Hollow, porous, solid,? Testing of Steam Sterilizers
|
|
|
- Kathryn Logan
- 6 years ago
- Views:
Transcription
1 Aspects of Steam Sterilization Process Monitoring: from Huckaback Towels to ETS and what Standards do and don't cover by Klaus Hahnen, 3M Medical Division Laboratory, Neuss, Germany Space for 3M Montage Hollow, porous, solid,? Testing of Steam Sterilizers
2 Contents Basic challenges of steam sterilization and testing principles Relevant standards Background to commonly used standard steam penetration test The hollow PCD Outlook 2
3 Space for 3M Montage Basic challenges of steam sterilization and testing principles
4 Why Process Monitoring? sterilization is a special process the result can not be tested on the end product a non-destructive, direct check of sterility is impossible YES --- it burns! 4
5 The basic challenge of a steam process If you don t get the air out, you can t get the steam in! This is why tests for air removal and steam penetration are carried out. See EN ISO (EN 554) 5
6 The challenges of a steam process Adequate steam penetration Based on adequate air removal and adequate steam quality To judge steam quality an object is necessary on which the steam can condense - the condensation point ). By this enough steam is attracted in order to accumulate NCG in such an amount that they are detectable. The condensation point can be a porous mass towel pack alternative paper based Bowie Dick Test packs or the challenge tube of the ETS 6
7 Load categories and product families 3 major groups of design characteristics: Solid Porous Hollow (or complex?) 7
8 Solid Typically found in many surgical instruments, e.g. hammer, retracting blades etc. Free access of steam to all surfaces Main influencing factors: thermal mass, steam quality NCG can form an insulating layer on the surface! 8
9 Is solid easy to sterilize? Peter Hooper: The inference may be that it is not cannulated, long, intricate instruments that will create operational difficulties. It is the orthopaedic mallet that requires our attention. Central Service, , 308 9
10 Porous Typically found with OR drapes and gowns, swabs and wound dressings Main influencing factors: air removal phase, steam quality Non-equilibrated cellulosic fibres will re-hydrate (exothermic process resulting in local superheat conditions) 10
11 Hollow (or complex?) Typically found in many surgical instruments, e.g. MIS Variety of challenges : gaps, hinges, hoses, insulation, sliding surfaces, threads Combination(s) of the above increases challenge dramatically Main influencing factors: Air removal phase, steam quality 11
12 Are temperature and pressure loggers enough? Kirk 1 has shown that pure physical measurement of pressure and temperature to judge steam quality isn t feasible and practical. A condensation point is necessary to accumulate noncondensable gases in a sufficient quantity to make a judgement. 1 B. Kirk, SGSV/EFHSS Conference 2003 Winterthur, CH 12
13 What are and why are PCD used PCD = Process Challenge Device device which challenges a process process challenge devices object which simulates the worst case of conditions for attainment of the specified sterilization conditions within the items to be sterilized See Central Service 2005/13, 320??? 13
14 Purpose of PCD create a (defined) challenge for the sterilant to reach a surface to be sterilized the overall kill capabilities of a sterilization process are defined by it s penetration and kill characteristics porous loads and torturous paths/tubes are the main designs 14
15 Porous is not Hollow! and vice versa! 15
16 The Standard Test Pack a porous PCD more than 40 years experience Lancet, 1963 Mar 16;1: The Bowie and Dick autoclave tape test, BOWIE JH, KELSEY JC, THOMPSON GR. constant development of standards (national, EN 285, EN 867-3/4, EN ISO 11140:2006) constant development of products disposable, limited reusable, 16
17 Relevance - The BDT and Your Car It is like kicking the tyre before you start the engine a quick basic test for road fitness of the car It is like watching the dashboard if no alarming light is on a reassuring test for safety 17 Btw: the German STVO (Straßenverkehrs-Ordnung) requires every driver to check the safety of his car before driving indepentenly from the TÜV test which needs to be carruied out every 2 years (= validation )
18 Tubular Test Devices hollow PCD more than 30 years experience S. J. Line and J. K. Pickerell (1973) Journal of Clinical Pathology 26, DIN test device for LTSF sterilization 18
19 Hollow test device for small steam sterilizers* EN 867-5** describes a hollow load process challenge device in the form of a PTFE tube (2mm inner diameter, 1.5m long) with an attached a capsule to contain a biological or chemical indicator. This is known as the Hollow A PCD. * EN ** Currently under revision (2008), will resurface as EN ISO
20 Adoption by EN 285 II The Hollow A test (EN 867-5) has been adopted as an additional test into EN 285. This Hollow A replaces the former Rubber Load Test of EN 285:1996. The Hollow A test complements, but does not replaces the Bowie Dick Test! According to pren ISO 17765:2, it shall be carried out at least annually, the Bowie Dick Test shall be carried out on every day the sterilizer is used. 21
21 Sterilizer testing using PCD 1950 onwards Are they representative for the items primarily sterilized at their time? Were/are they intended to be representative? 1970 onwards What are the limits of their representativeness? EN ISO (and EN 554) require every load to be monitored. 22
22 representative PCD s representativeness is limited only a syringe behaves like a syringe 23
23 Alternative Product Testing and EN ISO different types of test cycles are described: Sub-atmospheric Trans-atmospheric Super-atmospheric These are representing the real cycles found in hospital sterilizers and are used to qualify a BDT product. 3 different failure modes are described: Bad vacuum Leaks Air injection If a BDT product is not suitable for one of these defined cycles, the manufacturer has to state this. 24
24 Current Technologies incorporating the benefits of both Physical-electronic test device based on condensation load featuring hollow load calibrated against EN towel pack sensitivity non-interpretative result indication 25
25 Cycle from UK replicated in 3M research sterilizer 1 Chamber Pressure Chamber Temperature Internal temperature T5 Internal temperature T3 Thermal conductivity T5 Thermal conductivity T
26 Super atmospheric pulse converted to sub atmospheric
27 Cycle improvement easily visible
28 Outlook I Solid, hollow and porous items are and will be part of sterilizer loads Various PCD to represent product families, e.g. a porous one plus a hollow one??? Performance requirements (sensitivity level to detect failures) need to be defined for batch monitoring/load release/ Key question: What is the critical fail they need to detect? 29
29 Outlook II The user is responsible to prove his processes are safe: by validation and appropriate routine control mechanisms based on current (local) laws and applicable standards based on common sense and science Standards do not replace own thinking! 30
The Bowie and Dick Type Test Does size and penetration really matter?
 The Bowie and Dick Type Test Does size and penetration really matter? Presented by Terry McAuley Sterilisation and Infection Control Consultant Discuss the purpose of the Bowie Dick type test Examine the
The Bowie and Dick Type Test Does size and penetration really matter? Presented by Terry McAuley Sterilisation and Infection Control Consultant Discuss the purpose of the Bowie Dick type test Examine the
gke Steri-Record Testsets and PCDs for validation, process and batch monitoring of sterilization processes
 gke Steri-Record Testsets and PCDs for validation, process and batch monitoring of sterilization processes Application Process Monitoring Systems (PMS) are process challenge devices that are used to test
gke Steri-Record Testsets and PCDs for validation, process and batch monitoring of sterilization processes Application Process Monitoring Systems (PMS) are process challenge devices that are used to test
New methods to monitor steam penetration into complex medical devices (MD) using Medical Device Simulators (MDS) and Batch Monitoring Systems (BMS)
 New methods to monitor steam penetration into complex medical devices (MD) using Medical Device Simulators (MDS) and Batch Monitoring Systems (BMS) International Annual Meeting and Scientific Workshop
New methods to monitor steam penetration into complex medical devices (MD) using Medical Device Simulators (MDS) and Batch Monitoring Systems (BMS) International Annual Meeting and Scientific Workshop
New methods to monitor steam penetration into complex medical devices (MD) using Medical Device Simulators (MDS) and Batch Monitoring Systems (BMS)
 New methods to monitor steam penetration into complex medical devices (MD) using Medical Device Simulators (MDS) and Batch Monitoring Systems (BMS) WFHSS Workshop Bydgoszcz, Friday, December 7th, 2007
New methods to monitor steam penetration into complex medical devices (MD) using Medical Device Simulators (MDS) and Batch Monitoring Systems (BMS) WFHSS Workshop Bydgoszcz, Friday, December 7th, 2007
The Bowie & Dick test is used by manufacturers
 Keywords steam steriliser performance evaluation helix test Performance Evaluation of Hospital Steam Sterilisers Using the European Helix Test A.C.P. de Bruijn*, A.W. van Drongelen The Bowie & Dick test
Keywords steam steriliser performance evaluation helix test Performance Evaluation of Hospital Steam Sterilisers Using the European Helix Test A.C.P. de Bruijn*, A.W. van Drongelen The Bowie & Dick test
Lets Talk Indicators Challenging the process Sterilization. ICAN 5 th November Peter Newson
 Lets Talk Indicators Challenging the process Sterilization ICAN 5 th November Peter Newson Objectives In the context of the Decontamination process what is an indicator? What phases of the process do we
Lets Talk Indicators Challenging the process Sterilization ICAN 5 th November Peter Newson Objectives In the context of the Decontamination process what is an indicator? What phases of the process do we
MANAGEMENT AND REQUALIFICATION OF STERILISATION PROCESSES
 SUMMARY The Annexe 1 requirement for the annual requalification of sterilization processes is a huge resource requirement as well as a significant amount of downtime for many sites running complex sterilisation
SUMMARY The Annexe 1 requirement for the annual requalification of sterilization processes is a huge resource requirement as well as a significant amount of downtime for many sites running complex sterilisation
Dart Technical Bulletin. Evaluation of the Daily Air Removal Test (Dart) August 7, 1985
 Dart Technical Bulletin Evaluation of the Daily Air Removal Test (Dart) August 7, 1985 SUMMARY The data presented in this report show that Dart (Daily Air Removal Test) detects air at the same leak rate
Dart Technical Bulletin Evaluation of the Daily Air Removal Test (Dart) August 7, 1985 SUMMARY The data presented in this report show that Dart (Daily Air Removal Test) detects air at the same leak rate
Validation and Control of Porous Load Sterilisation
 Validation and Control of Porous Load Sterilisation Crown Plaza Hotel, Santry; October 14 th 2010 Gerard Sheridan 14th October 2010 Slide 1 14th October 2010 Slide 2 14th October 2010 Slide 3 Overview
Validation and Control of Porous Load Sterilisation Crown Plaza Hotel, Santry; October 14 th 2010 Gerard Sheridan 14th October 2010 Slide 1 14th October 2010 Slide 2 14th October 2010 Slide 3 Overview
Standards: Overview and Up-date
 Standards: Overview and Up-date Quality Assurance in CSSD by Compliance to Standards Klaus Hahnen, 3M, Germany Purpose of the Presentation Provide a brief introduction to standards, standardization bodies
Standards: Overview and Up-date Quality Assurance in CSSD by Compliance to Standards Klaus Hahnen, 3M, Germany Purpose of the Presentation Provide a brief introduction to standards, standardization bodies
PRODUCT CATALOGUE. for gke Steri-Record and gke Clean-Record Chemical indicators, PCDs and accessories for cleaning and sterilization
 PRODUCT CATALOGUE for gke Steri-Record and gke Clean-Record Chemical indicators, PCDs and accessories for cleaning and sterilization Bowie-Dick Simulation Test Batch and Process Monitoring Systems, Documentation
PRODUCT CATALOGUE for gke Steri-Record and gke Clean-Record Chemical indicators, PCDs and accessories for cleaning and sterilization Bowie-Dick Simulation Test Batch and Process Monitoring Systems, Documentation
Steam sterilization Process Challenge
 220 REVIEW Central Service 5/2016 Performance of Hollow Load Process Challenge Devices (HLPCDs) for the determination of air removal and steam penetration in porous load steam sterilization processes Part
220 REVIEW Central Service 5/2016 Performance of Hollow Load Process Challenge Devices (HLPCDs) for the determination of air removal and steam penetration in porous load steam sterilization processes Part
PRODUCT CATALOGUE. for gke Steri-Record and gke Clean-Record Chemical indicators, PCDs and accessories for cleaning and sterilization
 PRODUCT CATALOGUE for gke Steri-Record and gke Clean-Record Chemical indicators, PCDs and accessories for cleaning and sterilization Bowie-Dick Simulation Test Batch and Process Monitoring Systems, Documentation
PRODUCT CATALOGUE for gke Steri-Record and gke Clean-Record Chemical indicators, PCDs and accessories for cleaning and sterilization Bowie-Dick Simulation Test Batch and Process Monitoring Systems, Documentation
Hygiene Monitoring. Chemical Indicators, BD-Test and Batch- Product Information. CI_01_Eng
 Hygiene Monitoring Chemical Indicators, BD-Test and Batch- Control (PCD) Product Information CI_01_Eng verkauf@bag-healthcare.com service@bag-healthcare.com BAG-BD-Check II / BAG-BD-Check Disposable Bowie
Hygiene Monitoring Chemical Indicators, BD-Test and Batch- Control (PCD) Product Information CI_01_Eng verkauf@bag-healthcare.com service@bag-healthcare.com BAG-BD-Check II / BAG-BD-Check Disposable Bowie
Biological and chemical indicators
 Medical Market Laboratory EMEA Space for 3M Montage Biological and chemical indicators Josephus P.C.M van Doornmalen Gomez Hoyos Croatia, March 28, 2009) Outline Sterilization definitions Purpose of monitoring
Medical Market Laboratory EMEA Space for 3M Montage Biological and chemical indicators Josephus P.C.M van Doornmalen Gomez Hoyos Croatia, March 28, 2009) Outline Sterilization definitions Purpose of monitoring
Steam Quality Test Kit
 Steam Quality Test Kit Page 1 The Steam Quality Test Kit is equipped with all the component parts that are necessary to perform the required tests and report the results in accordance with ISO EN 285,
Steam Quality Test Kit Page 1 The Steam Quality Test Kit is equipped with all the component parts that are necessary to perform the required tests and report the results in accordance with ISO EN 285,
ISO INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD ISO 11140-3 Second edition 2007-03-15 Sterilization of health care products Chemical indicators Part 3: Class 2 indicator systems for use in the Bowie and Dick-type steam penetration
INTERNATIONAL STANDARD ISO 11140-3 Second edition 2007-03-15 Sterilization of health care products Chemical indicators Part 3: Class 2 indicator systems for use in the Bowie and Dick-type steam penetration
Options for load release
 Approaches to Release of Sterile Goods from Steam Sterilization: Parametric Release and Routine Monitoring Craig Wallace Senior Technical Specialist 3M Infection Prevention Division Today s Topics 1. Options
Approaches to Release of Sterile Goods from Steam Sterilization: Parametric Release and Routine Monitoring Craig Wallace Senior Technical Specialist 3M Infection Prevention Division Today s Topics 1. Options
ISO INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD ISO 11140-3 Second edition 2007-03-15 Sterilization of health care products Chemical indicators Part 3: Class 2 indicator systems for use in the Bowie and Dick-type steam penetration
INTERNATIONAL STANDARD ISO 11140-3 Second edition 2007-03-15 Sterilization of health care products Chemical indicators Part 3: Class 2 indicator systems for use in the Bowie and Dick-type steam penetration
Steam Sterilization Validation for Healthcare Institutions
 Steam Sterilization Validation for Healthcare Institutions Jonathan A. Wilder, Ph.D. President, H & W Technology, LLC/Stericert Co. Managing Director, Quality Processing Resource Group, LLC 1 Introduction
Steam Sterilization Validation for Healthcare Institutions Jonathan A. Wilder, Ph.D. President, H & W Technology, LLC/Stericert Co. Managing Director, Quality Processing Resource Group, LLC 1 Introduction
Technical Data Monograph. VERIFY All-In-One STEAM Reusable Test Pack
 Technical Data Monograph VERIFY All-In-One STEAM Reusable Test Pack Table of Contents 1. Introduction...3 2. Product....3 Use Application...3 Device Description...3 VERIFY All-In-One STEAM Reusable Test
Technical Data Monograph VERIFY All-In-One STEAM Reusable Test Pack Table of Contents 1. Introduction...3 2. Product....3 Use Application...3 Device Description...3 VERIFY All-In-One STEAM Reusable Test
CAN STERILE REALLY BE STERILE?: EFFECTIVE AUTOCLAVING METHODS
 Vet Times The website for the veterinary profession https://www.vettimes.co.uk CAN STERILE REALLY BE STERILE?: EFFECTIVE AUTOCLAVING METHODS Author : James Gasson Categories : Vets Date : February 2, 2009
Vet Times The website for the veterinary profession https://www.vettimes.co.uk CAN STERILE REALLY BE STERILE?: EFFECTIVE AUTOCLAVING METHODS Author : James Gasson Categories : Vets Date : February 2, 2009
International and European Standardization of Sterilization Processes and Equipment
 Standardization of Sterilization International and European Standardization of Sterilization Processes and Equipment Standards for sterilization in health care settings EN (16) EN ISO (29) ISO (3) ISO/TS
Standardization of Sterilization International and European Standardization of Sterilization Processes and Equipment Standards for sterilization in health care settings EN (16) EN ISO (29) ISO (3) ISO/TS
MEDICAL TRADING knows the meaning of sterilization very well: it can be
 STERILIZATION Wikipedia defines sterilization as any chemical or physical process that leads to the removal of any living organism. MEDICAL TRADING knows the meaning of sterilization very well: it can
STERILIZATION Wikipedia defines sterilization as any chemical or physical process that leads to the removal of any living organism. MEDICAL TRADING knows the meaning of sterilization very well: it can
Hong Kong Sterile Packaging Process & Packaging materials THE BACTERIAL BARRIER FOR HUMAN PROTECTION
 Hong Kong 2016 Sterile Packaging Process & Packaging materials THE BACTERIAL BARRIER FOR HUMAN PROTECTION Packaging material evolution Evolution axes of CSSD practice in Europe Technical progress Better
Hong Kong 2016 Sterile Packaging Process & Packaging materials THE BACTERIAL BARRIER FOR HUMAN PROTECTION Packaging material evolution Evolution axes of CSSD practice in Europe Technical progress Better
Endoscope Washer Disinfector Cleaning Process Challenge Devices Research & Validation
 Endoscope Washer Disinfector Cleaning Process Challenge Devices Research & Validation Richard Bancroft, B.Sc (Hons), FRSB Science & Technical Director STERIS Corporation Registered Authorising Engineer
Endoscope Washer Disinfector Cleaning Process Challenge Devices Research & Validation Richard Bancroft, B.Sc (Hons), FRSB Science & Technical Director STERIS Corporation Registered Authorising Engineer
Hong Kong Sterile Services Management Association Limited. Seminar. 25 th June 2014
 Hong Kong Sterile Services Management Association Limited Seminar 25 th June 2014 1. Bowie-Dick-Test: Use of Compact-PCD vs. Bowie-Dick-Test Pack 2. How to Monitor and Evaluate Steam Sterilization Processes
Hong Kong Sterile Services Management Association Limited Seminar 25 th June 2014 1. Bowie-Dick-Test: Use of Compact-PCD vs. Bowie-Dick-Test Pack 2. How to Monitor and Evaluate Steam Sterilization Processes
The Reusable Surgical Instrument Cycle
 Daily and Periodic Tests for Washer-Disinfectors and Sterilizers: Regulations, Technical and Operational Characteristics, and Updates on Electronic Bowie Dick Tests Richard Bancroft, B.Sc (Hons), FRSB
Daily and Periodic Tests for Washer-Disinfectors and Sterilizers: Regulations, Technical and Operational Characteristics, and Updates on Electronic Bowie Dick Tests Richard Bancroft, B.Sc (Hons), FRSB
Steam Sterilization Monitoring in the New CSA Standard (Z )
 Speaker Disclaimer Steam Sterilization Monitoring in the New CSA Standard (Z314.3-14) Rick Bauer, Ph.D. Senior Technical Services Advisor 3M Canada Employed in technical services for 3M Canada Education,
Speaker Disclaimer Steam Sterilization Monitoring in the New CSA Standard (Z314.3-14) Rick Bauer, Ph.D. Senior Technical Services Advisor 3M Canada Employed in technical services for 3M Canada Education,
Data Loggers for Routine Control. CSSD Medical Hygiene Control
 Data Loggers for Routine Control Washer-Disinfectors / Washer-Disinfectors for Endoscopes / Steam Sterilizers / Bedpan Washers CSSD Medical Hygiene Control Routine control made easy with ebro data loggers
Data Loggers for Routine Control Washer-Disinfectors / Washer-Disinfectors for Endoscopes / Steam Sterilizers / Bedpan Washers CSSD Medical Hygiene Control Routine control made easy with ebro data loggers
المملكة العربية السعودية وزارة الصحة اإلدارة العامة لمكافحة عدوى المنشئات الصحية
 1. POLICY STATEMENT: Sterilization process should be strictly monitored using, physical, chemical and biological indicator and the results of indicators should be supervised by infection control on daily
1. POLICY STATEMENT: Sterilization process should be strictly monitored using, physical, chemical and biological indicator and the results of indicators should be supervised by infection control on daily
GETINGE 633HC STEAM STERILIZERS. Always with you
 GETINGE 633HC STEAM STERILIZERS Always with you GETINGE 633HC Steam Sterilizers 3 MAKING EVERY SQUARE FOOT COUNT With the ever increasing demands being placed on today s surgical workplaces, space limitations
GETINGE 633HC STEAM STERILIZERS Always with you GETINGE 633HC Steam Sterilizers 3 MAKING EVERY SQUARE FOOT COUNT With the ever increasing demands being placed on today s surgical workplaces, space limitations
Bowie Dick Helix Test
 Technical Data Sheet Product name: Bowie Dick Helix Test Product reference: 101.201.0100 STEAM 134ºC - 3,5 min 121ºC - 15 min 101.202.0250 STEAM 134ºC - 3,5 min 121ºC - 15 min Applicable standards: ISO
Technical Data Sheet Product name: Bowie Dick Helix Test Product reference: 101.201.0100 STEAM 134ºC - 3,5 min 121ºC - 15 min 101.202.0250 STEAM 134ºC - 3,5 min 121ºC - 15 min Applicable standards: ISO
CERTIFICATE OF CONFORMANCE Mesa Laboratories, Inc. 625 Zoot Way, Bozeman, MT Phone: (303)
 Expiration : Bowie-Dick Test Sheets 77-05-CI-BD110 The ProChem Lead-Free Bowie-Dick Test Sheet is for daily monitoring of pre-vacuum sterilizers operating at 134 C for 3.5 minutes. The test sheet is designed
Expiration : Bowie-Dick Test Sheets 77-05-CI-BD110 The ProChem Lead-Free Bowie-Dick Test Sheet is for daily monitoring of pre-vacuum sterilizers operating at 134 C for 3.5 minutes. The test sheet is designed
Packaging & Sterilization
 Packaging & Sterilization Packaging & Sterilization 1 Dr. Norman Clinical Consultant General Sterilization Packaging System in Hospitals Sterilization Packaging System Disposable Reusable Crepe paper Woven
Packaging & Sterilization Packaging & Sterilization 1 Dr. Norman Clinical Consultant General Sterilization Packaging System in Hospitals Sterilization Packaging System Disposable Reusable Crepe paper Woven
QUALITY ASSURANCE IN AN MDRD
 QUALITY ASSURANCE IN AN MDRD MAINTENANCE AND PREVENTATIVE MAINTENANCE MDRD is a machine dependent department washers, pasteurizers, cart washer, sterilizer, ultrasonic, heat sealers, RO water systems All
QUALITY ASSURANCE IN AN MDRD MAINTENANCE AND PREVENTATIVE MAINTENANCE MDRD is a machine dependent department washers, pasteurizers, cart washer, sterilizer, ultrasonic, heat sealers, RO water systems All
S.M.A.R.T.Green Test Pack for Prevacuum Sterilizers Technology Overview
 Technology Overview S.M.A.R.T.Green Test Pack for Prevacuum Sterilizers Technology Overview Getinge Assure S.M.A.R.T.Green Test Pack is a lead, latex, heavy metal, and toxic metal free Bowie and Dick test
Technology Overview S.M.A.R.T.Green Test Pack for Prevacuum Sterilizers Technology Overview Getinge Assure S.M.A.R.T.Green Test Pack is a lead, latex, heavy metal, and toxic metal free Bowie and Dick test
A UT OCL A VE VE OCLA UT A _AutoClave_D1102EN_1703.indd /03/01 13:45
 228 229 More Safety, More Capacity Optimizing Handpiece Performance with The Long-Awaited Debut of NSK s Autoclave Series Even the best handpiece means nothing if you cannot use and decontaminate it safely.
228 229 More Safety, More Capacity Optimizing Handpiece Performance with The Long-Awaited Debut of NSK s Autoclave Series Even the best handpiece means nothing if you cannot use and decontaminate it safely.
6 Handling, collection, and transport of contaminated items General rationale Separation of waste and reusable items at point of
 Contents Glossary of equivalent standards... ix Committee representation... xi Acknowledgments...xiii Foreword... xiv Introduction: Need for the recommended practice...1 1 Scope...5 1.1 General...5 1.2
Contents Glossary of equivalent standards... ix Committee representation... xi Acknowledgments...xiii Foreword... xiv Introduction: Need for the recommended practice...1 1 Scope...5 1.1 General...5 1.2
SP MEDIKAL NEWS. We do hope, despite the economic situation, we again will gain a few distributors.
 SP MEDIKAL NEWS Fifth newsletter by SP Medikal San. Ltd In this Edition: Another year, another booth. It is Medica time!!! A 50 sqm big booth was our showroom for only 4 days. We feel blessed by the many
SP MEDIKAL NEWS Fifth newsletter by SP Medikal San. Ltd In this Edition: Another year, another booth. It is Medica time!!! A 50 sqm big booth was our showroom for only 4 days. We feel blessed by the many
Data Loggers for Routine Control. CSSD Hygiene Control
 Data Loggers for Routine Control Washer-Disinfectors / Washer-Disinfectors for Endoscopes / Steam Sterilizers / Bedpan Washers CSSD Hygiene Control Electronic Bowie Dick Test System Together with the evaluation
Data Loggers for Routine Control Washer-Disinfectors / Washer-Disinfectors for Endoscopes / Steam Sterilizers / Bedpan Washers CSSD Hygiene Control Electronic Bowie Dick Test System Together with the evaluation
Instructions for Validation of Autoclaves
 Instructions for Validation of Autoclaves A. Validation of autoclaves may accomplished by the following methods: 1. Autoclave tape applied to the outside of all packs to validate that the correct temperature
Instructions for Validation of Autoclaves A. Validation of autoclaves may accomplished by the following methods: 1. Autoclave tape applied to the outside of all packs to validate that the correct temperature
Bulk Steam Autoclaves. Laboratory & Scientific Line
 Bulk Steam Autoclaves Laboratory & Scientific Line Bulk Steam Autoclaves for Life Sciences The bulk autoclave line is designed for the sterilization of laboratory products and instruments including cages,
Bulk Steam Autoclaves Laboratory & Scientific Line Bulk Steam Autoclaves for Life Sciences The bulk autoclave line is designed for the sterilization of laboratory products and instruments including cages,
Steam Sterilizer. SA Series. for hospitals, CSSD and medical centers
 Steam Sterilizer SA Series for hospitals, CSSD and medical centers Driven by customer needs Colussi is a leading infection control solution provider, supplying the healthcare, laboratory research and pharma
Steam Sterilizer SA Series for hospitals, CSSD and medical centers Driven by customer needs Colussi is a leading infection control solution provider, supplying the healthcare, laboratory research and pharma
6 Handling, collection, and transport of contaminated items General rationale Separation of waste and reusable items at point of
 Contents Glossary of equivalent standards... ix Committee representation... xi Acknowledgments...xiii Background on Amendment 1...xiii Foreword... xiv Introduction: Need for the recommended practice...1
Contents Glossary of equivalent standards... ix Committee representation... xi Acknowledgments...xiii Background on Amendment 1...xiii Foreword... xiv Introduction: Need for the recommended practice...1
STERILIZATION DEVICES
 STERILIZATION DEVICES I. Central Sterilization Units II. Prismatic Steam Sterilizer III. Washing Steam Sterilizer IV. Laboratory Type Vertical Steam Sterilizer 2195 13485 14001 9001 About us Sümer A.Ş.
STERILIZATION DEVICES I. Central Sterilization Units II. Prismatic Steam Sterilizer III. Washing Steam Sterilizer IV. Laboratory Type Vertical Steam Sterilizer 2195 13485 14001 9001 About us Sümer A.Ş.
How PrePak Supports the Process of Converting Hospitals From Disposable to Reusable Surgical Linen Packs
 Case Study Title: Author: How PrePak Supports the Process of Converting Hospitals From Disposable to Reusable Surgical Linen Packs Jim Sprout PrePak Business Manager BACKGROUND & PURPOSE Standard Textile
Case Study Title: Author: How PrePak Supports the Process of Converting Hospitals From Disposable to Reusable Surgical Linen Packs Jim Sprout PrePak Business Manager BACKGROUND & PURPOSE Standard Textile
Mediclave Autoclaves 6, 18 & 23 Litre B, S & N Class
 Mediclave Autoclaves 6, 18 & 23 Litre B, S & N Class LTE Mediclave range of autoclaves are some of the most comprehensively equipped medical steam sterilizers available. Designed to incorporate features
Mediclave Autoclaves 6, 18 & 23 Litre B, S & N Class LTE Mediclave range of autoclaves are some of the most comprehensively equipped medical steam sterilizers available. Designed to incorporate features
TOP FIVE TIPS. Autoclave Validation. Create & Maintain Clear Validation Protocols. Begin Your Regimen with a Bowie-Dick Test
 Autoclave Validation TOP FIVE TIPS Time and Money Saving Validation Strategies 1 2 Create & Maintain Clear Validation Protocols Begin Your Regimen with a Bowie-Dick Test 3 Use Autoclave-Friendly Data Loggers
Autoclave Validation TOP FIVE TIPS Time and Money Saving Validation Strategies 1 2 Create & Maintain Clear Validation Protocols Begin Your Regimen with a Bowie-Dick Test 3 Use Autoclave-Friendly Data Loggers
COLLEGE OF PHARMACY STERILE PRODUCTS PHT 434. Dr. Mohammad Javed Ansari, PhD. Contact info:
 COLLEGE OF PHARMACY STERILE PRODUCTS PHT 434 Dr. Mohammad Javed Ansari, PhD. Contact info: javedpharma@gmail.com OBJECTIVES OF THE LECTURE At the end of this lecture, you will be aware of: What are Sterile
COLLEGE OF PHARMACY STERILE PRODUCTS PHT 434 Dr. Mohammad Javed Ansari, PhD. Contact info: javedpharma@gmail.com OBJECTIVES OF THE LECTURE At the end of this lecture, you will be aware of: What are Sterile
They can be used on washer-disinfectors, ultrasonic cleaners, surgical instruments and endoscopes. Page 5
 Rev. 0 October 2017 Terragene offers a wide range of simple and effective products to precisely control the washing, hygiene and sterilization processes in veterinary clinics. Available in a variety of
Rev. 0 October 2017 Terragene offers a wide range of simple and effective products to precisely control the washing, hygiene and sterilization processes in veterinary clinics. Available in a variety of
The Significance of Validation to US CSSD Managers
 The Significance of Validation to US CSSD Managers Jonathan A. Wilder, Ph.D. President, H & W Technology, LLC/Stericert Co. Managing Director, Quality Processing Resource Group, LLC 1 Introduction This
The Significance of Validation to US CSSD Managers Jonathan A. Wilder, Ph.D. President, H & W Technology, LLC/Stericert Co. Managing Director, Quality Processing Resource Group, LLC 1 Introduction This
E9 Next. Taking Class B sterilization to the next level. Classification:
 E9 Next Taking Class B sterilization to the next level E9 Next combines stunning design with a new traceability concept, which is expandable also thanks to the three Print Sets that can be added at any
E9 Next Taking Class B sterilization to the next level E9 Next combines stunning design with a new traceability concept, which is expandable also thanks to the three Print Sets that can be added at any
New modern comprehensive solution siemens.cz/waste
 Hazardous waste New modern comprehensive solution siemens.cz/waste How to beat the problem of disposal of infectious waste Filling Grinding Evaporation Heating Waste is placed into the operating chamber
Hazardous waste New modern comprehensive solution siemens.cz/waste How to beat the problem of disposal of infectious waste Filling Grinding Evaporation Heating Waste is placed into the operating chamber
E9 Med. The strength of simplicity. Classification:
 E9 Med The strength of simplicity Simple, effective and compact, E9 Med is an autoclave that makes Class B sterilization accessible to everyone. Hidden behind its simple, user-friendly interface is an
E9 Med The strength of simplicity Simple, effective and compact, E9 Med is an autoclave that makes Class B sterilization accessible to everyone. Hidden behind its simple, user-friendly interface is an
ISO INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD ISO 11140-5 Second edition 2007-03-15 Sterilization of health care products Chemical indicators Part 5: Class 2 indicators for Bowie and Dicktype air removal tests Stérilisation
INTERNATIONAL STANDARD ISO 11140-5 Second edition 2007-03-15 Sterilization of health care products Chemical indicators Part 5: Class 2 indicators for Bowie and Dicktype air removal tests Stérilisation
 Rev. 1 May 2018 Terragene offers a range of effective and easy-to-handle products to control the washing, hygiene, disinfection and sterilization processes in medical diagnostic laboratories. Available
Rev. 1 May 2018 Terragene offers a range of effective and easy-to-handle products to control the washing, hygiene, disinfection and sterilization processes in medical diagnostic laboratories. Available
ANSI/AAMI ST79 A2:2009 Key Changes in the 2009 Amendment
 3M Attest TM TM Products Sterile U Network T U T O R I A L S ANSI/AAMI ST79 A2:2009 Key Changes in the 2009 Amendment Background: A second amendment (A2:2009) to the Association for the Advancement of
3M Attest TM TM Products Sterile U Network T U T O R I A L S ANSI/AAMI ST79 A2:2009 Key Changes in the 2009 Amendment Background: A second amendment (A2:2009) to the Association for the Advancement of
Sirona competency in hygiene DAC PROFESSIONAL. Hygiene systems. Vacuum autoclave class B to satisfy the most demanding requirements
 Sirona competency in hygiene Hygiene systems Vacuum autoclave class B to satisfy the most demanding requirements Professional and fast Sirona s is an autoclave which satisfies the most demanding medical
Sirona competency in hygiene Hygiene systems Vacuum autoclave class B to satisfy the most demanding requirements Professional and fast Sirona s is an autoclave which satisfies the most demanding medical
Monitoring your decontamination and reprocessing cycle
 Monitoring your decontamination and reprocessing cycle 1er Journee National sur la Sterilisation La Societe Marocaine de Sterilisation Maroc 2011 Ahmed Ghatwary/SteriGMS/Pharcomedic Index 1-Cleaning step,
Monitoring your decontamination and reprocessing cycle 1er Journee National sur la Sterilisation La Societe Marocaine de Sterilisation Maroc 2011 Ahmed Ghatwary/SteriGMS/Pharcomedic Index 1-Cleaning step,
Written By Dr. Robert Ramaley (Univ. NE Med CTR), Mia Ware, Dan Pearson, and Christopher R. Dwyer* (Raven Biological Laboratories)
 Performance Comparison for Three Types of Biological Indicators Used in Steam Sterilization Processes: Spore Strips, Crushable Self-contained, and Sealed Glass Ampoule Written By Dr. Robert Ramaley (Univ.
Performance Comparison for Three Types of Biological Indicators Used in Steam Sterilization Processes: Spore Strips, Crushable Self-contained, and Sealed Glass Ampoule Written By Dr. Robert Ramaley (Univ.
Putting Confidence in Your Hands.
 Putting Confidence in Your Hands. 3M Sterilization Products and Services Full Line Catalogue 1 Trusted 3M Sterilization Solutions Equipment Monitoring Exposure Monitoring Pack Monitoring Load Monitoring
Putting Confidence in Your Hands. 3M Sterilization Products and Services Full Line Catalogue 1 Trusted 3M Sterilization Solutions Equipment Monitoring Exposure Monitoring Pack Monitoring Load Monitoring
SERCON 300 STEAM STERILIZERS. Full steam ahead. Reliable, flexible & efficient solutions. Central Sterile Services
 Central Sterile Services SERCON 300 STEAM STERILIZERS Reliable, flexible & efficient solutions Full steam ahead One Integrated Approach to Healthcare Throughput and choice Seven sizes standard and fast
Central Sterile Services SERCON 300 STEAM STERILIZERS Reliable, flexible & efficient solutions Full steam ahead One Integrated Approach to Healthcare Throughput and choice Seven sizes standard and fast
Advantages. Steam sterilization
 Rev. 1 November 2016 Steam sterilization Sterilization is the total destruction of all microorganisms, whether or not pathogenic, and their spores. Moist heat in the form of saturated steam under pressure
Rev. 1 November 2016 Steam sterilization Sterilization is the total destruction of all microorganisms, whether or not pathogenic, and their spores. Moist heat in the form of saturated steam under pressure
Advantages. Steam sterilization
 Rev. 2 March 2017 Steam sterilization Sterilization is the total destruction of all microorganisms, whether or not pathogenic, and their spores. Moist heat in the form of saturated steam under pressure
Rev. 2 March 2017 Steam sterilization Sterilization is the total destruction of all microorganisms, whether or not pathogenic, and their spores. Moist heat in the form of saturated steam under pressure
Facility: Sterile Processing Assessment Date: CRITERIA ANSI/AAMI COMPLIANT Number Y N N/A Point of Use Gross contaminant is removed with
 Point of Use 6.2-6.3 Gross contaminant is removed with water Instruments are sorted Sharps removed Instruments placed back in original container Un-used instruments placed in bottom of basket Towel placed
Point of Use 6.2-6.3 Gross contaminant is removed with water Instruments are sorted Sharps removed Instruments placed back in original container Un-used instruments placed in bottom of basket Towel placed
INSTRUMENT CLEANING & STERILISATION FOAMING BATH FORMULATION. Surgistain. Rapid Multi-Enzyme Cleaner. Kimguard Sterile Wrap. Instrument brushes $32.
 1Ltr Rapid Multi-Enzyme Cleaner Ÿ Proprietary combination enzyme Ÿ concentrate Ÿ Unparalleled combination of speed Ÿ and efficacy Ÿ Fully effective against proteins, Ÿ carbohydrates, lipids and Ÿ mucopolysaccharides
1Ltr Rapid Multi-Enzyme Cleaner Ÿ Proprietary combination enzyme Ÿ concentrate Ÿ Unparalleled combination of speed Ÿ and efficacy Ÿ Fully effective against proteins, Ÿ carbohydrates, lipids and Ÿ mucopolysaccharides
LEADING TECHNOLOGY IN STERILIZATION
 LEADING TECHNOLOGY IN STERILIZATION E9 RECORDER E9 MED E5 FAST, EFFICIENT, USER-FRIENDLY B TYPE VACUUM AND NON-VACUUM AUTOCLAVES Medisafe International, Twyford Road, Bishop s Stortford, Hertfordshire
LEADING TECHNOLOGY IN STERILIZATION E9 RECORDER E9 MED E5 FAST, EFFICIENT, USER-FRIENDLY B TYPE VACUUM AND NON-VACUUM AUTOCLAVES Medisafe International, Twyford Road, Bishop s Stortford, Hertfordshire
Plasma Low Temperature Sterilization. With vaporized hydrogen peroxide. PlazMax Line
 Plasma Low Temperature Sterilization With vaporized hydrogen peroxide PlazMax Line Low Temperature Sterilization PlazMax Line For heat and moisture sensitive equipment PlazMax provides an efficient sterilization
Plasma Low Temperature Sterilization With vaporized hydrogen peroxide PlazMax Line Low Temperature Sterilization PlazMax Line For heat and moisture sensitive equipment PlazMax provides an efficient sterilization
Active Learning and Lab Activity Bioman Conference 2014
 Active Learning and Lab Activity Bioman Conference 2014 A. What is validation? Establishing documented evidence which provides a high degree of assurance that a specific process will consistently produce
Active Learning and Lab Activity Bioman Conference 2014 A. What is validation? Establishing documented evidence which provides a high degree of assurance that a specific process will consistently produce
Wireless Data Logger System
 Wireless Data Logger System for Thermal Process Validation Medical / CSSD / Pharmaceutical / Food Flexible Data Logger System For Thermal Validations ebro offers a flexible measurement and documentation
Wireless Data Logger System for Thermal Process Validation Medical / CSSD / Pharmaceutical / Food Flexible Data Logger System For Thermal Validations ebro offers a flexible measurement and documentation
E10. The must of Euronda sterilization, in one single autoclave
 E10 The must of Euronda sterilization, in one single autoclave E10 is the latest Euronda Class B autoclave. A more powerful vacuum pump and an optimized drying cycle make E10 the best performing Euronda
E10 The must of Euronda sterilization, in one single autoclave E10 is the latest Euronda Class B autoclave. A more powerful vacuum pump and an optimized drying cycle make E10 the best performing Euronda
Manner of Assessment Code (check all that apply) & Surveyor Notes. Yes N/A. Yes. Yes N/A N/A. Yes N/A. N/A Yes N/A
 Section 4. L Reprocessing of Semi-Critical Equipment Elements to be assessed Manner of Assessment Code (check all that apply) & Surveyor Notes Manner of Assessment Code (check all that apply) & Surveyor
Section 4. L Reprocessing of Semi-Critical Equipment Elements to be assessed Manner of Assessment Code (check all that apply) & Surveyor Notes Manner of Assessment Code (check all that apply) & Surveyor
Contact: To sterilize or decontaminate uniformly, superheated steam must contact all areas of the load.
 Published on UC Davis Safety Services (https://safetyservices.ucdavis.edu) Effective Use of Autoclaves SafetyNet #: 26 Autoclave Use Autoclaves (steam sterilizers) are metal pressure vessels that are used
Published on UC Davis Safety Services (https://safetyservices.ucdavis.edu) Effective Use of Autoclaves SafetyNet #: 26 Autoclave Use Autoclaves (steam sterilizers) are metal pressure vessels that are used
Best Practice Monitoring for Sterile Processing Professionals
 Best Practice Monitoring for Sterile Processing Professionals Sterile processing departments are faced with greater challenges than ever. Increasing demands for quick turnarounds. Pressure to streamline
Best Practice Monitoring for Sterile Processing Professionals Sterile processing departments are faced with greater challenges than ever. Increasing demands for quick turnarounds. Pressure to streamline
Effective Use of Autoclaves
 Published on UC Davis Safety Services (https://safetyservices.ucdavis.edu) Effective Use of Autoclaves SafetyNet #: 26 Autoclave Use Autoclaves (steam sterilizers) are metal pressure vessels that are used
Published on UC Davis Safety Services (https://safetyservices.ucdavis.edu) Effective Use of Autoclaves SafetyNet #: 26 Autoclave Use Autoclaves (steam sterilizers) are metal pressure vessels that are used
Product and Package Stability Studies The Application of FDA Guidance
 Product and Package Stability Studies The Application of FDA Guidance Introduction In February 2008, the FDA issued Guidance to Industry dealing with physical tests for packaging for sterile medical devices
Product and Package Stability Studies The Application of FDA Guidance Introduction In February 2008, the FDA issued Guidance to Industry dealing with physical tests for packaging for sterile medical devices
Data Logger Systems. Medical CSSD Pharmaceutical Food
 Data Logger Systems for Thermal Process Validation Medical CSSD Pharmaceutical Food Flexible Data Logger Systems For Thermal Validations ebro offers flexible measurement and documentation systems for validating
Data Logger Systems for Thermal Process Validation Medical CSSD Pharmaceutical Food Flexible Data Logger Systems For Thermal Validations ebro offers flexible measurement and documentation systems for validating
Steam Sterilizers. DLOV & DLOM series. more than sixty years of experience in sterilization. DE LAMA s Quality Management System is certified:
 Steam Sterilizers DLOV & DLOM series DE LAMA s Quality Management System is certified: more than sixty years of experience in sterilization UNI EN ISO 9001 - Cert. n 0351 UNI EN ISO 13485 - Cert. n 1275
Steam Sterilizers DLOV & DLOM series DE LAMA s Quality Management System is certified: more than sixty years of experience in sterilization UNI EN ISO 9001 - Cert. n 0351 UNI EN ISO 13485 - Cert. n 1275
The Production of Electricity Power from Natural Gas. Image Source: Mscalora
 The Production of Electricity Power from Natural Gas Image Source: Mscalora How does it work? Natural Gas can be converted to Electricity using a Gas Turbine Power Plant While gas turbines can power up
The Production of Electricity Power from Natural Gas Image Source: Mscalora How does it work? Natural Gas can be converted to Electricity using a Gas Turbine Power Plant While gas turbines can power up
Not to be reproduced - AGPAL & QIP
 Keep it Clean! Practical methods for implementing correct sterilisation processes advanced level Presented by: Lin Lochead, B. App. Sc Validation A Documented Procedure for obtaining, recording and interpreting
Keep it Clean! Practical methods for implementing correct sterilisation processes advanced level Presented by: Lin Lochead, B. App. Sc Validation A Documented Procedure for obtaining, recording and interpreting
Process Filtration From Pure to Sterile
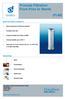 From Pure to Sterile MAIN FEATURES & BENEFITS: High mechanical and thermal stability Excellent flow rate Integrity testable according to HIMA Thermal stability up to 200 C Approved for Food Contact Use
From Pure to Sterile MAIN FEATURES & BENEFITS: High mechanical and thermal stability Excellent flow rate Integrity testable according to HIMA Thermal stability up to 200 C Approved for Food Contact Use
VALIDATION OF BUNG PROCESSOR
 243 P a g e International Standard Serial Number (ISSN): 2319-8141 International Journal of Universal Pharmacy and Bio Sciences 6(2): March-April 2017 INTERNATIONAL JOURNAL OF UNIVERSAL PHARMACY AND BIO
243 P a g e International Standard Serial Number (ISSN): 2319-8141 International Journal of Universal Pharmacy and Bio Sciences 6(2): March-April 2017 INTERNATIONAL JOURNAL OF UNIVERSAL PHARMACY AND BIO
STERILIZATION ON MSF EMERGENGY SETTING. Pascale CHOQUENET Médecins Sans Frontières Paris France Milan 5th June 2008
 STERILIZATION ON MSF EMERGENGY SETTING Pascale CHOQUENET Médecins Sans Frontières Paris France Milan 5th June 2008 MSF = Doctors Without Borders International Humanitarian Organisation Medical NGO (Non
STERILIZATION ON MSF EMERGENGY SETTING Pascale CHOQUENET Médecins Sans Frontières Paris France Milan 5th June 2008 MSF = Doctors Without Borders International Humanitarian Organisation Medical NGO (Non
Getinge HS55 Hospital sterilizer
 Getinge HS55 Hospital sterilizer The most versatile, reliable and Easy-to-use hospital sterilizer in the world The Getinge HS55 is a fully automatic steam sterilizer. It has pre-set programs for the most
Getinge HS55 Hospital sterilizer The most versatile, reliable and Easy-to-use hospital sterilizer in the world The Getinge HS55 is a fully automatic steam sterilizer. It has pre-set programs for the most
Make acquaintance with our further offers...
 For more information see our internet web site Medium-sized steam sterilizer for health care Make acquaintance with our further offers... Small steam sterilizers 15 25 litre Steam sterilizers 140 1 490
For more information see our internet web site Medium-sized steam sterilizer for health care Make acquaintance with our further offers... Small steam sterilizers 15 25 litre Steam sterilizers 140 1 490
Biological Monitoring
 Biological Monitoring Prof. Duygu PERÇİN, MD Department of Clinical Microbiology Erciyes University Faculty of Medicine, Kayseri-TURKEY duygu.percin@hotmail.com Learning Objectives The attandee will be
Biological Monitoring Prof. Duygu PERÇİN, MD Department of Clinical Microbiology Erciyes University Faculty of Medicine, Kayseri-TURKEY duygu.percin@hotmail.com Learning Objectives The attandee will be
Medical Device Reprocessing Quality Assurance. 3M All Rights Reserved.
 Medical Device Reprocessing Quality Assurance Additional Content Qualification Testing Flash Sterilization Extended Cycles Recalls Wet Packs Finish 2 Qualification Testing and Required Monitoring Installation
Medical Device Reprocessing Quality Assurance Additional Content Qualification Testing Flash Sterilization Extended Cycles Recalls Wet Packs Finish 2 Qualification Testing and Required Monitoring Installation
STERIVAP. Excellent Price High Quality Engineering. protecting human health. Large steam sterilizer for health care
 STERIVAP Large steam sterilizer for health care Excellent Price High Quality Engineering protecting human health MMM Group leading service supplier for health care Since 1954 the MMM Group has been active
STERIVAP Large steam sterilizer for health care Excellent Price High Quality Engineering protecting human health MMM Group leading service supplier for health care Since 1954 the MMM Group has been active
Determination of the Effect of Superheat on Class 6 Indicators Report No 286/
 Determination of the Effect of Superheat on Class 6 Indicators Report No 286/728424 Testing conducted by BSI Product Services, Hertfordshire, Great Britain 1. PURPOSE This study is intended to determine
Determination of the Effect of Superheat on Class 6 Indicators Report No 286/728424 Testing conducted by BSI Product Services, Hertfordshire, Great Britain 1. PURPOSE This study is intended to determine
Resistance Temperature Detectors Mineral Insulated Thermocouples Thermoelements, Accessories and Service All-round supplier
 Resistance Temperature Detectors Mineral Insulated Thermocouples Thermoelements, Accessories and Service All-round supplier of temperature sensors and component parts for all industrial applications Manufactured
Resistance Temperature Detectors Mineral Insulated Thermocouples Thermoelements, Accessories and Service All-round supplier of temperature sensors and component parts for all industrial applications Manufactured
Validation and Complete Routine Control Sets
 Validation and Routine Control Sets Complete Validation Sets Complete Routine Control Sets Description: Validation data loggers with evaluation software and an extensive range of accessories German TÜV
Validation and Routine Control Sets Complete Validation Sets Complete Routine Control Sets Description: Validation data loggers with evaluation software and an extensive range of accessories German TÜV
Meeting the challenges of medical device packaging
 Meeting the challenges of medical device packaging Gisela Cristina Mendes Bastos Viegas, S.A. October 14, 2011 Agenda Evaluation and selection Validation Test methods Sterile Barrier Systems EN 868 Parts
Meeting the challenges of medical device packaging Gisela Cristina Mendes Bastos Viegas, S.A. October 14, 2011 Agenda Evaluation and selection Validation Test methods Sterile Barrier Systems EN 868 Parts
VARIO XX28 Stainless Steel Time And Temperature Controlled Electric Model
 VARIO XX28 Stainless Steel Time And Temperature Controlled Electric Model Operators Manual Issue 5 For correct operation of this autoclave it is essential that the chamber be filled with water up to the
VARIO XX28 Stainless Steel Time And Temperature Controlled Electric Model Operators Manual Issue 5 For correct operation of this autoclave it is essential that the chamber be filled with water up to the
INSTRUMENT PROCESSING FROM PREP/PACKAGING TO STORAGE. January 2016
 INSTRUMENT PROCESSING FROM PREP/PACKAGING TO STORAGE January 2016 Disclosures The opinions expressed in this presentation are those of the author and do not necessarily reflect the official position of
INSTRUMENT PROCESSING FROM PREP/PACKAGING TO STORAGE January 2016 Disclosures The opinions expressed in this presentation are those of the author and do not necessarily reflect the official position of
ST 19 Aluminium Manually Controlled Electric Model
 ST 19 Aluminium Manually Controlled Electric Model Operators Manual Issue 5 For correct operation of this autoclave it is essential that the chamber be filled with water up to the height of the studs before
ST 19 Aluminium Manually Controlled Electric Model Operators Manual Issue 5 For correct operation of this autoclave it is essential that the chamber be filled with water up to the height of the studs before
UNISTERI HP. Medium-sized Steam Sterilizer for Health Care - Efficient, Intelligent, Exceptional. protecting human health
 Medium-sized Steam Sterilizer for Health Care - Efficient, Intelligent, Exceptional protecting human health MMM Group Leading Service Supplier for Health Care Individual Assembly of The Sterilizer General
Medium-sized Steam Sterilizer for Health Care - Efficient, Intelligent, Exceptional protecting human health MMM Group Leading Service Supplier for Health Care Individual Assembly of The Sterilizer General
EBI 12 The new data logger generation
 EBI 12 The new data logger generation 3 EBI 12 The new data logger generation High quality stainless steel housing Application range from -90 C to 150 C High temperature accuracy up to 0.05 C Extended
EBI 12 The new data logger generation 3 EBI 12 The new data logger generation High quality stainless steel housing Application range from -90 C to 150 C High temperature accuracy up to 0.05 C Extended
LARGE CAPACITY PHARMACEUTICAL AUTOCLAVES
 LARGE CAPACITY PHARMACEUTICAL AUTOCLAVES www.eryigit.com.tr Our Vision Our vision is to make Eryiğit an example among its class on its way to become the first medical device manufacturer that comes to
LARGE CAPACITY PHARMACEUTICAL AUTOCLAVES www.eryigit.com.tr Our Vision Our vision is to make Eryiğit an example among its class on its way to become the first medical device manufacturer that comes to
Welcome! How do I get a CE Certificate?
 M Sterile U Network M Sterile U Web Meeting November 0, 0 Today s meeting times: 9:00 a.m., :00 a.m., and :00 p.m. Central Standard Time To hear audio, call 800-97-00 and enter access code 7 Phone lines
M Sterile U Network M Sterile U Web Meeting November 0, 0 Today s meeting times: 9:00 a.m., :00 a.m., and :00 p.m. Central Standard Time To hear audio, call 800-97-00 and enter access code 7 Phone lines
