Department Of Pathology Point of Care Testing POC Quick Vue In-Line Strep A Version#4
|
|
|
- Brianna May
- 5 years ago
- Views:
Transcription
1 Department Of Pathology Point of Care Testing POC Quick Vue In-Line Strep A Version#4 Printed copies are for reference only. Please refer to the electronic copy for the latest version. A. Purpose: The QuickVue In-Line One-Step Strep A test allows for the rapid detection of group A streptococcal antigen directly from patient throat swab specimens. The test is intended for use as an aid in the diagnosis of group A streptococcal infection. B. Principle: To perform the test, a throat swab is collected and inserted into the test cartridge. The extraction solutions are mixed, resulting in a green color change, and added to the sample well. The extracted sample migrates by capillary action through a labeled pad consisting of a pink label containing rabbit polyclonal anti-strep A antibody and a blue control label. If the extracted solution contains strep antigen, the antigen will bind to the antibody coupled to the pink test label, which will, in turn, bind a second rabbit polyclonal anti-step A antibody spotted on the membrane, resulting in the formation of a pink-to-purple test line next to the imprinted T on the Test Cassette (positive result). The blue control line will also appear next to the letter C on the Test Cassette indicating that the reagents were mixed and added properly, proper volume of fluid entered the Test Cassette, and capillary flow occurred. A blue Control Line should always appear in a proper functioning Test Cassette. If strep A is not present, or at very low levels, only a blue Control Line will be visible (negative result). C. Policy: 1. A physician/pa s order is required 2. LifeBridge Health Trainers will be identified and trained on each unit performing Gastroccult and Strep A. 3. Certified Trainers will include an in service, satisfactory completion of a written examination and performance of a test. 4. Recertification will be performed yearly. 5. A record of Certified Operators and Trainers will be maintained on each nursing unit, Staff Development, and with the Department of Pathology. 6. Bedside Testing Team Leader/Technical Specialist will supply a report identifying problems to the Director of each unit. Effective Date: 09/04/2012 1
2 7. Documentation of follow-up investigation and corrective actions taken will be returned to Pathology to be maintained by Bedside Testing. 8. Results will be recorded in Powerform or on the Point-of-Care Testing Result Sheet. On Nursing Units at Sinai results will be entered as Other Tests in the Glucose Meter. 9. Any data entry or corrections of data entry made by operators will need to see Nursing Policies and Procedures for electronic entry and corrections when mistakes are made. 10. Specimens for testing at bedside and In-Lab are not to be obtained at the same time. Specimens need to be obtained individually. D. Specimen: 1. Specimen Type: Throat swab specimen 2. Patient Preparation: N/A 3. Handling Conditions: a. Sampling: Hold the tongue down with a sterile tongue depressor. Both tonsillar areas, the posterior pharynx, and any other areas of inflammation, ulceration or exudation should be sampled. Use only rayon tipped swabs on solid plastic shafts for specimen collection. Do not use calcium alginate, cotton tipped, hollow shafts or wooden shaft swabs. b. If a culture is desired, dual swabs or two sequential swabs can be taken to ensure adequate sample for the culture procedure. One swab alone cannot be used to perform the QuickVue test followed by a culture, as the extraction solution will destroy any viable bacteria on the swab. 4. Storage Requirements: Swab specimens should be processed as soon as possible after collection. 5. Rejection Criteria: N/A E. Equipment and Materials: 1. Equipment: N/A 2. Materials/ Supplies: a. Individually packaged test cassettes (25 or 75) containing a rabbit polyclonal antibody to Strep A (Test Line) and a control rabbit polyclonal antibody capable of binding the blue-colored control label (Control Line). b. Extraction Solution Bottles (25 or 75) containing 4M sodium nitrite with 0.01% thimerosal (0.6mL) and 0.2M acetic acid (0.65mL) inside a glass ampule. c. Individually packaged sterile rayon-tipped swabs on solid shafts (25 or 75). 1. Positive control swabs (+) (1 or 3) containing heat inactivated group A Streptococcus. 2. Negative control swabs (-) (1 or 3) containing heat inactivated group C Streptococcus. F. Preparation: N/A G. Performance Parameters: Effective Date: 09/04/2012 2
3 1. For in vitro diagnostic use only. 2. Do not use beyond the expiration date printed on the outside of the box. 3. Use appropriate precaution in the collection, handling, storage, and disposal of specimens, control swabs, and used kit contents. Discard used materials in a proper biohazard container. 4. The test cassette must remain sealed in the protective foil pouch until just prior to use. 5. If the Extraction Solution bottle is missing the glass ampule, or the solution is green prior to breaking the ampule, discard and use another Extraction Solution bottle. H. Storage Requirements: Store kit at room temperature (15 o C) out of direct sunlight. Kit contents are stable until the expiration date printed on the outer box. Do not freeze. I. Quality Control: The QuickVue In-Line One-Step Strep A test contains built-in control features. The manufacturer s recommendation for daily quality control is to document these controls for the first sample run each shift. 1. A control of the extraction procedure is provided by a color change from clear to green as the Extraction Reagents are mixed. The color change is an indication of extraction integrity and is also an indication that the extraction procedure was correctly performed. 2. The two color result format provides a clear-cut readout for positive and negative results. The appearance of a blue Control Line next to the letter C provides several forms of control. 3. First, detection components for the specimen and internal control are processed concurrently using identical procedures; therefore, the appearance of the Control Line assures that functional activity of the detection component is maintained. 4. Secondly, the appearance of the Control Line also ensures that the foil pouch integrity has been maintained and the Test Cassette has been stored in a manner so as to not compromise it s functionality. 5. Third, the appearance of the Control Line indicates that proper volume of fluid entered the Test Cassette and capillar flow occurred. This would indicate that the Test Cassette was assembled properly, by acting as a check for membrane interfaces and proper positioning of components. 6. Lastly, if the Control Line fails to develop, the test result is invalid. 7. A negative background control is provided by the clearing of background color in the Result Window and indicates that there were no immunological interfering substances in the specimen. The background color in the result window should be white to light pink within 5 minutes, and not interfere with the reading of the test result. If background color remains in the Result Window which interferes with your ability to read the test result, the test result may be invalid. Contact the Bedside Testing Coordinator or call Quindel Technical Effective Date: 09/04/2012 3
4 Assistance. 8. Materials Used: External controls may also be used to assure that reagent and assay procedure is performing properly. a. Positive and Negative Control Swabs are supplied in the kit. The positive (+) control swab is stored in the pink-capped tube; the negative (-) control swab is stored in the blue-capped tube. The positive control used must be traceable to an ATCC strain from 19615, and the negative control must be traceable to an ATCC non-group A Strep organism. To test: remove control swab from it s container and insert it into the Test Cassette Swab chamber, and continue with the assay as instructed in PROCEDURE step 6.3 below. b. Liquid Controls (Catalog #0354) may also be utilized. Hold the bottle vertically and place one free falling drop of the liquid control onto a sterile swab provided in the kit. Insert the swab into a Test Cassette Swab Chamber, and continue testing as outlined in PROCEDURE step 6.3 below. J. Proficiency Testing Surveys. a. Place a clean tube from the extraction kit into a test tube rack. b. Prepare the Extraction Solution Bottle as described in steps 6.3 and 6.4 below. c. Dispense 8 drops from the extraction solution bottle into the tube. Place the proficiency swab into the tube. Hold the bottom of the tube so the swab head is slightly compressed. Rotate the swab three times. d. Wait one minute. e. Express all the liquid from the swab head in the tube by rolling the swab against the inside of the tube and pressing slightly as it is withdrawn from the tube. Discard the swab. g. Fill the disposable dropper to the fill line with the solution from the tube and add the contents into the test Cassette Swab Chamber. h. Read the result at 5 minutes. K. Preparation and Handling: N/A L. Frequency Run: Positive and Negative controls will be tested for each 25 test kits. Bedside Testing Team Leader/Technical Specialist prior to distribution to units will perform Quality Control on each box of 25 tests. Each box will be labeled that QC was performed and that patient testing can now be performed. M. Tolerance Limits: N/A N. Corrective Action: N/A O. Recording and Storage of Data: N/A Effective Date: 09/04/2012 4
5 P. Procedure: 1. Apply gloves 2. Remove the Test Cassette from the foil pouch and place on a clean, dry, level surface. 3. Using the notch at the back of the chamber as a guide, gently insert the specimen into the Test Cassette Swab Chamber. 4. Hold the Extraction Solution bottle vertically, so that the tip is pointed upward. Carefully crush the glass ampule inside the bottle. 5. Invert the bottle, and shake vigorously four or five times to mix the solutions. Solution will turn green after the ampule is broken. Do not use the bottle if solution is green prior to breaking the ampule. Note: Solution must be used immediately. 6. Remove the cap. Hold the bottle pointing downward, approx. 1 to 1½ inches above the swab chamber. Using constant pressure, express 8 drops onto the swab tip located in the swab chamber. 7. Begin timing. If liquid has not moved across the Result Window in 1 minute, remove swab and gently re-insert it. The test cassette should not be moved or jarred again until the assay is complete and ready for interpretation. 8. Read results at 5 minutes. (Note: some positive results may be seen earlier). Q. Calculations: N/A R. Reporting Results: 1. Positive results: The appearance of any pink-to-purple line next to the letter T in the result window, along with a blue Control Line next to the letter C indicates a positive result for group A Streptococcus. 2. Negative Results: The appearance of only the blue Control Line next to the letter C in the results window indicates a negative result. A negative QuickVue test indicates that the specimen is a presumptive negative for the presences of group A Streptococcus. (Note: A negative test result might occur if the level of extracted antigen in a sample is below the sensitivity of the test. A follow up culture is recommended for all negative patient samples. 3. Invalid Results: The test result is invalid if the blue Control Line is not visible at 5 minutes. If this occurs either: retest using a new specimen and a new Test Cassette; call for assistance by contacting the Bedside Testing Coordinator; or alternatively, contact Quidel Technical Assistance at Patient Results: Are charted in Powerform or using the Point-of-Care result sheets. The white copy goes on the patient s chart, and the yellow copy is to be sent to the Bedside Testing Coordinator in pathology. S. Procedure Notes: T. Reference Range: N/A U. Critical Value: N/A Effective Date: 09/04/2012 5
6 V. Reporting Format: Results will in Powerform or recorded on the Point-of-Care Result Sheet. W. Hazardous Materials: 1. The Extraction Solution bottle contains an acidic solution. If the solution contacts the skin or eyes, flush with large volumes of water. 2. The Extraction Solution bottle contains glass, break cautiously. X. Protective Equipment Requirements: Gloves V. Performance Characteristics: Multi-center field study evaluation of the QuickVue test was conducted,using sheep blood agar cultures followed by a latex agglutination assay confirmation as the standard. A total of 537 specimens were analyzed. 301 specimens were tested by users in the field, and 236 were frozen and shipped overnight to QUIDEL for testing. Specificity: in the field: 94% At QUIDEL: 99% Sensitivity: in the field: 87% At QUIDEL: 92% Overall Agreement between SBA culture and QuickVue was 96% (See Reference for details.) W. Liminations of Procedure X. Linearity: N/A Y. Interfering Substances Z. Chemical Interference: N/A AA. In vivo Interference: 1. Heavy colonization by Staphylococcus aureaus (>10 10 ) can yield false positive results. 2. Respiratory infections, including pharyngitis, can be caused by Streptococcus from serogroups other than groupa, as well as by other Pathogens 3. The QuickVue test will not differentiate asymptomatic carriers of group A Streptococcus from those exhibiting streptococcal infection. BB. References: QuickVue In-Line One-Step Strep A Test Physician s Office Laboratory Procedure Manual, March 1996 QUIDEL McKellar Court San Diego, CA Effective Date: 09/04/2012 6
CLIA Complexity: MODERATE
 CLIA Complexity: MODERATE INTENDED USE The is intended for the rapid detection of Group A Streptococcal antigen directly from throat swabs and beta-hemolytic colonies recovered from culture. This test
CLIA Complexity: MODERATE INTENDED USE The is intended for the rapid detection of Group A Streptococcal antigen directly from throat swabs and beta-hemolytic colonies recovered from culture. This test
SUMMARY AND EXPLANATION
 This Procedural Bulletin is intended to provide a ready outline reference for performance of the assay. These abbreviated directions for use are not intended to replace the complete package insert. It
This Procedural Bulletin is intended to provide a ready outline reference for performance of the assay. These abbreviated directions for use are not intended to replace the complete package insert. It
PROCEDURE MANUAL. Prepared By Date Adopted Supersedes Procedure # Review Date Revision Date Signature
 Procedure: CLIA Complexity: Moderate Procedure #: Prepared By Date Adopted Supersedes Procedure # Review Date Revision Date Signature Distributed to # of Copies Distributed to # of Copies This Procedural
Procedure: CLIA Complexity: Moderate Procedure #: Prepared By Date Adopted Supersedes Procedure # Review Date Revision Date Signature Distributed to # of Copies Distributed to # of Copies This Procedural
PROCEDURE MANUAL. Prepared By Date Adopted Supersedes Procedure # Review Date Revision Date Signature
 Procedure: CLIA Complexity: Waived Procedure #: Prepared By Date Adopted Supersedes Procedure # Review Date Revision Date Signature Distributed to # of Copies Distributed to # of Copies This Procedural
Procedure: CLIA Complexity: Waived Procedure #: Prepared By Date Adopted Supersedes Procedure # Review Date Revision Date Signature Distributed to # of Copies Distributed to # of Copies This Procedural
SAMPLE PROCEDURE 928-8, 07/11
 SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
CLIA Complexity: Waived for Finger-stick Whole Blood. Instructional video available online at quidel.com/sofia2lyme.
 Study the Package Insert and User Manual thoroughly before using Quick Reference Instructions. This is not a complete Package Insert. CLIA Complexity: Waived for Finger-stick Whole Blood Instructional
Study the Package Insert and User Manual thoroughly before using Quick Reference Instructions. This is not a complete Package Insert. CLIA Complexity: Waived for Finger-stick Whole Blood Instructional
QUICK REFERENCE INSTRUCTIONS For use with Sofia and Sofia 2. Rx only
 QUICK REFERENCE INSTRUCTIONS For use with Sofia and Sofia 2. Rx only Test Procedure Study the Package Insert and User Manual thoroughly before using Quick Reference Instructions. This is not a complete
QUICK REFERENCE INSTRUCTIONS For use with Sofia and Sofia 2. Rx only Test Procedure Study the Package Insert and User Manual thoroughly before using Quick Reference Instructions. This is not a complete
SAMPLE PROCEDURE , 11/07
 SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
REAGENTS AND MATERIALS SUPPLIED Control 2: 25-OH vitamin D in human serum with <0.1% Streptomycin Sulfate (2)
 For use with the Sofia Quantitative Vitamin D FIA test system only For in vitro diagnostic use INTENDED USE The Sofia Quantitative Vitamin D Control Set is intended for use as assayed quality control materials
For use with the Sofia Quantitative Vitamin D FIA test system only For in vitro diagnostic use INTENDED USE The Sofia Quantitative Vitamin D Control Set is intended for use as assayed quality control materials
Rapid-VIDITEST. RSV Card. One step RSV Card test for the detection of Respiratory Syncytial Virus from nasal specimens. Instruction manual
 Rapid-VIDITEST RSV Card One step RSV Card test for the detection of Respiratory Syncytial Virus from nasal specimens. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42
Rapid-VIDITEST RSV Card One step RSV Card test for the detection of Respiratory Syncytial Virus from nasal specimens. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42
1. APPROVED MALARIA RDTS
 1. APPROVED MALARIA RDTS There are many different Rapid Diagnostic Tests available in the market. To assist partners of the Three Diseases Fund in the selection of the appropriate RDT, the Fund Manager
1. APPROVED MALARIA RDTS There are many different Rapid Diagnostic Tests available in the market. To assist partners of the Three Diseases Fund in the selection of the appropriate RDT, the Fund Manager
QUICK REFERENCE INSTRUCTIONS For use with Sofia and Sofia 2.
 QUICK REFERENCE INSTRUCTIONS For use with Sofia and Sofia 2. Study the Package Insert and User Manual thoroughly before using Quick Reference Instructions. This is not a complete Package Insert. CLIA Complexity:
QUICK REFERENCE INSTRUCTIONS For use with Sofia and Sofia 2. Study the Package Insert and User Manual thoroughly before using Quick Reference Instructions. This is not a complete Package Insert. CLIA Complexity:
Strep A Test. Rev ,02/17
 Strep A Test Rev. 3812-6,02/17 Strep A Test CLIA Complexity: Waived 23-200-276 INTENDED USE The Sure-Vue Signature Strep A Test is a color immunochromatographic assay intended for the qualitative detection
Strep A Test Rev. 3812-6,02/17 Strep A Test CLIA Complexity: Waived 23-200-276 INTENDED USE The Sure-Vue Signature Strep A Test is a color immunochromatographic assay intended for the qualitative detection
Rapid-VIDITEST RSV. One step RSV Blister test for the detection of Respiratory Syncytial Virus from nasal specimens. Instruction manual
 Rapid-VIDITEST RSV One step RSV Blister test for the detection of Respiratory Syncytial Virus from nasal specimens. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice,
Rapid-VIDITEST RSV One step RSV Blister test for the detection of Respiratory Syncytial Virus from nasal specimens. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice,
RNAPro SAL ORAL SPECIMEN COLLECTION SYSTEM. Catalog Number RPSAL-701. Transparent Plastic Compression Tube. Compression Seal
 RNAPro SAL ORAL SPECIMEN COLLECTION SYSTEM Catalog Number RPSAL-701 Transparent Plastic Compression Tube Splitting Unit Equipped with Variable Filtration Media Compression Seal Ergonomically Correct Handle
RNAPro SAL ORAL SPECIMEN COLLECTION SYSTEM Catalog Number RPSAL-701 Transparent Plastic Compression Tube Splitting Unit Equipped with Variable Filtration Media Compression Seal Ergonomically Correct Handle
E. coli O26 Latex Test Kit
 Importance of STEC Determination Shiga toxin-producing Escherichia coli strains (non-o157 STEC) have become an increasing public health concern. Some of the non-o157 STEC possess the same range of virulence
Importance of STEC Determination Shiga toxin-producing Escherichia coli strains (non-o157 STEC) have become an increasing public health concern. Some of the non-o157 STEC possess the same range of virulence
QuickVue In-Line Strep A Test Safety Data Sheet Revision Date: April 27, Section 1 Product and Company Identification
 Manufacturer Information Quidel Corporation Phone: 1.800.874.1517 Web: quidel.com 10165 McKellar Court Fax: 1.858.453.4338 E-mail: qehs@quidel.com San Diego, CA 92121 Emergency # (24-Hour): 1.866.519.4752
Manufacturer Information Quidel Corporation Phone: 1.800.874.1517 Web: quidel.com 10165 McKellar Court Fax: 1.858.453.4338 E-mail: qehs@quidel.com San Diego, CA 92121 Emergency # (24-Hour): 1.866.519.4752
E.coli O157:H7 Rapid Test (Card)
 E.coli O157:H7 Rapid Test (Card) Cat. No.:DTS518 Pkg.Size: Intended use The E.coli O157:H7 Rapid Test is a one step coloured chromatographic immunoassay for the qualitative detection of Escherichia coli
E.coli O157:H7 Rapid Test (Card) Cat. No.:DTS518 Pkg.Size: Intended use The E.coli O157:H7 Rapid Test is a one step coloured chromatographic immunoassay for the qualitative detection of Escherichia coli
Accutest ValuPak Immunological Fecal Occult Blood Test
 INTENDED USE The Accutest ValuPak Immunological Fecal Occult Blood (IFOB) Test is a rapid chromatographic immunoassay for the qualitative detection of human occult blood in human fecal specimens. The device
INTENDED USE The Accutest ValuPak Immunological Fecal Occult Blood (IFOB) Test is a rapid chromatographic immunoassay for the qualitative detection of human occult blood in human fecal specimens. The device
INVALID. Rev /15 POSITIVE NEGATIVE. Ultra Strep A Test. 2 min 5 min. 10x. 3 drops 3 drops
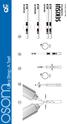 1 3 drops 3 drops Ultra Strep A Test 2 3 4 5 10x 2 min 5 min POSITIVE NEGATIVE INVALID Rev. 3101-2 06/15 Ultra Strep A Test CLIA Complexity: Waived INTENDED USE The OSOM Ultra Strep A Test is a color immunochromatographic
1 3 drops 3 drops Ultra Strep A Test 2 3 4 5 10x 2 min 5 min POSITIVE NEGATIVE INVALID Rev. 3101-2 06/15 Ultra Strep A Test CLIA Complexity: Waived INTENDED USE The OSOM Ultra Strep A Test is a color immunochromatographic
Hepatitis C virus Ab ELISA Kit
 Hepatitis C virus Ab ELISA Kit Catalog Number KA0291 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Principle of the Assay... 3 General Information...
Hepatitis C virus Ab ELISA Kit Catalog Number KA0291 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Principle of the Assay... 3 General Information...
HBeAg and HBeAg Ab ELISA Kit
 HBeAg and HBeAg Ab ELISA Kit Catalog Number KA0290 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay... 3 General
HBeAg and HBeAg Ab ELISA Kit Catalog Number KA0290 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay... 3 General
Human Myostatin, ELISA Kit (MSTN)
 Human Myostatin, ELISA Kit (MSTN) 96 Tests Catalog Number: MBS733837 Store all reagents at 2-8 C Valid Period: six months For samples: Cell culture fluid & body fluid & tissue homogenate Serum or blood
Human Myostatin, ELISA Kit (MSTN) 96 Tests Catalog Number: MBS733837 Store all reagents at 2-8 C Valid Period: six months For samples: Cell culture fluid & body fluid & tissue homogenate Serum or blood
Goat IgG ELISA Catalog #:
 INTENDED USE The IMMUNO-TEK Goat IgG ELISA Kit is a rapid, easy to use enzyme linked immunosorbent assay (ELISA * ) designed for the measurement of goat IgG in goat colostrum, milk, serum, plasma or other
INTENDED USE The IMMUNO-TEK Goat IgG ELISA Kit is a rapid, easy to use enzyme linked immunosorbent assay (ELISA * ) designed for the measurement of goat IgG in goat colostrum, milk, serum, plasma or other
NATIONAL FOOD SAFETY FOUNDATION
 NATIONAL FOOD SAFETY FOUNDATION RAPID TESTING KITS DETECTION E.COLI 0157 IN FOOD MATICES AND LIQUIDS USING E.COLI RAPID TESTING KIT INTRODUCTION E. coli serotype O157 is a gram-negative rod-shaped bacterium.
NATIONAL FOOD SAFETY FOUNDATION RAPID TESTING KITS DETECTION E.COLI 0157 IN FOOD MATICES AND LIQUIDS USING E.COLI RAPID TESTING KIT INTRODUCTION E. coli serotype O157 is a gram-negative rod-shaped bacterium.
Rat IgG ELISA Catalog #:
 INTENDED USE The IMMUNO-TEK Rat IgG ELISA* Kit is a rapid, easy to use enzyme linked immunosorbent assay (ELISA) designed for the measurement of rat IgG in rat serum, plasma, saliva, mucosa, cell culture
INTENDED USE The IMMUNO-TEK Rat IgG ELISA* Kit is a rapid, easy to use enzyme linked immunosorbent assay (ELISA) designed for the measurement of rat IgG in rat serum, plasma, saliva, mucosa, cell culture
Guinea pig Carboxy Terminal. Propeptide of Type 1 Procollagen. (PICP) ELISA kit
 19. If using an automated washer, the operating instructions for washing equipment should be carefully followed. 20. Assay Procedure Preliminary notes: Do not mix reagents from different lots. It is recommended
19. If using an automated washer, the operating instructions for washing equipment should be carefully followed. 20. Assay Procedure Preliminary notes: Do not mix reagents from different lots. It is recommended
RapidChek SELECT Salmonella Test Kit
 RapidChek SELECT Salmonella Test Kit Part #: 7000188-7000197 AOAC Approved Protocols: This test kit s performance was reviewed by AOAC Research Institute and was found to perform to the manufacturer s
RapidChek SELECT Salmonella Test Kit Part #: 7000188-7000197 AOAC Approved Protocols: This test kit s performance was reviewed by AOAC Research Institute and was found to perform to the manufacturer s
Department of Laboratories St. Louis, MO ISSUE DATE: June 2007 REVISION DATE: May 2012 REVIEWED DATE: May 2018 PRINCIPLE:
 Page 1 of 7 PROCEDURE: LEAD ESA LEADCARE II / HEALTHY KIDS EXPRESS ISSUE DATE: June 2007 REVISION DATE: May 2012 REVIEWED DATE: May 2018 PRINCIPLE: The LEADCARE II System relies on electrochemistry and
Page 1 of 7 PROCEDURE: LEAD ESA LEADCARE II / HEALTHY KIDS EXPRESS ISSUE DATE: June 2007 REVISION DATE: May 2012 REVIEWED DATE: May 2018 PRINCIPLE: The LEADCARE II System relies on electrochemistry and
Mouse Peptide YY (PYY) ELISA
 Mouse Peptide YY (PYY) ELISA For the quantitative determination of mouse PYY in serum, plasma, cell culture fluid and other biological fluids Cat. No. KT-58705 For Research Use Only. Not for use in diagnostic
Mouse Peptide YY (PYY) ELISA For the quantitative determination of mouse PYY in serum, plasma, cell culture fluid and other biological fluids Cat. No. KT-58705 For Research Use Only. Not for use in diagnostic
E. coli O157 Test Strip Kit Catalog #: 1062
 BIOO FOOD AND FEED SAFETY E. coli O157 Test Strip Kit Catalog #: 1062 BIOO Scientific Corp. 2011 TABLE OF CONTENTS GENERAL INFORMATION... 1 Product Description... 1 Procedure Overview... 1 Kit Contents,
BIOO FOOD AND FEED SAFETY E. coli O157 Test Strip Kit Catalog #: 1062 BIOO Scientific Corp. 2011 TABLE OF CONTENTS GENERAL INFORMATION... 1 Product Description... 1 Procedure Overview... 1 Kit Contents,
Human Angiotensin 2 (Ang2) ELISA
 Human Angiotensin 2 (Ang2) ELISA For the quantitative determination of human Ang2 in serum, plasma, cell culture fluid and other biological fluids Cat. No. KT-52748 For Research Use Only. Not for use in
Human Angiotensin 2 (Ang2) ELISA For the quantitative determination of human Ang2 in serum, plasma, cell culture fluid and other biological fluids Cat. No. KT-52748 For Research Use Only. Not for use in
Quick Reference Instructions
 PROFILE -V Quick Reference Instructions Reader System Before performing the test, refer to the PROFILE -V MEDTOXScan Drugs of Abuse Test System Insert and MEDTOXScan User Manual for complete operating
PROFILE -V Quick Reference Instructions Reader System Before performing the test, refer to the PROFILE -V MEDTOXScan Drugs of Abuse Test System Insert and MEDTOXScan User Manual for complete operating
Immunoassay Kit Catalog # KCA0021. Canine. C-Reactive Protein
 Immunoassay Kit Catalog # KCA0021 Canine C-Reactive Protein BioSource International, Inc. 542 Flynn Road Camarillo, California 93012 USA Tel: 805-987-0086 800-242-0607 FAX: 805-987-3385 email: tech.support@biosource.com
Immunoassay Kit Catalog # KCA0021 Canine C-Reactive Protein BioSource International, Inc. 542 Flynn Road Camarillo, California 93012 USA Tel: 805-987-0086 800-242-0607 FAX: 805-987-3385 email: tech.support@biosource.com
Glyphosate ELISA Kit. Catalog Number KA assays Version: 07. Intended for research use only.
 Glyphosate ELISA Kit Catalog Number KA1419 96 assays Version: 07 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay... 3 General Information...
Glyphosate ELISA Kit Catalog Number KA1419 96 assays Version: 07 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay... 3 General Information...
The kit is a sandwich enzyme immunoassay for in vitro quantitative measurement of CCSA-4 in. Reagents Quantity Reagents Quantity
 Catalog No: YLA0017HU 96 Test Human colon cancer-specific antigen-4(ccsa-4)elisa Kit FOR RESEARCH USE ONLY; NOT FOR THERAPEUTIC OR DIAGNOSTIC APPLICATIONS! PLEASE READ THROUGH ENTIRE procedure BEFORE BEGINNING!
Catalog No: YLA0017HU 96 Test Human colon cancer-specific antigen-4(ccsa-4)elisa Kit FOR RESEARCH USE ONLY; NOT FOR THERAPEUTIC OR DIAGNOSTIC APPLICATIONS! PLEASE READ THROUGH ENTIRE procedure BEFORE BEGINNING!
Respiratory Syncytial Virus IgM ELISA
 Respiratory Syncytial Virus IgM ELISA For the quantitative determination of IgM antibodies to Respiratory Syncytial Virus in serum. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog
Respiratory Syncytial Virus IgM ELISA For the quantitative determination of IgM antibodies to Respiratory Syncytial Virus in serum. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog
I-STAT POCT System PRINCIPLE:
 PRINCIPLE: I-STAT POCT System The i-stat system uses a single disposable cartridge. The cartridge, which has been filled with fresh blood, is placed into a hand-held analyzer for analysis. The results
PRINCIPLE: I-STAT POCT System The i-stat system uses a single disposable cartridge. The cartridge, which has been filled with fresh blood, is placed into a hand-held analyzer for analysis. The results
Rapid-VIDITEST E. coli O157:H7
 Rapid-VIDITEST E. coli O157:H7 One Step E.coli O157:H7 Card test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565, www.vidia.cz
Rapid-VIDITEST E. coli O157:H7 One Step E.coli O157:H7 Card test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565, www.vidia.cz
DRG International, Inc., USA Fax: (908)
 Please use only the valid version of the package insert provided with the kit. This kit is intended for Research Use Only. Not for use in diagnostic procedures. 1 INTENDED USE AND PRINCIPLE OF THE TEST
Please use only the valid version of the package insert provided with the kit. This kit is intended for Research Use Only. Not for use in diagnostic procedures. 1 INTENDED USE AND PRINCIPLE OF THE TEST
Human Selenoprotein P1, Plasma (SEPP1) ELISA
 Human Selenoprotein P1, Plasma (SEPP1) ELISA For the quantitative determination of human SEPP1 in serum, plasma, cell culture fluid and other biological fluids Cat. No. KT-50806 For Research Use Only.
Human Selenoprotein P1, Plasma (SEPP1) ELISA For the quantitative determination of human SEPP1 in serum, plasma, cell culture fluid and other biological fluids Cat. No. KT-50806 For Research Use Only.
Human Collagen Type III (COL3) ELISA
 Human Collagen Type III (COL3) ELISA For the quantitative determination of human COL3 in serum, plasma, cell culture fluid and other biological fluids Cat. No. KT-61018 For Research Use Only. Not for use
Human Collagen Type III (COL3) ELISA For the quantitative determination of human COL3 in serum, plasma, cell culture fluid and other biological fluids Cat. No. KT-61018 For Research Use Only. Not for use
Luteinizing Hormone ELISA kit
 55R-IB19104 Luteinizing Hormone ELISA kit Enzyme Immunoassay for the determination of LH in human serum 1. Principle of the test: The LH ELISA kit is a solid phase direct sandwich method. The samples and
55R-IB19104 Luteinizing Hormone ELISA kit Enzyme Immunoassay for the determination of LH in human serum 1. Principle of the test: The LH ELISA kit is a solid phase direct sandwich method. The samples and
Mouse Gonadotropin Releasing Hormone (GnRH) ELISA
 KAMIYA BIOMEDICAL COMPANY Mouse Gonadotropin Releasing Hormone (GnRH) ELISA For the quantitative determination of mouse GnRH in serum, plasma, cell culture fluid and other biological fluids Cat. No. KT-58182
KAMIYA BIOMEDICAL COMPANY Mouse Gonadotropin Releasing Hormone (GnRH) ELISA For the quantitative determination of mouse GnRH in serum, plasma, cell culture fluid and other biological fluids Cat. No. KT-58182
This kit is intended for Research Use Only. Not for use in diagnostic procedures.
 This kit is intended for Research Use Only. Not for use in diagnostic procedures. Introduction The DRG Herpes Simplex Virus Type 1+2 IgG Enzyme Immunoassay Kit provides materials for determination of IgG-class
This kit is intended for Research Use Only. Not for use in diagnostic procedures. Introduction The DRG Herpes Simplex Virus Type 1+2 IgG Enzyme Immunoassay Kit provides materials for determination of IgG-class
Chicken IgY (IgG) ELISA Catalog Number:
 Chicken IgY (IgG) ELISA : 0801010 INTENDED USE Chicken IgY is the designation for the IgG-like molecule found in avian species. The IMMUNOtek Chicken IgY (IgG) ELISA is a rapid, easy to use enzyme linked
Chicken IgY (IgG) ELISA : 0801010 INTENDED USE Chicken IgY is the designation for the IgG-like molecule found in avian species. The IMMUNOtek Chicken IgY (IgG) ELISA is a rapid, easy to use enzyme linked
Influenza A IgG ELISA
 Influenza A IgG ELISA For the quantitative determination of IgG-class antibodies against Influenza A virus in human serum or plasma (citrate). For Research use Only. Not For Use In Diagnostic Procedures.
Influenza A IgG ELISA For the quantitative determination of IgG-class antibodies against Influenza A virus in human serum or plasma (citrate). For Research use Only. Not For Use In Diagnostic Procedures.
DiviTum V2. User s Manual IVD. This User s Manual is intended for DiviTum V2. For use in the United States and Japan only for research purposes
 DiviTum V2 User s Manual This User s Manual is intended for DiviTum V2 For use in the United States and Japan only for research purposes IVD 40 Ref. 933 rev. EN 2 1 Contents DIVITUM ASSAY FOR MEASURING
DiviTum V2 User s Manual This User s Manual is intended for DiviTum V2 For use in the United States and Japan only for research purposes IVD 40 Ref. 933 rev. EN 2 1 Contents DIVITUM ASSAY FOR MEASURING
INSTRUCTIONS FOR USE
 INSTRUCTIONS FOR USE INTENDED USE KWIK-STIK, KWIK-STIK Plus and LYFO DISK microorganisms are intended to be used as controls to verify the performance of assays, reagents or media that are intended to
INSTRUCTIONS FOR USE INTENDED USE KWIK-STIK, KWIK-STIK Plus and LYFO DISK microorganisms are intended to be used as controls to verify the performance of assays, reagents or media that are intended to
Bovine IgG ELISA Catalog #:
 INTENDED USE The IMMUNO-TEK Bovine IgG ELISA Kit is a rapid, easy to use enzyme linked immunosorbent assay (ELISA) designed for the measurement of bovine IgG in bovine colostrum, milk, serum, plasma or
INTENDED USE The IMMUNO-TEK Bovine IgG ELISA Kit is a rapid, easy to use enzyme linked immunosorbent assay (ELISA) designed for the measurement of bovine IgG in bovine colostrum, milk, serum, plasma or
PERFUSION TECHNOLOFISTS OF GREATER CHICAGO, INC. POLICY AND PROCEDURE MEDTRONIC HEMOTEC ACTIVATED CLOTTING TIME (ACT) OPERATING PROCEDURE
 DATE OF REVISION Page 1 of 13 (ACT) OPERATING PROCEDURE PRINCIPLE: The ACT test is a definitive test used to monitor the anticoagulant effect of heparin. It measures the clotting time of fresh whole blood
DATE OF REVISION Page 1 of 13 (ACT) OPERATING PROCEDURE PRINCIPLE: The ACT test is a definitive test used to monitor the anticoagulant effect of heparin. It measures the clotting time of fresh whole blood
1 MICROTITER PLATE 96 wells 2 ENZYME CONJUGATE 10.0 ml 1 vial 3 STANDARD.1 0 pg/ml 1 vial 4 STANDARD pg/ml 1 vial 5 STANDARD.
 Rabbit Thyrotropin Releasing Hormone (TRH) Elisa Kit 96 Tests Catalog Number: MBS725153 Store all reagents at 2-8 C Valid Period:six months FOR LABORATORY RESEARCH USE ONLY. NOT FOR THERAPEUTIC OR DIAGNOSTIC
Rabbit Thyrotropin Releasing Hormone (TRH) Elisa Kit 96 Tests Catalog Number: MBS725153 Store all reagents at 2-8 C Valid Period:six months FOR LABORATORY RESEARCH USE ONLY. NOT FOR THERAPEUTIC OR DIAGNOSTIC
Human IL-6 ELISA Set
 Human IL-6 ELISA Set Catalog No. CDK082B Quantity: 10 x 96 tests PRODUCT SPECIFICATIONS : Specificity: Recognizes both natural and recombinant human IL-6 Range: 6.25 pg / ml - 200 pg / ml Sensitivity:
Human IL-6 ELISA Set Catalog No. CDK082B Quantity: 10 x 96 tests PRODUCT SPECIFICATIONS : Specificity: Recognizes both natural and recombinant human IL-6 Range: 6.25 pg / ml - 200 pg / ml Sensitivity:
Sample for Use as a Guide Revised IDEXX ENTEROLERT TEST METHOD FOR THE DETECTION OF ENTEROCOCCI IN WATER
 IDEXX ENTEROLERT TEST METHOD FOR THE DETECTION OF ENTEROCOCCI IN WATER 1 IDEXX ENTEROLERT TEST METHOD FOR THE DETECTION OF ENTEROCOCCI IN WATER 1. Scope and Application 1.1. This method is intended for
IDEXX ENTEROLERT TEST METHOD FOR THE DETECTION OF ENTEROCOCCI IN WATER 1 IDEXX ENTEROLERT TEST METHOD FOR THE DETECTION OF ENTEROCOCCI IN WATER 1. Scope and Application 1.1. This method is intended for
Escherichia coli O157:H7 (E.coli O157:H7) ELISA
 Product information User s Manual Escherichia coli O157:H7 (E.coli O157:H7) ELISA Enzyme-linked immune-sorbent assay for the determination of E.coli O157:H7 in food or water. Catalog No.: BE69221 96 Storage:
Product information User s Manual Escherichia coli O157:H7 (E.coli O157:H7) ELISA Enzyme-linked immune-sorbent assay for the determination of E.coli O157:H7 in food or water. Catalog No.: BE69221 96 Storage:
NT-proBNP (Human) ELISA Kit
 NT-proBNP (Human) ELISA Kit Catalog Number KA3099 96 assays Version: 21 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
NT-proBNP (Human) ELISA Kit Catalog Number KA3099 96 assays Version: 21 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Human Bordetella Pertussis IgG ELISA kit
 Human Bordetella Pertussis IgG ELISA kit Catalog number: NR-R10157 (96 wells) The kit is designed to qualitatively detect Bordetella Pertussis IgG in Human serum or plasma. FOR RESEARCH USE ONLY. NOT FOR
Human Bordetella Pertussis IgG ELISA kit Catalog number: NR-R10157 (96 wells) The kit is designed to qualitatively detect Bordetella Pertussis IgG in Human serum or plasma. FOR RESEARCH USE ONLY. NOT FOR
Immunoperoxidase Assay for Determination of C3 in Guinea Pig Sera Lot # 2F
 GUINEA PIG C3 ELISA Immunoperoxidase Assay for Determination of C3 in Guinea Pig Sera 40-374-130032 Lot # 2F Directions for Use For Research Use Only For In Vitro Use Only GUINEA PIG COMPLEMENT FACTOR
GUINEA PIG C3 ELISA Immunoperoxidase Assay for Determination of C3 in Guinea Pig Sera 40-374-130032 Lot # 2F Directions for Use For Research Use Only For In Vitro Use Only GUINEA PIG COMPLEMENT FACTOR
Mouse Coagulation Factor X (F10) ELISA
 Mouse Coagulation Factor X (F10) ELISA For the quantitative determination of mouse F10 in biological samples. Cat. No. KT-52133 For Research Use Only. Not for use in diagnostic procedures. Page 1 of 5
Mouse Coagulation Factor X (F10) ELISA For the quantitative determination of mouse F10 in biological samples. Cat. No. KT-52133 For Research Use Only. Not for use in diagnostic procedures. Page 1 of 5
RapidChek SELECT Salmonella Enteritidis Test System
 RapidChek SELECT Salmonella Enteritidis Test System Part #: 7000220, 7000221, 7000222, 7000228 AOAC Approved Protocols: This test kit s performance was reviewed by AOAC Research Institute and was found
RapidChek SELECT Salmonella Enteritidis Test System Part #: 7000220, 7000221, 7000222, 7000228 AOAC Approved Protocols: This test kit s performance was reviewed by AOAC Research Institute and was found
Sample collection & laboratory methods for identification bacteria. Lab. 2 A.T. Samira
 Sample collection & laboratory methods for identification bacteria Lab. 2 A.T. Samira Learning objectives At the end of the presentation, participants should understand the: Procedures, preparation, processing
Sample collection & laboratory methods for identification bacteria Lab. 2 A.T. Samira Learning objectives At the end of the presentation, participants should understand the: Procedures, preparation, processing
HP EASE. Qualitative assay for IgG antibodies to Helicobacter pylori Product code GD21 (96 tests)
 HP EASE Qualitative assay for IgG antibodies to Helicobacter pylori Product code GD21 (96 tests) For Research Use Only. Not For Use In Diagnostic Procedures. Updates from previous versions are non-critical
HP EASE Qualitative assay for IgG antibodies to Helicobacter pylori Product code GD21 (96 tests) For Research Use Only. Not For Use In Diagnostic Procedures. Updates from previous versions are non-critical
Human Herpes Simplex Virus Type 1 (HSV-1) IgM ELISA Kit
 Human Herpes Simplex Virus Type 1 Catalog No: IRAPKT1412 (HSV-1) IgM ELISA Kit Lot No: SAMPLE INTENDED USE The HSV-1 IgM ELISA is intended for the detection of IgM antibodies to herpes simplex virus HSV-1.
Human Herpes Simplex Virus Type 1 Catalog No: IRAPKT1412 (HSV-1) IgM ELISA Kit Lot No: SAMPLE INTENDED USE The HSV-1 IgM ELISA is intended for the detection of IgM antibodies to herpes simplex virus HSV-1.
Human Beta 2-Microglobulin ELISA Kit
 Human Beta 2-Microglobulin ELISA Kit CATALOG NO: IRKTAH1125 LOT NO: SAMPLE INTENDED USE The Beta 2-Microglobulin test kit is a highly sensitive two-site enzyme linked immunoassay (ELISA) for measuring
Human Beta 2-Microglobulin ELISA Kit CATALOG NO: IRKTAH1125 LOT NO: SAMPLE INTENDED USE The Beta 2-Microglobulin test kit is a highly sensitive two-site enzyme linked immunoassay (ELISA) for measuring
Pig Fibrinogen ELISA
 K-ASSAY Pig Fibrinogen ELISA For the quantitative determination of Fibrinogen in pig biological samples Cat. No. KT-484 For Research Use Only. 1 Rev. 9925484 K-ASSAY PRODUCT INFORMATION Pig Fibrinogen
K-ASSAY Pig Fibrinogen ELISA For the quantitative determination of Fibrinogen in pig biological samples Cat. No. KT-484 For Research Use Only. 1 Rev. 9925484 K-ASSAY PRODUCT INFORMATION Pig Fibrinogen
Manual. Catalog number:
 LLAMA IGG ELISA Quantitation Kit Manual Catalog number: 40-374-130033 Immunoperoxidase Assay for Determination of Llama IgG in Llama serum/plasma. This kit is for research use only, and is not for use
LLAMA IGG ELISA Quantitation Kit Manual Catalog number: 40-374-130033 Immunoperoxidase Assay for Determination of Llama IgG in Llama serum/plasma. This kit is for research use only, and is not for use
BIOSCAN* ATP method for monitoring of biofilm/sessile microorganisms in cooling systems
 Water Technologies & Solutions microbiological procedure MB013 BIOSCAN* ATP method for monitoring of biofilm/sessile microorganisms in cooling systems summary of method This method can be used to determine
Water Technologies & Solutions microbiological procedure MB013 BIOSCAN* ATP method for monitoring of biofilm/sessile microorganisms in cooling systems summary of method This method can be used to determine
Transport Medium for Storage and Transportation of Respiratory Swabs
 For Professional Use Only Transport Medium for Storage and Transportation of Respiratory Swabs Reagent for sampling, transportation, and storage of upper respiratory tract swabs Instruction Manual AmpliSens
For Professional Use Only Transport Medium for Storage and Transportation of Respiratory Swabs Reagent for sampling, transportation, and storage of upper respiratory tract swabs Instruction Manual AmpliSens
Human anti-myocardial antibody (IgM) ELISA Kit
 Human anti-myocardial antibody (IgM) ELISA Kit Catalog Number. MBS704679 For the qualitative determination of human Anti-myocardial antibody (IgM) concentrations in serum. This package insert must be read
Human anti-myocardial antibody (IgM) ELISA Kit Catalog Number. MBS704679 For the qualitative determination of human Anti-myocardial antibody (IgM) concentrations in serum. This package insert must be read
INSTRUCTIONS FOR USE LYFO DISK KWIK-STIK KWIK-STIK PLUS. n LYFO DISK Microorganisms n KWIK-STIK Microorganisms n KWIK-STIK Plus Microorganisms
 INSTRUCTIONS FOR USE n LYFO DISK Microorganisms n KWIK-STIK Microorganisms n KWIK-STIK Plus Microorganisms LYFO DISK INTENDED USE LYFO DISK, KWIK-STIK and KWIK-STIK Plus Microorganisms are lyophilized,
INSTRUCTIONS FOR USE n LYFO DISK Microorganisms n KWIK-STIK Microorganisms n KWIK-STIK Plus Microorganisms LYFO DISK INTENDED USE LYFO DISK, KWIK-STIK and KWIK-STIK Plus Microorganisms are lyophilized,
Please consult the Product Package Insert before running the test
 Please consult the Product Package Insert before running the test Sample Collection, Preparation, Storage and Volume Q. What type of blood sample should be used with the PIFA Heparin/PF4 Rapid Assay? A.
Please consult the Product Package Insert before running the test Sample Collection, Preparation, Storage and Volume Q. What type of blood sample should be used with the PIFA Heparin/PF4 Rapid Assay? A.
PictArray ToRCH ELISA Kit
 TORCH Instructions PictArray ToRCH ELISA Kit Instructions for testing human serum samples on PictArray ToRCH Panel consisting of Toxoplasma gondii, Rubella, Cytomegalovirus (CMV), Herpes Simplex Virus
TORCH Instructions PictArray ToRCH ELISA Kit Instructions for testing human serum samples on PictArray ToRCH Panel consisting of Toxoplasma gondii, Rubella, Cytomegalovirus (CMV), Herpes Simplex Virus
Human IL-1 alpha ELISA Kit
 Human IL-1 alpha ELISA Kit Catalog No: CDK025B Quantity: 2 x 96 tests SPECIFICITY : RANGE : SENSITIVITY : INCUBATION : SAMPLE TYPES : Recognizes both natural and recombinant human IL-1 alpha 31.2 pg /
Human IL-1 alpha ELISA Kit Catalog No: CDK025B Quantity: 2 x 96 tests SPECIFICITY : RANGE : SENSITIVITY : INCUBATION : SAMPLE TYPES : Recognizes both natural and recombinant human IL-1 alpha 31.2 pg /
Mouse Fibrinogen ELISA
 K-ASSAY Mouse Fibrinogen ELISA For the quantitative determination of fibrinogen in mouse biological samples Cat. No. KT-397 For research use only. 1 Rev. 9994397 K-ASSAY PRODUCT INFORMATION Mouse Fibrinogen
K-ASSAY Mouse Fibrinogen ELISA For the quantitative determination of fibrinogen in mouse biological samples Cat. No. KT-397 For research use only. 1 Rev. 9994397 K-ASSAY PRODUCT INFORMATION Mouse Fibrinogen
Fibrinogen ELISA. For the quantitative determination of fibrinogen in biological fluids, serum, and plasma.
 Fibrinogen ELISA For the quantitative determination of fibrinogen in biological fluids, serum, and plasma. Please read carefully due to Critical Changes, e.g., Changes to preparation of standard volume
Fibrinogen ELISA For the quantitative determination of fibrinogen in biological fluids, serum, and plasma. Please read carefully due to Critical Changes, e.g., Changes to preparation of standard volume
DRG Toxoplasma gondii IgM. Revised 29 Jan rm (Vers. 12.1) RUO in the USA
 This kit is intended for Research Use Only. This kit is not intended for diagnostic procedures. Introduction The DRG Toxoplasma gondii IgM Enzyme Immunoassay Kit provides materials for determination of
This kit is intended for Research Use Only. This kit is not intended for diagnostic procedures. Introduction The DRG Toxoplasma gondii IgM Enzyme Immunoassay Kit provides materials for determination of
Mouse Sclerostin ELISA
 Mouse Sclerostin ELISA For the quantitative determination of sclerostin in mouse serum and plasma. For Research use Only. Not For Use In Diagnostic Procedures. Catalog Number: Size: 41-SCLMS-E01 96 Wells
Mouse Sclerostin ELISA For the quantitative determination of sclerostin in mouse serum and plasma. For Research use Only. Not For Use In Diagnostic Procedures. Catalog Number: Size: 41-SCLMS-E01 96 Wells
GI DNA Extraction Kit
 GI DNA Extraction Kit REF: 655-01; 48 extractions (32 wells plate) 656-01; 48 extractions (96 wells plate) Store at 15 C-35 C For use with the Nextractor Instrument For Professional Use Only Savyon Diagnostics
GI DNA Extraction Kit REF: 655-01; 48 extractions (32 wells plate) 656-01; 48 extractions (96 wells plate) Store at 15 C-35 C For use with the Nextractor Instrument For Professional Use Only Savyon Diagnostics
Human IgG ELISA Kit. Strip well format. Reagents for up to 96 tests
 Human IgG ELISA Kit Strip well format. Reagents for up to 96 tests Catalog No. CS222A Quantity: 1 x 96 tests CS222B 5 x 96 tests Intended Use: Background: Assay Principle: This human immunoglobulin G antigen
Human IgG ELISA Kit Strip well format. Reagents for up to 96 tests Catalog No. CS222A Quantity: 1 x 96 tests CS222B 5 x 96 tests Intended Use: Background: Assay Principle: This human immunoglobulin G antigen
Anti- Tissue Transglutaminase TTG IgG ELISA Kit
 Anti- Tissue Transglutaminase TTG IgG ELISA Kit Catalog number: NR-R10142 (96 wells) The kit is designed to quantitatively detect TTG IgG in Human serum or plasma. FOR RESEARCH USE ONLY. NOT FOR DIAGNOSTIC
Anti- Tissue Transglutaminase TTG IgG ELISA Kit Catalog number: NR-R10142 (96 wells) The kit is designed to quantitatively detect TTG IgG in Human serum or plasma. FOR RESEARCH USE ONLY. NOT FOR DIAGNOSTIC
Sheep IgG ELISA Kit. Innovative Research, Inc. Catalog No: ISHIGGKT. Lot No: SAMPLE
 Catalog No: ISHIGGKT Sheep IgG ELISA Kit Lot No: SAMPLE INTENDED USE The total Sheep IgG test kits are a highly sensitive two-site enzyme linked immunoassay (ELISA) for measuring IgG in sheep biological
Catalog No: ISHIGGKT Sheep IgG ELISA Kit Lot No: SAMPLE INTENDED USE The total Sheep IgG test kits are a highly sensitive two-site enzyme linked immunoassay (ELISA) for measuring IgG in sheep biological
B2M (Human) ELISA Kit
 B2M (Human) ELISA Kit Catalog Number KA0222 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
B2M (Human) ELISA Kit Catalog Number KA0222 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
MSES consultants, inc.
 USER INSTRUCTIONS CHEMICAL TESTING SPECIAL Before using this Chemical Kit READ ALL INSTRUCTIONS. This kit only contains NOTE: enough materials to test for EITHER SOLIDS OR FILM/SLIME - NOT BOTH. CHEMICAL
USER INSTRUCTIONS CHEMICAL TESTING SPECIAL Before using this Chemical Kit READ ALL INSTRUCTIONS. This kit only contains NOTE: enough materials to test for EITHER SOLIDS OR FILM/SLIME - NOT BOTH. CHEMICAL
Acetyl-p53 (K381) Cell-Based Colorimetric ELISA Kit
 Acetyl-p53 (K381) Cell-Based Colorimetric ELISA Kit Catalog No. KA8015C Detection and Quantification of Acetyl-p53 (K381) Protein Concentration in Cell. Research Purposes Only. Not Intended for Diagnostic
Acetyl-p53 (K381) Cell-Based Colorimetric ELISA Kit Catalog No. KA8015C Detection and Quantification of Acetyl-p53 (K381) Protein Concentration in Cell. Research Purposes Only. Not Intended for Diagnostic
Liste des Symboles. Attention, voir notice d utilisation Tests par coffret Fabricant. Pour diagnostic in vitro uniquement Péremption Usage unique
 Liste des Symboles Attention, voir notice d utilisation Tests par coffret Fabricant Pour diagnostic in vitro uniquement Péremption Usage unique Conserver entre 2-30 C No. de lot REF Code produit Index
Liste des Symboles Attention, voir notice d utilisation Tests par coffret Fabricant Pour diagnostic in vitro uniquement Péremption Usage unique Conserver entre 2-30 C No. de lot REF Code produit Index
Clostridium difficile GDH ELISA
 Clostridium difficile GDH ELISA For the qualitative determination of C. difficile glutamate dehydrogenase in human stool. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: Size:
Clostridium difficile GDH ELISA For the qualitative determination of C. difficile glutamate dehydrogenase in human stool. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: Size:
i-stat iccnet CHSA Edition 1.2 Page 1 of 7 i-stat Rosy Tirimacco / Paul Simpson
 Page 1 of 7 i-stat Edition No: 1.2 Author: Rosy Tirimacco / Paul Simpson Date: January, 2012 Operative Date: January, 2012 Review Date: January, 2013 Location: Country Health SA Integrated Cardiovascular
Page 1 of 7 i-stat Edition No: 1.2 Author: Rosy Tirimacco / Paul Simpson Date: January, 2012 Operative Date: January, 2012 Review Date: January, 2013 Location: Country Health SA Integrated Cardiovascular
Contamination Prevention and Decontamination
 Contamination Prevention and Decontamination Introduction FilmArray is an automated in vitro diagnostic (IVD) system that utilizes nested multiplex PCR (nmpcr) and high-resolution melting analysis to detect
Contamination Prevention and Decontamination Introduction FilmArray is an automated in vitro diagnostic (IVD) system that utilizes nested multiplex PCR (nmpcr) and high-resolution melting analysis to detect
Human titin antibody(igg) ELISA Kit
 Human titin antibody(igg) ELISA Kit Catalog Number. CSB-E13356h For the qualitative determination of human titin antibody(igg) concentrations in serum. This package insert must be read in its entirety
Human titin antibody(igg) ELISA Kit Catalog Number. CSB-E13356h For the qualitative determination of human titin antibody(igg) concentrations in serum. This package insert must be read in its entirety
Dog Fibrinogen ELISA
 K-ASSAY Dog Fibrinogen ELISA For the quantitative determination of Fibrinogen in dog biological samples Cat. No. KT-457 For Research Use Only. 1 Rev. 010209 K-ASSAY PRODUCT INFORMATION Dog Fibrinogen ELISA
K-ASSAY Dog Fibrinogen ELISA For the quantitative determination of Fibrinogen in dog biological samples Cat. No. KT-457 For Research Use Only. 1 Rev. 010209 K-ASSAY PRODUCT INFORMATION Dog Fibrinogen ELISA
Human clonorchis sinensis (CS) antibody (IgG) ELISA kit
 Human clonorchis sinensis (CS) antibody (IgG) ELISA kit Catalog Number. CSB-EQ027680HU For the qualitative determination of human clonorchis sinensis (CS) antibody (IgG) concentrations in serum. This package
Human clonorchis sinensis (CS) antibody (IgG) ELISA kit Catalog Number. CSB-EQ027680HU For the qualitative determination of human clonorchis sinensis (CS) antibody (IgG) concentrations in serum. This package
DRG Anti-Tissue Transglutaminase
 Please use only the valid version of the package insert provided with the kit. This kit is intended for Research Use Only. Not for use in diagnostic procedures. NAME AND INTENDED USE Anti-Tissue-Transglutaminase
Please use only the valid version of the package insert provided with the kit. This kit is intended for Research Use Only. Not for use in diagnostic procedures. NAME AND INTENDED USE Anti-Tissue-Transglutaminase
Sheep oxytocin (OT) ELISA Kit
 Sheep oxytocin (OT) ELISA Kit Catalog Number. CSB-E13236Sh For the quantitative determination of sheep oxytocin (OT) concentrations in serum, plasma. This package insert must be read in its entirety before
Sheep oxytocin (OT) ELISA Kit Catalog Number. CSB-E13236Sh For the quantitative determination of sheep oxytocin (OT) concentrations in serum, plasma. This package insert must be read in its entirety before
For the quantitative determination of Complement Factor 3 in guinea pig serum and plasma
 For the quantitative determination of Complement Factor 3 in guinea pig serum and plasma For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 41-CO3GP-E01 Size: 96 wells Version:
For the quantitative determination of Complement Factor 3 in guinea pig serum and plasma For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 41-CO3GP-E01 Size: 96 wells Version:
BD GeneOhm MRSA ACP Lysis Kit
 REF 441638 100 Tests DOPS09-09-V1E1 USA Date: 2010-01 TABLE OF CONTENTS INTENDED USE...3 SUMMARY AND EXPLANATION OF THE PROCEDURE...3 REAGENTS...3 PRECAUTIONS...3 MATERIALS PROVIDED...4 STORAGE, HANDLING
REF 441638 100 Tests DOPS09-09-V1E1 USA Date: 2010-01 TABLE OF CONTENTS INTENDED USE...3 SUMMARY AND EXPLANATION OF THE PROCEDURE...3 REAGENTS...3 PRECAUTIONS...3 MATERIALS PROVIDED...4 STORAGE, HANDLING
Procine CRP ELISA Kit
 Procine CRP ELISA Kit For the quantitative in vitro determination of Procine C-Reactive Protein concentrations in serum - plasma - celiac fluid - tissue homogenate - body fluid FOR LABORATORY RESEARCH
Procine CRP ELISA Kit For the quantitative in vitro determination of Procine C-Reactive Protein concentrations in serum - plasma - celiac fluid - tissue homogenate - body fluid FOR LABORATORY RESEARCH
Guinea pig PGI2 ELISA Kit
 Guinea pig PGI2 ELISA Kit For the quantitative in vitro determination of Guinea pig PGI2 concentrations in serum - plasma - celiac fluid - tissue homogenate - body fluid FOR LABORATORY RESEARCH USE ONLY.
Guinea pig PGI2 ELISA Kit For the quantitative in vitro determination of Guinea pig PGI2 concentrations in serum - plasma - celiac fluid - tissue homogenate - body fluid FOR LABORATORY RESEARCH USE ONLY.
Armenian Hamster IgG Antigen ELISA Kit
 Armenian Hamster IgG Antigen ELISA Kit Catalog No: IAHTIGGKT Lot No: SAMPLE INTENDED USE This Armenian hamster Immunoglobulin G (IgG) antigen assay is intended for the quantitative determination of total
Armenian Hamster IgG Antigen ELISA Kit Catalog No: IAHTIGGKT Lot No: SAMPLE INTENDED USE This Armenian hamster Immunoglobulin G (IgG) antigen assay is intended for the quantitative determination of total
