COMPANY OVERVIEW. Global Pharmatech started as a specialized Sterile Liquid Injection manufacturing company in 1996.
|
|
|
- Georgina Davidson
- 6 years ago
- Views:
Transcription
1 OVERVIEW
2 COMPANY OVERVIEW Global Pharmatech started as a specialized Sterile Liquid Injection manufacturing company in Manufacturer of a wide range of products across various therapeutic segments. Total plant area of 5 acres is located in Hosur, Tamil Nadu (outskirts of Bangalore).
3 VISION To be recognized as a world class manufacturer of pharmaceutical formulations and create value for our business partners and stakeholders through continuous improvement and innovation to improve Quality of Life.
4 QUALITY POLICY We are committed to comply with customer requirements and with all applicable regulations, and to ensuring product quality, safety and efficacy. To achieve this goal, we shall maintain and continually improve the effectiveness of our Quality Management System.
5 APPROVALS WHO GMP: Valid up to NAFDAC: Approved in Sept 2014 DPM: GMP from Cote d Ivoire EU GMP compliant: Under Renewal ISO 9001:2008: Valid up to ISO 13485:2003: Valid up to Periodically audited by GSK, PMS Canada and our other customers.
6 IMPORTANT AUDITS - TRIGGERED Health Canada Audit 17 to 23 Sept 2015 EU GMP - Hungary (PICS) 11 to 15 Jan 2016 MOH- Malaysia (PICS) Q MCC South Africa SMF accepted Ethiopian Food, Medicine & Healthcare Administration & Control Authority (EFMHACA) awaiting audit schedule TGA / TFDA / NDA will be triggered soon
7 SITE LAYOUT
8 ORGANIZATION STRUCTURE Mr. T. Thomas Chairman Dr. Bhaskar Krishna Managing Director Operation Mr. Arul Prasadh Quality operation Mr. Chezhian Supply Chain Mr. Viswanathan R&D Business Development Mr. Deepak Vagale Finance & HR Mr. Hegde Production Mr. Chandran Engineering FR&D Mr. Raja G AR&D Mr. Karthik N Regulatory Mrs. Sindhu Hari Instrumentation Mrs. Sumathi Microbiology Mr. Shyam Quality Assurance Mr. Sethurajan G
9 MANAGEMENT TEAM Name Designation Qualification Responsibility Experience Dr. BHASKAR KRISHNA Managing Director PhD, MBA General Management 19 Yrs. SHIVANANDA HEGDE Chief Finance Controller FCA, ACS Finance, Accounts & HR 16 Yrs. S. ARUL PRASADH DGM Operation B.Pharm., MS Head of Production & Engineering 17 Yrs. S. CHEZHIAN DGM Q.O. M.Sc. Quality Operation 20 Yrs. A.VISWANATHAN DGM-Supply Chain B.Sc. MBA Supply Chain Management 25 Yrs. DEEPAK VAGALE SINDHU HARI DGM Business Development DGM Regulatory B.Sc., D. Pharm Business Development 20 Yrs. B.Sc., Diploma in Regulatory Regulatory Affairs 20 Yrs G. RAJA Sr. Manager FR&D B.Pharm., MS Formulation Department 14 Yrs. N. KARTHIK Sr. Manager AR&D M.Sc Analytical Development 9 Yrs
10 EMPLOYEE STRENGTH Department Staff Workmen Total Quality Assurance Quality Control Production Stores and Distribution Technical, Engineering, and other support services, FR&D/AR&D, Regulatory Affairs TOTAL EMPLOYEES Total
11 RAW MATERIAL WAREHOUSE QUARANTINE (AMBIENT) 70 sq.mt (with multi level storage racks) QUARANTINE (BELOW 25 O C) 30 sq.mt (with multi level storage racks) APPROVED MATL (BELOW 25 O C) 130 sq.mt (with multi level storage racks)
12 SAMPLING AND DISPENSING SAMPLING DISPENSING Dedicated sampling booths and dispensing booths with RLAF under Class D area
13 AMPOULE LINE Integrated Washing, Sterilization, and Filling line Terminal Sterilizer in place Aseptically filtered with 0.2 micron filters Fill volume: 1mL to 10mL Batch size: 100 liters to 400 liters Speed: 250 Ampoules/minute Annual Capacity: 72 million Ampoules
14 VIAL LINE Integrated Washing, Sterilization and Filling line Aseptically filtered with 0.2 micron filters Batch size: 100 liters to 400 liters Fill volume: 2mL to 100mL Speed:150 vials / minute Annual Capacity: 36 million vials
15 INSPECTION 100% visual inspection of all filled containers. Visual inspection conducted manually and with semi automatic inspection machines by trained and qualified personnel by validated method. Visual inspection designed to remove all defective containers.
16 PACKING CAPACITY Ampoules: 250,000 per shift Vials: 100,000 per shift BLISTER MACHINES Form Pack: 400 Ampoules / minute Rapid Pack: 400 Ampoules / minute PACKING Blisters and Vials manually packed in cartons QUALITY In process check carried out to eliminate packing errors
17 FINISHED GOODS WAREHOUSE AMBIENT TEMPERATURE : Storage area of 125 sq.mt TEMPERATURE BELOW 25 O C : Storage area of 150 sq.mt
18 Air Handling Unit Total 22 AHUs to provide clean air as per criticality of the process Separate Air Handling Units with HEPA filters are installed at each processing room. Aseptic area temperature is maintained between 18 C to 25 C and Relative Humidity (RH) is maintained between 40% to 60%. Temperature of storage areas is maintained based on material or product requirements. The entire plant is maintained at a positive differential pressure with respect to the external atmosphere. A minimum pressure differential of 1.6mm/16 Pascal's is maintained between two different classified areas and positive pressure is maintained from critical to non critical rooms of the same classification All AHUs are designed to have sufficient air-changes with 10% fresh air intake during each cycle. Filling room Air Changes is NLT 70 ACPH
19 Classification of Manufacturing Areas Grade-A (Filling) is provided by laminar airflow stations, HEPA of % efficiency and is surrounded by Grade-B Grade-B ( Back ground of Filling): 20 micron, 10 micron, 5 micron, 0.3 micron plenum HEPA of % and 0.3 micron with terminal HEPA of % efficiency is series Grade-C (Compounding): 20 micron, 10 micron, 5 micron, 0.3 micron plenum HEPA of % and 0.3 micron with terminal HEPA of % efficiency is series Grade-C (Washing): 20 micron, 10 micron, 5 micron with plenum HEPA of % efficiency
20 Utilities: Water System Purified Water RO/EDI system Capacity: 5000 liters/hr Water for Injection (WFI) Multi-column evaporator with continuous circulation loop with WFI at > 80 C. Capacity: 1500 liters/hr Pure Steam Capacity: 300 kilograms/hr
21 Raw Steam 2 MT briquette boiler 800 KG standby diesel boiler Utilities: Other Electricity Sanctioned load of 950 KVA supplied by State Electricity Board Full Diesel Generator backup (500 KVA, 380 KVA, 250 KVA) Cooling 250 TR Vapour Absorption Machine Backup Screw Chillers (150 TR and 45 TR)
22 Utilities: Other Compressed Air Capacity: 300 CFM Nitrogen Capacity: 25 Nm3/hr Complies with EP and USP Effluent Treat Plant Onsite Zero Liquid Discharge (ZLD) Sewage Treatment Plant (STP)
23 QUALITY Global Pharmatech s strength is providing products of the highest quality. In keeping with the requirements of our customers, the quality assurance department adopts the best of procedures and practices. The plant is scrupulously operated through built-in rigorous Quality Assurance procedures, strict documentation and internal audits.
24 QUALITY CONTROL INSTRUMENTS UV Spectrophotometer HPLC with Auto sampler Liquid Particle counter Micro Balance IR Spectrophotometer Mini API Analyzer Atomic Absorption Spectrophotometer (AAS)
25 RESEARCH AND DEVELOPMENT Formulation R&D Develop solution formulations in vials and ampoules Enhancing capability to develop emulsions, suspensions, and DEPO products Will acquire capability to develop lyophilization products Tech Transfer Experienced with good project management Analytical R&D Responsible for Method Development and validation
26 LYOPHILIZATION New Capability for Global Pharmatech To be constructed by incorporating part of Unit 1 into Unit 2 Vial line: 200 vpm Lyo Chamber: 8.6 m2 Expected to complete by next 15 to 18 months
27 STABILITY CHAMBERS Climatic Zone Zone Temperature Humidity Real Time II 25 ºC ± 2 ºC 60% RH ± 5% RH Real Time IV a 30 ºC ± 2 ºC 65% RH ± 5% RH Real Time IV b 30 ºC ± 2 ºC 75% RH ± 5% RH Real Time (under installation) Cold Chain 5 ºC ± 3 ºC - Accelerated - 40 ºC ± 2 ºC 75% RH ± 5% RH
28 PRODUCT REGISTRATION PRODUCTS REGISTERED Product Promethazine Hcl 2 ml Ampoule Country Algeria PRODUCTS UNDER REGISTRATION ASIA AFRICA LATAM Philippines Nigeria, Cote d Ivoire Dominican Republic Vietnam South Africa, Namibia, Zambia Jamaica Malaysia Togo, Burkina Faso, Senegal, Mali Peru Myanmar Ethiopia, Kenya, Tanzania, Uganda
29 THANK YOU
Actavis Italy. Nerviano Plant
 Actavis Italy Nerviano Plant Starting out in 1901 in Jerusalem The company known today as Teva was established as a small wholesale drug business by Chaim Salomon, Moshe Levin and Yitschak Elstein. (They
Actavis Italy Nerviano Plant Starting out in 1901 in Jerusalem The company known today as Teva was established as a small wholesale drug business by Chaim Salomon, Moshe Levin and Yitschak Elstein. (They
MEDIS INTERNATIONAL a.s Praha 1, Olivova 4/2096 MEDIS PHARMA Bolatice. Industrial zone Bolatice, region of Opava, Czech Republic
 MEDIS INTERNATIONAL a.s. 110 00 Praha 1, Olivova 4/2096 MEDIS PHARMA Bolatice Industrial zone Bolatice, region of Opava, Czech Republic In October 2008 MEDIS International a.s. started the building of
MEDIS INTERNATIONAL a.s. 110 00 Praha 1, Olivova 4/2096 MEDIS PHARMA Bolatice Industrial zone Bolatice, region of Opava, Czech Republic In October 2008 MEDIS International a.s. started the building of
PERFORMANCE QUALIFICATION PROTOCOL HVAC SYSTEM
 Page 1 of 24 PERFORMANCE QUALIFICATION PROTOCOL FOR HVAC SYSTEM Signing of this Performance Qualification Protocol indicates agreement with the Validation Master Plan approach of the equipment. Further
Page 1 of 24 PERFORMANCE QUALIFICATION PROTOCOL FOR HVAC SYSTEM Signing of this Performance Qualification Protocol indicates agreement with the Validation Master Plan approach of the equipment. Further
Review Validation of aseptic processes for pharmaceuticals
 OPEM www.opem.org Oriental Pharmacy and Experimental Medicine 2010 10(4), 231-238 DOI 10.3742/OPEM.2010.10.4.231 Review Validation of aseptic processes for pharmaceuticals Lincy Joseph*, Mathew George
OPEM www.opem.org Oriental Pharmacy and Experimental Medicine 2010 10(4), 231-238 DOI 10.3742/OPEM.2010.10.4.231 Review Validation of aseptic processes for pharmaceuticals Lincy Joseph*, Mathew George
ABOUT HAMELN PHARMA About us
 Welcome ABOUT HAMELN PHARMA About us With over 60 years of experience hameln pharma is the specialist for contract manufacturing of sterile solutions and suspensions filled in ampoules and vials. 2 ABOUT
Welcome ABOUT HAMELN PHARMA About us With over 60 years of experience hameln pharma is the specialist for contract manufacturing of sterile solutions and suspensions filled in ampoules and vials. 2 ABOUT
S terile. Injections HANDBOOK OF PHARMACEUTICAL. VOLUME 20 - Part One Drug Development - Sterile Injections GENERIC DEVELOPMENT
 HANDBOOK OF PHARMACEUTICAL GENERIC DEVELOPMENT S terile Injections VOLUME 20 - Part One Drug Development - Sterile Injections GENERIC DEVELOPMENT Handbook of Pharmaceutical Generic Development Series Sterile
HANDBOOK OF PHARMACEUTICAL GENERIC DEVELOPMENT S terile Injections VOLUME 20 - Part One Drug Development - Sterile Injections GENERIC DEVELOPMENT Handbook of Pharmaceutical Generic Development Series Sterile
USP Chapter 823 USP 32 (old) vs. USP 35 (new)
 USP Chapter 823 USP 32 (old) vs. USP 35 (new) Sally W. Schwarz, MS, BCNP Research Associate Professor of Radiology Washington University School of Medicine St. Louis, MO Why USP Chapter ? FDA has
USP Chapter 823 USP 32 (old) vs. USP 35 (new) Sally W. Schwarz, MS, BCNP Research Associate Professor of Radiology Washington University School of Medicine St. Louis, MO Why USP Chapter ? FDA has
Whitepaper. High temperature HEPA filtration. Dr.-Ing. Marc Schmidt, Dr.-Ing. Lothar Gail, Hugo Hemel MSc. Preview
 High temperature HEPA filtration Air filtration challenges and answers for dry heat sterilization tunnels Whitepaper Preview Dr.-Ing. Marc Schmidt, Dr.-Ing. Lothar Gail, Hugo Hemel MSc. Air filtration
High temperature HEPA filtration Air filtration challenges and answers for dry heat sterilization tunnels Whitepaper Preview Dr.-Ing. Marc Schmidt, Dr.-Ing. Lothar Gail, Hugo Hemel MSc. Air filtration
General European OMCL Network (GEON) QUALITY MANAGEMENT DOCUMENT
 General European OMCL Network (GEON) QUALITY MANAGEMENT DOCUMENT PA/PH/OMCL (16) 86 R2 MANAGEMENT OF ENVIRONMENTAL CONDITIONS Full document title and reference Document type Management of Environmental
General European OMCL Network (GEON) QUALITY MANAGEMENT DOCUMENT PA/PH/OMCL (16) 86 R2 MANAGEMENT OF ENVIRONMENTAL CONDITIONS Full document title and reference Document type Management of Environmental
Prequalification Team WHO PUBLIC INSPECTION REPORT Vaccine Manufacturer
 Part 1: General information Name of Manufacturer Production Block Physical address Contact address Prequalification Team WHO PUBLIC INSPECTION REPORT Vaccine Manufacturer. Clean Utilities in the Basement.
Part 1: General information Name of Manufacturer Production Block Physical address Contact address Prequalification Team WHO PUBLIC INSPECTION REPORT Vaccine Manufacturer. Clean Utilities in the Basement.
Management Team. Over 35 years experience in the pharmaceuticals industry
 2017 1 Overview Located in Bangalore, a Dedicated cgmp and cglp Compliance Laboratory Established in September 2010 Approved Contract research Organization by India FDA Approved by Health Canada since
2017 1 Overview Located in Bangalore, a Dedicated cgmp and cglp Compliance Laboratory Established in September 2010 Approved Contract research Organization by India FDA Approved by Health Canada since
Compounding Pharmacies and the Contract Testing Lab
 Compounding Pharmacies and the Contract Testing Lab Scott Sutton, Ph.D. scott.sutton@microbiol.org 1 Disclaimer I am making this presentation as an independent agent I am not making this presentation as
Compounding Pharmacies and the Contract Testing Lab Scott Sutton, Ph.D. scott.sutton@microbiol.org 1 Disclaimer I am making this presentation as an independent agent I am not making this presentation as
Where Quality Meets Flexibility
 Where Quality Meets Flexibility is an industry leading 503B Outsourcing Facility providing sterile and non-sterile compounding services to hospitals, surgery centers, clinics, researchers & patients nationwide.
Where Quality Meets Flexibility is an industry leading 503B Outsourcing Facility providing sterile and non-sterile compounding services to hospitals, surgery centers, clinics, researchers & patients nationwide.
Page 1
 Engineers to Healthcare Industry INTERTECH EQUIPTECHNOLOGIES PVT. LTD. Company Presentation 2014 www.intertechequip.com Page 1 INTERTECH Production facility Total Fabrication Area 75,000 ft2 & Clean room
Engineers to Healthcare Industry INTERTECH EQUIPTECHNOLOGIES PVT. LTD. Company Presentation 2014 www.intertechequip.com Page 1 INTERTECH Production facility Total Fabrication Area 75,000 ft2 & Clean room
Prequalification of medicines
 Prequalification of medicines Wondiyfraw Worku WHO Prequalification Team 3.2.S.3.2 Impurities, Malaysia, 1 29 September 2011 CPHI Mumbai 2017 1 Background In early 2000, there was an increasing demand
Prequalification of medicines Wondiyfraw Worku WHO Prequalification Team 3.2.S.3.2 Impurities, Malaysia, 1 29 September 2011 CPHI Mumbai 2017 1 Background In early 2000, there was an increasing demand
Annex 14 WHO guidelines for drafting a site master file 136
 World Health Organization WHO Technical Report Series, No.961, 2011 Annex 14 WHO guidelines for drafting a site master file 136 1. Introduction 2. Purpose 3. Scope 4. Content of site master fi le Appendix
World Health Organization WHO Technical Report Series, No.961, 2011 Annex 14 WHO guidelines for drafting a site master file 136 1. Introduction 2. Purpose 3. Scope 4. Content of site master fi le Appendix
Quality is Our Promise.
 Quality is Our Promise. Our goal at KRS Global Biotechnology is to provide the highest quality pharmaceutical preparations. We accomplish this with an unrivaled quality assurance and quality control program
Quality is Our Promise. Our goal at KRS Global Biotechnology is to provide the highest quality pharmaceutical preparations. We accomplish this with an unrivaled quality assurance and quality control program
Your partner in the pharmaceutical industry
 English Micronisation- and milling service Micronised by GfM Your API Particle size (µm) Your partner in the pharmaceutical industry The... always that little bit finer. We are a family business in the
English Micronisation- and milling service Micronised by GfM Your API Particle size (µm) Your partner in the pharmaceutical industry The... always that little bit finer. We are a family business in the
On-Site GMP Training GMP COMPLIANCE TECHNICAL
 PharmaNet On-Site GMP Training GMP COMPLIANCE TECHNICAL 284 E Lake Mead Pkwy Suite C-278 Henderson, NV 89015 Phone: 702-558-0094 Fax: 702-558-0079 www.gmpseminars.com Key to Level of Program Level A: Program
PharmaNet On-Site GMP Training GMP COMPLIANCE TECHNICAL 284 E Lake Mead Pkwy Suite C-278 Henderson, NV 89015 Phone: 702-558-0094 Fax: 702-558-0079 www.gmpseminars.com Key to Level of Program Level A: Program
Prequalification Team WHO PUBLIC INSPECTION REPORT Vaccine Manufacturer
 Prequalification Team WHO PUBLIC INSPECTION REPORT Vaccine Manufacturer Part 1: General information Name of Manufacturer / Iksan Plant Unit number Production Block Physical address Contact person and email
Prequalification Team WHO PUBLIC INSPECTION REPORT Vaccine Manufacturer Part 1: General information Name of Manufacturer / Iksan Plant Unit number Production Block Physical address Contact person and email
Sterility Assurance Level and Aseptic Manufacturing Process in Pharmaceuticals
 Review Article ISSN 2277-3657 Available online at www.ijpras.com Volume 3, Issue 4 (2014),10-15 International Journal of Pharmaceutical Research & Allied Sciences Sterility Assurance Level and Aseptic
Review Article ISSN 2277-3657 Available online at www.ijpras.com Volume 3, Issue 4 (2014),10-15 International Journal of Pharmaceutical Research & Allied Sciences Sterility Assurance Level and Aseptic
National Institute of Solar Energy. National Institute of Solar Energy
 ABOUT THE INSTITUTE, an autonomous institution of Ministry of New and Renewable (MNRE), is the apex National R&D institution in the field Solar Energy Assist the Ministry in implementing the National Solar
ABOUT THE INSTITUTE, an autonomous institution of Ministry of New and Renewable (MNRE), is the apex National R&D institution in the field Solar Energy Assist the Ministry in implementing the National Solar
STERIS FINN-AQUA Pure Steam Generator for Pharmaceutical Manufacturing
 cgmp MANUFACTURING STERIS FINN-AQUA Pure Steam Generator for Pharmaceutical Manufacturing Life Sciences STERIS Corporation has achieved worldwide leadership in the pharmaceutical and biotech industries
cgmp MANUFACTURING STERIS FINN-AQUA Pure Steam Generator for Pharmaceutical Manufacturing Life Sciences STERIS Corporation has achieved worldwide leadership in the pharmaceutical and biotech industries
MEDIS International, a.s., Karlovo náměstí 3, Praha 2, tel :
 MEDIS INTERNATIONAL a.s. 110 00 Praha 1, Olivova 4/2096 MEDIS PHARMA Bolatice Industrial zone Bolatice, region of Opava, Czech Republic MEDIS International a.s. started to build a new facility in October
MEDIS INTERNATIONAL a.s. 110 00 Praha 1, Olivova 4/2096 MEDIS PHARMA Bolatice Industrial zone Bolatice, region of Opava, Czech Republic MEDIS International a.s. started to build a new facility in October
GMP On Site Series. GMP Essentials
 GMP On Site Series GMP Essentials GMP Basics Objectives 1. State the critical definitions of the pharmaceutical industry. 2. Describe the law as it applies to various critical functions. 3. State the history
GMP On Site Series GMP Essentials GMP Basics Objectives 1. State the critical definitions of the pharmaceutical industry. 2. Describe the law as it applies to various critical functions. 3. State the history
Annex A2. Guidance on Process Validation Scheme for Aseptically Processed Products
 Annex A2 Guidance on Process Validation Scheme for Aseptically Processed Products 1 Table of content 1 PURPOSE... 3 2 SCOPE... 3 3 GENERAL INFORMATION... 3 4 INFORMATION NEEDED FOR ASEPTIC PROCESSES VALIDATION...
Annex A2 Guidance on Process Validation Scheme for Aseptically Processed Products 1 Table of content 1 PURPOSE... 3 2 SCOPE... 3 3 GENERAL INFORMATION... 3 4 INFORMATION NEEDED FOR ASEPTIC PROCESSES VALIDATION...
PRAXIS. A publication by Bioengineering AG
 PRAXIS A publication by Bioengineering AG Portrait of Rentschler Biotechnologie GmbH, a globally active service company that supports its clients in the development, production, and registration of biopharmaceuticals.
PRAXIS A publication by Bioengineering AG Portrait of Rentschler Biotechnologie GmbH, a globally active service company that supports its clients in the development, production, and registration of biopharmaceuticals.
SCHOTT is an international technology group with more than 125 years of experience in the areas of specialty glasses and materials and advanced
 Ampoules 2 SCHOTT is an international technology group with more than 125 years of experience in the areas of specialty glasses and materials and advanced technologies. With our high-quality products and
Ampoules 2 SCHOTT is an international technology group with more than 125 years of experience in the areas of specialty glasses and materials and advanced technologies. With our high-quality products and
Inspections, Compliance, Enforcement, and Criminal Investigations
 Page 1 of 8 Inspections, Compliance, Enforcement, and Criminal Investigations Lupin Limited 5/7/09 Department of Health and Human Services Public Health Service Food and Drug Administration CENTER FOR
Page 1 of 8 Inspections, Compliance, Enforcement, and Criminal Investigations Lupin Limited 5/7/09 Department of Health and Human Services Public Health Service Food and Drug Administration CENTER FOR
for IND and RDRC Regulated PET Compounding
 Overview of USP Chapter for IND and RDRC Regulated PET Compounding Distributed Manufacturing of PET Radiopharmaceuticals for Multi-Center Clinical Trials SNM, Annual Meeting Toronto, Ontario, Canada
Overview of USP Chapter for IND and RDRC Regulated PET Compounding Distributed Manufacturing of PET Radiopharmaceuticals for Multi-Center Clinical Trials SNM, Annual Meeting Toronto, Ontario, Canada
Compounding Pharmacies and Water
 Compounding Pharmacies and Water Scott Sutton, PhD scott.sutton@microbiol.org August 21, 2014 46 Disclaimer I am an independent consultant. I have been involved with USP for many years. I do not represent
Compounding Pharmacies and Water Scott Sutton, PhD scott.sutton@microbiol.org August 21, 2014 46 Disclaimer I am an independent consultant. I have been involved with USP for many years. I do not represent
UNIVERSITY OF KANSAS Office of Institutional Research and Planning
 10/13 TABLE 4-170 FALL - TOTAL 1,624 1,740 1,926 2,135 2,134 2,138 2,246 Male 927 968 1,076 1,191 1,188 1,179 1,262 Female 697 772 850 944 946 959 984 Undergraduate 685 791 974 1,181 1,189 1,217 1,281
10/13 TABLE 4-170 FALL - TOTAL 1,624 1,740 1,926 2,135 2,134 2,138 2,246 Male 927 968 1,076 1,191 1,188 1,179 1,262 Female 697 772 850 944 946 959 984 Undergraduate 685 791 974 1,181 1,189 1,217 1,281
FUCHS Pharmaceutical and Cosmetics Products. Soft Paraffins BP/EP, Liquid & Light Liquid Paraffins BP/EP (White Oils)
 FUCHS Pharmaceutical and Cosmetics Products Soft Paraffins BP/EP, Liquid & Light Liquid Paraffins BP/EP (White Oils) FUCHS has over 40 years experience in the production of White & Yellow Soft Paraffins
FUCHS Pharmaceutical and Cosmetics Products Soft Paraffins BP/EP, Liquid & Light Liquid Paraffins BP/EP (White Oils) FUCHS has over 40 years experience in the production of White & Yellow Soft Paraffins
Global Food Security Index
 Global Food Security Index Sponsored by 26 September 2012 Agenda Overview Methodology Overall results Results for India Website 2 Overview The Economist Intelligence Unit was commissioned by DuPont to
Global Food Security Index Sponsored by 26 September 2012 Agenda Overview Methodology Overall results Results for India Website 2 Overview The Economist Intelligence Unit was commissioned by DuPont to
GLOBAL QUALITY SOLUTIONS PACKAGE INTEGRITY SEAL INTEGRITY LEAK DETECTION
 GLOBAL QUALITY SOLUTIONS PACKAGE INTEGRITY SEAL INTEGRITY LEAK DETECTION Redefining the standards for accuracy and reliability. About PTI PTI Packaging Technologies & Inspection is headquartered in Tuckahoe,
GLOBAL QUALITY SOLUTIONS PACKAGE INTEGRITY SEAL INTEGRITY LEAK DETECTION Redefining the standards for accuracy and reliability. About PTI PTI Packaging Technologies & Inspection is headquartered in Tuckahoe,
GMP Inspection Process. Types of GMP Inspection. Module 18 Slide 1 of 14 WHO - EDM
 GMP Inspection Process Types of GMP Inspection Module 18 Slide 1 of 14 WHO - EDM Objectives 1. To review the different types of inspection 2. To examine when each is appropriate 3. To discuss inspections
GMP Inspection Process Types of GMP Inspection Module 18 Slide 1 of 14 WHO - EDM Objectives 1. To review the different types of inspection 2. To examine when each is appropriate 3. To discuss inspections
THE R&D LABORATORY THAT BOOSTS YOUR DEVELOPMENT PROJECTS
 THE R&D LABORATORY THAT BOOSTS YOUR DEVELOPMENT PROJECTS CREATED IN 2011 WITH A PROVEN EXPERTISE IN AUDIT AND REGULATORY AFFAIRS, BIOLABEX NORTH AMERICA PARIS EUROPE ASIA EXTENDS ITS OFFERINGS AND IS NOW
THE R&D LABORATORY THAT BOOSTS YOUR DEVELOPMENT PROJECTS CREATED IN 2011 WITH A PROVEN EXPERTISE IN AUDIT AND REGULATORY AFFAIRS, BIOLABEX NORTH AMERICA PARIS EUROPE ASIA EXTENDS ITS OFFERINGS AND IS NOW
Critical Path to TB drug Regimens 2016 Workshop
 Critical Path to TB drug Regimens 2016 Workshop AT A GLANCE Quality Pharmaceutical manufacturing company from Kenya Sales KES 2.26B (USD 22.6M) 2015 Unaudited 65% Business from Donors Portfolio of more
Critical Path to TB drug Regimens 2016 Workshop AT A GLANCE Quality Pharmaceutical manufacturing company from Kenya Sales KES 2.26B (USD 22.6M) 2015 Unaudited 65% Business from Donors Portfolio of more
KSP ID. Chairman. Dr. Abdul Rahman Abdullah Al-Awadi. Date of Establishment. 15 th May Ownership
 KSP ID Chairman Dr. Abdul Rahman Abdullah Al-Awadi Date of Establishment 15 th May 1994 Ownership A joint venture between National Pharmaceutical Industries Co. & Securities Group Co. Products Manufacturing,
KSP ID Chairman Dr. Abdul Rahman Abdullah Al-Awadi Date of Establishment 15 th May 1994 Ownership A joint venture between National Pharmaceutical Industries Co. & Securities Group Co. Products Manufacturing,
Country CAPEXIL Description HS Codes Value Qty AFGHANISTAN TIS Asbestos cement pipes
 Country-wise and Item-wise Exports of Cement, Clinkers and Asbestos Cement Products Value Rs. Lakh Quantity in '000 Unit: Kgs Source: MoC Export Import Data Bank Country CAPEXIL Description HS Codes Value
Country-wise and Item-wise Exports of Cement, Clinkers and Asbestos Cement Products Value Rs. Lakh Quantity in '000 Unit: Kgs Source: MoC Export Import Data Bank Country CAPEXIL Description HS Codes Value
API Stability Protocols and. Chris Byrne Tasmanian Alkaloids
 API Stability Protocols and Evaluations Chris Byrne Tasmanian Alkaloids API Stability Overview APIs = 100% pure Limited (if any) degradation No interactions with other agents in drug products Less likelihood
API Stability Protocols and Evaluations Chris Byrne Tasmanian Alkaloids API Stability Overview APIs = 100% pure Limited (if any) degradation No interactions with other agents in drug products Less likelihood
Supplier Assurance Program. CBE Pty Ltd
 Supplier Assurance Program CBE Pty Ltd This training program is copyright to CBE Pty Ltd and may not be modified, reproduced, sold, loaned, hired or traded in any form without its express written permission.
Supplier Assurance Program CBE Pty Ltd This training program is copyright to CBE Pty Ltd and may not be modified, reproduced, sold, loaned, hired or traded in any form without its express written permission.
Tests to Support Sterility Claim. Imtiaz Ahmed
 Tests to Support Sterility Claim Imtiaz Ahmed Sterile Product As per TGO 77, a sterile product must comply with the requirements of the following tests: Sterility Test Bacterial Endotoxins Test Appendix
Tests to Support Sterility Claim Imtiaz Ahmed Sterile Product As per TGO 77, a sterile product must comply with the requirements of the following tests: Sterility Test Bacterial Endotoxins Test Appendix
Visible Particles: Regulatory and Compendial Requirements
 Visible Particles: Regulatory and Compendial Requirements John G. Shabushnig, Ph.D. Insight Pharma Consulting, LLC June 2014 Agenda Visible Particle Definitions US FDA US Pharmacopeia (USP) EC GMP s European
Visible Particles: Regulatory and Compendial Requirements John G. Shabushnig, Ph.D. Insight Pharma Consulting, LLC June 2014 Agenda Visible Particle Definitions US FDA US Pharmacopeia (USP) EC GMP s European
Soft Paraffins BP/EP, Liquid & Light Liquid Paraffins BP/EP (White Oils)
 FUCHS Pharmaceutical and Cosmetics Products Innovative solutions need experienced advice FUCHS Lubricants (UK) plc Special Products Team will be glad to advise on the right product for each application
FUCHS Pharmaceutical and Cosmetics Products Innovative solutions need experienced advice FUCHS Lubricants (UK) plc Special Products Team will be glad to advise on the right product for each application
PDA: A Global. Association. Matrix Approach to Media Fills. (c) 2012 Catalent Pharma Solutions. All rights reserved.
 PDA: A Global Matrix Approach to Media Fills Association (c) 2012 Catalent Pharma Solutions. All rights reserved. Overview Guidance Overview Matrix Introduction Why Matrix Media Fills Sample Matrix New
PDA: A Global Matrix Approach to Media Fills Association (c) 2012 Catalent Pharma Solutions. All rights reserved. Overview Guidance Overview Matrix Introduction Why Matrix Media Fills Sample Matrix New
Dynamic Future. Reliable Partnership. Company Profile of the Aenova Group
 Dynamic Future. Reliable Partnership. Company Profile of the Aenova Group Dynamic Future. Reliable Partnership. With a turnover of approximately 1 billion USD, the Aenova Group is one of the leading companies
Dynamic Future. Reliable Partnership. Company Profile of the Aenova Group Dynamic Future. Reliable Partnership. With a turnover of approximately 1 billion USD, the Aenova Group is one of the leading companies
Aseptic Process Validation
 Aseptic Process Validation IMB GMP Information Seminar 27 th September 2012 Gerard Sheridan, Inspector Date Insert on Master Slide Slide 1 Overview Guidance Best Practices Common Deficiencies Slide 2 Aseptic
Aseptic Process Validation IMB GMP Information Seminar 27 th September 2012 Gerard Sheridan, Inspector Date Insert on Master Slide Slide 1 Overview Guidance Best Practices Common Deficiencies Slide 2 Aseptic
EU and FDA GMP Regulations: Overview and Comparison
 THE QUALITY ASSURANCE JOURNAL, VOL. 2, 55 60 (1997) EU and FDA GMP Regulations: Overview and Comparison The increasing emphasis on global supply of drug products, as well as starting materials and investigational
THE QUALITY ASSURANCE JOURNAL, VOL. 2, 55 60 (1997) EU and FDA GMP Regulations: Overview and Comparison The increasing emphasis on global supply of drug products, as well as starting materials and investigational
SCHOTT Vials. Perfection in Every Detail
 SCHOTT Vials Perfection in Every Detail 2 3 SCHOTT is a leading international technology group in the areas of specialty glass and glass-ceramics. With more than 130 years of outstanding development, materials
SCHOTT Vials Perfection in Every Detail 2 3 SCHOTT is a leading international technology group in the areas of specialty glass and glass-ceramics. With more than 130 years of outstanding development, materials
Automatic inspection for vials, ampules and cartridges
 CS-series Automatic inspection for vials, ampules and cartridges CS-Series Purity and sterility of parenterals vital to patients health. In the pharmaceutical industry, quality standards are becoming more
CS-series Automatic inspection for vials, ampules and cartridges CS-Series Purity and sterility of parenterals vital to patients health. In the pharmaceutical industry, quality standards are becoming more
Partner with the Global Leader in Drug Delivery Systems
 3M DRUG DELIVERY SYSTEMS Partner with the Global Leader in Drug Delivery Systems Northridge, CA, USA Manufacturing Facility Experts at Commercializing Innovation 3M: Transforming New Ideas into Thousands
3M DRUG DELIVERY SYSTEMS Partner with the Global Leader in Drug Delivery Systems Northridge, CA, USA Manufacturing Facility Experts at Commercializing Innovation 3M: Transforming New Ideas into Thousands
ISPE Aseptic Conference February 2014 Washington, D.C.
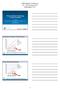 Trends of Barrier Technology in China and Japan Alex Cheng: Shanghai Tofflon Airex Science and Technology Co.,Ltd. Koji Kawaskai: Airex CoLtd. Japan Filling Barrier Isolators Global Deliveries 500 450
Trends of Barrier Technology in China and Japan Alex Cheng: Shanghai Tofflon Airex Science and Technology Co.,Ltd. Koji Kawaskai: Airex CoLtd. Japan Filling Barrier Isolators Global Deliveries 500 450
PROJECT REPORT FOR MANUFACTURING THINQ PHARMA CRO LTD PITHAMPUR -3 BADGAON MADHYA PRADESH
 PROJECT REPORT FOR MANUFACTURING THINQ PHARMA CRO LTD PITHAMPUR -3 BADGAON MADHYA PRADESH CONTENTS 1. INTRODUCTION 2. GENERAL SECTION 3. PARTICULARS OF PROJECT 4. COST OF PROJECT AND MEANS OF FINANCING
PROJECT REPORT FOR MANUFACTURING THINQ PHARMA CRO LTD PITHAMPUR -3 BADGAON MADHYA PRADESH CONTENTS 1. INTRODUCTION 2. GENERAL SECTION 3. PARTICULARS OF PROJECT 4. COST OF PROJECT AND MEANS OF FINANCING
Critical Environment Products and Services
 Critical Environment Products and Services For over two decades, EP Scientific products and services have defined clean for environmental sampling containers and services. Our proprietary cleaning methods
Critical Environment Products and Services For over two decades, EP Scientific products and services have defined clean for environmental sampling containers and services. Our proprietary cleaning methods
WHO PUBLIC INSPECTION REPORT (WHOPIR) Quality Control Laboratory
 SOP 408.4 Annex D 20, avenue Appia CH-1211 Geneva 27 Switzerland Tel central +41 22 791 2111 Fax central +41 22 791 3111 www.who.int WHO PUBLIC INSPECTION REPORT (WHOPIR) Quality Control Laboratory Part
SOP 408.4 Annex D 20, avenue Appia CH-1211 Geneva 27 Switzerland Tel central +41 22 791 2111 Fax central +41 22 791 3111 www.who.int WHO PUBLIC INSPECTION REPORT (WHOPIR) Quality Control Laboratory Part
FDA S GUIDANCE FOR INDUSTRY ANDAS: STABILITY TESTING OF DRUG SUBSTANCES AND PRODUCTS
 FDA S GUIDANCE FOR INDUSTRY ANDAS: STABILITY TESTING OF DRUG SUBSTANCES AND PRODUCTS 02-December-2014 San Diego, CA Kim Huynh-Ba Executive Director PHARMALYTIK Kim.huynhba@pharmalytik.com Overview Stability
FDA S GUIDANCE FOR INDUSTRY ANDAS: STABILITY TESTING OF DRUG SUBSTANCES AND PRODUCTS 02-December-2014 San Diego, CA Kim Huynh-Ba Executive Director PHARMALYTIK Kim.huynhba@pharmalytik.com Overview Stability
Environmental Monitoring of Aseptic Processing Areas - 1
 Environmental Monitoring of Aseptic Processing Areas - 1 A war against an invisible enemy DCVMN - Víctor Maqueda - May 30, 31, June 1 2016 1 Training Course Agenda Overview of Environmental Monitoring
Environmental Monitoring of Aseptic Processing Areas - 1 A war against an invisible enemy DCVMN - Víctor Maqueda - May 30, 31, June 1 2016 1 Training Course Agenda Overview of Environmental Monitoring
GUIDELINES ON THE STABILITY DATA REQUIRED FOR REGISTRATION OF STOCK REMEDIES IN SOUTH AFRICA
 GUIDELINES ON THE STABILITY DATA REQUIRED FOR REGISTRATION OF STOCK REMEDIES IN SOUTH AFRICA Issued by the Registrar: Act No. 36 of 1947, Private Bag X343, Pretoria 0001 Republic of South Africa Tel. (**27
GUIDELINES ON THE STABILITY DATA REQUIRED FOR REGISTRATION OF STOCK REMEDIES IN SOUTH AFRICA Issued by the Registrar: Act No. 36 of 1947, Private Bag X343, Pretoria 0001 Republic of South Africa Tel. (**27
Zydus Hospira Oncology Pvt. Ltd.
 Company, Position & Person Profile Zydus Hospira Oncology Pvt. Ltd. 6/7/15 About Zydus Hospira Oncology Pvt. Ltd. Zydus Hospira Oncology Private Limited (ZHOPL) is a joint venture company in Ahmedabad,
Company, Position & Person Profile Zydus Hospira Oncology Pvt. Ltd. 6/7/15 About Zydus Hospira Oncology Pvt. Ltd. Zydus Hospira Oncology Private Limited (ZHOPL) is a joint venture company in Ahmedabad,
Table D.1: Time Spent Collecting Wood (hours/day)
 Fuelwood Collection and Cooking Table D.1 shows that the time spent collecting wood each day varies quite considerably. But the average time is more than 2.5 hours, and it can be eight times that. The
Fuelwood Collection and Cooking Table D.1 shows that the time spent collecting wood each day varies quite considerably. But the average time is more than 2.5 hours, and it can be eight times that. The
Overview of a sterility assurance program for PET drugs
 Coalition for PET Drug Approval Radiopharmaceutical Sciences Council Overview of a sterility assurance program for PET drugs Eric Webster, PETNET Solutions Disclosures Employee of PETNET Solutions, a Siemens
Coalition for PET Drug Approval Radiopharmaceutical Sciences Council Overview of a sterility assurance program for PET drugs Eric Webster, PETNET Solutions Disclosures Employee of PETNET Solutions, a Siemens
Preparing Your Aseptic Processing Facility for an FDA Inspection. Valerie Welter, Director Quality Bayer HealthCare March 10, 2014
 Preparing Your Aseptic Processing Facility for an FDA Inspection Valerie Welter, Director Quality Bayer HealthCare March 10, 2014 Agenda Regulatory Requirements Establishing your Approach Aseptic Controls
Preparing Your Aseptic Processing Facility for an FDA Inspection Valerie Welter, Director Quality Bayer HealthCare March 10, 2014 Agenda Regulatory Requirements Establishing your Approach Aseptic Controls
Media Fill A Process Simution. Presented By Shikha Chauhan
 Media Fill A Process Simution Presented By Shikha Chauhan 21 CFR 211.113 Validation of Aseptic Processing and Sterilization Process Simulation / Media Fill Filtration Efficacy Sterilization of Equipment,
Media Fill A Process Simution Presented By Shikha Chauhan 21 CFR 211.113 Validation of Aseptic Processing and Sterilization Process Simulation / Media Fill Filtration Efficacy Sterilization of Equipment,
KINGSMANN CARE GROUP
 PHARMA CONSULTANTS KINGSMANN CARE GROUP KINGSMANN CONSULTANCY SERVICES Thank you for taking interest in Kingsmann Consultancy Services. Kingsmann Consultancy (KC) is a leading business development-consulting
PHARMA CONSULTANTS KINGSMANN CARE GROUP KINGSMANN CONSULTANCY SERVICES Thank you for taking interest in Kingsmann Consultancy Services. Kingsmann Consultancy (KC) is a leading business development-consulting
UNICEF Quality Assurance in the procurement of medicines
 UNICEF Quality Assurance in the procurement of medicines Peter S. Jakobsen 30 October 2006 Quality Assurance Centre SUPPLY DIVISION Today s presentation addresses 3 questions: How is UNICEF involved in
UNICEF Quality Assurance in the procurement of medicines Peter S. Jakobsen 30 October 2006 Quality Assurance Centre SUPPLY DIVISION Today s presentation addresses 3 questions: How is UNICEF involved in
FITOCLIMA WALK-IN STABILITY
 FITOCLIMA WALK-IN TEMPERATURE AND HUMIDITY CHAMBERS ICH, GMP, WHO, FDA COMPLIANT Common applications include: Pharmaceuticals Food and Beverages Cosmetics Veterinary Storage and Conservation Quality control
FITOCLIMA WALK-IN TEMPERATURE AND HUMIDITY CHAMBERS ICH, GMP, WHO, FDA COMPLIANT Common applications include: Pharmaceuticals Food and Beverages Cosmetics Veterinary Storage and Conservation Quality control
In Agriculture. UN-Water Project on. and 2 nd Regional Workshops; Scope of the 3 rd Regional Workshop. Africa Asia Latin America
 UN-Water Project on Safe Safe Use Use of Wastewater of Wastewater in Agriculture In Agriculture Africa Asia Latin America Recap 1st Regional of the Workshop International for Francophone Kick-off, Africa
UN-Water Project on Safe Safe Use Use of Wastewater of Wastewater in Agriculture In Agriculture Africa Asia Latin America Recap 1st Regional of the Workshop International for Francophone Kick-off, Africa
Quality Agreements with CMO s. Presented at ASQ Orange Empire Meeting May 13, 2014 By Luke Foo Sr. Director QA/QC Spectrum Pharmaceuticals
 Quality Agreements with CMO s Presented at ASQ Orange Empire Meeting May 13, 2014 By Luke Foo Sr. Director QA/QC Spectrum Pharmaceuticals FDA Guidance Contract Manufacturing Arrangements for Drugs: Quality
Quality Agreements with CMO s Presented at ASQ Orange Empire Meeting May 13, 2014 By Luke Foo Sr. Director QA/QC Spectrum Pharmaceuticals FDA Guidance Contract Manufacturing Arrangements for Drugs: Quality
Request for Quote RFQ #
 Request for Quote RFQ # 2017-007 Drug Product Manufacture: GMP Fill/Finish of Monoclonal Antibody I. Summary of Deadlines Release of Request for quote Feb 13, 2017 Confirmation of interest due by Feb 20,
Request for Quote RFQ # 2017-007 Drug Product Manufacture: GMP Fill/Finish of Monoclonal Antibody I. Summary of Deadlines Release of Request for quote Feb 13, 2017 Confirmation of interest due by Feb 20,
BLISTER PACKING MACHINES AND SERVICES THAT OFFER FLEXIBILITY AND HIGH QUALITY BLISTER PACKS FROM A WIDE VARIETY OF PACKAGING MATERIALS.
 BLISTER PACKING MACHINES AND SERVICES THAT OFFER FLEXIBILITY AND HIGH QUALITY BLISTER PACKS FROM A WIDE VARIETY OF PACKAGING MATERIALS. 06 1. PACKAGING SOLUTIONS 07 1. PACKAGING SOLUTIONS SMALL SCALE,
BLISTER PACKING MACHINES AND SERVICES THAT OFFER FLEXIBILITY AND HIGH QUALITY BLISTER PACKS FROM A WIDE VARIETY OF PACKAGING MATERIALS. 06 1. PACKAGING SOLUTIONS 07 1. PACKAGING SOLUTIONS SMALL SCALE,
ENCON IN THERMAL SYSTEM
 ENCON IN THERMAL SYSTEM Orchid chemicals & Pharmaceuticals ltd. Bulk API Alathur facility Bulk API Aurangabad facility OHC (Formulations) Alathur facility R&D Centre Corporate Office OHC (Formulations)
ENCON IN THERMAL SYSTEM Orchid chemicals & Pharmaceuticals ltd. Bulk API Alathur facility Bulk API Aurangabad facility OHC (Formulations) Alathur facility R&D Centre Corporate Office OHC (Formulations)
USP Perspective on Pharmaceutical Waters. Antonio Hernandez-Cardoso, M.Sc. Senior Scientific Liaison September 7, 2017
 USP Perspective on Pharmaceutical Waters Antonio Hernandez-Cardoso, M.Sc. Senior Scientific Liaison September 7, 2017 Why is Water Important? Raw material Solvent Ingredient Reagent Cleaning agent (hot
USP Perspective on Pharmaceutical Waters Antonio Hernandez-Cardoso, M.Sc. Senior Scientific Liaison September 7, 2017 Why is Water Important? Raw material Solvent Ingredient Reagent Cleaning agent (hot
For Safe and Delicious Water
 RESIDUAL CHLORINE ANALYZER 400G NON-REAGENT FREE CHLORINE ANALYZER FC400G 400G RESIDUAL CHLORINE ANALYZER FC400G NON-REAGENT FREE CHLORINE ANALYZER Bulletin 12F01A01-01E www.yokogawa.com/an/ For Safe and
RESIDUAL CHLORINE ANALYZER 400G NON-REAGENT FREE CHLORINE ANALYZER FC400G 400G RESIDUAL CHLORINE ANALYZER FC400G NON-REAGENT FREE CHLORINE ANALYZER Bulletin 12F01A01-01E www.yokogawa.com/an/ For Safe and
Regulatory Expectations, Standards & Guidelines
 Regulatory Expectations, Standards & Guidelines Regulatory Requirements Pharmacopeias Good Automated Manufacturing Practice (GAMP) 21 CFR Part 11 and Annex 11 Consequences of Non-Compliance 22 Regulatory
Regulatory Expectations, Standards & Guidelines Regulatory Requirements Pharmacopeias Good Automated Manufacturing Practice (GAMP) 21 CFR Part 11 and Annex 11 Consequences of Non-Compliance 22 Regulatory
Pharmaceutical Packaging
 Pharmaceutical Packaging 2017 2021 Section I: Introduction A. Pharmaceuticals defined B. Pharmaceutical packaging defined C. Study organization D. Methodology E. Geographic regions F. Conventions Section
Pharmaceutical Packaging 2017 2021 Section I: Introduction A. Pharmaceuticals defined B. Pharmaceutical packaging defined C. Study organization D. Methodology E. Geographic regions F. Conventions Section
African Export-Import Bank Afreximbank
 African Export-Import Bank Afreximbank Gwen Mwaba Director Trade Finance Geneva, 2017 African Export-Import Bank Banque Africaine D Import-Export Transforming Africa s Trade Trade Finance The Trade Finance
African Export-Import Bank Afreximbank Gwen Mwaba Director Trade Finance Geneva, 2017 African Export-Import Bank Banque Africaine D Import-Export Transforming Africa s Trade Trade Finance The Trade Finance
Pharmacopoeial Reference Standards
 Pharmacopoeial Reference Standards Industry view point Antony Raj Gomes Head- Quality Management 1 Presentation Overview Indian Industry an update Available guidelines Industry practices Current challenges
Pharmacopoeial Reference Standards Industry view point Antony Raj Gomes Head- Quality Management 1 Presentation Overview Indian Industry an update Available guidelines Industry practices Current challenges
Emcure Pharmaceuticals Limited 3/3/16
 1 of 7 05/06/2016 8:22 AM U.S. Food and Drug Administration Protecting and Promoting Your Health Emcure Pharmaceuticals Limited 3/3/16 Department of Health and Human Services Public Health Service Food
1 of 7 05/06/2016 8:22 AM U.S. Food and Drug Administration Protecting and Promoting Your Health Emcure Pharmaceuticals Limited 3/3/16 Department of Health and Human Services Public Health Service Food
Amazon FILTRATION SOLUTIONS PHARMACEUTICAL MANUFACTURING
 Amazon FILTRATION SOLUTIONS PHARMACEUTICAL MANUFACTURING FILTRATION SOLUTIONS FOR PHARMACEUTICAL MANUFACTURING Delivering quality filtration products As one of Europe s leading manufacturers of process
Amazon FILTRATION SOLUTIONS PHARMACEUTICAL MANUFACTURING FILTRATION SOLUTIONS FOR PHARMACEUTICAL MANUFACTURING Delivering quality filtration products As one of Europe s leading manufacturers of process
Quality Risk Management (ICH Q9): WHO Model for Sustainable Quality Medicines
 Quality Risk Management (ICH Q9): WHO Model for Sustainable Quality Medicines Longe Sunday Anthony Head- Quality Assurance May & Baker Nigeria Plc. Pharmacentre, Ota, Nigeria eaglesconsult@gmail.com; Slonge@may-
Quality Risk Management (ICH Q9): WHO Model for Sustainable Quality Medicines Longe Sunday Anthony Head- Quality Assurance May & Baker Nigeria Plc. Pharmacentre, Ota, Nigeria eaglesconsult@gmail.com; Slonge@may-
Impacto de las Nuevas Tendencias Regulatorias en la Producción de Parenterales. Innovacion en Packaging Primario
 Impacto de las Nuevas Tendencias Regulatorias en la Producción de Parenterales Innovacion en Packaging Primario 1 Contents Company Highlight What s Changing in Pharma Pharma Trends Harmonisation New Requirements
Impacto de las Nuevas Tendencias Regulatorias en la Producción de Parenterales Innovacion en Packaging Primario 1 Contents Company Highlight What s Changing in Pharma Pharma Trends Harmonisation New Requirements
Reference Standards for Monoclonal Antibodies: Key Challenges Addressed
 CASSS WCBP 2012: 16th Symposium on the Interface of Regulatory and Analytical Sciences for Biotechnology Health Products January 23-25, 2012 Reference Standards for Monoclonal Antibodies: Key Challenges
CASSS WCBP 2012: 16th Symposium on the Interface of Regulatory and Analytical Sciences for Biotechnology Health Products January 23-25, 2012 Reference Standards for Monoclonal Antibodies: Key Challenges
Validation Study on How to Avoid Microbial Contamination during Pharmaceutical Production
 Biocontrol Science, 2015, Vol. 20, No.1, 1 10 Review Validation Study on How to Avoid Microbial Contamination during Pharmaceutical Production HIDEHARU SHINTANI 1-13-27, Kasuga, Bunkyo, Tokyo 112-0003,
Biocontrol Science, 2015, Vol. 20, No.1, 1 10 Review Validation Study on How to Avoid Microbial Contamination during Pharmaceutical Production HIDEHARU SHINTANI 1-13-27, Kasuga, Bunkyo, Tokyo 112-0003,
(To be filled up by the applicant) 01. Name or style in which the applicant is assessed or assessable to Sales Tax Addresses or assessment.
 ANNEXURE-I Ref. Clause No. 4.(1) (m) FORM OR CERTIFICATE OF SALES TAX VERIFICATION TO BE PRODUCED BY AN APPLICANT FROM THE CONTRACT OR OTHER PATRONAGE AT THE DISPOSAL OF THE GOVERNMENT OF TAMILNADU. (To
ANNEXURE-I Ref. Clause No. 4.(1) (m) FORM OR CERTIFICATE OF SALES TAX VERIFICATION TO BE PRODUCED BY AN APPLICANT FROM THE CONTRACT OR OTHER PATRONAGE AT THE DISPOSAL OF THE GOVERNMENT OF TAMILNADU. (To
CHINA. Australia - China Life Science Summit. . Bio & Pharma Logistics
 CHINA. Bio & Pharma Logistics Andrew McDonald Operations Director, Asia Pacific Melbourne, Australia, 27th March 2013 Australia - China Life Science Summit Responsibilities Who takes responsibility on
CHINA. Bio & Pharma Logistics Andrew McDonald Operations Director, Asia Pacific Melbourne, Australia, 27th March 2013 Australia - China Life Science Summit Responsibilities Who takes responsibility on
Gas Generator ULTRA HIGH PURE - HIGH FLOW. Gas Generator
 Gas Generator ULTRA HIGH PURE - HIGH FLOW Gas Generator Zero Air Generator for GC Zero Air Generator produces a continuous flow of high purity Zero Air at selected pressure. The modular pressure swing
Gas Generator ULTRA HIGH PURE - HIGH FLOW Gas Generator Zero Air Generator for GC Zero Air Generator produces a continuous flow of high purity Zero Air at selected pressure. The modular pressure swing
Our manufacturing facility is designed to comply in accordance to the stringent Schedule M GMP Guidelines.
 Dales Remedies Pvt Ltd was founded in 1994 as a pioneer in the manufacture of wide range of pharmaceutical formulations such as creams, gels, ointments, lotions and mouthwash. Currently we have a separate
Dales Remedies Pvt Ltd was founded in 1994 as a pioneer in the manufacture of wide range of pharmaceutical formulations such as creams, gels, ointments, lotions and mouthwash. Currently we have a separate
 I. DEFINITION This training under Kilimanjaro School of Pharmacy Industrial Pharmacy Teaching Unit (IPTU) is an Advanced Training Program in Drug Development, Drug Manufacturing, Regulatory & Quality Compliance.
I. DEFINITION This training under Kilimanjaro School of Pharmacy Industrial Pharmacy Teaching Unit (IPTU) is an Advanced Training Program in Drug Development, Drug Manufacturing, Regulatory & Quality Compliance.
International Water Standards
 International Water Standards How they effect the design of a water treatment Dr. Stephan Krietemeyer Lauer Membran Wassertechnik GmbH Industriegebiet Speichermatt D 79599 Wittlingen 1 //KR International
International Water Standards How they effect the design of a water treatment Dr. Stephan Krietemeyer Lauer Membran Wassertechnik GmbH Industriegebiet Speichermatt D 79599 Wittlingen 1 //KR International
Product Permission Document (PPD) of Botulinum Toxin Type A for Injection Ph.Eur Purified Neurotoxin Complex
 Product Permission Document (PPD) of Botulinum Toxin Type A for Injection Ph.Eur Purified Neurotoxin Complex Brand Name : BOTO GENIE 1. Introduction : BOTO GENIE (Botulinum Toxin Type A for Injection Ph.Eur)
Product Permission Document (PPD) of Botulinum Toxin Type A for Injection Ph.Eur Purified Neurotoxin Complex Brand Name : BOTO GENIE 1. Introduction : BOTO GENIE (Botulinum Toxin Type A for Injection Ph.Eur)
Application of Quality Risk Management Tools for Cell Therapy Manufacturing
 Application of Quality Risk Management Tools for Cell Therapy Manufacturing 17 th ISCT Annual Meeting May 20, 2011 Jean Stanton Associate Director, Product Quality Management Janssen Supply Chain Conflict/Disclaimer
Application of Quality Risk Management Tools for Cell Therapy Manufacturing 17 th ISCT Annual Meeting May 20, 2011 Jean Stanton Associate Director, Product Quality Management Janssen Supply Chain Conflict/Disclaimer
FDA s Guidance for Industry
 Sterile Drug Products Produced by Aseptic Processing - CGMPs Midwest FDC Conference November 4 2005 Susan Bruederle, Investigator FDA / ORA / Central Region Chicago District / Hinsdale IL FDA s Guidance
Sterile Drug Products Produced by Aseptic Processing - CGMPs Midwest FDC Conference November 4 2005 Susan Bruederle, Investigator FDA / ORA / Central Region Chicago District / Hinsdale IL FDA s Guidance
Aseptic Processing Current Issues & Trends
 Aseptic Processing Current Issues & Trends 1 Introduction Richard M. Johnson Member, PDA for 20+ years President & CEO since 2009 Ladies and Gentlemen, I am happy to be here with you. Senhoras e Senhores,
Aseptic Processing Current Issues & Trends 1 Introduction Richard M. Johnson Member, PDA for 20+ years President & CEO since 2009 Ladies and Gentlemen, I am happy to be here with you. Senhoras e Senhores,
Situation as of November 2016
 Situation as of November 2016 - - The FAO/GIEWS Country Cereal Balance System (CCBS) is a database of annual supply and utilization balances for main cereals, covering all countries of the world. It has
Situation as of November 2016 - - The FAO/GIEWS Country Cereal Balance System (CCBS) is a database of annual supply and utilization balances for main cereals, covering all countries of the world. It has
Product Questionnaire
 Product Questionnaire Let us help you find the SOLUTION for your product needs This product questionnaire has been designed to help you initiate a successful transfer of your product/process to an appropriate
Product Questionnaire Let us help you find the SOLUTION for your product needs This product questionnaire has been designed to help you initiate a successful transfer of your product/process to an appropriate
Isolators v. RABS: Facility Design Considerations for a Fill-Finish Finish Suite
 APV 2008 Basle Conference Basle, Switzerland: May 28 & 29 2008 Isolators v. RABS: Facility Design Considerations for a Fill-Finish Finish Suite John R. Chester Principal Engineer Global Pharmaceutical
APV 2008 Basle Conference Basle, Switzerland: May 28 & 29 2008 Isolators v. RABS: Facility Design Considerations for a Fill-Finish Finish Suite John R. Chester Principal Engineer Global Pharmaceutical
Guiding Principles for the implementation of fluid management technologies for modern single use aseptic processing
 Guiding Principles for the implementation of fluid management technologies for modern single use aseptic processing Jean-Marc Cappia, Vice President Marketing Fluid Management Technologies Agenda 1. Aseptic
Guiding Principles for the implementation of fluid management technologies for modern single use aseptic processing Jean-Marc Cappia, Vice President Marketing Fluid Management Technologies Agenda 1. Aseptic
Isolator Technology TRUKING TECHNOLOGY LIMITED
 Isolator TRUKING TECHNOLOGY LIMITED Add No.1 xinkang Road,Yutan Town,Ningxiang,Changsha,China P.C 410600 Tel 86 731-87938222 87938255 Fax 86 731-879338201 Web www.truking.cn Any technical renewal will
Isolator TRUKING TECHNOLOGY LIMITED Add No.1 xinkang Road,Yutan Town,Ningxiang,Changsha,China P.C 410600 Tel 86 731-87938222 87938255 Fax 86 731-879338201 Web www.truking.cn Any technical renewal will
