I «disease-modifying drugs» nello stato preclinico dell AD: quali evidenze?
|
|
|
- Amice Watson
- 6 years ago
- Views:
Transcription
1 I «disease-modifying drugs» nello stato preclinico dell AD: quali evidenze? Daniela Galimberti Università di Milano, Fondazione Ca Granda, IRCCS Ospedale Maggiore Policlinico
2 DEFICIT COLINERGICO Scarpini et al, Lancet Neurol 2003
3 Current symptomatic drugs for Alzheimer s disease Tacrine Cognex Cholinesterase inhibition Donepezil Rivastigmine Galantamine Memantine Aricept Memac Exelon Prometax Razadyne Reminyl Namenda, Ebixa Cholinesterase inhibition Cholinesterase inhibition Butyrylcholinesterase inhibition Cholinesterase inhibition Nicotinic receptor modulation Glutamate receptor modulation Not used due to hepatotoxicity FDA: 1996, EMEA: 1997; QD dosing; for mild, mod, severe AD BID; patch formulation released; for mild to mod AD BID; XR formulation released; for mild to mod AD Approved 2003; for moderate to severe AD
4 GLOBAL RESPONDERS to ChEIs Study Rogers 1998 ChEI Donepezil ChEI responder s 107/305 Placebo responder s 27/150 Total subj. 455 Favours ChEI Favours placebo Rogers 1998b Donepezil 76/298 17/ Burns 1999 Donepezil 125/544 38/ Rosler 1999 Rivastigmine 149/467 44/ Raskind 2000 Galantamine 64/357 27/ Wilcock 2000 Galantamine 84/414 33/ Rockwood 2001 Galantamine 61/240 24/ Wilkinson 2001 Galantamine 59/179 23/ % 16 % 262 Meta-analytic difference 30% 20% 10% 0% -10% Responders: improvement or better (excluded unchanged) on a global assessment scale (type 1) Lanctot et al., 2003
5 Recommendations of the 4th Canadian Consensus Conf. on the Diagnosis and Treatment of Dementia (2012) Serge Gauthier, MD, Christopher Patterson, MD, Howard Chertkow, MD, Michael Gordon, MD, Nathan Herrmann, MD, Kenneth Rockwood, MD, Pedro Rosa-Neto, md, phd, Jean-Paul Soucy, MD on behalf of the cccdtd4 participants* McGill Center for Studies in Aging, Montreal, QC Donepezil Aricept Cholinesterase Inhibition FDA: 1996, EMEA: 1997; QD dosing; for mild, mod, severe AD Rivastigmine Exelon Cholinesterase Inhibition BID; patch formulation released; for mild to mod AD Galantamine Reminyl Cholinesterase Inhibition BID; XR formulation released; for mild to mod AD - all three ChEIs have demonstrated efficacy for mild to severe AD - improvement is statistically significant but clinically marginal - a trial for most patients is recommended (Grade 1A) - direct comparisons do not suggest differences (Grade 2B)
6 Disease-modifying treatments AD pathogenesis: the amyloid cascade hypothesis
7 AD: disease-modifying strategies 1. Anti-amyloid production and aggregation agents:. -secretase inhibition. BACE-1 inhibition 2. Amyloid removal: immunotherapy 3. Anti-tau protein compounds Golde et al, 2010
8 From the first preclinical evidence of effectiveness of vaccination against Ab in mice Nature, 1999 to the development of many disease-modifying drugs.
9 Active immunization HUMAN TRIAL: AN AS21 adjuvant phase IIa study probable AD (mild to moderate) Study discontinuation! Aseptic meningoencephalitis in 18 of 300 (6%) immunized patients By the time of discontinuation: - 24 patients received 3 immunizations received 2 immunizations
10 Results after 6 yrs: clearance of amyloid plaques, but no evidence of increased survival and no improvement in the time to severe dementia!
11 New approach: passive immunization
12 Mangialasche et al, Lancet Neurol. 2010
13 2016: state of the art 2003: AN 1792 failed due to adverse events 2012: Bapineuzumab failed to reach primary endpoints (Salloway et al., 2014) 2014: Gantenerumab withdrawn in Dec : i.v. Ig failed (Dodel et al., 2013) 2014: Solanezumab in moderate AD: failed (Doody et al., 2014) semagacestat avagacestat -secretase inhibitors failed Doody et al., 2013, Coric et al., 2012 CAD 106 phase II complete Solanezumab phase III: positive results only in mild AD MK-8931 BACE inhibitor, phase II Aducanumab phase Ib completed
14 Decreased amyloid load on PiB-PET Aug 31, 2016
15 Why did so many trials fail? Population recruited: - patients may not have AD but other dementias instead - too late treatment (removal of plaques is not sufficient to halt the disease progression) treat preclinical/prodromal AD
16 Pre-dementia phase Use of biomarkers to enrich the cohort studied with subjects with an ongoing AD pathology MCI with a positive biomarker Prodromal AD (Dubois et al., 2010)
17 NINCS-ADRDA 1984 Time line AD criteria
18 IWG 2010 Biomarkers: - MRI - fdgpet - CSF - mutation NINCS-ADRDA 1984 IWG Time line AD criteria
19 Biomarkers of underlying pathology: - CSF (amyloid and tau) - PET with amyloid (and tau) tracers - mutation IWG NINCS-ADRDA 1984 NIA-AA 2011 Time line AD criteria
20 Which biomarker? Reflecting Ab deposition: - CSF low Ab - PET Ab tracers (PiB, Fluorbetapir, Fluorbetaben, ecc) - presence of a known causal mutation (APP, PSEN 1 and 2) Reflecting tau deposition: - CSF high tau - PET tau tracers ([ 18 F]AV-1451) Attempts to select cohort have been done so far targeting Ab, on the basis of the «amyloid hypothesis» BUT Ab accumulation does not correlate well with extent of neuronal loss or cognitive dysfunction
21 Amyloid CSF levels: changed 10 years before symptoms appearance Tau CSF levels: changed 5 years before symptoms appearance
22 Use of BMs in clinical trials Ideally: treat subjects with low CSF Ab but normal tau (no cell death yet) prevent all pathogenic mechanisms related to Ab deposition, responsible for neurodegeneration In practice: considering the duration of a trial (2-3 years) and the lack of outcome measures in asymptomatics or surrogate biomarkers for progression treat «MCI due to AD» (outcome measure: conversion from MCI to AD) Sperling et al., 2014
23 PET TAU tracers: [ 18 F]AV-1451 The pattern of increased [ 18 F]AV-1451 retention highly overlapped with regions that showed decreased [ 18 F]FDG uptake hypometabolism and symptomatology are more closely linked to tau than to Ab
24 PRE-MCI: AD trajectory Ab Ab+Tau asymptomatic Sterling et al., 2014
25 Stage 0: primary prevention - About 40-50% of people - Cardiovascular risk factors - Mediterranean diet - Physical activity... Sterling et al., 2014; Jack et al., 2011, mod.
26 Nature and nurture: the case of Romania Cornutiu et al, 2011
27 - Express Ab Monkeys as models for AD - With aging: develop senile plaques, although to a lesser extent as compared with humans, but no NFTs - High amount of senile plaques and NFTs in an obese monkey Bufill et al., 2013
28 Stage 1: secondary prevention Stage 1 (10-15%): secondary prevention with a treatment interfering with Ab deposition Immunization: from treatment for symptomatics to prevention in asymptomatics Sterling et al., 2014
29 Cognitive trajectory in normal aging and AD Amyloid treatments Sterling et al., 2014
30 PREVENTION
31 Alzheimer Prevention Initiative (API) - Phase II: Crenezumab tested in a large Colombian PSEN1 cohort Dominantly Inherited Alzheimer Network (DIAN) - Phase II: Solanezumab vs gantenerumab in carriers of mutations in APP, PSEN1 and PSEN2 Anti-Amyloid Treatment in Asymptomatic Alzheimer s disease (A4) - Phase III: Solanezumab vs placebo in cognitively normal people (65-85 years) with evidence of Ab accumulation
32 Additional vaccines waiting for clinical trials ACI-24 (AC Immune): Ab1-15 peptide to which on both ends two lysines were attached. Antigen embedded in a liposome membrane. Currently evaluated in a phase I/II clinical trial in Denmark, Finland and Sweden Affitope AD-02 (Affiris, Vienna AT and licensee GSK Biol.): is 6-aminoacid peptide vaccine targeting the N-terminus of Ab. European phase II clinical trial in 420 patients ongoing Affitope-AD-03 (Affiris and licensee GSK Biol.): phase I completed in 2011, results not yet published ACC-001 (vanutide cridificar; Pfizer). Two Phase II studies in patients with mild to moderate AD: one in Japan (completed) and one ongoing in USA. UB-311 (United Biomedical): intramuscularly administered vaccine targeting N- terminal amino acids 1-14 of Ab in phase I clinical trials in Taiwan in patients with mild to moderate AD V-950 (Merck): Phase II, double-blind, randomized, placebo-controlled, dose escalating study completed, but results not yet published. D. Galimberti, E. Scarpini, J. Neurol Sci, 2013
33 Additional monoclonal antibodies waiting for clinical trials PF (Pfizer, phase II completed) GSK933776A (GlaxoSmithKline, phase I completed) NI-101 (Biogen Idec, phase I clinical trial in patients with mild to moderate AD ongoing) PF (Janssen Alzheimer Immunotherapy and Pfizer, phase I trial ongoing) RN6G (Rinat Neuroscience Corp., New York, now Pfizer, phase I completed) SAR (Sanofi, phase I ongoing) BAN-2401 (Eisai, phase I trial in 80 patients with mild to moderate AD ongoing). D. Galimberti, E. Scarpini, J. Neurol. Sci., 2013
34 The future: vaccination as primary prevention
35 Stages 2-3: acting on tau Strategies (preclinical): - Inhibition of GSK3b* (kinase phosphorylating tau) - Activation of phosphatases (PP2A) - Vaccination (more difficult as tau is intracellular)
36 Chronic, peripheral administration of anti-tau/ps422 antibody reduces the accumulation of tau pathology Triple transgenic mouse model of AD Anti-tau/pS422 anti-body binds to membrane-associated tau/ps422 and antigen-antibody complexes are cleared intracellularly
37 Stages 2-3: acting on tau Strategies (preclinical): Ab- Tau+ Other pathogenic proteins (TDP-43, FUS, Synuclein)? - Inhibition of GSK3b* (kinase phosphorylating tau) - Activation of phosphatases (PP2A) - Vaccination (more difficult as tau is intracellular) - Methylene blue (Rember), Phase II - ongoing - *Tideglusib, Phase II failed - AADvac1, Phase I, ongoing but not recruiting
38 Conclusions: open questions for early intervention Are subjects in early intervention trials early enough in disease process to respond to interventions? How early is early enough? What are the key variables that determines when a subject responds to a disease modifying drug, such as an anti amyloid agent? Cognitive impairment? Neurodegeneration? Tau or Ab levels? Are earlier trials feasible? Prodromal AD trials are hard to enroll Earlier stages of illness: TBD (use of BMs? Feasibility?) What is optimal design of early intervention trials - duration (more than 2-3 years)? - costs - ethical issues
39 Active immunotherapy: CAD106 K K K + + DAEFRH-GGC = K K Carrier: Qb-VLP Linker: SMPH Peptide: Ab1-6 - Spacer Qb-Ab1-6 (CAD106) - Drug substance: no T cellmediated immune reaction! New antigen designed to generate high Ab antibody titers without inducing Ab-reactive T-cells Abeta 1-6 peptide, shorter than known human T-cell epitopes Its sequence is predicted not to activate T-cell responses Experimentally it contains B-cell but not T-cell epitopes Monsonego, et al. 2003, Staufenbiel, M. et al., Abst O ICAD 2006 Madrid
40 Age of mice (months) Reduction of amyloid accumulation 25 mg CAD106, subcutaneously End of Experiment Cortex Control - VLP) (Qb) CAD106 (Qb-Ab1-6) Hippocampus APP24 mice Thalamus Caudate putamen Cortical plaque area Plaque area (%), Cortex ** Brain Ab42 content 0 PBS Qb CAD106A - Amyloid plaque area: ~80 % lower than in control mice - Longer treatment and start at earlier age: increased effect! Staufenbiel, M. et al., Abst O ICAD 2006 Madrid
41 Ab 1-40 Titers (OD50%) Immunization of Rhesus Monkeys Day 0 CAD106, 50 g CAD106, 25 g CAD106, 25 g Day 28 Day 54 Day 110 CAD106, 25 g Monkey #99 Monkey #110 Monkey #112 Monkey #134 Histology with antibodies from CAD 106 immunized Rhesus monkeys Time (days) CAD106-induced Ab antibodies: APP23 Brain - selectively bind to amyloid plaques in mouse and human brain - do not crossreact with cellular APP or brain cells - no reactivity with peripheral tissues: specific! AD Brain Staufenbiel, M. et al., 2006
42 Active immunotherapy: CAD106 first study in AD patients - phase I, 52-ws study in 58 mild to moderate AD pts (2 cohorts), aged yrs, randomly allocated to receive either CAD 106 or placebo - primary objectives: safety and tolerability and to identify Ab-antibody response (responders: patients with Ab-IgG serum titers>16 units at least once during the study) - 56/58 reported minor adverse events. No cases of meningoencephalitis - 67% of treated patients in cohort I and 82% in cohort 2 developed Ab antibody response. - Phase II: safety and tolerability confirmed (2014)
43 Ongoing active Phase III Adaption trial CAD : Novartis partnered with the Banner Alzheimer Research Institute to conduct a secondary prevention trial within the Alzheimer Prevention Initiative. - This Phase 2/3 trial began in November 2015 and is set to run until 2023, with a 5-year treatment period. - Aims to enroll 1,340 homozygous ApoE4 carriers between the ages of 60 and 75 who are cognitively normal. About half of participants will be randomized to compare CAD106 to matching placebo injected intramuscularly at weeks 1, 7, 13, 24 and then quarterly - Primary outcome: ability to delay diagnosis to MCI or AD dementia and change on the APICC cognitive composite (Langbaum et al., 2015). - Secondary outcomes: change on the Clinical Dementia Rating Scale sum of boxes (CDR-SB) along with other cognitive/functional scales, fluid biomarkers including CSF Aβ and tau, brain imaging including volumetric MRI plus amyloid PET and tau PET ClinicalTrials.gov Identifier: NCT Verified: Aug 2016
44 Caveat Are there concerns on inappropriately labeling individuals with «preclinical Alzheimer s disease» people who might never progress to manifest dementia in spite of biomarker evidence of amyloid deposition? In other fields, terms as «precancerous lesions» or «prediabetes» did not raise any ire. Apprehension over preclinical AD terminology reflects the stigma of the clinical syndrome of AD Sperling et al., 2014
45 Redundant research in AD drug development could be avoided by increased openness. Detailed results, outcomes, and databases of clinical trials should be made broadly available immediately after studies have been completed Early-phase clinical trials need to be replicated before drug development moves to later phases
46 Thank you for your attention
Engaging stakeholders for a holistic therapy in Alzheimer s disease
 Engaging stakeholders for a holistic therapy in Alzheimer s disease Brain Forum l Lausanne, May 26, 2016 l Prof. Andrea Pfeifer 2016 AC Immune. Not to be used or reproduced without permission. www.acimmune.com
Engaging stakeholders for a holistic therapy in Alzheimer s disease Brain Forum l Lausanne, May 26, 2016 l Prof. Andrea Pfeifer 2016 AC Immune. Not to be used or reproduced without permission. www.acimmune.com
12-Month Interim Analysis of APOE4 Carriers for Fixed and Titration Dosing Regimens in PRIME, a Phase 1b study of Aducanumab
 12-Month Interim Analysis of APOE4 Carriers for Fixed and Titration Dosing Regimens in PRIME, a Phase 1b study of Aducanumab Vissia Viglietta, 1 John O Gorman, 1 Leslie Williams, 1 Tianle Chen, 1 Ahmed
12-Month Interim Analysis of APOE4 Carriers for Fixed and Titration Dosing Regimens in PRIME, a Phase 1b study of Aducanumab Vissia Viglietta, 1 John O Gorman, 1 Leslie Williams, 1 Tianle Chen, 1 Ahmed
The Importance of Early Identification of Alzheimer s Disease
 The Importance of Early Identification of Alzheimer s Disease Alessandro Padovani Professor of Neurology, Director of the Institute of Neurology University of Brescia X No, nothing to disclose Yes, please
The Importance of Early Identification of Alzheimer s Disease Alessandro Padovani Professor of Neurology, Director of the Institute of Neurology University of Brescia X No, nothing to disclose Yes, please
Alzheimer's disease immunization strategies : a review of current research
 The University of Toledo The University of Toledo Digital Repository Master s and Doctoral Projects Alzheimer's disease immunization strategies : a review of current research Laura Ona Sirgedas The University
The University of Toledo The University of Toledo Digital Repository Master s and Doctoral Projects Alzheimer's disease immunization strategies : a review of current research Laura Ona Sirgedas The University
Capo Therapeutics, Inc.
 Capo Therapeutics, Inc. Vaccine Therapies for Debilitating Diseases of the Brain San Diego, CA 1 SCIENTIFIC ADVISORY BOARD M. Flint Beal, M.D. Dr. Beal is an internationally recognized authority on neurodegenerative
Capo Therapeutics, Inc. Vaccine Therapies for Debilitating Diseases of the Brain San Diego, CA 1 SCIENTIFIC ADVISORY BOARD M. Flint Beal, M.D. Dr. Beal is an internationally recognized authority on neurodegenerative
AD/PD Conference, Nice, Fr, 2015
 AD/PD Conference, Nice, Fr, 2015 Overview, novelties and conclusions for domestic research NAP B MTA TTK MS Neuroproteomics Group Overlaping molecular mechanisms of miss-folded protein based neurodegenerative
AD/PD Conference, Nice, Fr, 2015 Overview, novelties and conclusions for domestic research NAP B MTA TTK MS Neuroproteomics Group Overlaping molecular mechanisms of miss-folded protein based neurodegenerative
ADAPTIVE PHASE II STUDY OF BAN2401 IN EARLY ALZHEIMER S DISEASE CONTINUES TOWARD 18-MONTH ENDPOINT
 FOR IMMEDIATE RELEASE December 21, 2017 Eisai Co., Ltd. Biogen Inc. ADAPTIVE PHASE II STUDY OF BAN2401 IN EARLY ALZHEIMER S DISEASE CONTINUES TOWARD 18-MONTH ENDPOINT CRITERIA FOR SUCCESS AT 12-MONTH ANALYSIS
FOR IMMEDIATE RELEASE December 21, 2017 Eisai Co., Ltd. Biogen Inc. ADAPTIVE PHASE II STUDY OF BAN2401 IN EARLY ALZHEIMER S DISEASE CONTINUES TOWARD 18-MONTH ENDPOINT CRITERIA FOR SUCCESS AT 12-MONTH ANALYSIS
Innovative therapies for Alzheimer s disease
 WHITE PAPER IMS DISEASE INSIGHTS Innovative therapies for Alzheimer s disease New treatments will drive significant market value growth over the next ten years Author: Peter Alfinito, Ph.D., lead AnAlyst,
WHITE PAPER IMS DISEASE INSIGHTS Innovative therapies for Alzheimer s disease New treatments will drive significant market value growth over the next ten years Author: Peter Alfinito, Ph.D., lead AnAlyst,
Priavoid. A life without dementia. Company Presentation, BIO, June, Prof. Dr. Dieter Willbold
 Priavoid A life without dementia Company Presentation, BIO, June, 2017 Prof. Dr. Dieter Willbold (co-founder and future Chairman of the Supervisory Board) The Company At a glance Established: probably
Priavoid A life without dementia Company Presentation, BIO, June, 2017 Prof. Dr. Dieter Willbold (co-founder and future Chairman of the Supervisory Board) The Company At a glance Established: probably
Gunilla Osswald, PhD, CEO RedEye CNS Event, October 4, 2018
 BioArctic AB Company presentation Gunilla Osswald, PhD, CEO RedEye CNS Event, October 4, 2018 Disclaimer This presentation has been prepared and produced by BioArctic AB (publ) ( BioArctic ) solely for
BioArctic AB Company presentation Gunilla Osswald, PhD, CEO RedEye CNS Event, October 4, 2018 Disclaimer This presentation has been prepared and produced by BioArctic AB (publ) ( BioArctic ) solely for
Alzheimer s disease research in the 21 st century: Past and current failures and the way forward
 The European Commission s science and knowledge service Joint Research Centre Alzheimer s disease research in the 21 st century: Past and current failures and the way forward Francesca Pistollato francesca.pistollato@ec.europa.eu
The European Commission s science and knowledge service Joint Research Centre Alzheimer s disease research in the 21 st century: Past and current failures and the way forward Francesca Pistollato francesca.pistollato@ec.europa.eu
No new drugs for Alzheimer's disease in 15 years
 Published on ScienceNordic (http://sciencenordic.com) Home > Printer-friendly PDF > Printer-friendly PDF No new drugs for Alzheimer's disease in 15 years Health[1] Health[1]Alzheimer's disease [2]Health
Published on ScienceNordic (http://sciencenordic.com) Home > Printer-friendly PDF > Printer-friendly PDF No new drugs for Alzheimer's disease in 15 years Health[1] Health[1]Alzheimer's disease [2]Health
TARGETING ALZHEIMER S AND OTHER NEURODEGENERATIVE DISEASES WITH NOVEL THERAPEUTICS AND DIAGNOSTICS
 TARGETING ALZHEIMER S AND OTHER NEURODEGENERATIVE DISEASES WITH NOVEL THERAPEUTICS AND DIAGNOSTICS NASDAQ: ACIU Credit Suisse Healthcare Conference Prof. Andrea Pfeifer Nov 7, 2017 2017 AC Immune. Not
TARGETING ALZHEIMER S AND OTHER NEURODEGENERATIVE DISEASES WITH NOVEL THERAPEUTICS AND DIAGNOSTICS NASDAQ: ACIU Credit Suisse Healthcare Conference Prof. Andrea Pfeifer Nov 7, 2017 2017 AC Immune. Not
Tau-directed treatment approaches in Alzheimer s disease How far have we come?
 Tau-directed treatment approaches in Alzheimer s disease How far have we come? Lutz Frölich Department of Geriatric Psychiatry Central Institute of Mental Health University of Heidelberg, Germany EADC
Tau-directed treatment approaches in Alzheimer s disease How far have we come? Lutz Frölich Department of Geriatric Psychiatry Central Institute of Mental Health University of Heidelberg, Germany EADC
Application of Modeling and Simulation to Support Clinical Drug Development Decisions in Alzheimer's Disease
 Application of Modeling and Simulation to Support Clinical Drug Development Decisions in Alzheimer's Disease Marc R. Gastonguay, Ph.D. Scientific Director, Metrum Institute marcg@metruminstitute.org President
Application of Modeling and Simulation to Support Clinical Drug Development Decisions in Alzheimer's Disease Marc R. Gastonguay, Ph.D. Scientific Director, Metrum Institute marcg@metruminstitute.org President
BioArctic. Gunilla Osswald, PhD, CEO December 4, 2017 Lars Lannfelt, Professor, MD, Senior Advisor and Co-Founder
 BioArctic Gunilla Osswald, PhD, CEO December 4, 2017 Lars Lannfelt, Professor, MD, Senior Advisor and Co-Founder Disclaimer This presentation has been prepared and produced by BioArctic AB (publ) ( BioArctic
BioArctic Gunilla Osswald, PhD, CEO December 4, 2017 Lars Lannfelt, Professor, MD, Senior Advisor and Co-Founder Disclaimer This presentation has been prepared and produced by BioArctic AB (publ) ( BioArctic
A PHARMA MATTERS REPORT.
 REUTERS / Todd Korol SPOTLIGHT ON ALZHEIMER S DISEASE A PHARMA MATTERS REPORT. A REVIEW OF JULY-SEPTEMBER 2012. PUBLISHED NOVEMBER 2012. Expert therapy area review of the key market players and deals highlights
REUTERS / Todd Korol SPOTLIGHT ON ALZHEIMER S DISEASE A PHARMA MATTERS REPORT. A REVIEW OF JULY-SEPTEMBER 2012. PUBLISHED NOVEMBER 2012. Expert therapy area review of the key market players and deals highlights
The Big Picture of FTD Research: Funding, Collaboration and the Scoop on Participation
 The Big Picture of FTD Research: Funding, Collaboration and the Scoop on Participation 1 st Annual FTD Caregiver Conference Raleigh, NC July 12, 2011 Our objectives today Snapshot of medical research process
The Big Picture of FTD Research: Funding, Collaboration and the Scoop on Participation 1 st Annual FTD Caregiver Conference Raleigh, NC July 12, 2011 Our objectives today Snapshot of medical research process
New family of Alzheimer s disease-modifying agents that hit multiple biological targets
 New family of Alzheimer s disease-modifying agents that hit multiple biological targets Barcelona, 4 de julio de 2013 Content 1. The Research Group 2. The Product a) Target Indications b) Innovative mechanisms
New family of Alzheimer s disease-modifying agents that hit multiple biological targets Barcelona, 4 de julio de 2013 Content 1. The Research Group 2. The Product a) Target Indications b) Innovative mechanisms
LETTER OF INTENT Rapid Response: Canada 2019 Parkinson s & Related Diseases Round 2
 LETTER OF INTENT Rapid Response: Canada 2019 Parkinson s & Related Diseases Round 2 DEADLINE: March 13, 2019, 2:00pm EDT Applicants will be notified of Proposal invitations in May 2019 This Letter of Intent
LETTER OF INTENT Rapid Response: Canada 2019 Parkinson s & Related Diseases Round 2 DEADLINE: March 13, 2019, 2:00pm EDT Applicants will be notified of Proposal invitations in May 2019 This Letter of Intent
LETTER OF INTENT Rapid Response: Canada 2019 Parkinson s & Related Diseases Round 2
 LETTER OF INTENT Rapid Response: Canada 2019 Parkinson s & Related Diseases Round 2 DEADLINE: May 15, 2019, 2:00pm EDT Applicants will be notified of Proposal invitations in August 2019 This Letter of
LETTER OF INTENT Rapid Response: Canada 2019 Parkinson s & Related Diseases Round 2 DEADLINE: May 15, 2019, 2:00pm EDT Applicants will be notified of Proposal invitations in August 2019 This Letter of
PharmaCog Alzheimer s Disease
 PharmaCog Alzheimer s Disease Esther Schenker Institut de Recherches Servier France The issue People with dementia Total costs Billions US$ Western Europe 210.12 Central Europe 14.19 Eastern Europe 14.33
PharmaCog Alzheimer s Disease Esther Schenker Institut de Recherches Servier France The issue People with dementia Total costs Billions US$ Western Europe 210.12 Central Europe 14.19 Eastern Europe 14.33
LETTER OF INTENT Early Phase Clinical Trials 2018
 LETTER OF INTENT Early Phase Clinical Trials 2018 Applications are being accepted on a rolling basis. This Letter of Intent is an example only. Do not complete this paper application. Please submit the
LETTER OF INTENT Early Phase Clinical Trials 2018 Applications are being accepted on a rolling basis. This Letter of Intent is an example only. Do not complete this paper application. Please submit the
TARGETING ALZHEIMER S AND OTHER NEURODEGENERATIVE DISEASES WITH NOVEL THERAPEUTICS AND DIAGNOSTICS
 TARGETING ALZHEIMER S AND OTHER NEURODEGENERATIVE DISEASES WITH NOVEL THERAPEUTICS AND DIAGNOSTICS NASDAQ: ACIU KOL Event Tau Prof. Andrea Pfeifer Dec 1, 2017 2017 AC Immune. Not to be used or reproduced
TARGETING ALZHEIMER S AND OTHER NEURODEGENERATIVE DISEASES WITH NOVEL THERAPEUTICS AND DIAGNOSTICS NASDAQ: ACIU KOL Event Tau Prof. Andrea Pfeifer Dec 1, 2017 2017 AC Immune. Not to be used or reproduced
Engage with us on Twitter: #Molecule2Miracle
 Engage with us on Twitter: #Molecule2Miracle Kassy Perry President & CEO Perry Communications Group PhRMA Alliance Development Consultant.@kassyperry Emily Burke, Ph.D. Director of Curriculum BioTech
Engage with us on Twitter: #Molecule2Miracle Kassy Perry President & CEO Perry Communications Group PhRMA Alliance Development Consultant.@kassyperry Emily Burke, Ph.D. Director of Curriculum BioTech
Actinogen Medical Best Risk vs Reward Play in Alzheimer s Dementia
 ACTINOGEN LIMITED ACTINOGEN LIMITED 27 September 2016 Analyst: Alastair Murray +618 8236 8866 Authorisation: Alan Young +618 8236 8888 RECOMMENDATION Buy PRICE $0.057 TARGET PRICE $0.39 RISK High (Speculative)
ACTINOGEN LIMITED ACTINOGEN LIMITED 27 September 2016 Analyst: Alastair Murray +618 8236 8866 Authorisation: Alan Young +618 8236 8888 RECOMMENDATION Buy PRICE $0.057 TARGET PRICE $0.39 RISK High (Speculative)
Bapineuzumab Phase 3 trials in mild to moderate Alzheimer s disease dementia in apolipoprotein E e4 carriers (Study 302) and non-carriers (Study 301)
 Phase 3 trials in mild to moderate Alzheimer s disease dementia in apolipoprotein E e4 carriers (Study 302) and non-carriers (Study 301) Safety and PiB PET Amyloid Imaging Sperling R, Salloway S, Raskind
Phase 3 trials in mild to moderate Alzheimer s disease dementia in apolipoprotein E e4 carriers (Study 302) and non-carriers (Study 301) Safety and PiB PET Amyloid Imaging Sperling R, Salloway S, Raskind
BioArctic AB Interim Report January - June Gunilla Osswald, CEO Jan Mattsson, CFO August 23, 2018
 BioArctic AB Interim Report January - June 2018 Gunilla Osswald, CEO Jan Mattsson, CFO August 23, 2018 Disclaimer This presentation has been prepared and produced by BioArctic AB (publ) ( BioArctic ) solely
BioArctic AB Interim Report January - June 2018 Gunilla Osswald, CEO Jan Mattsson, CFO August 23, 2018 Disclaimer This presentation has been prepared and produced by BioArctic AB (publ) ( BioArctic ) solely
Evaluation of AMG 531 Efficacy in Splenectomized Patients with Chronic ITP in a Randomized Placebo- Controlled Phase 3 Study
 Evaluation of AMG 531 Efficacy in Splenectomized Patients with Chronic ITP in a Randomized Placebo- Controlled Phase 3 Study Plenary Session at ASH 2007 Presented by Terry B. Gernsheimer Madeleine Verhovsek
Evaluation of AMG 531 Efficacy in Splenectomized Patients with Chronic ITP in a Randomized Placebo- Controlled Phase 3 Study Plenary Session at ASH 2007 Presented by Terry B. Gernsheimer Madeleine Verhovsek
Genitope Corporation. Summary of MyVax Personalized Immunotherapy Phase 3 Clinical Trial Results
 Genitope Corporation Summary of MyVax Personalized Immunotherapy Phase 3 Clinical Trial Results Introduction In December 27, Genitope Corporation ( Genitope ) obtained data indicating that its pivotal
Genitope Corporation Summary of MyVax Personalized Immunotherapy Phase 3 Clinical Trial Results Introduction In December 27, Genitope Corporation ( Genitope ) obtained data indicating that its pivotal
Oral NT-KO-003 for the Treatment of Multiple Sclerosis
 Oral for the Treatment of Multiple Sclerosis Madrid, 27 de noviembre de 2013 Content 1. The Company 2. The Product a) Target Indications b) Innovative mechanisms of action c) Differential features facing
Oral for the Treatment of Multiple Sclerosis Madrid, 27 de noviembre de 2013 Content 1. The Company 2. The Product a) Target Indications b) Innovative mechanisms of action c) Differential features facing
Grifols demonstrates a significant reduction (61%) in the progression of moderate Alzheimer s disease using its AMBAR treatment protocol
 Grifols demonstrates a significant reduction (61%) in the progression of moderate Alzheimer s disease using its AMBAR treatment protocol Grifols presents AMBAR (Alzheimer Management by Albumin Replacement)
Grifols demonstrates a significant reduction (61%) in the progression of moderate Alzheimer s disease using its AMBAR treatment protocol Grifols presents AMBAR (Alzheimer Management by Albumin Replacement)
Klaus Romero MD MS FCP. (Supported by the CAMD AD Modeling & Simulation Team)
 Baseline ICV-adjusted Hippocampal Volume as a Biomarker for Enrichment in Alzheimer s Disease Trials Co-Chairs: Patricia Cole (Takeda) & Derek Hill (IXICO) A Modeling Approach to Demonstrate Trial Enrichment
Baseline ICV-adjusted Hippocampal Volume as a Biomarker for Enrichment in Alzheimer s Disease Trials Co-Chairs: Patricia Cole (Takeda) & Derek Hill (IXICO) A Modeling Approach to Demonstrate Trial Enrichment
Introducing MN-166 Multiple Sclerosis. July 9, 2008
 Introducing MN-166 A New Treatment Paradigm for Multiple Sclerosis July 9, 2008 MediciNova, Inc. 2008 Forward-Looking Statements Statements in this presentation that are not historical in nature constitute
Introducing MN-166 A New Treatment Paradigm for Multiple Sclerosis July 9, 2008 MediciNova, Inc. 2008 Forward-Looking Statements Statements in this presentation that are not historical in nature constitute
BioArctic AB Interim Report Jan Sep Jan Mattsson, CFO November 8, 2018
 BioArctic AB Interim Report Jan Sep 2018 Gunilla Osswald, CEO Nasdaq Stockholm: BIOA B Jan Mattsson, CFO November 8, 2018 Disclaimer This presentation has been prepared and produced by BioArctic AB (publ)
BioArctic AB Interim Report Jan Sep 2018 Gunilla Osswald, CEO Nasdaq Stockholm: BIOA B Jan Mattsson, CFO November 8, 2018 Disclaimer This presentation has been prepared and produced by BioArctic AB (publ)
LATEST RESEARCH IN ALZHEIMER S DISEASE THERAPIES
 LATEST RESEARCH IN ALZHEIMER S DISEASE THERAPIES Associate Professor Michael Woodward Austin Health Alzheimer s Australia national conference Brisbane May 2011 1 Third generation immunotherapy Beyond active
LATEST RESEARCH IN ALZHEIMER S DISEASE THERAPIES Associate Professor Michael Woodward Austin Health Alzheimer s Australia national conference Brisbane May 2011 1 Third generation immunotherapy Beyond active
Harmonizing Regulatory Requirements to Benefit Alzheimer s Disease Patients:
 Harmonizing Regulatory Requirements to Benefit Alzheimer s Disease Patients: Lessons learned from Critical Path Institute Diane Stephenson, PhD Executive Director, Coalition Against Major Diseases Critical
Harmonizing Regulatory Requirements to Benefit Alzheimer s Disease Patients: Lessons learned from Critical Path Institute Diane Stephenson, PhD Executive Director, Coalition Against Major Diseases Critical
Immunogenicity of Therapeutic Proteins. Steven J Swanson, Ph.D. Executive Director, Clinical Immunology
 Immunogenicity of Therapeutic Proteins Steven J Swanson, Ph.D. Executive Director, Clinical Immunology swanson@amgen.com Causes of Immunogenicity Sequence differences between therapeutic protein and endogenous
Immunogenicity of Therapeutic Proteins Steven J Swanson, Ph.D. Executive Director, Clinical Immunology swanson@amgen.com Causes of Immunogenicity Sequence differences between therapeutic protein and endogenous
BioArctic Gunilla Osswald, CEO December 5, Carnegie Nordic Healthcare Day
 BioArctic Gunilla Osswald, CEO December 5, 2017 Carnegie Nordic Healthcare Day Disclaimer This presentation has been prepared and produced by BioArctic AB (publ) ( BioArctic ) solely for the benefit of
BioArctic Gunilla Osswald, CEO December 5, 2017 Carnegie Nordic Healthcare Day Disclaimer This presentation has been prepared and produced by BioArctic AB (publ) ( BioArctic ) solely for the benefit of
LETTER OF INTENT Rapid Response: Canada 2019 Parkinson s & Related Diseases
 LETTER OF INTENT Rapid Response: Canada 2019 Parkinson s & Related Diseases DEADLINE: Wednesday, August 1, 2018, 2:00pm EDT Applicants will be notified of Proposal invitations in September 2018. This Letter
LETTER OF INTENT Rapid Response: Canada 2019 Parkinson s & Related Diseases DEADLINE: Wednesday, August 1, 2018, 2:00pm EDT Applicants will be notified of Proposal invitations in September 2018. This Letter
Novavax RSV F Vaccine is composed of a recombinant near full length F protein
 Magnitude and Durability of Anti-F IgG and Palivizumab-Competitive Antibody (PCA) Responses One Year Following Immunization with RSV F Nanoparticle Vaccine Adjuvanted with Aluminum Phosphate, or a Novel
Magnitude and Durability of Anti-F IgG and Palivizumab-Competitive Antibody (PCA) Responses One Year Following Immunization with RSV F Nanoparticle Vaccine Adjuvanted with Aluminum Phosphate, or a Novel
Biomarkers in the differential diagnosis of Dementia: which role for Omics? Sergio Bernardini, Biochimica Clinica Tor Vergata
 Biomarkers in the differential diagnosis of Dementia: which role for Omics? Sergio Bernardini, Biochimica Clinica Tor Vergata FTD VaD AD a-syn Alzheimer s disease Slowly progressive memory loss and dementia
Biomarkers in the differential diagnosis of Dementia: which role for Omics? Sergio Bernardini, Biochimica Clinica Tor Vergata FTD VaD AD a-syn Alzheimer s disease Slowly progressive memory loss and dementia
From Biomarkers to Diagnostics: Applications from Target Engagement to Patient Stratification
 Session: Wednesday February 22, 2017 at 08.40-09:10 am From Biomarkers to Diagnostics: Applications from Target Engagement to Patient Stratification Andrew Satlin, M.D. EVP Eisai Neurology Business Group
Session: Wednesday February 22, 2017 at 08.40-09:10 am From Biomarkers to Diagnostics: Applications from Target Engagement to Patient Stratification Andrew Satlin, M.D. EVP Eisai Neurology Business Group
Actinogen Medical Limited (ASX: ACW)
 Actinogen Medical Limited (ASX: ACW) July 5, 2016 Target Price: A$0.27 Recent Price: A$0.07 Market Data Fiscal Year June Industry BioTech Market Cap A$42.5M Price/Earnings (ttm) N/A Price/Book (mrq) 2.9x
Actinogen Medical Limited (ASX: ACW) July 5, 2016 Target Price: A$0.27 Recent Price: A$0.07 Market Data Fiscal Year June Industry BioTech Market Cap A$42.5M Price/Earnings (ttm) N/A Price/Book (mrq) 2.9x
Supplementary Figure 1. APP cleavage assay. HEK293 cells were transfected with various
 Supplementary Figure 1. APP cleavage assay. HEK293 cells were transfected with various GST-tagged N-terminal truncated APP fragments including GST-APP full-length (FL), APP (123-695), APP (189-695), or
Supplementary Figure 1. APP cleavage assay. HEK293 cells were transfected with various GST-tagged N-terminal truncated APP fragments including GST-APP full-length (FL), APP (123-695), APP (189-695), or
Data Sharing and Models in Pre(Non)- Competitive Space in Addressing Unmet Medical Needs. Critical Path Institute s Alzheimers Drug Development Tool
 Data Sharing and Models in Pre(Non)- Competitive Space in Addressing Unmet Medical Needs Critical Path Institute s Alzheimers Drug Development Tool Vikram Sinha, PhD Director, Division of Pharmacometrics
Data Sharing and Models in Pre(Non)- Competitive Space in Addressing Unmet Medical Needs Critical Path Institute s Alzheimers Drug Development Tool Vikram Sinha, PhD Director, Division of Pharmacometrics
Update on Alzheimer s Disease Therapy and Prevention Strategies
 ANNUAL REVIEWS Further Click here to view this article's online features: Download figures as PPT slides Navigate linked references Download citations Explore related articles Search keywords Update on
ANNUAL REVIEWS Further Click here to view this article's online features: Download figures as PPT slides Navigate linked references Download citations Explore related articles Search keywords Update on
TARGET VALIDATION. Maaike Everts, PhD (with slides from Dr. Suto)
 TARGET VALIDATION Maaike Everts, PhD (with slides from Dr. Suto) Drug Discovery & Development Source: http://dlab.cl/molecular-design/drug-discovery-phases/ How do you identify a target? Target: the naturally
TARGET VALIDATION Maaike Everts, PhD (with slides from Dr. Suto) Drug Discovery & Development Source: http://dlab.cl/molecular-design/drug-discovery-phases/ How do you identify a target? Target: the naturally
MY PERSONAL MEMORIES OF RITA LEVI-MONTALCINI G. NISTICÒ
 Vol. 2, no. 1, 1-3 (2013) MY PERSONAL MEMORIES OF RITA LEVI-MONTALCINI G. NISTICÒ General Director of the European Brain Research Institute (EBRI, Rita Levi-Montalcini Foundation) Vol. 2, no. 1, 5-17 (2013)
Vol. 2, no. 1, 1-3 (2013) MY PERSONAL MEMORIES OF RITA LEVI-MONTALCINI G. NISTICÒ General Director of the European Brain Research Institute (EBRI, Rita Levi-Montalcini Foundation) Vol. 2, no. 1, 5-17 (2013)
Immunotherapeutic Approaches for Alzheimer s Disease
 Immunotherapeutic Approaches for Alzheimer s Disease Thomas Wisniewski 1,2,3, * and Fernando Goñi 1 1 Department of Neurology, Center for Cognitive Neurology 2 Department of Pathology 3 Department of Psychiatry
Immunotherapeutic Approaches for Alzheimer s Disease Thomas Wisniewski 1,2,3, * and Fernando Goñi 1 1 Department of Neurology, Center for Cognitive Neurology 2 Department of Pathology 3 Department of Psychiatry
CENTRAL NERVOUS SYSTEM (CNS) BIOMARKERS: TECHNOLOGIES AND GLOBAL MARKETS
 CENTRAL NERVOUS SYSTEM (CNS) : TECHNOLOGIES AND GLOBAL MARKETS BIO074C March 2016 Jackson Highsmith Project Analyst ISBN: 1-62296-246-X BCC Research 49 Walnut Park, Building 2 Wellesley, MA 02481 USA 866-285-7215
CENTRAL NERVOUS SYSTEM (CNS) : TECHNOLOGIES AND GLOBAL MARKETS BIO074C March 2016 Jackson Highsmith Project Analyst ISBN: 1-62296-246-X BCC Research 49 Walnut Park, Building 2 Wellesley, MA 02481 USA 866-285-7215
Stealth BioTherapeutics Mission:
 The following is a summary of a live presentation offered through joint collaboration with UMDF, MitoAction and the Foundation for Mitochondrial Medicine to the mitochondrial disease patient and family
The following is a summary of a live presentation offered through joint collaboration with UMDF, MitoAction and the Foundation for Mitochondrial Medicine to the mitochondrial disease patient and family
Alzheimer s Disease. Joachim Herz. STARS January 11, 2010
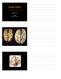 Alzheimer s Disease Joachim Herz STARS January 11, 2010 1 The Hallmarks of Alzheimer Disease Amyloid Plaques Neurofibrillary Tangles From: Medical Library of Utah 1906 First description of Alzheimer s
Alzheimer s Disease Joachim Herz STARS January 11, 2010 1 The Hallmarks of Alzheimer Disease Amyloid Plaques Neurofibrillary Tangles From: Medical Library of Utah 1906 First description of Alzheimer s
Treating and Preventing Infectious Disease. November 2011 Nasdaq: INHX
 Treating and Preventing Infectious Disease November 2011 Nasdaq: INHX Safe Harbor This presentation contains forward looking statements about Inhibitex and its business, business prospects, strategy and
Treating and Preventing Infectious Disease November 2011 Nasdaq: INHX Safe Harbor This presentation contains forward looking statements about Inhibitex and its business, business prospects, strategy and
CNS Gene Regulation Platform
 CNS Gene Regulation Platform 2018 Forward Looking Statements This presentation contains forward-looking statements within the meaning of the "safe harbor" provisions of the Private Securities Litigation
CNS Gene Regulation Platform 2018 Forward Looking Statements This presentation contains forward-looking statements within the meaning of the "safe harbor" provisions of the Private Securities Litigation
Plenary Session: An update of brain circuits in Parkinson s and Deep Brain Stimulation
 Plenary Session: An update of brain circuits in Parkinson s and Deep Brain Stimulation Deep Brain Stimulation Andres M Lozano, University of Toronto Deep brain stimulation is the delivery of an electrical
Plenary Session: An update of brain circuits in Parkinson s and Deep Brain Stimulation Deep Brain Stimulation Andres M Lozano, University of Toronto Deep brain stimulation is the delivery of an electrical
FDA Perspective on the Preclinical Development of Cancer Vaccines
 FDA Perspective on the Preclinical Development of Cancer Vaccines Richard D. McFarland Ph.D., M.D. Medical Officer CBER/OCTGT/DCEPT mcfarlandr@cber.fda.gov Cancer Vaccine Clinical Trials Workshop Alexandria,
FDA Perspective on the Preclinical Development of Cancer Vaccines Richard D. McFarland Ph.D., M.D. Medical Officer CBER/OCTGT/DCEPT mcfarlandr@cber.fda.gov Cancer Vaccine Clinical Trials Workshop Alexandria,
Passive vaccination as a global strategy for preventing RSV disease in infants. Filip Dubovsky MD MPH FAAP MedImmune March 2016
 Passive vaccination as a global strategy for preventing RSV disease in infants Filip Dubovsky MD MPH FAAP MedImmune March 2016 Outline for Presentation Rationale for passive immunization for RSV prophylaxis
Passive vaccination as a global strategy for preventing RSV disease in infants Filip Dubovsky MD MPH FAAP MedImmune March 2016 Outline for Presentation Rationale for passive immunization for RSV prophylaxis
Contract Research Offering
 Contract Research Offering Helping you understand the therapeutic in vivo effects of your Alzheimer drug candidates the partner you can trust to help you understand as fast as possible the therapeutic
Contract Research Offering Helping you understand the therapeutic in vivo effects of your Alzheimer drug candidates the partner you can trust to help you understand as fast as possible the therapeutic
DepoVax TM : A novel delivery formulation for cancer immunotherapy and infectious disease vaccines
 DepoVax TM : A novel delivery formulation for cancer immunotherapy and infectious disease vaccines May 10, 2017 The DepoVax Platform A patented oil-based formulation NOT an adjuvant Creates powerful vaccines
DepoVax TM : A novel delivery formulation for cancer immunotherapy and infectious disease vaccines May 10, 2017 The DepoVax Platform A patented oil-based formulation NOT an adjuvant Creates powerful vaccines
Clinical studies. BARBORA VLKOVÁ INSTITUTE OF MOLECULAR BIOMEDICINE
 Clinical studies BARBORA VLKOVÁ INSTITUTE OF MOLECULAR BIOMEDICINE www.imbm.sk barboravlk@gmail.com Biomedical research 1. Basic research a. In vitro = cell cultures b. In vivo = experimental animals c.
Clinical studies BARBORA VLKOVÁ INSTITUTE OF MOLECULAR BIOMEDICINE www.imbm.sk barboravlk@gmail.com Biomedical research 1. Basic research a. In vitro = cell cultures b. In vivo = experimental animals c.
Clinical Trials in the Age of Technology
 Clinical Trials in the Age of Technology Christopher G. Tarolli, M.D. Senior Instructor of Neurology Center for Health + Technology and Department of Neurology SCORE Seminar 6/5/2018 Disclosures I receive
Clinical Trials in the Age of Technology Christopher G. Tarolli, M.D. Senior Instructor of Neurology Center for Health + Technology and Department of Neurology SCORE Seminar 6/5/2018 Disclosures I receive
Do anti-amyloid beta protein antibody cross reactivities confound Alzheimer disease research?
 Hunter and Brayne Journal of Negative Results in BioMedicine (2017) 16:1 DOI 10.1186/s12952-017-0066-3 MINI-REVIEW Open Access Do anti-amyloid beta protein antibody cross reactivities confound Alzheimer
Hunter and Brayne Journal of Negative Results in BioMedicine (2017) 16:1 DOI 10.1186/s12952-017-0066-3 MINI-REVIEW Open Access Do anti-amyloid beta protein antibody cross reactivities confound Alzheimer
Lessons Learned from Alzheimer Disease: Clinical Trials with Negative Outcomes
 Citation: Clin Transl Sci (2018) 11, 147 152; C 2017 ASCPT. All rights reserved doi:10.1111/cts.12491 REVIEW Lessons Learned from Alzheimer Disease: Clinical Trials with Negative Outcomes Jeffrey INTRODUCTION
Citation: Clin Transl Sci (2018) 11, 147 152; C 2017 ASCPT. All rights reserved doi:10.1111/cts.12491 REVIEW Lessons Learned from Alzheimer Disease: Clinical Trials with Negative Outcomes Jeffrey INTRODUCTION
There are currently 4 US Food and Drug
 DISEASE-MODIFYING THERAPIES IN RELAPSING-REMITTING MULTIPLE SCLEROSIS* Benjamin M. Greenberg, MD, MHS ABSTRACT Four major disease-modifying therapies are discussed within the context of relapsing and remitting
DISEASE-MODIFYING THERAPIES IN RELAPSING-REMITTING MULTIPLE SCLEROSIS* Benjamin M. Greenberg, MD, MHS ABSTRACT Four major disease-modifying therapies are discussed within the context of relapsing and remitting
EPAD Proof-of-concept Study
 EPAD Proof-of-concept Study Value Proposition for Prospective Compound Nominators Fall, 2015 EPAD Goal The European Prevention of Alzheimer's Dementia (EPAD) project aims to develop an infrastructure and
EPAD Proof-of-concept Study Value Proposition for Prospective Compound Nominators Fall, 2015 EPAD Goal The European Prevention of Alzheimer's Dementia (EPAD) project aims to develop an infrastructure and
Pfizer Program in DMD. Beth Belluscio, MD-Ph.D. Pfizer Rare Disease September 9, 2017
 Pfizer Program in DMD Beth Belluscio, MD-Ph.D. Pfizer Rare Disease September 9, 2017 Myostatin Inhibitor for the Potential Treatment of Duchenne Muscular Dystrophy Disclaimer This presentation includes
Pfizer Program in DMD Beth Belluscio, MD-Ph.D. Pfizer Rare Disease September 9, 2017 Myostatin Inhibitor for the Potential Treatment of Duchenne Muscular Dystrophy Disclaimer This presentation includes
CONCORD 2010: Presentation
 CONCORD 2010: Presentation 4 March 2010 1 Disclaimer This document includes only summary information and does not intend to be comprehensive. Facts, figures and opinions contained herein, other than historical,
CONCORD 2010: Presentation 4 March 2010 1 Disclaimer This document includes only summary information and does not intend to be comprehensive. Facts, figures and opinions contained herein, other than historical,
Collaboration for Alzheimer s Prevention Data and sample sharing in preclinical AD trials
 Collaboration for Alzheimer s Prevention Data and sample sharing in preclinical AD trials Stacie Weninger, Maria C. Carrillo, Billy Dunn, Paul S. Aisen, Randall J. Bateman, Joanne D. Kotz, Jessica B. Langbaum,
Collaboration for Alzheimer s Prevention Data and sample sharing in preclinical AD trials Stacie Weninger, Maria C. Carrillo, Billy Dunn, Paul S. Aisen, Randall J. Bateman, Joanne D. Kotz, Jessica B. Langbaum,
Biologic Drug Delivery Across the Blood-Brain Barrier with IgG Fusion Proteins. William M. Pardridge, M.D. University of California ArmaGen, Inc.
 Biologic Drug Delivery Across the Blood-Brain Barrier with IgG Fusion Proteins William M. Pardridge, M.D. University of California ArmaGen, Inc. 1 The Brain and Biotechnology In 2017, there are no biologics
Biologic Drug Delivery Across the Blood-Brain Barrier with IgG Fusion Proteins William M. Pardridge, M.D. University of California ArmaGen, Inc. 1 The Brain and Biotechnology In 2017, there are no biologics
Biogen Idec and Collaborative Brain Research. Dominic Paes PhD EU Medical Sciences, Neurology Zug, Switzerland
 Biogen Idec and Collaborative Brain Research Dominic Paes PhD EU Medical Sciences, Neurology Zug, Switzerland AREAS OF HIGH UNMET MEDICAL NEED NEUROLOGY IMMUNOLOGY HEMATOLOGY MS - Repair / Progressive
Biogen Idec and Collaborative Brain Research Dominic Paes PhD EU Medical Sciences, Neurology Zug, Switzerland AREAS OF HIGH UNMET MEDICAL NEED NEUROLOGY IMMUNOLOGY HEMATOLOGY MS - Repair / Progressive
Immunogenicity. How to deal with? Nathalie Macé Sanofi, Biomarkers & Biological analyses Unit
 Immunogenicity How to deal with? Nathalie Macé Sanofi, Biomarkers & Biological analyses Unit Club Phase I, 22 March 2016 1 Outline Introduction to immunogenicity Analytical challenges for immunogenicity
Immunogenicity How to deal with? Nathalie Macé Sanofi, Biomarkers & Biological analyses Unit Club Phase I, 22 March 2016 1 Outline Introduction to immunogenicity Analytical challenges for immunogenicity
Successful Modeling and Simulation in a Consortium: A Recipe for Success
 Successful Modeling and Simulation in a Consortium: A Recipe for Success Brian Corrigan, PhD Head, Clinical Pharmacology, Global Product Development Pfizer, Groton, CT USA November 6, 2017 1 Conflict of
Successful Modeling and Simulation in a Consortium: A Recipe for Success Brian Corrigan, PhD Head, Clinical Pharmacology, Global Product Development Pfizer, Groton, CT USA November 6, 2017 1 Conflict of
Monoclonal Antibodies for Treatment and Prevention of HIV-1 CROI Charles Boucher Erasmus MC
 Monoclonal Antibodies for Treatment and Prevention of HIV-1 CROI 2018 Charles Boucher Erasmus MC Introduction: Human monoclonal antibodies Monoclonal Antibodies: Are identical immunoglobulins made by clone
Monoclonal Antibodies for Treatment and Prevention of HIV-1 CROI 2018 Charles Boucher Erasmus MC Introduction: Human monoclonal antibodies Monoclonal Antibodies: Are identical immunoglobulins made by clone
EARLY DIAGNOSIS OF BRAIN DISEASES via liquid biopsy, based on mirna patterns in microglial microvesicles
 EARLY DIAGNOSIS OF BRAIN DISEASES via liquid biopsy, based on mirna patterns in microglial microvesicles BrainDTech team Dr. Fabio Bianco, PhD Chief Executive Officier PhD in Pharmacology, Degree in Biotech
EARLY DIAGNOSIS OF BRAIN DISEASES via liquid biopsy, based on mirna patterns in microglial microvesicles BrainDTech team Dr. Fabio Bianco, PhD Chief Executive Officier PhD in Pharmacology, Degree in Biotech
Subgroup Identification
 Assessing and Communicating Heterogeneity of Treatment Effects (HTE) for Patient Subpopulations: Challenges and Opportunities Subgroup Identification The Hardest Problem There Is Stephen J. Ruberg, PhD
Assessing and Communicating Heterogeneity of Treatment Effects (HTE) for Patient Subpopulations: Challenges and Opportunities Subgroup Identification The Hardest Problem There Is Stephen J. Ruberg, PhD
Considering a New Paradigm for Alzheimer s Disease Research
 The Humane Society Institute for Science and Policy Animal Studies Repository 8-2014 Considering a New Paradigm for Alzheimer s Disease Research Gillian R. Langley glangley@hsi.org Follow this and additional
The Humane Society Institute for Science and Policy Animal Studies Repository 8-2014 Considering a New Paradigm for Alzheimer s Disease Research Gillian R. Langley glangley@hsi.org Follow this and additional
Ethical and Practical Dilemmas of Research with Nonhuman Primates
 N.A.S. / I.O.M. U.S. and European Animal Research Regulations: Impact on Neuroscience Research Ethical and Practical Dilemmas of Research with Nonhuman Primates Stuart Zola Yerkes National Primate Research
N.A.S. / I.O.M. U.S. and European Animal Research Regulations: Impact on Neuroscience Research Ethical and Practical Dilemmas of Research with Nonhuman Primates Stuart Zola Yerkes National Primate Research
Immunogenicity Assay Strategies for Antibody-Drug Conjugates
 Immunogenicity Assay Strategies for Antibody-Drug Conjugates 8th World ADC Conference, San Diego 20 Sep 2017 Seema Kumar, PhD Associate Scientific Director Global Early Development (GED) EMD Serono Research
Immunogenicity Assay Strategies for Antibody-Drug Conjugates 8th World ADC Conference, San Diego 20 Sep 2017 Seema Kumar, PhD Associate Scientific Director Global Early Development (GED) EMD Serono Research
Methods in Clinical Cancer Research Workshop Format for Protocol Concept Synopsis Sheet (blank) SYNOPSIS
 B Methods in Clinical Cancer Research Workshop Format for Protocol Concept Synopsis Sheet (blank) elow is the format to follow for the concept sheet for protocols. It is based on the International Committee
B Methods in Clinical Cancer Research Workshop Format for Protocol Concept Synopsis Sheet (blank) elow is the format to follow for the concept sheet for protocols. It is based on the International Committee
Regulatory Perspective on Developing Long Acting ARVs for HIV Treatment/Prevention. FDA Division of Antiviral Products
 Regulatory Perspective on Developing Long Acting ARVs for HIV Treatment/Prevention FDA Division of Antiviral Products None Financial Disclosures FDA Disclaimer The views in this presentation represent
Regulatory Perspective on Developing Long Acting ARVs for HIV Treatment/Prevention FDA Division of Antiviral Products None Financial Disclosures FDA Disclaimer The views in this presentation represent
Grifols has acquired 51% of the capital of Araclon Biotech to ensure the viability of the purchased company projects
 Alzheimer s disease acquires a growing role in Grifols R&D Grifols has acquired 51% of the capital of Araclon Biotech to ensure the viability of the purchased company projects Araclon Biotech, established
Alzheimer s disease acquires a growing role in Grifols R&D Grifols has acquired 51% of the capital of Araclon Biotech to ensure the viability of the purchased company projects Araclon Biotech, established
Alzheimer s Disease. Table of Contents. Differential processing of APP Hyperphosphorylation of Tau
 Alzheimer s Disease Alzheimer s disease (AD) is one of the most common neurodegenerative diseases worldwide. Clinically, it is characterized by the presence of extracellular amyloid plaques and intracellular
Alzheimer s Disease Alzheimer s disease (AD) is one of the most common neurodegenerative diseases worldwide. Clinically, it is characterized by the presence of extracellular amyloid plaques and intracellular
STRATIFICATION OF PRE-SYMPTOMATIC AND COGNITIVELY NORMAL INDIVIDUALS USING POLYGENIC SCORING
 The UK s innovation agency Maryam Shoai PhD Richard Pither PhD Valentina Escott-Price PhD, Simon M Laws PhD, Harald Hampel MD PhD, Simone Lista PhD, Rik Vandenberghe PhD, Isabelle Cleynen PhD, David Irwin
The UK s innovation agency Maryam Shoai PhD Richard Pither PhD Valentina Escott-Price PhD, Simon M Laws PhD, Harald Hampel MD PhD, Simone Lista PhD, Rik Vandenberghe PhD, Isabelle Cleynen PhD, David Irwin
Biogen Idec Neurology Pipeline. Alfred Sandrock, MD, PhD SVP, Neurology Research & Development
 Biogen Idec Neurology Pipeline Alfred Sandrock, MD, PhD SVP, Neurology Research & Development March 25, 2009 Robust Neurology Pipeline Neurology Multiple Sclerosis Discovery Pre-Clinical Phase 1 Phase
Biogen Idec Neurology Pipeline Alfred Sandrock, MD, PhD SVP, Neurology Research & Development March 25, 2009 Robust Neurology Pipeline Neurology Multiple Sclerosis Discovery Pre-Clinical Phase 1 Phase
Designed Zinc Finger Protein Transcription Factors for Single-Gene Regulation Throughout the Central Nervous System
 Designed Zinc Finger Protein Transcription Factors for Single-Gene Regulation Throughout the Central Nervous System Bryan J Zeitler 1 *, Sarah L DeVos 2 *, Susanne K Wegmann 2 *, Kimberly Marlen 1, Qi
Designed Zinc Finger Protein Transcription Factors for Single-Gene Regulation Throughout the Central Nervous System Bryan J Zeitler 1 *, Sarah L DeVos 2 *, Susanne K Wegmann 2 *, Kimberly Marlen 1, Qi
Guideline for the quality, safety and efficacy of follow-on biological medicinal products
 Guideline for the quality, safety and efficacy of follow-on biological medicinal products 1. Introduction A follow-on biological medicinal product (hereinafter referred to as FOBMP) is considered as a
Guideline for the quality, safety and efficacy of follow-on biological medicinal products 1. Introduction A follow-on biological medicinal product (hereinafter referred to as FOBMP) is considered as a
Regulatory Challenges for the Licensure of Future Vaccines
 Regulatory Challenges for the Licensure of Future Vaccines Tong Wu, Ph.D. Bacterial & Combination Vaccine Division, BGTD, Health Canada June 26-29, 2018, Seoul, Korea, the Global Bio Conference 1 Disclaimer
Regulatory Challenges for the Licensure of Future Vaccines Tong Wu, Ph.D. Bacterial & Combination Vaccine Division, BGTD, Health Canada June 26-29, 2018, Seoul, Korea, the Global Bio Conference 1 Disclaimer
Facilitating the Use of Imaging Biomarkers in Therapeutic Clinical Trials. Michael Graham, PhD, MD President, SNM Co-chair, Clinical Trials Network
 Facilitating the Use of Imaging Biomarkers in Therapeutic Clinical Trials Michael Graham, PhD, MD President, SNM Co-chair, Clinical Trials Network Facilitating the Use of Imaging Biomarkers in Therapeutic
Facilitating the Use of Imaging Biomarkers in Therapeutic Clinical Trials Michael Graham, PhD, MD President, SNM Co-chair, Clinical Trials Network Facilitating the Use of Imaging Biomarkers in Therapeutic
FDA Draft Guidance on Immunogenicity Testing
 FDA Draft Guidance on Immunogenicity Testing Susan Kirshner, Ph.D. Associate Chief, Laboratory of Immunology Division of Therapeutic Proteins OBP/CDER/FDA EBF 2010 Guidance for Industry Assay Development
FDA Draft Guidance on Immunogenicity Testing Susan Kirshner, Ph.D. Associate Chief, Laboratory of Immunology Division of Therapeutic Proteins OBP/CDER/FDA EBF 2010 Guidance for Industry Assay Development
Clinical Evaluation Phases 1,2,3,4
 Clinical Evaluation Phases 1,2,3,4 Matt Laurens, MD MPH Associate Professor of Pediatrics Center for Vaccine Development Institute for Global Health University of Maryland School of Medicine February 1,
Clinical Evaluation Phases 1,2,3,4 Matt Laurens, MD MPH Associate Professor of Pediatrics Center for Vaccine Development Institute for Global Health University of Maryland School of Medicine February 1,
chronic leukemia lymphoma myeloma differentiated 14 September 1999 Transformed Pre- Ig Surface Surface Secreted B- ALL Macroglobulinemia Myeloma
 Disease Usual phenotype acute leukemia precursor chronic leukemia lymphoma myeloma differentiated Pre- B-cell B-cell Transformed B-cell Plasma cell Ig Surface Surface Secreted Major malignant counterpart
Disease Usual phenotype acute leukemia precursor chronic leukemia lymphoma myeloma differentiated Pre- B-cell B-cell Transformed B-cell Plasma cell Ig Surface Surface Secreted Major malignant counterpart
Recombinant, Insect Cell-Derived RSV Nanoparticle Vaccine
 Recombinant, Insect Cell-Derived RSV Nanoparticle Vaccine Gregory Glenn Chief Medical Officer MVADS-Copenhagen 4 July 2012 1 Agenda for RSV Discussion Overview of Insect Cell Technology Respiratory Syncytial
Recombinant, Insect Cell-Derived RSV Nanoparticle Vaccine Gregory Glenn Chief Medical Officer MVADS-Copenhagen 4 July 2012 1 Agenda for RSV Discussion Overview of Insect Cell Technology Respiratory Syncytial
University of Eastern Finland (UEF) Main Research Lines
 University of Eastern Finland (UEF) Main Research Lines Asla Pitkänen, MD, PhD Epilepsy Research Laboratory A.I.Virtanen Institute for Molecular Sciences University of Eastern Finland Kuopio, Finland E-mail:
University of Eastern Finland (UEF) Main Research Lines Asla Pitkänen, MD, PhD Epilepsy Research Laboratory A.I.Virtanen Institute for Molecular Sciences University of Eastern Finland Kuopio, Finland E-mail:
PATHWAYS TO BIOMARKER QUALIFICATION AND ACCEPTANCE
 PATHWAYS TO BIOMARKER QUALIFICATION AND ACCEPTANCE CHRISTOPHER LEPTAK, M.D., PH.D. DIRECTOR, OND REGULATORY SCIENCE PROGRAM CO-DIRECTOR, BIOMARKER QUALIFICATION PROGRAM NASH Biomarkers Workshop May 5,
PATHWAYS TO BIOMARKER QUALIFICATION AND ACCEPTANCE CHRISTOPHER LEPTAK, M.D., PH.D. DIRECTOR, OND REGULATORY SCIENCE PROGRAM CO-DIRECTOR, BIOMARKER QUALIFICATION PROGRAM NASH Biomarkers Workshop May 5,
HARNESSING THE POWER OF PRECISION MEDICINE TO TREAT ALZHEIMER S DISEASE AND ALS (LOU GEHRIG S DISEASE)
 HARNESSING THE POWER OF PRECISION MEDICINE TO TREAT ALZHEIMER S DISEASE AND ALS (LOU GEHRIG S DISEASE) Toronto Stock Exchange (TSX) ticker: PMN.TO June 2017 1 Forward Looking Statement: Safe Harbor This
HARNESSING THE POWER OF PRECISION MEDICINE TO TREAT ALZHEIMER S DISEASE AND ALS (LOU GEHRIG S DISEASE) Toronto Stock Exchange (TSX) ticker: PMN.TO June 2017 1 Forward Looking Statement: Safe Harbor This
Corporate Presentation. Roberto Bellini President and Chief Executive Officer
 Corporate Presentation Roberto Bellini President and Chief Executive Officer September 2012 Forward Looking Statement Certain statements contained in this presentation, other than statements of fact that
Corporate Presentation Roberto Bellini President and Chief Executive Officer September 2012 Forward Looking Statement Certain statements contained in this presentation, other than statements of fact that
Biomarkers in Clinical Development: Implications for Personalized Medicine and Streamlining R&D in the UK and EU region
 Biomarkers in Clinical Development: Implications for Personalized Medicine and Streamlining R&D in the UK and EU region Dr. Christian Rohlff Oxford Genome Sciences Biomarkers in Clinical Development: Implications
Biomarkers in Clinical Development: Implications for Personalized Medicine and Streamlining R&D in the UK and EU region Dr. Christian Rohlff Oxford Genome Sciences Biomarkers in Clinical Development: Implications
Credit Suisse 7 th Annual European Large Cap Pharmaceutical Conference. November 6, 2007
 Credit Suisse 7 th Annual European Large Cap Pharmaceutical Conference November 6, 2007 Safe Harbor Statement This presentation contains forward-looking statements about Elan s financial condition, results
Credit Suisse 7 th Annual European Large Cap Pharmaceutical Conference November 6, 2007 Safe Harbor Statement This presentation contains forward-looking statements about Elan s financial condition, results
NDA Advisory Services Ltd
 Declaration of Interest statement: Paul Chamberlain has received Consulting fees from different companies in respect of strategic and operational advice relating to biopharmaceutical development; he is
Declaration of Interest statement: Paul Chamberlain has received Consulting fees from different companies in respect of strategic and operational advice relating to biopharmaceutical development; he is
