JAK2 p.val617phe (V617F) Mutation Status
|
|
|
- Lizbeth Rich
- 6 years ago
- Views:
Transcription
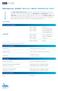
1 JAK2 p.val67phe (V67F) Mutation Status Distribution Participant ID - Date Issued - 9 October 206 Closing Date - 8 November 206 Trial Comments In this trial, there were 25 participating laboratories; 2 (98.%) of participants returned results. Sample Comments Two lyophilised samples (JAK2 43 and JAK2 44) were distributed to participants for JAK2 p.val67 Phe mutation analysis. JAK2 43 and 44 were duplicate samples formulated to be positive for the JAK2 V67F mutation. Results and Performance Your Results JAK2 Mutation Status Your Results Consensus Result Sample JAK2 43 Mutation Detected Sample JAK2 44 All Participant Results Your Performance Performance N/A = Not Applicable Mutation Detected () Mutation Detected No Mutation Detected () Sample JAK Sample JAK Performance Status for this Trial Performance Status Classification Over 3 Trial Period Satisfactory Critical Report Issue Date: 8 Jan 207 ; Distribution: JAK2 6702; Version:.0.0 Page of 6
2 Template Type JAK2 p.val67phe (V67F) Mutation Status PCR Type Protocol Type DNA 208 cdna 3 Allele Specific PCR 89 Real-Time PCR 83 Single PCR 5 Melting Curve Analysis Multiplex PCR 4 Droplet Digital PCR 3 Allele Specific Competitive Blocker PCR 2 LNA PCR 2 COLD PCR Nested PCR In-house Assay Ipsogen JAK2 MutaQuant Kit CE Ipsogen JAK2 MutaScreen Kit CE Ipsogen JAK2 MutaSearch Kit Ipsogen JAK2 RGQ PCR Kit CE AB Analitica REALQUALITY RS-JAK-2 V67F Q AmoyDx JAK2 Mutation Detection Kit Rotor-Gene Q MDx Tib Molbiol LightMix kit Report Issue Date: 8 Jan 207 ; Distribution: JAK2 6702; Version:.0.0 Page 2 of 6
3 Analysis Type JAK2 p.val67phe (V67F) Mutation Status Real-Time PCR Fluorescent Detection 30 Agarose Gel Electrophoresis 50 Capillary Electrophoresis High Resolution Melt Droplet Digital PCR Next Generation Sequencing (Ion Torrent) Pyrosequencing Mass Spectometry Restriction Enzyme Digest Sanger Sequencing Journal Reference for Assay Baxter et al (2005) Lancet; 365 (9464): Levine et al (2005) Cancer Cell 7(4): Tefferi et al Leukemia 22 (): Larsen et al (2007)BJH, 36, Jones et al (2005) Blood 06(6): Lippert et al (2006), Blood 08(6): Denys B et al (200)J Mol Diagn 2(4):52-9 Passamonti et al (2006) Blood 07 (9): Chen et al (2007) J Mol Diagn (9): Vannucchi et al (2009) 33(2): Kroger et al (2007) Blood 09(3): James et al (2006) Leukemia (2) Sidon et al (2006) Clin Chem :52(7): Goerttler et al Blood (2005) 06(8): McClure et al (2006) Leukemia 20 () Merker et al (200) J Mol Diagn 2(): Murugesan et al (2006) AM J Clin Pathol :25(4): Olsen et al (2006) Arch Pathol Lab Med;30(7): Schnittger et al (2006) Leukemia (2): Zhou et al (2004) Clin Chem 50(8): Report Issue Date: 8 Jan 207 ; Distribution: JAK2 6702; Version:.0.0 Page 3 of 6
4 JAK2 p.val67phe (V67F) Mutation Status Frequency distribution histogram showing % JAK2 mutation load in sample JAK2 43, classified by analysis method Capillary Electrophoresis Next Generation Sequencing (Ion Torrent) % JAK2 V67F Real-Time PCR Fluorescent Detection Droplet Digital PCR Pyrosequencing Mass Spectometry Frequency distribution histogram showing % JAK2 mutation load in sample JAK2 44, classified by analysis method Median = 9.3% Upper Quartile = 2.0% Lower Quartile = 6.0% High Resolution Melt Median = 9.3% Upper Quartile = 2.4% Lower Quartile = 6.0% % JAK2 V67F Capillary Electrophoresis Real-Time PCR Fluorescent Detection Pyrosequencing High Resolution Melt Next Generation Sequencing (Ion Torrent) Droplet Digital PCR Mass Spectometry Report Issue Date: 8 Jan 207 ; Distribution: JAK2 6702; Version:.0.0 Page 4 of 6
5 Trial comments JAK2 p.val67phe (V67F) Mutation Status In line with sample formulation, 207 laboratories (98.% of returning participants) detected the JAK2 p.val67phe (V67F) mutation in sample JAK2 43. In line with sample formulation, 208 laboratories (98.6% of returning participants) detected the JAK2 p.val67phe (V67F) mutation in sample JAK2 44. All three participants who didn t detect the mutation in JAK2 44 also failed to detect it in JAK2 43. Two of the four participants who didn t detect the mutation used Real-Time PCR Fluorescent Detection as their analysis method with the other two participants utilising Agarose Gel Electrophoresis. Quantification comments: Ninety-five laboratories (45.0% of returning participants) submitted quantification data for JAK2 43, mutation levels ranged from.%-26.9% (median 9.3%, inter quartile range (IQR) 6%). Ninety-six participants (45.5% of returning participants) submitted quantitative results for sample JAK2 44 with levels ranging from.%-25.4% (median 9.3%, IQR 6.4%). The median difference in mutation load reported by participants between the duplicate samples 42 and 43 was 0.7% (min 0.0%; max 9.9%). Overall the most commonly utilised quantitative methods were real time PCR (n=73), followed by agarose gel electrophoresis (n=7), capillary electrophoreses (n=5), droplet digital PCR, high resolution melt, NGS (n=3), pyrosequencing (n=2) and mass spectometry (n=). Report Issue Date: 8 Jan 207; Distribution: JAK2 6702; Version: v.0.0 Page 5 of 6 Sheffield Teaching Hospitals NHS Foundation Trust, a UKAS proficiency testing provider No. 7804, operating UK NEQAS for Leucocyte Immunophenotyping
6 JAK2 p.val67phe (V67F) Mutation Status Information with respect to compliance with standards BS EN ISO/IEC 7043: a) The proficiency testing provider for this programme is: UK NEQAS for Leucocyte Immunophenotyping Pegasus House, 4th Floor Suite 463A Glossop Road Sheffield, S0 2QD United Kingdom Tel: +44 (0) , Fax: +44 (0) nicola.rose@ukneqasli.co.uk b) The coordinators of UK NEQAS LI programmes are Prof David Barnett and Mr Liam Whitby c) Person(s) authorizing this report: Prof David Barnett, Director or Mr Liam Whitby, Operations Manager of UK NEQAS LI d) No activities in relation to this EQA exercise were subcontracted g) The UK NEQAS LI Confidentiality Policy can be found in the Quality Manual which is available by contacting the UK NEQAS LI office. Participant details, their results and their performance data remain confidential unless revealed to the relevant NQAAP when a UK participant is identified as having performance issues i) All EQA samples are prepared in accordance with strict Standard Operational Procedures by trained personnel proven to ensure homogeneity and stability. Where appropriate/possible EQA samples are tested prior to issue. Where the sample(s) issued is stabilised blood or platelets, pre and post stability testing will have proved sample suitability prior to issue l), n), o), r) & s) Please refer to the UK NEQAS LI website at for detailed information on each programme including the scoring systems applied to assess performance (for BS EN ISO/IEC 7043:200 accredited programmes only). Where a scoring system refers to the consensus result this means the result reported by the majority of participants for that trial issue. Advice on the interpretation of statistical analyses and the criteria on which performance is measured is also given. Please note that where different methods/procedures are used by different groups of participants these may be displayed within your report, but the same scoring system is applied to all participants irrespective of method/procedure used m) We do not assign values against reference materials or calibrants q) Details of the programme designs as authorized by The Steering Committee and Specialist Advisory Group can be found on our website at The proposed trial issue schedule for each programme is also available t) If you would like to discuss the outcomes of this trial issue, please contact UK NEQAS LI using the contact details provided. Alternatively, if you are unhappy with your performance classification for this trial, please find the appeals procedure at Report Issue Date: 8 Jan 207; Distribution: JAK2 6702; Version: v.0.0 Page 6 of 6 Sheffield Teaching Hospitals NHS Foundation Trust, a UKAS proficiency testing provider No. 7804, operating UK NEQAS for Leucocyte Immunophenotyping
CD34 Stem Cell Programme Sample All Participant Report. Date Issued - 06 March 2017 Closing Date - 24 March 2017 Machine Used - Facscanto II
 Distribution - 16176 CD3 Stem Cell Programme - 3 All Participant Report Participant ID - XXXX Date Issued - 6 March 17 Closing Date - March 17 Machine Used - Facscanto II Trial Comments This programme
Distribution - 16176 CD3 Stem Cell Programme - 3 All Participant Report Participant ID - XXXX Date Issued - 6 March 17 Closing Date - March 17 Machine Used - Facscanto II Trial Comments This programme
IgH/TCR Clonality Status. Performance Monitoring Cover Sheet. Final IgH Clonality Result IGH 131. Uncontrolled Copy
 Performance Monitoring Cover Sheet Participant No: Trial No: IgH161703 Issue Date: 20 th December 2016 Performance Monitoring: Your Result N Consensus Clonality Result Final IgH Clonality Result IGH 131
Performance Monitoring Cover Sheet Participant No: Trial No: IgH161703 Issue Date: 20 th December 2016 Performance Monitoring: Your Result N Consensus Clonality Result Final IgH Clonality Result IGH 131
Translating Droplet Digital PCR into Clinical Use. Christopher Campbell West Midlands Regional Genetics Laboratory
 Translating Droplet Digital PCR into Clinical Use Christopher Campbell West Midlands Regional Genetics Laboratory Introduction to digital PCR Applications in the West Midlands Regional Genetics Laboratory
Translating Droplet Digital PCR into Clinical Use Christopher Campbell West Midlands Regional Genetics Laboratory Introduction to digital PCR Applications in the West Midlands Regional Genetics Laboratory
Marker types. Potato Association of America Frederiction August 9, Allen Van Deynze
 Marker types Potato Association of America Frederiction August 9, 2009 Allen Van Deynze Use of DNA Markers in Breeding Germplasm Analysis Fingerprinting of germplasm Arrangement of diversity (clustering,
Marker types Potato Association of America Frederiction August 9, 2009 Allen Van Deynze Use of DNA Markers in Breeding Germplasm Analysis Fingerprinting of germplasm Arrangement of diversity (clustering,
AmoyDx TM JAK2 V617F Mutation Detection Kit
 AmoyDx TM JAK2 V617F Mutation Detection Kit Detection of V617F mutation in the JAK2 oncogene Instructions For Use Instructions Version: B1.0 Date of Revision: July 2012 Store at -20±2 o C -1/5- Background
AmoyDx TM JAK2 V617F Mutation Detection Kit Detection of V617F mutation in the JAK2 oncogene Instructions For Use Instructions Version: B1.0 Date of Revision: July 2012 Store at -20±2 o C -1/5- Background
Borrelia burgdorferi
 Borrelia burgdorferi 2013 EQA Programme Final Report QAB114147 (BbDNA13) Dr Volker Fingerle Scientific Expert on behalf of QCMD Report authorised by the QCMD Executive in February 2014 A UKAS accredited
Borrelia burgdorferi 2013 EQA Programme Final Report QAB114147 (BbDNA13) Dr Volker Fingerle Scientific Expert on behalf of QCMD Report authorised by the QCMD Executive in February 2014 A UKAS accredited
ESCMID Online Lecture Library. by author
 Molecular diagnostics: when to use (commercial vs in house) Marijke Raymaekers (Belgium) Kate Templeton (UK) Are you mainly using home-brew or commercial assays? In house diagnostics home brew assays Commercial
Molecular diagnostics: when to use (commercial vs in house) Marijke Raymaekers (Belgium) Kate Templeton (UK) Are you mainly using home-brew or commercial assays? In house diagnostics home brew assays Commercial
Clinical Molecular Biology A Danish Perspective
 Clinical Molecular Biology A Danish Perspective Niels Pallisgaard Molecular biologist Clinical Biochemistry, Vejle Sygehus niels.pallisgaard@slb.regionsyddanmark.dk Here s my DNA sequence The Epidermal
Clinical Molecular Biology A Danish Perspective Niels Pallisgaard Molecular biologist Clinical Biochemistry, Vejle Sygehus niels.pallisgaard@slb.regionsyddanmark.dk Here s my DNA sequence The Epidermal
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Molecular Pathology Duthie Link Building, Mailpoint 225 University Hospital NHS Foundation Trust Tremona Road Hampshire SO16 6YD Contact:
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Molecular Pathology Duthie Link Building, Mailpoint 225 University Hospital NHS Foundation Trust Tremona Road Hampshire SO16 6YD Contact:
TECHNICAL SHEET IDYLLA BRAF MUTATION TEST
 CE IVD TECHNICAL SHEET IDYLLA MUTATION TEST The Idylla, performed on the Biocartis Idylla System, is an in vitro diagnostic Test for the qualitative detection of V600E/ and V600 mutations in codon 600
CE IVD TECHNICAL SHEET IDYLLA MUTATION TEST The Idylla, performed on the Biocartis Idylla System, is an in vitro diagnostic Test for the qualitative detection of V600E/ and V600 mutations in codon 600
Functional DNA Quality Analysis Improves the Accuracy of Next Generation Sequencing from Clinical Specimens
 Functional DNA Quality Analysis Improves the Accuracy of Next Generation Sequencing from Clinical Specimens Overview We have developed a novel QC, the SuraSeq DNA Quantitative Functional Index (QFI ).
Functional DNA Quality Analysis Improves the Accuracy of Next Generation Sequencing from Clinical Specimens Overview We have developed a novel QC, the SuraSeq DNA Quantitative Functional Index (QFI ).
AmoyDx JAK2 Mutation Detection Kit
 AmoyDx JAK2 Mutation Detection Kit Detection of V617F mutation in the JAK2 oncogene Instructions For Use For Research Use Only and For Reference Only Instructions Version: B1.2 Date of Revision: June 2016
AmoyDx JAK2 Mutation Detection Kit Detection of V617F mutation in the JAK2 oncogene Instructions For Use For Research Use Only and For Reference Only Instructions Version: B1.2 Date of Revision: June 2016
Page 1. AMPTask Force on MLS Molecular Pathology Curriculum Dear Molecular Diagnostics Laboratory Manager,
 AMPTask Force on MLS Molecular Pathology Curriculum Dear Molecular Diagnostics Laboratory Manager, Recently the Association for Molecular Pathology (AMP) has decided to develop a suggested curriculum in
AMPTask Force on MLS Molecular Pathology Curriculum Dear Molecular Diagnostics Laboratory Manager, Recently the Association for Molecular Pathology (AMP) has decided to develop a suggested curriculum in
MIqPCR: Minimum Information about a Quantitative Polymerase Chain Reaction experiment Version 1.0, 4th April, 2008.
 MIqPCR: Minimum Information about a Quantitative Polymerase Chain Reaction experiment Version 1.0, 4th April, 2008. RDML-Consortium This module identifies the minimum information required to report the
MIqPCR: Minimum Information about a Quantitative Polymerase Chain Reaction experiment Version 1.0, 4th April, 2008. RDML-Consortium This module identifies the minimum information required to report the
SolCAP. Executive Commitee : David Douches Walter De Jong Robin Buell David Francis Alexandra Stone Lukas Mueller AllenVan Deynze
 SolCAP Solanaceae Coordinated Agricultural Project Supported by the National Research Initiative Plant Genome Program of USDA CSREES for the Improvement of Potato and Tomato Executive Commitee : David
SolCAP Solanaceae Coordinated Agricultural Project Supported by the National Research Initiative Plant Genome Program of USDA CSREES for the Improvement of Potato and Tomato Executive Commitee : David
Mycobacterium tuberculosis
 Mycobacterium tuberculosis 2012 EQA Programme Final Report QAB014129 (MTBDNA12) Prof. Udo Reischl Scientific Expert on behalf of QCMD Report authorised by the QCMD Executive in September 2012 A UKAS accredited
Mycobacterium tuberculosis 2012 EQA Programme Final Report QAB014129 (MTBDNA12) Prof. Udo Reischl Scientific Expert on behalf of QCMD Report authorised by the QCMD Executive in September 2012 A UKAS accredited
VALIDATION OF HLA TYPING BY NGS
 VALIDATION OF HLA TYPING BY NGS Eric T. Weimer, Ph.D., D(ABMLI) Assistant Professor, Pathology and Laboratory Medicine Associate Director, Clinical Flow Cytometry, HLA, and Immunology Laboratories CONFLICT
VALIDATION OF HLA TYPING BY NGS Eric T. Weimer, Ph.D., D(ABMLI) Assistant Professor, Pathology and Laboratory Medicine Associate Director, Clinical Flow Cytometry, HLA, and Immunology Laboratories CONFLICT
CT = control s = sample
 PANFLAVIVIRUS RT-qPCR Trainer: Dr. Cristina Domingo Assistants: Pranav Patel/Ravish Paliwal 1. Thaw all reagents except the Enzyme Mix (keep at -20 C) 2. Prepare maser mix for the RT-qPCR assay (NO DNA
PANFLAVIVIRUS RT-qPCR Trainer: Dr. Cristina Domingo Assistants: Pranav Patel/Ravish Paliwal 1. Thaw all reagents except the Enzyme Mix (keep at -20 C) 2. Prepare maser mix for the RT-qPCR assay (NO DNA
Aromatase Activity and CYP19 Gene Expression in male adult Xenopus laevis Exposed to Atrazine
 Aromatase Activity and CYP19 Gene Expression in male adult Xenopus laevis Exposed to Atrazine June-Woo Park*, Markus Hecker*, Margaret Murphy*, Paul Jones*, Glen van der Kraak**, Keith Solomon**, Ron Kendall***,
Aromatase Activity and CYP19 Gene Expression in male adult Xenopus laevis Exposed to Atrazine June-Woo Park*, Markus Hecker*, Margaret Murphy*, Paul Jones*, Glen van der Kraak**, Keith Solomon**, Ron Kendall***,
A Crash Course in NGS for GI Pathologists. Sandra O Toole
 A Crash Course in NGS for GI Pathologists Sandra O Toole The Sanger Technique First generation sequencing Uses dideoxynucleotides (dideoxyadenine, dideoxyguanine, etc) These are molecules that resemble
A Crash Course in NGS for GI Pathologists Sandra O Toole The Sanger Technique First generation sequencing Uses dideoxynucleotides (dideoxyadenine, dideoxyguanine, etc) These are molecules that resemble
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Royal Liverpool and Broadgreen University Hospitals NHS Trust Liverpool Clinical Laboratories (LCL) Department of Histocompatibility and Immunogenetics
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Royal Liverpool and Broadgreen University Hospitals NHS Trust Liverpool Clinical Laboratories (LCL) Department of Histocompatibility and Immunogenetics
BIOLOGY Dr.Locke Lecture# 27 An Introduction to Polymerase Chain Reaction (PCR)
 BIOLOGY 207 - Dr.Locke Lecture# 27 An Introduction to Polymerase Chain Reaction (PCR) Required readings and problems: Reading: Open Genetics, Chapter 8.1 Problems: Chapter 8 Optional Griffiths (2008) 9
BIOLOGY 207 - Dr.Locke Lecture# 27 An Introduction to Polymerase Chain Reaction (PCR) Required readings and problems: Reading: Open Genetics, Chapter 8.1 Problems: Chapter 8 Optional Griffiths (2008) 9
Successful gene expression studies using validated qpcr assays. Jan Hellemans, CEO Biogazelle webinar October 28 th, 2015
 Successful gene expression studies using validated qpcr assays Jan Hellemans, CEO Biogazelle webinar October 28 th, 2015 Agenda Requirements for high quality qpcr assays Approaches for qpcr assay validation
Successful gene expression studies using validated qpcr assays Jan Hellemans, CEO Biogazelle webinar October 28 th, 2015 Agenda Requirements for high quality qpcr assays Approaches for qpcr assay validation
TOYTEST - Toy Safety Analytes Proficiency Scheme Report Round: 51 Group: Flux testing
 TOYTEST - Toy Safety Analytes Proficiency Scheme Report Group: Flux testing Issue Number 1 Issued 01 October 2012 LGC Standards Proficiency Testing 1 Chamberhall Business Park, Chamberhall Green, Bury
TOYTEST - Toy Safety Analytes Proficiency Scheme Report Group: Flux testing Issue Number 1 Issued 01 October 2012 LGC Standards Proficiency Testing 1 Chamberhall Business Park, Chamberhall Green, Bury
AmoyDx JAK2 Mutation Detection Kit
 AmoyDx JAK2 Mutation Detection Kit Detection of V617F mutation in the JAK2 oncogene Instruction for Use For Research Use Only Instruction Version: B2.6 Revision Date: October 2013 Store at -20±5 Xiamen,
AmoyDx JAK2 Mutation Detection Kit Detection of V617F mutation in the JAK2 oncogene Instruction for Use For Research Use Only Instruction Version: B2.6 Revision Date: October 2013 Store at -20±5 Xiamen,
Annex to the Accreditation Certificate D PL according to DIN EN ISO/IEC 17025:2005
 Deutsche Akkreditierungsstelle GmbH German Accreditation Body Annex to the Accreditation Certificate D PL 13372 01 00 according to DIN EN ISO/IEC 17025:2005 Period of validity: 22.05.2014 to 25.03.2017
Deutsche Akkreditierungsstelle GmbH German Accreditation Body Annex to the Accreditation Certificate D PL 13372 01 00 according to DIN EN ISO/IEC 17025:2005 Period of validity: 22.05.2014 to 25.03.2017
Instruction manual for product # PNAC Version 2.0
 PNAClamp JAK2 Mutation Detection Kit For in vitro diagnostic use Instruction manual for product # PNAC-6001 Version 2.0 Store at -15 to -20 Instruction Version: 2.0 Date of Revision: Sep. 2016 1 / 19 PNG-PCJUM001
PNAClamp JAK2 Mutation Detection Kit For in vitro diagnostic use Instruction manual for product # PNAC-6001 Version 2.0 Store at -15 to -20 Instruction Version: 2.0 Date of Revision: Sep. 2016 1 / 19 PNG-PCJUM001
Clinical Performance of JAK2 V617F Mutation Detection Assays in a Molecular Diagnostics Laboratory Evaluation of Screening and Quantitation Methods
 Hematopathology / Method Comparison for JAK2 V617F Detection Clinical Performance of JAK2 V617F Mutation Detection Assays in a Molecular Diagnostics Laboratory Evaluation of Screening and Quantitation
Hematopathology / Method Comparison for JAK2 V617F Detection Clinical Performance of JAK2 V617F Mutation Detection Assays in a Molecular Diagnostics Laboratory Evaluation of Screening and Quantitation
2100 Bioanalyzer. Overview & News. Ralph Beneke Dec 2010
 2100 Bioanalyzer Overview & News Ralph Beneke Dec 2010 RNA QC in Gene Expression Workflows Cited in over 10,000 peer-reviewed papers RIN is the industry standard for RNA QC For any size and at low sample
2100 Bioanalyzer Overview & News Ralph Beneke Dec 2010 RNA QC in Gene Expression Workflows Cited in over 10,000 peer-reviewed papers RIN is the industry standard for RNA QC For any size and at low sample
Rapid Cycle PCR, Real Time Analysis, and Hi-Res Melting
 Rapid Cycle PCR, Real Time Analysis, and Hi-Res Melting Carl Wittwer Department of Pathology University of Utah ARUP Idaho Technology AMP, Oct. 31, 2008 Impatient, Lazy, and Cheap Rapid Cycle PCR Fast
Rapid Cycle PCR, Real Time Analysis, and Hi-Res Melting Carl Wittwer Department of Pathology University of Utah ARUP Idaho Technology AMP, Oct. 31, 2008 Impatient, Lazy, and Cheap Rapid Cycle PCR Fast
Monitoring Assay Traceability through EQAS Surveillance
 FOR CLINICAL CHEMISTRY UNITED KINGDOM NATIONAL EXTERNAL QUALITY ASSESSMENT SCHEMES Monitoring Assay Traceability through EQAS Surveillance AACC Edutrak 3305, Orlando, 30 July 2002 Dr David G Bullock Director,
FOR CLINICAL CHEMISTRY UNITED KINGDOM NATIONAL EXTERNAL QUALITY ASSESSMENT SCHEMES Monitoring Assay Traceability through EQAS Surveillance AACC Edutrak 3305, Orlando, 30 July 2002 Dr David G Bullock Director,
PARTICIPANTS MANUAL. Point of Care RhD typing. UK NEQAS for Blood Transfusion Laboratory Practice
 PARTICIPANTS MANUAL Point of Care RhD typing UK NEQAS for Blood Transfusion Laboratory Practice UK NEQAS (BTLP) PO Box 133 Watford WD18 0WP CONTENTS PAGE General Organisation of the Scheme 1-3 Location
PARTICIPANTS MANUAL Point of Care RhD typing UK NEQAS for Blood Transfusion Laboratory Practice UK NEQAS (BTLP) PO Box 133 Watford WD18 0WP CONTENTS PAGE General Organisation of the Scheme 1-3 Location
AmoyDx JAK2 Mutation Detection Kit
 AmoyDx JAK2 Mutation Detection Kit Detection of V617F mutation in the JAK2 oncogene Instruction for Use For Research Use Only and For Reference Only 8.01.20801X024E 24 tests For Rotor-Gene Q/6000 (72 Wells)
AmoyDx JAK2 Mutation Detection Kit Detection of V617F mutation in the JAK2 oncogene Instruction for Use For Research Use Only and For Reference Only 8.01.20801X024E 24 tests For Rotor-Gene Q/6000 (72 Wells)
Principals of Real-Time PCR. Amira A. T. AL-Hosary Lecturer of Infectious Diseases, Faculty of Veterinary Medicine, Assiut University, Egypt
 Principals of Real-Time PCR Amira A. T. AL-Hosary Lecturer of Infectious Diseases, Faculty of Veterinary Medicine, Assiut University, Egypt What Is Real-Time PCR? Nucleic acid (DNA) amplification and detection
Principals of Real-Time PCR Amira A. T. AL-Hosary Lecturer of Infectious Diseases, Faculty of Veterinary Medicine, Assiut University, Egypt What Is Real-Time PCR? Nucleic acid (DNA) amplification and detection
PAXgene Blood DNA PAXgene Blood DNA Tube (IVD) for clinical use PAXgene Blood DNA System (RUO) for research use. Explore more at
 PAXgene Blood DNA PAXgene Blood DNA Tube (IVD) for clinical use PAXgene Blood DNA System (RUO) for research use Explore more at www.preanalytix.com Situation The composition, amount, quality and integrity
PAXgene Blood DNA PAXgene Blood DNA Tube (IVD) for clinical use PAXgene Blood DNA System (RUO) for research use Explore more at www.preanalytix.com Situation The composition, amount, quality and integrity
Outline General NGS background and terms 11/14/2016 CONFLICT OF INTEREST. HLA region targeted enrichment. NGS library preparation methodologies
 Eric T. Weimer, PhD, D(ABMLI) Assistant Professor, Pathology & Laboratory Medicine, UNC School of Medicine Director, Molecular Immunology Associate Director, Clinical Flow Cytometry, HLA, and Immunology
Eric T. Weimer, PhD, D(ABMLI) Assistant Professor, Pathology & Laboratory Medicine, UNC School of Medicine Director, Molecular Immunology Associate Director, Clinical Flow Cytometry, HLA, and Immunology
Current Status of Forensic DNA Laboratory in Bangkok Thailand
 8 th National Grad Research Conference 1 Current Status of Forensic DNA Laboratory in Bangkok Thailand Samart Panthuen, Nathinee Panvisavas and Prapin Wilairat Forensic Science Graduate Programme, Faculty
8 th National Grad Research Conference 1 Current Status of Forensic DNA Laboratory in Bangkok Thailand Samart Panthuen, Nathinee Panvisavas and Prapin Wilairat Forensic Science Graduate Programme, Faculty
A portable microfluidic platform for rapid molecular diagnostic. testing of patients with myeloproliferative neoplasms
 A portable microfluidic platform for rapid molecular diagnostic testing of patients with myeloproliferative neoplasms Hua Wang 1, Xinju zhang 1, Xiao Xu 1, Qunfeng Zhang 1, Hengliang Wang 2, Dong Li 3,
A portable microfluidic platform for rapid molecular diagnostic testing of patients with myeloproliferative neoplasms Hua Wang 1, Xinju zhang 1, Xiao Xu 1, Qunfeng Zhang 1, Hengliang Wang 2, Dong Li 3,
Precise quantification of Ion Torrent libraries on the QuantStudio 3D Digital PCR System
 APPLICATION NOTE QuantStudio 3D Digital PCR System Precise quantification of Ion Torrent libraries on the QuantStudio 3D Digital PCR System Introduction The template preparation step in the next-generation
APPLICATION NOTE QuantStudio 3D Digital PCR System Precise quantification of Ion Torrent libraries on the QuantStudio 3D Digital PCR System Introduction The template preparation step in the next-generation
MOLECULAR TESTING: VERIFYING/VALIDATING INSTRUMENTS, REAGENTS AND ASSAYS. Richard L. Hodinka, Ph.D.
 MOLECULAR TESTING: VERIFYING/VALIDATING INSTRUMENTS, REAGENTS AND ASSAYS Richard L. Hodinka, Ph.D. University of South Carolina School of Medicine Greenville Greenville Health System, Greenville, SC hodinka@greenvillemed.sc.edu
MOLECULAR TESTING: VERIFYING/VALIDATING INSTRUMENTS, REAGENTS AND ASSAYS Richard L. Hodinka, Ph.D. University of South Carolina School of Medicine Greenville Greenville Health System, Greenville, SC hodinka@greenvillemed.sc.edu
AmoyDx TM RET 14 Mutations Detection Kit
 For: ADx-RE0 AmoyDx TM RET Mutations Detection Kit Detection of rearrangement mutations of RET gene Instructions For Use Instructions Version: B. Date of Revision: April 0 Store at -0± o C Website:www.amoydx.com/en
For: ADx-RE0 AmoyDx TM RET Mutations Detection Kit Detection of rearrangement mutations of RET gene Instructions For Use Instructions Version: B. Date of Revision: April 0 Store at -0± o C Website:www.amoydx.com/en
Multiplex PCR Assay Kit Ver.2
 Cat. # RR062A For Research Use Multiplex PCR Assay Kit Ver.2 Product Manual Table of Contents I. Description... 3 II. Components... 3 III. Storage... 3 IV. Precautions... 3 V. Designing Primers for Multiplex
Cat. # RR062A For Research Use Multiplex PCR Assay Kit Ver.2 Product Manual Table of Contents I. Description... 3 II. Components... 3 III. Storage... 3 IV. Precautions... 3 V. Designing Primers for Multiplex
Blood direct 2x PCR Mastermix. Data sheet. Order No. BS reactions x 20 µl. (For research and in vitro applications only) Batch No.
 Data sheet Order No. BS91.222.0250 250 reactions x 20 µl Order No. BS91.222.1250 1250 reactions x 20 µl (For research and in vitro applications only) Batch No.: Best before: Appearance: Colour: 1 Description
Data sheet Order No. BS91.222.0250 250 reactions x 20 µl Order No. BS91.222.1250 1250 reactions x 20 µl (For research and in vitro applications only) Batch No.: Best before: Appearance: Colour: 1 Description
Diagnosis of MDD was based on criteria of the Diagnostic and Statistical Manual of Mental
 Data Supplement for Luo and Zhang, Down-Regulation of SIRT1 Gene Expression in Major Depressive Disorder. Am J Psychiatry (doi: 10.1176/appi.ajp.2016.16040394) MDD diagnosis and detailed demographic information
Data Supplement for Luo and Zhang, Down-Regulation of SIRT1 Gene Expression in Major Depressive Disorder. Am J Psychiatry (doi: 10.1176/appi.ajp.2016.16040394) MDD diagnosis and detailed demographic information
CBFB-MYH11 REAL TIME QUANTITATIVE PCR DETECTION KIT ONKOTEST RQ
 CBFB-MYH11 REAL TIME QUANTITATIVE PCR DETECTION KIT ONKOTEST RQ3021-20 Product Information Inv(16)(p13q22) or the variant t(16;16)(p13;q22) are frequent recurring chromosomal rearrangements reported to
CBFB-MYH11 REAL TIME QUANTITATIVE PCR DETECTION KIT ONKOTEST RQ3021-20 Product Information Inv(16)(p13q22) or the variant t(16;16)(p13;q22) are frequent recurring chromosomal rearrangements reported to
FLT3-ITD REAL TIME PCR DETECTION KIT
 FLT3-ITD REAL TIME PCR DETECTION KIT ONKOTEST R3030-20 Keep the kit at -15 C to -25 C Rev. 1.4 Product Information FLT3 (fms-related tyrosine kinase 3) gene frequently mutated in acute myeloid leukemia
FLT3-ITD REAL TIME PCR DETECTION KIT ONKOTEST R3030-20 Keep the kit at -15 C to -25 C Rev. 1.4 Product Information FLT3 (fms-related tyrosine kinase 3) gene frequently mutated in acute myeloid leukemia
JAK2 V617F mutation mutation detection by quantitative allele specific amplification (quasa) JAK2(V617F)
 Primerdesign TM Ltd JAK2 V617F mutation mutation detection by quantitative allele specific amplification (quasa) JAK2(V617F) 50 tests For general laboratory and research use only 1 Contents Introduction
Primerdesign TM Ltd JAK2 V617F mutation mutation detection by quantitative allele specific amplification (quasa) JAK2(V617F) 50 tests For general laboratory and research use only 1 Contents Introduction
BIOO LIFE SCIENCE PRODUCTS. NEXTflex TM 16S V4 Amplicon-Seq Kit 4 (Illumina Compatible) BIOO Scientific Corp V13.01
 BIOO LIFE SCIENCE PRODUCTS NEXTflex TM 16S V4 Amplicon-Seq Kit 4 (Illumina Compatible) Catalog #: 4201-01 (16 reactions) BIOO Scientific Corp. 2013 V13.01 TABLE OF CONTENTS GENERAL INFORMATION... 1 Product
BIOO LIFE SCIENCE PRODUCTS NEXTflex TM 16S V4 Amplicon-Seq Kit 4 (Illumina Compatible) Catalog #: 4201-01 (16 reactions) BIOO Scientific Corp. 2013 V13.01 TABLE OF CONTENTS GENERAL INFORMATION... 1 Product
PrimePCR Assay Validation Report
 Gene Information Gene Name Gene Symbol Organism Gene Summary Gene Aliases RefSeq Accession No. UniGene ID Ensembl Gene ID coagulation factor XIII, A1 polypeptide F13A1 Human This gene encodes the coagulation
Gene Information Gene Name Gene Symbol Organism Gene Summary Gene Aliases RefSeq Accession No. UniGene ID Ensembl Gene ID coagulation factor XIII, A1 polypeptide F13A1 Human This gene encodes the coagulation
Real Time Quantitative PCR Assay Validation, Optimization and Troubleshooting
 Real Time Quantitative PCR Assay Validation, Optimization and Troubleshooting Dr Steffen Muller Field Applications Scientist Stratagene Europe Overview The Importance of Controls Validation and Optimization
Real Time Quantitative PCR Assay Validation, Optimization and Troubleshooting Dr Steffen Muller Field Applications Scientist Stratagene Europe Overview The Importance of Controls Validation and Optimization
Roche Molecular Biochemicals Technical Note No. LC 9/2000
 Roche Molecular Biochemicals Technical Note No. LC 9/2000 LightCycler Optimization Strategy Introduction Purpose of this Note Table of Contents The LightCycler system provides different detection formats
Roche Molecular Biochemicals Technical Note No. LC 9/2000 LightCycler Optimization Strategy Introduction Purpose of this Note Table of Contents The LightCycler system provides different detection formats
AmpFlSTR Identifiler PCR Amplification Kit
 Product Bulletin Human Identification AmpFlSTR Identifiler PCR Amplification Kit Fifteen STR (short tandem repeat) loci and Amelogenin co-amplified in a single tube Incorporation of the same proven, reliable
Product Bulletin Human Identification AmpFlSTR Identifiler PCR Amplification Kit Fifteen STR (short tandem repeat) loci and Amelogenin co-amplified in a single tube Incorporation of the same proven, reliable
Technical Review. Real time PCR
 Technical Review Real time PCR Normal PCR: Analyze with agarose gel Normal PCR vs Real time PCR Real-time PCR, also known as quantitative PCR (qpcr) or kinetic PCR Key feature: Used to amplify and simultaneously
Technical Review Real time PCR Normal PCR: Analyze with agarose gel Normal PCR vs Real time PCR Real-time PCR, also known as quantitative PCR (qpcr) or kinetic PCR Key feature: Used to amplify and simultaneously
OIE Reference Laboratory Reports Activities
 OIE Reference Laboratory Reports Activities Activities in 2017 This report has been submitted : 2018-01-18 22:45:15 Name of disease (or topic) for which you are a designated OIE Reference Laboratory: Infectious
OIE Reference Laboratory Reports Activities Activities in 2017 This report has been submitted : 2018-01-18 22:45:15 Name of disease (or topic) for which you are a designated OIE Reference Laboratory: Infectious
LaktaseCheck real time PCR Kit
 Instruction for Use Dr. Schröders LaktaseCheck real time PCR Kit Test for the analysis of the C/T polymorphism at position -13910 within the regulatory region of the lactase gen in man. G01010-96 96 gerbion
Instruction for Use Dr. Schröders LaktaseCheck real time PCR Kit Test for the analysis of the C/T polymorphism at position -13910 within the regulatory region of the lactase gen in man. G01010-96 96 gerbion
BEIPH Final Report. EQA Programme 2011 Hepatitis B Virus (HBVDNA11A) William G Mackay on behalf of QCMD and its Scientific Council July 2011
 BEIPH Final Report EQA Programme 2011 Hepatitis B Virus (HBVDNA11A) William G Mackay on behalf of QCMD and its Scientific Council July 2011 Not to be reproduced or quoted without permission of QCMD. Any
BEIPH Final Report EQA Programme 2011 Hepatitis B Virus (HBVDNA11A) William G Mackay on behalf of QCMD and its Scientific Council July 2011 Not to be reproduced or quoted without permission of QCMD. Any
Dedicated to Molecular Diagnostics
 Dedicated to Molecular Diagnostics For Europe Sample & Assay Technologies QIAGEN s growing role in molecular diagnostics At QIAGEN, one of our key goals is to support clinical decision-making and improve
Dedicated to Molecular Diagnostics For Europe Sample & Assay Technologies QIAGEN s growing role in molecular diagnostics At QIAGEN, one of our key goals is to support clinical decision-making and improve
Product Description SALSA MLPA Probemix P438-D2 HLA
 Product Description SALSA Probemix P438-D2 HLA To be used with the MLPA General Protocol. Version D2. Catalogue numbers: P438-025R: SALSA MLPA Probemix P438 HLA, 25 reactions. P438-050R: SALSA MLPA Probemix
Product Description SALSA Probemix P438-D2 HLA To be used with the MLPA General Protocol. Version D2. Catalogue numbers: P438-025R: SALSA MLPA Probemix P438 HLA, 25 reactions. P438-050R: SALSA MLPA Probemix
Course Competencies Template Form 112
 Course Competencies Template Form 112 GENERAL INFORMATION Course Prefix/Number: BSC-2426L Number of Credits: 2 Degree Type Course Title: Biotechnology Methods and Applications Lab-I B.A. B.S. B.A.S A.A.
Course Competencies Template Form 112 GENERAL INFORMATION Course Prefix/Number: BSC-2426L Number of Credits: 2 Degree Type Course Title: Biotechnology Methods and Applications Lab-I B.A. B.S. B.A.S A.A.
Processing Ion AmpliSeq Data using NextGENe Software v2.3.0
 Processing Ion AmpliSeq Data using NextGENe Software v2.3.0 July 2012 John McGuigan, Megan Manion, Kevin LeVan, CS Jonathan Liu Introduction The Ion AmpliSeq Panels use highly multiplexed PCR in order
Processing Ion AmpliSeq Data using NextGENe Software v2.3.0 July 2012 John McGuigan, Megan Manion, Kevin LeVan, CS Jonathan Liu Introduction The Ion AmpliSeq Panels use highly multiplexed PCR in order
European Union Reference Laboratory for Genetically Modified Food and Feed
 Report on the Verification of the Performance of MON 87751, MON 87701, MON 87708 and MON 89788 Event-specific PCR-based Methods Applied to DNA Extracted from GM Stack MON 87751 x MON 87701 x MON 87708
Report on the Verification of the Performance of MON 87751, MON 87701, MON 87708 and MON 89788 Event-specific PCR-based Methods Applied to DNA Extracted from GM Stack MON 87751 x MON 87701 x MON 87708
Principles of Real Time PCR Ameer Effat M. Elfarash
 Principles of Real Time PCR Ameer Effat M. Elfarash Dept. of Genetics Fac. of Agriculture, Assiut Univ. aelfarash@aun.edu.eg Types of PCR Standard PCR (conventional ) RT-PCR (Reverse Transcriptase PCR)
Principles of Real Time PCR Ameer Effat M. Elfarash Dept. of Genetics Fac. of Agriculture, Assiut Univ. aelfarash@aun.edu.eg Types of PCR Standard PCR (conventional ) RT-PCR (Reverse Transcriptase PCR)
CAPTURE-BASED APPROACH FOR COMPREHENSIVE DETECTION OF IMPORTANT ALTERATIONS
 CAPTURE-BASE APPROACH FOR COMPREHENSIVE ETECTION OF IMPORTANT ALTERATIONS SEQUENCE MUTATIONS MICROSATELLITE INSTABILITY AMPLIFICATIONS GENOMIC REARRANGEMENTS For Research Use Only. Not for iagnostic Purposes.
CAPTURE-BASE APPROACH FOR COMPREHENSIVE ETECTION OF IMPORTANT ALTERATIONS SEQUENCE MUTATIONS MICROSATELLITE INSTABILITY AMPLIFICATIONS GENOMIC REARRANGEMENTS For Research Use Only. Not for iagnostic Purposes.
Roche Molecular Biochemicals Technical Note No. LC 10/2000
 Roche Molecular Biochemicals Technical Note No. LC 10/2000 LightCycler Overview of LightCycler Quantification Methods 1. General Introduction Introduction Content Definitions This Technical Note will introduce
Roche Molecular Biochemicals Technical Note No. LC 10/2000 LightCycler Overview of LightCycler Quantification Methods 1. General Introduction Introduction Content Definitions This Technical Note will introduce
Lecture 8: Sequencing and SNP. Sept 15, 2006
 Lecture 8: Sequencing and SNP Sept 15, 2006 Announcements Random questioning during literature discussion sessions starts next week for real! Schedule changes Moved QTL lecture up Removed landscape genetics
Lecture 8: Sequencing and SNP Sept 15, 2006 Announcements Random questioning during literature discussion sessions starts next week for real! Schedule changes Moved QTL lecture up Removed landscape genetics
Factorial TM 30: Evaluate 30 Transcription Factors in single experiment
 Factorial TM 30: Evaluate 30 Transcription Factors in single experiment Attagene Inc. proudly introduces FACTORIAL 30, the founding product line that is based on our proprietary technology and enables
Factorial TM 30: Evaluate 30 Transcription Factors in single experiment Attagene Inc. proudly introduces FACTORIAL 30, the founding product line that is based on our proprietary technology and enables
UK NEQAS FOR BLOOD COAGULATION: Programmi EQAS nel laboratorio di coagulazione
 UK NEQAS FOR BLOOD COAGULATION: Programmi EQAS nel laboratorio di coagulazione Tim Woods, Manager UK NEQAS for Blood Coagulation STH Royal Hallamshire Hospital Sheffield U.K. www.ukneqasbc.org Quality
UK NEQAS FOR BLOOD COAGULATION: Programmi EQAS nel laboratorio di coagulazione Tim Woods, Manager UK NEQAS for Blood Coagulation STH Royal Hallamshire Hospital Sheffield U.K. www.ukneqasbc.org Quality
Additional File 1. Method 1: Restriction digestion of the TB Control Plasmid
 Additional File 1 Method 1: Restriction digestion of the TB Plasmid Approximately 1 ng of the control plasmid was linearised with 1 Units of HindIII restriction enzyme (New England Biolabs), in a final
Additional File 1 Method 1: Restriction digestion of the TB Plasmid Approximately 1 ng of the control plasmid was linearised with 1 Units of HindIII restriction enzyme (New England Biolabs), in a final
Evaluation of Restriction Enzymes for the Analysis of Circulating Free DNA by Droplet Digital PCR
 Evaluation of Restriction Enzymes for the Analysis of Circulating Free DNA by Droplet Digital PCR Amanda Weaver Cancer Genomics Consortium 2017 Summer Meeting August 7, 2017 Acknowledgements Everyone at
Evaluation of Restriction Enzymes for the Analysis of Circulating Free DNA by Droplet Digital PCR Amanda Weaver Cancer Genomics Consortium 2017 Summer Meeting August 7, 2017 Acknowledgements Everyone at
Combining Techniques to Answer Molecular Questions
 Combining Techniques to Answer Molecular Questions UNIT FM02 How to cite this article: Curr. Protoc. Essential Lab. Tech. 9:FM02.1-FM02.5. doi: 10.1002/9780470089941.etfm02s9 INTRODUCTION This manual is
Combining Techniques to Answer Molecular Questions UNIT FM02 How to cite this article: Curr. Protoc. Essential Lab. Tech. 9:FM02.1-FM02.5. doi: 10.1002/9780470089941.etfm02s9 INTRODUCTION This manual is
FLT3 Conventional PCR Detection Kit ONKOTEST K
 FLT3 Conventional PCR Detection Kit ONKOTEST K3030-20 Product Information FLT3 (fms-related tyrosine kinase 3) gene frequently mutated in acute myeloid leukemia (AML) is associated with disease pathogenesis.
FLT3 Conventional PCR Detection Kit ONKOTEST K3030-20 Product Information FLT3 (fms-related tyrosine kinase 3) gene frequently mutated in acute myeloid leukemia (AML) is associated with disease pathogenesis.
qpcr Quantitative PCR or Real-time PCR Gives a measurement of PCR product at end of each cycle real time
 qpcr qpcr Quantitative PCR or Real-time PCR Gives a measurement of PCR product at end of each cycle real time Differs from endpoint PCR gel on last cycle Used to determines relative amount of template
qpcr qpcr Quantitative PCR or Real-time PCR Gives a measurement of PCR product at end of each cycle real time Differs from endpoint PCR gel on last cycle Used to determines relative amount of template
KAPA hgdna QUANTIFICATION AND QC KIT:
 Poster Note As presented at AGBT 2015, Marco Island, FL KAPA hgdna QUANTIFICATION AND QC KIT: The KAPA Human Genomic DNA Quantification and QC Kit Enables Prediction of Sequencing Performance through User-Defined
Poster Note As presented at AGBT 2015, Marco Island, FL KAPA hgdna QUANTIFICATION AND QC KIT: The KAPA Human Genomic DNA Quantification and QC Kit Enables Prediction of Sequencing Performance through User-Defined
Real-Time PCR: An Essential Guide
 Real-Time PCR: An Essential Guide Publisher: Horizon Bioscience Editor: Kirstin Edwards, Julie Logan and Nick Saunders Genomics Proteomics and Bioinformatics Unit, Health Protection Agency, London Publication
Real-Time PCR: An Essential Guide Publisher: Horizon Bioscience Editor: Kirstin Edwards, Julie Logan and Nick Saunders Genomics Proteomics and Bioinformatics Unit, Health Protection Agency, London Publication
Protocol Page 1 of 11. Protocol for Proficiency testing scheme for Bitumen Programme No. GPTS/PT 05
 Tel Nos. +91 79 2656 5405, 2656 1104, Mob Nos. +91 98254 13148, 93270 17517, Fax Nos. +91 79 2656 3240 Email sales@globalptservices.com, nilesh@globalptservices.com Protocol Page 1 of 11 Protocol for Proficiency
Tel Nos. +91 79 2656 5405, 2656 1104, Mob Nos. +91 98254 13148, 93270 17517, Fax Nos. +91 79 2656 3240 Email sales@globalptservices.com, nilesh@globalptservices.com Protocol Page 1 of 11 Protocol for Proficiency
Analytical verification methods for the Oncomine Lung cfdna Assay using the Ion S5 XL System
 WHITE PAPER Oncomine Lung cfdna Assay and Ion S5 XL System Analytical verification methods for the Oncomine Lung cfdna Assay using the Ion S5 XL System Key highlights Investigate tumor heterogeneity and
WHITE PAPER Oncomine Lung cfdna Assay and Ion S5 XL System Analytical verification methods for the Oncomine Lung cfdna Assay using the Ion S5 XL System Key highlights Investigate tumor heterogeneity and
HELINI Hepatitis B virus [HBV] Real-time PCR Kit (Genotype A to H)
![HELINI Hepatitis B virus [HBV] Real-time PCR Kit (Genotype A to H) HELINI Hepatitis B virus [HBV] Real-time PCR Kit (Genotype A to H)](/thumbs/87/95094490.jpg) HELINI Hepatitis B virus [HBV] Real-time PCR Kit (Genotype A to H) Quantitative In vitro diagnostics Instruction manual Cat. No: 8001-25/50/100 tests Compatible with: Agilent, Bio-Rad, Applied Bio systems
HELINI Hepatitis B virus [HBV] Real-time PCR Kit (Genotype A to H) Quantitative In vitro diagnostics Instruction manual Cat. No: 8001-25/50/100 tests Compatible with: Agilent, Bio-Rad, Applied Bio systems
Introducing: 3Zomy Aneuploidity Test
 Introducing: 3Zomy Aneuploidity Test QF-PCR Life Technologies (India) Pvt. Ltd. 306, Aggarwal City Mall, Opposite M2K Pitampura, Delhi 110034 (INDIA). Ph: +91-11-42208000, 4220811, 42208222 Mobile: +91-9810521400
Introducing: 3Zomy Aneuploidity Test QF-PCR Life Technologies (India) Pvt. Ltd. 306, Aggarwal City Mall, Opposite M2K Pitampura, Delhi 110034 (INDIA). Ph: +91-11-42208000, 4220811, 42208222 Mobile: +91-9810521400
amplification High Resolution Melt Parameter Considerations for Optimal Data Resolution tech note 6009
 amplification tech note 6009 High Resolution Melt Parameter Considerations for Optimal Data Resolution Carl Fisher, Ray Meng, Francisco Bizouarn, and Rachel Scott Gene Expression Division, Bio-Rad Laboratories,
amplification tech note 6009 High Resolution Melt Parameter Considerations for Optimal Data Resolution Carl Fisher, Ray Meng, Francisco Bizouarn, and Rachel Scott Gene Expression Division, Bio-Rad Laboratories,
Applied Biosystems Real-Time PCR Rapid Assay Development Guidelines
 Applied Biosystems Real-Time PCR Rapid Assay Development Guidelines Description This tutorial will discuss recommended guidelines for designing and running real-time PCR quantification and SNP Genotyping
Applied Biosystems Real-Time PCR Rapid Assay Development Guidelines Description This tutorial will discuss recommended guidelines for designing and running real-time PCR quantification and SNP Genotyping
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK University College London Hospitals NHS Foundation Trust National Hospital for Neurology & Neurosurgery Unit, 6 th Floor UCL Institute of
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK University College London Hospitals NHS Foundation Trust National Hospital for Neurology & Neurosurgery Unit, 6 th Floor UCL Institute of
AKESOgen, Inc, 3155 Northwoods Place, NW, Norcross, GA 30071, USA Tel: +1 (770) Page 1
 Page 1 HLA Sequencing Based Typing Workflow: A- Sample Collection: iswab-discovery (Mawi DNA Technologies) - Sample was transported by standard US postal services mail, no cold chain of any sort was involved
Page 1 HLA Sequencing Based Typing Workflow: A- Sample Collection: iswab-discovery (Mawi DNA Technologies) - Sample was transported by standard US postal services mail, no cold chain of any sort was involved
SeCore SBT Sequence Based Typing
 MOLECULAR TYPING Product Brochure SeCore SBT Sequence Based Typing Key Benefits SeCore HLA typing kits combine the accuracy of bidirectional sequencing and the power and flexibility of our improved sequence
MOLECULAR TYPING Product Brochure SeCore SBT Sequence Based Typing Key Benefits SeCore HLA typing kits combine the accuracy of bidirectional sequencing and the power and flexibility of our improved sequence
Comparison of ExoSAP-IT and ExoSAP-IT Express reagents to alternative PCR cleanup methods
 WHITE PAPER ExoSAP-IT PCR cleanup reagents Comparison of ExoSAP-IT and ExoSAP-IT Express reagents to alternative PCR cleanup methods Abstract Here we present superior workflow advantages of enzymatic PCR
WHITE PAPER ExoSAP-IT PCR cleanup reagents Comparison of ExoSAP-IT and ExoSAP-IT Express reagents to alternative PCR cleanup methods Abstract Here we present superior workflow advantages of enzymatic PCR
Representing Errors and Uncertainty in Plasma Proteomics
 Representing Errors and Uncertainty in Plasma Proteomics David J. States, M.D., Ph.D. University of Michigan Bioinformatics Program Proteomics Alliance for Cancer Genomics vs. Proteomics Genome sequence
Representing Errors and Uncertainty in Plasma Proteomics David J. States, M.D., Ph.D. University of Michigan Bioinformatics Program Proteomics Alliance for Cancer Genomics vs. Proteomics Genome sequence
Embrace the Future of Electrophoresis
 Embrace the Future of Electrophoresis Analysis of 12 samples in as little as 3 minutes Unattended analysis of up to 96 samples Resolution down to 3 5 bp for fragments
Embrace the Future of Electrophoresis Analysis of 12 samples in as little as 3 minutes Unattended analysis of up to 96 samples Resolution down to 3 5 bp for fragments
Microsatellite markers
 Microsatellite markers Review of repetitive sequences 25% 45% 8% 21% 13% 3% Mobile genetic elements: = dispersed repeat included: transposition: moving in the form of DNA by element coding for transposases.
Microsatellite markers Review of repetitive sequences 25% 45% 8% 21% 13% 3% Mobile genetic elements: = dispersed repeat included: transposition: moving in the form of DNA by element coding for transposases.
Supporting Information for
 Supporting Information for Ultra-selective and sensitive DNA detection by a universal apurinic/apyrimidinic probe-based Endonuclease IV signal amplification system Xianjin Xiao, Chen Song, Chen Zhang,
Supporting Information for Ultra-selective and sensitive DNA detection by a universal apurinic/apyrimidinic probe-based Endonuclease IV signal amplification system Xianjin Xiao, Chen Song, Chen Zhang,
Changing the world of laboratory developed testing
 Changing the world of laboratory developed testing Solution overview Explore the solution Experience a convenient and easy workflow FLOW Primary Sample Handling Instrument MagNA Pure 96 Instrument FLOW
Changing the world of laboratory developed testing Solution overview Explore the solution Experience a convenient and easy workflow FLOW Primary Sample Handling Instrument MagNA Pure 96 Instrument FLOW
BIOO LIFE SCIENCE PRODUCTS
 BIOO LIFE SCIENCE PRODUCTS FOR REFERENCE PURPOSES This manual is for Reference Purposes Only. DO NOT use this protocol to run your assays. Periodically, optimizations and revisions are made to the kit
BIOO LIFE SCIENCE PRODUCTS FOR REFERENCE PURPOSES This manual is for Reference Purposes Only. DO NOT use this protocol to run your assays. Periodically, optimizations and revisions are made to the kit
MOLECULAR SOLUTIONS FOR BIOTECHNOLOGY
 MOLECULAR SOLUTIONS FOR BIOTECHNOLOGY OUR PCR TECHNOLOGY OUR CAPABILITIES Over 20 years of molecular biology research has given Bioline scientists a deep understanding of the profound influence that buffer
MOLECULAR SOLUTIONS FOR BIOTECHNOLOGY OUR PCR TECHNOLOGY OUR CAPABILITIES Over 20 years of molecular biology research has given Bioline scientists a deep understanding of the profound influence that buffer
Next Generation Oncology Sequencing in your Laboratory. Built by pioneers in cancer genomics and liquid biopsy approaches PROGENEUS
 PROGENEUS Next Generation Oncology Sequencing in your Laboratory Built by pioneers in cancer genomics and liquid biopsy approaches For Research Use Only. Not for iagnostic Purposes. personalgenome.com/progeneus
PROGENEUS Next Generation Oncology Sequencing in your Laboratory Built by pioneers in cancer genomics and liquid biopsy approaches For Research Use Only. Not for iagnostic Purposes. personalgenome.com/progeneus
ipsog Kit Version Quantitati ive in vitro LightCycle er instruments GERMANY EN
 January 2013 ipsog gen JAK2 MutaQuant Handbook Kit 12 (catalog no. 673522) 24 (catalog no. 673523) Version 1 Quantitati ive in vitro diagnostics For use with Rotor-Gene Q, ABI PRISM 7900HT SDS, Applied
January 2013 ipsog gen JAK2 MutaQuant Handbook Kit 12 (catalog no. 673522) 24 (catalog no. 673523) Version 1 Quantitati ive in vitro diagnostics For use with Rotor-Gene Q, ABI PRISM 7900HT SDS, Applied
Setting up a Run of Real-time PCR cycler Rotor Gene 3000
 Version: 1.0 Last Revision Date: 4. 11. 2014 Setting up a Run of Real-time PCR cycler Rotor Gene 3000 (Corbett Research) generi biotech OBSAH: 1. Setting of a new temperature profile and running the cycler...
Version: 1.0 Last Revision Date: 4. 11. 2014 Setting up a Run of Real-time PCR cycler Rotor Gene 3000 (Corbett Research) generi biotech OBSAH: 1. Setting of a new temperature profile and running the cycler...
MASSEY GENOME SERVICE SEPTEMBER 2014 NEWSLETTER
 MASSEY GENOME SERVICE SEPTEMBER 2014 NEWSLETTER Welcome to the September 2014 issue of the Massey Genome Service (MGS) Newsletter. ILLUMINA MiSEQ NEXT GENERATION SEQUENCING A reminder, all enquiries regarding
MASSEY GENOME SERVICE SEPTEMBER 2014 NEWSLETTER Welcome to the September 2014 issue of the Massey Genome Service (MGS) Newsletter. ILLUMINA MiSEQ NEXT GENERATION SEQUENCING A reminder, all enquiries regarding
Laboratory Validation. Chapter 16
 Laboratory Validation Chapter 16 Importance The DNA profile is used to convict or free a suspect It must be perfect! No mistakes like we have in regular laboratories all the time Also DNA evidence must
Laboratory Validation Chapter 16 Importance The DNA profile is used to convict or free a suspect It must be perfect! No mistakes like we have in regular laboratories all the time Also DNA evidence must
Amplicon size (bp) E ± SD
 Development of a probe-based qpcr assay for the detection of constitutional and somatic deletions in the NF1 gene. Application to genetic testing and tumor analysis. Terribas E, Garcia-Linares C, Lázaro
Development of a probe-based qpcr assay for the detection of constitutional and somatic deletions in the NF1 gene. Application to genetic testing and tumor analysis. Terribas E, Garcia-Linares C, Lázaro
AmoyDx PIK3CA Five Mutations Detection Kit
 AmoyDx PIK3CA Five Mutations Detection Kit Detection of five mutations in PIK3CA gene Instruction for Use Instruction Version: B 1.0 Revision Date: May 2013 Store at -20±2 Manufacturer: Amoy Diagnostics
AmoyDx PIK3CA Five Mutations Detection Kit Detection of five mutations in PIK3CA gene Instruction for Use Instruction Version: B 1.0 Revision Date: May 2013 Store at -20±2 Manufacturer: Amoy Diagnostics
How to do successful gene expression analysis
 How to do successful gene expression analysis Jan Hellemans, PhD Center for Medical Genetics Biogazelle qpcr meeting June 25 th 2010 Sienna, Italy Introduction qpcr: reference technology for nucleic acid
How to do successful gene expression analysis Jan Hellemans, PhD Center for Medical Genetics Biogazelle qpcr meeting June 25 th 2010 Sienna, Italy Introduction qpcr: reference technology for nucleic acid
FAQs: PCR Polymerases from Takara Bio
 FAQs: PCR Polymerases from Takara Bio Contents: PCR Basics Q1 Q2 Q3 What parameters do I need to consider when designing primers? What is the optimum amount of template to use? Which conditions are particularly
FAQs: PCR Polymerases from Takara Bio Contents: PCR Basics Q1 Q2 Q3 What parameters do I need to consider when designing primers? What is the optimum amount of template to use? Which conditions are particularly
