LAMINAR FLOW FUEL CELLS & FLUID DYNAMICS IN THE HIGH SCHOOL CLASSROOM
|
|
|
- Emery Franklin
- 5 years ago
- Views:
Transcription
1 LAMINAR FLOW FUEL CELLS & FLUID DYNAMICS IN THE HIGH SCHOOL CLASSROOM Jim Holman Asotin High School Asotin, WA & Alice Hinck Hillcrest High School Idaho Falls, ID Washington State University Mentors Dr. Prashanta Dutta Mechanical Engineering Isaac Sprague Graduate Research Assistant Zaki Jubery Graduate Research Assistant July 2008 The project herein was supported by the National Science Foundation Grant Award No. EEC : Dr. Richard L. Zollars, Principal Investigator. The module was developed by the authors and does not necessarily represent an official endorsement by the National Science Foundation.
2 Table of Contents Page Project summary... Overview of project Intended audience... Estimated duration... Introduction Rationale for module... Science Engineering. Goals... Prerequisite student skills/knowledge Procedures. Safety precautions.. Lab Equipment... Lab activity #1 Lab activity #2 Pictures... References.. Addendum.. Acknowledgements
3 SUMMARY Overview of the Project: The purpose of this project is to explore the dynamics of laminar flow within a fuel cell to generate electricity. Laminar flow is dynamic characteristic of fluids that will eliminate the use of membranes within the fuel cell. This will allow electrons to move more freely between the anode and cathode by not being limited due to the membrane in the fuel cell. This fuel cell would be ideal for powering small portable electronics such as hand-held radios, cellular telephones, and laptop computers. This module will teach students about laminar flow and show how the concept is used in various applications; however, this module will focus on the use of laminar flow in micro-fluidic mixing in order to generate power. Intended Audience: The intended audience for this module is middle and high school students in a science classroom. It is important to ensure that all students participating in this module have the appropriate pre-requisite knowledge required in order to be successful at completing this module. The labs are intended for use during physical science and physics classes covering concepts in electricity. This module may also be used in an Earth Science class to discuss alternative fuels. Estimated Duration: The duration for this module should be 4-5 class periods. This module includes the following lessons/labs: 1. The basics of electricity 2. Use of a multi-meter 3. Laminar Flow 4. Laminar Flow Fuel Cell 3
4 INTRODUCTION In the past decade, scientists have questioned the amount of petroleum products available to fuel the world s transportation sector at the rate of current consumption. Researchers have been tasked with not only finding more petroleum but also finding alternative energy sources. Methanol and ethanol have been heavily researched and have been shown to be able to produce energy on a very small scale. The fuel cell has been developed as an alternative energy source. Fuel cells are similar to batteries which may be fueled by a variety of fuels to include methanol and ethanol. Many types of fuel cells have been developed to include but not limited to: direct methanol fuel cells, alkaline fuel cells, microbial fuel cells, and laminar flow fuel cells. These fuel cells use the same basic principles of generating power using an oxidant electrode (cathode) and a fuel electrode (anode), an electrolyte which carries the charged particles, a catalyst used to speed up the reaction, and a fuel source. The design of the fuel cell must take into account what it will be used when determining the types of electrodes, electrolytes, catalyst, and fuel type. The waste product must also be considered when determining the materials to be used. The laminar flow fuel cell has been developed as a small-scale energy source to power small portable electronics. The biggest difference between the laminar flow fuel cell and the other fuel cells is the use of a membrane. Researchers have found that it is often difficult to develop a membrane that separates the anode and cathode but still allows electrons to pass through. The laminar flow fuel cell eliminates this problem as it does not have a membrane. The oxidant and fuel are pumped into the fuel cell in small amounts at an equal rate, which minimizes the mixing of the two fluids and thus does not require a membrane. 4
5 The laminar-flow fuel cell works by creating a fluid stream consisting of the fuel and oxidant in which the two liquids do not mix. By controlling the rate of flow, the fuel and oxidant are able to enter the channel without mixing initially. There is some mixing towards the end of the channel depending on the depth and length of the channel. The fluids interact with the catalyst that is in the channel, releasing electrons and creating measurable micro-volts. Figure 1 illustrates the input of the oxidant, the yellow stream, and the fuel, the green stream. There is no mixing when the fluid enters the channel. As the fluids move down the channel there is minimal mixing. The fluids interact with the catalysts and Figure 1: Laminar Flow Fuel Cell power is created. The power generated may be measured using a multi-meter. RATIONALE FOR MODULE This module is intended to introduce students to engineering by allowing them to be exposed to what engineers do. By exploring the laminar-flow fuel cell, students will not only get a taste of what engineering is about but will also have the opportunity to be exposed to developing technology that may help end the fuel crisis. 5
6 SCIENCE The scientific basis for this module focuses on fluid dynamics. The following concepts will be addressed in this module: Electricity: The energy made available by moving electrons. Laminar Flow: Laminar flow occurs when fluids are able to flow parallel with virtually little to no mixing. It is thought as being smooth flow and is the opposite of turbulent flow, where mixing occurs. Power: Work done or energy transferred per unit of time. Voltage: The unit of measurement of electrical potential Current: The flow of electrical charge Conductivity: Measure of a material s ability to conduct an electrical current Anode: The positive electrode Cathode: The negative electrode Electrolyte: Any substance that contains free ions that behaves as an electrically conductive medium Oxidant: A chemical compound that readily transfers oxygen atoms 6
7 ENGINEERING The engineering aspects of the module cover a broad range of areas from fabrication to electricity production. Micro-channel fabrication, for example, requires computer automated design and fabrication machines (CAD/CNC). Mixing is the primary challenge for most micro fluidic systems, resulting in innovations in how materials are pumped and channel design. The small diameter of the channels provide for only laminar flow under most conditions. Thus, diffusion rather than turbulence is the primary method for mixing. Controlling the flow through micro and nano- channels is also a challenge. Normal pressure pumps lack the precision necessary on these small scales. Thus, this technology requires the application of micro scale voltage pumps to use electromagnetic charges to generate the pressure to move fluids through the micro channels. (2006 module) However, innovative methods of producing a suitable method for pumping the fluid, therefore replacing the costly and delicate pumps, are introduced in this module. GOALS PREREQUISITE STUDENT SKILLS/KNOWLEDGE In order to successfully complete this module, students should have a basic understanding of the following terms and concepts: Basic chemistry to include: atoms, molecules, electrons, protons, Basic physical science to include: energy transformation (chemical to electrical), mass, volume, density, pressure Basic mathematic skills: graphical analysis, unit conversions, 7
8 EQUIPMENT A classroom set of 10 fabricated fuel cells 700 ml of fuel (0.5 M Methanol M Sulfuric Acid) 700 ml of oxidant (0.5 M sulfuric acid, bubbled with 0 2 gas) ml syringes 10 syringe pumps (mechanical or use the design described below) paper towels 1500 ml deionized water 1 or more multi-meters that measure in milli-volts and milli-ohms. 2 blunt-tipped needles attached to 25 cm of capillary tubing Fabrication of Classroom Supplies Laminar-Flow Channels: The channel was constructed in a rectangle piece of plexi-glass that was 7.5 cm long, 0.5 cm tall, and 3.8 cm wide. Within the rectangle, the channel was created. The two inlet sites were 5 mm in diameter and 2 mm in depth. The length of the two individual channels before joining was 5 mm. The main channel was 2 mm wide, 4 cm in length, and 1 mm in depth. The outlet was 10 mm in diameter and 2 mm in depth. Four screw holes where drilled in the corners and two dowel holes were drilled centered on the long side of the bottom piece. Please see picture in the Picture section of this module. Electrodes and Catalyst: Gold-Leaf was applied using Mona Lisa Gold Leaf Adhesive inside of the side wall of the channel and on top of the channel leading from the channel to the side of the plexi-glass. Gold-Leaf was applied as shown on the picture. Inside the channel on the side-walls, catalyst 8
9 was applied on top of the gold leaf. Platinum black was applied on the cathode side and Platinum-Ruthenium black was applied on the anode side. Two to three applications of the catalyst was applied. After each application, allow about 15 minutes for the catalyst to set prior to applying the next layer. Ideally, wait 24 hours prior to using the fuel cell. (Please refer to the Addendum portion of this module to view the recipe used to produce the catalysts). Electric wires were applied to anode and cathode sides of the channel using silver conducting epoxy. Follow the directions on the package. IMPORTANT: Be sure to use conducting epoxy when applying the wires to the gold-leaf on the anode and cathode sides. Channel Lid: The channel lid was constructed using a rectangle piece of plexi-glass that was identical in size to the piece containing the channel. Screws are used to hold the channel and lid together, with a gasket made of polydimethyl siloxane (PDMS). Follow the directions on the container to make a rectangle sheet of PDMS. Cut out a small rectangle shape large enough to cover both the inlet and outlet of the channel. It should not extend past the screw or dowel holes. Holes should be cut where the tubing for the inlets and outlet will flow through. Please see Pictures section for photos of the lid. Clear plastic tubing was used at both the inlet and outlet sites. The tubing was applied to the lid by cutting the tubing ends at an angle and using pliers to pull the tubing through their designated holes. Once pulled through, the tubing was cut flush with the channel lid using a flat razor. The inlet sites contained tubing that was 1/8 Id (1/16 wall) ¼ Od. The outlet site contained a tube that was 5/16 Id x 1/16 wall(7/16 Od). The length of the tubing will 9
10 depend on where the fuel cell is used and the setup that is designed for the pump and outlet disposal. We used tubing that was 20 cm long. Syringe Pump This syringe pump design was designed by John Johnson of La Center High School and George Petrina of Gonzaga Prep High School. They were SWEET program members in We followed their protocol with minor adjustments in order to keep costs minimal for this project. This syringe pump will replace the syringe pump mentioned in the lab activities that follow in this module. Equipment for pump: 1 Box of copper BBs (6000 count) 3-8 oz cups (Solo Style) 1 Science ring stand with 2 rings and 2 test tube holders Stabilizing Sleeve Syringe Pump Set-Up: 1. Fill syringe to 2 ml of water. 2. With needle of syringe pointed up, depress the syringe until you are certain that all air bubbles have been removed. 3. Place syringe in clamp on ring stand with the needle end pointing down and tighten so it will not move. 4. Use empty cup to adjust the stabilizing sleeve so that the cup can be centered on the syringe but not tip over. 5. Remove cup and fill to approximately ¼ of the cup with BBs. Syringe Friction Setup 10
11 6. Carefully place the cup onto the syringe and observe the end of the needle to look for a drop to begin to form. If a drop forms immediately, then you have too much weight. Remove cup and remove some of the BBs until the cup does not cause a drop to form immediately. 7. Add BBs slowly until a drop begins to form at the end of the needle. 8. Remove cup and record its weight as the Zero weight. PROCEDURES FOR ACTIVITIES (Procedure are on the following pages. The gaps are for page breaks for easier printing) 11
12 Activity/Lab #1 Teacher protocol Safety Precautions: This lab activity does not require additional safety guidelines beyond the normal requirements for any lab activity. Instructional Strategies: The purpose of this activity is to teach students the proper use of a multi-meter. Students will measure conductivity, current and voltage of the different types and sizes of batteries and compare their results. Students will work in groups of 2-4 in order to complete this activity. Materials/Equipment for each group: 1 multi-meter 1 9V battery 1 AA alkaline battery 1 A alkaline battery 1 C alkaline battery 1 D alkaline battery 1.5 V lithium battery 3 V lithium battery 1 activity sheet per student Procedures: Testing Conductivity Testing Current When measuring current: o The black wire should be connected to the COM port. o The red wire should be connected to the Ω o The other side of the red test probe should be connected to the positive side of the battery (cathode). Students should be aware that they should only use the ones marked with == NOT ~ Testing Voltage When measuring voltage: 12
13 o The red wire should be connected to the V o The other side of the red test probe should be connected to the positive side of the battery (cathode). Students should be aware that they should only use the ones marked with == NOT ~ Data Collection: Students will fill in the questions as they follow the directions. Data Analysis: How to use a multi-meter: Students will compare the voltage on the side of the battery with the voltage on their multi-meter. They will then compare their current reading with the teacher s result (can be shared on the overhead for each battery type). Evaluation Protocols: Worksheet/Handout to be Given to Students: (on next page) 13
14 Names Period LAB ACTIVITY #1: USING A MULTIMETER Purpose: To learn how to use a multi-meter to measure voltage and current of various batteries. Materials/Equipment: Multi-meter 1 C alkaline battery 9-V battery 1 D alkaline battery 1 AA alkaline battery 1.5 V lithium battery 1 AAA alkaline battery Electrical tape (if needed) Directions/Procedures: INSTRUCTIONS FOR MEASURING VOLTAGE: 1. Plug the black test probe into the black terminal on the multi-meter labeled COM 2. Plug the red test probe into the red voltage socket labeled V 3. Turn the dial to the V== segment. You may have several numbers to choose from (2, 20, or 200 for example). These are all voltage ranges. A maximum of 2 Volts, 20 volts, and 200 volts. Choose the one that fits the battery. Remember you are using various batteries so be sure to check the voltage on each battery prior to testing for voltage. 4. Take the black test probe and attach it to the negative (--) side of the battery 5. Take the red test probe and attach it to the positive (+) side of the battery. (If you do not get a reading ask your teacher for help.) 6. Record your data on the table on the following page. 14
15 Lab Activity #1: Using a multi-meter continued Battery Type/Size Voltage on Battery Multi-Meter Reading Any differences? 9-Volt AAA alkaline AA alkaline C alkaline D alkaline Large Lithium Small Lithium 1. What would cause the actual voltage to be less than the voltage reading on the side of the battery? 15
16 Lab Activity #1: Using a multi-meter continued INSTRUCTIONS FOR MEASURING CURRENT 1. When you connect the probes, do NOT leave them attached for more than 5 seconds. This will draw energy from the battery. 2. Plug the red test probe into the red A socket. Current is measured in amps. 3. Turn the multi-meter to the A == setting. 4. Take the black test probe and attach it to the negative (--) side of the battery 5. Take the red test probe and attach it to the positive (+) side of the battery. 6. Record your data on the table below. Battery Type/Size Multi-Meter Reading 9-Volt AAA alkaline AA alkaline C alkaline D alkaline Large Lithium Small Lithium 16
17 Lab Activity 1: Post Lab Questions 1. True or False: To measure voltage of a battery the multi-meter should be turned to V~. If false change the answer so it is true: 2. True or False: To measure the current of a battery the multi-meter should be turned to A~. If false change the answer so it is true: 3. Answer the following questions by checking the appropriate box: This test probe plugs into the V/Ω socket. This test probe plugs into the A socket This test probe is ALWAYS plugged into the COM socket To measure voltage this test probe must be plugged into the V== socket To measure current this test probe must be plugged into the 20A socket This test probe touches the positive (+) side of the battery This test probe touches the negative (--) side of the battery Black Test Probe Red Test Probe 17
18 Lab Activity 2: Fluid dynamics and Laminar Flow Teacher protocol Safety Precautions: During this lab activity, students should use eye protection and wear aprons. The dyes used during this lab have the potential to stain clothing. Students with long hair should have it pulled behind their shoulders. Normal lab safety guidelines should be followed as well. Instructional Strategies: The purpose of this activity is to introduce students to fluid dynamics and laminar flow. Laminar flow occurs when a fluid flows uninterrupted. It is referred to as being smooth. This is opposite of turbulent flow which is often referred to as being rough. Students will first observe the teacher s demonstration of laminar flow. They will then create laminar flow in groups of 2-4 in order to complete this activity. Materials/Equipment the class, using 10 cells: Syringe pump (See above directions for a different option to the pump used here) 70 ml of colored water 70 ml of a different colored water 150 ml of deionized water 1-2 fabricated fuel cells 2 syringes with capillary tubing attached Lab activity student sheet for each student Procedures: The teacher should first demonstrate the use of the syringe pump. The teacher should next demonstrate laminar flow. This may be done using the fuel cell with colored water. Worksheet/Handout to be Given to Students: (on next page) 18
19 Names Period LAB ACTIVITY #2: LAMINAR FLOW AND THE LAMINAR FLOW CELL Part A: Laminar Flow Purpose: To visually observe the effect of laminar flow with in a fuel cell channel. Materials/Equipment: Multi-meter Waste Collector Syringe Pump Blunt tipped capillary tube 70 ml of Blue- dyed water Safety Goggles 70 ml of Red-dyed water Papertowels 2-12 ml Syringes Latex or nitrile exam gloves Prepared Fuel Cell Procedure 1. Observe teacher s demonstration on the correct use of syringe pump and blunt tipped capillary tubes. 2. Using correct blunt tipped capillary tube, fill syringe labeled as Fuel/Anode with 10 ml of Red water. Ensure that all air is removed from syringe. 3. Using correct blunt tipped capillary tube, fill syringe labeled as Oxidant/Cathode with 10 ml of Blue water. Ensure that all air is removed from syringe. 4. Attach filled syringes to appropriate fuel cell tubes. It is critical that the Fuel/Anode syringe is attached to the Anode fuel cell tube and the Oxidant/Cathode syringe is attached to the Cathode fuel cell tube. 5. Load syringes into syringe pump and place waste collector at the end of the waste tube. 6. Attach multi-meter to red wires attached to fuel cell. 7. Program syringe pump to desired settings. - Volume: 10 ml, Rate: 1mL/min 19
20 8. Press Run/Stop button on syringe pump. 9. Wait until you see the blue and red liquids collecting in waste tube. 10. Turn multi-meter to the millivolts setting, observe data, then record. 11. Turn multi-meter to milliamps setting, observe data, then record. 12. Stop syringe pump. 13. Change Rate setting to 0.1 ml/min, observe laminar flow and record. 14. Press Run/Stop button on syringe pump. 15. Repeat Step #7-14, but with the Rate at 2 ml/min, 5 ml/min and 10 ml/min. (Refill syringe as necessary). 16. Flush Fuel Cell with deionized water to remove all traces of food coloring. Part B: Measuring Output of the Laminar Fuel Cell Purpose: To create power and analyze data of the Laminar Flow Fuel Cell. Materials/Equipment: Multi-meter 2-12 ml Syringes Syringe Pump Prepared Fuel Cell 70 ml of Blue- dyed Oxidant (0.5M Waste Collector Sulfuric Acid bubbled with O 2 ) Blunt tipped capillary tube 70 ml of Red-dyed Fuel (0.5M Methanol + 0.5M Sulfiric Acid) 20
21 Lab Activity 2 Part B Continued Procedure 1. Using correct blunt tipped capillary tube, fill syringe labeled as Fuel/Anode with 10 ml of Red-dyed Fuel. Ensure that all air is removed from syringe. 2. Using correct blunt tipped capillary tube, fill syringe labeled as Oxidant/Cathode with 10 ml of Blue-dyed Oxidant. Ensured that all air is removed from syringe. 3. Attach filled syringes to appropriate fuel cell tubes. It is critical that the Fuel/Anode syringe is attached to the Anode fuel cell tube and the Oxidant/Cathode syringe is attached to the Cathode fuel cell tube. 4. Load syringes into syringe pump and place waste collector at the end of the waste tube. 5. Attach multi-meter to red wires attached to fuel cell. 6. Program syringe pump to desired settings. a. Volume: 10 ml b. Rate: 1 ml/min 7. Press Run/Stop button on syringe pump. 8. Wait until you see the blue and red liquids collecting in waste tube. 9. Turn multi-meter to the millivolts setting, observe data, then record. 10. Turn multi-meter to milliamps setting, observe data, then record. 11. Stop syringe pump. 12. Change Rate setting to 0.1 ml/min. 13. Press Run/Stop button on syringe pump. Wait one minute, then observe and record millivolt and milliamps data. 21
22 14. Repeat Step #7-14, but with the Rate at 0.01 ml/min, 0.5 ml/min and 2 ml/min. (Refill syringe as necessary). 15. Calculate power and record on data table. 16. Determine which flow rate produces the best Power. 17. Follow procedure steps #1 and # Program syringe pump to desired settings. a. Volume: 10 ml b. Rate: Best Power from step # Start syringe pump. 20. Record the millivolts and milliamps every minute for 15 minutes in data table. 21. Calculate the Power produced at each minute interval. 22. Graph Power-Time. 23. Determine when the Fuel Cell reached it s Peak Performance. 24. Flush Fuel Cell with deionized water. 22
23 Lab Activity 2: Data Sheet Name: Date: Class: Part A: Flow Rate (ml/min) Color(s) Observed Laminar or Turbulent Voltage (volts) Current (amps) Part B: Flow Rate (ml/min) Voltage (volts) Current (amps) Power (volts x amps)
24 Lab Activity 2: Part B Continued: Optimal Flow Rate (ml/min): 0 Time (minutes) Voltage (volts) Current (amps) Power (volts x amps)
25 Lab Activity 2: Post-Lab Questions Name: Date: Class: 1. Define laminar flow and turbulent flow. 2. What electrical terms are volts and amps the units of? 3. At the fuel cells Peak Performance, how many of them would you have to hook together to replace a 9 volt battery? 4. According to the data collected in this lab, at the present time, are fuel cells capable of replacing batteries? Why or why not? 25
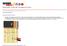
26 Pictures Initial Channel Channel with gold leaf Wires cold soldered to channel and catalyst applied Top piece, with tubes Channel assembled Blunt-tipped needle with attached capillary tube attached PDMS layer (gasket) Fuel cell ready for syringe Initial channel with dowels pump Fuel cell connected to syringe pump that we used in the lab Fuel cell pumping blue and red colored water to show laminar flow 26
27 REFERENCES Bledsoe, Jason & Jonathon Heflick. (Jul 2006). Fluid Dynamics & Micro-Fluidic Mixing Using Micro-Channels in High School Science and Mathematics. Pullman, WA: WSU Electricity. 29 Jul Jul 2008 Fuel Cells 2000 : Fuel Cell Basics : How They Work. 23 Oct Jul Johnson, John & George Petrina. (Jul 2005). Microfluidic Mixing Using Microchannels in High School Science and Math. Pullman, WA: WSU. Membraneless fuel cell is tiny, versatile. 23 Mar Jul
28 ADDENDUM Here are some additional notes from our experiences with this module. -The syringe pump that we used in our research was an expensive model in the lab at WSU. The pump using BB s is a good opportunity to work engineering into this lab. -There is some research that claims that instead of using oxygen bubbled into sulfuric acid as the catalyst, it is possible to use hydrogen peroxide mixed with sulfuric acid. It is recommendation that you use 1 M hydrogen peroxide in 0.5 sulfuric acid. If you are ambitious also try 2M hydrogen peroxide in 0.5M sulfuric acid. For the anode stream, stick with the 0.5M methanol and the 0.5M sulfuric acid. -The directions for the multi-meters may vary based on the brand and type of multi-meter that you have. -The recipe that was used to prepare the platinum black and platinum-ruthenium black catalyst is as follows: -1 part catalyst (say 10 mg) -10 parts milliper water (100mg) -30% by weight Nafion (3mg but because its 5% solution you need to put in 60 mg. The excess water in this step is okay because it evaporates. Nafion is basically the glue) -Mix and sonicate. -Please feel free to contact either of us with questions that you may have with this module. contacts; Jim Holman- jholman@aasd.wednet.edu Alice Hinck- aahinck@hotmail.com 28
29 ACKNOWLEDGEMENTS We would like to thank the people who helped us out with this module. Dr. Prashanta Dutta for his help setting up our project, Isaac Sprague for his guidance, Zaki Jubery for his help in the lab and Sean in Dr. Su Ha s lab for making our catalyst and providing us the chemicals that we needed. 29
LAMINAR FLOW FUEL CELLS & FLUID DYNAMICS IN THE HIGH SCHOOL CLASSROOM
 LAMINAR FLOW FUEL CELLS & FLUID DYNAMICS IN THE HIGH SCHOOL CLASSROOM Chayne Ibrahim Sunnyside High School Sunnyside, WA Joseph Forsman Lewis-Clark State College Lewiston, ID Washington State University
LAMINAR FLOW FUEL CELLS & FLUID DYNAMICS IN THE HIGH SCHOOL CLASSROOM Chayne Ibrahim Sunnyside High School Sunnyside, WA Joseph Forsman Lewis-Clark State College Lewiston, ID Washington State University
APPLICATIONS FOR HYDROGEN FUEL CELL TECHNOLOGY IN HIGH SCHOOL SCIENCE AND MATHEMATICS. EugeneWinkler Pullman High School Pullman, WA
 APPLICATIONS FOR HYDROGEN FUEL CELL TECHNOLOGY IN HIGH SCHOOL SCIENCE AND MATHEMATICS. EugeneWinkler Pullman High School Pullman, WA Marcus McAleer Pullman High School Pullman, WA Jason Bledsoe Pullman
APPLICATIONS FOR HYDROGEN FUEL CELL TECHNOLOGY IN HIGH SCHOOL SCIENCE AND MATHEMATICS. EugeneWinkler Pullman High School Pullman, WA Marcus McAleer Pullman High School Pullman, WA Jason Bledsoe Pullman
Electrochemistry. 2Mg + O 2
 Goals Assemble and run a salt water battery Maximize the generated electric current Make calculations based on data Background is a branch of scientific study that has been around for hundreds of years.
Goals Assemble and run a salt water battery Maximize the generated electric current Make calculations based on data Background is a branch of scientific study that has been around for hundreds of years.
Characteristic and efficiency of PEM fuel cell and PEM electrolyser
 Related topics Electrolysis, electrode polarisation, decomposition voltage, galvanic elements, Faraday s law. Principle and task In a PEM electrolyser, the electrolyte consists of a protonconducting membrane
Related topics Electrolysis, electrode polarisation, decomposition voltage, galvanic elements, Faraday s law. Principle and task In a PEM electrolyser, the electrolyte consists of a protonconducting membrane
Solution Concentrations
 Next Generation Science Standards NGSS Science and Engineering Practices: Asking questions and defining problems Developing and using models Planning and carrying out investigations Analyzing and interpreting
Next Generation Science Standards NGSS Science and Engineering Practices: Asking questions and defining problems Developing and using models Planning and carrying out investigations Analyzing and interpreting
Electricity. Characteristic and efficiency of PEM fuel cell and PEM electrolyser Stationary currents. What you need:
 Stationary currents Electricity Characteristic and efficiency of PEM fuel cell and PEM electrolyser What you can learn about Electrolysis Electrode polarisation Decomposition voltage Galvanic elements
Stationary currents Electricity Characteristic and efficiency of PEM fuel cell and PEM electrolyser What you can learn about Electrolysis Electrode polarisation Decomposition voltage Galvanic elements
Electrolysis, electrode polarisation, decomposition voltage, galvanic elements, Faraday s law.
 Characteristics and efficiency of PEM fuel cell TEP Related Topics Electrolysis, electrode polarisation, decomposition voltage, galvanic elements, Faraday s law. Principle In a PEM electrolyser, the electrolyte
Characteristics and efficiency of PEM fuel cell TEP Related Topics Electrolysis, electrode polarisation, decomposition voltage, galvanic elements, Faraday s law. Principle In a PEM electrolyser, the electrolyte
An Introduction to Sediment Microbial Fuel Cells: Can Electricity really be Dirt Cheap?
 An Introduction to Sediment Microbial Fuel Cells: Can Electricity really be Dirt Cheap? Norlinda Connolly Mansfield High School Mansfield, WA & Patrick Yecha Lyle High School Lyle, WA Washington State
An Introduction to Sediment Microbial Fuel Cells: Can Electricity really be Dirt Cheap? Norlinda Connolly Mansfield High School Mansfield, WA & Patrick Yecha Lyle High School Lyle, WA Washington State
Fluid Mechanics. Vishal Tandon. March 12, 2010
 Fluid Mechanics Vishal Tandon March 12, 2010 1 Introduction In this lab demonstration, you will learn about some of the basic properties of fluids, and how we describe them. In particular, you will learn
Fluid Mechanics Vishal Tandon March 12, 2010 1 Introduction In this lab demonstration, you will learn about some of the basic properties of fluids, and how we describe them. In particular, you will learn
Fuel Cell Car Chem. Lab
 This content should not be copied, translated or distributed in any manner Fuel Cell Car Chem. Lab Name: Class: Date: Chemistry High School 4 hours Objective Perform experiments with a hydrogen fuel cell
This content should not be copied, translated or distributed in any manner Fuel Cell Car Chem. Lab Name: Class: Date: Chemistry High School 4 hours Objective Perform experiments with a hydrogen fuel cell
Energy Conservation and Transformation
 Next Generation Science Standards NGSS Science and Engineering Practices: Asking questions and defining problems Developing and using models Planning and carrying out investigations Analyzing and interpreting
Next Generation Science Standards NGSS Science and Engineering Practices: Asking questions and defining problems Developing and using models Planning and carrying out investigations Analyzing and interpreting
Aluminum air battery activity
 Aluminum air battery activity Overview: Students design and build the simplest possible battery with aluminum foil, salt water and copper wire. Essential Question: How can we capture the energy released
Aluminum air battery activity Overview: Students design and build the simplest possible battery with aluminum foil, salt water and copper wire. Essential Question: How can we capture the energy released
INTRODUCTION TO ELECTROCHEMISTRY: CURRENT, VOLTAGE, & BATTERIES. Introduction. Electrochemistry Revised 4/28/14
 INTRODUCTION TO ELECTROCHEMISTRY: CURRENT, VOLTAGE, & BATTERIES Introduction Electrochemical Cells In this part of the experiment, four half cells are created by immersing metal strips of zinc, copper,
INTRODUCTION TO ELECTROCHEMISTRY: CURRENT, VOLTAGE, & BATTERIES Introduction Electrochemical Cells In this part of the experiment, four half cells are created by immersing metal strips of zinc, copper,
Procedure * You must wear gloves and safety glasses during this lab.
 LAB: Dye- Sensitized Solar Cells Lab Report Due April 25, 2011 Summary: In this lab, you will construct three nanocrystalline dye- sensitized solar cells. You will then measure and compare your solar cells
LAB: Dye- Sensitized Solar Cells Lab Report Due April 25, 2011 Summary: In this lab, you will construct three nanocrystalline dye- sensitized solar cells. You will then measure and compare your solar cells
Heat and Thermal Energy
 Next Generation Science Standards NGSS Science and Engineering Practices: Asking questions and defining problems Developing and using models Planning and carrying out investigations Analyzing and interpreting
Next Generation Science Standards NGSS Science and Engineering Practices: Asking questions and defining problems Developing and using models Planning and carrying out investigations Analyzing and interpreting
Stoichiometry. Lab. FCJJ 16 - Solar Hydrogen Science Kit. Next Generation Science Standards. Initial Prep Time. Lesson Time. Assembly Requirements
 Next Generation Science Standards NGSS Science and Engineering Practices: Asking questions and defining problems Developing and using models Planning and carrying out investigations Analyzing and interpreting
Next Generation Science Standards NGSS Science and Engineering Practices: Asking questions and defining problems Developing and using models Planning and carrying out investigations Analyzing and interpreting
EXTRA CREDIT - EXPERIMENT G ELECTROCHEMISTRY ACTIVITY OF METALS
 EXTRA CREDIT - EXPERIMENT G ELECTROCHEMISTRY ACTIVITY OF METALS INTRODUCTION The objective of this experiment is to develop an abbreviated activity series of metals using: 1. Displacement reactions 2.
EXTRA CREDIT - EXPERIMENT G ELECTROCHEMISTRY ACTIVITY OF METALS INTRODUCTION The objective of this experiment is to develop an abbreviated activity series of metals using: 1. Displacement reactions 2.
March Chemistry Project v3. Chemistry Project
 Chemistry Project Investigate and compare the quantitative effects of changing (a) metal types and (b) fruit/vegetable type on the emf (voltage) produced across two different metals, when the electrolytes
Chemistry Project Investigate and compare the quantitative effects of changing (a) metal types and (b) fruit/vegetable type on the emf (voltage) produced across two different metals, when the electrolytes
Chemical Reactions. Lab. FCJJ 11 - Fuel Cell Car Science Kit. Next Generation Science Standards. Initial Prep Time. Lesson Time. Assembly Requirements
 Next Generation Science Standards NGSS Science and Engineering Practices: Asking questions and defining problems Developing and using models Planning and carrying out investigations Analyzing and interpreting
Next Generation Science Standards NGSS Science and Engineering Practices: Asking questions and defining problems Developing and using models Planning and carrying out investigations Analyzing and interpreting
Manual. (979) T101 RFC Set. T105 HyRunnerBasic. T109 HySpeedster.
 Manual T101 RFC Set T105 HyRunnerBasic T107 HyRunner T109 HySpeedster www.fuelcellstore.com sales@fuelcellstore.com (979) 703-1925 Contents 04 04 05 06 08 11 12 14 16 19 23 24 25 26 28 29 Your Duties as
Manual T101 RFC Set T105 HyRunnerBasic T107 HyRunner T109 HySpeedster www.fuelcellstore.com sales@fuelcellstore.com (979) 703-1925 Contents 04 04 05 06 08 11 12 14 16 19 23 24 25 26 28 29 Your Duties as
COC Biotechnology Program
 COC Biotechnology Program DNA FINGERPRINTING: VERSION C In the time it takes you to complete this lab, your DNA could be extracted, amplified, analyzed and compared. Everything from a criminal past to
COC Biotechnology Program DNA FINGERPRINTING: VERSION C In the time it takes you to complete this lab, your DNA could be extracted, amplified, analyzed and compared. Everything from a criminal past to
Materials. Materials. NOTE Delta Education Customer Service can be reached at
 w Weather and Water Measuring Matter Materials Materials Contents Kit Inventory List... 42 Materials Supplied by the Teacher... 45 Preparing the Kit for Your Classroom... 47 Care, Reuse, and Recycling...
w Weather and Water Measuring Matter Materials Materials Contents Kit Inventory List... 42 Materials Supplied by the Teacher... 45 Preparing the Kit for Your Classroom... 47 Care, Reuse, and Recycling...
A. Introduction. Figure 1 Figure 2
 Varsity Biology Molecular Biotechnology Lab Decode the Candy Dye Essential Questions: How can we use Gel electrophoresis to separate molecules? How do we use the equipment (which will be important in the
Varsity Biology Molecular Biotechnology Lab Decode the Candy Dye Essential Questions: How can we use Gel electrophoresis to separate molecules? How do we use the equipment (which will be important in the
Pre-Lab Exercises Lab 5: Oxidation and Reduction
 Pre-Lab Exercises Lab 5: Oxidation and Reduction Name Date Section 1. What is oxidation? 2. What is reduction? 3. Look at the reaction 2 H 2O 2 H 2 + O 2. Is this an oxidation-reduction reaction? If so,
Pre-Lab Exercises Lab 5: Oxidation and Reduction Name Date Section 1. What is oxidation? 2. What is reduction? 3. Look at the reaction 2 H 2O 2 H 2 + O 2. Is this an oxidation-reduction reaction? If so,
PORTABLE ph METER PRICE CODE NUMBER MODEL 100
 PORTABLE METER MODEL 100 PRICE CODE NUMBER 56-0032 Refer to Bulletin A-301. OPERATION AND SERVICE GUIDE O-670A DEC. 1997 GENERAL INFORMATION Model 100 meter is designed for measuring over the entire 0
PORTABLE METER MODEL 100 PRICE CODE NUMBER 56-0032 Refer to Bulletin A-301. OPERATION AND SERVICE GUIDE O-670A DEC. 1997 GENERAL INFORMATION Model 100 meter is designed for measuring over the entire 0
Ethanol Combustion C 2 H 5
 Goals ᄏᄏ ᄏᄏ ᄏᄏ Use an ethanol fuel cell to produce an electric current Conduct experiments to study its performance Make calculations based on data Background Any organic molecule with an OH group attached
Goals ᄏᄏ ᄏᄏ ᄏᄏ Use an ethanol fuel cell to produce an electric current Conduct experiments to study its performance Make calculations based on data Background Any organic molecule with an OH group attached
Basic Biotechnology Kit
 Basic Biotechnology Kit GEL ELECTROPHORESIS OF DYES Partnership for Biotechnology and Genomics Education Barbara Soots Linda Curro Education Coordinator University of California Davis Assistant Education
Basic Biotechnology Kit GEL ELECTROPHORESIS OF DYES Partnership for Biotechnology and Genomics Education Barbara Soots Linda Curro Education Coordinator University of California Davis Assistant Education
Light. Lab. FCJJ 16 - Solar Hydrogen Science Kit. Next Generation Science Standards. Initial Prep Time. Lesson Time. Assembly Requirements
 Next Generation Science Standards NGSS Science and Engineering Practices: Asking questions and defining problems Developing and using models Planning and carrying out investigations Analyzing and interpreting
Next Generation Science Standards NGSS Science and Engineering Practices: Asking questions and defining problems Developing and using models Planning and carrying out investigations Analyzing and interpreting
Parts Per Million Analysis (PPM)
 Parts Per Million Analysis (PPM) Randy Sharpe, H2 Sciences Inc. What Is "PPM"? PPM is an acronym for "Parts Per Million", a unit of measure that describes how much of one substance (called the "solute")
Parts Per Million Analysis (PPM) Randy Sharpe, H2 Sciences Inc. What Is "PPM"? PPM is an acronym for "Parts Per Million", a unit of measure that describes how much of one substance (called the "solute")
Energy Conservation and Transformation
 Next Generation Science Standards NGSS Science and Engineering Practices: Asking questions and defining problems Developing and using models Planning and carrying out investigations Analyzing and interpreting
Next Generation Science Standards NGSS Science and Engineering Practices: Asking questions and defining problems Developing and using models Planning and carrying out investigations Analyzing and interpreting
Calcium-EDTA Tutorial for the MicroCal ITC200
 Calcium-EDTA Tutorial for the MicroCal ITC200 Getting Started 1. The computer and the ITC200 are normally left on. If they have been switched off for any reason makes sure that you boot them in the order
Calcium-EDTA Tutorial for the MicroCal ITC200 Getting Started 1. The computer and the ITC200 are normally left on. If they have been switched off for any reason makes sure that you boot them in the order
Compact Electro Plating Machine - CEPM-2A USER MANUAL
 1 Compact Electro Plating Machine - CEPM-2A USER MANUAL PRODUCT INTRODUCTION: This compact electroplating machine is a high performance compact plating unit that is especially designed for coating all
1 Compact Electro Plating Machine - CEPM-2A USER MANUAL PRODUCT INTRODUCTION: This compact electroplating machine is a high performance compact plating unit that is especially designed for coating all
REVERSE OSMOSIS SYSTEM USER'S MANUAL. Excellent Product, Excellent Water! My PurePro X6
 REVERSE OSMOSIS SYSTEM USER'S MANUAL UV Plus! Excellent Product, Excellent Water! My PurePro X6 PUREPRO X6 Thank you very much for selecting Pure-Pro Water Corp. In order to bring the best use of your
REVERSE OSMOSIS SYSTEM USER'S MANUAL UV Plus! Excellent Product, Excellent Water! My PurePro X6 PUREPRO X6 Thank you very much for selecting Pure-Pro Water Corp. In order to bring the best use of your
From Turbid to Clear: How Flocculation Cleans Up Drinking Water
 From Turbid to Clear: How Flocculation Cleans Up Drinking Water https://www.sciencebuddies.org/science-fair-projects/project-ideas/enveng_p039/environmental-engineering/clean-drinking-water-flocculation
From Turbid to Clear: How Flocculation Cleans Up Drinking Water https://www.sciencebuddies.org/science-fair-projects/project-ideas/enveng_p039/environmental-engineering/clean-drinking-water-flocculation
Module 2, Add on Lesson Conductivity Sensor. Teacher. 90 minutes
 Module 2, Add on Lesson Conductivity Sensor 90 minutes Teacher Purpose of this lesson Investigate the variation of resistance with conductivity Construct a sensor to measure the conductivity of water Graph
Module 2, Add on Lesson Conductivity Sensor 90 minutes Teacher Purpose of this lesson Investigate the variation of resistance with conductivity Construct a sensor to measure the conductivity of water Graph
Let s Move It! Gel Electrophoresis Using Food Dye Student Guide
 Let s Move It! Gel Electrophoresis Using Food Dye Student Guide Purpose This lab explores the principle of electrophoresis, an important technique used in biochemistry and molecular biology. You will:
Let s Move It! Gel Electrophoresis Using Food Dye Student Guide Purpose This lab explores the principle of electrophoresis, an important technique used in biochemistry and molecular biology. You will:
Nickel Electroplating
 Nickel Electroplating In a galvanic or voltaic electrochemical cell, the spontaneous reaction occurs and electrons flow from the anode (oxidation) to the cathode (reduction). In an electrolytic cell, a
Nickel Electroplating In a galvanic or voltaic electrochemical cell, the spontaneous reaction occurs and electrons flow from the anode (oxidation) to the cathode (reduction). In an electrolytic cell, a
EPK- EasyTest Patch Test Kit Manual
 EPK- EasyTest Patch Test Kit Manual L-2730 The Patch Test Kit from Schroeder provides the necessary tools to determine levels of both water and solid particulate contamination present in a particular fluid
EPK- EasyTest Patch Test Kit Manual L-2730 The Patch Test Kit from Schroeder provides the necessary tools to determine levels of both water and solid particulate contamination present in a particular fluid
Hydrocar Earth Sci. Lab
 Hydrocar Earth Sci. Lab Name: Class: Date: Earth Sciences High School 6 hours Objective Build and modify a hydrogen fuel cell car to explore the concepts of renewable energy and human effects on global
Hydrocar Earth Sci. Lab Name: Class: Date: Earth Sciences High School 6 hours Objective Build and modify a hydrogen fuel cell car to explore the concepts of renewable energy and human effects on global
I. INTRODUCTION. II. OBJECTIVE OF THE EXPERIMENT. III. THEORY
 I. INTRODUCTION. Chemical pollution is a serious problem that demands the attention of the scientific community in the early 21 st century. The consequences of pollution are numerous: heating of the atmosphere
I. INTRODUCTION. Chemical pollution is a serious problem that demands the attention of the scientific community in the early 21 st century. The consequences of pollution are numerous: heating of the atmosphere
Name Honors Chemistry / /
 Name Honors Chemistry / / SOL Questions Chapter 1 Each of the following questions below appeared on an SOL Chemistry Exam. For each of the following bubble in the correct answer on your scantron. 1. The
Name Honors Chemistry / / SOL Questions Chapter 1 Each of the following questions below appeared on an SOL Chemistry Exam. For each of the following bubble in the correct answer on your scantron. 1. The
PLANS for the Chesapeake Bay A Teacher s Guide
 1 Nutrient Enrichment of Phytoplankton in the Chesapeake Estuary A MWEE for 9 th Grade Environmental Science Classes Fifth Day of PLANS Teacher led classroom activity supported by PLANS staff via online
1 Nutrient Enrichment of Phytoplankton in the Chesapeake Estuary A MWEE for 9 th Grade Environmental Science Classes Fifth Day of PLANS Teacher led classroom activity supported by PLANS staff via online
LCD INSTALLATION MANUAL V.1
 X2 LCD INSTALLATION MANUAL V.1 www.odesutvs.com INDEX Steps 1-5: Uncrating ------------------------------------------------------------------------- Pgs. 1 /2 Steps 6-9: Dash Removal ------------------------------------------------------------------
X2 LCD INSTALLATION MANUAL V.1 www.odesutvs.com INDEX Steps 1-5: Uncrating ------------------------------------------------------------------------- Pgs. 1 /2 Steps 6-9: Dash Removal ------------------------------------------------------------------
Installing Equipment for the Hydric Soil Technical Standard
 Installing Equipment for the Hydric Soil Technical Standard Mike Vepraskas, NC State Univ. April 2005 Procedure: 1. Sites: Select at least two spots to monitor. One should be within a hydric soil, or at
Installing Equipment for the Hydric Soil Technical Standard Mike Vepraskas, NC State Univ. April 2005 Procedure: 1. Sites: Select at least two spots to monitor. One should be within a hydric soil, or at
Activity 5.1.4: Gram Staining
 Activity 5.1.4: Gram Staining Introduction In the last activity, you isolated the bacteria that was responsible for Anna s illness and performed a gross examination of the resultant colonies. While gross
Activity 5.1.4: Gram Staining Introduction In the last activity, you isolated the bacteria that was responsible for Anna s illness and performed a gross examination of the resultant colonies. While gross
Water-Water Titration for the MicroCal ITC200
 Water-Water Titration for the MicroCal ITC200 Goal: Water-Water titrations are a good way to check for possible problems, especially the cleanliness of the ITC and your skills in cell filling. If a water-water
Water-Water Titration for the MicroCal ITC200 Goal: Water-Water titrations are a good way to check for possible problems, especially the cleanliness of the ITC and your skills in cell filling. If a water-water
AQUATERR I N S T R U M E N T S. M, T & EC -300 User s Manual
 AQUATERR I N S T R U M E N T S M, T & EC -300 User s Manual Aquaterr Digital Soil Moisture, Temperature and Salinity Meters AQUATERR INSTRUMENTS Leader in Portable Soil Measuring Technology 2 It is important
AQUATERR I N S T R U M E N T S M, T & EC -300 User s Manual Aquaterr Digital Soil Moisture, Temperature and Salinity Meters AQUATERR INSTRUMENTS Leader in Portable Soil Measuring Technology 2 It is important
Chlorine Dioxide Amperometric 4-20mA Sensors
 Chlorine Dioxide Amperometric 4-20mA Sensors Product Instructions Section 1.0 Theory of Operation 1.0 Chlorine Dioxide Introduction Chlorine dioxide is considered one of the most powerful biocides available
Chlorine Dioxide Amperometric 4-20mA Sensors Product Instructions Section 1.0 Theory of Operation 1.0 Chlorine Dioxide Introduction Chlorine dioxide is considered one of the most powerful biocides available
Dissolved Oxygen Probe (Order Code: DO-BTA)
 Dissolved Oxygen Probe (Order Code: DO-BTA) The Vernier Dissolved Oxygen Probe uses a Clark-type polarographic electrode to measure the concentration of dissolved oxygen in water. You can use this sensor
Dissolved Oxygen Probe (Order Code: DO-BTA) The Vernier Dissolved Oxygen Probe uses a Clark-type polarographic electrode to measure the concentration of dissolved oxygen in water. You can use this sensor
Wet Cells, Dry Cells, Fuel Cells
 page 2 page 3 Teacher's Notes Wet Cells, Dry Cells, Fuel Cells How the various electrochemical cells work Grades: 7-12 Duration: 33 mins Program Summary This video is an introductory program outlining
page 2 page 3 Teacher's Notes Wet Cells, Dry Cells, Fuel Cells How the various electrochemical cells work Grades: 7-12 Duration: 33 mins Program Summary This video is an introductory program outlining
DETERMINATION of the EMPIRICAL FORMULA
 DETERMINATION of the EMPIRICAL FORMULA One of the fundamental statements of the atomic theory is that elements combine in simple whole number ratios. This observation gives support to the theory of atoms,
DETERMINATION of the EMPIRICAL FORMULA One of the fundamental statements of the atomic theory is that elements combine in simple whole number ratios. This observation gives support to the theory of atoms,
Module 2, Add on Lesson Turbidity Sensor. Teacher. 90 minutes
 Module 2, Add on Lesson Turbidity Sensor Teacher 90 minutes Purpose Construct a sensor to measure the turbidity of water Graph data and reason about curves and linear relationships Calibrate the turbidity
Module 2, Add on Lesson Turbidity Sensor Teacher 90 minutes Purpose Construct a sensor to measure the turbidity of water Graph data and reason about curves and linear relationships Calibrate the turbidity
ME 280 Lab #1. Lüders Strain Evaluation of Stress Concentration and Electrochemical Embrittlement of Spring Steel
 ME 280 Lab #1 Lüders Strain Evaluation of Stress Concentration and Electrochemical Embrittlement of Spring Steel Overview: In one portion of this lab, you will observe the propagation of a Lüder s band
ME 280 Lab #1 Lüders Strain Evaluation of Stress Concentration and Electrochemical Embrittlement of Spring Steel Overview: In one portion of this lab, you will observe the propagation of a Lüder s band
F U C H S L U B R I C A N T S C O.
 F U C H S L U B R I C A N T S C O. SENSOR INFORMATION, CARE AND WARRANTY RM CONCENTRATION / BRIX SENSOR RANGE: 0-35 BRIX GENERAL RM CONCENTRATION SENSOR CARE AND MAINTENANCE RECOMMENDATIONS BY MANUFACTURER
F U C H S L U B R I C A N T S C O. SENSOR INFORMATION, CARE AND WARRANTY RM CONCENTRATION / BRIX SENSOR RANGE: 0-35 BRIX GENERAL RM CONCENTRATION SENSOR CARE AND MAINTENANCE RECOMMENDATIONS BY MANUFACTURER
Experiment 3: Determination of an Empirical Formula
 Background Information The composition of a compound is defined by its chemical formula, which gives the number ratio of the different elements in the compound. For example, water has a fixed composition
Background Information The composition of a compound is defined by its chemical formula, which gives the number ratio of the different elements in the compound. For example, water has a fixed composition
Evaluation copy. Energy Content of Fuels. computer OBJECTIVES MATERIALS
 Energy Content of Fuels Computer 9 Energy content is an important property of fuels. This property helps scientists and engineers determine the usefulness of a fuel. Energy content is the amount of heat
Energy Content of Fuels Computer 9 Energy content is an important property of fuels. This property helps scientists and engineers determine the usefulness of a fuel. Energy content is the amount of heat
Introduction to Green Chemistry: Solar Energy Devices Made with Natural Dyes
 Introduction Introduction to Green Chemistry: Solar Energy Devices Made with Natural Dyes In the course of your lifetime, you have observed significant dependence of the economy on hydrocarbon-based fuels.
Introduction Introduction to Green Chemistry: Solar Energy Devices Made with Natural Dyes In the course of your lifetime, you have observed significant dependence of the economy on hydrocarbon-based fuels.
Module 2, Add on Lesson Conductivity Sensor. Student. 90 minutes
 Module 2, Add on Lesson Conductivity Sensor 90 minutes Student Purpose of this lesson Investigate the variation of resistance with conductivity Construct a sensor to measure the conductivity of water Graph
Module 2, Add on Lesson Conductivity Sensor 90 minutes Student Purpose of this lesson Investigate the variation of resistance with conductivity Construct a sensor to measure the conductivity of water Graph
SUN Angle Science Kit Copyright 2011
 SUN Angle Science Kit Copyright 2011 Kidwind Project 800 Transfer Rd Suite 30B Saint Paul, MN 55114 www.kidwind.org This work may not be reproduced by mechanical or electronic means without written permission
SUN Angle Science Kit Copyright 2011 Kidwind Project 800 Transfer Rd Suite 30B Saint Paul, MN 55114 www.kidwind.org This work may not be reproduced by mechanical or electronic means without written permission
Semiconductors. Lab. FCJJ 16 - Solar Hydrogen Science Kit. Next Generation Science Standards. Initial Prep Time. Lesson Time. Assembly Requirements
 Next Generation Science Standards NGSS Science and Engineering Practices: Asking questions and defining problems Developing and using models Planning and carrying out investigations Analyzing and interpreting
Next Generation Science Standards NGSS Science and Engineering Practices: Asking questions and defining problems Developing and using models Planning and carrying out investigations Analyzing and interpreting
Voltaic Cells. An Energizing Experience. Using the energy of a spontaneous redox reaction to do work. aka Galvanic Cells.
 Voltaic Cells aka Galvanic Cells Using the energy of a spontaneous redox reaction to do work. Chapter 20 An Energizing Experience 1 A piece of copper is dropped into an aqueous solution of zinc nitrate,
Voltaic Cells aka Galvanic Cells Using the energy of a spontaneous redox reaction to do work. Chapter 20 An Energizing Experience 1 A piece of copper is dropped into an aqueous solution of zinc nitrate,
Fabrication of Oxygenation Microfluidic Devices for Cell Cultures
 1 Fabrication of Oxygenation Microfluidic Devices for Cell Cultures Shauharda Khadka skhadka@ramapo.edu Department of Engineering Physics, Ramapo College of New Jersey Gerardo Mauleon mauleon2@uic.edu
1 Fabrication of Oxygenation Microfluidic Devices for Cell Cultures Shauharda Khadka skhadka@ramapo.edu Department of Engineering Physics, Ramapo College of New Jersey Gerardo Mauleon mauleon2@uic.edu
Cu (s) Cu 2+ (aq) Cu(OH) 2 (s) CuO (s) Cu 2+ (aq) Cu (s)
 Cycle of Copper Reactions Lab Exercise The following is a protocol for the multi-step transformation of copper metal based upon the following chemical transformations: Cu (s) Cu 2+ (aq) Cu(OH) 2 (s) CuO
Cycle of Copper Reactions Lab Exercise The following is a protocol for the multi-step transformation of copper metal based upon the following chemical transformations: Cu (s) Cu 2+ (aq) Cu(OH) 2 (s) CuO
Operating Manual for Digital ph-meter GPHR 1400
 Operating Manual for Digital ph-meter GPHR 1400 Specification Measuring range: Resolution: ph-electrode: conductivity Input resistance: Accuracy (unit only): Working temperature: Nominal temperature: Power
Operating Manual for Digital ph-meter GPHR 1400 Specification Measuring range: Resolution: ph-electrode: conductivity Input resistance: Accuracy (unit only): Working temperature: Nominal temperature: Power
CH 112 Special Assignment #6 The Case of the Phony Fruit
 CH 112 Special Assignment #6 The Case of the Phony Fruit PRE-LAB ASSIGNMENT: Make sure that you read this handout and bring the essentials to lab with you. You may find it helpful to read parts of Chapter
CH 112 Special Assignment #6 The Case of the Phony Fruit PRE-LAB ASSIGNMENT: Make sure that you read this handout and bring the essentials to lab with you. You may find it helpful to read parts of Chapter
INSTRUCTION MANUAL WARNING: BEFORE USING THE MACHINE READ THE INSTRUCTION MANUAL CAREFULLY!
 INSTRUCTION MANUAL WARNING: BEFORE USING THE MACHINE READ THE INSTRUCTION MANUAL CAREFULLY! SECTION 1 : PRODUCT AND COMPANY IDENTIFICATION PRODUCT NAME : SYNONYMS : PRODUCT CODES : Weldability ARC140i
INSTRUCTION MANUAL WARNING: BEFORE USING THE MACHINE READ THE INSTRUCTION MANUAL CAREFULLY! SECTION 1 : PRODUCT AND COMPANY IDENTIFICATION PRODUCT NAME : SYNONYMS : PRODUCT CODES : Weldability ARC140i
RAINBOW GELS: AN INTRODUCTION TO ELECTROPHORESIS. STANDARDS 3.1.7, , Westminster College 3.3.7, , 3.3.
 RAINBOW GELS: AN INTRODUCTION TO ELECTROPHORESIS STANDARDS 3.1.7, 3.1.10, 3.1.12 Westminster College 3.3.7, 3.3.10, 3.3.12 INTRODUCTION This laboratory will demonstrate the basics of electrophoresis and
RAINBOW GELS: AN INTRODUCTION TO ELECTROPHORESIS STANDARDS 3.1.7, 3.1.10, 3.1.12 Westminster College 3.3.7, 3.3.10, 3.3.12 INTRODUCTION This laboratory will demonstrate the basics of electrophoresis and
SUNSTONE WELDERS AC WELDER USER MANUAL
 SUNSTONE WELDERS AC WELDER USER MANUAL 2 Sunstone Welders - AC Welder User Manual TABLE OF CONTENTS Forward... p.4 Introduction... p.5 Welder Setup... p.5 Welder Interface... p.6 Welding Attachments...
SUNSTONE WELDERS AC WELDER USER MANUAL 2 Sunstone Welders - AC Welder User Manual TABLE OF CONTENTS Forward... p.4 Introduction... p.5 Welder Setup... p.5 Welder Interface... p.6 Welding Attachments...
Electroelution. Teachers Handbook. (Cat. # BE 602) think proteins! think G-Biosciences
 PR097 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Electroelution Teachers Handbook (Cat. # BE 602) think proteins! think G-Biosciences
PR097 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Electroelution Teachers Handbook (Cat. # BE 602) think proteins! think G-Biosciences
Portable Power. Find Out
 TOPIC 5 Portable Power Figure 4.29 Voltaic piles like these, invented in 1800, were the first electric batteries. How many battery-powered devices are in your household? How many batteries do you buy each
TOPIC 5 Portable Power Figure 4.29 Voltaic piles like these, invented in 1800, were the first electric batteries. How many battery-powered devices are in your household? How many batteries do you buy each
Hydro-wind Education Kit ASSEMBLY GUIDE
 Hydro-wind Education Kit ASSEMBLY GUIDE Battery operation instructions: Model No.: FCJJ-56 Warning To avoid the risk of property damage, serious injury or death: This kit should only be used by persons
Hydro-wind Education Kit ASSEMBLY GUIDE Battery operation instructions: Model No.: FCJJ-56 Warning To avoid the risk of property damage, serious injury or death: This kit should only be used by persons
Platinum Nanostructures by Template Wetting Nanofabrication and Their Use in a Miniature Fuel Cell Membrane Electrode Assembly
 Platinum Nanostructures by Template Wetting Nanofabrication and Their Use in a Miniature Fuel Cell Membrane Electrode Assembly Eric Broaddus, Jared Fernandez, and Scott A. Gold Institute for Micromanufacturing,
Platinum Nanostructures by Template Wetting Nanofabrication and Their Use in a Miniature Fuel Cell Membrane Electrode Assembly Eric Broaddus, Jared Fernandez, and Scott A. Gold Institute for Micromanufacturing,
OWNER S MANUAL. Model 845. RFV Dye Marking System /15
 OWNER S MANUAL Model 845 RFV Dye Marking System 845-15 5/15 HARVEST TEC 845 TABLE OF CONTENTS PAGE INTRODUCTION & TOOLS NEEDED 3 INSTALLATION OF DYE SPAYER TANK ASSEMBLY 4 DYE SPRAYER SYSTEM WIRING DIAGRAMS
OWNER S MANUAL Model 845 RFV Dye Marking System 845-15 5/15 HARVEST TEC 845 TABLE OF CONTENTS PAGE INTRODUCTION & TOOLS NEEDED 3 INSTALLATION OF DYE SPAYER TANK ASSEMBLY 4 DYE SPRAYER SYSTEM WIRING DIAGRAMS
Western Immunoblotting Standard Protocol:
 Gel Preparation: Clean plates, combs and casting frames and bottom strips with water and alcohol and allow to dry Place a short plate on top of the spacer plate Slide the two plates into the casting frame,
Gel Preparation: Clean plates, combs and casting frames and bottom strips with water and alcohol and allow to dry Place a short plate on top of the spacer plate Slide the two plates into the casting frame,
Family Secrets. Part 4 Testing for the HD Gene
 Family Secrets Part 4 Testing for the HD Gene Narrator: Jenny, Jeremy, and their father have decided to undergo genetic testing for Huntington s disease. Today, our lab groups will act as gene testing
Family Secrets Part 4 Testing for the HD Gene Narrator: Jenny, Jeremy, and their father have decided to undergo genetic testing for Huntington s disease. Today, our lab groups will act as gene testing
iworx Sample Lab Experiment AN-9: Membrane Potentials
 Experiment AN-9: Membrane Potentials Background The aim of this laboratory exercise is to record resting potentials across the membranes of fast extensor muscle fibers in the tail of crayfish. Microelectrodes
Experiment AN-9: Membrane Potentials Background The aim of this laboratory exercise is to record resting potentials across the membranes of fast extensor muscle fibers in the tail of crayfish. Microelectrodes
Making Milk Curdle with Pineapple Enzymes
 Making Milk Curdle with Pineapple Enzymes https://www.sciencebuddies.org/science-fair-projects/project-ideas/biochem_p032/biotechnology-techniques/enzymes-milk-curdle (http://www.sciencebuddies.org/sciencefair-projects/project-ideas/biochem_p032/biotechnology-techniques/enzymes-milk-curdle)
Making Milk Curdle with Pineapple Enzymes https://www.sciencebuddies.org/science-fair-projects/project-ideas/biochem_p032/biotechnology-techniques/enzymes-milk-curdle (http://www.sciencebuddies.org/sciencefair-projects/project-ideas/biochem_p032/biotechnology-techniques/enzymes-milk-curdle)
Aluminazing One of the most common minerals on the planet is also one of the most versatile. GRADES
 Aluminazing One of the most common minerals on the planet is also one of the most versatile. GRADES 9-12 Time Required: 2 class periods (90 minutes) without extension Gather materials (see activity pages).
Aluminazing One of the most common minerals on the planet is also one of the most versatile. GRADES 9-12 Time Required: 2 class periods (90 minutes) without extension Gather materials (see activity pages).
Experiment 13: Determination of Molecular Weight by Freezing Point Depression
 1 Experiment 13: Determination of Molecular Weight by Freezing Point Depression Objective: In this experiment, you will determine the molecular weight of a compound by measuring the freezing point of a
1 Experiment 13: Determination of Molecular Weight by Freezing Point Depression Objective: In this experiment, you will determine the molecular weight of a compound by measuring the freezing point of a
Inquiry Question Solid Copper, Silver, Gold! Or is it? Exploring electroplating!
 Inquiry Question Solid Copper, Silver, Gold! Or is it? Exploring electroplating! Name: Date: As much as solid precious metals are great to have, not many of us can afford them. Many items we use such as
Inquiry Question Solid Copper, Silver, Gold! Or is it? Exploring electroplating! Name: Date: As much as solid precious metals are great to have, not many of us can afford them. Many items we use such as
Significance of Water Observation Lab
 Significance of Water Observation Lab It has been said that the chemistry of life is water chemistry. Because of its chemical properties, water is the medium in which most of life s chemical reactions
Significance of Water Observation Lab It has been said that the chemistry of life is water chemistry. Because of its chemical properties, water is the medium in which most of life s chemical reactions
Final Year Progress Report
 Final Year Progress Report Student: Stephen Mulryan Student ID: 06583725 Discipline: Electronic & computer Engineering Supervisor: Dr. Maeve Duffy Co-Supervisor: Professor Ger Hurley Project Title: Energy
Final Year Progress Report Student: Stephen Mulryan Student ID: 06583725 Discipline: Electronic & computer Engineering Supervisor: Dr. Maeve Duffy Co-Supervisor: Professor Ger Hurley Project Title: Energy
USER MANUAL ON PROFESSIONAL ELECTROCHEMICAL STATION EPS-30A
 1 USER MANUAL ON PROFESSIONAL ELECTROCHEMICAL STATION EPS-30A PRODUCT INTRODUCTION: This electrochemical station is a professional, high performance plating/forming equipment designed to plate, form, strip,
1 USER MANUAL ON PROFESSIONAL ELECTROCHEMICAL STATION EPS-30A PRODUCT INTRODUCTION: This electrochemical station is a professional, high performance plating/forming equipment designed to plate, form, strip,
DNA RESTRICTION ANALYSIS
 DNA RESTRICTION ANALYSIS In this experiment, DNA from the bacteriophage Lambda (48,502 base pairs in length) is cut with a variety of restriction enzymes and the resulting fragments are separated using
DNA RESTRICTION ANALYSIS In this experiment, DNA from the bacteriophage Lambda (48,502 base pairs in length) is cut with a variety of restriction enzymes and the resulting fragments are separated using
Let s Move It! Gel Electrophoresis Using Food Dye Student Guide
 Let s Move It! Gel Electrophoresis Using Food Dye Student Guide Purpose This lab explores the principle of electrophoresis, an important technique used in biochemistry and molecular biology. You will:
Let s Move It! Gel Electrophoresis Using Food Dye Student Guide Purpose This lab explores the principle of electrophoresis, an important technique used in biochemistry and molecular biology. You will:
15A Carbon Dioxide and Living Things
 Carbon Dioxide and Living Things Investigation 15A 15A Carbon Dioxide and Living Things How do living things exchange carbon dioxide? The carbon cycle is a series of pathways through which carbon atoms
Carbon Dioxide and Living Things Investigation 15A 15A Carbon Dioxide and Living Things How do living things exchange carbon dioxide? The carbon cycle is a series of pathways through which carbon atoms
Activity of metals SCIENTIFIC. Demonstration and Inquiry. Introduction. Concepts. Background. Inquiry Approach. Demonstration Questions
 Activity of Metals Demonstration and Inquiry SCIENTIFIC Introduction Chemical reactions are not formulas on a piece of paper they are dynamic and exciting events! The demonstration of aluminum with copper(ii)
Activity of Metals Demonstration and Inquiry SCIENTIFIC Introduction Chemical reactions are not formulas on a piece of paper they are dynamic and exciting events! The demonstration of aluminum with copper(ii)
Biochemical Oxygen Demand
 Biochemical Oxygen Demand Computer 20 Oxygen available to aquatic organisms is found in the form of dissolved oxygen. Oxygen gas is dissolved in a stream through aeration, diffusion from the atmosphere,
Biochemical Oxygen Demand Computer 20 Oxygen available to aquatic organisms is found in the form of dissolved oxygen. Oxygen gas is dissolved in a stream through aeration, diffusion from the atmosphere,
Thermocouple & Fine Wire Welder Model No. L60+
 Thermocouple & Fine Wire Welder Model No. L60+ Web e-mail Telephone www.labfacility.com sales@labfacility.com +44(0)1243 871280 Issue: 050004.OM.G Web e-mail Telephone www.labfacility.com sales@labfacility.com
Thermocouple & Fine Wire Welder Model No. L60+ Web e-mail Telephone www.labfacility.com sales@labfacility.com +44(0)1243 871280 Issue: 050004.OM.G Web e-mail Telephone www.labfacility.com sales@labfacility.com
Fuel Cells Futuristic Battery
 Understanding Solar Energy Teacher Page Fuel Cells Futuristic Battery Student Objectives The student: will be able to explain the chemical reaction in the electrolysis procedure will be able to explain
Understanding Solar Energy Teacher Page Fuel Cells Futuristic Battery Student Objectives The student: will be able to explain the chemical reaction in the electrolysis procedure will be able to explain
Types of Magnets. force: a push or a pull
 Imagine that you have a superpower. Your power allows you to move things without touching them. You can even move things located on the other side of a wall! Your power to move things without touching
Imagine that you have a superpower. Your power allows you to move things without touching them. You can even move things located on the other side of a wall! Your power to move things without touching
Significance of Water Observation Lab
 Significance of Water Observation Lab It has been said that the chemistry of life is water chemistry. Because of its chemical properties, water is the medium in which most of life s chemical reactions
Significance of Water Observation Lab It has been said that the chemistry of life is water chemistry. Because of its chemical properties, water is the medium in which most of life s chemical reactions
KOBRA CELL Art. No. K01
 R-Biopharm Rhône Ltd KOBRA CELL Art. No. K01 Instruction Manual R-Biopharm for the safety of your analysis. R-Biopharm Rhone Ltd 2 Contents 1. Introduction to the KOBRA CELL... 4 1.1 Intended Use... 4
R-Biopharm Rhône Ltd KOBRA CELL Art. No. K01 Instruction Manual R-Biopharm for the safety of your analysis. R-Biopharm Rhone Ltd 2 Contents 1. Introduction to the KOBRA CELL... 4 1.1 Intended Use... 4
Chlorine Dioxide Amperometric 4-20mA Sensors
 Chlorine Dioxide Amperometric 4-20mA Sensors SECTION 1.0 THEORY OF OPERATION 1.0 CHLORINE DIOXIDE INTRODUCTION. Chlorine dioxide is considered one of the most powerful biocides available today. It is so
Chlorine Dioxide Amperometric 4-20mA Sensors SECTION 1.0 THEORY OF OPERATION 1.0 CHLORINE DIOXIDE INTRODUCTION. Chlorine dioxide is considered one of the most powerful biocides available today. It is so
WATER LEVEL INDICATOR. Model CPR. Roctest Limited, All rights reserved.
 INSTRUCTION MANUAL WATER LEVEL INDICATOR Model Roctest Limited, 2006. All rights reserved. This product should be installed and operated only by qualified personnel. Its misuse is potentially dangerous.
INSTRUCTION MANUAL WATER LEVEL INDICATOR Model Roctest Limited, 2006. All rights reserved. This product should be installed and operated only by qualified personnel. Its misuse is potentially dangerous.
Human DNA Alu Amplification by Polymerase Chain Reaction (PCR)* Laboratory Procedure
 Human DNA Alu Amplification by Polymerase Chain Reaction (PCR)* Laboratory Procedure *Polymerase Chain Reaction is covered by patents owned by Hoffmann-La Roche, Inc. This experiment was adapted from Laboratory
Human DNA Alu Amplification by Polymerase Chain Reaction (PCR)* Laboratory Procedure *Polymerase Chain Reaction is covered by patents owned by Hoffmann-La Roche, Inc. This experiment was adapted from Laboratory
VAN LONDON COMPANY COMBINATION ph ELECTRODE INSTRUCTION MANUAL
 VAN LONDON COMPANY COMBINATION ph ELECTRODE INSTRUCTION MANUAL GENERAL INFORMATION Van London combination electrodes offer the convenience of having the reference and measuring electrodes combined in a
VAN LONDON COMPANY COMBINATION ph ELECTRODE INSTRUCTION MANUAL GENERAL INFORMATION Van London combination electrodes offer the convenience of having the reference and measuring electrodes combined in a
Electrophoresis 101 MiniLab
 Electrophoresis 101 MiniLab Student s Guide Cat# M3001 Version 020718 Table of Contents Laboratory Safety 2 Objectives and Background 3 Instructions 4 Results and Analysis 7 Appendix A - TBE Concentrate
Electrophoresis 101 MiniLab Student s Guide Cat# M3001 Version 020718 Table of Contents Laboratory Safety 2 Objectives and Background 3 Instructions 4 Results and Analysis 7 Appendix A - TBE Concentrate
Single Pulse Resistance Welder Instruction Pamphlet CD125SP High Accuracy CD Welder
 EN G IN EERI N G Single Pulse Resistance Welder Instruction Pamphlet CD125SP High Accuracy CD Welder Fundamentals of Capacitive Discharge Resistance Welding deliver repeatable welds even during line voltage
EN G IN EERI N G Single Pulse Resistance Welder Instruction Pamphlet CD125SP High Accuracy CD Welder Fundamentals of Capacitive Discharge Resistance Welding deliver repeatable welds even during line voltage
ANG 111 Summer 2009 Laboratory 0: Basic Laboratory Procedures. To provide a foundation in basic laboratory techniques and safety issues
 ANG 111 Summer 2009 Laboratory 0: Basic Laboratory Procedures Objective: To provide a foundation in basic laboratory techniques and safety issues Overview: Students enter this laboratory with a wide range
ANG 111 Summer 2009 Laboratory 0: Basic Laboratory Procedures Objective: To provide a foundation in basic laboratory techniques and safety issues Overview: Students enter this laboratory with a wide range
