Adeno-Associated Virus as a Vector for Liver-Directed Gene Therapy
|
|
|
- Ralf French
- 6 years ago
- Views:
Transcription
1 JOURNAL OF VIROLOGY, Dec. 1998, p Vol. 72, No X/98/$ Copyright 1998, American Society for Microbiology. All Rights Reserved. Adeno-Associated Virus as a Vector for Liver-Directed Gene Therapy WEIDONG XIAO, SCOTT C. BERTA, MIN MIN LU, A. DAVID MOSCIONI, JOHN TAZELAAR, AND JAMES M. WILSON* Institute for Human Gene Therapy and Departments of Molecular and Cellular Engineering and of Medicine, University of Pennsylvania, and the Wistar Institute, Philadelphia, Pennsylvania Received 10 April 1998/Accepted 2 September 1998 Factors relevant to the successful application of adeno-associated virus (AAV) vectors for liver-directed gene therapy were evaluated. Vectors with different promoters driving expression of human -1-antitrypsin ( -1AT) were injected into the portal circulation of immunodeficient mice. -1AT expression was stable but dependent on the promoter. Southern analysis of liver DNA revealed approximately 0.1 to 2.0 provirus copies/diploid genome in presumed head-to-tail concatamers. In situ hybridization and immunohistochemical analysis revealed expression in approximately 5% of hepatocytes clustered in the pericentral region. These results support the use of AAV as a vector for diseases treatable by targeting of hepatocytes. The liver is an excellent target for in vivo gene therapy because hepatocytes are easily accessible to vectors injected into the circulation through large pores in liver capillaries (6). Nonviral gene delivery vehicles, based on liposomes and DNAprotein complexes, target hepatocytes in vivo, although gene transfer efficiency is low and expression is transient (20, 24). Recombinant adenoviruses target hepatocytes with higher efficiency but suffer from destructive cellular and blocking humoral immune responses (7, 11, 22). Adeno-associated virus (AAV) has shown promise for in vivo gene therapy in a number of organs, such as skeletal muscle, central nervous system, and retina, where expression is efficient, stable, and associated with little inflammation or cellular immune response (1, 4, 5, 10, 12, 21). Results in the liver have been less consistent, with Snyder et al. providing the most impressive results by achieving sustained and therapeutic levels of factor IX in mice from an AAV vector utilizing a retroviral long terminal repeat (LTR) promoter (18). The goal of this study was to further evaluate the potential of AAV as a vector for in vivo gene therapy of the liver. Studies were performed with human -1-antitrypsin ( -1AT) as a reporter gene in murine models, because it allows quantitation of overall expression of the transgene through enzyme-linked immunosorbent assay (ELISA) of -1AT in serum. Vectors in which -1AT is driven from one of a number of promoters were constructed; they included (i) the 5 flanking sequence and upstream enhancer sequences of the albumin gene (Alb), (ii) the 5 flanking region of the immediate early gene of cytomegalovirus (CMV), (iii) the 5 flanking region of the human phosphoglycerate kinase gene (PGK), (iv) the 5 LTR of the Moloney murine leukemia virus, and (v) the chicken -actin promoter fused to the enhancer region of the CMV immediate early gene (CB). Recombinant stocks of AAV for each vector were created by a transfection approach that does not require coinfection with adenovirus. Hepatocytes were transduced in vivo following infusion of genome equivalents of AAV into the portal circulation * Corresponding author. Mailing address: 204 Wistar, 3601 Spruce St., Philadelphia, PA Phone: (215) Fax: (215) wilsonjm@mail.med.upenn.edu. of immunodeficient mice. Figure 1 shows the level of human -1AT in serum of recipient mice for 18 weeks following gene transfer. The profile of expression with each vector was essentially the same, with the concentration of serum -1AT increasing during the first 6 weeks and thereafter stabilizing for the duration of the experiment. Total production of -1AT was consistent within each group but varied substantially between the different promoters. The highest expression was noted with the albumin and LTR constructs, with levels of -1AT ranging from5to50 g/ml. The CMV enhanced -actin promoter (CB) yielded intermediate levels of -1AT (i.e., 1 to 10 g/ml), whereas expression from the CMV and PGK promoters was substantially lower, at approximately 0.1 to 1 g/ml. Additional experiments conducted with the albumin construct demonstrated a direct relationship between the dose of vector and transgene expression over a range of to vector genomes (data not shown). The impact of the promoter on the level of transgene expression, noted in our study, may explain some discrepancies in the literature. Two groups failed to demonstrate significant expression of lacz in the liver with AAV vectors containing the CMV promoter (3, 17). The performance of the murine leukemia virus LTR is more complex. The high level of activity of the LTR demonstrated in our study is consistent with the data of Snyder et al., who achieved substantial levels of factor IX with a similar vector (18), but is discordant with the results of Koeberl et al., where expression of alkaline phosphatase and factor IX from the LTR was low (13). Additional studies were performed to evaluate the molecular state of the AAV genome in recipients. Livers were harvested from selected animals at 14 weeks after vector infusion and subjected to DNA hybridization analysis, the results of which are presented in Fig. 2. The total content of proviral DNA in liver was evaluated by digesting cellular DNA with enzymes that release an internal 800-bp fragment followed by DNA hybridization with human -1AT used as a probe (Fig. 2A). Proviral DNA was detected in livers from each animal, ranging from approximately 0.1 to 2 proviral copies per diploid genome. Undigested DNA produced a smear without a discrete band (Fig. 2B, lane 1). DNA restricted with endonucleases that contain a single site within the provirus yielded discrete bands whose apparent molecular weights were equivalent 10222
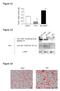
2 VOL. 72, 1998 NOTES FIG. 1. Levels of Human -1AT in serum of mice after intraportal infusion of AAV vector. RAG-1 mice were injected with genomes of AAV vector, bearing different promoters, directly into the portal circulation via the spleen. Vector constructs contained the EcoRI fragment of pat85 (from the American Type Culture Collection), spanning human -1AT cdna fused to the polyadenylation sequence from simian virus 40. Each panel presents serum -1AT levels from multiple animals for vectors with different promoters measured up to 18 weeks after vector infusion. -1AT was measured by an ELISA with -1AT antibodies purchased from Sigma. The different promoters were as follows: CB, chicken -actin promoter ( 1to 275) fused to enhancer sequences from the immediate early gene of CMV (23); CMV, promoter and enhancer of the immediate early gene of CMV spanning 795 bp of the 5 flanking sequence; LTR, 2.3-kb Moloney murine leukemia virus LTR promoter isolated from pbr-mfg containing the LTR and intron (2); Alb, 2.4-kb murine albumin promoter from palbupa2 (NotI-to-KpnI fragment) containing 300 bp of the 5 flanking sequence and the enhancer region from 8.5 to 10.4 kb (16); PGK, 0.5-kb PGK promoter from PGK-neo (from M. McBurney and K. Jardine). Experiments were repeated with a number of different vector preparations (CB [n 1], CMV, [n 3], LTR [n 1], Alb [n 5], and PGK [n 2]), all with identical results. Recombinant AAV based on AAV serotype 2 was produced by simultaneously transfecting three plasmids into 293 cells and purifying vector from the cell lysate by sequential cesium chloride sedimentation. The three plasmids were vector, pjwx500 (inverted terminal repeats deleted from wild-type AAV and a 500-bp stuffer inserted between p5 and rep), and PF 13 (the adenovirus [Ad] helper construct created by deleting the RsrII fragment from pfg140 [Microbix, Toronto, Canada]). These preparations contained varying levels of replication-competent AAV (rcaav), ranging from 1/10 3 to 1/10 5 (rcaav genomes/vector AAV genomes), and no detectable Ad based on PFU assay ( 1 Ad PFU/10 11 vector AAV genomes). FIG. 2. Evaluation of the status of AAV vector genome in mouse liver by DNA hybridization analysis. RAG-1 mice were injected with vector genomes of AAV vectors containing different promoters. Livers were harvested at week 16 post-vector administration. DNA hybridization studies were performed with the whole cdna of human -1AT as a probe following the loading of 10 g of restricted total cellular DNA per lane. (A) Lanes 1 to 4, control mouse genomic DNA spiked with 4, 0.4, 0.04, and 0 copies of human -1AT plasmid digested with BamHI and EcoRV per diploid genome. This combination of enzymes releases an 800-bp fragment from the -1AT cdna. Lanes 5 to 9, genomic DNA from mice that received vectors with different promoters, as described in the legend to Fig. 1. DNA was digested with BamHI and EcoRV. The vectorspecific fragment from DNA of transduced liver is 800 bp in length. The other two bands represent hybridization to endogenous mouse -1AT sequence. (B) The liver genomic DNA from a mouse injected with vector expressing -1AT from the Alb promoter was further analyzed. Lane 1, undigested genomic DNA; lane 2, genomic DNA digested with single-cut enzyme EcoRV; lane 3, genomic DNA digested with single-cut enzyme HindIII; lane 4, genomic DNA digested with both EcoRV and HindIII, which gives rise to a 3.1-kb internal fragment.
3 10224 NOTES J. VIROL. FIG. 3. Localization of -1AT expression in liver. The liver was harvested from a mouse 16 weeks after it received vector expressing -1AT from the Alb promoter and was analyzed by histochemical staining for -1AT protein by using rabbit anti-human -1AT (Sigma) as the primary antibody and biotinylated goat anti-rabbit immunoglobulin (Sigma) and the Vector Labs alkaline phosphatase kit (Vector Laboratories, Burlingame, Calif.). Specific binding was visualized by the alkaline phosphatase colorimetric assay. (A) Low magnification ( 40) illustrating representative pericentral (insets B and C) and periportal (insets D and E) regions. (B and C) High magnification ( 200) of representative pericentral regions. (D and E) High magnification ( 200) of periportal regions. The liver from a mouse that received the CB vector was harvested 18 weeks later, and paraffin sections were subjected to in situ hybridization with sense and antisense -1AT probes. Sections were viewed under light and dark-field microscopy. Representative sections hybridizing to the antisense probe are shown. No signal was observed in sections hybridized with the sense probe. (F) Low magnification ( 40) of a section of liver. Representative pericentral (G and H) and periportal (I and J) regions are demarcated. (H) High magnification ( 200) of representative pericentral region. (J) High magnification ( 200) of representative periportal region. to that of the provirus (Fig. 2B, lanes 2 and 3). A smaller band of equal intensity was observed when total cellular DNA was restricted with two endonucleases that together release a 3.1- kb internal fragment (Fig. 2B, lane 4). These data are most consistent with a model in which AAV exists as an integrated head-to-tail concatamer similar to what is seen with latent AAV infection (14, 15, 19) and muscle transduced with AAV vectors (4, 21). A number of histological analyses were performed to evaluate the frequency and distribution of AAV transduction fol-
4 VOL. 72, 1998 NOTES FIG. 3 Continued. lowing intraportal infusion. Immunohistochemical hybridization analysis with an antibody to human -1AT probe revealed staining over 5% of hepatocytes throughout the liver (Fig. 3A to E) that was not found in mock-infected animals (data not shown). More transgene-expressing cells were clustered around central veins (Fig. 3B and C) than around portal triads (Fig. 3D and E). Similar frequency and distribution of transgene-expressing cells were found by in situ hybridization with a probe to human -1AT (Fig. 3F to J). This does not agree with the findings of Snyder et al., who claimed expression of AAVencoded tyrosine hydroxylase in cells around periportal regions (18). In our study, the pericentral localization of transduction was observed with each promoter, including the one used by Snyder et al., suggesting that other differences must play a role (e.g., method of delivery). The reasons for the observed gradient of transduction are unclear although not unexpected, considering that many genes demonstrate a gradient of expression along the portal to the central hepatic axis (9). Data provided in this report and in the report of Snyder et al. (18) clearly support the use of AAV for targeting to hepatocytes genes that encode secreted proteins. What is the potential of AAV for treatment of metabolic diseases where the hepatocyte serves a catabolic or metabolic function, such as in familial hypercholesterolemia or urea cycle disorders? In situ
5 10226 NOTES J. VIROL. hybridization and immunohistochemistry revealed transduction in 5% of hepatocytes following infusion of vector genomes of recombinant AAV. Increased catabolic/metabolic capacity would best be achieved if transgene expression was distributed throughout a greater number of hepatocytes. Transgene expression appears to be proportional to dose up to vector genomes per injection, suggesting that increased doses will lead to more transduced hepatocytes. Direct confirmation of this hypothesis will require improved methods of AAV production to allow injection of higher vector doses. In summary, AAV appears to be a viable alternative to adenoviruses and nonviral vectors for in vivo gene therapy to the liver. The advantages of immune evasion that attend their use in muscle (8) need to be confirmed in the liver, although preliminary results in immunocompetent mice suggest that elimination of AAV-transduced hepatocytes by cytotoxic T lymphocytes will not be as much of a problem as has been observed with adenoviruses (data not shown). A better understanding of the mechanism and distribution of proviral integration is needed. We acknowledge the Vector and Cell Morphology Cores of the Institute for Human Gene Therapy and the technical support of Marcia Houston-Leslie, Rosalind Barr, Melissa Casey, and Sarah Ehlen- Haecker. This work was supported by grants from the NIH (P01 HD and P30 DK [to J. M. Wilson]) and by Genovo, Inc., a company J. M. Wilson founded and has equity in. REFERENCES 1. Bennett, J., D. Duan, J. Engelhardt, and A. M. Maguire Real-time non-invasive in vivo assessment of adeno-associated virus-mediated retinal transduction. Investig. Ophthalmol. Vis. Sci. 38: Dranoff, G., E. Jaffee, A. Lazenby, P. Golumbek, H. Levitsky, K. Brose, V. Jackson, H. Hamada, D. Pardoll, and R. C. Mulligan Vaccination with irradiated tumor cells engineered to secrete murine granulocyte-macrophage colony-stimulating factor stimulates potent, specific, and long-lasting antitumor immunity. Proc. Natl. Acad. Sci. USA 90: Fisher, K. J., G.-P. Gao, M. D. Weitzman, R. DeMatteo, J. F. Burda, and J. M. Wilson Transduction with recombinant adeno-associated virus for gene therapy is limited by leading-strand synthesis. J. Virol. 70: Fisher, K. J., K. Jooss, J. Alston, Y. Yang, S. E. Haecker, K. High, R. Pathak, S. E. Raper, and J. M. Wilson Recombinant adeno-associated virus for muscle-directed gene therapy. Nat. Med. 3: Flannery, J. G., S. Zolotukhin, M. I. Vaquero, M. M. LaVail, N. Muzyczka, and W. W. Hauswirth Efficient photoreceptor-targeted gene expression in vivo by recombinant adeno-associated virus. Proc. Natl. Acad. Sci. USA 94: Fraser, R., B. R. Dobbs, and G. W. T. Rogers Lipoproteins and the liver sieve: the role of fenestrated sinusoidal endothelium in lipoprotein metabolism, atherosclerosis, and cirrhosis. Hepatology 3: Jaffe, H. A., C. Danel, G. Longenecker, M. Metzger, Y. Setoguchi, M. A. Rosenfeld, T. W. Gant, S. S. Thorgeirsson, L. D. Stratford-Perricaudet, M. Perricaudet, A. Pavirani, J.-P. Lecocq, and R. G. Crystal Adenovirusmediated in vivo gene transfer and expression in normal rat liver. Nat. Genet. 1: Jooss, K., Y. Yang, K. J. Fisher, and J. M. Wilson Transduction of dendritic cells by DNA viral vectors directs the immune response to transgene products in muscle fibers. J. Virol. 72: Jungerman, K Zonation of metabolism and gene expression in liver. Histochemistry 103: Kaplitt, M. G., P. Leone, R. J. Samulski, X. Xiao, D. W. Pfaff, K. L. O Malley, and M. J. During Long-term gene expression and phenotypic correction using adeno-associated virus vectors in the mammalian brain. Nat. Genet. 8: Kay, M. A., F. Graham, F. Leland, and S. L. C. Woo Therapeutic serum concentrations of human alpha-1-antitrypsin after adenoviral-mediated gene transfer into mouse hepatocytes. Hepatology 21: Kessler, P. D., G. M. Podsakoff, X. Chen, S. A. McQuiston, P. C. Colosi, L. A. Matelis, G. J. Kurtzman, and B. J. Byrne Gene delivery to skeletal muscle results in sustained expression and systemic delivery of a therapeutic protein. Proc. Natl. Acad. Sci. USA 93: Koeberl, D. D., I. E. Alexander, C. L. Halbert, D. W. Russell, and A. D. Miller Persistent expression of human clotting factor IX from mouse liver after intravenous injection of adeno-associated virus vectors. Proc. Natl. Acad. Sci. USA 94: McLaughlin, S. K., P. Collis, P. L. Hermonat, and N. Muzcyzka Adeno-associated virus general transduction vectors: analysis of proviral structures. J. Virol. 62: Muzcyzka, N Use of adeno-associated virus as a general transduction vector in mammalian cells. Curr. Top. Microbiol. Immunol. 158: Pinkert, C. A., D. M. Ornitz, R. L. Brinster, and R. D. Palmiter An albumin enhancer located 10 kb upstream functions along with its promoter to direct efficient, liver-specific expression in transgenic mice. Genes Dev. 1: Ponnazahan, S., P. Mukherjee, M. C. Yoder, X.-S. Wang, S. Z. Zhou, J. Kaplan, S. Wadsworth, and A. Srivastava Adeno-associated virus 2-mediated gene transfer in vivo: organ-tropism and expression of transduced sequences in mice. Gene 190: Snyder, R. O., C. H. Miao, G. A. Patijn, S. K. Spratt, O. Danos, D. Nagy, A. M. Brown, B. Winther, L. Meuse, L. K. Cohen, A. R. Thompson, and M. A. Kay Persistent and therapeutic concentrations of human factor IX in mice after hepatic gene transfer of recombinant AAV vectors. Nat. Genet. 16: Tratschin, J.-D., I. L. Miller, M. G. Smith, and B. J. Carter Adenoassociated virus vector for high-frequency integration, expression, and rescue of genes in mammalian cells. Mol. Cell. Biol. 5: Wu, G., and C. H. Wu Receptor-mediated gene delivery and expression in vivo. J. Biol. Chem. 263: Xiao, X., J. Li, and R. J. Samulski Efficient long-term gene transfer into muscle tissue of immunocompetent mice by adeno-associated virus vectors. J. Virol. 70: Yang, Y., F. A. Nunes, K. Berencsi, E. E. Furth, E. Gönczöl, and J. M. Wilson Cellular immunity to viral antigens limits E1-deleted adenoviruses for gene therapy. Proc. Natl. Acad. Sci. USA 91: Ye, X., M. B. Robinson, M. L. Batshaw, E. E. Furth, I. Smith, and J. M. Wilson Prolonged metabolic correction in adult ornithine transcarbamylase deficient mice with adenoviral vectors. J. Biol. Chem. 271: Zhu, N., D. Liggett, Y. Liu, and R. Debs Systemetric gene expression after intravenous DNA delivery into adult mice. Science 261:
Mutagenesis and Expression of Mammalian Clotting Factor IX. Mark McCleland The Children s Hospital of Philadelphia and Lycoming College
 Mutagenesis and Expression of Mammalian Clotting Factor IX Mark McCleland The Children s Hospital of Philadelphia and Lycoming College Hemophilia B X-linked blood clotting disorder characterized by a deficiency
Mutagenesis and Expression of Mammalian Clotting Factor IX Mark McCleland The Children s Hospital of Philadelphia and Lycoming College Hemophilia B X-linked blood clotting disorder characterized by a deficiency
VECTOR SYSTEMS. Vector Systems
 VECTOR SYSTEMS Vector Systems The vector systems currently available through Penn Vector Core are those derived from adenoassociated virus (AAV), adenovirus and lentivirus. The Core offers AAV-based vectors
VECTOR SYSTEMS Vector Systems The vector systems currently available through Penn Vector Core are those derived from adenoassociated virus (AAV), adenovirus and lentivirus. The Core offers AAV-based vectors
Utility of a Recombinant Adeno-Associated Viral Vector Reference Standard BY Terence R. Flotte, Parris Burd, and Richard O. Snyder
 Utility of a Recombinant Adeno-Associated Viral Vector Reference Standard BY Terence R. Flotte, Parris Burd, and Richard O. Snyder Recombinant Adeno-associated viral (raav) vectors are known to be efficient
Utility of a Recombinant Adeno-Associated Viral Vector Reference Standard BY Terence R. Flotte, Parris Burd, and Richard O. Snyder Recombinant Adeno-associated viral (raav) vectors are known to be efficient
Quality Control Assays
 QUALITY CONTROL An integral part of the Penn Vector Core is its robust quality control program which is carried out by a separate quality control group. Quality control assays have been developed and optimized
QUALITY CONTROL An integral part of the Penn Vector Core is its robust quality control program which is carried out by a separate quality control group. Quality control assays have been developed and optimized
Manufacturing Viral Gene Therapy Vectors: General Approaches and Challenges John T. Gray
 Manufacturing Viral Gene Therapy s: 1 Dr. Vice President, Research & Development Audentes Therapeutics, Inc. San Francisco, USA Natural virus life cycle Late Early Viral Virally infected cell Naïve cell
Manufacturing Viral Gene Therapy s: 1 Dr. Vice President, Research & Development Audentes Therapeutics, Inc. San Francisco, USA Natural virus life cycle Late Early Viral Virally infected cell Naïve cell
Hemophilia and Gene Therapy
 Hemophilia and Gene Therapy Jackie Chu June 4, 2008 Overview Hemophilia, the disease Gene therapy Hemophilia as a target for gene therapy Gene delivery systems Clinical trials New methods Future of gene
Hemophilia and Gene Therapy Jackie Chu June 4, 2008 Overview Hemophilia, the disease Gene therapy Hemophilia as a target for gene therapy Gene delivery systems Clinical trials New methods Future of gene
Hybrid Vectors Based on Adeno-Associated Virus Serotypes 2 and 5 for Muscle-Directed Gene Transfer
 JOURNAL OF VIROLOGY, July 2001, p. 6199 6203 Vol. 75, No. 13 0022-538X/01/$04.00 0 DOI: 10.1128/JVI.75.13.6199 6203.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved. Hybrid Vectors
JOURNAL OF VIROLOGY, July 2001, p. 6199 6203 Vol. 75, No. 13 0022-538X/01/$04.00 0 DOI: 10.1128/JVI.75.13.6199 6203.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved. Hybrid Vectors
Hepatocyte-specific gene expression by a recombinant adeno-associated virus vector carrying the apolipoprotein E enhancer and 1-antitrypsin promoter
 Gene Therapy and Molecular Biology Vol 3, page 67 Gene Ther Mol Biol Vol 3, 67-74. August 1999. Hepatocyte-specific gene expression by a recombinant adeno-associated virus vector carrying the apolipoprotein
Gene Therapy and Molecular Biology Vol 3, page 67 Gene Ther Mol Biol Vol 3, 67-74. August 1999. Hepatocyte-specific gene expression by a recombinant adeno-associated virus vector carrying the apolipoprotein
Sustained Expression of Therapeutic Level of Factor IX in Hemophilia B Dogs by AAV-Mediated Gene Therapy in Liver
 doi:10.1006/mthe.2000.0031, available online at http://www.idealibrary.com on IDEAL Sustained Expression of Therapeutic Level of Factor IX in Hemophilia B Dogs by AAV-Mediated Gene Therapy in Liver Lili
doi:10.1006/mthe.2000.0031, available online at http://www.idealibrary.com on IDEAL Sustained Expression of Therapeutic Level of Factor IX in Hemophilia B Dogs by AAV-Mediated Gene Therapy in Liver Lili
10/07/2018. Liver directed gene therapy. - An overview. DNA/RNA therapies for Wilson s disease. The liver, organ of choice. Highly vascularised
 DNA/RNA therapies for Wilson s disease Julien Baruteau - Great Ormond Street Institute of Child Health, University College London, UK Metabolic Medicine, Great Ormond Street Hospital, London, UK PLAN Landscape
DNA/RNA therapies for Wilson s disease Julien Baruteau - Great Ormond Street Institute of Child Health, University College London, UK Metabolic Medicine, Great Ormond Street Hospital, London, UK PLAN Landscape
Gene Therapy: Fellow s Conference Fred Hutchinson Cancer Research Center. Zandra K. Klippel
 Gene Therapy: Did we finally get it right? Fellow s Conference Fred Hutchinson Cancer Research Center University it of Washington Zandra K. Klippel March 2012 Introduction >6000 human diseases are monogenic
Gene Therapy: Did we finally get it right? Fellow s Conference Fred Hutchinson Cancer Research Center University it of Washington Zandra K. Klippel March 2012 Introduction >6000 human diseases are monogenic
GC GC - AAV2/8-4E10
 SUPPLEMENTARY INFORMATION doi:.38/nature660 a Luciferase Expression (Photons/Sec) c f 12 11 11 GC - AAV2/8-Luciferase 9 GC - AAV2/8-Luciferase 0 8 16 24 32 40 48 56 64 Weeks Post-AAV Injection Human IgG
SUPPLEMENTARY INFORMATION doi:.38/nature660 a Luciferase Expression (Photons/Sec) c f 12 11 11 GC - AAV2/8-Luciferase 9 GC - AAV2/8-Luciferase 0 8 16 24 32 40 48 56 64 Weeks Post-AAV Injection Human IgG
Stable gene transfer and expression of human blood coagulation factor IX after intramuscular injection of recombinant adeno-associated virus
 Proc. Natl. Acad. Sci. USA Vol. 94, pp. 5804 5809, May 1997 Medical Sciences Stable gene transfer and expression of human blood coagulation factor IX after intramuscular injection of recombinant adeno-associated
Proc. Natl. Acad. Sci. USA Vol. 94, pp. 5804 5809, May 1997 Medical Sciences Stable gene transfer and expression of human blood coagulation factor IX after intramuscular injection of recombinant adeno-associated
VECTOR SAFETY INFORMATION. AAV Vectors: Material Information
 VECTOR SAFETY INFORMATION AAV Vectors: Material Information AAV vectors contain recombinant transgene sequences (e.g. encoding reporter or therapeutic genes) flanked by the AAV inverted terminal repeats
VECTOR SAFETY INFORMATION AAV Vectors: Material Information AAV vectors contain recombinant transgene sequences (e.g. encoding reporter or therapeutic genes) flanked by the AAV inverted terminal repeats
Lecture 7 Adeno-associated viral vectors
 Lecture 7 Adeno-associated viral vectors Viral vectors, continued 19th November 2012 AAV adenovirus 2 Infectious cycle of AAV 3 Adeno-associated viruses AAV Small, non-pathogenic single stranded DNA viruses.
Lecture 7 Adeno-associated viral vectors Viral vectors, continued 19th November 2012 AAV adenovirus 2 Infectious cycle of AAV 3 Adeno-associated viruses AAV Small, non-pathogenic single stranded DNA viruses.
CRN 18-mo progress update
 CRN 18-mo progress update Gene transfer studies for cystinosis Principal investigator: Vasiliki Kalatzis, Ph.D. Co-investigator: Eric J. Kremer, Ph.D. Affiliation: Institut Génétique Moléculaire de Montpellier
CRN 18-mo progress update Gene transfer studies for cystinosis Principal investigator: Vasiliki Kalatzis, Ph.D. Co-investigator: Eric J. Kremer, Ph.D. Affiliation: Institut Génétique Moléculaire de Montpellier
The Molecular Virology Support Core: Adenoviral Vectors and Beyond
 The : Adenoviral Vectors and Beyond Christoph A. Kahl, Ph.D. Viral Vector Workshop, May 5th, 2011 Outline 1) Overview of the MVSC 2) Adenoviral Vectors 3) Expertise and Services Overview What does the
The : Adenoviral Vectors and Beyond Christoph A. Kahl, Ph.D. Viral Vector Workshop, May 5th, 2011 Outline 1) Overview of the MVSC 2) Adenoviral Vectors 3) Expertise and Services Overview What does the
Baculovirus Adenovirus Adeno-associated virus Lentivirus/Retrovirus Many more: Vaccinia virus, Semliki Forest virus
 Recombinant Viruses Viruses produced by recombinant DNA technology Naturally occuring recombinant virus (= when more than one virus strain infects the same cell and genetic information is exchanged) Common
Recombinant Viruses Viruses produced by recombinant DNA technology Naturally occuring recombinant virus (= when more than one virus strain infects the same cell and genetic information is exchanged) Common
Plasmid Features: Coordinates Feature ITR: Ad5: and hsyn1 Promoter SV40: Ampicillin:
 4600 G0864 pad5hsyn1mcssv40pa Plasmid Features: 6155bp Coordinates Feature ITR: 16-118 Ad5: 16-365 and 903-3357 hsyn1 Promoter 399-862 SV40: 948-1385 Ampicillin: 5092-5952 Antibiotic Resistance: Ampicillin
4600 G0864 pad5hsyn1mcssv40pa Plasmid Features: 6155bp Coordinates Feature ITR: 16-118 Ad5: 16-365 and 903-3357 hsyn1 Promoter 399-862 SV40: 948-1385 Ampicillin: 5092-5952 Antibiotic Resistance: Ampicillin
Supplementary Methods
 Supplementary Methods Reverse transcribed Quantitative PCR. Total RNA was isolated from bone marrow derived macrophages using RNeasy Mini Kit (Qiagen), DNase-treated (Promega RQ1), and reverse transcribed
Supplementary Methods Reverse transcribed Quantitative PCR. Total RNA was isolated from bone marrow derived macrophages using RNeasy Mini Kit (Qiagen), DNase-treated (Promega RQ1), and reverse transcribed
ARTICLE FIG. 1. Heterodimer strategy. (A) Map of the B-domain (3.3 kb, the whole exon 14) deleted human F8 cdna. The arrow and the dotted line represe
 Expression of Human Factor VIII by Splicing between Dimerized AAV Vectors Hengjun Chao, 1,2 Liangwu Sun, 3 Andrew Bruce, 1 Xiao Xiao, 3 and Christopher E. Walsh 1,2, * 1 UNC Gene Therapy Center, University
Expression of Human Factor VIII by Splicing between Dimerized AAV Vectors Hengjun Chao, 1,2 Liangwu Sun, 3 Andrew Bruce, 1 Xiao Xiao, 3 and Christopher E. Walsh 1,2, * 1 UNC Gene Therapy Center, University
Concatamerization of Adeno-Associated Virus Circular Genomes Occurs through Intermolecular Recombination
 JOURNAL OF VIROLOGY, Nov. 1999, p. 9468 9477 Vol. 73, No. 11 0022-538X/99/$04.00 0 Copyright 1999, American Society for Microbiology. All Rights Reserved. Concatamerization of Adeno-Associated Virus Circular
JOURNAL OF VIROLOGY, Nov. 1999, p. 9468 9477 Vol. 73, No. 11 0022-538X/99/$04.00 0 Copyright 1999, American Society for Microbiology. All Rights Reserved. Concatamerization of Adeno-Associated Virus Circular
Production of High-Titer Recombinant Adeno-Associated Virus Vectors in the Absence of Helper Adenovirus
 JOURNAL OF VIROLOGY, Mar. 1998, p. 2224 2232 Vol. 72, No. 3 0022-538X/98/$04.00 0 Copyright 1998, American Society for Microbiology Production of High-Titer Recombinant Adeno-Associated Virus Vectors in
JOURNAL OF VIROLOGY, Mar. 1998, p. 2224 2232 Vol. 72, No. 3 0022-538X/98/$04.00 0 Copyright 1998, American Society for Microbiology Production of High-Titer Recombinant Adeno-Associated Virus Vectors in
Issues in production of viral gene transfer vectors. Stefan Kochanek Department of Gene Therapy Ulm University
 Issues in production of viral gene transfer vectors Stefan Kochanek Department of Gene Therapy Ulm University Only few positive results in gene therapy so far - many early phase, few late phase clinical
Issues in production of viral gene transfer vectors Stefan Kochanek Department of Gene Therapy Ulm University Only few positive results in gene therapy so far - many early phase, few late phase clinical
Adeno-Associated Viral (AAV) Vectors. Biosafety office
 Adeno-Associated Viral (AAV) Vectors Biosafety office April 2014 Copyright 2014 University of Cincinnati, All Rights Reserved. All rights reserved. No part of this publication may be reproduced, distributed,
Adeno-Associated Viral (AAV) Vectors Biosafety office April 2014 Copyright 2014 University of Cincinnati, All Rights Reserved. All rights reserved. No part of this publication may be reproduced, distributed,
TRANSGENIC ANIMALS. -transient transfection of cells -stable transfection of cells. - Two methods to produce transgenic animals:
 TRANSGENIC ANIMALS -transient transfection of cells -stable transfection of cells - Two methods to produce transgenic animals: 1- DNA microinjection - random insertion 2- embryonic stem cell-mediated gene
TRANSGENIC ANIMALS -transient transfection of cells -stable transfection of cells - Two methods to produce transgenic animals: 1- DNA microinjection - random insertion 2- embryonic stem cell-mediated gene
7.012 Problem Set 5. Question 1
 Name Section 7.012 Problem Set 5 Question 1 While studying the problem of infertility, you attempt to isolate a hypothetical rabbit gene that accounts for the prolific reproduction of rabbits. After much
Name Section 7.012 Problem Set 5 Question 1 While studying the problem of infertility, you attempt to isolate a hypothetical rabbit gene that accounts for the prolific reproduction of rabbits. After much
Certificate of Analysis
 Certificate of Analysis Catalog No. Amount Lot Number 631986 10 μg Specified on product label. Product Information plvx-ef1α-mcherry-n1 is a lentiviral expression vector that can be used to generate high-titer
Certificate of Analysis Catalog No. Amount Lot Number 631986 10 μg Specified on product label. Product Information plvx-ef1α-mcherry-n1 is a lentiviral expression vector that can be used to generate high-titer
Recombination between Two Identical Sequences within the Same Retroviral RNA Molecule
 JOURNAL OF VIROLOGY, July 1999, p. 5912 5917 Vol. 73, No. 7 0022-538X/99/$04.00 0 Copyright 1999, American Society for Microbiology. All Rights Reserved. Recombination between Two Identical Sequences within
JOURNAL OF VIROLOGY, July 1999, p. 5912 5917 Vol. 73, No. 7 0022-538X/99/$04.00 0 Copyright 1999, American Society for Microbiology. All Rights Reserved. Recombination between Two Identical Sequences within
SV40pA. Plasmid Features: Antibiotic Resistance: Ampicillin Backbone: pbr322
 G1072 pacad5cagmcswtiresmcherry- SV40pA Plasmid Features: Coordinates Feature ITR: 16-118 Ad5: 16-368 and 897-3361 CAG 441-2112 MCS: 2121-2177 wtires 2179-2769 mcherry 2765-3475 SV40pA: 3484-3923 Ampicillin:
G1072 pacad5cagmcswtiresmcherry- SV40pA Plasmid Features: Coordinates Feature ITR: 16-118 Ad5: 16-368 and 897-3361 CAG 441-2112 MCS: 2121-2177 wtires 2179-2769 mcherry 2765-3475 SV40pA: 3484-3923 Ampicillin:
Ideal vector for gene delivery:
 Ideal vector for gene delivery: High transduction efficiency High and long-lasting lasting transgene expression No cytotoxicity No induction of immune response Cell-type specificity High transgene capacity
Ideal vector for gene delivery: High transduction efficiency High and long-lasting lasting transgene expression No cytotoxicity No induction of immune response Cell-type specificity High transgene capacity
BRIEF COMMUNICATION Alpha-1-antitrypsin expression in the lung is increased by airway delivery of gene-transfected macrophages
 (2003) 10, 2148 2152 & 2003 Nature Publishing Group All rights reserved 0969-7128/03 $25.00 www.nature.com/gt BRIEF COMMUNICATION Alpha-1-antitrypsin expression in the lung is increased by airway delivery
(2003) 10, 2148 2152 & 2003 Nature Publishing Group All rights reserved 0969-7128/03 $25.00 www.nature.com/gt BRIEF COMMUNICATION Alpha-1-antitrypsin expression in the lung is increased by airway delivery
Pei et al. Supplementary Figure S1
 Pei et al. Supplementary Figure S1 C H-CUL9: + + + + + Myc-ROC1: - - + + + U2OS/pcDN3 U2OS/H-CUL9 U2OS/ + H-CUL9 IP: -H -myc input -H -myc 1 2 3 4 5 H-CUL9 Myc-ROC1 -H -H -H -H H-CUL9: wt RR myc-roc1:
Pei et al. Supplementary Figure S1 C H-CUL9: + + + + + Myc-ROC1: - - + + + U2OS/pcDN3 U2OS/H-CUL9 U2OS/ + H-CUL9 IP: -H -myc input -H -myc 1 2 3 4 5 H-CUL9 Myc-ROC1 -H -H -H -H H-CUL9: wt RR myc-roc1:
Revised: RG-RV2 by Fukuhara et al.
 Supplemental Figure 1 The generation of Spns2 conditional knockout mice. (A) Schematic representation of the wild type Spns2 locus (Spns2 + ), the targeted allele, the floxed allele (Spns2 f ) and the
Supplemental Figure 1 The generation of Spns2 conditional knockout mice. (A) Schematic representation of the wild type Spns2 locus (Spns2 + ), the targeted allele, the floxed allele (Spns2 f ) and the
Gene Therapy for Hemophilia A: Are we really getting better?
 Gene Therapy for Hemophilia A: Are we really getting better? XXIV Congresso Nazionale SISET Novembre 10, 2016 Abano Terme Valder R. Arruda The Children s Hospital of Philadelphia University of Pennsylvania
Gene Therapy for Hemophilia A: Are we really getting better? XXIV Congresso Nazionale SISET Novembre 10, 2016 Abano Terme Valder R. Arruda The Children s Hospital of Philadelphia University of Pennsylvania
G0411 pfiv3.2mcsegfp. Plasmid Features
 G0411 pfiv3.2mcsegfp Plasmid Features Coordinates Feature CMV/5 LTR hybrid 1:682 Major Splice Donor 926:957 Partial gag 958:1468 Central Polypurine Tract 1494:1653 Multiple cloning sites 1653:1714 egfp
G0411 pfiv3.2mcsegfp Plasmid Features Coordinates Feature CMV/5 LTR hybrid 1:682 Major Splice Donor 926:957 Partial gag 958:1468 Central Polypurine Tract 1494:1653 Multiple cloning sites 1653:1714 egfp
AAVanced Concentration Reagent
 AAVanced Concentration Reagent Catalog # AAV100A-1, AAV110A-1 A limited-use label license covers this product. By use of this product, you accept the terms and conditions outlined in the Licensing and
AAVanced Concentration Reagent Catalog # AAV100A-1, AAV110A-1 A limited-use label license covers this product. By use of this product, you accept the terms and conditions outlined in the Licensing and
Pre-made expression Adenovirus product manual
 Pre-made expression Adenovirus product manual Catalog# Product Name Amounts AVP001 RFP adenovirus AVP002 AVP004 AVP005 AVP011 AVP012 AVP017 AVP010 AVP013 AVP014 AVP015 AVP016 AVP-Null AVP001-PBS AVP002-PBS
Pre-made expression Adenovirus product manual Catalog# Product Name Amounts AVP001 RFP adenovirus AVP002 AVP004 AVP005 AVP011 AVP012 AVP017 AVP010 AVP013 AVP014 AVP015 AVP016 AVP-Null AVP001-PBS AVP002-PBS
Plasmid Features: Coordinates Feature Ad5: and mcmv: MCS: SV40: Ampicillin: (Complimentary)
 G0688 pacad5cmvmcssv40pa Plasmid Features: 6210bp Coordinates Feature Ad5: 16-368 and 1441-3899 mcmv: 385-907 MCS: 913-1002 SV40: 1003-1470 Ampicillin: 6007-5147 (Complimentary) Antibiotic Resistance:
G0688 pacad5cmvmcssv40pa Plasmid Features: 6210bp Coordinates Feature Ad5: 16-368 and 1441-3899 mcmv: 385-907 MCS: 913-1002 SV40: 1003-1470 Ampicillin: 6007-5147 (Complimentary) Antibiotic Resistance:
2054, Chap. 14, page 1
 2054, Chap. 14, page 1 I. Recombinant DNA technology (Chapter 14) A. recombinant DNA technology = collection of methods used to perform genetic engineering 1. genetic engineering = deliberate modification
2054, Chap. 14, page 1 I. Recombinant DNA technology (Chapter 14) A. recombinant DNA technology = collection of methods used to perform genetic engineering 1. genetic engineering = deliberate modification
Directed Evolution of Adeno-associated Virus Targeting Heart and Skeletal muscle. Vivian Xu. Principle Investigator: Xiao Xiao
 Directed Evolution of Adeno-associated Virus Targeting Heart and Skeletal muscle By Vivian Xu Principle Investigator: Xiao Xiao Biology Faculty Advisor: Terry Furey Senior Honors Thesis Department of Biology
Directed Evolution of Adeno-associated Virus Targeting Heart and Skeletal muscle By Vivian Xu Principle Investigator: Xiao Xiao Biology Faculty Advisor: Terry Furey Senior Honors Thesis Department of Biology
HH67.2, A PROMISING NOVEL ADENO-ASSOCIATED VIRUS (AAV) VECTOR, SHOWS IMPROVED MUSCLE SPECIFICITY AND INFECTIVITY COMPARED TO AAV9. Carrie B.
 HH67.2, A PROMISING NOVEL ADENO-ASSOCIATED VIRUS (AAV) VECTOR, SHOWS IMPROVED MUSCLE SPECIFICITY AND INFECTIVITY COMPARED TO AAV9 Carrie B. Martin A thesis submitted to the faculty at the University of
HH67.2, A PROMISING NOVEL ADENO-ASSOCIATED VIRUS (AAV) VECTOR, SHOWS IMPROVED MUSCLE SPECIFICITY AND INFECTIVITY COMPARED TO AAV9 Carrie B. Martin A thesis submitted to the faculty at the University of
GENE THERAPISTS have a special fondness for hematology.
 REVIEW ARTICLE Adeno-Associated Virus Vectors and Hematology GENE THERAPISTS have a special fondness for hematology. Initially, this was due to the many inherited diseases that could potentially be cured
REVIEW ARTICLE Adeno-Associated Virus Vectors and Hematology GENE THERAPISTS have a special fondness for hematology. Initially, this was due to the many inherited diseases that could potentially be cured
G0796 pacad5tretightmcssv40pa
 G0796 pacad5tretightmcssv40pa Plasmid Features: 6009bp Coordinates Feature ITR: 16-118 Ad5 wt 16-365 and 1240-3698 TRE-tight: 402-717 SV40: 802-1239 Ampicillin: 5806-4946 Antibiotic Resistance: Ampicillin
G0796 pacad5tretightmcssv40pa Plasmid Features: 6009bp Coordinates Feature ITR: 16-118 Ad5 wt 16-365 and 1240-3698 TRE-tight: 402-717 SV40: 802-1239 Ampicillin: 5806-4946 Antibiotic Resistance: Ampicillin
Schematic representation of the endogenous PALB2 locus and gene-disruption constructs
 Supplementary Figures Supplementary Figure 1. Generation of PALB2 -/- and BRCA2 -/- /PALB2 -/- DT40 cells. (A) Schematic representation of the endogenous PALB2 locus and gene-disruption constructs carrying
Supplementary Figures Supplementary Figure 1. Generation of PALB2 -/- and BRCA2 -/- /PALB2 -/- DT40 cells. (A) Schematic representation of the endogenous PALB2 locus and gene-disruption constructs carrying
Adenoviral Expression Systems. Lentivirus is not the only choice for gene delivery. Adeno-X
 Adenoviral Expression Systems Lentivirus is not the only choice for gene delivery 3 Adeno-X Why choose adenoviral gene delivery? Table I: Adenoviral vs. Lentiviral Gene Delivery Lentivirus Adenovirus Infects
Adenoviral Expression Systems Lentivirus is not the only choice for gene delivery 3 Adeno-X Why choose adenoviral gene delivery? Table I: Adenoviral vs. Lentiviral Gene Delivery Lentivirus Adenovirus Infects
Plaque Size Phenotype as a Selectable Marker To Generate Vaccinia Virus Recombinants
 JOURNAL OF VIROLOGY, Feb. 1989, p. 997-1001 0022-538X/89/020997-05$02.00/0 opyright 0 1989, American Society for Microbiology Vol. 63, No. 2 Plaque Size Phenotype as a Selectable Marker To Generate Vaccinia
JOURNAL OF VIROLOGY, Feb. 1989, p. 997-1001 0022-538X/89/020997-05$02.00/0 opyright 0 1989, American Society for Microbiology Vol. 63, No. 2 Plaque Size Phenotype as a Selectable Marker To Generate Vaccinia
J. Fraser Wright, Ph.D.
 Towards Better Characterization of Recombinant AAV Gene Transfer Vectors: Case Studies Illustrating Challenges with Dose Determining Vector Concentration methods J. Fraser Wright, Ph.D. Well Characterized
Towards Better Characterization of Recombinant AAV Gene Transfer Vectors: Case Studies Illustrating Challenges with Dose Determining Vector Concentration methods J. Fraser Wright, Ph.D. Well Characterized
Received 23 May 2002/Accepted 7 August 2002
 JOURNAL OF VIROLOGY, Nov. 2002, p. 11343 11349 Vol. 76, No. 22 0022-538X/02/$04.00 0 DOI: 10.1128/JVI.76.22.11343 11349.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. A Limited
JOURNAL OF VIROLOGY, Nov. 2002, p. 11343 11349 Vol. 76, No. 22 0022-538X/02/$04.00 0 DOI: 10.1128/JVI.76.22.11343 11349.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. A Limited
Article: "Noninvasive and Targeted Gene Delivery into the Brain Using Microbubble-Facilit 1 / 16. Ultrasound" Malova Anna
 Article: "Noninvasive and Targeted Gene Delivery into the Brain Using Microbubble-Facilitated Focused Ultrasound" 06.04.2013 Article: "Noninvasive and Targeted Gene Delivery into the Brain Using 06.04.2013
Article: "Noninvasive and Targeted Gene Delivery into the Brain Using Microbubble-Facilitated Focused Ultrasound" 06.04.2013 Article: "Noninvasive and Targeted Gene Delivery into the Brain Using 06.04.2013
High-Fidelity Correction of Mutations at Multiple Chromosomal Positions by Adeno-Associated Virus Vectors
 JOURNAL OF VIROLOGY, Sept. 1999, p. 7376 7380 Vol. 73, No. 9 0022-538X/99/$04.00 0 Copyright 1999, American Society for Microbiology. All Rights Reserved. High-Fidelity Correction of Mutations at Multiple
JOURNAL OF VIROLOGY, Sept. 1999, p. 7376 7380 Vol. 73, No. 9 0022-538X/99/$04.00 0 Copyright 1999, American Society for Microbiology. All Rights Reserved. High-Fidelity Correction of Mutations at Multiple
Recombinant Adeno- Associated Virus (raav) Delivered shrna. - A Technical Guide
 Efficient Gene Knockdown via Recombinant Adeno- Associated Virus (raav) Delivered shrna - A Technical Guide Revision 201602.01 ViGene Biosciences 2016 RESEARCH USE ONLY. Not for use in diagnostic procedures
Efficient Gene Knockdown via Recombinant Adeno- Associated Virus (raav) Delivered shrna - A Technical Guide Revision 201602.01 ViGene Biosciences 2016 RESEARCH USE ONLY. Not for use in diagnostic procedures
G1059 pacad5mcswtiresmcherry SV40pA
 G1059 pacad5mcswtiresmcherry SV40pA Plasmid Features: 6958bp Coordinates Feature ITR: 16-118 Ad5: 16-368 and 2189-4647 MCS: 375-456 wtires 445-1031 mcherry 1032-1742 SV40pA: 1751-2188 Ampicillin: 6755-5895
G1059 pacad5mcswtiresmcherry SV40pA Plasmid Features: 6958bp Coordinates Feature ITR: 16-118 Ad5: 16-368 and 2189-4647 MCS: 375-456 wtires 445-1031 mcherry 1032-1742 SV40pA: 1751-2188 Ampicillin: 6755-5895
TRANSGENIC ANIMALS. transient. stable. - Two methods to produce transgenic animals:
 Only for teaching purposes - not for reproduction or sale CELL TRANSFECTION transient stable TRANSGENIC ANIMALS - Two methods to produce transgenic animals: 1- DNA microinjection 2- embryonic stem cell-mediated
Only for teaching purposes - not for reproduction or sale CELL TRANSFECTION transient stable TRANSGENIC ANIMALS - Two methods to produce transgenic animals: 1- DNA microinjection 2- embryonic stem cell-mediated
Ribosomal DNA Integrating raav-rdna Vectors Allow for Stable Transgene Expression
 original article The American Society of Gene & Cell Therapy Ribosomal DNA Integrating raav-rdna Vectors Allow for Stable Transgene Expression Leszek Lisowski, Ashley Lau,2, Zhongya Wang 3, Yue Zhang,
original article The American Society of Gene & Cell Therapy Ribosomal DNA Integrating raav-rdna Vectors Allow for Stable Transgene Expression Leszek Lisowski, Ashley Lau,2, Zhongya Wang 3, Yue Zhang,
Isolation of Highly Infectious and Pure Adeno-Associated Virus Type 2 Vectors with a Single-Step Gravity-Flow Column
 HUMAN GENE THERAPY 12:71 76 (January 1, 2001) Mary Ann Liebert, Inc. Isolation of Highly Infectious and Pure Adeno-Associated Virus Type 2 Vectors with a Single-Step Gravity-Flow Column ALBERTO AURICCHIO,*
HUMAN GENE THERAPY 12:71 76 (January 1, 2001) Mary Ann Liebert, Inc. Isolation of Highly Infectious and Pure Adeno-Associated Virus Type 2 Vectors with a Single-Step Gravity-Flow Column ALBERTO AURICCHIO,*
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10163 Supplementary Table 1 Efficiency of vector construction. Process wells recovered efficiency (%) Recombineering* 480 461 96 Intermediate plasmids 461 381 83 Recombineering efficiency
doi:10.1038/nature10163 Supplementary Table 1 Efficiency of vector construction. Process wells recovered efficiency (%) Recombineering* 480 461 96 Intermediate plasmids 461 381 83 Recombineering efficiency
Improved adeno-associated virus (AAV) serotype 1 and 5 vectors for gene therapy
 Improved adeno-associated virus (AAV) serotype 1 and 5 vectors for gene therapy Dwaipayan Sen 1, Balaji Balakrishnan 1, Nishanth Gabriel 1, Prachi Agrawal 2, Vaani Roshini 1, Alok Srivastava 1,2, Giridhara
Improved adeno-associated virus (AAV) serotype 1 and 5 vectors for gene therapy Dwaipayan Sen 1, Balaji Balakrishnan 1, Nishanth Gabriel 1, Prachi Agrawal 2, Vaani Roshini 1, Alok Srivastava 1,2, Giridhara
Antibiotic Resistance: Ampicillin Bacterial Backbone: pbluescriptksii(+), Agilent Technologies
 G0656 pfiv3.2rsvmcs Coordinates Plasmid Features 1-682 CMV/5 LTR hybrid 958-1468 Partial Gag 1494-1653 Central Polypurine Tract 1695-2075 RSV Promoter 2104-2222 Multiple Cloning Sites 2244-2839 WPRE 2868-3009
G0656 pfiv3.2rsvmcs Coordinates Plasmid Features 1-682 CMV/5 LTR hybrid 958-1468 Partial Gag 1494-1653 Central Polypurine Tract 1695-2075 RSV Promoter 2104-2222 Multiple Cloning Sites 2244-2839 WPRE 2868-3009
sirna Overview and Technical Tips
 1 sirna Overview and Technical Tips 2 CONTENTS 3 4 5 7 8 10 11 13 14 18 19 20 21 Introduction Applications How Does It Work? Handy Tips Troubleshooting Conclusions Further References Contact Us 3 INTRODUCTION
1 sirna Overview and Technical Tips 2 CONTENTS 3 4 5 7 8 10 11 13 14 18 19 20 21 Introduction Applications How Does It Work? Handy Tips Troubleshooting Conclusions Further References Contact Us 3 INTRODUCTION
Chapter 20 Recombinant DNA Technology. Copyright 2009 Pearson Education, Inc.
 Chapter 20 Recombinant DNA Technology Copyright 2009 Pearson Education, Inc. 20.1 Recombinant DNA Technology Began with Two Key Tools: Restriction Enzymes and DNA Cloning Vectors Recombinant DNA refers
Chapter 20 Recombinant DNA Technology Copyright 2009 Pearson Education, Inc. 20.1 Recombinant DNA Technology Began with Two Key Tools: Restriction Enzymes and DNA Cloning Vectors Recombinant DNA refers
AAVpro Helper Free System
 For Research Use Cat. # 6230, 6650-6657, 6668, 6669, 6673 AAVpro Helper Free System Product Manual For China/Korea/India/Europe Table of Contents I. Description... 4 II. Components... 7 III. IV. Storage...13
For Research Use Cat. # 6230, 6650-6657, 6668, 6669, 6673 AAVpro Helper Free System Product Manual For China/Korea/India/Europe Table of Contents I. Description... 4 II. Components... 7 III. IV. Storage...13
BIO 202 Midterm Exam Winter 2007
 BIO 202 Midterm Exam Winter 2007 Mario Chevrette Lectures 10-14 : Question 1 (1 point) Which of the following statements is incorrect. a) In contrast to prokaryotic DNA, eukaryotic DNA contains many repetitive
BIO 202 Midterm Exam Winter 2007 Mario Chevrette Lectures 10-14 : Question 1 (1 point) Which of the following statements is incorrect. a) In contrast to prokaryotic DNA, eukaryotic DNA contains many repetitive
Potent and durable RNAi mediated suppression of Hepatitis B Virus infection in a mouse model by a nonviral Closed Ended Linear DNA (CELID) vector
 Potent and durable RNAi mediated suppression of Hepatitis B Virus infection in a mouse model by a nonviral Closed Ended Linear DNA (CELID) vector November 11, 2018 Dr. Peter W. Roelvink Senior Director
Potent and durable RNAi mediated suppression of Hepatitis B Virus infection in a mouse model by a nonviral Closed Ended Linear DNA (CELID) vector November 11, 2018 Dr. Peter W. Roelvink Senior Director
Adenovirus Titration Kit
 Adenovirus Titration Kit Catalog # LF-RK0001(1 kit) Immunostaining method for Quantitative Detection of Adenovirus For research use only Not for diagnostic or therapeutic procedures AbFrontier Science
Adenovirus Titration Kit Catalog # LF-RK0001(1 kit) Immunostaining method for Quantitative Detection of Adenovirus For research use only Not for diagnostic or therapeutic procedures AbFrontier Science
Biology of Adenovirus Vectors with E1 and E4 Deletions for Liver-Directed Gene Therapy
 JOURNAL OF VIROLOGY, Dec. 1996, p. 8934 8943 Vol. 70, No. 12 0022-538X/96/$04.00 0 Copyright 1996, American Society for Microbiology Biology of Adenovirus Vectors with E1 and E4 Deletions for Liver-Directed
JOURNAL OF VIROLOGY, Dec. 1996, p. 8934 8943 Vol. 70, No. 12 0022-538X/96/$04.00 0 Copyright 1996, American Society for Microbiology Biology of Adenovirus Vectors with E1 and E4 Deletions for Liver-Directed
ICH Considerations. Oncolytic Viruses September 17, 2009
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH Considerations Oncolytic Viruses September 17, 2009 1. Introduction Oncolytic viruses
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH Considerations Oncolytic Viruses September 17, 2009 1. Introduction Oncolytic viruses
MicroRNAs Modulate Hematopoietic Lineage Differentiation
 Chen et al., page 1 MicroRNAs Modulate Hematopoietic Lineage Differentiation Chang-Zheng Chen, Ling Li, Harvey F. Lodish, David. Bartel Supplemental Online Material Methods Cell isolation Murine bone marrow
Chen et al., page 1 MicroRNAs Modulate Hematopoietic Lineage Differentiation Chang-Zheng Chen, Ling Li, Harvey F. Lodish, David. Bartel Supplemental Online Material Methods Cell isolation Murine bone marrow
Enzyme that uses RNA as a template to synthesize a complementary DNA
 Biology 105: Introduction to Genetics PRACTICE FINAL EXAM 2006 Part I: Definitions Homology: Comparison of two or more protein or DNA sequence to ascertain similarities in sequences. If two genes have
Biology 105: Introduction to Genetics PRACTICE FINAL EXAM 2006 Part I: Definitions Homology: Comparison of two or more protein or DNA sequence to ascertain similarities in sequences. If two genes have
Improved Adeno-Associated Virus Vector Production with Transfection of a Single Helper Adenovirus Gene, E4orf6
 doi:10.1006/mthe.1999.0010, available on IDEAL at http://www.idealibrary.com Improved Adeno-Associated Virus Vector Production with Transfection of a Single Helper Adenovirus Gene, E4orf6 James M. Allen,*
doi:10.1006/mthe.1999.0010, available on IDEAL at http://www.idealibrary.com Improved Adeno-Associated Virus Vector Production with Transfection of a Single Helper Adenovirus Gene, E4orf6 James M. Allen,*
How fast? How fast can you clone your gene into a small puc based shuttle plasmid and prepare 100ug plasmid DNA? Add 7 to 10 days to that!
 AdMax AdMax Hi-IQ Helper Dependent Adenovirus AdCre NautCell Price List Ordering Info Technical Support AdMax "We continue to have great success with the AdMax system for producing a wide range of vectors
AdMax AdMax Hi-IQ Helper Dependent Adenovirus AdCre NautCell Price List Ordering Info Technical Support AdMax "We continue to have great success with the AdMax system for producing a wide range of vectors
M Keramatipour 2. M Keramatipour 1. M Keramatipour 4. M Keramatipour 3. M Keramatipour 5. M Keramatipour
 Molecular Cloning Methods Mohammad Keramatipour MD, PhD keramatipour@tums.ac.ir Outline DNA recombinant technology DNA cloning co Cell based PCR PCR-based Some application of DNA cloning Genomic libraries
Molecular Cloning Methods Mohammad Keramatipour MD, PhD keramatipour@tums.ac.ir Outline DNA recombinant technology DNA cloning co Cell based PCR PCR-based Some application of DNA cloning Genomic libraries
AD_. TITLE: Targeted Retroviral Infection of mammary Cells in Viral Receptor Transgenics
 «, AD_ Grant Number DAMD17-96-1-6264 TITLE: Targeted Retroviral Infection of mammary Cells in Viral Receptor Transgenics PRINCIPAL INVESTIGATOR: Paul Bates, Ph.D. CONTRACTING ORGANIZATION: University of
«, AD_ Grant Number DAMD17-96-1-6264 TITLE: Targeted Retroviral Infection of mammary Cells in Viral Receptor Transgenics PRINCIPAL INVESTIGATOR: Paul Bates, Ph.D. CONTRACTING ORGANIZATION: University of
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/317/5837/477/dc1 Supporting Online Material for AAV Vector Integration Sites in Mouse Hepatocellular Carcinoma Anthony Donsante, Daniel G. Miller, Yi Li, Carole Vogler,
www.sciencemag.org/cgi/content/full/317/5837/477/dc1 Supporting Online Material for AAV Vector Integration Sites in Mouse Hepatocellular Carcinoma Anthony Donsante, Daniel G. Miller, Yi Li, Carole Vogler,
G0752 pfiv3.2mcswtiresegfp
 G0752 pfiv3.2mcswtiresegfp Plasmid Features Coordinates Feature 1-682 CMV/5 LTR Hybrid 958-1468 Partial Gag 1494-1653 Central Polypurine Tract 1653-1738 Multiple Cloning Sites 1739-2325 wtires 2326-3045
G0752 pfiv3.2mcswtiresegfp Plasmid Features Coordinates Feature 1-682 CMV/5 LTR Hybrid 958-1468 Partial Gag 1494-1653 Central Polypurine Tract 1653-1738 Multiple Cloning Sites 1739-2325 wtires 2326-3045
Molecular Cloning. Restriction Enzymes and Ligases
 Tools in Genetic engineering The science of using living systems to benefit humankind is called biotechnology. Technically speaking, the domestication of plants and animals through farming and breeding
Tools in Genetic engineering The science of using living systems to benefit humankind is called biotechnology. Technically speaking, the domestication of plants and animals through farming and breeding
United States Patent (19) Russell et al.
 United States Patent (19) Russell et al. 54 ADENO-ASSOCIATED VIRUS (AAV) SOLATES AND AAV VECTORS DERIVED THEREFROM 75 Inventors: David W. Russell; Elizabeth A. Rutledge, both of Seattle, Wash. 73 Assignee:
United States Patent (19) Russell et al. 54 ADENO-ASSOCIATED VIRUS (AAV) SOLATES AND AAV VECTORS DERIVED THEREFROM 75 Inventors: David W. Russell; Elizabeth A. Rutledge, both of Seattle, Wash. 73 Assignee:
University of Lethbridge GUIDELINES FOR USE OF ADENO-ASSOCIATED VIRAL VECTORS (AAV VECTORS)
 GUIDELINES FOR USE OF ADENO-ASSOCIATED VIRAL VECTORS (AAV VECTORS) Abstract All laboratories using AAV vectors must adhere to this code of practice December 2013 Purpose: To provide guidelines for safely
GUIDELINES FOR USE OF ADENO-ASSOCIATED VIRAL VECTORS (AAV VECTORS) Abstract All laboratories using AAV vectors must adhere to this code of practice December 2013 Purpose: To provide guidelines for safely
Red Type Indicates Unique Site
 3600 G0605 pscaavmcmvmcsbghpa Plasmid Features: Coordinates Feature 980-1084 AAV2 5 ITR 1144-1666 modified CMV 1667-1761 MCS 1762-1975 BgHpA 1987-2114 AAV2 3 ITR 3031-3891 B-lactamase (Ampicillin) Antibiotic
3600 G0605 pscaavmcmvmcsbghpa Plasmid Features: Coordinates Feature 980-1084 AAV2 5 ITR 1144-1666 modified CMV 1667-1761 MCS 1762-1975 BgHpA 1987-2114 AAV2 3 ITR 3031-3891 B-lactamase (Ampicillin) Antibiotic
AAVpro Helper Free System
 For Research Use Cat. # 6230, 6652, 6655 AAVpro Helper Free System Product Manual For USA Table of Contents I. Description... 4 II. Components... 6 III. Storage... 9 IV. Materials Required but not Provided...
For Research Use Cat. # 6230, 6652, 6655 AAVpro Helper Free System Product Manual For USA Table of Contents I. Description... 4 II. Components... 6 III. Storage... 9 IV. Materials Required but not Provided...
COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) <DRAFT>
 European Medicines Agency Pre-authorisation Evaluation of Medicines for Human Use London, 17 November 2005 Doc. Ref. EMEA/273974/2005 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) NOTE
European Medicines Agency Pre-authorisation Evaluation of Medicines for Human Use London, 17 November 2005 Doc. Ref. EMEA/273974/2005 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) NOTE
Why do we care about homologous recombination?
 Why do we care about homologous recombination? Universal biological mechanism Bacteria can pick up new genes Biotechnology Gene knockouts in mice via homologous recombination 1 DNA of interest in mouse
Why do we care about homologous recombination? Universal biological mechanism Bacteria can pick up new genes Biotechnology Gene knockouts in mice via homologous recombination 1 DNA of interest in mouse
Biology 105: Introduction to Genetics PRACTICE FINAL EXAM Part I: Definitions. Homology: Reverse transcriptase. Allostery: cdna library
 Biology 105: Introduction to Genetics PRACTICE FINAL EXAM 2006 Part I: Definitions Homology: Reverse transcriptase Allostery: cdna library Transformation Part II Short Answer 1. Describe the reasons for
Biology 105: Introduction to Genetics PRACTICE FINAL EXAM 2006 Part I: Definitions Homology: Reverse transcriptase Allostery: cdna library Transformation Part II Short Answer 1. Describe the reasons for
510 Index. 22 rationale, 15, 16, 25 stability of complexes, 21 Atomic force microscopy (AFM), DNA conjugate particle size analysis, 41, 42
 Index AAV, see Adeno-associated virus Acellular dermis, see Skin substitute Adeno-associated virus (AAV), genome structure, 446, 462 herpes simplex virus amplicons for hightiter stock production, amplicon
Index AAV, see Adeno-associated virus Acellular dermis, see Skin substitute Adeno-associated virus (AAV), genome structure, 446, 462 herpes simplex virus amplicons for hightiter stock production, amplicon
pbroad3-lacz An optimized vector for mouse and rat transgenesis Catalog # pbroad3-lacz
 pbroad3-lacz An optimized vector for mouse and rat transgenesis Catalog # pbroad3-lacz For research use only Version # 03B04-MT PRODUCT INFORMATION Content: - 20 µg of pbroad3-lacz provided as lyophilized
pbroad3-lacz An optimized vector for mouse and rat transgenesis Catalog # pbroad3-lacz For research use only Version # 03B04-MT PRODUCT INFORMATION Content: - 20 µg of pbroad3-lacz provided as lyophilized
ICH Considerations Oncolytic Viruses ONCOLYTIC VIRUSES (EMEA/CHMP/ICH/607698/2008) TRANSMISSION TO CHMP November 2008
 European Medicines Agency October 2009 EMEA/CHMP/ICH/607698/2008 ICH Considerations Oncolytic Viruses ONCOLYTIC VIRUSES (EMEA/CHMP/ICH/607698/2008) TRANSMISSION TO CHMP November 2008 TRANSMISSION TO INTERESTED
European Medicines Agency October 2009 EMEA/CHMP/ICH/607698/2008 ICH Considerations Oncolytic Viruses ONCOLYTIC VIRUSES (EMEA/CHMP/ICH/607698/2008) TRANSMISSION TO CHMP November 2008 TRANSMISSION TO INTERESTED
species- Mus musculus Engineering the mouse genome David Ornitz
 species- Mus musculus Engineering the mouse genome David Ornitz How do we analyze gene function in mice? Gene addition (transgenic approach) Permits GOF, DN and knockdown experiments Ectopic (spatial or
species- Mus musculus Engineering the mouse genome David Ornitz How do we analyze gene function in mice? Gene addition (transgenic approach) Permits GOF, DN and knockdown experiments Ectopic (spatial or
Formation of Adeno-Associated Virus Circular Genomes Is Differentially Regulated by Adenovirus E4 ORF6 and E2a Gene Expression
 JOURNAL OF VIROLOGY, Jan. 1999, p. 161 169 Vol. 73, No. 1 0022-538X/99/$04.00 0 Copyright 1999, American Society for Microbiology. All Rights Reserved. Formation of Adeno-Associated Virus Circular Genomes
JOURNAL OF VIROLOGY, Jan. 1999, p. 161 169 Vol. 73, No. 1 0022-538X/99/$04.00 0 Copyright 1999, American Society for Microbiology. All Rights Reserved. Formation of Adeno-Associated Virus Circular Genomes
Kinetics of Recombinant Adeno-Associated Virus-Mediated Gene Transfer
 JOURNAL OF VIROLOGY, Apr. 2000, p. 3555 3565 Vol. 74, No. 8 0022-538X/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Kinetics of Recombinant Adeno-Associated Virus-Mediated
JOURNAL OF VIROLOGY, Apr. 2000, p. 3555 3565 Vol. 74, No. 8 0022-538X/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Kinetics of Recombinant Adeno-Associated Virus-Mediated
Somatic gene therapy for hypertension
 Brazilian Journal of Medical and Biological Research (2000) 33: 715-721 Gene therapy ISSN 0100-879X 715 Somatic gene therapy for hypertension Department of Physiology, College of Medicine, University of
Brazilian Journal of Medical and Biological Research (2000) 33: 715-721 Gene therapy ISSN 0100-879X 715 Somatic gene therapy for hypertension Department of Physiology, College of Medicine, University of
KNOW HOW & TECHNOLOGY LICENSING OPPORTUNITIES
 Vector platform developed at SAN RAFFAELE TELETHON INSTITUTE FOR GENE THERAPY SAN RAFFAELE HOSPITAL AND SCIENTIFIC INSTITUTE KNOW HOW & TECHNOLOGY LICENSING OPPORTUNITIES -regulated vectors and their uses
Vector platform developed at SAN RAFFAELE TELETHON INSTITUTE FOR GENE THERAPY SAN RAFFAELE HOSPITAL AND SCIENTIFIC INSTITUTE KNOW HOW & TECHNOLOGY LICENSING OPPORTUNITIES -regulated vectors and their uses
Comparability Is Not a Nightmare, Just Think Ahead!
 Comparability Is Not a Nightmare, Just Think Ahead! Margarida Menezes Ferreira Senior Assessor at INFARMED PT expert at BWP/CHMP - EMA member of the CAT - EMA (margarida.menezes@infarmed.pt) I attend this
Comparability Is Not a Nightmare, Just Think Ahead! Margarida Menezes Ferreira Senior Assessor at INFARMED PT expert at BWP/CHMP - EMA member of the CAT - EMA (margarida.menezes@infarmed.pt) I attend this
By two mechanisms: Mutation Genetic Recombination
 Genetics (see text pages 257-259, 267-298) Remember what it is we want to address: How is it that prokaryotes gain new genetic ability? The cells are haploid and reproduce by fission...so how does an genetic
Genetics (see text pages 257-259, 267-298) Remember what it is we want to address: How is it that prokaryotes gain new genetic ability? The cells are haploid and reproduce by fission...so how does an genetic
Manipulating genes and cells (Kap. 10)
 Manipulating genes and cells (Kap. 10) restriction enzymes and agarose gel electrophoresis DNA sequencing nucleic acid hybridization techniques genomic and cdna libraries cloning of DNA PCR and PCR applications
Manipulating genes and cells (Kap. 10) restriction enzymes and agarose gel electrophoresis DNA sequencing nucleic acid hybridization techniques genomic and cdna libraries cloning of DNA PCR and PCR applications
Figure S1. Figure S2. Figure S3 HB Anti-FSP27 (COOH-terminal peptide) Ab. Anti-GST-FSP27(45-127) Ab.
 / 36B4 mrna ratio Figure S1 * 2. 1.6 1.2.8 *.4 control TNFα BRL49653 Figure S2 Su bw AT p iw Anti- (COOH-terminal peptide) Ab Blot : Anti-GST-(45-127) Ab β-actin Figure S3 HB2 HW AT BA T Figure S4 A TAG
/ 36B4 mrna ratio Figure S1 * 2. 1.6 1.2.8 *.4 control TNFα BRL49653 Figure S2 Su bw AT p iw Anti- (COOH-terminal peptide) Ab Blot : Anti-GST-(45-127) Ab β-actin Figure S3 HB2 HW AT BA T Figure S4 A TAG
Integrating Adenovirus Adeno-Associated Virus Hybrid Vectors Devoid of All Viral Genes
 JOURNAL OF VIROLOGY, Nov. 1999, p. 9314 9324 Vol. 73, No. 11 0022-538X/99/$04.00 0 Copyright 1999, American Society for Microbiology. All Rights Reserved. Integrating Adenovirus Adeno-Associated Virus
JOURNAL OF VIROLOGY, Nov. 1999, p. 9314 9324 Vol. 73, No. 11 0022-538X/99/$04.00 0 Copyright 1999, American Society for Microbiology. All Rights Reserved. Integrating Adenovirus Adeno-Associated Virus
Integrating Adenovirus Adeno-Associated Virus Hybrid Vectors Devoid of All Viral Genes
 JOURNAL OF VIROLOGY, Nov. 1999, p. 9314 9324 Vol. 73, No. 11 0022-538X/99/$04.00 0 Copyright 1999, American Society for Microbiology. All Rights Reserved. Integrating Adenovirus Adeno-Associated Virus
JOURNAL OF VIROLOGY, Nov. 1999, p. 9314 9324 Vol. 73, No. 11 0022-538X/99/$04.00 0 Copyright 1999, American Society for Microbiology. All Rights Reserved. Integrating Adenovirus Adeno-Associated Virus
Induction of immune tolerance to coagulation factor IX antigen by in vivo hepatic gene transfer
 Induction of immune tolerance to coagulation factor IX antigen by in vivo hepatic gene transfer Federico Mingozzi,, Katherine A. High, Roland W. Herzog J Clin Invest. 2003;111(9):1347-1356. https://doi.org/10.1172/jci16887.
Induction of immune tolerance to coagulation factor IX antigen by in vivo hepatic gene transfer Federico Mingozzi,, Katherine A. High, Roland W. Herzog J Clin Invest. 2003;111(9):1347-1356. https://doi.org/10.1172/jci16887.
MCB 4211, Fall 2018, Practice Exam 1 Last, First name Student ID # Seat No. ***NOTE: Exam will have 40 multiple choice questions.
 MCB 4211, Fall 2018, Practice Exam 1 Last, First name Student ID # Seat No. ***NOTE: Exam 1 2018 will have 40 multiple choice questions. READ ALL THE CHOICES AND SELECT THE BEST 1. Which of the following
MCB 4211, Fall 2018, Practice Exam 1 Last, First name Student ID # Seat No. ***NOTE: Exam 1 2018 will have 40 multiple choice questions. READ ALL THE CHOICES AND SELECT THE BEST 1. Which of the following
a. Hypoxanthine was present in the media. MCB 4211, Fall 2018, Practice Exam 1 Last, First name Student ID # Seat No.
 MCB 4211, Fall 2018, Practice Exam 1 Last, First name Student ID # Seat No. ***NOTE: Exam 1 2018 will have 40 multiple choice questions. READ ALL THE CHOICES AND SELECT THE BEST 1. Which of the following
MCB 4211, Fall 2018, Practice Exam 1 Last, First name Student ID # Seat No. ***NOTE: Exam 1 2018 will have 40 multiple choice questions. READ ALL THE CHOICES AND SELECT THE BEST 1. Which of the following
