Seventh International Water Technology Conference Egypt 1-3 April
|
|
|
- Ralf Reeves
- 6 years ago
- Views:
Transcription
1 Seventh International Water Technology Conference Egypt 1-3 April RECVERY F EAVY METALS FRM WASTES WATERS BY SLVENT EXTRACTIN USING TE PSPRIC ACID (D2EPA) N. BELKUCE and M. A. DIDI Laboratory of Catalysis and synthesis in organic chemistry, Department of Chemistry, Faculty of Science University of Tlemcen - BP 119 ALGERIA. nbelkhouche@yahoo.fr ABSTRACT The solvent extraction of metallic cations by di(2-ethylhexyl)phosphoric acid (D2EPA), diluted in n-heptan, is discussed in a way of the extraction efficiency. The solvent extraction was carried out starting from an acetate aqueous solution. By study of the acetate aqueous phase containing metal to be extracted (volumetric analysis) and while varying the following conditions: Q, [(D2EPA) 2 ], volumes ratio aqueous to organic (V w /V org ), we could : - To optimize the extraction yield of each metal ion taken separately, - To establish a equilibrium mechanism of extraction for each metal, - To establish an order of selectivity, during the extraction starting from synthetic mixture of the metallic cations and show the synergy effect, during the extraction with extractants mixture (D2EPA + TBP). Keywords: Copper Nickel Separation, Solvent Extraction, D2EPA, Synergy Effect INTRDUCTIN The phosphorus chemistry offers broad fields of scientific and technological applications. The organophosphorus compounds are excellent metal ions extractants because are chelating agents; this is why they are frequently employed in the solvent extraction process. In particular, they are used for the purification of the liquid effluents who can contain heavy metals [1,2]. Among the commercial phosphoric acid extractants, di(2-ethylhexyl)phosphoric acid (abbreviated as D2EPA or simply R) has been extensively studied as an extractant reagent in hydrometallurgical processes for the separation and purification of a number of metals. It can extract heavy metals such as copper, cobalt, nickel and zinc [2,3]. Di(2-ethylhexyl)phosphoric acid continues to be studied in the solvent extraction process, for the separation and the purification of the metal liquid effluents [3,4]. The solutions of D2EPA in organic solvents can be charged strongly by the divalent metal ions, without the formation of a third phase and viscosity becomes inaccessible when the moles number ratio of ligand /metal = 2 (Q) is held in account.
2 918 Seventh International Water Technology Conference Egypt 1-3 April 2003 Many studies treat solvent extraction of nickel and copper in aqueous mediums such as sulphate, chloride and nitrate but our work was based on the choice of an acetate medium. EXPERIMENTAL 1. Realization of the extractions Each experimental point was obtained by magnetic shaking of extract aqueous phase (10 cm 3 ) and extracting organic phase (5 cm 3 ) in funnels separating, followed by a separation. The shaking time, in each case, determined from experiments and was chosen largely higher than time necessary to reach the extraction equilibrium. In all the cases, thirty minutes of stirring appeared sufficient. The variations of volume were supposed to be negligible and all experiments were carried out at ambient temperature (20 C). 2. Reagents and produced The aqueous solutions were prepared with distilled water and the following reagents were used: copper acetate (II), nickel acetate (II), murexide (C 8 6 N 5 5 ) and 1-[pyridyl-(2)-azo]-2-naphtol (C N 3 ) (PAN) provided by MERK, ethylen diamine tetraacetic bisodium acid (C N 2 Na ), (EDTA) provided by FLUKA. The organic phase is made up: The n-heptan diluent and di(2-ethylhexyl)phosphoric acid (D2EPA) extractant provided by FLUKA of which the proportion in D2EPA / M2EPA is 60 / 40. % 3. Analytical methods During this work, we were brought for determine the copper and nickel concentrations in the aqueous solutions. For this reason, the following analytical methods were used: - p-metric analysis of D2EPA, - Volumetric analysis of Cu (II) and Ni (II). RESULTS AND DISCUSSIN The D2EPA being a mixture of mono and di- phosphoric acids, we takes 10 ml of D2EPA diluted in the n-heptan (10-3 M) to which we adds 20 ml of distilled water, follow-up of a strong stirring and phases separation, in order to check if the monophosphoric acid remained or not in organic phase. We analyze a well defined volume of the organic phase (after separation of the two phases) by Na 5 x 10-2 N. Figure 1 shows only one turn, which confirms that all the M2EPA passes in the aqueous phase and only the D2EPAensures the extraction of the metal ions.
3 Seventh International Water Technology Conference Egypt 1-3 April E(m volts) Volume of Na (ml) Fig. 1: Potentiometry curve of D2EPA I. EXTRACTIN F CPPER BY TE D2EPA The concentration of the cupric ions after extraction was determined by volumetric analysis in the presence of indicator "PAN" (twice to recristallize in ethanol to 96 %, p 5). The results are presented in Fig. 2, which shows an extraction yield maximum of 94 %. 94 Extraction efficiency (%) [(D2EPA) 2 ] x 10 3 F Fig. 2: Effect of D2EPA concentration on extraction efficiency of copper, [Cu 2+ ] i = 10-3 M, p= 6.17
4 920 Seventh International Water Technology Conference Egypt 1-3 April 2003 The extraction of copper (II) by D2EPA, diluted in n-heptan can be expressed by the general equation (1) [5-7]. M w n+ + (x+n)/2 ( 2 R 2 ) M R n.xr + n w + (1) The corresponding equilibrium constant is: K x = [(MR n.xr)][ + ] n [M +n ] -1 [( 2 R 2 )] -(n+x)/2 (2) The distribution ration of copper is given by: E = C M,org /C M,w = [(MR n xr)]/c M,w (3) E = K x [( 2 R 2 )] (n+x)/2 [ + ] -n α (M) -1 (4) α (M) : is the coefficient, which takes account of the secondary reactions due to the hydrolysis and the formation of metal complexes in the aqueous phase [8]. R = (C 8 17 ) 2 P() - M n+ w = metal ion extracted of load n + in aqueous phase 2 R 2 = dimerous form of D2EPA in organic phase R = monomeric form of D2EPA From equation (4), distribution ratio depends on the p and the extractant concentration. The logarithmic plots of the distribution ration according to each variable of the equation (4) by maintaining the others constant, give an estimate of the stoechiometries values x and n. A plot of log E vs. log [ + ] gives a straight line with a slope of 0.83 i.e., n = 1. The experimental results are shown in Fig. 3. Figure 4 shows a straight line between log E and log [ 2 R 2 ] with a slope of 0.96 i.e., x = 1. 1,4 Log E 1,3 1,2 slope = 0.83 R 2 = ,9-1,8-1,7-1,6-1,5 Log [ + ] Fig. 3: Plots of log E vs. log [ + ] at extraction equilibrium of copper, [Cu 2+ ] i = 10-3 M.
5 Seventh International Water Technology Conference Egypt 1-3 April ,4 Log E 1,3 1,2 slope = 0.96 R 2 = ,6-2,5-2,4-2,3 Log [(D2EPA) 2 ] Fig. 4: Influence of D2EPA concentration on distribution ratio of copper, [Cu 2+ ] i = 10-3 M. The values obtained (x and n) shows that the extraction equilibrium of Cu +2 in acetate medium by the D2EPA, diluted in n-heptan obeys at following equation: CuC 3 C + w + ( 2 R 2 ) CuC 3 CR.R + w + The suggested structure of the formed complex, in the organic phase during the extraction, is given by Fig.5. (5) R R R P P R Cu C C 3 Fig. 5: Schematic representation of Cu-D2EPA complex
6 922 Seventh International Water Technology Conference Egypt 1-3 April Extraction efficiency (%) [(D2EPA) 2 ] x 10 3 F Fig. 6: Influence of D2EPA concentration on extraction efficiency of nickel, [Ni +2 ] = 10-3 M owever, it should be announced that the hydrolysis product of copper (II) is the dimerous species (Cu 2 () 2 ) 2+ [9,10]. A series of monomolecular species: Cu() +, Cu() 2, Cu() 3 +, can also be formed but are formed in considerable proportions only in the solutions very diluted between p 7 and 12. II. EXTRACTIN F NICKEL BY D2EPA The extraction of nickel (II) by the D2EPA, diluted in the n-heptan, is carried out in neutral medium with a volumes ratio (V w /V org = 2). It takes place in an engine stirred perfectly until equilibrium reached (t = 30 min). ne separates the two phases using a funnel of separation. A volume of 4 ml taken of the aqueous phase is titrated. The results are represented in Fig. 6. The choice of n-heptan is that it presents a synergy effect toward the metal, because according to the principle of Taube the molecules of the thinner do not block extractant. Then the D2EPA preserves its extractive character. By observing Fig. 6, one notices that the extraction yield of nickel increases with the concentration of extractant dimerous until becoming constant by reaching the maximum value of 80 %. The stoechiometries coefficients of the extraction equilibrium equation of nickel (II) by the D2EPA, in neutral medium are deduced from the slopes of logarithmic plots. ne distinguishes that the nickel is chelated by the half of dimer. Firstly: when extractant is in its monomeric form, equilibrium is written:
7 Seventh International Water Technology Conference Egypt 1-3 April NiC 3 C w (R) NiC 3 C.( 2 ) 2 R + w + (6) The structure of complex is represented as follows (Fig. 7): R R C 3 P C Ni C C 3 R R 2 P Ni 2 P R R Fig. 7: Schematic representations of Ni-D2EPA complexes Secondly: when extractant is in its dimeric form, equilibrium is written: NiC 3 C w ½ ( 2 R ) NiC 3 C.( 2 ) 4.R + w + (7) Dimer 2 R 2 is related to two water molecules because of the great solubility of water in the D2EPA (0.051 mol.l -1 at 20 C). Structure of complex is represented by Fig. 7. III. SELECTIVE EXTRACTIN F CPPER BY D2EPA III. 1. Calibration curves The calibration curves of nickel, copper and their mixture were carried out starting from the aqueous acetate solutions. For each solution one takes a volume of 2.5 ml, titrated by the EDTA (5 x 10-3 M) in the presence of the indicator meruxide. The results are deferred in the Fig 8.
8 924 Seventh International Water Technology Conference Egypt 1-3 April 2003! "!! " #!$ " Fig. 8: Calibration curves of Cu +2, Ni +2 and their mixture, p = 6 From Fig. 8, one notices that beyond the concentration 1.5 x 10-3 M in copper and nickel, the titrated volume of the two mixed cations is slightly higher than the sum of titrated volume of each taken cation separately (relative error equalizes to 5%). III. 2. Selective extraction of copper in neutral medium The extractions were carried out starting from an initial aqueous solution 10-3 M in copper and nickel. The concentrations of extractant are selected in such way that the difference between the extraction yield of each cation taken separately is highest (maximum for copper and minimum for nickel). The others conditions of extraction like the equilibrium time = 30 min, V w /Vorg = 2 and T = 20 C, were maintained. From aqueous phase recovered after extraction, a volume equal to 2.5 ml is titrated by the EDTA (5 x 10-3 M) in the presence of meruxide. The results are gathered in Figs. (9-12) and Table (1). Table. 1: Influence of extractant concentration on ydrogen ion concentration at extraction equilibrium of metal Cu (II) Ni (II) [(D2EPA) 2 ] x 10 3 F P p p * where p * = p(cu) - p(ni)
9 Seventh International Water Technology Conference Egypt 1-3 April copper Nickel mixture (copper and nickel) Extraction efficiency (%) [(D2EPA)2] x 10(3) F Fig. 9: Influence of D2EPA concentration on extraction efficiency of copper, nickel and their mixture, p = Extraction efficiency (%) copper nickel mixture (copper and nickel) p Fig. 10: Variation of extraction efficiency of copper, nickel and their mixture with p of equilibrium
10 926 Seventh International Water Technology Conference Egypt 1-3 April 2003 At a concentration of D2EPA equal to 4.8 x 10-3 F, we obtain under the same experimental conditions the same p of equilibrium for both cations (Table 1). The extraction yields of copper and nickel which results from this, show a good extrability of copper (93.5 %) compared to nickel (83%) (Fig. 10). In the order, we have the extrability of Cu (II) is higher than Ni (II). This result was obtained by Yukio Nagaosa and al [11] in sulphated medium. For the extraction of the mixture (Cu +2, Ni +2 ), the yield decreases with the increase in the extractant concentration (Fig. 9) copper nickel mixture 4.00 p [(D2EPA)2] x 10(3) F Fig. 11: Variation of p at the extraction equilibrium of copper, nickel and their mixture with the extractant concentration Fig. 12: p of separation for both metal-ions at one stage
11 Seventh International Water Technology Conference Egypt 1-3 April From Table (1) and Figure 11, the values of p of extraction equilibrium for the metal ions decrease with the increase in the extractant concentration. In the range 0.6 x 10-3 F at 8 x 10-3 F, the values of p change as follows: Cu (II) (range of p = 2.35) > Ni (II) (range of p = 0.31). The value of p * indicates that the effectiveness of copper separation decreases when the extractant concentration increases. The separation process for both ions is discussed in a way of distribution ratio for each one of these two metals, under the conditions identical to those of their mixture, by: SF = E Cu+2 / E Ni+2 where E Cu+2 : Distribution ratio of copper extraction E Ni+2 : Distribution ratio of nickel extraction This method is valid only when two metals have the same oxidation step. The two plots log E vs. p of equilibrium have slopes equal to 1 for Cu 2+ and Ni 2+ respectively. We can separate both ions in an extraction at only stage by determining of p s (p of extraction at only stage), with log E = +2 for copper and log E = -2 for nickel. The separation minimum of these two plots should be equal to 4/n. As n=1 (number of protons exchanged during the copper or nickel extraction) then there is 4/n = 4 from where one finds p S = 5.71 (Fig. 12). At this p, the D2EPA concentration must be lower than 0.5 x 10-3 F. III. 3. Effect of extractants mixture on the selective extraction of copper The mixture of two extractants, one cationnic (D2EPA) and the other neutral (TBP), produces one of the two effects: synergy effect or antagonism effect. Effect of the TBP (tri-butylphosphate) is discussed of its action capacity on the copper selective extraction. The extractions have been carried out on the mixture (Ni 2+, Cu 2+ ) in neutral medium (p = 6.24). Concentration of TBP was constant and equal at x 10-3 M throughout our experiments, only the concentration of D2EPA varies. Mixture was prepared in the n-heptan. The experimental conditions are maintained, identical to those of the preceding experiments (T= 20 C, V w /V org = 2, stirring time = 30 min, [Ni 2+ ] i =[Cu 2+ ] i = 10-3 M, [(D2EPA) 2 ]= 0.6 x 10-3 F at 7.2 x 10-3 F). Extraction results of Cu 2+ and Ni 2+ by the D2EPA in the presence of TBP are presented in Fig. 13.
12 928 Seventh International Water Technology Conference Egypt 1-3 April Extraction efficiency (%) [(D2EPA)2] x 10(3) F Fig.13: Influence of concentration of extractants mixture on selective extraction efficiency of copper Result obtained shows that when the percentage of TBP in the extractants mixture is lower than 0.76%, we have an increase in the extrability due to the solvatant character of the TBP. While, when the percentage of TBP is higher than 0.76%, we have a decrease in the extrability due to the polar character of the TBP and chemical reaction between D2EPA and TBP which lowers the free D2EPA concentration. CNCLUSIN The solvent extraction of various metal ions (copper and nickel), starting from the aqueous solutions containing the organic ion (C 3 C - ) using an organophosphorus acid (D2EPA) diluted in n-heptan, gave us a maximum yield of 94 % for copper and 80 % for nickel. ur experimental study enabled us to determine the extrability order from where we found that the extrability rate of copper (93.5%) is higher than nickel (83%). These last results were given when the metal ions are together. It is necessary to use an optimal concentration of D2EPA lower than 0.5 x 10-3 F to extract copper instead of nickel. Moreover, synergy effect was observed with 0.76 % of the TBP in the extractants mixture (D2EPA + TBP). In this present work, we used simple means and a traditional method which is the solvent extraction. The organophosphorus acid agents showed their effectiveness in the depollution of contaminated waters by heavy metals. Two successive extractions are sufficient to purify an industrial or urban liquid wastes containing copper, nickel or both at the same time.
13 Seventh International Water Technology Conference Egypt 1-3 April REFERENCES [1] Y. Chengye and CE. Deliang., Synthesis (stut; ISSN , coden SYNTBF, No. 6; DA pp (1992). [2] R. S. Juang and Y. T. Chang, Ind. Eng. Chem, 32. Res. pp (1993). [3] L. F. Cook and V. V. Szmokaluk, In Proc. Int. Solvent Extr. Conf., The ague April, 1971 (Edited by J. G. Gregory, B. Evans and P. C. Weston) p Soc. Chem. Ind, London (1971). [4] M. L. Brisk and W. J. McManamey, J. Appl. Chem. 19, 103 (1969). [5] Y. rashima and J. Shirokawa, Proceeding of the 12 th Rare Earth Research Conf., 1, , 1976; Inis-Atomindex, 9(6), , (1978). [6] D. Dyrssen, Acta chem. Scand, 11, p (1957). [7] Vogel, Quantitative Inorg. Analy, Chap IV.9; p 432. [8] J.. Forserberg and al., andbook of Inorg. Chem. Vol. D6, Ed. Springer- Verlag, Berlin, p. 88 (1983). [9] C. F. Baes, J. Inorg. Nucl. Chem, Vol. 24, pp (1962). [10] I. Kojima, J. Fukuta and M. Tanaka, J. Inorg. Nucl. Chem., Vol. 31, pp (1969). [11] Y. Nagaosa and al., J.Chem. Eng. Japan, Vol. 24; No. 1, pp (1991).
Estimation of Iron in Pharmaceutical samples-a Solvent extraction study
 International Journal of ChemTech Research CODEN( USA): IJCRGG ISSN : 0974-4290 Vol. 3, No.2, pp 703-707, April-June 2011 Estimation of Iron in Pharmaceutical samples-a Solvent extraction study A.V.L.N.S.H.Hariharan*,
International Journal of ChemTech Research CODEN( USA): IJCRGG ISSN : 0974-4290 Vol. 3, No.2, pp 703-707, April-June 2011 Estimation of Iron in Pharmaceutical samples-a Solvent extraction study A.V.L.N.S.H.Hariharan*,
COMPLEX FORMATION TITRATION
 1 COMPLEX FORMATION TITRATION Complexometry Volumetric method involves reaction of metal with ligand to form complex M + Electron acceptor Coordinate bond Ligand Electron donor Complex formation is a type
1 COMPLEX FORMATION TITRATION Complexometry Volumetric method involves reaction of metal with ligand to form complex M + Electron acceptor Coordinate bond Ligand Electron donor Complex formation is a type
Recovery of Copper, Nickel and Zinc from Sulfate Solutions by Solvent Extraction Using LIX 984N
 http://www.e-journals.net ISSN: 0973-4945; CODEN ECJHAO E- Chemistry 2011, 8(S1), S434-S438 Recovery of Copper, Nickel and Zinc from Sulfate Solutions by Solvent Extraction Using LIX 984N VAISHNAVI SRIDHAR
http://www.e-journals.net ISSN: 0973-4945; CODEN ECJHAO E- Chemistry 2011, 8(S1), S434-S438 Recovery of Copper, Nickel and Zinc from Sulfate Solutions by Solvent Extraction Using LIX 984N VAISHNAVI SRIDHAR
Effects of Gamma Radiation on Iron (III) Complex with Sodium Salicylate in Aqueous Solutions
 Effects of Gamma Radiation on Iron (III) Complex with Sodium Salicylate in Aqueous Solutions M.F. Barakat (1), M.El-Banna (2) (1) Nuclear and Radiological Regularity Authority, (2) National Center of Radiation
Effects of Gamma Radiation on Iron (III) Complex with Sodium Salicylate in Aqueous Solutions M.F. Barakat (1), M.El-Banna (2) (1) Nuclear and Radiological Regularity Authority, (2) National Center of Radiation
Solvent Extraction of Copper(I) and (II) as Thiocyanate
 ANALYTICAL SCIENCES DECEMBER 1997, VOL. 13 903 Solvent Extraction of Copper(I) and (II) as Thiocyanate Complexes with Tetrabutylammonium Ions into Chloroform and with Trioctylphosphine Oxide into Hexane
ANALYTICAL SCIENCES DECEMBER 1997, VOL. 13 903 Solvent Extraction of Copper(I) and (II) as Thiocyanate Complexes with Tetrabutylammonium Ions into Chloroform and with Trioctylphosphine Oxide into Hexane
Conductometric Studies on Terbium Soaps
 Portugaliae Electrochimica Acta 2012, 30(1), 45-51 OI: 10.4152/pea.201201045 PORTUGALIAE ELECTROCHIMICA ACTA ISSN 1647-1571 Conductometric Studies on Terbium Soaps Kamal Kishore * and S.K Upadhyaya P.G.
Portugaliae Electrochimica Acta 2012, 30(1), 45-51 OI: 10.4152/pea.201201045 PORTUGALIAE ELECTROCHIMICA ACTA ISSN 1647-1571 Conductometric Studies on Terbium Soaps Kamal Kishore * and S.K Upadhyaya P.G.
Experiment 18 Determination of Iron by Visible Spectrophotometry
 Experiment 18 Determination of Iron by Visible Spectrophotometry PRELAB DISCUSSION Visible spectrophotometry (spectral analysis) is a method of measuring the concentration of colored solutions by the amount
Experiment 18 Determination of Iron by Visible Spectrophotometry PRELAB DISCUSSION Visible spectrophotometry (spectral analysis) is a method of measuring the concentration of colored solutions by the amount
ISEC The 21st International Solvent Extraction Conference
 Selective Extraction of Scandium from ther REEs Using Binary Extractant of PC-88A and Versatic 10 from Nitrate Media Maha Sharaf 1, Wataru Yoshida 1, Fukiko Kubota 1 1, 2* and Masahiro Goto 1 Department
Selective Extraction of Scandium from ther REEs Using Binary Extractant of PC-88A and Versatic 10 from Nitrate Media Maha Sharaf 1, Wataru Yoshida 1, Fukiko Kubota 1 1, 2* and Masahiro Goto 1 Department
SOLVENT EXTRACTION TEST WORK TO EVALUATE A VERSATIC 10/NICKSYN SYNERGISTIC SYSTEM FOR NICKEL-CALCIUM SEPARATION
 SOLVENT EXTRACTION TEST WORK TO EVALUATE A VERSATIC 1/NICKSYN SYNERGISTIC SYSTEM FOR NICKEL-CALCIUM SEPARATION René du Preez a, Marthie Kotze b, Gerhard Nel c, Simon Donegan d and Huziel Masiiwa d a Mintek,
SOLVENT EXTRACTION TEST WORK TO EVALUATE A VERSATIC 1/NICKSYN SYNERGISTIC SYSTEM FOR NICKEL-CALCIUM SEPARATION René du Preez a, Marthie Kotze b, Gerhard Nel c, Simon Donegan d and Huziel Masiiwa d a Mintek,
SOLVENT EXTRACTION AND SEPARATION OF COPPER AND ZINC FROM A PICKLING SOLUTION
 15th International Conference on Non-ferrous metals, Hotel Oberoi Grand, Kolkata, July 8-9,2011 Eds: Dr. T. R. Mankhand, Dr.Abhilash, S. Majumdar SOLVENT EXTRACTION AND SEPARATION OF COPPER AND ZINC FROM
15th International Conference on Non-ferrous metals, Hotel Oberoi Grand, Kolkata, July 8-9,2011 Eds: Dr. T. R. Mankhand, Dr.Abhilash, S. Majumdar SOLVENT EXTRACTION AND SEPARATION OF COPPER AND ZINC FROM
Solvent Extraction Study of Rare Earths from Nitrate Medium by the Mixtures of TBP and D2EHPA in Kerosene
 Journal of Metals, Materials and Minerals. Vol. 15 No.2 pp.89-95, 25 Solvent Extraction Study of Rare Earths from Nitrate Medium by the Mixtures of TBP and D2EHPA in Kerosene J.Kraikaew*, W.Srinuttrakul,
Journal of Metals, Materials and Minerals. Vol. 15 No.2 pp.89-95, 25 Solvent Extraction Study of Rare Earths from Nitrate Medium by the Mixtures of TBP and D2EHPA in Kerosene J.Kraikaew*, W.Srinuttrakul,
 TitleSolvent Extraction of Metal Acetyla Author(s) Tabushi, Masayuki Citation Bulletin of the Institute for Chemi University (1959), 37(4): 252-259 Issue Date 1959-11-25 URL http://hdl.handle.net/2433/75719
TitleSolvent Extraction of Metal Acetyla Author(s) Tabushi, Masayuki Citation Bulletin of the Institute for Chemi University (1959), 37(4): 252-259 Issue Date 1959-11-25 URL http://hdl.handle.net/2433/75719
Numerical investigation of separation of metals by non-steady-state cyclic counter-current liquid-liquid extraction.
 Research Article http://www.alliedacademies.org/journal-industrial-environmental-chemistry/ Numerical investigation of separation of metals by non-steady-state cyclic counter-current liquid-liquid extraction.
Research Article http://www.alliedacademies.org/journal-industrial-environmental-chemistry/ Numerical investigation of separation of metals by non-steady-state cyclic counter-current liquid-liquid extraction.
Separation of nickel and cobalt from calcium, magnesium and manganese by solvent extraction with synergistic mixtures of carboxylic acids
 Separation of nickel and cobalt from calcium, magnesium and manganese by solvent extraction with synergistic mixtures of carboxylic acids by A.C. du Preez* and J.S. Preston* Synopsis The separation of
Separation of nickel and cobalt from calcium, magnesium and manganese by solvent extraction with synergistic mixtures of carboxylic acids by A.C. du Preez* and J.S. Preston* Synopsis The separation of
Experiment 8. Determination of Iron in an Ore by Potentiometric Titration. Iron ores are often completely decomposed in hot concentrated HCl.
 Experiment 8 Determination of Iron in an Ore by Potentiometric Titration Introduction The common iron ores are hematite (Fe 2 O 3 ), magnetite (Fe 3 O 4 ), and limonite (3Fe 2 O 3 3H 2 O). Volumetric methods
Experiment 8 Determination of Iron in an Ore by Potentiometric Titration Introduction The common iron ores are hematite (Fe 2 O 3 ), magnetite (Fe 3 O 4 ), and limonite (3Fe 2 O 3 3H 2 O). Volumetric methods
Separation of Cobalt and Nickel with 2-Ethylhexyl Phosphonic Acid Mono-2-Ethylhexyl Ester Using a Countercurrent Mixer-Settler Cascade
 Solvent Extraction Research and Development, Japan, Vol. 4, No, 131 140 (017) Separation of balt and ckel with -Ethylhexyl Phosphonic Acid Mono--Ethylhexyl Ester Using a untercurrent Mixer-Settler Cascade
Solvent Extraction Research and Development, Japan, Vol. 4, No, 131 140 (017) Separation of balt and ckel with -Ethylhexyl Phosphonic Acid Mono--Ethylhexyl Ester Using a untercurrent Mixer-Settler Cascade
TitleSolvent Extraction of Beryllium as.
 TitleSolvent Extraction of Beryllium as Author(s) Tabushi, Masayuki Citation Bulletin of the Institute for Chemi University (1958), 36(6): 156-162 Issue Date 1958-11-30 URL http://hdl.handle.net/2433/75675
TitleSolvent Extraction of Beryllium as Author(s) Tabushi, Masayuki Citation Bulletin of the Institute for Chemi University (1958), 36(6): 156-162 Issue Date 1958-11-30 URL http://hdl.handle.net/2433/75675
Solubility Equilibrium
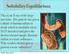 Solubility Equilibrium This is an X-ray of the large intestine. The patient was given a drink of barium sulfate to drink which is insoluble (does NOT dissolve) and gives the doctor a better image. Barium
Solubility Equilibrium This is an X-ray of the large intestine. The patient was given a drink of barium sulfate to drink which is insoluble (does NOT dissolve) and gives the doctor a better image. Barium
Introduction The Van London Co. Cadmium Ion Electrode is used to measure Cadmium ions in aqueous solutions.
 GENERAL INSTRUCTIONS Introduction The Van London Co. Cadmium Ion Electrode is used to measure Cadmium ions in aqueous solutions. Required Equipment 1. An ion meter 2. Cadmium Ion Sensing Electrode 3. Polishing
GENERAL INSTRUCTIONS Introduction The Van London Co. Cadmium Ion Electrode is used to measure Cadmium ions in aqueous solutions. Required Equipment 1. An ion meter 2. Cadmium Ion Sensing Electrode 3. Polishing
Journal of Chemical and Pharmaceutical Research
 Available on line www.jocpr.com Journal of Chemical and Pharmaceutical Research ISSN No: 0975-7384 CODEN(USA): JCPRC5 J. Chem. Pharm. Res., 2011, 3(4):945-950 Solvent Extraction of Iron(III) by Tri-n-Octyl
Available on line www.jocpr.com Journal of Chemical and Pharmaceutical Research ISSN No: 0975-7384 CODEN(USA): JCPRC5 J. Chem. Pharm. Res., 2011, 3(4):945-950 Solvent Extraction of Iron(III) by Tri-n-Octyl
The Synthesis of Copper Metal
 CHEM 109 Introduction to Chemistry Revision 1.0 The Synthesis of Copper Metal To learn about Oxidation-Reduction reactions. To learn about Half-Reactions and Half-Cells. To learn about the Activity of
CHEM 109 Introduction to Chemistry Revision 1.0 The Synthesis of Copper Metal To learn about Oxidation-Reduction reactions. To learn about Half-Reactions and Half-Cells. To learn about the Activity of
SULFURATION TREATMENT OF ELECTROPLATING WASTEWATER FOR SELECTIVE RECOVERY OF COPPER, ZINC AND NICKEL RESOURCE
 Global NEST Journal, Vol 8, No 2, pp 131-136, 20 Copyright 20 Global NEST Printed in Greece. All rights reserved SULFURATION TREATMENT OF ELECTROPLATING WASTEWATER FOR SELECTIVE RECOVERY OF COPPER, ZINC
Global NEST Journal, Vol 8, No 2, pp 131-136, 20 Copyright 20 Global NEST Printed in Greece. All rights reserved SULFURATION TREATMENT OF ELECTROPLATING WASTEWATER FOR SELECTIVE RECOVERY OF COPPER, ZINC
Spectrophotometric Determination of Cobalt in Water and Agricultural Samples
 Available online at www.joac.info ISSN: 2278-1862 Journal of Applicable Chemistry, 2012, 1 (4):500-504 (International Peer Reviewed Journal) Spectrophotometric Determination of Cobalt in Water and Agricultural
Available online at www.joac.info ISSN: 2278-1862 Journal of Applicable Chemistry, 2012, 1 (4):500-504 (International Peer Reviewed Journal) Spectrophotometric Determination of Cobalt in Water and Agricultural
39 Acid base titration: Alanine (HA2) (2- aminopropanoic acid) with sodium hydroxide
 Chemistry Sensors: Loggers: ph, Drop / Bubble counter Any EASYSENSE Logging time: EasyLog Teacher s notes 39 Acid base titration: Alanine (HA2) (2- aminopropanoic acid) with sodium hydroxide Read Amino
Chemistry Sensors: Loggers: ph, Drop / Bubble counter Any EASYSENSE Logging time: EasyLog Teacher s notes 39 Acid base titration: Alanine (HA2) (2- aminopropanoic acid) with sodium hydroxide Read Amino
Available online at ScienceDirect. Procedia Chemistry 19 (2016 ) M.Z. Mubarok a, L.I. Hanif a
 Available online at www.sciencedirect.com ScienceDirect Procedia Chemistry 19 (216 ) 743 75 5th International Conference on Recent Advances in Materials, Minerals and Environment (RAMM) & 2nd International
Available online at www.sciencedirect.com ScienceDirect Procedia Chemistry 19 (216 ) 743 75 5th International Conference on Recent Advances in Materials, Minerals and Environment (RAMM) & 2nd International
Formation Rate and Gas-Liquid Equilibrium of RuO4
 Journal of NUCLEAR SCIENCE and TECHNOLOGY, 25(5), pp. 472-478 (May 1988). Formation Rate and Gas-Liquid Equilibrium of RuO4 Akira SASAHIRA and Fumio KAWAMURA Energy Research Laboratory, Hitachi, Ltd.*
Journal of NUCLEAR SCIENCE and TECHNOLOGY, 25(5), pp. 472-478 (May 1988). Formation Rate and Gas-Liquid Equilibrium of RuO4 Akira SASAHIRA and Fumio KAWAMURA Energy Research Laboratory, Hitachi, Ltd.*
Solvent extraction behaviour of scandium from lateritic nickel-cobalt ores using different organic reagents
 Physicochem. Probl. Miner. Process., 54(2), 2018, 538-545 http://www.journalssystem.com/ppmp/ Physicochemical Problems of Mineral Processing ISSN 1643-1049 Wroclaw University of Science and Technology
Physicochem. Probl. Miner. Process., 54(2), 2018, 538-545 http://www.journalssystem.com/ppmp/ Physicochemical Problems of Mineral Processing ISSN 1643-1049 Wroclaw University of Science and Technology
UTILIZATION OF CALCIUM SULFIDE DERIVED FROM WASTE GYPSUM BOARD FOR METAL-CONTAINING WASTEWATER TREATMENT
 Global NEST Journal, Vol 1, No 1, pp 11-17, 28 Copyright 28 Global NEST Printed in Greece. All rights reserved UTILIZATION OF CALCIUM SULFIDE DERIVED FROM WASTE GYPSUM BOARD FOR METAL-CONTAINING WASTEWATER
Global NEST Journal, Vol 1, No 1, pp 11-17, 28 Copyright 28 Global NEST Printed in Greece. All rights reserved UTILIZATION OF CALCIUM SULFIDE DERIVED FROM WASTE GYPSUM BOARD FOR METAL-CONTAINING WASTEWATER
International Journal of Chemical Studies
 ISSN: 2321-4902 Volume 1 Issue 1 Online Available at www.chemijournal.com International Journal of Chemical Studies Calculation of Diffusion Coefficients and Layer Thickness for Oxidation the Ferrocene
ISSN: 2321-4902 Volume 1 Issue 1 Online Available at www.chemijournal.com International Journal of Chemical Studies Calculation of Diffusion Coefficients and Layer Thickness for Oxidation the Ferrocene
Precipitation Reactions and Gravimetric Analysis
 Precipitation Reactions and Gravimetric Analysis Titrations where the titrant forms a precipitate with the analyte. Not always so straightforward a number of requirements need to be met. Precipitation
Precipitation Reactions and Gravimetric Analysis Titrations where the titrant forms a precipitate with the analyte. Not always so straightforward a number of requirements need to be met. Precipitation
Chem 222 #11 Ch 16 Feb 15, 2005
 Chem 222 #11 Ch 16 Feb 15, 2005 Announcement Download your handout and answer sheet for Exp KH 3-14 starting in the next week at http://www.chem.uic.edu/chem222/cslist.htm Exp 7 Plot a graph of ph versus
Chem 222 #11 Ch 16 Feb 15, 2005 Announcement Download your handout and answer sheet for Exp KH 3-14 starting in the next week at http://www.chem.uic.edu/chem222/cslist.htm Exp 7 Plot a graph of ph versus
SENSORS FOR MANGANESE
 SENSORS FOR MANGANESE Contents 4.1 Synthesis of the Ionophore 4.2 Sensors based on TNCR 4.3 Fabrication of the sensors 4.4 Potential measurement and calibration 4.5 Optimization studies of the two types
SENSORS FOR MANGANESE Contents 4.1 Synthesis of the Ionophore 4.2 Sensors based on TNCR 4.3 Fabrication of the sensors 4.4 Potential measurement and calibration 4.5 Optimization studies of the two types
DISSOLUTION OF A COPPER ELECTRODE IN SULFURIC ACID AT POLARIZATION BY AN INDUSTRIAL ALTERNATING CURRENT
 Int. J. Chem. Sci.: 12(3), 2014, 9-1014 ISSN 0972-768X www.sadgurupublications.com DISSOLUTION OF A COPPER ELECTRODE IN SULFURIC ACID AT POLARIZATION BY AN INDUSTRIAL ALTERNATING CURRENT A. B. BAESHOV,
Int. J. Chem. Sci.: 12(3), 2014, 9-1014 ISSN 0972-768X www.sadgurupublications.com DISSOLUTION OF A COPPER ELECTRODE IN SULFURIC ACID AT POLARIZATION BY AN INDUSTRIAL ALTERNATING CURRENT A. B. BAESHOV,
METAL FINISHING. (As per revised VTU syllabus: )
 METAL FINISHING (As per revised VTU syllabus: 2015-16) Definition: It is a process in which a specimen metal (article) is coated with another metal or a polymer in order to modify the surface properties
METAL FINISHING (As per revised VTU syllabus: 2015-16) Definition: It is a process in which a specimen metal (article) is coated with another metal or a polymer in order to modify the surface properties
SERVA IMAC Ni-IDA Test Kit Agarose for Affinity Purification of His-Tag Fusion Proteins
 INSTRUCTION MANUAL SERVA IMAC Ni-IDA Test Kit Agarose for Affinity Purification of His-Tag Fusion Proteins (Cat. No.42164, 42165) SERVA Electrophoresis GmbH - Carl-Benz-Str. 7-69115 Heidelberg Phone +49-6221-138400,
INSTRUCTION MANUAL SERVA IMAC Ni-IDA Test Kit Agarose for Affinity Purification of His-Tag Fusion Proteins (Cat. No.42164, 42165) SERVA Electrophoresis GmbH - Carl-Benz-Str. 7-69115 Heidelberg Phone +49-6221-138400,
AFFINITY HIS-TAG PURIFICATION
 DESCRIPTION Test kits are a fast and easy way to screen different IMAC resins before choosing the most appropriate product for each target-protein. All the products supplied in the Test kit are suitable
DESCRIPTION Test kits are a fast and easy way to screen different IMAC resins before choosing the most appropriate product for each target-protein. All the products supplied in the Test kit are suitable
The Release of Base Metals During Acidic Leaching of Fly Ash
 The Release of Base Metals During Acidic Leaching of Fly Ash George Kazonich and Ann G. Kim U.S. Department of Energy Federal Energy Technology Center P.O. Box 19 Pittsburgh, PA 153 ABSTRACT Since 199,
The Release of Base Metals During Acidic Leaching of Fly Ash George Kazonich and Ann G. Kim U.S. Department of Energy Federal Energy Technology Center P.O. Box 19 Pittsburgh, PA 153 ABSTRACT Since 199,
Recovery of Cu and EDTA from EDTA-Cu Solution by use of Electrodialysis Accompanied by Electrochemical Reaction
 Vol., No. () Article Recovery of Cu and from Solution by use of Electrodialysis Accompanied by Electrochemical Reaction Hiroshi TAKAHASHI *, Etsuko KASHIUCHI and Kenzo MUNAKATA Department of Engineering
Vol., No. () Article Recovery of Cu and from Solution by use of Electrodialysis Accompanied by Electrochemical Reaction Hiroshi TAKAHASHI *, Etsuko KASHIUCHI and Kenzo MUNAKATA Department of Engineering
QUANTITATIVE DETERMINATION OF METALS IONS USING Fe (III)/ Fe (II) REDOX TITRATION SYSTEM WITH A PLATINUM ELECTRODE
 Journal of the University of M. Chemical M. El Jamal, Technology H. H. Hammud and Metallurgy, 42, 1, 27, 97-14 QUANTITATIVE DETERMINATION OF METALS IONS USING Fe (III/ Fe (II REDOX TITRATION SYSTEM WITH
Journal of the University of M. Chemical M. El Jamal, Technology H. H. Hammud and Metallurgy, 42, 1, 27, 97-14 QUANTITATIVE DETERMINATION OF METALS IONS USING Fe (III/ Fe (II REDOX TITRATION SYSTEM WITH
Quantitative Analysis of Phosphorus Removal Based on its As-Beneficiated Content, Iron Extraction Rate and Input Concentration of H 2 O 2
 INTERNATIONAL JOURNAL OF MULTIDISCIPLINARY SCIENCES AND ENGINEERING, VOL. 4, NO. 6, JULY 213 Quantitative Analysis of Phosphorus Removal Based on its As-Beneficiated Content, Iron Extraction Rate and Input
INTERNATIONAL JOURNAL OF MULTIDISCIPLINARY SCIENCES AND ENGINEERING, VOL. 4, NO. 6, JULY 213 Quantitative Analysis of Phosphorus Removal Based on its As-Beneficiated Content, Iron Extraction Rate and Input
Electrochemistry LEC Electrogravimetric determination of copper. What you need: What you can learn about. Principle and tasks
 Electrochemistry LEC 06 What you can learn about Quantitative analysis Electrolysis Gravimetry Overpotential and electrode polarisation Principle and tasks Electrogravimetry is an important analytical
Electrochemistry LEC 06 What you can learn about Quantitative analysis Electrolysis Gravimetry Overpotential and electrode polarisation Principle and tasks Electrogravimetry is an important analytical
COPPER CYCLE EXPERIMENT 3
 COPPER CYCLE EXPERIMENT 3 INTRODUCTION One simple way to state the aim of chemistry is: The study of matter and its transformations. In this experiment, a copper sample will appear in five different forms
COPPER CYCLE EXPERIMENT 3 INTRODUCTION One simple way to state the aim of chemistry is: The study of matter and its transformations. In this experiment, a copper sample will appear in five different forms
Preparation of Cyclohexene From Cyclohexanol
 EXPERIMENT 9 Alkene Synthesis From Alcohol Preparation of Cyclohexene From Cyclohexanol Purpose: a) Preparation of an alkene by dehydration (elimination of water) of an alcohol in the presence of an acid
EXPERIMENT 9 Alkene Synthesis From Alcohol Preparation of Cyclohexene From Cyclohexanol Purpose: a) Preparation of an alkene by dehydration (elimination of water) of an alcohol in the presence of an acid
Standards of Ethanol in Water. Analytical Volumetric Solutions
 Standards of Ethanol in Water These standards are intented for use as reference solutions for alcohol content measuring instruments. They are prepared using organic-free water, and the certified concentrations
Standards of Ethanol in Water These standards are intented for use as reference solutions for alcohol content measuring instruments. They are prepared using organic-free water, and the certified concentrations
Gypsum Solubility Simulating Real World Aqueous Problems
 Simulating Real World Aqueous Problems or Getting an edge on the competition! Reliable simulation of complex processes can provide insights for process optimization and reduced operating costs. The solubility
Simulating Real World Aqueous Problems or Getting an edge on the competition! Reliable simulation of complex processes can provide insights for process optimization and reduced operating costs. The solubility
International Journal of Technical Research and Applications e Konaem Wasamon, Kittisupakorn Paisan Abstract
 International Journal of Technical Research and Applications e-issn: 30-8163, SIMULATION FOR SYNERGISTIC EXTRACTION OF NEODYMIUM IONS WITH HOLLOW FIBER SUPPORTED LIQUID MEMBRANE Konaem Wasamon, Kittisupakorn
International Journal of Technical Research and Applications e-issn: 30-8163, SIMULATION FOR SYNERGISTIC EXTRACTION OF NEODYMIUM IONS WITH HOLLOW FIBER SUPPORTED LIQUID MEMBRANE Konaem Wasamon, Kittisupakorn
SERVA Ni-NTA Magnetic Beads
 INSTRUCTION MANUAL SERVA Ni-NTA Magnetic Beads Magnetic beads for Affinity Purification of His-Tag Fusion Proteins (Cat. No. 42179) SERVA Electrophoresis GmbH - Carl-Benz-Str. 7-69115 Heidelberg Phone
INSTRUCTION MANUAL SERVA Ni-NTA Magnetic Beads Magnetic beads for Affinity Purification of His-Tag Fusion Proteins (Cat. No. 42179) SERVA Electrophoresis GmbH - Carl-Benz-Str. 7-69115 Heidelberg Phone
Carbide Capacity of CaO SiO 2 CaF 2 ( Na 2 O) Slags at K
 ISIJ International, Vol. 44 (004), No., pp. 3 8 Carbide Capacity of CaO SiO CaF ( Na O) Slags at 1 773 K Joo Hyun PARK and Dong Joon MIN 1) Stainless Steel Research Group, Technical Research Laboratory,
ISIJ International, Vol. 44 (004), No., pp. 3 8 Carbide Capacity of CaO SiO CaF ( Na O) Slags at 1 773 K Joo Hyun PARK and Dong Joon MIN 1) Stainless Steel Research Group, Technical Research Laboratory,
A cobalt solvent extraction investigation in Africa s Copper Belt
 KÖNIGHOFER, T., ARCHER, S.J., and BRADFORD, L. A cobalt solvent extraction investigation in Africa s Copper Belt. Hydrometallurgy Conference 2009, The Southern African Institute of Mining and Metallurgy,
KÖNIGHOFER, T., ARCHER, S.J., and BRADFORD, L. A cobalt solvent extraction investigation in Africa s Copper Belt. Hydrometallurgy Conference 2009, The Southern African Institute of Mining and Metallurgy,
Experiment 1 Hard-Soft Acids and Bases: Altering the Cu + /Cu 2+ Equilibrium with Nitrogen, Oxygen, or Halide Ligands
 Experiment 1 Hard-Soft Acids and Bases: Altering the Cu + /Cu 2+ Equilibrium with Nitrogen, Oxygen, or Halide Ligands *Parts of the experiment are based on C. E. Ophardt & G. Wollaston, J. Chem. Ed. 1991,
Experiment 1 Hard-Soft Acids and Bases: Altering the Cu + /Cu 2+ Equilibrium with Nitrogen, Oxygen, or Halide Ligands *Parts of the experiment are based on C. E. Ophardt & G. Wollaston, J. Chem. Ed. 1991,
BORABU-MASABA DISTRICTS JOINT EVALUATION TEST 2012 Kenya Certificate of Secondary Education (K.C.S.E)
 Name. School Candidate s Signature. Index No /. Date. 233/2 CHEMISTRY Paper 2 (Theory) JULY / AUGUST - 2012 Time: 2 Hours BORABU-MASABA DISTRICTS JOINT EVALUATION TEST 2012 Kenya Certificate of Secondary
Name. School Candidate s Signature. Index No /. Date. 233/2 CHEMISTRY Paper 2 (Theory) JULY / AUGUST - 2012 Time: 2 Hours BORABU-MASABA DISTRICTS JOINT EVALUATION TEST 2012 Kenya Certificate of Secondary
Stern-Volmer Quenching of Conjugated Polymers: A Study of Fluorophore Concentration
 V Stern-Volmer Quenching of Conjugated Polymers: A Study of Fluorophore Concentration An Undergraduate Thesis by Christopher Vaughns Faculty Advisor: Dr. Uwe Bunz Georgia Institute of Technology School
V Stern-Volmer Quenching of Conjugated Polymers: A Study of Fluorophore Concentration An Undergraduate Thesis by Christopher Vaughns Faculty Advisor: Dr. Uwe Bunz Georgia Institute of Technology School
Leaching of Waste Hydroxide Sludges by Complex-Forming Agents
 International Conference with Elements of School for Young Scientists on Recycling and Utilization of Technogenic Formations (2017) Conference Paper Leaching of Waste Hydroxide Sludges by Complex-Forming
International Conference with Elements of School for Young Scientists on Recycling and Utilization of Technogenic Formations (2017) Conference Paper Leaching of Waste Hydroxide Sludges by Complex-Forming
Modified Zincex Process by Técnicas Reunidas
 The Modified ZINCEX TM Process is an advantageous smelting hydrometallurgical process licensed by Técnicas Reunidas to produce ultra-pure Zinc cathodes and enable the recovery of other valuable metals.
The Modified ZINCEX TM Process is an advantageous smelting hydrometallurgical process licensed by Técnicas Reunidas to produce ultra-pure Zinc cathodes and enable the recovery of other valuable metals.
MAROOCHYDORE SHS. Student booklet. Including. ame: Teacher: Name. er: Maroochydore SHS Chemistry Department
 MAROOCHYDORE SHS Including Student booklet Name ame: Teacher: er: EXPERIMENT 3.1 AIM: LEACHING THE COPPER ORE STUDENT ACTIVITY SHEET To simulate the leaching of oxidised copper ores using dilute sulphuric
MAROOCHYDORE SHS Including Student booklet Name ame: Teacher: er: EXPERIMENT 3.1 AIM: LEACHING THE COPPER ORE STUDENT ACTIVITY SHEET To simulate the leaching of oxidised copper ores using dilute sulphuric
AFFINITY HIS-TAG PURIFICATION
 DESCRIPTION Resins are products that allow batch or column purifications. This product is supplied as a suspension in 20% ethanol. INSTRUCTIONS The resins are adapted to work mainly in native conditions
DESCRIPTION Resins are products that allow batch or column purifications. This product is supplied as a suspension in 20% ethanol. INSTRUCTIONS The resins are adapted to work mainly in native conditions
Liquid-liquid extraction of aluminum (III) from sodium hydroxide solutions by alkylated hydroxyquinoline
 Vol. 40, No. 6 429 RESEARCH REPORT Liquid-liquid extraction of aluminum (III) from sodium hydroxide solutions by alkylated hydroxyquinoline Taichi SATO* In the extraction of aluminum (III) from sodium
Vol. 40, No. 6 429 RESEARCH REPORT Liquid-liquid extraction of aluminum (III) from sodium hydroxide solutions by alkylated hydroxyquinoline Taichi SATO* In the extraction of aluminum (III) from sodium
Extraction and Spectrophotometric Assay of Yttrium (III) with TOPO, Application to Water Samples and Alloys
 Science Journal of Chemistry 2017; 5(6): 80-86 http://www.sciencepublishinggroup.com/j/sjc doi: 10.11648/j.sjc.20170506.12 ISSN: 2330-0981 (Print); ISSN: 2330-099X (Online) Extraction and Spectrophotometric
Science Journal of Chemistry 2017; 5(6): 80-86 http://www.sciencepublishinggroup.com/j/sjc doi: 10.11648/j.sjc.20170506.12 ISSN: 2330-0981 (Print); ISSN: 2330-099X (Online) Extraction and Spectrophotometric
hydroxynitrile ester dihaloalkane alkane alkene haloalkane alcohol amine nitrile ketone HCN + KCN Nucleophilic addition carboxylic acid
 6.2.5 Synthesis dihaloalkane poly(alkene) high pressure atalyst polymerization Br 2, l 2 Electrophilic addition alkene alkane 2, Nickel atalyst addition/reduction Br 2, l 2 UV light Free radical Substitution
6.2.5 Synthesis dihaloalkane poly(alkene) high pressure atalyst polymerization Br 2, l 2 Electrophilic addition alkene alkane 2, Nickel atalyst addition/reduction Br 2, l 2 UV light Free radical Substitution
AFFINITY HIS-TAG PURIFICATION
 DESCRIPTION Resins are products that allow batch or column purifications. This product is supplied as a suspension in 20% ethanol. INSTRUCTIONS The resins are adapted to work mainly in native conditions
DESCRIPTION Resins are products that allow batch or column purifications. This product is supplied as a suspension in 20% ethanol. INSTRUCTIONS The resins are adapted to work mainly in native conditions
AFFINITY HIS-TAG PURIFICATION
 DESCRIPTION Nickel Affinity Cartridges 5ml are used for purification of histidine-tagged proteins in native or denaturing conditions. This cartridge can be used with an automated chromatography system,
DESCRIPTION Nickel Affinity Cartridges 5ml are used for purification of histidine-tagged proteins in native or denaturing conditions. This cartridge can be used with an automated chromatography system,
Minerals Engineering
 Minerals Engineering 23 (21) 492 497 Contents lists available at ScienceDirect Minerals Engineering journal homepage: www.elsevier.com/locate/mineng Development of a process for the separation of zinc
Minerals Engineering 23 (21) 492 497 Contents lists available at ScienceDirect Minerals Engineering journal homepage: www.elsevier.com/locate/mineng Development of a process for the separation of zinc
Research Journal of Pharmaceutical, Biological and Chemical Sciences
 Research Journal of Pharmaceutical, Biological and Chemical Sciences Electrochemical Simultaneous Removal of Copper and Zinc from Bimetallic Solution Using Packed-Bed Cathode Made of Carbon Particles -
Research Journal of Pharmaceutical, Biological and Chemical Sciences Electrochemical Simultaneous Removal of Copper and Zinc from Bimetallic Solution Using Packed-Bed Cathode Made of Carbon Particles -
PROCEDURE FOR USE NICKEL NTA Magnetic Agarose Beads (5%)
 1 AFFINITY HIS-TAG PURIFICATION PROCEDURE FOR USE NICKEL NTA Magnetic Agarose Beads (5%) DESCRIPTION Nickel NTA Magnetic Agarose Beads are products that allow rapid and easy small-scale purification of
1 AFFINITY HIS-TAG PURIFICATION PROCEDURE FOR USE NICKEL NTA Magnetic Agarose Beads (5%) DESCRIPTION Nickel NTA Magnetic Agarose Beads are products that allow rapid and easy small-scale purification of
2,6-Lutidine-N-oxide complexes of rare-earth bromides
 Prec. Indian Aead. Sei. (Chem. Sci.), Vol. 90, Number 2, April 1981, pp. 131-135. Priated in India. 2,6-Lutidine-N-oxide complexes of rare-earth bromides P V SIVAPULLAIAH* and S SOUNDARARAJAN* Department
Prec. Indian Aead. Sei. (Chem. Sci.), Vol. 90, Number 2, April 1981, pp. 131-135. Priated in India. 2,6-Lutidine-N-oxide complexes of rare-earth bromides P V SIVAPULLAIAH* and S SOUNDARARAJAN* Department
PRACTICAL STUDY OF A SOLVENT EXTRACTION PROCESS FOR SEPARATION OF COBALT AND NICKEL WITH 2-ETHYLHEXYLPHOSPHONIC ACID MONO-2-ETHYLHEXYL ESTER
 =organic-phase species or organic-phase concentration Literature Cited 1) Cotton, F. A. and G. Wilkinson: "Advanced Inorganic Chemistry," 3rd ed., p. 874 and 890, Interscience Publishers, New York (1972).
=organic-phase species or organic-phase concentration Literature Cited 1) Cotton, F. A. and G. Wilkinson: "Advanced Inorganic Chemistry," 3rd ed., p. 874 and 890, Interscience Publishers, New York (1972).
Solubility Worksheet #2 Level 3
 Name Date We have considered some conditions that allow solutes to dissolve in solvents, but we have not yet considered the solubility of a solute. Solubility is defined as the maximum mass of solute that
Name Date We have considered some conditions that allow solutes to dissolve in solvents, but we have not yet considered the solubility of a solute. Solubility is defined as the maximum mass of solute that
O 3, SIO 2. O, CAO, Al 2 AND MG IN ANT-HILL SOIL SAMPLES WITHIN ABRAKA TOWN IN NIGERIA
 Int. J. Agric.Sc & Vet.Med. 2014 Ekakitie A O and Osakwe A A, 2014 Research Paper ISSN 2320-3730 www.ijasvm.com Vol. 2, No. 3, August 2014 2014 www.ijasvm.com. All Rights Reserved DETERMINATION OF FE 2,
Int. J. Agric.Sc & Vet.Med. 2014 Ekakitie A O and Osakwe A A, 2014 Research Paper ISSN 2320-3730 www.ijasvm.com Vol. 2, No. 3, August 2014 2014 www.ijasvm.com. All Rights Reserved DETERMINATION OF FE 2,
Test sticks and test papers for semi-quantitative determinations
 QUANTOFIX test sticks for semi-quantitative determinations QUANTOFIX test sticks meet the most important requirements for a modern quick-test: rapid dip and read convenient the analysis can be carried
QUANTOFIX test sticks for semi-quantitative determinations QUANTOFIX test sticks meet the most important requirements for a modern quick-test: rapid dip and read convenient the analysis can be carried
LIQUID-LIQUID EXTRACTION OF IRON (III) FROM OUENZA IRON ORE LEACH LIQUOR BY TRIBUTYLPHOSPHATE. A. Nouioua*, D. Barkat
 Journal of Fundamental and Applied Sciences Research Article ISSN 1112-9867 Available online at http://www.jfas.info LIQUID-LIQUID EXTRACTION OF IRON (III) FROM OUENZA IRON ORE LEACH LIQUOR BY TRIBUTYLPHOSPHATE
Journal of Fundamental and Applied Sciences Research Article ISSN 1112-9867 Available online at http://www.jfas.info LIQUID-LIQUID EXTRACTION OF IRON (III) FROM OUENZA IRON ORE LEACH LIQUOR BY TRIBUTYLPHOSPHATE
Empirical Evaluation of Removed Phosphorus Concentration Based on Its As- Beneficiated Content and Grain Size of Iron Ore Leached in H 2 O 2
 Empirical Evaluation of Removed Phosphorus Concentration Based on Its As- Beneficiated Content and Grain Size of Iron Ore Leached in H 2 O 2 T. O. Chime, O.D. Onukwuli and A.J. Ujam Department of Chemical
Empirical Evaluation of Removed Phosphorus Concentration Based on Its As- Beneficiated Content and Grain Size of Iron Ore Leached in H 2 O 2 T. O. Chime, O.D. Onukwuli and A.J. Ujam Department of Chemical
AP*Chemistry Solubility Equilibria
 AP*Chemistry Solubility Equilibria SOLUBILITY EQUILIBRIA (K sp, THE SOLUBILITY PRODUCT) Saturated solutions of salts are another type of chemical equilibria. Remember those solubility rules? The fine print
AP*Chemistry Solubility Equilibria SOLUBILITY EQUILIBRIA (K sp, THE SOLUBILITY PRODUCT) Saturated solutions of salts are another type of chemical equilibria. Remember those solubility rules? The fine print
ELUTION BEHAVIOR OF PHOSPHATE CONTAINED IN Mg/Fe AND Zn/Fe LAYERED DOUBLE HYDROXIDES
 Advanced Materials Development and Performance (AMDP11) International Journal of Modern Physics: Conference Series Vol. 6 (12) 156-161 World Scientific Publishing Company DOI: 1.112/S11951318 ELUTION BEHAVIOR
Advanced Materials Development and Performance (AMDP11) International Journal of Modern Physics: Conference Series Vol. 6 (12) 156-161 World Scientific Publishing Company DOI: 1.112/S11951318 ELUTION BEHAVIOR
Estimation of copper in brass by iodo-potentiometric technique of analysis
 Trade Science Inc. October 2008 Volume 7 Issue 9 ACAIJ, 7(9) 2008 [694-702] Estimation of copper in brass by iodo-potentiometric technique of analysis H.C.Anandamurthy*, O.G.Palanna R & D Centre, Department
Trade Science Inc. October 2008 Volume 7 Issue 9 ACAIJ, 7(9) 2008 [694-702] Estimation of copper in brass by iodo-potentiometric technique of analysis H.C.Anandamurthy*, O.G.Palanna R & D Centre, Department
Preparation of potassium nitrate from potassium chloride and magnesium nitrate in a laboratory scale using industrial raw materials
 Preparation of potassium nitrate from potassium chloride and magnesium nitrate in a laboratory scale using industrial raw materials Jana Jurišová a, Pavel Fellner a, Vladimír Danielik a, Marek Lencsés
Preparation of potassium nitrate from potassium chloride and magnesium nitrate in a laboratory scale using industrial raw materials Jana Jurišová a, Pavel Fellner a, Vladimír Danielik a, Marek Lencsés
Recovery of cobalt sulfate from spent lithium ion batteries by reductive leaching and solvent extraction with Cyanex-272
 Accepted Manuscript Recovery of cobalt sulfate from spent lithium ion batteries by reductive leaching and solvent extraction with Cyanex-272 Jingu Kang, Gamini Senanayake, Jeongsoo Sohn, Shun Myung Shin
Accepted Manuscript Recovery of cobalt sulfate from spent lithium ion batteries by reductive leaching and solvent extraction with Cyanex-272 Jingu Kang, Gamini Senanayake, Jeongsoo Sohn, Shun Myung Shin
Complex formation metal ion=lewis acid=electron-pair acceptor ligand = Lewis base= electron-pair donor
 Complex formation titrations Complex formation Chelate effect EDTA Conditional formation constant EDTA titration Indicators Types of titrations Auxiliary complexing agents Complex formation metal ion=lewis
Complex formation titrations Complex formation Chelate effect EDTA Conditional formation constant EDTA titration Indicators Types of titrations Auxiliary complexing agents Complex formation metal ion=lewis
Corrosion and inhibition of Cu-Zn alloys in NaCl solution by using permanganate and phosphate anions
 Int. J. Electrochem. Sci., 2 (2007) 563-571 www.electrochemsci.org Corrosion and inhibition of Cu-Zn alloys in NaCl solution by using permanganate and phosphate anions S. A. M. Refaey 1, *, A. M. Abd El
Int. J. Electrochem. Sci., 2 (2007) 563-571 www.electrochemsci.org Corrosion and inhibition of Cu-Zn alloys in NaCl solution by using permanganate and phosphate anions S. A. M. Refaey 1, *, A. M. Abd El
Complies with the monograph for Parenteral Preparations and with that for Radiopharmaceuticals.
 January 2009 THALLOUS ( 201 Tl) CHLORIDE INJECTION: Final text for addition to The International Pharmacopoeia (January 2009) This monograph was adopted at the Forty-third WHO Expert Committee on Specifications
January 2009 THALLOUS ( 201 Tl) CHLORIDE INJECTION: Final text for addition to The International Pharmacopoeia (January 2009) This monograph was adopted at the Forty-third WHO Expert Committee on Specifications
Ultrasonic investigation of induced dipole-induced dipole interactions in binary liquid mixtures at 298K
 Indian Journal of Pure & Applied Physics Vol. 44, November 2006, pp. 815-819 Ultrasonic investigation of induced dipole-induced dipole interactions in binary liquid mixtures at 298K V Kannappan # & R Jaya
Indian Journal of Pure & Applied Physics Vol. 44, November 2006, pp. 815-819 Ultrasonic investigation of induced dipole-induced dipole interactions in binary liquid mixtures at 298K V Kannappan # & R Jaya
Recovery of Cu, Zn, Ni and Cr from Plating Sludge by Combined Sulfidation and Oxidation Treatment
 Recovery of Cu, Zn, Ni and Cr from Plating Sludge by Combined Sulfidation and Oxidation Treatment D. Kuchar, T. Fukuta, M. Kubota, and H. Matsuda Abstract The selective recovery of heavy metals of Cu,
Recovery of Cu, Zn, Ni and Cr from Plating Sludge by Combined Sulfidation and Oxidation Treatment D. Kuchar, T. Fukuta, M. Kubota, and H. Matsuda Abstract The selective recovery of heavy metals of Cu,
Recovery of Cu, Zn, Ni and Cr from Plating Sludge by Combined Sulfidation and Oxidation Treatment
 Recovery of Cu, Zn, Ni and Cr from Plating Sludge by Combined Sulfidation and Oxidation Treatment D. Kuchar, T. Fukuta, M. Kubota, and H. Matsuda Abstract The selective recovery of heavy metals of Cu,
Recovery of Cu, Zn, Ni and Cr from Plating Sludge by Combined Sulfidation and Oxidation Treatment D. Kuchar, T. Fukuta, M. Kubota, and H. Matsuda Abstract The selective recovery of heavy metals of Cu,
Electronic Supplementary Information (ESI) for
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information (ESI) for Enhancement of the coercivity in Co-Ni
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information (ESI) for Enhancement of the coercivity in Co-Ni
DETERMINATION OF HARDNESS IN A WATER SAMPLE
 22 DETERMINATION OF ARDNESS IN A WATER SAMPLE Purpose: To learn the EDTA titration method for determining the hardness of a water sample BACKGROUND In the past, water hardness was defined as a measure
22 DETERMINATION OF ARDNESS IN A WATER SAMPLE Purpose: To learn the EDTA titration method for determining the hardness of a water sample BACKGROUND In the past, water hardness was defined as a measure
Synthesis of Erichrome Black T-Zn 2+ complex by electrochemical method, Characterization and Kinetic study of the formation of complex
 International Journal of Chemistry and Applications. ISSN 0974-3111 Volume 5, Number 3 (2013), pp. 169-178 International Research Publication House http://www.irphouse.com Synthesis of Erichrome Black
International Journal of Chemistry and Applications. ISSN 0974-3111 Volume 5, Number 3 (2013), pp. 169-178 International Research Publication House http://www.irphouse.com Synthesis of Erichrome Black
A Cycle of Copper Reactions
 EXPERIMENT A Cycle of Copper Reactions PURPOSE To demonstrate a series of copper reactions: starting with copper metal, oxidizing the metal to put it into solution and then, form a copper hydroxide, an
EXPERIMENT A Cycle of Copper Reactions PURPOSE To demonstrate a series of copper reactions: starting with copper metal, oxidizing the metal to put it into solution and then, form a copper hydroxide, an
EXPERIMENT 5. Molecular Absorption Spectroscopy: Determination of Iron with 1,10-Phenanthroline
 EXPERIMENT 5 Molecular Absorption Spectroscopy: Determination of Iron with 1,10-Phenanthroline UNKNOWN Submit a clean, labeled 100-mL volumetric flask to the instructor so that your unknown iron solution
EXPERIMENT 5 Molecular Absorption Spectroscopy: Determination of Iron with 1,10-Phenanthroline UNKNOWN Submit a clean, labeled 100-mL volumetric flask to the instructor so that your unknown iron solution
THALLOUS ( 201 Tl) CHLORIDE INJECTION: Revised Final text for addition to The International Pharmacopoeia (January September 2009)
 September 2009 RESTRICTED THALLOUS ( 201 Tl) CHLORIDE INJECTION: Revised Final text for addition to The International Pharmacopoeia (January September 2009) [Note from the Secretariat: This monograph was
September 2009 RESTRICTED THALLOUS ( 201 Tl) CHLORIDE INJECTION: Revised Final text for addition to The International Pharmacopoeia (January September 2009) [Note from the Secretariat: This monograph was
1. Bloomsbury BBSRC Centre for Structural Biology, Birkbeck College and University College London.
 Purification/Polishing of His-tagged proteins - Application of Centrifugal Vivapure Ion-exchange Membrane Devices to the Purification/Polishing of Histagged Background Multi-milligram quantities of highly
Purification/Polishing of His-tagged proteins - Application of Centrifugal Vivapure Ion-exchange Membrane Devices to the Purification/Polishing of Histagged Background Multi-milligram quantities of highly
FOR 3457 Forest Watershed & Water Quality Watershed Management Tools & Methods. Water Quality Lab
 FOR 3457 Forest Watershed & Water Quality Watershed Management Tools & Methods Water Quality Lab In this lab you will be using the water, snow, and soil samples to determine the general quality of the
FOR 3457 Forest Watershed & Water Quality Watershed Management Tools & Methods Water Quality Lab In this lab you will be using the water, snow, and soil samples to determine the general quality of the
Solution Chemistry of Molten Amide-Nitrate Eutectics
 Solution Chemistry of Molten Amide-Nitrate Eutectics a E. I. EWEKA and b D. H. KERRIDGE a Department of Chemistry, University of Southampton, Highfield, Southampton, SO17 1BJ United Kingdom b Department
Solution Chemistry of Molten Amide-Nitrate Eutectics a E. I. EWEKA and b D. H. KERRIDGE a Department of Chemistry, University of Southampton, Highfield, Southampton, SO17 1BJ United Kingdom b Department
Lecture 11. Solubility of Ionic Compounds in Water
 Lecture 11 Solubility of Ionic Compounds in Water Soluble Compounds Exceptions Acetates, Nitrates, Chlorates Na +, K +, NH 4 + compounds None None Halides ( Cl -, Br -, I - ) Ag +, Hg 2 2+, Cu +, Pb 2+
Lecture 11 Solubility of Ionic Compounds in Water Soluble Compounds Exceptions Acetates, Nitrates, Chlorates Na +, K +, NH 4 + compounds None None Halides ( Cl -, Br -, I - ) Ag +, Hg 2 2+, Cu +, Pb 2+
Compounds & Reactions Week 1. Writing Formulas & Balancing Equations. Write the chemical formula for each molecular (covalent) compound.
 Compounds & Reactions Week 1 Name Writing Formulas & Balancing Equations Write the chemical formula for each ionic compound. 1. Lithium fluoride 2. Copper (II) chloride 3. Manganese (II) oxide 4. Potassium
Compounds & Reactions Week 1 Name Writing Formulas & Balancing Equations Write the chemical formula for each ionic compound. 1. Lithium fluoride 2. Copper (II) chloride 3. Manganese (II) oxide 4. Potassium
Nitrogen, Simplified TKN (s-tkn ) DOC
 Nitrogen, Simplified Total Kjeldahl, TNTplus 880, 10242 DOC316.53.01258 s-tkn Method Method 10242 0 to 16 mg/l TKN TNTplus 880 Scope and Application: For water and wastewater. Digestion is required. Test
Nitrogen, Simplified Total Kjeldahl, TNTplus 880, 10242 DOC316.53.01258 s-tkn Method Method 10242 0 to 16 mg/l TKN TNTplus 880 Scope and Application: For water and wastewater. Digestion is required. Test
NITEC CRYSTAL 1. Elektrolyt to achieve uniform and non reflective Nickel deposits
 Postfach 169 CH-9545 Wängi TG 25.07.2014 NITEC CRYSTAL 1 Elektrolyt to achieve uniform and non reflective Nickel deposits In contrast to bright nickel, the NITEC CRYSTAL electroplating process produces
Postfach 169 CH-9545 Wängi TG 25.07.2014 NITEC CRYSTAL 1 Elektrolyt to achieve uniform and non reflective Nickel deposits In contrast to bright nickel, the NITEC CRYSTAL electroplating process produces
Pitting Corrosion of Some Stainless Steel Alloys Preoxidized at Different Conditions
 Portugaliae Electrochimica Acta 25 (2007) 237-248 PORTUGALIAE ELECTROCHIMICA ACTA Pitting Corrosion of Some Stainless Steel Alloys Preoxidized at Different Conditions S.S. Mahmoud * and M.M. Ahmed a Chemistry
Portugaliae Electrochimica Acta 25 (2007) 237-248 PORTUGALIAE ELECTROCHIMICA ACTA Pitting Corrosion of Some Stainless Steel Alloys Preoxidized at Different Conditions S.S. Mahmoud * and M.M. Ahmed a Chemistry
Shigematsu, Tsunenobu; Tabushi, Mas Masakazu. Citation University (1963), 40(5-6):
 TitleCoprecipitation of Scandium with Ca Author(s) Shigematsu, Tsunenobu; Tabushi, Mas Masakazu Citation Bulletin of the Institute for Chemi University (1963), 40(5-6): 381-387 Issue Date 1963-01-30 URL
TitleCoprecipitation of Scandium with Ca Author(s) Shigematsu, Tsunenobu; Tabushi, Mas Masakazu Citation Bulletin of the Institute for Chemi University (1963), 40(5-6): 381-387 Issue Date 1963-01-30 URL
INTERDISCIPLINARY INVESTIGATION (IDI)-LAB LABORATORY HANDOUT
 INTERDISCIPLINARY INVESTIGATION (IDI)-LAB LABORATORY HANDOUT Research-based Interdisciplinary Science Education ION CHROMATOGRAPHY TO INVESTIGATE WATER QUALITY ISSUES: FLUORIDE IN DRINKING WATER Introduction
INTERDISCIPLINARY INVESTIGATION (IDI)-LAB LABORATORY HANDOUT Research-based Interdisciplinary Science Education ION CHROMATOGRAPHY TO INVESTIGATE WATER QUALITY ISSUES: FLUORIDE IN DRINKING WATER Introduction
ESTIMATION OF MATERIAL BALANCE IN PYROMETALLURGICAL PARTITIONING PROCESS FOR TRUs FROM HLLW
 ESTIMATION OF MATERIAL BALANCE IN PYROMETALLURGICAL PARTITIONING PROCESS FOR TRUs FROM HLLW Kensuke Kinoshita, Masaki Kurata, Koichi Uozumi, and Tadashi Inoue Central Research Institute of Electric Power
ESTIMATION OF MATERIAL BALANCE IN PYROMETALLURGICAL PARTITIONING PROCESS FOR TRUs FROM HLLW Kensuke Kinoshita, Masaki Kurata, Koichi Uozumi, and Tadashi Inoue Central Research Institute of Electric Power
The Effect of Reducing Agents on Electroless Copper Plating Process
 Universities Research Journal 2011, Vol. 4, No. 3 The Effect of Reducing Agents on Electroless Copper Plating Process May Zin Oo 1, Sandar Tun 2 and Kyaw Myo Naing 3 Abstract This work is aimed mainly
Universities Research Journal 2011, Vol. 4, No. 3 The Effect of Reducing Agents on Electroless Copper Plating Process May Zin Oo 1, Sandar Tun 2 and Kyaw Myo Naing 3 Abstract This work is aimed mainly
