Global impact of hydrogen the future energy source
|
|
|
- Douglas Jordan
- 6 years ago
- Views:
Transcription
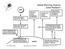
1 Global impact of hydrogen the future energy source Violeta-Carolina Niculescu, Mihai Anghel, Ioan Stefanescu Abstract In a future sustainable energy system based on renewable energy, environmentally harmless energy carriers like hydrogen, will be of crucial importance. One of the major impediments for the transition to a hydrogen based energy system is the lack of satisfactory hydrogen storage alternatives. Hydrogen storage in nanostructured materials has been proposed as a solution for adequate hydrogen storage for a number of applications, in particular for transportation. Also, hydrogen is an important, though little studied, trace component of the atmosphere. It exists at the mixing ratio of about 510 ppb. Hydrogen is an indirect greenhouse gas with a global warming potential GWP of 5.8 over a 100-year time horizon. A future hydrogen economy would therefore have greenhouse consequences and would not be free from climate perturbations. Our study focuses on the environmental impact of using hydrogen as a energy source. If a global hydrogen economy replaced the current fossil fuel-based energy system and exhibited a leakage rate of 1%, then it would produce a climate impact of 0.6% of the current fossil fuel based system. Hydrogen can be produced from a variety of feed stocks; from fossil resources such as natural gas and coal, and from renewable resources such as biomass and water with input from renewable energy sources (e.g. sunlight, wind, wave or hydro-power) [], []. Local availability of feedstock, maturity of technology, market applications and demand, policy issues and costs will influence the choice and timing of the various options for hydrogen production. An overview of the various feed stocks and process technologies is presented in Fig. 1.a [4] [5]: Keywords energy, environment, greenhouse effect, hydrogen economy, impact. I. INTRODUCTION Hydrogen seems to have low impact on the environment, but there are a number of uncertainties concerning the consequences of a large-scale shift towards a hydrogen economy. Hydrogen is forecast to become a major source of energy in the future, thus offering a potentially non-polluting, inexhaustible, efficient, and cost-attractive energy carrier [1]. In the last 10 years, the defining issues with respect to H economics have changed dramatically. However, refineries now have become net consumers of H in an effort to reduce pollution and meet environmental regulations. Manuscript received July 8, 010. This work was supported by Romanian Ministry of Education and Research, National Authority for Scientific Research, National Centre for Programmes Management under Project No. -154/008: Environment impact analysis regarding use on wide scale of hydrogen technologies. Violeta-Carolina Niculescu is with the National Research and Development Institute for Cryogenics and Isotopic Technologies ICIT, Ramnicu Valcea, Romania (phone: /ext. 19; fax: ; violeta@icsi.ro). Mihai Anghel is with the National Research and Development Institute for Cryogenics and Isotopic Technologies ICIT, Ramnicu Valcea, Romania ( mihai.anghel@icsi.ro). Ioan Stefanescu is with the National Research and Development Institute for Cryogenics and Isotopic Technologies ICIT, Ramnicu Valcea, Romania ( istef@icsi.ro). Fig. 1 Some feedstock, process alternatives (a) and main hydrogen pathways (b) 8
2 In Fig. 1.b a future hydrogen pathway is illustrated. Largescale hydrogen production is probable on the longer time scale. In the current and medium term the production options for hydrogen are first based on distributed hydrogen production from electrolysis of water and reforming of natural gas and coal. Larger centralized hydrogen production plants are more likely to be introduced at a later stage. These plants will probably be based on biomass or fossil fuels with CO - capture and storage. Scientists, engineers, industry and policy makers think that there are two ways to make a vehicle run on hydrogen: by using the hydrogen in an internal combustion engine or by using the hydrogen in a fuel cell. Hydrogen economy needs to solve the challenging problems involved with the synthesis of hydrogen, its storage, distribution and utilization in all of the sectors of the energy economy, transport included. But, first of all, it must be very good understand the fate and behaviour of hydrogen in the atmosphere and its major sources and sinks. Hydrogen itself, in contrast to most expectations, is a greenhouse gas and can be quantified by its global warming potential relative to carbon dioxide. II. POTENTIAL IMPACT OF HYDROGEN ON ATMOSPHERE Hydrogen is one of the major trace gases in the lower atmosphere or troposphere. The global mixing ratio of hydrogen is currently about 510 ppb, 500 ppb in the northern hemisphere and 50 ppb in the southern hemisphere [6]. Although its life cycle has been heavily influenced by human activities, its mixing ratios in the northern hemisphere are lower than those in the southern hemisphere, hydrogen being unusual among trace gases. This is caused by the main sink for hydrogen, surface uptake by soils, which accounts for 80% of the total loss of hydrogen from the atmosphere. The majority of the sink is therefore occurring over the continental land masses in the northern hemisphere. Recent analyses of long-term observations of hydrogen in the troposphere indicate that mixing ratios have remained fairly constant during the last decade [7]. The (incomplete) combustion of fossil fuel and biomass in boilers and internal combustion engines generates hydrogen along with carbon monoxide and carbon dioxide. This source represents about 40% of all the hydrogen released into the atmosphere. Another important source, accounting for an estimated 50% of atmospheric hydrogen emissions, is the atmospheric photochemical oxidation of methane ( 4 ) and non-methane hydrocarbons (NMHCs). Emissions from volcanoes, oceans and nitrogen-fixing legumes account for the remaining 10% [8], [9]. Movement of hydrogen into the upper atmosphere and then to space is negligible in terms of the global hydrogen budget. Instead, hydrogen is removed from the atmosphere largely through dry deposition at the surface and subsequent microbiological uptake in soils. The rate of uptake depends on microbial activity, soil texture and moisture content. This sink is largest in the northern hemisphere because of its larger landmass. TABLE I GLOBAL SOURCES AND SINKS FOR HYDROGEN Sources and sinks (Tg per year) a [1] [9] [1] Sources Methane oxidation NMHC b Biomass burning Anthropogenic c Nitrogen fixation Oceans Total sources Sinks OH-oxidation Soil uptake Total sinks a Tg = 10 1 g Non-methane hydrocarbons Hydrogen from fuel combustion The first analysis [10] estimated a global production rate of.9 Tg per year, largely from human activities and a global sink strength of 17.5 Tg per year, due to the reaction below: OH H H H O (1) In order to estimate the production of hydrogen from methane, through the photolysis of formaldehyde HO, a global box model has been used [11]: OH 4 H O () O O M () O NO O NO (4) O O HO HO (5) HO h H CO (6) The uptake of hydrogen by soils [10] is known like an additional sink for hydrogen. Also, global hydrogen budgets have been based on the oxidation rates of the primary emitted hydrocarbons and removal by OH-oxidation and uptake by soils [9], [1], [1] see table 1. For the representation of various sources and sinks of hydrogen in troposphere, it was used a D chemistry transport model STOEM [1]. Regarding man-made sources, these include fossil fuel combustion, mainly from motor vehicles with petrol, through the water gas reaction: CO H (7) O H CO Another source of hydrogen in troposphere is the biomass burning (incomplete combustion). Also, the surface ocean waters are generally supersaturated with hydrogen and so act as a small source [10]. Hydrogen is also formed as a byproduct of nitrogen fixation in leguminous plants [14]. The modelling study using STOEM [1] shows that the OH-oxidation of methane produces 15. Tg per year of 84
3 hydrogen and that of isoprene, 11.0 Tg per year and other organic compounds account for a further 4.1 Tg per year. The main chemical sink for hydrogen in the troposphere is the reaction with hydroxyl radicals - see reaction (1). The largest source of hydroxyl radicals is the reaction of electronically excited oxygen atoms O 1 D from the photolysis of ozone O, with water vapour H O: O (8) O 1 OH (9) 1 h O D O H O OH Another major sink for hydrogen is uptake by soils at the earth s surface, the rates depending on the nature of the soils, the properties of each trace gas and the transport of the trace gas through the atmospheric boundary layer by turbulence to the earth s surface. All these atmosphere reactions, closely linked one each other, determine the capacity of the atmosphere to neutralise pollutants. Field studies (Table 1) have indicated a dependence of the soil uptake of hydrogen on soil moisture content and on the ecosystem type growing on the soil [15] [17]. Soil uptake rates, expressed as dry deposition velocities, range from 1. mm s 1 for savannah systems to 0.1 mm s 1 for semi-desert systems. For an observed global hydrogen burden of 18 Tg [6], the global sink strength of 74.4 Tg per year, implies an atmospheric lifetime of.5 years or thereabouts for hydrogen [1]. Usually, the measured concentration of hydrogen in troposphere (the part of the atmosphere extending from the earth`s surface up to about 17 km) is 0.5 ppm and the measured concentration in stratosphere (17-50 km altitude) is ppm, but this concentration has an year cycle. For the northern hemisphere, the amplitude of variation is about ppm and the southern one is about ppm. These variations are due to the increased 4 and NMHC (nonmethane hydrocarbons) values in the winter and high values of oxidation rates in the summer. Another approach for modelling atmospheric chemistry of hydrogen uses Caltech/JPL D model [18] [0]. This model solves the continuity equation for all important long-lived species and includes all the chemistry recommended by NASA for stratospheric modelling [1]. In order to highlight the potential impact of an increase of H in the atmosphere, this model was used for two cases: (a) concentrations of H and 4 are assumed to equal their approximate current global annual means at Earth s surface; and (b), the concentration of H at Earth s surface is raised to. ppm. Intermediate cases are discussed below. Fig. presents simulated vertical profiles in concentrations of H and 4 in the atmosphere for these two cases and a comparison of case (a) with recent measurements. The model prediction for the vertical profile for case (b) is the dashed line in Fig.. Mixing ratios of 4 (solid line) and H (long-dashed line) are simulated by the two-dimensional model for January at 60 to 70 N for case (a). The H mixing ratio for case (b) is given by the short dashed line. The data (asterisks and crosses) are from the SOLVE balloon measurements [1]. The model uses a pressure coordinate, with altitude z = H ln( p 0 /p), where the scale height (H) = 6.95 km, p = pressure, and p 0 =surface pressure; z is approximately equal to the geometric altitude. The results show that the model correctly describes the relation between H and 4 concentrations in the lower stratosphere, which reflects the balance of photo-oxidative reactions that are important for converting H and 4 to H O. The simulation model suggests that a fourfold rise in surface H concentrations, such as might occur because of large rises in anthropogenic emissions, will lead to substantial moistening and cooling of the lower stratosphere and substantial decreases in stratospheric O. Fig. Simulated concentrations of H and 4 III. GREENHOUSE EFFECT OF HYDROGEN Scientists consider that any trace gas can be quantified as a greenhouse gas if, once released in the atmosphere, it interacts with the incoming solar radiation or with the outgoing terrestrial radiation [7]. Greenhouse gases exist in tiny fractions in the atmosphere only parts per million and even per billion. A minor change in concentration could, trigger big, unanticipated, and possibly traumatic change in the atmosphere. It is a highly leveraged situation, said John Firor, an atmospheric scientist at the National Center for Atmospheric Research in Boulder. Alan Lloyd, chairman of the California Air Resources Board and one of the 1990s pivotal personages on the American hydrogen scene, appointed in early 1999: Environmental pollution will likely represent the cold war of the next century []. The greenhouse effect of hydrogen was also simulated using STOEM model for global Lagrangian chemistry transport model []. The data used for this model were collected for 4 years and for three months, respectively. In the second case, the hydrogen emission source strength was increased so that a pulse containing an additional 40 Tg of hydrogen was emitted. The impact of the additional hydrogen on the composition of the model troposphere was followed by taking differences between the base and transient cases. These differences in composition between the two experiments were 85
4 termed excess concentrations. The effect of the additional emissions is the raise of the hydrogen burden, which decreases with an e-folding time of years (close to the atmospheric lifetime of hydrogen). The result of the increase in the atmospheric burden of hydrogen following the emission pulse was the variation of the concentrations for all the major troposphere free radical species and ultimately for troposphere ozone. Ozone production was stimulated in the transient case and that an excess ozone burden quickly developed following the emission pulse. The excess ozone burden decayed away with an e-folding time of about.5 years, close to the atmospheric lifetime of hydrogen. In this model [], the time-integrated radiative forcings were converted into Global Warming Potentials (GWPs) by comparison with the time-integrated radiative forcing of a reference gas (CO ). So, the GWP of hydrogen was expressed as the ratio of the time-integrated radiative forcing resulting from the emission of 1 Tg of hydrogen compared with that from the emission of 1 Tg of CO over a 100-year time horizon. The overall GWP for hydrogen was 5.8 over a 100- year time horizon [7]. The emission of hydrogen to the troposphere causes changes in the global distributions of methane and ozone the second and third most important greenhouse gases. Emissions of hydrogen lead to increased burdens of methane and ozone and increase the global radiative forcing. That is way we can say that hydrogen is an indirect radiatively active trace gas with a global warming potential [7]. These affirmations can lead to ask a simple question: a global hydrogen economy would have consequences for global warming and it would be better or worse than the fossil-fuel economy that it replaces? As an answer, there are some estimations regarding the global hydrogen production capacity required to replace the entire current fossil-fuel based energy system (about 500 Tg H per year). The global hydrogen economy with a leakage rate of 1% can have a climate impact of 0.6% of the fossil fuel system it replaces. At a leakage rate of 10%, the climate impact would be 6% of that of the fossil fuel system. IV. CONCLUSIONS One can ask a simple question: if hydrogen s benefits as a fuel are so great, why didn t hydrogen make significant inroads into our energy systems years or even decades ago? The answer is not so simple. It involves a complex array of interlocking factors. First, there was no real need for hydrogen as long as there were ample supplies of oil and natural gas and as long as environmental worries were not a concern. Fossil fuels were cheap, and hydrogen was as much as several times more expensive. Liquid hydrogen, the coldly exotic stuff that powers the Space Shuttle and experimental BMW sedans today, was a laboratory curiosity four or five decades ago []. Used as a fuel, it would reduce and eventually eliminate at least the man-made share of CO deposited in the atmosphere. Switching to hydrogen energy even perhaps to hydrogen from fossil fuels as a stopgap measure may help save our children s health and perhaps their lives. The potential environmental impact of hydrogen can be highlighted in few lines: increased hydrogen concentration can lower the natural oxidizing capacity of the atmosphere, increasing in this way the lifetime of air pollutants and greenhouse gases; it can increase water vapour concentration in the atmosphere, having consequences in cloud formation, stratospheric temperatures and ozone loss; it could exceed the uptake capacity of hydrogen by microorganisms in the soil, currently the main way in which hydrogen is removed from the atmosphere; if hydrogen is generated using electricity derived from burning coal, nitrogen oxides emissions could increase significantly; generating hydrogen from fossil fuels could lead to increased emissions of carbon dioxide, which would accelerate global warming, unless the CO is captured and stored; generating hydrogen from sustainable sources would reduce emissions of carbon monoxide and NO x, with a consequent fall in tropospheric ozone levels. This would improve air quality in many regions of the world. Furthermore, CO emissions would be reduced, thereby slowing the global warming trend. We can say that there is a confluence of drivers that point in the same direction towards hydrogen. These drivers include the requirement for a reduction in CO emissions, appalling urban air quality, legislation dictating zero-emission vehicles, progress in fuel cell technology, a move toward the use of local resources for energy production, the need to store intermittent renewable energy concerns about fossil-fuel resources, as well as the security of energy supplies. REFERENCES [1] J. N. Armor, Catalysis and the hydrogen economy, Catalysis Letters, vol. 101, no. -4, pp , June 005. [] H. Gunardson, Industrial Gases in Petrochemical Processing, New York: Marcel Dekker Inc., 1989, pp [] L. Schlapbach, A. Zuttel, Hydrogen-storage materials for mobile applications, Nature, vol. 414, pp. 5-57, Nov [4] A. Niedzwiecki, Storage, presented at National Hydrogen Vision Meeting, Washington, USA, Nov , 001. [5] R. Teitel, Hydrogen Storage in Glass Microspheres, Brookhaven National Laboratories, Long Island, Rep. BNL-5149, [6] P. G. Simmonds, R. G. Derwent, S. O Doherty, D. B. Ryall, L. P. Steele, R. L. Langenfelds, P. Salameh, H. J. Wang, C. H. Dimmer, and L. E. Hudson. (000, May). Continuous high-frequency observations of hydrogen at the Mace Head baseline atmospheric monitoring station over the period, J. Geophys. Res., vol. 105, no. D10, pp Available doi:10.109/000jd [7] R. G. Derwent, P. G. Simmonds, S. O Doherty, A. Manning, W. Collins, and D. Stevenson, Global environmental impacts of the hydrogen economy, Int. J. Nuclear Hydrogen Production and Application, vol. 1, no. 1, pp , May 006. [8] D. A. Hauglastine and D. H. Ehhalt. (00, Sept.). A three-dimensional model of molecular hydrogen in the troposphere, J. Geophys. Res., vol. 107, no. D17, pp Available doi:10.109/001jd [9] P. C. Novelli, M. L. Patricia, A. M. Kenneth, F. H. Dale, M. Richard, and W. E. James. (1999, July). Molecular hydrogen in the troposphere: Global distribution and budget, J. Geophys. Res., vol. 104, no. D, pp Available doi:10.109/1999jd [10] U. Schmidt, Molecular hydrogen in the atmosphere, Tellus, vol. 6, pp.78 90, Feb
5 [11] P. J. Crutzen and J. Fishman, Average concentrations of OH in the troposphere, and the budgets of 4, CO, H and CCl, Geophys. Res. Lett., vol. 4, no. 8, pp. 1-4, June [1] W. Seiler and R. Conrad, Contribution of tropical ecosystems to the global budgets of trace gases, especially 4, H, CO and N O, in The Geophysiology of Amazonia, R. E. Dickinson, Ed. New York: John Wiley and Sons, 1987, pp [1] M. G. Sanderson, W. J. Collins, R. G. Derwent, and C. E. Johnson, Simulation of global hydrogen levels using a Lagrangian threedimensional model, J. Atmos. Chem., vol. 46, no. 1, pp. 15 8, Sept. 00. [14] R. Conrad and W. Seiler. (1980, May). Contribution of hydrogen production by biological nitrogen fixation to the global hydrogen budget, J. Geophys. Res., vol. 85, no. C10, pp Available doi:10.109/jc085ic10p0549. [15] R. Conrad and W. Seiler. (1985, Jan.). Influence of temperature, moisture and organic carbon on the flux of H and CO between soil and atmosphere: field studies in subtropical regions, J. Geophys. Res., vol. 90, no. D, pp Available doi:10.109/jd090id0p [16] S. Yonemura, S. Kawashima, and H. Tsuruta, Continuous measurement of CO and H deposition velocities onto an andisol: uptake control by soil moisture, Tellus, vol. 51B, no., pp , July [17] S. Yonemura, S. Kawashima, and H. Tsuruta. (000, May) Carbon monoxide, hydrogen and methane uptake by soils in a temperate arable field and a forest, J. Geophys. Res., vol. 105, no. D11, pp Available doi:10.109/1999jd [18] K. T. Tromp, R.-L. Shia, M. Allen, J. M. Eiler, and Y. L. Yung. (00, June). Potential environmental impact of a hydrogen economy on the stratosphere, Science, vol. 00, pp , doi: /science Available: [19] Y. L. Yung and C. E. Miller. (1997, Dec.). Isotopic fractionation of stratospheric nitrous oxide, Science, vol. 78, pp , doi: /science Available: [0] Y. L. Yung and W. D. DeMore, Photochemistry of Planetary Atmospheres, New York: Oxford Univ. Press, 1999, ch.10. [1] W. B. DeMore, C. J. Howard, S. P. Sander A. R. Ravishankara, D. M. Golden, C. E. Kolb, R. F. Hampson, M. J. Molina, and M. J. Kurylo, Chemical Kinetics and Photochemical Data for Use in Stratospheric Modeling, Jet Propulsion Laboratory, California Institute of Technology, Pasadena, CA, JPL Publication 97-4, January [] Peter Hoffmann, Tomorrow s Energy: Hydrogen, Fuel Cells, and the Prospects for a Cleaner Planet, The Cambridge, Massachusetts: MIT Press, 001, pp [] R. G. Derwent, W. J. Collins, C. E. Johnson, and D. S. Stevenson, Transient behaviour of tropospheric ozone precursors in a global -D CTM and their indirect greenhouse effects, Climatic Change, vol. 49, no. 4, pp , June
Global Environmental Impacts of the Hydrogen Economy
 Global Environmental Impacts of the Hydrogen Economy Richard Derwent 1, Peter Simmonds 2, Simon O Doherty 2, Alistair Manning 3, William Collins 3, Colin Johnson 3, Michael Sanderson 3, David Stevenson
Global Environmental Impacts of the Hydrogen Economy Richard Derwent 1, Peter Simmonds 2, Simon O Doherty 2, Alistair Manning 3, William Collins 3, Colin Johnson 3, Michael Sanderson 3, David Stevenson
The next 2 weeks. Reading: IPCC (2007), Chap 7 (sections 7.4 and 7.5)
 PCC 588 Jan 15 The next 2 weeks Th. Jan 15: non-co 2 greenhouse gases CH 4 and N 2 O Tu. Jan 20: non-co 2 greenhouse gases: ozone, halocarbons Th. Jan 22: Aerosols and Climate Tu. Jan 27: Paper discussion
PCC 588 Jan 15 The next 2 weeks Th. Jan 15: non-co 2 greenhouse gases CH 4 and N 2 O Tu. Jan 20: non-co 2 greenhouse gases: ozone, halocarbons Th. Jan 22: Aerosols and Climate Tu. Jan 27: Paper discussion
Other GHGs. IPCC Climate Change 2007: The Physical Science Basis
 Other GHGs IPCC Climate Change 2007: The Physical Science Basis 1 Atmospheric Chemistry and other long-lived GHG during the industrial period 1750-2000 The radiative forcing of climate during the period
Other GHGs IPCC Climate Change 2007: The Physical Science Basis 1 Atmospheric Chemistry and other long-lived GHG during the industrial period 1750-2000 The radiative forcing of climate during the period
Aerosol from biomass burning and mineral aerosols. 1. What are aerosols from biomass burning?
 Lectures 40-41. Global change due to anthropogenic aerosols: Aerosol from biomass burning and mineral aerosols. Objectives: 1. What are aerosols from biomass burning? 2. What is mineral aerosol? 3. Direct
Lectures 40-41. Global change due to anthropogenic aerosols: Aerosol from biomass burning and mineral aerosols. Objectives: 1. What are aerosols from biomass burning? 2. What is mineral aerosol? 3. Direct
Module 7: Combustion and Environment Lecture 36: Atmosphere. The Lecture Contains: Atmosphere. Chemical Emission From Combustion
 The Lecture Contains: Atmosphere Chemical Emission From Combustion Chemicals From Combustion (Contd..) file:///d /Web%20Course/Dr.%20D.P.%20Mishra/Local%20Server/FOC/lecture36/36_1.htm[10/5/2012 4:32:17
The Lecture Contains: Atmosphere Chemical Emission From Combustion Chemicals From Combustion (Contd..) file:///d /Web%20Course/Dr.%20D.P.%20Mishra/Local%20Server/FOC/lecture36/36_1.htm[10/5/2012 4:32:17
Global Warming Science Solar Radiation
 SUN Ozone and Oxygen absorb 190-290 nm. Latent heat from the surface (evaporation/ condensation) Global Warming Science Solar Radiation Turbulent heat from the surface (convection) Some infrared radiation
SUN Ozone and Oxygen absorb 190-290 nm. Latent heat from the surface (evaporation/ condensation) Global Warming Science Solar Radiation Turbulent heat from the surface (convection) Some infrared radiation
POTENTIAL ENVIRONMENTAL IMPACTS OF HYDROGEN-BASED TRANSPORTATION & POWER SYSTEMS
 POTENTIAL ENVIRONMENTAL IMPACTS OF HYDROGEN-BASED TRANSPORTATION & POWER SYSTEMS Tetra Tech, Inc. Stanford University Potomac Hudson Engineering, Inc. Tom Grieb Tetra Tech, Inc. Research & Development
POTENTIAL ENVIRONMENTAL IMPACTS OF HYDROGEN-BASED TRANSPORTATION & POWER SYSTEMS Tetra Tech, Inc. Stanford University Potomac Hudson Engineering, Inc. Tom Grieb Tetra Tech, Inc. Research & Development
GLOBAL WARMING COMPUTER LAB
 GLOBAL WARMING COMPUTER LAB A COMPUTER SIMULATION PROGRAM ON TEMPERATURE CHANGE AND SEA LEVEL RISING After performing this computer simulation lab you will be able to: 1) understand the greenhouse effect
GLOBAL WARMING COMPUTER LAB A COMPUTER SIMULATION PROGRAM ON TEMPERATURE CHANGE AND SEA LEVEL RISING After performing this computer simulation lab you will be able to: 1) understand the greenhouse effect
The role of carbon dioxide in climate forcing from 1979 to 2004: introduction of the Annual Greenhouse Gas Index
 Tellus (2006), 58B, 614 619 Copyright C Blackwell Munksgaard, 2006 Printed in Singapore. All rights reserved TELLUS The role of carbon dioxide in climate forcing from 1979 to 2004: introduction of the
Tellus (2006), 58B, 614 619 Copyright C Blackwell Munksgaard, 2006 Printed in Singapore. All rights reserved TELLUS The role of carbon dioxide in climate forcing from 1979 to 2004: introduction of the
CURRENT AND FUTURE SURFACE OZONE LEVELS OVER EUROPE.
 CURRENT AND FUTURE SURFACE OZONE LEVELS OVER EUROPE Michael G. Sanderson 1, William J. Collins 1, Colin. E. Johnson 1, Richard G. Derwent 2, David S. Stevenson 3 1 Hadley Centre, Met Office, Fitzroy Road,
CURRENT AND FUTURE SURFACE OZONE LEVELS OVER EUROPE Michael G. Sanderson 1, William J. Collins 1, Colin. E. Johnson 1, Richard G. Derwent 2, David S. Stevenson 3 1 Hadley Centre, Met Office, Fitzroy Road,
Major Volcanic Eruptions in the past. Major Volcanic Eruptions in the past. Volcanic Eruptions and Global Temperature
 Mechanism of Volcanic Perturbation Amount of sunlight scattered depends greatly on size and amount of aerosol particles The global monitoring of aerosols began in ~1980 Hence, the history of the amplitude
Mechanism of Volcanic Perturbation Amount of sunlight scattered depends greatly on size and amount of aerosol particles The global monitoring of aerosols began in ~1980 Hence, the history of the amplitude
Energy, Greenhouse Gases and the Carbon Cycle
 Energy, Greenhouse Gases and the Carbon Cycle David Allen Gertz Regents Professor in Chemical Engineering, and Director, Center for Energy and Environmental Resources Concepts for today Greenhouse Effect
Energy, Greenhouse Gases and the Carbon Cycle David Allen Gertz Regents Professor in Chemical Engineering, and Director, Center for Energy and Environmental Resources Concepts for today Greenhouse Effect
Ozone air quality and radiative forcing consequences of changes in ozone precursor emissions
 Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 34, L06806, doi:10.1029/2006gl029173, 2007 Ozone air quality and radiative forcing consequences of changes in ozone precursor emissions J.
Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 34, L06806, doi:10.1029/2006gl029173, 2007 Ozone air quality and radiative forcing consequences of changes in ozone precursor emissions J.
Radiative forcing of climate change
 Radiative forcing of climate change Joanna D. Haigh Imperial College of Science, Technology and Medicine, London Radiative forcing concept, definition and applications On a global and annual average, and
Radiative forcing of climate change Joanna D. Haigh Imperial College of Science, Technology and Medicine, London Radiative forcing concept, definition and applications On a global and annual average, and
Biogenic and Biomass Burning Sources of Acetone to the Troposphere
 UCRL-ID-127304 Biogenic and Biomass Burning Sources of Acetone to the Troposphere Cynthia S. Atherton April 1997 Lawrence Livermore National Laboratory This is an informal report intended primarily for
UCRL-ID-127304 Biogenic and Biomass Burning Sources of Acetone to the Troposphere Cynthia S. Atherton April 1997 Lawrence Livermore National Laboratory This is an informal report intended primarily for
Lecture 2: Greenhouse Gases - Basic Background on Atmosphere - GHG Emission and Concentration Rise - California Regulation (AB32)
 Lecture 2: Greenhouse Gases - Basic Background on Atmosphere - GHG Emission and Concentration Rise - California Regulation (AB32) METR 113/ENVS 113 Spring Semester 2011 February 15, 2011 Suggested Reading
Lecture 2: Greenhouse Gases - Basic Background on Atmosphere - GHG Emission and Concentration Rise - California Regulation (AB32) METR 113/ENVS 113 Spring Semester 2011 February 15, 2011 Suggested Reading
Figure 1 - Global Temperatures - A plot from the EarthScience Centre at
 GLOBAL WARMING Global warming is evidenced by a steady rise in average global temperatures, changing climate, the fact that snow cover has decreased 10% over the past half-century and that glaciers have
GLOBAL WARMING Global warming is evidenced by a steady rise in average global temperatures, changing climate, the fact that snow cover has decreased 10% over the past half-century and that glaciers have
Leif Backman HENVI Seminar February 19, 2009
 Methane Sources and Sinks Leif Backman HENVI Seminar February 19, 2009 Background Atmospheric methane Sources & Sinks Concentration variations & trends Objective & methods Objective & Goals Research plan
Methane Sources and Sinks Leif Backman HENVI Seminar February 19, 2009 Background Atmospheric methane Sources & Sinks Concentration variations & trends Objective & methods Objective & Goals Research plan
TODAY: TOPIC #6 WRAP UP!! Atmospheric Structure & Composition
 TODAY: TOPIC #6 WRAP UP!! Atmospheric Structure & Composition There s one more thing to correct in our the depiction of incoming Solar....... the atmosphere is NOT totally TRANSPARENT to INCOMING Solar
TODAY: TOPIC #6 WRAP UP!! Atmospheric Structure & Composition There s one more thing to correct in our the depiction of incoming Solar....... the atmosphere is NOT totally TRANSPARENT to INCOMING Solar
Radiative forcing of gases, aerosols and, clouds.
 Lecture 23. Radiative forcing of gases, aerosols and, clouds. 1. Concepts of radiative forcing, climate sensitivity, and radiation feedbacks. 2. Radiative forcing of anthropogenic greenhouse gases. 3.
Lecture 23. Radiative forcing of gases, aerosols and, clouds. 1. Concepts of radiative forcing, climate sensitivity, and radiation feedbacks. 2. Radiative forcing of anthropogenic greenhouse gases. 3.
2. Climate Change: Projections of Climate Change: 2100 and beyond
 Global Warming: Science, Projections and Uncertainties Global Warming: Science, Projections and Uncertainties An overview of the basic science An overview of the basic science 1. A Brief History of Global
Global Warming: Science, Projections and Uncertainties Global Warming: Science, Projections and Uncertainties An overview of the basic science An overview of the basic science 1. A Brief History of Global
Global Climate Change. The sky is falling! The sky is falling!
 Global Climate Change The sky is falling! The sky is falling! 1 Global Climate Change Radiative Equilibrium, Solar and Earth Radiation Atmospheric Greenhouse Effect Greenhouse Gases Global Climate Change
Global Climate Change The sky is falling! The sky is falling! 1 Global Climate Change Radiative Equilibrium, Solar and Earth Radiation Atmospheric Greenhouse Effect Greenhouse Gases Global Climate Change
4-9 Chemistry/5-9 Trilogy Chemistry of the atmosphere
 4-9 Chemistry/5-9 Trilogy Chemistry of the atmosphere.0 This question is about fuels.. There are two main types of diesel fuel used for cars: biodiesel, made from vegetable oils petroleum diesel, made
4-9 Chemistry/5-9 Trilogy Chemistry of the atmosphere.0 This question is about fuels.. There are two main types of diesel fuel used for cars: biodiesel, made from vegetable oils petroleum diesel, made
Atmospheric chemistry Summary
 Atmospheric chemistry Summary Pontus Roldin Div. Nuclear Physics Department of Physics Lund University Summary 1 Chapman mechanism (1930) The Chapman mechanism for stratospheric ozone (1) O 2 + hn O +
Atmospheric chemistry Summary Pontus Roldin Div. Nuclear Physics Department of Physics Lund University Summary 1 Chapman mechanism (1930) The Chapman mechanism for stratospheric ozone (1) O 2 + hn O +
Hydrogen as an Energy Carrier
 CH2356 Energy Engineering Hydrogen as an Energy Carrier Dr. M. Subramanian 28-Feb-2011 Associate Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering Kalavakkam
CH2356 Energy Engineering Hydrogen as an Energy Carrier Dr. M. Subramanian 28-Feb-2011 Associate Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering Kalavakkam
Climate Change Vocabulary Global Challenges for the 21 st Century Tony Del Vecchio, M.Ed. Atmosphere
 Atmosphere The mixture of gases surrounding the Earth. The Earth's atmosphere consists of about 79.1% nitrogen (by volume), 20.9% oxygen, 0.036% carbon dioxide and trace amounts of other gases. The atmosphere
Atmosphere The mixture of gases surrounding the Earth. The Earth's atmosphere consists of about 79.1% nitrogen (by volume), 20.9% oxygen, 0.036% carbon dioxide and trace amounts of other gases. The atmosphere
Quantifying Molecular Hydrogen Emissions and an Industrial Leakage Rate for the South Coast Air Basin of California
 Quantifying Molecular Hydrogen Emissions and an Industrial Leakage Rate for the South Coast Air Basin of California MATT IRISH 1, JASON SCHROEDER 2, ANDREAS BEYERSDORF 3, and DONALD BLAKE 2 1 Climate &
Quantifying Molecular Hydrogen Emissions and an Industrial Leakage Rate for the South Coast Air Basin of California MATT IRISH 1, JASON SCHROEDER 2, ANDREAS BEYERSDORF 3, and DONALD BLAKE 2 1 Climate &
Page 2. Q1.Greenhouse gases affect the temperature of the Earth. Which gas is a greenhouse gas? Tick one box. Argon. Methane. Nitrogen.
 Q1.Greenhouse gases affect the temperature of the Earth. (a) Which gas is a greenhouse gas? Tick one box. Argon Methane Nitrogen Oxygen (b) An increase in global temperature will cause climate change.
Q1.Greenhouse gases affect the temperature of the Earth. (a) Which gas is a greenhouse gas? Tick one box. Argon Methane Nitrogen Oxygen (b) An increase in global temperature will cause climate change.
Climate Dynamics (PCC 587): Climate Forcings
 Climate Dynamics (PCC 587): Climate Forcings DARGAN M. W. FRIERSON UNIVERSITY OF WASHINGTON, DEPARTMENT OF ATMOSPHERIC SCIENCES DAY 7: 10-16-13 Outline of This Topic Climate forcings Things that directly
Climate Dynamics (PCC 587): Climate Forcings DARGAN M. W. FRIERSON UNIVERSITY OF WASHINGTON, DEPARTMENT OF ATMOSPHERIC SCIENCES DAY 7: 10-16-13 Outline of This Topic Climate forcings Things that directly
Effect of Aviation on Atmospheric Composition
 Effect of Aviation on Atmospheric Composition Cynthia Whaley, Kimberly Strong, Zen Mariani, and Steven Barrett (MIT) UTIAS Colloquium on Sustainable Aviation 15-16 May 2013 Outline Introduction Composition
Effect of Aviation on Atmospheric Composition Cynthia Whaley, Kimberly Strong, Zen Mariani, and Steven Barrett (MIT) UTIAS Colloquium on Sustainable Aviation 15-16 May 2013 Outline Introduction Composition
In 2011 burning of fossil fuels provided 83% of mankind s energy resource while nuclear electric power provided 9%, and renewable energy 8% (1).
 Hierarchy of Global Energy Sources and Related Risks John Bushell January 2015 johnbushell@jbvm.com.au Abstract This paper reviews future energy resource options required to provide mankind with the energy
Hierarchy of Global Energy Sources and Related Risks John Bushell January 2015 johnbushell@jbvm.com.au Abstract This paper reviews future energy resource options required to provide mankind with the energy
Climate Change and Air Quality
 Climate Change and Air Quality SW PA Air Quality Action June 6, 2007 Peter J. Adams Associate Professor Civil and Environmental Engineering Engineering and Public Policy Outline Climate Change Primer What
Climate Change and Air Quality SW PA Air Quality Action June 6, 2007 Peter J. Adams Associate Professor Civil and Environmental Engineering Engineering and Public Policy Outline Climate Change Primer What
GLOBAL CLIMATE CHANGE
 1 GLOBAL CLIMATE CHANGE From About Transportation and Climate Change (Source; Volpe center for Climate Change and Environmental forecasting, http://climate.volpe.dot.gov/trans.html Greenhouse effect has
1 GLOBAL CLIMATE CHANGE From About Transportation and Climate Change (Source; Volpe center for Climate Change and Environmental forecasting, http://climate.volpe.dot.gov/trans.html Greenhouse effect has
Physics 100 Lecture 17. The Greenhouse Effect and Global Warming April 2, 2018
 1 Physics 100 Lecture 17 The Greenhouse Effect and Global Warming April 2, 2018 2 Class Quiz Ch. 7: Suppose your car burned bituminous coal instead of gasoline. How much coal would provide the same energy
1 Physics 100 Lecture 17 The Greenhouse Effect and Global Warming April 2, 2018 2 Class Quiz Ch. 7: Suppose your car burned bituminous coal instead of gasoline. How much coal would provide the same energy
MAE 119 W2018 FINAL EXAM PROF. G.R..TYNAN Part I: MULTIPLE CHOICE SECTION 2 POINTS EACH
 MAE 119 W2018 FINAL EXAM PROF. G.R..TYNAN Part I: MULTIPLE CHOICE SECTION 2 POINTS EACH 1. Which best describes the working definition of energy used in class: a. Energy can be transformed and in doing
MAE 119 W2018 FINAL EXAM PROF. G.R..TYNAN Part I: MULTIPLE CHOICE SECTION 2 POINTS EACH 1. Which best describes the working definition of energy used in class: a. Energy can be transformed and in doing
Effects of Greenhouse Gas Emission
 Effects of Greenhouse Gas Emission Reshmi Banerjee Assistant Professor, Dept. of EE, Guru Nanak Institute of Technology, Kolkata, W.B., India ABSTRACT: Gases that trap heat in the atmosphere are called
Effects of Greenhouse Gas Emission Reshmi Banerjee Assistant Professor, Dept. of EE, Guru Nanak Institute of Technology, Kolkata, W.B., India ABSTRACT: Gases that trap heat in the atmosphere are called
/ Past and Present Climate
 MIT OpenCourseWare http://ocw.mit.edu 12.842 / 12.301 Past and Present Climate Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 12.842 Climate
MIT OpenCourseWare http://ocw.mit.edu 12.842 / 12.301 Past and Present Climate Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 12.842 Climate
Greenhouse Effect. The Greenhouse Effect
 Greenhouse Effect The Greenhouse Effect Greenhouse gases let short-wavelength radiation come into the Earth s atmosphere from the sun. However, they absorb and re-radiate Earth s long-wavelength radiation
Greenhouse Effect The Greenhouse Effect Greenhouse gases let short-wavelength radiation come into the Earth s atmosphere from the sun. However, they absorb and re-radiate Earth s long-wavelength radiation
Human nitrogen fixation and greenhouse gas emissions: a global assessment
 Human nitrogen fixation and greenhouse gas emissions: a global assessment Wim de Vries 1,2, Enzai Du 3, Klaus Butterbach-Bahl 4, Lena Schulte-Uebbing 2, Frank Dentener 5 1 Alterra Wageningen University
Human nitrogen fixation and greenhouse gas emissions: a global assessment Wim de Vries 1,2, Enzai Du 3, Klaus Butterbach-Bahl 4, Lena Schulte-Uebbing 2, Frank Dentener 5 1 Alterra Wageningen University
ENVIS- IITM NEWSLETTER The Air Quality: A Global Challenge
 ENVIS- IITM NEWSLETTER The Air Quality: A Global Challenge GLOBAL WARMING Editorial Prof. B.N. Goswami (Director, IITM, Pune) Dr. G. Beig (ENVIS Co-ordinetor) Ms. Neha S. Parkhi (Program Officer) Mr. Rajnikant
ENVIS- IITM NEWSLETTER The Air Quality: A Global Challenge GLOBAL WARMING Editorial Prof. B.N. Goswami (Director, IITM, Pune) Dr. G. Beig (ENVIS Co-ordinetor) Ms. Neha S. Parkhi (Program Officer) Mr. Rajnikant
Atmospheric Chemistry
 Atmospheric Chemistry Research Dr. Husam T. Majeed Department of Atmospheric Sciences 2nd Year Class First Semester 2018-2019 Course Syllabus 1. Introduction & Review Atmospheric Chemistry Box Models and
Atmospheric Chemistry Research Dr. Husam T. Majeed Department of Atmospheric Sciences 2nd Year Class First Semester 2018-2019 Course Syllabus 1. Introduction & Review Atmospheric Chemistry Box Models and
Global Warming Potentials in AR4. V. Ramaswamy. NOAA/ Geophysical Fluid Dynamics Laboratory, Princeton University
 Global Warming Potentials in AR4 V. Ramaswamy NOAA/ Geophysical Fluid Dynamics Laboratory, Princeton University GWP Definition Defined as the ratio of the time-integrated radiative forcing from the instantaneous
Global Warming Potentials in AR4 V. Ramaswamy NOAA/ Geophysical Fluid Dynamics Laboratory, Princeton University GWP Definition Defined as the ratio of the time-integrated radiative forcing from the instantaneous
Chemical mechanisms and kinetics in atmospheric chemistry Lecture 8: Global budgets and emissions inventories
 Chemical mechanisms and kinetics in atmospheric chemistry Lecture 8: Global budgets and emissions inventories Mike Pilling University of Leeds UK Synopsis Global budgets for CH 4, CO, NO x, VOCs (tomorrow
Chemical mechanisms and kinetics in atmospheric chemistry Lecture 8: Global budgets and emissions inventories Mike Pilling University of Leeds UK Synopsis Global budgets for CH 4, CO, NO x, VOCs (tomorrow
Concentrations of several of these greenhouse gases (CO 2, CH 4, N 2 O and CFCs) have increased dramatically in the last hundred years due to human
 Global Warming 1.1 The facts: With no atmosphere surrounding the earth the surface temperature would be 17 o C. However, due to the greenhouse gases in the atmosphere that absorb infrared radiation emitted
Global Warming 1.1 The facts: With no atmosphere surrounding the earth the surface temperature would be 17 o C. However, due to the greenhouse gases in the atmosphere that absorb infrared radiation emitted
A Hydrogen Economy. Dr. Mazen Abualtayef. Environmental Engineering Department. Islamic University of Gaza, Palestine
 A Hydrogen Economy Dr. Mazen Abualtayef Environmental Engineering Department Islamic University of Gaza, Palestine Adapted from a presentation by Professor S.R. Lawrence, Leeds School of Business, Environmental
A Hydrogen Economy Dr. Mazen Abualtayef Environmental Engineering Department Islamic University of Gaza, Palestine Adapted from a presentation by Professor S.R. Lawrence, Leeds School of Business, Environmental
Session 14 Unit VI CLIMATIC CHANGE AND GLOBAL WARMING
 Session 14 Unit VI CLIMATIC CHANGE AND GLOBAL WARMING Dr. H.S. Ramesh Professor of Environmental Engineering S.J. College of Engineering, Mysore 570 006 Carbon di-oxide is a natural constituent of atmosphere,
Session 14 Unit VI CLIMATIC CHANGE AND GLOBAL WARMING Dr. H.S. Ramesh Professor of Environmental Engineering S.J. College of Engineering, Mysore 570 006 Carbon di-oxide is a natural constituent of atmosphere,
Interesting points from presentations
 Interesting points from presentations The topics can be broken down, roughly, into the following categories Emissions of NOx, CO, VOCs, and other gases that influence ozone (and, thus, OH) Electric Vehicles
Interesting points from presentations The topics can be broken down, roughly, into the following categories Emissions of NOx, CO, VOCs, and other gases that influence ozone (and, thus, OH) Electric Vehicles
Climate Change. Air Quality. Stratospheric. Ozone. NAS study on International Transport of Air Pollution NOAA Perspective
 Climate Change Air Quality Stratospheric Ozone NAS study on International Transport of Air Pollution NOAA Perspective A.R. Ravishankara NOAA Climate Research and Modeling Program David Parrish NOAA/ESRL/Chemical
Climate Change Air Quality Stratospheric Ozone NAS study on International Transport of Air Pollution NOAA Perspective A.R. Ravishankara NOAA Climate Research and Modeling Program David Parrish NOAA/ESRL/Chemical
THE ROLE OF THE HYDROGEN FUEL CELL IN PROVIDING CLEAN ENERGY OF THE FUTURE
 THE ROLE OF THE HYDROGEN FUEL CELL IN PROVIDING CLEAN ENERGY OF THE FUTURE ENVIRONMENTAL SUSTAINABILITY 1983 Brundtland Commission- Our Common Future 1987 Sustainable Development Development that meets
THE ROLE OF THE HYDROGEN FUEL CELL IN PROVIDING CLEAN ENERGY OF THE FUTURE ENVIRONMENTAL SUSTAINABILITY 1983 Brundtland Commission- Our Common Future 1987 Sustainable Development Development that meets
UN Climate Council Words in red are defined in vocabulary section (pg. 9)
 UN Climate Council Words in red are defined in vocabulary section (pg. 9) To minimize the negative effects of global climate change, scientists have advocated for action to limit global warming to no more
UN Climate Council Words in red are defined in vocabulary section (pg. 9) To minimize the negative effects of global climate change, scientists have advocated for action to limit global warming to no more
Emission of Carbon Monoxide during Composting of Dung and Green Waste
 - 1 - Emission of Carbon Monoxide during Composting of Dung and Green Waste H. J. Hellebrand and W.-D. Kalk Institute of Agricultural Engineering Bornim, Max-Eyth-Allee 1, D-1449 Potsdam Introduction Carbon
- 1 - Emission of Carbon Monoxide during Composting of Dung and Green Waste H. J. Hellebrand and W.-D. Kalk Institute of Agricultural Engineering Bornim, Max-Eyth-Allee 1, D-1449 Potsdam Introduction Carbon
Chapter 19 Global Change. Wednesday, April 18, 18
 Chapter 19 Global Change Module 62 Global Climate Change and the Greenhouse Effect After reading this module you should be able to distinguish among global change, global climate change, and global warming.
Chapter 19 Global Change Module 62 Global Climate Change and the Greenhouse Effect After reading this module you should be able to distinguish among global change, global climate change, and global warming.
The Chemistry of Climate Change. Reading: Chapter 8 Environmental Chemistry, G. W. vanloon. S. J. Duffy
 The Chemistry of Climate Change Reading: Chapter 8 Environmental Chemistry, G. W. vanloon. S. J. Duffy The Science of Global Climate There's a lot of differing data, but as far as I can gather, over the
The Chemistry of Climate Change Reading: Chapter 8 Environmental Chemistry, G. W. vanloon. S. J. Duffy The Science of Global Climate There's a lot of differing data, but as far as I can gather, over the
Normal equatorial flow. Climate Variability. El Niño-Southern Oscillation Human-caused climate change Alternative Energy sources
 Normal equatorial flow Climate Variability El Niño-Southern Oscillation Human-caused climate change Alternative Energy sources Vertical normal flow ENSO disruption of flow Vertical ENSO flow Normal vs.
Normal equatorial flow Climate Variability El Niño-Southern Oscillation Human-caused climate change Alternative Energy sources Vertical normal flow ENSO disruption of flow Vertical ENSO flow Normal vs.
Earth s Dynamic Climate
 UNIT 3 Earth s Dynamic Climate Topic 3.1: What is climate, and how has it changed during Earth s history? Topic 3.2 : Where are the effects of climate change felt, and what is their impact? Topic 3.5:
UNIT 3 Earth s Dynamic Climate Topic 3.1: What is climate, and how has it changed during Earth s history? Topic 3.2 : Where are the effects of climate change felt, and what is their impact? Topic 3.5:
Chapter 6 of WGI AR6: Intention at the scoping meeting and the outline
 Chapter 6 of WGI AR6: Intention at the scoping meeting and the outline William Collins 1 and Hong Liao 2 Chapter 6 CLAs 1 Lawrence Berkeley Laboratory and University of California, Berkeley 2 Nanjing University
Chapter 6 of WGI AR6: Intention at the scoping meeting and the outline William Collins 1 and Hong Liao 2 Chapter 6 CLAs 1 Lawrence Berkeley Laboratory and University of California, Berkeley 2 Nanjing University
Atmospheric chemistry Summary Presented by Pontus Roldin Material from Erik Swietlicki Avd. för Kärnfysik Fysiska institutionen Lunds universitet
 Atmospheric chemistry Summary Presented by Pontus Roldin Material from Erik Swietlicki Avd. för Kärnfysik Fysiska institutionen Lunds universitet Summary 1 Chapman mechanism (1930) The Chapman mechanism
Atmospheric chemistry Summary Presented by Pontus Roldin Material from Erik Swietlicki Avd. för Kärnfysik Fysiska institutionen Lunds universitet Summary 1 Chapman mechanism (1930) The Chapman mechanism
Figure 1 CO2 Levels vs. Volcanic Activity
 Figure 1 CO2 Levels vs. Volcanic Activity Note: Stratospheric Optical Thickness is the measure of aerosols (e.g., urban haze, smoke particles, desert dust, sea salt) distributed within a column of air
Figure 1 CO2 Levels vs. Volcanic Activity Note: Stratospheric Optical Thickness is the measure of aerosols (e.g., urban haze, smoke particles, desert dust, sea salt) distributed within a column of air
Dr David Karoly School of Meteorology
 Global warming: Is it real? Does it matter for a chemical engineer? Dr David Karoly School of Meteorology Email: dkaroly@ou.edu Recent global warming quotes Senator James Inhofe (R, Oklahoma), Chair, Senate
Global warming: Is it real? Does it matter for a chemical engineer? Dr David Karoly School of Meteorology Email: dkaroly@ou.edu Recent global warming quotes Senator James Inhofe (R, Oklahoma), Chair, Senate
climate change Contents CO 2 (ppm)
 climate change CO 2 (ppm) 2007 Joachim Curtius Institut für Physik der Atmosphäre Universität Mainz Contents 1. Summary 2. Background 3. Climate change: observations 4. CO 2 : ocean acidification 5. OtherGreenhouse
climate change CO 2 (ppm) 2007 Joachim Curtius Institut für Physik der Atmosphäre Universität Mainz Contents 1. Summary 2. Background 3. Climate change: observations 4. CO 2 : ocean acidification 5. OtherGreenhouse
Monday, October 29, Coming up: Short-term organic carbon cycle (p ) Marine organic carbon cycle and nutrient limitation (p )
 Monday, October 29, 2017 Assessment available until Friday Topic - The Carbon Cycle (Chapter 8) Overview of the carbon cycle (p 149-151) Reservoirs (p 151-152) Steady State (p 152-153) Residence Time (p
Monday, October 29, 2017 Assessment available until Friday Topic - The Carbon Cycle (Chapter 8) Overview of the carbon cycle (p 149-151) Reservoirs (p 151-152) Steady State (p 152-153) Residence Time (p
Climate Change, Greenhouse Gases and Aerosols
 Climate Change, Greenhouse Gases and Aerosols J Srinivasan J Srinivasan is a Professor at the Centre for Atmospheric and Oceanic Sciences at Indian Institute of Science, Bangalore. He was a lead author
Climate Change, Greenhouse Gases and Aerosols J Srinivasan J Srinivasan is a Professor at the Centre for Atmospheric and Oceanic Sciences at Indian Institute of Science, Bangalore. He was a lead author
A note on New Zealand s methane emissions from livestock
 A note on New Zealand s methane emissions from livestock August 2018 This report has been produced pursuant to subsections 16(1)(a) to (c) of the Environment Act 1986. The Parliamentary Commissioner for
A note on New Zealand s methane emissions from livestock August 2018 This report has been produced pursuant to subsections 16(1)(a) to (c) of the Environment Act 1986. The Parliamentary Commissioner for
Lightning and Atmospheric Chemistry
 Lightning and Atmospheric Chemistry 1785 Cavendish performed the first experiments with a spark discharge in glass tube. Discovered that oxidized nitrogen (NO x =NO + NO 2 ) compounds resulted from the
Lightning and Atmospheric Chemistry 1785 Cavendish performed the first experiments with a spark discharge in glass tube. Discovered that oxidized nitrogen (NO x =NO + NO 2 ) compounds resulted from the
1. The diagram below shows a greenhouse.
 1. The diagram below shows a greenhouse. 5. A gradual increase in atmospheric carbon dioxide would warm Earth s because carbon dioxide is a A) poor reflector of ultraviolet radiation B) good reflector
1. The diagram below shows a greenhouse. 5. A gradual increase in atmospheric carbon dioxide would warm Earth s because carbon dioxide is a A) poor reflector of ultraviolet radiation B) good reflector
Lesson 9. Objectives: ocus: Subjects: 1. To understand that carbon cycles from one form. 1. Science: Ecology & Chemistry 2. Reading / Language Arts
 Lesson 9 Forests, Carbon & Our Climate Objectives: 1. To understand that carbon cycles from one form to another and the role that forests play in this process. 2. To understand where and how carbon is
Lesson 9 Forests, Carbon & Our Climate Objectives: 1. To understand that carbon cycles from one form to another and the role that forests play in this process. 2. To understand where and how carbon is
Fuels. N4 & N5 Homework Questions
 St Peter the Apostle High school Chemistry Department Fuels N4 & N5 Homework Questions Answer questions as directed by your teacher. National 4 level questions are first followed by National 5 level questions.
St Peter the Apostle High school Chemistry Department Fuels N4 & N5 Homework Questions Answer questions as directed by your teacher. National 4 level questions are first followed by National 5 level questions.
Global Climatic Change. GEOG/ENST 2331 Lecture 22 Ahrens: Chapter 16
 Global Climatic Change GEOG/ENST 2331 Lecture 22 Ahrens: Chapter 16 Global Climatic Change! Review: Radiation balance! Enhanced greenhouse effect! human-induced change! Climate feedbacks Climatic change!
Global Climatic Change GEOG/ENST 2331 Lecture 22 Ahrens: Chapter 16 Global Climatic Change! Review: Radiation balance! Enhanced greenhouse effect! human-induced change! Climate feedbacks Climatic change!
air pollution air pollution atmospheric pollution atmosphere unit 9
 air pollution unit 9 air pollution health effects WHO estimates that air pollution killed 7 million people in 2012 - more than double previous estimates indoor vs. outdoor (ambient) household pollution
air pollution unit 9 air pollution health effects WHO estimates that air pollution killed 7 million people in 2012 - more than double previous estimates indoor vs. outdoor (ambient) household pollution
Interconnections between Air Pollution, Climate Change and Health
 Interconnections between Air Pollution, Climate Change and Health Denise Mauzerall Princeton University National Academies Institute of Medicine San Francisco, CA September 10, 2007 Air Pollution Adversely
Interconnections between Air Pollution, Climate Change and Health Denise Mauzerall Princeton University National Academies Institute of Medicine San Francisco, CA September 10, 2007 Air Pollution Adversely
What the Snowball Earth Events of the Past May Offer in 2018 in the Accelerating Face of Excessive Greenhouse Gases and Continued Ozone Layer Issues
 What the Snowball Earth Events of the Past May Offer in 2018 in the Accelerating Face of Excessive Greenhouse Gases and Continued Ozone Layer Issues Viva Cundliffe, September 3, 2018 pre publication copy
What the Snowball Earth Events of the Past May Offer in 2018 in the Accelerating Face of Excessive Greenhouse Gases and Continued Ozone Layer Issues Viva Cundliffe, September 3, 2018 pre publication copy
Carbon Dioxide and Global Warming Case Study
 Carbon Dioxide and Global Warming Case Study Key Concepts: Greenhouse Gas Carbon dioxide El Niño Global warming Greenhouse effect Greenhouse gas La Niña Land use Methane Nitrous oxide Radiative forcing
Carbon Dioxide and Global Warming Case Study Key Concepts: Greenhouse Gas Carbon dioxide El Niño Global warming Greenhouse effect Greenhouse gas La Niña Land use Methane Nitrous oxide Radiative forcing
ATM S 211 Final Examination June 4, 2007
 ATM S 211 Final Examination June 4, 2007 Name This examination consists of a total of 100 points. In each of the first two sections, you have a choice of which questions to answer. Please note that you
ATM S 211 Final Examination June 4, 2007 Name This examination consists of a total of 100 points. In each of the first two sections, you have a choice of which questions to answer. Please note that you
Chapter 5. The Earth s Atmosphere
 Chapter 5 The Earth s Atmosphere Layers of the Earth Earth largest of the inner planets Gravity strong enough to hold gases. Lots of spheres Equator divided the Earth into two hemispheres Lithosphere-
Chapter 5 The Earth s Atmosphere Layers of the Earth Earth largest of the inner planets Gravity strong enough to hold gases. Lots of spheres Equator divided the Earth into two hemispheres Lithosphere-
Earth s Climate from Space. Richard Allan Department of Meteorology University of Reading
 Earth s Climate from Space Richard Allan Department of Meteorology University of Reading Earth s energy balance in space S πr 2 4πr 2 Outgoing Thermal Radiative Energy Absorbed Solar Radiative Energy
Earth s Climate from Space Richard Allan Department of Meteorology University of Reading Earth s energy balance in space S πr 2 4πr 2 Outgoing Thermal Radiative Energy Absorbed Solar Radiative Energy
Effects of Precursor Compounds on Natural and Anthropogenic Emissions of Ozone : A Review
 Effects of Precursor Compounds on Natural and Anthropogenic Emissions of Ozone : A Review + Dr. Bindu Khare, x Dr. Kanchan Khare + Dr. Bindu Khare, Lecturer sl. Gr, Dr. B.R.A. Polytechnic College, Gwalior.
Effects of Precursor Compounds on Natural and Anthropogenic Emissions of Ozone : A Review + Dr. Bindu Khare, x Dr. Kanchan Khare + Dr. Bindu Khare, Lecturer sl. Gr, Dr. B.R.A. Polytechnic College, Gwalior.
Lecture 28: Radiative Forcing of Climate Change
 Lecture 28: Radiative Forcing of Climate Change 1. Radiative Forcing In an unperturbed state, the net incoming solar radiation at the top of the atmosphere (Sn) must be balanced by the outgoing longwave
Lecture 28: Radiative Forcing of Climate Change 1. Radiative Forcing In an unperturbed state, the net incoming solar radiation at the top of the atmosphere (Sn) must be balanced by the outgoing longwave
Evidence and implications of anthropogenic climate change
 Evidence and implications of anthropogenic climate change Earth s Climate has always been changing 1) Is climate changing now? Global Warming? Sea level rising IPCC 2007 Fig. 5.13 (p. 410) Recontructed
Evidence and implications of anthropogenic climate change Earth s Climate has always been changing 1) Is climate changing now? Global Warming? Sea level rising IPCC 2007 Fig. 5.13 (p. 410) Recontructed
N-cycle: biogeochemistry. Biological flows of Nitrogen
 N-cycle: biogeochemistry SWES 410/510 April 4, 2014 I. N cycling A. simplest possible B. Global N budget C. Effects of N-cycling ( the Nitrogen Cascade ) II. Nitrous Oxide (N 2 O) budgets III. Data-Model
N-cycle: biogeochemistry SWES 410/510 April 4, 2014 I. N cycling A. simplest possible B. Global N budget C. Effects of N-cycling ( the Nitrogen Cascade ) II. Nitrous Oxide (N 2 O) budgets III. Data-Model
The September Equinox is today: Sep 23rd! It s considered the traditional end of Summer and the beginning of Fall
 More coming up in Topic #11 (class notes p 61) The September Equinox is today: Sep 23rd! It s considered the traditional end of Summer and the beginning of Fall The Sun s rays have greatest intensity right
More coming up in Topic #11 (class notes p 61) The September Equinox is today: Sep 23rd! It s considered the traditional end of Summer and the beginning of Fall The Sun s rays have greatest intensity right
RENEWABLE ENERGY NON-RENEWABLE ENERGY
 Hydro Solar Biomass RENEWABLE ENERGY The motion associated with rapidly falling water, waves and tidal currents can be harnessed to drive turbines and generate electricity. RENEWABLE ENERGY Energy from
Hydro Solar Biomass RENEWABLE ENERGY The motion associated with rapidly falling water, waves and tidal currents can be harnessed to drive turbines and generate electricity. RENEWABLE ENERGY Energy from
Three Connected Interactives
 Three Connected Interactives Carbon Dioxide and the Carbon Cycle Earth s Energy Flows and Climate Impacts of Climate Change in the Pacific Region Climate Change: Causes and Impacts Human Activities More
Three Connected Interactives Carbon Dioxide and the Carbon Cycle Earth s Energy Flows and Climate Impacts of Climate Change in the Pacific Region Climate Change: Causes and Impacts Human Activities More
Global temperature changes: relative importance of greenhouse constituents of the atmosphere as calculated with a 1-d radiative-convective model
 Proe. Indian Acad. Sci. (Earth Planet. Sci.), Vol. 94, No. 2, July 1985, pp. 139-145. Printed in India. Global temperature changes: relative importance of greenhouse constituents of the atmosphere as calculated
Proe. Indian Acad. Sci. (Earth Planet. Sci.), Vol. 94, No. 2, July 1985, pp. 139-145. Printed in India. Global temperature changes: relative importance of greenhouse constituents of the atmosphere as calculated
Lecture 27: Radiative Forcing of Climate Change
 Lecture 27: Radiative Forcing of Climate Change 1. Radiative Forcing In an unperturbed state, the net incoming solar radiation at the top of the atmosphere (Sn) must be balanced by the outgoing longwave
Lecture 27: Radiative Forcing of Climate Change 1. Radiative Forcing In an unperturbed state, the net incoming solar radiation at the top of the atmosphere (Sn) must be balanced by the outgoing longwave
Topic # 7 ATMOSPHERIC STRUCTURE & CHEMICAL COMPOSITION
 Topic # 7 ATMOSPHERIC STRUCTURE & CHEMICAL COMPOSITION All about the GASES IN THE ATMOSPHERE, esp. GREENHOUSE GASES! Class Notes pp 37-41 OBJECTIVES: To understand: -- the VERTICALSTRUCTURE of the atmosphere
Topic # 7 ATMOSPHERIC STRUCTURE & CHEMICAL COMPOSITION All about the GASES IN THE ATMOSPHERE, esp. GREENHOUSE GASES! Class Notes pp 37-41 OBJECTIVES: To understand: -- the VERTICALSTRUCTURE of the atmosphere
Temperature and moisture dependence of soil H 2 uptake measured in the laboratory
 GEOPHYSICAL RESEARCH LETTERS, VOL. 33, L14813, doi:10.1029/2006gl026749, 2006 Temperature and moisture dependence of soil H 2 uptake measured in the laboratory Nicole V. Smith-Downey, 1,2 James T. Randerson,
GEOPHYSICAL RESEARCH LETTERS, VOL. 33, L14813, doi:10.1029/2006gl026749, 2006 Temperature and moisture dependence of soil H 2 uptake measured in the laboratory Nicole V. Smith-Downey, 1,2 James T. Randerson,
Lecture 11: Global Warming. Human Acticities. Natural Climate Changes. Global Warming: Natural or Man-Made CO 2 CH 4
 Lecture 11: Global Warming Human Acticities CO 2 CH 4 The initial appearance of human species: last 100,000 to 200,000 years Development of the first civilization: the last 10,000 years What is the sensitivity
Lecture 11: Global Warming Human Acticities CO 2 CH 4 The initial appearance of human species: last 100,000 to 200,000 years Development of the first civilization: the last 10,000 years What is the sensitivity
LESSON 9: CONCEPTUALIZING MODULE II Factors Influencing Temperature
 LESSON 9: CONCEPTUALIZING MODULE II Factors Influencing Temperature PURPOSE/QUESTION To understand the need to compare data sets over different parameters thus increasing the strength for students climate
LESSON 9: CONCEPTUALIZING MODULE II Factors Influencing Temperature PURPOSE/QUESTION To understand the need to compare data sets over different parameters thus increasing the strength for students climate
Topic # 7 Part II ATMOSPHERIC STRUCTURE & CHEMICAL COMPOSITION
 Topic # 7 Part II ATMOSPHERIC STRUCTURE & CHEMICAL COMPOSITION All about the GASES IN THE ATMOSPHERE, esp. GREENHOUSE GASES! Class Notes pp 37-41 REVIEW: ATMOSPHERIC STRUCTURE The changes in temperature
Topic # 7 Part II ATMOSPHERIC STRUCTURE & CHEMICAL COMPOSITION All about the GASES IN THE ATMOSPHERE, esp. GREENHOUSE GASES! Class Notes pp 37-41 REVIEW: ATMOSPHERIC STRUCTURE The changes in temperature
The Carbon Cycle. the atmosphere the landmass of Earth (including the interior) all of Earth s water all living organisms
 The Carbon Cycle Carbon is an essential part of life on Earth. About half the dry weight of most living organisms is carbon. It plays an important role in the structure, biochemistry, and nutrition of
The Carbon Cycle Carbon is an essential part of life on Earth. About half the dry weight of most living organisms is carbon. It plays an important role in the structure, biochemistry, and nutrition of
Main Natural Sources of Greenhouse Gases
 Main Natural Sources of Greenhouse Gases Content Atmospheric Composition Composition of the Earth s Atmosphere Greenhouse Gases The Radiative Forcing bar chart: AR5 version Natural Greenhouse Gases Water
Main Natural Sources of Greenhouse Gases Content Atmospheric Composition Composition of the Earth s Atmosphere Greenhouse Gases The Radiative Forcing bar chart: AR5 version Natural Greenhouse Gases Water
Layers of the Atmosphere. Troposphere Stratosphere Mesosphere Thermosphere
 Air Pollution Layers of the Atmosphere Troposphere Stratosphere Mesosphere Thermosphere Troposphere Composition Sea level 17km Composition 78% Nitrogen 20% Oxygen Other 2%... Water vapor Argon gas Carbon
Air Pollution Layers of the Atmosphere Troposphere Stratosphere Mesosphere Thermosphere Troposphere Composition Sea level 17km Composition 78% Nitrogen 20% Oxygen Other 2%... Water vapor Argon gas Carbon
Topic 6 National Chemistry Summary Notes. Fuels. Fuels and Combustion
 Topic 6 National Chemistry Summary Notes Fuels LI 1 Fuels and Combustion Coal, oil, gas and wood can all be used as fuels. These fuels have energy-rich chemical bonds which were created using the energy
Topic 6 National Chemistry Summary Notes Fuels LI 1 Fuels and Combustion Coal, oil, gas and wood can all be used as fuels. These fuels have energy-rich chemical bonds which were created using the energy
3. If the power plant were to suddenly cease operations, how long would it take for m to decrease from its steady state value m to 5% of that value?
 35 PROBLEMS 3. 1 Atmospheric steady state A power plant emits a pollutant X to the atmosphere at a constant rate E (kg s -1 ) starting at time t = 0. X is removed from the atmosphere by chemical reaction
35 PROBLEMS 3. 1 Atmospheric steady state A power plant emits a pollutant X to the atmosphere at a constant rate E (kg s -1 ) starting at time t = 0. X is removed from the atmosphere by chemical reaction
2 Atmospheric Heating
 CHAPTER 15 2 Atmospheric Heating SECTION The Atmosphere BEFORE YOU READ After you read this section, you should be able to answer these questions: How does energy travel from the sun to Earth? What are
CHAPTER 15 2 Atmospheric Heating SECTION The Atmosphere BEFORE YOU READ After you read this section, you should be able to answer these questions: How does energy travel from the sun to Earth? What are
9th Period Environmental Science Chapter 15: The Atmosphere
 Section 15.1: Earth s Atmosphere 9th Period Environmental Science Chapter 15: The Atmosphere Properties of the Atmosphere: nitrogen, oxygen, water vapor and 1% other gases. air pressure is higher at the
Section 15.1: Earth s Atmosphere 9th Period Environmental Science Chapter 15: The Atmosphere Properties of the Atmosphere: nitrogen, oxygen, water vapor and 1% other gases. air pressure is higher at the
Introduction to the Role of Tropospheric Ozone and Arctic Climate. Ellen Baum May 8, 2008
 Introduction to the Role of Tropospheric Ozone and Arctic Climate Ellen Baum May 8, 2008 There is a significant global role for tropospheric ozone and climate 1.4 Temperature impact from CO2 compared to
Introduction to the Role of Tropospheric Ozone and Arctic Climate Ellen Baum May 8, 2008 There is a significant global role for tropospheric ozone and climate 1.4 Temperature impact from CO2 compared to
Risk and Uncertainties in Anthropogenic. Control over Greenhouse Forcing in the TAR. with a focus on Atmospheric Chemistry
 FCCC, Bonn, 4-6 Apr 2002 SBSTA IPCC/TAR Michael Prather University of California at Irvine Risk and Uncertainties in Anthropogenic Control over Greenhouse Forcing in the TAR with a focus on Atmospheric
FCCC, Bonn, 4-6 Apr 2002 SBSTA IPCC/TAR Michael Prather University of California at Irvine Risk and Uncertainties in Anthropogenic Control over Greenhouse Forcing in the TAR with a focus on Atmospheric
Environmental Engineering Atmosphere & pollution 2
 Environmental Engineering Atmosphere & pollution 2 Global radiation Greenhouse effect Kyoto protocol David Zumr Dpt. of Drainage, Irrigation and Landscape Eng. 1/ insolation from the Sun Electromagnetic
Environmental Engineering Atmosphere & pollution 2 Global radiation Greenhouse effect Kyoto protocol David Zumr Dpt. of Drainage, Irrigation and Landscape Eng. 1/ insolation from the Sun Electromagnetic
CALIFORNIA EDUCATION AND THE ENVIRONMENT INITIATIVE
 Water Vapor: A GHG Lesson 3 page 1 of 2 Water Vapor: A GHG Water vapor in our atmosphere is an important greenhouse gas (GHG). On a cloudy day we can see evidence of the amount of water vapor in our atmosphere.
Water Vapor: A GHG Lesson 3 page 1 of 2 Water Vapor: A GHG Water vapor in our atmosphere is an important greenhouse gas (GHG). On a cloudy day we can see evidence of the amount of water vapor in our atmosphere.
