Proquina A Bayer Company
|
|
|
- Linda Garrison
- 6 years ago
- Views:
Transcription
1 Proquina A Bayer Company Producing Active Pharmaceutical Ingredients and Advanced Intermediates
2 Proquina ensuring high quality in line with Bayer standards Reliable what we do, we do right As a subsidiary of Bayer AG, Proquina adheres to strict standards for quality, environmental protection, safety, and production. We ensure the reliable high quality of our products and processes by means of GMP rules and ISO 9001/14001-based processes and regular inspections. Experience our employees know-how Approximately 400 highly-qualified employees work at the production plant in Orizaba. This is where we combine experience with extensive skills. Training programs in GMP, safety, and environmental protection ensure compliance with the latest quality and HSE standards. Quality Know-how Service Versatile tailored to suit your requirements Proquina specializes in the production of active pharmaceutical ingredients and intermediates, particularly for the field of steroid hormones. We find custom-tailored solutions for your products. Proquina company profile The Proquina production plant is located in Orizaba, Mexico, about 280 kilometers (174 miles) east of Mexico City, at 1,200 meters (3,937 feet) above sea level. Proquina is a subsidiary of Bayer. It began operations in 1963 as part of the Germanybased company Schering at its 15-hectare (37-acre) site, producing steroid intermediates and active pharmaceutical ingredients based on the root of the barbasco plant, which grew abundantly in the region. The company supplies products to Bayer s internal and external customers. In collaboration with the Pharma Chemicals business area in Berlin, Germany, we serve external customers in South and Central America, North America, Europe, and other regions. Nearly 400 employees work for Proquina. 3
3 Production and services offering active pharmaceutical ingredients and intermediates as well as custom-tailored services Proquina is acknowledged on the global markets as a reliable company and partner producing to the highest quality standards. We strictly adhere to both national norms and best international standards. Our product portfolio includes a wide variety of steroid hormones and intermediates. The extensive experience of our staff allows us to offer process development services, new molecule production, and tailored product manufacture including the development of analytical methods, compilation of regulatory documentation, isolation and characterization of impurities, and stability testing. Our company is part of Bayer where we play a key role as a low-cost supplier of high-quality active pharmaceutical ingredients and intermediates. We are also considered a reliable supply partner for external clients on the international market for many decades. Close collaboration with the Pharma Chemicals business area of Bayer in commercial activities, personalized customer service, and technical support from experts in the field have allowed us to extend our global presence successfully. Partnering with Proquina, customers benefit from our integrated quality and HSE (Health, Safety, Environment) systems as well as from our ISO 9001 and certifications. These systems ensure production processes with consistently high product quality backed by the required documentation and supply reliability. We are proud to have a company culture oriented toward operational excellence, which includes process optimization through continuous improvement. This allows us to respond quickly to the current and future needs of our customers by providing the necessary resources and capabilities without delay. In addition to standard technical equipment, we also use systems and facilities that have been specifically developed for the field of steroid chemistry from small-scale development in the laboratory to pilot- and industrial-scale production. We have successfully passed a large number of inspections by the FDA as well as other national and international authorities clear recognition of our commitment and a success guarantee for our customers. certified tailor-made strategic Product solutions tailor-made development and production We are the right choice when it comes to the professional production of active pharmaceutical ingredients and their intermediates. Our specialty is the development and production of steroid hormones. We offer the option of customized production of active pharmaceutical ingredients according to the specific needs of our customers. After carrying out feasibility studies and adapting our facilities to the specific requirements of each product, we ensure the supply of necessary raw materials in order to guarantee consistent quality. Our steroid manufacturing processes are backed by modern analytic control and meticulous documentation. At Proquina, we are in constant communication with our customers to efficiently coordinate all production-related activities. We offer integrated technical, chemical, and strategic product solutions, all from a single source. Inspections and audits > FDA (Food and Drug Administration, USA), EMA (European Medical Agency) > TGA (Therapeutic Goods Administration, Australia) > PMDA (Pharmaceuticals and Medical Devices Agency, Japan) > COFEPRIS (Federal Commission for the Protection against Sanitary Risks, Mexico) > ISO 9001 & ISO audits > Frequent customer audits 4 5
4 Our portfolio Our product portfolio includes a wide variety of active pharmaceutical ingredients and intermediates, specifically for the field of steroid hormones. Proquina is acknowledged on the global markets as a reliable company and partner producing to the highest quality standards. We strictly adhere to both national norms and international standards. Our services CHEMICAL and TECHNICAL REGULATORY AFFAIRS FACILITIES AND EQUIPMENT CAS NO. PRODUCT REGULATORY DOCUMENTS ANDROGENS Testosterone USDMF, AUSTRALIAN DMF, NEW ZEALAND DMF, CANADIAN DMF & CEP Testosterone Enanthate USDMF & CEP Testosterone Undecanoate USDMF & ASMF Prasterone USDMF, CANADIAN DMF, ASMF Prasterone Enanthate Technical PACKAGE ESTROGENS alpha Estradiol USDMF Estradiol Benzoate Technical PACKAGE Estradiol Enanthate Technical PACKAGE Estriol CEP PROGESTOGENS Dihydroxyprogesterone Acetophenide Technical PACKAGE (Algestone Acetophenide) Progesterone USDMF & CANADIAN DMF, cep Danazol USDMF INTERMEDIATES Alpha Methyl Acetoxy Pregnenolone (MAP) Technical PACKAGE AD 17-Carboxylic acid Technical PACKAGE Boldenone Technical PACKAGE Ethisterone Technical PACKAGE Mestanolone Technical PACKAGE We also offer the customized production of active pharmaceutical ingredients and intermediates, beyond the products listed, to meet the specific needs of our customers. DEVELOPMENT Chemical development of manufacturing processes and syntheses Optimization of existing manufacturing processes and syntheses Scale-up work Micronization Analytical development and validation Regulatory support LABORATORY SUPPORT Synthesis and development of reference standards and working standards Synthesis, isolation, and characterization of impurities Studies of stability Photo stability Stress test We offer the technical support necessary to meet the requirements of authorities and customers. This includes the preparation, maintenance, and updating of all necessary regulatory documents for: Product registrations and management in conjunction with the relevant authorities (DMFs) Technical packages Certificates of suitability to the monographs of the European Pharmacopoeia (CEPs) MAIN REACTIONS Epoxidation Halogenation Hydrogenation Hydride-based reduction Organometallic reductions Esterification Ethinylization Oxidation Production facilities 2 plants for intermediates 3 plants for APIs 1 kilo lab 1 plant for solvent recovery Process development lab Equipment available Reactors (250 to 7,500 liters) Centrifuges Vacuum dryers Nutsche filter/dryers Homogenizers Micronizers Distillation columns Our staff Some 400 highly qualified employees work at our production plant in Orizaba, Mexico. A comprehensive training system ensures broad knowledge and required skills at all times. We guarantee a high level of experience in our production and support processes. Our customers benefit from Proquina s extensive experience in the field of steroids, our competitive production costs and our reliable product quality. 6 7
5 For more information, please contact our customer service department: Customer service Productos Químicos Naturales S.A. de C.V. (Proquina) Domicilio Conocido Ojo de Agua Ixtaczoquitlán, Veracruz México Phone: Fax: Pharma Chemicals Bayer AG Müllerstraße 178 D Berlin Germany Phone: or Legal disclaimer: This information and our technical advice whether verbal, in writing or by way of trials are given in good faith but without warranty. This also applies where proprietary rights of third parties are involved. Our advice does not release you from your obligation to verify current information in particular, information relating to our safety data or our technical information sheets and to test our products as to their suitability for the intended processes and uses. The application, use, and processing of our products and the products manufactured by you on the basis of our technical advice are beyond our control and, therefore, entirely your own responsibility. Our products are sold in accordance with the current version of our General Conditions of Sale and Delivery. January 2017 Bayer AG Pharma Chemicals Berlin, Germany
Proquina A Bayer Company
 Proquina A Bayer Company Producing Active Pharmaceutical Ingredients and Advanced Intermediates Quality Know-how Service Proquina ensuring high quality in line with Bayer standards Reliable what we do,
Proquina A Bayer Company Producing Active Pharmaceutical Ingredients and Advanced Intermediates Quality Know-how Service Proquina ensuring high quality in line with Bayer standards Reliable what we do,
APIs and Intermediates
 APIs and Intermediates Over 100 years of experience Bayer is one of the world s foremost innovators in health care and a leading supplier of prescription products, non-prescription medicines and medical
APIs and Intermediates Over 100 years of experience Bayer is one of the world s foremost innovators in health care and a leading supplier of prescription products, non-prescription medicines and medical
Switzerland EXPERTS TAKING CARE
 CordenPharma Switzerland EXPERTS TAKING CARE CordenPharma Switzerland Eichenweg 1 4410 Liestal Switzerland Phone +41 61 906 59 59 Fax +41 61 906 59 58 sales@cordenpharma.com www.cordenpharma.com Our History
CordenPharma Switzerland EXPERTS TAKING CARE CordenPharma Switzerland Eichenweg 1 4410 Liestal Switzerland Phone +41 61 906 59 59 Fax +41 61 906 59 58 sales@cordenpharma.com www.cordenpharma.com Our History
News Release. Even more attractive to life science and fine chemical industries. Saltigo at CPhI 2017, Frankfurt, October 24-26, 2017, Stand 111H21
 Saltigo at CPhI 2017, Frankfurt, October 24-26, 2017, Stand 111H21 Even more attractive to life science and fine chemical industries Saltigo GmbH Contact: Ilona Kawan LANXESS AG Corporate Communications
Saltigo at CPhI 2017, Frankfurt, October 24-26, 2017, Stand 111H21 Even more attractive to life science and fine chemical industries Saltigo GmbH Contact: Ilona Kawan LANXESS AG Corporate Communications
Your Complete Quality Solution Provider for the Life Science Industry
 Your Complete Quality Solution Provider for the Life Science Industry PQE at-a-glance Our broad services portfolio, extensive experience, effective project management, and exceptional cost effectiveness,
Your Complete Quality Solution Provider for the Life Science Industry PQE at-a-glance Our broad services portfolio, extensive experience, effective project management, and exceptional cost effectiveness,
Lonza Nansha, China For Global Excellence in Advanced Chemical Synthesis
 Pharma&Biotech Lonza Nansha, China For Global Excellence in Advanced Chemical Synthesis Lonza Guangzhou Nansha Ltd «Twenty-five years ago Lonza built one of the world s most advanced cgmp chemical manufacturing
Pharma&Biotech Lonza Nansha, China For Global Excellence in Advanced Chemical Synthesis Lonza Guangzhou Nansha Ltd «Twenty-five years ago Lonza built one of the world s most advanced cgmp chemical manufacturing
Jefferies 2018 Global Healthcare Conference. June 7, 2018
 Jefferies 2018 Global Healthcare Conference June 7, 2018 Forward-Looking Statements Statements in this presentation regarding the future financial and operating results, outlook, growth, prospects, business
Jefferies 2018 Global Healthcare Conference June 7, 2018 Forward-Looking Statements Statements in this presentation regarding the future financial and operating results, outlook, growth, prospects, business
Post approval change of Japanese registration dossiers and impact on market supply
 Post approval change of Japanese registration dossiers and impact on market supply Table of content Table of content Business model evolution of API sales Challenge on Regulatory compliance Risks of Regulatory
Post approval change of Japanese registration dossiers and impact on market supply Table of content Table of content Business model evolution of API sales Challenge on Regulatory compliance Risks of Regulatory
3M Drug Delivery Systems. April 26, 2011
 3M MDI Manufacturing April 26, 2011 3M Drug Delivery Systems Drug Delivery Systems Overview 3M Drug Delivery Systems is a division of 3M that is dedicated to the development and manufacturing of inhalation
3M MDI Manufacturing April 26, 2011 3M Drug Delivery Systems Drug Delivery Systems Overview 3M Drug Delivery Systems is a division of 3M that is dedicated to the development and manufacturing of inhalation
Jefferies 2015 Global Healthcare Conference June 3, 2015
 Jefferies 2015 Global Healthcare Conference June 3, 2015 Forward-Looking Statements This document may contain forward-looking statements within the meaning of the Private Securities Litigation Reform Act
Jefferies 2015 Global Healthcare Conference June 3, 2015 Forward-Looking Statements This document may contain forward-looking statements within the meaning of the Private Securities Litigation Reform Act
QUALITY AGREEMENT. The following Agreement has been concluded between
 QUALITY AGREEMENT The following Agreement has been concluded between LLC LABORATORIES The contract laboratory and service provider 1625 Trinity Dr., Unit 11 Mississauga, Ontario Canada L5T 1W9 Hereinafter
QUALITY AGREEMENT The following Agreement has been concluded between LLC LABORATORIES The contract laboratory and service provider 1625 Trinity Dr., Unit 11 Mississauga, Ontario Canada L5T 1W9 Hereinafter
Table of Contents. Welcome to Dishman. Our Mission. Our Focus: Client Satisfaction
 Welcome to Dishman Our Mission Dishman Group continually invests in the global pharmaceutical industry, ensuring Dishman s business can provide pharmaceutical customers with high-value, high-quality products
Welcome to Dishman Our Mission Dishman Group continually invests in the global pharmaceutical industry, ensuring Dishman s business can provide pharmaceutical customers with high-value, high-quality products
Investor Presentation RBC Capital Markets Global Healthcare Conference February 22, 2017
 Investor Presentation 2017 RBC Capital Markets Global Healthcare Conference February 22, 2017 Forward-Looking Statements This document may contain forward-looking statements within the meaning of the Private
Investor Presentation 2017 RBC Capital Markets Global Healthcare Conference February 22, 2017 Forward-Looking Statements This document may contain forward-looking statements within the meaning of the Private
Jefferies 2017 Global Healthcare Conference. June 7, 2017
 Jefferies 2017 Global Healthcare Conference June 7, 2017 Forward-Looking Statements This document may contain forward-looking statements within the meaning of the Private Securities Litigation Reform Act
Jefferies 2017 Global Healthcare Conference June 7, 2017 Forward-Looking Statements This document may contain forward-looking statements within the meaning of the Private Securities Litigation Reform Act
Validation of Pharmaceutical Manufacturing Process Focus: APIs.
 Validation of Pharmaceutical Manufacturing Process Focus: APIs Validations: Regulatory History (US FDA) Sterilisation 1977 Aseptic processing 1979 Water Systems 1981 Non-sterile Mfg Process 1987 Revised
Validation of Pharmaceutical Manufacturing Process Focus: APIs Validations: Regulatory History (US FDA) Sterilisation 1977 Aseptic processing 1979 Water Systems 1981 Non-sterile Mfg Process 1987 Revised
Stephens Fall Investment Conference
 Stephens Fall Investment Conference November 8, 2017 Steven M. Klosk President & CEO Forward-Looking Statements This document may contain forward-looking statements within the meaning of the Private Securities
Stephens Fall Investment Conference November 8, 2017 Steven M. Klosk President & CEO Forward-Looking Statements This document may contain forward-looking statements within the meaning of the Private Securities
Grindeks years of growth
 Grindeks years of growth Mission, vision and values of Grindeks Focus of Grindeks Business fields Turnover and profit TOP products of Grindeks Manufacturing value final dosage forms with high added value
Grindeks years of growth Mission, vision and values of Grindeks Focus of Grindeks Business fields Turnover and profit TOP products of Grindeks Manufacturing value final dosage forms with high added value
VASUDHA PHARMA CHEM LIMITED, Hyderabad, India
 , Hyderabad, India USFDA accepted facility Korean FDA inspected Mexican MoH - GMP certified WHO-GMP certified , Hyderabad, India Contributing to affordable healthcare Head office : Vasudha Pharma Chem
, Hyderabad, India USFDA accepted facility Korean FDA inspected Mexican MoH - GMP certified WHO-GMP certified , Hyderabad, India Contributing to affordable healthcare Head office : Vasudha Pharma Chem
MDI Manufacturing Services
 MDI Manufacturing Services Presenter, Date Who we are 3M Drug Delivery Systems is a division of 3M dedicated to working together with pharmaceutical and biotech companies to bring new and improved products
MDI Manufacturing Services Presenter, Date Who we are 3M Drug Delivery Systems is a division of 3M dedicated to working together with pharmaceutical and biotech companies to bring new and improved products
COMPANY PROFILE. Dishman offers innovation, security of supply and value for money. Dishman Group. Research EARLY STAGE API. Preclinical.
 COMPANY PROFILE Dishman is a global outsourcing partner for the pharmaceutical industry offering a portfolio of development, scale-up and services. Dishman improves its customers businesses by providing
COMPANY PROFILE Dishman is a global outsourcing partner for the pharmaceutical industry offering a portfolio of development, scale-up and services. Dishman improves its customers businesses by providing
Stellence Pharmscience Pvt Ltd. 456, Road #3, Industrial Area, Jigani, Bangalore South,
 Stellence Pharmscience Pvt Ltd 456, Road #3, Industrial Area, Jigani, Bangalore South, 562 105 1 Stellence Background Stellence Pharmscience Pvt Limited (SPPL) is promoted by Ramachandran Radhakrishnan
Stellence Pharmscience Pvt Ltd 456, Road #3, Industrial Area, Jigani, Bangalore South, 562 105 1 Stellence Background Stellence Pharmscience Pvt Limited (SPPL) is promoted by Ramachandran Radhakrishnan
GUIDANCE ON THE GMP CLEARANCE OF OVERSEAS MEDICINE MANUFACTURERS
 GUIDANCE ON THE GMP CLEARANCE OF OVERSEAS MEDICINE MANUFACTURERS 16 th Edition March 2008 www.tga.gov.au/manuf/gmpsom.htm Page 1 of 18 Table of Contents Introduction... 3 International Arrangements...
GUIDANCE ON THE GMP CLEARANCE OF OVERSEAS MEDICINE MANUFACTURERS 16 th Edition March 2008 www.tga.gov.au/manuf/gmpsom.htm Page 1 of 18 Table of Contents Introduction... 3 International Arrangements...
DIFFERING CMC REQUIREMENTS: US AND EU
 WHITEPAPER DIFFERING CMC REQUIREMENTS: US AND EU Torsten Schnetgoeke M.Sc. Senior Manager, Regulatory Affairs torsten.schnetgoeke@pharmalex.com Torsten joined PharmaLex in 2015 as a Senior Manager in Regulatory
WHITEPAPER DIFFERING CMC REQUIREMENTS: US AND EU Torsten Schnetgoeke M.Sc. Senior Manager, Regulatory Affairs torsten.schnetgoeke@pharmalex.com Torsten joined PharmaLex in 2015 as a Senior Manager in Regulatory
Enforcement of Compliance with GMPs in API manufacture
 Enforcement of Compliance with GMPs in API manufacture Guy Villax Copyright Hovione 2004 No one is more active in pharmaceutical ingredients Agenda What are APIs, GMP What is the legal framework What is
Enforcement of Compliance with GMPs in API manufacture Guy Villax Copyright Hovione 2004 No one is more active in pharmaceutical ingredients Agenda What are APIs, GMP What is the legal framework What is
Quality Agreement. by and between. Supplier Name. Address: and. Client Name: Address:
 NOTE TO USERS This Quality Agreement template was developed by the Bulk Pharmaceutical Task Force (BPTF), an affiliate organization of the Society of Chemical Manufacturers and Affiliates (SOCMA), as a
NOTE TO USERS This Quality Agreement template was developed by the Bulk Pharmaceutical Task Force (BPTF), an affiliate organization of the Society of Chemical Manufacturers and Affiliates (SOCMA), as a
Medical Device Single Audit Program (MDSAP) Copyright 2014 BSI. All rights reserved.
 Medical Device Single Audit Program (MDSAP) International Medical Device Regulatory Forum (IMDRF) IMDRF Management Committee (MC) regulators: Australia, Brazil, Canada, China, the European Union, Japan
Medical Device Single Audit Program (MDSAP) International Medical Device Regulatory Forum (IMDRF) IMDRF Management Committee (MC) regulators: Australia, Brazil, Canada, China, the European Union, Japan
The largest network of harmonized bio/ pharmaceutical GMP product testing labs worldwide, Eurofins BioPharma Product Testing enables companies to
 The largest network of harmonized bio/ pharmaceutical GMP product testing labs worldwide, Eurofins BioPharma Product Testing enables companies to advance candidates from development through commercialization
The largest network of harmonized bio/ pharmaceutical GMP product testing labs worldwide, Eurofins BioPharma Product Testing enables companies to advance candidates from development through commercialization
Manufacturers of. Pharmaceutical Formulations Active Pharmaceutical Ingredients Drug Intermediates ULTRATECH INDIA LIMITED
 Manufacturers of Pharmaceutical Formulations Active Pharmaceutical Ingredients Drug Intermediates ULTRATECH INDIA LIMITED Profile Ultratech India Limited is a WHO-GMP and ISO certified Pharmaceutical company
Manufacturers of Pharmaceutical Formulations Active Pharmaceutical Ingredients Drug Intermediates ULTRATECH INDIA LIMITED Profile Ultratech India Limited is a WHO-GMP and ISO certified Pharmaceutical company
Company Overview and Products & Services
 Company verview and Products & Services amari Chemicals - Content 1. amari Chemicals: Company verview 2. ur Products & Services Peptides R&D Services Manufacturing Services Corporate istory 1948 amari
Company verview and Products & Services amari Chemicals - Content 1. amari Chemicals: Company verview 2. ur Products & Services Peptides R&D Services Manufacturing Services Corporate istory 1948 amari
Drug Quality Assurance: Systems at ChemCon Author: Dr. Peter Gockel
 Drug Quality Assurance: Systems at ChemCon Author: Dr. Peter Gockel On February 13th, 2006, the FOOD AND DRUG ADMINISTRATION (FDA) implemented a revision to the Compliance Program Guidance Manual for active
Drug Quality Assurance: Systems at ChemCon Author: Dr. Peter Gockel On February 13th, 2006, the FOOD AND DRUG ADMINISTRATION (FDA) implemented a revision to the Compliance Program Guidance Manual for active
Business Units contract development and manufacturing cost effective processes reliability of supply. total API solution retroviral APIs
 Business Units Building on our strong capabilities in chemical development and manufacturing, Laurus Labs has developed an in-house range of APIs and related intermediates. Focusing on chemistries where
Business Units Building on our strong capabilities in chemical development and manufacturing, Laurus Labs has developed an in-house range of APIs and related intermediates. Focusing on chemistries where
DOW PuraGuard Propylene Glycol USP/EP. Your Security for Purity
 DOW PuraGuard Propylene Glycol USP/EP Your Security for Purity Purity Plus A very high dedication to consistent quality and purity governs the entire product supply process for DOW PuraGuard Propylene
DOW PuraGuard Propylene Glycol USP/EP Your Security for Purity Purity Plus A very high dedication to consistent quality and purity governs the entire product supply process for DOW PuraGuard Propylene
Anushri Global Pharma Consultants
 Anushri Global Pharma Consultants Your Knowledge Partner In Training, Consulting & Placement Anushri Global Pharma Consultants A Catalyst to sustainable growth Page 1 of 9 About Us Training is important
Anushri Global Pharma Consultants Your Knowledge Partner In Training, Consulting & Placement Anushri Global Pharma Consultants A Catalyst to sustainable growth Page 1 of 9 About Us Training is important
THE R&D LABORATORY THAT BOOSTS YOUR DEVELOPMENT PROJECTS
 THE R&D LABORATORY THAT BOOSTS YOUR DEVELOPMENT PROJECTS CREATED IN 2011 WITH A PROVEN EXPERTISE IN AUDIT AND REGULATORY AFFAIRS, BIOLABEX NORTH AMERICA PARIS EUROPE ASIA EXTENDS ITS OFFERINGS AND IS NOW
THE R&D LABORATORY THAT BOOSTS YOUR DEVELOPMENT PROJECTS CREATED IN 2011 WITH A PROVEN EXPERTISE IN AUDIT AND REGULATORY AFFAIRS, BIOLABEX NORTH AMERICA PARIS EUROPE ASIA EXTENDS ITS OFFERINGS AND IS NOW
EMPROVE For Raw and Starting Materials & For Filtration Devices and Single Use Systems. Jan Thomsen Warsaw, November 15 th, 2016
 EMPROVE For Raw and Starting Materials & For Filtration Devices and Single Use Systems Jan Thomsen Warsaw, November 15 th, 2016 2 Content Emprove - An Introduction Emprove for Raw and Starting Materials
EMPROVE For Raw and Starting Materials & For Filtration Devices and Single Use Systems Jan Thomsen Warsaw, November 15 th, 2016 2 Content Emprove - An Introduction Emprove for Raw and Starting Materials
KINGSMANN CARE GROUP
 PHARMA CONSULTANTS KINGSMANN CARE GROUP KINGSMANN CONSULTANCY SERVICES Thank you for taking interest in Kingsmann Consultancy Services. Kingsmann Consultancy (KC) is a leading business development-consulting
PHARMA CONSULTANTS KINGSMANN CARE GROUP KINGSMANN CONSULTANCY SERVICES Thank you for taking interest in Kingsmann Consultancy Services. Kingsmann Consultancy (KC) is a leading business development-consulting
Performance. concerns?
 Performance concerns? There is hardly anything in the world that some man can t make a little worse and sell a little cheaper, and the people who consider price only are this man s lawful prey. John Ruskin
Performance concerns? There is hardly anything in the world that some man can t make a little worse and sell a little cheaper, and the people who consider price only are this man s lawful prey. John Ruskin
Exclusive Manufacturing Solutions
 Exclusive Manufacturing Solutions Helsinn Advanced Synthesis SA Making the difference with highest quality manufacturing facilities, expertise, investments and commitment 1 Index of Contents Building
Exclusive Manufacturing Solutions Helsinn Advanced Synthesis SA Making the difference with highest quality manufacturing facilities, expertise, investments and commitment 1 Index of Contents Building
IMPLEMENTATION OF THE EU FALSIFIED MEDICINES DIRECTIVE, 2011/62/EU. IPA/EDQM/WHO 2012, Mumbai
 IMPLEMENTATION OF THE EU FALSIFIED MEDICINES DIRECTIVE, 2011/62/EU from the perspective of Indian manufacturers and Indian authorities IPA/EDQM/WHO 2012, Mumbai S.M.MUDDA Micro Labs Limited, Bangalore
IMPLEMENTATION OF THE EU FALSIFIED MEDICINES DIRECTIVE, 2011/62/EU from the perspective of Indian manufacturers and Indian authorities IPA/EDQM/WHO 2012, Mumbai S.M.MUDDA Micro Labs Limited, Bangalore
CREATING TOMORROW S SOLUTIONS BIOPHARMACEUTICALS I CONTRACT MANUFACTURING WACKER BIOTECH: THE MICROBIAL CMO
 CREATING TOMORROW S SOLUTIONS BIOPHARMACEUTICALS I CONTRACT MANUFACTURING WACKER BIOTECH: THE MICROBIAL CMO STATE-OF-THE-ART GMP FACILITIES Our sites in Jena and Halle (Germany) provide a complete range
CREATING TOMORROW S SOLUTIONS BIOPHARMACEUTICALS I CONTRACT MANUFACTURING WACKER BIOTECH: THE MICROBIAL CMO STATE-OF-THE-ART GMP FACILITIES Our sites in Jena and Halle (Germany) provide a complete range
We make drugs smarter
 We make drugs smarter Brookwood Pharmaceutical Services WE MAKE DRUGS SMARTER Using our 40 years of experience, our state of the art facilities and our IP protected technologies, we leverage existing capabilities
We make drugs smarter Brookwood Pharmaceutical Services WE MAKE DRUGS SMARTER Using our 40 years of experience, our state of the art facilities and our IP protected technologies, we leverage existing capabilities
API Assessment Activities
 API Assessment Activities Antony Fake PhD WHO Medicines Prequalification Programme 1 3.2.S.3.2 Impurities, Malaysia, 29 September 2011 2 Overview APIMF Procedure API PQ Procedure What is new? 3 Abbreviations
API Assessment Activities Antony Fake PhD WHO Medicines Prequalification Programme 1 3.2.S.3.2 Impurities, Malaysia, 29 September 2011 2 Overview APIMF Procedure API PQ Procedure What is new? 3 Abbreviations
BRUSSELS CORDENPHARMA EXPERTS TAKING CARE
 CORDENPHARMA BRUSSELS EXPERTS TAKING CARE Corden Pharma Brussels S.A. 310 rue de Ransbeek 1120 Brussels Belgium Phone +32 2 263 1411 www.cordenpharma.com/contact-us/ www.cordenpharma.com Our History 1987:
CORDENPHARMA BRUSSELS EXPERTS TAKING CARE Corden Pharma Brussels S.A. 310 rue de Ransbeek 1120 Brussels Belgium Phone +32 2 263 1411 www.cordenpharma.com/contact-us/ www.cordenpharma.com Our History 1987:
CORDENPHARMA BRUSSELS EXPERTS TAKING CARE
 CORDENPHARMA BRUSSELS EXPERTS TAKING CARE Our History 1987: Peptisyntha founded by Solvay 1996: First pre-approval FDA inspection for a major peptide made by Peptisyntha for the worldwide market. Following
CORDENPHARMA BRUSSELS EXPERTS TAKING CARE Our History 1987: Peptisyntha founded by Solvay 1996: First pre-approval FDA inspection for a major peptide made by Peptisyntha for the worldwide market. Following
Innovative Pharmaceutical Solutions for Discovery Chemistry, Biology and cgmp Manufacturing
 Innovative Pharmaceutical Solutions for Discovery Chemistry, Biology and cgmp Manufacturing Overview Serving biotech and pharma community since 1998 Proven track record of advancing small molecules from
Innovative Pharmaceutical Solutions for Discovery Chemistry, Biology and cgmp Manufacturing Overview Serving biotech and pharma community since 1998 Proven track record of advancing small molecules from
Evonik Birmingham Laboratories
 Evonik Birmingham Laboratories An integrated CDMO for advanced parenterals with a portfolio of delivery technologies, formulation development services and GMP manufacturing Evonik Birmingham Laboratories
Evonik Birmingham Laboratories An integrated CDMO for advanced parenterals with a portfolio of delivery technologies, formulation development services and GMP manufacturing Evonik Birmingham Laboratories
Best-in-class API / HPAPI / Small Molecules Custom Manufacturing
 Best-in-class API / HPAPI / Small Molecules Custom Manufacturing Tippecanoep Ham Hanau Slove enska L upča Dossenh senheim Nanning Production and process development Six production locations countless options.
Best-in-class API / HPAPI / Small Molecules Custom Manufacturing Tippecanoep Ham Hanau Slove enska L upča Dossenh senheim Nanning Production and process development Six production locations countless options.
"Saltigo - Customized Competence"
 News Release Saltigo GmbH Contact: Udo Erbstößer LANXESS Deutschland GmbH Corporate Communications Trade & Technical Press 51369 Leverkusen Germany Phone +49 214 30-54529 Fax +49 214 30-44865 udo.erbstoesser@lanxess.com
News Release Saltigo GmbH Contact: Udo Erbstößer LANXESS Deutschland GmbH Corporate Communications Trade & Technical Press 51369 Leverkusen Germany Phone +49 214 30-54529 Fax +49 214 30-44865 udo.erbstoesser@lanxess.com
RISE Research Institutes of Sweden Division Bioscience and Materials
 RISE Surface, Process & Formulation William Mackintosh RISE Research Institutes of Sweden Division Bioscience and Materials Agenda Description of the Active Pharmaceutical Ingredient (API) production equipment.
RISE Surface, Process & Formulation William Mackintosh RISE Research Institutes of Sweden Division Bioscience and Materials Agenda Description of the Active Pharmaceutical Ingredient (API) production equipment.
The Future of Pharmaceuticals for Human Use in Europe Cefic Input to the Consultation Process: Deadline for Submission 12/10/2007 Date: 10/10/07
 The Future of Pharmaceuticals for Human Use in Europe Cefic Input to the Consultation Process: Deadline for Submission 12/10/2007 Date: 10/10/07 CEFIC is the organisation that represents national federations,
The Future of Pharmaceuticals for Human Use in Europe Cefic Input to the Consultation Process: Deadline for Submission 12/10/2007 Date: 10/10/07 CEFIC is the organisation that represents national federations,
MADE WITH CARE. FDF Generics Chlorhexidine API
 MADE WITH CARE FDF Generics Chlorhexidine API MEDICHEM MADE WITH CARE EXPERTISE VALUE-ADDED SOLUTIONS RELIABILITY At a Glance Meet an independently-owned developer and manufacturer of Finished Dosage Forms
MADE WITH CARE FDF Generics Chlorhexidine API MEDICHEM MADE WITH CARE EXPERTISE VALUE-ADDED SOLUTIONS RELIABILITY At a Glance Meet an independently-owned developer and manufacturer of Finished Dosage Forms
PRAXIS. A publication by Bioengineering AG
 PRAXIS A publication by Bioengineering AG Portrait of Rentschler Biotechnologie GmbH, a globally active service company that supports its clients in the development, production, and registration of biopharmaceuticals.
PRAXIS A publication by Bioengineering AG Portrait of Rentschler Biotechnologie GmbH, a globally active service company that supports its clients in the development, production, and registration of biopharmaceuticals.
Your bridge to. better medicines
 Your bridge to better medicines At a Glance Anapharm Bioanalytics is a client-oriented, GLP-certified, FDA-inspected, GCP-compliant and ANVISA-certified bioanalytical contract research organization (CRO)
Your bridge to better medicines At a Glance Anapharm Bioanalytics is a client-oriented, GLP-certified, FDA-inspected, GCP-compliant and ANVISA-certified bioanalytical contract research organization (CRO)
Your partner in the pharmaceutical industry
 English Micronisation- and milling service Micronised by GfM Your API Particle size (µm) Your partner in the pharmaceutical industry The... always that little bit finer. We are a family business in the
English Micronisation- and milling service Micronised by GfM Your API Particle size (µm) Your partner in the pharmaceutical industry The... always that little bit finer. We are a family business in the
Materials Management Traceability, CEPs and managing non-conforming sites
 Materials Management Traceability, CEPs and managing non-conforming sites IMB Information Day, 14 th October 2010 Dr Cormac Dalton Inspector 13-Oct-10 Slide 1 Managing Supply Supply Demand Supply chain
Materials Management Traceability, CEPs and managing non-conforming sites IMB Information Day, 14 th October 2010 Dr Cormac Dalton Inspector 13-Oct-10 Slide 1 Managing Supply Supply Demand Supply chain
catalogue Key Organics Chemistry Innovation Quality
 2013 catalogue Key Organics Chemistry Innovation Quality www.keyorganics.net 1 Key Organics Chemistry Innovation Quality The Key Organics portfolio offers a diverse range of chemistry products and services
2013 catalogue Key Organics Chemistry Innovation Quality www.keyorganics.net 1 Key Organics Chemistry Innovation Quality The Key Organics portfolio offers a diverse range of chemistry products and services
Europe s new approach to assurance of API quality and its implications for manufacturers and producing countries
 Europe s new approach to assurance of API quality and its implications for manufacturers and producing countries IPA / EDQM / WHO Mumbai Conference 28 September 2012 Dr Florence Benoit-Guyod, EDQM Inspector,
Europe s new approach to assurance of API quality and its implications for manufacturers and producing countries IPA / EDQM / WHO Mumbai Conference 28 September 2012 Dr Florence Benoit-Guyod, EDQM Inspector,
EFFECTIVE PROCESS VALIDATION NUSAGE PharmEng Pharmaceutical and Biotechnology Training Program Copyright 2014 NUSAGE-PharmEng.
 EFFECTIVE PROCESS VALIDATION NUSAGE PharmEng Pharmaceutical and Biotechnology Training Program Shaping Human Capital for Challenges in the Pharmaceutical Industry EFFECTIVE PROCESS VALIDATION Objective
EFFECTIVE PROCESS VALIDATION NUSAGE PharmEng Pharmaceutical and Biotechnology Training Program Shaping Human Capital for Challenges in the Pharmaceutical Industry EFFECTIVE PROCESS VALIDATION Objective
Chromeleon CDS delivers efficiency gains and compliance and data integrity improvements to UK CRO/CMO
 CASE STUDY Sterling Pharma Solutions Chromeleon CDS delivers efficiency gains and compliance and data integrity improvements to UK CRO/CMO Brian Alliston is currently a Data Integrity Expert and CDS Specialist
CASE STUDY Sterling Pharma Solutions Chromeleon CDS delivers efficiency gains and compliance and data integrity improvements to UK CRO/CMO Brian Alliston is currently a Data Integrity Expert and CDS Specialist
Making the Right Chemistry Happen. Expertise in Hazardous Chemistry
 Making the Right Chemistry Happen. Expertise in Hazardous Chemistry Elppe, over the past 22 years has carved a niche for itself in the chemical industry handling range of projects involving hazardous reactions
Making the Right Chemistry Happen. Expertise in Hazardous Chemistry Elppe, over the past 22 years has carved a niche for itself in the chemical industry handling range of projects involving hazardous reactions
The procedure of Certification of Suitability to the Monographs of the European Pharmacopoeia (CEP) and the EDQM Inspection Programme
 18/10/2016 The procedure of Certification of Suitability to the Monographs of the European Pharmacopoeia (CEP) and the EDQM Inspection Programme Chinese & European Pharmacopoeias Workshop 17 October 2016
18/10/2016 The procedure of Certification of Suitability to the Monographs of the European Pharmacopoeia (CEP) and the EDQM Inspection Programme Chinese & European Pharmacopoeias Workshop 17 October 2016
Amber Somoza, Technical Services, Gilead San Dimas 21
 Amber Somoza, Technical Services, Gilead San Dimas 21 Each of the products Gilead researches and develops aligns with therapeutic areas that together impact millions of people in both the developed and
Amber Somoza, Technical Services, Gilead San Dimas 21 Each of the products Gilead researches and develops aligns with therapeutic areas that together impact millions of people in both the developed and
CU Chemie Uetikon GmbH
 CU Chemie Uetikon GmbH Challenges in API manufacturing UETIKN Dr. Hendrik Baumann, Commercial Director www.uetikon.com First of all... UETIKN... CNGRATULATINS to the 5th anniversary of Rapid Pharma Development
CU Chemie Uetikon GmbH Challenges in API manufacturing UETIKN Dr. Hendrik Baumann, Commercial Director www.uetikon.com First of all... UETIKN... CNGRATULATINS to the 5th anniversary of Rapid Pharma Development
ICH Q9 Q uality R isk M anagement
 National University of Singapore Academy of GxP Excellence (NUSAGE) & PharmEng Technology Presents ICH Q9 Q uality R isk M anagement Part of the Pharmaceutical and Biotechnology Training Courses Instructor
National University of Singapore Academy of GxP Excellence (NUSAGE) & PharmEng Technology Presents ICH Q9 Q uality R isk M anagement Part of the Pharmaceutical and Biotechnology Training Courses Instructor
Avra Laboratories Pvt. Ltd.
 Avra Laboratories Pvt. Ltd. Company Overview Introduction Avra Laboratories Private Limited is amongst the first companies in India to focus on Providing high-end contract research Custom synthesis of
Avra Laboratories Pvt. Ltd. Company Overview Introduction Avra Laboratories Private Limited is amongst the first companies in India to focus on Providing high-end contract research Custom synthesis of
API Assessment Activities. Antony Fake, PhD
 API Assessment Activities Antony Fake, PhD 2 Overview APIMF Procedure API PQ Procedure What is new? 3 Abbreviations API Active Pharmaceutical Ingredient FPP Finished Pharmaceutical Product API PQ API Prequalification
API Assessment Activities Antony Fake, PhD 2 Overview APIMF Procedure API PQ Procedure What is new? 3 Abbreviations API Active Pharmaceutical Ingredient FPP Finished Pharmaceutical Product API PQ API Prequalification
GLOBAL SUPPORT FOR A QUALITY SOLUTION
 GLOBAL SUPPORT FOR A QUALITY SOLUTION THE ULTIMATE PEPTIDE PARTNER Dear Customer, Thank you for your continued support and for the confidence you have placed in the PolyPeptide Group. Our goal is to be
GLOBAL SUPPORT FOR A QUALITY SOLUTION THE ULTIMATE PEPTIDE PARTNER Dear Customer, Thank you for your continued support and for the confidence you have placed in the PolyPeptide Group. Our goal is to be
NUVISAN GROUP. Our Philosophy
 NUVISAN GROUP Our Philosophy 2 EARLY DEVELOPMENT Introduction to Nuvisan Fully integrated Contract Research Organisation headquarted in Neu-Ulm, southern part of Germany. Nuvisan - Early Development Headquartered
NUVISAN GROUP Our Philosophy 2 EARLY DEVELOPMENT Introduction to Nuvisan Fully integrated Contract Research Organisation headquarted in Neu-Ulm, southern part of Germany. Nuvisan - Early Development Headquartered
A NUSAGE-PharmEng Pharmaceutical and Biotechnology Training Program
 A NUSAGE-PharmEng Pharmaceutical and Biotechnology Training Program Shaping Human Capital for Challenges in the Pharmaceutical Industry EFFECTIVE PROCESS VALIDATION Instructor Rick Ng Date and Time 10-11
A NUSAGE-PharmEng Pharmaceutical and Biotechnology Training Program Shaping Human Capital for Challenges in the Pharmaceutical Industry EFFECTIVE PROCESS VALIDATION Instructor Rick Ng Date and Time 10-11
EU Unannounced Audits (UAV) Experience To Date Medical Device Single Audit Program (MDSAP) Copyright 2015 BSI. All rights reserved.
 EU Unannounced Audits (UAV) Experience To Date Medical Device Single Audit Program (MDSAP) Copyright 2015 BSI. All rights reserved. Background & Requirements - See Webinars BSI Website Copyright 2015 BSI.
EU Unannounced Audits (UAV) Experience To Date Medical Device Single Audit Program (MDSAP) Copyright 2015 BSI. All rights reserved. Background & Requirements - See Webinars BSI Website Copyright 2015 BSI.
NUSAGE PharmEng. Pharmaceutical and Biotechnology Training Program FUNDAMENTALS OF MEDICAL DEVICE REGULATORY AFFAIRS
 FUNDAMENTALS OF MEDICAL DEVICE REGULATORY AFFAIRS NUSAGE PharmEng Pharmaceutical and Biotechnology Training Program Shaping Human Capital for Challenges in the Pharmaceutical Industry FUNDAMENTALS OF MEDICAL
FUNDAMENTALS OF MEDICAL DEVICE REGULATORY AFFAIRS NUSAGE PharmEng Pharmaceutical and Biotechnology Training Program Shaping Human Capital for Challenges in the Pharmaceutical Industry FUNDAMENTALS OF MEDICAL
SCHOTT is an international technology group with more than 125 years of experience in the areas of specialty glasses and materials and advanced
 Ampoules 2 SCHOTT is an international technology group with more than 125 years of experience in the areas of specialty glasses and materials and advanced technologies. With our high-quality products and
Ampoules 2 SCHOTT is an international technology group with more than 125 years of experience in the areas of specialty glasses and materials and advanced technologies. With our high-quality products and
Complete Service for Product Development
 We can help make your ideas become reality Complete Service for Product Development Hydromer Inc. Facilities, Laboratories Analysis Testing Polymer Synthesis Microbiology Polymer Production Web Coating/Film
We can help make your ideas become reality Complete Service for Product Development Hydromer Inc. Facilities, Laboratories Analysis Testing Polymer Synthesis Microbiology Polymer Production Web Coating/Film
Vetter Development Service
 A unique blend of expertise for clinical manufacturing Vetter Development Service Supporting your compound s success Answers that work 1 Your molecule s future starts here. By the time your new injectable
A unique blend of expertise for clinical manufacturing Vetter Development Service Supporting your compound s success Answers that work 1 Your molecule s future starts here. By the time your new injectable
Cooperation in the field of toxicology, analytics, R&D and production
 Cooperation in the field of toxicology, analytics, R&D and production Where you can find us 2 2 VUOS PARTNER FOR COOPERATION History 3 3 VUOS PARTNER FOR COOPERATION Organizational structure 1. R&D and
Cooperation in the field of toxicology, analytics, R&D and production Where you can find us 2 2 VUOS PARTNER FOR COOPERATION History 3 3 VUOS PARTNER FOR COOPERATION Organizational structure 1. R&D and
For personal use only. AGM PRESENTATION November 2018
 AGM PRESENTATION November 2018 Investment Focus Eve s vision is to be a vertically integrated Health, Food and Wellness Investment company. Investing and owning early growth phase companies with the aim
AGM PRESENTATION November 2018 Investment Focus Eve s vision is to be a vertically integrated Health, Food and Wellness Investment company. Investing and owning early growth phase companies with the aim
ABF Pharmaceutical Services GmbH
 ABF Pharmaceutical Services GmbH Facts and Figures GBA is a European laboratory group with an annual turnover of +40 M. Founded in 1989, Hamburg Our business covers Pharma, Environmental, Foodstuff, Consumer
ABF Pharmaceutical Services GmbH Facts and Figures GBA is a European laboratory group with an annual turnover of +40 M. Founded in 1989, Hamburg Our business covers Pharma, Environmental, Foodstuff, Consumer
CONVENIENCE FOR PATIENTS
 the dermal experience Co-Development CONVENIENCE FOR PATIENTS The dermal delivery route is well established. Millions of patients globally rely on delivery systems for topical and systemic treatments administered
the dermal experience Co-Development CONVENIENCE FOR PATIENTS The dermal delivery route is well established. Millions of patients globally rely on delivery systems for topical and systemic treatments administered
A NUSAGE-PharmEng Pharmaceutical and Biotechnology Training Program
 A NUSAGE-PharmEng Pharmaceutical and Biotechnology Training Program Shaping Human Capital for Challenges in the Pharmaceutical Industry STERILIZATION TECHNIQUES: STEAM & ETHYLENE OXIDE Instructor Dennis
A NUSAGE-PharmEng Pharmaceutical and Biotechnology Training Program Shaping Human Capital for Challenges in the Pharmaceutical Industry STERILIZATION TECHNIQUES: STEAM & ETHYLENE OXIDE Instructor Dennis
CHINOIN PROFESSIONAL RELIABLE DEDICATED PROSTAGLANDIN EXPERTISE
 CHINOIN PROFESSIONAL RELIABLE DEDICATED PROSTAGLANDIN EXPERTISE PROSTAGLANDIN EXPERTISE WITHIN CEPiA/SANOFI Sanofi is more than a pharmaceutical laboratory: it is a diversified healthcare company. It acts
CHINOIN PROFESSIONAL RELIABLE DEDICATED PROSTAGLANDIN EXPERTISE PROSTAGLANDIN EXPERTISE WITHIN CEPiA/SANOFI Sanofi is more than a pharmaceutical laboratory: it is a diversified healthcare company. It acts
Course Title ID Duration Basic Premium. Biological Evaluation of Medical Devices: A Risk-Based Approach N mins
 Pre-Clinical Basic Level : CMDA Certified Medical Device Associate Biological Evaluation of Medical Devices: A Risk-Based Approach N134 63 mins 186.00 144.00 Introduction to Process Validation N135 75
Pre-Clinical Basic Level : CMDA Certified Medical Device Associate Biological Evaluation of Medical Devices: A Risk-Based Approach N134 63 mins 186.00 144.00 Introduction to Process Validation N135 75
Pharmacopoeial Reference Standards
 Pharmacopoeial Reference Standards Industry view point Antony Raj Gomes Head- Quality Management 1 Presentation Overview Indian Industry an update Available guidelines Industry practices Current challenges
Pharmacopoeial Reference Standards Industry view point Antony Raj Gomes Head- Quality Management 1 Presentation Overview Indian Industry an update Available guidelines Industry practices Current challenges
CelaSYS Customized PEGylation Solutions BOS CMC Copenhagen c e l a r e s gmbh Slide 1
 CelaSYS Customized PEGylation Solutions BOS CMC Copenhagen 2012 c e l a r e s gmbh Slide 1 Overview Established in 2003 100% privately owned > 10 years of experience in PEGylation 18 employees, 7 with
CelaSYS Customized PEGylation Solutions BOS CMC Copenhagen 2012 c e l a r e s gmbh Slide 1 Overview Established in 2003 100% privately owned > 10 years of experience in PEGylation 18 employees, 7 with
Your Prestigious Partner to reach your business objectives
 Medical Devices Project Management, QA/RA, Lean & Support Services Your Prestigious Partner to reach your business objectives This document or any portion thereof shall not be reproduced or used in any
Medical Devices Project Management, QA/RA, Lean & Support Services Your Prestigious Partner to reach your business objectives This document or any portion thereof shall not be reproduced or used in any
THERAPEUTIC AREAS CARDIOVASCULAR RESEARCH
 THERAPEUTIC AREAS CARDIOVASCULAR RESEARCH PPD S CARDIOVASCULAR TEAM: OPTIMIZING TRIALS AND IMPROVING OUTCOMES APPLYING EXPERIENCE, INNOVATION AND EFFICIENCY IN THE FIGHT AGAINST CARDIOVASCULAR DISEASE
THERAPEUTIC AREAS CARDIOVASCULAR RESEARCH PPD S CARDIOVASCULAR TEAM: OPTIMIZING TRIALS AND IMPROVING OUTCOMES APPLYING EXPERIENCE, INNOVATION AND EFFICIENCY IN THE FIGHT AGAINST CARDIOVASCULAR DISEASE
SCHOTT Ampoules. Dedicated to Superior Fill + Finish
 SCHOTT Ampoules Dedicated to Superior Fill + Finish 2 SCHOTT is a leading international technology group in the areas of specialty glass and glass-ceramics. With more than 130 years of outstanding development,
SCHOTT Ampoules Dedicated to Superior Fill + Finish 2 SCHOTT is a leading international technology group in the areas of specialty glass and glass-ceramics. With more than 130 years of outstanding development,
SCHOTT Ampoules. Dedicated to Superior Fill + Finish
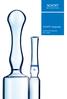 SCHOTT Ampoules Dedicated to Superior Fill + Finish 2 SCHOTT is a leading international technology group in the areas of specialty glass and glass-ceramics. With more than 130 years of outstanding development,
SCHOTT Ampoules Dedicated to Superior Fill + Finish 2 SCHOTT is a leading international technology group in the areas of specialty glass and glass-ceramics. With more than 130 years of outstanding development,
The Quality to Trust On
 The Quality to Trust On Our Quality Policy IS YOUR SAFETY The Quality of our products is an essential requisite for us, prior to release it to the market. So GMPs are applied from the early steps of sourcing
The Quality to Trust On Our Quality Policy IS YOUR SAFETY The Quality of our products is an essential requisite for us, prior to release it to the market. So GMPs are applied from the early steps of sourcing
EUROPEAN INDUSTRIAL PHARMACISTS GROUP. Guidance on CPD for QUALIFIED PERSONS
 EUROPEAN INDUSTRIAL PHARMACISTS GROUP Guidance on CPD for QUALIFIED PERSONS EIPG Guidance on CPD for QP Continuing Professional Development for Qualified Persons, Technical Directors and other Responsible
EUROPEAN INDUSTRIAL PHARMACISTS GROUP Guidance on CPD for QUALIFIED PERSONS EIPG Guidance on CPD for QP Continuing Professional Development for Qualified Persons, Technical Directors and other Responsible
Above & Beyond Quality
 Above & Beyond Quality Science & Testing Quality Ingredients Manufacturing Excellence Natural, Organic & Non-GMO Vegetarian, Allergy-Friendly & Gluten-Free Products Manufacturing Location & Country of
Above & Beyond Quality Science & Testing Quality Ingredients Manufacturing Excellence Natural, Organic & Non-GMO Vegetarian, Allergy-Friendly & Gluten-Free Products Manufacturing Location & Country of
in full-service contract pharmaceutical development and manufacturing
 in full-service contract pharmaceutical development and manufacturing THE Norwich Advantage Norwich is a recognized leader in full service contract pharmaceutical development and manufacturing. As a single-source
in full-service contract pharmaceutical development and manufacturing THE Norwich Advantage Norwich is a recognized leader in full service contract pharmaceutical development and manufacturing. As a single-source
Current Regulatory Considerations and Challenges for Continuous Manufacturing of Pharmaceuticals
 15th DIA Japan Annual Meeting 2018 Promoting Better Collaboration to Drive Global Health and Innovation in an Era of Medical and Scientific Transformation November 11-13, 2018 Tokyo Big Sight V5-S3 Current
15th DIA Japan Annual Meeting 2018 Promoting Better Collaboration to Drive Global Health and Innovation in an Era of Medical and Scientific Transformation November 11-13, 2018 Tokyo Big Sight V5-S3 Current
Corporate Presentation. October 2017
 Corporate Presentation October 2017 Company Overview OPIS is an international CRO providing: - A wide range of clinical and e-clinical services for Pharmaceutical and Biotechnology Industries, Medical
Corporate Presentation October 2017 Company Overview OPIS is an international CRO providing: - A wide range of clinical and e-clinical services for Pharmaceutical and Biotechnology Industries, Medical
Your German source for oncology products
 Your German source for oncology products Dr. Stephan Zinzen, Head of R&D You are looking for a German supplier for the most important oncology products? You need a flexible supply, sometimes even for small
Your German source for oncology products Dr. Stephan Zinzen, Head of R&D You are looking for a German supplier for the most important oncology products? You need a flexible supply, sometimes even for small
Regulatory requirements and registration process of Generic Drugs in China
 13 Regulatory requirements and registration process of Generic Drugs in China Jitendra Kumar Badjatya Deputy Manager-DRA, Montajat Pharmaceutical Company Limited, Dammam, KSA jeetbadjatya@gmail.com ABSTRACT
13 Regulatory requirements and registration process of Generic Drugs in China Jitendra Kumar Badjatya Deputy Manager-DRA, Montajat Pharmaceutical Company Limited, Dammam, KSA jeetbadjatya@gmail.com ABSTRACT
MARKETS & CUSTOMERS. Progressing globalization. Evolving markets
 Peptides are used worldwide in medical research. They contribute immensely to the understanding of diseases and have proven to be effective in medicinal products. Today, peptides can be produced at low
Peptides are used worldwide in medical research. They contribute immensely to the understanding of diseases and have proven to be effective in medicinal products. Today, peptides can be produced at low
CONTRACT RESEARCH SERVICES
 1537 NW 65th Avenue Plantation, FL 33313 USA Phone: (954) 321-8988 Fax: (954) 321-9778 info@receptopharm.com www.receptopharm.com ReceptoPharm has installed the pathways to develop, produce and supply
1537 NW 65th Avenue Plantation, FL 33313 USA Phone: (954) 321-8988 Fax: (954) 321-9778 info@receptopharm.com www.receptopharm.com ReceptoPharm has installed the pathways to develop, produce and supply
HIKMA PHARMACEUTICALS PLC. Merrill Lynch Middle East & North Africa (MENA) Conference 13 December r 2005
 HIKMA PHARMACEUTICALS PLC Merrill Lynch Middle East & North Africa (MENA) Conference 13 December r 2005 Important notice This document and its contents are confidential and may not be redistributed or
HIKMA PHARMACEUTICALS PLC Merrill Lynch Middle East & North Africa (MENA) Conference 13 December r 2005 Important notice This document and its contents are confidential and may not be redistributed or
ABOUT KYMOS. Our commitments. Quality assurance. Location
 Animal Health ABOUT KYMOS Kymos is a CRO that provides high quality analytical services in small molecule and biologic fields for diferent industries: Biotechnology Pharma Veterinary Phytopharma Nutraceuticals
Animal Health ABOUT KYMOS Kymos is a CRO that provides high quality analytical services in small molecule and biologic fields for diferent industries: Biotechnology Pharma Veterinary Phytopharma Nutraceuticals
Empowering innovation.
 Empowering innovation. RESOMER is a synonym for innovation and vision, enabling both pharma and medical device companies to shape and design a better future. 30 years of RESOMER The history of RESOMER
Empowering innovation. RESOMER is a synonym for innovation and vision, enabling both pharma and medical device companies to shape and design a better future. 30 years of RESOMER The history of RESOMER
