Density Determi nations
|
|
|
- Madeleine Strickland
- 5 years ago
- Views:
Transcription
1 f EXPERIMENT 3 Density Determi nations OBJECTIVE Density is an important characteristic property of matter, and may be used as a method of identification. In this experiment, you will determine the densities of regularly and irregularly shaped solids as well as of pure liquids and solutions. INTRODUCTION The density of a sample of maffer represents the mass contained within a unit volume of space within the sample. For most samples, a "unit volume" means 1.0 ml (1.0 cm3). The units of density, therefore, are quoted in terms of grams per milliliter (g/ml) or grams per cubic centimeter (g/cm3) for most solid and liquid samples of matter. Since we seldom deal with exactly 1.0 ml of substance in the general chemistry laboratory, we usually say that the density of a sample represents the mass of the specific sample divided by its particular volume. densttv= f- " InASS volume Because the density does in fact represent a ratio, the mass of any size sample, divided by the volume of that sample, gives the mass of 1.0 ml of the same sample. Densities are usually determined and reported at20"c (around room temperature) because the volume of a sample, and hence the density, will often vary with temperature. This is especially true for gases, with smaller (but still often significant) changes for liquids and solids. References (such as the various chemical handbooks) always speciff the temperature at which a density was measured. Density is often used as a point of identification in the determination of an unknown substance. In later experiments, you will study several other physical properties of substances that are used in the identification of unknown substances. For example, the boiling and melting points of a given substance are characteristic of that substance and are used routinely in identification of unknown substances. S/ppose an unknown's boiling and melting points have been determined, but on consulting the literature, it is found that more than one substance has these boiling and melting points. The density of the unknown might then be used to distinguish the unknown. It is very unlikely that two substances would have the same boiling point, melting point, and density. Density can also be used to determine the concentration of solutions in certain instances. When a solute is dissolved in a solvent, the density of the solutionwill be different from that of the pure solvent itself. Handbooks list detailed information about the densities of solutions as a function of their composition (typically, in terms of the percent solute in the solution). If a sample is known to contain only a single solute, the density of the solution could be measured experimentally, and then the handbook could be consulted to determine what concenhation of the solute gives rise to the measured density of the solution. tf. The determination of the density of certain physiological liquids is often an important screening tool in medical diagnosis. For example, if the density of urine differs from normal values, this Brooks/Cole, Cengage Learning 21
2 --E 22 Experiment 3: Density Determinations indicate a problem with the kidneys secreting substances that should not be lost from the body. A determination of specific gravity is almost always performed during a urinalysis (specific gravity represents the ratio of a sample's density to the density of water under the same conditions). r Several techniques are used for the determination of density. The method used will depend on the type of sample and on the level of precr'sion desired for the measirrement. For example, devices have been constructed for determinations of the density of urine that permit a quick, reliable, routine determination. In general, a density determination will involve the determination of the mass of the sample with a balance, but the method used to determine the volume of the sample will differ from situation to situation. Several methods of volume determination are explored in this experiment. For solid samples, different methods may be needed for the determination of the volume, depending on whether or not the solid is regularly shaped. If a solid has a regular shape (e.g., cubic, rectangular, cylindrical), the volume of the solid may be determined by geometry: For a cubic solid, volume = (edge)' For a rectangular solid, volume = length x width x height For a cylindrical solid, volume = fi x (radius)'?x height If a solid does not have a regular shape, it may be possible to determine the volume of the solid from Archimedes' principle, which states that an insoluble, non-reactive solid wlll displace a volume of liquid equal to its own volume. Typically, an irregularly shaped solid is added to a liquid in a volumetric container (such as a graduated cylinder) and the change in liquid level determined. For liquids, very precise values of density may be determined by pipeting an exact volume of liquid into a sealable weighing bottle (this is especially useful for highly volatile liquids), and then determining the mass of liquid that was transferred with the pipet. A more convenient method for routine density determinations for liquids is to weigh a particular volume of liquid as contained in a graduated cylinder. \-a SAFETY PRECAUTIONS o o o Pr:otective eyewear approved by your institution must be worn at all times while you are in the laboratory. The unknown liquids may be flammable, and their vapors may be toxic. Keep the unknown liquids away from open flames, and do not inhale their vapors. Dispose of the unknown liquids as directed by the instructor. Dispose of the metal samples in the container designated for their collection. Do not put into the trash container. Do not spill the metal samples on the floor or benchtop. AP PARATUS/REAG E NTS REQU I RE D Unknown liquid sample, metal samplei (regularly-shaped and irregularly-shaped), sodium chloride Brooks/Cole, Cengage Learning
3 Experiment 3: Density Determinations 23 PROCEDURE lecord all data and observations directly in your notebook in ink. l- Determination of the Density of Solids Obtain a regularly-shaped solid and record its identification number. With a ruler, determine the physical dimensions (e.g., length, width, height, radius) of the solid to the nearest 0.2 mm. From the physical dimensions, calculate the volume of the solid. Determine the mass of the regularly shaped solid to at least the nearest mg (0.001 g). From the mass and volume, calculate the density of the solid. Obtain a sample of irregularly-shaped metal pellets (shot) and record the identification code number. Weigh a sample ofthe metal of approximately 50 g, but record the actual mass of metal taken to the nearest mg (0.001 g). Add water to your 100-mL graduated cylinder to approximately the 50-mL mark. Record the exact volume of water in the cylinder to the precision permitted by the calibration marks of the cylinder, remembering to read the water level tangent to the meniscus. Pour the metal s4mple into the graduated cylinder, making sure that none of the pellets sticl<s to the walls of the cylinder above the water level. Stirlshake the cylinder to make certain that no air bubbles have"been trapped among the metal pellets. (See Figure 3-1.) Solvent level after addition of solid Solvent level before addition of solid --l--- Volume of solid I Liquid Solid material settled on bottom Figure 3-1. Measurement of volume by displacement. A non-soluble object displaces a volume of liquid equalto its own volume. Read the level of the water in the graduated cylinder, again making your determination to the precision permitted by the calibration marks of the cylinder. Assuming that the metal sample does not dissolve in or react with water, the change in water levels represents the volume of the metal pellets. f Calculate the density of the unknown metal pellets. After blotting them dry with apaper towel, turn in the metal pellets to your instructor (do not discard). 9 Brooks/Cole, Cengage Learning
4 24 Experiment 3: Density Determinations 2. Density of Pure Liquids Clean and dry a25-rnl graduated cylinder (a rotled-up paper towel or a stream of compressed air may be used). Weigh the dry graduated cylinder to the nearest mg (0.001 g). Add distilled water to the cylinder so that the water level is above the 2O-ntLmark but below the 25-mL mark. Use a thermometer to determine the temperature of the water in the cylinder to the nearest degree. Reweigh the cylinder to the nearest milligram. Record the exact volume of water in the cylinder, to the level of precision permitted by the calibration marks on the barrel of the cylinder, remembering to read the water level tangent to the meniscus. Calculate the density of the water. Compare the measured density of the water with the value listed in the handbook for the temperature of your experiment. Clean and dry the graduated cylinder. Obtain an unknown liquid and record its identification number. Determine the density of the unknown liquid, using the method just described for water. 3. Densit-v of Solutions The concentration of solutions is often conveniently described interms of ths solutions' percentage composition on a weight basis. For example, a 5olo sodium chloride solution contains 5 g of sodium chloride in every 100 g of solution (which corresponds to 5 g of sodium chloride for every 95 g of water present). Prepare solutions of sodium chloride in distilled water consisting of the following percentages by weight: 5yo, llyo, l5yo,20o,25yo.prepare25 g ofeach solution (you do nothave to prepare 100 g of each solution to be able to use the percentage composition). Make the weight determinations of solute and solvent to the nearest milligram. Using the method described earlier for samples of pure liquids, determine the density of each of your sodium chloride solutions. Record the temperature of each'solution while determining its density. t Construct a graph of the density of your solutions versus the percentage of NaCl the solution contains. What sort of relationship exists between density and composition? Use a handbook of chemical data to determine the true density of each of the solutions you prepared. Calculate the error in each of the densities you determined. Example: Suppose it is desired to prepare 50. g of 7.5% calcium chloride solution. When we say we have a "7.5 o c'alcium chloride solution," we are implying this conversion factor: 7.5 g calcium chloride 100. g solution 7.5 g calcium chloride 7.5 g calcium chloride g water So, to prepare 50. g of 7.5%:o calcium chloride solution, 50. g solution x 7.5 g calcium chloride : 3.8 g calcium chloride needed 100. g solution So, 3.8 g of calcium chloride and 46.2 g of water would be Brooks/Cole, Cengage Learning
5 \ame: Lab Instructor: Section: Date: EXPERIMENT 3 Density Determ i nations RESULTS/OBSERVATIONS 1. Density of Solids ID number of regular solid Shape ofregular solid Dimensions of regular solid Calculated volume of solid Mass of regular solid Density of regular solid ID number of metal pellets Mass of metal pellets taken Initial water level Final water level Volume of metal pellets Calculated density of metal pellets 2. Density of Pure Liquids Mass of empty graduated cylinder Mass of cylinder plus water Volume of water Density of water Temperature of water Handbook density of water.id number of unknown liquid Mass of cylinder plus liquid Mass of liquid Volume of unknown liquid Density of unknown liquid :) O Brooks/Cole, Cengage Learning 27
6 Experiment 3: Report J. Density of Solutions 7o NaCl Density measured Temperature Handbook value Vo err0,t QUESTIONS l. What error would be introduced into the determination of the density of the regularly shaped solid if the solid were hollow? Would the apparent volume of the solid be larger or smaller than the actual volume? Would the density calculated be too high or too low? 2. What error would be introduced into the determination of the density of the inegularly shaped metal pellets if you had not stirred/shaken the pellets to remove adhering air bubbles? Would the density be too high or too low? 3. Your data for the density of sodium chloride solutions should have produced a straight line when plotted. How could this plot be used to determine the density of any concentration of sodium chloride solution? 4. lvhy was it necessary to determine the temperature dttringthe determination of the density of the sodium chloride solutions? Which factor-mass or volume-used in calculating the density would be affected by'temperature? Explain. O Brooks/Cole, Cengage Learning
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS
 DETERMINING THE DENSITY OF LIQUIDS & SOLIDS Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter. Density
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter. Density
EXPERIMENT 3: Identification of a Substance by Physical Properties
 EXPERIMENT 3: Identification of a Substance by Physical Properties Materials: Hot plate Digital balance Capillary tubes (3) Thermometer Beakers (250 ml) Watch glass Graduated Cylinder (10 ml) Mel-Temp
EXPERIMENT 3: Identification of a Substance by Physical Properties Materials: Hot plate Digital balance Capillary tubes (3) Thermometer Beakers (250 ml) Watch glass Graduated Cylinder (10 ml) Mel-Temp
Density Measurements Background
 Density Measurements Background The density (ρ) of a substance or an object is defined by its mass (m) divided by its volume (V):!"#$%&' =!"##!"#$%&!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!! =!! The units used
Density Measurements Background The density (ρ) of a substance or an object is defined by its mass (m) divided by its volume (V):!"#$%&' =!"##!"#$%&!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!! =!! The units used
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS - worksheet
 DETERMINING THE DENSITY OF LIQUIDS & SOLIDS - worksheet 17 Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter.
DETERMINING THE DENSITY OF LIQUIDS & SOLIDS - worksheet 17 Density, like color, odor, melting point, and boiling point, is a physical property of matter. Therefore, density may be used in identifying matter.
Experiment: Measurements
 Experiment: Measurements I. INTRODUCTION Measurements are essential to experimental sciences such as chemistry, physics, biology, and geology. The measurements are usually made using the metric system
Experiment: Measurements I. INTRODUCTION Measurements are essential to experimental sciences such as chemistry, physics, biology, and geology. The measurements are usually made using the metric system
M. It is expressed in units such a g/cc,
 Experiment 2 Density Part I: Density of a solid Density is defined as mass per unit volume, D = V M. It is expressed in units such a g/cc, g/ml, lb/ft 3, etc. Determination of mass is done by weighing
Experiment 2 Density Part I: Density of a solid Density is defined as mass per unit volume, D = V M. It is expressed in units such a g/cc, g/ml, lb/ft 3, etc. Determination of mass is done by weighing
Measurement and Density - Experiment 1
 Measurement and Density - Experiment 1 Purpose: The purpose of this experiment is to acquaint you with some common metric units of length, volume, and mass. You will also measure the density of an unknown
Measurement and Density - Experiment 1 Purpose: The purpose of this experiment is to acquaint you with some common metric units of length, volume, and mass. You will also measure the density of an unknown
Density. The purpose of this experiment is to investigate the topic of density by determining the densities of some materials.
 Density Goals The purpose of this experiment is to investigate the topic of density by determining the densities of some materials. Introduction There is a lesson in the old bromide: Which is heavier,
Density Goals The purpose of this experiment is to investigate the topic of density by determining the densities of some materials. Introduction There is a lesson in the old bromide: Which is heavier,
Experiment 1: The Densities of Liquids and Solids (from Masterson & Hurley)
 Experiment 1: The Densities of Liquids and Solids (from Masterson & Hurley) One of the fundamental properties of any sample of matter is its density, which is its mass per unit of volume. The density of
Experiment 1: The Densities of Liquids and Solids (from Masterson & Hurley) One of the fundamental properties of any sample of matter is its density, which is its mass per unit of volume. The density of
Density (d) is a property of a substance equal to the ratio of its mass (m) to its volume (V): d = m V
 Experiment 2 Name: DEN 14 Si TY In this experiment, you will learn about density and practice the proper way to report measurements and calculated quantities. Properties Properties of a substance can be
Experiment 2 Name: DEN 14 Si TY In this experiment, you will learn about density and practice the proper way to report measurements and calculated quantities. Properties Properties of a substance can be
Introduction to Density
 Introduction to Density INTRODUCTION Which is heavier, a pound of aluminum or a pound of lead? The answer, of course, is neither, but many people confuse the words "heavy" and "dense". "Heavy" refers to
Introduction to Density INTRODUCTION Which is heavier, a pound of aluminum or a pound of lead? The answer, of course, is neither, but many people confuse the words "heavy" and "dense". "Heavy" refers to
Name Honors Chemistry / /
 Name Honors Chemistry / / SOL Questions Chapter 1 Each of the following questions below appeared on an SOL Chemistry Exam. For each of the following bubble in the correct answer on your scantron. 1. The
Name Honors Chemistry / / SOL Questions Chapter 1 Each of the following questions below appeared on an SOL Chemistry Exam. For each of the following bubble in the correct answer on your scantron. 1. The
EXPERIMENT 5 Chemistry 110 COMPOSITION OF A MIXTURE
 EXPERIMENT 5 Chemistry 110 PURPOSE: The purpose of this experiment is to determine the percent composition of a mixture. COMPOSITION OF A MIXTURE Most matter is a mixture of many substances. For example,
EXPERIMENT 5 Chemistry 110 PURPOSE: The purpose of this experiment is to determine the percent composition of a mixture. COMPOSITION OF A MIXTURE Most matter is a mixture of many substances. For example,
Revised Molar Mass Measurement Lab Purpose: Background:
 Revised Molar Mass Measurement Lab (Based on prior Ch b experiment, incorporating revision suggestions from Tsze Tsang) Purpose: To use the colligative properties of cyclohexane solutions to determine
Revised Molar Mass Measurement Lab (Based on prior Ch b experiment, incorporating revision suggestions from Tsze Tsang) Purpose: To use the colligative properties of cyclohexane solutions to determine
Experiment #3. Density
 Experiment #3. Density Goals 1. To measure and calculate the density of various substances. 2. To use significant figures correctly in calculations. Background Density is the mass per unit volume of a
Experiment #3. Density Goals 1. To measure and calculate the density of various substances. 2. To use significant figures correctly in calculations. Background Density is the mass per unit volume of a
IDENTIFYING UNKNOWN SUBSTANCES
 IDENTIFYING UNKNOWN SUBSTANCES LAB 15 EXPERIMENT STUDENT BOOK Chapter 1, page 25 TOOLBOX Page 4 and 36 Goal Identify unknown substances with the help of different tests. 1. What is the independent variable
IDENTIFYING UNKNOWN SUBSTANCES LAB 15 EXPERIMENT STUDENT BOOK Chapter 1, page 25 TOOLBOX Page 4 and 36 Goal Identify unknown substances with the help of different tests. 1. What is the independent variable
Experiment #3. Density and Specific Gravity.
 Experiment #3. Density and Specific Gravity. Goals 1. To measure and calculate the density and specific gravity of various substances. 2. To use significant figures correctly in calculations. Background
Experiment #3. Density and Specific Gravity. Goals 1. To measure and calculate the density and specific gravity of various substances. 2. To use significant figures correctly in calculations. Background
CHAPTER 2 HW PRACTICE SOLUTIONS
 CHAPTER 2 HW PRACTICE SOLUTIONS SIGNIFICANT FIGURES 1.) How many significant figures are shown in each of the following numbers? a. 0.0320 3 SF c. 503.10 5 SF e. 0.00702 3 SF b. 8000 1 SF d. 91,000,000
CHAPTER 2 HW PRACTICE SOLUTIONS SIGNIFICANT FIGURES 1.) How many significant figures are shown in each of the following numbers? a. 0.0320 3 SF c. 503.10 5 SF e. 0.00702 3 SF b. 8000 1 SF d. 91,000,000
Salinity in Seawater
 Salinity in Seawater Objective To familiarize students with the different methods used for measuring salinity of water. Introduction: Salinity exerts profound impacts on the marine environment. It controls
Salinity in Seawater Objective To familiarize students with the different methods used for measuring salinity of water. Introduction: Salinity exerts profound impacts on the marine environment. It controls
DETERMINATION OF DENSITY
 DETERMINATION OF DENSITY Dr. Barbara B. Bunn. Adapted from Experiments in General Chemistry ; Wiley 1996J.G. Wardeska, T.T-S. Huang, R.W. Kopp; used by permission. Tested by Ms. Janice Orr s Chemistry
DETERMINATION OF DENSITY Dr. Barbara B. Bunn. Adapted from Experiments in General Chemistry ; Wiley 1996J.G. Wardeska, T.T-S. Huang, R.W. Kopp; used by permission. Tested by Ms. Janice Orr s Chemistry
DENSITY of Solids and Liquids
 Regents Earth Science Name: Date: Lab # DENSITY of Solids and Liquids Introduction: Density is the term used to describe the relationship between the mass of an object and its volume. Under given conditions
Regents Earth Science Name: Date: Lab # DENSITY of Solids and Liquids Introduction: Density is the term used to describe the relationship between the mass of an object and its volume. Under given conditions
Determining the Density of Unknown Substances
 Determining the Density of Unknown Substances Introduction: Most of the elements on the periodic table are metals and solids. These elements have observable properties that make it possible to identify
Determining the Density of Unknown Substances Introduction: Most of the elements on the periodic table are metals and solids. These elements have observable properties that make it possible to identify
SIGNIFICANT FIGURES WORKSHEET
 SIGNIFICANT FIGURES WORKSHEET PART 1 - Determine the number of significant figures in the following numbers. 1.) 0.02 2.) 0.020 3.) 501 4.) 501.0 5.) 5,000 6.) 5,000. 7.) 6,051.00 8.) 0.0005 9.) 0.1020
SIGNIFICANT FIGURES WORKSHEET PART 1 - Determine the number of significant figures in the following numbers. 1.) 0.02 2.) 0.020 3.) 501 4.) 501.0 5.) 5,000 6.) 5,000. 7.) 6,051.00 8.) 0.0005 9.) 0.1020
Specific Heat. q = csm T (1)
 INTRODUCTION Just as people can be identified by their appearance and their behavior, substances are described and identified by their physical (appearance) and chemical (behavior) properties. Physical
INTRODUCTION Just as people can be identified by their appearance and their behavior, substances are described and identified by their physical (appearance) and chemical (behavior) properties. Physical
Experiment 1 MOLAR MASS DETERMINATION BY FREEZING POINT DEPRESSION
 1 Experiment 1 MOLAR MASS DETERMINATION BY FREEZING POINT DEPRESSION Whenever a substance is dissolved in a solvent, the vapor pressure of the solvent is lowered. As a result of the decrease in the vapor
1 Experiment 1 MOLAR MASS DETERMINATION BY FREEZING POINT DEPRESSION Whenever a substance is dissolved in a solvent, the vapor pressure of the solvent is lowered. As a result of the decrease in the vapor
Lab Section. Experiment 2 Density. Introduction
 Experiment 2 Density Introduction Density (given the symbol d or ρ depending upon which book you read) is an intrinsic property of materials. The term intrinsic means that it is independent upon the amount
Experiment 2 Density Introduction Density (given the symbol d or ρ depending upon which book you read) is an intrinsic property of materials. The term intrinsic means that it is independent upon the amount
SOLIDS: Mass, Volume and Density Measurements
 CHEM 1411 Lab Solids: Mass, Volume, and Density 23 SOLIDS: Mass, Volume and Density Measurements A. Overview Review Sections 1.4-1.6 in your textbook (Chemistry: The Central Science, 9th Ed., Brown, LeMay,
CHEM 1411 Lab Solids: Mass, Volume, and Density 23 SOLIDS: Mass, Volume and Density Measurements A. Overview Review Sections 1.4-1.6 in your textbook (Chemistry: The Central Science, 9th Ed., Brown, LeMay,
Copyright 2014 Science Doodles
 Copyright 2014 Science Doodles If a scientist comes across an unknown element, could she use density to identify the element? A. Yes, because each element has a specific density. B. No, because density
Copyright 2014 Science Doodles If a scientist comes across an unknown element, could she use density to identify the element? A. Yes, because each element has a specific density. B. No, because density
CHM Gravimetric Chloride Experiment (r7) 1/5
 CHM 111 - Gravimetric Chloride Experiment (r7) 1/5 Purpose You will perform one of the basic types of quantitative analysis - the gravimetric analysis. You will be asked to determine the percentage of
CHM 111 - Gravimetric Chloride Experiment (r7) 1/5 Purpose You will perform one of the basic types of quantitative analysis - the gravimetric analysis. You will be asked to determine the percentage of
Determination of the Mass Percentage of Copper in a Penny
 Determination of the Mass Percentage of Copper in a Penny Introduction This experiment will cost you one penny ($0.01). The penny must be minted after 1983. Any penny will do; for best results the penny
Determination of the Mass Percentage of Copper in a Penny Introduction This experiment will cost you one penny ($0.01). The penny must be minted after 1983. Any penny will do; for best results the penny
A Hydrogen Powered Bottle Rocket
 A Hydrogen Powered Bottle Rocket Rockets are made by filling plastic water bottles with a mixture of hydrogen gas and oxygen gas. The rocket is launched by igniting the mixture with a flame. The bottle
A Hydrogen Powered Bottle Rocket Rockets are made by filling plastic water bottles with a mixture of hydrogen gas and oxygen gas. The rocket is launched by igniting the mixture with a flame. The bottle
T. Trimpe Lesson 1: Length
 T. Trimpe 2008 http://sciencespot.net/ Lesson 1: Length Metric Units The basic unit of length in the metric system is the meter and is represented by a lowercase m. Metric Units 1 Kilometer (km) = 1000
T. Trimpe 2008 http://sciencespot.net/ Lesson 1: Length Metric Units The basic unit of length in the metric system is the meter and is represented by a lowercase m. Metric Units 1 Kilometer (km) = 1000
The Crystal Forest Favorite Holiday Demonstrations
 The Crystal Forest Favorite Holiday Demonstrations SCIENTIFIC Introduction Put a new twist on crystal growing. In this class participation demonstration, students cut out and assemble miniature trees and
The Crystal Forest Favorite Holiday Demonstrations SCIENTIFIC Introduction Put a new twist on crystal growing. In this class participation demonstration, students cut out and assemble miniature trees and
Changes for Organic Chemistry 2521 Labs
 Changes for Organic Chemistry 2521 Labs Chapter 3 Crystallization Part 1 (Starts on page 56) Test the solubility of three compounds with three solvents. There are four compounds to choose from: 1. Resorcinol
Changes for Organic Chemistry 2521 Labs Chapter 3 Crystallization Part 1 (Starts on page 56) Test the solubility of three compounds with three solvents. There are four compounds to choose from: 1. Resorcinol
CHM-202 General Chemistry and Laboratory II Laboratory 2 Molar Mass by Freezing Point Depression
 Purpose CHM-202 General Chemistry and Laboratory II Laboratory 2 Molar Mass by Freezing Point Depression In this experiment, you will determine the molar mass of a nonelectrolytic solvent by measuring
Purpose CHM-202 General Chemistry and Laboratory II Laboratory 2 Molar Mass by Freezing Point Depression In this experiment, you will determine the molar mass of a nonelectrolytic solvent by measuring
1 Elements. TAKE A LOOK 2. Identify Look at the illustration and identify one source of iron that comes to Earth from somewhere else.
 CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
CHAPTER 4 1 Elements SECTION Elements, Compounds, and Mixtures BEFORE YOU READ After you read this section, you should be able to answer these questions: What is an element? How do elements differ from
Creation Settings. can be separated into two or more pure substances by distillation. can be separated into two or more substances by chromatography.
 Page 1 of 10 TEST BANK (ACCT3321_201_1220) > CONTROL PANEL > POOL MANAGER > POOL CANVAS Pool Canvas Add, modify, and remove questions. Select a question type from the Add drop-down list and click Go to
Page 1 of 10 TEST BANK (ACCT3321_201_1220) > CONTROL PANEL > POOL MANAGER > POOL CANVAS Pool Canvas Add, modify, and remove questions. Select a question type from the Add drop-down list and click Go to
Alchemy: A Cross-Curricular Activity Copper, Silver, and Gold Redox Reactions
 Alchemy: A Cross-Curricular Activity Copper, Silver, and Gold Redox Reactions SCIENTIFIC Introduction Turn an ordinary copper penny into silver and then into gold! Get rich quick by demonstrating this
Alchemy: A Cross-Curricular Activity Copper, Silver, and Gold Redox Reactions SCIENTIFIC Introduction Turn an ordinary copper penny into silver and then into gold! Get rich quick by demonstrating this
Copper Odyssey. Chemical Reactions of Copper
 Name Lab Partner(s) Copper Odyssey Chemical Reactions of Copper Date Period Elemental copper metal will be converted into copper (II) ion and then brought through a series of compound conversions until
Name Lab Partner(s) Copper Odyssey Chemical Reactions of Copper Date Period Elemental copper metal will be converted into copper (II) ion and then brought through a series of compound conversions until
COPPER CYCLE EXPERIMENT 3
 COPPER CYCLE EXPERIMENT 3 INTRODUCTION One simple way to state the aim of chemistry is: The study of matter and its transformations. In this experiment, a copper sample will appear in five different forms
COPPER CYCLE EXPERIMENT 3 INTRODUCTION One simple way to state the aim of chemistry is: The study of matter and its transformations. In this experiment, a copper sample will appear in five different forms
Periodic Trends and the Properties of Elements The Alkaline Earth Metals
 Introduction Periodic Trends and the Properties of Elements The Alkaline Earth Metals The periodic table is the most recognized symbol of chemistry across the world. It is a valuable tool that allows scientists
Introduction Periodic Trends and the Properties of Elements The Alkaline Earth Metals The periodic table is the most recognized symbol of chemistry across the world. It is a valuable tool that allows scientists
THE MOLECULAR WEIGHT OF A SOLUTE*
 Name Per THE MOLECULAR WEIGHT OF A SOLUTE* One of the most helpful things that can be known about an element or compound is its molecular weight (molecular mass, gram molecular mass, atomic mass, or gram
Name Per THE MOLECULAR WEIGHT OF A SOLUTE* One of the most helpful things that can be known about an element or compound is its molecular weight (molecular mass, gram molecular mass, atomic mass, or gram
GRAVIMETRIC DETERMINATION OF SULFATE IN AN UNKNOWN SOLUTION
 GRAVIMETRIC DETERMINATION OF SULFATE IN AN UNKNOWN SOLUTION AIM The main objective of this experiment is to determine the concentration of sulfate ion in an unknown solution by using gravimetry. INTRODUCTION
GRAVIMETRIC DETERMINATION OF SULFATE IN AN UNKNOWN SOLUTION AIM The main objective of this experiment is to determine the concentration of sulfate ion in an unknown solution by using gravimetry. INTRODUCTION
The table below shows some of the properties of the elements cobalt and nickel.
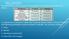 BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
BELL RINGER The table below shows some of the properties of the elements cobalt and nickel. A scientist has a sample of metal that could be either cobalt or nickel. Which of the following properties could
An Oxidation-Reduction Titration: The Reaction of Fe 2+ and Ce 4+
 An Oxidation-Reduction Titration: The Reaction of Fe 2+ and Ce 4+ LAB ADV COMP 8 From Advanced Chemistry with Vernier, Vernier Software & Technology, 2004 INTRODUCTION A titration, as you recall, is a
An Oxidation-Reduction Titration: The Reaction of Fe 2+ and Ce 4+ LAB ADV COMP 8 From Advanced Chemistry with Vernier, Vernier Software & Technology, 2004 INTRODUCTION A titration, as you recall, is a
Density. How tightly the atoms are packed together in an object
 Density How tightly the atoms are packed together in an object Mass vs Weight Mass is the amount of matter in an object. (it doesn t change) Weight is determined based on the amount of gravity pulling
Density How tightly the atoms are packed together in an object Mass vs Weight Mass is the amount of matter in an object. (it doesn t change) Weight is determined based on the amount of gravity pulling
AQA Chemistry A-level
 AQA Chemistry A-level Required Practical 10 Preparation of a pure organic solid, test of its purity, and preparation of a pure organic liquid Reflux Reflux: continuous boiling and condensing of a reaction
AQA Chemistry A-level Required Practical 10 Preparation of a pure organic solid, test of its purity, and preparation of a pure organic liquid Reflux Reflux: continuous boiling and condensing of a reaction
Gravimetric Analysis: Determination of % Sulfur in Fertilizer
 Gravimetric Analysis: Determination % Sulfur in Fertilizer This is another "real world" sample experiment in this case we will analyze a fertilizer sample for the sulfate content and express the result
Gravimetric Analysis: Determination % Sulfur in Fertilizer This is another "real world" sample experiment in this case we will analyze a fertilizer sample for the sulfate content and express the result
E24 PURIFICATION OF ORGANIC COMPOUNDS Distillation, recrystallisation, melting and boiling point determination
 E24 PURIFICATION OF ORGANIC COMPOUNDS Distillation, recrystallisation, melting and boiling point determination THE TASK To learn the main techniques of purifying organic compounds. THE SKILLS By the end
E24 PURIFICATION OF ORGANIC COMPOUNDS Distillation, recrystallisation, melting and boiling point determination THE TASK To learn the main techniques of purifying organic compounds. THE SKILLS By the end
Activity of metals SCIENTIFIC. Demonstration and Inquiry. Introduction. Concepts. Background. Inquiry Approach. Demonstration Questions
 Activity of Metals Demonstration and Inquiry SCIENTIFIC Introduction Chemical reactions are not formulas on a piece of paper they are dynamic and exciting events! The demonstration of aluminum with copper(ii)
Activity of Metals Demonstration and Inquiry SCIENTIFIC Introduction Chemical reactions are not formulas on a piece of paper they are dynamic and exciting events! The demonstration of aluminum with copper(ii)
Colligative Properties of Solutions: Freezing Point Depression
 E1 Colligative Properties of Solutions: Freezing Point Depression PURPOSE The experiment to be performed is divided into three sections: (a) In part 1, the FP of the pure solvent, cyclohexane, is determined
E1 Colligative Properties of Solutions: Freezing Point Depression PURPOSE The experiment to be performed is divided into three sections: (a) In part 1, the FP of the pure solvent, cyclohexane, is determined
UW Department of Chemistry Lab Lectures Online
 Lab 2: Chemical Composition of Compounds Part I. Identification Based on Percent Metal Compositions Part II. Determination of Percent KClO 3 in a KCl/KClO 3 Mixture Procedure Overview Inspect and clean
Lab 2: Chemical Composition of Compounds Part I. Identification Based on Percent Metal Compositions Part II. Determination of Percent KClO 3 in a KCl/KClO 3 Mixture Procedure Overview Inspect and clean
A Hydrogen Powered Bottle Rocket
 A Hydrogen Powered Bottle Rocket Rockets are made by filling plastic water bottles with a mixture of hydrogen gas and oxygen gas. The rocket is launched by igniting the mixture with a flame. The bottle
A Hydrogen Powered Bottle Rocket Rockets are made by filling plastic water bottles with a mixture of hydrogen gas and oxygen gas. The rocket is launched by igniting the mixture with a flame. The bottle
TYPES OF CHEMICAL REACTIONS PART I INTRODUCTION
 EXPERIMENT 10 (2 Weeks) Chemistry 100 Laboratory TYPES OF CHEMICAL REACTIONS PART I INTRODUCTION It is useful to classify reactions into different types, because products of reactions can be predicted.
EXPERIMENT 10 (2 Weeks) Chemistry 100 Laboratory TYPES OF CHEMICAL REACTIONS PART I INTRODUCTION It is useful to classify reactions into different types, because products of reactions can be predicted.
Lab 0.4: Density and Thickness of Aluminum Foil
 Name Block Lab 0.4: Density and Thickness of Aluminum Foil Student will be able to: Correctly use measuring instruments with accuracy Correctly apply the principles of significant figures in measurements
Name Block Lab 0.4: Density and Thickness of Aluminum Foil Student will be able to: Correctly use measuring instruments with accuracy Correctly apply the principles of significant figures in measurements
Experiment 3 MELTING POINTS AND RANGES. New Technique. Discussion. Melting points and ranges.
 E3-1 Experiment 3 Fig. 3-1 MELTING POINTS AND RANGES New Technique Antoine Lavoisier (1743-1794) One of first scientists to make chemistry quantitative. http://www.homepages.hetnet.nl/~b1beukema/wohler.html
E3-1 Experiment 3 Fig. 3-1 MELTING POINTS AND RANGES New Technique Antoine Lavoisier (1743-1794) One of first scientists to make chemistry quantitative. http://www.homepages.hetnet.nl/~b1beukema/wohler.html
Archer G11 Partner: Judy Aug Gravimetric Analysis of a Metal Carbonate
 Gravimetric Analysis of a Metal Carbonate Purpose The purpose of this lab is to identify the unknown carbonate. This can be done by finding the mass of the product carbonate and using stoichiometry on
Gravimetric Analysis of a Metal Carbonate Purpose The purpose of this lab is to identify the unknown carbonate. This can be done by finding the mass of the product carbonate and using stoichiometry on
Station Two Guide. Endothermic and exothermic reactions. A Vocabulary
 Part One: Baking Soda and Vinegar Station Two Guide How is the temperature of vinegar affected when combined with baking soda? Baking soda Measuring cups Vinegar expand kinetic energy potential energy
Part One: Baking Soda and Vinegar Station Two Guide How is the temperature of vinegar affected when combined with baking soda? Baking soda Measuring cups Vinegar expand kinetic energy potential energy
Practice with Properties of Matter. Practice with Physical and Chemical Changes. Practice with Mixtures and Pure Substances
 Chapter 1 Practice Problems Practice with Properties of Matter Indicate whether each of the following statements describes a physical or a chemical property. a) the normal color of elemental bromine is
Chapter 1 Practice Problems Practice with Properties of Matter Indicate whether each of the following statements describes a physical or a chemical property. a) the normal color of elemental bromine is
Experiment 2: The Chromatography of Organic Compounds
 Experiment 2: The Chromatography of Organic Compounds INTRODUCTION When performing an organic reaction, it is very common to observe the formation of other compounds in addition to your desired product;
Experiment 2: The Chromatography of Organic Compounds INTRODUCTION When performing an organic reaction, it is very common to observe the formation of other compounds in addition to your desired product;
CEEN Geotechnical Engineering - Laboratory Session 1 Mechanical Sieve Analysis, Specific Gravity of Solids, Volumetric/Gravimetric Relations
 OBJECTIVE: EQUIPMENT: To obtain data necessary for the classification of a soil sample. A set of sieves (3/8", Nos. 4, 10, 16, 40, 100, 200 and Pan), brushes for cleaning sieves, balance, sieve shaker,
OBJECTIVE: EQUIPMENT: To obtain data necessary for the classification of a soil sample. A set of sieves (3/8", Nos. 4, 10, 16, 40, 100, 200 and Pan), brushes for cleaning sieves, balance, sieve shaker,
Lab Report: Thermal Energy Transfer in Mixtures
 Lab Report: Thermal Energy Transfer in Mixtures Purpose: The purpose of this experiment is to discover how exactly the final temperature of a mixture, involving a substance and hot water, is affected and
Lab Report: Thermal Energy Transfer in Mixtures Purpose: The purpose of this experiment is to discover how exactly the final temperature of a mixture, involving a substance and hot water, is affected and
Demonstration of osmosis
 Demonstration of osmosis We will try to carry out a classical experiment on demonstration of osmosis. The principle is shown in the figure. Water moves from the solution of lower osmolality, across the
Demonstration of osmosis We will try to carry out a classical experiment on demonstration of osmosis. The principle is shown in the figure. Water moves from the solution of lower osmolality, across the
CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK. Due Date Assignment On-Time (100) Late (70)
 Name Period CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK Due Date Assignment On-Time (100) Late (70) 1.1 1.2 1.3 Warm Ups Extra Credit Notes, Homework, Exam Reviews and Their KEYS located
Name Period CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK Due Date Assignment On-Time (100) Late (70) 1.1 1.2 1.3 Warm Ups Extra Credit Notes, Homework, Exam Reviews and Their KEYS located
EXPERIMENT 7A. Chemical Separation by Filtration and Recrystallization INTRODUCTION
 EXPERIMENT 7A Chemical Separation by Filtration and Recrystallization INTRODUCTION The solubilities of solid substances in different kinds of liquid solvents vary widely. Substances that we call salts
EXPERIMENT 7A Chemical Separation by Filtration and Recrystallization INTRODUCTION The solubilities of solid substances in different kinds of liquid solvents vary widely. Substances that we call salts
Unit-2. Properties of Matter. Solutions 2.1 Matter & Density page V 1 =4.3.6=72 cm 3. Volume of the removed triangular prism (V 2 )
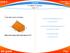 2.1 & Density page - 61 1. A solid object is given in the figure. Volume of the rectangular prism (V 1 ) V 1 =4.3.6=72 cm 3 Volume of the removed triangular prism (V 2 ) V 2 =((2.3)/2).6=18 cm 3 What is
2.1 & Density page - 61 1. A solid object is given in the figure. Volume of the rectangular prism (V 1 ) V 1 =4.3.6=72 cm 3 Volume of the removed triangular prism (V 2 ) V 2 =((2.3)/2).6=18 cm 3 What is
European Pharmacopoeia
 i PHARMACOPOEIAL DISCUSSION GROUP REV 1 - CORR 1 G02 Bulk density and tapped density of powders It is understood that sign-off covers the technical content of the draft and each party will adapt it as
i PHARMACOPOEIAL DISCUSSION GROUP REV 1 - CORR 1 G02 Bulk density and tapped density of powders It is understood that sign-off covers the technical content of the draft and each party will adapt it as
EXPERIMENT 15 FREEZING POINT: A COLLIGATIVE PROPERTY OF SOLUTIONS
 EXPERIMENT 15 FREEZING POINT: A COLLIGATIVE PROPERTY OF SOLUTIONS INTRODUCTION Scientists seldom work alone today. Instead they work in teams on projects, sharing the labor of carrying out the experiments
EXPERIMENT 15 FREEZING POINT: A COLLIGATIVE PROPERTY OF SOLUTIONS INTRODUCTION Scientists seldom work alone today. Instead they work in teams on projects, sharing the labor of carrying out the experiments
C3 USE OF AMOUNT OF SUBSTANCE IN VOLUME OF GASES
 C3 USE OF AMOUNT OF SUBSTANCE IN VOLUME OF GASES Question Practice Name: Class: Date: Time: 24 minutes Marks: 24 marks Comments: GCSE CHEMISTRY ONLY Page of 2 Lead bromide is a solid. Some students were
C3 USE OF AMOUNT OF SUBSTANCE IN VOLUME OF GASES Question Practice Name: Class: Date: Time: 24 minutes Marks: 24 marks Comments: GCSE CHEMISTRY ONLY Page of 2 Lead bromide is a solid. Some students were
Kidney Donor Teacher Information
 STO-102 Kidney Donor Teacher Information Summary Students collect and analyze data to determine which potential living kidney donor could safely donate a kidney to a patient with kidney failure. They perform
STO-102 Kidney Donor Teacher Information Summary Students collect and analyze data to determine which potential living kidney donor could safely donate a kidney to a patient with kidney failure. They perform
Contact Information: Laboratory Supervisor: Denise Kind Laboratory Manager: Mat Ashby
 Biology and Wildlife STANDARD OPERATING PROCEDURE Autoclaving Location(s): Murie 215 Chemical(s): None Specific Hazards: o steam improper use of autoclave can expose user to dangerous steam burns o extremely
Biology and Wildlife STANDARD OPERATING PROCEDURE Autoclaving Location(s): Murie 215 Chemical(s): None Specific Hazards: o steam improper use of autoclave can expose user to dangerous steam burns o extremely
Item 36 has not been slated for public release in 2011.
 S Use the power-plant diagrams and information below to answer questions 35 37. Power Plants 35. Which describes a chemical change and a physical change that take place during the production of electricity
S Use the power-plant diagrams and information below to answer questions 35 37. Power Plants 35. Which describes a chemical change and a physical change that take place during the production of electricity
CHM101 Lab - Energy Grading Rubric
 Name Team Name CHM101 Lab - Energy Grading Rubric To participate in this lab you must have splash- proof goggles, proper shoes and attire. Criteria Points possible Points earned Lab Performance Printed
Name Team Name CHM101 Lab - Energy Grading Rubric To participate in this lab you must have splash- proof goggles, proper shoes and attire. Criteria Points possible Points earned Lab Performance Printed
EXPERIMENT. The Reaction of Magnesium with Hydrochloric Acid; The Molar Volume of Hydrogen
 EXPERIMENT The Reaction of Magnesium with Hydrochloric Acid; The Molar Volume of Hydrogen PURPOSE In this experiment you will determine the volume of the hydrogen gas which is produced when a sample of
EXPERIMENT The Reaction of Magnesium with Hydrochloric Acid; The Molar Volume of Hydrogen PURPOSE In this experiment you will determine the volume of the hydrogen gas which is produced when a sample of
Colligative Properties of Solutions: Freezing Point Depression
 E1 Colligative Properties of Solutions: Freezing Point Depression PURPOSE The experiment to be performed is divided into three sections: (a) In part 1, the FP of the pure solvent, cyclohexane, is determined
E1 Colligative Properties of Solutions: Freezing Point Depression PURPOSE The experiment to be performed is divided into three sections: (a) In part 1, the FP of the pure solvent, cyclohexane, is determined
2 Chemicals, Apparatus and Unit Operations of Analytical Chemistry
 2 Chemicals, Apparatus and Unit Operations of Analytical Chemistry 2A Selecting and Handling Reagents and Other Chemicals NIST (The National Institute of Standards and Technology) 2A-1 Classifying Chemicals
2 Chemicals, Apparatus and Unit Operations of Analytical Chemistry 2A Selecting and Handling Reagents and Other Chemicals NIST (The National Institute of Standards and Technology) 2A-1 Classifying Chemicals
The following are the completed but unbalanced equations. Each equation is numbered to match each step of the cycle:
 REACTIONS OF COPPER Copper will undergo many types of reactions. In this experiment you will observe a sequence of copper reactions. The sequence begins with copper metal and ends with copper metal, so
REACTIONS OF COPPER Copper will undergo many types of reactions. In this experiment you will observe a sequence of copper reactions. The sequence begins with copper metal and ends with copper metal, so
PHARMACOPOEIAL DISCUSSION GROUP BULK AND TAPPED DENSITY OF SOLIDS
 PHARMACOPOEIAL DISCUSSION GROUP BULK AND TAPPED DENSITY OF SOLIDS It is understood that sign-off covers the technical content of the draft and each party will adapt it as necessary to conform to the usual
PHARMACOPOEIAL DISCUSSION GROUP BULK AND TAPPED DENSITY OF SOLIDS It is understood that sign-off covers the technical content of the draft and each party will adapt it as necessary to conform to the usual
Cu (s) Cu 2+ (aq) Cu(OH) 2 (s) CuO (s) Cu 2+ (aq) Cu (s)
 Cycle of Copper Reactions Lab Exercise The following is a protocol for the multi-step transformation of copper metal based upon the following chemical transformations: Cu (s) Cu 2+ (aq) Cu(OH) 2 (s) CuO
Cycle of Copper Reactions Lab Exercise The following is a protocol for the multi-step transformation of copper metal based upon the following chemical transformations: Cu (s) Cu 2+ (aq) Cu(OH) 2 (s) CuO
Evaluate long- range plans concerning resource use and by- product disposal for environmental, economic, and political impact
 Title: Author: Course: A Fluid State Of Mind Amanda Brewer Brookland High School Brookland Physical Science, Chemistry Duration: Three 45 minute class periods Grade Level: 9-12 Objective: The process of
Title: Author: Course: A Fluid State Of Mind Amanda Brewer Brookland High School Brookland Physical Science, Chemistry Duration: Three 45 minute class periods Grade Level: 9-12 Objective: The process of
Determination of the Molar Mass of a Compound by Freezing Point Depression
 Determination of the Molar Mass of a Compound by Freezing Point Depression Objective: The objective of this experiment is to determine the molar mass of an unknown solute by measuring the freezing point
Determination of the Molar Mass of a Compound by Freezing Point Depression Objective: The objective of this experiment is to determine the molar mass of an unknown solute by measuring the freezing point
To identify and classify various types of chemical reactions.
 Cycle of Copper Reactions Minneapolis Community and Technical College v.11.17 Objectives: To observe and document copper s chemical changes in five different reactions and verify that copper is conserved
Cycle of Copper Reactions Minneapolis Community and Technical College v.11.17 Objectives: To observe and document copper s chemical changes in five different reactions and verify that copper is conserved
Topic 1 Review Questions Set 1
 Topic 1 Review Questions Set 1 Base your answers to questions 1-5 on your knowledge of Earth Science, the Earth Science Reference Tables, and the diagrams below. The diagrams represent three samples of
Topic 1 Review Questions Set 1 Base your answers to questions 1-5 on your knowledge of Earth Science, the Earth Science Reference Tables, and the diagrams below. The diagrams represent three samples of
CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK. Due Date Assignment On-Time (100) Late (70)
 Name KEY Period CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK Due Date Assignment On-Time (100) Late (70) 1.1 1.2 1.3 Warm Ups Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic
Name KEY Period CRHS Academic Chemistry Unit 1 Matter and Change HOMEWORK Due Date Assignment On-Time (100) Late (70) 1.1 1.2 1.3 Warm Ups Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic
Science 8. Unit 1. Booklet
 Science 8 Unit 1 Mixture and Flow of Matter Booklet Name: Class: 1 TOPIC 1 REINFORCEMENT The Particle Model Goal Demonstrate your understanding of the particle model and changes of state. BLM 1-1 Answer
Science 8 Unit 1 Mixture and Flow of Matter Booklet Name: Class: 1 TOPIC 1 REINFORCEMENT The Particle Model Goal Demonstrate your understanding of the particle model and changes of state. BLM 1-1 Answer
CEEN Laboratory 1 Mechanical Sieve Analysis Specific Gravity of Soil Solids Gravimetric/Volumetric Relations
 INTRODUCTION CEEN 3160 - Laboratory 1 Mechanical Sieve Analysis Specific Gravity of Soil Solids Gravimetric/Volumetric Relations Grain size analysis is widely used for the classification of soils and for
INTRODUCTION CEEN 3160 - Laboratory 1 Mechanical Sieve Analysis Specific Gravity of Soil Solids Gravimetric/Volumetric Relations Grain size analysis is widely used for the classification of soils and for
Geometric formulas for volume include: Where r is the radius of the cylinder and h is the height. Volume of a rectangular prism = LxWxH
 Supplement Density The density of an object is defined as its mass per unit of volume. The formula below may be used to calculate the density of an object: Density = mass/volume Volume may be measured
Supplement Density The density of an object is defined as its mass per unit of volume. The formula below may be used to calculate the density of an object: Density = mass/volume Volume may be measured
Practice Problems; Chapter#1
 Practice Problems; Chapter#1 #1. Determine the density of an object that has a mass of 149.8 g and displaces 12.1 ml of water when placed in a graduated cylinder. A) 8.08 g/ml B) 1.38 g/ml C) 12.4 g/ml
Practice Problems; Chapter#1 #1. Determine the density of an object that has a mass of 149.8 g and displaces 12.1 ml of water when placed in a graduated cylinder. A) 8.08 g/ml B) 1.38 g/ml C) 12.4 g/ml
Chemistry. Freezing and Melting of Water ID: By Texas Instruments TEACHER GUIDE
 Freezing and Melting of Water ID: 16150 By Texas Instruments TEACHER GUIDE Time required 90 minutes Topic: Phase Changes Relate freezing point and melting point. Interpret a heating or cooling curve. Describe
Freezing and Melting of Water ID: 16150 By Texas Instruments TEACHER GUIDE Time required 90 minutes Topic: Phase Changes Relate freezing point and melting point. Interpret a heating or cooling curve. Describe
DOWNLOAD PDF CYCLE OF COPPER REACTIONS
 Chapter 1 : Copperâ chlorine cycle - Wikipedia CYCLE OF COPPER REACTIONS. PURPOSE: The goal of the experiment is to observe a series of reactions involving copper that form a cycle and calculate the percent
Chapter 1 : Copperâ chlorine cycle - Wikipedia CYCLE OF COPPER REACTIONS. PURPOSE: The goal of the experiment is to observe a series of reactions involving copper that form a cycle and calculate the percent
Chemistry: The Central Science, 12e (Brown et al.) Chapter 1 Introduction: Matter and Measurement. 1.1 Multiple-Choice Questions
 Chemistry: The Central Science, 12e (Brown et al.) Chapter 1 Introduction: Matter and Measurement 1.1 Multiple-Choice Questions 1) In the following list, only is not an example of matter. A) planets B)
Chemistry: The Central Science, 12e (Brown et al.) Chapter 1 Introduction: Matter and Measurement 1.1 Multiple-Choice Questions 1) In the following list, only is not an example of matter. A) planets B)
Practice Problems; Chapter#1
 Practice Problems; Chapter#1 #1. Determine the density of an object that has a mass of 149.8 g and displaces 12.1 ml of water when placed in a graduated cylinder. A) 8.08 g/ml B) 1.38 g/ml C) 12.4 g/ml
Practice Problems; Chapter#1 #1. Determine the density of an object that has a mass of 149.8 g and displaces 12.1 ml of water when placed in a graduated cylinder. A) 8.08 g/ml B) 1.38 g/ml C) 12.4 g/ml
Chemical Reactions Lab. Please remember, this power point is posted online
 Chemical Reactions Lab Please remember, this power point is posted online Chemical Reactions Lab This lab will be a partial formal write up, the data table and conclusion will be HIGH Priority! You may
Chemical Reactions Lab Please remember, this power point is posted online Chemical Reactions Lab This lab will be a partial formal write up, the data table and conclusion will be HIGH Priority! You may
Material Safety Data Sheet
 Material Safety Data Sheet CALCIUM-MAGNESIUM CHLORIDE SOLUTION 29% TO 35% DustMaster 20 A. GENERAL INFORMATION TRADE ME (COMMON ME): CALCIUM-MAGNESIUM CHLORIDE SOLUTION- DM29 to DM35 CHEMICAL ME AND/OR
Material Safety Data Sheet CALCIUM-MAGNESIUM CHLORIDE SOLUTION 29% TO 35% DustMaster 20 A. GENERAL INFORMATION TRADE ME (COMMON ME): CALCIUM-MAGNESIUM CHLORIDE SOLUTION- DM29 to DM35 CHEMICAL ME AND/OR
Material Safety Data Sheet
 Material Safety Data Sheet CALCIUM CHLORIDE SOLUTION DustMaster 20 A. GENERAL INFORMATION TRADE ME (COMMON ME): CALCIUM CHLORIDE SOLUTION DUSTMASTER CHEMICAL ME AND/OR SYNONYM: LIQUID CALCIUM CHLORIDE,
Material Safety Data Sheet CALCIUM CHLORIDE SOLUTION DustMaster 20 A. GENERAL INFORMATION TRADE ME (COMMON ME): CALCIUM CHLORIDE SOLUTION DUSTMASTER CHEMICAL ME AND/OR SYNONYM: LIQUID CALCIUM CHLORIDE,
Quiz 2 practice Quiz
 Name: ate: 1. The following data were recorded while determining the solubility of a certain salt. Temp. ( C) 10 20 30 40 50 Grams Solute/ 100 g H 2 O 30 33 36 39 42 Which graph best represents the solubility
Name: ate: 1. The following data were recorded while determining the solubility of a certain salt. Temp. ( C) 10 20 30 40 50 Grams Solute/ 100 g H 2 O 30 33 36 39 42 Which graph best represents the solubility
Chem 2115 Experiment #9. Consumer Chemistry: Determining the Iron Content in Supplements
 Chem 2115 Experiment #9 Consumer Chemistry: Determining the Iron Content in Supplements OBJECTIVE: The goal of this experiment is to use the quantitative technique of spectrophotometry to determine the
Chem 2115 Experiment #9 Consumer Chemistry: Determining the Iron Content in Supplements OBJECTIVE: The goal of this experiment is to use the quantitative technique of spectrophotometry to determine the
Experiment 3: The Chromatography of Organic Compounds
 Experiment 3: The Chromatography of Organic Compounds INTRODUCTION Very often, in an organic synthesis, a reaction will proceed to produce multiple products or perhaps will only partially form the desired
Experiment 3: The Chromatography of Organic Compounds INTRODUCTION Very often, in an organic synthesis, a reaction will proceed to produce multiple products or perhaps will only partially form the desired
Single/Double Displacement Lab
 Pre-lab Answer this question at your first lab station only. Table 1: 1. Take a few pellets, one piece or tweezer full of each metal and put them each in a separate well in the well plate. Place well plate
Pre-lab Answer this question at your first lab station only. Table 1: 1. Take a few pellets, one piece or tweezer full of each metal and put them each in a separate well in the well plate. Place well plate
Density What is density? How do you measure density?
 Density What is density? How do you measure density? What is density? Density describes how much mass is in a given volume of a material. Mass: the amount of matter in an object. Volume: the amount of
Density What is density? How do you measure density? What is density? Density describes how much mass is in a given volume of a material. Mass: the amount of matter in an object. Volume: the amount of
