They can be used on washer-disinfectors, ultrasonic cleaners, surgical instruments and endoscopes. Page 5
|
|
|
- Delilah Lloyd
- 6 years ago
- Views:
Transcription
1 Rev. 0 October 2017
2
3 Terragene offers a wide range of simple and effective products to precisely control the washing, hygiene and sterilization processes in veterinary clinics. Available in a variety of formats, these high performance control devices provide fast and consistent results in the control of veterinary infections. Page 3
4 Nowadays, ultrasonic washing machines are a fundamental tool for mechanical cleaning of veterinary instruments. The ultrasonic washing takes place through a process called cavitation, a microscopic phenomenon responsible for the removal of soil from the instruments. Cavitation energy can not be measured and depends on the integration of a complex series of parameters, thus the use of Chemdye CDWU indicator becomes indispensable for the performance control of the ultrasonic washing machines. Chemdye CDWU indicator consists of a transparent vial, with a blue-colored reactive solution and glass beads immersed in it. During cavitation, vibration of the glass pearls triggers a color change in the solution, from blue to yellow, through a range of green color intermediates. When cavitation energy is high enough to guarantee a correct washing of the instruments, the final result will be a yellow coloration, otherwise the indicator will remain greenish, which will show a weak cavitation zone located in that area of the washing machine. Page 4
5 For control of hygiene instruments and endoscopes for veterinary use, Terragene offers the Chemdye KPRO2-E69 and KPRO2-E250 Protein Monitoring Systems, designed to test the correct cleaning of materials by detecting protein residues. The systems have a high absorption swab, allowing the collection of samples from different surfaces with the same efficiency. These are compatible with cleaning verification of endoscopes for veterinary use and other reusable cannulated instruments. They can be used on washer-disinfectors, ultrasonic cleaners, surgical instruments and endoscopes. Page 5
6 Biological Indicators (BIs) are the only devices internationally accepted as direct evidence of microbial lethality after a sterilization cycle. Terragene offers its line of Conventional Self-Contained BIs to control the sterilization processes of veterinary materials and instruments. Its innovation technology allows results to be obtained within 24 hours (Vapor and VH2O2) or 48 hours (Ethylene Oxide and Dry Heat). In addition, Terragene offers a line of Rapid Colorimetric BIs, which ensure reliable results in shorter times, 8 hs (BT91, BT200) and 10 hrs (BT202). In orden to incubate Conventional and Rapid Colorimetric Self-Contained BIs, Terragene offers the Bionova IC10/20 Dual Incubator, which provides optimal conditions for accurate readout of a wide range of biological indicators (both at 37 C and 60 C) due to its block heater, furthermore allows to incubate culture media and biological ampoules for sterilization of liquid loads. This incubator has a hole for external temperature control (Bionova TB-IC1020). Page 6
7 Bowie & Dick Test Pack Chemdye Bowie & Dick Test Pack was developed to control air removal and steam penetration performance in vacuum-assisted steam sterilizers. The product Chemdye BD125X/2 is a single-use device which consist of a lead-free chemical indicator, placed between porous sheets of paper, wrapped with crepe paper, with a steam indicator label (Type 1) on the top of the package. This test pack must be routinely used in the first cycle of the sterilizer, to ensure performance of the sterilizer in the rest of the loaded cycles, provided the result is accepted. Bowie & Dick Test Card Chemdye Bowie & Dick Test Card has been designed to monitor the effectiveness of air removal in vacuum-assisted steam sterilizers at 132 C, 4 minutes and at 134 ºC, 3.5 minutes. This card consists of a Type 2 Metals free chemical indicator that changes from purple to green when processed. An heterogeneous color change, i.e the presence of purple/grey color, indicates presence of an air pocket during the sterilization cycle thus indicating sterilizer malfunction. This warns the user that it is not advisable to use the sterilizer to process the load. BD8948H is a stainless steel re-usable Holder for Bowie & Dick Test Card. The holder keeps BD8948X Test Card in place for proper assessment of sterilization cycle. Page 7
8 Self-adhesive tapes Cintape Self-adhesive tapes have been designed to wrap and seal sterilization packages as well as to distinguish between items that have been exposed to Steam sterilization processes from those that have not. Terragene has tapes for every sterilization process: Steam (CT22), Ethylene Oxide (CT10), Plasma or Vaporized Hydrogen Peroxide (CT40) and Dry Heat (CT30). Self-adhesive labels for automatic record system Chemdye Automatic record system labels have been designed to monitor Ethylene Oxide (CD13), Steam (CD23), Dry Heat (CD33) and Plasma or Vaporized Hydrogen Peroxide (CD43) sterilization processes. These self-adhesive labels are usually placed out of the plate, stuck to packages or pouches, allowing differentiation between processed and unprocessed items. Their double-adhesive technology allows easy label removal from the sterilization package for data documentation. Chemdye CG3 is a Three-line Automatic Labeler that allows a quick and easy labelling of sterilization packages through the use of special documentation labels, preventing the traceability errors of the packs. Page 8
9 CD CD40 Gas Plasma or lighter Multi-variable indicators for internal use Simple and double strips Chemdye Type 4 Internal control strips are Multi-variable Indicators that rapidly show if critical parameters of the sterilization process have been reached, besides ensuring sterilizing agent appropriate penetration of the items inside the packages. This chemical indicators show a significant colour change when exposed to the stated values (SVs) of the critical process variables. Terragene offers various presentations of Type 4 Strips, according to the needs of the users. or lighter Contents Initials Date Multi-variable Indicator Contents Initials D ate CD CD15 Multi-variable Indicator CD16 EO or lighter or darker or darker or darker STEAM CD Contents Initials Date Multi-variable Indicator Contents Initials Date STEAM Multi-variable Indicator CD CD25 CD29 ISO Type 4 CD Contents Initials D ate STEAM Multi-variable indicator CD202 CD CD15 Multi-variable Indicator CD Contents Initials Date CD or lighter Multi-variable Indicator CD16 EO or lighter Contents Initials D ate CD25 Multi-variable Indicator STEAM CD Contents Initials Date CD CD29 Multi-variable Indicator STEAM ISO Type 4 Contents Initials D ate STEAM CD202 Multi-variable indicator CD Contents Initials D ate or darker or darker or darker Page 9
10 Moving front integrators for Steam Integron IT26-C It was developed for monitoring Steam sterilization processes between 118 ºC and 138 ºC and to ensure an adequate control of the effectiveness of sterilization processes by monitoring all critical parameters of steam sterilization (temperature, time, steam quality). Chemical pellet melts and migrates as a dark bar along the paper wick. Migration occurs through a zone marked as ACCEPT or REJECT, thus indicating whether sterilization conditions were met or not. The ACCEPT result is reached when a theoretical spore population reaches its kill time, indicating integration condition has been reached. Unique point integrator for Steam Integron IT26-1YS Integron IT26-1YS indicator allows the verification of Steam sterilization cycles between 121ºC y 135ºC, ensuring that all critical process parameters (temperature, time, steam quality) have been met. Two-level integrator for EO Integron IT12 It was developed to control Ethylene Oxide sterilization processes. It is a two-level indicator: Level 1 is the exposure level, which indicates exposure to Ethylene Oxide, while Level 2 is the integration level. This second level consists of a purple/brown ink dot that turns to green as it integrates all critical parameters of the sterilization process (time, temperature, humidity and Ethylene Oxide concentration). This indicator mimics the death curve of a theoretical Bacillus atrophaeus spore population. Page 10
11
12
 Rev. 0 October 2017 Terragene offers a range of simple and effective products to precisely control washing and sterilization processes of different instruments in tattoo and body piercing studios. Available
Rev. 0 October 2017 Terragene offers a range of simple and effective products to precisely control washing and sterilization processes of different instruments in tattoo and body piercing studios. Available
Best Practice Monitoring for Sterile Processing Professionals
 Best Practice Monitoring for Sterile Processing Professionals Sterile processing departments are faced with greater challenges than ever. Increasing demands for quick turnarounds. Pressure to streamline
Best Practice Monitoring for Sterile Processing Professionals Sterile processing departments are faced with greater challenges than ever. Increasing demands for quick turnarounds. Pressure to streamline
PRODUCT CATALOGUE. for gke Steri-Record and gke Clean-Record Chemical indicators, PCDs and accessories for cleaning and sterilization
 PRODUCT CATALOGUE for gke Steri-Record and gke Clean-Record Chemical indicators, PCDs and accessories for cleaning and sterilization Bowie-Dick Simulation Test Batch and Process Monitoring Systems, Documentation
PRODUCT CATALOGUE for gke Steri-Record and gke Clean-Record Chemical indicators, PCDs and accessories for cleaning and sterilization Bowie-Dick Simulation Test Batch and Process Monitoring Systems, Documentation
PRODUCT CATALOGUE. for gke Steri-Record and gke Clean-Record Chemical indicators, PCDs and accessories for cleaning and sterilization
 PRODUCT CATALOGUE for gke Steri-Record and gke Clean-Record Chemical indicators, PCDs and accessories for cleaning and sterilization Bowie-Dick Simulation Test Batch and Process Monitoring Systems, Documentation
PRODUCT CATALOGUE for gke Steri-Record and gke Clean-Record Chemical indicators, PCDs and accessories for cleaning and sterilization Bowie-Dick Simulation Test Batch and Process Monitoring Systems, Documentation
Instructions for Validation of Autoclaves
 Instructions for Validation of Autoclaves A. Validation of autoclaves may accomplished by the following methods: 1. Autoclave tape applied to the outside of all packs to validate that the correct temperature
Instructions for Validation of Autoclaves A. Validation of autoclaves may accomplished by the following methods: 1. Autoclave tape applied to the outside of all packs to validate that the correct temperature
Hygiene Monitoring. Chemical Indicators, BD-Test and Batch- Product Information. CI_01_Eng
 Hygiene Monitoring Chemical Indicators, BD-Test and Batch- Control (PCD) Product Information CI_01_Eng verkauf@bag-healthcare.com service@bag-healthcare.com BAG-BD-Check II / BAG-BD-Check Disposable Bowie
Hygiene Monitoring Chemical Indicators, BD-Test and Batch- Control (PCD) Product Information CI_01_Eng verkauf@bag-healthcare.com service@bag-healthcare.com BAG-BD-Check II / BAG-BD-Check Disposable Bowie
QUALITY ASSURANCE IN AN MDRD
 QUALITY ASSURANCE IN AN MDRD MAINTENANCE AND PREVENTATIVE MAINTENANCE MDRD is a machine dependent department washers, pasteurizers, cart washer, sterilizer, ultrasonic, heat sealers, RO water systems All
QUALITY ASSURANCE IN AN MDRD MAINTENANCE AND PREVENTATIVE MAINTENANCE MDRD is a machine dependent department washers, pasteurizers, cart washer, sterilizer, ultrasonic, heat sealers, RO water systems All
gke Steri-Record Testsets and PCDs for validation, process and batch monitoring of sterilization processes
 gke Steri-Record Testsets and PCDs for validation, process and batch monitoring of sterilization processes Application Process Monitoring Systems (PMS) are process challenge devices that are used to test
gke Steri-Record Testsets and PCDs for validation, process and batch monitoring of sterilization processes Application Process Monitoring Systems (PMS) are process challenge devices that are used to test
Hygiene Monitoring. Biological Indicators for Monitoring Sterilization Processes. Product Information. BI_07_en
 Hygiene Monitoring Biological Indicators for Monitoring Sterilization Processes Product Information BI_07_en BAG-BioStrip Biological Indicators for Monitoring all Types of Sterilization Processes STEAM
Hygiene Monitoring Biological Indicators for Monitoring Sterilization Processes Product Information BI_07_en BAG-BioStrip Biological Indicators for Monitoring all Types of Sterilization Processes STEAM
HemoCheck TM Sample Policy. SUBJECT: Detection of blood residue on various surfaces
 HemoCheck TM Sample Policy SUBJECT: Detection of blood residue on various surfaces DEPARTMENT: Central Service APPROVED BY: EFFECTIVE: REVISED: 07/2016 PURPOSE: To test for detection of blood residue on
HemoCheck TM Sample Policy SUBJECT: Detection of blood residue on various surfaces DEPARTMENT: Central Service APPROVED BY: EFFECTIVE: REVISED: 07/2016 PURPOSE: To test for detection of blood residue on
One such problem is blood residual inside the channels of a robotic arm.
 SUBJECT: Robotic Arm Testing for residue blood DEPARTMENT: Central Service APPROVED BY: EFFECTIVE: REVISED: 07/2016 PURPOSE: To test for detection of blood residue inside and outside of a robotic arm and
SUBJECT: Robotic Arm Testing for residue blood DEPARTMENT: Central Service APPROVED BY: EFFECTIVE: REVISED: 07/2016 PURPOSE: To test for detection of blood residue inside and outside of a robotic arm and
ISO INTERNATIONAL STANDARD. Sterilization of health care products Chemical indicators Part 1: General requirements
 INTERNATIONAL STANDARD ISO 11140-1 Second edition 2005-07-15 Sterilization of health care products Chemical indicators Part 1: General requirements Stérilisation des produits de santé Indicateurs chimiques
INTERNATIONAL STANDARD ISO 11140-1 Second edition 2005-07-15 Sterilization of health care products Chemical indicators Part 1: General requirements Stérilisation des produits de santé Indicateurs chimiques
PART 5. Sterilisation Consumables RFTHPV
 PART 5 STATEMENT OF REQUIREMENTS Sterilisation Consumables RFTHPV2015 010 Part 5 Statement of Requirements TABLE OF CONTENTS INTRODUCTION... 3 5.1 Purpose... 3 5.2 Scope... 3 5.3 Product Categories...
PART 5 STATEMENT OF REQUIREMENTS Sterilisation Consumables RFTHPV2015 010 Part 5 Statement of Requirements TABLE OF CONTENTS INTRODUCTION... 3 5.1 Purpose... 3 5.2 Scope... 3 5.3 Product Categories...
sterilisation monitoring products
 sterilisation monitoring products Known for their consistent, reliable performance, Excelsior Scientific sterilisation monitoring products are used by pharmaceutical and medical device manufacturers, contract
sterilisation monitoring products Known for their consistent, reliable performance, Excelsior Scientific sterilisation monitoring products are used by pharmaceutical and medical device manufacturers, contract
Slide 1. Slide 2. Slide 3. Sponsored by Webber Training Inc. For more information contact Paul Webber
 Slide 1 QUALITY ASSURANCE IN STERILIZATION Susan Hadfield Director, Sterile Processing Department Health Sciences Centre Winnipeg, Manitoba Hosted by Paul Webber (paul@webbertraining.com) A teleclass sponsored
Slide 1 QUALITY ASSURANCE IN STERILIZATION Susan Hadfield Director, Sterile Processing Department Health Sciences Centre Winnipeg, Manitoba Hosted by Paul Webber (paul@webbertraining.com) A teleclass sponsored
Biological Monitoring
 Biological Monitoring Prof. Duygu PERÇİN, MD Department of Clinical Microbiology Erciyes University Faculty of Medicine, Kayseri-TURKEY duygu.percin@hotmail.com Learning Objectives The attandee will be
Biological Monitoring Prof. Duygu PERÇİN, MD Department of Clinical Microbiology Erciyes University Faculty of Medicine, Kayseri-TURKEY duygu.percin@hotmail.com Learning Objectives The attandee will be
STERILIZATION VALIDATION IN THIS SECTION REPROCESSING VALIDATIONS FOR REUSABLE MEDICAL DEVICES. Radiation Sterilization Validation
 STERILIZATION VALIDATION All sterilization processes require validation of the efficacy and reproducibility of the process. Depending on the type of sterilization, this may be accomplished by partial,
STERILIZATION VALIDATION All sterilization processes require validation of the efficacy and reproducibility of the process. Depending on the type of sterilization, this may be accomplished by partial,
Products and Guidelines for Enhanced Office Safety
 infection prevention IMS WRAP SporeCheck STERILIZATION solutions Products and Guidelines for Enhanced Office Safety Bagette pouches sterilization monitoring CDC recommends placing an indicator inside each
infection prevention IMS WRAP SporeCheck STERILIZATION solutions Products and Guidelines for Enhanced Office Safety Bagette pouches sterilization monitoring CDC recommends placing an indicator inside each
MEDICAL TRADING knows the meaning of sterilization very well: it can be
 STERILIZATION Wikipedia defines sterilization as any chemical or physical process that leads to the removal of any living organism. MEDICAL TRADING knows the meaning of sterilization very well: it can
STERILIZATION Wikipedia defines sterilization as any chemical or physical process that leads to the removal of any living organism. MEDICAL TRADING knows the meaning of sterilization very well: it can
Innovative. Smart. Trusted.
 Innovative. Smart. Trusted. We re your partner for sterilization products. Monitoring Products for Vaporized Hydrogen Peroxide Table of Contents Crosstex Industrial is a division of Crosstex International,
Innovative. Smart. Trusted. We re your partner for sterilization products. Monitoring Products for Vaporized Hydrogen Peroxide Table of Contents Crosstex Industrial is a division of Crosstex International,
Secure. Tested. Certified.
 Secure. Tested. Certified. We solve problems with confidence and creativity. Monitoring Products for Dry Heat Steam-Formaldehyde FORM Chlorine Dioxide Table of Contents Crosstex Industrial is a division
Secure. Tested. Certified. We solve problems with confidence and creativity. Monitoring Products for Dry Heat Steam-Formaldehyde FORM Chlorine Dioxide Table of Contents Crosstex Industrial is a division
8/27/2015. Learning Objectives. Purpose of Sterilization. To destroy all living pathogenic microorganisms
 Learning Objectives Compare and contrast the available methods of sterilization for the healthcare setting Describe the appropriate use and monitoring for sterilization methods Purpose of Sterilization
Learning Objectives Compare and contrast the available methods of sterilization for the healthcare setting Describe the appropriate use and monitoring for sterilization methods Purpose of Sterilization
CERTIFICATE OF CONFORMANCE Mesa Laboratories, Inc. 625 Zoot Way, Bozeman, MT Phone: (303)
 Expiration : Bowie-Dick Test Sheets 77-05-CI-BD110 The ProChem Lead-Free Bowie-Dick Test Sheet is for daily monitoring of pre-vacuum sterilizers operating at 134 C for 3.5 minutes. The test sheet is designed
Expiration : Bowie-Dick Test Sheets 77-05-CI-BD110 The ProChem Lead-Free Bowie-Dick Test Sheet is for daily monitoring of pre-vacuum sterilizers operating at 134 C for 3.5 minutes. The test sheet is designed
Responses to Questions for SYSTEM 1E Process Monitoring and Validation Webinar
 Responses to Questions for SYSTEM 1E Process Monitoring and Validation Webinar MONITORING THE SYSTEM 1E PROCESSOR Q: I want to confirm that there is no need for a Biological Indicator (BI) and that use
Responses to Questions for SYSTEM 1E Process Monitoring and Validation Webinar MONITORING THE SYSTEM 1E PROCESSOR Q: I want to confirm that there is no need for a Biological Indicator (BI) and that use
Sterilization. Euronda Spa t (+39) m euronda.com
 Sterilization The purpose of sterilization is to remove all microbial life forms, including spores and fungi, whether they are pathogenic or not. The sterilization process as a whole has to be in full
Sterilization The purpose of sterilization is to remove all microbial life forms, including spores and fungi, whether they are pathogenic or not. The sterilization process as a whole has to be in full
INSTRUMENT PROCESSING FROM PREP/PACKAGING TO STORAGE. January 2016
 INSTRUMENT PROCESSING FROM PREP/PACKAGING TO STORAGE January 2016 Disclosures The opinions expressed in this presentation are those of the author and do not necessarily reflect the official position of
INSTRUMENT PROCESSING FROM PREP/PACKAGING TO STORAGE January 2016 Disclosures The opinions expressed in this presentation are those of the author and do not necessarily reflect the official position of
Specialty Testing of Steam Sterilizers
 Specialty Testing of Steam Sterilizers Nancy Chobin, RN, AAS, ACSP, CSPM, CFER 2017 Sterile Processing University, LLC. **This in-service has been Approved by the CBSPD for 1.5 CEUs. Objectives 1. To describe
Specialty Testing of Steam Sterilizers Nancy Chobin, RN, AAS, ACSP, CSPM, CFER 2017 Sterile Processing University, LLC. **This in-service has been Approved by the CBSPD for 1.5 CEUs. Objectives 1. To describe
In the Know about.appropriate Use of 3M Attest Rapid Readout Biological Indicators
 3M Attest Sterile U Web Meeting Jan 22, 2009 In the Know about.appropriate Use of 3M Attest Rapid Readout Biological Indicators ww.3m.com/atteststerileuonline 7/15 Modified per Inma s input. SDF Prepared
3M Attest Sterile U Web Meeting Jan 22, 2009 In the Know about.appropriate Use of 3M Attest Rapid Readout Biological Indicators ww.3m.com/atteststerileuonline 7/15 Modified per Inma s input. SDF Prepared
AS/NZS 4187 RELEVANT STANDARDS. Robyn Williams NUM CSSD LBHHS & Standards Australia Chair HE-023
 AS/NZS 4187 RELEVANT STANDARDS Robyn Williams NUM CSSD LBHHS & Standards Australia Chair HE-023 Relevant Standards AS 2514 AS 2774 AS 2773 AS 2773.1 Drying cabinets for medical equipment Drying cabinets
AS/NZS 4187 RELEVANT STANDARDS Robyn Williams NUM CSSD LBHHS & Standards Australia Chair HE-023 Relevant Standards AS 2514 AS 2774 AS 2773 AS 2773.1 Drying cabinets for medical equipment Drying cabinets
Sterilization Service Revolution Service Enhancement in HA Hospitals
 Sterilization Service Revolution Service Enhancement in HA Hospitals Hong Kong Hospital Authority Convention 2014 7 May 2014 LAW Tat Hong Samuel Cluster Operations Manager Central Sterile Supplies Department,
Sterilization Service Revolution Service Enhancement in HA Hospitals Hong Kong Hospital Authority Convention 2014 7 May 2014 LAW Tat Hong Samuel Cluster Operations Manager Central Sterile Supplies Department,
Medline Gemini Sterilization Wrap. Test Studies
 Medline Gemini Sterilization Wrap Test Studies Steam Efficacy : Steam PreVac Steam Efficacy: Steam Gravity Shelf Life: 180 Days Plasma Efficacy: STERRAD 100NX Plasma Efficacy: STERRAD NX Plasma Efficacy:
Medline Gemini Sterilization Wrap Test Studies Steam Efficacy : Steam PreVac Steam Efficacy: Steam Gravity Shelf Life: 180 Days Plasma Efficacy: STERRAD 100NX Plasma Efficacy: STERRAD NX Plasma Efficacy:
CAN STERILE REALLY BE STERILE?: EFFECTIVE AUTOCLAVING METHODS
 Vet Times The website for the veterinary profession https://www.vettimes.co.uk CAN STERILE REALLY BE STERILE?: EFFECTIVE AUTOCLAVING METHODS Author : James Gasson Categories : Vets Date : February 2, 2009
Vet Times The website for the veterinary profession https://www.vettimes.co.uk CAN STERILE REALLY BE STERILE?: EFFECTIVE AUTOCLAVING METHODS Author : James Gasson Categories : Vets Date : February 2, 2009
Effective Use of Autoclaves
 Published on UC Davis Safety Services (https://safetyservices.ucdavis.edu) Effective Use of Autoclaves SafetyNet #: 26 Autoclave Use Autoclaves (steam sterilizers) are metal pressure vessels that are used
Published on UC Davis Safety Services (https://safetyservices.ucdavis.edu) Effective Use of Autoclaves SafetyNet #: 26 Autoclave Use Autoclaves (steam sterilizers) are metal pressure vessels that are used
LEADING TECHNOLOGY IN STERILIZATION
 LEADING TECHNOLOGY IN STERILIZATION E9 RECORDER E9 MED E5 FAST, EFFICIENT, USER-FRIENDLY B TYPE VACUUM AND NON-VACUUM AUTOCLAVES Medisafe International, Twyford Road, Bishop s Stortford, Hertfordshire
LEADING TECHNOLOGY IN STERILIZATION E9 RECORDER E9 MED E5 FAST, EFFICIENT, USER-FRIENDLY B TYPE VACUUM AND NON-VACUUM AUTOCLAVES Medisafe International, Twyford Road, Bishop s Stortford, Hertfordshire
Bovine IgG ELISA Kit
 Bovine IgG ELISA Kit Cat. No. E11-118 Components Supplied Bovine IgG Pre-Coated 96-well Strip Plate, 1 each Bovine IgG Standard, 500 ng/vial, 2 each Bovine IgG Detection Antibody, 12 ml 20X Dilution Buffer
Bovine IgG ELISA Kit Cat. No. E11-118 Components Supplied Bovine IgG Pre-Coated 96-well Strip Plate, 1 each Bovine IgG Standard, 500 ng/vial, 2 each Bovine IgG Detection Antibody, 12 ml 20X Dilution Buffer
Disinfection and sterilisation
 Disinfection and sterilisation Mongolia 2011 Prof. Dr. Walter Popp Hospital Hygiene, University Clinics Essen, Germany 1 Term Definition Reduction factor of germs Cleaning Remove dirt including 10-100
Disinfection and sterilisation Mongolia 2011 Prof. Dr. Walter Popp Hospital Hygiene, University Clinics Essen, Germany 1 Term Definition Reduction factor of germs Cleaning Remove dirt including 10-100
COLORIMETRIC SANDWICH ELISA KIT INSTRUCTION MANUAL
 Page 1 of 7 COLORIMETRIC SANDWICH ELISA KIT INSTRUCTION MANUAL This product is for research use ONLY and not for human or animal therapeutic or diagnostic use. Page 2 of 7 Contents Page 3 I. Supplied Materials:
Page 1 of 7 COLORIMETRIC SANDWICH ELISA KIT INSTRUCTION MANUAL This product is for research use ONLY and not for human or animal therapeutic or diagnostic use. Page 2 of 7 Contents Page 3 I. Supplied Materials:
Bowie Dick Helix Test
 Technical Data Sheet Product name: Bowie Dick Helix Test Product reference: 101.201.0100 STEAM 134ºC - 3,5 min 121ºC - 15 min 101.202.0250 STEAM 134ºC - 3,5 min 121ºC - 15 min Applicable standards: ISO
Technical Data Sheet Product name: Bowie Dick Helix Test Product reference: 101.201.0100 STEAM 134ºC - 3,5 min 121ºC - 15 min 101.202.0250 STEAM 134ºC - 3,5 min 121ºC - 15 min Applicable standards: ISO
Packaging Systems for Central Service Operations
 Lesson No. CRCST 132 (Technical Continuing Education - TCE) Sponsored by: by Carol Petro, RN, CRCST, CIS, CNOR OR Educator and Sterile Processing Educator Indiana University Health North Hospital Carmel,
Lesson No. CRCST 132 (Technical Continuing Education - TCE) Sponsored by: by Carol Petro, RN, CRCST, CIS, CNOR OR Educator and Sterile Processing Educator Indiana University Health North Hospital Carmel,
Getinge da Vinci Solution
 Getinge da Vinci Solution A comprehensive cleaning solution for da Vinci EndoWrist instruments This document is intended to provide information to an international audience outside of the US. Validated,
Getinge da Vinci Solution A comprehensive cleaning solution for da Vinci EndoWrist instruments This document is intended to provide information to an international audience outside of the US. Validated,
Your Sterile Processing Partner. Duraholder IPS
 Your Sterile Processing Partner Table of Contents Item Page Duraholder Case Study 17 AAMI ST79 Reference 20 Steam Sterilization Efficacy Testing 21 PRODUCT INDEX Product Page Standard Pouches DURA050/100
Your Sterile Processing Partner Table of Contents Item Page Duraholder Case Study 17 AAMI ST79 Reference 20 Steam Sterilization Efficacy Testing 21 PRODUCT INDEX Product Page Standard Pouches DURA050/100
Meeting the challenges of medical device packaging
 Meeting the challenges of medical device packaging Gisela Cristina Mendes Bastos Viegas, S.A. October 14, 2011 Agenda Evaluation and selection Validation Test methods Sterile Barrier Systems EN 868 Parts
Meeting the challenges of medical device packaging Gisela Cristina Mendes Bastos Viegas, S.A. October 14, 2011 Agenda Evaluation and selection Validation Test methods Sterile Barrier Systems EN 868 Parts
Sirona competency in hygiene DAC PROFESSIONAL. Hygiene systems. Vacuum autoclave class B to satisfy the most demanding requirements
 Sirona competency in hygiene Hygiene systems Vacuum autoclave class B to satisfy the most demanding requirements Professional and fast Sirona s is an autoclave which satisfies the most demanding medical
Sirona competency in hygiene Hygiene systems Vacuum autoclave class B to satisfy the most demanding requirements Professional and fast Sirona s is an autoclave which satisfies the most demanding medical
EH&S. Sheet. Fact. Safe and Effective Use of Autoclaves. What are autoclaves? Factors for effective sterilization. Dry heat cycle - when to use
 Please post or circulate Fact heet nvironment, ealth and afety Information for the Berkeley Campus No. 33 Revised 04/04/11 afe and ffective Use of Autoclaves Autoclaves are easy to use but can pose a safety
Please post or circulate Fact heet nvironment, ealth and afety Information for the Berkeley Campus No. 33 Revised 04/04/11 afe and ffective Use of Autoclaves Autoclaves are easy to use but can pose a safety
Steam Sterilization Monitoring in the New CSA Standard (Z )
 Speaker Disclaimer Steam Sterilization Monitoring in the New CSA Standard (Z314.3-14) Rick Bauer, Ph.D. Senior Technical Services Advisor 3M Canada Employed in technical services for 3M Canada Education,
Speaker Disclaimer Steam Sterilization Monitoring in the New CSA Standard (Z314.3-14) Rick Bauer, Ph.D. Senior Technical Services Advisor 3M Canada Employed in technical services for 3M Canada Education,
Principles of Chlorine Dioxide Gas as a Decontamination Method
 Principles of Chlorine Dioxide Gas as a Decontamination Method In order for any decontamination method to be effective, the following points must be satisfied The decontamination method must: Be able to
Principles of Chlorine Dioxide Gas as a Decontamination Method In order for any decontamination method to be effective, the following points must be satisfied The decontamination method must: Be able to
BEST PARTER & BEST SOLUTION RENO Plasma Sterilizer
 Web: www.medcon.com.au Email: info@medcon.com.au MED-CON PTY LTD SOLUTIONS FOR BIO-SECURITY Phone: 03 9753 2710 Fax: 03 9753 2722 BEST PARTER & BEST SOLUTION RENO Series - Benefits Rapid Sterilization
Web: www.medcon.com.au Email: info@medcon.com.au MED-CON PTY LTD SOLUTIONS FOR BIO-SECURITY Phone: 03 9753 2710 Fax: 03 9753 2722 BEST PARTER & BEST SOLUTION RENO Series - Benefits Rapid Sterilization
Important Terminology (pg )
 Number of living microbes 10/18/2016 Chapter 9: Control of Microbial Growth 1. Physical Methods 2. Chemical methods Important Terminology (pg. 263-264) sterilization > commercial sterilization > disinfection
Number of living microbes 10/18/2016 Chapter 9: Control of Microbial Growth 1. Physical Methods 2. Chemical methods Important Terminology (pg. 263-264) sterilization > commercial sterilization > disinfection
ENDOSCOPIC REPROCESSING ETD DOUBLE Advancing the Efficiency of Reprocessing.
 11157 ENDOSCOPIC REPROCESSING ETD DOUBLE Advancing the Efficiency of Reprocessing. ETD DOUBLE: INTRODUCING THE FIRST OLYMPUS PASS- THROUGH WASHER-DISINFECTOR With the ETD Double, Olympus Has Advanced Its
11157 ENDOSCOPIC REPROCESSING ETD DOUBLE Advancing the Efficiency of Reprocessing. ETD DOUBLE: INTRODUCING THE FIRST OLYMPUS PASS- THROUGH WASHER-DISINFECTOR With the ETD Double, Olympus Has Advanced Its
The Bowie & Dick test is used by manufacturers
 Keywords steam steriliser performance evaluation helix test Performance Evaluation of Hospital Steam Sterilisers Using the European Helix Test A.C.P. de Bruijn*, A.W. van Drongelen The Bowie & Dick test
Keywords steam steriliser performance evaluation helix test Performance Evaluation of Hospital Steam Sterilisers Using the European Helix Test A.C.P. de Bruijn*, A.W. van Drongelen The Bowie & Dick test
We ve built a STRONG CASE. for changing the way you sterilize. Reusable Sterilization Container System
 We ve built a STRONG CASE for changing the way you sterilize. Reusable Sterilization Container System Reusable Sterilization Container System Introducing the DePuy Synthes REUSABLE STERILIZATION CONTAINER
We ve built a STRONG CASE for changing the way you sterilize. Reusable Sterilization Container System Reusable Sterilization Container System Introducing the DePuy Synthes REUSABLE STERILIZATION CONTAINER
3M Products and Services for Medical Sterilization Processes. Full Line Catalog. Medical
 3M Products and Services for Medical Sterilization Processes Full Line Catalog Medical 3M Medical Sterilization Specialties 3M Medical Sterilization Specialties is a global leader in the manufacture and
3M Products and Services for Medical Sterilization Processes Full Line Catalog Medical 3M Medical Sterilization Specialties 3M Medical Sterilization Specialties is a global leader in the manufacture and
NO 2 Gas Sterilization and Decontamination: Keeping Pace, from Start to Finish
 NO 2 Gas Sterilization and Decontamination: Keeping Pace, from Start to Finish Evan Goulet, Ph.D., VP of Microbiology Applications Laboratory PDA Midwest: Sterility Assurance and Risk Mitigation October
NO 2 Gas Sterilization and Decontamination: Keeping Pace, from Start to Finish Evan Goulet, Ph.D., VP of Microbiology Applications Laboratory PDA Midwest: Sterility Assurance and Risk Mitigation October
Choose a category. You will be given the answer. You must give the correct question. Click to begin. Instructions for using this template.
 Instructions for using this template. Remember this is Jeopardy, so where I have written Answer this is the prompt the players will see, and where I have Question should be the player s response. To enter
Instructions for using this template. Remember this is Jeopardy, so where I have written Answer this is the prompt the players will see, and where I have Question should be the player s response. To enter
TUBES WITH INTERNAL THREAD BROCHURE. Non-Coded and Alphanumeric Coded 30 YEARS OF EXPERIENCE IN TRACEABLE SAMPLE STORAGE
 BROCHURE 30 YEARS OF EXPERIENCE IN TRACEABLE SAMPLE STORAGE CERTIFIED CLASS 7 CLEAN ROOM INJECTION MOLDING AND ASSEMBLY PRODUCTION IN THE NETHERLANDS AND THE UNITED STATES TUBES WITH INTERNAL THREAD NON-CODED
BROCHURE 30 YEARS OF EXPERIENCE IN TRACEABLE SAMPLE STORAGE CERTIFIED CLASS 7 CLEAN ROOM INJECTION MOLDING AND ASSEMBLY PRODUCTION IN THE NETHERLANDS AND THE UNITED STATES TUBES WITH INTERNAL THREAD NON-CODED
PRODUCT CATALOGUE. for gke Steri-Record Biological Indicators, Process Challenge Devices and Accessories
 PRODUCT CATALOGUE for gke Steri-Record Biological Indicators, Process Challenge Devices and Accessories Spore strips, Ampoules, Suspensions, Self-contained Mini-Bio-Plus Indicators to monitor steam, formaldehyde,
PRODUCT CATALOGUE for gke Steri-Record Biological Indicators, Process Challenge Devices and Accessories Spore strips, Ampoules, Suspensions, Self-contained Mini-Bio-Plus Indicators to monitor steam, formaldehyde,
AIOS ASSOCIAZIONE ITALIANA OPERATORI SANITARI ADDETTI ALLA STERILIZZAZIONE
 AIOS ASSOCIAZIONE ITALIANA OPERATORI SANITARI ADDETTI ALLA STERILIZZAZIONE (Italian Association of Sterilization Operators working in health care facilities) 1 WFHSS 2007 - WORLD CONFERENCE FOR HOSPITAL
AIOS ASSOCIAZIONE ITALIANA OPERATORI SANITARI ADDETTI ALLA STERILIZZAZIONE (Italian Association of Sterilization Operators working in health care facilities) 1 WFHSS 2007 - WORLD CONFERENCE FOR HOSPITAL
REUSABLE MEDICAL DEVICE VALIDATION
 All sterilization processes require validation of the efficacy and reproducibility of the process. Depending on the type of sterilization, this may be accomplished by partial, sub-lethal, or repetitive
All sterilization processes require validation of the efficacy and reproducibility of the process. Depending on the type of sterilization, this may be accomplished by partial, sub-lethal, or repetitive
Product Catalogue. for gke Steri-Record Biological Indicators, Process Challenge Devices and Accessories
 Product Catalogue for gke Steri-Record Biological Indicators, Process Challenge Devices and Accessories Spore Strips, Ampoules, Suspensions, Self-contained Mini-Bio-Plus Indicators to monitor steam, formaldehyde,
Product Catalogue for gke Steri-Record Biological Indicators, Process Challenge Devices and Accessories Spore Strips, Ampoules, Suspensions, Self-contained Mini-Bio-Plus Indicators to monitor steam, formaldehyde,
Welcome! How do I get a CE Certificate?
 M Sterile U Network M Sterile U Web Meeting November 0, 0 Today s meeting times: 9:00 a.m., :00 a.m., and :00 p.m. Central Standard Time To hear audio, call 800-97-00 and enter access code 7 Phone lines
M Sterile U Network M Sterile U Web Meeting November 0, 0 Today s meeting times: 9:00 a.m., :00 a.m., and :00 p.m. Central Standard Time To hear audio, call 800-97-00 and enter access code 7 Phone lines
Hollow, porous, solid,? Testing of Steam Sterilizers
 Aspects of Steam Sterilization Process Monitoring: from Huckaback Towels to ETS and what Standards do and don't cover by Klaus Hahnen, 3M Medical Division Laboratory, Neuss, Germany Space for 3M Montage
Aspects of Steam Sterilization Process Monitoring: from Huckaback Towels to ETS and what Standards do and don't cover by Klaus Hahnen, 3M Medical Division Laboratory, Neuss, Germany Space for 3M Montage
EQUIPMENT FOR AUTOCLAVING
 EQUIPMENT FOR AUTOCLAVING BY D.NARENDAR M. Pharm-II sem DEPARTMENT OF PHARMACEUTICS UNIVERSITY COLLEGE OF PHARMACEUTICAL SCIENCES KAKATIYA UNIVERSITY, WARANGAL CONTENTS INTRODUCTION ADVANTAGES DISADVANTAGES
EQUIPMENT FOR AUTOCLAVING BY D.NARENDAR M. Pharm-II sem DEPARTMENT OF PHARMACEUTICS UNIVERSITY COLLEGE OF PHARMACEUTICAL SCIENCES KAKATIYA UNIVERSITY, WARANGAL CONTENTS INTRODUCTION ADVANTAGES DISADVANTAGES
3M s Sterilization Tech Line FAQs Sandra Velte, BA, CSPDT
 3M Sterile U Sterilization Assurance Continuing Education 3M s Sterilization Tech Line FAQs Sandra Velte, BA, CSPDT May 2013 Objectives After completion of this self-study activity, the learner will be
3M Sterile U Sterilization Assurance Continuing Education 3M s Sterilization Tech Line FAQs Sandra Velte, BA, CSPDT May 2013 Objectives After completion of this self-study activity, the learner will be
breaking load N/cm elongation at break %
 PRINTED TAPES backing thickness µm total thickness µm adhesion N/cm breaking load N/cm elongation at break % available colours medium weight light weight packaging packaging automatic sealing machine suitable
PRINTED TAPES backing thickness µm total thickness µm adhesion N/cm breaking load N/cm elongation at break % available colours medium weight light weight packaging packaging automatic sealing machine suitable
Safe to Handle? Comparing Manually and Machine-Washed Medical Devices
 Safe to Handle? Comparing Manually and Machine-Washed Medical Devices Kaumudi Kulkarni, Dzelma Kaczorowski, Alex Bonkowski, Stephen Kovach, and Ralph Basile About the Authors Kaumudi Kulkarni, MS, is manager
Safe to Handle? Comparing Manually and Machine-Washed Medical Devices Kaumudi Kulkarni, Dzelma Kaczorowski, Alex Bonkowski, Stephen Kovach, and Ralph Basile About the Authors Kaumudi Kulkarni, MS, is manager
BLISTER PACKING MACHINES AND SERVICES THAT OFFER FLEXIBILITY AND HIGH QUALITY BLISTER PACKS FROM A WIDE VARIETY OF PACKAGING MATERIALS.
 BLISTER PACKING MACHINES AND SERVICES THAT OFFER FLEXIBILITY AND HIGH QUALITY BLISTER PACKS FROM A WIDE VARIETY OF PACKAGING MATERIALS. 06 1. PACKAGING SOLUTIONS 07 1. PACKAGING SOLUTIONS SMALL SCALE,
BLISTER PACKING MACHINES AND SERVICES THAT OFFER FLEXIBILITY AND HIGH QUALITY BLISTER PACKS FROM A WIDE VARIETY OF PACKAGING MATERIALS. 06 1. PACKAGING SOLUTIONS 07 1. PACKAGING SOLUTIONS SMALL SCALE,
What Dentistry Can Learn from AAMI ST79. Best Practices for device and instrument reprocessing
 What Dentistry Can Learn from AAMI ST79 Best Practices for device and instrument reprocessing Learning goals: Purpose and goals of AAMI organization. Overview of ST79. Review selected topics: IFUs, tools
What Dentistry Can Learn from AAMI ST79 Best Practices for device and instrument reprocessing Learning goals: Purpose and goals of AAMI organization. Overview of ST79. Review selected topics: IFUs, tools
Clostridium difficile Update Dr. Michelle Alfa, Winnipeg Sponsored by ARJO
 Clostridium difficile: Update Dr. Michelle Alfa Ph.D, FCCM Diagnostic Services of Manitoba St. Boniface General Hospital Site Hosted by Paul Webber paul@webbertraining.com Sponsored by www.arjo.com www.webbertraining.com
Clostridium difficile: Update Dr. Michelle Alfa Ph.D, FCCM Diagnostic Services of Manitoba St. Boniface General Hospital Site Hosted by Paul Webber paul@webbertraining.com Sponsored by www.arjo.com www.webbertraining.com
Sterilization of Schweickhardt Instruments
 Sterilization of Schweickhardt Instruments INTRODUCTION l Most dental instruments are made of corrosion-resistant highgrade steels. The requirements for grades of steel are defined in national and international
Sterilization of Schweickhardt Instruments INTRODUCTION l Most dental instruments are made of corrosion-resistant highgrade steels. The requirements for grades of steel are defined in national and international
xdental x the sterilization company Domina Plus B Aptica Plus B Atoma Plus
 xdental x the sterilization company Domina Plus B Aptica Plus B Atoma Plus THE PREMIUM LINE Dental X Sterilization an important choice The growing concern for the spreading of infection diseases and the
xdental x the sterilization company Domina Plus B Aptica Plus B Atoma Plus THE PREMIUM LINE Dental X Sterilization an important choice The growing concern for the spreading of infection diseases and the
Temperature mapping and autoclave validation specialists
 www.thermalcompliance.co.uk Temperature mapping and autoclave validation specialists Who are we? We are Thermal Compliance, specialists in temperature mapping and autoclave validation. Founded in 2007,
www.thermalcompliance.co.uk Temperature mapping and autoclave validation specialists Who are we? We are Thermal Compliance, specialists in temperature mapping and autoclave validation. Founded in 2007,
1.02 DESCRIPTION A. This Section specifies furnishing and installing Surface Applied Detectable/Tactile Warning Surface Tiles where indicated.
 SURFACE APPLIED DETECTABLE/TACTILE WARNING SURFACE TILE PART 1 GENERAL 1.01 RELATED DOCUMENTS A. Drawings and general provisions of Contract, including General and Special Conditions and Division 1 Specifications
SURFACE APPLIED DETECTABLE/TACTILE WARNING SURFACE TILE PART 1 GENERAL 1.01 RELATED DOCUMENTS A. Drawings and general provisions of Contract, including General and Special Conditions and Division 1 Specifications
If All Else Fails Read the Directions. Disclosure. Manufacturer s Instructions. Presenter: Sue Lafferty CHICA Canada Conference, May 2010
 If All Else Fails Read the Directions The Importance of Medical Device Manufacturer s Instructions Presenter: Sue Lafferty CHICA Canada Conference, May 2010 Disclosure Member of 3M Canada Speakers Bureau:
If All Else Fails Read the Directions The Importance of Medical Device Manufacturer s Instructions Presenter: Sue Lafferty CHICA Canada Conference, May 2010 Disclosure Member of 3M Canada Speakers Bureau:
Effect of Storage Conditions upon the Resistance (D-value) of Spore Strip Biological Indicators (BIs) used in EtO Sterilization Monitoring
 Effect of Storage Conditions upon the Resistance (D-value) of Spore Strip Biological Indicators (BIs) used in EtO Sterilization Monitoring Wendy Royalty*, Mia Ware, Russell Nyberg Raven Biological Laboratories,
Effect of Storage Conditions upon the Resistance (D-value) of Spore Strip Biological Indicators (BIs) used in EtO Sterilization Monitoring Wendy Royalty*, Mia Ware, Russell Nyberg Raven Biological Laboratories,
Shur-Shot X-Proof Hydrogen Fluoride Alarm Operations Manual
 Shur-Shot X-Proof Hydrogen Fluoride Alarm Operations Manual P/N 1000006053 Rev E $7%$QDO\WLFV// //& 733 Dairy Rd. Parkton, Md. 21120 www.atbanalytics.com (410) 733-6365 Table of Contents Chapter 1: Getting
Shur-Shot X-Proof Hydrogen Fluoride Alarm Operations Manual P/N 1000006053 Rev E $7%$QDO\WLFV// //& 733 Dairy Rd. Parkton, Md. 21120 www.atbanalytics.com (410) 733-6365 Table of Contents Chapter 1: Getting
SERIE 4. rapid, reliable, efficient
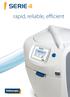 SERIE 4 rapid, reliable, efficient SERIE 4 Intuitive an machine 12/07/2014 15:23:00 1 35 C The power at your fingertips A NEW GENERATION OF INTERFACE The Serie 4 PA brings the benefit of a new intuitive
SERIE 4 rapid, reliable, efficient SERIE 4 Intuitive an machine 12/07/2014 15:23:00 1 35 C The power at your fingertips A NEW GENERATION OF INTERFACE The Serie 4 PA brings the benefit of a new intuitive
Competency Guide: Care and Handling of Rigid Endoscopes
 Competency Guide: Care and Handling of Rigid Endoscopes This document details the basic steps to correctly process and maintain a rigid endoscope. This document can be used to perform a competency review
Competency Guide: Care and Handling of Rigid Endoscopes This document details the basic steps to correctly process and maintain a rigid endoscope. This document can be used to perform a competency review
PRODUCT CATALOGUE. for gke Steri-Record Biological Indicators, Process Challenge Devices and Accessories
 PRODUCT CATALOGUE for gke Steri-Record Biological Indicators, Process Challenge Devices and Accessories Spore strips, Ampoules, Suspensions, Self-contained Mini-Bio-Plus Indicators to monitor steam, formaldehyde,
PRODUCT CATALOGUE for gke Steri-Record Biological Indicators, Process Challenge Devices and Accessories Spore strips, Ampoules, Suspensions, Self-contained Mini-Bio-Plus Indicators to monitor steam, formaldehyde,
HEAD. 5. Materials and preformed sterile barrier systems Microbial barrier properties...10
 DUPONT TYVEK COMPLIANCE TO ISO 11607-1:2006 CONTENTS HEAD Introduction.... 3 ISO 11607-1:2006 Requirements.... 4 4. General requirements.... 4 4.2 Quality systems.... 4 4.3 Sampling.... 4 4.4 Test methods....
DUPONT TYVEK COMPLIANCE TO ISO 11607-1:2006 CONTENTS HEAD Introduction.... 3 ISO 11607-1:2006 Requirements.... 4 4. General requirements.... 4 4.2 Quality systems.... 4 4.3 Sampling.... 4 4.4 Test methods....
For Research Use Only Please read all instructions carefully before beginning this assay. Store kit at 4ºC at all times.
 Kit Instruction Progesterone Enzyme Immunoassay Product Number: EA 74 Storage: 4 C Document Contol #: 010603 pg. 1 of 8 Enzyme Immunoassay for Progesterone Product No. EA 74 For Research Use Only Please
Kit Instruction Progesterone Enzyme Immunoassay Product Number: EA 74 Storage: 4 C Document Contol #: 010603 pg. 1 of 8 Enzyme Immunoassay for Progesterone Product No. EA 74 For Research Use Only Please
AUTOMATED ENDOSCOPE REPROCESSOR. OER-Pro Easy. Fast. Reliable.
 AUTOMATED ENDOSCOPE REPROCESSOR OER-Pro Easy. Fast. Reliable. Olympus Endoscope Reprocessor The only reprocessor designed by an endoscope manufacturer. The OER-Pro provides high-level disinfection of Olympus
AUTOMATED ENDOSCOPE REPROCESSOR OER-Pro Easy. Fast. Reliable. Olympus Endoscope Reprocessor The only reprocessor designed by an endoscope manufacturer. The OER-Pro provides high-level disinfection of Olympus
VHP Sterilization and Prion Inactivation
 WFHSS Workshop Opatija Croatia March 27 th 2009 VHP Sterilization and Prion Inactivation Dr. Georgia Alevizopoulou Clinical Specialist Eastern Europe, Middle East & Africa VHP Sterilization and Prion Inactivation
WFHSS Workshop Opatija Croatia March 27 th 2009 VHP Sterilization and Prion Inactivation Dr. Georgia Alevizopoulou Clinical Specialist Eastern Europe, Middle East & Africa VHP Sterilization and Prion Inactivation
Health Service Executive Code of Practice for Decontamination of Reusable Invasive Medical Devices
 PART 3: RECOMMENDED PRACTICES FOR CENTRAL DECONTAMINATION UNITS Health Service Executive Code of Practice for Decontamination of Reusable Invasive Medical Devices Reader Information Directorate: Title:
PART 3: RECOMMENDED PRACTICES FOR CENTRAL DECONTAMINATION UNITS Health Service Executive Code of Practice for Decontamination of Reusable Invasive Medical Devices Reader Information Directorate: Title:
Testing of routinely sterilized products should
 Product Quality Assurance Testing: One Hospital s Experience Testing of routinely sterilized products should be part of a healthcare facility s quality control program, and ANSI/AAMI ST79:2006, Comprehensive
Product Quality Assurance Testing: One Hospital s Experience Testing of routinely sterilized products should be part of a healthcare facility s quality control program, and ANSI/AAMI ST79:2006, Comprehensive
Plasma Low Temperature Sterilization. With vaporized hydrogen peroxide. PlazMax Line
 Plasma Low Temperature Sterilization With vaporized hydrogen peroxide PlazMax Line Low Temperature Sterilization PlazMax Line For heat and moisture sensitive equipment PlazMax provides an efficient sterilization
Plasma Low Temperature Sterilization With vaporized hydrogen peroxide PlazMax Line Low Temperature Sterilization PlazMax Line For heat and moisture sensitive equipment PlazMax provides an efficient sterilization
PREVENTING WOUND INFECTION BEGINS BEFORE THE PATIENT
 Lesson No. CRCST 132 (Technical Continuing Education - TCE) Sponsored by: by Carol Petro, RN, CRCST, CIS, CNOR OR Educator and Sterile Processing Educator Indiana University Health North Hospital Carmel,
Lesson No. CRCST 132 (Technical Continuing Education - TCE) Sponsored by: by Carol Petro, RN, CRCST, CIS, CNOR OR Educator and Sterile Processing Educator Indiana University Health North Hospital Carmel,
FREQUENTLY ASKED QUESTIONS IPAC Lapses
 FREQUENTLY ASKED QUESTIONS IPAC Lapses Updated: December 15, 2017 Reprocessing in Community Health Care Settings Medication Administration Laboratory Office design/facilities Reprocessing in Community
FREQUENTLY ASKED QUESTIONS IPAC Lapses Updated: December 15, 2017 Reprocessing in Community Health Care Settings Medication Administration Laboratory Office design/facilities Reprocessing in Community
Sterilization & Chemical Resistance of Healthcare Polymers
 Sterilization & Chemical Resistance of Healthcare Polymers Key Terms & Definitions Sterilization - A process that eliminates or kills all forms of life, including transmissible agents, on a medical device;
Sterilization & Chemical Resistance of Healthcare Polymers Key Terms & Definitions Sterilization - A process that eliminates or kills all forms of life, including transmissible agents, on a medical device;
3 Screen Printable Sheet Vinyl Label Material
 3 Screen Printable Sheet Vinyl Label Material Technical Data February, 2008 Product Description 3M Screen Printable Sheet Vinyl Label Materials 7200, 7902 and 7906 are durable, high performance materials
3 Screen Printable Sheet Vinyl Label Material Technical Data February, 2008 Product Description 3M Screen Printable Sheet Vinyl Label Materials 7200, 7902 and 7906 are durable, high performance materials
Bovine Albumin ELISA Kit
 Bovine Albumin ELISA Kit Cat. No. E11-113 Components Supplied Bovine Albumin Pre-Coated 96-well Strip Plate, 1 each Bovine Albumin Standard, 500 ng/vial, 2 each Bovine Albumin Detection Antibody, 12 ml
Bovine Albumin ELISA Kit Cat. No. E11-113 Components Supplied Bovine Albumin Pre-Coated 96-well Strip Plate, 1 each Bovine Albumin Standard, 500 ng/vial, 2 each Bovine Albumin Detection Antibody, 12 ml
35.0 Sterile Supply Unit (SSU)
 35.0 Sterile Supply Unit (SSU) 35.1 Introduction 35.1.1 General A Hospital must provide adequate facilities for cleaning, sterilisation and storage of equipment and instruments to ensure the care and safety
35.0 Sterile Supply Unit (SSU) 35.1 Introduction 35.1.1 General A Hospital must provide adequate facilities for cleaning, sterilisation and storage of equipment and instruments to ensure the care and safety
Sterilization & Infection Control Products
 Sterilization & Infection Control Products Mechanical Autoclaves Tuttnauer semi-automatic (M) autoclave is designed to complement any healthcare and laboratory facility. Time tested reliability, durability
Sterilization & Infection Control Products Mechanical Autoclaves Tuttnauer semi-automatic (M) autoclave is designed to complement any healthcare and laboratory facility. Time tested reliability, durability
Guidelines for Selection and Use of Disinfectants
 Guidelines for Selection and Use of Disinfectants Ref: (a) APIC Guidelines for Infection Control Practice, American Journal of Infection Control; April 1990, Vol 18, 99-113. To assist health care professionals
Guidelines for Selection and Use of Disinfectants Ref: (a) APIC Guidelines for Infection Control Practice, American Journal of Infection Control; April 1990, Vol 18, 99-113. To assist health care professionals
Endoscope Washer Disinfector Cleaning Process Challenge Devices Research & Validation
 Endoscope Washer Disinfector Cleaning Process Challenge Devices Research & Validation Richard Bancroft, B.Sc (Hons), FRSB Science & Technical Director STERIS Corporation Registered Authorising Engineer
Endoscope Washer Disinfector Cleaning Process Challenge Devices Research & Validation Richard Bancroft, B.Sc (Hons), FRSB Science & Technical Director STERIS Corporation Registered Authorising Engineer
ISO INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD ISO 14161 Second edition 2009-09-15 Sterilization of health care products Biological indicators Guidance for the selection, use and interpretation of results Stérilisation des produits
INTERNATIONAL STANDARD ISO 14161 Second edition 2009-09-15 Sterilization of health care products Biological indicators Guidance for the selection, use and interpretation of results Stérilisation des produits
ETD4. More than meets the eye.
 ETD4 More than meets the eye. More than meets the eye ETD4 intelligent reliable safe Setting endoscope reprocessing standards With ETD4, Olympus has made significant progress in providing you with even
ETD4 More than meets the eye. More than meets the eye ETD4 intelligent reliable safe Setting endoscope reprocessing standards With ETD4, Olympus has made significant progress in providing you with even
GLOBAL QUALITY SOLUTIONS PACKAGE INTEGRITY SEAL INTEGRITY LEAK DETECTION
 GLOBAL QUALITY SOLUTIONS PACKAGE INTEGRITY SEAL INTEGRITY LEAK DETECTION Redefining the standards for accuracy and reliability. About PTI PTI Packaging Technologies & Inspection is headquartered in Tuckahoe,
GLOBAL QUALITY SOLUTIONS PACKAGE INTEGRITY SEAL INTEGRITY LEAK DETECTION Redefining the standards for accuracy and reliability. About PTI PTI Packaging Technologies & Inspection is headquartered in Tuckahoe,
PACKAGE TESTING AND VALIDATION SERVICES
 PACKAGE TESTING AND VALIDATION SERVICES A sterile medical device, which includes the packaging, must function as labeled for the shelf-life claimed for the device. The primary package must maintain its
PACKAGE TESTING AND VALIDATION SERVICES A sterile medical device, which includes the packaging, must function as labeled for the shelf-life claimed for the device. The primary package must maintain its
Infection Control. Low Temperature Sterilization (LTS) Critical Aspects on Selection and Validation of LTS-Processes
 Infection Control Low Temperature Sterilization (LTS) Critical Aspects on Selection and Validation of LTS-Processes Dr. Tronje von dem Hagen tronje@vondemhagen.com Lübeck (Germany) T. v. dem Hagen, Lübeck/Germany
Infection Control Low Temperature Sterilization (LTS) Critical Aspects on Selection and Validation of LTS-Processes Dr. Tronje von dem Hagen tronje@vondemhagen.com Lübeck (Germany) T. v. dem Hagen, Lübeck/Germany
GETINGE 633HC STEAM STERILIZERS. Always with you
 GETINGE 633HC STEAM STERILIZERS Always with you GETINGE 633HC Steam Sterilizers 3 MAKING EVERY SQUARE FOOT COUNT With the ever increasing demands being placed on today s surgical workplaces, space limitations
GETINGE 633HC STEAM STERILIZERS Always with you GETINGE 633HC Steam Sterilizers 3 MAKING EVERY SQUARE FOOT COUNT With the ever increasing demands being placed on today s surgical workplaces, space limitations
