Solubility Worksheet #2 Level 3
|
|
|
- Alaina Hunt
- 5 years ago
- Views:
Transcription
1 Name Date We have considered some conditions that allow solutes to dissolve in solvents, but we have not yet considered the solubility of a solute. Solubility is defined as the maximum mass of solute that can dissolve in a certain quantity of solvent at a specified temperature. Below is a data table that shows the solubility of ammonium chloride (NH 4 Cl) at various temperatures. Use the data table to construct a line graph. Remember to set up your axes and label appropriately. Water Temperature ( C) Maximum Mass of Ammonium Chloride that can be dissolved in a 100 grams of water (g) We want to know how many grams of ammonium chloride can dissolve in water at various temperatures. In this case, what are the independent and dependent variables? Independent variable Dependent variable 2. Label your x and y axes. 3. An appropriate scale and increments should be chosen. The x values range from The y values range from You may use increments of 5, 10, 20 or whatever you think is appropriate. Remember that every data point we have does not have to fall exactly on one of your numbered lines. 4. Plot the data points and draw s best-fit line through the data points. 5. Add a title to the graph 1
2 2
3 Analysis 1. Use your best fit line to determine how many grams of ammonium chloride will dissolve at a temperature of 70 C grams 2. What temperature would be necessary to dissolve 50 grams of ammonium chloride? C 3. As the temperature increases from 20 C to 100 C on your best-fit line, the mass of the solute dissolved in grams increases by. 4. The unit rate of change is the change in the maximum amount of ammonium chloride in grams that can be dissolved for every 1 C change in temperature. Rate of Change= Δ Grams of Solute Dissolved Δ Temperature Use the data table to calculate the unit rate of change in dissolved ammonium chloride between 20 C and 100 C. Rate of Change= (grams at 100 C grams at 20 C) = = (100 C - 20 C) 5. You can also calculate the unit rate of change using the graph by calculating Unit Rate of Change (slope) = Δ y (grams) Δ x ( C) Pick an ordered pair on your best-fit line and calculate the unit rate of change. Use the chart on the next page. 3
4 Ordered Pair used for calculation (x 1, y 1 ) (x 2, y 2 ) Δ grams of solute dissolved Δ y Δ temperature ( C) Δ x Unit Rate of Change (slope) Δ y/δ x 6. Use the information from the data table to calculate the rate of change between 20 C and 40 C. Show formula and work. 7. Use the information from the data table to calculate the rate of change between 60 C and 80 C. Show formula and work. 8. Are your calculated rates of change for the two temperature ranges (20 C 40 C) and (60 C - 80 C) the same or different? What does this suggest? 4
5 9. How many grams of ammonium chloride can dissolve in 100 ml of water at 0 C?. On your graph where does your line cross the Y-axis?. This is the Y-intercept. 10. The general equation for a line is y= mx+b when m represents the unit rate of change (slope) and b represents the y-intercept. What is the equation for your line? 11. Based on the equation above fill in the chart below to predict the amount of solute dissolved at various temperatures. *Use your graph to check your work for the first two. x (Temperature in C) y (Solute in grams) Show your calculations. 5
6 12. Suppose you tested a different salt (for example potassium nitrate) instead of ammonium chloride and found that more of that salt could dissolved in 100ml of water at 50 C. Does that necessarily mean that more potassium nitrate would dissolve in 100 ml of water at 10 C?. Explain your answer. 13. Suppose you tested a different salt (for example potassium chloride) and found that less of that salt could dissolved in 100ml of water at every temperature tested. Add a line on your graph representing the solubility curve of this salt. (Use a different color.) Is the line representing the solubility curve of this salt necessarily parallel to that of ammonium chloride? Explain your answer. 6
MiSP Insolation Worksheet #1
 MiSP Insolation Worksheet #1 Name Date Insolation Absorption and Radiation by Land and Water (L 3) Introduction The word insolation comes from three words: incoming solar radiation. Insolation is the electromagnetic
MiSP Insolation Worksheet #1 Name Date Insolation Absorption and Radiation by Land and Water (L 3) Introduction The word insolation comes from three words: incoming solar radiation. Insolation is the electromagnetic
Solutions and Crystallisation exam question
 Solutions and Crystallisation exam question 2012 - Higher An experiment was performed to investigate the effect of temperature on the solubility of carbon dioxide in water. The data obtained from this
Solutions and Crystallisation exam question 2012 - Higher An experiment was performed to investigate the effect of temperature on the solubility of carbon dioxide in water. The data obtained from this
Quiz 2 practice Quiz
 Name: ate: 1. The following data were recorded while determining the solubility of a certain salt. Temp. ( C) 10 20 30 40 50 Grams Solute/ 100 g H 2 O 30 33 36 39 42 Which graph best represents the solubility
Name: ate: 1. The following data were recorded while determining the solubility of a certain salt. Temp. ( C) 10 20 30 40 50 Grams Solute/ 100 g H 2 O 30 33 36 39 42 Which graph best represents the solubility
M. It is expressed in units such a g/cc,
 Experiment 2 Density Part I: Density of a solid Density is defined as mass per unit volume, D = V M. It is expressed in units such a g/cc, g/ml, lb/ft 3, etc. Determination of mass is done by weighing
Experiment 2 Density Part I: Density of a solid Density is defined as mass per unit volume, D = V M. It is expressed in units such a g/cc, g/ml, lb/ft 3, etc. Determination of mass is done by weighing
Double Award Science: Chemistry
 New Specification Centre Number Candidate Number General Certificate of Secondary Education 2014 2015 Double Award Science: Chemistry Unit C1 Higher Tier ML [GSD22] WEDNESDAY 25 FEBRUARY 2015, MORNING
New Specification Centre Number Candidate Number General Certificate of Secondary Education 2014 2015 Double Award Science: Chemistry Unit C1 Higher Tier ML [GSD22] WEDNESDAY 25 FEBRUARY 2015, MORNING
Chapter 10 Regression Analysis
 Chapter 10 Regression Analysis Goal: To become familiar with how to use Excel 2007/2010 for Correlation and Regression. Instructions: You will be using CORREL, FORECAST and Regression. CORREL and FORECAST
Chapter 10 Regression Analysis Goal: To become familiar with how to use Excel 2007/2010 for Correlation and Regression. Instructions: You will be using CORREL, FORECAST and Regression. CORREL and FORECAST
CHEMISTRY WESTMINSTER SCHOOL THE CHALLENGE Thursday 28 April Time allowed: 30 minutes. Please write in black or blue ink.
 WESTMINSTER SCHOOL THE CHLLENGE 2016 CHEMISTRY Thursday 28 pril 2016 Time allowed: 30 minutes Please write in black or blue ink. Write your answers in the spaces provided. For examiner use only Total Mark
WESTMINSTER SCHOOL THE CHLLENGE 2016 CHEMISTRY Thursday 28 pril 2016 Time allowed: 30 minutes Please write in black or blue ink. Write your answers in the spaces provided. For examiner use only Total Mark
Solutions Unit Exam Name Date Period
 Name Date Period Ms. Roman Page 1 Regents Chemistry 1. Which mixture can be separated by using the equipment shown below? 6. Which ion, when combined with chloride ions, Cl, forms an insoluble substance
Name Date Period Ms. Roman Page 1 Regents Chemistry 1. Which mixture can be separated by using the equipment shown below? 6. Which ion, when combined with chloride ions, Cl, forms an insoluble substance
Solubility Equilibrium
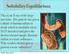 Solubility Equilibrium This is an X-ray of the large intestine. The patient was given a drink of barium sulfate to drink which is insoluble (does NOT dissolve) and gives the doctor a better image. Barium
Solubility Equilibrium This is an X-ray of the large intestine. The patient was given a drink of barium sulfate to drink which is insoluble (does NOT dissolve) and gives the doctor a better image. Barium
Quantitive Chemistry Question paper
 Quantitive Chemistry Question paper Level Subject Exam Board Topic Sub-Topic Booklet GCSE Chemistry CCEA Quantitative Chemistry Quantitive Chemistry Question paper Time Allowed: 93 minutes Score: /77 Percentage:
Quantitive Chemistry Question paper Level Subject Exam Board Topic Sub-Topic Booklet GCSE Chemistry CCEA Quantitative Chemistry Quantitive Chemistry Question paper Time Allowed: 93 minutes Score: /77 Percentage:
function relations and domain of functions
 function relations and domain of functions Module 3 : Investigation 2 MAT 170 Precalculus September 7, 2016 what is a function? Recall from last time, that a function consists of 3 parts : (1) The set
function relations and domain of functions Module 3 : Investigation 2 MAT 170 Precalculus September 7, 2016 what is a function? Recall from last time, that a function consists of 3 parts : (1) The set
Experiment 18 Determination of Iron by Visible Spectrophotometry
 Experiment 18 Determination of Iron by Visible Spectrophotometry PRELAB DISCUSSION Visible spectrophotometry (spectral analysis) is a method of measuring the concentration of colored solutions by the amount
Experiment 18 Determination of Iron by Visible Spectrophotometry PRELAB DISCUSSION Visible spectrophotometry (spectral analysis) is a method of measuring the concentration of colored solutions by the amount
Econ Intermediate Microeconomic Theory College of William and Mary December 16, 2013 John Parman. Final Exam
 Econ 303 - Intermediate Microeconomic Theory College of William and Mary December 16, 2013 John Parman Final Exam You have until 3:30pm to complete the exam, be certain to use your time wisely. Answer
Econ 303 - Intermediate Microeconomic Theory College of William and Mary December 16, 2013 John Parman Final Exam You have until 3:30pm to complete the exam, be certain to use your time wisely. Answer
i. The profit maximizing output of firm B is smaller than the profit maximizing output of firm A.
 Short Questions 1. A firm is currently producing output using units of labor and units of other materials. The isoquant that corresponds to output level and the firm s optimal input choices are given in
Short Questions 1. A firm is currently producing output using units of labor and units of other materials. The isoquant that corresponds to output level and the firm s optimal input choices are given in
Consumer and Producer Surplus and Deadweight Loss
 Consumer and Producer Surplus and Deadweight Loss The deadweight loss, value of lost time or quantity waste problem requires several steps. A ceiling or floor price must be given. We call that price the
Consumer and Producer Surplus and Deadweight Loss The deadweight loss, value of lost time or quantity waste problem requires several steps. A ceiling or floor price must be given. We call that price the
) and it s ideal van t Hoff factor is 4. Note that polyatomic ions do not break up into their constituent elements.
 Freezing Point Depression: Determining CaCl2 Van t Hoff Factor Minneapolis Community and Technical College C1152 v.12.15 I. Introduction The physical properties of solutions that depend on the number of
Freezing Point Depression: Determining CaCl2 Van t Hoff Factor Minneapolis Community and Technical College C1152 v.12.15 I. Introduction The physical properties of solutions that depend on the number of
Agry 465 Exam November 17, 2004 (100 points) (8 pages)
 Agry 465 Exam November 17, 2004 (100 points) (8 pages) Name (4) 1. Tensiometers measure what in the soil? (circle all true statements) a) hydrogen concentration b) gravimetric water content c) volumetric
Agry 465 Exam November 17, 2004 (100 points) (8 pages) Name (4) 1. Tensiometers measure what in the soil? (circle all true statements) a) hydrogen concentration b) gravimetric water content c) volumetric
3. [7 points] How many significant figures should there be in the answer to the following problem?
![3. [7 points] How many significant figures should there be in the answer to the following problem? 3. [7 points] How many significant figures should there be in the answer to the following problem?](/thumbs/83/87492512.jpg) 1 of 6 10/20/2009 3:52 AM 1. [7 points] What is the correct name for ZnF 2? (a) zinc fluorine (b) zinc (II) fluoride (c) zinc fluoride (d) fluoride zinc 2. [7 points] What is the correct name for P 2 O
1 of 6 10/20/2009 3:52 AM 1. [7 points] What is the correct name for ZnF 2? (a) zinc fluorine (b) zinc (II) fluoride (c) zinc fluoride (d) fluoride zinc 2. [7 points] What is the correct name for P 2 O
Linear Cost, Revenue, Profit, Supply, and Demand
 Linear Cost, Revenue, Profit, Supply, and Demand Complete the following questions to investigate different types of linear models. Record your responses on this worksheet and the answer sheet. Turn in
Linear Cost, Revenue, Profit, Supply, and Demand Complete the following questions to investigate different types of linear models. Record your responses on this worksheet and the answer sheet. Turn in
Determination of the Mass Percentage of Copper in a Penny
 Determination of the Mass Percentage of Copper in a Penny Introduction This experiment will cost you one penny ($0.01). The penny must be minted after 1983. Any penny will do; for best results the penny
Determination of the Mass Percentage of Copper in a Penny Introduction This experiment will cost you one penny ($0.01). The penny must be minted after 1983. Any penny will do; for best results the penny
Total Grade /150 Checked by
 FIRST LETTER OF YOUR LAST NAME CHEMISTRY 1127 EXAM I NAME (PRINT) SECTION SIGNATURE TA PLEASE READ THE FOLLOWING INSTRUCTIONS Do NOT begin the exam until asked to do so. There are 8 numbered pages, a useful
FIRST LETTER OF YOUR LAST NAME CHEMISTRY 1127 EXAM I NAME (PRINT) SECTION SIGNATURE TA PLEASE READ THE FOLLOWING INSTRUCTIONS Do NOT begin the exam until asked to do so. There are 8 numbered pages, a useful
Lecture 11. Solubility of Ionic Compounds in Water
 Lecture 11 Solubility of Ionic Compounds in Water Soluble Compounds Exceptions Acetates, Nitrates, Chlorates Na +, K +, NH 4 + compounds None None Halides ( Cl -, Br -, I - ) Ag +, Hg 2 2+, Cu +, Pb 2+
Lecture 11 Solubility of Ionic Compounds in Water Soluble Compounds Exceptions Acetates, Nitrates, Chlorates Na +, K +, NH 4 + compounds None None Halides ( Cl -, Br -, I - ) Ag +, Hg 2 2+, Cu +, Pb 2+
ADVANCED AP PLACEMENT CHEMISTRY. Activity Series. Introduction. Objective. Chemicals and Equipment
 ADVANCED AP PLACEMENT CHEMISTRY Introduction Activity Series An activity series of metals is a table of metals arranged in the order of their decreasing chemical activity or the ease at which the metal
ADVANCED AP PLACEMENT CHEMISTRY Introduction Activity Series An activity series of metals is a table of metals arranged in the order of their decreasing chemical activity or the ease at which the metal
Determining the Density of Unknown Substances
 Determining the Density of Unknown Substances Introduction: Most of the elements on the periodic table are metals and solids. These elements have observable properties that make it possible to identify
Determining the Density of Unknown Substances Introduction: Most of the elements on the periodic table are metals and solids. These elements have observable properties that make it possible to identify
Duncan. UNIT 8 - Chemical Equations BALANCING EQUATIONS PRACTICE WORKSHEET 14.) C2H6 + O2 CO2 + H2O. 2.) Na + I2 NaI 3.) N2 + O2 N2O 4.
 BALANCING EQUATIONS PRACTICE WORKSHEET 1.) CH4 + O2 CO2 + H2O 2.) Na + I2 NaI 3.) N2 + O2 N2O 4.) N2 + H2 NH3 5.) KI + Cl2 KCl + I2 6.) HCl + Ca(OH)2 CaCl2 + H2O 7.) KClO3 KCl + O2 8.) K3PO4 + HCl KCl
BALANCING EQUATIONS PRACTICE WORKSHEET 1.) CH4 + O2 CO2 + H2O 2.) Na + I2 NaI 3.) N2 + O2 N2O 4.) N2 + H2 NH3 5.) KI + Cl2 KCl + I2 6.) HCl + Ca(OH)2 CaCl2 + H2O 7.) KClO3 KCl + O2 8.) K3PO4 + HCl KCl
Look at the measuring cylinders. What happened to the volume of the water and the wax after freezing? the volume of water... the volume of wax...
 1. Meera poured 7 cm 3 of water into a measuring cylinder. She poured 7 cm 3 of melted wax into another measuring cylinder. She put both measuring cylinders into a freezer for 24 hours. water before freezing
1. Meera poured 7 cm 3 of water into a measuring cylinder. She poured 7 cm 3 of melted wax into another measuring cylinder. She put both measuring cylinders into a freezer for 24 hours. water before freezing
LEIBNIZ SHIFTS IN DEMAND AND SUPPLY
 8.6.1 SHIFTS IN DEMAND AND SUPPLY The competitive equilibrium price and quantity lie at the point where the supply and demand curves cross. If a shock occurs that shifts one of the curves, the equilibrium
8.6.1 SHIFTS IN DEMAND AND SUPPLY The competitive equilibrium price and quantity lie at the point where the supply and demand curves cross. If a shock occurs that shifts one of the curves, the equilibrium
Quadratic Regressions Group Acitivity 2 Business Project Week #4
 Quadratic Regressions Group Acitivity 2 Business Project Week #4 In activity 1 we created a scatter plot on the calculator using a table of values that were given. Some of you were able to create a linear
Quadratic Regressions Group Acitivity 2 Business Project Week #4 In activity 1 we created a scatter plot on the calculator using a table of values that were given. Some of you were able to create a linear
Experimental Procedure. Lab 402
 Experimental Procedure Lab 402 Procedure Overview Cyclohexane is the solvent selected for this experiment although other solvents may be just as effective for the determination of the molar mass of a solute.
Experimental Procedure Lab 402 Procedure Overview Cyclohexane is the solvent selected for this experiment although other solvents may be just as effective for the determination of the molar mass of a solute.
IGCSE(A*-G) Edexcel - Chemistry
 IGCSE(A*-G) Edexcel - Chemistry Principles of Chemistry States of Matter NOTES 1.1 Understand the arrangement, movement and energy of the particles in each of the three states of matter: solid, liquid
IGCSE(A*-G) Edexcel - Chemistry Principles of Chemistry States of Matter NOTES 1.1 Understand the arrangement, movement and energy of the particles in each of the three states of matter: solid, liquid
BORABU-MASABA DISTRICTS JOINT EVALUATION TEST 2012 Kenya Certificate of Secondary Education (K.C.S.E)
 Name. School Candidate s Signature. Index No /. Date. 233/2 CHEMISTRY Paper 2 (Theory) JULY / AUGUST - 2012 Time: 2 Hours BORABU-MASABA DISTRICTS JOINT EVALUATION TEST 2012 Kenya Certificate of Secondary
Name. School Candidate s Signature. Index No /. Date. 233/2 CHEMISTRY Paper 2 (Theory) JULY / AUGUST - 2012 Time: 2 Hours BORABU-MASABA DISTRICTS JOINT EVALUATION TEST 2012 Kenya Certificate of Secondary
Reliability vs. Validity
 Reliability vs. Validity Reliability (precision) is how _ the results are. Is there a large enough sample size? Are the measuring procedures accurate? Etc. Validity (accuracy) is whether the data measures.
Reliability vs. Validity Reliability (precision) is how _ the results are. Is there a large enough sample size? Are the measuring procedures accurate? Etc. Validity (accuracy) is whether the data measures.
Microeconomic Theory -1- Introduction and maximization
 Microeconomic Theory -- Introduction and maximization Introduction Maximization. Profit maximizing firm with monopoly power 6. General results on maximizing with two variables 3. Non-negativity constraints
Microeconomic Theory -- Introduction and maximization Introduction Maximization. Profit maximizing firm with monopoly power 6. General results on maximizing with two variables 3. Non-negativity constraints
General Oceanography Geology 105 Expedition 12 - The Briny Deep
 General Oceanography Geology 105 Expedition 12 - The Briny Deep Name Not attempting to answer questions on expeditions will result in point deductions on course workbook (two or more blank answers will
General Oceanography Geology 105 Expedition 12 - The Briny Deep Name Not attempting to answer questions on expeditions will result in point deductions on course workbook (two or more blank answers will
1. What volume of water is required to make a 4.65 M solution from 5.2 g of NaBr (MM = g/mol)?
 CHEMISTRY 110 EXAM II Answer Key March 22, 2013 1. What volume of water is required to make a 4.65 M solution from 5.2 g of NaBr (MM = 102.894 g/mol)? A. 4.65 ml B. 10.9 ml C. 11 ml D. 235 ml E. 240 ml
CHEMISTRY 110 EXAM II Answer Key March 22, 2013 1. What volume of water is required to make a 4.65 M solution from 5.2 g of NaBr (MM = 102.894 g/mol)? A. 4.65 ml B. 10.9 ml C. 11 ml D. 235 ml E. 240 ml
Salinity in Seawater
 Salinity in Seawater Objective To familiarize students with the different methods used for measuring salinity of water. Introduction: Salinity exerts profound impacts on the marine environment. It controls
Salinity in Seawater Objective To familiarize students with the different methods used for measuring salinity of water. Introduction: Salinity exerts profound impacts on the marine environment. It controls
Think about the connections among graphs, data patterns, function rules, and problem conditions for linear relationships.
 Unit 2 Lesson 1 Investigation 1 Pay for Soliciting Credit Card Customers Name: Think About This Situation Think about the connections among graphs, data patterns, function rules, and problem conditions
Unit 2 Lesson 1 Investigation 1 Pay for Soliciting Credit Card Customers Name: Think About This Situation Think about the connections among graphs, data patterns, function rules, and problem conditions
Statistics: Data Analysis and Presentation. Fr Clinic II
 Statistics: Data Analysis and Presentation Fr Clinic II Overview Tables and Graphs Populations and Samples Mean, Median, and Standard Deviation Standard Error & 95% Confidence Interval (CI) Error Bars
Statistics: Data Analysis and Presentation Fr Clinic II Overview Tables and Graphs Populations and Samples Mean, Median, and Standard Deviation Standard Error & 95% Confidence Interval (CI) Error Bars
19. H, S, C, and G Diagrams Module
 HSC - HSC Diagrams 15012-ORC-J 1 (8) 19. H, S, C, and G Diagrams Module The diagram module presents the basic thermochemical data for the given species in graphical format. Eight different diagram types
HSC - HSC Diagrams 15012-ORC-J 1 (8) 19. H, S, C, and G Diagrams Module The diagram module presents the basic thermochemical data for the given species in graphical format. Eight different diagram types
Materials Required for Lab Report Resubmissions: Original graded report with all required components
 Appendix III: What to Turn in for Each Lab and Resubmission Policies Lab Report Guidelines All lab reports are due in lecture. Graded reports will be handed back in the laboratory section. If you miss
Appendix III: What to Turn in for Each Lab and Resubmission Policies Lab Report Guidelines All lab reports are due in lecture. Graded reports will be handed back in the laboratory section. If you miss
INTERDISCIPLINARY INVESTIGATION (IDI)-LAB LABORATORY HANDOUT
 INTERDISCIPLINARY INVESTIGATION (IDI)-LAB LABORATORY HANDOUT Research-based Interdisciplinary Science Education ION CHROMATOGRAPHY TO INVESTIGATE WATER QUALITY ISSUES: FLUORIDE IN DRINKING WATER Introduction
INTERDISCIPLINARY INVESTIGATION (IDI)-LAB LABORATORY HANDOUT Research-based Interdisciplinary Science Education ION CHROMATOGRAPHY TO INVESTIGATE WATER QUALITY ISSUES: FLUORIDE IN DRINKING WATER Introduction
CH241 Experiment #1 (Weeks of September 11, 18, and 25, 2017)
 CH241 Experiment #1 (Weeks of September 11, 18, and 25, 2017) SEPARATION AND RECOVERY OF ORGANIC COMPOUNDS, THIN LAYER CHROMATOGRAPHY, COLUMN CHROMATOGRAPHY, CRYSTALLIZATION, AND MELTING POINTS Overview
CH241 Experiment #1 (Weeks of September 11, 18, and 25, 2017) SEPARATION AND RECOVERY OF ORGANIC COMPOUNDS, THIN LAYER CHROMATOGRAPHY, COLUMN CHROMATOGRAPHY, CRYSTALLIZATION, AND MELTING POINTS Overview
Revised Molar Mass Measurement Lab Purpose: Background:
 Revised Molar Mass Measurement Lab (Based on prior Ch b experiment, incorporating revision suggestions from Tsze Tsang) Purpose: To use the colligative properties of cyclohexane solutions to determine
Revised Molar Mass Measurement Lab (Based on prior Ch b experiment, incorporating revision suggestions from Tsze Tsang) Purpose: To use the colligative properties of cyclohexane solutions to determine
Kinetic vs. Thermodynamic Control
 Experiment: Kinetic vs. Thermodynamic Control of rganic Reactions Kinetic vs. Thermodynamic Control During your study of reactions this year you have examined many mechanisms. You have used these mechanisms
Experiment: Kinetic vs. Thermodynamic Control of rganic Reactions Kinetic vs. Thermodynamic Control During your study of reactions this year you have examined many mechanisms. You have used these mechanisms
1. The equation that represents the equilibrium in a saturated solution of Fe 2 (SO 4 ) 3 is.. The precipitate which forms first is
 CHEMISTRY 12 UNIT 3 - REVIEW Ksp Part A - Multiple Choice 1. The equation that represents the equilibrium in a saturated solution of Fe 2 (SO 4 ) 3 is A. Fe 2 (SO 4 ) 3 (s) 3 Fe 2+ (aq) + 2 SO 4 3 (aq)
CHEMISTRY 12 UNIT 3 - REVIEW Ksp Part A - Multiple Choice 1. The equation that represents the equilibrium in a saturated solution of Fe 2 (SO 4 ) 3 is A. Fe 2 (SO 4 ) 3 (s) 3 Fe 2+ (aq) + 2 SO 4 3 (aq)
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS
 DETERMINING THE DENSITY OF LIQUIDS & SOLIDS Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter. Density
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter. Density
THE MOLECULAR WEIGHT OF A SOLUTE*
 Name Per THE MOLECULAR WEIGHT OF A SOLUTE* One of the most helpful things that can be known about an element or compound is its molecular weight (molecular mass, gram molecular mass, atomic mass, or gram
Name Per THE MOLECULAR WEIGHT OF A SOLUTE* One of the most helpful things that can be known about an element or compound is its molecular weight (molecular mass, gram molecular mass, atomic mass, or gram
WATER QUALITY ASSESSMENT AND REPORTING
 - 3.7 - WATER QUALITY ASSESSMENT AND REPORTING 1 3.7. WATER QUALITY ASSESSMENT AND REPORTING Diederik Rousseau UNESCO-IHE Institute for Water Education Online Module Water Quality Assessment 2 CONTENTS
- 3.7 - WATER QUALITY ASSESSMENT AND REPORTING 1 3.7. WATER QUALITY ASSESSMENT AND REPORTING Diederik Rousseau UNESCO-IHE Institute for Water Education Online Module Water Quality Assessment 2 CONTENTS
(3) The compound boron nitride (BN) has a high melting point (2967 ºC), high density, and is very hard. What is the best classification of this solid?
 Solids and Liquids Name: Period: (1) Identify the type of solid formed by each compound. (a) Ag (b) CO 2 (c) SiO 2 (d) wax (e) MgCl 2 (f) Fe (g) graphite (h) SO 2 (i) CaCO 3 (j) I 2 (k) rubber (l) SiC
Solids and Liquids Name: Period: (1) Identify the type of solid formed by each compound. (a) Ag (b) CO 2 (c) SiO 2 (d) wax (e) MgCl 2 (f) Fe (g) graphite (h) SO 2 (i) CaCO 3 (j) I 2 (k) rubber (l) SiC
It is not essential for students to conduct an investigation for every question they generate or generate questions based on prior investigations.
 Scientific Inquiry 5-1 The student will demonstrate an understanding of scientific inquiry, including the foundations of technological design and the processes, skills, and mathematical thinking necessary
Scientific Inquiry 5-1 The student will demonstrate an understanding of scientific inquiry, including the foundations of technological design and the processes, skills, and mathematical thinking necessary
Answers to the Take-Home Midterm Examination
 Answers to the Take-Home Midterm Examination Econ 111s Spring/Summer 2009 Economics Department, Queen s University Instructor: Jean-Denis Garon Posted: June 19 Here are the main elements of the answers.
Answers to the Take-Home Midterm Examination Econ 111s Spring/Summer 2009 Economics Department, Queen s University Instructor: Jean-Denis Garon Posted: June 19 Here are the main elements of the answers.
Section Class.notebook. June 06, Nov 26 5:13 PM. Sep 23 11:05 AM. Nov 26 5:20 PM. Nov 26 5:22 PM. Nov 26 5:20 PM.
 Our objective is to solve problems such as... This is a system of equations. Our goal is to find values of x and y that make both equations true! ) My son, Jimmy, takes his coin jar into the bank. He pours
Our objective is to solve problems such as... This is a system of equations. Our goal is to find values of x and y that make both equations true! ) My son, Jimmy, takes his coin jar into the bank. He pours
LAB II CRYSTAL STRUCTURE AND CRYSTAL GROWTH PART 1: CRYSTAL GROWTH. I. Introduction
 LAB II CRYSTAL STRUCTURE AND CRYSTAL GROWTH This lab will be divided into two parts. In the first part, you will be growing crystals from a seed crystal in a very visual demonstration of heterogeneous
LAB II CRYSTAL STRUCTURE AND CRYSTAL GROWTH This lab will be divided into two parts. In the first part, you will be growing crystals from a seed crystal in a very visual demonstration of heterogeneous
Intermediate Microeconomics 301 Problem Set # 2 Due Wednesday June 29, 2005
 Intermediate Microeconomics 301 Problem Set # 2 Due Wednesday June 29, 2005 1. A new chemical cleaning solution is introduced to the market. Initially, demand is Q D = 100 + 2p p 2 and supply is Q S =
Intermediate Microeconomics 301 Problem Set # 2 Due Wednesday June 29, 2005 1. A new chemical cleaning solution is introduced to the market. Initially, demand is Q D = 100 + 2p p 2 and supply is Q S =
AGEC 3333 Practice Questions for Exam 2 Fall Semester, 2009 Set A: Material From Chapter 2 (55 56) & Chapter 3
 The charts below show supply and demand curves. Solid lines are original supply and demand curves, and dotted lines are changes in supply and demand. Use the chart below to answer 1 4. (1) In the chart
The charts below show supply and demand curves. Solid lines are original supply and demand curves, and dotted lines are changes in supply and demand. Use the chart below to answer 1 4. (1) In the chart
WET ANALYSIS OF GOLD-SILVER ALLOYS OF HIGH GOLD CONTENT 1
 WET ANALYSIS OF GOLD-SILVER ALLOYS OF HIGH GOLD CONTENT EARLE R. CALEY AND LOWELL W. SHANK Department of Chemistry, The Ohio State University, Columbus, Ohio ABSTRACT -silver alloys dissolve completely
WET ANALYSIS OF GOLD-SILVER ALLOYS OF HIGH GOLD CONTENT EARLE R. CALEY AND LOWELL W. SHANK Department of Chemistry, The Ohio State University, Columbus, Ohio ABSTRACT -silver alloys dissolve completely
Department of Chemistry University of Texas at Austin
 Colligative Properties Supplemental Worksheet PROBLEM #1: Give the ecular formula, the van t hoff factor for the following Ionic Compounds as well as guess the solubility of the compounds. If you cannot
Colligative Properties Supplemental Worksheet PROBLEM #1: Give the ecular formula, the van t hoff factor for the following Ionic Compounds as well as guess the solubility of the compounds. If you cannot
Lab 4: Recrystallization
 Lab 4: Recrystallization Pre Lab Question: (Answer submitted in a separate piece of paper at the beginning of lab) 1. Calculate how much 95% ethanol will be required to dissolve 0.8 g of sulfanilamide
Lab 4: Recrystallization Pre Lab Question: (Answer submitted in a separate piece of paper at the beginning of lab) 1. Calculate how much 95% ethanol will be required to dissolve 0.8 g of sulfanilamide
Econ 2113: Principles of Microeconomics. Spring 2009 ECU
 Econ 2113: Principles of Microeconomics Spring 2009 ECU Two Big Economic Questions Two big questions summarize the scope of economics: 1. How society determines what, how, and for whom goods and services
Econ 2113: Principles of Microeconomics Spring 2009 ECU Two Big Economic Questions Two big questions summarize the scope of economics: 1. How society determines what, how, and for whom goods and services
» Please take out your periodic table.
 » Please take out your periodic table. Ionic Compounds Before we begin What are ions? How are they made? (Hint: Remember what happens to VALENCE electrons.) (Put a line down the middle of the white board.)
» Please take out your periodic table. Ionic Compounds Before we begin What are ions? How are they made? (Hint: Remember what happens to VALENCE electrons.) (Put a line down the middle of the white board.)
Chapter 2 Production Possibilities, Opportunity Cost,
 Chapter 2 Production Possibilities, Opportunity Cost, and Economic Growth CHAPTER IN A NUTSHELL In this chapter, you continue your quest to learn the economic way of thinking. The chapter begins with the
Chapter 2 Production Possibilities, Opportunity Cost, and Economic Growth CHAPTER IN A NUTSHELL In this chapter, you continue your quest to learn the economic way of thinking. The chapter begins with the
Quantity Retail Price Marketing Cost Farm Price. Doz/cap/yr. $/doz. $/doz. $/doz. 17 $1.30 $0.50 $ $1.20 $0.50 $ $1.10 $0.50 $0.
 Derived Demand The basic idea of derived demand is that farmers don t experience retail demand, but instead they experience farm-level demand which is the demand for farm products by the marketing sector,
Derived Demand The basic idea of derived demand is that farmers don t experience retail demand, but instead they experience farm-level demand which is the demand for farm products by the marketing sector,
MINISTRY OF EDUCATION AND HUMAN RESOURCES, TERTIARY EDUCATION AND SCIENTIFIC RESEARCH MAURITIUS EXAMINATIONS SYNDICATE. CHEMISTRY OCTOBER hour
 MINISTRY OF EDUCATION AND HUMAN RESOURCES, TERTIARY EDUCATION AND SCIENTIFIC RESEARCH MAURITIUS EXAMINATIONS SYNDICATE CANDIDATE NAME SCHOOL NAME CLASS/SECTION NATIONAL ASSESSMENT AT FORM III CHEMISTRY
MINISTRY OF EDUCATION AND HUMAN RESOURCES, TERTIARY EDUCATION AND SCIENTIFIC RESEARCH MAURITIUS EXAMINATIONS SYNDICATE CANDIDATE NAME SCHOOL NAME CLASS/SECTION NATIONAL ASSESSMENT AT FORM III CHEMISTRY
ICSE-Science 2 (Chemistry) 2000
 ICSE-Science 2 (Chemistry) 2000 Answers to this Paper must be written on the paper provided separately. You will not be allowed to write during the first 15 minutes. This time is to be spent in reading
ICSE-Science 2 (Chemistry) 2000 Answers to this Paper must be written on the paper provided separately. You will not be allowed to write during the first 15 minutes. This time is to be spent in reading
Does Myoglobin Unfold in the Body? 1
 Does Myoglobin Unfold in the Body? 1 Myoglobin is a protein found in muscles that binds oxygen when in the proper conformation. In the denatured (unfolded) form, myoglobin cannot bind oxygen. Our body
Does Myoglobin Unfold in the Body? 1 Myoglobin is a protein found in muscles that binds oxygen when in the proper conformation. In the denatured (unfolded) form, myoglobin cannot bind oxygen. Our body
19. H, S, C, and G Diagrams Module
 HSC 8 - HSC Diagrams November 5, 4 43-ORC-J (8 9. H, S, C, and G Diagrams Module The diagram module presents the basic thermochemical data for the given species in graphical format. Eight different diagram
HSC 8 - HSC Diagrams November 5, 4 43-ORC-J (8 9. H, S, C, and G Diagrams Module The diagram module presents the basic thermochemical data for the given species in graphical format. Eight different diagram
Approved for NPDES (Editorial Revision 1978) Silica, Dissolved (Colorimetric)
 METHOD #: 370.1 TITLE: Approved for NPDES (Editorial Revision 1978) Silica, Dissolved (Colorimetric) ANALYTE: Silica, SiO 2 INSTRUMENTATION: Spectrophotometer STORET No. Dissolved 00955 1.0 Scope and Application
METHOD #: 370.1 TITLE: Approved for NPDES (Editorial Revision 1978) Silica, Dissolved (Colorimetric) ANALYTE: Silica, SiO 2 INSTRUMENTATION: Spectrophotometer STORET No. Dissolved 00955 1.0 Scope and Application
Phase diagram of carbon. Lecture 10. Simple eutectic systems. Greek, Eutektos easily melting Eu good, well + tekein to melt
 Phase diagram of carbon Lecture 10 Simple eutectic systems Greek, Eutektos easily melting Eu good, well + tekein to melt Motivation: A two component system is the simplest of multicomponent systems. A
Phase diagram of carbon Lecture 10 Simple eutectic systems Greek, Eutektos easily melting Eu good, well + tekein to melt Motivation: A two component system is the simplest of multicomponent systems. A
Laboratory Demonstration No. 1
 Laboratory Demonstration No. 1 Purpose: 1. to investigate the role of volume and flow rate in the cleansing of a contaminated water body 2. to determine the residence time 3. to apply the residence time
Laboratory Demonstration No. 1 Purpose: 1. to investigate the role of volume and flow rate in the cleansing of a contaminated water body 2. to determine the residence time 3. to apply the residence time
1.4 Applications of Functions to Economics
 CHAPTER 1. FUNCTIONS AND CHANGE 18 1.4 Applications of Functions to Economics Definition. The cost function gives the total cost of producing a quantity of some good. The standard notation is: q = quantity,
CHAPTER 1. FUNCTIONS AND CHANGE 18 1.4 Applications of Functions to Economics Definition. The cost function gives the total cost of producing a quantity of some good. The standard notation is: q = quantity,
Enzyme kinetics. Irreversible inhibition inhibitor is bound tightly to enzyme - very slow dissociation can be covalent or non covalently bound
 Enzymes can be regulated by acceleration and inhibition inhibition very common - several different mechanisms competitive / non competitive reversible / irreversible Irreversible inhibition inhibitor is
Enzymes can be regulated by acceleration and inhibition inhibition very common - several different mechanisms competitive / non competitive reversible / irreversible Irreversible inhibition inhibitor is
CRYSTAL LATTICE. Defining lattice: Mathematical construct; ideally infinite arrangement of points in space.
 CRYSTAL LATTICE How to form a crystal? 1. Define the structure of the lattice 2. Define the lattice constant 3. Define the basis Defining lattice: Mathematical construct; ideally infinite arrangement of
CRYSTAL LATTICE How to form a crystal? 1. Define the structure of the lattice 2. Define the lattice constant 3. Define the basis Defining lattice: Mathematical construct; ideally infinite arrangement of
Determination of the Molar Mass of a Compound by Freezing Point Depression
 Determination of the Molar Mass of a Compound by Freezing Point Depression Objective: The objective of this experiment is to determine the molar mass of an unknown solute by measuring the freezing point
Determination of the Molar Mass of a Compound by Freezing Point Depression Objective: The objective of this experiment is to determine the molar mass of an unknown solute by measuring the freezing point
CHAPTER 4, SECTION 1
 DAILY LECTURE CHAPTER 4, SECTION 1 Understanding Demand What Is Demand? Demand is the willingness and ability of buyers to purchase different quantities of a good, at different prices, during a specific
DAILY LECTURE CHAPTER 4, SECTION 1 Understanding Demand What Is Demand? Demand is the willingness and ability of buyers to purchase different quantities of a good, at different prices, during a specific
Experiment 13: Determination of Molecular Weight by Freezing Point Depression
 1 Experiment 13: Determination of Molecular Weight by Freezing Point Depression Objective: In this experiment, you will determine the molecular weight of a compound by measuring the freezing point of a
1 Experiment 13: Determination of Molecular Weight by Freezing Point Depression Objective: In this experiment, you will determine the molecular weight of a compound by measuring the freezing point of a
Lab 4: Recrystallization
 Lab 4: Recrystallization Objectives: - Purify an impure sample of an antibiotic. - Practice the crystallization technique. Introduction: The purpose of this experiment is to introduce the technique of
Lab 4: Recrystallization Objectives: - Purify an impure sample of an antibiotic. - Practice the crystallization technique. Introduction: The purpose of this experiment is to introduce the technique of
EXAMINATION #2 VERSION A Consumers and Demand September 28, 2017
 William M. Boal Signature: Printed name: EXAMINATION #2 VERSION A Consumers and Demand September 28, 2017 INSTRUCTIONS: This exam is closed-book, closed-notes. Calculators, mobile phones, and wireless
William M. Boal Signature: Printed name: EXAMINATION #2 VERSION A Consumers and Demand September 28, 2017 INSTRUCTIONS: This exam is closed-book, closed-notes. Calculators, mobile phones, and wireless
Clever crystals and solubility
 19 Clever crystals and solubility Bunsen Burner Badge car 20 Clever crystals and solubility Suitable for: 11 14 years Curriculum and learning links: States of matter, separating mixtures Learning objectives:
19 Clever crystals and solubility Bunsen Burner Badge car 20 Clever crystals and solubility Suitable for: 11 14 years Curriculum and learning links: States of matter, separating mixtures Learning objectives:
Chapter 2 - Introduction to Spreadsheet Modeling
 1. Which of the following is not one of the components of a mathematical model? a. Inputs b. Outputs c. Decision variables d. None of these options 2. Which of the following is not one of the features
1. Which of the following is not one of the components of a mathematical model? a. Inputs b. Outputs c. Decision variables d. None of these options 2. Which of the following is not one of the features
EXAMINATION 1 VERSION A "Labor Supply and Demand" February 27, 2018
 William M. Boal Signature: Printed name: EXAMINATION 1 VERSION A "Labor Supply and Demand" February 27, 2018 INSTRUCTIONS: This exam is closed-book, closed-notes. Simple calculators are permitted, but
William M. Boal Signature: Printed name: EXAMINATION 1 VERSION A "Labor Supply and Demand" February 27, 2018 INSTRUCTIONS: This exam is closed-book, closed-notes. Simple calculators are permitted, but
Gypsum Solubility Simulating Real World Aqueous Problems
 Simulating Real World Aqueous Problems or Getting an edge on the competition! Reliable simulation of complex processes can provide insights for process optimization and reduced operating costs. The solubility
Simulating Real World Aqueous Problems or Getting an edge on the competition! Reliable simulation of complex processes can provide insights for process optimization and reduced operating costs. The solubility
Year 7 Chemistry HW Questions
 Year 7 Chemistry HW Questions 37 minutes 56 marks Page 1 of 15 Q1. Molly used a ph sensor to test different liquids. She dipped the probe of the sensor into each liquid and recorded the ph value in a table.
Year 7 Chemistry HW Questions 37 minutes 56 marks Page 1 of 15 Q1. Molly used a ph sensor to test different liquids. She dipped the probe of the sensor into each liquid and recorded the ph value in a table.
CHM Gravimetric Chloride Experiment (r7) 1/5
 CHM 111 - Gravimetric Chloride Experiment (r7) 1/5 Purpose You will perform one of the basic types of quantitative analysis - the gravimetric analysis. You will be asked to determine the percentage of
CHM 111 - Gravimetric Chloride Experiment (r7) 1/5 Purpose You will perform one of the basic types of quantitative analysis - the gravimetric analysis. You will be asked to determine the percentage of
Lecture 3: Section 1.2 Linear Functions and Applications
 L3-1 Lecture 3: Section 1.2 Linear Functions and Applications In Lecture 2, we explored linear equations of the form Ax + By = C. Solving for y, we have the equivalent form y = mx + b. This is a functional
L3-1 Lecture 3: Section 1.2 Linear Functions and Applications In Lecture 2, we explored linear equations of the form Ax + By = C. Solving for y, we have the equivalent form y = mx + b. This is a functional
Q1. From the following list of substances, choose the substances which meet the description given in parts (i) to (v) below :
 Questions:- Q1. From the following list of substances, choose the substances which meet the description given in parts (i) to (v) below : Ammonium chloride, ammonium nitrate, chlorine, dilute hydrochloric
Questions:- Q1. From the following list of substances, choose the substances which meet the description given in parts (i) to (v) below : Ammonium chloride, ammonium nitrate, chlorine, dilute hydrochloric
Find the quadratic function with y intercept (0,4) and passes through the points (2, 10) and ( 1, 7).
 Our objective is to solve problems such as... 1) My son, Jimmy, takes his coin jar into the bank. He pours all the coins into the "coin counting" machine. It reports that he had 273 coins (nickels and
Our objective is to solve problems such as... 1) My son, Jimmy, takes his coin jar into the bank. He pours all the coins into the "coin counting" machine. It reports that he had 273 coins (nickels and
SPSS Guide Page 1 of 13
 SPSS Guide Page 1 of 13 A Guide to SPSS for Public Affairs Students This is intended as a handy how-to guide for most of what you might want to do in SPSS. First, here is what a typical data set might
SPSS Guide Page 1 of 13 A Guide to SPSS for Public Affairs Students This is intended as a handy how-to guide for most of what you might want to do in SPSS. First, here is what a typical data set might
Additional Applied Science
 Centre Number Surname Candidate Number For Examiner s Use Other Names Candidate Signature Examiner s Initials General Certificate of Secondary Education Higher Tier June 2013 Additional Applied Science
Centre Number Surname Candidate Number For Examiner s Use Other Names Candidate Signature Examiner s Initials General Certificate of Secondary Education Higher Tier June 2013 Additional Applied Science
*20GSD5201* Double Award Science: Chemistry. Unit C2 Higher Tier WEDNESDAY 15 JUNE 2016, AFTERNOON [GSD52] *GSD52* *G5802* TIME 1 hour 15 minutes.
![*20GSD5201* Double Award Science: Chemistry. Unit C2 Higher Tier WEDNESDAY 15 JUNE 2016, AFTERNOON [GSD52] *GSD52* *G5802* TIME 1 hour 15 minutes. *20GSD5201* Double Award Science: Chemistry. Unit C2 Higher Tier WEDNESDAY 15 JUNE 2016, AFTERNOON [GSD52] *GSD52* *G5802* TIME 1 hour 15 minutes.](/thumbs/84/89360002.jpg) Centre Number Candidate Number General Certificate of Secondary Education 2016 Double Award Science: Chemistry Unit C2 Higher Tier [GSD52] *GSD52* *G5802* *GSD52* WEDNESDAY 15 JUNE 2016, AFTERNOON TIME
Centre Number Candidate Number General Certificate of Secondary Education 2016 Double Award Science: Chemistry Unit C2 Higher Tier [GSD52] *GSD52* *G5802* *GSD52* WEDNESDAY 15 JUNE 2016, AFTERNOON TIME
Example: Compute the wavelength of a 1 [kg] block moving at 1000 [m/s].
![Example: Compute the wavelength of a 1 [kg] block moving at 1000 [m/s]. Example: Compute the wavelength of a 1 [kg] block moving at 1000 [m/s].](/thumbs/95/123245062.jpg) Example: Calculate the energy required to excite the hydrogen electron from level n = 1 to level n = 2. Also calculate the wavelength of light that must be absorbed by a hydrogen atom in its ground state
Example: Calculate the energy required to excite the hydrogen electron from level n = 1 to level n = 2. Also calculate the wavelength of light that must be absorbed by a hydrogen atom in its ground state
Total Cost of Materials:
 Worksheet 1: Cost Inventors have to do more than just come up with brilliant ideas. They then need to take those ideas and implement them using available money, resources, and time. Now that you have created
Worksheet 1: Cost Inventors have to do more than just come up with brilliant ideas. They then need to take those ideas and implement them using available money, resources, and time. Now that you have created
10. Validated Normal Phase HPLC Method for the Determination. Fulvestrant is primarily used in the treatment of hormone receptor
 229 10. Validated Normal Phase HPLC Method for the Determination of Fulvestrant in Pharmaceutical Dosage Forms 10.1 Introduction Fulvestrant is primarily used in the treatment of hormone receptor positive
229 10. Validated Normal Phase HPLC Method for the Determination of Fulvestrant in Pharmaceutical Dosage Forms 10.1 Introduction Fulvestrant is primarily used in the treatment of hormone receptor positive
EXPERIMENT III. Determination of Iron in Iron Oxide, (Fe 2 O 3 ), Using Dichromate Method. Chemical Overview
 EXPERIMENT III Determination of Iron in Iron Oxide, (Fe 2 O 3 ), Using Dichromate Method Chemical Overview This is a direct titration using K 2 Cr 2 O 7, a primary standard, as the titrant. As such the
EXPERIMENT III Determination of Iron in Iron Oxide, (Fe 2 O 3 ), Using Dichromate Method Chemical Overview This is a direct titration using K 2 Cr 2 O 7, a primary standard, as the titrant. As such the
SAARC Training Workshop Program Identification, Comparison and Scenario Based Application of Power Demand/ Load Forecasting Tools
 SAARC Training Workshop Program Identification, Comparison and Scenario Based Application of Power Demand/ Load Forecasting Tools Long Term Power Demand Forecasting using Regression Model Contents Growth
SAARC Training Workshop Program Identification, Comparison and Scenario Based Application of Power Demand/ Load Forecasting Tools Long Term Power Demand Forecasting using Regression Model Contents Growth
Total Dissolved Solids
 Total Dissolved Solids LabQuest 12 INTRODUCTION Solids are found in streams in two forms, suspended and dissolved. Suspended solids include silt, stirred-up bottom sediment, decaying plant matter, or sewage-treatment
Total Dissolved Solids LabQuest 12 INTRODUCTION Solids are found in streams in two forms, suspended and dissolved. Suspended solids include silt, stirred-up bottom sediment, decaying plant matter, or sewage-treatment
Exercise 6a. Balancing equations
 Exercise 6a Balancing equations Balance the following equations. To get you started _ indicates the first six questions where numbers need to be inserted to achieve the balance. In one or two difficult
Exercise 6a Balancing equations Balance the following equations. To get you started _ indicates the first six questions where numbers need to be inserted to achieve the balance. In one or two difficult
Copper Odyssey. Chemical Reactions of Copper
 Name Lab Partner(s) Copper Odyssey Chemical Reactions of Copper Date Period Elemental copper metal will be converted into copper (II) ion and then brought through a series of compound conversions until
Name Lab Partner(s) Copper Odyssey Chemical Reactions of Copper Date Period Elemental copper metal will be converted into copper (II) ion and then brought through a series of compound conversions until
Demand The Demand curve answers this question: What quantity of a good would consumers buy at each possible price?
 1 of 6 30.4.2006 20:25 DANBY IA&S 324 Demand, Supply, and Surpluses Demand, Supply, and Surpluses Demand The Demand curve answers this question: What quantity of a good would consumers buy at each possible
1 of 6 30.4.2006 20:25 DANBY IA&S 324 Demand, Supply, and Surpluses Demand, Supply, and Surpluses Demand The Demand curve answers this question: What quantity of a good would consumers buy at each possible
McCord CH302 Exam 1 Spring 2017
 112 version last name first name signature McCord CH302 Exam 1 Spring 2017 50375 / 50380 Reminder: Be sure and correctly bubble in your name, uteid, and version number on your bubblesheet. The Periodic
112 version last name first name signature McCord CH302 Exam 1 Spring 2017 50375 / 50380 Reminder: Be sure and correctly bubble in your name, uteid, and version number on your bubblesheet. The Periodic
Topic Reacting masses Level GCSE Outcomes 1. To calculate reacting masses 2. To set out mole calculations in a grid format
 Topic Reacting masses Level GCSE Outcomes 1. To calculate reacting masses 2. To set out mole calculations in a grid format Problems on Reacting Masses of Solids Section 1 1. What is the mass of magnesium
Topic Reacting masses Level GCSE Outcomes 1. To calculate reacting masses 2. To set out mole calculations in a grid format Problems on Reacting Masses of Solids Section 1 1. What is the mass of magnesium
