacterial Textiles for Medical Use FTTS-FA-002 FTTS-FA-002 Antibacterial Textiles for Medical Use Antibacterial Textiles suppress and even kill harmful bacteria
Test Method of Specified Requirements of Antibacterial Textiles for Medical Use FTTS-FA-002 FTTS-FA-002 Antibacterial Textiles for Medical Use Antibacterial Textiles suppress and even kill harmful bacteria
Foot-and-Mouth Disease Virus: Selection by
 INFECTION AND IMMUNITY, Jan. 1972, p. 65-69 Copyright 1972 American Society for Microbiology Foot-and-Mouth Disease Virus: Selection by Homogenized Calf Kidney Adsorption and Cell Culture Passage Vol.
INFECTION AND IMMUNITY, Jan. 1972, p. 65-69 Copyright 1972 American Society for Microbiology Foot-and-Mouth Disease Virus: Selection by Homogenized Calf Kidney Adsorption and Cell Culture Passage Vol.
SEROLOGICAL CHARACTERIZATION OF PATHOGENIC FUNGI
 SEROLOGICAL CHARACTERIZATION OF PATHOGENIC FUNGI BY MEANS OF FLUORESCENT ANTIBODIES I. ANTIGENIC RELATIONSHIPS BETWEEN YEAST AND MYCELIAL FORMS OF HISTOPLASMA CAPSULATUM AND BLASTOMYCES DERMATITIDIS LEO
SEROLOGICAL CHARACTERIZATION OF PATHOGENIC FUNGI BY MEANS OF FLUORESCENT ANTIBODIES I. ANTIGENIC RELATIONSHIPS BETWEEN YEAST AND MYCELIAL FORMS OF HISTOPLASMA CAPSULATUM AND BLASTOMYCES DERMATITIDIS LEO
TITRATION OF ANTISERA TO SOLUBLE PROTEINS ON THE BASIS OF AN AGGLUTINATION REACTION:
 390 TITRATION OF ANTISERA TO SOLUBLE PROTEINS ON THE BASIS OF AN AGGLUTINATION REACTION: CONJUGATION OF EGG ALBUMIN AND CHICKEN SERUM GLOBULIN TO THE INCOMPLETE RH ANTIBODY AND THE SUBSEQUENT USE OF RH-POSITIVE
390 TITRATION OF ANTISERA TO SOLUBLE PROTEINS ON THE BASIS OF AN AGGLUTINATION REACTION: CONJUGATION OF EGG ALBUMIN AND CHICKEN SERUM GLOBULIN TO THE INCOMPLETE RH ANTIBODY AND THE SUBSEQUENT USE OF RH-POSITIVE
Document No. FTTS-FA-001. Specified Requirements of Antibacterial Textiles for General Use
 1. Purpose and Scope This criterion is applicable to the evaluation and testing of antibacterial activity of textile for general use. The quantitative evaluation of antibacterial activity is judged by
1. Purpose and Scope This criterion is applicable to the evaluation and testing of antibacterial activity of textile for general use. The quantitative evaluation of antibacterial activity is judged by
DETECTION OF BRUCELLAE AND THEIR ANTIBODIES BY FLUORESCENT ANTIBODY AND AGGLUTINATION TESTS
 DETECTION OF BRUCELLAE AND THEIR ANTIBODIES BY FLUORESCENT ANTIBODY AND AGGLUTINATION TESTS MAX D. MOODY, JOSEPH Z. BIEGELEISEN, JR., AND GERALD C. TAYLOR U. S. Department of Health, Education and Welfare,
DETECTION OF BRUCELLAE AND THEIR ANTIBODIES BY FLUORESCENT ANTIBODY AND AGGLUTINATION TESTS MAX D. MOODY, JOSEPH Z. BIEGELEISEN, JR., AND GERALD C. TAYLOR U. S. Department of Health, Education and Welfare,
Chapter 17: Immunization & Immune Testing. 1. Immunization 2. Diagnostic Immunology
 Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
1. Immunization. What is Immunization? 12/9/2016. Chapter 17: Immunization & Immune Testing. 1. Immunization 2. Diagnostic Immunology
 Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
Chapter 17: Immunization & Immune Testing 1. Immunization 2. Diagnostic Immunology 1. Immunization Chapter Reading pp. 505-511 What is Immunization? A method of inducing artificial immunity by exposing
Method for Identifying Salmonella and Shigella Directly from
 JOURNAL OF CLINICAL MICROBIOLOGY, Mar. 1976, p. 339-343 Copyright 1976 American Society for Microbiology Vol. 3, No. 3 Printed in U-SA. Method for Identifying Salmonella and Shigella Directly from the
JOURNAL OF CLINICAL MICROBIOLOGY, Mar. 1976, p. 339-343 Copyright 1976 American Society for Microbiology Vol. 3, No. 3 Printed in U-SA. Method for Identifying Salmonella and Shigella Directly from the
Method for Identifying Salmonella and Shigella Directly from
 JOURNAL OF CLINICAL MICROBIOLOGY, Mar. 1976, p. 339-343 Copyright 1976 American Society for Microbiology Vol. 3, No. 3 Printed in U-SA. Method for Identifying Salmonella and Shigella Directly from the
JOURNAL OF CLINICAL MICROBIOLOGY, Mar. 1976, p. 339-343 Copyright 1976 American Society for Microbiology Vol. 3, No. 3 Printed in U-SA. Method for Identifying Salmonella and Shigella Directly from the
AN ELISA FOR THE DETECTION OF ANTIBODIES AGAINST NEWCASTLE DISEASE VIRUS IN AFRICAN VILLAGE POULTRY
 AN ELISA FOR THE DETECTION OF ANTIBODIES AGAINST NEWCASTLE DISEASE VIRUS IN AFRICAN VILLAGE POULTRY J.G. BELL, M. LELENTA Animal Production Unit, Food and Agriculture International Atomic Energy Agency,
AN ELISA FOR THE DETECTION OF ANTIBODIES AGAINST NEWCASTLE DISEASE VIRUS IN AFRICAN VILLAGE POULTRY J.G. BELL, M. LELENTA Animal Production Unit, Food and Agriculture International Atomic Energy Agency,
Identification of Strains of Sf reptococcus pyogenes of Types 5, 11, 12, 27 and 44 by the Precipitin Test for the T Antigen
 110 MCLEAN, S. J. (1953). J. gen. Mkro~l. 9, 110118. Identification of Strains of Sf reptococcus pyogenes of Types 5, 11, 12, 27 and 44 by the Precipitin Test for the T Antigen BY SIBELY J. McLEAN* Streptococcal
110 MCLEAN, S. J. (1953). J. gen. Mkro~l. 9, 110118. Identification of Strains of Sf reptococcus pyogenes of Types 5, 11, 12, 27 and 44 by the Precipitin Test for the T Antigen BY SIBELY J. McLEAN* Streptococcal
STAINING BACTERIAL SMEARS WITH FLUORESCENT ANTIBODY
 STAINING BACTERIAL SMEARS WITH FLUORESCENT ANTIBODY III. ANTIGENIC ANALYSIS OF Salmonella typhosa BY MEANS OF FLUORESCENT ANTIBODY AND AGGLUTINATION REACTIONS BERENICE M. THOMASON, WILLIAM B. CHERRY, AND
STAINING BACTERIAL SMEARS WITH FLUORESCENT ANTIBODY III. ANTIGENIC ANALYSIS OF Salmonella typhosa BY MEANS OF FLUORESCENT ANTIBODY AND AGGLUTINATION REACTIONS BERENICE M. THOMASON, WILLIAM B. CHERRY, AND
days at 24 C is, within limits, proportional to
 IN VITRO STUDIES ON STAPHYLOCOCCAL ENTEROTOXIN PRODUCTION' H. SUGIYAMA, M. S. BERGDOLL, AND G. M. DACK Food Research Institute, University of Chicago, Chicago, Illinois Received for publication January
IN VITRO STUDIES ON STAPHYLOCOCCAL ENTEROTOXIN PRODUCTION' H. SUGIYAMA, M. S. BERGDOLL, AND G. M. DACK Food Research Institute, University of Chicago, Chicago, Illinois Received for publication January
days at 24 C is, within limits, proportional to
 IN VITRO STUDIES ON STAPHYLOCOCCAL ENTEROTOXIN PRODUCTION' H. SUGIYAMA, M. S. BERGDOLL, AND G. M. DACK Food Research Institute, University of Chicago, Chicago, Illinois Received for publication January
IN VITRO STUDIES ON STAPHYLOCOCCAL ENTEROTOXIN PRODUCTION' H. SUGIYAMA, M. S. BERGDOLL, AND G. M. DACK Food Research Institute, University of Chicago, Chicago, Illinois Received for publication January
THE TECHNIC OF THE KOLMER COMPLEMENT FIXATION TESTS FOR SYPHILIS EMPLOYING ONE-FIFTH AMOUNTS OF REAGENTS JOHN A. KOLMER
 THE TECHNIC OF THE KOLMER COMPLEMENT FIXATION TESTS FOR SYPHILIS EMPLOYING ONE-FIFTH AMOUNTS OF REAGENTS JOHN A. KOLMER WITH THE TECHNICAL ASSISTANCE OP ELSA R. LYNCH Department of Bacteriology and Immunology
THE TECHNIC OF THE KOLMER COMPLEMENT FIXATION TESTS FOR SYPHILIS EMPLOYING ONE-FIFTH AMOUNTS OF REAGENTS JOHN A. KOLMER WITH THE TECHNICAL ASSISTANCE OP ELSA R. LYNCH Department of Bacteriology and Immunology
AN EXAMINATION OF THE EFFECTS OF SIMVASTATIN ON INNATE IMMUNE RESPONSES TO S. AUREUS A RESEARCH PAPER BY TRACI STANKIEWICZ
 AN EXAMINATION OF THE EFFECTS OF SIMVASTATIN ON INNATE IMMUNE RESPONSES TO S. AUREUS A RESEARCH PAPER BY TRACI STANKIEWICZ SUBMITTED TO THE GRADUATE SCHOOL IN PARTIAL FULFILLMENT OF THE REQUIRMENTS SET
AN EXAMINATION OF THE EFFECTS OF SIMVASTATIN ON INNATE IMMUNE RESPONSES TO S. AUREUS A RESEARCH PAPER BY TRACI STANKIEWICZ SUBMITTED TO THE GRADUATE SCHOOL IN PARTIAL FULFILLMENT OF THE REQUIRMENTS SET
Antibody Response to Bacterial Antigens: Characteristics of Antibody Response to Somatic
 INFECTION AND IMMUNITY, Mar. 1970, p. 219-225 Copyright 1970 American Society for Microbiology Vol. 1, No. 3 Printed in U.S.A. Antibody Response to Bacterial Antigens: Characteristics of Antibody Response
INFECTION AND IMMUNITY, Mar. 1970, p. 219-225 Copyright 1970 American Society for Microbiology Vol. 1, No. 3 Printed in U.S.A. Antibody Response to Bacterial Antigens: Characteristics of Antibody Response
APPENDIXES APPENDIX - I. Buffer, Reagent and Culture Media Preparation. 5 gm. 5 gm. 15 gm
 APPENDIXES APPENDIXES APPENDIX - I Buffer, Reagent and Culture Media Preparation a. Tryptone soy broth Casein triptic digest NaCl Soy peptone 1 b. Tryptone soy agar Casein triptic digest NaCl Soy peptone
APPENDIXES APPENDIXES APPENDIX - I Buffer, Reagent and Culture Media Preparation a. Tryptone soy broth Casein triptic digest NaCl Soy peptone 1 b. Tryptone soy agar Casein triptic digest NaCl Soy peptone
Lab. 7: Serological Tests ELISA. 320 MIC Microbial Diagnosis 320 MBIO PRACTICAL. Amal Alghamdi 2018
 Lab. 7: 320 MIC Microbial Diagnosis Serological Tests ELISA. 320 MBIO PRACTICAL Amal Alghamdi 2018 1 Infection and Immunity Serology is the study of immune bodies in human blood. These are products of
Lab. 7: 320 MIC Microbial Diagnosis Serological Tests ELISA. 320 MBIO PRACTICAL Amal Alghamdi 2018 1 Infection and Immunity Serology is the study of immune bodies in human blood. These are products of
Mouse C-Reactive Protein and Endotoxin-Induced Resistance
 JOURNAL OF BACTERIOLOGY, Dec., 1965 Copyright @ 1965 American Society for Microbiology Vol. 90, No. 6 Printed in U.S.A. Mouse C-Reactive Protein and Endotoxin-Induced Resistance L. T. PATTERSON' AND R.
JOURNAL OF BACTERIOLOGY, Dec., 1965 Copyright @ 1965 American Society for Microbiology Vol. 90, No. 6 Printed in U.S.A. Mouse C-Reactive Protein and Endotoxin-Induced Resistance L. T. PATTERSON' AND R.
sline, and the cells were killed by heating at 56 C for 1 hour. One to 2 ml of
 THE APPLICATION OF BACTERIOPHAGE AND SEROLOGY IN THE DIFFERENTIATION OF STRAINS OF LEUCONOSTOC MESENTEROIDES ALVARO LEIVA-QUIROS AND C. S. McCLESKEY Louisiana Stats University, Baton Rouge, Louisiana Received
THE APPLICATION OF BACTERIOPHAGE AND SEROLOGY IN THE DIFFERENTIATION OF STRAINS OF LEUCONOSTOC MESENTEROIDES ALVARO LEIVA-QUIROS AND C. S. McCLESKEY Louisiana Stats University, Baton Rouge, Louisiana Received
obtained from the infected and treated tissues, Fleming's2 technic of hemolytic streptococcus B. Immediately following the infection, 1.0 ml.
 THE SENSITIVITY OF STREPTOCOCCI TO PENICILLIN G AFTER EXPOSURE TO THE ANTIBIOTIC IN VIVO* E. GRUNBERG, C. UNGER, AND D. ELDRIDGE Previous investigations by Grunberg, Schnitzer, and Unger3 on the topical
THE SENSITIVITY OF STREPTOCOCCI TO PENICILLIN G AFTER EXPOSURE TO THE ANTIBIOTIC IN VIVO* E. GRUNBERG, C. UNGER, AND D. ELDRIDGE Previous investigations by Grunberg, Schnitzer, and Unger3 on the topical
COMPARATIVE STUDY OF THE AGGLUTINOGENS OF THE ENDOSPORES OF
 COMPARATIVE STUDY OF THE AGGLUTINOGENS OF THE ENDOSPORES OF BACILLUS ANTHRACIS AND BACILLUS CEREUS' CARL LAMANNA AND DANIEL EISLER The Naval Biological Laboratory, School of Public Health, University of
COMPARATIVE STUDY OF THE AGGLUTINOGENS OF THE ENDOSPORES OF BACILLUS ANTHRACIS AND BACILLUS CEREUS' CARL LAMANNA AND DANIEL EISLER The Naval Biological Laboratory, School of Public Health, University of
E. A. EDWARDS' AND G. L. LARSON
 APPLIED MICROBIOLOGY, Dec. 1974, p. 972-976 Copyright 0 1975 American Society for Microbiology Vol. 28, No. 6 Printed in U.S.A. New Method of Grouping Beta-Hemolytic Streptococci Directly on Sheep Blood
APPLIED MICROBIOLOGY, Dec. 1974, p. 972-976 Copyright 0 1975 American Society for Microbiology Vol. 28, No. 6 Printed in U.S.A. New Method of Grouping Beta-Hemolytic Streptococci Directly on Sheep Blood
Instructions for use of 1 % Sheep-Erythrocytes-Suspension
 Instructions for use of 1 % Sheep-Erythrocytes-Suspension Ready to use for CFT Order No.: E-420, E-423, E-430, E-433, E-450, E-453 Intended Use The 1 % Sheep-Erythrocytes-Suspension is a ready-to-use reagent
Instructions for use of 1 % Sheep-Erythrocytes-Suspension Ready to use for CFT Order No.: E-420, E-423, E-430, E-433, E-450, E-453 Intended Use The 1 % Sheep-Erythrocytes-Suspension is a ready-to-use reagent
Rapid Detection of Bacterial Growth in Blood Cultures by Bioluminescent Assay of Bacterial ATP
 JOURNAL OF CLINICAL MICROBIOLOGY, Sept. 1983, p. 521-525 0095-1137/83/090521-05$02.00/O Copyright C 1983, American Society for Microbiology Vol. 18, No. 3 Rapid Detection of Bacterial Growth in Blood Cultures
JOURNAL OF CLINICAL MICROBIOLOGY, Sept. 1983, p. 521-525 0095-1137/83/090521-05$02.00/O Copyright C 1983, American Society for Microbiology Vol. 18, No. 3 Rapid Detection of Bacterial Growth in Blood Cultures
Trachoma-Inclusion Conjunctivitis-Lymphogranuloma
 INFZCTON AND IMMUNITr, Mar. 973, p. 3536 Copyright 973 American Society for Microbiology Vol. 7, No. 3 Printed in U.S.A. A Simplified Method for Immunological Typing of Trachoma-Inclusion Conjunctivitis-Lymphogranuloma
INFZCTON AND IMMUNITr, Mar. 973, p. 3536 Copyright 973 American Society for Microbiology Vol. 7, No. 3 Printed in U.S.A. A Simplified Method for Immunological Typing of Trachoma-Inclusion Conjunctivitis-Lymphogranuloma
Generation of Non-typeable Haemophilus influenzae Directed Gene Deletion Mutants Jeroen D. Langereis *
 Generation of Non-typeable Haemophilus influenzae Directed Gene Deletion Mutants Jeroen D. Langereis * Laboratory of Pediatric Infectious Diseases, Department of Pediatrics and Laboratory of Medical Immunology,
Generation of Non-typeable Haemophilus influenzae Directed Gene Deletion Mutants Jeroen D. Langereis * Laboratory of Pediatric Infectious Diseases, Department of Pediatrics and Laboratory of Medical Immunology,
RAPID IDENTIFICATION OF CHOLERA VIBRIOS WITH FLUORESCENT ANTIBODY
 RAPID IDENTIFICATION OF CHOLERA VIBRIOS WITH FLUORESCENT ANTIBODY RICHARD A. FINKELSTEIN AND EUGENE H. LABREC Division of Immunology, Walter Reed Army Institute of Research, Washington, D. C. Received
RAPID IDENTIFICATION OF CHOLERA VIBRIOS WITH FLUORESCENT ANTIBODY RICHARD A. FINKELSTEIN AND EUGENE H. LABREC Division of Immunology, Walter Reed Army Institute of Research, Washington, D. C. Received
Report BerryMeat. Antimicrobial effect for different preparations from 8 plants during storage at 18 C for 1½ year. Flemming Hansen.
 Report BerryMeat Antimicrobial effect for different preparations from 8 plants during storage at 18 C for 1½ year. 19. marts 2014 Project 2000248-13 FH/ Flemming Hansen Summary Introduction In the project,
Report BerryMeat Antimicrobial effect for different preparations from 8 plants during storage at 18 C for 1½ year. 19. marts 2014 Project 2000248-13 FH/ Flemming Hansen Summary Introduction In the project,
The Bentonite Flocculation Test for Detection of Plant Viruses and Titration of Antibody. (28801)
 Copyright, 1963, by the Society for Experimental Biology and Medicine. Reprinted from PROCEEDINGS OF THE SOCIETY FOR EXPERIMENTAL BIOLOGY AND MEDICINE, 1963, v114, 75-754 The Bentonite Flocculation Test
Copyright, 1963, by the Society for Experimental Biology and Medicine. Reprinted from PROCEEDINGS OF THE SOCIETY FOR EXPERIMENTAL BIOLOGY AND MEDICINE, 1963, v114, 75-754 The Bentonite Flocculation Test
An indirect haemagglutination test to detect serum antibodies to Giardia lamblia
 J. Biosci., Vol. 10, Number 4, December 1986, pp. 475-480. Printed in India. An indirect haemagglutination test to detect serum antibodies to Giardia lamblia K. N. JALAN, TUSHER MAITRA and RITA DAS Kothari
J. Biosci., Vol. 10, Number 4, December 1986, pp. 475-480. Printed in India. An indirect haemagglutination test to detect serum antibodies to Giardia lamblia K. N. JALAN, TUSHER MAITRA and RITA DAS Kothari
Transduction of Staphylococcus aureus to
 JOURNAL OF BACTPERIOLOGY, May, 1965 Copyright 1965 American Society for Microbiology Vol. 89, No. 5 Printed in U.S.A. Transduction of Staphylococcus aureus to Tetracycline Resistance In Vivo HOWARD JAROLMEN,
JOURNAL OF BACTPERIOLOGY, May, 1965 Copyright 1965 American Society for Microbiology Vol. 89, No. 5 Printed in U.S.A. Transduction of Staphylococcus aureus to Tetracycline Resistance In Vivo HOWARD JAROLMEN,
See external label 2 C-8 C = ANTISTREPTOLYSIN O (ASO) REAGENT SET A LATEX SLIDE TEST
 CORTEZ DIAGNOSTICS, INC. 21250 Califa Street, Suite 102 and 116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
CORTEZ DIAGNOSTICS, INC. 21250 Califa Street, Suite 102 and 116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
Analysis of Corynebacterium vaginale by an Immunodiffusion
 APPLIED MICROBIOLOGY, Mar. 1974, p. 469-474 Copyright 0 1974 American Society for Microbiology Vol. 27, No. 3 Printed in U.S.A. Analysis of Corynebacterium vaginale by an Immunodiffusion Technique MARY
APPLIED MICROBIOLOGY, Mar. 1974, p. 469-474 Copyright 0 1974 American Society for Microbiology Vol. 27, No. 3 Printed in U.S.A. Analysis of Corynebacterium vaginale by an Immunodiffusion Technique MARY
Cross-Protection among Serotypes of Group A Streptococci
 THE JOURNAL OF INFECTIOUS DISEASES VOL. 125 NO.4 APRIL 1972 1972 by the University of Chicago. All rights reserved. ' Cross-Protection among Serotypes of Group A Streptococci Sonia Bergner-Rabinowitz,
THE JOURNAL OF INFECTIOUS DISEASES VOL. 125 NO.4 APRIL 1972 1972 by the University of Chicago. All rights reserved. ' Cross-Protection among Serotypes of Group A Streptococci Sonia Bergner-Rabinowitz,
Lafayette, Indiana. Animals. Adult albino mice of the Purdue. Swiss and Rockland Swis strains were used. Toxin and antiserum. The toxin and antitoxin
 THE EFFECT OF CALCIUM BINDING AGENTS ON THE VIRULENCE OF CLOSTRIDIUM PERFRINGENS FOR THE WHITE MOUSE' ERNEST ALAN MEYER AND MERWIN MOSKOWITZ Division of Bacteriology, Department of Biological Sciences,
THE EFFECT OF CALCIUM BINDING AGENTS ON THE VIRULENCE OF CLOSTRIDIUM PERFRINGENS FOR THE WHITE MOUSE' ERNEST ALAN MEYER AND MERWIN MOSKOWITZ Division of Bacteriology, Department of Biological Sciences,
The Cat s Out of the Bag: Microbiological Investigations of Acute Transfusion Reactions.
 The Cat s Out of the Bag: Microbiological Investigations of Acute Transfusion Reactions. Philippe Lagacé-Wiens, MD FRCPC, DTM&H plagacewiens@sharedhealthmb.ca COI declaration I have no conflicts, real
The Cat s Out of the Bag: Microbiological Investigations of Acute Transfusion Reactions. Philippe Lagacé-Wiens, MD FRCPC, DTM&H plagacewiens@sharedhealthmb.ca COI declaration I have no conflicts, real
TYPING OF GROUP A STREPTOCOCCI BY IMMUNOFLUORESCENCE
 JOURNAL OF BACTERIOLOGY Vol. 87, No. 6, pp. 1377-1382 June, 1964 Copyright 1964 by the Anmerican Society for Microbiology Printed in U.S.A. TYPING OF GROUP A STREPTOCOCCI BY IMMUNOFLUORESCENCE I. PREPARATION
JOURNAL OF BACTERIOLOGY Vol. 87, No. 6, pp. 1377-1382 June, 1964 Copyright 1964 by the Anmerican Society for Microbiology Printed in U.S.A. TYPING OF GROUP A STREPTOCOCCI BY IMMUNOFLUORESCENCE I. PREPARATION
Enzyme-Linked Immunosorbent Assays for
 INFECON AND IMMUNrrY, May 1973, p. 759-763 Copyright i 1973 American Society for Microbiology Vol. 7, No. 5 Printed in U.S.A. Enzyme-Linked Immunosorbent Assays for Cholera Serology J. HOLMGREN AND A.-M.
INFECON AND IMMUNrrY, May 1973, p. 759-763 Copyright i 1973 American Society for Microbiology Vol. 7, No. 5 Printed in U.S.A. Enzyme-Linked Immunosorbent Assays for Cholera Serology J. HOLMGREN AND A.-M.
(From the Laboratories of The Rockefeller Institute for Medical Research)
 STUDIES ON THE FLEXNER GROUP OF DYSENTERY BACILLI IV. THE SEROLOGICAL AND Toxic PROPERTIES OF THE SOMATIC ANTIGENS* BY ELY PERLMAN, M.D., ANY WALTHER F. GOEBEL, PH.D. (From the Laboratories of The Rockefeller
STUDIES ON THE FLEXNER GROUP OF DYSENTERY BACILLI IV. THE SEROLOGICAL AND Toxic PROPERTIES OF THE SOMATIC ANTIGENS* BY ELY PERLMAN, M.D., ANY WALTHER F. GOEBEL, PH.D. (From the Laboratories of The Rockefeller
Replication of Vesicular Stomatitis Virus Facilitated by Shope
 INFECTION AND IMMUNITY, July 1979, p. 213-219 0019-9567/79/07-0213/07 $02.00/0 Vol. 25, No. 1 Replication of Vesicular Stomatitis Virus Facilitated by Shope Fibroma Virus In Vivo NORMAN A. CROUCHt* AND
INFECTION AND IMMUNITY, July 1979, p. 213-219 0019-9567/79/07-0213/07 $02.00/0 Vol. 25, No. 1 Replication of Vesicular Stomatitis Virus Facilitated by Shope Fibroma Virus In Vivo NORMAN A. CROUCHt* AND
(From the Laboratories of The Rockefeller Institute for Medical Research)
 Published Online: 1 September, 1946 Supp Info: http://doi.org/1.184/jem.84.3.223 Downloaded from jem.rupress.org on January 5, 219 STUDIES ON THE FLEXNER GROUP OF DYSENTERY BACILLI IV. THE SEROLOGICAL
Published Online: 1 September, 1946 Supp Info: http://doi.org/1.184/jem.84.3.223 Downloaded from jem.rupress.org on January 5, 219 STUDIES ON THE FLEXNER GROUP OF DYSENTERY BACILLI IV. THE SEROLOGICAL
Culture of Serum-induced Spheroplasts from
 JOURNAL OF BACTERIOLOGY, May 1967, p. 1688-1692 Copyright ( 1967 American Society for Microbiology Vol. 93, No. 5 Printed in U.S.A. Culture of Serum-induced Spheroplasts from Vibrio cholerae ANTOINETTE
JOURNAL OF BACTERIOLOGY, May 1967, p. 1688-1692 Copyright ( 1967 American Society for Microbiology Vol. 93, No. 5 Printed in U.S.A. Culture of Serum-induced Spheroplasts from Vibrio cholerae ANTOINETTE
A membrane filter technique for testing disinfectants
 J. clin. Path., 1975, 28, 71-76 A membrane filter technique for testing disinfectants JEAN PRINCE', C. E. A. DEVERILL, AND G. A. J. AYLIFFE From the Hospital Infection Research Laboratory, Birmingham SYNOPSIS
J. clin. Path., 1975, 28, 71-76 A membrane filter technique for testing disinfectants JEAN PRINCE', C. E. A. DEVERILL, AND G. A. J. AYLIFFE From the Hospital Infection Research Laboratory, Birmingham SYNOPSIS
Effect of Diuresis on Staphylococcus aureus Kidney
 INFECTION AND IMMUNITY, Dec. 1971, p. 74-746 Copyright 1971 American Society for Microbiology Vol. 4, No. 6 Printed in U.S.A. Effect of Diuresis on Staphylococcus aureus Kidney Infections in Mice DOLORES
INFECTION AND IMMUNITY, Dec. 1971, p. 74-746 Copyright 1971 American Society for Microbiology Vol. 4, No. 6 Printed in U.S.A. Effect of Diuresis on Staphylococcus aureus Kidney Infections in Mice DOLORES
meningitidis passages from the original source material by storing the cultures at -65 C (18) in Trypticase Soy Broth
 JOURNAL OP BACTERIOLOGY, Apr. 1968, p. 1300-1304 Vol. 95, No. 4 Copyright @ 1968 American Society for Microbiology Printed in U.S.A. A New Serological Group (E) of Neisseria meningitidis NEYLAN A. VEDROS,
JOURNAL OP BACTERIOLOGY, Apr. 1968, p. 1300-1304 Vol. 95, No. 4 Copyright @ 1968 American Society for Microbiology Printed in U.S.A. A New Serological Group (E) of Neisseria meningitidis NEYLAN A. VEDROS,
INTRODUCTION water-soluble Figure 1.
 INTRODUCTION Natural waters contain bacteria. The aerobic gram negative bacillus of the genera Psedomonas, Alcalignes, and Flavobacterium are common in natural waters. Many of these bacteria are able to
INTRODUCTION Natural waters contain bacteria. The aerobic gram negative bacillus of the genera Psedomonas, Alcalignes, and Flavobacterium are common in natural waters. Many of these bacteria are able to
Jlexneri. The first portion compares the specificity
 SEROLOGICAL IDENTIFICATION OF SHIGELLA FLEXNERI BY MEANS OF FLUORESCENT ANTIBODY EUGENE H. LABREC, SAMUEL B. FORMAL, AND HERMAN SCHNEIDER Immunology Division, Walter Reed Army Institute of Research, Walter
SEROLOGICAL IDENTIFICATION OF SHIGELLA FLEXNERI BY MEANS OF FLUORESCENT ANTIBODY EUGENE H. LABREC, SAMUEL B. FORMAL, AND HERMAN SCHNEIDER Immunology Division, Walter Reed Army Institute of Research, Walter
Microbial Survival. Created on 2/25/ :05 AM
 Microbial Survival Introduction The main objectives of this practical are: Verify, or otherwise, the Department of Health recommendation that food be cooked to 70 o C for 2 minutes or an equivalent time-temperature
Microbial Survival Introduction The main objectives of this practical are: Verify, or otherwise, the Department of Health recommendation that food be cooked to 70 o C for 2 minutes or an equivalent time-temperature
Interferon-Producing Capacity of Germfree Mice
 INFECTION AND IMMUNrrY, Feb. 1976, p. 332-336 Copyright 1976 American Society for Microbiology Vol. 13, No. 2 Printed in USA. Interferon-Producing Capacity of Germfree Mice YASUHIKO ITO, YUKIHIRO NISIYAMA,
INFECTION AND IMMUNrrY, Feb. 1976, p. 332-336 Copyright 1976 American Society for Microbiology Vol. 13, No. 2 Printed in USA. Interferon-Producing Capacity of Germfree Mice YASUHIKO ITO, YUKIHIRO NISIYAMA,
Relation of Mucoid Growth of Staphylococcus aureus
 JOURNAL OF BACTERIOLOGY, OCt. 1968, p. 902-908 Copyright @ 1968 American Society for Microbiology Vol. 96, No. 4 Printed in U.S.A. Relation of Mucoid Growth of Staphylococcus aureus to Clumping Factor
JOURNAL OF BACTERIOLOGY, OCt. 1968, p. 902-908 Copyright @ 1968 American Society for Microbiology Vol. 96, No. 4 Printed in U.S.A. Relation of Mucoid Growth of Staphylococcus aureus to Clumping Factor
Ammonium Sulfate Fractionation of Sera: Mouse, Hamster,
 APuLED MICROBIOLoGY, Feb. 1974, p. 389-393 Copyright 0 1974 American Society for Microbiology Vol. 27, No. 2 Printed in U.SA. Ammonium Sulfate Fractionation of Sera: Mouse, Hamster, Guinea Pig, Monkey,
APuLED MICROBIOLoGY, Feb. 1974, p. 389-393 Copyright 0 1974 American Society for Microbiology Vol. 27, No. 2 Printed in U.SA. Ammonium Sulfate Fractionation of Sera: Mouse, Hamster, Guinea Pig, Monkey,
DNA: THE GENETIC MATERIAL
 DNA: THE GENETIC MATERIAL This document is licensed under the Attribution-NonCommercial-ShareAlike 2.5 Italy license, available at http://creativecommons.org/licenses/by-nc-sa/2.5/it/ 1. Which macromolecule
DNA: THE GENETIC MATERIAL This document is licensed under the Attribution-NonCommercial-ShareAlike 2.5 Italy license, available at http://creativecommons.org/licenses/by-nc-sa/2.5/it/ 1. Which macromolecule
ELECTRON-MICROSCOPIC OBSERVATIONS OF POLYOMA VIRUS-TRANSFORMED MOUSE CELLS TREATED WITH SPECIFIC IMMUNE SERUM
 J. Cell Sci. 7, 711-718(1970) 7II Printed in Great Britain ELECTRON-MICROSCOPIC OBSERVATIONS OF POLYOMA VIRUS-TRANSFORMED MOUSE CELLS TREATED WITH SPECIFIC IMMUNE SERUM G.NEGRONI AND RITA TILLY Department
J. Cell Sci. 7, 711-718(1970) 7II Printed in Great Britain ELECTRON-MICROSCOPIC OBSERVATIONS OF POLYOMA VIRUS-TRANSFORMED MOUSE CELLS TREATED WITH SPECIFIC IMMUNE SERUM G.NEGRONI AND RITA TILLY Department
EXPERIMENTAL BLASTOMYCOSIS IN MICE' JOSEPH M. HITCH, M.D. Wim THE TECHNICAL ASSISTANCE OF JANE S. SHARP, M.S.
 EXPERIMENTAL BLASTOMYCOSIS IN MICE' JOSEPH M. HITCH, M.D. Raleigh, N. C.; Instructor in Dermatology and Syphilology, Duke University School of Medicine Wim THE TECHNICAL ASSISTANCE OF JANE S. SHARP, M.S.
EXPERIMENTAL BLASTOMYCOSIS IN MICE' JOSEPH M. HITCH, M.D. Raleigh, N. C.; Instructor in Dermatology and Syphilology, Duke University School of Medicine Wim THE TECHNICAL ASSISTANCE OF JANE S. SHARP, M.S.
LECTURE: 26 SIMPLE SEROLOGICAL LABORATORY TECHNIQUES LEARNING OBJECTIVES:
 LECTURE: 26 Title SIMPLE SEROLOGICAL LABORATORY TECHNIQUES LEARNING OBJECTIVES: The student should be able to: Define the term "simple serological techniques". Describe the benefit of the use of serological
LECTURE: 26 Title SIMPLE SEROLOGICAL LABORATORY TECHNIQUES LEARNING OBJECTIVES: The student should be able to: Define the term "simple serological techniques". Describe the benefit of the use of serological
CHAPTER 24. Immunology
 CHAPTER 24 Diagnostic i Microbiology and Immunology Growth-Dependent Diagnostic Methods Isolation of Pathogens from Clinical Specimens Proper sampling and culture of a suspected pathogen is the most reliable
CHAPTER 24 Diagnostic i Microbiology and Immunology Growth-Dependent Diagnostic Methods Isolation of Pathogens from Clinical Specimens Proper sampling and culture of a suspected pathogen is the most reliable
Determination of Pseudomonas aeruginosa by Biochemical Test Methods Test, a Modified Biochemical Test for
 Japan. J. Microbiol. Vol. 14 (4), 279-284, 1970 Determination of Pseudomonas aeruginosa II. Acylamidase by Biochemical Test Methods the Identification Test, a Modified Biochemical Test for of Pseudomonas
Japan. J. Microbiol. Vol. 14 (4), 279-284, 1970 Determination of Pseudomonas aeruginosa II. Acylamidase by Biochemical Test Methods the Identification Test, a Modified Biochemical Test for of Pseudomonas
APPLICATION OF THE GDV-GRAPHY TECHNIQUE FOR THE ESTIMATION OF ANTIGEN-ANTIBODY REACTION
 In: Konstantin G. Korotkov (Ed.). Measuring Energy Fields: Current Research. Backbone Publishing Co. Fair Lawn, USA, 2004. pp.39-42. APPLICATION OF THE GDV-GRAPHY TECHNIQUE FOR THE ESTIMATION OF ANTIGEN-ANTIBODY
In: Konstantin G. Korotkov (Ed.). Measuring Energy Fields: Current Research. Backbone Publishing Co. Fair Lawn, USA, 2004. pp.39-42. APPLICATION OF THE GDV-GRAPHY TECHNIQUE FOR THE ESTIMATION OF ANTIGEN-ANTIBODY
RESISTANCE TO LYSIS FROM WITHOUT IN BACTERIA INFECTED WITH T2 BACTERIOPHAGE'
 RESISTANCE TO LYSIS FROM WITHOUT IN BACTERIA INFECTED WITH T2 BACTERIOPHAGE' N. VISCONTI Department of Genetics, Carnegie Institution of Washington, Cold Spring Harbor, New York Received for publication
RESISTANCE TO LYSIS FROM WITHOUT IN BACTERIA INFECTED WITH T2 BACTERIOPHAGE' N. VISCONTI Department of Genetics, Carnegie Institution of Washington, Cold Spring Harbor, New York Received for publication
A rapid and efficient single-cell manipulation method for screening antigen-specific antibody-secreting cells from human peripheral blood
 Supplementary information A rapid and efficient single-cell manipulation method for screening antigen-specific antibody-secreting cells from human peripheral blood Aishun Jin 1,2,6, Tatsuhiko Ozawa 1,6,
Supplementary information A rapid and efficient single-cell manipulation method for screening antigen-specific antibody-secreting cells from human peripheral blood Aishun Jin 1,2,6, Tatsuhiko Ozawa 1,6,
Diagnostic Microbiology
 Diagnostic Microbiology 320 MIC Lecture: 4 Identification of Microbes 3/8/2014 1 Agglutination and Precipitation Reactions Agglutination testing: Antibody cross links whole-cell antigens, forming complexes
Diagnostic Microbiology 320 MIC Lecture: 4 Identification of Microbes 3/8/2014 1 Agglutination and Precipitation Reactions Agglutination testing: Antibody cross links whole-cell antigens, forming complexes
Secretory Immunoglobulin A and Herpes Keratitis
 INFECTION AND IMMuNrnr, Dec. 1970, p. 778-782 Copyright 1970 American Society for Microbiology Vol. 2, No. 6 Printed in U.S.A Secretory Immunoglobulin A and Herpes Keratitis Y. M. CENTIFANTO AND H. E.
INFECTION AND IMMuNrnr, Dec. 1970, p. 778-782 Copyright 1970 American Society for Microbiology Vol. 2, No. 6 Printed in U.S.A Secretory Immunoglobulin A and Herpes Keratitis Y. M. CENTIFANTO AND H. E.
AGGLUTINATION BY PRECIPITIN. (From the Department of Animal Pathology of The Rockefeller Institute for Medical Research, Princeton, N. Y.
 Published Online: 1 August, 1927 Supp Info: http://doi.org/10.1084/jem.46.2.303 Downloaded from jem.rupress.org on November 11, 2018 AGGLUTINATION BY PRECIPITIN. BY F. S. JONES, V.M.D. (From the Department
Published Online: 1 August, 1927 Supp Info: http://doi.org/10.1084/jem.46.2.303 Downloaded from jem.rupress.org on November 11, 2018 AGGLUTINATION BY PRECIPITIN. BY F. S. JONES, V.M.D. (From the Department
Result:COMPLETE Report Date: December 28 th, 2015
 Send to: Clean Water Environmental, LLC 1939 Talamore Court Southeast, Grand Rapids, MI 49546 Dr. Dale Williams Result:COMPLETE Report Date: December 28 th, 2015 Customer Name: Clean Water Environmental,
Send to: Clean Water Environmental, LLC 1939 Talamore Court Southeast, Grand Rapids, MI 49546 Dr. Dale Williams Result:COMPLETE Report Date: December 28 th, 2015 Customer Name: Clean Water Environmental,
Determination of MIC & MBC
 1 Determination of MIC & MBC Minimum inhibitory concentrations (MICs) are defined as the lowest concentration of an antimicrobial that will inhibit the visible growth of a microorganism after overnight
1 Determination of MIC & MBC Minimum inhibitory concentrations (MICs) are defined as the lowest concentration of an antimicrobial that will inhibit the visible growth of a microorganism after overnight
Determination of MIC & MBC
 1 Determination of MIC & MBC Minimum inhibitory concentrations (MICs) are defined as the lowest concentration of an antimicrobial that will inhibit the visible growth of a microorganism after overnight
1 Determination of MIC & MBC Minimum inhibitory concentrations (MICs) are defined as the lowest concentration of an antimicrobial that will inhibit the visible growth of a microorganism after overnight
Agar-Gel Precipitin-Inhibition Technique for
 APPLIED MICROBIOLOGY, July 1967, p. 794-799 Copyright 1967 American Society for Microbiology Agar-Gel Precipitin-Inhibition Technique for Histoplasmosis Antibody Determinations JOHN G. RAY, JR. Medical
APPLIED MICROBIOLOGY, July 1967, p. 794-799 Copyright 1967 American Society for Microbiology Agar-Gel Precipitin-Inhibition Technique for Histoplasmosis Antibody Determinations JOHN G. RAY, JR. Medical
INHIBITION OF A STAPHYLOCOCCAL HEMOLYSIN BY A SOLUBLE SUBSTANCE PRODUCED BY A NONHEMOLYTIC MICROCOCCUS SPECIES
 INHIBITION OF A STAPHYLOCOCCAL HEMOLYSIN BY A SOLUBLE SUBSTANCE PRODUCED BY A NONHEMOLYTIC MICROCOCCUS SPECIES PINGHUI LIU' Kitasato Institute for Infectious Diseases, Tokyo, Japan Received for publication
INHIBITION OF A STAPHYLOCOCCAL HEMOLYSIN BY A SOLUBLE SUBSTANCE PRODUCED BY A NONHEMOLYTIC MICROCOCCUS SPECIES PINGHUI LIU' Kitasato Institute for Infectious Diseases, Tokyo, Japan Received for publication
typing phages were obtained from John Blair
 SEROLOGY AND TRANSDUCTION IN STAPHYLOCOCCAL PHAGE C. E. DOWELL' AND E. D. ROSENBLUM Department of Microbiology, The University of Texas Southwestern Medical School, Dallas, Texas ABSTRACT DOWELL, C. E.
SEROLOGY AND TRANSDUCTION IN STAPHYLOCOCCAL PHAGE C. E. DOWELL' AND E. D. ROSENBLUM Department of Microbiology, The University of Texas Southwestern Medical School, Dallas, Texas ABSTRACT DOWELL, C. E.
Blocking of Bacteriophage Receptor Sites by Concanavalin A
 Jorrr.iral of General Microbiologj. (19721, 73, 58 I -585 PrititPd in Great Britain Blocking of Bacteriophage Receptor Sites by Concanavalin A By A. R. ARCHIBALD AND HILARY E. COAPES Microbiological Chemistry
Jorrr.iral of General Microbiologj. (19721, 73, 58 I -585 PrititPd in Great Britain Blocking of Bacteriophage Receptor Sites by Concanavalin A By A. R. ARCHIBALD AND HILARY E. COAPES Microbiological Chemistry
Test Method for the Continuous Reduction of Bacterial Contamination on Copper Alloy Surfaces
 Test Method for the Continuous Reduction of Bacterial Contamination on Copper Alloy Surfaces Test Organisms: Staphylococcus aureus (ATCC 6538) Enterobacter aerogenes (ATCC 13048) Pseudomonas aeruginosa
Test Method for the Continuous Reduction of Bacterial Contamination on Copper Alloy Surfaces Test Organisms: Staphylococcus aureus (ATCC 6538) Enterobacter aerogenes (ATCC 13048) Pseudomonas aeruginosa
Human IL-10 ELISA MAX Set Deluxe
 Human IL-10 ELISA MAX Set Deluxe Cat. No. 430604 (5 plates) 430605 (10 plates) 430606 (20 plates) ELISA Set for Accurate Cytokine Quantification from Cell Culture Supernatant, Serum, Plasma or Other Body
Human IL-10 ELISA MAX Set Deluxe Cat. No. 430604 (5 plates) 430605 (10 plates) 430606 (20 plates) ELISA Set for Accurate Cytokine Quantification from Cell Culture Supernatant, Serum, Plasma or Other Body
INOCULATION GROUPS' these groups, has made advisable some study of the antigenic
 INVESTIGATIONS UPON THE ANTIGENIC RELATION- SHIPS AMONG THE ROOT-NODULE BACTERIA OF THE SOYBEAN, COWPEA, AND LUPINE CROSS- INOCULATION GROUPS' 0. A. BUSHNELL AND W. B. SARLES Department of Agricultural
INVESTIGATIONS UPON THE ANTIGENIC RELATION- SHIPS AMONG THE ROOT-NODULE BACTERIA OF THE SOYBEAN, COWPEA, AND LUPINE CROSS- INOCULATION GROUPS' 0. A. BUSHNELL AND W. B. SARLES Department of Agricultural
MICROBIOLOGICAL EXAMINATION OF NON-STERILE PRODUCTS: TEST FOR SPECIFIED MICRO-ORGANISMS Test for specified micro-organisms
 5-2-3. Most-probable-number method Prepare and dilute the sample using a method that has been shown to be suitable as described in section 4. Incubate all tubes at 30-35 C for 3-5 days. Subculture if necessary,
5-2-3. Most-probable-number method Prepare and dilute the sample using a method that has been shown to be suitable as described in section 4. Incubate all tubes at 30-35 C for 3-5 days. Subculture if necessary,
Interferon Immunosuppression: Mediation by a Suppressor
 INFECTION AND IMMUNITY, Aug. 1980, p. 301-305 0019-9567/80/08-0301/05$02.00/0 Vol. 29, No. 2 Interferon Immunosuppression: Mediation by a Suppressor Factor HOWARD M. JOHNSON* AND J. EDWIN BLALOCK Department
INFECTION AND IMMUNITY, Aug. 1980, p. 301-305 0019-9567/80/08-0301/05$02.00/0 Vol. 29, No. 2 Interferon Immunosuppression: Mediation by a Suppressor Factor HOWARD M. JOHNSON* AND J. EDWIN BLALOCK Department
PSEUDOMONAS-CF-IgG ELISA KIT
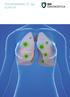 PSEUDOMONAS-CF-IgG ELISA KIT PSEUDOMONAS-CF-IgG ELISA Kit is for the quantitative measurement of the antibody level to P. aeruginosa in human serum samples. Application Chronic P. aeruginosa infection
PSEUDOMONAS-CF-IgG ELISA KIT PSEUDOMONAS-CF-IgG ELISA Kit is for the quantitative measurement of the antibody level to P. aeruginosa in human serum samples. Application Chronic P. aeruginosa infection
Antigen-Antibody. reactions (2) By: Masheal Aljumaah OCT 2018
 Antigen-Antibody reactions (2) By: Masheal Aljumaah OCT 2018 Learning objectives: introduction to Antigen Antibody reactions. Antigen Antibody reactions part1: Precipitation, Flocculation and Immunodiffusion.
Antigen-Antibody reactions (2) By: Masheal Aljumaah OCT 2018 Learning objectives: introduction to Antigen Antibody reactions. Antigen Antibody reactions part1: Precipitation, Flocculation and Immunodiffusion.
