Import Health Standard
|
|
|
- Cecily Murphy
- 6 years ago
- Views:
Transcription
1 Import Health Standard Hides and Skins HIDESKIN.ALL 7 August 2015 An import health standard issued under the Biosecurity Act 1993
2 TITLE Import Health Standard: Hides and Skins COMMENCEMENT This Import Health Standard comes into force on 7 August REVOCATION This Import Health Standard revokes and replaces Hides and Skins (HIDCOMIC.ALL dated 30 June 2011). ISSUING AUTHORITY This Import Health Standard is issued under section 24A of the Biosecurity Act Dated at Wellington this 7 th day of August 2015 Howard Pharo Manager, Import and Export Animals Ministry for Primary Industries (acting under delegated authority of the Director General) Contact for further information Ministry for Primary Industries (MPI) Regulation and Assurance Branch Animal Imports PO Box 2526 Wellington animalimports@mpi.govt.nz Ministry for Primary Industries Page 1 of 14
3 Contents Page Introduction 3 Part 1: General Requirements Application Incorporation of material by reference Definitions Packaging Import permit The documentation that must accompany goods Transitional facility Biosecurity clearance 6 Part 2: Specified Requirements for Identified Risk Organisms Fully tanned hides and skins Processed hides and skins Salted or dried hides and skins from specified animals Casualty (slink) skins Macropod hides and skins New Zealand returning hides and skins 9 Schedule 1 Document History 11 Schedule 2 Definitions 12 Schedule 3 Processing Requirements at the Transitional Facility 14 Ministry for Primary Industries Page 2 of 14
4 Introduction This introduction is not part of the legal requirements of this import health standard (IHS), but is intended to indicate its general effect. Purpose (1) This IHS specifies the minimum requirements that must be met when importing hides and skins into New Zealand. Background (1) The Biosecurity Act 1993 (the Act) provides the legal basis for excluding, eradicating and effectively managing pests and unwanted organisms. (2) Import health standards issued under the Act set out requirements to be met to effectively manage biosecurity risks associated with importing goods. They include requirements that must be met in the exporting country, during transit, and during importation, before biosecurity clearance can be given. (3) A guidance document accompanies this IHS providing information on how the requirements may be met. Who should read this import health standard? (1) This IHS applies to importers of eligible consignments of hides and skins. Why is this important? (1) It is the importer s responsibility to ensure the requirements of this IHS are met. Consignments that do not comply with the requirements of this IHS may not be cleared for entry into New Zealand and/or further information may be sought from importers. Consignments that do not comply with the requirements of this IHS may be re-shipped or destroyed under the Act or treated in accordance with this IHS prior to release or equivalence determined. Importers are liable for all associated expenses. See guidance document for more information about importer responsibilities. Equivalence (1) The Chief Technical Officer (CTO) may approve measures under section 27(1)(d) of the Act, different from those set out in this IHS, that may be applied to effectively manage risks associated with the importation of these goods. If an equivalent measure is approved an import permit may be issued under section 24D(2) of the Act, if the Director-General considers it appropriate to do so. See guidance document for more information about equivalence and permits. Document history (1) Refer to Schedule 1. Other information (1) This is not an exhaustive list of compliance requirements and it is the importer s responsibility to be familiar with and comply with all New Zealand laws. See guidance document for more information about inspection and verification of consignments. Ministry for Primary Industries Page 3 of 14
5 Part 1: General Requirements 1.1 Application (1) This import health standard (IHS) applies to: a) Fully tanned hides and skins. b) Processed hides and skins. c) Salted or dried hides and skins. d) Casualty skins. e) Macropod hides and skins. (2) The following products are not eligible for importation under this IHS: a) Manufactured products containing fully tanned hides and skins (see IHS: Ornamental Products of Animal Origin). b) Rawhide articles and handicrafts (see IHS: Ornamental Products of Animal Origin). c) Animal fibre (see IHS: Animal Fibre). 1.2 Incorporation of material by reference (1) The following international standards are incorporated by reference in this IHS under section 142M of the Act: a) The OIE Terrestrial Animal Health Code (the Code). (2) The following material is incorporated by reference in this IHS under section 142M of the Act: a) MPI Approved Diagnostic Tests, Vaccines, Treatments and Post-arrival Testing Laboratories for Animal Import Health Standards (MPI-STD-TVTL). b) MPI List of FMD-Free Countries and Zones. (3) Under section 142O(3) of the Act it is declared that section 142O(1) does not apply, that is, a notice under section 142O(2) of the Act is not required to be published before material that amends or replaces the above listed standards, guideline or lists has legal effect as part of these documents. See guidance document for more information about incorporation by reference and section 142O(1). 1.3 Definitions (1) For the purposes of this standard and the attached guidance document, terms used that are defined in the Act have the meanings set out there. The Act is available at the following website: (2) Refer to Schedule 2 for additional definitions that apply. 1.4 Packaging (1) The consignment must be clearly identified and identifiable to the veterinary certificate. (2) The packaging of all consignments of hides and skins must be clean and secure. (3) The criteria to be applied to determine whether the outcome in clause (2) has been achieved are as follows: a) The exterior of the package is free from any organic contaminants. b) The hides and skins are adequately contained. Ministry for Primary Industries Page 4 of 14
6 1.5 Import permit (1) An import permit under section 24D of the Act is required if: a) It is required in the IHS. b) Prior to import, a CTO has approved an equivalent measure under section 27(1)(d) of the Act that is different from those in this IHS that may be applied to effectively manage risks. (2) A permit is not required if, prior to import, a CTO has approved an equivalent measure under section 27(1)(d) of the Act that is different from those in this IHS in the form of a negotiated veterinary certificate. See guidance document for more information about applying for a permit, equivalence and countryspecific veterinary certificates. 1.6 The documentation that must accompany goods (1) The consignment must arrive in New Zealand with the following: a) An import permit (copy acceptable) where required. The importer must supply the following information to obtain a permit: i) The name and address of exporter. ii) The type of product. iii) The name and address of the New Zealand importer. iv) The name and address of the transitional facility in New Zealand to which the consignment is to proceed following importation. b) A veterinary certificate where required in this IHS, that must include the following: i) A unique consignment identifier. ii) The description, species, and amount of product. iii) The country of origin (or zone as defined by the Code). iv) The name and address of the importer (consignee) and exporter (consignor). v) The name, signature and contact details of the Official Veterinarian. vi) Certification and endorsement by the Official Veterinarian that the general requirements outlined in Part 1 of this IHS have been met. vii) Certification and endorsement by the Official Veterinarian that the specified requirements outlined in Part 2 of this IHS have been met. c) A manufacturer s declaration where required in this IHS. The manufacturer s declaration must: i) Be prepared by the manufacturer on letterhead paper. ii) Contain the correct statements as required by the import requirements specific to the product. iii) Be signed and dated by the quality manager or equivalent. (2) Where equivalent measures have been negotiated and agreed with MPI, and a CTO has, prior to import, approved an equivalent measure under section 27(1)(d) of the Act that is different from those in this standard in the form of a negotiated veterinary certificate, a country-specific veterinary certificate must accompany the consignment. See guidance document for more information about country-specific veterinary certificates that have been agreed for trade and a model veterinary certificates. (3) All documents must: a) Be original, unless otherwise stated. b) Accompany the imported goods. c) Be in English or have an English translation that is clear and legible. Ministry for Primary Industries Page 5 of 14
7 d) Be endorsed on every page by the Official Veterinarian with their original stamp, signature and date or be endorsed in the space allocated and all pages have paper based alternative security features. 1.7 Transitional facility (1) Following biosecurity authorisation being given, the hides and skins (where applicable) must proceed directly to the transitional facility named on the import permit. (2) The consignment must meet all the requirements noted in the General Requirements in Part 1, and must be processed to the requirements in Part 2 (Specified Requirements) and in Schedule 3 Processing Requirements at the Transitional Facility, of this IHS in order to be eligible for biosecurity clearance. 1.8 Biosecurity clearance (1) A biosecurity clearance, under section 26 of the Act, may be issued when the hides and skins meet all the requirements of this IHS, provided the applicable requirements of section 27 in the Act are met. Ministry for Primary Industries Page 6 of 14
8 Part 2: Specified Requirements for Identified Risk Organisms See guidance document for more information about hides and skins samples. 2.1 Fully tanned hides and skins Documentation (1) No documentation is required for fully tanned hides and skins Requirements (1) Consignments of fully tanned hides and skins from any species may be imported from any country without specific requirements. 2.2 Processed hides and skins Documentation (1) Consignments of processed hides and skins must be accompanied by the following documentation: a) A veterinary certificate certifying that the processed hides and skins meet the requirements in clause of this IHS; or b) A manufacturer s declaration that the processed hides and skins meet the requirements in clause of this IHS Requirements (1) Consignments of processed hides and skins must be treated by: a) A liming process at a ph equal to or greater than 12.5; and/or b) A pickling process in a solution of salt and mineral acid at a ph equal to or less than Salted or dried hides and skins from specified animals Important information for importers CTO [B] gives direction to replace the MPI List of Foot & Mouth Disease (FMD) Free Countries with the OIE List of FMD Free Member Countries for clearance of specified products. For the purposes of this IHS, FMD free includes countries listed in these sections: FMD free where vaccination is not practised; and FMD free where vaccination is practised. It does not include these sections: FMD free zone where vaccination is not practised; or FMD free zone where vaccination is practised. Ministry for Primary Industries Page 7 of 14
9 2.3.1 Documentation (1) Consignments of salted or dried hides and skins from specified animals (see Schedule 2 for definition) must be accompanied by the following documentation: a) A valid import permit. b) A veterinary certificate certifying that the salted or dried hides and skins meet the requirements in clauses 2.3.2(1) and (2) of this IHS. c) A manufacturer s declaration stating that the salted or dried hides and skins have been packaged in new, clean, and secure packaging that is free from any extraneous contamination Requirements (1) Consignments of salted or dried hides and skins must originate from animals that have been slaughtered in a government licensed processing establishment. (2) Consignments of salted or dried hides and skins from specified animals must meet the following requirements for the specified risk organisms: a) Anthrax requirements: i) Originate from a country or zone officially free from anthrax; or ii) Originate from establishments that were not subject to restrictions imposed for the control of anthrax; or iii) Originate from animals that have been slaughtered in a government-licensed processing establishment under the direct supervision of an official veterinarian, subjected to anteand post-mortem inspection, and certified as fit for human consumption. b) Foot and mouth disease (FMD) requirements: i) Originate from a country or zone that is officially free from FMD in accordance with the Code and as referenced in the MPI List of FMD-Free Countries or Zones; or ii) Treated for 28 days with salt containing 2% sodium carbonate. (3) Salted or dried hides and skins must be directed on arrival by an inspector to the transitional facility listed on the import permit. While in the transitional facility the hides and skins must be processed in accordance with Schedule 3 of this IHS. 2.4 Casualty (slink) skins Documentation (1) Consignments of casualty (slink) skins (see Schedule 2 for definition) must be accompanied by the following documentation: a) A valid import permit. b) A veterinary certificate certifying that the casualty skins meet the requirements in clause 2.4.2(1) of this IHS. c) A manufacturer s declaration stating that the casualty skins have been packaged in new, clean, and secure packaging that is free from any extraneous contamination Requirements (1) Consignments of casualty (slink) skins must meet the following requirements for the specified risk organisms: a) Anthrax requirements: i) Originate from a country or zone officially free from anthrax; or ii) Originate from establishments that were not subject to restrictions imposed for the control of anthrax. Ministry for Primary Industries Page 8 of 14
10 b) FMD requirements: i) Originate from a country or zone that is officially free from FMD in accordance with the Code and as referenced in the MPI List of FMD-Free Countries or Zones; or ii) Treated for 28 days with salt containing 2% sodium carbonate. (2) Casualty skins must be directed on arrival by an inspector to the transitional facility listed on the import permit. While in the transitional facility the skins must be processed in accordance with Schedule 3 of this IHS. 2.5 Macropod hides and skins Documentation (1) Consignments of macropod hides and skins (see Schedule 2 for definition) must be accompanied by the following documentation: a) A valid import permit. b) A veterinary certificate certifying that the macropod hides and skins meet the requirements in clause 2.5.2(3) of this IHS. c) A manufacturer s declaration stating that the macropod hides and skins have been packaged in new, clean, and secure packaging that is free from any extraneous contamination Requirements (1) Consignments of macropod hides and skins may only be imported from Australia. (2) Consignments of macropod hides and skins may be dried, salted, fresh, frozen or chilled. (3) Consignments of macropod hides and skins must meet the following anthrax requirements for the specified risk organisms: a) Originate from a zone officially free from anthrax; or b) Originate from establishments that were not subject to restrictions imposed for the control of anthrax; or c) Originate from animals that have been processed under the Australian Standard for the Hygienic Production of Game Meat for Human Consumption, and have passed a post-mortem inspection in a government-licensed processing establishment. (4) Macropod hides and skins must be directed on arrival by an inspector to the transitional facility listed on their import permit. While in the transitional facility the skins must be processed in accordance with Schedule 3 of this IHS. 2.6 New Zealand returning hides and skins Important information for importers and MPI Border 28 April 2017 Clause 2.6 in relation to New Zealand returning hides and skins has been revoked. This clause is replaced by IHS: Returned New Zealand Animal Products (RETURNAP.ALL, dated 26 April 2017), which is available at: Documentation (1) Consignments of New Zealand returning hides and skins must be accompanied by documentation which identifies the hides and skins to be of New Zealand origin. Ministry for Primary Industries Page 9 of 14
11 2.6.2 Requirements (1) All consignments New Zealand returning hides and skins is eligible for import provided that the product is: a) Identifiable as being of New Zealand origin. b) Packed in the original packaging. c) Free from extraneous organic contamination. Ministry for Primary Industries Page 10 of 14
12 Schedule 1 Document History Date First Issued Title Shortcode 30 June 2011 Hides and Skins HIDCOMIC.ALL Date of Issued Amendments Title Shortcode 7 August 2015 Hides and Skins HIDESKIN.ALL Ministry for Primary Industries Page 11 of 14
13 Schedule 2 Definitions Casualty Skin A skin derived from a lamb or calf up to the age of 6 months that has died of natural causes, also known as a slink skin. Competent Authority The Veterinary or other Governmental Authority of an OIE Member, that has the responsibility and competence for ensuring or supervising the implementation of animal health and welfare measures, international veterinary certification and other standards and recommendations in the Code in the whole territory. CTO Direction Chief Technical Officer (CTO) Direction - equivalent measures recorded by number under section 27(1)d(iii) of the Act, to enable border staff to clear the goods and record the number in the MPI database. Fully Tanned Hides and Skins Hides and skins that have undergone a full tanning process so that they are preserved from putrescence and are thermally stable and are ready for use in a manufactured product. Hides and skins can be tanned using a variety of methods. The many processes involved in tanning are explained in MPI s risk analysis document for hides and skins ( See also wet blues, browns and whites below. Import Permit A permit issued by the Director-General of MPI pursuant to section 24D(2) of the Act. Macropod A species from the marsupial family Macropodidiae which includes kangaroos, wallabies and wallaroos. MPI Ministry for Primary Industries. Official Veterinarian A veterinarian authorised by the Competent Authority of the country to perform certain designated official tasks associated with animal health and/or public health and inspections of commodities and, when appropriate, to certify in conformity with the provisions of the OIE Code Chapter for certification procedures. OIE The World Organisation for Animal Health. Pickling The process of soaking a hide or skin in an acid based solution in preparation for tanning. Processed Hides and Skins Hides and skins that have been pickled and/or limed, this includes wet whites, wet blues, and wet browns. Ministry for Primary Industries Page 12 of 14
14 Salted or Dried Hides and Skins from Specified Animals Hides and skins that have been air dried or dry/wet salted derived from the following farmed animals: alpacas, buffalo, cattle, deer, emus, goats, horses, llamas, ostriches, pigs, and sheep. The Code The OIE Terrestrial Animal Health Code as found on the OIE website. Veterinary Certificate A certificate, issued in conformity with the provisions of the Code Chapter for certification procedures, describing the animal health and/or public health requirements which are fulfilled by the exported commodities. Wet Blue Wet blue hides have been tanned with a chromium salt, which imparts its characteristic colour. It is thermally stable but not usable in this form, requiring further tanning (retanning) before being suitable for end-use. Wet Brown Wet brown hides have been tanned with a vegetable tanning agent or agents, which imparts its characteristic colour. It is thermally stable but not usable in this form, requiring further tanning (retanning) before being suitable for end-use. Wet White Wet white hides have been tanned with various organic and/or inorganic agents, which leave it white in colour. It is thermally stable but not usable in this form, requiring further tanning (retanning) before being suitable for end-use. Ministry for Primary Industries Page 13 of 14
15 Schedule 3 Processing Requirements at the Transitional Facility (1) This schedule applies to consignments of hides and skins imported to New Zealand that have been authorised to be moved for further processing at the transitional facility listed on the import permit. (2) While in the transitional facility, and prior to processing, imported hides and skins must be clearly marked, stored, and handled in accordance with the MPI Standard for General Transitional Facilities for Uncleared Goods, MPI-STD-TFGEN, issued under section 39(1) of the Act (or any standard that replaces that standard), and any associated requirements listed in Annex F of the guidance document to that standard. (3) While in the transitional facility, the facility operator must ensure that the hides and skins be processed (limed and/or pickled) in accordance with clause of this IHS. (4) The facility operator must ensure that all trimmings and solid waste associated with hides and skins are treated, destroyed or disposed of at an approved transitional facility by: a) Rendering; or b) Incineration. (5) The facility operator must ensure that all packaging that has been in contact with hides and skins is destroyed or disposed of at an approved transitional facility by: a) Incineration. (6) The facility operator must ensure that all effluent generated during processing is: a) Discharged into a municipal sewage system; or b) Discharged into a securely fenced and isolated soak-pit; or c) Treated with a biocide that is listed in the standard known as MPI Approved Diagnostic Tests, Vaccines, Treatments and Post-Arrival Testing Laboratories for Animal Import Health Standards (MPI-STD-TVTL). Ministry for Primary Industries Page 14 of 14
IMPORT HEALTH STANDARD FOR MILK AND MILK PRODUCTS FOR HUMAN CONSUMPTION FROM THE EUROPEAN UNION AND SWITZERLAND
 IMPORT HEALTH STANDARD FOR MILK AND MILK PRODUCTS FOR HUMAN CONSUMPTION FROM THE EUROPEAN UNION AND SWITZERLAND Issued pursuant to Section 22 of the Biosecurity Act 1993 Dated: 19 July 2010 Important information
IMPORT HEALTH STANDARD FOR MILK AND MILK PRODUCTS FOR HUMAN CONSUMPTION FROM THE EUROPEAN UNION AND SWITZERLAND Issued pursuant to Section 22 of the Biosecurity Act 1993 Dated: 19 July 2010 Important information
Import Health Standard
 Import Health Standard Oncidium Cut Flowers MPI.IHS.CFF.ONCIDIUM 21 December 2017 Issued under the Biosecurity Act 1993 TITLE Import Health Standard: Oncidium Cut Flowers COMMENCEMENT This Import Health
Import Health Standard Oncidium Cut Flowers MPI.IHS.CFF.ONCIDIUM 21 December 2017 Issued under the Biosecurity Act 1993 TITLE Import Health Standard: Oncidium Cut Flowers COMMENCEMENT This Import Health
IMPORT HEALTH STANDARD FOR THE IMPORTATION INTO NEW ZEALAND OF DAIRY PRODUCTS AND PRODUCTS CONTAINING DAIRY INGREDIENTS FROM SPECIFIED COUNTRIES
 IMPORT HEALTH STANDARD FOR THE IMPORTATION INTO NEW ZEALAND OF DAIRY PRODUCTS AND PRODUCTS CONTAINING DAIRY INGREDIENTS FROM SPECIFIED COUNTRIES Issued pursuant to Section 22 of the Biosecurity Act 1993
IMPORT HEALTH STANDARD FOR THE IMPORTATION INTO NEW ZEALAND OF DAIRY PRODUCTS AND PRODUCTS CONTAINING DAIRY INGREDIENTS FROM SPECIFIED COUNTRIES Issued pursuant to Section 22 of the Biosecurity Act 1993
Overseas Market Access Requirements Notification - Animal Products Act 1999
 Overseas Market Access Requirements Notification - Animal Products Act 1999 Regulation and Assurance Branch, Animal and Animal Products Directorate, Ministry for Primary Industries Ref: AE-PH-25 Date:
Overseas Market Access Requirements Notification - Animal Products Act 1999 Regulation and Assurance Branch, Animal and Animal Products Directorate, Ministry for Primary Industries Ref: AE-PH-25 Date:
Import Health Standard
 Import Health Standard Specified Foods for Human Consumption Containing Animal Products EDIPROIC.ALL Issued under the Biosecurity Act 1993 TITLE Import Health Standard: Specified Foods for Human Consumption
Import Health Standard Specified Foods for Human Consumption Containing Animal Products EDIPROIC.ALL Issued under the Biosecurity Act 1993 TITLE Import Health Standard: Specified Foods for Human Consumption
Animal Products Notice
 Animal Products Notice OMAR- Animal Products to Canada.. An animal products notice issued under the Animal Products Act 1999 TITLE Animal Products Notice: OMAR- Animal Products to Canada COMMENCEMENT This
Animal Products Notice OMAR- Animal Products to Canada.. An animal products notice issued under the Animal Products Act 1999 TITLE Animal Products Notice: OMAR- Animal Products to Canada COMMENCEMENT This
Import Health Standard
 Import Health Standard Sea Containers SEACO CTO Directions for Sea Containers from Italy Additional CTO requirements were issued 22 December 2017, 19 January 2018 and 2 March 2018 for all sea containers
Import Health Standard Sea Containers SEACO CTO Directions for Sea Containers from Italy Additional CTO requirements were issued 22 December 2017, 19 January 2018 and 2 March 2018 for all sea containers
Guidance Document. Meeting Requirements as a Registered Food Importer. A guidance document issued by the Ministry for Primary Industries
 Guidance Document Meeting Requirements as a Registered Food Importer. 23 February 2016 A guidance document issued by the Ministry for Primary Industries Title Meeting Requirements as a Registered Food
Guidance Document Meeting Requirements as a Registered Food Importer. 23 February 2016 A guidance document issued by the Ministry for Primary Industries Title Meeting Requirements as a Registered Food
Animal Products (Transport of Export Animal Products and Handling at Point of Export) Notice 2009
 Animal Products (Transport of Export Animal Products and Handling at Point of Export) Notice 2009 Pursuant to sections 60(1)(a) and 60(1)(c) of the Animal Products Act 1999, I, Carol Barnao, Director (Standards)
Animal Products (Transport of Export Animal Products and Handling at Point of Export) Notice 2009 Pursuant to sections 60(1)(a) and 60(1)(c) of the Animal Products Act 1999, I, Carol Barnao, Director (Standards)
Animal Products (Export Requirements for Branding, Marking and Security Devices) Notice 2012
 Animal Products (Export Requirements for Branding, Marking and Security Devices) Notice 2012 Pursuant to section 60 of the Animal Products Act 1999, I, Paul Dansted, Manager Food Assurance issue the following
Animal Products (Export Requirements for Branding, Marking and Security Devices) Notice 2012 Pursuant to section 60 of the Animal Products Act 1999, I, Paul Dansted, Manager Food Assurance issue the following
IMPORT HEALTH STANDARD FOR SEA CONTAINERS FROM ALL COUNTRIES
 IMPORT HEALTH STANDARD FOR SEA CONTAINERS FROM ALL COUNTRIES New Zealand MAF Biosecurity Authority Standard: BMG-STD-SEACO SEPTEMBER 2003 TABLE OF CONTENTS 1. Introduction 2. Review 3. Definitions 4. Information
IMPORT HEALTH STANDARD FOR SEA CONTAINERS FROM ALL COUNTRIES New Zealand MAF Biosecurity Authority Standard: BMG-STD-SEACO SEPTEMBER 2003 TABLE OF CONTENTS 1. Introduction 2. Review 3. Definitions 4. Information
ACVM Requirement. Registration Information Requirements
 ACVM Requirement Registration Information Requirements Information required to register (or vary a registration of) a trade name product under the Agricultural Compounds and Veterinary Medicines Act 1997
ACVM Requirement Registration Information Requirements Information required to register (or vary a registration of) a trade name product under the Agricultural Compounds and Veterinary Medicines Act 1997
Import Health Standard
 Import Health Standard Fertilisers and Growing Media of Plant Origin MPI.STD.FERTGRO Issued under the Biosecurity Act 1993 TITLE Import Health Standard: Fertilisers and Growing Media of Plant Origin COMMENCEMENT
Import Health Standard Fertilisers and Growing Media of Plant Origin MPI.STD.FERTGRO Issued under the Biosecurity Act 1993 TITLE Import Health Standard: Fertilisers and Growing Media of Plant Origin COMMENCEMENT
Import Health Standard
 Import Health Standard Vehicles Machinery and Tyres.. An import health standard issued under the Biosecurity Act 1993 TITLE Import Health Standard: Vehicles Machinery and Tyres COMMENCEMENT This Import
Import Health Standard Vehicles Machinery and Tyres.. An import health standard issued under the Biosecurity Act 1993 TITLE Import Health Standard: Vehicles Machinery and Tyres COMMENCEMENT This Import
Food and Agricultural Import Regulations and Standards - Certification
 THIS REPORT CONTAINS ASSESSMENTS OF COMMODITY AND TRADE ISSUES MADE BY USDA STAFF AND NOT NECESSARILY STATEMENTS OF OFFICIAL U.S. GOVERNMENT POLICY Required Report - public distribution Date: 12/10/2010
THIS REPORT CONTAINS ASSESSMENTS OF COMMODITY AND TRADE ISSUES MADE BY USDA STAFF AND NOT NECESSARILY STATEMENTS OF OFFICIAL U.S. GOVERNMENT POLICY Required Report - public distribution Date: 12/10/2010
STAATSKOERANT, 1 DESEMBER 2017 No DEPARTMENT OF TRADE AND INDUSTRY NO DECEMBER 2017
 1329 National Regulator for Compulsory Specifications Act (5/2008), as amended: Amendment of the Compulsory Specification for canned fish, canned marine molluscs and canned crustaceans and products derived
1329 National Regulator for Compulsory Specifications Act (5/2008), as amended: Amendment of the Compulsory Specification for canned fish, canned marine molluscs and canned crustaceans and products derived
CLASSIFICATION OF NON-TARIFF MEASURES FEBRUARY 2012 VERSION
 CLASSIFICATION OF NON-TARIFF MEASURES FEBRUARY 2012 VERSION A SANITARY AND PHYTOSANITARY MEASURES Measures that are applied: to protect human or animal life from risks arising from additives, contaminants,
CLASSIFICATION OF NON-TARIFF MEASURES FEBRUARY 2012 VERSION A SANITARY AND PHYTOSANITARY MEASURES Measures that are applied: to protect human or animal life from risks arising from additives, contaminants,
ACVM Notice. Agricultural Compounds Exempt from Registration. Requirements for conditions of exemption. 1 August 2017
 ACVM Notice Agricultural Compounds Exempt from Registration Requirements for conditions of exemption Issued under the Agricultural Compounds and Veterinary Medicines Act 1997 TITLE ACVM Notice: Agricultural
ACVM Notice Agricultural Compounds Exempt from Registration Requirements for conditions of exemption Issued under the Agricultural Compounds and Veterinary Medicines Act 1997 TITLE ACVM Notice: Agricultural
Animal Products (Export Requirements Dairy Products) Notice 2005
 Animal Products (Export Requirements Dairy Products) Notice 2005 Pursuant to section 60 of the Animal Products Act 1999, I, Carol Barnao, Director (Dairy & Plant Products), issue the following Notice for
Animal Products (Export Requirements Dairy Products) Notice 2005 Pursuant to section 60 of the Animal Products Act 1999, I, Carol Barnao, Director (Dairy & Plant Products), issue the following Notice for
(Non-legislative acts) REGULATIONS
 26.2.2011 Official Journal of the European Union L 54/1 II (Non-legislative acts) REGULATIONS COMMISSION REGULATION (EU) No 142/2011 of 25 February 2011 implementing Regulation (EC) No 1069/2009 of the
26.2.2011 Official Journal of the European Union L 54/1 II (Non-legislative acts) REGULATIONS COMMISSION REGULATION (EU) No 142/2011 of 25 February 2011 implementing Regulation (EC) No 1069/2009 of the
COMMISSION REGULATION (EC)
 L 313/40 Official Journal of the European Union 28.11.2009 COMMISSION REGULATION (EC) No 1152/2009 of 27 November 2009 imposing special conditions governing the import of certain foodstuffs from certain
L 313/40 Official Journal of the European Union 28.11.2009 COMMISSION REGULATION (EC) No 1152/2009 of 27 November 2009 imposing special conditions governing the import of certain foodstuffs from certain
RESOLUTION # 5 LIVESTOCK IN NEW JERSEY AGRICULTURE
 RESOLUTION # 5 LIVESTOCK IN NEW JERSEY AGRICULTURE 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 WHEREAS, livestock has been a part of New Jersey agriculture from the state s earliest days as
RESOLUTION # 5 LIVESTOCK IN NEW JERSEY AGRICULTURE 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 WHEREAS, livestock has been a part of New Jersey agriculture from the state s earliest days as
CIRCULAR GUIDING ON THE FOOD HYGIENE AND SAFETY CONTROL FOR IMPORTED FOODSTUFFS OF ANIMAL ORIGIN
 Unofficial translation MINISTRY OF AGRICULTURE AND RURAL DEVELOPMENT No. 25/2010/TT-BNNPTNT SOCIALIST REPUBLIC OF VIET NAM Independence - Freedom Happiness Hanoi, April 8, 2010 CIRCULAR GUIDING ON THE
Unofficial translation MINISTRY OF AGRICULTURE AND RURAL DEVELOPMENT No. 25/2010/TT-BNNPTNT SOCIALIST REPUBLIC OF VIET NAM Independence - Freedom Happiness Hanoi, April 8, 2010 CIRCULAR GUIDING ON THE
Animal Products Notice
 Animal Products Notice Specifications for Products Intended for Human Consumption.. An animal products notice issued under the Animal Products Act 1999 TITLE Animal Products Notice: Specifications for
Animal Products Notice Specifications for Products Intended for Human Consumption.. An animal products notice issued under the Animal Products Act 1999 TITLE Animal Products Notice: Specifications for
INFECTIOUS MYONECROSIS
 CHAPTER 9.4. INFECTIOUS MYONECROSIS Article 9.4.1. For the purposes of the Aquatic Code, infectious myonecrosis (IMN) means infection with infectious myonecrosis virus (IMNV). This virus is similar to
CHAPTER 9.4. INFECTIOUS MYONECROSIS Article 9.4.1. For the purposes of the Aquatic Code, infectious myonecrosis (IMN) means infection with infectious myonecrosis virus (IMNV). This virus is similar to
Implementing Procedures
 Implementing Procedures 1. Certificate of origin Rule 1 Document (a) A certificate of origin should be on ISO A4 size paper in conformity with the format shown in Appendix 1. It should be completed in
Implementing Procedures 1. Certificate of origin Rule 1 Document (a) A certificate of origin should be on ISO A4 size paper in conformity with the format shown in Appendix 1. It should be completed in
Guidance Document. RMP Template for Farm Dairies Export Eligible Milk. 11 December 2015
 Guidance Document RMP Template for Farm Dairies Export Eligible Milk 11 December 2015 A guidance document issued by the Ministry for Primary Industries Title Guidance Document: RMP Template for Farm Dairies
Guidance Document RMP Template for Farm Dairies Export Eligible Milk 11 December 2015 A guidance document issued by the Ministry for Primary Industries Title Guidance Document: RMP Template for Farm Dairies
Guidance Document. Guidance for Risk Management Programme (RMP) Template for Dairy Processors. Liquid Milk, Domestic Supply.
 Guidance Document Guidance for Risk Management Programme (RMP) Template for Dairy Processors Liquid Milk, Domestic Supply 30 October 2015 A guidance document issued by the Ministry for Primary Industries
Guidance Document Guidance for Risk Management Programme (RMP) Template for Dairy Processors Liquid Milk, Domestic Supply 30 October 2015 A guidance document issued by the Ministry for Primary Industries
AUSTRALIAN MEDICAL DEVICES GUIDANCE DOCUMENT NUMBER 5. The Declaration of Conformity
 AUSTRALIAN MEDICAL DEVICES GUIDANCE DOCUMENT NUMBER 5 The Declaration of Conformity 30 October 2003 DISCLAIMER This document is provided for guidance only. It should not be relied upon to address every
AUSTRALIAN MEDICAL DEVICES GUIDANCE DOCUMENT NUMBER 5 The Declaration of Conformity 30 October 2003 DISCLAIMER This document is provided for guidance only. It should not be relied upon to address every
Post Entry Quarantine Facility Operator Training Resource
 Post Entry Quarantine Facility Operator Training Resource December 2016 Publisher Ministry for Primary Industries Pastoral House, 25 The Terrace PO Box 2526, Wellington, 6140, New Zealand Tel: 0800 00
Post Entry Quarantine Facility Operator Training Resource December 2016 Publisher Ministry for Primary Industries Pastoral House, 25 The Terrace PO Box 2526, Wellington, 6140, New Zealand Tel: 0800 00
Certificate of Area or Property Freedom Based on Monitoring by the Accrediting Authority ICA 23. ICA-23 Version 4.0 October 2004
 ICA 23 Certificate of Area or Property Freedom Based on Monitoring by the Accrediting Authority Version 4.0 October 2004 Controlled Copy # Signature Uncontrolled copies of this document are not subject
ICA 23 Certificate of Area or Property Freedom Based on Monitoring by the Accrediting Authority Version 4.0 October 2004 Controlled Copy # Signature Uncontrolled copies of this document are not subject
Guidance Document. Guidance for Risk Management Programme (RMP) Template for Dairy Processors - Cheese Domestic Supply
 Guidance Document Guidance for Risk Management Programme (RMP) Template for Dairy Processors - Cheese Domestic Supply. 23 May 2014 A guidance document issued by the Ministry for Primary Industries Title
Guidance Document Guidance for Risk Management Programme (RMP) Template for Dairy Processors - Cheese Domestic Supply. 23 May 2014 A guidance document issued by the Ministry for Primary Industries Title
Guidance Document. Guidance for Risk Management Programme (RMP) Template for Dairy Processors - Yoghurt Domestic Supply
 Guidance Document Guidance for Risk Management Programme (RMP) Template for Dairy Processors - Yoghurt Domestic Supply. 23 May 2014 A guidance document issued by the Ministry for Primary Industries Title
Guidance Document Guidance for Risk Management Programme (RMP) Template for Dairy Processors - Yoghurt Domestic Supply. 23 May 2014 A guidance document issued by the Ministry for Primary Industries Title
EURASIAN ECONOMIC COMMISSION COUNCIL
 EURASIAN ECONOMIC COMMISSION COUNCIL Working translation: DECISION October 9, 2014 No.94 Minsk On the Regulation on the Harmonized Procedure of Joint On-Site Inspections and of Taking Samples of Goods
EURASIAN ECONOMIC COMMISSION COUNCIL Working translation: DECISION October 9, 2014 No.94 Minsk On the Regulation on the Harmonized Procedure of Joint On-Site Inspections and of Taking Samples of Goods
Agenda Item 9 CX/FICS 07/16/9 September 2007
 Agenda Item 9 CX/FICS 07/16/9 September 2007 Background JOINT FAO/WHO FOOD STANDARDS PROGRAMME CODEX COMMITTEE ON FOOD IMPORT AND EXPORT INSPECTION AND CERTIFICATION SYSTEMS Sixteenth Session Surfer s
Agenda Item 9 CX/FICS 07/16/9 September 2007 Background JOINT FAO/WHO FOOD STANDARDS PROGRAMME CODEX COMMITTEE ON FOOD IMPORT AND EXPORT INSPECTION AND CERTIFICATION SYSTEMS Sixteenth Session Surfer s
National TFS Office, Dublin City Council [NTFSO] Guidance for Completing Notification Document (Annex 1A) & Movement Document (Annex 1B)
![National TFS Office, Dublin City Council [NTFSO] Guidance for Completing Notification Document (Annex 1A) & Movement Document (Annex 1B) National TFS Office, Dublin City Council [NTFSO] Guidance for Completing Notification Document (Annex 1A) & Movement Document (Annex 1B)](/thumbs/75/72492936.jpg) National TFS Office, Dublin City Council [NTFSO] Guidance for Completing Notification Document (Annex 1A) & Movement Document (Annex 1B) Introduction A planned shipment subject to the procedure of prior
National TFS Office, Dublin City Council [NTFSO] Guidance for Completing Notification Document (Annex 1A) & Movement Document (Annex 1B) Introduction A planned shipment subject to the procedure of prior
Plant Protection Rules 2031
 Plant Protection Rules 2031 In exercise of the powers conferred by Section 9 of the Plant Protection Act, 2029 His Majesty's Government is pleased to make the following rules: 1. Short title, and commencement:
Plant Protection Rules 2031 In exercise of the powers conferred by Section 9 of the Plant Protection Act, 2029 His Majesty's Government is pleased to make the following rules: 1. Short title, and commencement:
AUDIT REPORT. Audit of Offi cial Controls carried out by the Local Authority Veterinary Service Leitrim County Council
 AUDIT REPORT Audit of Offi cial Controls carried out by the Local Authority Veterinary Service AUDIT REPORT Audit of Official Controls carried out by the Local Authority Veterinary Service TABLE OF CONTENTS
AUDIT REPORT Audit of Offi cial Controls carried out by the Local Authority Veterinary Service AUDIT REPORT Audit of Official Controls carried out by the Local Authority Veterinary Service TABLE OF CONTENTS
The Prerequisites Program includes:
 STATEMENT OF COMPLIANCE (HACCP-PREERQUISITES LETTER) January, 2015 1 APPROVALS AND CERTIFICATIONS Frigorífico INALER S.A., Establishment N9 55 is approved by the European Commission (Food and Veterinary
STATEMENT OF COMPLIANCE (HACCP-PREERQUISITES LETTER) January, 2015 1 APPROVALS AND CERTIFICATIONS Frigorífico INALER S.A., Establishment N9 55 is approved by the European Commission (Food and Veterinary
SPRING VIRAEMIA OF CARP
 CHAPTER 10.9. SPRING VIRAEMIA OF CARP Article 10.9.1. For the purposes of the Aquatic Code, spring viraemia of carp (SVC) means infection with the viral species SVC virus (SVCV) tentatively placed in the
CHAPTER 10.9. SPRING VIRAEMIA OF CARP Article 10.9.1. For the purposes of the Aquatic Code, spring viraemia of carp (SVC) means infection with the viral species SVC virus (SVCV) tentatively placed in the
Background paper. Animal welfare in the EU
 Background paper Animal welfare in the EU January 2018 1 The European Court of Auditors is currently examining whether action by the European Commission and the Member States has made an effective contribution
Background paper Animal welfare in the EU January 2018 1 The European Court of Auditors is currently examining whether action by the European Commission and the Member States has made an effective contribution
Biosecurity Services Group
 Biosecurity Services Group Verification of Ante and Post Mortem Inspection On-Plant Management System Standard Operating Procedure 3.03 Page 1 Export Meat Program Page 1 of 7 CONTENTS 1. PURPOSE...3 2.
Biosecurity Services Group Verification of Ante and Post Mortem Inspection On-Plant Management System Standard Operating Procedure 3.03 Page 1 Export Meat Program Page 1 of 7 CONTENTS 1. PURPOSE...3 2.
Requirements for Beef and Beef Products to be Exported to Japan from the United States of America January 25, 2013
 Requirements for Beef and Beef Products to be Exported to Japan from the United States of America January 25, 2013 Requirements for beef and beef products to be exported to Japan from the United States
Requirements for Beef and Beef Products to be Exported to Japan from the United States of America January 25, 2013 Requirements for beef and beef products to be exported to Japan from the United States
INVESTING IN GUYANA, LIVESTOCK REARING. Ms. Monique Ifill Guyana Livestock Development Authority June 29, 2017
 INVESTING IN GUYANA, LIVESTOCK REARING Ms. Monique Ifill Guyana Livestock Development Authority June 29, 2017 OVERVIEW One of Guyana s main economic activity is Agriculture. As it relates to Agriculture
INVESTING IN GUYANA, LIVESTOCK REARING Ms. Monique Ifill Guyana Livestock Development Authority June 29, 2017 OVERVIEW One of Guyana s main economic activity is Agriculture. As it relates to Agriculture
EXPLANATORY NOTES TO ANNEX I 1 TO THE FREE TRADE AGREEMENT BETWEEN THE EFTA STATES AND MEXICO
 EXPLANATORY NOTES TO ANNEX I 1 TO THE FREE TRADE AGREEMENT BETWEEN THE EFTA STATES AND MEXICO (REFERRED TO IN ARTICLE 37 OF ANNEX I TO THE AGREEMENT) The Parties have agreed to apply the following Explanatory
EXPLANATORY NOTES TO ANNEX I 1 TO THE FREE TRADE AGREEMENT BETWEEN THE EFTA STATES AND MEXICO (REFERRED TO IN ARTICLE 37 OF ANNEX I TO THE AGREEMENT) The Parties have agreed to apply the following Explanatory
STATE DEPARTMENT OF LIVESTOCK UPDATE ON DISEASE FREE ZONING IN KENYA ZONE 1: COAST
 STATE DEPARTMENT OF LIVESTOCK UPDATE ON DISEASE FREE ZONING IN KENYA ZONE 1: COAST GOAL To contribute toward poverty reduction and food security. To comply with sanitary measures in line with Kenya s obligations
STATE DEPARTMENT OF LIVESTOCK UPDATE ON DISEASE FREE ZONING IN KENYA ZONE 1: COAST GOAL To contribute toward poverty reduction and food security. To comply with sanitary measures in line with Kenya s obligations
FINAL REPORT Electronic system
 FINAL REPORT Electronic system for Alternate protocol for managing illegible or missing shipping marks for the USA Project code: 2016.1047 Prepared by: Des Bowler Date Submitted: August 2016 Date Published:
FINAL REPORT Electronic system for Alternate protocol for managing illegible or missing shipping marks for the USA Project code: 2016.1047 Prepared by: Des Bowler Date Submitted: August 2016 Date Published:
Release of Fish or Fish Product Detained or Recalled for Marine Biotoxin Reasons Guide
 Release of Fish or Fish Product Detained or Recalled for Marine Biotoxin Reasons Guide January 2012 Disclaimer Every effort has been made to ensure the information in this report is accurate. MAF does
Release of Fish or Fish Product Detained or Recalled for Marine Biotoxin Reasons Guide January 2012 Disclaimer Every effort has been made to ensure the information in this report is accurate. MAF does
By Adelaida Harries and Joseph Cortes Seed Science Center Iowa State University
 SEED IMPORT/ EXPORT PROCEDURES MANUAL FOR GOVERNMENT AGENCIES By Adelaida Harries and Joseph Cortes Seed Science Center Iowa State University Copyright 2005 Seed Science Center Iowa State University. All
SEED IMPORT/ EXPORT PROCEDURES MANUAL FOR GOVERNMENT AGENCIES By Adelaida Harries and Joseph Cortes Seed Science Center Iowa State University Copyright 2005 Seed Science Center Iowa State University. All
Risk Management Programme Page: 1 of 8 Date: / / 1. Business Identification. 2. Operator Name, Business Address and Contact Details
 Risk Management Programme Page: 1 of 8 Date: / / 1. Business Identification 1.1 Business ID: 1.2 RMP No.: 1.3 Unique Location Identifier for Dairy Business Operators: 2. Operator Name, Business Address
Risk Management Programme Page: 1 of 8 Date: / / 1. Business Identification 1.1 Business ID: 1.2 RMP No.: 1.3 Unique Location Identifier for Dairy Business Operators: 2. Operator Name, Business Address
OVERVIEW OF THE RESULTS OF A SERIES OF MISSIONS CARRIED OUT DURING TO EVALUATE CONTROLS OVER PIG MEAT PRODUCTION IN MEMBER STATES
 EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate F - Food and Veterinary Office Doc. No. DG(SANCO)/9005/2002 OVERVIEW OF THE RESULTS OF A SERIES OF MISSIONS CARRIED OUT
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate F - Food and Veterinary Office Doc. No. DG(SANCO)/9005/2002 OVERVIEW OF THE RESULTS OF A SERIES OF MISSIONS CARRIED OUT
Import Health Standard
 Impt Health Standard Vehicles, Machinery and Tyres VEHICLE.ALL Please Note: This document is the Impt Health Standard from 25 August 2017 that has been re-fmatted. It also includes requirements f used
Impt Health Standard Vehicles, Machinery and Tyres VEHICLE.ALL Please Note: This document is the Impt Health Standard from 25 August 2017 that has been re-fmatted. It also includes requirements f used
AUSTRALIAN RUMINANT FEED BAN NATIONAL UNIFORM GUIDELINES. For ensuring compliance through inspection, sampling and testing programs
 AUSTRALIAN RUMINANT FEED BAN NATIONAL UNIFORM GUIDELINES For ensuring compliance through inspection, sampling and testing programs JULY 2016 TO JUNE 2017 Published November 2016 Animal Health Australia
AUSTRALIAN RUMINANT FEED BAN NATIONAL UNIFORM GUIDELINES For ensuring compliance through inspection, sampling and testing programs JULY 2016 TO JUNE 2017 Published November 2016 Animal Health Australia
Structural Assistance Act
 Issuer: Riigikogu Type: act In force from: 01.09.2015 In force until: 04.12.2015 Translation published: 10.08.2015 Amended by the following acts Passed 04.06.2014 RT I, 21.06.2014, 1 Entry into force 01.07.2014
Issuer: Riigikogu Type: act In force from: 01.09.2015 In force until: 04.12.2015 Translation published: 10.08.2015 Amended by the following acts Passed 04.06.2014 RT I, 21.06.2014, 1 Entry into force 01.07.2014
Proceedings, The Range Beef Cow Symposium XXIII December 3-5, 2013 Rapid City, South Dakota OPPORTUNITIES AND OBSTACLES FOR BREEDING CATTLE EXPORTS
 Proceedings, The Range Beef Cow Symposium XXIII December 3-5, 2013 Rapid City, South Dakota OPPORTUNITIES AND OBSTACLES FOR BREEDING CATTLE EXPORTS Tony Clayton, President Clayton Agri-Marketing, Inc.
Proceedings, The Range Beef Cow Symposium XXIII December 3-5, 2013 Rapid City, South Dakota OPPORTUNITIES AND OBSTACLES FOR BREEDING CATTLE EXPORTS Tony Clayton, President Clayton Agri-Marketing, Inc.
Dental Products (Subsidiary Hazard) Group Standard HSR002558
 Dental Products (Subsidiary Hazard) Group Standard 2017 - HSR002558 GROUP STANDARD UNDER THE HAZARDOUS SUBSTANCES AND NEW ORGANISMS ACT 1996 Dental Products (Subsidiary Hazard) Group Standard 2017 Pursuant
Dental Products (Subsidiary Hazard) Group Standard 2017 - HSR002558 GROUP STANDARD UNDER THE HAZARDOUS SUBSTANCES AND NEW ORGANISMS ACT 1996 Dental Products (Subsidiary Hazard) Group Standard 2017 Pursuant
CERTIFICATION OF AREA OR PROPERTY FREEDOM BASED ON MONITORING BY THE ACCREDITING AUTHORITY
 CERTIFICATION OF AREA OR PROPERTY FREEDOM BASED ON MONITORING BY THE ACCREDITING AUTHORITY REVISION REGISTER Revision No. Date of Issue Amendment Details New 01/01/2003 New Issue First 27/07/2005 All Pages
CERTIFICATION OF AREA OR PROPERTY FREEDOM BASED ON MONITORING BY THE ACCREDITING AUTHORITY REVISION REGISTER Revision No. Date of Issue Amendment Details New 01/01/2003 New Issue First 27/07/2005 All Pages
Animal identification and traceability in the European Union and international trade
 Animal identification and traceability in the European Union and international trade S. Pavón Directorate General for Health and Consumers, European Commission, Brussels, Belgium Summary International
Animal identification and traceability in the European Union and international trade S. Pavón Directorate General for Health and Consumers, European Commission, Brussels, Belgium Summary International
The best quality assurance for meat and animal feed comes from the Netherlands
 The best quality assurance for meat and animal feed comes from the Netherlands The Dutch livestock and meat sectors have a worldwide reputation to uphold as reliable suppliers of quality produce. To safeguard
The best quality assurance for meat and animal feed comes from the Netherlands The Dutch livestock and meat sectors have a worldwide reputation to uphold as reliable suppliers of quality produce. To safeguard
Overview Codex Alimentarius Guidance for Export Certification. Presented by Mary Stanley APEC Certification Roundtable February 25, 2010
 Overview Codex Alimentarius Guidance for Export Certification Presented by Mary Stanley APEC Certification Roundtable February 25, 2010 Codex Committee on Food Import and Export Certification and Inspection
Overview Codex Alimentarius Guidance for Export Certification Presented by Mary Stanley APEC Certification Roundtable February 25, 2010 Codex Committee on Food Import and Export Certification and Inspection
Guidance Document. CATDOG.TF Guidance Document for Cat and Dog Transitional Facilities. 16 January 2013
 Guidance Document CATDOG.TF Guidance Document for Cat and Dog Transitional Facilities 16 January 2013 This document is intended as a guidance document accompanying the Standard for Cat and Dog Transitional
Guidance Document CATDOG.TF Guidance Document for Cat and Dog Transitional Facilities 16 January 2013 This document is intended as a guidance document accompanying the Standard for Cat and Dog Transitional
ASEAN STANDARD FOR MANDARIN
 ASEAN Stan 9:2008 ASEAN STANDARD FOR MANDARIN 1. DEFINITION OF PRODUCE This standard applies to commercial varieties of mandarin grown from Citrus reticulata Blanco of the Rutaceae family, to be supplied
ASEAN Stan 9:2008 ASEAN STANDARD FOR MANDARIN 1. DEFINITION OF PRODUCE This standard applies to commercial varieties of mandarin grown from Citrus reticulata Blanco of the Rutaceae family, to be supplied
CFIA: Program Import Requirements
 CFIA: Program Import Requirements Food Safety & Labelling The Food and Drugs Act and Regulations (FDA & R ), and the Consumer Packaging and Labelling Act and Regulations (CPLA & R) place the legal responsibility
CFIA: Program Import Requirements Food Safety & Labelling The Food and Drugs Act and Regulations (FDA & R ), and the Consumer Packaging and Labelling Act and Regulations (CPLA & R) place the legal responsibility
Part 148. CAA Consolidation. 10 March Aircraft Manufacturing Organisations - Certification
 Part 148 CAA Consolidation 10 March 2017 Aircraft Manufacturing Organisations - Certification Published by the Civil Aviation Authority of New Zealand DESCRIPTION Part 148 prescribes rules governing the
Part 148 CAA Consolidation 10 March 2017 Aircraft Manufacturing Organisations - Certification Published by the Civil Aviation Authority of New Zealand DESCRIPTION Part 148 prescribes rules governing the
 Department for Environment, Food and Rural Affairs Scottish Executive Welsh Assembly Government Notes for Guidance for Completion of TRACES EXA31(Equidae) Application for Veterinary Health Certification
Department for Environment, Food and Rural Affairs Scottish Executive Welsh Assembly Government Notes for Guidance for Completion of TRACES EXA31(Equidae) Application for Veterinary Health Certification
1.1 Fruits shall be treated by using one of the following schedules: 2. Approval of facilities for pre-cooling, refrigeration and fumigation:
 Quarantine Requirements for The Importation of Fresh Fruits from Countries or Districts Where The Mediterranean Fruit Fly Is Known to Occur (In case of any discrepancy between the Chinese text and the
Quarantine Requirements for The Importation of Fresh Fruits from Countries or Districts Where The Mediterranean Fruit Fly Is Known to Occur (In case of any discrepancy between the Chinese text and the
Farm registration and movement of farm livestock
 Farm registration and movement of farm livestock Registering your land as an agricultural holding Before moving livestock to your school land (holding) you need a County Parish Holding (CPH) number for
Farm registration and movement of farm livestock Registering your land as an agricultural holding Before moving livestock to your school land (holding) you need a County Parish Holding (CPH) number for
Warehousing Process - Land
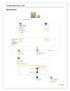 High Level Process Pre-clearance Process 1 2 Ministries Departments and Agencies (MDA) Process Scans Commercial Invoice Unique Consignment Reference (UCR) Bond Process Removal Bond Premises Bond MDA Permit
High Level Process Pre-clearance Process 1 2 Ministries Departments and Agencies (MDA) Process Scans Commercial Invoice Unique Consignment Reference (UCR) Bond Process Removal Bond Premises Bond MDA Permit
GUIDE TO COMPLETING THE NEW ZEALAND CUSTOMS & QUARANTINE (MPI) DECLARATION - NZCS218
 GUIDE TO COMPLETING THE NEW ZEALAND CUSTOMS & QUARANTINE (MPI) DECLARATION - NZCS218 The attached declaration must be completed by every person bringing household and personal effects into NZ. Some keys
GUIDE TO COMPLETING THE NEW ZEALAND CUSTOMS & QUARANTINE (MPI) DECLARATION - NZCS218 The attached declaration must be completed by every person bringing household and personal effects into NZ. Some keys
TEXTILE EXCHANGE. Policy and Template for issuing Transaction Certificates (TCs) Objective. Original Document. Multiple Shipments on a TC
 Policy and Template for issuing Transaction Certificates (TCs) Objective This policy provides for mandatory instructions and notes to be followed by approved Certification Bodies (CBs) to issue TCs in
Policy and Template for issuing Transaction Certificates (TCs) Objective This policy provides for mandatory instructions and notes to be followed by approved Certification Bodies (CBs) to issue TCs in
Meat Processor Quality Assurance Scheme Processor Standard Revision 01
 Meat Processor Quality Assurance Scheme Processor Standard Revision 01 Growing the success of Irish food & horticulture Growing the success of Irish food & horticulture Meat Processor Quality Assurance
Meat Processor Quality Assurance Scheme Processor Standard Revision 01 Growing the success of Irish food & horticulture Growing the success of Irish food & horticulture Meat Processor Quality Assurance
Rules governing the use of the AB mark
 Ministry of Agriculture and Fisheries Rules governing the use of the AB mark ARTICLE 1 - PURPOSE The AB mark may be used for the certification of products and/or for informational purposes. The AB mark,
Ministry of Agriculture and Fisheries Rules governing the use of the AB mark ARTICLE 1 - PURPOSE The AB mark may be used for the certification of products and/or for informational purposes. The AB mark,
Traceability: what to do in the context of mollusc production requirements under Directive 2006/88/EC
 Directorate-General for Health & Consumers Traceability: what to do in the context of mollusc production requirements under Directive 2006/88/EC Sigrid Cabot, DG SANCO, D1 sigrid.cabot@ec.europa.eu Annual
Directorate-General for Health & Consumers Traceability: what to do in the context of mollusc production requirements under Directive 2006/88/EC Sigrid Cabot, DG SANCO, D1 sigrid.cabot@ec.europa.eu Annual
SUBMISSION ON THE MPI IMPORT HEALTH STANDARD: VEHICLES, MACHINERY AND EQUIPMENT (VEHICLE-ALL) DATED DECEMBER 2017
 PO Box 10232, The Terrace, Wellington, 6143 Level 4, Co-operative Bank House 20 Ballance Street, Wellington, 6011 Phone: +64 4 472 3795 Fax: +64 4 471 2861 19 January 2018 Web: www.hortnz.co.nz Email:
PO Box 10232, The Terrace, Wellington, 6143 Level 4, Co-operative Bank House 20 Ballance Street, Wellington, 6011 Phone: +64 4 472 3795 Fax: +64 4 471 2861 19 January 2018 Web: www.hortnz.co.nz Email:
PROCEDURE FOR OFFICIAL RECOGNITION OF DISEASE STATUS UPDATE ON FOOT AND MOUTH DISEASE
 PROCEDURE FOR OFFICIAL RECOGNITION OF DISEASE STATUS UPDATE ON FOOT AND MOUTH DISEASE Introduction In 1994, Member Countries gave the World Organisation for Animal Health (OIE) the responsibility of compiling
PROCEDURE FOR OFFICIAL RECOGNITION OF DISEASE STATUS UPDATE ON FOOT AND MOUTH DISEASE Introduction In 1994, Member Countries gave the World Organisation for Animal Health (OIE) the responsibility of compiling
12 th International Training Course Harmonisation of Fruit and Vegetables Quality Assessment
 12 th International Training Course Harmonisation of Fruit and Vegetables Quality Assessment FOOD INSPECTION IN MAURITIUS with focus on fruit and vegetables Shalini A. NEELIAH Senior Research & Development
12 th International Training Course Harmonisation of Fruit and Vegetables Quality Assessment FOOD INSPECTION IN MAURITIUS with focus on fruit and vegetables Shalini A. NEELIAH Senior Research & Development
ORIGINATING OFFICE: FO:TCF DISTRIBUTION: S-01 CBP DIRECTIVE NO A DATE: AUGUST 7, 2003 SUPERSEDES: ,1/7/88 REVIEW DATE: AUGUST 2006
 CUSTOMS AND BORDER PROTECTION DIRECTIVE ORIGINATING OFFICE: FO:TCF DISTRIBUTION: S-01 CBP DIRECTIVE NO. 3240-036A DATE: AUGUST 7, 2003 SUPERSEDES: 3240-036,1/7/88 REVIEW DATE: AUGUST 2006 SUBJECT: IN-BOND
CUSTOMS AND BORDER PROTECTION DIRECTIVE ORIGINATING OFFICE: FO:TCF DISTRIBUTION: S-01 CBP DIRECTIVE NO. 3240-036A DATE: AUGUST 7, 2003 SUPERSEDES: 3240-036,1/7/88 REVIEW DATE: AUGUST 2006 SUBJECT: IN-BOND
CURRENT PROBLEMS OF THE LEATHER INDUSTRY. Yibralem Abadi
 CURRENT PROBLEMS OF THE LEATHER INDUSTRY Yibralem Abadi Ethiopian Tanners Association, P.O. Box 12898, Addis Ababa, Ethiopia Introduction The leather industry sector is one of the fast growing economic
CURRENT PROBLEMS OF THE LEATHER INDUSTRY Yibralem Abadi Ethiopian Tanners Association, P.O. Box 12898, Addis Ababa, Ethiopia Introduction The leather industry sector is one of the fast growing economic
UNITED STATES DEPARTMENT OF AGRICULTURE FOOD SAFETY AND INSPECTION SERVICE WASHINGTON, DC AVAILABILITY OF QUESTIONS AND ANSWERS EXPORT CERTIFICATION
 UNITED STATES DEPARTMENT OF AGRICULTURE FOOD SAFETY AND INSPECTION SERVICE WASHINGTON, DC FSIS NOTICE 65-06 10/5/06 AVAILABILITY OF QUESTIONS AND ANSWERS EXPORT CERTIFICATION This notice provides answers
UNITED STATES DEPARTMENT OF AGRICULTURE FOOD SAFETY AND INSPECTION SERVICE WASHINGTON, DC FSIS NOTICE 65-06 10/5/06 AVAILABILITY OF QUESTIONS AND ANSWERS EXPORT CERTIFICATION This notice provides answers
CHAPER 4 CUSTOMS PROCEDURES AND TRADE FACILITATION
 CHAPER 4 CUSTOMS PROCEDURES AND TRADE FACILITATION Article 4.1 : Scope and Objectives 1. This Chapter shall apply, in accordance with the Parties respective international obligations and customs law, to
CHAPER 4 CUSTOMS PROCEDURES AND TRADE FACILITATION Article 4.1 : Scope and Objectives 1. This Chapter shall apply, in accordance with the Parties respective international obligations and customs law, to
POULTRY HATCHERY Process day-old poultry chicks
 1 of 5 level: 3 credit: 10 planned review date: December 2008 sub-field: purpose: entry information: accreditation option: moderation option: Poultry Production People credited with this unit standard
1 of 5 level: 3 credit: 10 planned review date: December 2008 sub-field: purpose: entry information: accreditation option: moderation option: Poultry Production People credited with this unit standard
Land Transport Rule Dangerous Goods 2005
 WELLINGTON, NEW ZEALAND PURSUANT to Section 152 of the Land Transport Act 1998 I, Harry James Duynhoven, Minister for Transport Safety, HEREBY make the following ordinary Rule: Land Transport Rule: Dangerous
WELLINGTON, NEW ZEALAND PURSUANT to Section 152 of the Land Transport Act 1998 I, Harry James Duynhoven, Minister for Transport Safety, HEREBY make the following ordinary Rule: Land Transport Rule: Dangerous
Australian Import Permits
 Australian Import Permits It is the customer s responsibility to arrange the correct import permit. An import permit is a document issued by a national government authorising the importation of certain
Australian Import Permits It is the customer s responsibility to arrange the correct import permit. An import permit is a document issued by a national government authorising the importation of certain
E37. Genetically modified organisms [CIV : Federated Farmers of New Zealand Incorporated]
![E37. Genetically modified organisms [CIV : Federated Farmers of New Zealand Incorporated] E37. Genetically modified organisms [CIV : Federated Farmers of New Zealand Incorporated]](/thumbs/73/68458426.jpg) E37. Genetically modified organisms [CIV-2016-404-002299: Federated Farmers of New Zealand Incorporated] E37.1. Background The outdoor use of genetically modified organisms has the potential to cause adverse
E37. Genetically modified organisms [CIV-2016-404-002299: Federated Farmers of New Zealand Incorporated] E37.1. Background The outdoor use of genetically modified organisms has the potential to cause adverse
Law of the Republic of Kazakhstan «Standardization»
 Draft Law of the Republic of Kazakhstan «Standardization» The present Law shall establish legal basis for standardization and determine measures of state protection of consumers and state s interests by
Draft Law of the Republic of Kazakhstan «Standardization» The present Law shall establish legal basis for standardization and determine measures of state protection of consumers and state s interests by
When Certification Programs to Ensure Compliance with Foreign Standards Enhance Trade: The Case of Brazilian Beef Exports
 International Agricultural Trade Research Consortium Annual Meeting December 13-15, 2009 Ft Myers, Florida When Certification Programs to Ensure Compliance with Foreign Standards Enhance Trade: The Case
International Agricultural Trade Research Consortium Annual Meeting December 13-15, 2009 Ft Myers, Florida When Certification Programs to Ensure Compliance with Foreign Standards Enhance Trade: The Case
SOME ASPECTS OF AGRICULTURAL POLICY IN AUSTRALIA
 SOME ASPECTS OF AGRICULTURAL POLICY IN AUSTRALIA R. A. Sherwin, Agricultural Attache Australian Embassy, Washington, D. C. Before discussing government programs relating to agriculture in Australia I propose
SOME ASPECTS OF AGRICULTURAL POLICY IN AUSTRALIA R. A. Sherwin, Agricultural Attache Australian Embassy, Washington, D. C. Before discussing government programs relating to agriculture in Australia I propose
EUROPEAN COMMISSION DIRECTORATE-GENERAL FOR HEALTH AND FOOD SAFETY. EudraLex. Volume 4
 EUROPEAN COMMISSION DIRECTORATE-GENERAL FOR HEALTH AND FOOD SAFETY Ref. Ares(2015)4234460-12/10/2015 Medicinal products quality, safety and efficacy Brussels, 12 October 2015 EudraLex Volume 4 EU Guidelines
EUROPEAN COMMISSION DIRECTORATE-GENERAL FOR HEALTH AND FOOD SAFETY Ref. Ares(2015)4234460-12/10/2015 Medicinal products quality, safety and efficacy Brussels, 12 October 2015 EudraLex Volume 4 EU Guidelines
Code of Practice: Processing of Seafood Product. Part 3: HACCP Application, and the Identification of Other Risk Factors and their Controls
 Code of Practice: Processing of Seafood Product Part 3: HACCP Application, and the Identification of Other Risk Factors and their Controls July 2011 Introduction Disclaimer Every effort has been made to
Code of Practice: Processing of Seafood Product Part 3: HACCP Application, and the Identification of Other Risk Factors and their Controls July 2011 Introduction Disclaimer Every effort has been made to
A Case Study from EP3: Pollution Prevention Assessment from a Cattle Hide Tannery. HBI Pub. 10/31/94
 A Case Study from EP3: Pollution Prevention Assessment from a Cattle Hide Tannery HBI-94-003-01 Pub. 10/31/94 What is EP3? The amount of pollutants and waste generated by industrial facilities has become
A Case Study from EP3: Pollution Prevention Assessment from a Cattle Hide Tannery HBI-94-003-01 Pub. 10/31/94 What is EP3? The amount of pollutants and waste generated by industrial facilities has become
Guidelines for the import and export of drug and precursor reference standards
 INTERNATIONAL NARCOTICS CONTROL BOARD Guidelines for the import and export of drug and precursor reference standards for use by national drug testing laboratories and competent national authorities UNITED
INTERNATIONAL NARCOTICS CONTROL BOARD Guidelines for the import and export of drug and precursor reference standards for use by national drug testing laboratories and competent national authorities UNITED
Spotlight on Animal Health Business Operations: A Legal Update Husch Blackwell LLP. March 5, 2015
 Spotlight on Animal Health Business Operations: A Legal Update Husch Blackwell LLP March 5, 2015 Environmental Compliance, Planning and Management Systems I. Introduction A. Manufacturers and distributors,
Spotlight on Animal Health Business Operations: A Legal Update Husch Blackwell LLP March 5, 2015 Environmental Compliance, Planning and Management Systems I. Introduction A. Manufacturers and distributors,
B REGULATION (EC) No 183/2005 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 12 January 2005 laying down requirements for feed hygiene
 2005R0183 EN 16.09.2012 002.001 1 This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents B REGULATION (EC) No 183/2005 OF THE EUROPEAN PARLIAMENT
2005R0183 EN 16.09.2012 002.001 1 This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents B REGULATION (EC) No 183/2005 OF THE EUROPEAN PARLIAMENT
Streamlining and Automating Procedures for Agrifood Trade. TJCIQ LinJing
 Streamlining and Automating Procedures for Agrifood Trade TJCIQ LinJing Contents 1 Brief Introduction of AQSIQ and Tianjin CIQ 2 CIQ s promoting measure for Agrifood 3 The future plans of trade facilitation
Streamlining and Automating Procedures for Agrifood Trade TJCIQ LinJing Contents 1 Brief Introduction of AQSIQ and Tianjin CIQ 2 CIQ s promoting measure for Agrifood 3 The future plans of trade facilitation
GRAIN TRANSPORT CODE OF PRACTICE
 GRAIN TRADE AUSTRALIA LIMITED GRAIN TRANSPORT CODE OF PRACTICE 1. Introduction The Australian grain industry is heavily reliant on land transport for its viability. Transport is a critical component of
GRAIN TRADE AUSTRALIA LIMITED GRAIN TRANSPORT CODE OF PRACTICE 1. Introduction The Australian grain industry is heavily reliant on land transport for its viability. Transport is a critical component of
THE CORPORATION OF THE TOWNSHIPSHIP OF ADELAIDE METCALFE
 THE CORPORATION OF THE TOWNSHIPSHIP OF ADELAIDE METCALFE BY-LAW #69-2011 Being a by-law respecting Construction, Demolition and Change of Use Permits and Inspections for the Township of Adelaide Metcalfe
THE CORPORATION OF THE TOWNSHIPSHIP OF ADELAIDE METCALFE BY-LAW #69-2011 Being a by-law respecting Construction, Demolition and Change of Use Permits and Inspections for the Township of Adelaide Metcalfe
In New Zealand. ProAnd Ltd Stafford Street Feilding New Zealand Ph (++64)-(0) Fax (++64)-(0)
 COMPANY PROFILE Australia ProAnd Associates Australia Pty Ltd Suite 3,10 Wharf Road Gladesville, Sydney NSW 2111 Australia Ph (++61)-(0)2-9879- 5500 Fax (++61)-(0)2-9879- 5511 In New Zealand ProAnd Ltd
COMPANY PROFILE Australia ProAnd Associates Australia Pty Ltd Suite 3,10 Wharf Road Gladesville, Sydney NSW 2111 Australia Ph (++61)-(0)2-9879- 5500 Fax (++61)-(0)2-9879- 5511 In New Zealand ProAnd Ltd
Management for Technology Pty Ltd. Analysis of information flows and implementation of an e-business solution for Killarney Abattoir
 final report Project code: P.PIP.0079 Prepared by: Des Bowler Management for Technology Pty Ltd Date submitted: August 2006 PUBLISHED BY Meat & Livestock Australia Limited Locked Bag 991 NORTH SYDNEY NSW
final report Project code: P.PIP.0079 Prepared by: Des Bowler Management for Technology Pty Ltd Date submitted: August 2006 PUBLISHED BY Meat & Livestock Australia Limited Locked Bag 991 NORTH SYDNEY NSW
Guidance Document. Dairy Export Quota Products. A guide to the development of a Quota Compliance Programme (QCP) and Export Licence guidance
 Guidance Document Dairy Export Quota Products A guide to the development of a Quota Compliance Programme (QCP) and Export Licence guidance A guidance document issued by the Ministry for Primary Industries
Guidance Document Dairy Export Quota Products A guide to the development of a Quota Compliance Programme (QCP) and Export Licence guidance A guidance document issued by the Ministry for Primary Industries
