Penicillin: Reversible Inhibition of Forespore Septum Development in Bacillus megaterium Cells
|
|
|
- Kenneth Cross
- 6 years ago
- Views:
Transcription
1 ANTIMICROBAL AGENTS AND CHEMOTHERAPY, Dec. 1974, p Copyright American Society for Microbiology Vol. 6, No. 6 Printed in U.S.A. Penicillin: Reversible Inhibition of Forespore Septum Development in Bacillus megaterium Cells PAUL J. LAWRENCE Department of Biological Chemistry and Medicine, University of Utah Medical Center, Salt Lake City, Utah Received for publication 10 July 1974 Benzylpenicillin inhibits the development of the forespore septum in sporulating Bacillus megaterium cells. The inhibitory effect is a function of the duration of exposure to the antibiotic and is completely reversible by penicillinase. Under the incubation conditions employed, less than 20% of the covalently bound antibiotic is released from the cells. The penicillin which remains bound to the cells after treatment with penicillinase may be necessary but is not sufficient for the effect; unbound antibiotic in the sporulation medium is also required. In addition to their bactericidal effects on susceptible organisms, penicillins at concentrations similar to those required to prevent vegetative cell division can inhibit bacterial spore formation. The latter process has been described as a modified or atypical cell division (14) which involves an orderly sequence of seven well-defined morphological stages (14). Stages II and IV of bacterial sporulation include the formation of the forespore septum and the spore cortex, respectively. Penicillin, cycloserine, bacitracin, and other antibiotics which prevent vegetative cell division by inhibiting the synthesis of functional vegetative cell wall peptidoglycan can prevent development of either the forespore septum (15) or spore cortex (12, 21), depending upon the time of addition of the antibiotic to the sporulating culture. Two intervals of enhanced specific penicillin-binding capacity are observed in sporulating bacterial cultures (17). The first is seen during the formation of the primordial cell wall or forespore septum (stage II); the second is coincident with the formation of the spore cortex (stage IV). Asporogenous mutants blocked prior to the development of the forespore septum (stage II) fail to exhibit either interval of enhanced specific penicillin binding capacity. Similarly, asporogenous mutants blocked after stage II, but before stage IV, exhibit only the first peak of enhanced specific binding capacity (22). The above observations, when combined with those presented below, suggest that penicillins inhibit bacterial forespore septum formation by a mechanism similar to that believed to be responsible for their bactericidal effect: inhibition of the terminal stages of functional cell wall 815 peptidoglycan synthesis (24). Considerable experimental evidence indicates that penicillins irreversibly inhibit both glycopeptide transpeptidase and D-alanine carboxypeptidase activity (24). Inhibition of the latter enzyme appears neither necessary nor sufficient to cause vegetative cell death (2). Several penicillin-binding proteins have been identified in detergent extracts of vegetative Bacillus subtilis cells (3, 4, 24). The same proteins are also apparently present in sporulating B. subtilis cells, although their quantities differ somewhat from that of their vegetative counterparts (1). Reasonable correlation has been observed between the concentration of penicillins required to saturate rapidly growing bacterial cells and that required for cell death (6, 8-11, 23). These investigations suggested that bound antibiotic is responsible for the bactericidal effect (compare [4 ]). Despite the obvious importance of the penicillin target proteins in bacterial sporulation, the role of covalently bound antibiotic in inhibiting this process has received little attention. Although penicillins irreversibly inhibit glycopeptide transpeptidases, the biological effect of penicillin exposure is reversible. Addition of penicillinase to a susceptible bacterial culture exposed to a lethal concentration of the antibiotic prevents cell death (6). The reversibility of penicillin inhibition of bacterial forespore septum formation has not been examined. Additional information concerning the reversibility of the biological effects of penicillins may prove useful in the identification and purification of the target protein(s) responsible for these effects. The development of covalent affinity chromatography techniques (3) permits convenient examination of these proteins which react to form a covalent bond
2 816 LAWRENCE with penicillin. Identification of the proteins primarily responsible for the biological effects is thus of critical importance. Whereas no single, clear alteration of bacterial morphology is conveniently correlated with cell death, the formation of the forespore septum can readily be identified and quantitated. For these reasons, the inhibitory effect of penicillin on the formation of the forespore septum in sporulating B. megaterium cultures and the reversibility of the effect were examined. MATERIALS AND METHODS Organism and growth conditions. B. megaterium cells (ATCC 19213) were grown in the synthetic sucrose medium of Kolodziej and Slepecky (16). The culture (300 ml) was shaken at 30 C in 2-liter Fernbach flasks on a New Brunswick gyratory shaker. At appropriate intervals, 10-ml samples were removed and added to 100-ml Erlenmeyer flasks containing either benzylpenicillin (50 pug/ml, final concentration) or an equivalent volume of water. Unlabeled potassium benzylpenicillin and [14C]benzylpenicillin were obtained from Sigma and Amersham Searle, respectively. The cells were shaken at 30 C, and portions were removed for septum counting by the staining procedure of Gordon and Murrell (13). Effect of penicilhinase on forespore septum formation. Cells were grown as indicated above. At appropriate intervals, 10-ml samples were removed from the Fernbach flasks and added to 100-ml Erlenmeyer flasks containing either penicillin or water. Penicillinase (20,000 U) was added to individual flasks after 10, 20, 30, or 60 min. Samples were removed from each flask at appropriate intervals, and forespore septa were counted. A non-penicillinasetreated culture and a culture to which only penicillinase was added were included as controls. Effects of penicillinase on the binding of [14C]benzylpendllhin to sporulating B. megaterium cells. Cells were grown as described above. When approximately 5% of the cells contained forespore septa, the cells were harvested and washed with buffer containing: tris(hydroxymethyl)aminomethane, 0.01 M; succinate, 0.04 M; and magnesium acetate, 0.01 M; adjusted to ph 7.2. Washed cells were suspended in a volume of buffer (ml) four times their wet weight (in grams), and the suspension was divided into two equal fractions. One was preincubated for 5 min at 0 C with unlabeled benzylpenicillin (25 mg/ml), and the other was untreated. Thereafter, both samples were incubated with [14C ]benzylpenicillin (10 ;&g/ml, final concentration) for 5 min at 0 C. Both samples were washed to remove unbound radioactive antibiotic and resuspended in buffer at 0 C. Samples were removed for determination of ["C]benzylpenicillin bound to the cells (17). The cells were incubated with radioactive antibiotic at 0 C to inhibit further sporulation development. Independent studies indicate that both sporulating and vegetative B. subtilis cells bind the same quantity of radioactive antibiotic at 0, 30, or 37 C. (T. E. Hamilton and P. J. Lawrence, manuscript in preparation). The culture labeled with ANTMCROB. AG. CHEMOTHER. ["IC Jbenzylpenicillin was then divided into two equal fractions. Penicillinase (20,000 U) was added to one sample, and the other was untreated. The cells pretreated with unlabeled penicillin, those treated with [14C]benzylpenicillin alone, and the fraction treated with both ["CC]benzylpenicillin and penicillinase were shaken at 30 C. After, 5, 10, 20, and 30 min, 200-uliter samples were removed, the cells were washed three times with the above buffer, and the quantity of [14C]benzylpenicillin bound to the cells was determined (17). All determinations were performed in triplicate. Corrections were made for the quantity of penicillin bound nonspecifically to the cells by subtracting from the total ["4C lbenzylpenicillin bound to the cells that quantity of radioactive antibiotic bound in the presence of unlabeled benzylpenicillin (25 mg/ml). Protein was determined by the method of Lowry et al. (20). RESULTS Effect of benzylpenicillin on forespore septum formation in B. megaterium cells. Under the growth conditions employed, forespore septa were distinguishable after approximately 10 h of incubation at 30 C. Forespore septation continued until approximately 85% of the cells in the culture contained clearly recognized forespore septa (Fig. 1). Thereafter, cells containing forespore septa decreased in number as the sporulation process continued and forespores, sporangia, and mature spores were formed. If benzylpenicillin at a final concentration of 50,ug/ml, were added to the culture at 10 h, subsequent forespore septum formation was completely prevented (Fig. 1) as were all later stages of sporulation (data not presented). Penicillin at lower concentrations also prevented forespore septum formation, but in the initial experiments a comparatively high concentration of antibiotic was utilized to prevent hydrolysis of penicillin by cell-bound,-lactamase activity over the interval of study (T. E. Hamilton and P. J. Lawrence, unpublished data). The degree of inhibition of forespore septum formation was a function of the duration of penicillin exposure (Fig. 2). Addition of penicillin to the culture at 10 h completely inhibited subsequent forespore septum formation for at least 4 h (after which the experiments were terminated). A 14-h culture which had been exposed to penicillin for the last 3 h of growth produced far fewer forespore septum containing cells than an untreated culture. A similar inhibition of forespore septum formation by benzylpenicillin was seen in 13-, 12-, and 11-h cultures during the entire period of incubation in the presence of antibiotic (Fig. 2). Reversibility of penicillin inhibition of bacterial forespore septum formation. The culture was incubated at 30 C until approximately
3 VOL. 6, 1974 INCUBATION TIME (hours) FiG. 1. Effect of benzylpenicillin on forespore septum formation in stage I B. megaterium cells. Cells were grown and exposed to benzylpenicillin (50 Ag/ ml) as described in Materials and Methods. Samples (10 ml) were removed at the indicated times, stained, and forespore septa counted. Symbols: *, forespore septa in untreated cells; 0, forespore septa in cells exposed to benzylpenicillin. 10%o of the cells contained forespore septa (10 h). Several 10-ml samples were removed and added to individual Erlenmeyer flasks containing unlabeled benzylpenicillin (50 ug/ml, final concentration). Control flasks containing no antibiotic, or penicillinase (20,000 U) in.the absence of antibiotic, were also utilized. As described previously (Fig. 1), penicillin completely inhibited the formation of forespore septa, but penicillinase alone had no effect on the formation of this structure (Fig. 3). The addition of penicillinase to the antibiotic-treated cells, however, at 10, 20, 30, or 60 min after introduction of the antibiotic, completely mitigated the penicillin inhibition of forespore septum formation. The rate of forespore septum formation after the addition of penicillinase to the antibiotictreated cultures was at least as great as that of the control cultures (Table 1). Reversibility of the binding of ["Cjbenzylpenicillin to stage H B. megaterium cells. The chemistry of binding of ["4C ]benzylpenicillin to sporulating B. megaterium cells (17) is similar to that observed with vegetative B. subtilis cells or their membrane fragments (18, INHIBION OF FORESPORE SEPTUM DEVELOPMENT ). Stage II B. megaterium cells were incubated with [14C ]benzylpenicillin at a final antibiotic concentration of 10,g/ml at 0 C. The same amount of radioactive antibiotic is covalently bound to sporulating cells at 0 as at 30 C (T. E. Hamilton and P. J. Lawrence, manuscript in preparation). When the treated cells were washed free of unbound antibiotic and incubated at 0 C, no significant release of bound radioactive antibiotic was detected (Fig. 4). At 30 C, less than 20% of the bound antibiotic was removed from the cells over a 30-min interval. The kinetics of antibiotic removal was not significantly different in the presence of penicillinase (Fig. 4). The quantity of the radioactive antibiotic which remains bound to the cells ( >80%) at 30 C after a 30-min incubation may be necessary to inhibit forespore septum TIME OF INCUBATION WITH PENICILLIN (hrs) FIG. 2. Effect of duration of penicillin exposure on forespore septum development in B. megaterium cells. Cells were grown as described in Materials and Methods. At the times designated below, 10-mI samples were added to flasks containing benzylpenicillin (50 sg/ml) and grown in the presence of the antibiotic for the indicated interval. Symbols: 0, 14-h cells, penicillin added to separate samples at 10, 11, 12, and 13 h; 0, 13-h cells, penicillin added to separate samples at 9, 10, 11, and 12 h; A, 12-h cells, penicillin added to separate samples at 8, 9, 10, and 11 h; A, 11-h cells, penicillin added to separate samples at 7,8, 9, and 10 h; and 0, 10-h cells, penicillin added at 6, 7, 8, and 9 h. Samples were removed from each flask at hourly intervals, and the number of cells containing forespore septa was determined.
4 818 LAWRENCE tgc0 IN. ~~~ 25~ ~ ~ INCUBATION TIME (hours) FIG. 3. Effect of penicillinase on penicillin inhibition of forespore septum formation in stage II B. megaterium cells. Cells were incubated as described in Materials and Methods for 10 h. Samples were then removed, and the incubation continued: 0, with no additions; 0, with benzylpenicillin; or 0, with the addition of penicillinase alone. Benzylpenicillin (50 sg/ml) was present in all of the remaining flasks. To these flasks, penicillinase (20,000 U) was added at the following times: A, 10 min; A, 20 min; *, 30 min; and x, 60 min. Samples were removed after a 10-h incubation and at hourly intervals thereafter for determination of forespore septa. formation, but it is clearly insufficient to cause this effect. Removal of 20%o of the covalently bound antibiotic permits forespore septum development at a rate equal to that of the control culture (Fig. 3, Table 1). Several vegetative bacilli have been shown to contain multiple penicillin-binding proteins (3, 4, 24), and the penicillin-binding proteins of vegetative B. subtilis cells appear qualitatively the same as that of their sporulating counterparts (1). The data presented in Fig. 4 do not indicate whether the penicillin released from sporulating cells is removed from a single protein or from several proteins. Excess, unbound antibiotic may be required to exert the biological effect for a number of reasons. Conceivably, excess, unbound antibiotic in the media is needed to re-penicilloylate the protein(s) from which penicillin is lost during the incubation period. Alternatively, the free antibiotic may be required to penicilloylate target proteins synthesized as the sporulation process occurs. Finally, the antibiotic may interact with its target protein in a reversible manner. The available data do not distinguish between these alternatives. ANTIMICROB. AG. CHEMOTHER. DISCUSSION Several lines of experimental evidence suggest that functional cell wall peptidoglycan biosynthesis is required for the formation of forespore septa although the septum itself apparently contains no cell wall material (14). (i) Antibiotics known to prevent vegetative cell division by inhibiting specific enzymes in the synthesis of peptidoglycan (cycloserine, baci- TABLE. 1. Effect of penicillinase on the rate of forespore septum formation in cells exposed to benzylpenicillina Addition Rate of forespore septum synthesis Penicillinase 10.5 Benzylpenicillinc 0 Benzylpenicillin and penicillinase at: 10 min min min mind 11.1 a The procedures are the same as those described in the legend to Fig. 3. brate of forespore septum synthesis: (percent cells with forespore septa at 11 h - percent cells with forespore septa at 10 h)/(total incubation time - incubation time after the addition of penicillinase). c Rate of forespore septum synthesis: (percent cells with forespore septa at 11 h - percent cells with forespore septa at 10 h)/(60 min). d Rate of forespore septum formation: (percent cells with forespore septa at 12 h - percent cells with forespore septa at 11 h)/(60 min).?.~. 10 '75 XX co 3 O' zo- 3e5 z,.~~~~~~~~~~~~~ lw I 0O TIME AFTER ADDITION OF PENICILLINASE (min) FIG. 4. Release of covalently bound ['4C]benzylpenicillin from stage II sporulating B. megaterium cells. Stage H cells were harvested, washed, and treated with ['4C]benzylpenicillin as described in Materials and Methods. The cells were washed free of excess, unbound penicillin and incubated as follows: x, 0 C; 0, 30 C, no additions; and 0, 30 C, penicillinase (20,000 U). i
5 VOL. 6, 1974 tracin, and penicillin) inhibit formation of forespore septa (15). (ii) Five proteins known to react covalently with penicillins are present in both vegetative and sporulating cells, although the quantities of the individual components differ slightly (1, 3, 4, 24). (iii) While the specific penicillin-binding capacity of a nonsporulating organism (S. aureus) remains constant in the post-logarithmic phase of growth, that of sporulating bacilli exhibits two intervals of enhanced specific penicillin-binding capacity; the first increase corresponds to the formation of the forespore septum, but the second is coincident with the formation of the spore cortex (17). Asporogenous mutants blocked prior to stage fl show neither interval of enhanced specific binding capacity (22). (iv) The intracellular concentration of a nucleotide precursor of peptidoglycan biosynthesis (uridine diphosphate-n-acetylglucosamine) rises suddenly in wild-type sporulating B. subtilis cells at the time of forespore septum formation. The INHIBITION OF FORESPORE SEPTUM DEVELOPMENT 819 increase is not observed in asporogenous mutants blocked prior to formation of the forespore septum, but is seen in a Stage II mutant blocked after formation of this structure (5). The data presented indicate that unbound penicillin in the sporulation medium is required for penicillin inhibition of forespore septum formation (Fig. 3, Table 1). In this respect, the effect of penicillin on forespore septum formation is similar to that observed in its inhibition of vegetative cell division; the destruction of unbound antibiotic results in a reversal of the biological effect (6). After treatment with penicillinase, less than 20% of the ["4C ]benzylpenicillin covalently bound to stage II cells is removed during a 30-min incubation period at 30 C (Fig. 4). Nonetheless, the rate of forespore septum formation of cells exposed to penicillin for 30 min prior to the addition of penicillinase is essentially the same as that of untreated cells (Table 1). Whereas more than 80% of the penicillin remains bound to the cells under these conditions, bound antibiotic is clearly not sufficient to inhibit forespore septum synthesis. The penicillin released from the cells may be lost by one or several of the bacterial penicillinbinding proteins thus far identified (1, 3, 4, 24). Some of these proteins are present in comparatively small amounts (1, 3, 4, 24), but may be of critical biological importance. To explain the failure of covalently bound antibiotic alone to kill logarithmically growing bacteria, it has been suggested that rapid synthesis of new penicillin target proteins permits resumption of normal growth (6). In vegetatively growing B. subtilis cells, the total penicillin-binding capacity increases approximately 135% over a 30-min period (T. E. Hamilton and P. J. Lawrence, manuscript in preparation), whereas the increase in specific binding capacity of a stage II sporulating culture is less than 15% over the same interval (19). It is also possible that bound antibiotic is required, but is itself not sufficient to exert either biological effect. The role of excess, unbound penicillin on vegetative bacterial cell division and on forespore septum formation remains to be clarified. ACKNOWLEDGMENTIS This research was supported by Public Health Service grant AI from the National Institute of Allergy and Infectious Diseases, and by a General Research Support Fund grant from the University of Utah (Salt Lake City). The technical assistance of Mark Astill is, gratefully acknowledged. LITERATURE CITED 1. Anwar, R. A., P. M. Blumberg, and J. L. Strominger Penicillin binding components in Bacillus subtilis during sporulation. J. Bacteriol. 117: Blumberg, P. M., and J. L. Strominger Inactivation of D-alanine carboxypeptidase by penicillins and cephalosphorins is not lethal in Bacillus subtilis. Proc. Nat. Acad. Sci. U.S.A. 68: Blumberg, P. M., and J. L. Strominger Isolation by covalent affinity chromatography of the penicillinbinding components from membranes of Bacillus subtilis. Proc. Nat. Acad. Sci. U.S.A. 69: Blumberg, P. M., and J. L. Strominger Five penicillin-binding components occur in Bacillus subtilis membranes. J. Biol. Chem. 247: Chow, C. T., and I. Takahashi Acid-soluble nucleotides in an asporogenous mutant of Bacillus subtilis. J. Bacteriol. 109: Cooper, P. D Site of action of radiopenicillin. Bacteriol. Rev. 20: Cooper, P. D., and D. Rowley Investigations with radioactive penicillin. Nature (London) 163: Eagle, H The binding of penicillin in relation to its cytotoxic action. J. Exp. Med. 99: Eagle, H The multiple mechanisms of penicillin resistance. J. Bacteriol. 68: Eagle, H., N. Levy, and R. Fleischman The binding of penicillin in relation to its cytotoxic action. IV. The amounts bound by bacteria at ineffective, growth inhibitory, bactericidal, and maximally effective concentrations. J. Bacteriol. 69: Edwards, J. R., and J. T. Park Correlation between growth inhibition and the binding of various penicillins and cephalosporins to Staphylococcus aureus. J. Bacteriol. 99: Fitz-James, P. C Spore formation in wild and mutant strains of B. cereus and some effects of inhibitors, p In M. J. C. Senez (ed.), Regulations chez les microorganismes. Centre National Recherche Science, Paris. 13. Gordon, R. A., and W. G. Murrell Simple method of detecting spore septum formation and synchrony of sporulation. J. Bacteriol. 93: Hitchens, A. D., and R. A. Slepecky Bacterial sporulation as a modified procaryotic cell division. Nature (London) 223: Hitchens, A. D., and R. A. Slepecky Antibiotic inhibition of the septation stage in sporulation of Bacillus megaterium. J. Bacteriol. 97:
6 820 LAWRENCE ANTIMICROB. AG. CHOAHE. 16. Kolodziej, B. J., and R. A. Slepecky Trace metal requirements for sporulation of Bacillus megaterium. J. Bacteriol. 88: Lawrence, P. J., M. Rogolsky, and V. T. Hanh Binding of radioactive benzylpenicillin to sporulating Bacillus cultures: chemistry and fluctuations in specific binding capacity. J. Bacteriol. 108: Lawrence, P. J., and J. L. Strominger The binding of radioactive penicillin to the particulate enzyme preparation of Bacillus subtilis and its reversal with hydroxylamine or thiols. J. Biol. Chem. 245: Lawrence, P. J., and J. L. Strominger The reversible fixation of radioactive penicillin G to the D-alanine carboxypeptidase of Bacillus subtilis. J. Biol. Chem. 245: Lowry, 0. H., N. J. Rosebrough, A. L. Farr, and R. J. Randall Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193: Pearce, S. M., and P. C. Fitz-Jamnes Sporulation of a cortexless mutant of a varint of Bacillu cereus: J. Bacteriol. 106: Rogolsky, M., P. J. Lawrence, and V. T. Hanh Binding of radioactive benzylpenicillin to asporogenous mutants of Bacilus subtilis during postexponential growth. J. Bacteriol. 114: Rowley, D., P. D. Cooper, P. W. Roberts, and E. L. Smith The site of action of penicillin. Biochem. J. 46: Strominger, J. L., P. M. Blumberg, H. Suginaka, J. Umbreit, and G. G. Wickus How penicillins kill bacteria: progress and problems. Proc. Roy. Sci. London Ser. B. 179:
Antimicrobial and Antibacterial Agents
 Antimicrobial and Antibacterial Agents Contents Introduction Classification of antimicrobial drugs Special terms Mechanism of action Resistance of antimicrobial agent Introduction Joseph Lister 1867 -
Antimicrobial and Antibacterial Agents Contents Introduction Classification of antimicrobial drugs Special terms Mechanism of action Resistance of antimicrobial agent Introduction Joseph Lister 1867 -
By H. M. CHANDLER AND R. C. HAMILTON Common wealth Serum Laboratories, Parkville, Victoria, 3052, Australia INTRODUCTION
 Journal of General Microbiology (1975), 88, I 79-1 83 Printed in Great Britain = 79 SHORT COMMUNICATIONS The Protective Antigenicity of Protoplasts and Sphaeroplasts of a Highly Protective Strain of Clostridium
Journal of General Microbiology (1975), 88, I 79-1 83 Printed in Great Britain = 79 SHORT COMMUNICATIONS The Protective Antigenicity of Protoplasts and Sphaeroplasts of a Highly Protective Strain of Clostridium
Growth and Sporulation of Clostridium botulinum
 JOURNAL OF BACTERIOLOGY, Nov. 1973, p. 989-993 Copyright 0 1973 American Society for Microbiology Vol. 116, No. 2 Printed in U.S.A. Poly-f-Hydroxybutyrate Metabolism During Growth and Sporulation of Clostridium
JOURNAL OF BACTERIOLOGY, Nov. 1973, p. 989-993 Copyright 0 1973 American Society for Microbiology Vol. 116, No. 2 Printed in U.S.A. Poly-f-Hydroxybutyrate Metabolism During Growth and Sporulation of Clostridium
SYNTHESIS AND DEGRADATION OF POLY-3-HYDROXYBUTYRIC ACID IN CONNECTION WITH SPORULATION OF BACILLUS MEGATERIUM
 SYNTHESIS AND DEGRADATION OF POLY-3-HYDROXYBUTYRIC ACID IN CONNECTION WITH SPORULATION OF BACILLUS MEGATERIUM RALPH A. SLEPECKY AND JOHN H. LAW Department of Biological Sciences, Northwestern University,
SYNTHESIS AND DEGRADATION OF POLY-3-HYDROXYBUTYRIC ACID IN CONNECTION WITH SPORULATION OF BACILLUS MEGATERIUM RALPH A. SLEPECKY AND JOHN H. LAW Department of Biological Sciences, Northwestern University,
METABOLISM OF PENTOSES AND PENTITOLS
 JOURNAL OF BACTERIOLOGY Vol. 88, No. 4, p. 845-849 October, 1964 Copyright 1964 American Society for Microbiology Printed in U.S.A. II. METABOLISM OF PENTOSES AND PENTITOLS BY AEROBACTER AEROGENES MECHANISM
JOURNAL OF BACTERIOLOGY Vol. 88, No. 4, p. 845-849 October, 1964 Copyright 1964 American Society for Microbiology Printed in U.S.A. II. METABOLISM OF PENTOSES AND PENTITOLS BY AEROBACTER AEROGENES MECHANISM
(Accepted for publication, July 8, 1937)
 THE RELATIONSHIP BETWEEN BACTERIAL GROWTH AND PHAGE PRODUCTION B~ ALBERT P. KRUEGER A1~ro JACOB FONG* (From the Department of Bacteriology, Uni~erslty of California, Berkeley) (Accepted for publication,
THE RELATIONSHIP BETWEEN BACTERIAL GROWTH AND PHAGE PRODUCTION B~ ALBERT P. KRUEGER A1~ro JACOB FONG* (From the Department of Bacteriology, Uni~erslty of California, Berkeley) (Accepted for publication,
SECONDARY COLONY FORMATION BY BACILLUS SUBTILIS ON EOSINE
 SECONDARY COLONY FORMATION BY BACILLUS SUBTILIS ON EOSINE METHYLENE BLUE AGAR K. K. SHAH' AND V. N. IYER2 Microbiology Department, S. B. Garda College, Navsari, India Received for publication November
SECONDARY COLONY FORMATION BY BACILLUS SUBTILIS ON EOSINE METHYLENE BLUE AGAR K. K. SHAH' AND V. N. IYER2 Microbiology Department, S. B. Garda College, Navsari, India Received for publication November
RELATIONSHIPS BETWEEN INITIAL NUTRIENT CONCENTRATION ANDB TOTAL GROWTH1
 RELATIONSHIPS BETWEEN INITIAL NUTRIENT CONCENTRATION ANDB TOTAL GROWTH1 R. E. ECKER2 AND W. R. LOCKHART Department of Bacteriology, Iowa State University, Ames, Iowa Received for publication January 4,
RELATIONSHIPS BETWEEN INITIAL NUTRIENT CONCENTRATION ANDB TOTAL GROWTH1 R. E. ECKER2 AND W. R. LOCKHART Department of Bacteriology, Iowa State University, Ames, Iowa Received for publication January 4,
Effects of Dimethylsulfoxide on the Lactose Operon
 JOURNAL OF BACTERIOLOGY, August, 1966 Copyright @ 1966 American Society for Microbiology Vol. 92, No. 2 Printed in U.S.A. Effects of Dimethylsulfoxide on the Lactose Operon in Escherichia coli AUDREE V.
JOURNAL OF BACTERIOLOGY, August, 1966 Copyright @ 1966 American Society for Microbiology Vol. 92, No. 2 Printed in U.S.A. Effects of Dimethylsulfoxide on the Lactose Operon in Escherichia coli AUDREE V.
Effects of Dimethylsulfoxide on the Lactose Operon
 JOURNAL OF BACTERIOLOGY, August, 1966 Copyright @ 1966 American Society for Microbiology Vol. 92, No. 2 Printed in U.S.A. Effects of Dimethylsulfoxide on the Lactose Operon in Escherichia coli AUDREE V.
JOURNAL OF BACTERIOLOGY, August, 1966 Copyright @ 1966 American Society for Microbiology Vol. 92, No. 2 Printed in U.S.A. Effects of Dimethylsulfoxide on the Lactose Operon in Escherichia coli AUDREE V.
Synchronous Cultures of Bacillus subtilis
 JOURNAL OF BACTERIOLOGY, Nov. 1973, p. 736-740 Copyright 1973 American Society for Microbiology Vol. 116, No. 2 Printed in U.S.A., Synchronous Cultures of Bacillus subtilis Obtained by Filtration with
JOURNAL OF BACTERIOLOGY, Nov. 1973, p. 736-740 Copyright 1973 American Society for Microbiology Vol. 116, No. 2 Printed in U.S.A., Synchronous Cultures of Bacillus subtilis Obtained by Filtration with
obtained from the infected and treated tissues, Fleming's2 technic of hemolytic streptococcus B. Immediately following the infection, 1.0 ml.
 THE SENSITIVITY OF STREPTOCOCCI TO PENICILLIN G AFTER EXPOSURE TO THE ANTIBIOTIC IN VIVO* E. GRUNBERG, C. UNGER, AND D. ELDRIDGE Previous investigations by Grunberg, Schnitzer, and Unger3 on the topical
THE SENSITIVITY OF STREPTOCOCCI TO PENICILLIN G AFTER EXPOSURE TO THE ANTIBIOTIC IN VIVO* E. GRUNBERG, C. UNGER, AND D. ELDRIDGE Previous investigations by Grunberg, Schnitzer, and Unger3 on the topical
ACTIVATION OF BACTERIAL ENDOSPORES
 JOURNAL OF BACTERIOLOGY Vol. 88, No. 2, p. 313-318 August, 1964 Copyright @ 1964 American Society for Microbiology Printed in U.S.A. ACTIVATION OF BACTERIAL ENDOSPORES A. KEYNAN, Z. EVENCHIK, H. 0. HALVORSON,
JOURNAL OF BACTERIOLOGY Vol. 88, No. 2, p. 313-318 August, 1964 Copyright @ 1964 American Society for Microbiology Printed in U.S.A. ACTIVATION OF BACTERIAL ENDOSPORES A. KEYNAN, Z. EVENCHIK, H. 0. HALVORSON,
Factors Influencing Detection of Tolerance in Staphylococcus aureus
 ANTIMICROBIAL AGENTS AND CHEMOTHERAPY, Sept. 1982, p. 364-368 Vol. 22, No. 3 0066-4804/82/090364-0$02.00/0 Copyright 1982, American Society for Microbiology Factors Influencing Detection of Tolerance in
ANTIMICROBIAL AGENTS AND CHEMOTHERAPY, Sept. 1982, p. 364-368 Vol. 22, No. 3 0066-4804/82/090364-0$02.00/0 Copyright 1982, American Society for Microbiology Factors Influencing Detection of Tolerance in
Effect of Hypertonic Sucrose Upon the Immune Bactericidal Reaction
 INFECrION AND IMMUNITY, Jan. 1970, p. 51-55 Copyright 1970 American Society for Microbiology Vol. 1, No. 1 Printed in U.S.A. Effect of Hypertonic Sucrose Upon the Immune Bactericidal Reaction LOUIS H.
INFECrION AND IMMUNITY, Jan. 1970, p. 51-55 Copyright 1970 American Society for Microbiology Vol. 1, No. 1 Printed in U.S.A. Effect of Hypertonic Sucrose Upon the Immune Bactericidal Reaction LOUIS H.
Krzysztof Sieradzki and Alexander Tomasz* The Rockefeller University, 1230 York Avenue, New York, NY 10021, USA
 Journal of Antimicrobial Chemotherapy (1997) 39, Suppl. A, 47 51 JAC Suppression of -lactam antibiotic resistance in a methicillin-resistant Staphylococcus aureus through synergic action of early cell
Journal of Antimicrobial Chemotherapy (1997) 39, Suppl. A, 47 51 JAC Suppression of -lactam antibiotic resistance in a methicillin-resistant Staphylococcus aureus through synergic action of early cell
BIOSYNTHESIS OF MYCOBACILLIN, A NEW ANTIFUNGAL PEPTIDE
 JOURNAL OF BACTERIOLOGY Vol. 87, No. 6, pp. 1397-1401 June, 1964 Copyright 1964 by the American Society for Microbiology Printed in U.S.A. BIOSYNTHESIS OF MYCOBACILLIN, A NEW ANTIFUNGAL PEPTIDE I. ROLE
JOURNAL OF BACTERIOLOGY Vol. 87, No. 6, pp. 1397-1401 June, 1964 Copyright 1964 by the American Society for Microbiology Printed in U.S.A. BIOSYNTHESIS OF MYCOBACILLIN, A NEW ANTIFUNGAL PEPTIDE I. ROLE
antigen." 2 Moreover, when mixed populations of normal and sensitive cells
 DELA YED HYPERSENSITIVITY IN VITRO: ITS MEDIATION BY CELL-FREE SUBSTANCES FORMED BY LYMPHOID CELL-ANTIGEN INTERACTION* BY JOHN R. DAVIDt DEPARTMENT OF MEDICINE, NEW YORK UNIVERSITY SCHOOL OF MEDICINE Communicated
DELA YED HYPERSENSITIVITY IN VITRO: ITS MEDIATION BY CELL-FREE SUBSTANCES FORMED BY LYMPHOID CELL-ANTIGEN INTERACTION* BY JOHN R. DAVIDt DEPARTMENT OF MEDICINE, NEW YORK UNIVERSITY SCHOOL OF MEDICINE Communicated
GROWTH OF BACTERIA ON THE SURFACE ANION-EXCHANGE RESIN I. EXPERIMENT WITH BATCH CULTURE
 J. Gen. Appl. Microbiol., 18, 271-283 (1972) GROWTH OF BACTERIA ON THE SURFACE ANION-EXCHANGE RESIN OF I. EXPERIMENT WITH BATCH CULTURE REIKO HATTORI, TSUTOMU HATTORI, AND CHOSEKI FURUSAKA Institute for
J. Gen. Appl. Microbiol., 18, 271-283 (1972) GROWTH OF BACTERIA ON THE SURFACE ANION-EXCHANGE RESIN OF I. EXPERIMENT WITH BATCH CULTURE REIKO HATTORI, TSUTOMU HATTORI, AND CHOSEKI FURUSAKA Institute for
Staphylococcus aureus Cell Extract Transcription-Translation Assay: Firefly Luciferase Reporter System for Evaluating Protein Translation Inhibitors
 ANTIMICROBIAL AGENTS AND CHEMOTHERAPY, June 2001, p. 1900 1904 Vol. 45, No. 6 0066-4804/01/$04.00 0 DOI: 10.1128/AAC.45.6.1900 1904.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved.
ANTIMICROBIAL AGENTS AND CHEMOTHERAPY, June 2001, p. 1900 1904 Vol. 45, No. 6 0066-4804/01/$04.00 0 DOI: 10.1128/AAC.45.6.1900 1904.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved.
number or vitality. Spores from strain 62A were used for the major part of this EFFECT OF SUBTILIN ON SPORES OF CLOSTRIDIUM
 EFFECT OF SUBTILIN ON SPORES OF CLOSTRIDIUM BOTULINUM A. A. ANDERSEN Western Regional Research Laboratory,' Albany, California Received for publication January 9, 1952 Within the last few years considerable
EFFECT OF SUBTILIN ON SPORES OF CLOSTRIDIUM BOTULINUM A. A. ANDERSEN Western Regional Research Laboratory,' Albany, California Received for publication January 9, 1952 Within the last few years considerable
Isolation of L Forms from Group A Streptococci Exposed to Bacitracin
 JOURNAL OF BACTERIOLOGY, June, 1965 Copyright 0 1965 Amiierican Society for Microbiology Vol. 89, No. 6 Printed in U.S.A. Isolation of L Forms from Group A Streptococci Exposed to Bacitracin JIRI ROTTA,1
JOURNAL OF BACTERIOLOGY, June, 1965 Copyright 0 1965 Amiierican Society for Microbiology Vol. 89, No. 6 Printed in U.S.A. Isolation of L Forms from Group A Streptococci Exposed to Bacitracin JIRI ROTTA,1
MICROORGANISM AND CHEMOTHERAPEIC MATERIALS
 MICROORGANISM AND CHEMOTHERAPEIC MATERIALS Chemotherapeutic substances are antimicrobials derived from chemical substances. Antibiotics are antimicrobials obtained from bacteria or fungi CHEMOTHERAPYTIC
MICROORGANISM AND CHEMOTHERAPEIC MATERIALS Chemotherapeutic substances are antimicrobials derived from chemical substances. Antibiotics are antimicrobials obtained from bacteria or fungi CHEMOTHERAPYTIC
Section B and C. Volume-21. Contents 12. APPLIED BIOLOGY. Life Sciences For NET & SLET Exams Of UGC-CSIR. *Mudra*
 Section B and C Volume-21 Contents 12. APPLIED BIOLOGY A. MICROBIAL FERMENTATION AND PRODUCTION OF SMALL AND MACRO MOLECULES 1 B. APPLICATIONS OF IMMUNOLOGICAL PRINCIPLES 47 TISSUE AND CELL CULTURE METHODS
Section B and C Volume-21 Contents 12. APPLIED BIOLOGY A. MICROBIAL FERMENTATION AND PRODUCTION OF SMALL AND MACRO MOLECULES 1 B. APPLICATIONS OF IMMUNOLOGICAL PRINCIPLES 47 TISSUE AND CELL CULTURE METHODS
GROWTH OF CELLULAR FORMS IN CULTURES OF CHROMIATIN BODIES
 GROWTH OF CELLULAR FORMS IN CULTURES OF CHROMIATIN BODIES ISOLATED FROM BACILLUS MEGATERIUM1 B. R. CHATTERJEE AND ROBERT P. WILLIAMS Departnment of Microbiology, Baylor University College of Medicine,
GROWTH OF CELLULAR FORMS IN CULTURES OF CHROMIATIN BODIES ISOLATED FROM BACILLUS MEGATERIUM1 B. R. CHATTERJEE AND ROBERT P. WILLIAMS Departnment of Microbiology, Baylor University College of Medicine,
GROWTH OF CELLULAR FORMS IN CULTURES OF CHROMIATIN BODIES
 GROWTH OF CELLULAR FORMS IN CULTURES OF CHROMIATIN BODIES ISOLATED FROM BACILLUS MEGATERIUM1 B. R. CHATTERJEE AND ROBERT P. WILLIAMS Departnment of Microbiology, Baylor University College of Medicine,
GROWTH OF CELLULAR FORMS IN CULTURES OF CHROMIATIN BODIES ISOLATED FROM BACILLUS MEGATERIUM1 B. R. CHATTERJEE AND ROBERT P. WILLIAMS Departnment of Microbiology, Baylor University College of Medicine,
Low-pH Activation of Bacillus cereus Spores
 JOURNAL OF BACrERIOLOGY, Feb. 1970, p. 418-422 Vol. 101, No. 2 Copyright 1970 American Society for Microbiology Printed in U.S.A. Low-pH Activation of Bacillus cereus Spores GABRIELA ISSAHARY, Z. EVENCHIK,
JOURNAL OF BACrERIOLOGY, Feb. 1970, p. 418-422 Vol. 101, No. 2 Copyright 1970 American Society for Microbiology Printed in U.S.A. Low-pH Activation of Bacillus cereus Spores GABRIELA ISSAHARY, Z. EVENCHIK,
His-Spin Protein Miniprep
 INSTRUCTIONS His-Spin Protein Miniprep Catalog No. P2001 (10 purifications) and P2002 (50 purifications). Highlights Fast 5 minute protocol to purify His-tagged proteins from cell-free extracts Screen
INSTRUCTIONS His-Spin Protein Miniprep Catalog No. P2001 (10 purifications) and P2002 (50 purifications). Highlights Fast 5 minute protocol to purify His-tagged proteins from cell-free extracts Screen
Title. Author(s)MAKINO, Toshikazu. Issue Date Doc URL. Type. File Information. bulletin. 14(2)_P
 Title Effect of antibiotics inhibiting cell wall synthesis Author(s)MAKINO, Toshikazu CitationJournal of the Faculty of Science, Hokkaido Universi Issue Date 1988 Doc URL http://hdl.handle.net/2115/26438
Title Effect of antibiotics inhibiting cell wall synthesis Author(s)MAKINO, Toshikazu CitationJournal of the Faculty of Science, Hokkaido Universi Issue Date 1988 Doc URL http://hdl.handle.net/2115/26438
Requirement for a Functional int Product in Temperature Inductions of
 JOURNAL OF VIROLOGY, SePt. 1970, p. 320-325 Vol. 6, No. 3 Copyright 1970 American Society for Microbiology Prinited in U.S.A. Requirement for a Functional int Product in Temperature Inductions of Prophage
JOURNAL OF VIROLOGY, SePt. 1970, p. 320-325 Vol. 6, No. 3 Copyright 1970 American Society for Microbiology Prinited in U.S.A. Requirement for a Functional int Product in Temperature Inductions of Prophage
by using continuous flow paper electrophoresis of equilibrated plasma, which has permitted the demonstration
 BINDING OF CORTICOSTEROIDS BY PLASMA PROTEINS. IV. THE ELECTROPHORETIC DEMONSTRATION OF CORTICOSTEROID BINDING GLOBULIN 1, 2 By WILLIAM H. DAUGHADAY WITH THE TECHNICAL ASSISTANCE OF IDA KOZAK (From the
BINDING OF CORTICOSTEROIDS BY PLASMA PROTEINS. IV. THE ELECTROPHORETIC DEMONSTRATION OF CORTICOSTEROID BINDING GLOBULIN 1, 2 By WILLIAM H. DAUGHADAY WITH THE TECHNICAL ASSISTANCE OF IDA KOZAK (From the
MagExtactor -His-tag-
 Instruction manual MagExtractor-His-tag-0905 F0987K MagExtactor -His-tag- Contents NPK-701 100 preparations Store at Store at 4 C [1] Introduction [2] Components [3] Materials required [4] Protocol3 1.
Instruction manual MagExtractor-His-tag-0905 F0987K MagExtactor -His-tag- Contents NPK-701 100 preparations Store at Store at 4 C [1] Introduction [2] Components [3] Materials required [4] Protocol3 1.
Antimicrobial Drugs. Antimicrobial Drugs. The dawn of antibiotics. Alexander Fleming. Chain and Florey. Antibiotics
 Antimicrobial Drugs Antimicrobial Drugs Chemotherapy: The use of drugs to treat a disease Antimicrobial drugs: Interfere with the growth of microbes within a host Antibiotic: Substance produced by a microbe
Antimicrobial Drugs Antimicrobial Drugs Chemotherapy: The use of drugs to treat a disease Antimicrobial drugs: Interfere with the growth of microbes within a host Antibiotic: Substance produced by a microbe
01/08/2018. Control of Microbial Growth. Methods. Terminology. Disinfectants and Antiseptics. Three approaches. Cleaning. Chemical.
 Control of Microbial Growth Disinfectants and Antiseptics 1 Methods 2 Three approaches Chemical Disinfectants and antiseptics Physical Heat Ultraviolet Irradiations Mechanical elimination Cleaning Filtration
Control of Microbial Growth Disinfectants and Antiseptics 1 Methods 2 Three approaches Chemical Disinfectants and antiseptics Physical Heat Ultraviolet Irradiations Mechanical elimination Cleaning Filtration
Mechanisms of the post-antibiotic effects induced by rifampicin and gentamicin in Escherichia coli
 Journal of Antimicrobial Chemotherapy (26) 58, 444 448 doi:1.193/jac/dkl225 Advance Access publication 3 May 26 Mechanisms of the post-antibiotic effects induced by rifampicin and gentamicin in Escherichia
Journal of Antimicrobial Chemotherapy (26) 58, 444 448 doi:1.193/jac/dkl225 Advance Access publication 3 May 26 Mechanisms of the post-antibiotic effects induced by rifampicin and gentamicin in Escherichia
Bacterial PE LB. Bacterial Protein Extraction Lysis Buffer. (Cat. # , , , , , )
 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Bacterial PE LB Bacterial Protein Extraction Lysis Buffer (Cat. # 786-176, 786-177, 786-185,
G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Bacterial PE LB Bacterial Protein Extraction Lysis Buffer (Cat. # 786-176, 786-177, 786-185,
CHAPTER 4 DISCUSSION. Many types of suitable media can be used to support the fungal growth and there is no
 CHAPTER 4 DISCUSSION 4.1 Media Preparation and Subculture Many types of suitable media can be used to support the fungal growth and there is no specific medium ideally suited for the culture of species
CHAPTER 4 DISCUSSION 4.1 Media Preparation and Subculture Many types of suitable media can be used to support the fungal growth and there is no specific medium ideally suited for the culture of species
SHORT COMMUNICATION. (Received 10 February 1969)
 Journal of Neurochemistry. 1969, Vol. 16, pp. 1037 to 1041. Petearnon Press. Printed in Northern Ireland SHORT COMMUNICATION Evidence for RNA transport in rat optic nerve (Received 10 February 1969) A
Journal of Neurochemistry. 1969, Vol. 16, pp. 1037 to 1041. Petearnon Press. Printed in Northern Ireland SHORT COMMUNICATION Evidence for RNA transport in rat optic nerve (Received 10 February 1969) A
The Histidine Decarboxylase of a Species of LactobaciZZus; Apparent Dispensability of Pyridoxal Phosphate as Coenzyme
 233 RODWELL, A. W. (1953). J. gen. Microbiol. 8, 233-237. The Histidine Decarboxylase of a Species of LactobaciZZus; Apparent Dispensability of Pyridoxal Phosphate as Coenzyme BY A. W. RODWELL* Medical
233 RODWELL, A. W. (1953). J. gen. Microbiol. 8, 233-237. The Histidine Decarboxylase of a Species of LactobaciZZus; Apparent Dispensability of Pyridoxal Phosphate as Coenzyme BY A. W. RODWELL* Medical
Dehydrogenase Obtained from Heated Spores of
 JOURNAL OF BACTERIOLOGY, Aug. 1971, p. 442-447 Copyright 1971 American Society for Microbiology Vol. 17, No. 2 Printed in U.S.A. Reactivative Action of Ethylenediaminetetraacetic Acid or Dipicolinic Acid
JOURNAL OF BACTERIOLOGY, Aug. 1971, p. 442-447 Copyright 1971 American Society for Microbiology Vol. 17, No. 2 Printed in U.S.A. Reactivative Action of Ethylenediaminetetraacetic Acid or Dipicolinic Acid
E. A. EDWARDS' AND G. L. LARSON
 APPLIED MICROBIOLOGY, Dec. 1974, p. 972-976 Copyright 0 1975 American Society for Microbiology Vol. 28, No. 6 Printed in U.S.A. New Method of Grouping Beta-Hemolytic Streptococci Directly on Sheep Blood
APPLIED MICROBIOLOGY, Dec. 1974, p. 972-976 Copyright 0 1975 American Society for Microbiology Vol. 28, No. 6 Printed in U.S.A. New Method of Grouping Beta-Hemolytic Streptococci Directly on Sheep Blood
HOOK 6X His Protein Purification (Yeast)
 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name HOOK 6X His Protein Purification (Yeast) For The Purification of His Tagged Proteins from
G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name HOOK 6X His Protein Purification (Yeast) For The Purification of His Tagged Proteins from
Identification of the In Vivo Pharmacokinetics and Pharmacodynamic Drivers of Iclaprim
 AAC Accepted Manuscript Posted Online 29 January 2018 Antimicrob. Agents Chemother. doi:10.1128/aac.02550-17 Copyright 2018 American Society for Microbiology. All Rights Reserved. 1 Identification of the
AAC Accepted Manuscript Posted Online 29 January 2018 Antimicrob. Agents Chemother. doi:10.1128/aac.02550-17 Copyright 2018 American Society for Microbiology. All Rights Reserved. 1 Identification of the
Several Aspects of Microbial Technology for Food Pasteurization and Sterilization
 , Vol. 10, No. 4, pp. 183-190, Dec. 2009 Several Aspects of Microbial Technology for Food Pasteurization and Sterilization Tetsuaki TSUCHIDO Department of Life Science and Biotechnology, Faculty of Chemistry,
, Vol. 10, No. 4, pp. 183-190, Dec. 2009 Several Aspects of Microbial Technology for Food Pasteurization and Sterilization Tetsuaki TSUCHIDO Department of Life Science and Biotechnology, Faculty of Chemistry,
organisms which yield predominantly lactic acid in the
 THE LACTIC ACID FERMENTATION OF STREPTOCOCCI PAUL A. SMITH AND J. M. SHERMAN Laboratory of Bacteriology, College of Agriculture, Cornell Univer8ity, Ithaca, New York Received for publication August 21,
THE LACTIC ACID FERMENTATION OF STREPTOCOCCI PAUL A. SMITH AND J. M. SHERMAN Laboratory of Bacteriology, College of Agriculture, Cornell Univer8ity, Ithaca, New York Received for publication August 21,
Reduction of Ferric Compounds by Soil Bacteria
 No. 3, Volume 10 of the Jouml of General Microbiology was issued on 10 June 1954. BROMFIELD, S. M. (1954). J. gen. Microbiol. 11, 14. Reduction of Ferric Compounds by Soil Bacteria BY S. M. BROMFIELD Microbiology
No. 3, Volume 10 of the Jouml of General Microbiology was issued on 10 June 1954. BROMFIELD, S. M. (1954). J. gen. Microbiol. 11, 14. Reduction of Ferric Compounds by Soil Bacteria BY S. M. BROMFIELD Microbiology
How antimicrobial agents work
 Physical and Chemical Control of Microbes Physical Agents heat or radiation Chemical Agents disinfectants or antiseptics Important Terms 1. Sterilization process of killing all viable microbes 2. Bactericide
Physical and Chemical Control of Microbes Physical Agents heat or radiation Chemical Agents disinfectants or antiseptics Important Terms 1. Sterilization process of killing all viable microbes 2. Bactericide
Isolation of Mutants Affecting Tryptophanase Production in Escherichia coli
 JOURNAL OF BACTERIOLOGY, Feb., 1965 Copyright 1965 American Society for Microbiology Vol. 89, No. 2 Printed in U.S.A. Isolation of Mutants Affecting Tryptophanase Production in Escherichia coli THEODORE
JOURNAL OF BACTERIOLOGY, Feb., 1965 Copyright 1965 American Society for Microbiology Vol. 89, No. 2 Printed in U.S.A. Isolation of Mutants Affecting Tryptophanase Production in Escherichia coli THEODORE
INDUCTION OF CEPHALOSPORINASE PRODUCTION BY VARIOUS PENICILLINS IN ENTEROBACTERIACEAE
 VOL. xxxvi NO. 10 THE JOURNAL OF ANTIBIOTICS 187 INDUCTION OF CEPHALOSPORINASE PRODUCTION BY VARIOUS PENICILLINS IN ENTEROBACTERIACEAE SHINZABUROU MINAMI, NOBUYUKI MATSUBARA, AKIRA YOTSUJI. HARUMI ARAKI,
VOL. xxxvi NO. 10 THE JOURNAL OF ANTIBIOTICS 187 INDUCTION OF CEPHALOSPORINASE PRODUCTION BY VARIOUS PENICILLINS IN ENTEROBACTERIACEAE SHINZABUROU MINAMI, NOBUYUKI MATSUBARA, AKIRA YOTSUJI. HARUMI ARAKI,
Deoxyribonucleic Acid Synthesis in
 JOURNAL OF BACTERIOLOGY, Sept. 973, p. 966-97 Vol. 5. No. 3 Copyright 973 American Society for Microbiology Printed in U.S.A. Three Additional Genes Required for Deoxyribonucleic Acid Synthesis in Saccharomyces
JOURNAL OF BACTERIOLOGY, Sept. 973, p. 966-97 Vol. 5. No. 3 Copyright 973 American Society for Microbiology Printed in U.S.A. Three Additional Genes Required for Deoxyribonucleic Acid Synthesis in Saccharomyces
The GeneEditor TM in vitro Mutagenesis System: Site- Directed Mutagenesis Using Altered Beta-Lactamase Specificity
 Promega Notes Magazine Number 62, 1997, p. 02 The GeneEditor TM in vitro Mutagenesis System: Site- Directed Mutagenesis Using Altered Beta-Lactamase Specificity By Christine Andrews and Scott Lesley Promega
Promega Notes Magazine Number 62, 1997, p. 02 The GeneEditor TM in vitro Mutagenesis System: Site- Directed Mutagenesis Using Altered Beta-Lactamase Specificity By Christine Andrews and Scott Lesley Promega
Experimental Design. Margaret A. Daugherty. Fall Michaelis Menton Kinetics and Inhibition. Lecture 14: Enzymes & Kinetics III E + S ES E + P
 Lecture 14: Enzymes & Kinetics III Michaelis Menton Kinetics and Inhibition Margaret A. Daugherty Fall 2003 Experimental Design k 1 k cat E + S ES E + P k -1 I want to measure the reactivity of my enzyme
Lecture 14: Enzymes & Kinetics III Michaelis Menton Kinetics and Inhibition Margaret A. Daugherty Fall 2003 Experimental Design k 1 k cat E + S ES E + P k -1 I want to measure the reactivity of my enzyme
GERMINATION OF SPORES OF CERTAIN AEROBIC BACILLI
 GERMINATION OF SPORES OF CERTAIN AEROBIC BACILLI UNDER ANAEROBIC CONDITIONS NORMAN G. ROTH AND DAVID H. LIVELY Camp Detrick, Frederick, Maryland Received for publication June 24, 1955 Previous work by
GERMINATION OF SPORES OF CERTAIN AEROBIC BACILLI UNDER ANAEROBIC CONDITIONS NORMAN G. ROTH AND DAVID H. LIVELY Camp Detrick, Frederick, Maryland Received for publication June 24, 1955 Previous work by
-7 ::(teto) ::(teto) 120. GFP membranes merge. overexposed. Supplemental Figure 1 (Marquis et al.)
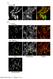 P spoiie -gfp -7 ::(teto) 120-91 ::(teto) 120 GFP membranes merge overexposed Supplemental Figure 1 (Marquis et al.) Figure S1 TetR-GFP is stripped off the teto array in sporulating cells that contain
P spoiie -gfp -7 ::(teto) 120-91 ::(teto) 120 GFP membranes merge overexposed Supplemental Figure 1 (Marquis et al.) Figure S1 TetR-GFP is stripped off the teto array in sporulating cells that contain
GeneMATRIX Gram Plus & Yeast Genomic DNA Purification Kit
 GeneMATRIX Gram Plus & Yeast Genomic DNA Purification Kit Universal kit for isolation of total DNA from gram positive bacteria and yeast. The kit contains glass beads for mechanical cell disruption. Cat.
GeneMATRIX Gram Plus & Yeast Genomic DNA Purification Kit Universal kit for isolation of total DNA from gram positive bacteria and yeast. The kit contains glass beads for mechanical cell disruption. Cat.
Transmission Electron Microscopic Study of Antibiotic Action on Klebsiella pneumoniae Biofilm
 ANTIMICROBIAL AGENTS AND CHEMOTHERAPY, Aug. 2002, p. 2679 2683 Vol. 46, No. 8 0066-4804/02/$04.00 0 DOI: 10.1128/AAC.46.8.2679 2683.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved.
ANTIMICROBIAL AGENTS AND CHEMOTHERAPY, Aug. 2002, p. 2679 2683 Vol. 46, No. 8 0066-4804/02/$04.00 0 DOI: 10.1128/AAC.46.8.2679 2683.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved.
Growth of Mycobacterium smegmatis in minimal and complete media
 J. Biosci., Vol. 2, Number 4, December 1980. pp. 337 348 Printed in India Growth of Mycobacterium smegmatis in minimal and complete media RAGHAVENDRA GADAGKAR* and K. P. GOPINATHAN Microbiology and Cell
J. Biosci., Vol. 2, Number 4, December 1980. pp. 337 348 Printed in India Growth of Mycobacterium smegmatis in minimal and complete media RAGHAVENDRA GADAGKAR* and K. P. GOPINATHAN Microbiology and Cell
Denis V. Kurek, Sergey A. Lopatin, *Vladimir E. Tikhonov, Valery P. Varlamov
 NEW AFFINITY SORBENTS FOR PURIFICATION OF RECOMBINANT PROTEINS WITH THE USE OF CHITIN-BINDING DOMAIN AS AN AFFINITY TAG Denis V. Kurek, Sergey A. Lopatin, *Vladimir E. Tikhonov, Valery P. Varlamov Centre
NEW AFFINITY SORBENTS FOR PURIFICATION OF RECOMBINANT PROTEINS WITH THE USE OF CHITIN-BINDING DOMAIN AS AN AFFINITY TAG Denis V. Kurek, Sergey A. Lopatin, *Vladimir E. Tikhonov, Valery P. Varlamov Centre
DNA recombination without ligase: TOPO TA Cloning. topoisomerase
 DNA recombination without ligase: TOPO TA Cloning topoisomerase Cloning strategies, cloning in bacteria other than E.coli Mitesh Shrestha Cloning strategies Cloning in bacteria other than E.coli Convenient
DNA recombination without ligase: TOPO TA Cloning topoisomerase Cloning strategies, cloning in bacteria other than E.coli Mitesh Shrestha Cloning strategies Cloning in bacteria other than E.coli Convenient
(0-D-galactosidase) of Escherichia coli.
 EFFECTS OF X-RAYS ON THE FORMATION OF LACTASE IN A LYSOGENIC STRAIN AND A SENSITIVE STRAIN OF ESCHERICHIA COLI1 CHARLES YANOFSKY Department of Microbiology, Osborn Botanical Laboratory, Yale University,
EFFECTS OF X-RAYS ON THE FORMATION OF LACTASE IN A LYSOGENIC STRAIN AND A SENSITIVE STRAIN OF ESCHERICHIA COLI1 CHARLES YANOFSKY Department of Microbiology, Osborn Botanical Laboratory, Yale University,
MagSi Beads. Magnetic Silica Beads for Life Science and Biotechnology study
 MagSi Beads Magnetic Silica Beads for Life Science and Biotechnology study MagnaMedics Diagnostics B.V. / Rev. 9.2 / 2012 Wide range of products for numerous applications MagnaMedics separation solutions
MagSi Beads Magnetic Silica Beads for Life Science and Biotechnology study MagnaMedics Diagnostics B.V. / Rev. 9.2 / 2012 Wide range of products for numerous applications MagnaMedics separation solutions
Virus-specific RNA: Suspension cultures of KB cells at 2-3 X 106 cells/ml were infected
 A METHOD FOR DETERMINING THE FRACTION OF THE VIRAL GENOME TRANSCRIBED DURING INFECTION AND ITS APPLICATION TO ADENOVIRUS-INFECTED CELLS* BY KEI FUJINAGA, STANLEY MAK4 AND MAURICE GREENt INSTITUTE FOR MOLECULAR
A METHOD FOR DETERMINING THE FRACTION OF THE VIRAL GENOME TRANSCRIBED DURING INFECTION AND ITS APPLICATION TO ADENOVIRUS-INFECTED CELLS* BY KEI FUJINAGA, STANLEY MAK4 AND MAURICE GREENt INSTITUTE FOR MOLECULAR
In Vitro Incorporation of Molybdate into Demolybdoproteins
 JOURNAL OF BACTERIOLOGY, Feb. 1979, p. 719-726 0021-9193/79/02-0719/08$02.00/0 Vol. 137, No. 2 In Vitro Incorporation of Molybdate into Demolybdoproteins in Escherichia coli RANDOLPH H. SCOTT, GEORGE T.
JOURNAL OF BACTERIOLOGY, Feb. 1979, p. 719-726 0021-9193/79/02-0719/08$02.00/0 Vol. 137, No. 2 In Vitro Incorporation of Molybdate into Demolybdoproteins in Escherichia coli RANDOLPH H. SCOTT, GEORGE T.
Summary of Mutagenic Toxicity Test Results for EvaGreen
 Summary of Mutagenic Toxicity Test Results for EvaGreen Compiled by Biotium, Inc. from the results of an independent testing service: Litron Laboratories, Inc., Rochester, NY Overview When our scientists
Summary of Mutagenic Toxicity Test Results for EvaGreen Compiled by Biotium, Inc. from the results of an independent testing service: Litron Laboratories, Inc., Rochester, NY Overview When our scientists
10/2/2016. Control of Microbial Growth. Method. Terminology. Disinfectants and Antiseptics
 Control of Microbial Growth Disinfectants and Antiseptics 1 Method Three approaches for the control of microbial growth Chemical Disinfectants and antiseptics Physical Heat Ultraviolet Irradiations Mechanical
Control of Microbial Growth Disinfectants and Antiseptics 1 Method Three approaches for the control of microbial growth Chemical Disinfectants and antiseptics Physical Heat Ultraviolet Irradiations Mechanical
FURTHER OBSERVATIONS ON THE AGGLUTINATION OF BACTERIA IN VIVO.
 FURTHER OBSERVATIONS ON THE AGGLUTINATION OF BACTERIA IN VIVO. BY CARROLL G. BULL, M.D. (From the Laboratories of The Rockefeller Institute for Medical Research.) PLATE 7. (Received for publication, April
FURTHER OBSERVATIONS ON THE AGGLUTINATION OF BACTERIA IN VIVO. BY CARROLL G. BULL, M.D. (From the Laboratories of The Rockefeller Institute for Medical Research.) PLATE 7. (Received for publication, April
revtersed by methionine, they postulate that 2-Cl-PAB inhibits only the
 INHIBITION OF METHIONINE SYNTHESIS IN ESCHERICHIA COLI BY 2-CHLORO-4-AMINOBENZOIC ACID AND SULFANILAMIDE FREDE B. STRANDSKOV The Research Department of Wallace and Tiernan Products, Inc., Belleville, New
INHIBITION OF METHIONINE SYNTHESIS IN ESCHERICHIA COLI BY 2-CHLORO-4-AMINOBENZOIC ACID AND SULFANILAMIDE FREDE B. STRANDSKOV The Research Department of Wallace and Tiernan Products, Inc., Belleville, New
revtersed by methionine, they postulate that 2-Cl-PAB inhibits only the
 INHIBITION OF METHIONINE SYNTHESIS IN ESCHERICHIA COLI BY 2-CHLORO-4-AMINOBENZOIC ACID AND SULFANILAMIDE FREDE B. STRANDSKOV The Research Department of Wallace and Tiernan Products, Inc., Belleville, New
INHIBITION OF METHIONINE SYNTHESIS IN ESCHERICHIA COLI BY 2-CHLORO-4-AMINOBENZOIC ACID AND SULFANILAMIDE FREDE B. STRANDSKOV The Research Department of Wallace and Tiernan Products, Inc., Belleville, New
Chapter 10. Antimicrobials. PowerPoint Lecture Slides for MICROBIOLOGY ROBERT W. BAUMAN
 PowerPoint Lecture Slides for MICROBIOLOGY ROBERT W. BAUMAN Chapter 10 Antimicrobials Antimicrobial Drugs Chemotherapy - The use of drugs to treat a disease Antimicrobial drugs - Interfere with the growth
PowerPoint Lecture Slides for MICROBIOLOGY ROBERT W. BAUMAN Chapter 10 Antimicrobials Antimicrobial Drugs Chemotherapy - The use of drugs to treat a disease Antimicrobial drugs - Interfere with the growth
HOOK 6X His Protein Spin Purification (Bacteria)
 222PR 03 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name HOOK 6X His Protein Spin Purification (Bacteria) For the Purification of His Tagged
222PR 03 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name HOOK 6X His Protein Spin Purification (Bacteria) For the Purification of His Tagged
I proline prior to irradiation in helium showed fewer chromosome aberrations
 REDUCTION OF RADIATION DAMAGE TO TRADESCANTIA CHROMOSOMES BY ADENOSINE TRIPHOSPHATE, PROLINE, AND HISTIDINE1 ALVIN V. BEATTY AND JEANNE W. BEATTY Department of Biology, Emory University, Atlanta, Georgia
REDUCTION OF RADIATION DAMAGE TO TRADESCANTIA CHROMOSOMES BY ADENOSINE TRIPHOSPHATE, PROLINE, AND HISTIDINE1 ALVIN V. BEATTY AND JEANNE W. BEATTY Department of Biology, Emory University, Atlanta, Georgia
Cell Growth and DNA Extraction- Technion igem HS
 Growing Cells and DNA Extraction Goals 1. Become familiar with the process of growing bacteria 2. Get to know the DNA extraction process 3. Perform miniprep in the lab Keywords 1. Growth stages 6. Techniques
Growing Cells and DNA Extraction Goals 1. Become familiar with the process of growing bacteria 2. Get to know the DNA extraction process 3. Perform miniprep in the lab Keywords 1. Growth stages 6. Techniques
The Activity of Glycopeptide Antibiotics Against Resistant Bacteria Correlates with their Ability to Induce the Resistance System
 Supplemental Material for The Activity of Glycopeptide Antibiotics Against Resistant Bacteria Correlates with their Ability to Induce the Resistance System Min Jung Kwun, Hee-Jeon Hong# Department of Biochemistry,
Supplemental Material for The Activity of Glycopeptide Antibiotics Against Resistant Bacteria Correlates with their Ability to Induce the Resistance System Min Jung Kwun, Hee-Jeon Hong# Department of Biochemistry,
BACTERIAL BIOFILMS FORMATION AT AIR LIQUID INTERFACES
 Innovative Romanian Food Biotechnology Vol. 5, Issue of December, 009 009 by Dunărea de Jos University Galaţi Received September 1, 009/ Accepted November 8, 009 RESEARCH ARTICLE BACTERIAL BIOFILMS FORMATION
Innovative Romanian Food Biotechnology Vol. 5, Issue of December, 009 009 by Dunărea de Jos University Galaţi Received September 1, 009/ Accepted November 8, 009 RESEARCH ARTICLE BACTERIAL BIOFILMS FORMATION
Electron Microscopy of Proteus vulgaris Exposed to
 JOURNAL OF BACTERIOLOGY, May 1968, p. 1956-1960 Copyright ( 1968 American Society for Microbiology Vol. 95, No. 5 Printed in U.S.A. Electron Microscopy of Proteus vulgaris Exposed to Cephalothin' NICHOLAS
JOURNAL OF BACTERIOLOGY, May 1968, p. 1956-1960 Copyright ( 1968 American Society for Microbiology Vol. 95, No. 5 Printed in U.S.A. Electron Microscopy of Proteus vulgaris Exposed to Cephalothin' NICHOLAS
Effect of Diuresis on Staphylococcus aureus Kidney
 INFECTION AND IMMUNITY, Dec. 1971, p. 74-746 Copyright 1971 American Society for Microbiology Vol. 4, No. 6 Printed in U.S.A. Effect of Diuresis on Staphylococcus aureus Kidney Infections in Mice DOLORES
INFECTION AND IMMUNITY, Dec. 1971, p. 74-746 Copyright 1971 American Society for Microbiology Vol. 4, No. 6 Printed in U.S.A. Effect of Diuresis on Staphylococcus aureus Kidney Infections in Mice DOLORES
Germination Properties as Marker Events Characterizing Later Stages of Bacillus subtilis Spore Formation
 JOURNAL OF BACTERIOLOGY, Feb. 198, p. 786-792 21-9193/8/2-786/7$2./ Vol. 141, No. 2 Germination Properties as Marker Events Characterizing Later Stages of Bacillus subtilis Spore Formation PATRICE DIONt
JOURNAL OF BACTERIOLOGY, Feb. 198, p. 786-792 21-9193/8/2-786/7$2./ Vol. 141, No. 2 Germination Properties as Marker Events Characterizing Later Stages of Bacillus subtilis Spore Formation PATRICE DIONt
ENVIRONMENTAL PARAMETERS OF GROWTH
 ENVIRONMENTAL PARAMETERS OF GROWTH The growth and survival of microorganisms are affected by the chemical and physical conditions of the external environment. Environmental factors which have significant
ENVIRONMENTAL PARAMETERS OF GROWTH The growth and survival of microorganisms are affected by the chemical and physical conditions of the external environment. Environmental factors which have significant
THE COMPARATIVE RESISTANCE OF BACTERIA AND HUMAN TISSUE CELLS TO CERTAIN COMMON ANTISEPTICS.
 Published Online: 1 December, 1916 Supp Info: http://doi.org/1.184/jem.24.6.683 Downloaded from jem.rupress.org on January 8, 219 THE COMPARATIVE RESISTANCE OF BACTERIA AND HUMAN TISSUE CELLS TO CERTAIN
Published Online: 1 December, 1916 Supp Info: http://doi.org/1.184/jem.24.6.683 Downloaded from jem.rupress.org on January 8, 219 THE COMPARATIVE RESISTANCE OF BACTERIA AND HUMAN TISSUE CELLS TO CERTAIN
Genetically Alterable Transport of Amethopterin
 JOURNAL OF BACTERIOLOGY, Jan., 1967, p. 315-319 Copyright ( 1967 American Society for Microbiology Vol. 93, No. I Printed in U.S.A. Genetically Alterable Transport of Amethopterin in Diplococcus pneumoniae
JOURNAL OF BACTERIOLOGY, Jan., 1967, p. 315-319 Copyright ( 1967 American Society for Microbiology Vol. 93, No. I Printed in U.S.A. Genetically Alterable Transport of Amethopterin in Diplococcus pneumoniae
Physical State in Which Naphthalene and Bibenzyl are Utilized by Bacteria
 APPLIED MicRosoLowy, June 1972, p. 1077-1081 Copyright i 1972 American Society for Microbiology Vol. 23, No. 6 Printed in U.S.A. Physical State in Which Naphthalene and Bibenzyl are Utilized by Bacteria
APPLIED MicRosoLowy, June 1972, p. 1077-1081 Copyright i 1972 American Society for Microbiology Vol. 23, No. 6 Printed in U.S.A. Physical State in Which Naphthalene and Bibenzyl are Utilized by Bacteria
(From the Laboratory of The Rockefeller Institute for Medical Researck, Department of Bacteriology, University of California, Berkeley)
 THE EFFECT OF TRYPSIN, CHYMOTRYPSIN, RIBONUCLEASE, AND DESOXYRIBONUCLEASE ON ACTIVE, INACTIVE, AND REVERSIBLY INACTIVATED MEGATHERIUM PHAGE BY JOHN H. NORTHROP WITH THE ~CHNICAL ASSISTANCE OF MAttE KL~G
THE EFFECT OF TRYPSIN, CHYMOTRYPSIN, RIBONUCLEASE, AND DESOXYRIBONUCLEASE ON ACTIVE, INACTIVE, AND REVERSIBLY INACTIVATED MEGATHERIUM PHAGE BY JOHN H. NORTHROP WITH THE ~CHNICAL ASSISTANCE OF MAttE KL~G
Penicillin-Induced Lysis of Streptococcus multans
 INFECTION AND IMMUNITY, Nov 1984, p 442-447 19-9567/84/11442-6$2/ Copyright 1984, American Society for Microbiology Vol 46, No 2 Penicillin-Induced Lysis of Streptococcus multans TIMOTHY A KRAL* AND MATTHEW
INFECTION AND IMMUNITY, Nov 1984, p 442-447 19-9567/84/11442-6$2/ Copyright 1984, American Society for Microbiology Vol 46, No 2 Penicillin-Induced Lysis of Streptococcus multans TIMOTHY A KRAL* AND MATTHEW
International Journal of Pharma and Bio Sciences
 International Journal of Pharma and Bio Sciences RESEARCH ARTICLE BIO TECHNOLOGY OPTIMIZATION OF PHYSICAL CONDITIONS FOR THE PRODUCTION OF L- GLUTAMIC ACID BY A MUTANT Micrococcus glutamicus AB 100. *
International Journal of Pharma and Bio Sciences RESEARCH ARTICLE BIO TECHNOLOGY OPTIMIZATION OF PHYSICAL CONDITIONS FOR THE PRODUCTION OF L- GLUTAMIC ACID BY A MUTANT Micrococcus glutamicus AB 100. *
PeriPreps Periplasting Kit
 Cat. No. PS81100 The PeriPreps Periplasting Kit facilitates the release of proteins contained within the periplasmic space of E. coli cells by combining digestion of the cell wall using Ready-Lyse Lysozyme
Cat. No. PS81100 The PeriPreps Periplasting Kit facilitates the release of proteins contained within the periplasmic space of E. coli cells by combining digestion of the cell wall using Ready-Lyse Lysozyme
Effects of Cell Wall Inhibitors on Cell Division in Streptococcus mutans
 INFECTION AND IMMUNITY, July 1983, P. 28-32 19-9567/83/728-5$2./ Copyright 1983, American Society for Microbiology Vol. 41, No. 1 Effects of Cell Wall Inhibitors on Cell Division in Streptococcus mutans
INFECTION AND IMMUNITY, July 1983, P. 28-32 19-9567/83/728-5$2./ Copyright 1983, American Society for Microbiology Vol. 41, No. 1 Effects of Cell Wall Inhibitors on Cell Division in Streptococcus mutans
Isolation, partial purification and characterization of α-amylase from Bacillus subtilis
 Journal of Microbiology and Biotechnology Research Scholars Research Library J. Microbiol. Biotech. Res., 2014, 4 (1):49-54 (http://scholarsresearchlibrary.com/archive.html) ISSN : 2231 3168 CODEN (USA)
Journal of Microbiology and Biotechnology Research Scholars Research Library J. Microbiol. Biotech. Res., 2014, 4 (1):49-54 (http://scholarsresearchlibrary.com/archive.html) ISSN : 2231 3168 CODEN (USA)
A Discovery Laboratory Investigating Bacterial Gene Regulation
 Chapter 8 A Discovery Laboratory Investigating Bacterial Gene Regulation Robert Moss Wofford College 429 N. Church Street Spartanburg, SC 29307 mosssre@wofford.edu Bob Moss is an Associate Professor of
Chapter 8 A Discovery Laboratory Investigating Bacterial Gene Regulation Robert Moss Wofford College 429 N. Church Street Spartanburg, SC 29307 mosssre@wofford.edu Bob Moss is an Associate Professor of
Gary Ketner, PhD Johns Hopkins University. Treatment of Infectious Disease: Drugs and Drug Resistance
 This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike License. Your use of this material constitutes acceptance of that license and the conditions of use of materials on this
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike License. Your use of this material constitutes acceptance of that license and the conditions of use of materials on this
INDUCTION OF LACTOSE UTILIZATION IN STAPHYLOCOCCUS AUREUS
 INDUCTION OF LACTOSE UTILIZATION IN STAPHYLOCOCCUS AUREUS J. K. McCLATCHY AND E. D. ROSENBLUM Department of Microbiology, The University of Texas Soutthwestern Medical School, ABSTRACT Received for publication
INDUCTION OF LACTOSE UTILIZATION IN STAPHYLOCOCCUS AUREUS J. K. McCLATCHY AND E. D. ROSENBLUM Department of Microbiology, The University of Texas Soutthwestern Medical School, ABSTRACT Received for publication
Protein isotopic enrichment for NMR studies
 Protein isotopic enrichment for NMR studies Protein NMR studies ARTGKYVDES sequence structure Structure of protein- ligand, protein-protein complexes Binding of molecules (perturbation mapping) Dynamic
Protein isotopic enrichment for NMR studies Protein NMR studies ARTGKYVDES sequence structure Structure of protein- ligand, protein-protein complexes Binding of molecules (perturbation mapping) Dynamic
Escherichia coli. then were added to give the desired medium. England Nuclear Corp., Boston, Mass. media (2, 3). The present study shows that the
 JOURNAL OF BACTERIOLOGY, Feb., 1966 Vol. 91, No. 2 Copyright @ 1966 American Society for Microbiology Printed hn U.S.A. Mechanism of Action of Nalidixic Acid on Escherichia coli III. Conditions Required
JOURNAL OF BACTERIOLOGY, Feb., 1966 Vol. 91, No. 2 Copyright @ 1966 American Society for Microbiology Printed hn U.S.A. Mechanism of Action of Nalidixic Acid on Escherichia coli III. Conditions Required
6/28/2016. Control of Microbial Growth. Method. Terminology. Disinfectants and Antiseptics
 Control of Microbial Growth Disinfectants and Antiseptics 1 Method Three approaches for the control of microbial growth Chemical Disinfectants and antiseptics Physical Heat Ultraviolet Irradiations Mechanical
Control of Microbial Growth Disinfectants and Antiseptics 1 Method Three approaches for the control of microbial growth Chemical Disinfectants and antiseptics Physical Heat Ultraviolet Irradiations Mechanical
Data Sheet. TCR activator / PD-L1 - CHO Recombinant Cell line Cat. #: 60536
 Data Sheet TCR activator / PD-L1 - CHO Recombinant Cell line Cat. #: 60536 Product Description Recombinant CHO-K1 cells constitutively expressing human PD-L1 (Programmed Cell Death 1 Ligand 1, CD274, B7
Data Sheet TCR activator / PD-L1 - CHO Recombinant Cell line Cat. #: 60536 Product Description Recombinant CHO-K1 cells constitutively expressing human PD-L1 (Programmed Cell Death 1 Ligand 1, CD274, B7
2120 Lab. Week 11. Experiments 13,14,21. Kirby Bauer, TDT, Chemicals
 2120 Lab Week 11 Experiments 13,14,21 Kirby Bauer, TDT, Chemicals Controlling Microorganisms Decontamination: Physical, chemical, and mechanical methods to destroy or reduce undesirable microbes in a given
2120 Lab Week 11 Experiments 13,14,21 Kirby Bauer, TDT, Chemicals Controlling Microorganisms Decontamination: Physical, chemical, and mechanical methods to destroy or reduce undesirable microbes in a given
Flagellum Growth and Regeneration in the True Slime Mould!Didymium nigripes
 Journal of General Microbiology (1972), 72,429437 Printed in Great Britain 429 Flagellum Growth and Regeneration in the True Slime Mould!Didymium nigripes By SYLVI J. KERR* Department of Zoology, University
Journal of General Microbiology (1972), 72,429437 Printed in Great Britain 429 Flagellum Growth and Regeneration in the True Slime Mould!Didymium nigripes By SYLVI J. KERR* Department of Zoology, University
Sebastian Hernandez and Justo M. Mata Compania Espanola de Penicilina y Antibioticos, S. A., Madrid, Spain
 42 THE JOURNAL OF ANTIBIOTICS JAN. 1971 AZIRINOMYCIN. MICROBIAL PRODUCTION AND BIOLOGICAL CHARACTERISTICS Edward O. Stapley, David Hendlin, Marion Jackson and A. Kathrine Miller Department of Basic Microbiological
42 THE JOURNAL OF ANTIBIOTICS JAN. 1971 AZIRINOMYCIN. MICROBIAL PRODUCTION AND BIOLOGICAL CHARACTERISTICS Edward O. Stapley, David Hendlin, Marion Jackson and A. Kathrine Miller Department of Basic Microbiological
Biofilm Protocol Optimization For Pseudomonas aeruginosa. Introduction. Materials and Methods. Culture Media, Incubation Time, and Biofilm Measurement
 Biofilm Protocol Optimization For Pseudomonas aeruginosa Culture Media, Incubation Time, and Biofilm Measurement Introduction In addition to the conventional arsenal of antibiotic resistance mechanisms
Biofilm Protocol Optimization For Pseudomonas aeruginosa Culture Media, Incubation Time, and Biofilm Measurement Introduction In addition to the conventional arsenal of antibiotic resistance mechanisms
