Note Application of semi-selective mediums in routine diagnostic testing of Pseudomonas savastanoi pv. phaseolicola on common bean seeds
|
|
|
- Clement Snow
- 5 years ago
- Views:
Transcription
1 265 Scientia Psp Agricola routine testing on bean seed Note Application of semi-selective mediums in routine diagnostic testing of Pseudomonas savastanoi pv. phaseolicola on common bean seeds Tatjana Popovi 1 *, Predrag Milovanovi 2, Goran Aleksi 1, Veljko Gavrilovi 1, Mira Starovi 1, Mirjana Vasi 3, Jelica Balaž 4 1 Institute for Plant Protection and Environment, Teodora Drajzera, 9, PO Box Belgrade Republic of Serbia. 2 Galenika/Fitofarmacija, Batajni ki drum b.b Belgrade Republic of Serbia. 3 Institute for Field and Vegetable Crops, Maksima Gorkog Novi Sad Republic of Serbia. 4 University of Novi Sad/Faculty of Agriculture, Dositeja Obradovi a Novi Sad Republic of Serbia. *Corresponding author<tanjaizbis@gmail.com> Edited by: Cláudio Marcelo Gonçalves de Oliveira Received March 03, 2011 Accepted November 22, 2011 ABSTRACT: Halo blight, caused by Pseudomonas savastanoi pv. phaseolicola (Psp), is considered to be an important bacterial disease on common bean (Phaseolus vulgaris L.) in Serbia. Use of pathogen-free seeds is one of the most effective control measures against this disease. The aim of this study was to evaluate a detection method for Psp on untreated common bean seeds (23 genotypes) from commercial crops grown within Serbia. Detection of this pathogen was made by plating onto the modified sucrose peptone (MSP) and Milk Tween (MT) semi-selective mediums from soaked whole common bean seed. Colonies growing on the MSP medium were light yellow, convex and shiny, whereas on the MT medium, they were creamy white, flat and circular. The pathogenicity of the obtained strains was confirmed by the inoculation of germinated bean seed. The isolates recovered from the seed assay were further confirmed to be Psp by using both Enzyme-linked immunosorbent assay (ELISA) and polymerase chain reaction (Nested-PCR) detection methodologies. The International Seed Testing Association (ISTA) method selected for this work was found to be effective in detecting the presence of Psp in common bean seed. The bacterium Psp was detected in only two of the 23 seed samples analyzed by this method, which shows that the bacterium is not widespread in Serbia. Keywords: MSP, MT, ELISA, Nested-PCR, halo blight, pathogenicity Introduction Pseudomonas savastanoi pv. phaseolicola (Burkholder) Gardan et al. (Psp) which causes halo blight of bean (Phaseolus vulgaris L.) is a pathogen present in all countries where beans are cultivated. The disease is induced by cool temperatures and humidity and is widespread in Europe and North America. Many states and countries have strict quarantine laws regarding Psp (Grogan and Kimble, 1967; Taylor, 1970; Taylor et al., 1979; Webster et al., 1983). Halo blight was the main problem in bean production in Serbia during the s (Balaž, 1990). Earlier cases have been reported where snap bean crops have completely collapsed in years where the spring weather was cool and rainy. However, in recent years, Psp has not been a major problem, which can probably be contributed to the weather conditions (the emergence and first phase of bean development generally follow warmer and drier weather conditions) and the use of resistant bean cultivars. Pseudomonas savastanoi pv. phaseolicola is a seedborne pathogen (Fahy and Lloyd, 1983). The detection of bacterium on seeds is essential for effective control of the disease. Several methods have been described for testing the presence of Psp in bean seed: a seed soakplant inoculation technique (Lahman and Schaad, 1985; Webster et al., 1983), immunological methods (Guthrie et al., 1965; Van Vuurde et al., 1983), plating on King B agar (Van Vuurde and Van den Bovenkamp, 1987) and semiselective modified sucrose peptone (MSP) agar (Mohan and Schaad, 1987). Various PCR assays have been proposed for the detection of Psp (Prosen et al., 1993; Schaad et al., 1995, 2001; Mosqueda and Herrera, 1997). All of them rely on the detection of DNA sequences involved in the biosynthesis of phaseolotoxin (Prosen et al., 1993; Schaad et al., 1995; Audy et al., 1996; Güven et al., 2004). Considering the necessity for the development of a practical and effective method for the routine analysis of bean seed in Serbia, this study was carried out using Psp isolation in semi-selective mediums developed for this purpose. Pathogenicity testing and rapid ELISA and PCR methods were used to confirm identification of obtained strains. Materials and Methods Untreated common bean seeds from 23 genotypes grown in Serbia (the Novi Sad area 45º15 N and E) were analyzed for the presence of Psp. There were 12 samples of Serbian dry bean cultivars (Balkan, Belko, Dvadesetica, Galeb, Maksa, Medijana, Oplenac, Panonski gradistanac, Panonski tetovac, Slavonski zutozeleni, Sremac, and Zlatko) and 11 samples of foreign common bean genotypes from collection (Oreol, Dobrudzanski rani, Dobrudzanski rani 7, KB 100 and KB 101 from Bulgaria; C 20, Naya Nayahit, HR 45, Xan 159, Xan 208 and Xan 273 from USA). All samples came from untreated commercial crops. Five 1000-seed sub-samples were gathered for each cultivar, with a total of 115 sub-samples. Pure culture of Psp reference strain (Ps12, Faculty of Agriculture, Novi Sad) was used in all assays.
2 266 Extraction of bacteria from the seed Each 1000-seed sub-sample was first washed in running water and then placed on sterile laboratory paper to dry. After that, sub-samples were weighed and soaked in seed extraction solution (0.85 % saline with Tween 20) in the proportion of 1:2 (1 g of seed in 2 ml of solution) for 24 h in the fridge (5 ºC). After soaking, the liquid was sampled. Extraction of the bacterium from whole bean seeds was done according to Jansing and Rudolph (1990), ISF (2006) and Kurowski and Remeeus (2007, 2008) methods. Isolation on semi-selective media After incubating, 0.1 ml of undiluted extracts obtained from the seeds and their 10-fold dilution series (to 10 5 ) were plated onto the surface of two semi-selective media, Modified Sucrose Peptone Agar (MSP) and Milk Tween Agar (MT), described by Mohan and Schaad (1987) and Goszczynska and Serfontein (1998), respectively. The series of dilution were prepared with the aim to get single colonies of Psp which could be morfologically differentiated from other non-targeted organisms. Two Petri dishes were plated for each concentration. A pure culture of Psp reference strain (Ps12) was also plated on both semi-selective media. The Petri dishes were incubated at ºC for five days. After that, the sample plates were visually assessed for the presence of colonies with typical Psp morphology by comparison with the reference strain. Suspected colonies, as well as the reference strain, were transferred onto a King s B agar (King et al., 1954) and incubated for three-four days at 27 C. The transferred colonies were again compared with the reference strain. Six typical strains from each positive sample (12 representative strains) were selected for identification (Table 1). Pathogenicity test Suspected Psp bacterial colonies were evaluated using pathogenicity testing on the susceptible bean cultivar Oplenac, by the inoculation of germinated seed (Fenwick and Guthrie, 1969; Van Vuurde and Van den Table 1 Isolation of Psp from naturally contaminated bean seeds. Strains Bean cultivar Medium TP106 Dvadesetica MSP TP108 Dvadesetica MSP TP127 Dvadesetica MSP TP114 Dvadesetica MT TP117 Dvadesetica MT TP118 Dvadesetica MT TP232 Oplenac MSP TP233 Oplenac MSP TP234 Oplenac MSP TP229 Oplenac MT TP230 Oplenac MT TP231 Oplenac MT Bovenkamp, 1987, 1989; ISF, 2006). Susceptibility of the cultivar Oplenac to the Psp strains from Serbia had been confirmed and described by Balaž (1990). The seeds were incubated in rolled germination paper for four days at 25 C in the dark ness. To provide better conditions for infection, seedlings were sprayed with water two hours before inoculation. Inoculation was made with a sterile toothpick, which was dipped into the bacterial culture of obtained strains growing on the King B medium for two days. The toothpick was pushed through the cotyledon. After that, the toothpick was turned slightly while being pulled out in order to release the bacteria. Four seedlings were inoculated per every representative strain. The positive (reference culture) and negative (sterile water) controls were prepared in a similar manner. The seedlings were placed in a phytotron at 20 C with 80 % relative humidity and light adequate for plant growth (light:dark 12:12). The symptoms on the inoculated seedlings were recorded after five and 10 days and compared with the positive and negative controls. Pathogenicity was evaluated by the inspection of the flat inner sides of the cotyledons to determine the presence of typical greasy spots at the point of inoculation. Serological and molecular identification All strains were identified as Psp by the rapid tests, ELISA (Enzyme-linked immunosorbent assay) and PCR (polymerase chain reaction), regardless of their pathogenicity. ELISA - Double-antibody sandwich (DAS)-ELISAs and Plate Trapped Antigen (PTA)-ELISAs were performed with commercial kits by Loewe Biochemica GmbH, Germany and ADGEN Phytodiagnostics, Neogen Europe Ltd., Scotland, U.K., respectively. The assays followed the manufacturer s instructions. Bacterial suspensions were prepared in sterile water with pure bacterial cultures grown on the King B for 48 h at 27 C. A reference strain of Psp was used as a positive control, while a reference strain of Erwinia amylovora (NCPPB 595) was used as the negative control. PCR - Nested PCR was conducted with DNA extracted from pure bacterial cultures (Schaad et al., 2001). Cultures were grown on the King B medium at 27 C for 48 h, and cells from 0.5 ml of water suspension ( CFU ml 1 ) were used for DNA extraction. The method of Schaad et al. (1995) for the detection of phaseolotoxin genes was modified by Güven et al. (2004). 0.5 ml of cell suspension were boiled for 15 min. The cell debris was removed by centrifugation for 10 min at rpm. 2 µl of supernatant were used for amplification. For the first PCR primers P 5.1: 5 -AGC TTC TCC TCA AAA CAC CTG C- 3 and P 3.1: 5 -TGT TCG CCA GAG GCA GTC ATG-3 were used, as suggested by Schaad et al. (1995). Primers P 5.1 and P 3.1 directed the amplification of the 500-bp DNA fragment. For the second PCR, primers P
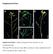
3 : 5 -TCG AAC ATC AAT CTG CCA GCC A-3 and P 3.2: 5 -GGC TTT TAT TAT TGC CGT GGG C-3 were used, as suggested by Schaad et al. (2001). Primers P 5.2 and P 3.2 directed the amplification of the 450-bp DNA fragment. A reference strain of Psp (Ps12) was used as a positive control and a reference strain of E. amylovora (NCPPB 595) was used as the negative control. The PCR amplification assay was performed in a 25 µl reaction mixture containing Taq DNA polymerase 1.25 U, 50 mm KCl, 30 mm Tris-HCl, 1.5 mm Mg 2+, 0.1 % igepal-ca630, 200 µm dntp, 0.4 µm of primers and 1 µl of DNA.A Mastercycler ep gradient S (Eppendorf, Germany) was used for PCR with the following profile amplifications: an initial 3 min incubation at 94 C, a manual hot start step at 80 C, 25 cycles (1 min at 94 C, 1 min at 58 C and 1 min at 72 C), and a final extension step of 10 min at 72 C. After the first PCR, products were diluted 10x and 2 µl were used for the second PCR. The amplified DNA fragments were electrophoresed in 2.0 % agarose gels in 1xTBE buffer and visualized with ultraviolet light after ethidium bromide staining. Results The presence of Psp on the common bean seeds was observed using the method of isolation on semi-selective media following the ISTA method. This method was supplemented with ELISA and Nested-PCR methods in order to confirm the identity of obtained strains. The method was effective in isolating Psp colonies from infected common bean seeds. All the obtained strains were identified as Psp by the pathogenicity and rapid tests ELISA and PCR. The analysis of 23 common bean seed samples from the Novi Sad locality in Serbia revealed that only two of them (Dvadesetica and Oplenac) were infected with Psp (Table 1). Not a single suspected bacterium colony formed on the 21 bean genotypes (Balkan, Belko, C 20, Dobrudzanski rani, Dobrudzanski rani 7, Galeb, HR 45, KB 100, KB 101, Maksa, Medijana, Naya Nayahit, Oreol, Panonski gradistanac, Panonski tetovac, Slavonski zutozeleni, Sremac, Xan 159, Xan 208, Xan 273 and Zlatko). After four days of incubation on the MSP medium colonies of Psp formed in dilutions. They were light yellow, convex, shiny, and 2-3 mm in diameter (Figure 1). The medium around the colony turned light yellow. On MT, colonies of Psp were formed in dilutions after four days of incubation. They were creamy white, flat, circular, and 3-5 mm in diameter (Figure 2). Furthermore, in both samples of common bean seeds (cultivar Dvadesetica and Oplenac) on the same medium, colonies of the bacterium Xanthomonas axonopodis pv. phaseoli (Smith) Vauterin et al. (Xap) also formed. Colonies were yellow, convex and mucoid and were surrounded by two zones of hydrolysis, a large clear zone of casein hydrolysis and a smaller milky zone of Tween 80 hydrolysis (, 2010), indicating that the seeds were infected with both bacteria (Figure 2). MT medium can also be used to develop colonies of the bacterium Pseudomonas syringae pv. syringae. This bacterium can be distinguished from Psp using a differential tests described by Lelliott and Stead (1987). The selected colonies grown on King B medium were creamy white and formed a green fluorescent pigment. The presence of the suspect strains (Table 1) was confirmed by pathogenicity testing. On the inoculated bean cotyledons characteristic dark green, greasy spots formed at the point of inoculation after five days (Figure 3a). After 10 days, the whole cotyledon was affected with small greasy spots (Figure 3b). Obtained strains of Psp were tested by DAS- and PTA-ELISA. The polyclonal antibody used reacted as expected with all the strains, producing clear positive reactions which indicate that strains of Psp can be detected using commercial ELISA tests. All bacterial strains submitted to the Nested-PCR test were identified as Psp. The used P 5.1/P 3.1 and P 5.2/P 3.2 primers pair directed the amplification of the 450-bp target DNA fragment (Figure 4). Discussion The bacterium Psp is a seed-borne pathogen (Taylor, 1970). Seeds contaminated internally or externally are the primary source of infection (Schwartz, 1989). Very low levels of inoculum, such as a single contaminated seed in 2000 or 5000, can result in an epidemic in the field under favorable environmental conditions (Webster et al., 1983). Use of healthy, pathogen-free seed is one of the most effective control measures against halo blight disease. In Serbia, commercial bean seed is used for planting, from native varieties predominate in the production. Certified seeds are seldom used, and even in the case of the new domestic cultivars for which seed production has been organized, it is used only in the first production year (Todorovi et al., 2008; Popov et al., 2010). The overall objective of this study was to evaluate the detection method and determine the occurrence of Psp on common bean seed collected from different genotypes and populations in Serbia. A number of plating assays and semi-selective media have been developed and improved to detect Psp in bean seed (Mohan and Schaad, 1987; Gozczynska and Serfontein, 1998). We conducted a detection method by sowing the extract obtained from the soaked whole bean seed onto the semi-selective media MSP and MT. This method was previously reported on the artificial infected bean seed by Balaž et al. (2008). The results of our study showed that the direct plating method can be routinely used for the detection of the Psp on bean seed, because of its reliability. The Psp colonies that formed on the semi-selective medium MSP after four days of incubation were light yellow, convex and shiny and medium around the colony became turned light yellow (Mohan and Schaad,
4 268 Figure 1 View of bacteria colonies from the bean cultivar Oplenac on MSP medium, different dilutions. Figure 2 View of bacteria colonies from the bean cultivar Oplenac on MT medium, dilutions Figure 3 Symptoms on inoculated cotyledon of bean, strain TP233; A) after five days, B) after ten days. Figure 4 Amplification of a 450 bp DNA fragment from the phaseolotoxin gene using P 5.1/P 3.1 and P 5.2/P 3.2 primers. 1987; Jansing and Rudolph, 1990, 1996; Kurowski and Remeeus, 2008). The colonies formed on the semi-selective medium MT are creamy white, flat and circular after four-five days of incubation (Goszczynska and Serfontein, 1998; ISF, 2006; Kurowski and Remeeus, 2008). Many colonies of Psp on MT can form a pale-blue fluorescent pigment when using UV light (ISF, 2006). Goszczynska and Serfontein (1998) described the use of the semi-selective medium MT for routine seed testing in the laboratory and for the detection and distinction of all bacterial pathogens screened for in dry bean seed and plant material. This study confirmed the advantage of the MT medium, which allowed simultaneous detection of Xap and Psp on the tested bean cultivar Oplenac, where seeds were infected with both of the bacteria. Other media such as King B (Taylor, 1970), KBC (Mohan and Schaad, 1987; NSHS, 2002) and LPGA can also be used for the isolation of Psp from bean seeds. Pathogenicity tests were carried out to establish if the obtained strains were typically pathogenic. Psp formed dark green, greasy spots on inoculated bean cotyledons. The rapid confirmation to identify the obtained strains in this assay as Psp was achieved by using ELISA and PCR tests. When using ELISA test in identifying Psp 10 4 bacteria in 1 ml can be detected (Barzic and Trigalet, 1982), while the use of the other serological method immunefluorescence (IF) makes it possible to detect 10 2 cells in 1 ml (Bazzi and Calzolari, 1982). Van Vuurde and Van den Bovenkamp (1987, 1989) reported that IF is a fast, simple and cheap method for the detection of Psp on bean seed, but they also noted that the reliability of the results depends on the specific and appropriate solutions of antisera. Van Vuurde et al. (1983) reported the application of IF and ELISA tests as potential routine tests for the detection of Psp and Xap in bean seed. Serological methods were considered to be an important diagnostic test in the detection of bacteria causing blight of bean. Certainly, there are numerous restrictions and warnings that must be done before a method is standardized and accepted for routine seed health testing (Weaver and Guthrie, 1978; Sheppard et al., 1986). The nested-pcr assay with the specific primers pair P5.1/P3.1 and P5.2/P3.2, which directed the amplification of the 500-bp and 450-bp DNA fragments, respectively (Schaad et al., 2001), was included to confirm the identity of all the obtained strains from this study. Audy
5 269 et al. (1996) reported the possibility of using the PCR method for simultaneous detection of Xap and Psp in bean seed. A combination of specific primers was used to detect two bacteria specifically, but the other tested pathogenic bacteria did not give positive results. The method is sensitive, can detect as few as 1 infected seed in seeds, and has a great potential for the detection of these two bacteria in commercial seeds (Audy et al., 1996). The presence of Psp was recorded on only two out of 23 common bean seed samples analyzed from the Novi Sad area in Serbia. The lower number of seed samples infected by Psp indicated that the bacterium is not widespread, which is attributed to the warmer and drier weather conditions in Serbia in recent years as well as to the use of halo blight resistant bean cultivars. Presented results provide a good basis for a widely routine use, because the detection method is applicable, fast, reliable and cheap and therefore recommended for the use by the phytosanitary service in Serbia. References Audy, P.; Braat, C.E.; Saindon, G.; Huang, H.C.; Laroche, A A rapid and sensitive PCR-based assay for concurrent detection of bacteria causing common and halo blights in bean seed. Phytopathology 86: Balaž, J Contribution to the study of string-bean and bean susceptibility to Pseudomonas syringae pv. phaseolicola (Burkholder) Young, Dye et Wilkie. Zaštita bilja 41: (in Serbian, with abstract in English). Balaž, J.; Popovi, T.; Vasi, M.; Nikoli, Z Elaboration of methods for detection of Pseudomonas savastanoi pv. phaseolicola on bean seeds. Pesticidi i Fitomedicina 23: (in Serbian, with abstract in English). Barzic, M.R.; Trigalet, A Detection de Pseudomonas phaseolicola (Burkh.) Dowson par la technique ELISA. Agronomie 2: Bazzi, C.; Calzolari, A Detection of Pseudomonas phaseolicola (Burkh.) Dowson in bean seed with immunofluorescence staining (indirect method). Informatore Fitopatologico 32: (in Italian). Fahy, P.C.; Lloyd, A.B Pseudomonas: The Flourescent Pseudomonads. p In: Fahy, P.C.; Persley, G.J., eds. Plant bacterial diseases: a diagnostic guide. Academic Press, Sydney, Australia. Fenwick, H.S.; Guthrie, J.W An improved Pseudomonas phaseolicola pathogenicity test. Phytopathology 59: 11. Goszczynska, T.; Serfontein, J.J Milk tween agar, a semiselective medium for isolation and differentiation of Pseudomonas syringae pv. syringae, Pseudomonas syringae pv. phaseolicola and Xanthomonas axonopodis pv. phaseoli. Journal of Microbiological Methods 32: Guthrie, J.W.; Huber, D.M.; Fenwick, H.S Serological detection of halo blight. Plant Disease Reporter 49: Grogan, R.G.; Kimble, K.A The role of seed contamination in the transmission of Pseudomonas phaseolicola in Phaseolis vulgaris. Phytopathology 57: Güven, K.; Jones, J.B.; Momol, M.T.; Dickstein, E.R Phenotypic and genetic diversity among Pseudomonas syringae pv. phaseolicola. Journal of Phytopathology 152: International Seed Testing (ISF) Method for the Detection of Pseudomonas savastanoi pv. phaseolicola on Bean Seed. ISHI- Veg, Nyon, Switzerland. Available at: org/cms/medias/file/tradeissues/phytosanitarymatters/ SeedHealthTesting/ISHI-Veg [Accessed Aug. 2, 2011] Jansing, H.; Rudolph, K A sensitive and quick test for determination of bean seed infestation by Pseudomonas syringae pv. phaseolicola. Journal Zeitschrift für Pflanzenkrankheiten und Pflanzenschutz 97: Jansing, H.; Rudolph, K Pseudomonas syringae pv. phaseolicola: ISTA Handbook on Seed Health Testing. International Seed Testing Association, Zurich, Switzerland. (Working Sheet, 66). King, E.O.; Ward, M.K.; Raney, D.E Two simple media for the demonstration of pyocyanin and fluorescin. Journal of Laboratory and Clinical Medicine 44: Kurowski, C.; Remeeus, P.M Proposal for a new method for detecting Pseudomonas savastanoi pv. phaseolicola on bean seeds. ISTA Method Validation Reports 4: Available at: [Accessed Jan. 1, 2007] Kurowski, C.; Remeeus, P.M Detection of Pseudomonas savastanoi pv. phaseolicola on Phaseolus vulgaris. International Seed Testing Association, Bassersdorf, Switzerland. (International Rules for Seed Testing, 7-023). Available at: [Accessed Jan. 1, 2008] Lahman, L.K.; Schaad, N.W Evaluation of the dome test as a reliable assay for seedborne bacterial blight pathogens of beans. Plant Disease 69: Lelliott, R.A.; Stead, D.E Methods for the Diagnosis of Bacterial Diseases of Plants. Blackwell, Oxford, UK. Mohan, S.K.; Schaad, N.W An improved agar plating assay for detecting Pseudomonas syringae pv. syringae and P. s. pv. phaseolicola in contaminated bean seed. Phytopathology 77: Mosqueda, C.G.; Herrera, E.L A simple and efficient PCR method for the specific detection of Pseudomonas syringae pv. phaseolicola in bean seeds. World Journal of Microbiology & Biotechnology 13: National Seed Health System [NSHS] Reference manual B (RM-B): seed health testing and phytosanitary field inspection methods manual. Available at: pdf/rmb0402.pdf. [Accessed Sep. 18, 2002] Popovi, T.; Balaž, J.; Nikoli, Z.; Starovi, M.; Gavrilovi, V.; Aleksi, G.; Vasi, M.; Živkovi, S Detection and identification of Xanthomonas axonopodis pv. phaseoli on bean seed collected in Serbia. African Journal of Agricultural Research 5: Prosen, D.; Hatziloukas, E.; Schaad, N.W.; Panopoulos, N.J Specific detection of Pseudomonas syringae pv. phaseolicola DNA in bean seed by polymerase chain reaction-based amplification of a phaseolotoxin gene region. Phytopathology 83:
6 270 Schaad, N.W.; Cheong, S.S.; Tamaki, S.; Hatziloukas, E.; Panopoulos, N.J A combined biological and enzymatic amplification (BIO-PCR) technique to detect Pseudomonas syringae pv. phaseolicola in bean seed extracts. Phytopathology 85: Schaad, N.W.; Jones, J.B.; Chun, W Laboratory Guide for Identification of Plant Pathogenic Bacteria. APS Press, St. Paul, MN, USA. Schwartz, H.F Halo blight. In: Schwartz, H.F.; Pastor- Coralles, M.A., eds. Bean production problems in the tropics. Centro Internacional de Agricultura Tropical, Cali, Colombia. Sheppard, J.W.; Wright, P.F.; DeSavingy, D.H Methods for the evaluation of EIA for use in the detection of seed-borne diseases. Seed Science and Technology 14: Taylor, J.D The quantitative estimation of the infection of bean seed with Pseudomonas phaseolicola (Burkh.) Dowson. Annals of Applied Biology 66: Taylor, J.D.; Dudley, C.L.; Presly, L Studies of halo-blight seed infection and disease transmission in dwarf beans. Annals of Applied Biology 93: Todorovi, J.; Vasi, M.; Todorovi, J Pasulj i Boranija. Institute for Vegetable and Field Crops. Novi Sad, Serbia.. Van Vuurde, J.W.L.; Van den Bovenkamp, G.W Pseudomonas syringae pv. phaseolicola:. ISTA Handbook on Seed Health Testing. International Seed Testing Association. Zurich, Switzerland. (Working Sheet, 65). Van Vuurde, J.W.L.; Van den Bovenkamp, G.W Detection of Pseudomonas syringae pv. phaseolicola in bean. p In: Saettler, A.W.; Schaad, N.W.; Roth, D.A., eds. Detection of bacteria in seed and other planting material, The American Phytopathological Society Press, St. Paul, USA. Van Vuurde, J.W.L.; Van den Bovenkamp, G.W.; Birnbaum, Y Immunofluorescence microscopy and enzymelinked immunosorbent assay as potential routine tests for the detection of Pseudomonas syringae pv. phaseolicola and Xanthomonas campestris pv. phaseoli in bean seed. Seed Science and Technology 11: Weaver, W.M.; Guthrie, J.W Enzyme linked immunospecific assay: application to the detection of seed-borne bacteria. Phytopathology News 12: 156. Webster, D.M.; Atkin, J.D.; Cross, J.E Bacterial blights of snap beans and their control. Plant Disease 67:
Detection and identification of Xanthomonas axonopodis pv. phaseoli on bean seed collected in Serbia
 African Journal of Agricultural Research Vol. 5(19), pp. 2730-2736, 4 October, 2010 Available online at http://www.academicjournals.org/ajar ISSN 1991-637X 2010 Academic Journals Full Length Research Paper
African Journal of Agricultural Research Vol. 5(19), pp. 2730-2736, 4 October, 2010 Available online at http://www.academicjournals.org/ajar ISSN 1991-637X 2010 Academic Journals Full Length Research Paper
ISTA Method Validation Reports
 ISTA Method Validation Reports Published by: The International Seed Testing Association P.O. Box 308 8303 Bassersdorf, CH-Switzerland Volume 2006 Copyright 2006 by the International Seed Testing Association
ISTA Method Validation Reports Published by: The International Seed Testing Association P.O. Box 308 8303 Bassersdorf, CH-Switzerland Volume 2006 Copyright 2006 by the International Seed Testing Association
ISTA Method Validation Reports
 ISTA Method Validation Reports Published by: The International Seed Testing Association (ISTA) P.O. Box 308 8303 Bassersdorf, CH-Switzerland Volume 2007/1: 500 copies Copyright 2007 by the International
ISTA Method Validation Reports Published by: The International Seed Testing Association (ISTA) P.O. Box 308 8303 Bassersdorf, CH-Switzerland Volume 2007/1: 500 copies Copyright 2007 by the International
Method for the Detection of Xanthomonas spp. on Pepper Seed
 Method for the Detection of Xanthomonas spp. on Pepper Seed Crop: Pathogen: Pepper (Capsicum annuum) Xanthomonas euvesicatoria, Xanthomonas vesicatoria and Xanthomonas gardneri See (1) and (2) for an account
Method for the Detection of Xanthomonas spp. on Pepper Seed Crop: Pathogen: Pepper (Capsicum annuum) Xanthomonas euvesicatoria, Xanthomonas vesicatoria and Xanthomonas gardneri See (1) and (2) for an account
7-024: Detection of Pea Early-Browning Virus and Pea Seed-borne Mosaic Virus on Pisum sativum (pea)
 International Rules for Seed Testing Annexe to Chapter 7: Seed Health Testing Methods 7-024: Detection of Pea Early-Browning Virus and Pea Seed-borne Mosaic Virus on Pisum sativum (pea) Published by the
International Rules for Seed Testing Annexe to Chapter 7: Seed Health Testing Methods 7-024: Detection of Pea Early-Browning Virus and Pea Seed-borne Mosaic Virus on Pisum sativum (pea) Published by the
Method for the Detection of Lettuce mosaic virus on Lettuce Seed and Seedlings
 Method for the Detection of Lettuce mosaic virus on Lettuce Seed and Seedlings Crop: Pathogen: Lactuca sativa Lettuce mosaic virus (LMV) Revision history: Version 4.3, February 2017 Sample and sub-sample
Method for the Detection of Lettuce mosaic virus on Lettuce Seed and Seedlings Crop: Pathogen: Lactuca sativa Lettuce mosaic virus (LMV) Revision history: Version 4.3, February 2017 Sample and sub-sample
Halo Blight. Photograph provided by H. F. Schwartz AgImage - Colorado State University
 Halo Blight Photograph provided by H. F. Schwartz AgImage - Colorado State University Halo blight, caused by Pseudomonas syringae pv. phaseolica (Burkh.) Dows, is an important seed-borne disease of common
Halo Blight Photograph provided by H. F. Schwartz AgImage - Colorado State University Halo blight, caused by Pseudomonas syringae pv. phaseolica (Burkh.) Dows, is an important seed-borne disease of common
Speed Detection of Pests in China
 INVASIVE ALIEN SPECIES AND THE IPPC, September 2003,Germany Speed Detection of Pests in China Ning Hong (Agriculture Ministry of China) Phytosanitary falls back on technology. Today, with the rapidly development
INVASIVE ALIEN SPECIES AND THE IPPC, September 2003,Germany Speed Detection of Pests in China Ning Hong (Agriculture Ministry of China) Phytosanitary falls back on technology. Today, with the rapidly development
Add 5µl of 3N NaOH to DNA sample (final concentration 0.3N NaOH).
 Bisulfite Treatment of DNA Dilute DNA sample to 2µg DNA in 50µl ddh 2 O. Add 5µl of 3N NaOH to DNA sample (final concentration 0.3N NaOH). Incubate in a 37ºC water bath for 30 minutes. To 55µl samples
Bisulfite Treatment of DNA Dilute DNA sample to 2µg DNA in 50µl ddh 2 O. Add 5µl of 3N NaOH to DNA sample (final concentration 0.3N NaOH). Incubate in a 37ºC water bath for 30 minutes. To 55µl samples
Supplemental Data. The Survival Advantage of Olfaction. in a Competitive Environment. Supplemental Experimental Procedures
 Supplemental Data The Survival Advantage of Olfaction in a Competitive Environment Kenta Asahina, Viktoryia Pavlenkovich, and Leslie B. Vosshall Supplemental Experimental Procedures Experimental animals
Supplemental Data The Survival Advantage of Olfaction in a Competitive Environment Kenta Asahina, Viktoryia Pavlenkovich, and Leslie B. Vosshall Supplemental Experimental Procedures Experimental animals
PHYTOSANITARY PROCEDURES
 EPPO Standards PHYTOSANITARY PROCEDURES PSEUDOMONAS SYRINGAE PV. PERSICAE SAMPLING AND TEST METHODS PM 3/44(1) English oepp eppo Organisation Européenne et Méditerranéenne pour la Protection des Plantes
EPPO Standards PHYTOSANITARY PROCEDURES PSEUDOMONAS SYRINGAE PV. PERSICAE SAMPLING AND TEST METHODS PM 3/44(1) English oepp eppo Organisation Européenne et Méditerranéenne pour la Protection des Plantes
Supplement 1: Sequences of Capture Probes. Capture probes were /5AmMC6/CTG TAG GTG CGG GTG GAC GTA GTC
 Supplementary Appendixes Supplement 1: Sequences of Capture Probes. Capture probes were /5AmMC6/CTG TAG GTG CGG GTG GAC GTA GTC ACG TAG CTC CGG CTG GA-3 for vimentin, /5AmMC6/TCC CTC GCG CGT GGC TTC CGC
Supplementary Appendixes Supplement 1: Sequences of Capture Probes. Capture probes were /5AmMC6/CTG TAG GTG CGG GTG GAC GTA GTC ACG TAG CTC CGG CTG GA-3 for vimentin, /5AmMC6/TCC CTC GCG CGT GGC TTC CGC
strain devoid of the aox1 gene [1]. Thus, the identification of AOX1 in the intracellular
![strain devoid of the aox1 gene [1]. Thus, the identification of AOX1 in the intracellular strain devoid of the aox1 gene [1]. Thus, the identification of AOX1 in the intracellular](/thumbs/72/66361212.jpg) Additional file 2 Identification of AOX1 in P. pastoris GS115 with a Mut s phenotype Results and Discussion The HBsAg producing strain was originally identified as a Mut s (methanol utilization slow) strain
Additional file 2 Identification of AOX1 in P. pastoris GS115 with a Mut s phenotype Results and Discussion The HBsAg producing strain was originally identified as a Mut s (methanol utilization slow) strain
MacBlunt PCR Cloning Kit Manual
 MacBlunt PCR Cloning Kit Manual Shipping and Storage MacBlunt PCR Cloning Kits are shipped on dry ice. Each kit contains a box with cloning reagents and an attached bag with Eco-Blue Competent Cells (optional).
MacBlunt PCR Cloning Kit Manual Shipping and Storage MacBlunt PCR Cloning Kits are shipped on dry ice. Each kit contains a box with cloning reagents and an attached bag with Eco-Blue Competent Cells (optional).
Cat. # Product Size DS130 DynaExpress TA PCR Cloning Kit (ptakn-2) 20 reactions Box 1 (-20 ) ptakn-2 Vector, linearized 20 µl (50 ng/µl) 1
 Product Name: Kit Component TA PCR Cloning Kit (ptakn-2) Cat. # Product Size DS130 TA PCR Cloning Kit (ptakn-2) 20 reactions Box 1 (-20 ) ptakn-2 Vector, linearized 20 µl (50 ng/µl) 1 2 Ligation Buffer
Product Name: Kit Component TA PCR Cloning Kit (ptakn-2) Cat. # Product Size DS130 TA PCR Cloning Kit (ptakn-2) 20 reactions Box 1 (-20 ) ptakn-2 Vector, linearized 20 µl (50 ng/µl) 1 2 Ligation Buffer
SUPPLEMENTARY MATERIALS AND METHODS. E. coli strains, plasmids, and growth conditions. Escherichia coli strain P90C (1)
 SUPPLEMENTARY MATERIALS AND METHODS E. coli strains, plasmids, and growth conditions. Escherichia coli strain P90C (1) dinb::kan (lab stock) derivative was used as wild-type. MG1655 alka tag dinb (2) is
SUPPLEMENTARY MATERIALS AND METHODS E. coli strains, plasmids, and growth conditions. Escherichia coli strain P90C (1) dinb::kan (lab stock) derivative was used as wild-type. MG1655 alka tag dinb (2) is
Kasetsart J. (Nat. Sci) 32 : (1998) Sutruedee Prathuangwong and Khanitha Khandej ABSTRACT
 Kasetsart J. (Nat. Sci) 32 : 84-89 (1998) Development of the New Methods for Ecological Study of Soybean Bacterial Pustule : An Artificial Inoculation Method of Soybean Seed with Xanthomonas campestris
Kasetsart J. (Nat. Sci) 32 : 84-89 (1998) Development of the New Methods for Ecological Study of Soybean Bacterial Pustule : An Artificial Inoculation Method of Soybean Seed with Xanthomonas campestris
Regional Disease Surveillance workshop for banana Rwanda 25 th -29 th January IITA-pathology,Uganda
 Regional Disease Surveillance workshop for banana Rwanda 25 th -29 th January 2010 Fen Beed Idd Ramathani IITA-pathology,Uganda Why lab-based diagnostic tools??? Field Diagnostic tools symptoms expression-bbtd
Regional Disease Surveillance workshop for banana Rwanda 25 th -29 th January 2010 Fen Beed Idd Ramathani IITA-pathology,Uganda Why lab-based diagnostic tools??? Field Diagnostic tools symptoms expression-bbtd
Bacterial 16S rdna PCR Kit Fast (800)
 Cat. # RR182A For Research Use Bacterial 16S rdna PCR Kit Fast (800) Product Manual Table of Contents I. Description... 3 II. Components... 3 III. Materials Required but not Provided... 4 IV. Storage...
Cat. # RR182A For Research Use Bacterial 16S rdna PCR Kit Fast (800) Product Manual Table of Contents I. Description... 3 II. Components... 3 III. Materials Required but not Provided... 4 IV. Storage...
Ohio Vegetable & Small Fruit Research & Development Program
 Ohio Vegetable & Small Fruit Research & Development Program Project Title: Controlling Angular Leaf Spot on pumpkin using seed treatments and foliar applications Jim Jasinski, OSUE IPM Program Sally Miller
Ohio Vegetable & Small Fruit Research & Development Program Project Title: Controlling Angular Leaf Spot on pumpkin using seed treatments and foliar applications Jim Jasinski, OSUE IPM Program Sally Miller
Method for the Detection of Clavibacter michiganensis subsp. michiganensis on Tomato seed
 Method for the Detection of Clavibacter michiganensis subsp. michiganensis on Tomato seed Crop: Tomato (Solanum lycopersicum ex. Lycopersicon esculentum L.) Pathogen: Clavibacter michiganensis subsp. michiganensis
Method for the Detection of Clavibacter michiganensis subsp. michiganensis on Tomato seed Crop: Tomato (Solanum lycopersicum ex. Lycopersicon esculentum L.) Pathogen: Clavibacter michiganensis subsp. michiganensis
Supplemental Data Supplemental Figure 1.
 Supplemental Data Supplemental Figure 1. Silique arrangement in the wild-type, jhs, and complemented lines. Wild-type (WT) (A), the jhs1 mutant (B,C), and the jhs1 mutant complemented with JHS1 (Com) (D)
Supplemental Data Supplemental Figure 1. Silique arrangement in the wild-type, jhs, and complemented lines. Wild-type (WT) (A), the jhs1 mutant (B,C), and the jhs1 mutant complemented with JHS1 (Com) (D)
7-026: Detection of Squash Mosaic Virus, Cucumber Green Mottle Mosaic Virus and Melon Necrotic Spot Virus in cucurbits
 International Rules for Seed Testing Annexe to Chapter 7: Seed Health Testing Methods 7-026: Detection of Squash Mosaic Virus, Cucumber Green Mottle Mosaic Virus and Melon Necrotic Spot Virus in cucurbits
International Rules for Seed Testing Annexe to Chapter 7: Seed Health Testing Methods 7-026: Detection of Squash Mosaic Virus, Cucumber Green Mottle Mosaic Virus and Melon Necrotic Spot Virus in cucurbits
Method for the Detection of Clavibacter michiganensis subsp. michiganensis on Tomato seed
 Method for the Detection of Clavibacter michiganensis subsp. michiganensis on Tomato seed Crop: Tomato (Solanum lycopersicum ex. Lycopersicon esculentum L.) Pathogen: Clavibacter michiganensis subsp. michiganensis
Method for the Detection of Clavibacter michiganensis subsp. michiganensis on Tomato seed Crop: Tomato (Solanum lycopersicum ex. Lycopersicon esculentum L.) Pathogen: Clavibacter michiganensis subsp. michiganensis
CHAPTER 24. Immunology
 CHAPTER 24 Diagnostic i Microbiology and Immunology Growth-Dependent Diagnostic Methods Isolation of Pathogens from Clinical Specimens Proper sampling and culture of a suspected pathogen is the most reliable
CHAPTER 24 Diagnostic i Microbiology and Immunology Growth-Dependent Diagnostic Methods Isolation of Pathogens from Clinical Specimens Proper sampling and culture of a suspected pathogen is the most reliable
Fungal rdna (D1/D2) PCR Kit Fast
 Cat. # RR184A For Research Use Fungal rdna (D1/D2) PCR Kit Fast Product Manual Table of Contents I. Description... 3 II. Components... 3 III. Materials Required but not Provided... 4 IV. Storage... 4 V.
Cat. # RR184A For Research Use Fungal rdna (D1/D2) PCR Kit Fast Product Manual Table of Contents I. Description... 3 II. Components... 3 III. Materials Required but not Provided... 4 IV. Storage... 4 V.
REFERENCE PROTOCOL NAKTUINBOUW. Detection of Clavibacter michiganensis subsp. michiganensis in seeds of tomato
 REFERENCE PROTOCOL NAKTUINBOUW Detection of Clavibacter michiganensis subsp. michiganensis in seeds of tomato Protocol number: SPN-B004 Version: 9.1UK Date: 18 March 2010 Validation: Dilution plating and
REFERENCE PROTOCOL NAKTUINBOUW Detection of Clavibacter michiganensis subsp. michiganensis in seeds of tomato Protocol number: SPN-B004 Version: 9.1UK Date: 18 March 2010 Validation: Dilution plating and
An engineered tryptophan zipper-type peptide as a molecular recognition scaffold
 SUPPLEMENTARY MATERIAL An engineered tryptophan zipper-type peptide as a molecular recognition scaffold Zihao Cheng and Robert E. Campbell* Supplementary Methods Library construction for FRET-based screening
SUPPLEMENTARY MATERIAL An engineered tryptophan zipper-type peptide as a molecular recognition scaffold Zihao Cheng and Robert E. Campbell* Supplementary Methods Library construction for FRET-based screening
Y-chromosomal haplogroup typing Using SBE reaction
 Schematic of multiplex PCR followed by SBE reaction Multiplex PCR Exo SAP purification SBE reaction 5 A 3 ddatp ddgtp 3 T 5 A G 3 T 5 3 5 G C 5 3 3 C 5 ddttp ddctp 5 T 3 T C 3 A 5 3 A 5 5 C 3 3 G 5 3 G
Schematic of multiplex PCR followed by SBE reaction Multiplex PCR Exo SAP purification SBE reaction 5 A 3 ddatp ddgtp 3 T 5 A G 3 T 5 3 5 G C 5 3 3 C 5 ddttp ddctp 5 T 3 T C 3 A 5 3 A 5 5 C 3 3 G 5 3 G
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Dissecting binding of a β-barrel outer membrane
Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Dissecting binding of a β-barrel outer membrane
HLA-DR TYPING OF GENOMIC DNA
 HLA-DR TYPING OF GENOMIC DNA Zofia SZCZERKOWSKA, Joanna WYSOCKA Institute of Forensic Medicine, Medical University, Gdañsk, Poland ABSTRACT: Advances in molecular biology techniques allowed for introduction
HLA-DR TYPING OF GENOMIC DNA Zofia SZCZERKOWSKA, Joanna WYSOCKA Institute of Forensic Medicine, Medical University, Gdañsk, Poland ABSTRACT: Advances in molecular biology techniques allowed for introduction
Occurrence of viruses in apple and pear orchards in Latvia.
 INTERNATIONAL SCIENTIFIC CONFERENCE Sustainable Fruit Growing: From Plant to Product Occurrence of viruses in apple and pear orchards in Latvia. Neda P pola, Anna K le, Inga Moro ko Bi evska. The economically
INTERNATIONAL SCIENTIFIC CONFERENCE Sustainable Fruit Growing: From Plant to Product Occurrence of viruses in apple and pear orchards in Latvia. Neda P pola, Anna K le, Inga Moro ko Bi evska. The economically
Supporting Information. Copyright Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2006
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Supporting Information for Expanding the Genetic
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2006 Supporting Information for Expanding the Genetic
Updated August well High-multiplexing Tagmentation and Amplification Robyn Tanny August Company Kit Catalog Number.
 96-well High-multiplexing Tagmentation and Amplification Robyn Tanny August 2015 This protocol uses the following purchased reagents: Company Kit Catalog Number Illumina Nextera DNA Sample Preparation
96-well High-multiplexing Tagmentation and Amplification Robyn Tanny August 2015 This protocol uses the following purchased reagents: Company Kit Catalog Number Illumina Nextera DNA Sample Preparation
Occurrence of Foliar Fungal and Bacterial Diseases of Dry Bean in North Dakota
 2006 Plant Management Network. Accepted for publication 7 June 2006. Published. Occurrence of Foliar Fungal and Bacterial Diseases of Dry Bean in North Dakota R. Harikrishnan, L. E. del Río, R. S. Lamppa,
2006 Plant Management Network. Accepted for publication 7 June 2006. Published. Occurrence of Foliar Fungal and Bacterial Diseases of Dry Bean in North Dakota R. Harikrishnan, L. E. del Río, R. S. Lamppa,
PHENOTYPING PROTOCOLS FOR TOLERANCE TO IMPORTANT CHERRY DISEASES. Fruit Growing Institute, Plovdiv; Department of Plant Protection
 STSM Scientific Report: Subject: PHENOTYPING PROTOCOLS FOR TOLERANCE TO IMPORTANT CHERRY DISEASES STSM Reference number: COST-STSM-FA1104-20571 Period: 2014-07-01 to 2014-07-31 COST Action number: FA1104
STSM Scientific Report: Subject: PHENOTYPING PROTOCOLS FOR TOLERANCE TO IMPORTANT CHERRY DISEASES STSM Reference number: COST-STSM-FA1104-20571 Period: 2014-07-01 to 2014-07-31 COST Action number: FA1104
Supporting Information
 Supporting Information Transfection of DNA Cages into Mammalian Cells Email: a.turberfield@physics.ox.ac.uk Table of Contents Supporting Figure 1 DNA tetrahedra used in transfection experiments 2 Supporting
Supporting Information Transfection of DNA Cages into Mammalian Cells Email: a.turberfield@physics.ox.ac.uk Table of Contents Supporting Figure 1 DNA tetrahedra used in transfection experiments 2 Supporting
Sample size required:
 VERSION: 1.0 DATE: 12/2012 PATHOGEN: Curtobacterium flaccumfaciens pv. flaccumfaciens (syn: Corynebacterium flaccumfaciens pv flaccumfaciens) HOST: common bean (Phaseolus vulgaris COMMON NAME: bacterial
VERSION: 1.0 DATE: 12/2012 PATHOGEN: Curtobacterium flaccumfaciens pv. flaccumfaciens (syn: Corynebacterium flaccumfaciens pv flaccumfaciens) HOST: common bean (Phaseolus vulgaris COMMON NAME: bacterial
Supplementary Information
 Supplementary Information A general solution for opening double-stranded DNA for isothermal amplification Gangyi Chen, Juan Dong, Yi Yuan, Na Li, Xin Huang, Xin Cui* and Zhuo Tang* Supplementary Materials
Supplementary Information A general solution for opening double-stranded DNA for isothermal amplification Gangyi Chen, Juan Dong, Yi Yuan, Na Li, Xin Huang, Xin Cui* and Zhuo Tang* Supplementary Materials
Bacterial Transformation: Unlocking the Mysteries of Genetic Material
 PR009 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Bacterial Transformation: Unlocking the Mysteries of Genetic Material Teacher s Guidebook
PR009 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Bacterial Transformation: Unlocking the Mysteries of Genetic Material Teacher s Guidebook
Supplemental material
 Supplemental material Diversity of O-antigen repeat-unit structures can account for the substantial sequence variation of Wzx translocases Yaoqin Hong and Peter R. Reeves School of Molecular Bioscience,
Supplemental material Diversity of O-antigen repeat-unit structures can account for the substantial sequence variation of Wzx translocases Yaoqin Hong and Peter R. Reeves School of Molecular Bioscience,
Rhonda Simmons, Bo-Ming Wu, Ken Johnson, and Lindsey dutoit
 Evaluating Actigard for Controlling Xanthomonas Blight in Carrot Seed Crops Rhonda Simmons, Bo-Ming Wu, Ken Johnson, and Lindsey dutoit Introduction Bacterial blight caused by Xanthomonas hortorum pv.
Evaluating Actigard for Controlling Xanthomonas Blight in Carrot Seed Crops Rhonda Simmons, Bo-Ming Wu, Ken Johnson, and Lindsey dutoit Introduction Bacterial blight caused by Xanthomonas hortorum pv.
Bean common mosaic virus (BCMV) ELISA Kit
 Bean common mosaic virus (BCMV) ELISA Kit Cat.No: DEIAPV17 Lot. No. (See product label) Size 5000T Intended use The test can be used to detect BCMV in infected host plants. General Description Bean common
Bean common mosaic virus (BCMV) ELISA Kit Cat.No: DEIAPV17 Lot. No. (See product label) Size 5000T Intended use The test can be used to detect BCMV in infected host plants. General Description Bean common
Sequencing of DNA lesions facilitated by site-specific excision via base. excision repair DNA glycosylases yielding ligatable gaps
 Supporting information Sequencing of DNA lesions facilitated by site-specific excision via base excision repair DNA glycosylases yielding ligatable gaps Jan Riedl, Aaron M. Fleming, and Cynthia J. Burrows*
Supporting information Sequencing of DNA lesions facilitated by site-specific excision via base excision repair DNA glycosylases yielding ligatable gaps Jan Riedl, Aaron M. Fleming, and Cynthia J. Burrows*
Mycobacterium paratuberculosis
 BACTOTYPE PCR Amplification Kit Mycobacterium paratuberculosis Labor Diagnostik Leipzig Manual Technology The product group BACTOTYPE PCR Amplification Kit comprises optimised systems for the identification
BACTOTYPE PCR Amplification Kit Mycobacterium paratuberculosis Labor Diagnostik Leipzig Manual Technology The product group BACTOTYPE PCR Amplification Kit comprises optimised systems for the identification
English Version. Animal feeding stuffs - PCR typing of probiotic strains of Saccharomyces cerevisiae (yeast)
 TECHNICAL SPECIFICATION SPÉCIFICATION TECHNIQUE TECHNISCHE SPEZIFIKATION CEN/TS 15790 December 2008 ICS 07.100.30; 65.120 English Version Animal feeding stuffs - PCR typing of probiotic strains of Saccharomyces
TECHNICAL SPECIFICATION SPÉCIFICATION TECHNIQUE TECHNISCHE SPEZIFIKATION CEN/TS 15790 December 2008 ICS 07.100.30; 65.120 English Version Animal feeding stuffs - PCR typing of probiotic strains of Saccharomyces
Taxonomy. Classification of microorganisms 3/12/2017. Is the study of classification. Chapter 10 BIO 220
 Taxonomy Is the study of classification Organisms are classified based on relatedness to each other Chapter 10 BIO 220 Fig. 10.1 1 Species Binomial nomenclature for species identification A eukaryotic
Taxonomy Is the study of classification Organisms are classified based on relatedness to each other Chapter 10 BIO 220 Fig. 10.1 1 Species Binomial nomenclature for species identification A eukaryotic
Application of Molecular Biology tools for cloning of a foreign gene
 IFM/Kemi Linköpings Universitet September 2013/LGM Labmanual Project course Application of Molecular Biology tools for cloning of a foreign gene Table of contents Introduction... 3 Amplification of a gene
IFM/Kemi Linköpings Universitet September 2013/LGM Labmanual Project course Application of Molecular Biology tools for cloning of a foreign gene Table of contents Introduction... 3 Amplification of a gene
DETERMINATION OF THE Rh FACTOR BY PCR
 DETERMINATION OF THE Rh FACTOR BY PCR Ref.: PCR2 1. EXPERIMENT OBJECTIVE The aim of this experiment is to introduce students to the principles and practice of the Polymerase Chain Reaction (PCR) by studying
DETERMINATION OF THE Rh FACTOR BY PCR Ref.: PCR2 1. EXPERIMENT OBJECTIVE The aim of this experiment is to introduce students to the principles and practice of the Polymerase Chain Reaction (PCR) by studying
pgm-t Cloning Kit For direct cloning of PCR products generated by Taq DNA polymerases For research use only Cat. # : GVT202 Size : 20 Reactions
 pgm-t Cloning Kit For direct cloning of PCR products generated by Taq DNA polymerases Cat. # : GVT202 Size : 20 Reactions Store at -20 For research use only 1 pgm-t Cloning Kit Cat. No.: GVT202 Kit Contents
pgm-t Cloning Kit For direct cloning of PCR products generated by Taq DNA polymerases Cat. # : GVT202 Size : 20 Reactions Store at -20 For research use only 1 pgm-t Cloning Kit Cat. No.: GVT202 Kit Contents
Protocol for mycoplasma detection in JCRB Cell Bank
 Protocol for mycoplasma detection in JCRB Cell Bank Translation and modification by Hideyuki Tanabe 1. Vero-Hoechst method: using indicator cells detected by staining with fluorescent dye (Indirect method:
Protocol for mycoplasma detection in JCRB Cell Bank Translation and modification by Hideyuki Tanabe 1. Vero-Hoechst method: using indicator cells detected by staining with fluorescent dye (Indirect method:
This Product Description is only valid for Lot No. 36V.
 HLA Wipe Test Negative Control Product Insert Page 1 of 8 Olerup SSP HLA Wipe Test Negative Control Product number: 102.101-01 including Taq polymerase Product number: 102.101-01u without Taq polymerase
HLA Wipe Test Negative Control Product Insert Page 1 of 8 Olerup SSP HLA Wipe Test Negative Control Product number: 102.101-01 including Taq polymerase Product number: 102.101-01u without Taq polymerase
500U. Unit Definition. Storage Buffer. 10X PCR Buffer with Mg 2+
 Hy-Taq 500U + dntps #EZ1012 500U Concentration: 5U/μl Contents: Hy-Taq DNA Polymerase 100μl 10xPCR Buffer(Mg 2+ Plus) 1.25ml dntps(2.5mm each) 1ml 6xLoading Buffer 1ml Store at -20 C For research only
Hy-Taq 500U + dntps #EZ1012 500U Concentration: 5U/μl Contents: Hy-Taq DNA Polymerase 100μl 10xPCR Buffer(Mg 2+ Plus) 1.25ml dntps(2.5mm each) 1ml 6xLoading Buffer 1ml Store at -20 C For research only
Mastermix 16S Complete, DNA-free
 Mastermix 16S Complete, DNA-free For the PCR detection and identification of bacteria using universal 16S rdna primers For research use only Cat. No. S-020-0100 Cat. No. S-020-0250 Cat. No. S-020-1000
Mastermix 16S Complete, DNA-free For the PCR detection and identification of bacteria using universal 16S rdna primers For research use only Cat. No. S-020-0100 Cat. No. S-020-0250 Cat. No. S-020-1000
Vibrio splendidus et V. aestuarianus detection by Real Time Polymerase Chain Reaction
 European Union Reference Laboratory for Molluscs Diseases Vibrio splendidus et V. aestuarianus detection by Real Time Polymerase Chain Reaction CONTENTS 1. SCOPE... 2 2. REFERENCES... 2 3. EQUIPMENT AND
European Union Reference Laboratory for Molluscs Diseases Vibrio splendidus et V. aestuarianus detection by Real Time Polymerase Chain Reaction CONTENTS 1. SCOPE... 2 2. REFERENCES... 2 3. EQUIPMENT AND
pgm-t Cloning Kit Cat. # : GVT202 Size : 20 Reactions Store at -20
 pgm-t Cloning Kit Cat. # : GVT202 Size : 20 Reactions Store at -20 1 Kit Contents Contents pgm-t Cloning Kit pgm-t Vector (50 ng/μl) 20 μl T4 DNA Ligase (3 U/μl) 20 μl 10X T4 DNA Ligation Buffer 30 μl
pgm-t Cloning Kit Cat. # : GVT202 Size : 20 Reactions Store at -20 1 Kit Contents Contents pgm-t Cloning Kit pgm-t Vector (50 ng/μl) 20 μl T4 DNA Ligase (3 U/μl) 20 μl 10X T4 DNA Ligation Buffer 30 μl
Quantitative reverse-transcription PCR. Transcript levels of flgs, flgr, flia and flha were
 1 Supplemental methods 2 3 4 5 6 7 8 9 1 11 12 13 14 15 16 17 18 19 21 22 23 Quantitative reverse-transcription PCR. Transcript levels of flgs, flgr, flia and flha were monitored by quantitative reverse-transcription
1 Supplemental methods 2 3 4 5 6 7 8 9 1 11 12 13 14 15 16 17 18 19 21 22 23 Quantitative reverse-transcription PCR. Transcript levels of flgs, flgr, flia and flha were monitored by quantitative reverse-transcription
Multiplex PCR Assay Kit Ver.2
 Cat. # RR062A For Research Use Multiplex PCR Assay Kit Ver.2 Product Manual Table of Contents I. Description... 3 II. Components... 3 III. Storage... 3 IV. Precautions... 3 V. Designing Primers for Multiplex
Cat. # RR062A For Research Use Multiplex PCR Assay Kit Ver.2 Product Manual Table of Contents I. Description... 3 II. Components... 3 III. Storage... 3 IV. Precautions... 3 V. Designing Primers for Multiplex
DNA Extraction and Real-Time PCR Detection Kit for Mycoplasma gallisepticum live vaccine strain ts-11.
 For in vitro Veterinary Diagnostics only. DNA Extraction and Real-Time PCR Detection Kit for Mycoplasma gallisepticum live vaccine strain ts-11 www.kylt.eu DIRECTION FOR USE Art. No. 31410 / 31411 Kylt
For in vitro Veterinary Diagnostics only. DNA Extraction and Real-Time PCR Detection Kit for Mycoplasma gallisepticum live vaccine strain ts-11 www.kylt.eu DIRECTION FOR USE Art. No. 31410 / 31411 Kylt
Figure S1. Characterization of the irx9l-1 mutant. (A) Diagram of the Arabidopsis IRX9L gene drawn based on information from TAIR (the Arabidopsis
 1 2 3 4 5 6 7 8 9 10 11 12 Figure S1. Characterization of the irx9l-1 mutant. (A) Diagram of the Arabidopsis IRX9L gene drawn based on information from TAIR (the Arabidopsis Information Research). Exons
1 2 3 4 5 6 7 8 9 10 11 12 Figure S1. Characterization of the irx9l-1 mutant. (A) Diagram of the Arabidopsis IRX9L gene drawn based on information from TAIR (the Arabidopsis Information Research). Exons
TECHNICAL SHEET No. 23. Virus Detection: Potato virus Y (PVY) and PVY N
 TECHNICAL SHEET No. 23 Virus Detection: Potato virus Y (PVY) and PVY N Method: RT-PCR General Virus detected: PVY from potato tubers and leaf. General method is reverse transcription PCR (RT-PCR). Developed
TECHNICAL SHEET No. 23 Virus Detection: Potato virus Y (PVY) and PVY N Method: RT-PCR General Virus detected: PVY from potato tubers and leaf. General method is reverse transcription PCR (RT-PCR). Developed
Cat. # MK600. For Research Use. ApopLadder Ex. Product Manual. v201608da
 Cat. # MK600 For Research Use ApopLadder Ex Product Manual Table of Contents I. Description... 3 II. Principle... 3 III. Features... 3 IV. Components... 3 V. Storage... 3 VI. Materials Required but not
Cat. # MK600 For Research Use ApopLadder Ex Product Manual Table of Contents I. Description... 3 II. Principle... 3 III. Features... 3 IV. Components... 3 V. Storage... 3 VI. Materials Required but not
ACP601: Plant Mycology and Bacteriology Plant Bacteriology
 ACP601: Plant Mycology and Bacteriology Plant Bacteriology James W. Muthomi Department of Plant Science and Crop Protection University of Nairobi Bacterial Diseases of Plant in East Africa Bacterial blight
ACP601: Plant Mycology and Bacteriology Plant Bacteriology James W. Muthomi Department of Plant Science and Crop Protection University of Nairobi Bacterial Diseases of Plant in East Africa Bacterial blight
Arabidopsis actin depolymerizing factor AtADF4 mediates defense signal transduction triggered by the Pseudomonas syringae effector AvrPphB
 Arabidopsis actin depolymerizing factor mediates defense signal transduction triggered by the Pseudomonas syringae effector AvrPphB Files in this Data Supplement: Supplemental Table S1 Supplemental Table
Arabidopsis actin depolymerizing factor mediates defense signal transduction triggered by the Pseudomonas syringae effector AvrPphB Files in this Data Supplement: Supplemental Table S1 Supplemental Table
Supplemental Data. mir156-regulated SPL Transcription. Factors Define an Endogenous Flowering. Pathway in Arabidopsis thaliana
 Cell, Volume 138 Supplemental Data mir156-regulated SPL Transcription Factors Define an Endogenous Flowering Pathway in Arabidopsis thaliana Jia-Wei Wang, Benjamin Czech, and Detlef Weigel Table S1. Interaction
Cell, Volume 138 Supplemental Data mir156-regulated SPL Transcription Factors Define an Endogenous Flowering Pathway in Arabidopsis thaliana Jia-Wei Wang, Benjamin Czech, and Detlef Weigel Table S1. Interaction
APPLICATION OF MOLECULAR TECHNICS FOR DIAGNOSIS OF VIRAL INFECTIONS
 APPLICATION OF MOLECULAR TECHNICS FOR DIAGNOSIS OF VIRAL INFECTIONS Hossein Keyvani Basic Diagnostic Methods in Virology Immunology and serology techniques (Antigen-Antibody Reactions) 1 ELISA ( Enzyme
APPLICATION OF MOLECULAR TECHNICS FOR DIAGNOSIS OF VIRAL INFECTIONS Hossein Keyvani Basic Diagnostic Methods in Virology Immunology and serology techniques (Antigen-Antibody Reactions) 1 ELISA ( Enzyme
Practical 4: PCR in Molecular Diagnosis
 PRINCIPLES What is PCR Practical 4: PCR in Molecular Diagnosis Instructors: Dr. Valerie C.L. Lin and Dr. Sze Chun Chau PCR stands for polymerase chain reaction. The PCR method was devised and named by
PRINCIPLES What is PCR Practical 4: PCR in Molecular Diagnosis Instructors: Dr. Valerie C.L. Lin and Dr. Sze Chun Chau PCR stands for polymerase chain reaction. The PCR method was devised and named by
Positive control kit for enzymatic mismatch cleavage and agarose gel visualization (version 2.4)
 28 January, 2010 Positive control kit for enzymatic mismatch cleavage and agarose gel visualization (version 2.4) Plant Breeding Unit Brad Till and Owen Huynh January, 2010 Kit Contents: Genomic DNA from
28 January, 2010 Positive control kit for enzymatic mismatch cleavage and agarose gel visualization (version 2.4) Plant Breeding Unit Brad Till and Owen Huynh January, 2010 Kit Contents: Genomic DNA from
Cat. # R100A. For Research Use. EpiScope MSP Kit. Product Manual. v1103da
 Cat. # R100A For Research Use EpiScope MSP Kit Product Manual Table of Contents I. Description... 3 II. Principle of MSP... 3 III. Kit Components... 4 IV. Materials Required but not Provided... 4 V. Storage...
Cat. # R100A For Research Use EpiScope MSP Kit Product Manual Table of Contents I. Description... 3 II. Principle of MSP... 3 III. Kit Components... 4 IV. Materials Required but not Provided... 4 V. Storage...
Taq + dntps #EZ U
 #EZ1012 500U Taq + dntps Concentration: 5U/μl Contents: Hy-Taq DNA polymerase 100μl 10xPCR Buffer (Mg2+ Plus) 1.25ml dntps(2.5mm each) 1ml 6xLoading Buffer 1ml Store at -20 C Description Hy-Taq 500U +
#EZ1012 500U Taq + dntps Concentration: 5U/μl Contents: Hy-Taq DNA polymerase 100μl 10xPCR Buffer (Mg2+ Plus) 1.25ml dntps(2.5mm each) 1ml 6xLoading Buffer 1ml Store at -20 C Description Hy-Taq 500U +
Cat. # R100A. For Research Use. EpiScope MSP Kit. Product Manual. v201712da_2
 Cat. # R100A For Research Use EpiScope MSP Kit Product Manual Table of Contents I. Description... 3 II. MSP Principle... 3 III. Components... 4 IV. Storage... 4 V. Materials Required but not Provided...
Cat. # R100A For Research Use EpiScope MSP Kit Product Manual Table of Contents I. Description... 3 II. MSP Principle... 3 III. Components... 4 IV. Storage... 4 V. Materials Required but not Provided...
I. Things to keep in mind before you start this protocol
 Inverse PCR and Sequencing Protocol on 5 Fly Preps For recovery of sequences flanking piggybac elements This protocol is an adaptation of "Inverse PCR and Cycle Sequencing Protocols" by E. Jay Rehm Berkeley
Inverse PCR and Sequencing Protocol on 5 Fly Preps For recovery of sequences flanking piggybac elements This protocol is an adaptation of "Inverse PCR and Cycle Sequencing Protocols" by E. Jay Rehm Berkeley
PROCEDURES USED BY THE GERMPLASM HEALTH LABORATORY (GHL) OF CIAT
 1 PROCEDURES USED BY THE GERMPLASM HEALTH LABORATORY (GHL) OF CIAT INTRODUCTION The main responsibility of the Germplasm Health Laboratory (GHL) is to test the health status of germplasm (beans, tropical
1 PROCEDURES USED BY THE GERMPLASM HEALTH LABORATORY (GHL) OF CIAT INTRODUCTION The main responsibility of the Germplasm Health Laboratory (GHL) is to test the health status of germplasm (beans, tropical
FMF NIRCA PROTOCOL STEP 1.
 FMF NIRCA PROTOCOL STEP 1. After you have isolated patient s DNA and DNA from a healthy donor (wild type), you perform a nested PCR. The primers used to amplify exon 2 and exon 10 of the mefv gene are
FMF NIRCA PROTOCOL STEP 1. After you have isolated patient s DNA and DNA from a healthy donor (wild type), you perform a nested PCR. The primers used to amplify exon 2 and exon 10 of the mefv gene are
Converting rabbit hybridoma into recombinant antibodies with effective transient production in an optimized human expression system
 Converting rabbit hybridoma into recombinant antibodies with effective transient production in an optimized human expression system Dr. Tim Welsink Molecular Biology Transient Gene Expression OUTLINE Short
Converting rabbit hybridoma into recombinant antibodies with effective transient production in an optimized human expression system Dr. Tim Welsink Molecular Biology Transient Gene Expression OUTLINE Short
Bacterial blight in field pea
 Bacterial blight in field pea Efficient phenotyping method developed in controlled environment for pre-breeding and breeding Dr Pragya Kant- Horsham, Vic Bacterial blight epidemic in Rupanyup, Vic, 2012
Bacterial blight in field pea Efficient phenotyping method developed in controlled environment for pre-breeding and breeding Dr Pragya Kant- Horsham, Vic Bacterial blight epidemic in Rupanyup, Vic, 2012
7-022: Agar method for the detection of Microdochium nivale and Microdochium majus on Triticum spp.
 International Rules for Seed Testing Annexe to Chapter 7: Seed Health Testing Methods 7-022: Agar method for the detection of Microdochium nivale and Microdochium majus on Triticum spp. Published by the
International Rules for Seed Testing Annexe to Chapter 7: Seed Health Testing Methods 7-022: Agar method for the detection of Microdochium nivale and Microdochium majus on Triticum spp. Published by the
HiPer RT-PCR Teaching Kit
 HiPer RT-PCR Teaching Kit Product Code: HTBM024 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 4 hours Agarose Gel Electrophoresis: 45 minutes Storage Instructions: The
HiPer RT-PCR Teaching Kit Product Code: HTBM024 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 4 hours Agarose Gel Electrophoresis: 45 minutes Storage Instructions: The
Lecture 10, 20/2/2002: The process of solution development - The CODEHOP strategy for automatic design of consensus-degenerate primers for PCR
 Lecture 10, 20/2/2002: The process of solution development - The CODEHOP strategy for automatic design of consensus-degenerate primers for PCR 1 The problem We wish to clone a yet unknown gene from a known
Lecture 10, 20/2/2002: The process of solution development - The CODEHOP strategy for automatic design of consensus-degenerate primers for PCR 1 The problem We wish to clone a yet unknown gene from a known
Pasteurella multocida
 BACTOTYPE PCR Amplification Kit Pasteurella multocida Labor Diagnostik Leipzig Manual Technology The product group BACTOTYPE PCR Amplification Kit comprises optimised systems for the identification of
BACTOTYPE PCR Amplification Kit Pasteurella multocida Labor Diagnostik Leipzig Manual Technology The product group BACTOTYPE PCR Amplification Kit comprises optimised systems for the identification of
HE Swift Cloning Kit
 HE Swift Cloning Kit For high-efficient cloning of PCR products either blunt or sticky-end Kit Contents Contents VTT-BB05 phe Vector (35 ng/µl) 20 µl T4 DNA Ligase (3 U/µl) 20 µl 2 Reaction Buffer 100
HE Swift Cloning Kit For high-efficient cloning of PCR products either blunt or sticky-end Kit Contents Contents VTT-BB05 phe Vector (35 ng/µl) 20 µl T4 DNA Ligase (3 U/µl) 20 µl 2 Reaction Buffer 100
Introduction. Kit components. 50 Preps GF-PC-050. Product Catalog No. 200 Preps GF-PC Preps GF-PC Preps SAMPLE
 Introduction The GF-1 PCR Clean Up Kit is a system designed for rapid clean up of DNA bands ranging from 100bp to 20kb. The GF-1 PCR Clean Up Kit contains special buffers to provide the correct salt concentration
Introduction The GF-1 PCR Clean Up Kit is a system designed for rapid clean up of DNA bands ranging from 100bp to 20kb. The GF-1 PCR Clean Up Kit contains special buffers to provide the correct salt concentration
HiPer Random Amplification of Polymorphic DNA (RAPD) Teaching Kit
 HiPer Random Amplification of Polymorphic DNA (RAPD) Teaching Kit Product Code: HTBM031 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 3.5 hours Agarose Gel Electrophoresis:
HiPer Random Amplification of Polymorphic DNA (RAPD) Teaching Kit Product Code: HTBM031 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 3.5 hours Agarose Gel Electrophoresis:
SpeedSTAR HS DNA Polymerase
 Cat. # RR070A For Research Use SpeedSTAR HS DNA Polymerase Product Manual Table of Contents I. Description... 3 II. Components... 3 III. Storage... 3 IV. Supplied buffers... 3 V. General reaction mixture...
Cat. # RR070A For Research Use SpeedSTAR HS DNA Polymerase Product Manual Table of Contents I. Description... 3 II. Components... 3 III. Storage... 3 IV. Supplied buffers... 3 V. General reaction mixture...
Legends for supplementary figures 1-3
 High throughput resistance profiling of Plasmodium falciparum infections based on custom dual indexing and Illumina next generation sequencing-technology Sidsel Nag 1,2 *, Marlene D. Dalgaard 3, Poul-Erik
High throughput resistance profiling of Plasmodium falciparum infections based on custom dual indexing and Illumina next generation sequencing-technology Sidsel Nag 1,2 *, Marlene D. Dalgaard 3, Poul-Erik
PCR analysis was performed to show the presence and the integrity of the var1csa and var-
 Supplementary information: Methods: Table S1: Primer Name Nucleotide sequence (5-3 ) DBL3-F tcc ccg cgg agt gaa aca tca tgt gac tg DBL3-R gac tag ttt ctt tca ata aat cac tcg c DBL5-F cgc cct agg tgc ttc
Supplementary information: Methods: Table S1: Primer Name Nucleotide sequence (5-3 ) DBL3-F tcc ccg cgg agt gaa aca tca tgt gac tg DBL3-R gac tag ttt ctt tca ata aat cac tcg c DBL5-F cgc cct agg tgc ttc
Distributed at The Empire State Fruit & Vegetable Expo; January 26,
 RESEARCH YIELDS GREATER UNDERSTANDING OF BACTERIAL DISEASES OF ONION IN NEW YORK Steven V. Beer a, Jo Ann E. Asselin b*, Jean M. Bonasera c*, and Ali M. Zaid d* Professor a, Post-doctoral Associate b,
RESEARCH YIELDS GREATER UNDERSTANDING OF BACTERIAL DISEASES OF ONION IN NEW YORK Steven V. Beer a, Jo Ann E. Asselin b*, Jean M. Bonasera c*, and Ali M. Zaid d* Professor a, Post-doctoral Associate b,
WORKING GROUP ON BIOCHEMICAL AND MOLECULAR TECHNIQUES AND DNA-PROFILING IN PARTICULAR. Eighth Session Tsukuba, Japan, September 3 to 5, 2003
 E BMT/8/25 ORIGINAL: English DATE: August 18, 2003 INTERNATIONAL UNION FOR THE PROTECTION OF NEW VARIETIES OF PLANTS GENEVA WORKING GROUP ON BIOCHEMICAL AND MOLECULAR TECHNIQUES AND DNAPROFILING IN PARTICULAR
E BMT/8/25 ORIGINAL: English DATE: August 18, 2003 INTERNATIONAL UNION FOR THE PROTECTION OF NEW VARIETIES OF PLANTS GENEVA WORKING GROUP ON BIOCHEMICAL AND MOLECULAR TECHNIQUES AND DNAPROFILING IN PARTICULAR
3. How can you test a food to find out if it contains materials derived from a GMO?
 GMO Investigator LAB Name Introduction With the world population exploding and farmable land disappearing, agricultural specialists are concerned about the world's ability to produce enough food to feed
GMO Investigator LAB Name Introduction With the world population exploding and farmable land disappearing, agricultural specialists are concerned about the world's ability to produce enough food to feed
Assessment of the sanitary status of pome fruit crops in Kosovo, with particular emphasis to the bacterial disease the Fire Blight
 ORIGINAL SCIENTIFIC PAPER Assessment of the sanitary status of pome fruit crops in Kosovo, with particular emphasis to the bacterial disease the Fire Blight Naim KRASNIQI 1, Arben MUSLIU 1, Kujtim LEPAJA
ORIGINAL SCIENTIFIC PAPER Assessment of the sanitary status of pome fruit crops in Kosovo, with particular emphasis to the bacterial disease the Fire Blight Naim KRASNIQI 1, Arben MUSLIU 1, Kujtim LEPAJA
High Pure Technology and Silica Adsorption High Pure PCR Product Purification Kit
 for purification of DNA from PCR reactions Cat. No. 1 73 668 (50 purifications) Cat. No. 1 73 676 (50 purifications) Principle In the presence of chaotropic salt, product DNA binds selectively to glass
for purification of DNA from PCR reactions Cat. No. 1 73 668 (50 purifications) Cat. No. 1 73 676 (50 purifications) Principle In the presence of chaotropic salt, product DNA binds selectively to glass
PlantDirect TM Multiplex PCR System
 PlantDirect TM Multiplex PCR System Technical Manual No. 0178 Version 10112010 I Description.. 1 II Applications 2 III Key Features.. 3 IV Shipping and Storage. 3 V Simplified Procedures. 3 VI Detailed
PlantDirect TM Multiplex PCR System Technical Manual No. 0178 Version 10112010 I Description.. 1 II Applications 2 III Key Features.. 3 IV Shipping and Storage. 3 V Simplified Procedures. 3 VI Detailed
Supporting Information
 Supporting Information Table S1. Oligonucleotide sequences used in this work Oligo DNA A B C D CpG-A CpG-B CpG-C CpG-D Sequence 5 ACA TTC CTA AGT CTG AAA CAT TAC AGC TTG CTA CAC GAG AAG AGC CGC CAT AGT
Supporting Information Table S1. Oligonucleotide sequences used in this work Oligo DNA A B C D CpG-A CpG-B CpG-C CpG-D Sequence 5 ACA TTC CTA AGT CTG AAA CAT TAC AGC TTG CTA CAC GAG AAG AGC CGC CAT AGT
Solid-phase PCR in a picowell array for immobilizing and arraying 100,000 PCR products to a microscope slide
 Solid-phase PCR in a picowell array for immobilizing and arraying 100,000 PCR products to a microscope slide Jochen Hoffmann a,b, Martin Trotter a, Felix von Stetten a,b, Roland Zengerle, a,b,c Günter
Solid-phase PCR in a picowell array for immobilizing and arraying 100,000 PCR products to a microscope slide Jochen Hoffmann a,b, Martin Trotter a, Felix von Stetten a,b, Roland Zengerle, a,b,c Günter
Samantha Thomas US-ISHI Chair Global Stewardship and Industry Affairs Lead, Monsanto
 Samantha Thomas US-ISHI Chair Global Stewardship and Industry Affairs Lead, Monsanto ISF Viewpoint on Indirect Seed Health Tests http://www.worldseed.org/cms/medias/file/ PositionPapers/OnSpecificTechnicalSubjects/I
Samantha Thomas US-ISHI Chair Global Stewardship and Industry Affairs Lead, Monsanto ISF Viewpoint on Indirect Seed Health Tests http://www.worldseed.org/cms/medias/file/ PositionPapers/OnSpecificTechnicalSubjects/I
Comparison of extraction procedures and determination of the detection threshold for Clavibacter michiganensis ssp. michiganensis in tomato seeds
 Plant Pathology (2005) 54, 643 649 Doi: 10.1111/j.1365-3059.2005.01230.x Blackwell Publishing, Ltd. Comparison of extraction procedures and determination of the detection threshold for Clavibacter michiganensis
Plant Pathology (2005) 54, 643 649 Doi: 10.1111/j.1365-3059.2005.01230.x Blackwell Publishing, Ltd. Comparison of extraction procedures and determination of the detection threshold for Clavibacter michiganensis
SuperTCRExpress TM Human TCR Vβ Repertoire CDR3 Diversity Determination (Spectratyping) and Quantitative Analysis Kit
 SuperTCRExpress TM Human TCR Vβ Repertoire CDR3 Diversity Determination (Spectratyping) and Quantitative Analysis Kit Cat. No. H0521 Size: 2 sets (22 Vβ families/each, with enzymes) H0522 Size: 4 sets
SuperTCRExpress TM Human TCR Vβ Repertoire CDR3 Diversity Determination (Spectratyping) and Quantitative Analysis Kit Cat. No. H0521 Size: 2 sets (22 Vβ families/each, with enzymes) H0522 Size: 4 sets
SOP for Quantitation of the Hepatitis C Virus (HCV) Genome by RT-PCR
 Virus Bank SOP-HCV-001 1. Scope SOP for Quantitation of the Hepatitis C Virus (HCV) Genome by RT-PCR 1.1 This procedure describes a method for quantitation of the HCV genome in HCV-infected cells by RT-PCR.
Virus Bank SOP-HCV-001 1. Scope SOP for Quantitation of the Hepatitis C Virus (HCV) Genome by RT-PCR 1.1 This procedure describes a method for quantitation of the HCV genome in HCV-infected cells by RT-PCR.
QualiPlate kit for Detection of Soybean Rust. Murali Bandla EnviroLogix Inc
 QualiPlate kit for Detection of Soybean Rust Murali Bandla EnviroLogix Inc Putting Science to the Test EnviroLogix Inc. WHAT DO WE DO? We develop, manufacture and market immunoassay test kits for the detection
QualiPlate kit for Detection of Soybean Rust Murali Bandla EnviroLogix Inc Putting Science to the Test EnviroLogix Inc. WHAT DO WE DO? We develop, manufacture and market immunoassay test kits for the detection
Amplification Products for PCR and RT-PCR
 Selection guide Polymerase Hot start Comment UptiTherm DNA pol. no Most economic. Lower error rate than Taq polymerase Available in several formats, master mix including or not dntp, Mg 2+..., in gel format
Selection guide Polymerase Hot start Comment UptiTherm DNA pol. no Most economic. Lower error rate than Taq polymerase Available in several formats, master mix including or not dntp, Mg 2+..., in gel format
